 Open Access Article
Open Access ArticleFluoride ion chelation via pnictogen bonding using a distibora[1.1]ferrocenophane
Arunabha
Thakur
*ab,
Brendan L.
Murphy
a,
You
Jiang
a,
Nattamai
Bhuvanesh
a and
François P.
Gabbaï
 *a
*a
aDepartment of Chemistry, Texas A&M University, College Station, TX 77843, USA. E-mail: francois@tamu.edu
bDepartment of Chemistry, Jadavpur University, Kolkata 700032, India
First published on 30th January 2026
Abstract
As part of our interest in anion binding platforms, we now report the synthesis and structure of a neutral distibora[1.1]ferrocenophane, which behaves as a bidentate Lewis acid as demonstrated by its ability to chelate a fluoride anion. This bonding mode, confirmed by single crystal X-ray diffraction, is associated with a fluoride ion affinity of 346 kJ mol−1.
As part of our efforts in the chemistry of bidentate bis-antimony Lewis acids1 for the chelation of anions via convergent pnictogen bonding,2 we have investigated a family of bifunctional systems featuring catecholatostiboranes as σ-hole donors.3–5 These systems can be accessed with high fidelity through a route involving oxidation of the corresponding distibine with an ortho-quinone, typically 3,4,5,6-tetrachloro-1,2-benzoquinone (o-chloranil).6 The success of this approach is illustrated by derivatives such as A3 and B4 (Fig. 1) which were shown to chelate the fluoride anion through the formation of an Sb-F-Sb bridge. To further elaborate this class of compounds, we wondered whether our approach would also be compatible with ferrocene as the backbone. This question was stimulated by prior reports exploring ferrocenylboranes as anion-binding platforms7 as well as more recent reports of ferrocenylstibines in which the ferrocene fragment survives antimony-centered oxidation by ortho-quinones.8 Against this backdrop and with applications in anion capture and transport as an ultimate objective,9 we have now set our eyes on ferrocene-based distibines in view of their conversion into the corresponding distiboranes.
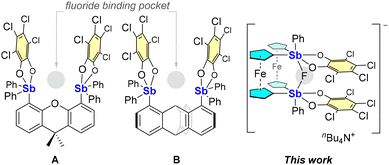 | ||
| Fig. 1 Selected examples of known catecholatostiborane-based bidentate Lewis acids (A) and (B) as fluoride anion chelators and structure of the fluoride complex obtained in this work. | ||
Aiming for rigidity, we decided to target a distiba[1.1]ferrocenophane, a class of compounds that has remained unknown even though the diphospha-10,11 and diarsa-congeners12 are precedented. In this communication, we report the synthesis and characterization of a distiba[1.1]ferrocenophane and its subsequent oxidation via treatment with o-chloranil. We further show that the resulting distibora[1.1]ferrocenophane is capable of fluoride anion chelation, which is evident by both NMR spectroscopy and cyclic voltammetry (CV).
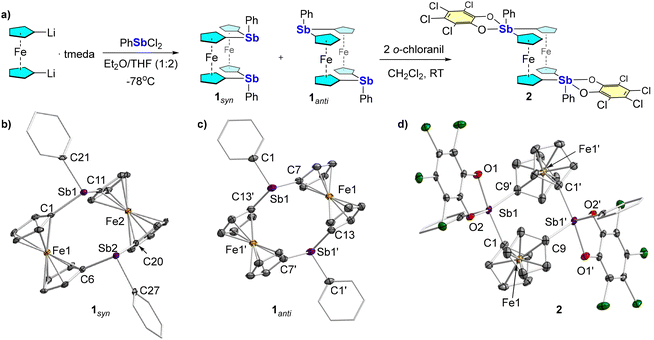
To begin, 1,1′-dilithioferrocene·tmeda (tmeda = N,N,N′,N′-tetramethylethylenediamine) was reacted with one equiv. of PhSbCl2 which, following column chromatography, afforded the distiba[1.1]ferrocenophane 1 as a yellow-orange solid (Fig. 2a). Compound 1 is a rare example of a ferrocenophane which was isolated directly from the reaction mixture at room temperature without light irradiation of its isolated monomer.11,13 In the course of fully characterizing this compound, 1H NMR spectroscopy indicated the formation of two distinct species that are differentiated by their cyclopentadienyl (Cp) proton resonances. As has been found for other heteroatom-bridged ferrocenophane species,11 this suggests the formation of syn and anti-conformations of distiba[1.1]ferrocenophane 1. This proposal was confirmed by the crystallization and determination of the solid-state structures of 1syn and 1anti by single-crystal X-ray diffraction (scXRD, Fig. 2b and c) analysis, which indicated that 1anti exists as a centrosymmetrical dimer. Unfortunately, the lack of an adequate amount of isolated pure 1anti precluded its characterization by 1H and 13C NMR spectroscopy. Nevertheless, examination of the solid-state structures demonstrates that the Cp ligands in both 1syn and 1anti are oriented in a nearly eclipsed conformation and that the two ferrocene units of each isomer stand in registry with one another. The two antimony centers are separated by 3.754(6) Å in the case of 1syn; however, the distance between the two stibine centers for 1anti is significantly greater at 5.221(6) Å. In both cases, the antimony atom adopts a trigonal pyramidal geometry with a slightly broader C(Cp)-Sb-C(Cp) angle for 1anti (103.8(1)°) compared to that for 1syn (101.8(2)°). We note that these isomers do not interconvert even upon heating a mixture of 1syn and 1anti to 100 °C in toluene-d8 (Fig. S5), in contrast to the phosphorus congener.11
We next attempted to access the distibora[1.1]ferrocenophane by treating a mixture of isomers of 1 with two equiv. of o-chloranil in CH2Cl2. This reaction cleanly oxidizes the stibine units to afford compound 2 as a single product in 80% yield (Fig. 2). scXRD analysis reveals that 2 crystallizes with two independent centrosymmetric dimers in the asymmetric unit. Each molecule situates its two Lewis acidic catecholatostiborane centers anti to one another (Fig. 2). Given the steric heft of the tetrachlorocatecholate ligand, we contend that the anti-conformation lessens the Pauli repulsions bewteen the bulky catecholatostiboranes. In the independent molecule depicted in Fig. 2, the antimony atoms adopt a distorted square-pyramidal geometry (τ5 = 0.06)14 with an Sb–Sb separation of 5.113(6) Å. However, we note that both antimony atoms in the second independent molecule adopt a distorted trigonal-bipyramidal geometry (τ5 = 0.68) with an Sb–Sb separation of 5.368(4) Å. Finally, no evidence for isomerization into the syn isomer was obtained, including by 1H NMR spectroscopy in toluene-d8 at 100 °C (Fig. S12).
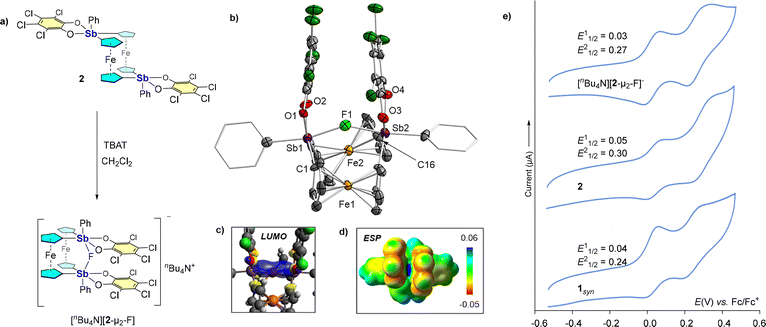
With this potential bidentate Lewis acid in hand, we aimed to investigate its fluoride ion affinity (FIA) both computationally and experimentally. Addition of [nBu4N][PF6] to a CDCl3 solution of 2 did not produce any fluoride adduct, indicating that 2 cannot abstract fluoride from the PF6− ion (Fig. S9–S11). However, addition of one equiv. of [nBu4N][Ph3SiF2] (TBAT) to 2 in CH2Cl2 generates the air- and moisture-stable pale-yellow solid [nBu4N][2-µ2-F] in 80% yield (Fig. 3a). 19F NMR of this material reveals a single resonance at −32.00 ppm, which is in the range of other fluoride-bridged bis-stiborane species.3,4 scXRD analysis confirms the chelation of the fluoride anion between the two catecholatostiborane units (Fig. 3b), which now adopt a syn orientation with respect to the ferrocenophane scaffold. At this geometry, fluoride anion chelation appears perfectly symmetrical, as indicated by equal Sb1-F1 and Sb2-F1 bonds of 2.147(9) Å, a distance that is on par with the Sb-F separations observed in previously described fluoride complexes of A and B.3,4 The resulting Sb1-F1-Sb2 angle of 147.8(4)° is quite oblique,5 reflecting the close proximity of the two stiborane units (Sb-Sb separation 4.125(1) Å) and the narrow fluoride binding pocket. This is also reflected in the computed FIA of 2 of 346 kJ mol−1 which is lower than that of other antimony-based fluoride chelators such a A (365 kJ mol−1) and B (395 kJ mol−1), which we calculated previously using a marginally different level of theory.9 To assess the electronic properties of the host at the adduct geometry, we computationally removed the fluoride anion and evaluated the electronic structure of 2 at that geometry. This approach reveals the presence of the antimony-based σ* orbitals that project into the binding pocket of 2 (Fig. 3c). Moreover, the electrostatic potential map (ESP) displays a region of positive potential between the two Lewis acidic centers (Fig. 3d). Both of these findings substantiate that 2 acts as a dual pnictogen bond donor to the fluoride anion.
But unlike A and B, which have their Lewis acidic centers rigidly predisposed to chelate fluoride, we contend that formation of [2-µ2-F]− is templated by the fluoride anion. That is, the fluoride likely first binds to a single antimony center in the anti configuration of the free receptor, which subsequently rearranges into the observed syn configuration, with the fluoride anion bridging the two antimony centers. We have computed a possible pathway for this process which begins with a highly favorable binding of the fluoride anion to one of the antimony centers of 2 (Fig. 4). Powered by the excess energy of this fluoride-binding step, the resulting intermediate (Int-1) proceeds over a 26.2 kcal mol−1 barrier, ultimately enabling the required conformational change and subsequent fluoride chelation. This mechanistic proposal hinges on the notion that the excess energy of the first step enables an otherwise prohibitive barrier to be overcome at room temperature. This notion is supported by previous mechanistic investigations where similar arguments have been convincingly advanced.15
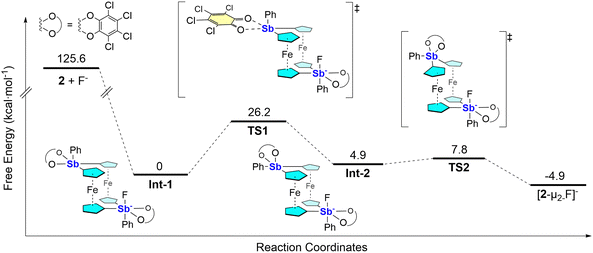 | ||
| Fig. 4 Computed mechanism leading to the formation of [2-µ2-F]−. All energies, the accuracy of which is affected by the level of theory employed, are in kcal mol−1. | ||
Finally, the electrochemical behavior of 1syn, 2, and crystalline [nBu4N][2-µ2-F] was studied by cyclic voltammetry. The voltammogram of 1syn shows two waves of almost equal height, separated by ∼0.20 V (Fig. 3e, vide supra), suggesting two sequential one-electron oxidations of the iron centers. Interestingly, compound 2 shows a perceptible shift towards anodic potential (ΔE1/21 = 10 mV and ΔE1/22 = 60 mV), indicating that the ferrocene centers in 2 become more electron-deficient after oxidation of the antimony center by o-chloranil. Furthermore, the CV of complex [2-µ2-F]− shows a shift towards cathodic potential (ΔE1/21 = 20 mV and ΔE1/22 = 30 mV) with respect to compound 2, consistent with the chelation of an anionic guest that partially screens positive charge development at the iron centers.16
This work provides access to a heretofore unknown distiba[1.1]ferrocenophane, which can be accessed by conventional synthetic routes and converted into a distibora[1.1]ferrocenophanes without involvement of the iron centers in the oxidation process. The anti structure presented by the latter rearranges upon fluoride binding, to position the catecholatostiborane units in a conformation compatible with the chelation of the fluoride anion via convergent pnictogen bonding. The narrow spacing of the Cp ring makes for a rather snug binding pocket, resulting in the puckering of the Sb-F-Sb bridge.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article can be found in the supplementary information (SI). Supplementary information: this SI contains synthetic details, characterization data, spectroscopic data, crystallographic details, and computational details. See DOI: https://doi.org/10.1039/d5cc06492a.CCDC: 2502484–2502487 contain the supplementary crystallographic data for this paper.17a–d
Note added in proof
We acknowledge a recent related publication18 that appeared after submission of this article, and that describes related ferrocene-antimony derivatives, including a stiba[1]ferrocenophane and its polymerization.Acknowledgements
This work was carried out at Texas A&M University with support from the National Science Foundation (CHE-2505795) and the Welch Foundation (A-1423). AT thanks the India-based Science and Engineering Research Board for awarding him an International Research Experience Fellowship (SIR/2022/000058). All calculations were conducted with the advanced computing resources provided by Texas A&M High Performance Research Computing. Finally, we thank Daniel Singleton for useful discussions.References
- (a) J. Qiu, B. Song, X. Li and A. F. Cozzolino, Phys. Chem. Chem. Phys., 2018, 20, 46–50 RSC; (b) J. L. Beckmann, J. Krieft, Y. V. Vishnevskiy, B. Neumann, H.-G. Stammler and N. W. Mitzel, Angew. Chem., Int. Ed., 2023, e202310439 Search PubMed.
- G. Resnati, D. L. Bryce, G. R. Desiraju, A. Frontera, I. Krossing, A. C. Legon, P. Metrangolo, F. Nicotra, K. Rissanen, S. Scheiner and G. Terraneo, Pure Appl. Chem., 2024, 96, 135–145 CrossRef CAS.
- M. Hirai and F. P. Gabbaï, Angew. Chem., Int. Ed., 2015, 54, 1205–1209 CrossRef CAS PubMed.
- C.-H. Chen and F. P. Gabbaï, Angew. Chem., Int. Ed., 2017, 56, 1799–1804 Search PubMed.
- D. You, B. Zhou, M. Hirai and F. P. Gabbaï, Org. Biomol. Chem., 2021, 19, 4949–4957 RSC.
- (a) R. R. Holmes, R. O. Day, V. Chandrasekhar and J. M. Holmes, Inorg. Chem., 1987, 26, 157–163 CrossRef CAS; (b) M. Yang, D. Tofan, C.-H. Chen, K. M. Jack and F. P. Gabbaï, Angew. Chem., Int. Ed., 2018, 57, 13868–13872 Search PubMed.
- (a) C. Dusemund, K. R. A. S. Sandanayake and S. Shinkai, J. Chem. Soc., Chem. Commun., 1995, 333–334 Search PubMed; (b) R. Boshra, K. Venkatasubbaiah, A. Doshi, R. A. Lalancette, L. Kakalis and F. Jäkle, Inorg. Chem., 2007, 46, 10174–10186 CrossRef CAS PubMed; (c) A. E. J. Broomsgrove, D. A. Addy, A. Di Paolo, I. R. Morgan, C. Bresner, V. Chislett, I. A. Fallis, A. L. Thompson, D. Vidovic and S. Aldridge, Inorg. Chem., 2010, 49, 157–173 CrossRef CAS PubMed; (d) M. J. Kelly, A. E. J. Broomsgrove, I. R. Morgan, I. Siewert, P. Fitzpatrick, J. Smart, D. Vidovic and S. Aldridge, Organometallics, 2013, 32, 2674–2684 CrossRef CAS; (e) D. Cao, H. Zhao and F. P. Gabbaï, New J. Chem., 2011, 35, 2299–2305 RSC.
- (a) J. Schulz, J. Antala, D. Rezazgui, I. Císařová and P. Štěpnička, Inorg. Chem., 2023, 62, 14028–14043 CrossRef CAS PubMed; (b) B. Zhou, S. Bedajna and F. P. Gabbaï, Chem. Commun., 2024, 60, 192–195 RSC; (c) J. Antala, J. Schulz, I. Císařová and P. Štěpnička, New J. Chem., 2024, 48, 5107–5119 RSC; (d) D. Rezazgui, J. Schulz and P. Štěpnička, Inorg. Chem., 2025, 64, 11075–11092 CrossRef CAS PubMed.
- B. L. Murphy and F. P. Gabbaï, J. Am. Chem. Soc., 2023, 145, 19458–19477 CrossRef CAS.
- H. Brunner, J. Klankermayer and M. Zabel, J. Organomet. Chem., 2000, 601, 211–219 CrossRef CAS.
- T. Mizuta, Y. Imamura, K. Miyoshi, H. Yorimitsu and K. Oshima, Organometallics, 2005, 24, 990–996 CrossRef CAS.
- C. Spang, F. T. Edelmann, M. Noltemeyer and H. W. Roesky, Chem. Ber., 1989, 122, 1247–1254 CrossRef CAS.
- D. E. Herbert, J. B. Gilroy, W. Y. Chan, L. Chabanne, A. Staubitz, A. J. Lough and I. Manners, J. Am. Chem. Soc., 2009, 131, 14958–14968 CrossRef CAS PubMed.
- A. W. Addison, T. N. Rao, J. Reedijk, J. van Rijn and G. C. Verschoor, J. Chem. Soc., Dalton Trans., 1984, 1349–1356 RSC.
- (a) Y. Oyola and D. A. Singleton, J. Am. Chem. Soc., 2009, 131, 3130–3131 CrossRef CAS PubMed; (b) L. M. M. Quijano and D. A. Singleton, J. Am. Chem. Soc., 2011, 133, 13824–13827 CrossRef CAS PubMed.
- (a) A. J. Taylor, R. Hein, S. C. Patrick, J. J. Davis and P. D. Beer, Angew. Chem., Int. Ed., 2024, 63, e202315959 Search PubMed; (b) R. Hein, A. Docker, J. J. Davis and P. D. Beer, J. Am. Chem. Soc., 2022, 144, 8827–8836 CrossRef CAS PubMed.
- (a) CCDC 2502484: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2q0193; (b) CCDC 2502485: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2q01b4; (c) CCDC 2502486: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2q01c5; (d) CCDC 2502487: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2q01d6.
- H. K. S. Young, H. N. L. Leslie, B. O. Patrick, E. A. LaPierre and I. Manners, Inorg. Chem., 2026, 65, 2833–2840 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: