Open Access Article
Open Access ArticleRecent developments in antimicrobial polymers for biofilm inhibition
Gillian A.
Kropp†
 ab,
Cassell N.
McMillian†
ab,
Jonathan D.
Mase
ab,
Sanjana
Kandagiri
a,
Elizabeth S.
Nowak
ab,
Cassell N.
McMillian†
ab,
Jonathan D.
Mase
ab,
Sanjana
Kandagiri
a,
Elizabeth S.
Nowak
 de and
Michael D.
Schulz
de and
Michael D.
Schulz
 *abc
*abc
aDepartment of Chemistry, Virginia Tech, Virginia, USA. E-mail: mdschulz@vt.edu
bMacromolecules Innovation Institute, Virginia Tech, Virginia, USA
cTranslational Biology, Medicine, and Health Graduate Program, Virginia Tech, Virginia, USA
dDivision of Infectious Diseases, Carilion Clinic, Virginia, USA
eVirginia Tech Carilion School of Medicine, Virginia, USA
First published on 26th March 2026
Abstract
Biofilm-associated infections continue to present a formidable clinical challenge, as surface-adhered microbial communities exhibit remarkable tolerance toward conventional antibiotics. Polymeric materials have emerged as a versatile platform for combating biofilms, offering chemical tunability and enabling diverse antimicrobial strategies. This feature review article highlights recent advances in polymeric materials designed to prevent biofilm-associated infections by resisting bacterial adhesion (passive inhibition) or exerting bactericidal effects (active inhibition). These approaches include antifouling surfaces, polymer–nanoparticle composites, and bioinspired materials. Particular attention is given to how polymer structure and functionality (e.g., hydrophobicity, charge, and network architecture) govern bacterial adhesion and viability at interfaces. Emerging glycomaterials are also discussed, where glycan motifs are integrated with nanoparticles or cationic domains to enhance biofilm penetration and antimicrobial efficacy. Collectively, these studies underscore the potential of polymeric materials to modulate microbe–surface interactions, thereby guiding the design of next-generation antibiofilm materials.
Introduction
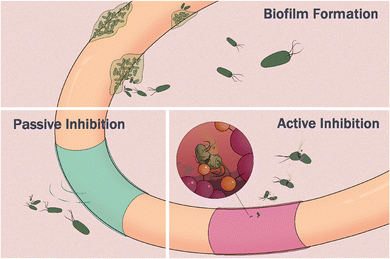
The human body hosts trillions of microbes, including bacteria, viruses, fungi, and archaea.1 This diverse community, collectively known as the microbiome, exists in a balanced symbiosis with the host. Bacteria are especially influential and abundant. They line our skin as an additional protective barrier and immune regulator,2,3 form biofilms in the oral cavity,4–6 and densely populate the gastrointestinal tract, where they support digestion,7,8 nutrient absorption, and metabolite production.9 However, even minor disruptions to this balance can have profound consequences, as microbial species can rapidly proliferate and cause disease. The 20th century saw a dramatic reduction in bacterial infections due to the development of antibiotics and improved sanitation. Today, many bacterial infections can be treated with short courses of antibiotics, which disrupt key processes such as cell wall synthesis, protein production, or nucleic acid replication.10 Yet two challenges limit their long-term effectiveness. First, because bacteria reproduce rapidly, even a few surviving cells can acquire mutations and develop resistance. Second, bacteria can transition from free-floating (planktonic) cells to surface-attached biofilms—a multicellular state that protects them from antibiotics and immune cells while anchoring them to surfaces such as tissues or medical devices (Fig. 1).11,12Biofilms are particularly problematic in clinical settings. On indwelling medical devices, such as urinary catheters, they resist clearance and seed chronic infections. In wounds, biofilms promote persistent inflammation and treatment failure; for example, most diabetic foot ulcers that progress to lower-limb amputation are biofilm-infected, with serious impacts on morbidity and short-term survival.13,14 Similarly, catheter-associated urinary tract infections (CAUTIs)—driven by biofilm formation that begins within days of catheter insertion—are among the most common hospital-acquired infections (HAIs). Annually, CAUTIs afflict hundreds of thousands of U.S. patients and contribute to thousands of deaths.15 In fact, CAUTIs account for 30–40% of all HAIs, globally.16 Current clinical strategies are largely reactive, relying on device removal, and prolonged antibiotic regimens. Unfortunately, these approaches are often ineffective, contribute to antimicrobial resistance, and disrupt the patient's microbiome. Preventing biofilm formation altogether represents an urgent yet unmet need in the medical field.
This review highlights emerging polymer-based strategies designed to prevent or disrupt biofilm formation on medical devices. First, we outline the biology of biofilm formation and the structural features that make these microbial communities so recalcitrant to treatment. We then discuss how polymer design has been leveraged to develop two main classes of antibiofilm materials: active polymers, which kill bacterial cells directly, and passive polymers, which create antifouling barriers that block bacterial adhesion (Fig. 1). By examining these approaches, we aim to clarify both the challenges and the opportunities for translating polymeric antibiofilm technologies into clinical practice.
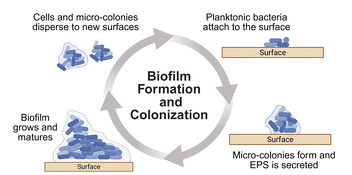
Biofilm formation
Bacterial biofilms contribute to the spread of infectious diseases and are frequently implicated in persistent chronic infections.17,18 Biofilm formation proceeds through several distinct stages (Fig. 2).3,19,20 Initially, free-roaming (planktonic) bacteria attach to a surface. This early attachment is followed by microcolony development, during which cells proliferate and secrete extracellular polymeric substances (EPS)—a matrix primarily composed of polysaccharides, proteins, and nucleic acids—to which additional bacteria can adhere.21 As the biofilm matures, cells embedded within the EPS can detach and revert to the planktonic state, enabling dispersal and the seeding of new biofilms.19,20 The EPS functions as both a physical and chemical barrier, impeding antibiotic penetration and shielding resident bacteria from host immune defenses.22,23 This protective environment contributes to the persistence and pathogenicity of device-associated infections.24–26Bacterial biofilms are a major contributor to HAIs, which substantially increase patient morbidity and mortality.27 Device-associated infections are responsible for 25.6% of HAIs,28 and are commonly associated with biofilm-forming pathogens, which can be endogenous bacteria or exogenous bacteria introduced during device insertion.27,29–32 Additionally, an estimated 80% of chronic and surgical site infections are attributable to biofilm-forming organisms.33 These biofilm-associated infections are most frequently associated with Staphylococcus aureus (S. aureus), Pseudomonas aeruginosa (P. aeruginosa), Escherichia coli (E. coli), Staphylococcus epidermidis (S. epidermidis), and Enterococcus faecalis (E. faecalis), all of which originate from either the patient's microbiota or hospital-acquired sources.33,34
Strategies to control biofilms generally focus on preventing their formation or eradicating established communities. Both approaches are hindered by the widespread emergence of antibiotic resistance.35 Most clinically used antibiotics are derived from microbial natural products and act by targeting essential cellular processes.36,37 However, these agents typically act on a single target, and many have already been compromised by resistance. This problem is further exacerbated by antibiotic pollution from hospitals, agriculture, aquaculture, and industry, which causes bacteria to be exposed to sub-minimum inhibitory concentrations (sub-MIC) of antibiotics. This low-level exposure drives the selection and dissemination of resistance genes.38–44
The distinct physiology of biofilms further limits antibiotic efficacy by restricting diffusion of drugs through the matrix and reducing metabolic activity such that metabolism-targeting drugs become less effective.45–48 The development of new antibiotics is slow, costly, and high risk; research into just one antibiotic can span 10–14 years and require a company to invest roughly $1 billion.49,50 However, only a few drug candidates ever enter clinical use.51 Most new agents are structural analogues of existing drugs rather than scaffolds with novel mechanisms, and high-throughput screening has likewise failed to substantially expand the scope of novel small-molecule antibiotics.52 These limitations have prompted growing interest in polymers as alternative antimicrobial materials, due to their structural versatility and ability to act as antibacterial or antifouling agents, or as coatings that integrate multiple antimicrobial mechanisms.
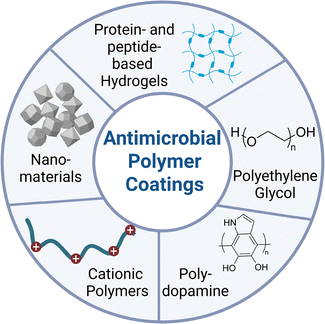
Antimicrobial coatings
Medical devices represent a persistent point of vulnerability for infection, as their surfaces readily serve as substrates for bacterial adhesion and subsequent biofilm formation. Biofilms are especially problematic because they display an intrinsic resistance to conventional antibiotic therapies, rendering many infections difficult to eradicate once established.53 To address this challenge, polymeric coatings have emerged as a particularly attractive strategy. These materials can act at the earliest stage of infection, primarily by preventing the adhesion of planktonic bacteria to device surfaces. Hydrogels, thin films, and related polymeric constructs not only provide a protective barrier between the device and its biological environment but can also be engineered to incorporate bioactive agents that actively interfere with microbial colonization (Fig. 3).54 In doing so, coatings on indwelling medical hardware offer a route to reducing infection risk by inhibiting bacteria before a mature biofilm can form.Relatedly, antimicrobial peptides (AMPs) are short, low-molecular-weight biopolymers (typically 12–50 amino acids long) that can be integrated into hydrogel networks as active antimicrobial components.61 Unlike passive antifouling coatings, AMP-containing hydrogels can exert direct antimicrobial activity.62 Their ability to disrupt bacterial membranes while inducing minimal resistance has made AMPs attractive candidates for biomedical applications.62–64 Moreover, many AMPs are also capable of self-assembly, enabling the creation of peptide-only hydrogel coatings that resist colonization while mimicking aspects of native extracellular environments. Finally, AMPs’ intrinsic properties—low immunogenicity, high structural versatility, and biodegradability—further facilitate their application in coatings for medical devices.65,66
Recent efforts have focused on developing synthetic peptide mimics with tunable functionality.67–72 For example, Kumar and coworkers reported an antibacterial hydrogel using naphthyl anthranilamide (NaA)-capped peptide mimics. These short peptide sequences, synthesized via ring-opening polymerization (ROP) of isatoic anhydride followed by the incorporation of cationic substituents, yielded a library of amphiphilic peptide analogues that could form nanofibrous networks. This library consisted of ultra-short NaA-capped peptides with varied cationic group, anthranilamide-core substituent, or counter ion. The authors observed that peptide mimics containing primary ammonium groups were the most effective antibacterials.73 Continuous nanofiber release was observed when testing against S. aureus, allowing antibacterial efficacy to last over nine days. The authors noted that the choice of cationic group significantly influenced the hydrophobicity–hydrophilicity balance of the overall material.73 This study demonstrates the potential of peptide mimics as dynamic antimicrobial coatings.
Although PEG effectively delays microbial adhesion, its protection is fundamentally passive. To expand its utility, PEG has frequently been combined with antimicrobial agents to provide both passive and active inhibition.27,78–82 For example, Bernthal and coworkers developed a “smart” antimicrobial implant coating using branched PEG-poly(propylene sulfide) that encapsulates and releases antibiotics (vancomycin and tigecycline).83 In a murine implant model, this coating significantly reduced bacterial load in both the implant and adjacent tissue, with tigecycline outperforming vancomycin.
Similarly, Sileika and colleagues engineered a PEG–silver nanoparticle (AgNP) coating on polydopamine-coated polycarbonate substrates.84 In this system, PEG slowed the release of AgNPs, extending their activity from 6 to ≥10 days while simultaneously enhancing bactericidal performance against E. coli, P. aeruginosa, and S. epidermidis. PEG grafting also improved antifouling by reducing bacterial attachment relative to the AgNPs or polydopamine alone. AgNPs and their incorporation into coatings have been well investigated for their broad-spectrum antimicrobial effects,85–90 largely driven by enzymes (particularly sulfhydryl-containing enzymes) interacting with silver, which blocks enzymatic processes and causes membrane destabilization.86 Together, these studies illustrate PEG's dual role as a passive antifouling barrier and as a versatile scaffold for incorporating antimicrobial agents.
However, PEG coatings have several drawbacks. First, they are insufficient once biofilms have become established, underscoring the need for more active approaches. Second, PEG's long-term clinical use is problematic due to the emergence of patients who express anti-PEG antibodies.91,92 Efforts have been made to design PEG-alternatives that maintain PEG's desirable properties (e.g., inertness, hydrophilicity) while avoiding such negative biological side effects: polyglycerols, poly(2-oxazolines), and poly(acrylamides) are some of many options.93,94
Furthermore, many PEG-alternatives have been investigated as antifouling coatings, and some coatings even display antibacterial activity.95 For example, recent studies have looked at sulfoxide-containing polymers for their antifouling properties in nanoparticle drug delivery,96,97 and sulfoxides are further known to possess antibacterial properties when incorporated into a PEG backbone, highlighting their potential in antibiofilm applications.98 As the field continues to investigate novel PEG-alternatives, those that display antibacterial activity may find further utility as biofilm inhibitors. Interested readers can consult several recent review articles for more expansive discussions on this growing field.99–102
| Antimicrobial agent | Polydopamine-coated surface | Ref. |
|---|---|---|
| a Nitric oxide was generated in situ assisted by copper. b Nitric oxide was generated in situ assisted by selenium. | ||
| H2O2-generating catechol | Polypropylene | 107 |
| Silver nanoparticles (AgNPs) | Silicone | 108 |
| Polycarbonate | 84 | |
| Polymer-ceramic composite | 109 | |
| Titanium | 110 and 111 | |
| Sulfobetaine | Silicone | 112 |
| ε-polylysine/gum arabic layer-by-layer assembly | Anodized titanium | 113 |
| Lipopeptide | Polyurethane | 114 |
| Nitric oxide | Glass | 115 |
| Titaniuma | 116 | |
| Iron oxide Nanoparticles | 105 | |
| Siliconea | 117 | |
| Selenium Nanoparticlesb | 118 | |
| Cu2+ | Titanium | 119 |
| Vancomycin | Titanium | 120 |
| Curcumin | Titanium | 121 |
For example, Schulz and co-workers developed an antibiofilm catheter coating using a polydopamine-antibiotic (gentamicin) composite (PD-Gent).106 The authors found that when synthesizing these coatings with dopamine concentrations >1.0 mg mL−1, the composite was unable to inhibit the growth of planktonic P. aeruginosa and biofilm formation. However, when these coatings were synthesized with less dopamine (0.25–1.0 mg mL−1), PD-Gent was consistently effective against bacterial colonies, despite containing less gentamicin. This change in bactericidal activity was attributed to polydopamine aggregates present at higher polydopamine concentrations that likely prevented antibiotic–bacteria interactions. The PD-Gent coating remained effective when changing the underlying substrate (polystyrene, polyvinyl chloride, or silicone) or the antibiotic (tobramycin, amikacin), demonstrating the versatility of this approach. Finally, the authors evaluated their coating's effectiveness by simulating bacteriuria flow through coated commercial foley urinary catheters. After 24 hours the PD-Gent–coated catheters generated a 3-fold reduction in biofilm formation compared to the uncoated catheters.
Polymer–nanoparticle composites are particularly attractive due to their tuneable physiochemical properties (i.e., particle size, morphology, metal identity, and polymeric surface coating), which can be adjusted to modulate stability and antimicrobial activity.132–137 For example, Rahaman et al. synthesized copper nanoparticles stabilized with poly(vinyl pyrrolidone) (PVP) or poly(vinyl alcohol) (PVA), demonstrating the advantages of polymer coatings in nanoparticle stabilization and antibiofilm performance.138 Both formulations significantly reduced the growth of planktonic bacterial cells (with slightly greater inhibition against S. aureus) and diminished exopolysaccharide production, suggesting that polymer coatings can not only stabilize copper nanoparticles against aggregation and oxidation, but also enhance overall antibiofilm performance.
Beyond metallic systems, non-metallic polymer-based nanomaterials have been increasingly explored for antibiofilm applications. These systems include nanoemulsions,139,140 nanogels,141,142 and polymeric nanoparticles that frequently incorporate biological or synthetic components such as cell membranes,143,144 lipids,145,146 natural polymers,147–149 or synthetic polymer matricies.142,150,151 Usually, the nanomaterial's primary function is to facilitate the targeted delivery of antimicrobial agents within the biofilm matrix. For example, Anjum et al. synthesized poly(lactic-co-glycolic acid) (PLGA) nanoparticles loaded with xylitol and functionalized with concanavalin A for treating S. aureus and P. aeruginosa biofilm infections.152 Free xylitol poorly penetrates the biofilm matrix; however, when delivered via PLGA nanoparticles, the authors observed improved penetration and biofilm inhibition. In fact, the xylitol-loaded nanoparticles successfully permeated S. aureus biofilm, P. aeruginosa biofilm, and a polymicrobial biofilm with both bacterial species after 30 minutes. Live/dead viability assays corroborated these findings, revealing markedly greater biofilm eradication by xylitol-loaded nanoparticles compared to free xylitol. The authors attributed this enhanced activity to the improved transport of the nanoparticles through the biofilm matrix, enabling localized drug release. This study highlights the promising ability for nanoparticles to overcome the physical and chemical barriers that impede conventional drug delivery.
