Open Access Article
Open Access ArticleHeteroatom constructed covalent organic frameworks for oxygen electrocatalysis and rechargeable zinc–air batteries
Anup Kumar Pradhan a,
Bhushan Kishor Nandrea,
Pandiyan Sivasakthia,
Sayan Halder
a,
Bhushan Kishor Nandrea,
Pandiyan Sivasakthia,
Sayan Halder a,
Sasanka Dalapatib,
Asim Bhaumik
a,
Sasanka Dalapatib,
Asim Bhaumik c,
Pralok K. Samanta*a and
Chanchal Chakraborty
c,
Pralok K. Samanta*a and
Chanchal Chakraborty *a
*a
aDepartment of Chemistry, Birla Institute of Technology and Science, Pilani, Hyderabad Campus, Jawahar Nagar, Kapra Mandal, Medchal District, Telangana 500078, India. E-mail: pralokkumar.samanta@hyderabad.bits-pilani.ac.in; chanchal@hyderabad.bits-pilani.ac.in
bDepartment of Materials Science, School of Technology, Central University of Tamil Nadu (CUTN), Thiruvarur, 610005, Tamil Nadu, India
cSchool of Materials Sciences, Indian Association for the Cultivation of Science (IACS), 2A & 2B Raja S. C. Mallick Road, Jadavpur, Kolkata, 700032, West Bengal, India
First published on 9th February 2026
Abstract
Two low-bandgap, metal-free donor–acceptor COFs exhibit bifunctional OER/ORR activity, with N,S-rich BTTh-TZ-COF delivering superior charge transport, abundant active sites, and enhanced solid-state ZAB performance.
Driven by fossil fuel depletion and associated environmental concerns, sustainable energy storage technologies such as rechargeable metal-air batteries (MABs) have attracted attention for their high theoretical energy density and eco-friendly nature. Here, the discharging and charging processes of the MABs are accompanied by the oxygen reduction reaction (ORR) and oxygen evolution reaction (OER) at the same electrode via multistep electron-transfer pathways.1a However, the ORR is intrinsically sluggish, mainly due to complex adsorption/desorption dynamics and a reaction pathway that involves multiple oxygen-containing intermediates, e.g., OOH, O, and OH*. State-of-the-art ORR catalysts rely on Pt-based nanomaterials, but their high cost, scarcity, susceptibility to fuel crossover, and poor durability have driven the development of low-cost transition-metal alternatives for oxygen electrocatalysis and MABs.1b–d
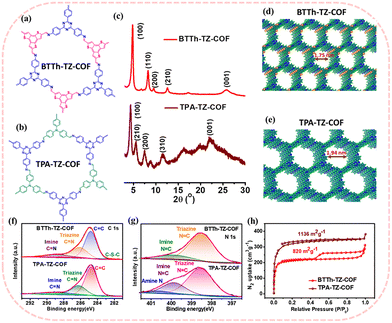
Covalent organic frameworks (COFs), a class of porous crystalline polymers constructed from covalently linked building blocks, have recently emerged as versatile materials with a wide range of applications.2 Guided by the reticular chemistry principles, COFs enable the construction of highly ordered networks in which moieties are precisely incorporated at the atomic scale. Additionally, the intrinsic physicochemical properties of COFs can be strategically tuned to achieve electrocatalytic activity comparable to that of noble metals.3 In this regard, very limited reports with heteroatom incorporated COFs like porphyrin-based COFs,4 a cobalt-centered COFs,5 quaternary phosphonium COFs with high hydroxide conductivity,6a and fluorinated COFs with enhanced oxygen-transport nanochannels6b are reported for high-performance ZABs. The heteroatom incorporation optimizes charge distribution and adsorption energies, enabling electrocatalytic activity comparable to that of conventional noble-metal-based catalysis.7 Herein, we have reported two heteroatom-containing COFs [benzene-trithiophene (BTTh) and triazine-based BTTh-TZ-COF, triphenyl amine (TPA) and triazine-based TPA-TZ-COF] (Fig. 1a and b) for fabricating ZABs. The synthesis protocols of the COFs are given in the Electronic Supplementary Information (ESI), Scheme S1. BTTh-TZ-COF displays superior bifunctional electrocatalytic activity with a higher ORR onset potential (0.92 V vs. RHE) and lower OER overpotential (373 mV), and delivers higher specific capacity, energy, and power density in solid-state ZABs than TPA-TZ-COF.
The powder X-ray diffraction (PXRD) of the BTTh-TZ-COF and TPA-TZ-COF (Fig. 1c) confirms high crystallinity in both the COFs. The extended 2D COF models for both COFs, as shown in Fig. 1d-e, were built and optimized using density functional theory, as discussed in the SI. The XRD pattern of BTTh-TZ-COF shows diffraction peaks at 2θ = 4.8°, 8.3°, 9.5°, 12.6°, and 25.8° corresponding to the 100, 110, 200, 210, and 001 crystalline planes, respectively, and is consistent with previous reports.8,9 In our previous report, the lattice dimensions of the hexagonal 2D BTTh-TZ-COF after Pawley refinement were reported as a = b = 21.80 Å, c = 3.51 Å, α = β = 90°, and γ = 120° with a profile residual (Rp) and weighted profile residual (Rwp) of 3.66% and 4.96%, respectively.9 Similarly, TPA-TZ-COF crystallizes in a 2D hexagonal structure and shows diffraction peaks at 4.35°, 7.47°, 8.79°, 11.62°, and 24.10°, corresponding to the 100, 110, 200, 210, and 001 crystalline planes, respectively, as shown in Fig. 1c. The lattice dimensions of the TPA-TZ-COF are estimated to be a = b = 22.58 Å, c = 3.62 Å, α = β = 90°, and γ = 120° (Fig. S1 and Table S1 in the SI), whereas Rp and Rwp are 3.83% and 5.21%.
The elemental analysis of the COFs through XPS spectra reveals the presence of C, N, and S in BTTh-TZ-COF, and C and N in TPA-TZ-COF, as depicted in Fig. S2. As shown in Fig. 1f, the C 1s core level XPS spectra of BTTh-TZ-COF are fitted into four distinct peaks at 288.6, 286.3, 284.3, and 283.1 eV, attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N (imine), C
N (imine), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N (triazine), C
N (triazine), C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (aromatic), and C–S–C bonds, respectively. The C 1s spectra of TPA-TZ-COF show three peaks at 284.5, 286.2, and 288.5 eV, corresponding to C
C (aromatic), and C–S–C bonds, respectively. The C 1s spectra of TPA-TZ-COF show three peaks at 284.5, 286.2, and 288.5 eV, corresponding to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (aromatic), N
C (aromatic), N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (triazine), and N
C (triazine), and N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (imine) carbon, respectively. The N 1s spectra of BTTh-TZ-COF are fitted into two peaks at 398.5 and 400.1 eV, corresponding to triazine N
C (imine) carbon, respectively. The N 1s spectra of BTTh-TZ-COF are fitted into two peaks at 398.5 and 400.1 eV, corresponding to triazine N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and imine N
C and imine N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, respectively. Similarly, the N 1s spectra of TPA-TZ-COF show three fitted peaks of N
C, respectively. Similarly, the N 1s spectra of TPA-TZ-COF show three fitted peaks of N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (triazine), N
C (triazine), N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C (imine), and amine N at 398.6, 399.8, and 400.5 eV, respectively, as shown in Fig. 1g. Additionally, the S 2p spectra of BTTh-TZ-COF are fitted at 163.7 and 165.4 eV, corresponding to 2p3/2 and 2p1/2, as depicted in Fig. S3.
C (imine), and amine N at 398.6, 399.8, and 400.5 eV, respectively, as shown in Fig. 1g. Additionally, the S 2p spectra of BTTh-TZ-COF are fitted at 163.7 and 165.4 eV, corresponding to 2p3/2 and 2p1/2, as depicted in Fig. S3.
The Brunauer–Emmett–Teller (BET) surface areas of BTTh-TZ-COF and TPA-TZ-COF, measured through N2 adsorption–desorption isotherms at 77 K, are estimated as 820 m2 g−1 and 1136 m2 g−1, respectively. BTTh-TZ-COF exhibits a predominant type-I isotherm with a clear H1-type desorption hysteresis, whereas TPA-TZ-COF discloses a type-I isotherm with a sharp capillary N2 uptake at low relative pressure (P/P0 < 0.04) and gradual high-pressure adsorption, indicating coexisting micropores and mesopores arising from interparticle voids in the COFs.10,11 The pore size distribution study shows the existence of 1D microporous channels within the 2D COF framework due to the π–π stacked layers, as shown in Fig. 1d and e. The total pore volumes and average pore sizes are calculated to be 0.25 cm3 g−1 and 1.17 nm for BTTh-TZ-COF, and 1.18 nm for TPA-TZ-COF, with stretching up to 2 nm, well aligned with the theoretical pore sizes of 1.75 nm in BTTh-TZ-COF and 1.94 nm in TPA-TZ-COF (Fig. 1d, e and Fig. S4 and Table S2), estimated from DFT-optimized structural models. The SEM study of BTTh-TZ-COF (Fig. S5) reveals a spherical aggregated morphology, whereas TPA-TZ-COF divulges the self-assembled aggregated structures. The substantial π–π stacking between the COF layers provides the highly aggregated morphologies in both cases. In the FT-IR spectra of BTTh-TZ-COF and TPA-TZ-COF, as shown in Fig. S6a and b, the characteristic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching frequencies at 1665 cm−1 and 1680 cm−1 attributed to the corresponding aldehyde precursors have disappeared in BTTh-TZ-COF and TPA-TZ-COF, respectively. The disappearance of the N–H stretching bands and the appearance of C
O stretching frequencies at 1665 cm−1 and 1680 cm−1 attributed to the corresponding aldehyde precursors have disappeared in BTTh-TZ-COF and TPA-TZ-COF, respectively. The disappearance of the N–H stretching bands and the appearance of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching peaks at ∼1581 and ∼1588 cm−1 confirm complete precursor consumption and successful formation of imine-linked COF networks.
N stretching peaks at ∼1581 and ∼1588 cm−1 confirm complete precursor consumption and successful formation of imine-linked COF networks.
The solid-state UV-visible spectra (Fig. S7a) demonstrate BTTh-TZ-COF with a strong absorption at 468 nm with a broad NIR tail, while TPA-TZ-COF reveals a maximum at 394 nm with a shoulder at 472 nm, attributed to π–π* and n–π* transitions and donor–acceptor charge transfer within the frameworks.12 The broader and more extended tail in BTTh-TZ-COF denotes the better donor–acceptor property in the BTTh-based donor than that of the TPA-based donor to the TZ-based acceptors in the framework. BTTh-TZ-COF exhibits a lower optical band gap (2.04 eV) than TPA-TZ-COF (2.31 eV) (Fig. S7b), consistent with the cyclic voltammetry (CV)-derived bandgap trend (Fig. S8, SI), indicating enhanced suitability of BTTh-TZ-COF for optoelectronic, electrical, and electrochemical applications. The HOMO and LUMO positions for BTTh-TZ-COF and TPA-TZ-COF are calculated to be −5.07 and −5.14 eV (HOMO) and −2.92 and −2.90 eV (LUMO), respectively.
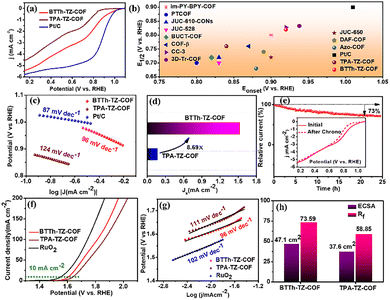
Cyclic voltammograms of BTTh-TZ-COF and TPA-TZ-COF exhibit reduction peaks at 0.81 V and 0.78 V, respectively, in O2-saturated 0.1 M KOH electrolyte (Fig. S9), confirming that the ORR on BTTh-TZ-COF is more thermodynamically favourable, as it occurs at a more positive potential.13 Cathodic linear sweep voltammetry (CLSV) curves, as depicted in Fig. 2a, disclose a promising onset potential (Eonset) of 0.92 V and a half-wave potential (E1/2) of 0.82 V for BTTh-TZ-COF. These values are much higher than those of the TPA-TZ-COF-based electrode (Eonset = 0.83 V and E1/2 = 0.77 V, Fig. 2b). Even the limiting current density of BTTh-TZ-COF (5.43 mA cm−2) is higher than that of TPA-TZ-COF (4.56 mA cm−2). Interestingly, the metal-free BTTh-TZ-COF reveals comparable ORR performance to a commercial 20% Pt/C catalyst (Eonset = 1.01 V, E1/2 = 0.90 V, and JL = 5.73 mA cm−2). Fig. 2a shows that, unlike Pt/C with a clear diffusion-limited plateau from fast 4e− ORR and high conductivity, BTTh-TZ-COF and TPA-TZ-COF exhibit continuously rising currents due to slower metal-free ORR kinetics and lower conductivity, indicating kinetic or mixed-control behaviour. Furthermore, the prepared BTTh-TZ-COF catalyst is more efficient in ORR activity than previously reported metal-free COF-based electrocatalysts, as depicted in Fig. 2b and Table S3. As shown in Fig. 2c, the BTTh-TZ-COF exhibits a lower Tafel slope (96 mV dec−1) than the TPA-TZ-COF (124 mV dec−1) and is comparable to Pt/C (87 mV dec−1), indicating enhanced ORR kinetics in the BTTh-TZ-COF.14 Consequently, the low Tafel slope of BTTh-TZ-COF also indicates a low oxygen coverage and faster oxygen dissociation rate on the surface of BTTh-TZ-COF during ORR activity.15 On the other hand, the kinetic current density (JK) of BTTh-TZ-COF is 8.69 times higher than the JK value of TPA-TZ-COF (Fig. 2d), implying the higher intrinsic ORR activity. The calculated number of transferred electrons during the ORR at 0.56 V using the Koutecky–Levich (K–L) equation is 3.24 and 2.89 for BTTh-TZ-COF and TPA-TZ-COF, respectively (Fig. S10a–c). These results indicate a more efficient dissociative pathway in BTTh-TZ-COF than in TPA-TZ-COF. Again, the BTTh-TZ-COF divulges high catalytic stability as it retains 73% relative current density even after 25 h (Fig. 2e). Anodic linear sweep voltammetry (ALSV) curves in Fig. 2f reveal that the BTTh-TZ-COF and TPA-TZ-COF require 52 mV and 90 mV more overpotential than that of commercial RuO2 (321 mV). The corresponding Tafel slopes (BTTh-TZ-COF: 96 mV dec−1, TPA-TZ-COF: 111 mV dec−1, and RuO2: 102 mV dec−1) confirm the better charge transfer over the electrocatalytic interface in BTTh-TZ-COF catalysts with enhanced OER kinetics (Fig. 2g). Again, the better catalytic performance of BTTh-TZ-COF is attributed to its higher ECSA of 47.1 cm2 and higher Rf of 73.59 than that of TPA-TZ-COF (ECSA: 37.6 cm2, Rf: 58.85), as shown in Fig. S11 and Fig. 2h. The ECSA normalized LSV curves in Fig. S12 disclosed quite similar current density in BTTh-TZ-COF compared to TPA-TZ-COF, indicating that the enhanced catalytic activity of BTTh-TZ-COF primarily originated from the higher active site. Moreover, the post-OER XPS analysis of BTTh-TZ-COF and TPA-TZ-COF, as shown in Fig. S13, indicates that the C1s, N1s, and S2p spectra of both COFs remain largely unchanged compared to the pristine samples, indicating preservation of chemical environments and negligible structural reconstruction. The appearance of a broad peak around 292 eV in the C 1s spectra was attributed to the presence of –CF2– in the sample, as Nafion was used as a binder.
The theoretical study based on the DFT method provides detailed information about the OER and ORR mechanisms for BTTh-TZ-COF and TPA-TZ-COF (Fig. 3a, b and Table S4 and Fig. S14–S18).
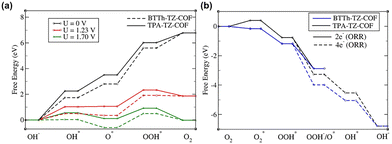 | ||
| Fig. 3 Energy profile for the (a) OER and (b) ORR, on BTTh-TZ-COF and TPA-TZ-COF using the PBE exchange–correlation functional. U is the applied bias for the OER. | ||
The rate-determining step for the OER is the *O to *OOH conversion, as it is the highest energy barrier for both BTTh-TZ-COF and TPA-TZ-COF. The overall lower energy reaction pathway for BTTh-TZ-COF makes BTTh-TZ-COF a more favourable catalyst for the OER and ORR than TPA-TZ-COF (Fig. 3a and b). The higher efficiency of BTTh-TZ-COF towards the OER and ORR is due to the favourable adsorption of different species (Tables S4 and S5). DFT analysis indicates that OH adsorption during the OER is dominated by the C1 site in BTTh-TZ-COF, while the C4 site is most active in TPA-TZ-COF (Fig. S14). The adsorption energies for OH− are 1.74 eV and 2.26 eV in BTTh-TZ-COF and TPA-TZ-COF, respectively. Although the energy barrier of *O to *OOH for BTTh-TZ-COF (2.79 eV) is higher than that of TPA-TZ-COF (2.51 eV), the presence of much higher density of active sites in BTTh-TZ-COF (6 and 3 active sites per unit in BTTh-TZ-COF and TPA-TZ-COF, respectively, Table S5) compensates for the higher barrier, resulting in a lower overpotential and better overall efficiency. Thus, the presence of S-containing donors in BTTh-TZ-COF, along with the N-containing moieties, generates better adsorption and higher active site density in the BTTh-TZ-COF framework than in the only N-containing TPA-TZ-COF. Again, the 4-electron transfer process is more favourable than the 2-electron process for the ORR of both COFs, consistent with our experimental data.
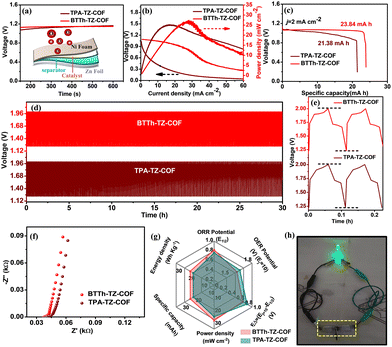
To meet the growing demand for energy, we fabricated a solid-state ZAB with COF on a Ni-foam air cathode, a Zn-plate anode, and a PVA–KOH–Zn(CH3COO)2 gel electrolyte.16 The single-cell OCV values are 1.13 V for BTTh-TZ-COF and 1.07 V for TPA-TZ-COF (Fig. 4a). The BTTh-TZ-COF-based ZAB delivers a higher areal power density of 26.36 mW cm−2 at 28.15 mA cm−2 compared to 25.02 mW cm−2 at 15.57 mA cm−2 for TPA-TZ-COF (Fig. 4b), indicating superior charge transfer on the electrode surface of BTTh-TZ-COF during discharging. The significant discharge activity in the solid-state device primarily stems from the systematically enhanced ORR activity of BTTh-TZ-COF.
BTTh-TZ-COF and TPA-TZ-COF-based solid-state ZABs exhibit specific capacities of 23.84 and 21.38 mAh, respectively, at 2 mA cm−2 (Fig. 4c). Both ZABs show stable cycling over 30 h (Fig. 4d), with BTTh-TZ-COF achieving a higher round-trip efficiency (64%) than TPA-TZ-COF (60%) (Fig. 4e). EIS results (Fig. 4f) confirm low charge transfer resistance (RCT) for both COFs; in particular, the RCT of BTTh-TZ-COF is lower than that of TPA-TZ-COF. The spider chart (Fig. 4g) compares the overall electrochemical performances of BTTh-TZ-COF and TPA-TZ-COF, revealing better oxygen electrocatalysis in BTTh-TZ-COF. Two BTTh-TZ-COF containing ZABs connected in series can light up a green LED bulb (Fig. 4h), signifying the practical demonstration of the BTTh-TZ-COF-based ZAB. Moreover, the battery performances of our COF-based ZABs are compared with those of previously reported COF-based ZABs in Table S6.
In summary, two low-bandgap, metal-free, donor–acceptor porous COFs are prepared to compare their bifunctional OER and ORR electrocatalytic activities. The S-containing BTTh-TZ-COF exhibits better donor–acceptor properties, a lower bandgap, improved transport properties, higher ECSA, and more active sites, surpassing the OER or ORR activity of TPA-TZ-COF. DFT calculations also demonstrate a lower reaction energy barrier for bifunctional electrocatalysis in BTTh-TZ-COF. Furthermore, the solid-state ZAB constructed with BTTh-TZ-COF as the air cathode exhibits higher specific capacity, energy, and power densities than BTTh-TZ-COF. This work offers new insights into the design of catalytic donor–acceptor COFs for advanced energy storage and conversion applications.
C. C. and B. K. N. acknowledge the financial support from the SERB-SURE grant from the Science & Engineering Research Board (SERB), Department of Science and Technology, Government of India. (SUR/2022/002293). The authors are grateful for DST-FIST and DST-PURSE (SR/PURSE/2020/20) (G) project funding from the Department of Science and Technology (DST), Govt. of India. A. K. P. acknowledges the Council of Scientific & Industrial Research (CSIR), Govt. of India, for his fellowship. The authors thank the high-performance computing facility, Sharanga, at the Birla Institute of Technology and Science - Pilani, Hyderabad Campus.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included in the article or as part of the supplementary information (SI). Supplementary information: details of the synthesis procedure, modelling, spectroscopic and electrochemical characterizations, XPS, DFT, and performance comparison tables. See DOI: https://doi.org/10.1039/d5cc06145h.References
- (a) S. Li, L. Shi, Y. Guo, J. Wang, D. Liu and S. Zhao, Chem. Sci., 2024, 15, 11188–11228 RSC; (b) Z. Pei, H. Zhang, Y. Guo, D. Luan, X. Gu and X. W. Lou, Adv. Mater., 2024, 36, 2306047 CrossRef CAS PubMed; (c) Z.-P. Wu, S. Zuo, R. Wu, C. Chen, X. Zhao, Y. Ren, X. T. Xiao, J. Zhang, J. Ruiz-Martinez, Z. P. Liu and H. Zhang, J. Am. Chem. Soc., 2025, 147, 36763–36773 CrossRef CAS PubMed; (d) X. F. Lu, S. L. Zhang, E. Shangguan, P. Zhang, S. Gao and X. W. Lou, Adv. Sci., 2020, 7, 2001178 CrossRef CAS PubMed.
- (a) A. P. Côté, A. I. Benin, N. W. Ockwig, M. O'Keeffe, A. J. Matzger and O. M. Yaghi, Science, 2005, 310, 1166–1170 CrossRef PubMed; (b) K. Geng, T. He, R. Liu, S. Dalapati, K. T. Tan, Z. Li, S. Tao, Y. Gong, Q. Jiang and D. L. Jiang, Chem. Rev., 2020, 120, 8814–8933 CrossRef CAS PubMed; X. T. Ren, M. Y. Wen, X. B. Hou, J. J. Sun, F. Bai and Y. S. Li, Chem. Commun., 2024, 60, 4423–4426 Search PubMed.
- R. Cheng, B. Ran, X. Zhang, Y. Han, X. Shao, H. Li and C. Fu, Adv. Funct. Mater., 2024, 34, 2406717 CrossRef CAS.
- (a) B. Li, S. Y. Zhang, B. Wang, Z. J. Xia, C. Tang and Q. Zhang, Energy Environ. Sci., 2018, 11, 1723–1729 Search PubMed; (b) S. Bhunia, A. Peña-Duarte, H. Li, H. Li, M. F. Sanad, P. Saha, M. A. Addicoat, K. Sasaki, T. A. Strom, M. J. Yacaman, C. R. Cabrera, S. Bhattacharya, J. Bredas and L. Echegoyen, ACS Nano, 2023, 17, 3492–3505 Search PubMed.
- H. Zhang, Z. Qu, H. Tang, X. Wang, R. Koehler, M. Yu, C. Gerhard, Y. Yin, M. Zhu, K. Zhang and O. G. Schmidt, ACS Energy Lett., 2021, 6, 2491–2498 CrossRef CAS.
- (a) Y. Tian, X. Hui, K. Wang, Y. Yuan, H. Chen, K. T. Bang, R. Tao, R. Wang, D. Shin, Y. Lan, Z. Xu and Y. Kim, Angew. Chem., Int. Ed., 2024, 137, e202419257 CrossRef; (b) Q. Cao, L. Wan, Z. Xu, W. Kuang, H. Liu, X. Zhang, W. Zhang, Y. Lu, Y. Yao, B. Wang and K. Liu, Adv. Mater., 2023, 35, 2210550 CrossRef CAS PubMed.
- (a) C. Liu, F. Liu, H. Li, J. Chen, J. Fei, Z. Yu, Z. Yuan, C. Wang, H. Zheng, Z. Liu, M. Xu, G. Henkelman, L. Wei and Y. Chen, ACS Nano, 2021, 15, 3309–3319 CrossRef CAS PubMed; (b) D. Li, C. Li, L. Zhang, H. Li, L. Zhu, D. Yang, Q. Fang, S. Qiu and X. Yao, J. Am. Chem. Soc., 2020, 142, 8104–8108 CrossRef CAS PubMed.
- H. Liu, X. Zheng, J. Xu, X. Jia, M. Chao, D. Wang and Y. Zhao, ACS Appl. Mater. Interfaces, 2023, 15, 16794–16800 Search PubMed.
- B. N. Nandre, S. Halder, S. Dalapati, A. Bhaumik and C. Chakraborty, Sol. Energy Mater. Sol. Cells, 2026, 295, 113979 Search PubMed.
- B. K. Nandre, S. Halder and C. Chakraborty, Chem. – Asian J., 2025, 20, e202401460 CrossRef CAS PubMed.
- S. Ruidas, A. Das, S. Kumar, S. Dalapati, U. Manna and A. Bhaumik, Angew. Chem., Int. Ed., 2022, 61, e202210507 CrossRef CAS PubMed.
- Z. M. Wang, Q. Song, C. He, P. Y. Feng, L. Zhao and C. Y. Duan, Chem. Commun., 2024, 60, 4793–4796 RSC.
- R. Cheng, K. Li, Z. Li, M. Jiang, F. Wang, Z. Yang, T. Zhao, P. Meng and C. Fu, J. Power Sources, 2023, 556, 232476 Search PubMed.
- I. Fareed, M. D. Khan, M. Firdous, T. Maqsood, M. U. H. Farooq, M. Tahir, F. K. Butt, J. J. Zou and S. Du, Adv. Sci., 2025, 12, e09902 CrossRef CAS PubMed.
- R. Brandiele, A. Guadagnini, M. Parnigotto, F. Pini, V. Coviello, D. Badocco, P. Pastore, G. A. Rizzi, A. Vittadini, D. Forrer, V. Amendola and C. Durante, J. Energy Chem., 2024, 92, 508 Search PubMed.
- A. K. Pradhan, S. Halder, S. G. Palani and C. Chakraborty, J. Power Sources, 2025, 659, 238425 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |