Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Recent progress in NiFe-based catalysts for the high current density oxygen evolution reaction
Jaira Neibel Y.
Bamba
a,
Maricor F.
Divinagracia-Luzadas
a,
Donghyun
Yoon
 bc,
Jung-goo
Choi
bc,
Joey D.
Ocon
*d and
Jaeyoung
Lee
bc,
Jung-goo
Choi
bc,
Joey D.
Ocon
*d and
Jaeyoung
Lee
 *bce
*bce
aLaboratory of Electrochemical Engineering (LEE), Department of Chemical Engineering, University of the Philippines Diliman, Quezon City, 1101, Philippines
bErtl Center for Electrochemistry and Catalysis, Gwangju Institute of Science and Technology (GIST), 123 Cheomdangwagi-Ro, Buk-gu, Gwangju, 61005, South Korea
cDepartment of Environment and Energy Engineering, GIST, 123 Cheomdangwagiro, Buk-gu, Gwangju, 61005, South Korea
dChemical Engineering Department, De La Salle University, 2401 Taft Avenue, Manila, 0922, Philippines
eInternational Future Research Center of Chemical Energy Storage and Conversion Processes (ifRC-CHESS), GIST, Gwangju, 61005, Republic of Korea
First published on 19th January 2026
Abstract
The global transition to green hydrogen via water electrolysis is constrained by the sluggish oxygen evolution reaction (OER), particularly at high current densities required for industrial applications. Among Earth-abundant materials, nickel–iron (NiFe)-based compounds have emerged as leading candidates, offering intrinsic activity, synergistic interactions, and cost advantages that reduce the OER energy barrier, positioning them as viable alternatives to noble-metal catalysts such as IrO2 and RuO2. Yet, achieving long-term activity and structural stability at high current densities (HCDs) remains a critical challenge. This review highlights strategies to advance NiFe-based OER catalysts for sustained high-current operation, focusing on recent innovations including heteroatom doping, vacancy engineering, heterostructure formation, active-site modulation, and self-healing mechanisms. Developments across oxides, (oxy)hydroxides, non-metallic heteroatomic composites, layered double hydroxides, metal–organic framework-derived materials, and noble-metal-integrated hybrids are examined to provide a rational design framework for robust and efficient OER catalysts. Key pathways to tune morphology, composition, and electronic structure are identified, offering insights to bridge the gap between laboratory-scale studies and scalable electrolyzer deployment.
1. Introduction
As the world shifts towards sustainable energy sources, hydrogen has emerged as a promising candidate for clean fuel due to its high energy density and zero-carbon emissions. A pivotal process for hydrogen production is water electrolysis, which splits water into oxygen and hydrogen. However, the efficiency of water electrolysis is limited by the sluggish oxygen evolution reaction (OER), a half-reaction involving a complex multielectron transfer process. Therefore, developing efficient, stable, and cost-effective OER catalysts is crucial for advancing hydrogen technologies.1 Noble metal-based catalysts (Ru, Ir, and their oxides) are known for their superior OER activity but are constrained by their high cost and limited availability.2 Consequently, there is a significant research interest in alternative catalysts composed of Earth-abundant materials. Among these, nickel- and iron-based compounds have garnered attention due to their inherent catalytic properties, affordability, tuneable properties, and synergistic effects that enhance OER activity.3,4 However, commercial viability demands not only high activity but also long-term stability at high current densities (≥200 mA cm−2).5,6 High-current-density (HCD) OER poses significant challenges due to the rapid formation of oxygen bubbles, which create concentration gradients, block active sites, and strain porous nanostructures.7 At the same time, harsh oxidative conditions accelerate corrosion, leaching, and structural degradation of the catalyst. Surface reconstruction can sometimes be beneficial, exposing new active sites, but uncontrolled changes often lead to a decline in performance.8,9Despite extensive efforts to enhance OER activity and elucidate the active sites of NiFe-based catalysts, relatively few studies have focused on achieving sustained high current densities (≥200 mA cm−2), which are equally vital for practical applications, and several critical gaps remain in the existing literature. Most existing studies and reviews primarily benchmark catalyst performance at low current densities (typically 10 mA cm−2), often for limited durations and at room temperature, conditions that do not adequately reflect the harsh environments encountered in industrial electrolyzers.10–12 Degradation mechanisms specific to high current regimes, particularly Fe leaching, surface reconstruction, and bubble-induced mechanical stress, remain insufficiently addressed, despite their critical impact on catalyst durability. Although Ni species are largely insoluble in alkaline media, Fe components are susceptible to leaching or segregation, potentially leading to uneven distribution of Fe elements or unwanted doping into the host lattice.13 In addition, most prior reviews predominantly emphasize half-cell OER measurements, with comparatively limited discussion of overall water splitting and full electrolyzer performance. These factors accentuate the need for continuous improvement and development of durable NiFe-based catalysts through advanced strategies, including introducing vacancies to optimize the electronic structure, constructing heterostructures, enhancing interfacial interactions, doping with heteroatoms, etc.13–17
Considering these factors, this work provides an overview of recent design strategies for NiFe-based catalysts that exhibit high activity, stability, and current density, as illustrated in Fig. 1. Moreover, the advancements across various systems, including oxides, (oxy)hydroxides, non-metallic heteroatomic composites, layered double hydroxides (LDHs), metal–organic frameworks (MOFs), and noble metal-doped materials, have been highlighted. Through a systematic examination of recent NiFe-based catalysts, this review elucidates the key factors governing HCD performance, specifically the necessity of balancing the adsorbate evolution mechanism (AEM) and the lattice oxygen mechanism (LOM) pathways, and the role of electronic structure modulation in stabilizing active species.
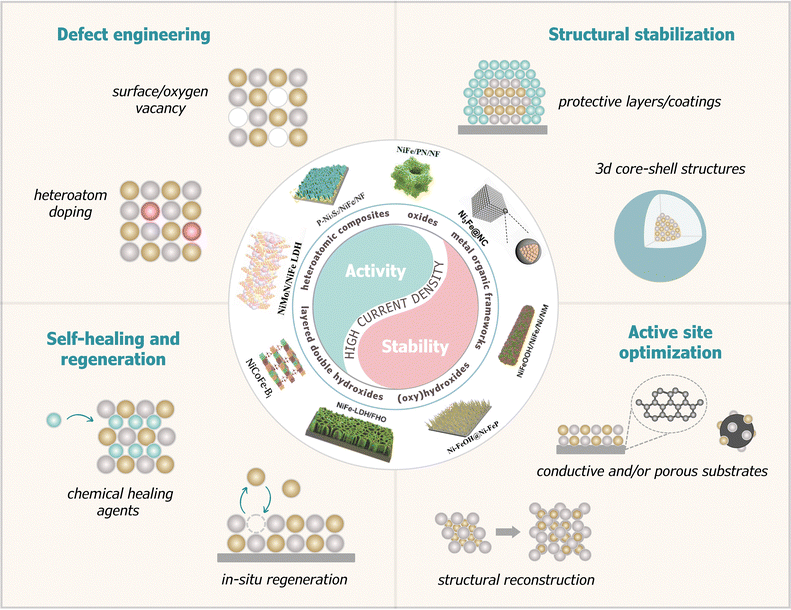 | ||
| Fig. 1 Overview of recent design strategies for NiFe-based catalysts aimed at enhancing OER activity and stability at high current density (HCD). | ||
2. The OER mechanism of NiFe-based catalysts
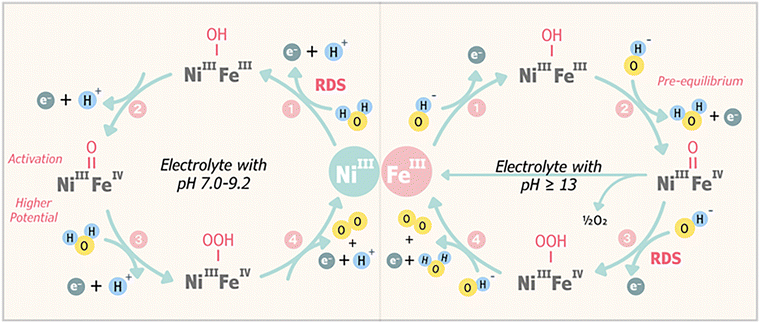
The OER under alkaline conditions constitutes a complex multielectron transfer process with O–O bond formation, rendering it more thermodynamically challenging compared to the two-electron hydrogen evolution reaction (HER). Thus, considerable efforts have been dedicated to deciphering the OER mechanism through kinetic studies, density functional theory (DFT) calculations, and operando or in situ spectroscopic techniques.18–21 DFT is widely used to calculate the Gibbs free energy barriers (ΔG‡) for reaction intermediates and identify the rate-determining step (RDS), offering theoretical insight into the preferred reaction channels. Operando and in situ spectroscopic methods (like Raman or X-ray absorption spectroscopy) are critical for observing dynamic changes in the catalyst structure and electronic environment during the OER, which helps validate computational models and track the formation of active species.22OER mechanisms are typically classified based on the source of oxygen in O–O coupling: the adsorbate evolution mechanism (AEM), where oxygen originates from water molecules adsorbed onto the catalyst surface with metal ions as active sites, and the lattice oxygen mechanism (LOM), where oxygen comes from lattice oxygen ions within the catalyst structure, which themselves serve as active sites.23 In the AEM, the reaction proceeds via sequential proton–electron transfer steps involving intermediates such as *OH, *O, and *OOH, with the metal cations facilitating adsorption and O–O bond formation. The activity of AEM catalysts is often limited by the intrinsic linear scaling relationship between *OH and *OOH adsorption energies, which imposes a theoretical overpotential threshold.24 In contrast, the LOM involves direct participation of lattice oxygen in O–O coupling, generating transient oxygen vacancies that are replenished by water molecules from the electrolyte.25 This pathway can bypass the scaling constraints of the AEM, potentially lowering the energy barrier for the OER, but may introduce structural instability if vacancy regeneration is insufficient. Recent DFT studies on the (001) facet of a nickel ferrite (NiFe2O4) catalyst revealed that the Fe-assisted LOM pathway is the preferred mechanism, exhibiting the lowest energy barrier (ΔG‡ = 0.84 eV at U = 1.63 VSHE) for the RDS involving O–O bond formation.2 Another study proposed an AEM pathway (Fig. 2) for NiFe-based LDH catalysts at different electrolyte pH levels using combined electrochemical and operando spectroscopic measurements.26 In alkaline media (pH ≥ 13), it was found that the adsorption of *OH occurs during the transformation from Ni(OH)2 to NiOOH. The adsorption coverage of OH* is high during the OER, making this step both fast and efficient. Furthermore, a portion of *OH is converted to *O concurrently with the formation of Fe4+, representing the pre-equilibrium step. The RDS involves the nucleophilic attack of OH− on *O (Fe4+![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) to generate M–OOH intermediates (M = Ni or Fe), with the final step being the rapid oxidation of *OOH and the release of O2.
O) to generate M–OOH intermediates (M = Ni or Fe), with the final step being the rapid oxidation of *OOH and the release of O2.
LOM-type mechanisms can enhance OER activity; however, catalysts typically undergo significant surface reconstruction during the LOM process due to the redox and migration of lattice oxygen, leading to potential catalyst dissolution and instability.27 Conversely, catalysts generally exhibit higher stability but lower activity when following the AEM process.28 Achieving sustained HCDs in NiFe-based materials critically relies on controlling the OER pathway between the high-activity, low-stability LOM and the high-stability, low-activity AEM. Specifically, advanced material engineering can be employed to tailor the catalyst's unique electronic structure, optimizing the adsorption of intermediates and effectively striking a balance between the LOM and the AEM to ensure optimal durability and performance.
3. Emerging NiFe-based materials for the high current density OER
Recent advancements have highlighted the potential of NiFe-based systems in achieving HCD with long-term stability for the OER. The synergistic interplay between Ni and Fe active sites within diverse structural frameworks, including oxides, (oxy)hydroxides, non-metallic heteroatomic composites, LDHs, MOF-derived materials, and noble metal-doped compounds, is believed to underpin this exceptional performance. While existing review papers have comprehensively covered NiFe-based electrocatalysts for the alkaline OER,29–31 a systematic analysis of catalysts capable of efficiently delivering HCDs remains limited. The succeeding sections address this gap by outlining robust NiFe-based materials reported over the last three years, prioritizing those demonstrating sustained HCD performance and detailing the synergistic design strategies that maximize stability and activity in commercially relevant environments.3.1. NiFe-based oxides
Studies have consistently demonstrated that NiFe-based oxides exhibit superior activity compared to oxides based solely on Ni or Fe, attributed to a synergistic interplay between Ni and Fe.32 Specifically, Ni lattices provide an ideal framework for anchoring Fe sites due to their exceptional electrical conductivity under anodic potentials.33 Ni thus serves as a conductive scaffold, while Fe incorporation induces structural transformations, creating additional active sites and leading to an enhanced OER at HCDs. A recent theoretical investigation by Friebel et al.34 explored the impact of bond lengths and Löwdin charges by varying Ni![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fe atomic ratios. The study found that Fe substitutes for Ni within the NiO2 structure, forming Ni–O–Fe motifs that minimize overpotential (η) for the OER. Furthermore, a minimum Fe content of 10% has been identified as crucial for significant OER improvements in NiFe-based oxide catalysts.35 Introducing oxygen vacancies (VO) through dual-defect engineering can also enhance the electrical conductivity and intrinsic activity of NiFe-based oxides, leading to low overpotentials and good durability. Recently, Khatun et al.36 developed a surface-modified Cr-doped FeNi3/NiFe2O4 heterojunction OER catalyst rich in VO. The generated VO is believed to modulate the electronic configurations, creating numerous unsaturated coordinated metal active sites (Fig. 3a). This enhances the adsorption ability of oxygen intermediates, resulting in robust OER performance (η500 = 360 mV) and achieving sustainable HCD (200 h at 1000 mA cm−2). It is also noteworthy that during the OER with NiFe-based oxides, intrinsic VO can induce spontaneous electrochemical surface reconstruction, generating highly reactive (oxy)hydroxide (M–OOH) species, which are also recognized as active OER sites.37
Fe atomic ratios. The study found that Fe substitutes for Ni within the NiO2 structure, forming Ni–O–Fe motifs that minimize overpotential (η) for the OER. Furthermore, a minimum Fe content of 10% has been identified as crucial for significant OER improvements in NiFe-based oxide catalysts.35 Introducing oxygen vacancies (VO) through dual-defect engineering can also enhance the electrical conductivity and intrinsic activity of NiFe-based oxides, leading to low overpotentials and good durability. Recently, Khatun et al.36 developed a surface-modified Cr-doped FeNi3/NiFe2O4 heterojunction OER catalyst rich in VO. The generated VO is believed to modulate the electronic configurations, creating numerous unsaturated coordinated metal active sites (Fig. 3a). This enhances the adsorption ability of oxygen intermediates, resulting in robust OER performance (η500 = 360 mV) and achieving sustainable HCD (200 h at 1000 mA cm−2). It is also noteworthy that during the OER with NiFe-based oxides, intrinsic VO can induce spontaneous electrochemical surface reconstruction, generating highly reactive (oxy)hydroxide (M–OOH) species, which are also recognized as active OER sites.37
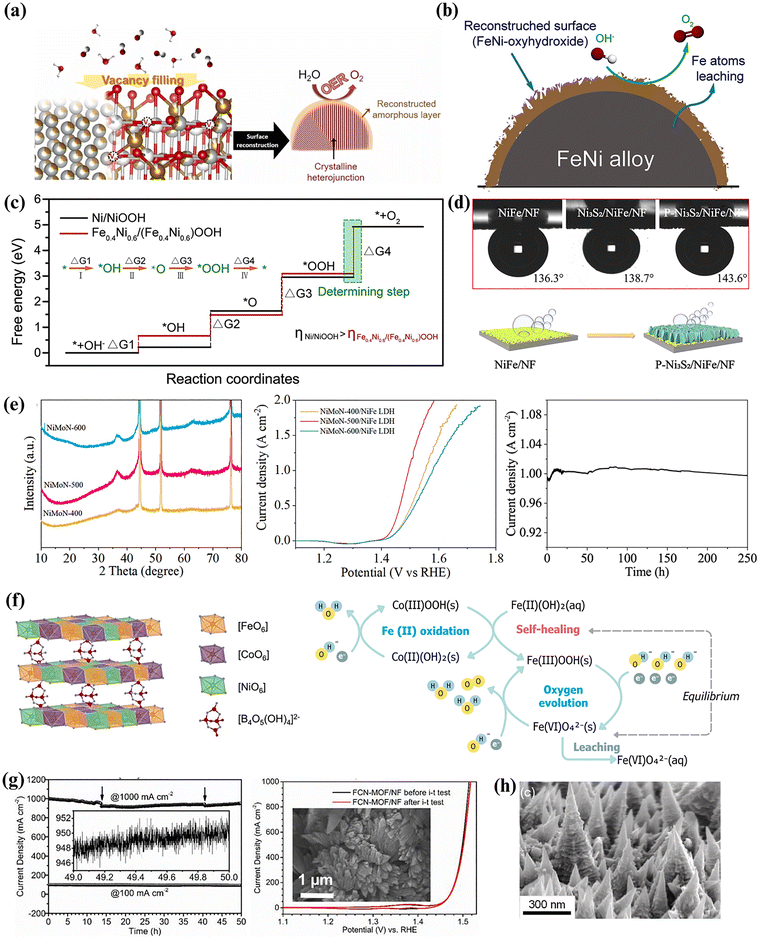 | ||
| Fig. 3 (a) Schematic illustration of vacancy filling in Cr-doped FeNi3/NiFe2O4 Mott–Schottky heterojunctions, accompanied by surface reconstruction that enhances OER performance. Reproduced from ref. 36. Copyright 2024, Elsevier. (b) Schematic illustration of partial Fe leaching and surface reconstruction in FexNi1−x alloy fiber paper. Reproduced from ref. 43. Copyright 2021, Elsevier. (c) Gibbs free energy diagram illustrating the four OER steps on Fe0.4Ni0.6 and Fe0Ni1.0 alloy fiber papers. Reproduced from Ref. 43. Copyright 2021, Elsevier. (d) Underwater aerophobicity of NiFe/NF, Ni3S2/NiFe/NF, and P–Ni3S2/NiFe/NF electrodes (top), and a schematic of their O2 bubble adhesion behaviour (bottom). Reproduced from ref. 46. Copyright 2022, Elsevier. (e) XRD pattern of NiMoN–T and OER polarization curve of NiMoN–T/NiFe LDH (T represents the nitridation temperature). Chronoamperometry curve of NiMoN/NiFe LDH for 250 h. Reproduced from Ref. 54. Copyright 2023, Springer Nature. (f) Schematic illustration of the structure of NiCoFe–Bi and its self-healing mechanism, catalysed by Co. Reproduced from ref. 68. Copyright 2021, Springer Nature. (g) Chronoamperometric stability and cyclic voltammetry curves of FCN-MOF/NF at 1000 mA cm−2. Reproduced from ref. 72. Copyright 2020, Elsevier. (h) Scanning electron microscopy (SEM) image of NiFe nanocones with a superhydrophilic surface, enabling rapid mass transfer in an industrial water electrolyzer. Reproduced from ref. 85. Copyright 2024, Elsevier. | ||
NiFe-based oxides exhibit superior OER performance due to synergistic Ni–Fe interactions, where Ni provides a conductive framework and Fe creates highly active sites that lower overpotential, particularly at HCDs. Optimal Fe incorporation and oxygen vacancy engineering further enhance activity and durability by tuning the electronic structure, promoting active Ni–O–Fe motifs, and enabling dynamic surface reconstruction into catalytically active (oxy)hydroxide species.
3.2. NiFe-based (oxy)hydroxides
Early research on NiFe (oxy)hydroxide catalysts predominantly focused on powder forms. To enhance their performance, researchers have previously explored various strategies, including optimizing composition,38 incorporating conductive carbon-based supports,39 introducing structural/lattice defects,40etc. However, the inherent limitations of powder-based catalysts, such as durability and mass transport challenges, have hindered their practical applications in demanding environments like HCD electrolysis.41 Due to these challenges, research has shifted towards developing alternative catalyst architectures. One promising approach involves the direct growth of catalysts on a conductive porous substrate/support. For instance, a recent study by Wu et al.42 demonstrated the formation of NiFeOOH on a NiFe/Ni film through anodic electro-oxidation. The NiFe/Ni film, fabricated via a dynamic hydrogen bubble-templated process, facilitated efficient electron transport due to its high conductivity and served as a robust support for the catalyst layer. Additionally, the three-dimensional laminar porous structure of the film enhanced mass transport, reduced interfacial resistance, and maximized exposure of active sites. These combined factors contributed to the catalyst's enhanced performance (η1000 = 300 mV), enduring 100 h of testing at 500 mA cm−2.Interestingly, while Fe leaching is often considered detrimental to the stability of NiFe-based catalysts, recent studies have revealed its potential to enhance catalytic performance. Zhang et al.43 showed that the partial dissolution/leaching of surface Fe atoms in a FeNi alloy exposes Ni sites (Fig. 3b), promoting lattice oxygen formation, which subsequently transforms into (oxy)hydroxides with low overpotential (η100 = 287 mV). Specifically, it activates the initially inactive Ni component, inducing the surface reconstruction of a FeNi–(oxy)hydroxide/alloy heterojunction with longer N–O bonds within the (oxy)hydroxide structure compared to pure NiOOH. This significantly lowered the energy barrier for *OOH intermediate decomposition (Fig. 3c), enabling stable operation at HCD (80 h at 250 mA cm−2). Meanwhile, Kang et al.44 recently demonstrated that surface functionalization of NiFe (oxy)hydroxide catalysts (FSN) with tetraphenylporphyrin (TPP) significantly enhances their durability. O-labelling experiments combined with in situ Raman spectroscopy revealed that TPP does not alter the OER pathway of FSN, indicating that TPP's stability enhancement mechanism is unrelated to a shift from the LOM to the AEM. Instead, TPP functions as a protective layer, inhibiting the protonation of Fe–O sites and consequently reducing Fe dissolution. This protective effect enabled the TPP–FSN to maintain HCD (110 h at 500 mA cm−2).
Another effective strategy to enhance performance in these systems is to design three-dimensional nanostructured catalysts that enlarge the specific surface area, expose more active sites, and create heterojunctions between the metallic core and the (oxy)hydroxide shell to improve charge transport. For example, hierarchical FeNi3@(Fe,Ni)S2 core–shell heterojunctions with a tunable shell thickness were fabricated through NiFe(OH)x reduction followed by partial sulfidization.45 The analysis revealed that the FeNi3 core substantially increases the conductivity, acting as an electron highway, while the (Fe,Ni)S2 shell provides an active surface for the in situ generation of S-doped NiFe (oxy)hydroxides with remarkable OER activity (η100 = 288 mV) and stability (1200 h at 200 mA cm−2). Notably, S-doping was also found to improve the conductivity of the catalyst, as evidenced by the reduced bandgap of S–NixFe1−xOOH compared to NixFe1−xOOH, contributing to the enhancement of catalytic performance.
NiFe-based (oxy)hydroxide catalysts have evolved from powder systems toward engineered architectures that address durability and mass-transport limitations at HCDs. Strategies such as direct growth on conductive porous substrates, controlled Fe leaching, surface protection to suppress Fe dissolution, and 3D core–shell or heterojunction designs collectively enhance conductivity, active-site exposure, and structural stability, enabling sustained OER operation under industrially relevant conditions.
3.3. Non-metallic heteroatomic combinations
To boost intrinsic activity and stability, pristine catalysts often require surface and/or structural modifications by introducing heteroatoms like P, S, N, and C to form stable functional groups. Oxidation states and electronic structures are modified, generating defects that aid HCD operation.Meanwhile, integrating P into the S structure induces electron transfer from Ni to S via heterointerface formation, creating favourable hydride-acceptor (Niδ+) and proton-acceptor (Sδ−–Pδ−) sites. This optimized electronic configuration, coupled with improved aerophobicity, enables the catalyst to achieve exceptional activity (η1000 = 372 mV) and stability at HCD (20 h at 1000 mA cm−2). Additionally, the –PO4 groups contribute by lowering the free-energy barrier for the OER at NiFe sites. Other works have also shown that phosphate ions in LDHs improve mass transfer and catalyst durability by mitigating metal leaching caused by local OH− depletion.49 Similarly, Sheng et al.50 observed improved overall durability in a NiFe–P catalyst despite slight structural instability. The catalyst films, synthesized via flame aerosol deposition, were initially encapsulated by a carbonaceous (soot-like) matrix. The controlled electrochemical removal of this carbon under OER conditions triggered a self-refreshing mechanism, causing gradual and mild detachment of the film material while continuously exposing fresh catalytic sites. This dynamic process mitigated the selective leaching of Fe, preserving an Fe-rich, highly active surface. As a result, high activity (η10 = 245 mV) and stability (100 h at 500 mA cm−2) were achieved at HCD.
