Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Kolbe radical-initiated electrochemical desulfurization of thioamides under aerobic conditions
Subban
Kathiravan
 * and
Ian A.
Nicholls
* and
Ian A.
Nicholls

Bioorganic & Biophysical Chemistry Laboratory, Linnaeus University Centre for Biomaterials Chemistry, Department of Chemistry & Biomedical Sciences, Linnaeus University, Kalmar, SE-391 82, Sweden. E-mail: suppan.kathiravan@lnu.se
First published on 26th January 2026
Abstract
A metal-free electrochemical desulfurization of thioamides is reported, enabled by an isobutyric acid-mediated Kolbe radical initiation under air. The protocol operates in an undivided cell, tolerates diverse substrates, and proceeds via radical activation and oxygen trapping, offering a mechanistically distinct, sustainable alternative to conventional oxidative desulfurization methods.
Thioamides are valuable synthetic intermediates and structural motifs that serve as amide bioisosteres in medicinal chemistry and are frequently encountered as ligands in coordination chemistry.1 Because amides are among the most prevalent functional groups in pharmaceuticals, natural products, and materials, the selective conversion of thioamides to amides, commonly referred to as desulfurization, has become a transformation of significant synthetic importance.2,3 Thioamides are synthetically versatile precursors since the C
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S bond is more nucleophilic and electronically distinct than the C
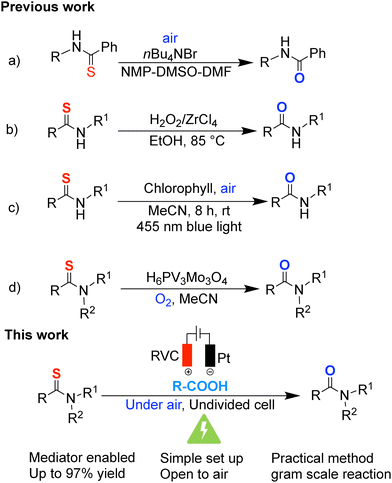
S bond is more nucleophilic and electronically distinct than the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond, enabling unique reactivity and selective functionalization strategies.4,5 However, conventional desulfurization methods typically rely on stoichiometric transition-metal reagents such as Ru- and Mn-based systems,6–8 harsh reducing agents, such as Raney® nickel,9,10 strong Lewis acids such as tris(pentafluorophenyl)borane.11 Established desulfurization protocols often depend on elevated temperatures, metal catalysts, external oxidants, or photochemical setups (Fig. 1a–d).12–15 In contrast, the present work enables catalyst-free electrochemical desulfurization under ambient conditions.
O bond, enabling unique reactivity and selective functionalization strategies.4,5 However, conventional desulfurization methods typically rely on stoichiometric transition-metal reagents such as Ru- and Mn-based systems,6–8 harsh reducing agents, such as Raney® nickel,9,10 strong Lewis acids such as tris(pentafluorophenyl)borane.11 Established desulfurization protocols often depend on elevated temperatures, metal catalysts, external oxidants, or photochemical setups (Fig. 1a–d).12–15 In contrast, the present work enables catalyst-free electrochemical desulfurization under ambient conditions.
Organic electrochemistry has emerged as a powerful tool for achieving redox transformations under environmentally benign conditions.16,17 By using electricity as a traceless redox reagent, electrochemical methods can replace stoichiometric oxidants or reductants, reduce waste generation, and enable unique reaction outcomes that are difficult to achieve under conventional thermal or photochemical conditions.18–20 Among these, Kolbe decarboxylation offers a predictable and controllable means of radical generation, as carboxylate oxidation occurs readily under constant-current conditions.21,22 As a result, for the substrates that does not undergo direct anodic oxidation, Kolbe electrolysis is an indirect method to generate radicals under electrochemical conditions.22 Electrochemical strategies for desulfurization offer an attractive alternative, as they can generate reactive intermediates in a controlled manner and allow fine-tuning of reaction selectivity through electrode material and applied potential. Despite these advantages, electrochemical desulfurization remains underexplored, with only limited activation modes reported to date. During the preparation of this manuscript, Rybicka-Jasinska and co-workers disclosed the first electrochemical desulfurization of thioamides, proceeding via direct anodic oxidation of the substrate.23 In contrast, the present work introduces a mechanistically distinct strategy based on carboxylate-mediated electrochemical activation, in which anodic Kolbe decarboxylation generates alkyl radicals that decouple substrate oxidation from electrode potential. This indirect activation mode enables selective C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S to C
S to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O conversion under constant-current electrolysis using air as the sole oxidant. As part of our continuing efforts toward developing sustainable electrochemical transformations,24–27 we herein report an isobutyric acid–enabled electrochemical desulfurization of thioamides under ambient air conditions.
O conversion under constant-current electrolysis using air as the sole oxidant. As part of our continuing efforts toward developing sustainable electrochemical transformations,24–27 we herein report an isobutyric acid–enabled electrochemical desulfurization of thioamides under ambient air conditions.
We began by investigating the electrochemical desulfurization of N,N-disubstituted thioamide (1) under various conditions (Table 1). The standard conditions, employing a reticulated vitreous carbon (RVC) anode, platinum cathode, nBu4NBF4 as the electrolyte, triethylamine (TEA) as base, isobutyric acid as additive, and a DCE/TFE solvent mixture under constant current (10 mA) in air, afforded the desired amide in 98% yield (entry 1). Control experiments revealed that electricity is essential, as no significant conversion was observed in its absence (entry 2). Similarly, omission of isobutyric acid, air, TFE, or TEA led to either complete loss of reactivity or significantly diminished yields (entries 3–6), underscoring the importance of each component. Solvent screening showed that replacing DCE with MeCN or EtOH completely suppressed product formation (entry 7), highlighting the necessity of a mixed solvent system with TFE as a co-solvent to stabilize key intermediates.28 Electrode material proved crucial as as replacing either the RVC anode with graphite or the platinum cathode with RVC completely suppressed reactivity (entries 8 & 9). Among the electrolytes examined, nBu4NBF4 was the optimal electrolyte, while other salts or reduced electrolyte loading resulted in diminished or no reactivity (entries 10–14). Reaction efficiency was strongly current-dependent, with 10 mA proving optimal, while lower currents led to diminished or negligible conversion (entry 15).
| Entry | Deviation from standard conditions | Yieldb (%) |
|---|---|---|
| a Reaction conditions: RVC anode, Pt plate cathode, 1 (0.26 mmol), iPrCO2H (2 equiv.), TFE (2 equiv.), TEA (2 equiv.), nBu4NBF4 (2 equiv.) 1,2-DCE (3 mL), 10 mA, undivided cell. b Isolated yield. NR = No reaction. | ||
| 1 | None | 98 |
| 2 | No electricity | Trace |
| 3 | No iPrCO2H | 2 |
| 4 | No air | 0 |
| 5 | No TFE | 13 |
| 6 | No TEA | 15 |
| 7 | CH3CN or EtOH instead of DCE | 0 |
| 8 | Graphite anode | 0 |
| 9 | RVC cathode | 0 |
| 10 | nBu4NPF6 | Trace |
| 11 | nBu4NBr | 58 |
| 12 | nBu4NOAc | 25 |
| 13 | LiClO4 | 32 |
| 14 | nBu4NBF4 (1 equiv.) | NR |
| 15 | 1, 3, 5 & 7 mA | Trace, 21, 33 & 22 |
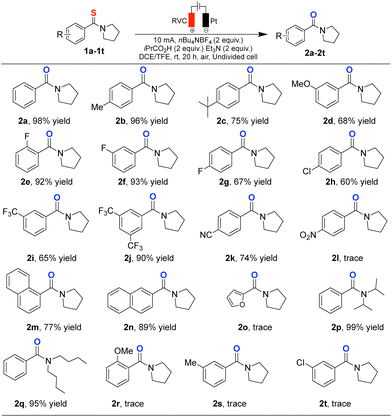
With the optimized electrochemical conditions in hand, we set out to explore the scope and limitations of this desulfurization protocol. Initially, unsubstituted benzothioamide (1a) underwent smooth conversion to the corresponding benzamide in 98% isolated yield (Scheme 1). Substrates bearing electron-donating groups on the aryl ring, such as 4-methyl and 4-tert-butyl, afforded the desired amides (2b–c) in 96% and 75% yields, respectively. The presence of a 3-methoxy group was tolerated, albeit with a slightly diminished yield of 68% (2d). Electron-withdrawing substituents also participated well: 2- and 3-fluorothiobenzamides delivered the products in 92% and 93% yields (2e–f), whereas 4-fluoro and 4-chloro provided moderate yields of 67% and 60% (2g–h), respectively. Strongly electron-withdrawing groups such as trifluoromethyl were tolerated; 3-CF3 gave 65% yield (2i), while 3,5-diCF3 led to a much improved 90% yield (2j). 4-Cyanothiobenzamide afforded the product in 74% yield (2k), whereas 4-nitro substrate resulted only in trace conversion (2l), likely due to its low oxidation potential under the electrochemical conditions. 1-Naphthyl thioamide gave a 77% yield (2m), and 2-naphthyl thioamide provided an even higher yield of 89% (2n). To evaluate the influence of the amine moiety, several secondary thioamides were examined. Thioamides derived from dibutylamine and diisopropylamine furnished the corresponding amides in 99% and 95% yields (2p–q), respectively, underscoring the compatibility of bulky secondary amines. Despite the broad applicability of the method, certain substrates failed to undergo productive desulfurization (2r–t). The present protocol is currently limited to aromatic benzothioamides. A representative aliphatic thioamide did not afford the corresponding amide under the optimized conditions, indicating that extension to aliphatic thioamides is not straightforward under the current electrochemical parameters (see SI).
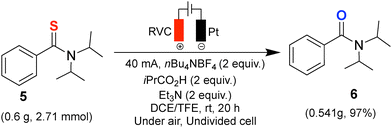
To demonstrate the scalability and practicality of this protocol, we performed the electrochemical desulfurization of N,N-diisopropylbenzothioamide (6) on a gram scale under the optimized conditions (Scheme 2). The reaction was conducted in an undivided cell equipped with an RVC anode and platinum cathode at a constant current of 40 mA for 20 h, providing the desired amide in 97% isolated yield (0.541 g) as a brown liquid.
A series of control experiments was conducted to gain insight into the reaction mechanism (Scheme 3). The addition of radical scavengers had a pronounced effect on the reaction outcome. In the presence of TEMPO, amide formation was completely suppressed, while BHT led to a significant decrease in yield. (Scheme 3a). Attempts to probe the oxygen source by H218O labelling did not result in detectable isotope incorporation, suggesting that water is not the primary oxygen donor (Scheme 3b). Product formation correlated with aliphatic carboxylic acids capable of Kolbe-type decarboxylation, indicating the acid functions as a radical precursor rather than a proton source (Scheme 3c). The reaction was also strongly inhibited in the presence of p-benzoquinone, a known scavenger of O2-derived reactive oxygen species, supporting the involvement of an oxygen-centered species such as superoxide in the later stages of the transformation (Scheme 3d).
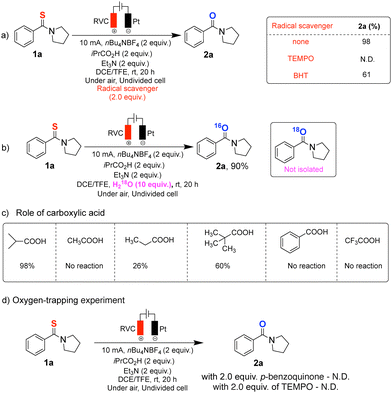 | ||
| Scheme 3 Mechanistic studies: (a) Radical scavenger studies. (b) Isotope incorporation experiment. (c) Evaluation of the role of carboxylic acids. (d) Oxygen-trapping experiments. | ||
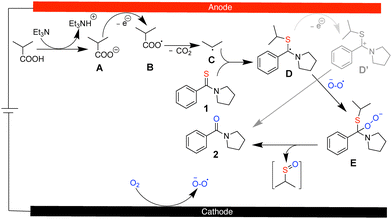
Based on the mechanistic studies and literature precedence,14,15,23 we propose the following mechanism for the isobutyric acid enabled electrochemical desulfurization of thioamides, as outlined in Scheme 4. Triethylamine (Et3N) first deprotonates isobutyric acid to generate the corresponding isobutyrate anion (iPrCO2−) (A). At the anode, iPrCO2− undergoes single-electron oxidation (Kolbe decarboxylation) to afford an isobutyryl radical (iPrCO2˙) (B), which rapidly decarboxylates to generate an isopropyl radical (iPr˙) (C) and CO2. The resulting iPr˙ then adds to the thioamide (1) in solution to produce an isopropylthio radical (D). Alternatively, radical D may undergo anodic oxidation to generate the corresponding cationic species D′. The radical is subsequently trapped by molecular oxygen radical under aerobic conditions to furnish a peroxyl radical intermediate (E). This intermediate undergoes fragmentation, effecting C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) S to C
S to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O conversion and producing the corresponding amide (2) along with sulfur-containing byproducts. At present, the detailed mechanism of the final oxygenation and sulfur extrusion remains unclear. At the cathode, either proton reduction or oxygen reduction may occur to maintain charge balance under constant current electrolysis. The requirement for aerobic conditions suggests that cathodic oxygen reduction could also contribute by forming superoxide, which may participate in the propagation steps.29
O conversion and producing the corresponding amide (2) along with sulfur-containing byproducts. At present, the detailed mechanism of the final oxygenation and sulfur extrusion remains unclear. At the cathode, either proton reduction or oxygen reduction may occur to maintain charge balance under constant current electrolysis. The requirement for aerobic conditions suggests that cathodic oxygen reduction could also contribute by forming superoxide, which may participate in the propagation steps.29
In summary, we have developed an Kolbe electrochemical protocol for the efficient desulfurization of thioamides to form amides under ambient air. Compared to simple aerobic oxidation of thioamides, which typically proceeds through direct oxidative activation of the substrate, the present Kolbe-enabled electrochemical strategy operates via indirect radical activation. This distinction allows the transformation to proceed under mild, constant-current conditions using air as the sole oxidant, avoiding the elevated temperatures, metal catalysts, or strongly oxidative reaction environments often required for direct aerobic oxidation. This study establishes a mechanistically distinct and sustainable electrochemical pathway for thioamide desulfurization and highlights the potential of carboxylic-acid-mediated radical generation as a general strategy for electroorganic synthesis.
SK conceived and designed the experiments and performed the data analysis. IAN acquired funding, validation and supervision. All authors contributed to manuscript writing and approved the final version.
We thank Linnaeus University, Swedish Research Council, grant number 2023-03406; the Helge Ax:son Johnson Foundation, grant numbers 2019-0318 and 2022-0317; the Swedish Knowledge Foundation, Synergi22, grant No. 20230019; and the Crafoord Foundation, grant numbers 2019-0925 and 2020-0775 for generous funding. We are thankful to Dr Subramanian Suriyanarayanan for his assistance with cyclic voltammetry experiments.
Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article (General procedure, cyclic voltammetric studies, NMR data and spectra) have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5cc05504k.References
- S. Kumari, A. V. Carmona, A. K. Tiwari and P. C. Trippier, J. Med. Chem., 2020, 63, 12290–12358 Search PubMed.
- A. K. Ghose, V. N. Viswanadhan and J. J. Wendoloski, J. Comb. Chem., 1999, 1, 55–68 CrossRef CAS PubMed.
- T. S. Jagodziński, Chem. Rev., 2002, 197–228 Search PubMed.
- J. Lou, Q. Wang, P. Wu, H. Wang, Y.-G. Zhou and Z. Yu, Chem. Soc. Rev., 2020, 49, 4307–4359 Search PubMed.
- S. G. Modha, V. P. Mehta and E. V. V. D. Eycken, Chem. Soc. Rev., 2013, 42, 5042–5055 Search PubMed.
- R. Sharma, G. Kenguva, R. Dandelab and P. Daw, Chem. Commun., 2025, 61, 9460–9463 Search PubMed.
- Z. Wang, S. Chen, C. Chen, Y. Yang and C. Wang, Angew. Chem., Int. Ed., 2022, 62, e202215963 Search PubMed.
- J. Luo, M. Rauch, L. Avram, Y. Ben-David and D. Milstein, J. Am. Chem. Soc., 2020, 142, 21628–21633 Search PubMed.
- E. C. Kornfeld, J. Org. Chem., 1951, 16, 131–138 Search PubMed.
- C. D. Hurd and B. Rudner, J. Am. Chem. Soc., 1951, 73, 5157–5160 CrossRef CAS.
- P. Nad, S. Goswami, H. K. Kisan and A. Mukherjee, Chem. – Eur. J., 2025, 31, e202500738 Search PubMed.
- M. S. K. Inamoto, K. Hiroya and T. Doi, Synthesis, 2010, 3087–3090 Search PubMed.
- M. M. K. K. Bahrami and Y. Tirandaz, Synthesis, 2009, 369–371 Search PubMed.
- Z. Yang, H. Zhou, L. Wang, J. Zhang, H. Xie, Y. Liu, J. Zengab and P. Cheng, RSC Adv., 2022, 12, 17190–17193 RSC.
- N. Xu, X. Jin, K. Suzuki, K. Yamaguchia and N. Mizuno, New J. Chem., 2016, 40, 4865–4869 Search PubMed.
- C. Zhu, N. W. J. Ang, T. H. Meyer, Y. Qiu and L. Ackermann, ACS Cent. Sci., 2021, 7, 415–431 CrossRef CAS PubMed.
- D. Polloka and S. R. Waldvogel, Chem. Sci., 2020, 11, 12386–12400 RSC.
- B. A. Frontana-Uribe, R. D. Little, J. G. Ibanez, A. Palmad and R. Vasquez-Medranoc, Green Chem., 2010, 12, 2099–2119 RSC.
- E. J. Horn, B. R. Rosen and P. S. Baran, ACS Cent. Sci., 2016, 2, 302–308 Search PubMed.
- S. Tang, L. Zeng and A. Lei, J. Am. Chem. Soc., 2018, 140, 13128–13135 Search PubMed.
- I. G. M. Quertenmont, K. Lam, I. Marko and O. Riant, Org. Lett., 2020, 22, 1771–1775 Search PubMed.
- K. L. M. C. Leech, Acc. Chem. Res., 2020, 53, 121–134 Search PubMed.
- Z. Bienkowska, I. Khan, M. Skompska and K. R. Janiska, Chem. – Eur. J., 2025, 31, e02278 Search PubMed.
- S. Kathiravan and P. Anaspure, Org. Biomol. Chem., 2023, 21, 2024–2033 Search PubMed.
- S. Kathiravan, S. Suriyanarayanan and I. A. Nicholls, Org. Lett., 2019, 21, 1968–1972 Search PubMed.
- S. Kathiravan and I. A. Nicholls, Curr. Res. Green Sustainable Chem., 2024, 8, 100405 Search PubMed.
- S. Kathiravan and I. A. Nicholls, ACS Org. Inorg. Au, 2025, 405–406 Search PubMed.
- L. Schulz, J.-Å. Husmann and S. R. Waldvogel, Electrochim. Acta, 2020, 337, 135786 CrossRef CAS.
- T. N. E. Katori, T. Kunieda and M. Hirobe, Chem. Pharm. Bull., 1981, 29, 3075–3077 CrossRef PubMed.
| This journal is © The Royal Society of Chemistry 2026 |

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Reaction conditions: RVC anode, Pt plate cathode, 1 (0.26 mmol), iPrCO2H (2 equiv.), TFE (2 equiv.), TEA (2 equiv.),
Reaction conditions: RVC anode, Pt plate cathode, 1 (0.26 mmol), iPrCO2H (2 equiv.), TFE (2 equiv.), TEA (2 equiv.),