Overall, nanomaterials provide a powerful toolkit for antibiofilm intervention by enhancing targeting and penetration. Nevertheless, several translational challenges remain. Many nanomaterial synthesis strategies are complex and difficult to scale up, and the toxicity profiles of these materials must be evaluated in a case-specific manner. Additionally, most advances remain confined to the laboratory scale at present. Bridging the gap between lab and clinic will require the development of reproducible, scalable synthetic methodologies, comprehensive long-term toxicity studies, and rigorous clinical evaluations to fully realize the therapeutic potential of nanomaterials.
Cationic polymers
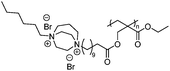
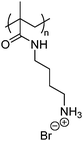
Cationic polymers are one of the most widely explored classes of antimicrobial materials because they can bypass conventional mechanisms of antibiotic resistance.153 Their activity typically relies on two synergistic features: a cationic moiety that promotes electrostatic adsorption to the negatively charged cell membrane of the bacteria, and hydrophobic groups that insert into the lipid bilayer to destabilize the membrane and cause cell lysis.154 Common cationic functionalities include quaternary ammonium (QAC), imidazolium, guanidinium, pyridinium, triazolium, and phosphonium groups, which can be introduced directly into polymer backbones or pendant side chains, or formulated as ionic liquids (Table 2).155,156 By varying charge density and hydrophobicity–hydrophilicity balance, these polymers can be tailored to optimize antimicrobial potency while maintaining biocompatibility. A wide range of structural motifs have been explored to exploit this charge–hydrophobicity balance, which highlights how variations in cationic group and backbone architecture influence antibacterial efficacy and cytocompatibility (Table 2).| Polymer backbone | Strain | MIC µg mL−1 | Ref. |
|---|---|---|---|
|
E. coli and S. aureus. | 1 | 157 |
|
E. coli and S. aureus. | 62.5 | 158 |
|
E. coli | 18 | 159 |
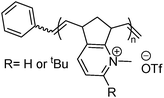
|
MRSA | 50 | 160 |
| E. coli | 25–50 | ||
|
B. subtills | 4 | 161 |
| S. aureus | 4 | ||
| E. coli | 2–4 | ||
| E. faecalis | 2 | ||
| E. faecium | 4 | ||
| K. pneumoniae | 2–4 | ||
| A. baumannii | 2 | ||
| P. aeruginosa | 4 | ||
| M. smegmatis | 2.5 | ||
| M. tuberculosis | 25 | ||
| E. cloacae | 2–4 | ||
| MRSA | 2 |
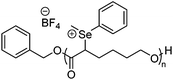
Recently, selenium-based cationic polymers have garnered significant interest due to their promising antimicrobial performance.162–165 For example, Zhu and coworkers developed a degradable family of selenium-containing polyesters via ring-opening polymerization of selenide-functionalized ε-caprolactone monomers, followed by methylation to yield polyselenonium salts.157 In this study, four variants with side chains of differing hydrophobicity—C3, C5, C10, and phenyl—were prepared, with degrees of polymerization between 12 and 13. All four polymers exhibited potent antibacterial activity against E. coli and S. aureus while maintaining good mammalian cell viability at biocidal concentrations. Among them, the phenyl-substituted polyselenonium salt displayed the lowest MIC (1 µg mL−1) and minimum bactericidal concentration (MBC) (2 µg mL−1), along with superior biocompatibility (before and after degradation) compared to its alkyl-substituted counterparts. Hydrophobicity was identified as a key determinant of efficacy: moderate hydrophobicity (C5 or phenyl) enhanced bacterial killing, whereas excessively long alkyl chains (C10) reduced solubility and biocompatibility (Fig. 4).
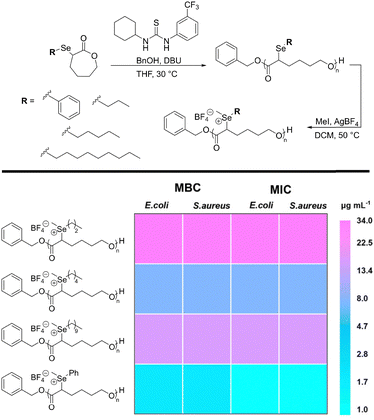 | ||
| Fig. 4 Heatmap of MBCs and MICs for polyselenoniums. Reproduced from ref. 157 with permission from the American Chemical Society, copyright 2022. | ||
In this report, mechanistic studies using zeta potential and quartz crystal microbalance revealed that cationic selenium species first adsorb electrostatically onto the bacterial surface, shifting cell surface potentials from –43.6 mV to –7.4 mV (E. coli) and –34.3 mV to –3.79 mV (S. aureus). Subsequent insertion of hydrophobic groups into the lipid bilayer caused membrane disruption, collapse, and cytoplasmic leakage. Importantly, cytotoxicity assays showed >80–90% fibroblast viability at antibacterial concentrations, and biocompatibility further improved upon polymer degradation. These results suggest that phenyl-substituted polyselenonium salts offer an optimal balance of hydrophobicity, antimicrobial potency, and safety, making them promising candidates for antibacterial coatings.157
Finally, there has been recent interest in developing polyurethane-based cationic biofilm inhibitors, as polyurethanes are already frequently used when fabricating medical devices.166–169 For example, work from the Joy lab has featured polyurethanes with fixed charge-to-hydrophobicity ratios (60:40) that incorporate amino acid mimetics: arginine or lysine mimetics for cationic functionality and alanine or phenylalanine mimetics for hydrophobic character. Though the charged polyurethanes generally exhibited poor activity against planktonic bacteria (MICs >250 µg mL−1 were predominately observed), the materials consistently inhibited biofilm colony growths of E. coli, P. aeruginosa, and S. aureus at significantly lower concentrations (MICs <8 µg mL−1). This stark difference in activity can be explained, in part, by the fact that the materials derive their activity not from typical bactericidal mechanisms (e.g., cell membrane rupturing) but instead from disrupting key processes in biofilm development. Primarily the polymers prevented colony formation by altering the bacteria's cell surface charge and by reducing bacteria swarming motility, or the bacteria's ability to migrate across surfaces.167
Antibiofilm glycopolymers
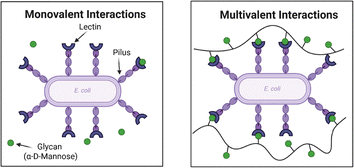
Cell surfaces are coated with a dense layer of glycosylated biomolecules, collectively known as the glycocalyx. These surface glycans mediate diverse molecular recognition events, and many bacteria display lectins that specifically bind to host glycans, enabling adhesion and initiating biofilm formation.170 These interactions between sugar-binding sites and cell-surface glycans are crucial biological events that have often been exploited to develop biomimetic molecules.171,172 Drawing inspiration from these natural recognition processes, synthetic glycopolymers—polymers bearing pendant carbohydrate moieties—have been developed to mimic the glycocalyx and modulate cell–microbe interactions.Across Gram-negative and Gram-positive species, bacteria typically develop resistance against conventional antibiotics through five mechanisms: enzymatic drug degradation, target modification, altered porin expression, efflux pump activity, and biofilm formation.173,174 However, bacteria are unable to circumvent interactions with glycopolymers through such mechanisms. By targeting highly conserved bacterial structures such as fimbriae, glycopolymers use an inhibition mechanism with a lower risk for resistance development.175–177
Glycopolymers leverage the principle of multivalency, wherein multiple carbohydrate units along the polymer backbone simultaneously engage multiple bacterial lectins (Fig. 5). These interactions are facilitated through cooperative binding, with affinities far exceeding those of individual monovalent interactions.178 For example, single carbohydrate–protein interactions typically exhibit dissociation constants (Kd) in the millimolar range. Multivalent analogues can enhance binding by several orders of magnitude, reaching micromolar to nanomolar affinities.179–181
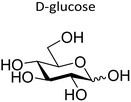
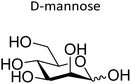
Antibacterial glycopolymers typically incorporate additional antimicrobial motifs, such as cationic side chains, to couple competitive binding with direct bactericidal activity (see Table 3 for a summary of these materials). However, unfunctionalized glycopolymers can also inhibit bacteria. By presenting sugars in a multivalent manner, glycopolymers can outcompete native glycans for bacterial lectins and inhibit growth. For example, Schulz and coworkers demonstrated this strategy using glycopolymers prepared via ring-opening metathesis polymerization. Polymers with mannose side-chains selectively inhibited E. coli growth, consistent with the high affinity of Type 1 fimbriae for α-D-mannose residues.85 Typically, Type 1 fimbriae on the E. coli pathogen selectively recognize terminal α-D-mannose residues on host polysaccharides to mediate adhesion and initiate biofilm formation.182 This study demonstrates the ability of unfunctionalized glycopolymers to inhibit bacterial growth without the addition of conventional antibacterial moieties.
| Glycan | α/β | Active moiety | Polymer backbone | Strain | Ref. |
|---|---|---|---|---|---|
|
β | N/A | Dendritic peptide | P. aeruginosa | 194 |
| β | QAC | Chitosan | P. aeruginosa | 200 | |
| β | PDT/QAC | Polyacrylate | P. aeruginosa, E. coli, S. aureus, and B. amyloliquefaciens | 203 | |
| β | CuS NP/ Photothermal therapy (PTT) | Polymethacrylate | P. aeruginosa, S. aureus | 207 | |
| β | Gold nanorods (AuNRs) | Polymethacrylate | P. aeruginosa | 211 | |
| β | AgNP | Polyacrylamide | S. mutans, E. coli, S. aureus, P. aeruginosa | 212 | |
| β | Tobramycin | Polyacrylamide | P. aeruginosa | 225 | |
| β | Spiropyran (PTT) | Polyacrylate | P. aeruginosa | 214 | |
| β | Guanidine (cationic)/BODIPY (PDT) | Polymethacrylate | S. mutans | 226 | |
| β | BODIPY (PDT) | Polymethacrylate | P. aeruginosa | 227 | |
| β | Graphene oxide NP | PEG linker | P. aeruginosa | 217 | |
|
α | QAC | Chitosan | P. aeruginosa | 200 |
| α/β | AuNRs | Polymethacrylate | P. aeruginosa | 211 | |
| β | Graphene oxide NP | PEG linker | P. aeruginosa | 217 | |
| α/β | AuNRs | Polymethacrylate | P. aeruginosa | 175 | |
| α | N/A | Oligo(amido amine) | P. aeruginosa | 228 | |
|
α | Graphene oxide NP | PEG linker | P. aeruginosa | 217 |
| N/A | Lysine (cationic) | Block co-beta peptide | A. aureus, MRSA, S. aureus, S. agalactiae, S. pyogenes, S. pneumonia, E. faecalis | 229 | |
| 230 | |||||
|
α | Tobramycin | Polyacrylamide | P. aeruginosa | 213 |
| α | Cu/AgNP | Pentaerythritol dendrimer core | E. coli | 208 | |
|
β | N/A | Silk fibroin | S. mutans | 177 |
|
α/β | AgNP/QAC | Polyacrylamide | P. aeruginosa, E. coli, S. aureus, and B. amyloliquefaciens | 210 |
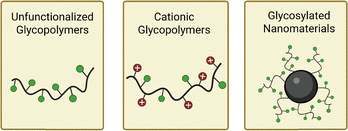
In this review we discuss only materials developed to inhibit or disrupt biofilm formation. For further information on antibacterial glycopolymers more broadly, we direct readers to several excellent reviews of the field.183–185 Here, we divide antibiofilm glycomaterials into three categories: unfunctionalized glycopolymers, cationic glycopolymers, and glycosylated nanomaterials (Fig. 6).
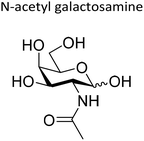
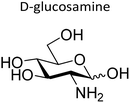
Several studies have drawn inspiration from natural, mucin-mediated suppression of bacterial colonization. For example, Werlang et al. developed silk-based glycopolymers functionalized with a series of different monosaccharides: N-acetyl glucosamine (GlcNAc), N-acetyl galactosamine (GalNAc), N-acetyl neuraminic acid (Neu5Ac), galactosamine (GalN), and glucosamine (GlcN) (Fig. 7).177 The authors then evaluated each polymer's ability to inhibit Streptococcus mutans (S. mutans) biofilm formation.177 Only glycopolymers that contained GalNAc or Neu5Ac demonstrated biofilm reduction compared to media controls. The authors also compared the polymers’ antibiofilm efficacy against that of native mucus. Native mucus (containing highly glycosylated serine, threonine, and proline domains) reduced biofilm formation by 70%, compared to GalNAc- and Neu5Ac-functionalized polymers, which showed a 98% and 40% reduction, respectively. Mechanistic studies revealed distinct modes of action. Neu5Ac-containing polymers decreased planktonic viability of S. mutans, while GalNAc-containing polymers suppressed biofilm formation by directly interfering with bacterial adhesion without affecting cell viability. On the other hand, GlcNAc-containing polymers induced biofilm growth. These findings illustrate how changing the glycan identity can generate distinct pro- or antibiofilm pathways.
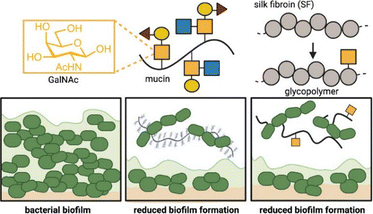 | ||
| Fig. 7 Silk-based glycopolymers to treat oral pathogenic S. mutans and commensal S. sanguinis. Reproduced from ref. 177 with permission from the American Chemical Society, copyright 2024. | ||
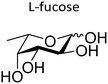
Glycopolymers have also been explored as synthetic mimics of host–bacteria interactions, leveraging sugar–lectin binding events to block bacterial adhesion and subsequent biofilm formation. In P. aeruginosa, the LecB lectin recognizes L-fucose and D-mannose residues on host cell surfaces. Inspired by these interactions, Hartmann and coworkers, synthesized α-L-fucose–functionalized polyamide oligomers (1.9–3.0 kDa) that outcompeted host glycans in binding LecB. These synthetic glycopolymers effectively disrupted bacterial adhesion and suppressed biofilm production by 15–20%, illustrating the potential of targeted lectin–ligand interactions as a design principle for antifouling materials.197 However, no clear trend in antibiofilm activity emerged from changing the fucose content, even though LecB-binding assays confirmed that polymers with a higher fucose density displayed stronger interactions between polymer and lectin, consistent with multivalent binding principles. These results suggest that P. aeruginosa biofilm development is governed by processes more complex than LecB–carbohydrate recognition alone, underscoring the limitations of purely passive glycopolymer strategies.
Though unfunctionalized glycopolymers demonstrate that glycan presentation alone can weaken bacterial adhesion and biofilm formation, these materials’ activity remains highly specific to the glycan–pathogen pairs they mimic. This specificity limits their utility as broad-spectrum antifouling agents. This limitation is potentially addressed by designing active glycopolymers that combine adhesion-blocking motifs with bactericidal or disruptive functionalities. Such dual-acting materials may offer more robust and generalizable antibiofilm performance, bridging the gap between passive mimics of mucin and fully synthetic antimicrobial polymers. Nevertheless, unfunctionalized glycomaterials have provided the glycopolymer community with key fundamental insights into polymer-bacteria interactions.
Zhang et al. highlighted the advantages of combining carbohydrates with cationic motifs by synthesizing block copolymers of glucose and lysine prepared via cationic ROP.201 β-Lactam-based monomers were used that enabled the formation of well-defined polymer blocks (Fig. 8). The initial antibacterial assays revealed that the lysine homopolymer was broadly active against most Gram-positive bacteria but was also highly cytotoxic and hemolytic. In contrast, a glucose:lysine block copolymer with a 7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13 ratio retained a comparable potency against S. aureus, while eliminating the hemolytic side effects. This modular strategy illustrates how glycan units can be incorporated within the polymer backbone, creating a framework to balance antibacterial potency with biocompatibility.201 This copolymer also displayed potency against community- and hospital-acquired methicillin-resistant S. aureus (MRSA) strains, including those resistant to vancomycin and daptomycin (Fig. 8). In addition to dispersing and eradicating MRSA biofilms with a 99.9% reduction (substantially outperforming vancomycin), the copolymer provided complete protection in vivo. All mice challenged with a lethal dose of MRSA survived when treated with the copolymer, compared to only a 67% survival rate for those treated with vancomycin. These results underscore the advantages of carbohydrate incorporation into cationic polymers, which enhances selectivity and antibiofilm performance while mitigating cytotoxicity. Subsequent mechanistic studies investigated this synergy. It was found that the cationic block intercalated into the bacterial cell envelope, imparting bactericidal activity, while the carbohydrate segment acted as an antifouling domain, reducing bacterial adhesion and disrupting biofilm matrix interactions. Together, these two mechanisms suppressed biofilm growth and promoted dispersion, demonstrating that the combination of glycans and cationic functionalities can overcome the toxicity and hemolysis issues that often limit cationic antimicrobial polymers.202
13 ratio retained a comparable potency against S. aureus, while eliminating the hemolytic side effects. This modular strategy illustrates how glycan units can be incorporated within the polymer backbone, creating a framework to balance antibacterial potency with biocompatibility.201 This copolymer also displayed potency against community- and hospital-acquired methicillin-resistant S. aureus (MRSA) strains, including those resistant to vancomycin and daptomycin (Fig. 8). In addition to dispersing and eradicating MRSA biofilms with a 99.9% reduction (substantially outperforming vancomycin), the copolymer provided complete protection in vivo. All mice challenged with a lethal dose of MRSA survived when treated with the copolymer, compared to only a 67% survival rate for those treated with vancomycin. These results underscore the advantages of carbohydrate incorporation into cationic polymers, which enhances selectivity and antibiofilm performance while mitigating cytotoxicity. Subsequent mechanistic studies investigated this synergy. It was found that the cationic block intercalated into the bacterial cell envelope, imparting bactericidal activity, while the carbohydrate segment acted as an antifouling domain, reducing bacterial adhesion and disrupting biofilm matrix interactions. Together, these two mechanisms suppressed biofilm growth and promoted dispersion, demonstrating that the combination of glycans and cationic functionalities can overcome the toxicity and hemolysis issues that often limit cationic antimicrobial polymers.202
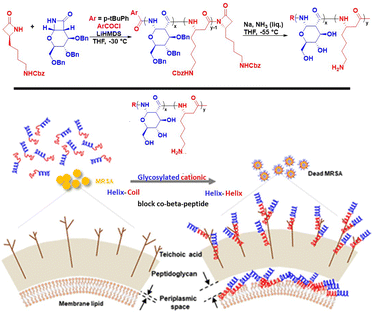 | ||
| Fig. 8 Interaction between the Co-β-peptide and Cell Envelope Components of MRSA. Reproduced from ref. 201 with permission from Springer Nature, copyright 2019. | ||
Similarly, Dai et al. developed cationic glycopolymers that incorporated a photodynamic therapy (PDT) unit to generate reactive oxygen species (ROS) (Fig. 9).203 The design involved thiazole-derived cations that anchored within bacterial membranes, while the PDT moiety produced ROS to induce bactericidal effects. Galactose units were incorporated to enhance biocompatibility and bacterial specificity. The resulting polymers inhibited biofilm formation and eradicated S. aureus and P. aeruginosa biofilms, with increasing potency with increasing glycan content. Notably, cytotoxicity assays revealed ∼80% cell viability at concentrations greater than 500-fold above the MIC, highlighting the material's large therapeutic window. Collectively, these studies illustrate the value of pairing cationic activity with carbohydrate functionalities to create potent, selective antibiofilm agents. By simultaneously disrupting bacterial membranes, dispersing biofilm matrices, and maintaining host compatibility, cationic glycopolymers address many of the limitations inherent to purely sugar- or cationic-based systems.
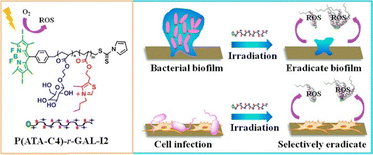 | ||
| Fig. 9 Cationic glycopolymers with PDT units to generate ROS for combating biofilm formation. Reproduced from ref. 203 with permission from the American Chemical Society, copyright 2018. | ||
However, significant obstacles still limit the translation of cationic glycopolymers into clinical practice. Carbohydrate incorporation often attenuates cytotoxicity of cationic scaffolds in vitro; however, this benefit can fail to manifest in vivo.204 Additionally, manipulating the material's structural parameters (i.e., charge density, hydrophilicity, molecular weight, and architecture) can strongly influence the in vivo toxicity of cationic polymers.205 However, these parameters have not been systematically examined for cationic glycopolymers.
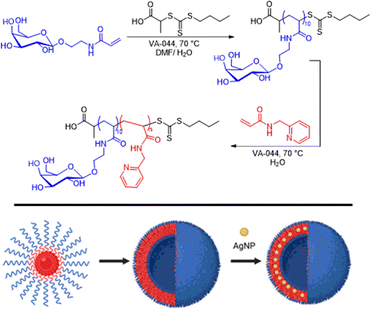 | ||
| Fig. 10 Illustration of glycopolymersome-supported AgNP composite.212 | ||
Against E. coli, the MIC decreased from >62 µg mL−1 for AgNPs to 16 µg mL−1 for the nanocomposites. Concentration-dependent biofilm inhibition was also observed, with 70–80% inhibition across E. coli, S. aureus, and P. aeruginosa. The strongest efficacy was against P. aeruginosa, which was attributed to galactose–LecA lectin interactions. The cytocompatibility also improved: hemolysis remained below 5% while cell viability remained above 70% at all concentrations tested, reflecting the protective role of the glycopolymer shell. In vivo evaluation using a rat dentin model with S. mutans showed that these glycosylated nanocomposites fully prevented biofilm formation over 14 days, whereas untreated controls or treatments with AgNPs or glycopolymers alone were ineffective. This work illustrates how encapsulating silver nanoparticles within glycopolymersomes can lower toxicity, enhance antibacterial potency, and improve biofilm inhibition, highlighting the potential of glyconanocomposites as multifunctional, biocompatible antibiofilm platforms.
While metallic nanomaterials have dominated glyconanotechnology research, non-metallic nanostructures conjugated with glycopolymers have also been explored for antibiofilm applications. A prevalent strategy leverages glycosylated nanoassemblies as targeted drug-delivery vehicles to enhance the penetration and efficacy of conventional antibiotics within biofilms.213
In parallel, glycopolymer-based nanoparticles have been engineered to deliver PDT agents. In these systems, the glycopolymer shell mediates selective biofilm targeting and penetration, after which irradiation induces the generation of ROS to eradicate resident bacteria while minimizing off-target damage.214–216 For instance, Tricomi et al. investigated glycopolymer-coated graphene oxide nanomaterials to combat P. aeruginosa biofilms.217 In this study, L-fucose, D-mannose, and D-galactose were conjugated to graphene oxide surfaces through a green mechanochemical ball-milling process and subsequently evaluated for their antibacterial efficacy. The fucose- and galactose-functionalized derivatives killed more than 99% of P. aeruginosa at half the concentration of unfunctionalized graphene oxide. Similarly, these derivatives significantly reduced biofilm attachment to polystyrene surfaces. Scanning electron microscopy revealed that the glycosylated graphene oxides were able to inhibit bacteria from forming mature biofilm colonies, whereas the control graphene oxide did not. This work reinforces the observation that strategic carbohydrate functionalization can enhance both the potency and biofilm-disrupting capabilities of antimicrobial nanomaterials.
Despite the rapid growth of glycosylated nanomaterials, some debate remains over their associated health risks.218 Designing non-toxic nanomaterials has proven challenging due to the intrinsic structural complexity and unique behaviors of nanomaterial structures (e.g., aggregation), making standard toxicological measurements potentially insufficient for capturing the material's full biological impact.219,220 This challenge is particularly relevant for the multifunctional systems discussed here, where surface coatings, ligands, and diverse functional groups can influence interactions with biological environments. Comprehensive and tailored toxicological studies are thus essential to assess the safety, biodistribution, and long-term effects of these emerging antimicrobial platforms.
Although not strictly classified as glycopolymers, polysaccharides such as cationic poly(acetyl, arginyl) glucosamine (PAAG) have been extensively studied for their ability to inhibit and disrupt biofilms across multiple bacterial species.221–224 These systems provide valuable mechanistic insights that may guide the rational design and safety optimization of future glycosylated materials. While polysaccharide-based materials such as PAAG have advanced into clinical trials, no synthetic glycopolymer systems are currently under such evaluation.
Key barriers to translation include the cost and complexity of synthesis, which can impede large-scale production and commercial viability. However, these challenges are not unique to glycomaterials and echo similar challenges that currently successful therapeutic materials also faced during their early development. Moving forward, establishing predictable, cost-effective, and scalable synthetic routes will be essential to enable broader application. Equally important will be the integration of glycopolymers into practical delivery platforms that align with clinical manufacturing and regulatory frameworks. By addressing these challenges, the field will be well-positioned to translate the unique advantages of glycopolymers from the laboratory into real-world antibiofilm intervention therapies.
Clinical perspectives
Antimicrobial resistance is one of the most significant global health challenges of the twenty-first century, projected to contribute to more than 8 million deaths per year by 2050.231 No major new classes of antibiotics have been discovered since the 1980s, and most antibiotic candidates are based on existing chemical frameworks.232–235 Importantly, reliance on conventional systemic antimicrobials not only drives drug resistance but also causes unintended off-target effects, ranging from potentially irreversible side effects to profound disruptions in the microbiome, including Clostridioides difficile infections.236,237 For example, it is estimated that nearly one in five emergency room visits in the United States are related to an antibiotic adverse event,238 and exposure to antimicrobials directly affects the risk of developing drug-resistance genes.239The overall prevalence of medical devices has increased in recent decades, coinciding with advancements in device development and an aging population.240,241 Devices now exist for almost every organ system, including central venous and urinary catheters, surgical meshes, vascular grafts, shunts, deep brain stimulators, cochlear implants, prosthetic valves, pacemaker-defibrillator systems, left ventricular assist devices, and prosthetic joints. This increase in device use has also led to a rise in associated infections.240,242 As expected, infections on devices that are difficult to remove surgically are associated with high rates of morbidity and mortality.240,242 For example, prosthetic-valve endocarditis is associated with over 30% mortality,242,243 and prosthetic hip infections carry a 21% mortality rate by 5-years, a 4-fold increase from age-based rates.244 Often, these types of infections require both invasive surgical procedures and prolonged courses of systemic antibiotics, posing a significant burden to patients and the healthcare system.241–243
Though some prophylactic techniques have been developed, approaches to biofilm-related infections are largely reactive, focusing on treatment only after an infection has been diagnosed. There is a critical need for biofilm prevention strategies that exist outside of these treatment paradigms. The design of polymers that prevent surface adhesion could offer multiple advantages for implanted devices, including a lower risk of colonization, reduced reliance on systemic antimicrobials, and more selective local targeting of specific pathogens or organ systems. To have a clinical impact, such surfaces would not necessarily need to be bactericidal, since planktonic cells are readily cleared by the immune system or targeted antimicrobials. Such an approach represents a treatment strategy that preserves conventional antimicrobials, combats multidrug-resistant pathogens, and protects the host microbiome.
Conclusion
Throughout this review, we have highlighted recent advances in the design and application of polymeric materials for antibiofilm purposes. Current research demonstrates two major approaches: (i) engineering polymers with intrinsic antibiofilm activity through chemical and structural modification, and (ii) employing polymers as platforms to enhance the delivery or efficacy of established antimicrobial agents.The diversity of polymer functionalities explored to date—including cationic moieties, nitric oxide donors, metallic and glycosylated systems, photosensitizers, and antibiotic conjugates—underscores the versatility of synthetic polymers in addressing biofilm-associated infections. Subtle changes in polymer parameters such as topology, molecular weight, backbone identity, and functional group chemistry can yield markedly different antibiofilm outcomes. Glycopolymers are a particularly interesting case, where small variations in glycan stereochemistry can switch their effects from inhibitory to promotive, revealing unique biofilm-modulating mechanisms beyond conventional bactericidal action.
Despite this promise, clinical translation of antibacterial polymers remains a major challenge. Synthetic complexity, high production costs, and limited scalability often hinder real-world implementation. Consequently, a growing emphasis has emerged on developing practical, application-driven materials. Moving forward, efforts to develop cost-effective and scalable fabrication methods must proceed in parallel with the discovery of new antibiofilm materials. Realizing the translational potential of these materials will require integrating polymer design with device engineering, thus ensuring that materials are optimized not only for performance but also for deployment in clinical and commercial settings.
Beyond these translational obstacles, major fundamental questions remain. The mechanisms by which polymers interact with bacterial membranes, EPS networks, and surface adhesins remain only partly understood, and even small changes in polymer structure can produce large effects. Mapping these (often nonlinear) structure–function relationships and integrating them into predictive models is both a major challenge and a major opportunity. A deeper mechanistic foundation will make it possible to move beyond empirical screening toward an increasingly rational and sophisticated design of antibiofilm polymers.
As scientific and translational challenges are addressed in parallel, the field is poised for rapid progress. The convergence of increasingly precise polymer synthesis tools, deeper microbiology insights, and growing clinical need creates an exceptional opportunity. Increasing interactions among historically siloed but scientifically interrelated fields allow us to ask—and answer—questions that were out of reach only a few years ago. As these advances come together, the prospect of creating robust, clinically deployable antibiofilm materials becomes increasingly realistic. The next generation of polymer-based strategies may therefore not only complement existing therapies, but fundamentally change how we prevent and treat biofilm-related infections.
Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included, and no new data were generated or analysed as part of this review.Acknowledgements
Our work in this area has been supported by GlycoMIP, an NSF Materials Innovation Platform funded through Cooperative Agreement DMR-1933525. Figures were created using Biorender.References
- M. Matijašić, T. Meštrović, H. Paljetak, M. Perić, A. Barešić and D. Verbanac, Gut Microbiota beyond Bacteria-Mycobiome, Virome, Archaeome, and Eukaryotic Parasites in IBD, Int. J. Mol. Sci., 2020, 21, 8 CrossRef PubMed.
- M. Eisenstein, The skin microbiome, Nature, 2020, 588(7838), S209–S209 CrossRef CAS.
- K. A. Floyd; A. R. Eberly and M. Hadjifrangiskou, 3 - Adhesion of bacteria to surfaces and biofilm formation on medical devices, In Biofilms and Implantable Medical Devices, ed Y. Deng; W. Lv, Woodhead Publishing, 2017, pp. 47–95 Search PubMed.
- L. Sedghi, V. DiMassa, A. Harrington, S. V. Lynch and Y. L. Kapila, The oral microbiome: Role of key organisms and complex networks in oral health and disease, Periodontology, 2021, 87(1), 107–131 CrossRef PubMed.
- K. Kriebel, C. Hieke, B. Müller-Hilke, M. Nakata and B. Kreikemeyer, Oral Biofilms from Symbiotic to Pathogenic Interactions and Associated Disease -Connection of Periodontitis and Rheumatic Arthritis by Peptidylarginine Deiminase, Front. Microbiol., 2018, 9, 53 CrossRef PubMed.
- A. Maddi and F. A. Scannapieco, Oral biofilms, oral and periodontal infections, and systemic disease, Am. J. Dent., 2013, 26(5), 249–254 Search PubMed.
- S. Kitamoto, H. Nagao-Kitamoto, R. Hein, T. M. Schmidt and N. Kamada, The Bacterial Connection between the Oral Cavity and the Gut Diseases, J. Dent. Res., 2020, 99(9), 1021–1029 CrossRef CAS PubMed.
- I. Rowland, G. Gibson, A. Heinken, K. Scott, J. Swann, I. Thiele and K. Tuohy, Gut microbiota functions: metabolism of nutrients and other food components, Eur. J. Nutr., 2018, 57(1), 1–24 CrossRef CAS PubMed.
- N. M. Delzenne, C. Knudsen, M. Beaumont, J. Rodriguez, A. M. Neyrinck and L. B. Bindels, Contribution of the gut microbiota to the regulation of host metabolism and energy balance: a focus on the gut–liver axis, Proc. Nutr. Soc., 2019, 78(3), 319–328 CrossRef CAS.
- E. M. Halawa, M. Fadel, M. W. Al-Rabia, A. Behairy, N. A. Nouh, M. Abdo, R. Olga, L. Fericean, A. M. Atwa, M. El-Nablaway and A. Abdeen, Antibiotic action and resistance: updated review of mechanisms, spread, influencing factors, and alternative approaches for combating resistance, Front. Pharmacol., 2024, 14, 2023 Search PubMed.
- J. L. Del Pozo and R. Patel, The Challenge of Treating Biofilm-associated Bacterial Infections, Clin. Pharmacol. Ther., 2007, 82(2), 204–209 CrossRef CAS PubMed.
- N. N. Tran, T. Morrisette, S. C. J. Jorgensen, J. M. Orench-Benvenutti and R. Kebriaei, Current therapies and challenges for the treatment of Staphylococcus aureus biofilm-related infections, Pharmacotherapy, 2023, 43(8), 816–832 CrossRef CAS PubMed.
- C. Pouget, C. Dunyach-Remy, A. Pantel, S. Schuldiner, A. Sotto and J. P. Lavigne, Biofilms in Diabetic Foot Ulcers: Significance and Clinical Relevance, Microorganisms, 2020, 8, 10 CrossRef PubMed.
- A. I. Adler, E. J. Boyko, J. H. Ahroni and D. G. Smith, Lower-extremity amputation in diabetes. The independent effects of peripheral vascular disease, sensory neuropathy, and foot ulcers, Diabetes Care, 1999, 22(7), 1029–1035 CrossRef CAS PubMed.
- G. T. Werneburg, Catheter-Associated Urinary Tract Infections: Current Challenges and Future Prospects, Res. Rep. Urol., 2022, 14, 109–133 CAS.
- K. M. Al-Sayaghi, T. A. H. Alqalah, S. A. Alkubati, S. A. Alshoabi, M. Alsabri, G. G. Alrubaiee, M. A. Almoliky, K. A. Saleh, A. K. Al-Sayaghi, R. A. Elshatarat, Z. T. Saleh, A. M. Saleh and H. R. Abdel-Aziz, Healthcare workers’ compliance with the catheter associated urinary tract infection prevention guidelines: an observational study in Yemen, Antimicrob. Resis. Infection Control, 2023, 12(1), 144 CrossRef PubMed.
- V. G. Preda and O. Săndulescu, Communication is the key: biofilms, quorum sensing, formation and prevention, Discoveries, 2019, 7(3), 2359–7232 CrossRef PubMed.
- S. Mendhe, A. Badge, S. Ugemuge and D. Chandi, Impact of Biofilms on Chronic Infections and Medical Challenges, Cureus, 2023, 15(11), e48204 Search PubMed.
- M. A. Rather, K. Gupta and M. Mandal, Microbial biofilm: formation, architecture, antibiotic resistance, and control strategies, Braz. J. Microbiol., 2021, 52(4), 1701–1718 CrossRef PubMed.
- S. Sharma, J. Mohler, S. D. Mahajan, S. A. Schwartz, L. Bruggemann and R. Aalinkeel, Microbial Biofilm: A Review on Formation, Infection, Antibiotic Resistance, Control Measures, and Innovative Treatment, Microorganisms, 2023, 11, 6 Search PubMed.
- J. Wang, P. Li, N. Wang, J. Wang and D. Xing, Antibacterial features of material surface: strong enough to serve as antibiotics?, J. Mater. Chem. B, 2023, 11(2), 280–302 RSC.
- S. Singh, S. Datta, K. B. Narayanan and K. N. Rajnish, Bacterial exo-polysaccharides in biofilms: role in antimicrobial resistance and treatments, J. Genet. Eng. Biotechnol., 2021, 19(1), 140 CrossRef PubMed.
- J. S. Gunn, L. O. Bakaletz and D. J. Wozniak, What's on the Outside Matters: The Role of the Extracellular Polymeric Substance of Gram-negative Biofilms in Evading Host Immunity and as a Target for Therapeutic Intervention, J. Biol. Chem., 2016, 291(24), 12538–12546 CrossRef CAS PubMed.
- A. Vertes, V. Hitchins and K. S. Phillips, Analytical Challenges of Microbial Biofilms on Medical Devices, Anal. Chem., 2012, 84(9), 3858–3866 CrossRef CAS.
- M. M. Zafer, G. A. Mohamed, S. R. M. Ibrahim, S. Ghosh, C. Bornman and M. A. Elfaky, Biofilm-mediated infections by multidrug-resistant microbes: a comprehensive exploration and forward perspectives, Arch. Microbiol., 2024, 206(3), 101 CrossRef CAS PubMed.
- D. Lebeaux, J. M. Ghigo and C. Beloin, Biofilm-related infections: bridging the gap between clinical management and fundamental aspects of recalcitrance toward antibiotics, Microbiol. Mol. Biol. Rev., 2014, 1098–5557 Search PubMed.
- D. Mitra, E.-T. Kang and K. G. Neoh, Polymer-Based Coatings with Integrated Antifouling and Bactericidal Properties for Targeted Biomedical Applications, ACS Appl. Polym. Mater., 2021, 3(5), 2233–2263 CrossRef CAS.
- S. Magill Shelley, R. Edwards Jonathan, W. Bamberg, G. Beldavs Zintars, G. Dumyati, A. Kainer Marion, R. Lynfield, M. Maloney, L. McAllister-Hollod, J. Nadle, M. Ray Susan, D. L. Thompson, L. E. Wilson and S. K. Fridkin, Multistate Point-Prevalence Survey of Health Care–Associated Infections, N. Engl. J. Med., 2014, 370(13), 1198–1208 CrossRef CAS PubMed.
- H. Wu, C. Moser, H.-Z. Wang, N. Høiby and Z.-J. Song, Strategies for combating bacterial biofilm infections, Int. J. Oral Sci., 2015, 7(1), 1–7 CrossRef CAS PubMed.
- Z. Song, L. Borgwardt, N. Høiby, H. Wu, T. S. Sørensen and A. Borgwardt, Prosthesis infections after orthopedic joint replacement: the possible role of bacterial biofilms, Orthop. Rev., 2013, 5(2), 65–71 Search PubMed.
- A. Silverstein and C. F. Donatucci, Bacterial Biofilms and implantable prosthetic devices, Int. J. Impotence Res., 2003, 15(S5), S150–S154 CrossRef PubMed.
- H. O. Gbejuade, A. M. Lovering and J. C. Webb, The role of microbial biofilms in prosthetic joint infections, Acta Orthop., 2015, 86(2), 147–158 CrossRef PubMed.
- A. Hrynyshyn, M. Simões and A. Borges, Biofilms in Surgical Site Infections: Recent Advances and Novel Prevention and Eradication Strategies, Antibiotics, 2022, 11, 1 CrossRef PubMed.
- A. Giacometti, O. Cirioni, A. M. Schimizzi, M. S. Del Prete, F. Barchiesi, M. M. D'Errico, E. Petrelli and G. Scalise, Epidemiology and microbiology of surgical wound infections, J. Clin. Microbiol., 2000, 38(2), 918–922 CrossRef CAS PubMed.
- E. A. Saverina, N. A. Frolov, O. A. Kamanina, V. A. Arlyapov, A. N. Vereshchagin and V. P. Ananikov, From Antibacterial to Antibiofilm Targeting: An Emerging Paradigm Shift in the Development of Quaternary Ammonium Compounds (QACs), ACS Infect. Dis., 2023, 9(3), 394–422 CrossRef CAS PubMed.
- M. A. Kohanski, D. J. Dwyer and J. J. Collins, How antibiotics kill bacteria: from targets to networks, Nat. Rev. Microbiol., 2010, 8(6), 423–435 CrossRef CAS PubMed.
- J. Davies, Are antibiotics naturally antibiotics?, J. Ind. Microbiol. Biotechnol., 2006, 33(7), 496–499 CrossRef CAS PubMed.
- S. Q. Shah, F. C. Cabello, T. M. L'Abée-Lund, A. Tomova, H. P. Godfrey, A. H. Buschmann and H. Sørum, Antimicrobial resistance and antimicrobial resistance genes in marine bacteria from salmon aquaculture and non-aquaculture sites, Environ. Microbiol., 2014, 16(5), 1310–1320 CrossRef CAS PubMed.
- D. Hocquet, A. Muller and X. Bertrand, What happens in hospitals does not stay in hospitals: antibiotic-resistant bacteria in hospital wastewater systems, J. Hosp. Infect., 2016, 93(4), 395–402 CrossRef CAS PubMed.
- J. E. M. Watts, H. J. Schreier, L. Lanska and M. S. Hale, The Rising Tide of Antimicrobial Resistance in Aquaculture: Sources, Sinks and Solutions, Mar. Drugs, 2017, 15(6), 158 CrossRef PubMed.
- A. Pruden, R. Pei, H. Storteboom and K. H. Carlson, Antibiotic Resistance Genes as Emerging Contaminants:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Studies in Northern Colorado, Environ. Sci. Technol., 2006, 40(23), 7445–7450 CrossRef CAS PubMed.
Studies in Northern Colorado, Environ. Sci. Technol., 2006, 40(23), 7445–7450 CrossRef CAS PubMed. - J. Wang, L. Chu, L. Wojnárovits and E. Takács, Occurrence and fate of antibiotics, antibiotic resistant genes (ARGs) and antibiotic resistant bacteria (ARB) in municipal wastewater treatment plant: An overview, Sci. Total Environ., 2020, 744, 140997 CrossRef CAS PubMed.
- F. C. Cabello, H. P. Godfrey, A. Tomova, L. Ivanova, H. Dölz, A. Millanao and A. H. Buschmann, Antimicrobial use in aquaculture re-examined: its relevance to antimicrobial resistance and to animal and human health, Environ. Microbiol., 2013, 15(7), 1917–1942 CrossRef PubMed.
- J. Bengtsson-Palme and D. G. Larsson, Concentrations of antibiotics predicted to select for resistant bacteria: Proposed limits for environmental regulation, Environ. Int., 2016, 86, 140–149 CrossRef CAS PubMed.
- D. Davies, Understanding biofilm resistance to antibacterial agents, Nat. Rev. Drug Discovery, 2003, 2(2), 114–122 CrossRef CAS PubMed.
- J. W. Costerton, P. S. Stewart and E. P. Greenberg, Bacterial biofilms: a common cause of persistent infections, Science, 1999, 284(5418), 1318–1322 CrossRef CAS PubMed.
- C. Sternberg, B. B. Christensen, T. Johansen, A. Toftgaard Nielsen, J. B. Andersen, M. Givskov and S. Molin, Distribution of bacterial growth activity in flow-chamber biofilms, Appl. Environ. Microbiol., 1999, 65(9), 4108–4117 CrossRef CAS PubMed.
- P. A. Suci, M. W. Mittelman, F. P. Yu and G. G. Geesey, Investigation of ciprofloxacin penetration into Pseudomonas aeruginosa biofilms, Antimicrob. Agents Chemother., 1994, 38(9), 2125–2133 CrossRef CAS PubMed.
- N. Gargate, M. Laws and K. M. Rahman, Current economic and regulatory challenges in developing antibiotics for Gram-negative bacteria, npj Antimicrob. Resist., 2025, 3(1), 50 CrossRef CAS PubMed.
- A new class of antibiotics is cause for cautious celebration — but the economics must be fixed, Nature, 2024, 6257993, 7 Search PubMed.
- G. S. Burrill, Personalized Medicine or Blockbusterology?, Cancer, 2002, 2(41.3), 5.2 Search PubMed.
- J. F. Barrett, Can biotech deliver new antibiotics?, Curr. Opin. Microbiol., 2005, 8(5), 498–503 CrossRef PubMed.
- A. Banerjee, P. Chowdhury, K. Bauri, B. Saha and P. De, Inhibition and eradication of bacterial biofilm using polymeric materials, Biomater. Sci., 2023, 11(1), 11–36 RSC.
- A. Valliammai, A. Selvaraj, P. Mathumitha, C. Aravindraja and S. K. Pandian, Polymeric antibiofilm coating comprising synergistic combination of citral and thymol prevents methicillin-resistant Staphylococcus aureus biofilm formation on titanium, Mater. Sci. Eng. C, 2021, 121, 111863 CrossRef CAS PubMed.
- A. Copling, M. Akantibila, R. Kumaresan, G. Fleischer, D. Cortes, R. S. Tripathi, V. J. Carabetta and S. L. Vega, Recent Advances in Antimicrobial Peptide Hydrogels, Int. J. Mol. Sci., 2023, 24, 8 Search PubMed.
- M. Wang, Y. Zheng, C. Yin, S. Dai, X. Fan, Y. Jiang, X. Liu, J. Fang, B. Yi, Q. Zhou and T. Wang, Recent Progress in antibacterial hydrogel coatings for targeting biofilm to prevent orthopedic implant-associated infections, Front. Microbiol., 2023, 14 CAS.
- T. Peng, Q. Shi, M. Chen, W. Yu and T. Yang, Antibacterial-Based Hydrogel Coatings and Their Application in the Biomedical Field-A Review, J. Funct. Biomater., 2023, 14, 5 Search PubMed.
- D. Pranantyo, C. K. Yeo, Y. Wu, C. Fan, X. Xu, Y. S. Yip, M. I. G. Vos, S. H. Mahadevegowda, P. L. K. Lim, L. Yang, P. T. Hammond, D. I. Leavesley, N. S. Tan and M. B. Chan-Park, Hydrogel dressings with intrinsic antibiofilm and antioxidative dual functionalities accelerate infected diabetic wound healing, Nat. Commun., 2024, 15(1), 954 CrossRef CAS PubMed.
- D. U. Lee, M. Kayumov, J. Park, S. K. Park, Y. Kang, Y. Ahn, W. Kim, S. H. Yoo, J.-K. Park, B.-G. Kim, Y. S. Oh, I.-S. Jeong and D. Y. Choi, Antibiofilm and antithrombotic hydrogel coating based on superhydrophilic zwitterionic carboxymethyl chitosan for blood-contacting devices, Bioact. Mater., 2024, 34, 112–124 CAS.
- S. V. Pavlukhina, J. B. Kaplan, L. Xu, W. Chang, X. Yu, S. Madhyastha, N. Yakandawala, A. Mentbayeva, B. Khan and S. A. Sukhishvili, Noneluting Enzymatic Antibiofilm Coatings, ACS Appl. Mater. Interfaces, 2012, 4(9), 4708–4716 CrossRef CAS PubMed.
- I. Negut, B. Bita and A. Groza, Polymeric Coatings and Antimicrobial Peptides as Efficient Systems for Treating Implantable Medical Devices Associated-Infections, Polymers, 2022, 14, 8 CrossRef.
- N. Raheem and S. K. Straus, Mechanisms of Action for Antimicrobial Peptides With Antibacterial and Antibiofilm Functions, Front. Microbiol., 2019, 10 Search PubMed.
- B. Zhang, D. Lu and H. Duan, Recent advances in responsive antibacterial materials: design and application scenarios, Biomater. Sci., 2023, 11(2), 356–379 RSC.
- H. Q. Karp, E. S. Nowak, G. A. Kropp, N. A. Col, M. D. Schulz, N. Sriranganathan and J. Rao, Broad Host Range Peptide Nucleic Acids Prevent Gram-negative Biofilms Implicated in Catheter-Associated Urinary Tract Infections, Microorganisms, 2025, 1948 CrossRef CAS.
- Z. Guo, Y. Wang, T. Tan, Y. Ji, J. Hu and Y. Zhang, Antimicrobial d-Peptide Hydrogels, ACS Biomater. Sci. Eng., 2021, 7(4), 1703–1712 CrossRef CAS PubMed.
- M. Yang, R. Xing, G. Shen, C. Yuan and X. Yan, A versatile cyclic dipeptide hydrogelator: Self-assembly and rheology in various physiological conditions, Colloids Surf., A, 2019, 572, 259–265 CrossRef CAS.
- Z. Jiang, J. Feng, F. Wang, J. Wang, N. Wang, M. Zhang, C.-Y. Hsieh, T. Hou, W. Cui and L. Ma, AI-Guided Design of Antimicrobial Peptide Hydrogels for Precise Treatment of Drug-resistant Bacterial Infections, Adv. Mater., 2025, 37(20), 2500043 CrossRef CAS.
- N. A. Obaid, S. Almarzoky Abuhussain, K. K. Mulibari, F. Alshanqiti, S. A. Malibari, S. S. Althobaiti, M. Alansari, E. Muneef, L. Almatrafi, A. Alqarzi, N. Alotaibi, A. M. Mostafa and A. Hagag, Antimicrobial-resistant pathogens related to catheter-associated urinary tract infections in intensive care units: A multi-center retrospective study in the Western region of Saudi Arabia, Clin. Epidemiol. Global Health, 2023, 21, 101291 CrossRef CAS.
- H. Zhang, Z. Wu, J. Zhou, Z. Wang, C. Yang, P. Wang, M. S. Fareed, Y. He, J. Su, R. Cha and K. Wang, The Antimicrobial, Hemostatic, and Anti-Adhesion Effects of a Peptide Hydrogel Constructed by the All-d-Enantiomer of Antimicrobial Peptide Jelleine-1, Adv. Healthcare Mater., 2023, 12(29), e2301612 CrossRef PubMed.
- S. Debnath, A. Shome, D. Das and P. K. Das, Hydrogelation Through Self-Assembly of Fmoc-Peptide Functionalized Cationic Amphiphiles: Potent Antibacterial Agent, J. Phys. Chem. B, 2010, 114(13), 4407–4415 CrossRef CAS PubMed.
- A. P. McCloskey, E. R. Draper, B. F. Gilmore and G. Laverty, Ultrashort self-assembling Fmoc-peptide gelators for anti-infective biomaterial applications, J. Pept. Sci., 2017, 23(2), 131–140 CrossRef CAS PubMed.
- E. R. Cross, S. M. Coulter, S. Pentlavalli and G. Laverty, Unravelling the antimicrobial activity of peptide hydrogel systems: current and future perspectives, Soft Matter, 2021, 17(35), 8001–8021 RSC.
- V. R. Aldilla, R. Chen, R. Kuppusamy, S. Chakraborty, M. D. P. Willcox, D. S. Black, P. Thordarson, A. D. Martin and N. Kumar, Hydrogels with intrinsic antibacterial activity prepared from naphthyl anthranilamide (NaA) capped peptide mimics, Sci. Rep., 2022, 12(1), 22259 CrossRef PubMed.
- N. P. Desai and J. A. Hubbell, Biological responses to polyethylene oxide modified polyethylene terephthalate surfaces, J. Biomed. Mater. Res., 1991, 25(7), 829–843 CrossRef CAS PubMed.
- J. Hoffmann, J. Groll, J. Heuts, H. Rong, D. Klee, G. Ziemer, M. Moeller and H. P. Wendel, Blood cell and plasma protein repellent properties of star-PEG-modified surfaces, J. Biomater. Sci., Polym. Ed., 2006, 17(9), 985–996 CrossRef CAS PubMed.
- W. R. Gombotz, W. Guanghui, T. A. Horbett and A. S. Hoffman, Protein adsorption to poly(ethylene oxide) surfaces, J. Biomed. Mater. Res., 1991, 25(12), 1547–1562 CrossRef CAS PubMed.
- P. Lundberg, A. Bruin, J. W. Klijnstra, A. M. Nyström, M. Johansson, M. Malkoch and A. Hult, Poly(ethylene glycol)-Based Thiol-ene Hydrogel Coatings−Curing Chemistry, Aqueous Stability, and Potential Marine Antifouling Applications, ACS Appl. Mater. Interfaces, 2010, 2(3), 903–912 CrossRef CAS PubMed.
- X. He, G. Sathishkumar, K. Gopinath, K. Zhang, Z. Lu, C. Li, E.-T. Kang and L. Xu, One-step self-assembly of biogenic Au NPs/PEG-based universal coatings for antifouling and photothermal killing of bacterial pathogens, Chem. Eng. J., 2021, 421, 130005 CrossRef CAS.
- M. Charnley, M. Textor and C. Acikgoz, Designed polymer structures with antifouling–antimicrobial properties, React. Funct. Polym., 2011, 71(3), 329–334 CrossRef CAS.
- W. Xi, V. Hegde, S. D. Zoller, H. Y. Park, C. M. Hart, T. Kondo, C. D. Hamad, Y. Hu, A. H. Loftin, D. O. Johansen, Z. Burke, S. Clarkson, C. Ishmael, K. Hori, Z. Mamouei, H. Okawa, I. Nishimura, N. M. Bernthal and T. Segura, Point-of-care antimicrobial coating protects orthopaedic implants from bacterial challenge, Nat. Commun., 2021, 12(1), 5473 CrossRef CAS PubMed.
- Z. Li, X. Yang, H. Liu, X. Yang, Y. Shan, X. Xu, S. Shang and Z. Song, Dual-functional antimicrobial coating based on a quaternary ammonium salt from rosin acid with in vitro and in vivo antimicrobial and antifouling properties, Chem. Eng. J., 2019, 374, 564–575 CrossRef CAS.
- A. M. C. Maan, A. H. Hofman, W. M. de Vos and M. Kamperman, Recent Developments and Practical Feasibility of Polymer-Based Antifouling Coatings, Adv. Funct. Mater., 2020, 30(32), 2000936 CrossRef CAS.
- A. I. Stavrakis, S. Zhu, V. Hegde, A. H. Loftin, A. G. Ashbaugh, J. A. Niska, L. S. Miller, T. Segura and N. M. Bernthal, In Vivo Efficacy of a “Smart” Antimicrobial Implant Coating, J. Bone Jt. Surg., 2016, 98(14), 1183–1189 CrossRef PubMed.
- T. S. Sileika, H.-D. Kim, P. Maniak and P. B. Messersmith, Antibacterial Performance of Polydopamine-Modified Polymer Surfaces Containing Passive and Active Components, ACS Appl. Mater. Interfaces, 2011, 3(12), 4602–4610 CrossRef CAS.
- B. A. Hall, O. J. Wadsworth, L. M. Breiner, J. C. Chappell, A. S. Brenner, J. P. McCord, A. N. Lowell and M. D. Schulz, Inherent antibacterial properties of mannose-containing polynorbornene glycomaterials, Chem. Commun., 2025, 61(23), 4539–4542 RSC.
- J. D. Denstedt and P. A. Cadieux, Eliminating biofilm from ureteral stents: the holy grail, Curr. Opin. Neurol., 2009, 19, 2 Search PubMed.
- J. S. Kim, E. Kuk, K. N. Yu, J.-H. Kim, S. J. Park, H. J. Lee, S. H. Kim, Y. K. Park, Y. H. Park, C.-Y. Hwang, Y.-K. Kim, Y.-S. Lee, D. H. Jeong and M.-H. Cho, Antimicrobial effects of silver nanoparticles, Nanomedicine, 2007, 3(1), 95–101 CrossRef CAS PubMed.
- T. Maneerung, S. Tokura and R. Rujiravanit, Impregnation of silver nanoparticles into bacterial cellulose for antimicrobial wound dressing, Carbohydr. Polym., 2008, 72(1), 43–51 CrossRef CAS.
- M. Rai, A. Yadav and A. Gade, Silver nanoparticles as a new generation of antimicrobials, Biotechnol. Adv., 2009, 27(1), 76–83 CrossRef CAS PubMed.
- D. R. Monteiro, L. F. Gorup, S. Silva, M. Negri, E. R. de Camargo, R. Oliveira, D. B. Barbosa and M. Henriques, Silver colloidal nanoparticles: antifungal effect against adhered cells and biofilms of Candida albicans and Candida glabrata, Biofouling, 2011, 27(7), 711–719 CrossRef CAS PubMed.
- W.-A. Chen, D.-Y. Chang, B.-M. Chen, Y.-C. Lin, Y. Barenholz and S. R. Roffler, Antibodies against Poly(ethylene glycol) Activate Innate Immune Cells and Induce Hypersensitivity Reactions to PEGylated Nanomedicines, ACS Nano, 2023, 17(6), 5757–5772 CrossRef CAS PubMed.
- A. Bianchi, P. Bottau, E. Calamelli, S. Caimmi, G. Crisafulli, F. Franceschini, L. Liotti, F. Mori, C. Paglialunga, F. Saretta, M. Tosca, F. Cardinale, A. Licari, M. Miraglia Del Giudice and C. Caffarelli, Hypersensitivity to polyethylene glycol in adults and children: An emerging challenge, Acta Biomed., 2021, 92(S7), e2021519 CAS.
- M. Barz, R. Luxenhofer, R. Zentel and M. J. Vicent, Overcoming the PEG-addiction: well-defined alternatives to PEG, from structure–property relationships to better defined therapeutics, Polym. Chem., 2011, 2(9), 1900–1918 RSC.
- T. T. Hoang Thi, E. H. Pilkington, D. H. Nguyen, J. S. Lee, K. D. Park and N. P. Truong, The Importance of Poly(ethylene glycol) Alternatives for Overcoming PEG Immunogenicity in Drug Delivery and Bioconjugation, Polymers, 2020, 298 CrossRef PubMed.
- H. Yang, Y. Wang, L. Yao, J. Wang and H. Chen, Antifouling Polymer Coatings for Bioactive Surfaces, Langmuir, 2025, 41(10), 6471–6496 CrossRef CAS.
- R. Qiao, C. Fu, Y. Li, X. Qi, D. Ni, A. Nandakumar, G. Siddiqui, H. Wang, Z. Zhang, T. Wu, J. Zhong, S.-Y. Tang, S. Pan, C. Zhang, M. R. Whittaker, J. W. Engle, D. J. Creek, F. Caruso, P. C. Ke, W. Cai, A. K. Whittaker and T. P. Davis, Sulfoxide-Containing Polymer-Coated Nanoparticles Demonstrate Minimal Protein Fouling and Improved Blood Circulation, Adv. Sci., 2020, 7(13), 2000406 CrossRef CAS PubMed.
- Y. Zhang, M. Hu, R. Xin, X. Xu, Z. Zhang, H. Peng, R. Cai, C. Chen, A. K. Whittaker and C. Fu, Enhanced Hydrophilicity and Antifouling Performance of PEG with Sulfoxide-Containing Side Chains for Nanomedicine Applications, Polym. Sci. Technol., 2025, 1(7), 640–650 CrossRef CAS PubMed.
- J. Yook, D. Jeong and J.-C. Lee, Synthesis of Citronellol-Derived Antibacterial Polymers and Effect of Thioether, Sulfoxide, Sulfone, and Ether Functional Groups on Their Bactericidal Activity, Macromolecules, 2023, 56(9), 3406–3420 CrossRef CAS.
- R. H. Pelton and A. Karami, Simulating Polydisperse Polymer Adsorption onto Porous Substrates, Langmuir, 2025, 41(25), 16625–16633 CrossRef CAS PubMed.
- A. S. Imbia, A. Ounkaew, X. Mao, H. Zeng, Y. Liu and R. Narain, Mussel-Inspired Polymer-Based Coating Technology for Antifouling and Antibacterial Properties, Langmuir, 2024, 40(21), 10957–10965 CrossRef PubMed.
- J. Wang, L. Li, Y. Wu and Y. Liu, Design and Application of Antifouling Bio-Coatings, Polymers, 2025, 793 CrossRef PubMed.
- C. Doyle and M. Ahmed, Antifouling Coatings Inspired by Biological Templates, Macromol. Rapid Commun., 2025, 46(12), 2400932 CrossRef CAS PubMed.
- Y. Fu, L. Yang, J. Zhang, J. Hu, G. Duan, X. Liu, Y. Li and Z. Gu, Polydopamine antibacterial materials, Mater. Horiz., 2021, 8(6), 1618–1633 RSC.
- L. Su, Y. Yu, Y. Zhao, F. Liang and X. Zhang, Strong Antibacterial Polydopamine Coatings Prepared by a Shaking-assisted Method, Sci. Rep., 2016, 6(1), 24420 CrossRef CAS PubMed.
- N. N. M. Adnan, Z. Sadrearhami, A. Bagheri, T.-K. Nguyen, E. H. H. Wong, K. K. K. Ho, M. Lim, N. Kumar and C. Boyer, Exploiting the Versatility of Polydopamine-Coated Nanoparticles to Deliver Nitric Oxide and Combat Bacterial Biofilm, Macromol. Rapid Commun., 2018, 39(13), 1800159 CrossRef PubMed.
- G. A. Kropp, H. Q. Karp, J. Rao, H. S. Muller, N. Sriranganathan, E. S. Nowak and M. D. Schulz, Polydopamine-Antibiotic Composite Coating for Antibiofilm Applications, Macromol. Biosci., 2025, e00501 Search PubMed.
- P. Kord Forooshani, E. Polega, K. Thomson, M. S. A. Bhuiyan, R. Pinnaratip, M. Trought, C. Kendrick, Y. Gao, K. A. Perrine, L. Pan and B. P. Lee, Antibacterial Properties of Mussel-Inspired Polydopamine Coatings Prepared by a Simple Two-Step Shaking-Assisted Method, Front. Chem., 2019, 7, 631 CrossRef PubMed.
- B.-B. Wang, Y.-H. Quan, Z.-M. Xu and Q. Zhao, Preparation of highly effective antibacterial coating with polydopamine/chitosan/silver nanoparticles via simple immersion, Prog. Org. Coat., 2020, 149, 105967 CrossRef CAS.
- W. Guo, Y. Wu, B. Li, L. Zhao, S. Feng, Y. Wei, C. Liu, Y. Huang, S. Wang, B. Liu, H. Mai, H. You and Y. Long, Silver-loaded polydopamine coating mediated dual-mode antibacterial and biological response for composite scaffolds, J. Am. Ceram. Soc., 2025, 108(11), e70066 CrossRef CAS.
- J. Cui, H. Shu, X. Gu, S. Wu, X. Liu and P. Cao, Enhancing antibacterial performance and stability of implant materials through surface modification with polydopamine/silver nanoparticles, Colloids Surf., B, 2025, 245, 114327 CrossRef CAS PubMed.
- H. Zhang, X. Shen, Z. Fei, X. Fan, L. Ma, H. Wang, C. Tian, B. Zhang, R. Luo, Y. Wang and S. Huang, Ag-Incorporated Polydopamine/Tannic Acid Coating on Titanium With Enhanced Cytocompatible and Antibacterial Properties, Front. Bioeng. Biotechnol., 2022, 10, 877738 CrossRef PubMed.
- Y.-J. Fan, M. T. Pham and C.-J. Huang, Development of Antimicrobial and Antifouling Universal Coating via Rapid Deposition of Polydopamine and Zwitterionization, Langmuir, 2019, 35(5), 1642–1651 CrossRef CAS PubMed.
- Y. Zhang, F. Wang, Q. Huang, A. B. Patil, J. Hu, L. Fan, Y. Yang, H. Duan, X. Dong and C. Lin, Layer-by-layer immobilizing of polydopamine-assisted ε-polylysine and gum Arabic on titanium: Tailoring of antibacterial and osteogenic properties, Mater. Sci. Eng. C, 2020, 110, 110690 CrossRef CAS PubMed.
- S. Lamba, K. Wang, J. Lu, A. R. J. Phillips, S. Swift and V. Sarojini, Polydopamine-Mediated Antimicrobial Lipopeptide Surface Coating for Medical Devices, ACS Appl. Bio Mater., 2024, 7(11), 7574–7584 CrossRef CAS PubMed.
- Z. Sadrearhami, F. N. Shafiee, K. K. K. Ho, N. Kumar, M. Krasowska, A. Blencowe, E. H. H. Wong and C. Boyer, Antibiofilm Nitric Oxide-Releasing Polydopamine Coatings, ACS Appl. Mater. Interfaces, 2019, 11(7), 7320–7329 CrossRef CAS PubMed.
- J. Mendhi, S. S. Ramachandra, I. Prasadam, S. Ivanovski, Y. Yang and Y. Xiao, Endogenous nitric oxide-generating surfaces via polydopamine-copper coatings for preventing biofilm dispersal and promoting microbial killing, Mater. Sci. Eng. C, 2021, 128, 112297 CrossRef CAS PubMed.
- L. Zhou, X. Li, K. Wang, F. Shen, L. Zhang, P. Li, T. Shang, J. Wang and N. Huang, Cu(‖)-loaded polydopamine coatings with in situ nitric oxide generation function for improved hemocompatibility, Regen. Biomater., 2020, 7(2), 153–160 CrossRef CAS PubMed.
- S. Geng, Q. Fan, K. Lin, F. Mazur and R. Chandrawati, Polydopamine-Coated Selenium Nanoparticles as a Stable Catalyst for Tunable and Sustained Nitric Oxide Generation, Small Sci., 2025, 5(8), 2500151 CrossRef PubMed.
- J. Li, B. Jiang, L. Yang, P. Zhang, J. Wu, Y. Yang, Y. Yang, G. Wang, J. Chen, L. Zhang, S. Huang, L. Zhang and E. Zhang, Dual-functional titanium implants via polydopamine-mediated lithium and copper co-incorporation: synergistic enhancement of osseointegration and antibacterial efficacy, Front. Bioeng. Biotechnol., 2025, 13, 1593545 CrossRef PubMed.
- T. Zhang, W. Zhou, Z. Jia, Q. Wei, D. Fan, J. Yan, C. Yin, Y. Cheng, H. Cai, X. Liu, H. Zhou, X. Yang, Y. Zheng and Z. Liu, Polydopamine-assisted functionalization of heparin and vancomycin onto microarc-oxidized 3D printed porous Ti6Al4V for improved hemocompatibility, osteogenic and anti-infection potencies, Sci. China Mater., 2018, 61(4), 579–592 CrossRef CAS.
- M. Chen, W. Yu, Q. Shi, H. Wen, G. Li, Y. Wang, T. Wang, S. Liu and T. Yang, Boosting the Anti-Infection Ability of Titanium Implants by Coating Polydopamine–Curcumin, Coatings, 2024, 640 CrossRef CAS.
- A. A. Ali, R. D. Al Bostami and A. Al-Othman, Nanogel-based composites for bacterial antibiofilm activity: advances, challenges, and prospects, RSC Adv., 2024, 14(15), 10546–10559 RSC.
- D. L. Bordón, E. Herrera, M. Laura González, L. I. Rossi, M. L. Aimar, A. M. Vázquez and A. M. Granados, Catalytic and biocidal activity of silver and gold nanoparticles obtained by green synthesis using aqueous extracts of glossy privet (Ligustrum lucidum) dry fruits, J. Mol. Liq., 2024, 415, 126378 CrossRef.
- R. Aguilar-Garay, L. F. Lara-Ortiz, M. Campos-López, D. E. Gonzalez-Rodriguez, M. M. Gamboa-Lugo, J. A. Mendoza-Pérez, Á. Anzueto-Ríos, D. E. Nicolás-Álvarez and A. Comprehensive, Review of Silver and Gold Nanoparticles as Effective Antibacterial Agents, Pharmaceuticals, 2024, 17(9), 1134 CrossRef CAS PubMed.
- A. Balestri, J. Cardellini and D. Berti, Gold and silver nanoparticles as tools to combat multidrug-resistant pathogens, Curr. Opin. Colloid Interface Sci., 2023, 66, 101710 CrossRef CAS.
- P. Singh and I. Mijakovic, Green synthesis and antibacterial applications of gold and silver nanoparticles from Ligustrum vulgare berries, Sci. Rep., 2022, 12(1), 7902 CrossRef CAS PubMed.
- M.-H. Moosavy, M. de la Guardia, A. Mokhtarzadeh, S. A. Khatibi, N. Hosseinzadeh and N. Hajipour, Green synthesis, characterization, and biological evaluation of gold and silver nanoparticles using Mentha spicata essential oil, Sci. Rep., 2023, 13(1), 7230 CrossRef CAS PubMed.
- N. Abed and P. Couvreur, Nanocarriers for antibiotics: A promising solution to treat intracellular bacterial infections, Int. J. Antimicrob. Agents, 2014, 43(6), 485–496 CrossRef CAS PubMed.
- P. J. Weldrick, M. J. Hardman and V. N. Paunov, Smart active antibiotic nanocarriers with protease surface functionality can overcome biofilms of resistant bacteria, Mater. Chem. Front., 2021, 5(2), 961–972 RSC.
- G. Prozorova, A. Emel'Yanov, A. Ivanova, T. Semenova, T. Y. Fadeeva, A. Nevezhina, S. Korzhova and A. Pozdnyakov, A novel water-soluble polymer nanocomposite containing ultra-small Fe3O4 nanoparticles with strong antibacterial and antibiofilm activity, Nanoscale, 2025, 17(3), 1458–1472 RSC.
- H. Yazdani-Ahmadabadi, K. Yu, S. Khoddami, D. F. Felix, H. H. Yeh, H. D. Luo, I. Moskalev, Q. Wang, R. Wang, D. Grecov, L. Fazli, D. Lange and J. N. Kizhakkedathu, Robust Nanoparticle-Derived Lubricious Antibiofilm Coating for Difficult-to-Coat Medical Devices with Intricate Geometry, ACS Nanosci. Au, 2023, 3(1), 67–83 CrossRef CAS PubMed.
- V. C. Ibarra, L. O. Vargas-Sanchez, A. Rodríguez-Moreno, N. V. Zavala-Alonso, M. Loredo-Tobias, M. E. García-Arreola and R. Manisekaran, Antibiofilm property of poly(methyl)methacrylate and silver nanocomposite on a mixed population of oral bacteria, Next Nanotechnol., 2025, 8, 100230 CrossRef CAS.
- A. S. Takamiya, D. R. Monteiro, L. F. Gorup, E. A. Silva, E. R. de Camargo, J. E. Gomes-Filho, S. H. P. de Oliveira and D. B. Barbosa, Biocompatible silver nanoparticles incorporated in acrylic resin for dental application inhibit Candida albicans biofilm, Mater. Sci. Eng. C, 2021, 118, 111341 CrossRef CAS PubMed.
- L. L. Hamid, R. S. Zakir, N. M. Abdel Rahman, R. H. Idrahim and S. M. Abdel Rahman, Construction and Characterization of Polyethylene Glycol/Sodium Alginate Hydrogel Loaded With Zirconia Nanoparticles: Potential Antibacterial and Antibiofilm Agent to Inhibit Dental Caries In Vitro and In Vivo, Microsc. Res. Tech., 2025, 88(8), 2268–2284 CrossRef CAS PubMed.
- W. Q. Hong, W.-H. Lee, S. H. Musa, N. A. Kamaruzaman and C.-Y. Loo, Evaluation of hydrogel loading with curcumin and silver nanoparticles: biocompatibilities and anti-biofilm activities, Biometals, 2025, 38(2), 663–682 CrossRef CAS PubMed.
- M. Solomon, A. M. Holban, B. Bălăceanu-Gurău, L. M. Ditu, A. Alberts, A. M. Grumezescu, L. S. Manolescu and M. M. Mihai, Silver Nanoparticles Functionalized with Polymeric Substances to Reduce the Growth of Planktonic and Biofilm Opportunistic Pathogens, Int. J. Mol. Sci., 2025, 3930 CrossRef CAS PubMed.
- Z. Chegini, M. Azizi, M. Safaiee, F. Kalhori, R. Zare Shahraki, J. Hemmati, S. M. Hosseini, A. Teimoori, A. Shariati and M. R. Arabestani, Wound healing properties and antibiofilm activity of hydrogel matrix containing nitric oxide, silver nanoparticles, and ciprofloxacin against Pseudomonas aeruginosa burn wound infection, Sci. Rep., 2025, 15(1), 26508 CrossRef CAS PubMed.
- I. Rahaman, N. Srivastava, D. Thakur, A. Jamwal and L. Kumar, Enhanced Antibiofilm Activity of Copper Nanoparticles Stabilized With Polyvinyl Pyrrolidone and Polyvinyl Alcohol Against Pseudomonas aeruginosa and Staphylococcus aureus, J. Biomed. Mater. Res., Part B, 2025, 113(8), e35633 CrossRef CAS PubMed.
- K. Razdan, V. S. Gondil, S. Chhibber, K. K. Singh and V. R. Sinha, Levofloxacin loaded clove essential oil nanoscale emulsion as an efficient system against Pseudomonas aeruginosa biofilm, J. Drug Delivery Sci. Technol., 2022, 68, 103039 CrossRef CAS.
- M. E. El-Naggar, A. M. Abdelgawad, R. Abdel-Sattar, A. A. Gibriel and B. A. Hemdan, Potential antimicrobial and antibiofilm efficacy of essential oil nanoemulsion loaded polycaprolactone nanofibrous dermal patches, Eur. Polym. J., 2023, 184, 111782 CrossRef CAS.
- T. Du, Z. Xiao, G. Zhang, L. Wei, J. Cao, Z. Zhang, X. Li, Z. Song, W. Wang, J. Liu, X. Du and S. Wang, An injectable multifunctional hydrogel for eradication of bacterial biofilms and wound healing, Acta Biomater., 2023, 161, 112–133 CrossRef CAS PubMed.
- L. Yang, F. Teles, W. Gong, S. A. Dua, L. Martin and M. H. Schoenfisch, Antibacterial action of nitric oxide-releasing hyperbranched polymers against ex vivo dental biofilms, Dent. Mater., 2020, 36(5), 635–644 CrossRef CAS PubMed.
- L. Weng, L. Wu, R. Guo, J. Ye, W. Liang, W. Wu, L. Chen and D. Yang, Lactobacillus cell envelope-coated nanoparticles for antibiotic delivery against cariogenic biofilm and dental caries, J. Nanobiotechnol., 2022, 20(1), 356 CrossRef CAS PubMed.
- X. Pang, X. Liu, Y. Cheng, C. Zhang, E. Ren, C. Liu, Y. Zhang, J. Zhu, X. Chen and G. Liu, Sono-Immunotherapeutic Nanocapturer to Combat Multidrug-Resistant Bacterial Infections, Adv. Mater., 2019, 31(35), 1902530 CrossRef PubMed.
- Z. Zhou, F. Hu, S. Hu, M. Kong, C. Feng, Y. Liu, X. Cheng, Q. Ji and X. Chen, pH-Activated nanoparticles with targeting for the treatment of oral plaque biofilm, J. Mater. Chem. B, 2018, 6(4), 586–592 RSC.
- M. Kluzek, Y. Oppenheimer-Shaanan, T. Dadosh, M. I. Morandi, O. Avinoam, C. Raanan, M. Goldsmith, R. Goldberg and J. Klein, Designer Liposomic Nanocarriers Are Effective Biofilm Eradicators, ACS Nano, 2022, 16(10), 15792–15804 CrossRef CAS PubMed.
- J. Cao, Y. Zhao, Y. Liu, S. Tian, C. Zheng, C. Liu, Y. Zhai, Y. An, H. J. Busscher, L. Shi and Y. Liu, Phosphorylcholine-Based Polymer Encapsulated Chitosan Nanoparticles Enhance the Penetration of Antimicrobials in a Staphylococcal Biofilm, ACS Macro Lett., 2019, 8(6), 651–657 CrossRef CAS PubMed.
- P. Wang, Y. Zeng, J. Liu, L. Wang, M. Yang and J. Zhou, Antimicrobial and anti-biofilm effects of dihydroartemisinin-loaded chitosan nanoparticles against methicillin-resistant Staphylococcus aureus, Microb. Pathog., 2025, 199, 107208 CrossRef CAS PubMed.
- Y. Tan, S. Ma, M. Leonhard, D. Moser, G. M. Haselmann, J. Wang, D. Eder and B. Schneider-Stickler, Enhancing antibiofilm activity with functional chitosan nanoparticles targeting biofilm cells and biofilm matrix, Carbohydr. Polym., 2018, 200, 35–42 CrossRef CAS PubMed.
- Z. Huang, T. Zhou, Y. Yuan, S. Natalie Kłodzińska, T. Zheng, C. Sternberg, H. Mørck Nielsen, Y. Sun and F. Wan, Synthesis of carbon quantum dot-poly lactic-co-glycolic acid hybrid nanoparticles for chemo-photothermal therapy against bacterial biofilms, J. Colloid Interface Sci., 2020, 577, 66–74 CrossRef CAS PubMed.
- J. Li, G. Pan, G. V. Zyryanov, Y. Peng, G. Zhang, L. Ma, S. Li, P. Chen and Z. Wang, Positively Charged Semiconductor Conjugated Polymer Nanomaterials with Photothermal Activity for Antibacterial and Antibiofilm Activities In Vitro and In Vivo, ACS Appl. Mater. Interfaces, 2023, 15(34), 40864–40876 CrossRef CAS PubMed.
- A. Anjum, P.-Y. Chung and S.-F. Ng, PLGA/xylitol nanoparticles enhance antibiofilm activity via penetration into biofilm extracellular polymeric substances, RSC Adv., 2019, 9(25), 14198–14208 RSC.
- H. Qiu, Z. Si, Y. Luo, P. Feng, X. Wu, W. Hou, Y. Zhu, M. B. Chan-Park, L. Xu and D. Huang, The Mechanisms and the Applications of Antibacterial Polymers in Surface Modification on Medical Devices, Front. Bioeng. Biotechnol., 2020, 8, 910 CrossRef PubMed.
- Y. Yang, Z. Cai, Z. Huang, X. Tang and X. Zhang, Antimicrobial cationic polymers: from structural design to functional control, Polym. J., 2018, 50(1), 33–44 CrossRef CAS.
- M. Abdollahi, A. Baharian, M. Mohamadhoseini, M. Hassanpour, P. Makvandi, M. Habibizadeh, B. Jafari, R. Nouri, Z. Mohamadnia and N. Nikfarjam, Advances in ionic liquid-based antimicrobial wound healing platforms, J. Mater. Chem. B, 2024, 12(38), 9478–9507 RSC.
- N. Nikfarjam, M. Ghomi, T. Agarwal, M. Hassanpour, E. Sharifi, D. Khorsandi, M. Ali Khan, F. Rossi, A. Rossetti, E. Nazarzadeh Zare, N. Rabiee, D. Afshar, M. Vosough, T. Kumar Maiti, V. Mattoli, E. Lichtfouse, F. R. Tay and P. Makvandi, Antimicrobial Ionic Liquid-Based Materials for Biomedical Applications, Adv. Funct. Mater., 2021, 31(42), 2104148 CrossRef CAS.
- Y. Li, X. Ma, J. Zhang, X. Pan, N. Li, G. Chen and J. Zhu, Degradable Selenium-Containing Polymers for Low Cytotoxic Antibacterial Materials, ACS Macro Lett., 2022, 11(12), 1349–1354 CrossRef CAS PubMed.
- B. Dizman, M. O. Elasri and L. J. Mathias, Synthesis and antimicrobial activities of new water-soluble bis-quaternary ammonium methacrylate polymers, J. Appl. Polym. Sci., 2004, 94(2), 635–642 CrossRef CAS.
- P. Salas-Ambrosio, S. Vexler, R. P S, I. A. Chen and H. D. Maynard, Caffeine and Cationic Copolymers with Antimicrobial Properties, ACS Bio Med Chem Au, 2023, 3(2), 189–200 CrossRef CAS PubMed.
- S. N. Hancock, N. Yuntawattana, E. Diep, A. Maity, A. Tran, J. D. Schiffman and Q. Michaudel, Ring-opening metathesis polymerization of N-methylpyridinium-fused norbornenes to access antibacterial main-chain cationic polymers, Proc. Natl. Acad. Sci. U. S. A., 2023, 120(51), e2311396120 CrossRef CAS PubMed.
- S. Bai, J. Wang, K. Yang, C. Zhou, Y. Xu, J. Song, Y. Gu, Z. Chen, M. Wang, C. Shoen, B. Andrade, M. Cynamon, K. Zhou, H. Wang, Q. Cai, E. Oldfield, S. C. Zimmerman, Y. Bai and X. Feng, A polymeric approach toward resistance-resistant antimicrobial agent with dual-selective mechanisms of action, Sci. Adva., 2021, 7(5), eabc9917 CrossRef CAS PubMed.
- A. R. Egorov, A. A. Artemjev, V. A. Kozyrev, D. N. Sikaona, V. V. Rubanik, V. V. Rubanik Jr, I. S. Kritchenkov, N. Z. Yagafarov, O. M. Khubiev, T. A. Tereshina, E. K. Kultyshkina, B. Medjbour, V. N. Khrustalev and A. S. Kritchenkov, Synthesis of Selenium-Containing Chitosan Derivatives and Their Antibacterial Activity, Appl. Biochem. Microbiol., 2022, 58(2), 132–135 CrossRef CAS.
- A. Dai, W. Wang, X. Heng, W. Shu, S. Lu, Y. Xu, D. Wang, X. Pan, N. Li, G. Chen and J. Zhu, High-Efficiency Bactericidal and Biofilm Elimination Ability of the Biodegradable Alternating Sequence Main-Chain Polyselenium Salt, ACS Appl. Polym. Mater., 2024, 6(9), 4975–4984 CrossRef CAS.
- Q. Wang, L.-M. Barnes, K. I. Maslakov, C. A. Howell, M. J. Illsley, P. Dyer and I. N. Savina, In situ synthesis of silver or selenium nanoparticles on cationized cellulose fabrics for antimicrobial application, Mater. Sci. Eng. C, 2021, 121, 111859 CrossRef CAS PubMed.
- T. Huang, J. A. Holden, E. C. Reynolds, D. E. Heath, N. M. O’Brien-Simpson and A. J. O’Connor, Multifunctional Antimicrobial Polypeptide-Selenium Nanoparticles Combat Drug-Resistant Bacteria, ACS Appl. Mater. Interfaces, 2020, 12(50), 55696–55709 CrossRef CAS PubMed.
- C. Peng, A. Vishwakarma, Z. Li, T. Miyoshi, H. A. Barton and A. Joy, Modification of a conventional polyurethane composition provides significant anti-biofilm activity against Escherichia coli, Polym. Chem., 2018, 9(23), 3195–3198 RSC.
- A. Vishwakarma, F. Dang, A. Ferrell, H. A. Barton and A. Joy, Peptidomimetic Polyurethanes Inhibit Bacterial Biofilm Formation and Disrupt Surface Established Biofilms, J. Am. Chem. Soc., 2021, 143(25), 9440–9449 CrossRef CAS PubMed.
- R. Gharibi and S. Agarwal, Polyurethanes from Hydrophobic Elastic Materials to Hydrogels with Potent Nonleaching Biocidal and Antibiofilm Activity, ACS Appl. Polym. Mater., 2021, 3(9), 4695–4707 CrossRef CAS.
- R. F. Menger, J. J. Beck, T. Borch and C. S. Henry, Colorimetric Paper-Based Analytical Device for Perfluorooctanesulfonate Detection. ACS ES&T, Water, 2022, 2(4), 565–572 CAS.
- A. L. Lewis; J. J. Kohler and M. Aebi, Microbial Lectins: Hemagglutinins, Adhesins, and Toxins, in Essentials of Glycobiology [Internet], ed. A. Varki, R. D. Cummings, J. D. Esko, P. Stanley, G. W. Hart and M. Aebi, Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY, 2022 Search PubMed.
- J. J. Lundquist and E. J. Toone, The Cluster Glycoside Effect, Chem. Rev., 2002, 102(2), 555–578 CrossRef CAS PubMed.
- Y. C. Lee and R. T. Lee, Carbohydrate-Protein Interactions: Basis of Glycobiology, Acc. Chem. Res., 1995, 28(8), 321–327 CrossRef CAS.
- E. van Duijkeren, A.-K. Schink, C. Roberts Marilyn, Y. Wang and S. Schwarz, Mechanisms of Bacterial Resistance to Antimicrobial Agents, Microbiol. Spectrum, 2018, 6(2) DOI:10.1128/microbiolspec.arba-0019-2017.
- E. M. Halawa, M. Fadel, M. W. Al-Rabia, A. Behairy, N. A. Nouh, M. Abdo, R. Olga, L. Fericean, A. M. Atwa, M. El-Nablaway and A. Abdeen, Antibiotic action and resistance: updated review of mechanisms, spread, influencing factors, and alternative approaches for combating resistance, Front. Pharmacol., 2023, 14, 1305294 CrossRef CAS PubMed.
- X. He, X. Sheng, X. Yao, Y. Wang, L. Zhang, H. Wang and L. Yuan, The anti-biofilm effect of α-amylase/glycopolymer-decorated gold nanorods, Colloids Surf., B, 2025, 246, 114393 CrossRef CAS PubMed.
- X. Chen, B. Wu, H. A. Perera and M. Yan, Synthesis of Glycopolymer Micelles for Antibiotic Delivery, Molecules, 2023, 28, 10 Search PubMed.
- C. A. Werlang, J. K. Sahoo, G. Cárcarmo-Oyarce, C. Stevens, D. Uzun, R. Putnik, O. Hasturk, J. Choi, D. L. Kaplan and K. Ribbeck, Selective Biofilm Inhibition through Mucin-Inspired Engineering of Silk Glycopolymers, J. Am. Chem. Soc., 2024, 146(50), 34661–34668 CrossRef CAS PubMed.
- L. L. Kiessling; T. Young; T. D. Gruber and K. H. Mortell, Multivalency in Protein–Carbohydrate Recognition, Springer Berlin Heidelberg, 2008, pp. 2483–2523 Search PubMed.
- H. M. Branderhorst, R. Ruijtenbeek, R. M. J. Liskamp and R. J. Pieters, Multivalent Carbohydrate Recognition on a Glycodendrimer-Functionalized Flow-Through Chip, ChemBioChem, 2008, 9(11), 1836–1844 CrossRef CAS PubMed.
- M. R. Thalji, A. A. Ibrahim, K. F. Chong, A. V. Soldatov and G. A. M. Ali, Glycopolymer-Based Materials: Synthesis, Properties, and Biosensing Applications, Top Curr. Chem., 2022, 380(5), 45 CrossRef CAS PubMed.
- R. Mei, X. Heng, X. Liu and G. Chen, Glycopolymers for Antibacterial and Antiviral Applications, Molecules, 2023, 28, 3 Search PubMed.
- D. J. Wurpel, S. A. Beatson, M. Totsika, N. K. Petty and M. A. Schembri, Chaperone-Usher Fimbriae of Escherichia coli, PLoS One, 2013, 8(1), e52835 CrossRef CAS PubMed.
- R. Mei, X. Heng, X. Liu and G. Chen, Glycopolymers for Antibacterial and Antiviral Applications, Molecules, 2023, 985 CrossRef CAS PubMed.
- Y. Miura, Y. Hoshino and H. Seto, Glycopolymer Nanobiotechnology, Chem. Rev., 2016, 116(4), 1673–1692 CrossRef CAS PubMed.
- U. I. M. Gerling-Driessen, M. Hoffmann, S. Schmidt, N. L. Snyder and L. Hartmann, Glycopolymers against pathogen infection, Chem. Soc. Rev., 2023, 52(8), 2617–2642 RSC.
- C. A. Werlang, W. G. Chen, K. Aoki, K. M. Wheeler, C. Tymm, C. J. Mileti, A. C. Burgos, K. Kim, M. Tiemeyer and K. Ribbeck, Mucin O-glycans suppress quorum-sensing pathways and genetic transformation in Streptococcus mutans, Nat. Microbiol., 2021, 6(5), 574–583 CrossRef CAS PubMed.
- B. X. Wang, K. M. Wheeler, K. C. Cady, S. Lehoux, R. D. Cummings, M. T. Laub and K. Ribbeck, Mucin Glycans Signal through the Sensor Kinase RetS to Inhibit Virulence-Associated Traits in Pseudomonas aeruginosa, Curr. Biol., 2021, 31(1), 90–102.e7 CrossRef CAS PubMed.
- L. Kavanaugh Nicole, Q. Zhang Angela, J. Nobile Clarissa, D. Johnson Alexander and K. Ribbeck, Mucins Suppress Virulence Traits of Candida albicans, mBio, 2014, 5(6), e01911-14, DOI:10.1128/mbio.01911-14.
- M. Caldara, R. S. Friedlander, N. L. Kavanaugh, J. Aizenberg, K. R. Foster and K. Ribbeck, Mucin biopolymers prevent bacterial aggregation by retaining cells in the free-swimming state, Curr. Biol., 2012, 22(24), 2325–2330 CrossRef CAS PubMed.
- E. S. Frenkel and K. Ribbeck, Salivary mucins protect surfaces from colonization by cariogenic bacteria, Appl. Environ. Microbiol., 2015, 81(1), 332–338 CrossRef PubMed.
- C. A. Werlang, W. G. Chen, K. Aoki, K. M. Wheeler, C. Tymm, C. J. Mileti, A. C. Burgos, K. Kim, M. Tiemeyer and K. Ribbeck, Mucin O-glycans suppress quorum-sensing pathways and genetic transformation in Streptococcus mutans, Nat. Microbiol., 2021, 6(5), 574–583 CrossRef CAS PubMed.
- C. Ligeour, O. Vidal, L. Dupin, F. Casoni, E. Gillon, A. Meyer, S. Vidal, G. Vergoten, J.-M. Lacroix, E. Souteyrand, A. Imberty, J.-J. Vasseur, Y. Chevolot and F. Morvan, Mannose-centered aromatic galactoclusters inhibit the biofilm formation of Pseudomonas aeruginosa, Org. Biomol. Chem., 2015, 13(31), 8433–8444 RSC.
- E. M. V. Johansson, S. A. Crusz, E. Kolomiets, L. Buts, R. U. Kadam, M. Cacciarini, K.-M. Bartels, S. P. Diggle, M. Cámara, P. Williams, R. Loris, C. Nativi, F. Rosenau, K.-E. Jaeger, T. Darbre and J.-L. Reymond, Inhibition and Dispersion of Pseudomonas aeruginosa Biofilms by Glycopeptide Dendrimers Targeting the Fucose-Specific Lectin LecB, Chem. Biol., 2008, 15(12), 1249–1257 CrossRef CAS PubMed.
- R. U. Kadam, M. Bergmann, M. Hurley, D. Garg, M. Cacciarini, M. A. Swiderska, C. Nativi, M. Sattler, A. R. Smyth, P. Williams, M. Cámara, A. Stocker, T. Darbre and J. L. Reymond, A glycopeptide dendrimer inhibitor of the galactose-specific lectin LecA and of Pseudomonas aeruginosa biofilms, Angew. Chem., Int. Ed., 2011, 50(45), 10631–10635 CrossRef CAS PubMed.
- E. Kolomiets, E. M. V. Johansson, O. Renaudet, T. Darbre and J.-L. Reymond, Neoglycopeptide Dendrimer Libraries as a Source of Lectin Binding Ligands, Org. Lett., 2007, 9(8), 1465–1468 CrossRef CAS PubMed.
- E. L. Dane, A. E. Ballok, G. A. O'Toole and M. W. Grinstaff, Synthesis of bioinspired carbohydrate amphiphiles that promote and inhibit biofilms, Chem. Sci., 2014, 5(2), 551–557 RSC.
- K. S. Bücher, N. Babic, T. Freichel, F. Kovacic and L. Hartmann, Monodisperse Sequence-Controlled α-l-Fucosylated Glycooligomers and Their Multivalent Inhibitory Effects on LecB, Macromol. Biosci., 2018, 18(12), 1800337 CrossRef PubMed.
- M. Liu, J. Li and B. Li, Mannose-Modificated Polyethylenimine: A Specific and Effective Antibacterial Agent against Escherichia coli, Langmuir, 2018, 34(4), 1574–1580 CrossRef CAS PubMed.
- M. Álvarez-Paino, A. Muñoz-Bonilla, F. López-Fabal, J. L. Gómez-Garcés, J. P. A. Heuts and M. Fernández-García, Effect of glycounits on the antimicrobial properties and toxicity behavior of polymers based on quaternized DMAEMA, Biomacromolecules, 2015, 16(1), 295–303 CrossRef PubMed.
- P. Sahariah, F. Papi, K. L. Merz, O. E. Sigurjonsson, R. L. Meyer and C. Nativi, Chitosan–saccharide conjugates for eradication of Pseudomonas aeruginosa biofilms, RSC Appl. Polym., 2024, 2(3), 461–472 RSC.
- K. Zhang, Y. Du, Z. Si, Y. Liu, M. E. Turvey, C. Raju, D. Keogh, L. Ruan, S. L. Jothy, S. Reghu, K. Marimuthu, P. P. De, O. T. Ng, J. R. Mediavilla, B. N. Kreiswirth, Y. R. Chi, J. Ren, K. C. Tam, X.-W. Liu, H. Duan, Y. Zhu, Y. Mu, P. T. Hammond, G. C. Bazan, K. Pethe and M. B. Chan-Park, Enantiomeric glycosylated cationic block co-beta-peptides eradicate Staphylococcus aureus biofilms and antibiotic-tolerant persisters, Nat. Commun., 2019, 10(1), 4792 CrossRef PubMed.
- K. Zhang, C. Raju, W. Zhong, K. Pethe, A. Gründling and M. B. Chan-Park, Cationic Glycosylated Block Co-β-peptide Acts on the Cell Wall of Gram-positive Bacteria as Anti-biofilm Agents, ACS Appl. Bio Mater., 2021, 4(5), 3749–3761 CrossRef CAS PubMed.
- X. Dai, X. Chen, Y. Zhao, Y. Yu, X. Wei, X. Zhang, C. Li and A. Water-Soluble Galactose-Decorated, Cationic Photodynamic Therapy Agent Based on BODIPY to Selectively Eliminate Biofilm, Biomacromolecules, 2018, 19(1), 141–149 CrossRef CAS PubMed.
- R. Toy, P. Pradhan, V. Ramesh, N. C. Di Paolo, B. Lash, J. Liu, E. L. Blanchard, C. J. Pinelli, P. J. Santangelo, D. M. Shayakhmetov and K. Roy, Modification of primary amines to higher order amines reduces in vivo hematological and immunotoxicity of cationic nanocarriers through TLR4 and complement pathways, Biomaterials, 2019, 225, 119512 CrossRef CAS PubMed.
- A. M. Weiss, M. A. Lopez, I. I. Rawe, B. W. Manna, S. Chen, Q. Mulder, E. J. Rowan and S. J. Esser-Kahn, A. P., Understanding How Cationic Polymers’ Properties Inform Toxic or Immunogenic Responses via Parametric Analysis, Macromolecules, 2023, 56(18), 7286–7299 CrossRef CAS PubMed.
- D. Kamath and S. H. Bossmann, Advances in glyconanotechnology based biomedical applications, J. Cancer Biol., 2025, 6(1), 4–9 Search PubMed.
- X. Wei, Y. Gao, Y. Hu, Y. Zhang and X. Zhang, A light-activated nanotherapeutic with broad-spectrum bacterial recognition to eliminate drug-resistant pathogens, J. Mater. Chem. B, 2021, 9(5), 1364–1369 RSC.
- A. El Riz, A. Tchoumi Neree, L. Mousavifar, R. Roy, Y. Chorfi and M. A. Mateescu, Metallo-Glycodendrimeric Materials against Enterotoxigenic Escherichia coli, Microorganisms, 2024, 966 CrossRef CAS PubMed.
- X. He, X. Sheng, X. Yao, Y. Wang, L. Zhang, H. Wang and L. Yuan, The anti-biofilm effect of α-amylase/glycopolymer-decorated gold nanorods, Colloids Surf., B, 2025, 246, 114393 CrossRef CAS PubMed.
- Q. Guo, Y. Zhao, X. Dai, T. Zhang, Y. Yu, X. Zhang and C. Li, Functional Silver Nanocomposites as Broad-Spectrum Antimicrobial and Biofilm-Disrupting Agents, ACS Appl. Mater. Interfaces, 2017, 9(20), 16834–16847 CrossRef CAS PubMed.
- Y. Zhao, Q. Guo, X. Dai, X. Wei, Y. Yu, X. Chen, C. Li, Z. Cao, X. Zhang and A. Biomimetic Non-Antibiotic, Approach to Eradicate Drug-Resistant Infections, Adv. Mater., 2019, 31(7), 1806024 CrossRef PubMed.
- H. Wei, C. Yang, F. Bi, B. Li, R. Xie, D. Yu, S. Fang, Z. Hua, Q. Wang and G. Yang, Structure-Controllable and Mass-Produced Glycopolymersomes as a Template of the Carbohydrate@Ag Nanobiohybrid with Inherent Antibacteria and Biofilm Eradication, Biomacromolecules, 2024, 25(1), 315–327 CrossRef CAS PubMed.
- D. Boffoli, F. Bellato, G. Avancini, P. Gurnani, G. Yilmaz, M. Romero, S. Robertson, F. Moret, F. Sandrelli, P. Caliceti, S. Salmaso, M. Cámara, G. Mantovani and F. Mastrotto, Tobramycin-loaded complexes to prevent and disrupt Pseudomonas aeruginosa biofilms, Drug Delivery Transl. Res., 2022, 12(8), 1788–1810 CrossRef CAS PubMed.
- Z. Ma, J. Li, Y. Bai, Y. Zhang, H. Sun and X. Zhang, A bacterial infection-microenvironment activated nanoplatform based on spiropyran-conjugated glycoclusters for imaging and eliminating of the biofilm, Chem. Eng. J., 2020, 399, 125787 CrossRef CAS.
- W. Wang, Y. Hu, Z. Chen, L. Yu, S. Huang, Y. Zhang, J. Li, Y. Xue, A. Li, Y. Wang, Z. Wu and X. Zhang, Guanidine and Galactose Decorated Nanophotosensitizer with Oxygen Self-Sufficient Capability for the Localized Ablation of Oral Biofilm, Adv. Funct. Mater., 2023, 33(37), 2300474 CrossRef CAS.
- Y. Zhao, Z. Lu, X. Dai, X. Wei, Y. Yu, X. Chen, X. Zhang and C. Li, Glycomimetic-Conjugated Photosensitizer for Specific Pseudomonas aeruginosa Recognition and Targeted Photodynamic Therapy, Bioconjugate Chem., 2018, 29(9), 3222–3230 CrossRef CAS PubMed.
- J. Tricomi, M. Cacaci, G. Biagiotti, L. Caselli, L. Niccoli, R. Torelli, A. Gabbani, M. Di Vito, F. Pineider, M. Severi, M. Sanguinetti, E. Menna, M. Lelli, D. Berti, S. Cicchi, F. Bugli and B. Richichi, Ball milled glyco-graphene oxide conjugates markedly disrupted Pseudomonas aeruginosa biofilms, Nanoscale, 2022, 14(28), 10190–10199 RSC.
- S. Medici, M. Peana, A. Pelucelli and M. A. Zoroddu, An updated overview on metal nanoparticles toxicity, Semin. Cancer Biol., 2021, 76, 17–26 CrossRef CAS PubMed.
- Z. Ferdous and A. Nemmar, Health Impact of Silver Nanoparticles: A Review of the Biodistribution and Toxicity Following Various Routes of Exposure, Int. J. Mol. Sci., 2020, 2375 CrossRef CAS PubMed.
- R. Kumar, A. Kumar, S. Bhardwaj, M. Sikarwar, S. Sriwastaw, G. Sharma and M. Gupta, Nanotoxicity unveiled: Evaluating exposure risks and assessing the impact of nanoparticles on human health, J. Trace Element Miner., 2025, 13, 100252 CrossRef.
- P. Narayanaswamy Vidya, P. Duncan Andrew, J. LiPuma John, P. Wiesmann William, M. Baker Shenda and M. Townsend Stacy, In Vitro Activity of a Novel Glycopolymer against Biofilms of Burkholderia cepacia Complex Cystic Fibrosis Clinical Isolates, Antimicrob. Agents Chemother., 2019, 63(6), e00498-19, DOI:10.1128/aac.00498-19.
- V. P. Narayanaswamy, S. M. Townsend, A. J. Loughran, W. Wiesmann and S. Baker, Polycationic Glycopolymer Demonstrates Activity Against Persisters and Biofilms of Non-tuberculosis Mycobacteria Cystic Fibrosis Clinical Isolates in vitro, Front. Microbiol., 2022, 13, 821820 CrossRef PubMed.
- B. A. Garcia, M. S. McDaniel, A. J. Loughran, J. D. Johns, V. Narayanaswamy, C. Fernandez Petty, S. E. Birket, S. M. Baker, R. Barnaby, B. A. Stanton, J. B. Foote, S. M. Rowe and W. E. Swords, Poly(acetyl, arginyl) glucosamine disrupts Pseudomonas aeruginosa biofilms and enhances bacterial clearance in a rat lung infection model, Microbiology, 2022, 168, 1 CrossRef PubMed.
- V. P. Narayanaswamy, L. L. Keagy, K. Duris, W. Wiesmann, A. J. Loughran, S. M. Townsend and S. Baker, Novel Glycopolymer Eradicates Antibiotic- and CCCP-Induced Persister Cells in Pseudomonas aeruginosa, Front. Microbiol., 2018, 9, 1724 CrossRef PubMed.
- D. Boffoli, F. Bellato, G. Avancini, P. Gurnani, G. Yilmaz, M. Romero, S. Robertson, F. Moret, F. Sandrelli, P. Caliceti, S. Salmaso, M. Cámara, G. Mantovani and F. Mastrotto, Tobramycin-loaded complexes to prevent and disrupt Pseudomonas aeruginosa biofilms, Drug Delivery Transl. Res., 2022, 12(8), 1788–1810 CrossRef CAS PubMed.
- W. Wang, Y. Hu, Z. Chen, L. Yu, S. Huang, Y. Zhang, J. Li, Y. Xue, A. Li, Y. Wang, Z. Wu and X. Zhang, Guanidine and Galactose Decorated Nanophotosensitizer with Oxygen Self-Sufficient Capability for the Localized Ablation of Oral Biofilm, Adv. Funct. Mater., 2023, 33 Search PubMed.
- Y. Zhao, Z. Lu, X. Dai, X. Wei, Y. Yu, X. Chen, X. Zhang and C. Li, Glycomimetic-Conjugated Photosensitizer for Specific Pseudomonas aeruginosa Recognition and Targeted Photodynamic Therapy, Bioconjugate Chem., 2018, 29(9), 3222–3230 CrossRef CAS PubMed.
- K. S. Bücher, N. Babic, T. Freichel, F. Kovacic and L. Hartmann, Monodisperse Sequence-Controlled α-l-Fucosylated Glycooligomers and Their Multivalent Inhibitory Effects on LecB, Macromol. Biosci., 2018, 18(12), e1800337 CrossRef PubMed.
- K. Zhang, Y. Du, Z. Si, Y. Liu, M. E. Turvey, C. Raju, D. Keogh, L. Ruan, S. L. Jothy, S. Reghu, K. Marimuthu, P. P. De, O. T. Ng, J. R. Mediavilla, B. N. Kreiswirth, Y. R. Chi, J. Ren, K. C. Tam, X. W. Liu, H. Duan, Y. Zhu, Y. Mu, P. T. Hammond, G. C. Bazan, K. Pethe and M. B. Chan-Park, Enantiomeric glycosylated cationic block co-beta-peptides eradicate Staphylococcus aureus biofilms and antibiotic-tolerant persisters, Nat. Commun., 2019, 10(1), 4792 CrossRef PubMed.
- K. Zhang, C. Raju, W. Zhong, K. Pethe, A. Gründling and M. B. Chan-Park, Cationic Glycosylated Block Co-β-peptide Acts on the Cell Wall of Gram-positive Bacteria as Anti-biofilm Agents, ACS Appl. Bio Mater., 2021, 4(5), 3749–3761 CrossRef CAS PubMed.
- M. Naghavi, S. E. Vollset, K. S. Ikuta, L. R. Swetschinski, A. P. Gray, E. E. Wool, G. Robles Aguilar, T. Mestrovic, G. Smith, C. Han, R. L. Hsu, J. Chalek, D. T. Araki, E. Chung, C. Raggi, A. Gershberg Hayoon, N. Davis Weaver, P. A. Lindstedt, A. E. Smith, U. Altay, N. V. Bhattacharjee, K. Giannakis, F. Fell, B. McManigal, N. Ekapirat, J. A. Mendes, T. Runghien, O. Srimokla, A. Abdelkader, S. Abd-Elsalam, R. G. Aboagye, H. Abolhassani, H. Abualruz, U. Abubakar, H. J. Abukhadijah, S. Aburuz, A. Abu-Zaid, S. Achalapong, I. Y. Addo, V. Adekanmbi, T. E. Adeyeoluwa, Q. E. S. Adnani, L. A. Adzigbli, M. S. Afzal, S. Afzal, A. Agodi, A. J. Ahlstrom, A. Ahmad, S. Ahmad, T. Ahmad, A. Ahmadi, A. Ahmed, H. Ahmed, I. Ahmed, M. Ahmed, S. Ahmed, S. A. Ahmed, M. A. Akkaif, S. Al Awaidy, Y. Al Thaher, S. O. Alalalmeh, M. T. AlBataineh, W. A. Aldhaleei, A. A. S. Al-Gheethi, N. B. Alhaji, A. Ali, L. Ali, S. S. Ali, W. Ali, K. Allel, S. Al-Marwani, A. Alrawashdeh, A. Altaf, A. B. Al-Tammemi, J. A. Al-Tawfiq, K. H. Alzoubi, W. A. Al-Zyoud, B. Amos, J. H. Amuasi, R. Ancuceanu, J. R. Andrews, A. Anil, I. A. Anuoluwa, S. Anvari, A. E. Anyasodor, G. L. C. Apostol, J. Arabloo, M. Arafat, A. Y. Aravkin, D. Areda, A. Aremu, A. A. Artamonov, E. A. Ashley, M. O. Asika, S. S. Athari, M. M. W. Atout, T. Awoke, S. Azadnajafabad, J. M. Azam, S. Aziz, A. Y. Azzam, M. Babaei, F.-X. Babin, M. Badar, A. A. Baig, M. Bajcetic, S. Baker, M. Bardhan, H. J. Barqawi, Z. Basharat, A. Basiru, M. Bastard, S. Basu, N. S. Bayleyegn, M. A. Belete, O. O. Bello, A. Beloukas, J. A. Berkley, A. S. Bhagavathula, S. Bhaskar, S. S. Bhuyan, J. A. Bielicki, N. I. Briko, C. S. Brown, A. J. Browne, D. Buonsenso, Y. Bustanji, C. G. Carvalheiro, C. A. Castañeda-Orjuela, M. Cenderadewi, J. Chadwick, S. Chakraborty, R. M. Chandika, S. Chandy, V. Chansamouth, V. K. Chattu, A. A. Chaudhary, P. R. Ching, H. Chopra, F. R. Chowdhury, D.-T. Chu, M. Chutiyami, N. Cruz-Martins, A. G. da Silva, O. Dadras, X. Dai, S. D. Darcho, S. Das, F. P. De la Hoz, D. M. Dekker, K. Dhama, D. Diaz, B. F. R. Dickson, S. G. Djorie, M. Dodangeh, S. Dohare, K. G. Dokova, O. P. Doshi, R. K. Dowou, H. L. Dsouza, S. J. Dunachie, A. M. Dziedzic, T. Eckmanns, A. Ed-Dra, A. Eftekharimehrabad, T. C. Ekundayo, I. El Sayed, M. Elhadi, W. El-Huneidi, C. Elias, S. J. Ellis, R. Elsheikh, I. Elsohaby, C. Eltaha, B. Eshrati, M. Eslami, D. W. Eyre, A. O. Fadaka, A. F. Fagbamigbe, A. Fahim, A. Fakhri-Demeshghieh, F. O. Fasina, M. M. Fasina, A. Fatehizadeh, N. A. Feasey, A. Feizkhah, G. Fekadu, F. Fischer, I. Fitriana, K. M. Forrest, C. Fortuna Rodrigues, J. E. Fuller, M. A. Gadanya, M. Gajdács, A. P. Gandhi, E. E. Garcia-Gallo, D. O. Garrett, R. K. Gautam, M. W. Gebregergis, M. Gebrehiwot, T. G. Gebremeskel, C. Geffers, L. Georgalis, R. M. Ghazy, M. Golechha, D. Golinelli, M. Gordon, S. Gulati, R. D. Gupta, S. Gupta, V. K. Gupta, A. D. Habteyohannes, S. Haller, H. Harapan, M. L. Harrison, A. I. Hasaballah, I. Hasan, R. S. Hasan, H. Hasani, A. H. Haselbeck, M. S. Hasnain, I. I. Hassan, S. Hassan, M. S. Hassan Zadeh Tabatabaei, K. Hayat, J. He, O. E. Hegazi, M. Heidari, K. Hezam, R. Holla, M. Holm, H. Hopkins, M. M. Hossain, M. Hosseinzadeh, S. Hostiuc, N. R. Hussein, L. D. Huy, E. D. Ibáñez-Prada, A. Ikiroma, I. M. Ilic, S. M. S. Islam, F. Ismail, N. E. Ismail, C. D. Iwu, C. J. Iwu-Jaja, A. Jafarzadeh, F. Jaiteh, R. Jalilzadeh Yengejeh, R. D. G. Jamora, J. Javidnia, T. Jawaid, A. W. J. Jenney, H. J. Jeon, M. Jokar, N. Jomehzadeh, T. Joo, N. Joseph, Z. Kamal, K. K. Kanmodi, R. S. Kantar, J. A. Kapisi, I. M. Karaye, Y. S. Khader, H. Khajuria, N. Khalid, F. Khamesipour, A. Khan, M. J. Khan, M. T. Khan, V. Khanal, F. F. Khidri, J. Khubchandani, S. Khusuwan, M. S. Kim, A. Kisa, V. A. Korshunov, F. Krapp, R. Krumkamp, M. Kuddus, M. Kulimbet, D. Kumar, E. A. P. Kumaran, A. Kuttikkattu, H. H. Kyu, I. Landires, B. K. Lawal, T. T. T. Le, I. M. Lederer, M. Lee, S. W. Lee, A. Lepape, T. L. Lerango, V. S. Ligade, C. Lim, S. S. Lim, L. W. Limenh, C. Liu, X. Liu, X. Liu, M. J. Loftus, M. H. I. Amin, K. L. Maass, S. B. Maharaj, M. A. Mahmoud, P. Maikanti-Charalampous, O. M. Makram, K. Malhotra, A. A. Malik, G. D. Mandilara, F. Marks, B. A. Martinez-Guerra, M. Martorell, H. Masoumi-Asl, A. G. Mathioudakis, J. May, T. A. McHugh, J. Meiring, H. N. Meles, A. Melese, E. B. Melese, G. Minervini, N. S. Mohamed, S. Mohammed, S. Mohan, A. H. Mokdad, L. Monasta, A. Moodi Ghalibaf, C. E. Moore, Y. Moradi, E. Mossialos, V. Mougin, G. D. Mukoro, F. Mulita, B. Muller-Pebody, E. Murillo-Zamora, S. Musa, P. Musicha, L. A. Musila, S. Muthupandian, A. J. Nagarajan, P. Naghavi, F. Nainu, T. S. Nair, H. H. R. Najmuldeen, Z. S. Natto, J. Nauman, B. P. Nayak, G. T. Nchanji, P. Ndishimye, I. Negoi, R. I. Negoi, S. A. Nejadghaderi, Q. P. Nguyen, E. A. Noman, D. C. Nwakanma, S. O’Brien, T. J. Ochoa, I. A. Odetokun, O. A. Ogundijo, T. R. Ojo-Akosile, S. R. Okeke, O. C. Okonji, A. T. Olagunju, A. Olivas-Martinez, A. A. Olorukooba, P. Olwoch, K. I. Onyedibe, E. Ortiz-Brizuela, O. Osuolale, P. Ounchanum, O. T. Oyeyemi, M. P. A. Padukudru, R. R. Parikh, J. Patel, S. Patil, S. Pawar, A. Y. Peleg, P. Peprah, J. Perdigão, C. Perrone, I.-R. Petcu, K. Phommasone, Z. Z. Piracha, D. Poddighe, A. J. Pollard, R. Poluru, A. Ponce-De-Leon, J. Puvvula, F. N. Qamar, N. H. Qasim, C. D. Rafai, P. Raghav, L. Rahbarnia, F. Rahim, V. Rahimi-Movaghar, M. Rahman, M. A. Rahman, H. Ramadan, S. K. Ramasamy, P. S. Ramesh, P. W. Ramteke, R. K. Rana, U. Rani, M.-M. Rashidi, D. Rathish, S. Rattanavong, S. Rawaf, E. M. M. Redwan, L. F. Reyes, T. Roberts, J. V. Robotham, V. D. Rosenthal, A. G. Ross, N. Roy, K. E. Rudd, C. J. Sabet, B. A. Saddik, M. R. Saeb, U. Saeed, S. Saeedi Moghaddam, W. Saengchan, M. Safaei, A. Saghazadeh, N. Saheb Sharif-Askari, A. Sahebkar, S. S. Sahoo, M. Sahu, M. Saki, N. Salam, Z. Saleem, M. A. Saleh, Y. L. Samodra, A. M. Samy, A. Saravanan, M. Satpathy, A. E. Schumacher, M. Sedighi, S. Seekaew, M. Shafie, P. A. Shah, S. Shahid, M. J. Shahwan, S. Shakoor, N. Shalev, M. A. Shamim, M. A. Shamshirgaran, A. Shamsi, A. Sharifan, R. P. Shastry, M. Shetty, A. Shittu, S. Shrestha, E. E. Siddig, T. Sideroglou, J. Sifuentes-Osornio, L. M. L. R. Silva, E. A. F. Simões, A. J. H. Simpson, A. Singh, S. Singh, R. Sinto, S. S. M. Soliman, S. Soraneh, N. Stoesser, T. Z. Stoeva, C. K. Swain, L. Szarpak, S. Tabatabai, S. S. T Y, Z. M.-A. Taha, K.-K. Tan, N. Tasak, N. Y. Tat, A. Thaiprakong, P. Thangaraju, C. C. Tigoi, K. Tiwari, M. R. Tovani-Palone, T. H. Tran, M. Tumurkhuu, P. Turner, A. J. Udoakang, A. Udoh, N. Ullah, S. Ullah, A. G. Vaithinathan, M. Valenti, T. Vos, H. T. L. Vu, Y. Waheed, A. S. Walker, J. L. Walson, T. Wangrangsimakul, K. G. Weerakoon, H. F. L. Wertheim, P. C. M. Williams, A. A. Wolde, T. M. Wozniak, F. Wu, Z. Wu, M. K. K. Yadav, S. Yaghoubi, Z. S. Yahaya, A. Yarahmadi, S. Yezli, Y. E. Yismaw, D. K. Yon, C.-W. Yuan, H. Yusuf, F. Zakham, G. Zamagni, H. Zhang, Z.-J. Zhang, M. Zielińska, A. Zumla, S. e H. H. Zyoud, S. H. Zyoud, S. I. Hay, A. Stergachis, B. Sartorius, B. S. Cooper, C. Dolecek and C. J. L. Murray, Global burden of bacterial antimicrobial resistance 1990–2021: a systematic analysis with forecasts to 2050, Lancet, 2024, 404(10459), 1199–1226 CrossRef PubMed.
- C. R. MacNair, S. T. Rutherford and M.-W. Tan, Alternative therapeutic strategies to treat antibiotic-resistant pathogens, Nat. Rev. Microbiol., 2024, 22(5), 262–275 CrossRef CAS PubMed.
- B. Plackett, Why big pharma has abandoned antibiotics, Nature, 2020, 586(7830), S50–S52 CrossRef CAS.
- G. A. Durand, D. Raoult and G. Dubourg, Antibiotic discovery: history, methods and perspectives, Int. J. Antimicrob. Agents, 2019, 53(4), 371–382 CrossRef CAS PubMed.
- R. I. Aminov, A brief history of the antibiotic era: lessons learned and challenges for the future, Front. Microbiol., 2010, 1, 134 Search PubMed.
- Centers for Diseases Control and Prevention, Antibiotic Resistance Threats in the United States, 2019, U.S. Department of Health and Human Services, Atlanta, GA, 2019 Search PubMed.
- P. Feuerstadt, N. Theriault and G. Tillotson, The burden of CDI in the United States: a multifactorial challenge, BMC Infect. Dis., 2023, 23(1), 132 CrossRef PubMed.
- N. Shehab, P. R. Patel, A. Srinivasan and D. S. Budnitz, Emergency department visits for antibiotic-associated adverse events, Clin. Infect. Dis., 2008, 47(6), 735–743 CrossRef PubMed.
- J. Curran, J. Lo, V. Leung, K. Brown, K. L. Schwartz, N. Daneman, G. Garber, J. H. C. Wu and B. J. Langford, Estimating daily antibiotic harms: an umbrella review with individual study meta-analysis, Clin. Microbiol. Infect., 2022, 28(4), 479–490 CrossRef PubMed.
- J. S. VanEpps and J. G. Younger, Implantable Device-Related Infection, Shock, 2016, 46(6), 597–608 CrossRef PubMed.
- M. Kuder, A. Gelman and J. M. Zenilman, Prevalence of Implanted Medical Devices in Medicine Inpatients, J. Patient Saf., 2018, 14(3), 153–156 CrossRef PubMed.
- R. O. Darouiche, Treatment of infections associated with surgical implants, N. Engl. J. Med., 2004, 350(14), 1422–1429 CrossRef CAS PubMed.
- A. Ramos-Martínez, F. Domínguez, P. Muñoz, M. Marín, Á. Pedraz, M. C. Fariñas, V. Tascón, A. De Alarcón, R. Rodríguez-García, J. M. Miró, J. Goikoetxea, G. Ojeda-Burgos, F. Escrihuela-Vidal and J. Calderón-Parra, Clinical presentation, microbiology, and prognostic factors of prosthetic valve endocarditis. Lessons learned from a large prospective registry, PLoS One, 2023, 18(9), e0290998 CrossRef PubMed.
- R. Patel, Periprosthetic Joint Infection, N. Engl. J. Med., 2023, 388(3), 251–262 CrossRef PubMed.
Footnote |
| † Equal contribution. |
| This journal is © The Royal Society of Chemistry 2026 |