NiFe–P catalysts have spurred considerable research interest in recent years due to their bifunctional activity towards both the OER and the HER across various pH conditions. Recently, Hu et al.51 developed a core–shell Ni–FeOH@Ni–FeP bifunctional catalyst for overall water splitting (OWS) and proposed a reaction mechanism in which FeOOH transiently forms at Fe sites with a low theoretical overpotential (0.14 eV). However, the subsequent O2 release from FeOOH is hindered by a high overpotential (0.63 eV), whereas the transition from FeOOH to NiOOH occurs with a lower energy barrier (≈0.43 eV), and O2 release from NiOOH is more favourable (0.2 eV). The presence of Fe in the Ni–FeOH@Ni–FeP electrode facilitates NiOOH formation, thereby enhancing OER activity. Additionally, the hydroxide layer interacts with the phosphide core, modulating electronic structures and surface microenvironments. This interaction regulates FeOOH formation rather than completely suppressing it, ensuring a balance between active site regeneration and catalytic efficiency. Hence, the catalyst exhibited remarkable stability at HCDs of 750 and 935 mA cm−2 in OWS.
NiFe-based phosphide catalysts enhance the HCD OER by optimizing triple-phase interfaces through improved aerophobicity, porous architectures, and interfacial electronic modulation. Phosphorus incorporation and heterointerface formation promote efficient charge transfer, favorable intermediate energetics, suppressed metal leaching, and self-refreshing or stabilized active surfaces, enabling high activity and durability at industrially relevant current densities.
Sulphide incorporation and sulphur vacancy engineering in NiFe catalysts enhance the HCD OER by tuning electronic structures, improving conductivity, and enabling in situ formation of active oxysulphide and high-valence Ni4+/Fe4+ species. These dynamic transformations expose new active sites, stabilize sulphur vacancies, and promote oxygen-bridged Ni–O–Fe intermediates, resulting in high activity and durability under demanding electrolysis conditions.
Nitride formation and N-doping enhance NiFe-based OER catalysts at HCDs by strengthening reactant interactions, tuning electronic structures, and improving conductivity and charge transport. Core–shell nitride or hydroxide architectures demonstrate exceptional HCD stability, although long-term durability data at industrially relevant current densities remain limited in the literature.
Carbon integration in NiFe-based catalysts enhances OER performance by improving electrical conductivity, exposing active sites, and stabilizing catalysts under harsh conditions. Graphitized or porous carbon structures, coupled with dopants or alloy encapsulation, protect active phases, suppress agglomeration, and lower OER energy barriers.
3.4. NiFe-based layered double hydroxides
For LDHs or other NiFe-based materials in general, the synergistic effect among multiple metal ions is not limited to Ni and Fe. Adding a third metal may further improve the catalytic performance by effectively changing the adsorption/desorption energy and surface electronic structure of LDH catalysts.60 Wu et al.61 employed a ternary NiFeCo LDH through operando incorporation of Co2+ into the NiFe LDH catalyst (NiFeCo-0.6), identifying 0.6 mmol as the optimal Co-doping content. Here, Co-doping reduced the Ni oxidation potential and accelerated the formation of highly active Ni(Co)1−xFexOOH species during the OER, resulting in lower overpotential (η1000 = 353 mV) compared to pristine NiFe LDH (η1000 = 502 mV). Co-doping also enabled NiFeCo-0.6 to follow the LOM pathway with a lower RDS Gibbs free energy (0.35 eV) than that of NiFeOOH (1.23 eV). Additionally, the Fe dissolution rate in NiFeCo-0.6 was lower than that of NiFe LDH, indicating greater stability. It showed that Co-doping stabilized the Fe local coordination environment, reducing dissolution and enhancing the overall stability of the NiFeCo-0.6 catalyst (300 h at 500 mA cm−2). Similarly, various metals have also been used to improve the performance of NiFe-based LDHs.62–64 To fully optimize trimetallic LDH systems, a comprehensive understanding of the interplay between metal ion species within the brucite layers and the nature of the interlayer anions is crucial, as these factors collectively influence activity and stability.Defect modification can also be an efficient and cost-effective way to achieve high-performance NiFe LDHs. According to the DFT study of Mohammed-Ibrahim,65 creating unsaturated NiFe coordination sites in LDHs through defect engineering of Ni–O–Fe coupling at the atomic scale can reduce the Gibbs free energy of the deprotonation step in the OER. In another study by Wang et al.,66 a hexagonal NiFe–LDH/FeOOH heterostructure with oxygen defects was synthesized using a facile hydrothermal method, based on the in situ competing growth strategy of bimetallic ions and OH− ions. Oxygen defects were found to enhance OH− nucleophilic attack and promote synergy at Fe–O sites, particularly between the AEM and the LOM, thereby maximizing the utilization of surface metals and oxygen atoms. The projected density of state (PDOS) analysis showed that introducing VO shifts the Fe-3d band center down, reducing over-strong adsorption and weakening the Fe–O bond, thus enhancing both AEM and LOM catalytic performance. As a result, the catalyst exhibited low overpotential (η1000 = 353 mV) and remarkable stability (20 h at 1000 mA cm−2) at HCDs.
Another emerging strategy to mitigate the instability of NiFe-based LDH materials involves investigating their capacity for self-repair or regeneration. Recent studies have demonstrated the potential of such systems to restore catalytic activity and maintain stability at HCDs.67–69 For instance, the presence of both ferrous and borate ions in the electrolyte was critical for the self-healing of a borate-intercalated NiCoFe–LDH (NiCoFe–Bi).68 The addition of ferrous ions restored Fe content by activating a self-healing mechanism (Fig. 3f) where Co catalyses the redeposition of Fe oxyhydroxide, compensating for the leaching of Fe active sites. Concurrently, borate ions prevented further degradation by replenishing the depleted borate ions within the LDH structure, resulting in sustained stability (100 h at 500 mA cm−2). However, the exploration of the self-healing ability of NiFe-based materials remains relatively untapped and requires further investigation, as their effectiveness is contingent on factors including catalyst properties, electrolyte and buffer composition, pH, applied current and potential, temperature, and mass transport conditions.
NiFe-based LDH performance at HCDs can be significantly enhanced through multimetal synergy, defect engineering, and emerging self-healing strategies. Trimetallic doping (e.g., Co) tunes the electronic structure, lowers overpotential, suppresses Fe dissolution, and stabilizes active oxyhydroxide species, while oxygen vacancies optimize adsorption energetics and promote dual AEM/LOM pathways; additionally, electrolyte-assisted self-repair mechanisms offer a promising route to sustain long-term stability under harsh OER conditions.
3.5. MOF-derived NiFe-based catalysts
NiFe-based MOFs often suffer from leaching/exfoliation under testing conditions, significantly impacting long-term stability.70 To address this, the interaction between catalysts and support should be reinforced, as the adhesion between them may be insufficient to withstand the stresses induced by gas bubble formation during HCD operations.71 One of the effective strategies to tackle this issue is the in situ growth of catalysts on conductive 3D porous substrates. For example, equimolar Fe-, Co-, and Ni(FCN)-based trimetallic MOFs, grown in situ on NF, exhibited exceptional OER activity (η1000 = 284 mV), operating at a HCD of 1000 mA cm−2 with only a 5% decay after 50 h (Fig. 3g).72 The binder-free, in situ growth strategy enhanced both mechanical adhesion and intrinsic activity by promoting synergistic interactions among the well-dispersed metal ions within the MOF structure. The intrinsically low electrical conductivity and mass permeability of many NiFe-based MOFs limit their direct use as catalysts. Instead, these materials are often used as precursors to derive composites containing NiFe derivatives as active species through high-temperature thermal treatments. The incorporation of good conductors, such as carbon nanotubes (CNTs) and graphene, has also been explored to improve OER performance, as their higher degree of graphitization provides better structural stability and resistance to carbon corrosion induced by oxidation currents. A recent study by Wang et al.73 demonstrated the effectiveness of anchoring FeNi LDH nanoflakes onto MOF-derived CNT networks to create stable, self-supported catalysts, achieving low overpotential (η10 = 200 mV) and robust stability for over 160 h at 1000 mA cm−2. Meanwhile, the scalability and cost-effectiveness of MOF-derived NiFe catalysts pose challenges for practical applications. Most of their metal oxides are produced via calcination with low yields.74 Thus, exploring new synthesis strategies to improve product yield and preparation efficiency is both important and urgent.MOF-derived NiFe catalysts address the instability and poor conductivity of pristine MOFs by strengthening catalyst–support interactions and enhancing charge transport. Strategies such as in situ growth on conductive 3D substrates and conversion into carbon- or CNT-integrated NiFe derivatives improve mechanical adhesion, activity, and durability at HCDs, although scalability and low-yield synthesis remain key challenges for practical deployment.
3.6. Noble metal-containing NiFe-based catalysts
In recent years, several groups have reported that integrating noble metals (NMs) into NiFe-based catalysts offers a promising avenue for enhancing activity and durability while mitigating the reliance on these scarce and costly resources.75,76 While NM oxides like RuO2 exhibit superior OER activity, their instability under harsh operating conditions remains a critical challenge.77 To address these, researchers have explored strategies to maximize the utilization of NMs through size reduction and structural optimization.76,78,79Single-atom catalysts (SACs) represent the ultimate limit in terms of particle size and offer the potential to overcome the cost and stability challenges associated with traditional NM-based catalysts.80 Recent studies have demonstrated the feasibility of incorporating NM single atoms (SAs) into complex nanostructures to achieve exceptional OER performance.81 For instance, Wang et al.78 developed a bifunctional catalyst comprising RuSAs integrated into a Ni(OH)2–FeOOH nanowire heterostructure with enhanced stability. The unique architecture exhibited superior aerophobicity, facilitating efficient mass transport and bubble removal, which is a common cause of active layer degradation. The synergistic interplay among RuSAs, FeOOH clusters, and the porous nanostructure resulted in enhanced active site exposure (η1000 = 386 mV) and accelerated reaction kinetics, leading to a minimal overpotential increase after 140 h of stability testing at 500 mA cm−2. In another aspect, the structural mismatch between LDHs can generate abundant defects, providing ideal supports and unique anchoring sites for NM atoms. Van et al.75 demonstrated that the strong electronic interaction between lanthanum (La) and the host metals in a NiFe LDH not only considerably increased the number of catalytic sites and reduced charge transfer resistance but also elevated Fe d-band levels and created more VO than in pristine NiFe catalysts, leading to superior activity (η500 = 309 mV) and long-term stability (600 h at 500 mA cm−2). Similarly, Hu et al.79 reported an IrSAC–NiFe–LDH catalyst with long-term stability (120 h at 200 mA cm−2), where charge redistribution at the active Ni sites (induced by the high-valence IrSAs) lowered the energy barrier of *OOH dissociation via the AEM pathway, thereby facilitating the fast reaction kinetics of IrSAC–NiFe–LDH. Beyond serving as anchoring sites, the interaction between monatomic NM atoms and the NiFe-based support in determining activity/stability at HCDs is still elusive for SACs, which should be of critical importance for maximizing the efficiency of NMs and exploring novel properties.
Incorporating noble metals into NiFe-based catalysts, especially as single-atom sites, enhances OER activity and durability by improving active-site exposure, accelerating reaction kinetics, and facilitating mass transport. Strategies like size reduction, defect-rich supports, and heterostructure engineering maximize noble-metal utilization, while electronic interactions with the NiFe host further optimize activity and stability at HCDs, though the full mechanisms in single-atom systems remain underexplored.
As a summary, Table 1 lists the catalytic activity and HCD stability of recently reported NiFe-based catalysts designed for HCD operations. For the alkaline OER, strategies such as heteroatom doping, heterostructure formation, and defect/vacancy engineering have proven to be effective in modulating the physical, electronic, chemical, and surface properties of catalysts. These modifications directly influence intermediate adsorption/desorption, the OER mechanism, and charge transfer kinetics, leading to substantial enhancements in intrinsic activity. Additionally, employing techniques like in situ growth on conductive substrates, surface wettability engineering, and the creation of 3D porous architectures can significantly extend the catalyst lifespan and boost overall performance, aligning with the commercial requirements of practical water electrolysis.
| Catalyst | Design strategy used | Overpotential | Stability test | Electrolyte | Remark after the stability test |
|---|---|---|---|---|---|
| a Catalyst performance was tested in an alkaline electrolyzer or under simulated industrial conditions. | |||||
NiFeOOH/NiFe/Ni42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
Core–shell heterostructure | 300 mV at 1000 mA cm−2 | 100 h at 500 mA cm−2 | 1 M KOH | Small voltage decrease |
| P–Ni3S2/NiFe/NF46 | Phosphorous-doped (or phosphide) heterostructure | 372 mV at 1000 mA cm−2 | 20 h at 1000 mA cm−2 | 1 M KOH | Pristine structure and morphology were retained |
| NiMoN/NiFe LDH54 | Core–shell heterostructure/nitrogen doping | 266 mV at 1000 mA cm−2 | 250 h at 1000 mA cm−2 | 1 M KOH | Negligible degradation |
| NiFe–P-1.8850 | Phosphorous (or phosphide) doping | 360 mV at 1000 mA cm−2 | 100 h at 500 mA cm−2 | 1 M KOH | Reasonable stability |
| CeF3–NiFe LDH91 | Heteroatom doping/surface modification | 340 mV at 1000 mA cm−2 | 90 h at 500 mA cm−2 | 1 M KOH | No significant change |
NiFe–S-0.652![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
Sulphur (or sulphide) doping | 235 mV at 1000 mA cm−2 | 140 h at 500 mA cm−2 | 6 M KOH | Only 8.9% increase in potential |
| NiFeCo-0.661 | Ternary LDH/co-doping/optimized LOM pathway | 353 mV at 1000 mA cm−2 | 300 h at 500 mA cm−2 | 1 M KOH | Negligible degradation |
WO2Ni17W3/NiFe(OH)x/NF92![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
Core–shell heterostructure/heteroatom doping | 338 mV at 1000 mA cm−2 | 120 h at 1000 mA cm−2 | 6 M KOH | Negligible potential rise |
| Ni3S4/FeS (FNF-0.5 V)93 | Sulphide-doped heterostructure | 395 mV at 1000 mA cm−2 | 200 h at 1500 mA cm−2 | 1 M KOH | No significant degradation |
| MoS2/NiFeS2/NF94 | Sulphide-doped heterostructure | 314 mV at 1000 mA cm−2 | 35 h at 500 mA cm−2 | 1 M KOH | Only a 76 mV increase in potential |
| NiFe–LDH/FHO95 | LDH heterostructure | 248 mV at 500 mA cm−2 | 1000 h at 500 mA cm−2 | 1 M KOH | Only a 30 mV increase in potential |
NiFeLa LDH-275![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
Heteroatom doping/defect engineering | 309 mV at 500 mA cm−2 | 600 h at 500 mA cm−2 | 1 M KOH | No significant degradation |
| Cr-doped (4 mol%) FeNi3/NiFe2O436 | Oxide heterojunction/dual-defect engineering | 360 mV at 500 mA cm−2 | 200 h at 1000 mA cm−2 | 1 M KOH | No noticeable decay |
| Fe,N–Ni(OH)2/NF6 | Nitrogen doping | 340 mV at 500 mA cm−2 | 10 h at 500 mA cm−2 | 1 M KOH | Excellent potential sustention |
NiFe/PN/NF96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a a |
Phosphorous/nitrogen-doped (dual-doping) | 297 mV at 500 mA cm−2 | 40 h at 500 mA cm−2 | 6 M KOH | Only a 20 mV increase in potential |
Importantly, the performance descriptors governing NiFe-based electrocatalysts vary significantly across different electrochemical processes. In half-cell OER measurements, activity is primarily governed by intrinsic parameters such as the Fe coordination environment, electronic structure modulation, and adsorption energetics of oxygen intermediates.37 However, under bifunctional operation or full-cell electrolysis, additional parameters, including catalyst–support adhesion, bubble management, mass transport, and structural robustness under continuous polarization, become equally critical.82 For instance, strategies that promote surface reconstruction and lattice oxygen participation may enhance intrinsic OER kinetics in half-cell tests but can accelerate Fe dissolution and structural degradation during prolonged high-current operation in electrolyzers.7 This process-dependent behavior highlights the necessity of tailoring NiFe catalyst design according to the targeted electrochemical configuration rather than relying solely on half-cell performance metrics.
4. Scale-up technologies
Shifting focus towards large-scale systems, the performance and durability of NiFe-based catalysts in a full electrolyzer cell may deviate from half-cell tests due to variations in pH, temperature, and pressure.While HCDs can enhance reaction kinetics, they also entail higher investment costs, requiring a balance between cost and efficiency to optimize current density. Moreover, concentration polarization and severe localized stresses induced by rapid gas bubble generation in HCDs can compromise catalyst stability, ultimately leading to inactivity.83,84 To ensure the longevity and efficiency of electrolyzers, it is essential to examine the catalyst performance within the context of the entire system's operations. Liu et al.85 employed an alkaline electrolyzer using an optimized NiFe alloy anode and NiMo cathode, operating at 80 °C with a 30 wt% KOH electrolyte. The superhydrophilic structure of the NiFe catalyst (Fig. 3h) helped enhance mass transfer and likely minimized gas bubbles that could obstruct active sites during operation. This led to a low cell voltage (1.841 V at 500 mA cm−2) and reduced energy consumption (4.4 kWh Nm−3 H2). In addition to its high activity, the NiFe//NiMo electrolyzer demonstrated exceptional durability, maintaining stable operation at HCD (50 h at 500 mA cm−2). Similarly, Ha et al.86 developed a NiFeV-oxide thin-film electrocatalyst via magnetron sputtering, introducing oxygen vacancies that enhanced catalytic activity. Operando Raman spectroscopy revealed that these vacancies, along with vanadium leaching, promoted surface reconstruction into NiFe(oxy)hydroxide, optimizing water adsorption and accelerating the OER. The single-stack NFV-0.7(−)‖NFV-0.7(+) electrolyzer, operating at 60 °C, achieved a low cell voltage (1.84 V at 1000 mA cm−2), attributed to the formation of VO species resulting from vanadium substitution into NiFe, as well as robust stability at the same current density for 50 h, demonstrating its potential for industrial-scale electrolysis. In another study, Fe ions were introduced as electrolyte additives to promote the activity of NiFe LDH in an anion exchange membrane water electrolyzer, resulting in a low cell voltage of 1.73 V and a high energy conversion efficiency of 69.4% at 3000 mA cm−2.87 Additional comprehensive stability assessments under simulated industrial conditions have recently been carried out, offering more reliable quantitative data and enabling a more effective comparison of the scalability potential of NiFe-based catalysts.88–90
5. Conclusions and future perspectives
The development of NiFe-based catalysts for the oxygen evolution reaction (OER) has progressed rapidly, yet achieving simultaneous high activity and long-term stability at high current densities (HCDs) remains a critical challenge. Stability is as crucial as intrinsic catalytic performance, particularly under harsh oxidative conditions encountered in industrial water electrolysis. Protective coatings, such as carbon-based layers and metal oxides, can shield NiFe catalysts from corrosion and structural degradation, but their effectiveness depends on chemical robustness and electrical conductivity. Core–shell architectures, in which a stable shell encases a reactive NiFe core, can extend the operational lifespan by preserving the active core, yet the precise mechanisms that govern activity retention remain poorly understood. To fully understand the precise mechanism by which performance is enhanced in these systems, further in-depth investigations are still necessary. Future research must employ advanced operando and in situ characterization techniques to definitively resolve the precise OER mechanism in complex systems, such as core–shell structures. Crucially, the location of the true active site, whether it resides on the conductive core, the protective shell, or the reconstructed interface, remains a subject of debate that must be clarified to enable rational catalyst optimization.High catalytic performance also requires large surface areas and dense active sites, ideally integrated on three-dimensional conductive porous substrates to enhance mass transport, electron conductivity, and bubble management. Gas bubble accumulation at HCDs can impede reaction kinetics and damage nanostructures. Strategies including surface (wettability) engineering, hierarchical porosity, superaerophobic or superhydrophilic interfaces, conductive scaffolds, and flow-assisted bubble removal can improve gas evacuation, stability, and overall performance. Structural reconstruction during the OER can either expose new active sites or degrade catalysts, and its beneficial aspects can be further optimized through defect engineering, alloying, heteroatom doping, and self-healing materials, enhancing durability under demanding conditions.
Despite these advances, key challenges remain. A comprehensive understanding of degradation mechanisms at HCDs, including corrosion, leaching, structural collapse, and bubble-induced stress, is lacking. Moreover, bridging the gap between the lab and industrial OER requires a standardized benchmark catalyst for HCD evaluation under industry-relevant conditions, including elevated temperatures (60–90 °C), high pressure (6–200 bar), and high alkaline electrolyte concentrations (20–30 wt%). Lastly, developing scalable and energy-efficient synthesis methods for the catalytic electrode is equally important for economic viability.
Conflicts of interest
There are no conflicts to declare.Data availability
As this is a review article, no primary research results, data, software, or code have been included.Acknowledgements
The authors would like to acknowledge the support from the CIPHER Project (IIID 2018-008) funded by the Commission on Higher Education – Philippine California Advanced Research Institutes (currently CHED–LAKAS Program). This research was also supported by the Korea Institute of Energy Technology Evaluation and Planning (KETEP) granted financial resources from the Ministry of Trade, Industry & Energy, Republic of Korea (20213030040590).Notes and references
- F. Zeng, C. Mebrahtu, L. Liao, A. K. Beine and R. Palkovits, J. Energy Chem., 2022, 69, 301–329 CrossRef CAS.
- Ö. N. Avcl, L. Sementa and A. Fortunelli, ACS Catal., 2022, 12, 9058–9073 CrossRef PubMed.
- C. Kong, C. Zhi, Z. Wu, W. Yang, J. Yang and Z. Sun, J. Colloid Interface Sci., 2024, 665, 863–870 Search PubMed.
- C. Gort, P. W. Buchheister, M. Klingenhof, S. D. Paul, F. Dionigi, R. van de Krol, U. I. Kramm, W. Jaegermann, J. P. Hofmann, P. Strasser and B. Kaiser, ChemCatChem, 2023, 15, e202201670 Search PubMed.
- M. R. Domalanta, J. N. Bamba, D. D. Matienzo, J. A. del Rosario-Paraggua and J. Ocon, ChemSusChem, 2023, 16, e202300310 CrossRef PubMed.
- P. Shi, X. Cheng and S. Lyu, Chin. Chem. Lett., 2021, 32, 1210–1214 Search PubMed.
- Y. Yao, J. Lyu, X. Li, C. Chen, F. Verpoort, J. Wang, Z. Pan and Z. Kou, DeCarbon, 2024, 5, 100062 Search PubMed.
- Y. Zeng, M. Zhao, Z. Huang, W. Zhu, J. Zheng, Q. Jiang, Z. Wang and H. Liang, Adv. Energy Mater., 2022, 12, 2201713 CrossRef CAS.
- X. Luo, X. Tan, P. Ji, L. Chen, J. Yu and S. Mu, EnergyChem, 2023, 5, 100091 CrossRef CAS.
- R. Chen, S. F. Hung, D. Zhou, J. Gao, C. Yang, H. Tao, H. Bin Yang, L. Zhang, L. Zhang, Q. Xiong, H. M. Chen and B. Liu, Adv. Mater., 2019, 31, 1903909 CrossRef CAS PubMed.
- T. Yang, J. Huang, Z. Liu and C. Chen, Chem. Commun., 2025, 61, 11681–11684 Search PubMed.
- E. Brim, K. K. Rücker, D. H. Taffa, D. W. Hayes, O. Bisen, S. Alia, M. Risch, J. Lorenz, C. Harms, M. Wark and R. M. Richards, Chem. Commun., 2026, 62, 763–774 RSC.
- Y. Han, J. Wang, Y. Liu, T. Li, T. Wang, X. Li, X. Ye, G. Li, J. Li, W. Hu and Y. Deng, Carbon Neutralization., 2024, 3, 172–198 CrossRef CAS.
- A. Hao, Z. Liu and X. Ning, J. Alloys Compd., 2024, 1003, 175523 CrossRef CAS.
- M. Görlin, P. Chernev, P. Paciok, C. W. Tai, J. Ferreira de Araújo, T. Reier, M. Heggen, R. Dunin-Borkowski, P. Strasser and H. Dau, Chem. Commun., 2019, 55, 818–821 RSC.
- S. Wang, A. Hao and Z. Liu, ACS Appl. Nano Mater., 2024, 7, 28602–28611 CrossRef CAS.
- Z. Cai, M. Xu, Y. Li, X. Zhou, K. Yin, L. Li, B. Jia, L. Guo and H. Zhao, Carbon Energy, 2024, 6, e543 CrossRef CAS.
- J. Geppert, P. Röse, S. Czioska, D. Escalera-López, A. Boubnov, E. Saraçi, S. Cherevko, J. D. Grunwaldt and U. Krewer, J. Am. Chem. Soc., 2022, 144, 13205–13217 Search PubMed.
- M. Berger, I. M. Popa, L. Negahdar, S. Palkovits, B. Kaufmann, M. Pilaski, H. Hoster and R. Palkovits, ChemElectroChem, 2023, 10, e202300235 CrossRef CAS.
- J. Qu, X. Hu, M. Deconinck, L. Liu, Y. Cheng, R. Zhao, M. Wang, H. Zhang, Y. Vaynzof, J. Schuster, A. Cabot, K. Leistner and F. Li, ChemCatChem, 2025, 17, e202401667 CrossRef CAS.
- M. Nozari-Asbemarz, H. Imanzadeh, L. Hazraty, B. Babaei, A. Abbasi, S. S. Mehrabi-Kalajahi, M. A. Vafolomeev, J. J. Leahy and M. Amiri, J. Power Sources, 2025, 652, 237555 CrossRef CAS.
- U. Shamraiz, B. Raza and A. Badshah, Mater. Today Energy, 2025, 53, 102006 Search PubMed.
- Y. J. Son, K. Kawashima, R. A. Márquez, L. A. Smith, C. E. Chukwuneke and C. B. Mullins, Curr. Opin. Electrochem., 2023, 39, 101298 Search PubMed.
- Y. Pan, X. Xu, Y. Zhong, L. Ge, Y. Chen, J. P. M. Veder, D. Guan, R. O’Hayre, M. Li, G. Wang, H. Wang, W. Zhou and Z. Shao, Nat. Commun., 2020, 11, 2002 CrossRef CAS PubMed.
- F. Wu, F. Tian, M. Li, S. Geng, L. Qiu, L. He, L. Li, Z. Chen, Y. Yu, W. Yang and Y. Hou, Angew. Chem., Int. Ed., 2025, 64, e202413250 CrossRef CAS PubMed.
- Q. Xie, D. Ren, L. Bai, R. Ge, W. Zhou, L. Bai, W. Xie, J. Wang, M. Grätzel and J. Luo, Chin. J. Catal., 2023, 44, 127–138 Search PubMed.
- Z. Wang, W. A. Goddard and H. Xiao, Nat. Commun., 2023, 14, 4228 CrossRef CAS PubMed.
- M. Lu, Y. Zheng, Y. Hu, B. Huang, D. Ji, M. Sun, J. Li, Y. Peng, R. Si, P. Xi and C.-H. Yan, Sci. Adv., 2022, 8, eabq3563 Search PubMed.
- Z. Feng, P. Wang, Y. Cheng, Y. Mo, X. Luo, P. Liu, R. Guo and X. Liu, J. Electroanal. Chem., 2023, 946, 117703 CrossRef CAS.
- L. Wei, D. Meng, Q. Jiang, W. Wang and J. Tang, J. Environ. Chem. Eng., 2022, 10, 108591 Search PubMed.
- S. Iqbal, J. C. Ehlers, I. Hussain, K. Zhang and C. Chatzichristodoulou, Chem. Eng. J., 2024, 499, 156219 CrossRef CAS.
- M. Gong and H. Dai, Nano Res., 2015, 8, 23–39 CrossRef CAS.
- J. Zhang, J. R. Winkler, H. B. Gray and B. M. Hunter, Energy and Fuels, 2021, 35, 19164–19169 Search PubMed.
- D. Friebel, M. W. Louie, M. Bajdich, K. E. Sanwald, Y. Cai, A. M. Wise, M. J. Cheng, D. Sokaras, T. C. Weng, R. Alonso-Mori, R. C. Davis, J. R. Bargar, J. K. Nørskov, A. Nilsson and A. T. Bell, J. Am. Chem. Soc., 2015, 137, 1305–1313 CrossRef CAS PubMed.
- S. Anantharaj, S. Kundu and S. Noda, Nano Energy, 2021, 80, 105514 CrossRef CAS.
- S. Khatun, S. Pal and P. Roy, J. Alloys Compd., 2024, 977, 173393 Search PubMed.
- X. Wang, Z. Jiang, Y. Ma, X. Su, X. Zhao, A. Zhu and Q. Zhang, J. Power Sources, 2024, 591, 233819 Search PubMed.
- E. Lee, A. H. Park, H. U. Park and Y. U. Kwon, Ultrason. Sonochem., 2018, 40, 552–557 CrossRef CAS PubMed.
- M. Gorlin, J. F. De Araujo, H. Schmies, D. Bernsmeier, S. Dresp, M. Gliech, Z. Jusys, P. Chernev, R. Kraehnert, H. Dau and P. Strasser, J. Am. Chem. Soc., 2017, 139, 2070–2082 Search PubMed.
- S. Zhang, S. Huang, F. Sun, Y. Li, L. Ren, H. Xu, Z. Li, Y. Liu, W. Li, L. Chong and J. Zou, J. Energy Chem., 2024, 88, 194–201 Search PubMed.
- C. Liang, P. Zou, A. Nairan, Y. Zhang, J. Liu, K. Liu, S. Hu, F. Kang, H. J. Fan and C. Yang, Energy Environ. Sci., 2020, 13, 86–95 Search PubMed.
- Y. Wu, H. Wang, S. Ji, X. Tian, G. Li, X. Wang and R. Wang, Appl. Surf. Sci., 2021, 564, 150440 Search PubMed.
- G. Zhang, J. Zeng, J. Yin, C. Zuo, P. Wen, H. Chen and Y. Qiu, Appl. Catal., B, 2021, 286, 119902 CrossRef CAS.
- S. Kang, C. Im, I. Spanos, K. Ham, A. Lim, T. Jacob, R. Schlögl and J. Lee, Angew. Chem., Int. Ed., 2022, 61, e202214541 CrossRef CAS PubMed.
- M. Yan, Z. Zhao, P. Cui, K. Mao, C. Chen, X. Wang, Q. Wu, H. Yang, L. Yang and Z. Hu, Nano Res., 2021, 14, 4220–4226 Search PubMed.
- Y. Zhang, L. Liu, J. Wang, R. Yao, Y. Wu, M. Wang, Q. Zhao, J. Li and G. Liu, J. Power Sources, 2022, 518, 230757 Search PubMed.
- L. Fan, Q. Geng, L. Ma, C. Wang, J. X. Li, W. Zhu, R. Shao, W. Li, X. Feng, Y. Yamauchi, C. Li and L. Jiang, Chem. Sci., 2023, 14, 13851–13859 Search PubMed.
- Q. Geng, L. Fan, H. Chen, C. Zhang, Z. Xu, Y. Tian, C. Yu, L. Kang, Y. Yamauchi, C. Li and L. Jiang, J. Am. Chem. Soc., 2024, 146, 10599–10607 Search PubMed.
- Y. Li, R. Tong, W. Zhang and S. Peng, J. Catal., 2022, 410, 22–30 Search PubMed.
- Y. Sheng, M. Manuputty, M. Kraft and R. Xu, ACS Appl. Energy Mater., 2023, 6, 2320–2332 Search PubMed.
- J. Hu, J. Yin, A. Peng, D. Zeng, J. Ke, J. Liu and K. Guo, Small, 2024, 20, 2402881 CrossRef CAS PubMed.
- L. He, N. Wang, M. Xiang, L. Zhong, S. Komarneni and W. Hu, Appl. Catal., B, 2024, 345, 123686 Search PubMed.
- Y. Zhao, W. Wan, N. Dongfang, C. A. Triana, L. Douls, C. Huang, R. Erni, M. Iannuzzi and G. R. Patzke, ACS Nano, 2022, 16, 15318–15327 CrossRef CAS PubMed.
- P. Zhai, C. Wang, Y. Zhao, Y. Zhang, J. Gao, L. Sun and J. Hou, Nat. Commun., 2023, 14, 1873 Search PubMed.
- M. Batool, A. Hameed and M. A. Nadeem, Coord. Chem. Rev., 2023, 480, 215029 Search PubMed.
- H. W. Choi, D. I. Jeong, S. Bin Kwon, S. Woo, J. Kim, J. H. Kim, W. S. Yang, B. Lim, B. K. Kang and D. H. Yoon, Appl. Surf. Sci., 2021, 566, 150706 Search PubMed.
- X. Wu, X. Gu, J. Tai, J. Tang, K. Yuan, L. Liu, Y. Zheng, A. Altaf, X. Shen and S. Cui, J. Power Sources, 2025, 633, 236430 CrossRef CAS.
- Y. Li, H. Guo, J. Zhao, Y. Zhang, L. Zhao and R. Song, Chem. Eng. J., 2023, 464, 142604 Search PubMed.
- Y. Wang, W. Nong, N. Gong, T. Salim, M. Luo, T. L. Tan, K. Hippalgaonkar, Z. Liu and Y. Huang, Small, 2022, 18, 2203340 Search PubMed.
- X. Liu, X. Fan, H. Huang, H. Lin and J. Gao, J. Colloid Interface Sci., 2021, 587, 385–392 Search PubMed.
- Y. Wu, Y. Li, Z. Xie, Y. Wang, Y. Wang and B. Wei, Chem. Eng. J., 2024, 488, 151086 Search PubMed.
- X. Wan, Y. Song, H. Zhou and M. Shao, Energy Mater. Adv., 2022, 2022, 9842610 Search PubMed.
- J. Yu, D. Huang, L. Zhou, W. Liu and D. Zhou, J. Power Sources, 2025, 647, 237341 Search PubMed.
- S. Assavachin, S. Ittisanronnachai, T. Atithep, N. Chitterisin and M. Sawangphruk, J. Power Sources, 2025, 650, 237494 Search PubMed.
- J. Mohammed-Ibrahim, J. Power Sources, 2020, 448, 227375 Search PubMed.
- Y. H. Wang, L. Li, J. Shi, M. Y. Xie, J. Nie, G. F. Huang, B. Li, W. Hu, A. Pan and W. Q. Huang, Adv. Sci., 2023, 10, 2303321 CrossRef CAS PubMed.
- H. Wang and S. Chen, Catalysts, 2024, 14, 81 Search PubMed.
- C. Feng, F. Wang, Z. Liu, M. Nakabayashi, Y. Xiao, Q. Zeng, J. Fu, Q. Wu, C. Cui, Y. Han, N. Shibata, K. Domen, I. D. Sharp and Y. Li, Nat. Commun., 2021, 12, 5980 CrossRef CAS PubMed.
- Y. Qiu, Z. Liu, A. Sun, X. Zhang, X. Ji and J. Liu, ACS Sustainable Chem. Eng., 2022, 10, 16417–16426 Search PubMed.
- J. Xing, S. Bliznakov, L. Bonville, R. Maric and A. Friedman, ChemCatChem, 2025, 17, e202402165 Search PubMed.
- H. Qiu, K. Obata, K. Tsuburaya, T. Nishimoto, K. Nagato and K. Takanabe, J. Power Sources, 2024, 611, 234765 Search PubMed.
- D. Senthil Raja, C. L. Huang, Y. A. Chen, Y. M. Choi and S. Y. Lu, Appl. Catal., B, 2020, 279, 119375 CrossRef CAS.
- X. Wang, Z. Qin, J. Qian, L. Chen and K. Shen, Appl. Catal., B, 2024, 359, 124506 Search PubMed.
- X. Dong, E. Yan, Y. Lv, Y. Zhou and X. Chu, Appl. Catal., A, 2024, 681, 119772 Search PubMed.
- C. Dang Van, S. Kim, M. Kim and M. H. Lee, ACS Sustainable Chem. Eng., 2023, 11, 1333–1343 Search PubMed.
- X. Liu, S. Jing, C. Ban, K. Wang, Y. Feng, C. Wang, J. Ding, B. Zhang, K. Zhou, L. Gan and X. Zhou, Nano Energy, 2022, 98, 107328 Search PubMed.
- Y. Huang, T. Zhou, Y. Hu, Y. Yang, F. Yang, W. Huang, L. He and W. Sun, Int. J. Hydrogen Energy, 2023, 48, 33411–33421 Search PubMed.
- B. Wang, H. Sun, M. Chen, T. Zhou, H. Zheng, M. Zhang, B. Xiao, J. Zhao, Y. Zhang, J. Zhang and Q. Liu, Chem. Eng. J., 2024, 479, 147500 Search PubMed.
- Y. Hu, T. Shen, Z. Song, Z. Wu, S. Bai, G. Liu, X. Sun, Y. Wang, S. Hu, L. Zheng and Y. F. Song, ACS Catal., 2023, 13, 11195–11203 CrossRef CAS.
- F. Jiang, Y. Li and Y. Pan, Adv. Mater., 2024, 36, 2306309 Search PubMed.
- Y. Zhu, Z. Li and Y. Zhou, ChemCatChem, 2025, e202500072 Search PubMed.
- N. Yang, H. Li, X. Lin, S. Georgiadou, L. Hong, Z. Wang, F. He, Z. Qi and W. F. Lin, J. Energy Chem., 2025, 105, 669–701 Search PubMed.
- J. N. Bamba, A. T. Dumlao, R. M. Lazaro, D. D. Matienzo and J. Ocon, Curr. Opin. Electrochem., 2024, 48, 101592 Search PubMed.
- Y. Li, X. Lin, Y. Yang, S. Georgiadou, B. Du, Z. Qi, S. Saremi-Yarahmadi and W. F. Lin, ChemCatChem, 2025, 17, e01040 CrossRef CAS.
- P. Liu, J. Wang, X. Wang, L. Liu, X. Yan, H. Wang, Q. Lu, F. Wang and Z. Ren, Int. J. Hydrogen Energy, 2024, 49, 285–294 Search PubMed.
- Q. N. Ha, C. H. Yeh, N. S. Gultom and D. H. Kuo, J. Mater. Chem. A, 2023, 12, 460–474 RSC.
- S. Kang, D. Yeol Lee, Y. Kim, S. Bae and J. Lee, Chem. Eng. J., 2024, 499, 156469 Search PubMed.
- Y. Son, J. Mo, E. Yong, J. Ahn, G. Kim, W. Lee, C. H. Kwon, H. Ju, S. W. Lee, B. H. Kim, M. Kim and J. Cho, Appl. Catal., B, 2024, 343, 123563 CrossRef CAS.
- Y. Zhao, Q. Wen, D. Huang, C. Jiao, Y. Liu, Y. Liu, J. Fang, M. Sun and L. Yu, Adv. Energy Mater., 2023, 13, 2203595 CrossRef CAS.
- N. Zhou, R. Liu, X. Wu, Y. Ding, X. Zhang, S. Liang, C. Deng, G. Qin, Z. Huang and B. Chen, J. Power Sources, 2023, 574, 233163 CrossRef CAS.
- R. Kaur, A. Gaur, V. Pundir, K. Arun and V. Bagchi, J. Colloid Interface Sci., 2024, 672, 736–743 CrossRef CAS PubMed.
- J. Liu, G. Qian, T. Yu, J. Chen, C. Zhu, Y. Li, J. He, L. Luo and S. Yin, Chem. Eng. J., 2022, 431, 134247 CrossRef CAS.
- P. Tan, Y. Wu, Y. Tan, Y. Xiang, L. Zhou, N. Han, Y. Jiang, S. J. Bao and X. Zhang, Small, 2024, 20, 2308371 CrossRef CAS PubMed.
- Y. Y. Feng, G. Deng, X. Y. Wang, M. Zhu, Q. N. Bian and B. S. Guo, Int. J. Hydrogen Energy, 2023, 48, 12354–12363 Search PubMed.
- Y. F. Song, Z. Y. Zhang, H. Tian, L. Bian, Y. Bai and Z. L. Wang, Chem. – Eur. J., 2023, 29, e202301124 Search PubMed.
- M. Liu, J. Wang, H. Wu, S. Li, Y. Shi and N. Cai, Int. J. Hydrogen Energy, 2024, 51, 626–637 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |