Open Access Article
Open Access ArticleTuning the viscoelasticity of sugar–silicone polymers using caramelization
Andrea
Heugenhauser
 *ab,
Emily
Lu
b,
Kyle
Faiczak
b,
Daniel
Hrabowyj
b and
Michael A.
Brook
*ab,
Emily
Lu
b,
Kyle
Faiczak
b,
Daniel
Hrabowyj
b and
Michael A.
Brook
 *b
*b
aChemistry and Physics of Materials, Paris-Lodron University Salzburg, Jakob-Haringer Straße 2a, 5020, Salzburg, Austria. E-mail: Andrea.Heugenhauser@plus.ac.at
bDepartment of Chemistry and Chemical Biology, McMaster University, 1280 Main St. W., Hamilton, ON, Canada L8S 4M1. E-mail: mabrook@mcmaster.ca
First published on 10th February 2026
Abstract
Covalent grafting of sugars onto silicones imparts viscoelasticity via hydrogen-bonding among sugar moieties. Catalyst-free caramelization enables programmed, permanent adjustment of these properties by first reducing the OH content on the sugar moieties, which initially lowers viscosity, followed by oligomerization to form viscoelastic materials.
The extremely low surface energy of silicones – manifested in their water repellency – makes them particularly useful in sealants, coatings and adhesives.1 The target viscoelastic properties of the vast majority of silicones destined for these applications are controlled by the introduction of covalent chemical crosslinks. Although some silicone thermoplastic materials are known2–6 that may utilize non-traditional bonding motifs, including ionic and hydrogen bonds,7,8 the vast majority of commercial materials use crosslinks generated by expensive platinum-catalyzed hydrosilylation cure, tin- or titanium-catalyzed moisture cure (room temperature vulcanization, RTV) or high temperature radical cure.9 Catalyst residues can leach from all three types of materials,10,11 and concerns about potential toxicity are particularly elevated in the latter two cases.12
Silicones can also be generated by using organic chemistry to form crosslinks, taking advantage of high atom economy reactions. For example, click reactions based on Huisgen 3+2 cycloadditions,13,14 Diels Alder reactions,15,16 2+2 photoinitiated cycloadditions,17 dynamic Schiff base imine formation,18,19 and aza-Michael reactions20 can be efficient, do not require catalysts, and can be induced at reasonable temperatures. However, it is still necessary to know the target modulus and other viscoelastic properties to encode the requisite crosslink density. Often achieving these targets is, at best, hit and miss and requires many experiments in order to optimize physical properties. It would be desirable to have a process that could, on-the-fly, allow one to tune the viscoelasticity of silicone polymers until the desired properties are achieved.
A very old, delightful process to manipulate viscoelastic properties involves the caramelization of sugar. On heating, sugars, including sucrose and many monosaccharides, undergo conversion in a series of not-perfectly-understood processes: from crystalline granules, to pale yellow, then brown soft malleable materials, to viscoelastic intermediates that become dark brown hard, brittle caramel; and, ultimately black carbon. It is known that dehydration, rearrangement, and polymerization processes occur during this sequence;21 starting from sucrose, the intermediacy of classes of low molecular weight oligomer products including caramelans (C24H36O18), caramelens (C36H50O25), and caramelins (C125H188O80) has been reported.22
We previously demonstrated that glucose-capped linear silicone polymers exhibit the ability to form transient polymer networks under compression due to the interactions of the sugars that dynamically are forced together to create dynamic crosslinks.23 Thus, fluids are readily converted, temporarily, to energy absorbing solids by virtue of these H-bonding interactions. The magnitude of the viscoelastic response is readily tuned by using residues with fewer OH groups,24 diluting the sugars either by increasing the molecular weight of the silicones, or by diluting the sugar–silicone with nonfunctional silicone oil; different sugars will have slightly different caramelization points. We report that caramelization – a catalyst-free process that is readily turned on/off – provides a convenient method to modify viscoelasticity of a given sugar silicone to desired values.
Aminopropylsilicones are traditionally made by the platinum-catalyzed hydrosilylation of allylamine with hydrosiloxane, although a more sustainable approach from less-expensive vinylsilicones and the natural compound cysteamine has been reported.25 Either type of aminosilicone works efficiently. Sugar silicones were made using the simple catalyst-free ring opening reaction of gluconolactone by aminopropylsilicones to form sugar–silicones (Tables S1 and S2 SI).23 Three telechelic and three pendant polymers with different ratios of sugar/silicone served as starting materials (Fig. 1).
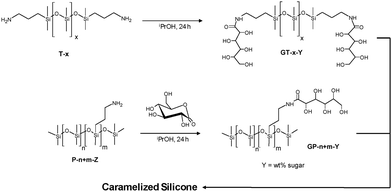 | ||
| Fig. 1 Preparation of sugar–silicones. Z is the weight% of amine monomers; Y is the weight% of amidosugar monomers (Z < Y). | ||
Caramelization is normally undertaken with pure saccharides rather than open-chain sugar–silicones. Therefore, an exploration of the best temperature to use for the caramelization process for these open chain sugars was initially undertaken with the model compound N-butyl gluconamide (BG, control). A balance was struck between the use of higher temperatures to elicit changes rapidly, but not so high such that the reaction would be uncontrollable. The use of 180 °C, ideal for glucose and saccharose caramelization21 (also to make soft caramel, according to cookbooks) was ultimately chosen for caramelization studies of sugar–silicones. The rate of caramelization was readily followed by color changes at 180 °C over different time periods (Fig. 2). The four rows show results from compounds in which the weight% of sugar moieties in the telechelic silicone polymers decreases from top to bottom, inversely correlating with the molar mass of the telechelic silicones, which ranged from 3000 to 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 g mol−1.
000 g mol−1.
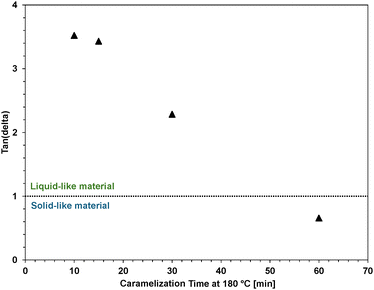
To a degree, the caramelization process could be followed by eye, and common practice was to heat until a dark brown (but not black) color was achieved. Heating beyond this stage led to ‘burnt’ and brittle samples. The time required for caramelization was inversely proportional to the weight% sugar in the starting sugar–silicone polymer. Determining a precise time for caramelization was challenging; colorimeters proved unhelpful in this regard so rheological measurements were employed to assess material transitions. Rheological measurements enabled tracking of the viscoelastic transition from liquid-like to solid-like behavior by measuring the storage and loss moduli (Tables S6–S11, SI) and by calculating tan(δ), representing the ratio of viscous to elastic response. Materials with tan(δ) > 1 are considered liquid-like, while those with tan(δ) < 1 are solid-like. Before caramelization, GT-37-15 was a solid and thus not able to be characterized by tan(δ). However, upon heating for 10 minutes at 180 °C, it transitioned into a soft, malleable viscoelastic fluid with tan(δ) = 3.5. Continued heating resulted in a progressive decrease in tan(δ), reaching 0.7 after 60 minutes, indicative of solid-like material formation (Fig. 3).
GT-334-2 and GT-672-1, which contain lower sugar-to-siloxane ratios than GT-37-15, were already viscoelastic fluids prior to heating. Both exhibited a decrease in tan(δ) upon caramelization (Fig. S11, SI). Notably, despite their lower sugar contents, GT-334-2 and GT-672-1 maintained a higher tan(δ) than GT-37-15 due to their greater viscosities. A similar behavior was observed for the prepared pendant sugar–silicone samples (Table S12, SI). Additional swelling and mobility experiments supporting the viscoelastic behavior a provided in Fig. S1, S2 and Table S5 in the SI. It should be noted that the influence of the silicone molecular weight on the material properties is inherently coupled to the sugar content of the polymers. Lower-molecular-weight telechelic silicones contain a higher proportion of sugar units, whereas higher-molecular-weight polymers contain correspondingly fewer sugar moieties. As a result, the viscoelastic behavior is governed predominantly by the density of hydrogen-bonding sugar groups rather than by the molecular weight of the silicone backbone. This relationship explains why the effect of molecular weight appears less pronounced than expected based on polymer-physics considerations.
The control N-butyl gluconamide showed a weight loss of 1.9% during caramelization, but the sugar–silicones lost less than 1 wt% (Fig. 4). Thus, changes in viscoelastic properties are not a consequence of material loss. Shore OO hardness values were taken for all samples that showed a tan(δ) below 1 after a certain caramelization time (Table S3 and S4, SI).
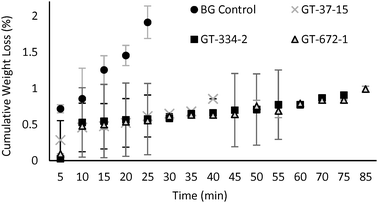 | ||
| Fig. 4 Cumulative weight loss during heating (caramelization) at 180 °C. Error bars represent standard deviation of 3 samples. | ||
DSC (differential scanning calorimetry) measurements were performed on samples before heating and after full caramelization. The GT-37-15, which is a solid at room temperature, exhibited a Tc of 43.2 °C and a transition (likely water evaporation) at ∼100 °C; neither transition was seen after caramelization (heating to 180 °C for 40 min, Fig. S3 SI)†. By contrast, TGA exhibited thermal transitions for GT-37-15 near 200 °C, which reflects the caramelization step and, for all the sugar–silicones, decomposition of the silicone chain near 400 °C. Note that further heating of caramelized samples still results in weight loss at 200 °C as further dehydration/crosslinking occurs to give rigid samples (Table S14, SI).
Anyone who has made caramel will understand the challenges with this system. As the process occurs, any solids initially present first melt; the resulting fluids increase in viscosity, become viscoelastic, then rigid and finally brittle, black solids. These changes challenged physical characterization. For example, adhesion of a sticky caramel to probe surfaces can impact the apparent results; multiple measurements were required. The outcomes when sugar was bound to silicone polymers correlated with sugar concentration and also structure. For example, the high sugar content GT-37-15 is a solid at room temperature, melts with heating to form a lower viscosity fluid and then, with further caramelization, a solid. The other two telechelic polymers, with lower sugar content, started as viscous fluids and ended as soft solids at the caramelization point.
The pendant polymers were all fluids before heating, even with much higher sugar content than the telechelic analogues. This suggests that individual pendant polymers can have sugars that undergo intramolecular, rather than intermolecular crosslinking interactions.
The chemistry of the caramelization process is complex and leads to small molecules that give caramel its typical (pleasant) odors and flavors, including a butterscotch flavor (diacetyl) and a ‘toasty flavor’ (e.g., in bread, maltol). Dehydration, molecular fragmentation and polymerization lead to increases in molecular weight and then crosslinking.26 In the case of glucose C6H12O6 as precursor, oligomers from C24–C125 have been reported.22 In the limit, the full dehydration of sugar, for example using sulfuric acid, leads to carbon, as in the ‘carbon snake demonstration’ often shown to undergraduates.27
The gluconamide residue on the GT and GP silicone polymers behaves as a typical sugar upon heating. Viscosity builds and then viscoelastic materials form. These programmed changes allow one to select the desired precursor viscosity of a sealant or coating, for example, by controlling both molar mass and sugar density. The GP-based polymers we prepared had much higher sugar content than the telechelic polymers, except GT-37-15, which was a solid before heating. Simply heating the powders or fluids, for example using a heat gun, leads in a straightforward manner to elastic materials that should perform in similar fashion to traditional silicones, particularly water repellency. The pendant samples would appear to be the most practical here, as liquid samples are converted in a few minutes to viscoelastic materials with starting viscosity and final Shore hardness (modulus) that correlate with sugar content – it is straightforward to identify the optimal properties. A limitation of the current study is that no long-term aging or storage-stability data are available. Sugar-based materials can undergo time-dependent changes in stiffness due to continued dehydration or crosslinking. Systematic long-term studies will be required to assess whether the caramelized sugar–silicones maintain their mechanical properties over extended periods.
We note that chewing gum based on silicones has been considered as an alternative to hydrocarbon based chewing gums; several patents exist. So a natural question would be, “What do the caramelized silicones taste like?” We don’t know – we are following our own admonition not to eat anything in the lab.
Conceptualization, A. H. and M. A. B.; methodology and investigation E. L., K. F., D. H.; writing M. A. B., A. H. and E. L.; supervision M. A. B. and A. H.; project administration A. H. and M. A. B.; funding acquisition, M. A. B. All authors have read and agreed to the published version of the manuscript.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: full experimental protocols; tables or recipes; details on caramelization, swelling behavior, and mobility experiments; changes in tan(δ) and Shore OO hardness as a function of heating; charts of rheology, TGA, and DSC of telechelic and pendant sugar–silicones before and after caramelization. See DOI: https://doi.org/10.1039/d5cc05934h.References
- W. J. Noll, Chemistry and Technology of Silicones, Academic Press, New York, 1968 Search PubMed.
- A. S. Fawcett and M. A. Brook, Macromolecules, 2014, 47, 1656–1663 CrossRef CAS.
- E. Ogliani, L. Yu, I. Javakhishvili and A. L. Skov, RSC Adv., 2018, 8, 8285–8291 RSC.
- O. Schaefer, A. Kneissl, S. Delica, J. Weis and F. Csellich, Abstr. Pap., Am. Chem. Soc., 2004, 227, U445–U445 Search PubMed.
- M. A. Brook, Chem. Commun., 2023, 59, 12813–12829 Search PubMed.
- R. S. Ward, Thermoplastic Silicone-Urethane Copolymers: A New Class of Biomedical Elastomers, https://www.mddionline.com/materials/thermoplastic-silicone-urethane-copolymers-a-new-class-of-biomedical-elastomers, (accessed accessed June 25, 2025) Search PubMed.
- S. Zheng, Y. Chen and M. A. Brook, Polym. Chem., 2020, 11, 7382–7392 RSC.
- K. Tamim, C. B. Gale, K. E. C. Silverthorne, G. Lu, C. H. Iao and M. A. Brook, ACS Sustainable Chem. Eng., 2023, 11, 7062–7071 Search PubMed.
- M. A. Brook, Silicon in Organic, Organometallic and Polymer Chemistry, Wiley, New York, 2000, ch. 9, pp. 256–308 Search PubMed.
- M. A. Brook, Biomaterials, 2006, 27, 3274–3286 CrossRef CAS PubMed.
- D. Flassbeck, B. Pfleiderer, P. Klemens, K. G. Heumann, E. Eltze and A. V. Hirner, Anal. Bioanal. Chem., 2003, 375, 356–362 Search PubMed.
- I. J. Boyer, Toxicology, 1989, 55, 253–298 CrossRef CAS PubMed.
- M. Pascoal, M. A. Brook, F. Gonzaga and L. Zepeda-Velazquez, Eur. Polym. J., 2015, 69, 429–437 Search PubMed.
- T. Rambarran, F. Gonzaga, A. Fatona, M. Coulson, S. Saem, J. Moran-Mirabal and M. A. Brook, J. Polym. Sci., Part A: Polym. Chem., 2018, 56, 589–597 CrossRef CAS.
- G. Heo, K.-h Pyo, D. H. Lee, Y. Kim and J.-W. Kim, Sci. Rep., 2016, 6, 25358 CrossRef CAS PubMed.
- G. Hu, F. J. LaRonde and M. A. Brook, Silicon Chem., 2002, 1, 215–222 CrossRef.
- A. S. Fawcett, T. C. Hughes, L. Zepeda-Velazquez and M. A. Brook, Macromolecules, 2015, 48, 6499–6507 Search PubMed.
- R. Bui and M. A. Brook, Polymer, 2019, 160, 282–290 CrossRef CAS.
- R. Bui and M. A. Brook, Adv. Funct. Mater., 2020, 30, 2000737 Search PubMed.
- A. Genest, D. Portinha, E. Fleury and F. Ganachaud, Prog. Polym. Sci., 2017, 72, 61–110 Search PubMed.
- A. Golon and N. Kuhnert, J. Agric. Food Chem., 2012, 60, 3266–3274 CrossRef CAS PubMed.
- G. Sengar and H. K. Sharma, J. Food Sci. Technol., 2014, 51, 1686–1696 Search PubMed.
- K. Faiczak, M. A. Brook and A. Feinle, Macromol. Rapid Commun., 2020, 41, 2000161 CrossRef CAS PubMed.
- A. Yepremyan, A. Osamudiamen, M. A. Brook and A. Feinle, Chem. Commun., 2020, 56, 13555–13558 RSC.
- A. Lusterio, M. Melendez-Zamudio and M. A. Brook, Ind. Eng. Chem. Res., 2021, 60, 3830–3838 CrossRef CAS.
- D. D. Miller and C. K. Yeung, Food Chemistry: A Laboratory Manual, Wiley-Interscience, 2nd edn, 2022 Search PubMed.
- A. Helmenstine, Carbon Snake Demo (Sugar and Sulfuric Acid), https://sciencenotes.org/carbon-snake-demo-sugar-and-sulfuric-acid/, (accessed June 26, 2025, 2025).
Footnote |
| † Similar thermal transitions as for GT-37-15 were not observed with GT-64-9, GT-334-2, and GT-672-1 prior to heating; the samples do not contain sufficient sugar content. |
| This journal is © The Royal Society of Chemistry 2026 |