Open Access Article
Open Access ArticleTriptycene-grafted helicenes: modular synthesis and key properties
Pattarakiat
Seankongsuk
a,
Martin
Vacek
a,
Jiří
Rybáček
 a,
Jaroslav
Vacek
a,
Katsiaryna
Kutsenka
a,
Lucie
Bednárová
a,
Radek
Pohl
a,
Ivana
Císařová
b,
Irena G.
Stará
a,
Jaroslav
Vacek
a,
Katsiaryna
Kutsenka
a,
Lucie
Bednárová
a,
Radek
Pohl
a,
Ivana
Císařová
b,
Irena G.
Stará
 *a and
Ivo
Starý
*a and
Ivo
Starý
 *a
*a
aInstitute of Organic Chemistry and Biochemistry, Czech Academy of Sciences, Flemingovo nám. 2, 166 10 Prague 6, Czech Republic. E-mail: stara@uochb.cas.cz; stary@uochb.cas.cz
bDepartment of Inorganic Chemistry, Faculty of Science, Charles University in Prague, Hlavova 2030/8, Prague 128 43, Czech Republic
First published on 7th April 2026
Abstract
Racemic and enantiopure ditriptyceno[n]helicenes (n = 5–7) were accessed via a modular, straightforward synthesis, enabling a systematic comparison of their conformational dynamics, solubility, electronic and (chir)optical properties with those of the parent helicenes.
Helicenes represent a prominent class of helically twisted polycyclic aromatic hydrocarbons1 whose unique structural chirality and extended π-conjugation endow them with properties attractive for, e.g., asymmetric catalysis,2,3 chiroptics4,5 and spintronics,6–8 spin-polarised charge transport,9–11 optoelectronics12,13 or covalent organic frameworks.14,15 Impressive progress in their synthesis now allows for the construction of hybrid nanographene architectures,16 yet triptycene-grafted helicenes have gained limited attention. There are only sporadic examples of molecules where triptycene is directly annulated to a (hetero)helicene scaffold.17,18 Also related propellane,19 helical ladder20 or triptycenyl helicene21 architectures are rare. Nevertheless, triptycene, a rigid three-dimensional aromatic scaffold, continues to attract interest for imposing spatially decoupled π-systems, providing internal free volume, directing crystal packing, and suppressing deleterious aggregation.22,23
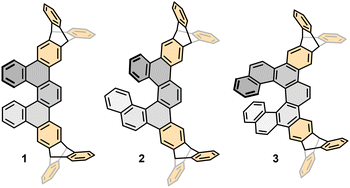
Herein, we report a concise synthetic route to a family of ditriptyceno[n]helicenes (n = 5–7) 1–3 (Fig. 1) and a comprehensive characterisation of their electronic and (chir)optical properties, barrier to racemisation, capricious solubility and crystal packing.
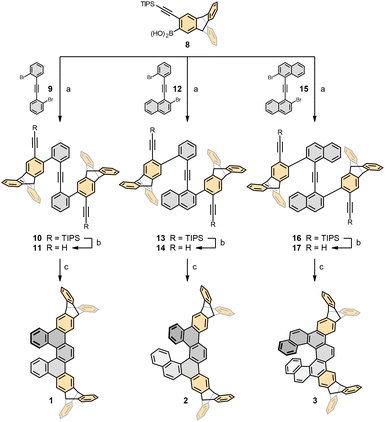
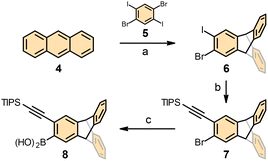
The preparation of racemic ditriptycenohelicenes 1–3 builds on our previously developed synthesis of dibenzohelicenes.24 In this study, we first addressed a straightforward access to o-alkynylated triptycene boronic acid 8 (Scheme 1). In the first step, addition of the aryne generated in situ from 1,4-dibromo-2,5-diiodobenzene 5 to anthracene 4 (both commercially available) furnished bromoiodotriptycene 6 in yields exceeding literature reports.25 After chemoselective alkynylation of 6 with (triisopropylsilyl)acetylene, the bromotriptycene derivative 7 was smoothly converted to the respective boronic acid 8. Thereafter, a unified synthetic sequence delivered ditriptycenohelicenes 1–3 (Scheme 2). First, o-alkynylated triptycene boronic acid 8 was subjected to Suzuki–Miyaura cross-coupling with the homologous tolane-type dibromides 9,261224 and 1526 to receive protected triynes 10, 13 and 16 in high yield. Desilylation proceeded smoothly; however, only compound 11 could be routinely purified, as 14 and 17 showed poor solubility. The compound 14 became sparingly soluble after short-column chromatography and solvent evaporation, while 17 precipitated during the reaction. We nonetheless exposed all triynes to RhCl(PPh3)3-catalysed [2 + 2 + 2] cycloisomerisation in chlorobenzene under high-temperature microwave conditions. While 11, being soluble, delivered the target ditriptyceno[5]helicene 1 in good yield, the aggregated triynes 14 and 17 afforded ditriptyceno[6]helicene 2 and ditriptyceno[7]helicene 3 in modest but acceptable yields.
The solubility of racemic ditriptycenohelicenes 1–3 in organic solvents deserves a brief comment. In general, grafting triptycene end-caps to termini of planar π-systems can markedly enhance solubility by suppressing π–π stacking and introducing free volume, whereas a single triptycene cap primarily modulates solid-state packing.25,27 For instance, a large improvement of solubility in organic solvents by the factor of 70 was described for indigo pigments.28 Unexpectedly, ditriptycenohelicene 1 and, in particular, the higher homologues 2 and 3 exhibited markedly reduced solubility relative to the parent helicenes 18–20. An exception was 2 in toluene, where the “triptycene effect” afforded increased solubility (Table 1).
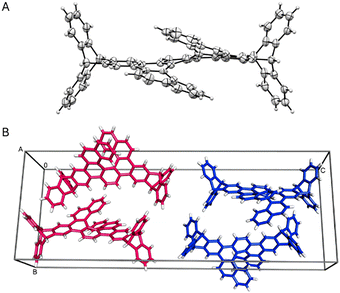
The single-crystal XRD analysis of the racemic ditriptyceno[7]helicene 3 rationalises its reduced solubility (Fig. 2). The compound crystallises as a racemic mixture with two M/P pairs per unit cell. Homochiral molecules assemble into columns along the A axis, interlaced with disordered THF. While the multiple C–H⋯π contacts produce a dense, interdigitated lattice, the parallel-displaced π–π interactions are not present in the crystal.
Racemic ditriptycenohelicenes 1–3 were resolved into enantiomers by chiral HPLC on analytical (1) and also semipreparative (2 and 3) scale (rac-1: Chiralpak IC (DAICEL), rac-2 and rac-3: ChiralArt Cellulose-SA (YMC)) to enable studies of configurational dynamics and chiroptical properties. The experimental barriers to racemisation, determined by dynamic chiral HPLC29 (1) or by monitoring reversible first-order racemisation kinetics (2), agree reasonably with the calculated values and are quite close to those of the parent [5]-, [6]- and [7]helicene 18–20 (Table S4).
Gas-phase DFT B3LYP-D3/cc-pVTZ calculations show that the frontier orbitals (HOMO/LUMO) of 1–3 are localised almost entirely on the central part of the native helicene scaffold, whereas the adjacent levels (HOMO−1/LUMO+1) extend onto the triptycene blades fused to the helix, as illustrated for ditriptyceno[7]helicene 3 (Fig. 3). Calculated HOMO–LUMO gaps (related to the first excited singlet state) and the optical HOMO–LUMO gaps (calculated from absorption spectra) fall in a narrow range of ca 3.50–3.72 eV and decrease slightly from 1 to 3 as the π-conjugated framework extends (Table S2). The electronic behaviour of ditriptycenohelicenes 1–3 follows that of the parent [5]-, [6]- and [7]helicene 18–20.
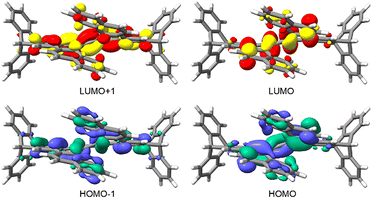 | ||
| Fig. 3 The frontier molecular orbitals of the ditriptyceno[7]helicene 3 calculated by DFT B3LYP/cc-pVTZ/GD3. Grey: carbon, white: hydrogen, orbital isovalue ±0.03 a.u. | ||
The UV–Vis spectra of ditriptycenohelicenes 1–3 show the characteristic multiband profile of π-extended systems, with intense bands across 250–360 nm. The absorption onset shifts modestly to longer wavelength with increasing helical length (≈400 nm for 1, 410 nm for 2 and 420 nm for 3), indicating progressive π-conjugation and a gradual narrowing of the HOMO–LUMO gap (Fig. 4, Table S2). The fluorescence spectra are devoid of multiband character or pronounced vibronic structure, except for the hexahelicene derivative 2, which displays a faint shoulder. The emission maxima follow a similar bathochromic trend, shifting from 431 nm for 1 and 432 nm for 2 to 446 nm for 3.
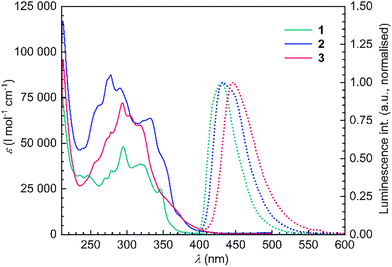 | ||
| Fig. 4 UV-Vis (full lines, 10−4 M in THF) and fluorescence spectra (dotted lines, 10−5 M in THF, exc. 345 nm for 1, 286 nm for 2, 282 nm for 3) of ditriptyceno-helicenes 1–3. | ||
Electronic circular dichroism (ECD) spectra of enantiopure 2 and 3 display clean, near-ideal mirror-image profiles for each enantiomeric pair (Fig. 5 and Fig. S23–S24). The longest- wavelength Cotton effect is positive for the P enantiomers and negative for the M enantiomers (ca. 330–380 nm for 2 and 340–410 for 3), in line with the empirical helicene sign rule and our TD-DFT simulations (see SI). The overall spectral intensity is markedly diminished compared to the parent helicenes, consistent with observations made for the dibenzohelicene series.24 The absolute configurations (−)-M and (+)-P were unambiguously assigned to the isolated enantiomers of 2 and 3 based on comparison of their ECD spectra with those of the reference parent helicenes 19 and 20 and the computed ones.
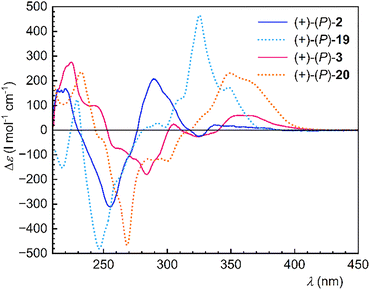 | ||
| Fig. 5 ECD spectra of ditriptycenohelicenes 2 and 3 (full lines) and parent helicenes 19 and 20 (dotted lines) (10−4 M in THF). | ||
In comparison to the parent helicenes 19 and 20, the chiroptical responses of the ditriptycenohelicenes 2 and 3 are attenuated, as evidenced by their smaller |Δε| values in the ECD spectra (vide supra) and markedly reduced specific rotations [α]D20: −155/+162 for (−)-(M)/(+)-(P)-2 (chloroform) vs. −3 629/+3 676 for (−)-(M)/(+)-(P)-19 (dichloromethane)30 or -415/+ 420 for (−)-(M)/(+)-(P)-3 (chloroform) vs. −5 164/+5 101 for (−)-(M)/(+)-(P)-20 (dichloromethane).30 On the other hand, the modest values of the luminescence dissymmetry factor |glum| obtained from the CPL spectra are comparable across the two classes of helicenes: 2.3 × 10−3 (425 nm, THF) for 2vs. 0.9 × 10−3 (ca 424 nm, dichloromethane) for 1931 and 0.9 × 10−3 (430 nm, THF) for 3vs. 6 × 10−3 (450 nm, chloroform) for 20.32
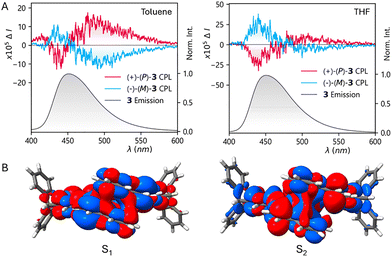
Similarly to the parent [6]helicene 19, the glum value for the ditriptyceno[6]helicene (+)-(P)-2 is negative. In contrast, the situation differs for ditriptyceno[7]helicene (+)-(P)-3 compared to the parent [7]helicene (+)-(P)-20. Whereas the latter reference compound exhibits a positive glum factor, (+)-(P)-3 displays a bisignate CPL spectrum, with a more intense blue-shifted negative band in THF and a red-shifted positive band that predominates in toluene (Fig. 6A). To clarify the behaviour of (+)-(P)-3, we calculated its excited-state properties and CPL luminescence dissymmetry factors for the lowest-energy states, S1 (“bright”) and S2 (“dark”), both possessing predominantly a π → π* character (Table S5). In addition, we recorded temperature-dependent CPL spectra of (+)-(P)-3 (Fig. S30) and measured its fluorescence lifetime, including that of 1 and 2 (Table S6). To explain the bisignate character of the CPL spectrum of (+)-(P)-3, we propose the coexistence of two distinct emissive pathways: (i) The S1 → S0 transition, observed and calculated at ca 490 nm, involves an S1 state that is structurally compressed, with the terminal helicene rings separated by approximately 3.5 Å (cf. 4.0 Å in S0), thereby promoting a transannular vibronic coupling (Fig. 6B, S31). (ii) The S2 → S0 transition, observed at ca 430 nm (calcd. at ca 390 nm), involves an S2 state that is structurally more relaxed, with the terminal helicene rings separated by approximately 4.3 Å. Then, non-adiabatic S1–S2 coupling renders a “dark” S2 state partially bright because, during internal conversion, vibronic mixing (activated by a low-frequency “breathing” mode of the helicene scaffold in the range of 28–40 cm−1) enables it to borrow part of the electric dipole intensity from a nearby bright S1 state (cf. Herzberg–Teller intensity borrowing).33 It is worth noting that the S1/S2 PES crossing occurs along the “breathing” mode coordinate. This vibronic coupling is further modulated by the solvent environment and temperature, lending additional support to this model (for a discussion, see Fig. S32).
In summary, we have prepared a series of racemic and enantiopure ditriptyceno[n]helicenes (n = 5–7) 1–3, a hybrid family hitherto unreported in the literature. Grafting two triptycene units onto the helicene scaffolds does not substantially perturb their conformational dynamics and electronic or optical properties relative to the parent helicenes. However, the ditriptycenohelicenes show some differences such as slightly reduced optical HOMO–LUMO gaps, mostly attenuated chiroptical responses and incoherent solubility.
P. Seankongsuk synthesised and characterised triptycene-grafted helicenes 2 and 3, carried out solubility tests and contributed to the SI. M. Vacek pioneered the synthesis and characterisation of triptycene-grafted helicene 1, calculated its racemisation barrier and contributed to the SI. J. Rybáček performed DFT calculations, supervised chiral separations, interpreted the ECD spectra, contributed to the SI and co-wrote the manuscript. J. Vacek carried out DFT calculations, interpreted the CPL spectra and co-wrote the manuscript. K. Kutsenka supervised the synthesis and characterisation of triptycene-grafted helicene 1, measured and interpreted fluorescence lifetimes. L. Bednárová measured and interpreted the (chir)optical spectra. R. Pohl recorded and interpreted the NMR spectra. I. Císařová conducted the single-crystal XRD analysis. I. G. Stará and Ivo Starý conceived and directed the project and co-wrote the manuscript. All authors contributed to the writing and verified the experimental data.
Conflicts of interest
There are no conflicts to declare.Data availability
The data that support this article have been included in the supplementary information (SI). supplementary information: synthesis and characterisation data of new compounds including HPLC resolution of enantiomers, (chir)optical spectra, racemisation barriers and computational details. See DOI: https://doi.org/10.1039/d5cc06240c.CCDC 2492687 contains the supplementary crystallographic data for this paper.34
Acknowledgements
We are grateful to the Czech Science Foundation (Reg. No. 24-10787S) and the Institute of Organic Chemistry and Biochemistry, Czech Academy of Sciences (RVO: 61388963) for their funding of this research. I.S. gratefully acknowledges the support of the Praemium Academiae of the Czech Academy of Sciences. We thank IOCB Core Facilities for technical assistance in the characterisation of the compounds.References
- Helicenes: Synthesis, Properties and Applications, ed. J. Crassous, I. G. Stará and I. Starý, John Wiley & Sons, Ltd, Weinheim, 2022 Search PubMed.
- D. Sakamoto, I. Gay Sánchez, J. Rybáček, J. Vacek, L. Bednárová, M. Pazderková, R. Pohl, I. Císařová, I. G. Stará and I. Starý, ACS Catal., 2022, 12, 10793–10800 CrossRef CAS.
- T. Edlová, J. Rybáček, H. Cattey, J. Vacek, L. Bednárová, P. Le Gendre, A. T. Normand, I. G. Stará and I. Starý, Angew. Chem., Int. Ed., 2025, 64, e202414698 CrossRef PubMed.
- J. Crassous, M. J. Fuchter, D. E. Freedman, N. A. Kotov, J. Moon, M. C. Beard and S. Feldmann, Nat. Rev. Mater., 2023, 365–371 CrossRef.
- I. Shioukhi, H. Batchu, G. Schwartz, L. Minion, Y. Deree, B. Bogoslavsky, L. J. W. Shimon, J. Wade, R. Hoffman, M. J. Fuchter, G. Markovich and O. Gidron, Angew. Chem., Int. Ed., 2024, 63, e202319318 CrossRef CAS PubMed.
- A. Borissov, P. J. Chmielewski, C. J. Gómez García, T. Lis and M. Stępień, Angew. Chem., Int. Ed., 2023, 62, e202309238 CrossRef CAS PubMed.
- S. Cadeddu, R. Chowdhury, C. Delpiano Cordeiro, S. Parmar, A. Kramer, M. Cordier, A. Pensel, N. Vanthuyne, R. Sessoli, M. Chiesa, Y.-K. Liao, R. H. Friend, E. Salvadori, J. Autschbach and J. Crassous, J. Am. Chem. Soc., 2025, 147, 23643–23653 CrossRef CAS PubMed.
- J. Borstelmann, S. Zank, M. Krug, G. Berger, N. Fröhlich, G. Glotz, F. Gnannt, L. Schneider, F. Rominger, F. Deschler, T. Clark, G. Gescheidt, D. M. Guldi and M. Kivala, Angew. Chem., Int. Ed., 2025, 64, e202423516 CrossRef CAS PubMed.
- V. Kiran, S. P. Mathew, S. R. Cohen, I. Hernández Delgado, J. Lacour and R. Naaman, Adv. Mater., 2016, 28, 1957–1962 CrossRef CAS PubMed.
- R. Rodríguez, C. Naranjo, A. Kumar, P. Matozzo, T. K. Das, Q. Zhu, N. Vanthuyne, R. Gómez, R. Naaman, L. Sánchez and J. Crassous, J. Am. Chem. Soc., 2022, 144, 7709–7719 CrossRef PubMed.
- M. R. Safari, F. Matthes, C. M. Schneider, K.-H. Ernst and D. E. Bürgler, Small, 2024, 20, 2308233 CrossRef CAS PubMed.
- Y. Yang, R. C. da Costa, M. J. Fuchter and A. J. Campbell, Nat. Photonics, 2013, 7, 634–638 CrossRef CAS.
- W.-C. Guo, W.-L. Zhao, K.-K. Tan, M. Li and C.-F. Chen, Angew. Chem., Int. Ed., 2024, 63, e202401835 CrossRef CAS PubMed.
- Q. Yan, S. Tao, R. Liu, Y. Zhi and D. Jiang, Angew. Chem., Int. Ed., 2024, 63, e202316092 CrossRef CAS PubMed.
- J. del, R. Monroy, T. Deshpande, J. Schlecht, C. Douglas, R. Stirling, N. Grabicki, G. J. Smales, Z. Kochovski, F. G. Fabozzi, S. Hecht, S. Feldmann and O. Dumele, J. Am. Chem. Soc., 2025, 147, 17750–17763 CrossRef PubMed.
- N. Martin and C. P. Nuckolls, Molecular Nanographenes: Synthesis, Properties, and Applications, John Wiley & Sons, 2025 Search PubMed.
- H. Mubarok, A. Amin, T. Lee, J. Jung, J.-H. Lee and M. H. Lee, Angew. Chem., Int. Ed., 2023, 62, e202306879 CrossRef CAS PubMed.
- K.-K. Tan, M. Li and C.-F. Chen, Chem. Commun., 2025, 61, 11191–11194 RSC.
- J. Ma, X. Liu, F. Hao and L. Zhang, Angew. Chem., Int. Ed., 2025, 64, e202503225 CrossRef CAS PubMed.
- T. Ikai, T. Yoshida, K. Shinohara, T. Taniguchi, Y. Wada and T. M. Swager, J. Am. Chem. Soc., 2019, 141, 4696–4703 CrossRef CAS PubMed.
- T. R. Kelly, Acc. Chem. Res., 2001, 34, 514–522 CrossRef CAS PubMed.
- M. N. Khan and T. Wirth, Chem. – Eur. J., 2021, 27, 7059–7068 CrossRef CAS PubMed.
- F. Ishiwari, Y. Shoji, C. J. Martin and T. Fukushima, Polym. J., 2024, 56, 791–818 CrossRef CAS.
- A. Jančařík, J. Rybáček, K. Cocq, J. Vacek Chocholoušová, J. Vacek, R. Pohl, L. Bednárová, P. Fiedler, I. Císařová, I. G. Stará and I. Starý, Angew. Chem., Int. Ed., 2013, 52, 9970–9975 CrossRef PubMed.
- L. Ueberricke, J. Schwarz, F. Ghalami, M. Matthiesen, F. Rominger, S. M. Elbert, J. Zaumseil, M. Elstner and M. Mastalerz, Chem. – Eur. J., 2020, 26, 12596–12605 CrossRef CAS PubMed.
- H.-K. Chang, S. Datta, A. Das, A. Odedra and R.-S. Liu, Angew. Chem., Int. Ed., 2007, 25, 4744–4747 CrossRef PubMed.
- L. Ueberricke and M. Mastalerz, Chem. Rec., 2021, 21, 558–573 CrossRef CAS PubMed.
- B. P. Benke, L. Hertwig, X. Yang, F. Rominger and M. Mastalerz, Eur. J. Org. Chem., 2021, 72–76 CrossRef CAS PubMed.
- O. Trapp, J. Chromatogr. B, 2008, 875, 42–47 CrossRef CAS PubMed.
- M. Šámal, J. Rybáček, J. Holec, J. Hanus, J. Vacek, M. Buděšínský, L. Bednárová, P. Fiedler, M. Š. Slušná, I. G. Stará and I. Starý, Chem. Commun., 2022, 58, 12732–12735 RSC.
- H. Tanaka, M. Ikenosako, Y. Kato, M. Fujiki, Y. Inoue and T. Mori, Commun. Chem., 2018, 1, 1–8 CAS.
- H. Kubo, T. Hirose, T. Nakashima, T. Kawai, J. Hasegawa and K. Matsuda, J. Phys. Chem. Lett., 2021, 12, 686–695 CrossRef CAS PubMed.
- Y. Liu, J. Cerezo, G. Mazzeo, N. Lin, X. Zhao, G. Longhi, S. Abbate and F. Santoro, J. Chem. Theory Comput., 2016, 12, 2799–2819 CrossRef CAS PubMed.
- CCDC 2492687: Experimental Crystal Structure Determination, 2026 DOI:10.5517/ccdc.csd.cc2pnv8h.
| This journal is © The Royal Society of Chemistry 2026 |

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
: