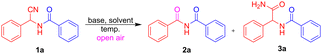Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Base-catalyzed switchable reactivity of N-acyl-α-aminonitriles: oxidative decyanation to imides and hydrolysis to amides in batch and flow conditions
Vikraman Ganesh
Moorthi
a,
Vijay Thavasianandam
Seenivasan
b,
Sharmila
Nokku
a,
Aron Manick
Joel
a,
Wei-Yu
Lin
 *b and
Gopal Chandru
Senadi
*b and
Gopal Chandru
Senadi
 *ab
*ab
aGreen and Sustainable Synthesis Laboratory, Department of Chemistry, College of Engineering and Technology, SRM Institute of Science and Technology, SRM Nagar, Kattankulathur - 603 203, Chengalpattu District, Tamil Nadu, India. E-mail: chandrug@srmist.edu.in
bDepartment of Medicinal and Applied Chemistry, Kaohsiung Medical University, Kaohsiung, Taiwan. E-mail: wylin@kmu.edu.tw
First published on 13th February 2026
Abstract
A base-catalyzed, transition-metal-free synthesis of imides and amides from N-acyl-α-aminonitriles is reported under batch and continuous-flow conditions. Imides are selectively formed in the presence of open air or an O2 atmosphere, while degassed H2O–DMSO conditions afford amides. The protocol shows good scalability and broad functional-group tolerance, and enables late-stage modification of pharmaceutically relevant, ibuprofen-derived substrates; mechanistic studies suggest oxidative decyanation and nitrile hydrolysis pathways for imides and amides, respectively.
The imide functional group is a key structural motif prevalent in numerous natural products and widely exploited in pharmaceuticals1a,b and materials science.1c Representative examples include penimide A, a fungitoxic natural product,2a berkeleyamide B,2b and approved drugs such as dermagen (a skin-care agent),2c fumaramidmycin (an antibiotic),2d and aniracetam (a cognitive enhancer),2e all defined by the imide scaffold (Fig. 1). Its broad utility in bioactive molecules and materials continues to drive the development of efficient and practical synthetic strategies for imide scaffolds.3
Classical approaches to imide synthesis primarily include the Mumm rearrangement, which involves a 1,3-(O–N) acyl transfer from an acyl imidate to an imide (Scheme 1a),4 and the direct acylation of amides with acid chlorides, anhydrides, or isocyanates (Scheme 1b).5 While effective, these strategies often rely on toxic reagents and generate significant chemical waste. To address these limitations, Bode and co-workers developed a chemoselective acylation of amides with potassium acyltrifluoroborates (KATs) under aqueous acidic conditions.6 In addition, a wide range of modern strategies for imide formation have emerged, including palladium-catalyzed aminocarbonylations, dicarbonylation of amines, hydroamidocarbonylation of olefins, and radical oxidative carbonylations (Scheme 1c),7 as well as oxidative cross-coupling (Scheme 1d),8 α-oxygenation (Scheme 1e),9 isocyanide-based multi-component reactions,10 visible-light and electrochemical approaches,11,12 and a few other notable methods.13 Despite these advances, many of these methodologies still depend on transition-metals, stoichiometric oxidants, or complex setups, which can restrict their practicality and broader synthetic applicability. This highlights the continued need for efficient, sustainable, and operationally simple approaches for imide synthesis. The motivation for this work arose from studies on the base-promoted Nef reaction (Scheme 1f)14 and the oxidative amidation of α-substituted malonitriles (Scheme 1g)15 using molecular oxygen, along with our ongoing investigations on α-aminonitriles.16a–c Herein, we report a base-catalyzed, condition-switchable strategy that enables oxidative imidation via decyanation or amide formation via nitrile hydrolysis under both batch and continuous-flow conditions (Schemes 1h and 1i).
Initial screening of model substrate 1a with Et3N delivered 2a, albeit in only 15% yield, whereas DBU gave a markedly improved 83% yield (Table 1, entries 1 and 2). To further enhance the efficiency, a range of bases was evaluated and Cs2CO3 emerged as the most effective, affording 2a in 87% yield, while DIPEA, DMAN, K2CO3, Na2CO3 and NaHCO3 were less competent (Table 1, entries 3–8). Examination of the solvent effects revealed that EtOH, 1,4-dioxane and ACN were inferior to DMSO (Table 1, entries 9–11). However, using H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (8
DMSO (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) (Table 1, entry 12), as the solvent, the expected product 2a was obtained in 57% yield, along with the nitrile-hydrolyzed product 3a in 25% yield. To improve the selectivity toward 3a, the reaction conditions were slightly modified. The mixture was degassed under N2 and then sealed (Table 1, entry 13), resulting in the selective formation of 3a in 78% yield, and the unreacted starting material was recovered. Variation of the catalyst loading showed that lowering Cs2CO3 reduced the yield, whereas increasing it to 20 mol% provided no additional benefit (Table 1, entries 14 and 15). Temperature optimization established 100 °C as optimal, with diminished efficiency at 80 °C and no improvement at 120 °C (Table 1, entries 16 and 17). Variation of the reaction atmosphere revealed the crucial role of air, as the yield dropped to 23% under Ar and showed no significant change under O2 (Table 1, entries 18 and 19). Taken together, these studies defined the optimal conditions as 10 mol% Cs2CO3 in DMSO (0.15 M) at 100 °C under open air, which were then employed in subsequent scope studies for imides.
2) (Table 1, entry 12), as the solvent, the expected product 2a was obtained in 57% yield, along with the nitrile-hydrolyzed product 3a in 25% yield. To improve the selectivity toward 3a, the reaction conditions were slightly modified. The mixture was degassed under N2 and then sealed (Table 1, entry 13), resulting in the selective formation of 3a in 78% yield, and the unreacted starting material was recovered. Variation of the catalyst loading showed that lowering Cs2CO3 reduced the yield, whereas increasing it to 20 mol% provided no additional benefit (Table 1, entries 14 and 15). Temperature optimization established 100 °C as optimal, with diminished efficiency at 80 °C and no improvement at 120 °C (Table 1, entries 16 and 17). Variation of the reaction atmosphere revealed the crucial role of air, as the yield dropped to 23% under Ar and showed no significant change under O2 (Table 1, entries 18 and 19). Taken together, these studies defined the optimal conditions as 10 mol% Cs2CO3 in DMSO (0.15 M) at 100 °C under open air, which were then employed in subsequent scope studies for imides.
| Entry | Base (x mol%) | Solvent | Temp. (°C) | Yieldb (%) (2a/3a) |
|---|---|---|---|---|
| a Reaction conditions: All reactions were carried out using 1a (0.5 mmol), base (x, mol%), and solvent (0.15 M) in a vial at the indicated temperature for 11 h under open air. b Isolated yield. c N2 degassed, then sealed. d Yield is based on recovered starting material. e Ar atmosphere. f O2 atmosphere. Abbreviations: DBU (1,8-diazabicyclo[5.4.0]undec-7-ene). DIPEA (N,N-diisopropylethylamine). DMAN (1,8-bis(dimethylamino)naphthalene). DMSO (dimethyl sulfoxide). EtOH (ethanol). ACN (acetonitrile). N.R. (no reaction). | ||||
| 1 | Et3N (10) | DMSO | 100 | 15/0 |
| 2 | DBU (10) | DMSO | 100 | 83/0 |
| 3 | DIPEA (10) | DMSO | 100 | 23/0 |
| 4 | DMAN (10) | DMSO | 100 | 56/0 |
| 5 | K2CO3 (10) | DMSO | 100 | 72/0 |
| 6 | Na2CO3 (10) | DMSO | 100 | 76/0 |
| 7 | NaHCO3 (10) | DMSO | 100 | 85/0 |
| 8 | Cs 2 CO 3 (10) | DMSO | 100 | 87/0 |
| 9 | Cs2CO3 (10) | EtOH | 75 | N.R. |
| 10 | Cs2CO3 (10) | 1,4-Dioxane | 100 | 21/0 |
| 11 | Cs2CO3 (10) | ACN | 80 | 16/0 |
| 12 | Cs2CO3 (10) | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (8 DMSO (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
100 | 57/25 |
| 13c | Cs2CO3 (10) | H2O![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMSO (8 DMSO (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
100 | 0/78d |
| 14 | Cs2CO3 (20) | DMSO | 100 | 83/0 |
| 15 | Cs2CO3 (05) | DMSO | 100 | 68/0 |
| 16 | Cs2CO3 (10) | DMSO | 80 | 64/0 |
| 17 | Cs2CO3 (10) | DMSO | 120 | 79/0 |
| 18e | Cs2CO3 (10) | DMSO | 100 | 23/0 |
| 19f | Cs2CO3 (10) | DMSO | 100 | 85/0 |
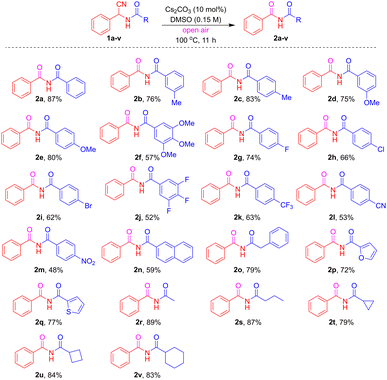
The synthetic scope of the N-acyl-α-aminonitrile framework was comprehensively investigated (Table 2) by systematically varying the substituents, demonstrating broad applicability for oxidative decyanation reactions. Initial exploration focused on the phenyl group, revealing that electron-donating substituents such as 3-Me (1b), 4-Me (1c), 3-MeO (1d), 4-MeO (1e), and 3,4,5-tri-MeO (1f) successfully afforded the desired imide products 2b–2f in good to excellent yields, ranging from 57% to 83%. Subsequently, halogen-substituted derivatives, including 4-F (1g), 4-Cl (1h), 4-Br (1i), and 3,4,5-tri-F (1j), also yielded the corresponding imides 2g–2j with satisfactory yields (52% to 74%). The methodology was further extended to electron-withdrawing groups such as 4-CF3 (1k), 4-CN (1l), and 4-NO2 (1m), which underwent oxidative decyanation to produce 2k–2m in 48% to 63% yields under standard conditions. To enhance the versatility of the reaction, diverse aromatic and heteroaromatic systems including 2-naphthyl (1n), benzyl (1o), 2-furyl (1p), and 2-thiophenyl (1q) were successfully converted into their respective imides 2n–2q, with yields ranging from 59% to 79%. Remarkably, the scope of the R-functionality was extensively evaluated with various linear and cyclic aliphatic derivatives, such as methyl (1r), n-propyl (1s), cyclopropyl (1t), cyclobutyl (1u), and cyclohexyl (1v), all of which afforded the desired compounds 2r–2v in good to excellent yields, specifically 79% to 89%. Notably, aliphatic derivatives generally demonstrated superior yields compared to their aromatic and heteroaromatic counterparts. Similarly, to extend the scope, the reaction conditions described in Table 1, entry 13, were tested for other representative derivatives (1a, 1b, 1d, 1l and 1o) to afford the nitrile hydrolysis products amides (see the SI, 2.5).
Enhanced mass and heat transfer in a microtubing flow reactor17 significantly accelerated the reaction; however, under flow conditions using catalytic Cs2CO3 in H2O–DMSO (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) in the presence of O2, a mixture of imide 2a and amide 3a was obtained in 25% and 37% yield, respectively (see the SI, 2.6). The formation of the amide is likely attributable to the presence of water used to dissolve Cs2CO3. To suppress amide formation, the next best conditions identified in Table 1 (entry 2) were employed, affording the desired imide 2a in 87% yield within a residence time of 1.27 min. The protocol was extended to substrates bearing a 4-Me substituent (1c) and a 4-Cl substituent (1h), delivering the corresponding imides in 65% and 51% yield, respectively (see the SI, 2.7). Unreacted starting material was recovered, and yields are reported based on recovered starting material; further increases in residence time were not explored. Furthermore, to improve the selectivity toward amide 3a, a systematic optimization in the absence of O2 (see the SI, Table S1) delivered amide 3a in 76% yield using flow rates (F1 = 20 µL min−1, F2 = 40 µL min−1) at 90 °C with a residence time of 20.9 min. The scope was further extended to electron-donating, halogen-substituted, electron-withdrawing, benzyl and heteroaryl substrates and all of them gave better yields in shorter reaction times as compared to the batch amide synthesis (Scheme 2).
2) in the presence of O2, a mixture of imide 2a and amide 3a was obtained in 25% and 37% yield, respectively (see the SI, 2.6). The formation of the amide is likely attributable to the presence of water used to dissolve Cs2CO3. To suppress amide formation, the next best conditions identified in Table 1 (entry 2) were employed, affording the desired imide 2a in 87% yield within a residence time of 1.27 min. The protocol was extended to substrates bearing a 4-Me substituent (1c) and a 4-Cl substituent (1h), delivering the corresponding imides in 65% and 51% yield, respectively (see the SI, 2.7). Unreacted starting material was recovered, and yields are reported based on recovered starting material; further increases in residence time were not explored. Furthermore, to improve the selectivity toward amide 3a, a systematic optimization in the absence of O2 (see the SI, Table S1) delivered amide 3a in 76% yield using flow rates (F1 = 20 µL min−1, F2 = 40 µL min−1) at 90 °C with a residence time of 20.9 min. The scope was further extended to electron-donating, halogen-substituted, electron-withdrawing, benzyl and heteroaryl substrates and all of them gave better yields in shorter reaction times as compared to the batch amide synthesis (Scheme 2).
To demonstrate scalability, gram-scale reactions were performed under the optimized conditions (Scheme 3a). The model substrate 1a (1.0 g) afforded imide 2a in 82% yield, while aliphatic (1s to 2s, 85%) and heteroaryl (1q to 2q, 72%) substrates also delivered good yields. Late-stage modification of a pharmaceutically relevant, ibuprofen-derived substrate furnished imide (1w to 2w) in 73% yield, further demonstrating the robustness of the protocol (Scheme 3b). Mechanistic studies revealed no product in the absence of base (Scheme 3c), and product suppression with TEMPO and TEMPOL suggested a radical pathway (Scheme 3d). H218O-labeling experiments were conducted to probe oxygen incorporation in products 2a (batch) and 3a (flow). LC–MS analysis revealed no 18O incorporation in imide 2a (Scheme 3e), whereas clear incorporation of 18O was observed in the amide product 3a (Scheme 3f) (see the SI, 10).
Furthermore, to verify the involvement of superoxide anion radicals in the formation of acyclic imides, we carried out room-temperature EPR studies (X-band, 9.65 GHz) using DMPO (0.1 mol L−1) as a spin trap (see the SI, Fig. S4). Spectra were recorded under three conditions: (i) DMPO in DMSO, (ii) DMPO with substrate 1a and base in DMSO, and (iii) DMPO with substrate 1a in DMSO. In the stacked spectra, the signal marked (*) corresponds to DMPO, while the feature labeled (Φ) arises from its oxidized adduct, consistent with the trapping of superoxide species. The characteristic hyperfine pattern together with a g value of 2.0035 provides strong evidence for the generation of superoxide under the reaction conditions.18
Based on control experiments, a plausible reaction mechanism is proposed (Scheme 4).16 Under basic conditions, deprotonation of N-acyl α-aminonitrile 1 generates the amidyl anion A, which undergoes a rapid [1,2]-H shift to afford the carbanion B. The latter engages in a single-electron transfer (SET) with molecular oxygen to produce radical intermediate C and a superoxide radical anion. Coupling of C with the superoxide species forms the peroxy intermediate D, which undergoes intramolecular cyclization to give the imino-dioxetane E. Proton transfer from E furnishes intermediate F, and subsequent C–C bond cleavage releases isocyanic acid G, affording the acyclic imide 2. On the other hand, the α-carbanion B undergoes delocalization into the nitrile group, giving a resonance-stabilized ketenimine intermediate II, which reacts with water to afford the iminol species III. Subsequent enol–keto tautomerization of III furnishes the corresponding amide product 3 (see the SI, Scheme S1).
In summary, a base-catalyzed, condition-switchable protocol enables the transformation of N-acyl-α-aminonitriles into imides or amides under both batch and continuous-flow conditions. Flow processing allows access to both product classes with significantly shorter residence times compared to batch reactions. Mechanistic investigations indicate that the carbonyl oxygen in the amide product originates from water, as demonstrated by H218O-labeling experiments, while radical inhibition and EPR studies support a radical pathway involving superoxide species for imide formation. The practicality of the method is demonstrated through gram-scale synthesis and late-stage modification of an ibuprofen-derived substrate. Further studies aimed at expanding the synthetic utility of N-acyl-α-aminonitriles are ongoing.
V. G. M., S. N., and A. M. J., thank the SRM Institute of Science and Technology for the PhD fellowship. Dr G. C. S. acknowledges Anusandhan National Research Foundation (ANRF) for the Core Research Grant (File No. CRG/2022/006963). The authors thank IIISM, SRM Institute of Science and Technology, for providing an NMR facility. The authors also thank the Department of Chemistry, SRM Institute of Science and Technology, for providing the HRMS facilities. The authors also thank the Centre for Research Resources and Development, Kaohsiung Medical University, for 400 MHz NMR and LC–MS analyses. The authors further acknowledge the Academia Sinica Small Molecule Mass Spectrometry Facility for mass spectrometric analyses.
Conflicts of interest
There are no conflicts to declare.Data availability
The supporting data have been provided as part of the supplementary information (SI). Supplementary information: Table S1, Scheme S1, and Fig. S1–S145. NMR spectra and further experimental details. See DOI: https://doi.org/10.1039/d5cc05918f.References
- (a) L. Raj, T. Ide, A. U. Gurkar, M. Foley, M. Schenone, X. Li, N. J. Tolliday, T. R. Golub, S. A. Carr, A. F. Shamji, A. M. Stern, A. Mandinova, S. L. Schreiber and S. W. Lee, Nature, 2011, 475, 231–234 CrossRef CAS PubMed; (b) M. Prudhomme, Eur. J. Med. Chem., 2003, 38, 123–140 CrossRef CAS PubMed; (c) Z. Cheng, X. Zhu, E. T. Kang and K. G. Neoh, Macromolecules, 2006, 39, 1660–1663 CrossRef CAS.
- (a) T. Pacher, A. Raninger, E. Lorbeer, L. Brecker, P. P.-H. But and H. Greger, J. Nat. Prod., 2010, 73, 1389–1393 CrossRef CAS PubMed; (b) A. A. Stierle, D. B. Stierle and B. Patacini, J. Nat. Prod., 2008, 71, 856–860 CrossRef CAS PubMed; (c) D. Vacher, Ann. Pharm. Fr., 2005, 63, 207–210 CrossRef CAS PubMed; (d) H. B. Maruyama, Y. Suhara, J. S. Watanabe, Y. Maeshima, N. Shimizu, M. O. Hamada, H. Fujimoto and K. Takano, J. Antibiot., 1975, 28, 636–647 CrossRef CAS; (e) K. Nakamura, CNS Drug Rev., 2006, 8, 70–89 Search PubMed.
- J. Sperry, Synthesis, 2011, 3569–3580 CrossRef CAS.
- O. Mumm, H. Hesse and H. Volquartz, Chem. Ber., 1915, 48, 379–391 CrossRef CAS.
- (a) Q. E. Thompson, J. Am. Chem. Soc., 1951, 73, 5841–5846 CrossRef CAS; (b) C. D. Hurd and A. G. Prapas, J. Org. Chem., 1959, 24, 388–392 CrossRef CAS.
- A. O. Gálvez, C. P. Schaack, H. Noda and J. W. Bode, J. Am. Chem. Soc., 2017, 139, 1826–1829 CrossRef.
- (a) A. Schnyder and A. F. Indolese, J. Org. Chem., 2002, 67, 594–597 CrossRef CAS; (b) H. Li, K. Dong, H. Neumann and M. Beller, Angew. Chem., Int. Ed., 2015, 127, 10377–10381 CrossRef; (c) H. Li, X. Fang, R. Jackstell, H. Neumann and M. Beller, Chem. Commun., 2016, 52, 7142–7145 Search PubMed; (d) C.-S. Kuai, L.-C. Wang, J.-X. Xu and X.-F. Wu, Org. Lett., 2022, 24, 451–456 Search PubMed; (e) L. Lu, D. Cheng, Y. Zhan, R. Shi, C.-W. Chiang and A. Lei, Chem. Commun., 2017, 53, 6852–6855 RSC.
- (a) H. Jiang, A. Lin, C. Zhu and Y. Cheng, Chem. Commun., 2013, 49, 819–821 RSC; (b) J. Wang, C. Liu, J. Yuan and A. Lei, Chem. Commun., 2014, 50, 4736–4739 RSC; (c) N. Xu, J. Liu, D. Li and L. Wang, Org. Biomol. Chem., 2016, 14, 4749–4757 RSC; (d) H. Aruri, U. Singh, S. Kumar, M. Kushwaha, A. P. Gupta, R. A. Vishwakarma and P. P. Singh, Org. Lett., 2016, 18, 3638–3641 CrossRef CAS.
- (a) L. Xu, S. Zhang and M. L. Trudell, Chem. Commun., 2004, 1668–1669 RSC; (b) K. C. Nicolaou and C. J. N. Mathison, Angew. Chem., Int. Ed., 2005, 44, 5992–5997 CrossRef CAS; (c) Z. Jin, B. Xu and G. B. Hammond, Tetrahedron Lett., 2011, 52, 1956–1959 CrossRef CAS; (d) Y. Zhang, L. Pan, Y. Zou, X. Xu and Q. Liu, Chem. Commun., 2014, 50, 14334–14337 RSC; (e) H. Yu, Y. Chen and Y. Zhang, Chin. J. Chem., 2015, 33, 531–534 Search PubMed; (f) C. Mei, Y. Hu and W. Lu, Synthesis, 2018, 2999–3005 CAS; (g) H.-H. Chang, X.-X. He, Z.-L. Zang, C.-H. Zhou and G.-X. Cai, Asian J. Org. Chem., 2022, 11, e202200500 CrossRef CAS; (h) N. Neerathilingam and R. Anandan, RSC Adv., 2022, 12, 8368–8373 RSC; (i) S.-H. Liu, Z.-C. Dong, Z.-L. Zang, C.-H. Zhou and G.-X. Cai, Org. Biomol. Chem., 2024, 22, 1205–1212 RSC; (j) C. Gu, T. Yatabe, K. Yamaguchi and K. Suzuki, Chem. Commun., 2024, 60, 4906–4909 RSC; (k) J. Qi, X. Wang, G. Wang, S. R. Dubbaka, P. O'Neill, H. T. Ang and J. Wu, Green Chem., 2024, 26, 306–311 RSC.
- (a) X. Ye, C. Xie, Y. Pan, L. Han and T. Xie, Org. Lett., 2010, 12, 4240–4243 CrossRef CAS; (b) T. Brandhofer, A. Gini, S. Stockerl, D. G. Piekarski and O. G. Mancheno, J. Org. Chem., 2019, 84, 12992–13002 CrossRef CAS PubMed; (c) B. Wang, D. He, B. Ren and T. Yao, Chem. Commun., 2020, 56, 900–903 RSC; (d) T.-T. Xu, Y. Xiao, J.-L. Zhou, L. Tang, F. Wu, J.-Y.-H. Sheng, W.-B. Wu and J.-J. Feng, Org. Chem. Front., 2023, 10, 4381–4387 RSC.
- (a) Y.-Y. Cheng, T. Lei, L. Su, X. Fan, B. Chen, C.-H. Tung and L.-Z. Wu, Org. Lett., 2019, 21, 8789–8794 CrossRef CAS PubMed; (b) B.-G. Cai, W.-Z. Yao, L. Li and J. Xuan, Org. Lett, 2022, 24, 6647–6652 CrossRef CAS PubMed; (c) R. Ma, Y. Ren, Z. Deng, K.-H. Wang, J. Wang, D. Huang, X. Lv and Y. Hu, Org. Lett., 2023, 25, 4080–4085 CrossRef CAS PubMed; (d) M. Hitt, A. Norris and A. N. Vedernikov, Org. Lett., 2023, 25, 5504–5508 CrossRef CAS PubMed.
- (a) X. Zhang, T. Cui, X. Zhao, P. Liu and P. Sun, Angew. Chem., Int. Ed., 2019, 132, 3493–3497 CrossRef; (b) Q. Chu, Z. Feng, S. Zhang, P. Liu and P. Sun, Green Chem., 2023, 25, 6728–6732 RSC; (c) W. Zhou, P. Chen, Z.-Q. Li, L.-T. Xiao, J. Bai, X.-R. Song, M.-J. Luo and Q. Xiao, Org. Lett., 2023, 25, 6919–6924 CrossRef CAS PubMed; (d) P. Jiang, C. Liang, T. He, R. Liu, X. Meng, Y. Zheng and S. Huang, Eur. J. Org. Chem., 2024, e202400469 CrossRef CAS.
- (a) D. A. Evans, P. Nagorny and R. Xu, Org. Lett., 2006, 8, 5669–5671 CrossRef CAS PubMed; (b) T. Tomizawa, K. Orimoto, T. Niwa and M. Nakada, Org. Lett., 2012, 14, 6294–6297 CrossRef CAS PubMed; (c) Y. Shen, Q. Li, G. Xu and S. Cui, Org. Lett., 2018, 20, 5194–5197 CrossRef CAS PubMed; (d) J.-L. Xu, H. Tian, J.-H. Kang, W.-X. Kang, W. Sun, R. Sun, Y.-M. Li and M. Sun, Org. Lett., 2020, 22, 6739–6743 CrossRef CAS PubMed; (e) X. Du, D. Xu, G. Xu, C. Yu and X. Jiang, Org. Lett., 2022, 24, 7323–7327 CrossRef CAS PubMed.
- (a) R. Ballini, G. Bosica, D. Fiorini and M. Petrini, Tetrahedron Lett., 2002, 43, 5233–5235 CrossRef CAS; (b) S. Umemiya, K. Nishino, I. Sato and Y. Hayashi, Chem. – Eur. J., 2014, 20, 15753–15759 CrossRef CAS PubMed.
- J. Li, M. J. Lear and Y. Hayashi, Angew. Chem., Int. Ed., 2016, 55, 9060–9064 CrossRef CAS PubMed.
- (a) S. Swetha and G. C. Senadi, Adv. Synth. Catal., 2022, 364, 2872–2882 CrossRef CAS; (b) S. Sathyendran, K. Muthu, K. Govindan, N.-Q. Chen, W.-Y. Lin and G. C. Senadi, Org. Lett., 2023, 25, 4086–4091 CrossRef CAS PubMed; (c) S. Sathyendran and G. C. Senadi, Asian J. Org. Chem., 2023, 12, e202300433 CrossRef CAS; (d) J.-P. Leblanc and H. W. Gibson, J. Org. Chem., 1994, 59, 1072–1077 CrossRef CAS.
- L. Capaldo, Z. Wen and T. Noël, Chem. Sci., 2023, 14, 4230–4247 RSC.
- W. Li, S. Li, L. Luo, Y. Ge, J. Xu, X. Zheng, M. Yuan, R. Li, H. Chen and H. Fu, Green Chem., 2021, 23, 3649–3655 RSC.
| This journal is © The Royal Society of Chemistry 2026 |