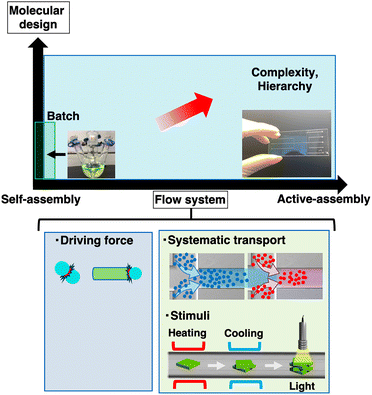
Flow-driven non-covalent synthesis
Munenori
Numata

Department of Biomolecular Chemistry, Graduate School of Life and Environmental Sciences, Kyoto Prefectural University, 1-5, Shimogamo, Sakyo-ku, Kyoto 606-8522, Japan. E-mail: numata@kpu.ac.jp
First published on 10th March 2026
Abstract
Flow systems enable continuous systematic solution transport. Under the concept of space-time regulation, parameters including concentration, mixing ratio, the encounter timing of different molecular components, solvent composition, pH, and the application of external stimuli, such as heat or light, can be precisely controlled in a reproducible manner and multi-step reactions have been realised in covalent synthesis. In non-covalent synthesis, flow systems provide established solution transport while affecting bond formation/dissociation. In this review, I present how flow systems can contribute to non-covalent synthesis, highlighting our recent achievements in this field. I focus on chemistry driven by the macroscopic motion of flow. Finally, I characterise flow-directed non-covalent synthesis by comparing it with associated chemistry fields, such as covalent synthesis, mechanochemistry, and non-equilibrium- and dissipative-assembly systems, and discuss its similarities and discrepancies with these fields. The insights obtained through flow-driven non-covalent synthesis enable us to expand supramolecular structure development and understand chemical events in a motion.
Introduction
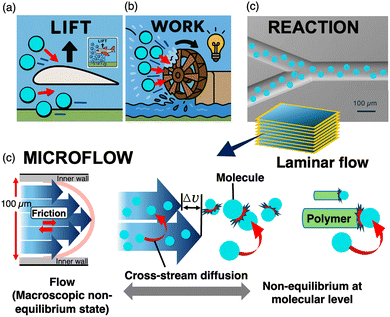
Non-covalent synthesis is widely accepted as a key concept in approaching hierarchically ordered molecular assemblies that possess inner complexities.1,2 In contrast to well-established synthetic organic chemistry, combining a selected number of multiple molecular components in a desired arrangement with control over the entire dimension and size remains challenging. The fundamental issue is the rational design of novel kinetic reaction pathways, coupled with energy consumption. Thus far, we have proposed a device-driven system for controlling complicated kinetic processes in a reproducible manner.3,4 Recent studies have revealed that microflow systems can provide an ideal reaction field for continuous multi-step reactions coupled with external energy consumption.The formal mathematical treatment of flow began in the 17th century with contributions from several key researchers.5 Fluid motion, which is influenced by pressure, gravity, and friction, has been successfully applied to technologies across multiple disciplines. Moreover, Beronoulli's principle states that pressure changes in a fluid drives motion. Under the restriction of the first law of thermodynamics, the energy of the flow can be converted to other types of energy, such as mechanical work, if any appropriate device exists in fluidic motion. For example, air flow on airplane wings generates different flow velocities above and below the wings, resulting in airplane lift (Fig. 1a). Moreover, water flow at the radiated wings on a water wheel revolves to generate electric power (Fig. 1b). In these processes, energy is extracted from fluid motion using an appropriately designed energy conversion system. In a thin channel, flow motion can be converted to molecular motion through viscosity, which might be a useful energy source for chemical conversion, particularly for non-covalent bonds (Fig. 1c).
 | ||
| Fig. 1 Mechanism for harnessing energy from flow: collisions between (a) gas molecules and an airplane wing, (b) water molecules and a water wheel, and (c) molecules through the cross-stream diffusion process in laminar flow. Laminar flow acts as a generator of mechanochemical force. Reproduced from ref. 3 with permission from Oxford University Press, Copyright 2023. | ||
Flow systems have been applied to covalent synthesis as a flowing vessel6–8 and to high-throughput analysis.9–11 For over a decade, flow systems have been used to create various supramolecular assemblies,3,4,12–16 including MOFs,17–19 COFs,20,21 supramolecular polymers,22–24 supramolecular gels,25–28 biomolecules or polymer assemblies29–32 and further higher-ordered hierarchical assemblies,33–35 chiral assemblies,36–39 fabrication on materials,40 and models of the origin of life.38,41 Laminar or turbulent flow can be used depending on the reaction conditions, assembled structures, and their sizes and robustness. In general, laminar flow is suitable for creating divergently ordered supramolecular structures, such as fibres or sheets, on the sub-micron scale, resulting in inner molecular complexity through the combination of different molecular components. Conversely, turbulent flow is effective for supramolecular reactions involving a limited number of molecular components, resulting in convergently closed assemblies, such as cages. Relatively stronger interactions, such as coordination or dynamic covalent bonds, can be facilitated under turbulent flow without dissociating the resulting assemblies. An alternative flow system for non-covalent bond formation uses a compartmentalised continuous flow space, such as droplets. This convective flow in a restricted space facilitates molecular assembly. Since the first study on these combined supramolecular concepts,42 a series of studies have been published.43–46
In general, flow systems have provided new opportunities for systematic solution transport, enabling chemists to precisely control the concentrations, solvent composition, temperature, and moment at which two solutions come into contact (Fig. 2). This approach is useful for both covalent and non-covalent syntheses.
So far, pioneering studies have demonstrated that flow systems can serve as powerful tools in a wide range of areas within supramolecular chemistry. Recently, several excellent reviews have summarized advances in the integration of flow systems with supramolecular assembly.14–16 In this Feature Article, I aim to highlight a different and emerging frontier by demonstrating the further potential of flow systems as non-equilibrium platforms, while also drawing comparisons with related studies in other disciplines.
From self- to active-assembly
Active-assembly through cross-stream diffusion in laminar flow systems
A thin-flowing solution contains only a small amount of fluidic energy. All the molecules gain kinetic energy from flow, which is primarily released as heat. Although fluidic energy appears to have no influence on covalent synthesis, it can be a driving force for the formation of non-covalent bonds. To apply a flow system to non-covalent synthesis, in addition to flow as a systematic mass transport, it is necessary to understand fluidic motion at the molecular scale.Hydrodynamic mechanics demonstrates that laminar flow has dynamic multilayered structures (Fig. 1c).5 Assuming a circular channel cross-section, the motion of a fluid can be described as Hagen–Poiseuille flow, which exhibits a parabolic velocity distribution owing to the friction at the inner wall of the channel. Thus, adjacent layers move at different velocities. If the channel diameter is sufficiently small, the entire solution has a lamellar structure. Unlike batch mixing, all solute molecules can move across different velocity streamlines (cross-stream diffusion) and collide with each other by adding a differential velocity. During cross-stream diffusion followed by molecular collisions, macroscopic flow energy is transferred to the molecules, increasing their kinetic energy and facilitating chemical reactions. Although this effect may be negligible for the rearrangement of covalent bonds, it significantly influences non-covalent bonds. Furthermore, a polymer chain with a high aspect ratio tends to orient along the streamline, indicating restricted motion. Thus, molecules interact with the polymer end via the stream-cross-diffusion mechanism, which differs from conventional self-assembly depending on thermal diffusion. This article defines the present assembly process as active-assembly driven by cross-stream diffusion (Fig. 2). I would like to briefly discuss non-covalent synthesis in flow from the viewpoint of active-assembly in comparison to similar strategies driven by external energy consumption, including dissipative self-assembly, and covalent synthesis such as mechanochemistry.
Active-assembly at the liquid–liquid interface
In 2012, we first demonstrated supramolecular chemistry in a microflow field.47 In particular, we designed Zn-chlorophyll-based amphiphilic molecules featuring dendritic tetra(ethylene glycol) units at position 17 and an isonicotinic acid (Chl-4Py; Fig. 3). A 1,2-dicholoroethane (DCE) solution of Chl-4Py and water was introduced into the Y-type inlet of a double-Y-shaped microflow channel with the expectation that Chl-4Py units would assemble at the water–DCE dynamic interface in the channel. The DCE layer was collected at the Y-type outlet. Although Chl-4Py did not self-assemble in DCE, atomic force microscopy (AFM) images confirmed that unique 1D structures were formed depending on the concentration and flow rate. Thus, laminar flow drove the formation of non-covalent bonds with the assistance of the liquid–liquid interface. However, the resulting assemblies were metastable owing to the loose π–π stacking interactions, which underwent decomposition within an hour after elusion. Re-injection of the solution into the same flow resulted in the spontaneous regeneration of the supramolecular 1D structures. The temporal assembly of less-active Chl-4Py was achieved in a laminar flow environment under the guidance of a dynamic interface.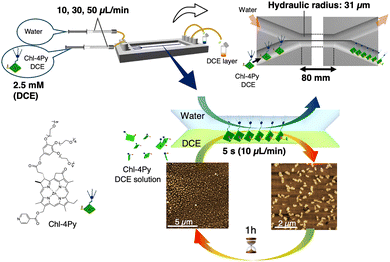 | ||
| Fig. 3 Molecular structure of Chl-4Py and schematic representation of the concept of temporal assembly at the flow interface. AFM images of the assembled structures of Chl-4Py before and after contact with the interface. Reproduced from ref. 47 with permission from Oxford University Press, copyright 2012. | ||
A similar temporal assembly was observed for a water-induced assembly in laminar flow. A THF solution of a p-phenlylenevinilene-based monomer (OPV-1; Fig. 4a) was injected into the central inlet of a cross-type microflow channel, and water was injected into two lateral inlets.48 Although the OPV-1 monomer had no self-assembly ability in a THF/water solvent in a vial, supramolecular polymerisation occurred in laminar flow through π–π stacking interactions to afford its temporal self-assembly in an eluted solution. With changing interactions from π–π stacking to hydrogen-bonds and CH–π interactions, the initial fibrous structure underwent drastic morphological changes and finally decomposed from the termini (Fig. 4b). The creation of metastable supramolecular polymers suggested an active-assembly process in laminar flow. Similar to the temporal assembly of Chl-4Py, the THF/water interface might assist in the creation of primitive assemblies. The dynamic interface in laminar flow had a parabolic velocity profile, enabling active-assembly through the cross-diffusion of OPV-1. To suppress decomposition of the resulting fibres, we designed another p-phenlylenevinilene-based monomer (OPV-2) with a hydrogen-bonding moiety complementary to the original OPV-1 monomer. Supramolecular co-polymerisation of the OPV-1/OPV-2 monomers in laminar flow resulted in the creation of an end-capped supramolecular co-polymer (Fig. 4c).49
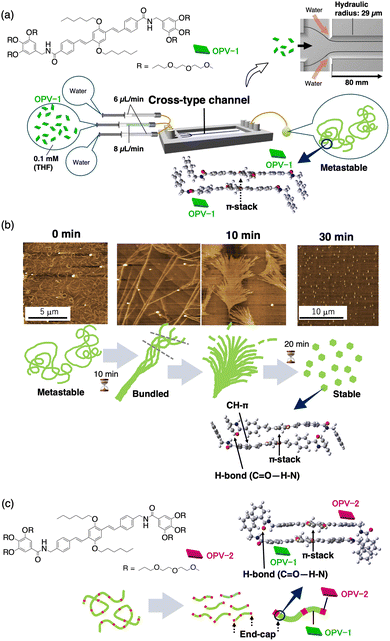 | ||
| Fig. 4 (a) Molecular structure of OPV-1 and illustration of its water-triggered active-assembly in a cross-type microflow channel, and DFT-calculated structures of the OPV-1 dimer in metastable nanofibres. (b) Time-dependent structural changes of metastable nanofibres observed using AFM, and DFT-calculated OPV-1 dimer structure after morphological changes. (c) Molecular structure of OPV-2 and illustration of its co-assembly with OPV-1. Decomposition of the metastable co-assembled nanofibre to form end-capped nanofibres. Reproduced from ref. 48 with permission from Oxford University Press, copyright 2015. | ||
Active-assembly in an aqueous solution
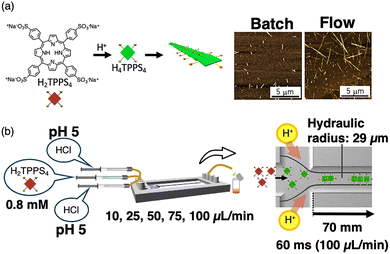
A water-soluble porphyrin, 5,10,15,20-tetrakis(4-sulfonatophenyl)porphyrin (H2TPPS4), was transformed into its active form, H4TPPS4, for self-assembly via protonation of the central pyrrole moiety. H4TPPS4 self-assembles in a slipped fashion through sulfonate–pyrrole interactions to produce a J-stacked supramolecular polymer (Fig. 5a).50,51 Moreover, H2TPPS4 can self-assemble in a batch owing to these strong sulfonate–pyrrole interactions. Thus, we investigated how H2TPPS4 underwent active-assembly in flow. We used double-Y-shaped glass-made microflow channel. The inner As shown in Fig. 5b, HCl was added to the side inlets of the cross-type microflow channel while an aqueous solution of H2TPPS4 was introduced at the centre inlet. UV-Vis spectral studies of the eluted solution revealed that the association efficiency increased compared with that of the batch sample, implying that the number of H4TPPS4 monomers participating in the J-stacks increased under active-assembly. Reflecting the increased interactions, the AFM images revealed that extended J-stack nanofibres were formed (Fig. 5a).52 According to the active-assembly mechanism, the collision frequency between the H2TPPS4 monomers increased in laminar flow. Furthermore, the collision frequency between the protons and H2TPPS4 likely increased with an increasing concentration of H4TPPS4, resulting in subsequent H2TPPS4 monomer collisions.53In situ spectroscopic study revealed that J-aggregate formation was finished within 48 ms. Furthermore, nucleation process finished at much earlier stage (within 2.9 ms). Both nucleation and elongation processes were accelerated comparing to the vial mixing as summarized in Table 1.
| Batch (0.27 mM, pH 5.0) | Flow (0.27 mM, pH 4.6, 30 µL min−1) |
|---|---|
| Ca. 80 min | 48 ms |
Attempt to emerge complexity in molecular arrangements
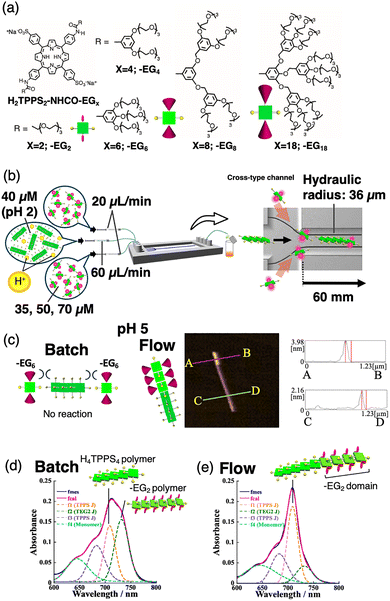
The active-assembly mechanism suggests that molecules encounter greater impacts and higher frequencies than those occurring under thermal diffusion. This encouraged us to create new non-covalent bonds were inaccessible through the conventional self-assembly because of steric bulkiness or different reactivities. We synthesised a series of H2TPPS4 derivatives in which two non-ionic ethylene glycol (EG) units were introduced at opposite meso (trans) positions through amide groups (Fig. 6a; H2TPPS2-NHCO-EGx; x = 2, 4, 6, 8, and 18).54 The reactivity of these porphyrins depended on the bulkiness of the EG units. For example, H2TPPS2-NHCO-EG6 with more EG units exhibited a reduced self-assembly capability owing to steric hindrance. Conversely, H2TPPS2-NHCO-EG2 or -EG4 with lesser steric bulkiness had a higher self-assembly capability, which suppressed its connection to different monomers with a moderate reaction rate. An acidic aqueous solution containing the pre-prepared H2TPPS4 J-stacked nanofibres was injected into the central channel, and H2TPPS2-NHCO-EGx monomers were injected into the lateral inlets such that they met at the cross-point (Fig. 6b). Under active-assembly conditions, less reactive H2TPPS2-NHCO-EG6 monomers were tandemly connected at the end of the parent H4TPPS4 J-stacked supramolecular polymer, yielding a supramolecular diblock co-polymer (Fig. 6c and d).55 Notably, once the H2TPPS2-NHCO-EG6 monomers were connected, the resulting non-covalent bond was stable in the EG6 domain. Using a similar mechanism, highly reactive H2TPPS2-NHCO-EG2 or -EG4 monomers were successfully connected at the end of the H4TPPS4 J-aggregated supramolecular polymer to afford H4TPPS4–H2TPPS2–NHCO–EG2 (or –EG4) bonds, overcoming the relatively faster homo-aggregation between -EG2 (or -EG4) monomers (self-sorting). In both cases, the collision impact and frequency increased at the end of the supramolecular polymer in laminar flow. For H2TPPS2-NHCO-EG6, the formation of the extended block domain was visually confirmed by AFM (Fig. 6c). Statistical analysis of the AFM images revealed that the formation ratio of EG6 diblock co-polymer was approximately 3% in the vial, which increased to 14% under flow conditions.Perylene bisimide (PBI) derivatives have been widely employed as monomers in supramolecular polymerisation.56 In particular, PBI-C3 and PBI-C4 possess peripheral moieties connected to the PBI scaffold through spacers of different lengths. The intramolecular hydrogen bonds at the bisimide edges enable these PBI derivatives to adopt two forms: open and closed. NMR studies have revealed that PBI-C4 is a less active monomer because of its closed form, in which peripheral moieties are present. Therefore, we introduced a mixed solution containing the pre-prepared PBI-C3 supramolecular polymer and the PBI-C4 monomer into a simple straight-type microflow channel. The system produced supramolecular diblock co-polymers, and the PBI-C4 domain grew from the terminus of the polymer facing the forward direction.57 Unlike the case of Y- or cross-type channels, no interface was generated in the channel, highlighting the essential role of laminar flow in active-assembly.
Emergence of geometrical complexity
A glass microflow channel provides an ideal system for coupling an active-assembly with photostimuli owing to its high light penetrability.58 When photo-responsive molecules undergo active-assembly in laminar flow, pinpoint in situ photoirradiation realises the co-assembly of two photo-isomerised monomers. This approach provides an alternative strategy for increasing molecular complexity. Among various photoresponsive molecules, azobenzene derivatives are the most widely investigated. In general, the trans-isomer serves as a reactive monomer in supramolecular polymers, whereas the cis-isomer has less self-assembly ability owing to its non-planar geometry. When a THF solution of trans-azobenzene derivative (trans-Azo-C3NO; Fig. 7a) was introduced into a cross-type channel and mixed with a water at the cross-point, π-stacking and hydrogen-bonding interactions drove 1D assembly to afford fine nanofibres. Irradiation with UV light at 365 nm during the active-assembly of trans-Azo-C3NO induced the in situ conversion from the trans- to cis-isomer in the very primitive aggregate stage (Fig. 7b). Consequently, cis-Azo-C3NO was forced to incorporate into trans-Azo-C3NO nanofibres, resulting in the formation of supramolecular co-polymers. AFM observations and XRD patterns revealed that the resulting supramolecular co-polymers rolled up to yield disk-like structures in the micrometre regime, probably due to the assistance of shear force in laminar flow (Fig. 7c and d). When the eluted solution containing the disc was photo-irradiated at 450 nm, the curvature of the disk was released, resulting in the formation of nanofibres. The morphological changes from disks to nanofibres were irreversible. For example, when the pre-prepared Azo-trans-C3NO nanofibres were irradiated with UV light at 365 nm in a batch, no significant morphological changes were observed. The active-assembly coupled with photo-stimuli in a laminar environment had a pronounced effect on the energy landscape and resulted in a new kinetic pathway.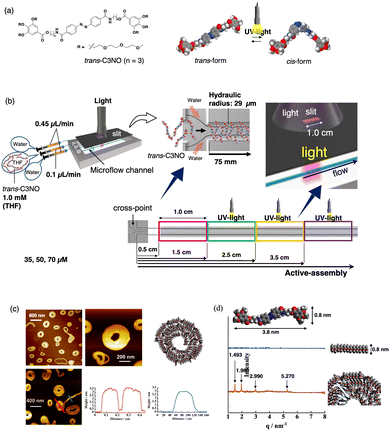 | ||
| Fig. 7 (a) Chemical structure of trans-C3NO, and schematic illustrations of the photo-isomerisation of trans- and cis-C3NO. (b) Illustration of the pinpoint photoirradiation system using a cross-type channel. The position of the 1.0 cm slit was fixed at 0.5, 1.5, 2.5, and 3.5 cm downstream from the cross-point of the microflow channel. (c) AFM images and height profiles of the disk structures prepared upon irradiation at 1.5 cm in the downstream region. (d) XRD patterns of the disk structure (orange line) and supramolecular polymers prepared through a microflow process without light (blue line). Reproduced from ref. 58 with permission from Wiley, copyright 2024. | ||
Active-threading
Successful supramolecular polymerisation using an active-assembly strategy encouraged us to establish active threading systems with a combination of polymer guests and ring-shaped hosts. We employed a well-established polyrotaxane synthesis using poly(ethylene glycol) (PEG) as the guest polymer and cyclodextrins (CDs) as host rings (Fig. 8a).59 According to the developed supramolecular polymerisation system, a mixed aqueous solution containing PEG, CDs, and water was introduced into a microflow channel. As expected, the number of CDs threaded onto PEG drastically increased in laminar flow. Therefore, similar to the supramolecular polymerization mechanism, CDs would collide at the terminus of PEG at a greater frequency than in a batch. Moreover, in the present system, the water phase was indispensable, which generated a CD concentration gradient across the streamline. We confirmed from this finding that the mobility of the CDs across the ends of PEG (cross-stream diffusion) was essential for active threading (Fig. 8b). The AFM images confirmed the formation of extended polyrotaxane nanofibers (Fig. 8c and d). As a reference experiment, the same solution was vigorously mixed in a vial; however, no acceleration effect on threading was observed. Although turbulent flow in a micromixer exhibited a certain acceleration effect, extended polyrotaxane fibers were not observed in the AFM images, indicating that laminar flow is essential for forming long-range ordered structures beyond the molecular scale.60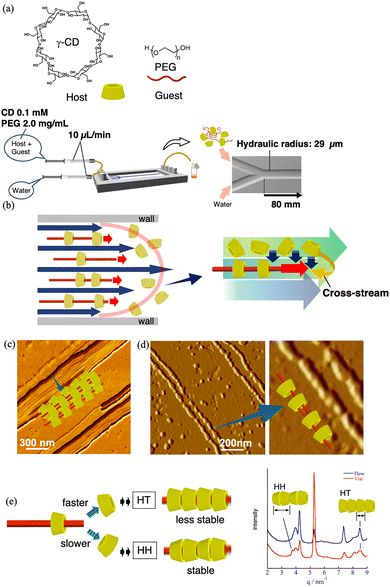 | ||
| Fig. 8 (a) Chemical structures of γ-CD and PEG, and experimental set-up using a cross-type channel. (b) Illustration of the cross-diffusion of molecular rings at the linearly mobbing polymer end in laminar flow. (c and d) AFM images of (c) polyrotaxane microfibres (bundled nanofibers) and (d) polyrotaxane nanofibres. (e) Illustration of the predominant formation of head-to-tail (HT) CD arrangement under active threading, and XRD patterns of the powder samples prepared through batch process (red) and flow (blue). Reproduced from ref. 60 with permission from ACS, copyright 2025. | ||
To investigate the threading rates, dansyl-modified γ-cyclodextrin (Dan-γ-CD) was employed. The guest inclusion phenomenon was evaluated using a parameter expressed as ΔI/I0, where ΔI = I0 − I, and I0 and I are the fluorescence intensities in the absence and presence of the guest, respectively. The ΔI/I0 value was used as an indicator of the inclusion efficiency. The reaction rates and corresponding ΔI/I0 values obtained under batch and flow conditions are summarized in Table 2. The reaction under flow conditions proceeded faster than that in the batch system, although the rate was still slower than the millisecond-order supramolecular polymerization observed in flow.
| Batch | Flow (20 µL min−1, 500 mm channel length) | |
|---|---|---|
| Reaction time | ca. 2 h | 4.8 s |
| Efficiency | ΔI/I0 = 0.011 | ΔI/I0 = 0.165 |
Under active threading, CDs were thread onto PEG under kinetic control. Wide-angle X-ray scattering (XRD) data revealed that active threading affected the orientation of the CDs (Fig. 8e). For example, the head-to-tail (HT) CD orientation became predominant in the resultant polyrotaxane, implying that CDs threaded onto PEG from their wider rims. This result contrasts with polyrotaxane formation under conventional thermodynamic conditions, where head-to-head (HH) and tail-to-tail arrangements are predominantly formed to maximise the hydrogen-bonding interactions between CDs.61 Due to the lack of hydrogen-bonding interactions, the HT polyrotaxane underwent slow dissociation in the eluted solution. Moreover, unlike the HH polyrotaxane, the HT polyrotaxane had a direction. Supramolecular diblock co-polymers obtained through active-assembly are also directional polymers, such as biopolymers. The observed symmetry breaking indicated that active-assembly and threading proceeded under non-equilibrium conditions.
Recently, we demonstrated that capillary tubes can provide a reaction field for polyrotaxane synthesis under active threading.62 An advantage of the capillary flow system is its ability to change the diameter and/or length. Various capillary diameters of 65, 125, 260, 500, and 750 µm were cut into an appropriate length and connected to a cross-type channel device for use as a cross-type capillary microflow channel. The primary parameter of the active-threading was thinness of flow, and a capillary diameter of <260 µm was required to transform the motion of the solution to molecular-level interactions. Thus far, host–guest chemistry has relied on the ‘lock and key’ principle under thermodynamic control. Thus, active threading will expand the repertoire of the combination of host and guest structures that do not fit inside the framework of the ‘lock and key’ principle.
Discussion
Extended one-dimensional supramolecular structures were preferentially formed in the microflow channel. In the present study, the typical hydrodynamic diameter was approximately 30 µm, and the highest flow rate used was 100 µL min−1. Although various solvents were employed, if we assume a water flow in a cylindrical channel, the Reynolds number is estimated to be around 30. This confirms that all experiments were consistently conducted under laminar flow conditions. When the same reaction was carried out under turbulent flow conditions, neither extended supramolecular polymers nor polyrotaxane fibers were obtained, as shown in Fig. 8c. Turbulent flow may facilitate bimolecular reactions such as simple host–guest interactions; however, it suppresses regular supramolecular growth, particularly for structures with high aspect ratios.Self-assembly under non-equilibrium conditions has received considerable attention.63–66 Thus far, chemical funnels and/or light irradiation have generally been used as external energy sources to transform self-assembly precursors into activated forms for self-assembly. To generate non-equilibrium state, the process is accompanied by structural transformation of the precursors. After the equilibrium shifts, non-covalent bond formation occurs via self-assembly without extra energy consumption. Cross-stream diffusion in laminar flow activates molecules toward non-covalent bond formation without any structural conversions. In covalent bond synthesis, heating, light, and a catalyst also activate molecules without any chemical modifications. In both cases, energy is consumed when the molecules collide. In this sense, active-assembly may resemble covalent synthesis. Moreover, since the mechanical friction is converted into molecular kinetic energy in a laminar flow, the energy source might be similar to that of a mechanochemical reaction, which is induced by direct mechanical energy adsorption.67–73 Analogous to covalent synthesis, laminar flow may be regarded as a space catalyst for non-covalent synthesis. Typical examples of space catalysis in covalent synthesis include molecular cages, zeolites, and MOFs.74–77 These are static, regular, and tailored spaces at the atomic scale, While laminar flow is characterised as dynamic space converting mechanical energy to kinetic energy, structurally temporal, and ambiguous; thus, the conversion process does not depend on chemical structures.
In the energy landscape, molecules in active-assembly are in a higher-energy state that is maintained until the solution is eluted from the channel. When passing through the channel, the flow environment supplies energy to the molecules, and active-assembly occurs along the uphill energy landscape (Fig. 9). The energy landscape after elution depends on the stability of the resulting assemblies, which are inherently affected by molecular structures and flow parameters such as flow rates, flow thickness, and residence time. In addition, final solvent composition and pH of the eluted solution affect the landscapes after flow, and three pathways have been identified. If the resultant assemblies are not sufficiently stable in the eluted solution, they undergo decomposition and return to the initial stage (Fig. 9a), indicating that nothing occurs in the flow. In contrast, if the energy barrier for returning to the initial state is sufficiently high, new assemblies that are inaccessible through self-assembly are formed. The active-assembly products can be isolated if the structure is in a kinetically trapped state (Fig. 9b). If the energy barrier toward a lower minimal state is relatively low, the initial structure is metastable and transformed into a more stable structure (Fig. 9c).
 | ||
| Fig. 9 Proposed energy landscape of active-assembly: (a) formation of no assemblies, (b) kinetic assembly, and (c) meta-stable assembly after elution. | ||
Perspective
In flow systems, unlike in batch reactions, we always need to consider the relationship between residence time and reaction time. Non-covalent reactions generally involve much lower energy processes than covalent bond formation, and their reaction rates are faster than those of typical covalent reactions. These characteristics make flow systems particularly promising for non-covalent synthesis. Laminar flow would restrict effective solution mixings. By partially combining turbulent flow, reactions would proceed more efficiently. From a practical standpoint, one of the most critical issues in flow-based non-covalent synthesis is the stability of the products. A general strategy that can be implemented within the same flow process – such as the spontaneous secondary assembly of preliminary products or their fixation through covalent bonding – is required to address this issue. If kinetically unique supramolecular structures could be isolated in a stable form, it would become possible to quantitatively evaluate the supramolecular production efficiency using parameters such as space-time yield (STY) and turnover frequency (TOF). This, in turn, would contribute to scaling up production in a manner analogous to conventional organic synthesis. These findings will also contribute to the more theoretical design of complex supramolecular reaction. At the molecular level, the detailed mechanism remains ambiguous. To fully understand the observed polymer–monomer non-covalent reaction, it is necessary to consider the timescales of polymer dynamics, such as tumbling and coil–stretch transitions. When the reaction rate between the polymer end and the monomer is significantly faster than the timescale of conformational transitions, the transiently aligned and stretched polymer chains can effectively react with monomers at their termini. Further experiments and discussions on the dynamic behavior of polymers under the present flow conditions, combined with hydrodynamic analysis, rheological studies, and simulations, will help clarify this issue.Findings obtained intersection at flow and non-covalent chemistry will correlate to whole solution chemistry in flow. Flow may perturb solvation structures. Changes in solvent polarity or in solubility may influence covalent reactions, and molecular and polymer assembly, although direct experimental verification remains challenging at present. It would also be interesting to investigate how perturbations of solvation structures affect photochemical reactions or analytical sensibility. Biological systems likewise utilize microflow environments. Beyond serving merely as transport pathways, it is worth examining whether such microflow systems actively enhance non-covalent reactions within living systems.
Conclusions
Hydrodynamics has a long history dating back to the 17th century. However, how hydrodynamics intersects with chemistry remains elusive. Understanding of non-covalent interactions under flow could serve as a bridge between these two fields. Herein, I demonstrate that flow can be a practical tool for non-covalent synthesis from the viewpoint of systematic solution transport and mixing systems, which create a desired kinetic pathway, as well as a non-equilibrium system that harnesses fine non-covalent bond energy from macroscopic flow. Because the diffusion of small molecules in laminar flow occurs across streamlines with different velocities, molecular assembly accelerated by differential stream velocity is defined as an active-assembly in flow. Under this mechanism, the number of molecules associated with the assemblies generally increases compared with that in a batch. Even unreacted molecules in the batch are forced to temporally interact under active-assembly to afford metastable assemblies. Only small molecules become active in motion through friction in laminar flow, whereas supramolecular assemblies with lower diffusion coefficients restrict their motion under a shear force. Furthermore, in laminar flow, the molecular collision becomes directional. These contrast with the random non-selective activation mechanism observed in a batch upon heating. For turbulent flow, a similar mechanism would be observed in the local flow space. Thus, the active-assembly mechanism in flow is ubiquitously observed in various flow systems, which might contribute to the acceleration of covalent-bond formation or improve analytical sensitivity. Moreover, molecular recognition in natural fluidic systems, such as blood vessels and primitive molecular assemblies found in thermal vents, might be related to fluidic motion. The present insights obtained through non-covalent synthesis driven in a flow motion will enable us to expand supramolecular structures and understand any chemical events in a motion, such as in a chip of µTAS, life, and geochemical events.Conflicts of interest
There are no conflicts to declare.Data availability
No original experimental data were generated or analyzed during the manuscript preparation. As such, there are no data sets associated with this work.Acknowledgements
The synchrotron radiation experiments were performed at the BL40B2 beamline of SPring-8 with the approval of the Japan Synchrotron Radiation Research Institute (JASRI). The series of study was supported by the ZE Research Program, IAE; the Asahi Glass Foundation and JSPS KAKENHI (grant no. 22K19070, 22H02065 and 23K17951).Notes and references
- L. J. Prins, J. Huskens, F. de Jong, P. Timmerman and D. N. Reinhoudt, Noncovalent synthesis using hydrogen bonding, Nature, 1999, 398, 498 CrossRef.
- G. Vantomme and E. W. Meijer, The construction of supramolecular systems, Science, 2019, 363, 1396 CrossRef PubMed.
- M. Numata and C. Kanzaki, Supramolecular chemistry of a moving solution: flow drives new non-covalent bond formation, Chem. Lett., 2023, 52, 602 CrossRef.
- M. Numata, Supramolecular chemistry in microflow fields: toward a new material world of precise kinetic control, Chem. – Asian J., 2015, 10, 2574 CrossRef PubMed.
- P. K. Panigrahi, Transport Phenomena in Microfluidic Systems, Wiley, Singapore, 2016 Search PubMed.
- Microreactors in Organic Synthesis and Catalysis, ed. T. Wirth, Wiley-VCH, Weinheim, 2008 Search PubMed.
- S. Suga, D. Yamada and J. Yoshida, Cationic three-component coupling involving an optically active enamine derivative. From time integration to space integration of reactions, Chem. Lett., 2010, 39, 404 Search PubMed.
- M. B. Plutschack, B. Pieber, K. Gilmore and P. H. Seeberger, The Hitchhiker's guide to flow chemistry, Chem. Rev., 2017, 117, 11796 Search PubMed.
- M. Goto, K. Sato, A. Murakami, M. Tokeshi and T. Kitamori, Development of a microchip-based bioassay system using cultured cells, Anal. Chem., 2005, 77, 2125 CrossRef PubMed.
- M. Mirasoli, M. Guardigli, E. Michelini and A. Roda, Recent advancements in chemical luminescence-based lab-on-chip and microfluidic platforms for bioanalysis, JPBA, 2014, 87, 36 Search PubMed.
- M. Sonker, V. Sahore and A. T. Woolley, Recent advances in microfluidic sample preparation and separation techniques for molecular biomarker analysis: a critical review, Anal. Chim. Acta, 2017, 986, 1 Search PubMed.
- M. Pumera, Nanomaterials meet microfluidics, Chem. Commun., 2011, 47, 5671 Search PubMed.
- L. O. Kononov, Chemical reactivity and solution structure: on the way to a paradigm shift?, RSC Adv., 2015, 5, 46718 Search PubMed.
- S. Sevim, A. Sorrenti, C. Franco, S. Furukawa, S. Pané, A. J. deMello and J. Puigmartí-Luis, Self-assembled materials and supramolecular chemistry within microfluidic environments: from common thermodynamic states to non-equilibrium structures, Chem. Soc. Rev., 2018, 47, 3788 Search PubMed.
- K. Ollerton, R. L. Greenaway and A. G. Slater, Enabling technology for supramolecular chemistry, Front. Chem., 2021, 9, 774987 CrossRef PubMed.
- L. Guo, Q. Zhu and A. G. Slater, Self-assembly under continuous flow conditions, Chem. Commun., 2025, 61, 10236 RSC.
- A. Carne-Sanchez, I. Imaz, M. Cano-Sarabia and D. Maspoch, A spray-drying strategy for synthesis of nanoscale metal-organic frameworks and their assembly into hollow superstructures, Chem, 2013, 5(3), 203 Search PubMed.
- M. Rubio-Martinez, M. P. Batten, A. Polyzos, K. C. Carey, J. I. Mardel, K. S. Lim and M. R. Hill, Versatile, high quality and scalable continuous flow production of metal-organic frameworks, Sci. Rep., 2014, 4, 5443 CrossRef PubMed.
- M. Taddei, D. A. Steitz, J. A. van Bokhoven and M. Ranocchiari, Continuous-flow microwave synthesis of metal–organic frameworks: a highly efficient method for large-scale production, Chem. – Eur. J., 2016, 22(10), 3245 CrossRef PubMed.
- D. Rodriguez-San-Miguel, A. Abrishamkar, J. A. R. Navarro, R. Rodriguez-Trujillo, D. B. Amabilino, R. Mas-Balleste, F. Zamora and J. Puigmarti-Luis, Crystalline fibres of a covalent organic framework through bottom-up microfluidic synthesis, Chem. Commun., 2016, 52(59), 9212 RSC.
- M. Traxler and W. R. Dichtel, Continuous flow synthesis and post-synthetic conversion of single-crystalline covalent organic frameworks, Chem. Sci., 2024, 15(20), 7545 RSC.
- F. Helmich and E. W. Meijer, Controlled perturbation of the thermodynamic equilibrium by microfluidic separation of porphyrin-based aggregates in a multi-component self-assembling system, Chem. Commun., 2013, 49, 1796 RSC.
- S. Ogi, A. Takamatsu, K. Matsumoto, S. Hasegawa and S. Yamaguchi, Biomimetic design of a robustly stabilized folded state enabling seed-initiated supramolecular polymerization under microfluidic mixing, Angew. Chem., Int. Ed., 2023, 62(34), e202306428 CrossRef PubMed.
- S. Kim, R. Ikebata, Y. Kuramochic, T. Uchihashi and A. Satake, Dynamics of controlled transformation between two states of coordination-organized supramolecular polymers composed of 1,3-Butadiyne-linked Bis-zinc-porphyrin, Coord. Chem. Res., 2025, 2, 100010 Search PubMed.
- W. Chen, Y. Yang, C. Rinadi, D. Zhou and A. Q. Shen, Formation of supramolecular hydrogel microspheres via microfluidic, Lab Chip, 2009, 9(20), 2947 RSC.
- D. Kiriya, M. Ikeda, H. Onoe, M. Takinoue, H. Komatsu, Y. Shimoyama, I. Hamachi and S. Takeuchi, Meter-long and robust supramolecular strands encapsulated in hydrogel jackets, Angew. Chem., Int. Ed., 2012, 51, 1553 CrossRef PubMed.
- Z. Yu, J. Zhang, R. J. Coulston, R. M. Parker, F. Biedermann, X. Liu, O. A. Scherman and C. Abell, Supramolecular hydrogel microcapsules viacucurbit[8]uril host–guest interactions with triggered and UV-controlled molecular permeability, Chem. Sci., 2015, 6, 4929 RSC.
- S. Seibt, S. With, A. Bernet, H.-W. Schmidt and S. Förster, Hydrogelation kinetics measured in a microfluidic device with in situ X-ray and fluorescence detection, Langmuir, 2018, 34, 5535 CrossRef PubMed.
- S. Köster, J. B. Leach, B. Struth, T. Pfohl and J. Y. Wong, Visualization of flow-aligned type I collagen self-assembly in tunable pH gradients, Langmuir, 2007, 23, 357 CrossRef PubMed.
- Z. A. Arnon, A. Vitalis, A. Levin, T. C. T. Michaels, A. Caflisch, T. P. J. Knowles, L. Adler-Abramovich and E. Gazit, Dynamic microfluidic control of supramolecular peptide self-assembly, Nat. Commun., 2016, 7, 13190 CrossRef PubMed.
- A. Kamada, A. Levin, Z. Toprakcioglu, Y. Shen, V. Lutz-Bueno, K. N. Baumann, P. Mohammadi, M. B. Linder, R. Mezzenga and T. P. J. Knowles, Modulating the mechanical performance of macroscale fibers through shear-induced alignment and assembly of protein nanofibrils, Small, 2020, 16, 1904190 CrossRef PubMed.
- A. Mendez-Ardoy, A. Bayón-Fernandez, Z. Yu, C. Abell, J. R. Granja and J. Montenegro, Spatially controlled supramolecular polymerization of peptide nanotubes by microfluidics, Angew. Chem., Int. Ed., 2020, 59, 6902 CrossRef PubMed.
- D. Witters, N. Vergauwe, R. Ameloot, S. Vermeir, D. De Vos, R. Puers, B. Sels and J. Lammertyn, Digital microfluidic high-throughput printing of single metal–organic framework crystals, Adv. Mater., 2012, 24(10), 1316 CrossRef PubMed.
- A. B. Marciel, M. Tanyeri, B. D. Wall, J. D. Tovar, C. M. Schroeder and W. L. Wilson, Concentration-driven assembly and sol–gel transition of π-conjugated oligopeptides, Adv. Mater., 2013, 25, 6398 CrossRef PubMed.
- M. T. Tolley, M. Krishnan, D. Erickson and H. Lipson, Advances towards programmable, matter, Appl. Phys. Lett., 2008, 93, 254105 CrossRef.
- O. Arteaga, A. Canillas, J. Crusats, Z. El-Hachemi, J. Llorens, A. Sorrenti and J. M. Ribo, Flow effects in supramolecular chirality, Isr. J. Chem., 2011, 51, 1007 CrossRef.
- A. Sorrenti, R. Rodriguez-Trujillo, D. B. Amabilino and J. Puigmartı-Luis, Milliseconds make the difference in the far-from-equilibrium self-assembly of supramolecular chiral nano-structures, J. Am. Chem. Soc., 2016, 138(22), 6920 CrossRef PubMed.
- J. Sun, Y. Li, F. Yan, C. Liu, Y. Sang, F. Tian, Q. Feng, P. Duan, L. Zhang, X. Shi, B. Ding and M. Liu, Control over the emerging chirality in supramolecular gels and solutions by chiral microvortices in milliseconds, Nat Commun., 2018, 9, 2599 CrossRef PubMed.
- S. Sevim, A. Sorrenti, J. P. Vale, Z. El-Hachemi, S. Pané, A. D. Flouris, T. S. Mayor and J. Puigmartí-Luis, Chirality transfer from a 3D macro shape to the molecular level by controlling asymmetric secondary flows, Nat. Commun., 2022, 13, 1766 CrossRef PubMed.
- J. Puigmarti-Luis, D. Schaffhauser, B. R. Burg and P. S. Dittrich, A microfluidic approach for the formation of conductive nanowires and hollow hybrid structures, Adv. Mater., 2010, 22(20), 2255–2259 CrossRef PubMed.
- F. M. Mçller, F. Kriegel, M. Kieß, V. Sojo and D. Braun, Steep pH gradients and directed colloid transport in a microfluidic alkaline hydrothermal pore, Angew. Chem., Int. Ed., 2017, 56, 2340 CrossRef PubMed.
- J. Zhang, R. J. Coulston, S. T. Jones, J. Geng, O. A. Scherman and C. Abell, Science, 2012, 335, 690 CrossRef PubMed.
- M. F. Thorne, F. Simkovic and A. G. Slater, Production of mono-disperse polyurea microcapsules using microfluidics, Sci. Rep., 2019, 9(1), 17983 CrossRef PubMed.
- R. Ameloot, F. Vermoortele, W. Vanhove, M. B. J. Roeffaers, B. F. Sels and D. E. De Vos, Interfacial synthesis of hollow metal–organic framework capsules demonstrating selective permeability, Nat. Commun., 2011, 3(5), 382 Search PubMed.
- A. MØndez-Ardoy, A. Bayón-Fernµndez, Z. Yu, C. Abell, J. R. Granja and J. Montenegro, Spatially controlled supramolecular polymerization of peptide nanotubes by microfluidics, Angew. Chem., Int. Ed., 2020, 59, 6902 CrossRef PubMed.
- L. Rao, X. Zhang, P. Liu, W.-T. Dou, H.-Y. Lin, F. Zhao, W. Zhu, X. Qian, H.-B. Yang and L. Xu, Regulating and deciphering the selective synthesis of metallacages in microdroplets, J. Am. Chem. Soc., 2025, 147(35), 31618 CrossRef PubMed.
- M. Numata, M. Takayama, S. Shoji and H. Tamiaki, Microflow-driven temporal self-assembly of amphiphilic molecules, Chem. Lett., 2012, 41, 1689 CrossRef.
- M. Numata, A. Sato and R. Nogami, Energy-dissipative self-assembly driven in microflow: a time-programmed self-organization and decomposition of metastable nanofibers, Chem. Lett., 2015, 44, 995 CrossRef.
- C. Kanzaki, T. Nakadozono and M. Numata, Creation of discrete 1D microstructures: directional dissociation from the ends of a metastable supramolecular polymer, ChemPlusChem, 2020, 85, 74 CrossRef.
- J. M. Ribo, J. Crusats, J.-A. Farrera and M. L. Valero, Aggregation in water solutions of tetrasodium diprotonated meso-tetrakis(4sulfonatophenyl)porphyrin, J. Chem. Soc., Chem. Commun., 1994, 681 RSC.
- Z. El-Hachemi, T. S. Balaban, J. L. Campos, S. Cespedes, J. Crusats, C. Escudero, C. S. Kamma-Lorger, J. Llorens, M. Malfois, G. R. Mitchell, A. P. Tojeira and J. M. Ribo, Effect of hydrodynamic forces on meso-(4-sulfonatophenyl)-substituted porphyrin J-aggregate nanoparticles: elasticity, plasticity and breaking, Chem. – Eur. J., 2016, 22, 9740 CrossRef PubMed.
- M. Numata and R. Sakai, Kinetically controllable supramolecular polymerization through synchronized activation of monomers, Bull. Chem. Soc. Jpn., 2014, 87, 858 CrossRef.
- C. Kanzaki, S. Matoba, A. Inagawa, G. Fukuhara, T. Okada, T. Narushima, H. Okamoto and M. Numata, Linear momentum of a microfluid realizes an anisotropic reaction at the ends of a supramolecular nanofiber, Bull. Chem. Soc. Jpn., 2021, 94, 579 CrossRef.
- C. Kanzaki, H. Yoneda, S. Nomura, T. Maeda and M. Numata, Ionic supramolecular polymerization of water-soluble porphyrins: balancing ionic attraction and steric repulsion to govern stacking, RSC Adv., 2022, 12, 30670 RSC.
- H. Takemori, C. Kanzaki, S. Nomura, T. Maeda and M. Numata, Catalytic effect of microflow space for supramolecular block co-polymerization of water-soluble porphyrins, Chem. Commun., 2024, 60, 7303 RSC.
- S. Ogi, V. Stepanenko, J. Thein and F. Würthner, Impact of alkyl spacer length on aggregation pathways in kinetically controlled supramolecular polymerization, J. Am. Chem. Soc., 2016, 138, 670 CrossRef CAS PubMed.
- S. Matoba, C. Kanzaki, K. Yamashita, T. Kusukawa, G. Fukuhara, T. Okada, T. Narushima, H. Okamoto and M. Numata, Directional s, J. Am. Chem. Soc., 2021, 143, 8731 CrossRef CAS PubMed.
- A. Kaneyoshi, S. Nomura, T. Maeda, T. Kusukawa, Y. Kikkawa and M. Numata, Photostimuli reacha selective intermediate in a microflow: one-shot transformation from a supramolecular co-polymer to a micro-disk structure, ChemSystemsChem, 2024, 6, e202400031 CrossRef CAS.
- A. Harada and M. Kamachi, Complex formation between cyclodextrin and poly(propylene glycol), J. Chem. Soc., Chem. Commun., 1990, 1322 RSC.
- M. Numata, K. Tanaka, A. Asai, M. Matsushita, H. Yoneda, S. Tanaka, T. Fukai, H. Taguchi and Y. Kikkawa, An oriented polymer in a dynamic microsolution pierces molecular rings: an approach toward polyrotaxane synthesis under precise kinetic control, J. Am. Chem. Soc., 2025, 147, 20612 CrossRef CAS PubMed.
- G. Wenz, B.-H. Han and A. Müller, Cyclodextrin rotaxanes and polyrotaxanes, Chem. Rev., 2006, 106, 782 CrossRef CAS PubMed.
- S. Fujiwara, S. Tanaka and M. Numata, Practical polyrotaxane synthesis using capillary tubes based on active-threading mechanism, Chem. Lett., 2025, 54, upaf123 CrossRef CAS.
- A. Sorrenti, J. Leira-Iglesias, A. J. Markvoort, T. F. A. de Greef and T. M. Hermans, Non-equilibrium supramolecular polymerization, Chem. Soc. Rev., 2017, 46, 5476 RSC.
- S. A. P. van Rossum, M. Tena-Solsona, J. H. van Esch, R. Eelkema and J. Boekhoven, Chem. Soc. Rev., 2017, 46, 5519 RSC.
- S. Borsley, D. A. Leigh and B. M. W. Roberts, Molecular ratchets and kinetic asymmetry: giving chemistry direction, Angew. Chem., Int. Ed., 2024, 63, e202400495 Search PubMed.
- I. Hwang, R. D. Mukhopadhyay, P. Dhasaiyan, S. Choi, S.-Y. Kim, Y. H. Ko, K. Baek and K. Kim, Audible sound-controlled spatiotemporal patterns in out-of-equilibrium systems, Nat. Chem., 2020, 12, 808 CrossRef CAS PubMed.
- T. Friščić, Supramolecular concepts and new techniques in mechanochemistry: cocrystals, cages, rotaxanes, open metal–organic frameworks, Chem. Soc. Rev., 2012, 41, 3493 RSC.
- G.-W. Wang, Mechanochemical organic synthesis, Chem. Soc. Rev., 2013, 42, 7668 RSC.
- A. Ikeda, M. Ishikawa, J.-I. Kikuchi and K. Nobusawa, Enantioselective recognition of [60]fullerene bisadducts by cyclodextrin: kinetic control of a mechanochemical high-speed vibration milling process, Chem. Lett., 2013, 42, 1137 CrossRef CAS.
- K. Kubota and H. Ito, Mechanochemical cross-coupling reactions, Trends Chem., 2020, 2, 1066 CrossRef CAS.
- H. Hu, Y. Zhou, B. Xi and Y. Li, Polymer mechanochemistry in confined spaces, Angew. Chem., Int. Ed., 2025, 64, e202417357 Search PubMed.
- K. Wada, K. Yasuzawa, S. Fa, Y. Nagata, K. Kato, S. Ohtani and T. Ogoshi, Diastereoselective rotaxane synthesis with pillar[5]arenes via co-crystallization and solid-state mechanochemical processes, J. Am. Chem. Soc., 2023, 145, 15324 Search PubMed.
- P. A. May and J. S. Moore, Polymer mechanochemistry: techniques to generate molecular force via elongational flows, Chem. Soc. Rev., 2013, 42, 7497 RSC.
- Y. Zhang and W.-Y. Sun, Rational design of organic ligands for metal–organic frameworks as electrocatalysts for CO2 reduction, Chem. Commun., 2024, 60, 8824 RSC.
- H.-f Cui, X.-j Liu, C. Liu, H.-w Zhu, M.-y Liu and R.-t Guo, Recent progress on Cu-based MOF electrocatalysts for CO2 reduction, Fuel, 2026, 403, 136133 Search PubMed.
- T. Li, Y.-T. Chen, X.-B. Zhang, R.-R. Du, L.-N. Ma and Y.-Q. Lan, Asymmetric heterogeneous catalysis using crystalline porous materials, Chem. Soc. Rev., 2025, 54, 5912 RSC.
- J. Li, G. Li and S. C. E. Tsang, Heterogeneous frustrated Lewis pair catalysts: rational structure design and mechanistic elucidation based on intrinsic properties of supports, Acc. Chem. Res., 2025, 58, 555 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |