Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Diverse hydrogen chemistry with perspectives for energy storage
Colin J.
Webb
 a,
Terry D.
Humphries
a,
Terry D.
Humphries
 b,
Joseph A.
Teprovich
b,
Joseph A.
Teprovich
 c,
Marek
Polanski
d,
Michael
Heere
e,
Hai-Wen
Li
f,
Michael
Felderhoff
c,
Marek
Polanski
d,
Michael
Heere
e,
Hai-Wen
Li
f,
Michael
Felderhoff
 g,
Yaroslav
Filinchuk
h,
Martin
Dornheim
g,
Yaroslav
Filinchuk
h,
Martin
Dornheim
 *i,
Mark
Paskevicius
*i,
Mark
Paskevicius
 b,
Craig E.
Buckley
b,
Craig E.
Buckley
 b and
Torben R.
Jensen
b and
Torben R.
Jensen
 *j
*j
aSchool of Environment and Science, Griffith University, Brisbane, Australia
bPhysics and Astronomy, School of Electrical Engineering, Computing and Mathematical Sciences, Curtin University, GPO Box U1987, Perth, WA 6845, Australia
cDepartment of Chemistry & Biochemistry, California State University Northridge, Northridge, CA 91330, USA
dFaculty of New Technologies and Chemistry, Military University of Technology, Kaliskiego 2 Str., 00-908 Warsaw, Poland
eTU Braunschweig, Institute of Internal Combustion Engines and Fuel Cells, Germany
fSchool of Advanced Energy, Sun Yat-sen University, Shenzhen 518107, China
gDepartment of Heterogeneous Catalysis, Max-Planck-Institut für Kohlenforschung, Kaiser-Wilhelm-Platz 1, 45470 Mülheim an der Ruhr, Germany
hInstitute of Condensed Matter and Nanosciences, Université catholique de Louvain, 1348 Louvain-la-Neuve, Belgium
iAdvanced Materials Research Group, Faculty of Engineering, University of Nottingham, Nottingham, NG7 2TU, UK. E-mail: Martin.Dornheim@nottingham.ac.uk
jDepartment of Chemistry and Interdisciplinary Nanoscience Center, Aarhus University, Langelandsgade 140, 8000 Aarhus C, Denmark. E-mail: trj@chem.au.dk
First published on 23rd January 2026
Abstract
The chemistry of hydrogen and its interaction with matter is remarkably diverse with new discoveries and materials continuously being uncovered. New types of chemical bonding and interactions allow for the preparation of new compounds with unusual compositions and properties. For instance, neutral hydrogen molecules may spontaneously form penta-dihydrogen clusters, (H2)5, in nanoporous materials with extremely dense packing, similar to metallic hydrogen at high pressure. Hydrides with extreme hydrogen densities – ‘superhydrides’ – have yielded record critical temperatures under pressure and now guide routes toward low-pressure high-temperature hydride superconductors. A well-known weak interaction identified in biological matter, the hydrogen bond, has an inorganic analogue: the dihydrogen bond. These two interactions have very similar bond lengths and bond strengths that are known to produce flexible and relatively open structures, which often have interesting functionalities. Recently, the di-hydrogen bond has come into focus for development of fast divalent magnesium and calcium cationic conductors. In this review, we highlight key advances in the synthesis and characterisation of novel hydrogen-based materials and illustrate how the compositional and structural versatility of hydrides leads to new functionalities. Hydrides are highly relevant materials with a diversity of energy applications such as solid-state hydrogen storage, solid-state batteries and superconductors, as well as future global hydrogen transportation.
1. Introduction
Hydrogen is the lightest and simplest element and exists as the smallest molecule with only two electrons. However, hydrogen can interact with other elements and compounds by forming a wide variety of bonding types spanning stronger covalent, ionic and metallic bonding along with different types of weaker interactions. In this highlight, we discuss how these weak interactions can lead to new materials and properties, which may help address global challenges such as solid-state energy storage. In addition, stronger interactions may provide hydrogen-based energy storage in the solid-state for mobile applications and large-scale energy transportation.1–7Complex hydrides can be synthesised from some late transition metals, i.e. Fe, Ni and Co,8,9 and some p-block elements, such as boron, aluminium and nitrogen.4,7,10 In particular, boron-based materials have received significant interest over the past decade owing to their fascinating structural chemistry and diversity of compositions and properties.10 Many of these compounds have very high hydrogen densities with potential for solid-state hydrogen storage. Ammonium borohydride, NH4BH4, which is isoelectronic to natural gas, has attained significant attention owing to its extreme gravimetric hydrogen density.11,12 The physical and chemical properties of this material can be modified by additional reactions with more stable metal borohydrides, M(BH4)x, to form (NH4)yM(BH4)x.13 Other types of compounds with extremely high hydrogen content can also be produced under high pressure (>1 GPa), called ‘superhydrides’. Some of these compounds have unique properties such as high-temperature superconductivity.14
Recently, weak interactions and low dimensional frameworks have been shown to have structural flexibility and very high divalent cationic conductivity. The progress in the field has been partially driven by the successful development of new synthesis strategies, e.g. combining several approaches such as solvent-based methods, solid–gas reactions and also mechano-chemistry methods, enabling reactions under more extreme conditions, i.e. high temperature and pressure.15
Dihydrogen interactions, Hδ−⋯Hδ+, can occur between partly positively charged hydrogen, Hδ+ bonded to N, and partly negatively charged hydrogen, Hδ− bonded to B, as found in ammine metal hydroborate structures (Fig. 1).16 A theoretical investigation of dihydrogen bonds revealed that N–Hδ+⋯−δH–B in Mg(BH4)2·NH3 has a bond energy of ∼20 kJ mol−1 and bond lengths of ∼2 Å.17 This is very similar to hydrogen bonds known from biological matter, i.e. proteins, DNA, and membranes. Initially, dihydrogen interactions were considered as a means to tune hydrogen release from solids. However, there is no direct relation between dihydrogen bond strength (energy), N–Hδ+⋯−δH–B, in the solid-state and the composition of the released gas from ammine metal borohydrides.18
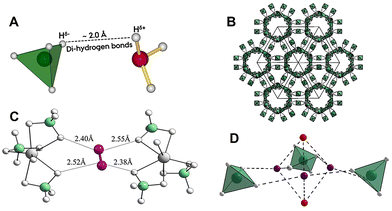 | ||
| Fig. 1 (A) A di-hydrogen bond formed between partly negative hydrogen bonded to boron and partly positive hydrogen bonded to nitrogen. (B) The 'empty' nanoporous structure of γ-Mg(BH4)2. The structure contains 1D channels of ∼9 Å diameter with apertures of ∼5.8 Å. (C) The hydrogen molecules form directional hexa-hydrogen bonds, (B–Hδ−)2⋯H–H⋯(Hδ−–B)2 with partially negatively charged hydrogen in the nanoporous framework (calculated with density functional theory methods). (D) Penta-dihydrogen clusters in the structure of γ-Mg(BH4)2·2.33H2 formed by five hydrogen molecules, (H2)5. Three hydrogen molecules (purple) in the penta-dihydrogen clusters are also forming hexa-hydrogen bonds, whereas the other two (red) only interact with the porous framework via weak dispersion interactions and behave as free rotors that rotate freely around their centre of mass. Colour code: boron is green and BH4− is shown as a tetrahedron, hydrogen is white and magnesium is shown as larger grey spheres. Fig. 1C and D are adapted from reference no. 47 with permission from Springer, Nature Chemistry, Volume 16, 2024, 809–816, copyright 2026 under a Creative Commons Attribution 4.0 International License. | ||
Nanoporous materials are potential candidates for hydrogen gas storage, due to their high surface area and tuneable pore structure. In contrast to absorption, where a chemical interaction occurs between the hydrogen atom and a material, adsorption involves the binding of hydrogen molecules to the internal surfaces of porous solids through van der Waals interactions,19 which is typically fast and readily reversed when the applied hydrogen pressure is removed.20
2. Porous materials for hydrogen adsorption
Molecules can adsorb to an interface by weak dispersion/van der Waals interactions,19 which are proportional to the number of electrons and the molecular surface area. Thus, molecular hydrogen (H2), the smallest molecule of all with only two electrons, forms the weakest dispersion interactions with solid matter. A wide variety of adsorptive materials, including carbons such as graphite and nanotubes, activated carbons (AC), covalent organic frameworks (COFs), and metal–organic frameworks (MOFs),20,21 among others, have been investigated. Pore size and pore volume affect the hydrogen storage capacity since micropores (<2 nm) increase the binding potential, while mesopores and hierarchical structures facilitate hydrogen diffusion and access, balancing storage density and kinetics.212.1. Challenges for storing hydrogen in porous materials
While hydrogen storage in porous materials generally requires less costly heat management than intermetallics, and some materials such as carbon are abundant and relatively inexpensive, there are still several disadvantages. The inherently low enthalpy of adsorption, typically around −4 to −8 kJ mol−1 for physisorption in materials such as activated carbons and many MOFs, requires a low or cryogenic temperature, usually 77 K, to obtain usable hydrogen capacities. This increases system costs through additional energy requirements of cooling and the supply of liquid nitrogen which increases the complexity of the storage system. This leads to the need for tank insulation and further heat management, thereby reducing the economic feasibility of large-scale implementation.The cost of materials, due to their synthesis and processing can also be high. While activated carbons can be typically produced relatively inexpensively from biomass or synthetic precursors, metal-organic frameworks (MOFs) and covalent organic frameworks (COFs) often rely on costly organic linkers and metals.22,23 Typical MOFs such as MOF-524 or HKUST-125 have promising storage capacities in laboratory experiments, but the cost of raw materials and the complexity of the synthesis processes when scaling to commercial quantities are still too high.26 Suggested methods for industrial-scale MOF production to improve sustainability and reduce energy costs have included assessment of MOF life cycles as well as the component costs, toxicity, safety, washing and activation.23
The volumetric capacity of porous materials is typically low, although the gravimetric capacity can be significant due to high surface areas. Volumetric hydrogen capacity, which is more important for mobile applications such as fuel cell vehicles, is limited by the low density of many porous materials. The porous material can be compacted or densified by pressing into discs or pellets; however, this can reduce the porosity and restrict the diffusion of hydrogen gas.27 Nanoporous materials adsorb hydrogen readily at low pressures; consequently, the majority of the hydrogen capacity is only released when the pressure is substantially reduced, typically lower than that required by fuel cells, meaning that the usable capacity is much lower than the full laboratory capacity reported in the literature.21 This may be improved by applying temperature–pressure swings, increasing the temperature of the vessel to increase the hydrogen back pressure to a usable value for a fuel cell.28
In addition, repeated adsorption–desorption cycles can damage the structural stability of some materials such as MOFs. For example, under mechanical stress due to the pelletisation necessary in the forming process, the porous framework of some MOFs may collapse or pores can be blocked, reducing the cycling capacity and operational lifetime.29 Research on more robust frameworks, particularly those with strong metal–ligand bonds, is on-going. Forming composite materials, where MOFs are combined with polymers or carbons, has been proposed to improve structural integrity while retaining a high surface area.20,30
At the system level, managing the application and removal of heat during adsorption and desorption, respectively, is another challenge for reducing costs. Efficient thermal management allows the maintenance of equilibrium pressures and prevents delays in fuelling and discharging.
2.2. Pore size, hydrogen density and increasing the hydrogen capacity
The relationship between pore size, hydrogen density, and overall storage capacity has been the subject of intensive investigation.28 Hydrogen adsorption is strongly influenced by confinement effects: at low pressures in narrow micropores (<1 nm), the overlap of potential energy fields from pore walls increases the binding energy compared to flat surfaces, effectively increasing local hydrogen density. At higher pressures, the maximum uptake is approximately proportional to the total pore volume.The total hydrogen capacity is determined by the area of the surface and the volume of pores available to hydrogen molecules, as well as the interaction of the hydrogen with the surface material under the applied pressure and temperature conditions. For adsorption largely defined by surface interactions, the excess hydrogen adsorption (the amount of hydrogen adsorbed on a material's surface beyond what would occupy the same pore volume as compressed bulk gas) scales with the surface area and has a maximum of approximately 1 wt% per 500 m2 g−1 at 77 K, known as Chahine's Rule,31 see Fig. 2. The maximum uptake at cryogenic temperatures typically occurs at hydrogen pressures of 20–50 bar. For microporous materials, the pore dimensions affect the density of hydrogen stored in the pore volume. Experimental studies employing the same porous activated carbon and carbide-derived carbon (but with different pore diameters) indicate that mesopores (pore sizes >∼25 nm) contribute very little to the hydrogen storage capacity and the majority of hydrogen is stored in micropores below 1.5 nm.32 Overall, pore sizes of 0.6–0.7 nm give the optimal uptake per unit surface area.33
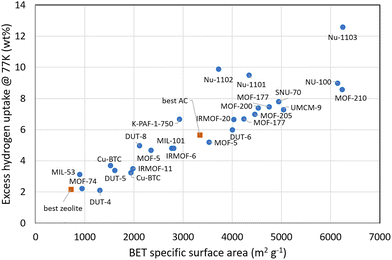 | ||
| Fig. 2 Excess gravimetric hydrogen uptake for various MOFs at 20 bar or above and 77 K (data shown as circles). For comparison, the data for the best zeolite and activated carbon (AC) are also included (data shown as squares). The slope is approximately 1 wt% per 500 m2 g−1, showing Chahine's Rule. Adapted from Broom et al.20 with permission from Springer, Applied Physics A, Volume 122, 2016, 151, copyright 2026 under the terms of the Creative Commons Attribution 4.0 International License (https://http:/creativecommons.org/licenses/by/4.0), with additional data from Li et al.34 and Ahmed et al.35 | ||
For a typical (rigid) microporous material, increasing the pressure of hydrogen increases the hydrogen content until a saturation is reached, yielding the type I isotherm. Higher temperatures reduce the adsorption capacity, and therefore, higher pressures are required to reach saturation. The density of hydrogen in the pores at the maximum capacity depends on both temperature and the nature of the gas–solid interaction. Efforts to determine the average density of hydrogen in a microporous material involve high pressure adsorption measurements substantially beyond the maximum capacity. At hydrogen pressures greater than that needed for saturation, a linear relationship between excess uptake and gas density allows the determination of the volume of the adsorbate, and hence the average density. For MOF materials at 77 K and pressures up to 4 MPa, the average density of hydrogen in the micropores was determined to be 50–70 g H2 L−1, approaching the density of liquid hydrogen, 71 g L−1.36 More recently, much higher pressures, up to 2000 bar, have been employed to investigate the hydrogen density in activated carbon at ambient temperature, yielding an average adsorbate density of 44.7 g H2 L−1 (compared to 38.7 g H2 L−1 for pure hydrogen gas at that pressure). This lower value reflects the much higher temperature (cf. 77 K), but still indicates that a higher capacity than that due to pressure alone is possible in porous materials at ambient temperature if the pressure is sufficiently high.37
Strategies to increase storage capacity of hydrogen physisorption include maximising a material's surface area, increasing pore volume and tailoring pore size distributions, introducing functional groups to strengthen interactions, doping with metal ions, catenation and incorporating open metal sites within MOFs. Open metal sites provide localised electrostatic interactions, which can increase adsorption enthalpies to greater than −15 kJ mol−1.38 Materials such as Co-based SNU-15 MOF with open Co(II) sites have demonstrated this effect, although the hydrogen uptake is only 0.74 wt% owing to the low density of metal sites.39 Additionally, doping carbon materials with heteroatoms such as nitrogen or boron has been reported to enhance hydrogen affinity and increase binding energies, thereby improving low-pressure storage capacities.40
Another promising direction involves densification of porous powders into monoliths or pellets without significantly reducing accessible pore volume,41,42 which can improve volumetric capacity while retaining the gravimetric capacity. In addition, advanced shaping methods, including sol–gel synthesis43 and 3D printing,44 have enabled the fabrication of mechanically stable adsorbents with tuned and optimised pore networks.43
Molecular simulations, including Grand Canonical Monte Carlo (GCMC) and density functional theory (DFT) calculations,45 have been pivotal in predicting optimal pore environments and guiding synthetic efforts. By screening large libraries of hypothetical MOFs, computational studies have revealed that the best-performing materials often feature ultramicropores combined with high densities of adsorption sites. Integration of computational design with experimental synthesis is thus a key avenue toward discovering materials that maximise hydrogen density.
It was shown in 2011 that porous materials can also consist of hydrides: a porous magnesium tetrahydridoborate, γ-Mg(BH4)2, framework has small pores and a unique partially negatively charged non-flat interior capable of reversibly adsorbing small guest molecules such as nitrogen, hydrogen and dichloromethane.46 More recently, this material was investigated for hydrogen and nitrogen uptake by using neutron powder diffraction, volumetric gas adsorption, inelastic neutron scattering, and first-principles calculations.47 γ-Mg(BH4)2 has ca. 30% void space in the solid crystal and contains a 3-dimensional network of channels with a pore diameter of ∼9 Å and smaller openings (apertures) of ∼5.8 Å, which enables the adsorption of smaller molecules.46 This porous substance, γ-Mg(BH4)2, is distinguished from other porous matter by having a partially negative inner surface formed by B–Hδ−–Mg bonds. Surprisingly, ∼3.5 times more hydrogen can be adsorbed in this framework than nitrogen, resulting in the compositions, γ-Mg(BH4)2·2.33H2 or γ-Mg(BH4)2·2/3N2.47 Powder neutron diffraction experiments have revealed extremely dense packing of hydrogen molecules (H2), which form trigonal bipyramidal units, denoted penta-dihydrogen clusters, (H2)5, within the channels. This packing structure leads to adsorbed H2 densities up to ∼144 g H2 L−1 at 25 K, which is about twice that of liquid hydrogen (71 g L−1).47 This corresponds to 8.0 wt% dihydrogen adsorbed in the porous substance and a total hydrogen content of 21.7 wt%. Furthermore, the first two hydrogen molecules adsorbed in each penta-dihydrogen cluster interact with the solid-state structure via non-directional dispersion forces (van der Waals interactions), and these molecules can rotate freely about their centre of mass. This is in contrast to the three following H2 molecules in the penta-dihydrogen clusters, (H2)5, which are bound with well-defined directional bonds and are located slightly closer to the lattice structure in porous γ-Mg(BH4)2. An interaction between molecular hydrogen and the partially negatively charged lattice structure, B–Hδ−⋯H2, is created. New types of hexa-hydrogen bonds are formed, (B–Hδ−)2⋯H–H⋯(Hδ−–B)2, which contribute to the very dense packing of molecular hydrogen.47 Neutron diffraction reveals that dihydrogen is adsorbed close to the covalently bound hydrogen in the porous host structure (shortest distance, H⋯H2 of 2.69 Å) and there is a very short distance between the molecules of the penta-dihydrogen units as H2⋯H2 of 2.64 Å.47 This can be compared to solid hydrogen in which the distance between dihydrogen molecules, H2⋯H2 is 2.66 Å at a pressure of 5.4 GPa.48 The high H2 density in the pores was attributed to the anisotropic shape of the H2 molecules normally seen at near-ambient pressures as close-packed spheroids. In contrast to hydrogen, nitrogen molecules are located in the middle of a pore, as far from the host structure as possible, whereas hydrogen prefers to be closer to the hydridic apertures. Nitrogen and hydrogen have a similar kinetic diameter, d(H2/D2) = 2.89 Å and d(N2) = 3.64 Å, which hints towards specific interactions between the negatively charged framework and neutral molecules. Moreover, macroscopic measurements of nitrogen and hydrogen gas adsorption in the porous substance, γ-Mg(BH4)2, provided very dissimilar results owing to the distinct adsorption sites of these gasses. The calculated Brunauer–Emmett–Teller (BET)-specific surface area for γ-Mg(BH4)2 determined from an N2 isotherm measured at 77 K is only SBET(N2) = 610 m2 g−1, whereas a H2 isotherm (measured at 20 K) gives the result SBET(H2) = 1787 m2 g−1. This highlights the probe-molecule dependence of BET analysis in small-pore frameworks, where H2 and N2 access distinct adsorption sites.47 Interestingly, larger molecules, such as ethane and noble gasses are capable of entering the pores reversibly, despite having very different kinetics.49,50
It was argued that porous γ-Mg(BH4)2 is the first step towards a new class of nanoporous materials resembling flexible metal–organic frameworks (MOFs).46 The similarity of these frameworks to MOFs provides a route to novel hybrid materials, where the combined use of tetrahydridoborate complexes, BH4−, with other directional ligands may produce new porous materials. This could include materials with a high hydrogen storage density like that demonstrated by γ-Mg(BH4)2. In particular, the same framework topology is observed for γ-Mg(BH4)2 and zinc imidazolate framework ZIF-72, and thus prompts the combined use of BH4− and imidazolate ions as ligands. Hence, attempts have been made to extend the family of hydridic porous solids by combining imidazolates (ligands known to build stable frameworks) and complex hydrides, yielding hybrid hydrides with unexpected dynamical properties.51 More complex ligands combining hydridoborates and imidazolates have also been explored.52
2.3. Future prospects for porous materials
Currently, porous materials suffer from low storage capacity under ambient conditions and the requirement for expensive cooling or high pressures, poor stability of some materials, particularly after densification or many cycles of hydrogen release and uptake, costs of synthesis and upscaling, low volumetric capacities and system integration challenges, such as low thermal conductivity and compatibility with existing infrastructure.To improve storage capacity, many MOFs, COFs and PAFs (porous aromatic frameworks) can be tuned to optimise pore size and chemistry, as well as some carbon materials such as carbide-derived carbons, activated and other porous carbons.53 Techniques such as sol–gel synthesis, hydrothermal synthesis, and chemical vapor deposition,54 as well as 3D-printing,44 enable precise pore size distributions. Current research developing adsorbents with enthalpies near the optimal range of ∼−15 to −25 kJ mol−1, which could enable storage under near-ambient conditions, might reduce or eliminate the need for cryogenic cooling. An enthalpy of −21 kJ mol−1 for an MOF with exposed vanadium(II) sites has been reported.55 An imine-based COF with incorporated copper(II) has been found to reversibly adsorb H2 above room temperature with an enthalpy of hydrogen adsorption of −15 kJ mol−1 (ref. 56) and a range of enthalpies from −23 to −33 kJ mol−1 have been reported for a Cu-based MOF undergoing post-synthetic modifications with different metals.57
Recent advances in continuous-flow synthesis of MOFs,58 including air–liquid segmented flow59 and droplet microfluidic systems,60 as well as new techniques, such as the use of artificial intelligence (AI) and improved packing arrangements;61 and the use of cheaper, renewable carbon sources, for example, biomass for porous carbons,62,63 may enable low-cost scalable synthesis. In addition, recent studies of the effects of densification by compaction have indicated that this can result in higher volumetric capacity without structural damage for MOFs such as UiO-66,42 ZIF-8,64 and HKUST-1.43 Repeated cycling of activated carbons63 and MOFs, such as MOF-5,65 along with databases and rules and guidelines for MOF design,66 also shows promise in the design and implementation of materials for long-term cycling.
For large-scale and commercial implementation, thermal management, compact storage and the use of existing hydrogen infrastructure will all need to be addressed. For thermal management, improving the effective thermal conductivity of the absorbent by adding a conductive material, such as graphite67/expanded natural graphite (ENG),68,69 or graphene70/graphene oxide,71 has proven effective, along with advances in the engineering of thermal management systems, such as microchannel cooling plates and liquid nitrogen as a separate cooling fluid.72 Hybrid approaches may provide further improvements. Hybrid materials, such as LiBH4 in a porous scaffold of carbon wrapped Fe3O4,73 MgH2 ball-milled with Ni/Fe based MOFs,74 and MOF-AC hybrids,75,76 generally provide higher capacity and/or better hydrogen diffusion due to synergistic effects between the two components. Hybrid techniques are also developments that have the potential to facilitate more applications, for example, cryo-adsorption coupled with cryo-compression has the advantages of higher hydrogen capacity than adsorption or compression alone, at a lower pressure than compression and a higher temperature than cryo-compression.77
3. Solid-state hydrogen storage
3.1. High hydrogen content hydrides
During the past decade, novel classes of complex hydrides have been reported, some of which have the highest known solid-state gravimetric and volumetric hydrogen densities. This is illustrated by a solid inorganic compound, ammonium tetrahydridoborate, NH4BH4, which is isoelectronic to natural gas (CH4). The two compounds, CH4 and NH4BH4, have similar gravimetric hydrogen densities (∼25 wt% H2), although NH4BH4 has a significantly higher solid-state volumetric density of hydrogen with ρV = 157 g L−1 (see Table 1). However, NH4BH4 is not stable and has a half-life of ∼6 hours owing to slow conversion to the diammoniate of diborane ([(NH3)2BH2]BH4), hydrogen and toxic gases such as ammonia and diborane.11,12,78 This is in contrast to the decomposition of ammonia borane, NH3BH3, which releases hydrogen through polymerisation via polyaminoborane [NH2BH2]n (at 70–100 °C) and polyiminoborane [NHBH]n (∼150 °C) and finally to boron nitride (BN) at temperatures approaching 500 °C.79 Quasielastic neutron scattering of ammonium tetrahydridoborate, NH4BH4, reveals that no reorientation dynamics are present in the low-temperature ordered polymorph at T < 50 K (within the timescale 0.1 to 100 ps). However, the ammonium cation is more dynamic than the BH4− anions in the high-temperature (T > 50 K) disordered polymorph. Initially, NH4+ exhibits tetrahedral tumbling, then cubic tumbling towards isotropic rotational motion at increasing temperatures.80 The highly dynamic structure contributes to the low thermal stability.| Compound | ρ m (wt%) | ρ V (g L−1) | T dec (°C) | Ref. |
|---|---|---|---|---|
| a Metastable compound. | ||||
| H2(l) | 100 | 71.0 | — | 86 |
| CH4(l) | 25.1 | 106.0 | — | 86 |
| MgH2(s) | 7.7 | 108.7 | ∼330 | 87 |
| LiBH4(s) | 18.4 | 122.5 | ∼400 | 88 |
| NH3BH3(s) | 19.6 | 143.6 | 70 | 79 |
| NH4BH4(s) | 24.5 | 157.0 | 11 | |
| NH4Li(BH4)2(s) | 22.1 | 153.1 | ∼52 | 83 |
| (NH4)3Mg(BH4)5(s) | 21.1 | 158.2 | ∼35 | 83 |
| (NH4)3Mn(BH4)5(s) | 17.6 | 154.4 | ∼50 | 83 |
| (NH4)2Y(BH4)5·NH3(s) | 17.0 | 144.3 | — | 83 |
Interestingly, it was found that NH4BH4 can be stabilised by mechano-chemical reactions with metal tetrahydridoborates, such as Ca(BH4)2, which forms a perovskite-type compound NH4Ca(BH4)3.81 Ammonium calcium tetrahydridoborate, NH4Ca(BH4)3, is the first example of efficient stabilisation of NH4BH4 since this compound decomposes to Ca(BH4)2·NH3BH3 and hydrogen at T ∼ 100 °C.81,82
A variety of ammonium metal tetrahydridoborates with new compositions and structure types have recently been discovered.13,83 Full solubility is observed for MBH4–NH4BH4, M = K, Rb, Cs, with formation of solid-solutions, (NH4)xM1−xBH4, by cryo-mechano-chemical treatment. The ammonium metal tetrahydridoborates stabilise NH4BH4 and suppress the aggressive decomposition and possibly alter the decomposition pathway. However, the thermal stability decreases with increasing ratios of NH4BH4 in the compounds owing to increasing numbers of di-hydrogen bonds in the solid-state, facilitating low temperature hydrogen elimination via di-hydrogen bonds.84 The thermal stability of ammonium metal tetrahydridoborates is governed by the Pauling electronegativity of the metal, the structural dimensionality, the dihydrogen bond length, the relative amount of NH4+ to BH4−, and the nearest coordination sphere of NH4+. Hydrogen release of ammonium metal tetrahydridoborates during constant heating often occurs in three steps, involving new intermediate compounds, resulting in crystalline, polymeric, and amorphous materials.83
Ammonium metal tetrahydridoborates may have extreme hydrogen densities even if they contain a transition metal (see Table 1). The compound (NH4)3Mn(BH4)5 has an extremely high gravimetric hydrogen content, ρm = 17.6 wt%, but a record high volumetric hydrogen density, ρV = 154.4 (g L−1). This latter value is more than twice the density of liquid hydrogen (71.0 g L−1). Ammonium–ammonia hydrido-closo-borates, such as (NH4)2B10H10·NH3 and (NH4)2B12H12·NH3 have recently received attention due to their potential application as proton conductors.85 This is owing to the formation of the complex cation, N2H7+, that is formed via a hydrogen bond between NH4+ and NH3.
However, although these new classes of hydrides possess high hydrogen densities, and often release hydrogen under moderate conditions, their rehydrogenation is not straightforward, and further research efforts are required. Another breakthrough is the discovery of chemical reactions between different hydrides and other compounds, which react to form other compounds during the release of hydrogen that more readily absorb hydrogen during the rehydrogenation process. This research provides new opportunities for the design and tailoring of novel functional materials with interesting properties.
3.2. Hydrogen in high entropy alloys
High entropy alloys (HEAs) are a class of multi-component metallic materials characterised by a high configurational disorder. They are defined as alloys containing five or more elements with an atomic content of between 5 and 35 at%89 and a mixing entropy larger or equal to 12.5 J mol−1 K−1.90 They offer tuneable thermodynamics and fast hydrogen sorption kinetics. It has been found that, in addition to configurational entropy, other entropic contributions (vibrational, magnetic and electronic entropy), and enthalpies of mixing, could play a significant role in the phase formation and stability of HEAs. This unique composition leads to a complex crystalline structure with a high degree of disorder at the atomic level, which gives HEAs their distinctive properties and makes them potentially suitable for hydrogen storage applications. A key aspect is the myriad of possibilities to customise the composition of these alloys, potentially leading to the development of alloys with excellent hydrogenation properties. In addition, light and inexpensive elements can be incorporated into the compositions, reducing the weight of the alloys along with the cost, and thus increasing the gravimetric hydrogen storage capacity. The literature indicates that, depending on composition and conditions, these alloys can incorporate large amounts of hydrogen into their lattice structure through processes such as interstitial solid solution formation or hydride formation. Due to the increasing number of compositions and complexity, a recent trend in the literature on HEAs is the use of modeling91 and machine learning (ML)92 for the prediction and/or optimisation of properties,93 which can accelerate development and optimisation of such high entropy hydrides.The studies on HEAs for hydrogen storage focus on three main groups: body-centred cubic (BCC) HEAs, lightweight HEAs and intermetallic HEAs. These groups are related to the traditional hydride-forming metal alloys and are influenced to some extent by the knowledge gained so far. BCC HEAs94 and intermetallic HEAs95,96 are named after the crystal structure they form (BCC solid solutions and ordered intermetallic structures). BCC alloys exhibit high hydrogen per metal ratios and can accommodate up to 2 H per M by transforming into fcc structures. Combining ML, DFT calculations and experiments, Agafonov et al. successfully optimised the composition of a Ti–V–Nb–Cr–Mo alloy and identified a promising HEA with a capacity of 2.6 wt% and an enthalpy of hydride formation of 38 kJ mol−1 H2.97 The third group, light HEAs, does not refer to a specific crystal structure, but to the presence of light elements, such as Mg, Al and Li in the HEA structure.98 Research on lightweight HEAs is mainly focused on improving the gravimetric hydrogen storage capacity by reducing the molar weight of the alloys. Since light HEAs are not bound to a specific crystal structure, they can contain HEAs with BCC, fcc and intermetallic structures. Nevertheless, most of the light HEAs reported so far crystallise in a BCC structure.
Overall, HEAs all have very high volumetric hydrogen density (>100 g H2 L−1), which is superior to that of liquid H2, 70 g L−1. They usually also have very fast kinetics for hydrogen release and uptake, and they are considered safe storage media.99 Furthermore, the gravimetric storage capacity and the thermodynamics for hydrogen release and uptake can be tailored. As an example, the enthalpy of hydride formation for the equimolar alloy Ti25V25Cr25Nb25 is ΔH = −51.6 kJ mol−1 H2,100 whereas that of Ti25V30Nb10Cr31Mo4 is ΔH = −40 kJ mol−1 H2 and this alloy stores 2.24 wt% H2 reversibly at room temperature.101
3.3. Reactive hydride composites for reversible hydrogen storage under moderate conditions
Classical interstitial metal hydrides exhibit favourable volumetric hydrogen storage densities; however, the reversible gravimetric storage capacity under gas-phase loading of such hydrides is typically limited to approximately 2 wt% under practical conditions.102,103 Such a value is insufficient for most mobile applications. In contrast, Mg-based hydrides such as MgH2 and Mg2FeH6 display significantly higher reversible gravimetric storage capacities of approximately 7 and 5 wt%, respectively.104,105 Notably, Mg2FeH6 possesses one of the highest volumetric storage densities reported, reaching 150 g H2 L−1.106 Despite these advantages, Mg-based hydrides suffer from unfavourable thermodynamics as their high hydrogenation enthalpies hinder large-scale implementation, even though kinetic limitations can be partly mitigated through suitable additives.107,108The discovery of reversible hydrogenation in Ti-doped NaAlH4109 initiated intensive research in the discovery and exploration of numerous new lightweight complex hydrides with markedly improved gravimetric storage densities. LiBH4 has been particularly noteworthy owing to its exceptionally high gravimetric hydrogen content of 18.5 wt%.110 Unfortunately, both the hydrogenation and dehydrogenation kinetics of such high-capacity complex hydrides remain insufficient for practical use. Partial rehydrogenation of LiH and B to form LiBH4 requires very harsh conditions of 350 bar and 600 °C.111 To overcome these challenges, Barkhordarian et al.112 demonstrated that rehydrogenation of tetrahydridoborate is considerably more feasible when lightweight hydrides such as LiH, NaH or CaH2 are combined, for example, with MgB2 rather than pure B. In such cases, instead of the thermodynamically hindered reaction (1), reaction (2) occurs at significantly lower temperatures and pressures, while reversible dehydrogenation reaction also proceeds under milder conditions. The use of layered MgB2 instead of icosahedral B plays a key role in facilitating hydrogen uptake and formation of the tetrahydridoborate.113
| xMH2/x + 2B + 3H2 → xM(BH4)2/x | (1) |
| xMH2/x + MgB2 + 4H2 → xM(BH4)2/x + MgH2 | (2) |
As early as 2004, enhanced reaction kinetics and thermodynamics in 2LiBH4/MgH2 composites were reported as compared to their single-phase counterparts.114 Such composites are denoted reactive hydride composites (RHC) and are illustrated in Fig. 3. A critical limitation, however, of such LiBH4/MgH2 composites remains: the high dehydrogenation temperature of more than 350 °C. Nanoconfinement strategies have since reduced hydrogen release temperatures to below 300 °C.115,116
Furthermore, replacing MgH2 in this composite with the thermodynamically less-stable Mg2NiH4 demonstrated a mutual destabilisation effect with LiBH4 lowering the operating temperature to 270 °C, below the melting temperature of LiBH4.117–119
Subsequent studies revealed that further optimisation is possible using different types of transition metal-based additives which promote nucleation, maintaining structural integrity of the nanocomposite and thus enabling faster hydrogenation and dehydrogenation kinetics under more moderate conditions.120,121
An alternative destabilisation approach was also reported in 2005.122 These materials exhibited surprisingly low hydrogen release and uptake temperatures of 170 °C for Mg(NH2)2/2LiH composites and reasonable reversibility under gas-phase loading. However, the reaction kinetics at 170 °C remained slow, with rehydrogenation times of almost 2 hours. However, the addition of small amounts of LiBH4 dramatically improved kinetics with rehydrogenation occurring within 12 min with the addition of 0.5 mol of LiBH4 to 6 mol Mg(NH2)2 + 9 mol LiH. Higher amounts of LiBH4 (1 or 2 mol) in this composite lead to shortened rehydrogenation times of 8 min and 4 min, respectively.123 Further increases in the LiBH4 content accelerated the reaction rates even at near-ambient temperatures. In the 6Mg(NH2)2 + 9LiH + 12LiBH4 composite, Wang et al. could demonstrate hydrogenation at temperatures as low as 53 °C with a rehydrogenation time of 5 hours at 98 °C.124 This kinetic destabilisation was accompanied by thermodynamic destabilisation as the equilibrium plateau pressure at 187 °C increased from 20 bar for the binary 6Mg(NH2)2 + 9LiH system to almost 100 bar in the case of the 3-component 6Mg(NH2)2 + 9LiH + 12LiBH4 system. Recent work suggests that further optimisation is possible by increasing the LiBH4 content.
Although most complex hydrides suffer from sluggish kinetics at moderate temperatures, these studies highlight the potential of using reacting mixtures of hydrides to tailor both thermodynamic and kinetic properties of lightweight and complex hydride-based materials as a promising alternative strategy for hydrogen storage materials design.
3.4. Solid-state hydrogen storage and transport
Sodium tetrahydridoborate, NaBH4, is a vital reducing agent in numerous organic chemistry syntheses but is also a well-known hydrogen-rich material.10,125 Over the last 20 years it has been widely investigated for hydrogen storage purposes, but it typically requires high temperatures (>500 °C) for hydrogen release and extreme conditions for rehydrogenation.126–129 Alternatively, hydrogen release can be obtained from NaBH4 at room temperature using hydrolysis or methanolysis reactions, which were first reported in 1953:130| NaBH4 + 2H2O → NaBO2 + 4H2 | (3) |
| NaBH4 + 4CH3OH → NaB(OCH3)4 + 4H2 | (4) |
The hydrolysis reaction is typically catalysed by metallic nanoparticles for controlled rapid rates of hydrogen gas release.131–133 The simplicity of sodium borohydride hydrolysis has marked it as a potential hydrogen carrier for energy export.134 Here, the cost of hydrogen export from Australia to Japan using powdered NaBH4 ($4.40 USD per kg H2) could be lower than that using ammonia ($5.50 USD per kg H2), liquid organic hydrogen carriers ($5.90 USD per kg H2), or liquid hydrogen ($7.10 USD per kg H2).134 These costs include the renewable production of the hydrogen and the carrier, along with transport and reconversion costs. Another cost-efficient aspect of the process is that the thermodynamics for the hydrolysis and methanolysis suggest that high hydrogen pressure is possible, and it has been demonstrated that up to 1000 bar H2 has been generated, which is comparable to mechanical compression technology.134
If NaBH4 is to become a hydrogen export material, a green, low-cost regeneration method from the NaBO2 hydrolysis product to NaBH4 must be realised. This excludes the typical synthesis method using the Brown–Schlesinger or Bayer processes, as the starting materials are B(OCH3)3 and Na2B4O7·7SiO2, respectively.135 Since 2003, regeneration using thermochemical, mechano-chemical and electrochemical techniques has been demonstrated.132,136 The highest yields have been achieved using magnesium or magnesium hydride as a reactant with the formation of MgO, although Si, Al, Mg17Al12, Mg2Si, Na2CO3 and other additives have also been reported.136,137 For the thermochemical method, temperature is the main barrier in the process, with 600 °C at a H2 pressure of 70 bar being required to achieve a 97% yield,138 although a H2 pressure of 21 bar at 600 °C has also shown to be effective to achieve 90% conversion.139 Mechano-chemistry at room temperature under an argon or hydrogen atmosphere can also achieve high yields, but long milling times are required.136 This method may also suffer from scale up issues due to the ball to powder ratio required and limited size of mills. Electrochemistry processes are carried out in an electrochemical cell which consists of an anode and a cathode according to eqn (5) and (6) below:
| Cathode: BO2− + 6H2O + 8e− → BO4− + 8OH− | (5) |
| Anode: 8OH− → 4H2O + 2O2 + 8e− | (6) |
There have been several publications on the electrochemical synthesis of NaBH4 using various electrolytes, membranes and electrodes, but replication of these data has been reported to be difficult.136,140 One important aspect to overcome is preventing the decomposition of water into hydrogen and oxygen, while at the same time improving electron efficiency to reduce costs. Once the cost-effective synthesis of NaBH4 has been achieved, the extraction of the NaBH4 powder from residual NaBO2 must also be optimised given their similar solubilities in water and general insolubility in aprotic solvents.141,142
4. Solid-state battery materials, hydrides as electrolytes
4.1. Overview
Traditional oxides and halides often have close-packed structures and only have cationic conductivity at very high temperatures owing to defect formation. New classes of hydridoborate-based materials have been discovered during the past decade, which have more open and flexible structures. These hydrides provide new routes to create fast cationic conducting materials as functional solid-state battery materials. Simultaneously, it has become clear that several new phenomena in the solid-state can enhance the ionic conductivity for this class of materials. Impressively, this can lead to novel fast cationic conductivity of divalent cations at moderate temperatures (T < 100 °C).Lithium tetrahydridoborate was recognised as a fast lithium ionic conducting material in 2007, but only at T > ∼110 °C, owing to a polymorphic transition forming the disordered high temperature variant, h-LiBH4 (with a hexagonal structure).143 This polymorph can be stabilised at room temperature by anion substitution resulting in the formation of solid solutions, LiBH4–LiX, X = Cl, Br, I, reaching high Li+ conductivities. The high conductivity was assigned to anionic dynamics in the solid-state.144
4.2. Hydroborate-based magnesium electrolytes
An ammonia derivative of magnesium tetrahydridoborate, Mg(BH4)2·NH3, was discovered recently, with a relatively open structure consisting of one-dimensional zig-zag chains of –BH4–Mg–BH4–Mg– running along the b-axis with one terminal and two bridging BH4− complexes. The chains are interlinked by dihydrogen bonds, B–Hδ−⋯+δH–N, created by partly positively charged hydrogen bonded to nitrogen and partly negatively charged hydrogen atoms bonded to boron. Dihydrogen bonds are the inorganic analogue to hydrogen bonds found in most biological matter with the same bond length (∼2 Å) and strength (∼20 kJ mol−1).17 The monoamine, Mg(BH4)2·NH3, derivative of the series of compounds, Mg(BH4)2·xNH3, x = 0, 1, 2, 3, 6, has the highest Mg2+ conductivity, σ(Mg2+) = 3.3 × 10−4 S cm−1 at 80 °C, which is approximately 8 orders of magnitude higher than that of Mg(BH4)2.145DFT calculations reveal that the fast cationic conductivity is owing to the weak di-hydrogen interactions, flexible coordination of the BH4− complex, and exchange of the neutral molecule, ammonia (NH3), between interstitial and framework magnesium. This is considered a new mechanism for cationic conductivity denoted ‘ligand-assisted cationic conductivity’, which is unique for hydridoborate-based electrolytes.17
Surprisingly, mixing any two compounds in the series, Mg(BH4)2·xNH3, x = 0, 1, 2, 3, 6, increases the conductivity by one to three orders of magnitude. In addition, the composite 0.4Mg(BH4)2·NH3–0.6Mg(BH4)2·2NH3, has a rather low eutectic melting point (T ∼ 55 °C). Also, the addition of larger amounts of inert, insulating nanoparticles, e.g. Al2O3 (∼75 wt%), stabilises the molten phase to become a solid at room temperature.145 Micrographs show that the molten phase is distributed over the nanoparticles as a surface layer, but the dynamic properties of the molten state are preserved providing high conductivity at low temperatures, e.g. σ(Mg2+) ∼ 2.5 × 10−5 S cm−1 at T = 22 °C for the composite, 0.4Mg(BH4)2·NH3–0.6Mg(BH4)2·2NH3–Al2O3, denoted Mg(BH4)2·1.6NH3–Al2O3.146 This phenomenon also increases the conductivity of divalent cations, at low temperatures, which is otherwise very difficult to achieve. These nanocomposites exhibit good mechanical and thermal stability and benefit from nanoconfinement strategies that preserve interfacial dynamics and enhance divalent-ion transport. A nanocomposite material was recently demonstrated as a functional electrolyte for an inorganic solid-state magnesium battery using Mg-metal as the anode, layered titanium disulfide (TiS2) as an intercalation cathode, and a nanocomposite as electrolyte, Mg(BH4)2·1.6NH3–MgO (75 wt%).147
Multiple derivatives of magnesium tetrahydridoborate have been investigated recently containing neutral molecules, which can introduce weak interactions (dispersion/van der Waals) between apolar molecular moieties along with dihydrogen bonds between partly positive and partly negative hydrogen atoms. A series of new compounds have been discovered, e.g. Mg(BH4)2·xCH3NH2, x = 1, 3, 6,148 Mg(BH4)2·x(CH2)4O, x = 0, 2/3, 2, and 3 ((CH2)4O, tetrahydrofuran, THF),149 and Mg(BH4)2·x(CH3)2CHNH2, x = 1, 2, 3 (isopropylamine, IPA).150 It has been noted that compounds with layered (two-dimensional) or chain-like (one-dimensional) structures often have the highest cationic conductivity. The structures of these materials are dominated by polar covalent bonding explaining their extremely low electronic conductivity (σe < 10−12 S cm−1 at RT). The weak bonding introduces structural flexibility, which promotes record high conductivity of divalent cations, e.g. σ(Mg2+) ∼ 1.5 × 10−4 S cm−1 at RT of β-Mg(BH4)2·CH3NH2 (Fig. 4).148 Metal tetrahydridoborates also have very high ionic selectivity, i.e. only one type of ion is mobile in the material, observed as very high cationic transport numbers (t+ ∼ 1).
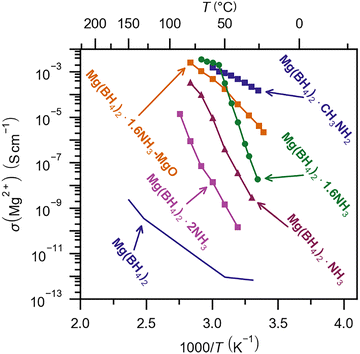 | ||
| Fig. 4 Magnesium ionic conductivity as a function of temperature for derivatives of magnesium tetrahydridoborate. Raw data presented in Fig. 4 are published in ref. 17, 145 and 148. | ||
The nanocomposite, Mg(BH4)2·1.5THF–MgO, displayed very high conductivity, σ(Mg2+) ∼ 10−4 S cm−1 at 70 °C, and a high ionic transport number of t+ = 0.99, and cyclic voltammetry revealed an oxidative stability of ∼1.2 V vs. Mg2+/Mg.149 The electrolyte was stable towards magnesium electrodes, which allowed for stable Mg plating/stripping for at least 100 cycles at 55 °C. The first rechargeable inorganic all-solid-state magnesium battery was presented using this electrolyte: Mg | Mg(BH4)2·1.5THF–MgO | MgyTiS2.149 These new derivatives of magnesium tetrahydridoborate display the highest Mg2+ cationic conductivities at moderate temperatures among all known solids and have an extremely large structural and compositional diversity and offer continued possibilities for the discovery of new compounds.1
Several derivatives of lithium tetrahydridoborate have also been investigated, such as LiBH4·0.5NH3 and LiBH4·CH3NH2, with fast Li+ ionic conductivity.151 A nanocomposite LiBH4·0.5NH3–Li2O–Al2O3, achieved a high conductivity of σ(Li+) = 1.1 × 10−3 S cm−1 at T = 30 °C.152,153 A new solid-state rechargeable lithium battery was assembled, Li | LiBH4·CH3NH2 | TiS2 (battery voltage 2.1 V), based on a LiBH4·CH3NH2 electrolyte with a conductivity of σ(Li+) = 1.24 × 10−4 S cm−1 at RT.154
However, the most remarkable findings are the extremely high calcium cationic conductivity of some derivatives of calcium tetrahydridoborates, which are approaching the relevant values of solid state batteries. A series of urea calcium tetrahydridoborates were recently discovered, Ca(BH4)2·xCO(NH2)2, x = 2, 4, and 6. The highest cationic conductivity is observed for a composite with average composition Ca(BH4)2–6.52CO(NH2)2, σ(Ca2+) = 1.23 × 10−4 S cm−1 at 70 °C.155 A series of crystalline methylamine calcium tetrahydridoborates, Ca(BH4)2·xCH3NH2, x = 1, 2, 4, 6 were also discovered. The compound Ca(BH4)2·2NH2CH3 has a conductivity of σ(Ca2+) ∼ 10−5 S cm−1 at T = 70 °C and high ionic transport number close to unity, tion = 0.9916. This is assigned to a relatively open and flexible octahedral structure built from layers of [Ca(NH2CH3)2(BH4)4] complexes in a cis-geometry, connected by apolar moieties and weak dihydrogen bonds. An electrochemical cell demonstrated plating and stripping of calcium.156 Although thermal stability generally decreases with increasing CH3NH2 content, the mechanical and thermal stability was increased for nanocomposites, which also showed an increase in the ionic conductivity, e.g. Ca(BH4)2·4CH3NH2–MgO (50 wt%), σ(Ca2+) = 1.3 × 10−4 S cm−1 at 60 °C.157 These studies indicate that higher calcium ionic conductivities are observed for derivatives with a higher content of neutral molecules, in contrast to magnesium tetrahydridoborate derivatives (Fig. 5).
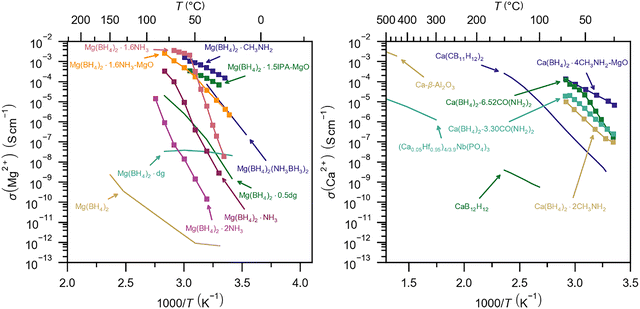 | ||
| Fig. 5 Calcium ionic conductivity as a function of temperature for derivatives of calcium tetrahydridoborate compared to that of known oxide, phosphate and hydrido-closo-(carba)borate based electrolytes. Raw data presented in Fig. 5 are published in ref. 155–157. | ||
4.3. Closo-Borates as electrolytes
Early studies focussed on derivatives of tetrahydridoborate (BH4−) systems as described above, but research has recently shifted towards the use of closo-decaborates (B10H102−) and closo-dodecaborates (B12H122−) (Fig. 6). The larger closo-borates and closo-carbaborates have polymorphic order/disorder transitions from low-temperature (LT) ordered to high-temperature (HT) disordered structures. These transitions are related to dynamics of the anion lattice, i.e. rotation, reorientation and/or vibration of anions. This motion in the anion lattice can be correlated with cation migration, denoted the ‘paddle-wheel’ mechanism, which is often observed as the step-wise increase of cationic conductivity.158,159The interest in metal closo-borates as electrolytes was initiated by the investigation of Li2B12H12 with significant anion disorder in the high temperature polymorph.160,161 Moreover, the large composite borate anions have a low charge, typically −1 or −2, which provides a low charge density and weaker interaction with cations.162 A recent study demonstrated that introducing strain and/or defects into the Li2B12H12 crystal lattice by ball milling can improve ionic conductivity, while partial thermal decomposition at 380 °C leads to an increase in Li+ conductivity by ∼ 2 orders of magnitude compared to the pristine material (σ(Li+) = 1.03 × 10−6 S cm−1 at 30 °C and σ(Li+) = 1.74 × 10−8 S cm−1, respectively).163 Future work on modifications of these closo-borates could improve their ionic conductivity and electrochemical stability as solid-state electrolytes.
The direct synthesis of double anion closo-borates, such as Na4(B12H12)(B10H10), has also been successfully used for the formation of functional electrolytes with fast sodium conductivity of σ(Na+) ∼ 1 mS cm−1 at 25 °C.164 Related advances in solid electrolytes have enabled anode-free all-solid-state sodium batteries with stable cycling, underscoring the relevance of hydride-based electrolytes for next-generation architectures.165
Over the past few decades, it has been shown that these higher borates have large structural flexibility and can provide high ionic conductivities. Further insights allowed for investigations of monovalent closo-carbaborates (e.g. CB11H12−) and nido-borates (e.g. B11H14−), which were potentially more weakly coordinated to cations due to their weaker anionic charge.
4.4. Nido-borates as electrolytes
Alkali-B11H14 salts can be conveniently prepared from NaBH4via a single step low-cost reaction.166 The hydrated LiB11H14 and NaB11H14 salts display high RT ionic conductivities near 1 × 10−4 S cm−1 and 1 × 10−3 S cm−1, respectively. Similar to the closo-carbadodecaborates, thermal decomposition results in hydroxylation of the anion, but even in the hydrated state the salts present an oxidative stability above 2 V vs. Li+/Li.166 Interestingly, LiB11H14·2H2O is an ionic liquid that melts at 70 °C and can be used as a liquid electrolyte.167 If water is removed from the material, resulting in LiB11H14·(H2O)n (n < 2), it remains solid at these temperatures, but exhibits an enhanced ionic conductivity of 3.2 × 10−2 S cm−1.167 Hybrid solid–liquid battery cells made from LiB11H14 composites show impressive cycling with a TiS2 cathode and metallic Li anode due to improved SSE/Li contact.167 Analogous to the B11H14− anion is the divalent B11H132−, where Na2B11H13 shows a similar ionic conductivity to that of the corresponding closo-carbadodecaborate, but lower than that of NaB11H14.168 The ionic conductivity of Na2B11H13 is improved at room temperature to almost 1 × 10−4 S cm−1 through anionic mixing to form Na11(B11H14)3(B11H13)4, which is a higher ionic conductivity than that of hydrated NaB11H14.1684.5. Derivatives of higher borates as electrolytes
CB11H12− is a carbon-substituted closo-dodecaborate anion. Other substitutions can also be used as weakly coordinating anions to form interesting derivatives. For example, the terminal hydrogen atom, B–H unit in B12H122−, can be substituted by a heavy metal atom, such as Pb, to form a B11H11Pb2− anion. Li2B11H11Pb·xH2O shows ionic conductivities of up to 7 mS cm−1 at 120 °C and an oxidative stability of 2.3 V vs. Li+/Li.169 Substitution can also be undertaken with Sn, where Li2B11H11Sn shows an ionic conductivity of 8 mS cm−1 at 130 °C and similar oxidative stability.170 Alternatively, instead of substituting a B–H unit, one can substitute a single H atom on the B12H122− anion with an ammonia group to form the monovalent B12H11NH3− anion. Structural similarities exist between Na2B12H12 and NaB12H11NH3 with high temperature polymorphic phase transitions resulting in the ionic conductivity of both Li and NaB12H11NH3 above 1 × 10−4 S cm−1 at 200 °C.171 These ionic conductivities far exceed those displayed by other derivatives such as the class of alkali metal B12(OH)122− compounds (<1 × 10−4 S cm−1 at 250 °C) or the halogenated variants, B12X122−, where X = Cl, Br, or I, which only reach high ionic conductivity (0.1 S cm−1) at 500 °C.172,173 It is not only the closo-borates that display interesting structural properties and ionic conductivities. The alkali alkoxyborates also show interesting ionic conductivities approaching 1 × 10−2 S cm−1 below 200 °C, which may offer cheaper options for future research studies that approach industrial requirements for cost and scalability.1744.6. Borates in organic liquid or polymeric matrix as electrolytes
To date, the majority of ion conductor research, with respect to boron-based hydrides, has been in the solid-state. However, an increasing amount of research has focused on the electrochemical properties of boron-based hydrides in organic solvents and polymer composites. The unique ability of boron-based hydrides to function as ion conductors in the pure solid-state, solution, and composites sets them apart from the other classes of electrolytes currently used in commercial batteries (LiPF6, LiClO4, LiN(SO2CF3)2) and those investigated for solid-state batteries (oxides, sulphides, and halides). This feature could enable the rational design and tailoring of the hydroborate electrolyte system for specific applications (i.e. vehicular, stationary, extreme conditions). The majority of research using hydroborate in liquid electrolytes has focused on divalent battery chemistries such as Mg2+ and Ca2+, while there are very few studies on Li+.175,176 The early seminal work of Mohtadi using Mg(BH4)2 as a liquid electrolyte in a Mg/Mo6S8 cell highlighted the importance of solvent selection and additives such as LiBH4 to achieve favourable electrochemical properties.177 Subsequent work evaluated the impact of various solvent systems and additives such as ionic liquids, glymes, and crown ethers on the electrochemical, solvation dynamics, and interface properties.178–183 The reactive nature of the BH4−, and incompatibility with high-voltage cathodes, led to the emergence of the magnesium monocarbaborate (MMC) electrolyte, e.g. Mg(CB11H12)2 in tetraglyme.184–186 Using the MMC dissolved in diglyme or tetraglyme as the liquid electrolyte, a highly stable and efficient battery with a high-voltage organic cathode was recently demonstrated.187 The development of calcium ion batteries has lagged behind that of magnesium given the first demonstration of reversible electrochemical plating and stripping of calcium was reported in 2018 for a Ca(BH4)2–THF liquid electrolyte.188 The interphase formation/evolution and solvation environment have also been evaluated at the molecular level.189–191 Mirroring the development of magnesium liquid electrolytes, the use of a calcium monocarborane (CMC) in a THF/DME solvent mixture facilitated the charge and discharge of a Ca/S battery.192The development of gel polymer and free-standing polymer electrolytes utilising boron hydrides has also been recently investigated. The use of polymer electrolytes could improve the energy density and reduce the interfacial resistance that often plagues many solid-state electrolytes. This could also help reduce the quantity of boron-hydride needed for a battery and allow for the use of tape casting for the electrolyte layer during cell assembly. Both advantages could lead to a significant reduction in the cost of battery manufacturing if commercialised. Free standing polyethylene oxide (PEO) based polymer electrolyte films have been demonstrated using Mg(BH4)2 and LiBH4.193–195 However, the ionic conductivity at or near room temperature is relatively low (<10−5 S cm−1) requiring the addition of heat to increase the conductivity and to facilitate reversible cation insertion/extraction with a cathode. To overcome these issues, recent work has turned to gel polymer electrolytes (GPE) which incorporate a plasticiser into the polymer and can achieve high ionic conduction at room temperature (>10−4 S cm−1) and reduced interfacial resistance. GPEs have been developed by incorporating Ca(BH4)2, Mg(BH4)2, LiBH4, or Li2B12H12 into polymers such as PVDF, p-THF, and PMMA.196–200 These GPEs have shown the ability to allow battery cycling at or below room temperature and to prepare flexible batteries. Recent work has also demonstrated that it is possible to anchor and immobilise lithium monocarborane on a polymer to form liquid crystal electrolytes which could lead to new opportunities for future electrolyte designs and opportunities.201,202
5. Hydrides as superconductors
Since the theoretical prediction of metallic hydrogen in 1935 and the subsequent prediction of it possessing superconductivity at or near room temperature, an ongoing quest has been undertaken to create this still elusive state of hydrogen.203,204 The concept of hydrogen “precompression”, through the use of metal hydrides, represented a possible avenue to overcome the extreme pressures needed to achieve the density of metallic hydrogen in materials under experimentally achievable pressures.205 This concept led to the prediction of stable hydrogen clathrate-like structures composed of weakly covalent bonding hydrogen atoms with a lanthanide (Ln) atom at the centre of the hydrogen cage.206 These structures were predicted to possess critical superconducting transition temperatures (Tc) approaching room temperature. However, these structures are only stable at pressures > 100 GPa requiring the use of diamond anvil cell (DAC) conditions. This theoretical prediction was then confirmed experimentally through the observation of superconductivity in a LaH10 species at 250 K under 150 GPa of pressure.207 Since this report, a number of other binary lanthanide hydride phases with Tc values greater than 100 K (YH9 at 243 K and 201 GPa,208 CeH9 at 115 K and 95 GPa,209 and ThH10 at 159 K and 174 GPa210) have been shown to be stable and display behaviour consistent with superconductivity (i.e. resistance, R = 0 Ω below the Tc). These clathrate-like phases of lanthanide hydrides significantly exceed the expected stoichiometry of typical lanthanide hydrides (LnHx, x ≤ 3) that can be readily achieved at pressures available from commercial hydrogen gas cylinders.Non-lanthanide hydride species have been experimentally verified as possessing properties consistent with superconductivity, such as CaH6 at 210 K and 160 GPa,211 H3S at 260 K and 160 GPa,212 and SnHx at 70 K and 200 GPa.213 Much like the lanthanide hydrides, these species also possess a metal to hydrogen stoichiometry that exceeds what would be expected for stable ambient pressure hydrides for these metals (i.e. CaH2 and SnH4). Alloys and multi-element hydrides have also been experimentally confirmed to behave like superconductors with notable examples including (La,Ce)H9 at 176 K and 100 GPa,214 (Y,Ce)H9 at 131 K and 114 GPa,215 (La,Y)H10 at 253 K and 183 GPa,216 and (LaCeY)H10 at 190 K and 112 GPa.217
Superconductivity in these materials is attributed to the contribution of high electronic densities of states at the Fermi level and strong electron–phonon coupling due to the high density of hydrogen, with some reports consistent with Bardeen–Cooper–Schrieffer (BCS) superconductivity theory.218 While the results on superconductivity in metal hydrides have led to the rapid expansion of this field, there are some who call into question these results and make counter arguments for these materials not being superconductive at all.219,220 This stems from the recent retraction of many high profile superconductivity claims, accusations of academic fraud, lack of reproducible results on such small sample sizes (typically on the microgram scale), and difficulty in measuring other hallmarks of superconductivity like the Meissner Effect in DAC on extremely small samples.221 This opens the door for further advancement in this field by developing advanced measurement techniques or devising new strategies to stabilise hydride superconductors and similar high-pressure phases for characterisation near ambient pressure. For example, recent theoretical predictions suggest that certain perovskite hydrides (ABH3) can be superconductive between 140 and 170 K below 10 GPa, while ternary hydrides such as a metastable Mg2IrH6 may be superconductive at 160 K at ambient pressure.222,223 Also, partial hole doping of Ca(BH4)2 through replacement of Ca with K in the structure could potentially achieve ambient pressure superconductivity at 110 K.224 In addition, recent studies have also demonstrated that nanosizing and nonconfinement of lanthanide hydrides could stabilise higher order hydrides at pressures much lower than the bulk lanthanide hydride.225,226 Most recently, it was predicted that Al substituted Mg2FeH6 hydride could exhibit superconductivity at a critical temperature of about 130 K under ambient pressure conditions.227 This finding provides a promising pathway toward the development of practical high-temperature superconductors.
6. New approaches to hydride synthesis
6.1. Overview
Mechanochemical techniques, e.g. ball milling, are widely used in various scientific fields, particularly in synthesising solid-state hydrogen storage materials.228 Despite significant advantages, historically, mechanochemistry has been associated with several limitations such as imprecise control over reaction conditions (e.g., temperature and energy input),229 leading to inconsistent outcomes and scalability issues. In addition, the fundamental mechanisms behind mechanochemical reactions were poorly understood, causing debate about the reliability and reproducibility of the results. However, monitoring the mechanochemical synthesis by in situ powder X-ray diffraction can reveal unexpected insight into the reaction mechanism such as short-lived, metastable intermediates, as observed in the synthesis of metal-organic framework ZIF-8.230 Advances in mill design now allow controlled temperature and pressure environments, while the development of in situ and operando diagnostic techniques (e.g. X-ray diffraction, Raman spectroscopy and calorimetry during milling) has significantly improved insight into reaction pathways and kinetics.231Recent advancements have shown that combining mechanochemical processes with other energy sources, such as light (photo-mechanochemistry), sound (sono-mechanochemistry), electrical fields or impulses (electro-mechanochemistry), and heat (thermo-mechanochemistry), can overcome traditional activation energy barriers and show promise for the synthesis of hydrogen storage materials.232 These hybrid approaches enhance control over reaction conditions, enable novel reactions, increase reaction rates and selectivity, and allow for the synthesis of products that are not achievable through conventional mechanochemical methods alone. As a result, these hybrid techniques can overcome traditional barriers233 and are becoming useful in the field of hydrogen storage materials. Alternatively, a counterintuitive way of performing mechanically induced reactions with theoretically very low energies (without using grinding media) has also gained popularity and can produce contamination-free products with yields higher than those of classical methods.234–236 Mechanochemistry also provides a way to eliminate the need for bulk solvents used in organic synthesis and can reduce both contamination and the generation of waste. Mechanochemistry approaches can enable different reaction mechanisms and thereby new strategies for synthesis and reaction products not readily accessible by solution based methods.237
6.2. High-temperature reactive milling
High-temperature reactive milling (HTRM) was among the first methods used for synthesising magnesium hydride, MgH2. In 1961, Dymova et al.238 used a rotating stainless steel autoclave in a furnace to synthesise MgH2 with a 97–98% yield, using iodine as a catalyst and solved issues previously observed by Wiberg et al.239 Huot et al.240 introduced a high-temperature milling method under hydrogen with the addition of graphite. This significantly accelerated the initial hydrogenation process of magnesium, achieving complete hydrogenation in about one hour. This method combined high temperature (300 °C), hydrogen pressure (4 bar), and graphite additives to enhance the synthesis efficiency and hydrogen sorption properties of magnesium hydride. Recently, Baran et al. proposed an original method using a high-energy planetary ball mill redesigned to control temperatures up to 450 °C and hydrogen pressure up to 100 bar.15 They synthesised magnesium hydride under controlled conditions, finding that higher temperatures enhance reaction rates and yields. However, they found that applying the same technique to synthesise ternary hydrides, such as Mg2FeH6, Mg2NiH4 or Mg2CoH5, encountered issues such as an agglomeration of transition metals, requiring further optimization of milling parameters and experimenting with different (more brittle) materials.241–2436.3. Low-energy reactive milling
Despite the advantages that ball milling provides (i.e. high stress and strain in reactant particles), it can also result in significant introduction of contamination, uncontrolled phase transformations, and local overheating. This variability has led to the development of low-energy mechanochemical approaches without grinding media. For instance, Pistidda et al. investigated the hydrogenation of magnesium diboride, MgB2, using low-energy milling, showing that particle collisions were sufficient to facilitate the reduction of crystallite size, increase lattice strain, and cause hydrogen uptake of 2.1 wt% using operating parameters of 550 rpm for 50 h.234 Patel et al. applied a similar approach to solve the difficulties of activation of intermetallic FeTi.235 Room temperature activation was demonstrated without modification of FeTi by alloying elements244 or heat treatment, with a near theoretical hydrogen capacity being achieved (1.8 wt%). Wyrebska et al. demonstrated the spontaneous room temperature reaction of titanium, and its alloys, with hydrogen during self-shearing reactive milling.236 Using this cost-effective method, titanium hydride was synthesised at close to a 100% yield. These studies highlight the potential of low-energy mechanochemistry in synthesising advanced hydrogen storage materials under moderate conditions, achieving contamination-free and cost-effective synthesis of various hydrogen storage materials.7. Conclusions and outlook
Hydrogen-based materials show highly interesting and promising properties for a wide range of energy-related applications including hydrogen storage and compression, electrochemical energy storage and superconductivity.Nanoporous materials such as carbons, MOFs, COFs, and hydride-based frameworks remain potential hydrogen storage materials, due to their tuneable pore structures and high surface areas, as well as good reversibility at moderate pressures. However, practical or commercial implementation is limited by the low adsorption enthalpies which require cryogenic conditions, low volumetric capacities, high synthesis costs, and poor structural stability with cycling. Despite this, progress in tuning pore size distributions, introducing functional groups, doping, densification, and forming hybrid or composite materials has advanced the scope of applications.
Hydrides offer the advantage of high volumetric hydrogen storage density and safe hydrogen storage even under ambient conditions and are also suitable for long time storage. Challenges remain to improve either the thermodynamics or kinetics, and reversibility of those hydrides that show the highest gravimetric storage densities. However, new approaches have been shown both to improve the key properties of hydrogen rich materials for hydrogen storage and to design completely new classes of materials and/or modify the mechanisms for hydrogen uptake and release.
New synthesis techniques, scalable processing, and improved thermal management strategies have the possibility of reducing costs, and computational modelling and AI provide inspiration for future avenues for development of optimised materials. Furthermore, theoretical approaches may also help to discover novel super-hydrides with unexpected, extremely high hydrogen contents, which may be superconducting at temperatures approaching ambient conditions.
Generally, many solid-state hydridoborates have been found to have extremely high cationic conductivity, both for monovalent cations, such as Li+, Na+, K+ or Ag+, and remarkably also for divalent cations, e.g. Mg2+ or Ca2+. They also have ionic transport numbers close to one (t+ ∼ 1), which contrasts with polymeric and liquid electrolytes (t+ ∼ 0.5).245 Solid state hydridoborate electrolytes also reveal very high charge carrier selectivity with no indication of any H+, H− or anion mobility and extremely low electronic conductivity.1 Thus, future solid-state batteries are expected to have faster charge and discharge as compared to the commercial lithium-ion batteries known today. Furthermore, hydridoborates are stable towards metallic anodes and have a tendency for increasing oxidative stability with increasing number of boron atoms in the anion. Thus, tetrahydridoborate has a relatively low oxidative stability but tends to be oxidised to closo-borates, e.g. B12H122−.246 As an example, lithium tetrahydridoborate, LiBH4, can be oxidised to form Li2B12H12, which may dimerise to B24H233− or trimerize to B36H344− polyanions. Metal hydridoborates are known to have low material density, which contributes to increasing the energy densities of the electrochemical cell.247 Metal hydridoborates are soft and compactable materials, with high deformability and good ability to form interfaces with good contact towards electrodes, e.g. upon mechanical cold pressing.158 They are also known to have high chemical stability and are compatible with halides and oxides and can form double anion compounds such as Ag3(B12H12)I and LiCa3(BH4)(BO3)2.248,249 Metal hydridoborates have high stability towards halides and oxides and provide a variety of possible combinations with known electrolytes. This highlights hydridoborates as a very promising class of solid-state electrolytes with a range of prolific properties, which may be the foundation for future solid-state batteries.
Conflicts of interest
There are no conflicts to declare.Data availability
Data for this review article are available via the references in the reference list in the manuscript. All of the information used in this review has been fully published. No primary research results have been included and no new data were generated or analysed as part of this review.Acknowledgements
The authors acknowledge the important contribution of the International Energy Agency (IEA) Hydrogen Technology Collaboration Programme (TCP) Task 51: Hydrogen Materials for Energy. T. R. J. gratefully acknowledges support from the independent research fund Denmark for technology and production via Calcium Metal Battery – CaMBat (DFF – 0217-00327B) and affiliation with the Center for Integrated Materials Research (iMAT) at Aarhus University. Aarhus Universitets Forskningsfond (AUFF-F-2024-6-8) is acknowledged for support. C. E. B., M. P. and T. D. H. would like to thank the Australian Renewable Energy Agency (ARENA) for Transformative Research Accelerating Commercialisation (TRAC) Program grant 2023/TRAC005 (PRO-1081). M. D. gratefully acknowledges the support from the Leverhulme Trust International Professorship Program grant (LIP-2021-018) and the ISPF UK-Japan Advanced Materials Programme UKRI664. Y. F. acknowledges FNRS (CC J.0078.24) and SPW-Economie Emploi Recherche for Win4Excellence project “TiNTHyN” under contract No. 2310142. Marek Polanski would like to acknowledge the National Centre of Science, Poland (2021/43/B/ST5/01299).References
- J. B. Grinderslev, M. B. Amdisen, L. N. Skov, K. T. Møller, L. G. Kristensen, M. Polanski, M. Heere and T. R. Jensen, J. Alloys Compd., 2022, 896, 163014 CrossRef CAS.
- F. Cuevas, M. B. Amdisen, M. Baricco, C. E. Buckley, Y. W. Cho, P. de Jongh, L. M. de Kort, J. B. Grinderslev, V. Gulino, B. C. Hauback, M. Heere, T. Humphries, T. R. Jensen, S. Kim, K. Kisu, Y.-S. Lee, H.-W. Li, R. Mohtadi, K. T. Møller, P. Ngene, D. Noréus, S. Orimo, M. Paskevicius, M. Polanski, S. Sartori, L. N. Skov, M. H. Sørby, B. C. Wood, V. A. Yartys, M. Zhu and M. Latroche, Prog. Energy, 2022, 4, 032001 CrossRef CAS.
- L. Pasquini, K. Sakaki, E. Akiba, M. D. Allendorf, E. Alvares, J. R. Ares, D. Babai, M. Baricco, J. Bellosta von Colbe, M. Bereznitsky, C. E. Buckley, Y. W. Cho, F. Cuevas, P. de Rango, E. M. Dematteis, R. V. Denys, M. Dornheim, J. F. Fernández, A. Hariyadi, B. C. Hauback, T. W. Heo, M. Hirscher, T. D. Humphries, J. Huot, I. Jacob, T. R. Jensen, P. Jerabek, S. Y. Kang, N. Keilbart, H. Kim, M. Latroche, F. Leardini, H. Li, S. Ling, M. V. Lototskyy, R. Mullen, S. Orimo, M. Paskevicius, C. Pistidda, M. Polanski, J. Puszkiel, E. Rabkin, M. Sahlberg, S. Sartori, A. Santhosh, T. Sato, R. Z. Shneck, M. H. Sørby, Y. Shang, V. Stavila, J.-Y. Suh, S. Suwarno, L. Thi Thu, L. F. Wan, C. J. Webb, M. Witman, C. Wan, B. C. Wood and V. A. Yartys, Prog. Energy, 2022, 4, 032007 Search PubMed.
- E. M. Dematteis, M. B. Amdisen, T. Autrey, J. Barale, M. E. Bowden, C. E. Buckley, Y. W. Cho, S. Deledda, M. Dornheim, P. de Jongh, J. B. Grinderslev, G. Gizer, V. Gulino, B. C. Hauback, M. Heere, T. W. Heo, T. D. Humphries, T. R. Jensen, S. Y. Kang, Y.-S. Lee, H.-W. Li, S. Li, K. T. Møller, P. Ngene, S. Orimo, M. Paskevicius, M. Polanski, S. Takagi, L. Wan, B. C. Wood, M. Hirscher and M. Baricco, Prog. Energy, 2022, 4, 032009 CrossRef CAS.
- M. Adams, C. E. Buckley, M. Busch, R. Bunzel, M. Felderhoff, T. W. Heo, T. D. Humphries, T. R. Jensen, J. Klug, K. H. Klug, K. T. Møller, M. Paskevicius, S. Peil, K. Peinecke, D. A. Sheppard, A. D. Stuart, R. Urbanczyk, F. Wang, G. S. Walker, B. C. Wood, D. Weiss and D. M. Grant, Prog. Energy, 2022, 4, 032008 CrossRef CAS.
- M. D. Allendorf, V. Stavila, J. L. Snider, M. Witman, M. E. Bowden, K. Brooks, B. L. Tran and T. Autrey, Nat. Chem., 2022, 14, 1214–1223 CrossRef CAS PubMed.
- M. Hirscher, V. A. Yartys, M. Baricco, J. Bellosta von Colbe, D. Blanchard, R. C. Bowman, D. P. Broom, C. E. Buckley, F. Chang, P. Chen, Y. W. Cho, J.-C. Crivello, F. Cuevas, W. I. F. David, P. E. de Jongh, R. V. Denys, M. Dornheim, M. Felderhoff, Y. Filinchuk, G. E. Froudakis, D. M. Grant, E. M. Gray, B. C. Hauback, T. He, T. D. Humphries, T. R. Jensen, S. Kim, Y. Kojima, M. Latroche, H.-W. Li, M. V. Lototskyy, J. W. Makepeace, K. T. Møller, L. Naheed, P. Ngene, D. Noréus, M. M. Nygård, S. Orimo, M. Paskevicius, L. Pasquini, D. B. Ravnsbæk, M. Veronica Sofianos, T. J. Udovic, T. Vegge, G. S. Walker, C. J. Webb, C. Weidenthaler and C. Zlotea, J. Alloys Compd., 2020, 827, 153548 CrossRef CAS.
- K. Yvon, in Encyclopedia of Materials: Science and Technology, ed. K. H. J. Buschow, R. W. Cahn, M. C. Flemings, B. Ilschner, E. J. Kramer, S. Mahajan and P. Veyssière, Elsevier, Oxford, 2004, pp. 1–9 DOI:10.1016/B0-08-043152-6%2F01905-7.
- T. D. Humphries, D. A. Sheppard and C. E. Buckley, Coord. Chem. Rev., 2017, 342, 19–33 CrossRef CAS.
- M. Paskevicius, L. H. Jepsen, P. Schouwink, R. Cerny, D. B. Ravnsbaek, Y. Filinchuk, M. Dornheim, F. Besenbacher and T. R. Jensen, Chem. Soc. Rev., 2017, 46, 1565–1634 RSC.
- M. Bowden, D. J. Heldebrant, A. Karkamkar, T. Proffen, G. K. Schenter and T. Autrey, Chem. Commun., 2010, 46, 8564–8566 RSC.
- A. Karkamkar, S. M. Kathmann, G. K. Schenter, D. J. Heldebrant, N. Hess, M. Gutowski and T. Autrey, Chem. Mater., 2009, 21, 4356–4358 CrossRef CAS.
- J. B. Grinderslev and T. R. Jensen, Dalton Trans., 2022, 51, 17762–17771 RSC.
- S. Di Cataldo, C. Heil, W. von der Linden and L. Boeri, Phys. Rev. B, 2021, 104, L020511 CrossRef CAS.
- A. Baran, M. Knioła, T. Rogala and M. Polanski, Int. J. Hydrogen Energy, 2022, 47, 35003–35016 Search PubMed.
- D. J. Wolstenholme, J. L. Dobson and G. S. McGrady, Dalton Trans., 2015, 44, 9718–9731 Search PubMed.
- Y. Yan, W. Dononelli, M. Jorgensen, J. B. Grinderslev, Y.-S. Lee, Y. W. Cho, R. Černý, B. Hammer and T. R. Jensen, Phys. Chem. Chem. Phys., 2020, 22, 9204–9209 RSC.
- L. H. Jepsen, Y. S. Lee, R. Černý, R. S. Sarusie, Y. W. Cho, F. Besenbacher and T. R. Jensen, ChemSusChem, 2015, 8, 3472–3482 CrossRef CAS PubMed.
- R. C. Lochan and M. Head-Gordon, Phys. Chem. Chem. Phys., 2006, 8, 1357–1370 RSC.
- D. P. Broom, C. J. Webb, K. E. Hurst, P. A. Parilla, T. Gennett, C. M. Brown, R. Zacharia, E. Tylianakis, E. Klontzas, G. E. Froudakis, T. A. Steriotis, P. N. Trikalitis, D. L. Anton, B. Hardy, D. Tamburello, C. Corgnale, B. A. van Hassel, D. Cossement, R. Chahine and M. Hirscher, Appl. Phys. A, 2016, 122, 151–171 CrossRef.
- D. P. Broom, C. J. Webb, G. S. Fanourgakis, G. E. Froudakis, P. N. Trikalitis and M. Hirscher, Int. J. Hydrogen Energy, 2019, 44, 7768–7779 CrossRef CAS.
- J. A. Villajos, M. Bienert, N. Gugin, F. Emmerling and M. Maiwald, Mater. Adv., 2023, 4, 4226–4237 Search PubMed.
- D. Chakraborty, A. Yurdusen, G. Mouchaham, F. Nouar and C. Serre, Adv. Funct. Mater., 2023, 34, 2309089 CrossRef.
- K. K. Gangu, S. Maddila and S. B. Jonnalagadda, RSC Adv., 2022, 12, 14282–14298 Search PubMed.
- T. Zhao, S. Nie, M. Luo, P. Xiao, M. Zou and Y. Chen, J. Alloys Compd., 2024, 974, 172897 CrossRef CAS.
- W. Luo, Z. Zhang, G. Zhu, X. Zhang, G. Huang, T. Zhou and X. Lu, J. Environ. Chem. Eng., 2025, 13, 118229 CrossRef CAS.
- C. Xu, J. Yang, M. Veenstra, A. Sudik, J. J. Purewal, Y. Ming, B. J. Hardy, J. Warner, S. Maurer, U. Müeller and D. J. Siegel, Int. J. Hydrogen Energy, 2013, 38, 3268–3274 CrossRef CAS.
- L. Zhang, M. D. Allendorf, R. Balderas-Xicohténcatl, D. P. Broom, G. S. Fanourgakis, G. E. Froudakis, T. Gennett, K. E. Hurst, S. Ling, C. Milanese, P. A. Parilla, D. Pontiroli, M. Riccò, S. Shulda, V. Stavila, T. A. Steriotis, C. J. Webb, M. Witman and M. Hirscher, Prog. Energy, 2022, 4, 042013 CrossRef CAS.
- A. R. Yuvaraj, A. Jayarama, D. Sharma, S. S. Nagarkar, S. P. Duttagupta and R. Pinto, Int. J. Hydrogen Energy, 2024, 59, 1434–1458 CrossRef CAS.
- X. Zhang, P. Liu and Y. Zhang, Inorg. Chim. Acta, 2023, 557, 121683 Search PubMed.
- R. Chahine and T. K. Bose, Proceedings: 11th World Hydrogen Energy Conference (WHEC), 1996, 2, 1259–1263.
- J. Huang, Y. Liang, H. Dong, H. Hu, P. Yu, L. Peng, M. Zheng, Y. Xiao and Y. Liu, Int. J. Hydrogen Energy, 2018, 43, 18077–18082 Search PubMed.
- Y. Gogotsi, C. Portet, S. Osswald, J. M. Simmons, T. Yildirim, G. Laudisio and J. E. Fischer, Int. J. Hydrogen Energy, 2009, 34, 6314–6319 CrossRef CAS.
- Y. Li, T. Ben, B. Zhang, Y. Fu and S. Qiu, Sci. Rep., 2013, 3, 2420 CrossRef PubMed.
- A. Ahmed, S. Seth, J. Purewal, A. G. Wong-Foy, M. Veenstra, A. J. Matzger and D. J. Siegel, Nat. Commun., 2019, 10, 1568 CrossRef PubMed.
- E. Poirier, RSC Adv., 2014, 4, 22848–22855 RSC.
- L. Naheed, K. E. Lamb, E. M. Gray and C. J. Webb, Adsorption, 2021, 27, 1251–1261 CrossRef CAS.
- M. P. Suh, H. J. Park, T. K. Prasad and D. W. Lim, Chem. Rev., 2012, 112, 782–835 CrossRef CAS PubMed.
- Y. E. Cheon and M. P. Suh, Chem. Commun., 2009, 2296–2298 RSC.
- Z. Wang, L. Sun, F. Xu, H. Zhou, X. Peng, D. Sun, J. Wang and Y. Du, Int. J. Hydrogen Energy, 2016, 41, 8489–8497 CrossRef CAS.
- M. Jordá-Beneyto, D. Lozano-Castelló, F. Suárez-García, D. Cazorla-Amorós and Á. Linares-Solano, Microporous Mesoporous Mater., 2008, 112, 235–242 Search PubMed.
- S. E. Bambalaza, H. W. Langmi, R. Mokaya, N. M. Musyoka, J. Ren and L. E. Khotseng, J. Mater. Chem. A, 2018, 6, 23569–23577 RSC.
- D. G. Madden, D. O'Nolan, N. Rampal, R. Babu, C. Camur, A. N. Al Shakhs, S. Y. Zhang, G. A. Rance, J. Perez, N. P. Maria Casati, C. Cuadrado-Collados, D. O'Sullivan, N. P. Rice, T. Gennett, P. Parilla, S. Shulda, K. E. Hurst, V. Stavila, M. D. Allendorf, J. Silvestre-Albero, A. C. Forse, N. R. Champness, K. W. Chapman and D. Fairen-Jimenez, J. Am. Chem. Soc., 2022, 144, 13729–13739 CrossRef CAS PubMed.
- A. J. A. Abubakar, R. L. S. Canevesi, D. A. L. Sanchez and C. A. Grande, Chem. Eng. J., 2024, 489, 151450 CrossRef CAS.
- M. D. Allendorf, Z. Hulvey, T. Gennett, A. Ahmed, T. Autrey, J. Camp, E. Seon Cho, H. Furukawa, M. Haranczyk, M. Head-Gordon, S. Jeong, A. Karkamkar, D.-J. Liu, J. R. Long, K. R. Meihaus, I. H. Nayyar, R. Nazarov, D. J. Siegel, V. Stavila, J. J. Urban, S. P. Veccham and B. C. Wood, Energy Environ. Sci., 2018, 11, 2784–2812 RSC.
- Y. Filinchuk, B. Richter, T. R. Jensen, V. Dmitriev, D. Chernyshov and H. Hagemann, Angew. Chem., Int. Ed., 2011, 50, 11162–11166 CrossRef CAS PubMed.
- H. Oh, N. Tumanov, V. Ban, X. Li, B. Richter, M. R. Hudson, C. M. Brown, G. N. Iles, D. Wallacher, S. W. Jorgensen, L. Daemen, R. Balderas-Xicohténcatl, Y. Cheng, A. J. Ramirez-Cuesta, M. Heere, S. Posada-Pérez, G. Hautier, M. Hirscher, T. R. Jensen and Y. Filinchuk, Nat. Chem., 2024, 16, 809–816 CrossRef CAS PubMed.
- R. Hazen, H. Mao, L. Finger and R. Hemley, Phys. Rev. B:Condens. Matter Mater. Phys., 1987, 36, 3944 CrossRef CAS PubMed.
- I. Dovgaliuk, V. Dyadkin, M. V. Donckt, Y. Filinchuk and D. Chernyshov, ACS Appl. Mater. Interfaces, 2020, 12, 7710–7716 CrossRef CAS PubMed.
- I. Dovgaliuk, I. Senkovska, X. Li, V. Dyadkin, Y. Filinchuk and D. Chernyshov, Angew. Chem., Int. Ed., 2021, 60, 5250–5256 CrossRef CAS PubMed.
- A. V. Skripov, M. Dimitrievska, O. A. Babanova, R. V. Skoryunov, A. V. Soloninin, F. Morelle, Y. Filinchuk, A. Faraone, H. Wu, W. Zhou and T. J. Udovic, J. Phys. Chem. C, 2019, 123, 20789–20799 CrossRef CAS.
- M. Reberc, M. Mazaj, J. Stare, M. Počkaj, G. Mali, X. Li, Y. Filinchuk, R. Černý and A. Meden, Inorg. Chem., 2022, 61, 12708–12718 CrossRef CAS PubMed.
- L. A. M. Mahmoud, J. L. Rowlandson, D. J. Fermin, V. P. Ting and S. Nayak, RSC Appl. Interfaces, 2025, 2, 25–55 RSC.
- K. Mondal, S. J. Malode, N. P. Shetti, S. A. Alqarni, S. Pandiaraj and A. Alodhayb, J. Energy Storage, 2024, 76, 109719 CrossRef.
- D. E. Jaramillo, H. Z. H. Jiang, H. A. Evans, R. Chakraborty, H. Furukawa, C. M. Brown, M. Head-Gordon and J. R. Long, J. Am. Chem. Soc., 2021, 143, 6248–6256 CrossRef CAS PubMed.
- W. A. Braunecker, S. Shulda, M. B. Martinez, K. E. Hurst, J. T. Koubek, S. Zaccarine, R. E. Mow, S. Pylypenko, A. Sellinger, T. Gennett and J. C. Johnson, ACS Mater. Lett., 2020, 2, 227–232 CrossRef CAS.
- Y. Yabuuchi, H. Furukawa, K. M. Carsch, R. A. Klein, N. V. Tkachenko, A. J. Huang, Y. Cheng, K. M. Taddei, E. Novak, C. M. Brown, M. Head-Gordon and J. R. Long, J. Am. Chem. Soc., 2024, 146, 22759–22776 CrossRef CAS PubMed.
- D. Senthil Raja and D. H. Tsai, Chem. Commun., 2024, 60, 8497–8515 RSC.
- Q. Fu, W. Niu, L. Yan, W. Xie, H. Jiang, S. Zhang, L. Yang, Y. Wang, Y. Xing and X. Zhao, Mater. Lett., 2023, 343, 134344 CrossRef CAS.
- H.-Y. Wu, C.-L. Wu, W. Liao, B. M. Matsagar, K.-Y. Chang, J.-H. Huang and K. C. W. Wu, J. Mater. Chem. A, 2023, 11, 9427–9435 RSC.
- L. Qiao, C. Lu, W. Fan, Z. Xue, X. Wang, Z. Kang and D. Sun, Int. J. Hydrogen Energy, 2024, 93, 805–821 CrossRef CAS.
- S. Elyasi, S. Saha, N. Hameed, P. J. Mahon, S. Juodkazis and N. Salim, Int. J. Hydrogen Energy, 2024, 62, 272–306 CrossRef CAS.
- J. Serafin, B. Dziejarski, C. Solis, P. Ramírez de la Piscina and N. Homs, Fuel, 2024, 363, 130975 CrossRef CAS.
- E. Tsalaporta and J. M. D. MacElroy, Heliyon, 2020, 6, e04883 CrossRef CAS PubMed.
- Y. Ming, J. Purewal, J. Yang, C. Xu, M. Veenstra, M. Gaab, U. Müller and D. J. Siegel, Int. J. Hydrogen Energy, 2016, 41, 9374–9382 CrossRef CAS.
- T. C. Wang, J. L. White, B. Bie, H. Deng, J. Edgington, J. D. Sugar, V. Stavila and M. D. Allendorf, ChemPhysChem, 2019, 20, 1305–1310 CrossRef CAS PubMed.
- J. Farrando-Pérez, M. Rodríguez-Castillo, M. Martínez-Escandell, M. Monge and J. Silvestre-Albero, Int. J. Hydrogen Energy, 2023, 48, 36474–36484 CrossRef.
- D. Menard, X. Py and N. Mazet, Chem. Eng. Process. Process Intensif., 2005, 44, 1029–1038 CrossRef CAS.
- D. Liu, J. J. Purewal, J. Yang, A. Sudik, S. Maurer, U. Mueller, J. Ni and D. J. Siegel, Int. J. Hydrogen Energy, 2012, 37, 6109–6117 CrossRef CAS.
- S. E. Bambalaza, H. W. Langmi, N. M. Musyoka, J. Ren and L. E. Khotseng, Mater. Today Proc., 2018, 5, 10431–10439 CrossRef CAS.
- M. C. Tapia, B. G. Alamani and T.-H. Bae, Sep. Purif. Technol., 2025, 363, 132313 CrossRef CAS.
- H. Wang and D. C. Miller, J. Electrochem. Eng. Conv. Stor., 2022, 19, 011002 Search PubMed.
- S. Wang, M. Gao, Z. Yao, Y. Liu, M. Wu, Z. Li, Y. Liu, W. Sun and H. Pan, Chem. Eng. J., 2022, 428, 131056 Search PubMed.
- J.-Q. Hu, W. Jiang, N. Si, Z. Wang and H. Zhang, Int. J. Hydrogen Energy, 2024, 86, 445–455 Search PubMed.
- Z. Yu, J. Deschamps, L. Hamon, P. Karikkethu Prabhakaran and P. Pré, Int. J. Hydrogen Energy, 2017, 42, 8021–8031 Search PubMed.
- L. Luo, Y. Zhou, W. Yan, L. Luo, J. Deng, M. Fan and W. Zhao, Int. J. Hydrogen Energy, 2022, 47, 39563–39571 Search PubMed.
- P. Ramirez-Vidal, G. Sdanghi, A. Celzard and V. Fierro, Int. J. Hydrogen Energy, 2022, 47, 8892–8915 Search PubMed.
- T. K. Nielsen, A. Karkamkar, M. Bowden, F. Besenbacher, T. R. Jensen and T. Autrey, Dalton Trans., 2013, 42, 680–687 RSC.
- V. Sit, R. Geanangel and W. Wendlandt, Thermochim. Acta, 1987, 113, 379–382 CrossRef CAS.
- M. S. Andersson, J. B. Grinderslev, T. R. Jensen, V. García Sakai, U. Häussermann, T. J. Udovic and M. Karlsson, Phys. Rev. Mater., 2020, 4, 085002 CrossRef CAS.
- P. Schouwink, M. B. Ley, A. Tissot, H. Hagemann, T. R. Jensen, Ľ. Smrčok and R. Černý, Nat. Commun., 2014, 5, 5706 CrossRef CAS PubMed.
- P. Schouwink, F. Morelle, Y. Sadikin, Y. Filinchuk and R. Černý, Energies, 2015, 8, 8286–8299 CrossRef CAS.
- J. B. Grinderslev, L. H. Jepsen, Y.-S. Lee, K. T. Møller, Y. W. Cho, R. Černý and T. R. Jensen, Inorg. Chem., 2020, 59, 12733–12747 CrossRef CAS PubMed.
- S. Filippov, J. B. Grinderslev, M. S. Andersson, J. Armstrong, M. Karlsson, T. R. Jensen, J. Klarbring, S. I. Simak and U. Häussermann, J. Phys. Chem. C, 2019, 123, 28631–28639 CrossRef CAS.
- J. B. Grinderslev, Y.-S. Lee, M. Paskevicius, K. T. Møller, Y. Yan, Y. W. Cho and T. R. Jensen, Inorg. Chem., 2020, 59, 11449–11458 Search PubMed.
- D. R. Lide, CRC Handbook of Chemistry and Physics, CRC Press, Boca Raton, USA, New York, 88th edn, 2007 Search PubMed.
- M. Dornheim, N. Eigen, G. Barkhordarian, T. Klassen and R. Bormann, Adv. Eng. Mater., 2006, 8, 377–385 CrossRef CAS.
- M. B. Smith and G. E. Bass Jr, J. Chem. Eng. Data, 1963, 8, 342–346 CrossRef CAS.
- F. Marques, M. Balcerzak, F. Winkelmann, G. Zepon and M. Felderhoff, Energy Environ. Sci., 2021, 14, 5191–5227 RSC.
- T. R. Somo, M. V. Lototskyy, V. A. Yartys, M. W. Davids and S. N. Nyamsi, J. Energy Storage, 2023, 73, 108969 CrossRef.
- G. Zepon, B. H. Silva, C. Zlotea, W. J. Botta and Y. Champion, Acta Mater., 2021, 215, 117070 CrossRef CAS.
- M. Witman, G. Ek, S. Ling, J. Chames, S. Agarwal, J. Wong, M. D. Allendorf, M. Sahlberg and V. Stavila, Chem. Mater., 2021, 33, 4067–4076 CrossRef CAS.
- A. Agafonov, N. Pineda-Romero, M. Witman, V. Nassif, G. B. M. Vaughan, L. Lei, S. Ling, D. M. Grant, M. Dornheim, M. Allendorf, V. Stavila and C. Zlotea, Acta Mater., 2024, 276, 120086 Search PubMed.
- L. Kong, B. Cheng, D. Wan and Y. Xue, Front. Mater., 2023, 10, 1135864 Search PubMed.
- H. Ma and C. H. Shek, J. Alloys Compd., 2020, 827, 154159 Search PubMed.
- Y. Long, X. Liang, K. Su, H. Peng and X. Li, J. Alloys Compd., 2019, 780, 607–617 CrossRef CAS.
- A. Agafonov, N. Pineda-Romero, M. D. Witman, V. Enblom, M. Sahlberg, V. Nassif, L. Lei, D. M. Grant, M. Dornheim, S. Ling, V. Stavila and C. Zlotea, ACS Appl. Mater. Interfaces, 2025, 17, 41991–42003 Search PubMed.
- Z. Wang, S. Chen, S. Yang, Q. Luo, Y. Jin, W. Xie, L. Zhang and Q. Li, J. Mater. Sci. Technol., 2023, 151, 41–65 Search PubMed.
- K. Chen, M. Y. Lau, X. Luo, J. Huang, L. Ouyang and X.-S. Yang, J. Mater. Sci. Technol., 2026, 246, 256–289 Search PubMed.
- M. M. Nygård, Ø. S. Fjellvåg, M. H. Sørby, K. Sakaki, K. Ikeda, J. Armstrong, P. Vajeeston, W. A. Sławiński, H. Kim, A. Machida, Y. Nakamura and B. C. Hauback, Acta Mater., 2021, 205, 116496 Search PubMed.
- B. Cheng, L. Kong, H. Cai, Y. Li, Y. Zhao, D. Wan and Y. Xue, Int. J. Hydrogen Energy, 2024, 60, 282–292 Search PubMed.
- M. Dornheim, L. Baetcke, E. Akiba, J.-R. Ares, T. Autrey, J. Barale, M. Baricco, K. Brooks, N. Chalkiadakis, V. Charbonnier, S. Christensen, J. Bellosta von Colbe, M. Costamagna, E. Dematteis, J.-F. Fernández, T. Gennett, D. Grant, T. W. Heo, M. Hirscher, K. Hurst, M. Lototskyy, O. Metz, P. Rizzi, K. Sakaki, S. Sartori, E. Stamatakis, A. Stuart, A. Stubos, G. Walker, C. J. Webb, B. Wood, V. Yartys and E. Zoulias, Prog. Energy, 2022, 4, 042005 Search PubMed.
- V. A. Yartys, M. V. Lototskyy, V. Linkov, S. Pasupathi, M. W. Davids, I. Tolj, G. Radica, R. V. Denys, J. Eriksen, K. Taube, J. Bellosta von Colbe, G. Capurso, M. Dornheim, F. Smith, D. Mathebula, D. Swanepoel and S. Suwarno, Int. J. Hydrogen Energy, 2021, 46, 35896–35909 CrossRef CAS.
- J. C. Crivello, R. V. Denys, M. Dornheim, M. Felderhoff, D. M. Grant, J. Huot, T. R. Jensen, P. de Jongh, M. Latroche, G. S. Walker, C. J. Webb and V. A. Yartys, Appl. Phys. A, 2016, 122, 85–101 CrossRef.
- V. A. Yartys, M. V. Lototskyy, E. Akiba, R. Albert, V. E. Antonov, J. R. Ares, M. Baricco, N. Bourgeois, C. E. Buckley, J. M. Bellosta von Colbe, J. C. Crivello, F. Cuevas, R. V. Denys, M. Dornheim, M. Felderhoff, D. M. Grant, B. C. Hauback, T. D. Humphries, I. Jacob, T. R. Jensen, P. E. de Jongh, J. M. Joubert, M. A. Kuzovnikov, M. Latroche, M. Paskevicius, L. Pasquini, L. Popilevsky, V. M. Skripnyuk, E. Rabkin, M. V. Sofianos, A. Stuart, G. Walker, H. Wang, C. J. Webb and M. Zhu, Int. J. Hydrogen Energy, 2019, 44, 7809–7859 CrossRef CAS.
- A.-L. Chaudhary, S. Dietzel, H.-W. Li, E. Akiba, N. Bergemann, C. Pistidda, T. Klassen and M. Dornheim, Int. J. Hydrogen Energy, 2017, 42, 11422–11428 Search PubMed.
- M. Dornheim, S. Doppiu, G. Barkhordarian, U. Boesenberg, T. Klassen, O. Gutfleisch and R. Bormann, Scripta Mater., 2007, 56, 841–846 CrossRef CAS.
- C. J. Webb, J. Phys. Chem. Solids, 2015, 84, 96–106 Search PubMed.
- B. Bogdanović and M. Schwickardi, J. Alloys Compd., 1997, 253–254, 1–9 Search PubMed.
- L. Mosegaard, B. Møller, J.-E. Jørgensen, U. Bösenberg, M. Dornheim, J. C. Hanson, Y. Cerenius, G. Walker, H. J. Jakobsen, F. Besenbacher and T. R. Jensen, J. Alloys Compd., 2007, 446–447, 301–305 Search PubMed.
- G. Li, M. Matsuo, S. Takagi, A.-L. Chaudhary, T. Sato, M. Dornheim and S. Orimo, Inorganics, 2017, 5, 81 Search PubMed.
- G. Barkhordarian, T. Klassen, M. Dornheim and R. Bormann, J. Alloys Compd., 2007, 440, L18–L21 CrossRef CAS.
- N. Kuganathan, M. Dornheim, D. M. Grant and S. Ling, Mater. Chem. Phys., 2024, 324, 129677 CrossRef CAS.
- J. J. Vajo, F. Mertens, C. C. Ahn, R. C. Bowman and B. Fultz, J. Phys. Chem. B, 2004, 108, 13977–13983 Search PubMed.
- T. T. Le, C. Pistidda, V. H. Nguyen, P. Singh, P. Raizada, T. Klassen and M. Dornheim, Int. J. Hydrogen Energy, 2021, 46, 23723–23736 CrossRef CAS.
- R. Utke, S. Thiangviriya, P. Javadian, T. R. Jensen, C. Milanese, T. Klassen and M. Dornheim, Mater. Chem. Phys., 2016, 169, 136–141 CrossRef CAS.
- N. Bergemann, C. Pistidda, M. Uptmoor, C. Milanese, A. Santoru, T. Emmler, J. Puszkiel, M. Dornheim and T. Klassen, J. Energy Chem., 2019, 34, 240–254 CrossRef.
- J. J. Vajo, W. Li and P. Liu, Chem. Commun., 2010, 46, 6687–6689 RSC.
- P. Dansirima, J. B. Grinderslev, L. G. Kristensen, R. Utke and T. R. Jensen, Int. J. Hydrogen Energy, 2025, 98, 908–914 CrossRef CAS.
- F. Karimi, S. Börries, P. K. Pranzas, O. Metz, A. Hoell, G. Gizer, J. A. Puszkiel, M. V. C. Riglos, C. Pistidda, M. Dornheim, T. Klassen and A. Schreyer, Adv. Eng. Mater., 2021, 23, 2100294 Search PubMed.
- F. Karimi, M. V. C. Riglos, A. Santoru, A. Hoell, V. S. Raghuwanshi, C. Milanese, N. Bergemann, C. Pistidda, P. Nolis, M. D. Baro, G. Gizer, T.-T. Le, P. K. Pranzas, M. Dornheim, T. Klassen, A. Schreyer and J. Puszkiel, J. Phys. Chem. C, 2018, 122, 11671–11681 CrossRef CAS.
- Z. Xiong, J. Hu, G. Wu, P. Chen, W. Luo, K. Gross and J. Wang, J. Alloys Compd., 2005, 398, 235–239 Search PubMed.
- G. Gizer, J. Puszkiel, H. Cao, C. Pistidda, T. T. Le, M. Dornheim and T. Klassen, Int. J. Hydrogen Energy, 2019, 44, 11920–11929 Search PubMed.
- H. Wang, G. Wu, H. Cao, C. Pistidda, A. L. Chaudhary, S. Garroni, M. Dornheim and P. Chen, Adv. Energy Mater., 2017, 7, 1602456 CrossRef.
- S. Orimo, Y. Nakamori and A. Zuttel, Mater. Sci. Eng., B, 2004, 108, 51–53 Search PubMed.
- M. Paskevicius, M. B. Ley, D. A. Sheppard, T. R. Jensen and C. E. Buckley, Phys. Chem. Chem. Phys., 2013, 15, 19774–19789 Search PubMed.
- P. Martelli, R. Caputo, A. Remhof, P. Mauron, A. Borgschulte and A. Züttel, J. Phys. Chem. C, 2010, 114, 7173–7177 Search PubMed.
- D. S. Stasinevich and G. A. Egorenko, Russ. J. Inorg. Chem., 1968, 13, 341–343 Search PubMed.
- T. D. Humphries, G. N. Kalantzopoulos, I. Llamas-Jansa, J. E. Olsen and B. C. Hauback, J. Phys. Chem. C, 2013, 117, 6060–6065 Search PubMed.
- H. I. Schlesinger, H. C. Brown, H. R. Hoekstra and L. R. Rapp, J. Am. Chem. Soc., 1953, 75, 199–204 CrossRef CAS.
- H. N. Abdelhamid, Int. J. Hydrogen Energy, 2021, 46, 726–765 Search PubMed.
- L. Ouyang, J. Jiang, K. Chen, M. Zhu and Z. Liu, Nano-Micro Lett., 2021, 13, 134 CrossRef CAS PubMed.
- U. B. Demirci, Int. J. Hydrogen Energy, 2023, 48, 29682–29698 Search PubMed.
- A. Ibrahim, M. Paskevicius and C. E. Buckley, Sustain. Energy Fuels, 2023, 7, 1196–1203 RSC.
- A. Ibrahim, M. Paskevicius, A. Patel, A. M. D’Angelo, T. D. Humphries and C. E. Buckley, ACS Appl. Energy Mater., 2024, 7, 11206–11217 CrossRef CAS.
- H. X. Nunes, D. L. Silva, C. M. Rangel and A. M. F. R. Pinto, Energies, 2021, 14, 3567 CrossRef CAS.
- Y. Liu, M. Paskevicius, T. D. Humphries and C. E. Buckley, Int. J. Hydrogen Energy, 2022, 47, 25347–25356 CrossRef CAS.
- Y. Kojima and T. Haga, Int. J. Hydrogen Energy, 2003, 28, 989–993 CrossRef CAS.
- T. Ou, A. Giuliano, M. Panizza, A. Barbucci and G. Cerisola, Int. J. Hydrogen Energy, 2013, 38, 15269–15274 CrossRef CAS.
- A. Patel, P. Ó. Conghaile, T. D. Humphries, M. Paskevicius and C. E. Buckley, Next Energy, 2025, 9, 100364 CrossRef.
- T. P. T. Nguyen, T. D. Humphries, H. Hagemann, C. E. Buckley, T. R. Jensen and M. Paskevicius, Dalton Trans., 2025, 54, 1199–1211 RSC.
- M. Wang, W. Guo, S. Wang, Y. Guo, J. Yang and T. Deng, J. Chem. Eng. Data, 2020, 65, 5184–5191 CrossRef CAS.
- M. Matsuo, Y. Nakamori, S. Orimo, H. Maekawa and H. Takamura, Appl. Phys. Lett., 2007, 91, 224103 CrossRef.
- H. Maekawa, M. Matsuo, H. Takamura, M. Ando, Y. Noda, T. Karahashi and S. Orimo, J. Am. Chem. Soc., 2009, 131, 894–895 Search PubMed.
- Y. Yan, J. B. Grinderslev, M. Jorgensen, L. N. Skov, J. Skibsted and T. R. Jensen, ACS Appl. Energy Mater., 2020, 3, 9264–9270 Search PubMed.
- Y. Yan, J. B. Grinderslev, T. Burankova, S. Wei, J. P. Embs, J. Skibsted and T. R. Jensen, J. Phys. Chem. Lett., 2022, 13, 2211–2216 Search PubMed.
- L. N. Skov, J. B. Grinderslev and T. R. Jensen, Batteries Supercaps, 2023, 6, e202300185 CrossRef CAS.
- M. B. Amdisen, J. B. Grinderslev, L. N. Skov and T. R. Jensen, Chem. Mater., 2023, 35, 1440–1448 CrossRef CAS.
- L. N. Skov, J. B. Grinderslev, A. Rosenkranz, Y. S. Lee and T. R. Jensen, Batteries Supercaps, 2022, 5, e202200163 CrossRef CAS.
- L. G. Kristensen, M. B. Amdisen, L. N. Skov and T. R. Jensen, Phys. Chem. Chem. Phys., 2022, 24, 18185–18197 RSC.
- Y. Yan, J. B. Grinderslev, Y.-S. Lee, M. Jørgensen, Y. W. Cho, R. Černý and T. R. Jensen, Chem. Commun., 2020, 56, 3971–3974 RSC.
- R. Zhang, W. Zhao, Z. Liu, S. Wei, Y. Yan and Y. Chen, Chem. Commun., 2021, 57, 2380–2383 Search PubMed.
- W. Zhao, R. Zhang, H. Li, Y. Zhang, Y. Wang, C. Wu, Y. Yan and Y. Chen, ACS Appl. Mater. Interfaces, 2021, 13, 31635–31641 Search PubMed.
- J. B. Grinderslev, L. N. Skov, J. G. Andreasen, S. Ghorwal, J. Skibsted and T. R. Jensen, Angew. Chem., Int. Ed., 2022, 134, e202203484 CrossRef.
- M. B. Amdisen and T. R. Jensen, Chem. Mater., 2025, 37, 1183–1194 CrossRef CAS.
- L. N. Skov, J. B. Grinderslev, T. S. S. Kjaer, L. R. Kristensen and T. R. Jensen, Angew. Chem., Int. Ed., 2025, 64, e202500613 CrossRef CAS PubMed.
- J. B. Grinderslev, L. N. Skov, L. R. Kristensen and T. R. Jensen, Angew. Chem., Int. Ed., 2025, 64, e202510493 CrossRef CAS PubMed.
- R. Černý, F. Murgia and M. Brighi, J. Alloys Compd., 2022, 895, 162659 Search PubMed.
- Z. Zhang and L. F. Nazar, Nat. Rev. Mater., 2022, 7, 389–405 CrossRef.
- M. Paskevicius, M. P. Pitt, D. H. Brown, D. A. Sheppard, S. Chumphongphan and C. E. Buckley, Phys. Chem. Chem. Phys., 2013, 15, 15825–15828 Search PubMed.
- N. Verdal, J.-H. Her, V. Stavila, A. V. Soloninin, O. A. Babanova, A. V. Skripov, T. J. Udovic and J. J. Rush, J. Solid State Chem., 2014, 212, 81–91 CrossRef CAS.
- I. M. Riddlestone, A. Kraft, J. Schaefer and I. Krossing, Angew. Chem., Int. Ed., 2018, 57, 13982–14024 Search PubMed.
- T. P. T. Nguyen, W. Gao, T. D. Humphries, Y. Zhong, C. E. Buckley and M. Paskevicius, J. Mater. Chem. A, 2025, 13, 28440–28450 RSC.
- L. Duchêne, D. H. Kim, Y. B. Song, S. Jun, R. Moury, A. Remhof, H. Hagemann, Y. S. Jung and C. Battaglia, Energy Storage Mater., 2020, 26, 543–549 CrossRef.
- G. Deysher, J. A. S. Oh, Y.-T. Chen, B. Sayahpour, S.-Y. Ham, D. Cheng, P. Ridley, A. Cronk, S. W.-H. Lin, K. Qian, L. H. B. Nguyen, J. Jang and Y. S. Meng, Nat. Energy, 2024, 9, 1161–1172 CAS.
- D. H. P. Souza, K. T. Møller, S. A. Moggach, T. D. Humphries, A. M. D'Angelo, C. E. Buckley and M. Paskevicius, J. Mater. Chem. A, 2021, 9, 15027–15037 RSC.
- D. H. P. Souza, T. D. Humphries, Y. Liu, A. Gradišek, A. M. D'Angelo, C. E. Buckley and M. Paskevicius, Sustain. Energy Fuels, 2022, 6, 4614–4625 RSC.
- D. H. P. Souza, A. M. D'Angelo, T. D. Humphries, C. E. Buckley and M. Paskevicius, Dalton Trans., 2022, 51, 13848–13857 Search PubMed.
- T. A. Hales, K. T. Møller, T. D. Humphries, A. M. D’Angelo, C. E. Buckley and M. Paskevicius, J. Phys. Chem. C, 2023, 127, 949–957 Search PubMed.
- T. A. Hales, K. T. Møller, T. D. Humphries, A. M. D’Angelo, C. E. Buckley and M. Paskevicius, Phys. Chem. Chem. Phys., 2023, 25, 31249–31256 RSC.
- S. R. H. Jensen, M. Jørgensen, T. P. T. Nguyen, G. Nolan, C. E. Buckley, T. R. Jensen and M. Paskevicius, Dalton Trans., 2024, 53, 7619–7627 Search PubMed.
- B. R. S. Hansen, M. Paskevicius, M. Jørgensen and T. R. Jensen, Chem. Mater., 2017, 29, 3423–3430 Search PubMed.
- M. Jørgensen, S. R. Jensen, T. D. Humphries, M. R. Rowles, M. V. Sofianos, C. E. Buckley, T. R. Jensen and M. Paskevicius, J. Phys. Chem. C, 2020, 124, 11340–11349 Search PubMed.
- A. Berger, A. Ibrahim, T. A. Hales, A. M. D'Angelo, C. E. Buckley and M. Paskevicius, Dalton Trans., 2024, 53, 3638–3653 Search PubMed.
- M. Green, H. Simonyan, K. Kaydanik and J. A. Teprovich, Appl. Sci., 2022, 12, 2273 CrossRef CAS.
- H. Simonyan, L. Zhong, M. M. Green, K. Movsesyan, A. Fraire, P. A. Ward, K. C. Lau and J. A. Teprovich, Jr., ACS Appl Mater Interfaces, 2024, 16, 70028–70037 Search PubMed.
- R. Mohtadi, M. Matsui, T. S. Arthur and S.-J. Hwang, Angew. Chem., Int. Ed., 2012, 51, 9780–9783 Search PubMed.
- I. Weber, J. Ingenmey, J. Schnaidt, B. Kirchner and R. J. Behm, ChemElectroChem, 2021, 8, 390–402 CrossRef CAS.
- F. Tuerxun, K. Yamamoto, M. Hattori, T. Mandai, K. Nakanishi, A. Choudhary, Y. Tateyama, K. Sodeyama, A. Nakao, T. Uchiyama, M. Matsui, K. Tsuruta, Y. Tamenori, K. Kanamura and Y. Uchimoto, ACS Appl. Mater. Interfaces, 2020, 12, 25775–25785 CrossRef CAS PubMed.
- S. Su, Y. NuLi, N. Wang, D. Yusipu, J. Yang and J. Wang, J. Electrochem. Soc., 2016, 163, D682 Search PubMed.
- D. Zhang, Y. Sun, X. Liu, Y. Zhang, R. Wang, Y. Zhao, M. Pan, Y. Wang, S. Chen, M. Zhou, Y. Chen, J. Yang, J. Wang and Y. NuLi, ACS Energy Lett., 2024, 9, 2685–2695 CrossRef CAS.
- N. T. Hahn, J. Self, K. S. Han, V. Murugesan, K. T. Mueller, K. A. Persson and K. R. Zavadil, J. Phys. Chem. B, 2021, 125, 3644–3652 CrossRef CAS PubMed.
- T. Watkins, A. Kumar and D. A. Buttry, J. Am. Chem. Soc., 2016, 138, 641–650 Search PubMed.
- O. Tutusaus, R. Mohtadi, T. S. Arthur, F. Mizuno, E. G. Nelson and Y. V. Sevryugina, Angew. Chem., Int. Ed., 2015, 54, 7900–7904 Search PubMed.
- O. Tutusaus, R. Mohtadi, N. Singh, T. S. Arthur and F. Mizuno, ACS Energy Lett., 2017, 2, 224–229 Search PubMed.
- R. Jay, A. W. Tomich, J. Zhang, Y. Zhao, A. De Gorostiza, V. Lavallo and J. Guo, ACS Appl. Mater. Interfaces, 2019, 11, 11414–11420 Search PubMed.
- H. Dong, O. Tutusaus, Y. Liang, Y. Zhang, Z. Lebens-Higgins, W. Yang, R. Mohtadi and Y. Yao, Nat. Energy, 2020, 5, 1043–1050 CrossRef CAS.
- D. Wang, X. Gao, Y. Chen, L. Jin, C. Kuss and P. G. Bruce, Nat. Mater., 2018, 17, 16–20 Search PubMed.
- S. A. McClary, D. M. Long, A. Sanz-Matias, P. G. Kotula, D. Prendergast, K. L. Jungjohann and K. R. Zavadil, ACS Energy Lett., 2022, 7, 2792–2800 CrossRef CAS.
- A. M. Melemed, D. A. Skiba, K. J. Steinberg, K.-H. Kim and B. M. Gallant, J. Phys. Chem. C, 2023, 127, 19886–19899 Search PubMed.
- Z. Yang, N. J. Leon, C. Liao, B. J. Ingram and L. Trahey, ACS Appl. Mater. Interfaces, 2023, 15, 25018–25028 Search PubMed.
- K. Kisu, S. Kim, T. Shinohara, K. Zhao, A. Züttel and S. Orimo, Sci. Rep., 2021, 11, 7563 CrossRef CAS PubMed.
- H. N. M. Sarangika, H. T. G. Shashintha, M. A. K. L. Dissanayake and G. K. R. Senadeera, J. Solid State Electrochem., 2024, 28, 2163–2173 Search PubMed.
- Y. Shao, N. N. Rajput, J. Hu, M. Hu, T. Liu, Z. Wei, M. Gu, X. Deng, S. Xu, K. S. Han, J. Wang, Z. Nie, G. Li, K. R. Zavadil, J. Xiao, C. Wang, W. A. Henderson, J.-G. Zhang, Y. Wang, K. T. Mueller, K. Persson and J. Liu, Nano Energy, 2015, 12, 750–759 Search PubMed.
- X. Zhang, T. Zhang, Y. Shao, H. Cao, Z. Liu, S. Wang and X. Zhang, ACS Sustain. Chem. Eng., 2021, 9, 5396–5404 CrossRef CAS.
- P. Wang, J. Trück, J. Häcker, A. Schlosser, K. Küster, U. Starke, L. Reinders and M. R. Buchmeiser, Energy Storage Mater., 2022, 49, 509–517 CrossRef.
- T. Shinohara, K. Kisu, A. Dorai, K. Zushida, H. Yabu, S. Takagi and S. Orimo, Adv. Sci., 2024, 11, 2308318 CrossRef CAS PubMed.
- M. Green, K. Kaydanik, M. Orozco, L. Hanna, M. A. T. Marple, K. A. S. Fessler, W. B. Jones, V. Stavila, P. A. Ward and J. A. Teprovich Jr., Adv. Sci., 2022, 9, 2106032 CrossRef CAS PubMed.
- K. Bao, Y. Pang, J. Yang, D. Sun, F. Fang and S. Zheng, Sci. China Mater., 2022, 65, 95–104 Search PubMed.
- A. Du, H. Zhang, Z. Zhang, J. Zhao, Z. Cui, Y. Zhao, S. Dong, L. Wang, X. Zhou and G. Cui, Adv. Mater., 2019, 31, 1805930 Search PubMed.
- J. Guschlbauer, L. Niedzicki, L. Jacob, E. Rzeszotarska, D. Pociecha and P. Kaszyński, J. Mol. Liq., 2023, 377, 121525 CrossRef CAS.
- L. Jacob, L. Niedzicki, R. Jakubowski, D. Pociecha and P. Kaszyński, Dalton Trans., 2024, 53, 10293–10302 RSC.
- E. Wigner and H. B. Huntington, J. Chem. Phys., 1935, 3, 764–770 Search PubMed.
- N. W. Ashcroft, Phys. Rev. Lett., 1968, 21, 1748–1749 CrossRef CAS.
- N. W. Ashcroft, Phys. Rev. Lett., 2004, 92, 187002 CrossRef CAS PubMed.
- F. Peng, Y. Sun, C. J. Pickard, R. J. Needs, Q. Wu and Y. Ma, Phys. Rev. Lett., 2017, 119, 107001 Search PubMed.
- A. P. Drozdov, P. P. Kong, V. S. Minkov, S. P. Besedin, M. A. Kuzovnikov, S. Mozaffari, L. Balicas, F. F. Balakirev, D. E. Graf, V. B. Prakapenka, E. Greenberg, D. A. Knyazev, M. Tkacz and M. I. Eremets, Nature, 2019, 569, 528–531 CrossRef CAS PubMed.
- P. Kong, V. S. Minkov, M. A. Kuzovnikov, A. P. Drozdov, S. P. Besedin, S. Mozaffari, L. Balicas, F. F. Balakirev, V. B. Prakapenka, S. Chariton, D. A. Knyazev, E. Greenberg and M. I. Eremets, Nat. Commun., 2021, 12, 5075 CrossRef CAS PubMed.
- W. Chen, D. V. Semenok, X. Huang, H. Shu, X. Li, D. Duan, T. Cui and A. R. Oganov, Phys. Rev. Lett., 2021, 127, 117001 Search PubMed.
- D. V. Semenok, A. G. Kvashnin, A. G. Ivanova, V. Svitlyk, V. Y. Fominski, A. V. Sadakov, O. A. Sobolevskiy, V. M. Pudalov, I. A. Troyan and A. R. Oganov, Mater. Today, 2020, 33, 36–44 Search PubMed.
- Z. Li, X. He, C. Zhang, X. Wang, S. Zhang, Y. Jia, S. Feng, K. Lu, J. Zhao, J. Zhang, B. Min, Y. Long, R. Yu, L. Wang, M. Ye, Z. Zhang, V. Prakapenka, S. Chariton, P. A. Ginsberg, J. Bass, S. Yuan, H. Liu and C. Jin, Nat. Commun., 2022, 13, 2863 Search PubMed.
- S. Mozaffari, D. Sun, V. S. Minkov, A. P. Drozdov, D. Knyazev, J. B. Betts, M. Einaga, K. Shimizu, M. I. Eremets, L. Balicas and F. F. Balakirev, Nat. Commun., 2019, 10, 2522 Search PubMed.
- F. Hong, P. F. Shan, L. X. Yang, B. B. Yue, P. T. Yang, Z. Y. Liu, J. P. Sun, J. H. Dai, H. Yu, Y. Y. Yin, X. H. Yu, J. G. Cheng and Z. X. Zhao, Mater. Today Phys., 2022, 22, 100596 Search PubMed.
- J. Bi, Y. Nakamoto, P. Zhang, K. Shimizu, B. Zou, H. Liu, M. Zhou, G. Liu, H. Wang and Y. Ma, Nat. Commun., 2022, 13, 5952 Search PubMed.
- L.-C. Chen, T. Luo, Z.-Y. Cao, P. Dalladay-Simpson, G. Huang, D. Peng, L.-L. Zhang, F. A. Gorelli, G.-H. Zhong, H.-Q. Lin and X.-J. Chen, Nat. Commun., 2024, 15, 1809 Search PubMed.
- D. V. Semenok, I. A. Troyan, A. G. Ivanova, A. G. Kvashnin, I. A. Kruglov, M. Hanfland, A. V. Sadakov, O. A. Sobolevskiy, K. S. Pervakov, I. S. Lyubutin, K. V. Glazyrin, N. Giordano, D. N. Karimov, A. L. Vasiliev, R. Akashi, V. M. Pudalov and A. R. Oganov, Mater. Today, 2021, 48, 18–28 Search PubMed.
- S. Chen, Y. Wang, F. Bai, X. Wu, X. Wu, A. Pakhomova, J. Guo, X. Huang and T. Cui, J. Am. Chem. Soc, 2024, 146, 14105–14113 CrossRef CAS PubMed.
- J. Bardeen, L. N. Cooper and J. R. Schrieffer, Phys. Rev., 1957, 106, 162–164 Search PubMed.
- J. E. Hirsch and F. Marsiglio, Matter Radiat. Extremes, 2022, 7, 058401 Search PubMed.
- J. E. Hirsch, Nat. Sci. Rev., 2023, 11, nwad174 CrossRef PubMed.
- P. Bhattacharyya, W. Chen, X. Huang, S. Chatterjee, B. Huang, B. Kobrin, Y. Lyu, T. J. Smart, M. Block, E. Wang, Z. Wang, W. Wu, S. Hsieh, H. Ma, S. Mandyam, B. Chen, E. Davis, Z. M. Geballe, C. Zu, V. Struzhkin, R. Jeanloz, J. E. Moore, T. Cui, G. Galli, B. I. Halperin, C. R. Laumann and N. Y. Yao, Nature, 2024, 627, 73–79 Search PubMed.
- M. Du, H. Huang, Z. Zhang, M. Wang, H. Song, D. Duan and T. Cui, Adv. Sci., 2024, 11, 2408370 Search PubMed.
- K. Dolui, L. J. Conway, C. Heil, T. A. Strobel, R. P. Prasankumar and C. J. Pickard, Phys. Rev. Lett., 2024, 132, 166001 CrossRef CAS PubMed.
- S. Di Cataldo and L. Boeri, Phys. Rev. B, 2023, 107, L060501 CrossRef CAS.
- N. Porcellino, C. Pakhanyan, T. Elmslie, M. Shivanna, S. Duwal, C. D. Spataru, R. Lindvall, M. Somayazulu, P. A. Ward, S. I. Kawaguchi, V. Stavila, P. A. Sharma and J. A. Teprovich, ACS Appl. Energy Mater., 2024, 7, 9608–9615 CrossRef CAS.
- M. Shivanna, T. A. Elmslie, C. D. Spataru, S. Duwal, N. Porcellino, S. Mouzaya, C. P. Pakhanyan, F. El Gabaly, S. Kawaguchi-Imada, J. Guo, Z. Zhuo, M.-J. Kim, B. T. Sturtevant, J. A. Teprovich, M. D. Allendorf, P. A. Sharma and V. Stavila, ACS Appl. Energy Mater., 2024, 8, 7–15 CrossRef.
- H. Xu, O. Schneble, R. Jaramillo, M. Polanski and J. Li, Phys. Rev. Lett., 2025 Search PubMed , submitted.
- J. Huot, F. Cuevas, S. Deledda, K. Edalati, Y. Filinchuk, T. Grosdidier, B. C. Hauback, M. Heere, T. R. Jensen, M. Latroche and S. Sartori, Materials, 2019, 12, 2778 Search PubMed.
- R. A. Varin, R. Parviz, M. Polanski and Z. S. Wronski, Int. J. Hydrogen Energy, 2014, 39, 10585–10599 Search PubMed.
- A. D. Katsenis, A. Puskaric, V. Strukil, C. Mottillo, P. A. Julien, K. Uzarevic, M. H. Pham, T. O. Do, S. A. Kimber, P. Lazic, O. Magdysyuk, R. E. Dinnebier, I. Halasz and T. Friscic, Nat. Commun., 2015, 6, 6662 Search PubMed.
- H. Li, J. Huang, K. Yang, Z. Lu, S. Yan, H. Su, C. Liu, X. Wang and B. Ren, J. Phys. Chem. Lett., 2022, 13, 479–485 Search PubMed.
- J. Batteas, K. G. Blank, E. Colacino, F. Emmerling, T. Friščić, J. Mack, J. Moore, M. E. Rivas and W. Tysoe, RSC Mechanochem., 2025, 2, 10–19 Search PubMed.
- V. Martinez, T. Stolar, B. Karadeniz, I. Brekalo and K. Užarević, Nat. Rev. Chem., 2023, 7, 51–65 Search PubMed.
- C. Pistidda, A. Santhosh, P. Jerabek, Y. Shang, A. Girella, C. Milanese, M. Dore, S. Garroni, S. Bordignon, M. R. Chierotti, T. Klassen and M. Dornheim, J. Phys. Energy, 2021, 3, 044001 Search PubMed.
- A. K. Patel, D. Siemiaszko, J. Dworecka-Wójcik and M. Polański, Int. J. Hydrogen Energy, 2022, 47, 5361–5371 Search PubMed.
- I. Wyrębska, K. Tomczyk, D. Siemiaszko, D. Zasada, J. Dworecka-Wójcik, M. Pęska, R. Chulist, S. Koter and M. Polański, Chem. Eng. J., 2024, 485, 149673 Search PubMed.
- T. Friscic, C. Mottillo and H. M. Titi, Angew. Chem., Int. Ed., 2020, 59, 1018–1029 Search PubMed.
- T. N. Dymova, Z. K. Sterlyadkina and V. G. Safronov, Russ. J. Inorg. Chem., 1961, 6, 389–395 Search PubMed.
- E. Wiberg, H. Goeltzer and R. Bauer, Z. Naturforsch., B, 1951, 394–395 Search PubMed.
- J. Huot, M.-L. Tremblay and R. Schulz, J. Alloys Compd., 2003, 356, 603–607 Search PubMed.
- A. Baran, T. R. Jensen and M. Polański, Adv. Eng. Mater., 2024, 27, 2400435 Search PubMed.
- A. Baran, T. R. Jensen and M. Polański, J. Energy Storage, 2024, 103, 114271 Search PubMed.
- A. Baran, T. R. Jensen and M. Polański, J. Energy Storage, 2024, 103, 114272 CrossRef.
- L. Yu, R. Qu, S. Jiang, C. Hu, Y. Wang, L. Xu, S. Li and X. Du, Renew. Sustain. Energy Rev., 2026, 225, 116151 Search PubMed.
- H. Gudla, A. Hockmann, D. Brandell and J. Mindemark, ACS Appl. Polym. Mater., 2025, 7, 4716–4724 Search PubMed.
- R. Asakura, Z. Łodziana, R. Grissa, D. Rentsch, C. Battaglia and A. Remhof, ACS Appl. Energy Mater., 2025, 8, 9637–9645 Search PubMed.
- M. Brighi, F. Murgia and R. Černý, Cell Rep. Phys. Sci., 2020, 1, 100217 CrossRef CAS.
- M. Paskevicius, B. R. S. Hansen, M. Jorgensen, B. Richter and T. R. Jensen, Nat Commun, 2017, 8, 15136 CrossRef PubMed.
- Y.-S. Lee, Y. Filinchuk, H.-S. Lee, J.-Y. Suh, J. W. Kim, J.-S. Yu and Y. W. Cho, J. Phys. Chem. C, 2011, 115, 10298–10304 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |