Open Access Article
Open Access ArticleLow-dimensional transition metal dichalcogenide heterostructure photoanodes for photoelectrochemical hydrogen evolution application: recent progress and prospects
Zhiyuan
Peng†
a,
Yilu
Su†
b,
Imane
En-Naji
a,
Amir
Khojastehnezhad
a and
Mohamed
Siaj
 *a
*a
aFaculty of Engineering, Université de Sherbrooke, 2500 Blvd de l’Université, Sherbrooke, QC J1K 2R1, Canada. E-mail: mohamed.siaj@usherbrooke.ca
bDepartment of Chemistry, Université du Québec à Montréal, Montréal, QC H3C 3P8, Canada
First published on 7th January 2026
Abstract
Solar-driven hydrogen evolution through photoelectrochemical (PEC) water splitting technology provides a prospective approach for green energy production. To accomplish reliable PEC systems with sufficient solar-to-hydrogen conversion efficiencies (STH, ≥10%), one of the current primary challenges lies in the design and fabrication of highly-performing semiconductor (SC) photoanodes to overcome its high overpotential requirement and sluggish surface oxidation kinetics. The emergence of low-dimensional layered transition metal dichalcogenides (TMDs) with extraordinary electronic and optical properties has gained considerable attention and they are inarguably promising photoanode candidates. The rational combination of TMDs interfaced with other SC photoabsorbers via energy band modulation and heterojunction formation can markedly improve PEC performance and solar conversion. In this context, this review begins with a description of the PEC water oxidation mechanism, efficiency-related parameters, band bending and charge transfer behavior within n-type SC photoanodes, followed by an overview of recent progress and our contributions in fabricating efficient TMD-based heterostructure photoanodes with various synthetic routes and architectures. Next, the unique superiorities and positive effects of TMD utilization, such as optimized light harvesting, regulated electron transfer channels, promoted charge separation and transport, and improved long-term photostability, were comprehensively summarized in various TMD/SC heterostructure photoanode systems. Finally, the remaining challenges and future opportunities in advancing TMD-based van der Waals heterostructure photoanodes for next generation PEC water splitting applications are addressed.
1. Introduction
Developing a global-scale green hydrogen economy is one of the most promising strategies to address the escalating energy supply challenges facing humanity and decarbonize. According to the Statistical Review of World Energy in 2024, approximately 81.3% of the total energy demand relies on the extensive combustion of non-renewable fossil fuels (e.g., oil, coal, and natural gas), leading to energy shortages and environmental degradation. Therefore, a swift transition from traditional fossil fuels to clean and sustainable energy sources is imperative. Hydrogen (H2), known for its eco-friendly characteristic of zero carbon emissions and excellent gravimetric heating value (142 MJ kg−1), is poised to be a viable alternative energy source.1–3 Sunlight-driven hydrogen generation through photoelectrochemical (PEC) water-splitting can directly convert renewable solar energy into storable hydrogen fuel in an environmentally friendly and cost-effective manner, attracting significant research interest.4 In a typical PEC system, semiconductor electrodes are photoexcited to generate electron–hole pairs under sunlight irradiation, driving the photocathodic hydrogen evolution reaction (HER) and the photoanodic oxygen evolution reaction (OER). Compared with conventional powder photocatalytic or photovoltaic–electrocatalytic hydrolysis, this system accomplishes direct solar-to-hydrogen (STH) conversion with a subtle balance between energy efficiency and device complexity. Over the past few decades, PEC water splitting technology has achieved significant progress but is still far from industrial scale-up due to insufficient STH efficiency and long-term durability. Overcoming these limitations necessitates ongoing innovation in photoelectrode fabrication and device architecture.5 Noteworthily, relative to the HER, the OER is particularly complex, involving a four-proton coupled electron exchange process, which necessitates a photoanode with superior charge transfer and injection properties to overcome the higher overpotential requirement and sluggish oxidation kinetics.6,7 Despite substantial efforts to enhance photoanodes, the OER remains the rate-limiting step and a major bottleneck in overall PEC water splitting.An ideal OER photoanode should be low-cost, physiochemically stable, and exhibit a suitable energy band structure that allows for visible-light absorption and effective water oxidation. On the basis of these principles, low-dimensional transition metal dichalcogenides (TMDs), especially two-dimensional (2D) TMDs, have been thoroughly investigated and present great potential in energy harvesting, conversion, and storage applications because of their narrow bandgaps, large surface-to-volume atomic ratio, stackability, high charge mobility, short carrier diffusion distance, and thickness-dependent photoelectronic properties.8–10 Specifically, TMDs crystallize in a layered structure comprising a transition metal plane sandwiched by two chalcogen planes, where adjacent layers are held together via weak van der Waals forces. Such interactions facilitate easy exfoliation of TMD materials into their few/mono layer, which generally exhibit superior conductivity, mechanical robustness, and structural stability, along with an indirect-to-direct bandgap transition relative to their bulk phase.11–13 In addition, the diverse combinations of transition metals (Mo, W, and Ti) and chalcogen atoms (S, Se, and Te) give TMDs versatile structural phases, where variations in atomic coordination yield metallic, semiconducting, or semimetallic properties.14 Thanks to these unique and extraordinary properties, low-dimensional layered TMDs have emerged as promising photoanode materials. However, in many cases, the single-component TMD photocatalysts show non-photoactive to very weak PEC performance due to the low utilization of solar energy and high recombination of photogenerated charge carriers. Fabricating TMD-based heterostructure photoanodes with synergistic effects, through rationally integrating with secondary photoabsorbers to form multi-heterojunctions, p–n junctions, or Schottky junctions, can effectively broaden spectral absorption, boost charge separation and transfer behavior, promote surface reaction, and improve long-term stability, and has been deemed as one of the most prevalent strategies to optimize photoanodes for achieving enhanced PEC conversion efficiency.13,15–17
This feature article majorly focuses on the photoanodic water oxidation reaction and describes the PEC water splitting mechanism, performance parameters, band bending and charge transfer behaviour within SC photoanodes in detail. Recent progress and our contributions in designing and fabricating TMD-based heterostructure photoanodes for efficient PEC water splitting are systematically reviewed and summarized. A particular emphasis on relevant synthetic methods and crucial roles of TMDs as photosensitizers, electron transporting mediators, passivation layers, cocatalysts, and protective layers in various TMDs/SC heterostructure PEC systems are also presented. Finally, a critical outlook on the future development of highly-efficient and stable TMD-based photoanodes, and the construction of tandem devices for unassisted solar water splitting are discussed.
2. Principles of PEC water splitting
PEC water splitting for hydrogen production is driven by the photoexcited charge carriers-induced redox reactions of water.18 Thermodynamically, the overall process of converting a water molecule into hydrogen and oxygen is energetically uphill, requiring a Gibbs free energy change of approximately 237 kJ mol−1 under standard conditions, which corresponds to a Nernstian potential of 1.23 V.19–21 The complete photo-assisted water splitting involves two fundamental half-reactions: a two-electron proton reduction and a four-electron water oxidation, which are represented by the following equations:1| Photoexcitation: SCs + hν (>Eg) → e− + h+ | (1) |
| HER (proton reduction): 2H+ + 2e− → H2 | (2) |
| OER (water oxidation): 2H2O + 4h+ → O2 + 4H+ | (3) |
| Overall water splitting: 2H2O → O2 + 2H2, ΔE° = 1.23 eV | (4) |
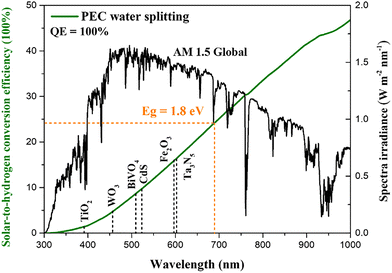 | ||
| Fig. 1 Air Mass 1.5 global solar spectrum (black) with the theoretical maximum STH efficiency at 1.8 eV (green) and bandgaps of some representative photoanode materials. | ||
2.1. Configuration of photoanodic water oxidation reaction and interfacial band bending
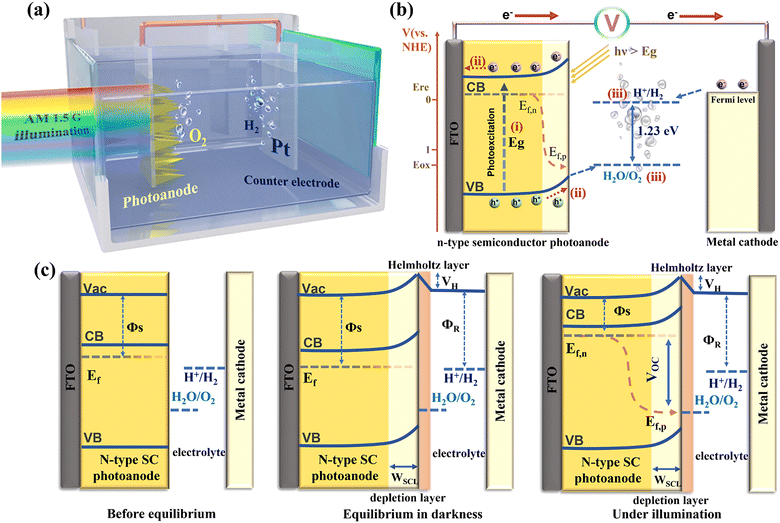
PEC hydrogen evolution is recognized as a promising strategy for converting abundant solar energy into storable chemical fuels, offering a pathway to alleviate the growing global energy and environmental challenges. SC photoabsorbers, serving as the core components, have long been at the centre of research interest. Actually, the OER phenomenon on illuminated rutile TiO2 electrodes was first observed in 1968,26 and shortly thereafter, Fujishima and Honda reported the pioneering demonstration of solar-driven electrochemical water splitting in 1972 using n-type TiO2 as a photoanode, which is widely regarded as the inception of PEC water splitting.27 Despite the excellent chemical stability of TiO2, its wide bandgap (∼3.2 eV) confines absorption to the ultraviolet (UV) region, thereby restricting its solar energy utilization and conversion efficiency. Therefore, extensive efforts have been dedicated to designing narrow-bandgap photoactive semiconductors to enhance PEC performance. Building on these foundational studies, PEC water-splitting systems have advanced considerably, with recent reports achieving solar-to-hydrogen (STH) conversion efficiencies approaching 19%.28Fig. 2a presents a schematic configuration of the representative PEC water-splitting system dominated by the photoanode. The setup typically includes an n-type semiconductor photoanode serving as the working electrode, a Pt counter electrode, and a simulated solar light source (AM 1.5G). As illustrated in Fig. 2b, the photoanodic OER proceeds through three main stages. (i) Photoexcitation: Upon illumination, the semiconductor absorbs photons with energies equal to or greater than its bandgap (hν ≥ Eg), leading to the generation of electron–hole pairs. The photoexcited electrons are promoted to the conduction band (CB), while the holes remain in the valence band (VB). (ii) Charge separation and transfer: driven by band bending and aided by an applied bias, the photogenerated carriers are spatially separated. Electrons (majority carriers) migrate through the external circuit to the counter electrode, while holes (minority carriers) move toward the surface of the photoanode. However, charge recombination may occur within the bulk semiconductor, at the surface, or across semiconductor/substrate and semiconductor/electrolyte interfaces, thereby diminishing the quantum efficiency.29 Hence, strategies that enhance charge separation and injection efficiency are crucial for improving PEC performance. (iii) Surface redox reactions: at the counter electrode, the transferred electrons reduce protons (H+) to hydrogen (H2), while holes accumulated at the photoanode surface participate in the oxygen evolution reaction. Meanwhile, proton transport through the electrolyte completes the circuit, enabling the overall water-splitting reaction.
In a conventional PEC cell employing a single photoabsorber as the photoanode, an external bias is typically required to compensate for the overpotential and other energy losses. Unlike purely electrocatalytic water splitting, the photoanode replaces the catalytic anode for the OER, thereby driving the corresponding half-reaction under illumination. The incorporation of a photoanode allows partial or complete reduction of the external bias, with the compensated portion corresponding to the photovoltage (VPh) generated by the semiconductor absorber.30 When VPh exceeds the combined potential of water oxidation (E°(O2/H2O)) and the associated overpotential requirements (∼1.8 V), this PEC device can then achieve an unassisted water-splitting process. To obtain sufficient photovoltage for spontaneous water splitting operation, advanced configurations such as photoanode–photocathode tandem cells or photoanode–photovoltaic (PV) tandem cells are often adopted. What is more, in some cases, anion or cation exchange membranes are introduced to separate the photoanode from the counter electrode, ensuring ionic conductivity while preventing product crossover. Additionally, conductive substrates such as silicon,31 fluorine-doped tin oxide (FTO), or indium tin oxide (ITO)-coated glass are commonly utilized to support the deposition or growth of photoactive layers during photoanode fabrication.
Except for the SC photoanode module, the electrolyte also plays a crucial role in facilitating charge transport within the PEC system. In reality, electrolyte selection is not strictly defined, as the stability of different materials varies significantly across specific pH ranges. Noteworthily, in specific cases such as metal sulfides as photoanodes (e.g., CdS and TMDs), redox couples like H2O2, Na2S, and Na2SO3 are generally introduced into the electrolyte as hole sacrificial agents, effectively replacing the photoanodic OER due to the lower oxidation potential of these species compared to water.32,33 The rapid harvesting of photogenerated holes by sacrificial agents helps to inhibit surface charge recombination, avoid back reactions, and protect the photoanode. Nevertheless, the use of sacrificial agents introduces additional costs. To enhance the economic feasibility, sacrificial organic compounds, including short-chain alcohols, carboxylic acids, and biomass-derived molecules, have been explored, offering the dual advantages of generating high-value-added chemicals alongside hydrogen production.20,34 Furthermore, this PEC reforming strategy can be applied for the treatment of organic-laden industrial wastewater, contributing to aquatic protection. Considering the mechanistic insights and operational principles of photoanode-based PEC systems discussed above, it is evident that the primary factors and challenges for a practical PEC hydrogen production device include efficiency optimization and long-term photostability, which remain the focal points of current research.
In photoanode-dominated OER processes, band bending at the semiconductor/electrolyte interface is critical for facilitating the charge carrier separation and transport, making a thorough understanding of this phenomenon essential. When a SC photoanode comes into contact with an electrolyte, the difference between the SC's Fermi level (Ef) and the electrolyte redox potential drives charge transfer until equilibrium is established, resulting in band bending within the semiconductor. Fig. 2c illustrates the band energetics at the photoanode/electrolyte interface under three conditions: before equilibration, after equilibration in the dark, and during quasi-static equilibration under steady-state illumination. Apparently, upon immersion of a typical n-type SC into the electrolyte, electrons flow across the interface until the Ef aligns with the redox potential of the electrolyte. This equilibration forms a depletion region, or space charge layer (WSCL), in the semiconductor containing an excess of positive charges, which leads to upward band bending. Simultaneously, a narrow, anion-rich Helmholtz layer develops at the interface.35 Under illumination, the generation of non-equilibrium electron and hole populations can be described using quasi-Fermi levels, which represent the electrochemical potentials of electrons and holes under steady-state light conditions. The resulting gradient of the quasi-Fermi levels induces a built-in electric field at the semiconductor surface,36 producing a photovoltage (or open-circuit voltage, VOC) that provides the powerful driving force for water oxidation. Experimentally, VOC can be determined from the potential difference between the quasi-Fermi levels of electrons and holes under open-circuit conditions, while the maximum power output (PMAX) can be estimated as the product of the short-circuit current (JSC) and VOC.
2.2. Charge separation and transfer behaviour within SC photoanodes
Photoanodic oxygen evolution is governed by several key processes, including photoexcitation, charge separation, and carrier transport. The efficiency of photoexcitation, which generates electron–hole pairs, is primarily determined by the intrinsic photoelectronic properties of the semiconductor, such as its bandgap and band-edge positions, as well as the spectral alignment of absorbed photons with the solar spectrum. Subsequent charge separation and transport critically influence the overall quantum efficiency. Ideally, radiative recombination within the bulk semiconductor is the only competing process, whereby emitted photons may be reabsorbed.37 However, in practice, nonradiative pathways, including Shockley–Read–Hall (trap-assisted) recombination, Auger recombination, and other mechanisms, account for the majority of energy losses during the photo-to-current conversion.38,39 The schematic illustration of various charge carrier recombination pathways and relevant description are shown in Fig. S1. Furthermore, a mismatch between the diffusion length of photoexcited carriers, particularly minority carriers, and the light penetration depth also exacerbates recombination, reducing PEC performance. For instance, in hematite, the hole diffusion length is reported to be only 2–4 nm, considerably shorter than the characteristic light penetration depth of ∼46 nm at 450 nm illumination.40,41 As a result, most of photogenerated holes recombine before reaching the semiconductor surface to participate in the redox reaction. Consequently, only minority carriers generated within approximately one diffusion length of the depletion layer can be efficiently separated and drift toward the semiconductor/electrolyte interface. Under the assumption of negligible recombination in the depletion region, the instantaneous current of photogenerated minority carriers (J) can be estimated using the Gartner equation:42,43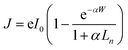 | (5) |
Charge carrier recombination occurs not only within the bulk semiconductor but also at the semiconductor surface or interfaces, where it manifests as a solid-state process.44,45 Even pristine semiconductors exhibit abundant surface electronic states, arising from defects such as structural imperfections and dangling bonds, regardless of whether the surface is in contact with a metal electrode or exposed to vacuum. These surface states are a primary source of charge recombination. Additional competing processes, including self-corrosion of the photoanode and side reactions, further impede surface charge transport, thereby reducing charge injection efficiency. Moreover, chemisorbed species, such as reactants, intermediates, or products formed during surface oxidation, can also serve as recombination centres, limiting the effective separation of photogenerated carriers.
The rational design of SC photoanodes with enhanced performance remains a central focus in advancing PEC technology for efficient solar hydrogen production. As outlined in the PEC water-splitting process, the primary factors limiting the performance of SC photoanodes are solar light absorption, charge separation, and charge injection efficiency. Over the past decades, numerous theoretical and experimental studies have explored a variety of modification strategies to address these limitations, including the development of narrow-bandgap photocatalysts, chemical doping or vacancy engineering,46,47 morphology design,48 surface reconstruction,49 noble metal coating,50 heterojunction construction,51–53 dye sensitization,54 and co-catalyst deposition.47 These approaches aim to broaden the light absorption range while minimizing both bulk and surface charge recombination, thereby improving quantum efficiency and overall PEC performance.55 However, individual strategies or materials rarely optimize all three aspects simultaneously; in practice, combining multiple modification techniques is often necessary to achieve superior photoanode performance.
2.3. Performance parameters in photoanodic water oxidation reaction
Standardized parameters and efficiency measurements for the photoanodic OER process provide quantitative information for comparison. The primary goal in designing photoanodes is to maximize the photocurrent density (Jph) while minimizing the onset potential (Von). Typically, the Jph measured at 1.23 VRHE is used as a benchmark to evaluate the PEC performance of a photoanode, with higher Jph values indicating a greater number of photogenerated carriers participating in the water-splitting reaction. To provide a clearer understanding, the measured photocurrent density can be expressed through a simplified representation of the charge separation and transfer processes:| Jph = Jabs × ηsep × ηinj | (6) |
In addition to bulk and surface charge recombination efficiencies, several other metrics are commonly used to evaluate the solar conversion efficiency, including the solar-to-hydrogen conversion efficiency (ηSTH), incident photon-to-current efficiency (IPCE), absorbed photon-to-current efficiency (APCE), applied bias photon-to-current efficiency (ABPE), and faradaic efficiency (FE). The corresponding calculation formulas for these parameters are provided below:
As a standard metric for evaluating and comparing PEC system performance, the solar-to-hydrogen conversion efficiency (ηSTH) is calculated as the ratio of the chemical energy output to the total incident solar energy (AM 1.5G), as expressed below:
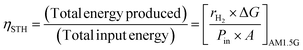 | (7) |
The incident photon-to-current efficiency (IPCE) is a widely employed metric for assessing the photoelectric conversion performance of photoanodes at a specific illumination wavelength. It is defined as the ratio of the number of photoexcited electrons contributing to the photocurrent to the number of incident photons, as expressed by the following equation:
 | (8) |
In this expression, J (λ), e, h, c, λ, and P (λ) denote the photocurrent density at the specified incident wavelength (mA cm−2), the elementary charge (1.602 × 10−19 C), Planck's constant (6.626 × 10−34 J s), the speed of light (3 × 108 m s−1), the wavelength of the incident light (nm), and the corresponding light intensity (mW cm−2), respectively.
The IPCE calculation described above is based on the total number of incident photons and does not account for optical losses due to reflection or transmission, which may introduce deviations in the measured efficiency. To address this limitation, the absorbed photon-to-current efficiency (APCE) is often used to more accurately evaluate the intrinsic quantum efficiency. APCE is defined as the ratio of photoexcited electrons contributing to the photocurrent to the number of photons actually absorbed by the photoanode, as expressed by the following equation:
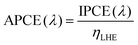 | (9) |
| ηLHE = 1 − 10−α | (10) |
In practical PEC measurements, a conventional three-electrode setup is commonly employed to evaluate photoanode performance. By excluding the contribution from externally applied bias, the applied bias photon-to-current efficiency (ABPE) can be determined using the following expression:
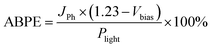 | (11) |
Faradaic efficiency (FE) is widely employed to verify whether the measured photocurrent density arises from water-splitting reactions rather than photoelectrode corrosion or other parasitic processes. It is defined as the ratio of the experimentally evolved gas quantity to the theoretical amount of gas calculated from the detected photocurrent density, as expressed by the following equation:
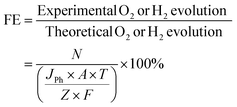 | (12) |
In this expression, N denotes the experimentally measured gas amount determined by gas chromatography (GC, mmol), JPh is the photocurrent density (mA cm−2), A represents the illuminated area of photoelectrode (cm2), T is the duration of the photoreactions (s), Z corresponds to the number of electrons required to generate one molecule of gas (2 for H2, 4 for O2), and F is the Faraday constant (96![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 485.33 C mol−1).
485.33 C mol−1).
Among the various efficiency metrics, ηSTH is regarded as the most reliable parameter for rigorously benchmarking PEC water-splitting devices. Nonetheless, auxiliary metrics, such as ABPE and IPCE, serve as valuable diagnostic tools, providing additional insight into the development and characterization of PEC materials.
3. Structure and advantages of TMD-based heterostructure photoanodes
SC-based photoabsorbers, as the key component of PEC devices, have consistently been the focus of photoanode design. Upon the pioneering development of TiO2 photoanodes in 1979, a variety of photoactive SC materials, e.g., CdS,50 α-Fe2O3,56 BiVO4,57 Ag3PO4,58 Si,59etc., and relevant heterojunctions, have been explored and implemented for PEC water splitting, leading to significant advancements in device performance. However, their practical photoconversion efficiencies are still far from satisfactory. Fortunately, the emergence and investigation of low-dimensional TMDs injects renewed impetus into the construction of highly-performing photoanodes for PEC water splitting, due to their extraordinary physicochemical, photoelectronic, and catalytic properties.In general, TMDs are composed of hexagonal layers in which a transition metal plane is sandwiched between two chalcogen planes, with an individual layer thickness of approximately 6–7 Å, as shown in Fig. 3a. The stacking arrangement of these layers dictates both the crystal structure and the resulting physicochemical properties of the materials.11 The most prevalent polymorphs include the 1T, 1T′, 2H, and 3R phases, corresponding to trigonal, distorted trigonal (octahedral), hexagonal, and rhombohedral structures, respectively (Fig. 3b).60 These structural configurations arise from the electronic interactions between the valence d-orbitals of the transition metal atoms and the p-orbitals of the chalcogen atoms.61 Specifically, the 1T and 1T′ polymorphs adopt octahedral and distorted octahedral coordination, respectively, and generally exhibit metallic behavior characterized by high electrical conductivity. In contrast, the 2H phase, which contains two hexagonal layers per unit cell, and the 3R phase, comprising three rhombohedral layers, typically display semiconducting properties with bulk bandgaps ranging from 0.3 to 2.0 eV. The band alignment of the most representative monolayer TMDs used for photoanode fabrications, and the origins of the conduction band minimum and valence band maximum for these phases are illustrated in Fig. 3c and d.62 Overall, the electronic and optical characteristics of TMDs are strongly governed by the coordination geometry of the transition metal centers and their d-electron occupancy.63
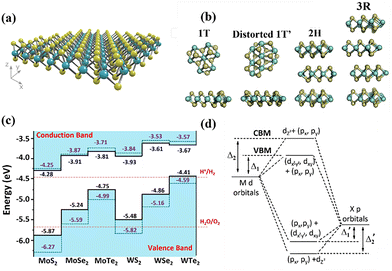 | ||
| Fig. 3 (a) The typical crystal structure of monolayer TMDs, showing a transition metal plane (blue) is sandwiched between two chalcogen planes (yellow); (b) different polymorphs or phases of single-layer TMDs: 1T, 1T′, 2H, and 3R-phases. Reproduced from ref. 60 with permission. Copyright 2015 Royal Society of Chemistry. (c) Bandgap alignment of most representative monolayer TMDs used for photoanode fabrications; (d) schematic illustration for the origin of conductance band maximum (CBM) and valence band maximum (VBM). Adapted from ref. 62 with permission. Copyright 2013 AIP Publishing. | ||
As typical layered materials, a defining characteristic of TMDs is that their bulk crystals are composed of multiple stacked layers held together by weak van der Waals (vdW) interactions. These layers can be exfoliated into mono- or few-layer structures through approaches such as mechanical exfoliation, liquid-phase exfoliation, and a ball milling method, thereby increasing the density of catalytically accessible sites. The intrinsically large surface-to-volume ratio of 2D TMDs provides abundant photoreactive centers for light harvesting and interfacial reactions. Among them, molybdenum (Mo)-based TMDs, particularly MoS2, have attracted considerable interest as promising alternatives to platinum (Pt) for HER catalysis, due to their comparable catalytic activity and superior stability. While numerous studies indicate that the edge sites are generally more catalytically active than the basal planes, plenty of efforts are thus employed to expose more active edge sites and activate basal planes through surface modification, phase or defecting engineering.64–67 Next, TMDs exhibiting superior solar absorption capability and their bandgaps can be easily modulated via thickness variation, chemical doping, and strain engineering.68,69 Due to their narrow bandgaps and having band positions well matched with the redox potentials of water, TMDs are ideal photosensitizers to optimize the overall light harvesting efficiency. In addition, 2D TMD materials also exhibit several other distinct advantages, including high light transmission with enhanced photon–matter interactions, enormous charge mobility (∼200 cm2 V−1 s−1 for MoS2), and short carrier diffusion pathway, which is beneficial for yielding electron–hole pairs and fast charge transport across materials.70 More importantly, van der Waals interactions allow TMD materials to be assembled onto diverse substrates or stacked into multilayers without inducing lattice mismatch or atomic interdiffusion, unlike covalently bonded materials that often cause mechanical strain and structural distortion.17 All the above structural features and property superiorities provide TMD materials with potential and flexibility to incorporate with other SCs for van der Waals heterojunction fabrication.
Among the family of TMDs, molybdenum/tungsten disulfides, like MoS2 and WS2, have obtained much more attention and serve as the most common photoanode materials. However, since TMD nanomaterials are susceptible to desulfurization or corrosion under alkaline conditions and high overpotentials, their long-term stability and durability remain a critical limitation for OER applications in PEC water splitting. To address this issue, recent research has shifted toward employing TMDs as cocatalysts or photosensitizers in PEC-OER systems, rather than as the primary OER photocatalysts. Ingeniously assembling low-dimensional TMDs onto other photoactive semiconductor scaffolds to construct heterojunctions is the most prevalent and useful method to optimize PEC performance. Their synergistic effects and formed built-in electrical field within interfacial junctions can effectively improve the overall light absorption, promote charge separation and transfer, increase surface reaction sites, and enhance photocorrosion resistance. The positive roles of TMDs in various heterojunction architectures can be summarized as photosensitizers, electron transporting mediators, passivation layers, cocatalysts, and protective layers. The relevant synthetic routes, recent advancements, and our contributions in designing and manufacturing TMD-based heterostructure photoanodes are reviewed below.
4. Design and fabrication of TMD-based heterostructure photoanodes
A heterojunction is established when two or more semiconductors are brought into direct contact, creating an interface that facilitates efficient charge transfer between the constituent materials. Elaborately integrating low-dimensional TMDs with other primary semiconductor photoabsorbers to form heterojunctions can tackle the weakness of each component well, synergistically contributing to the overall PEC performance. Generally speaking, the fabrication of heterojunctions typically requires semiconductors with compatible crystal structures, lattice parameters, and thermal expansion coefficients. Lattice mismatch at the interface can lead to weak interfacial bonding and the formation of defects, which hinder efficient charge transfer.71 Therefore, diverse synthetic methods and preparation routes are employed to improve the quality of heterojunctions with intimate interfacial contact and high-speed charge transport channels between semiconductors.4.1. General synthetic strategies
Unlike other 2D nanomaterials, low-dimensional TMDs with different microstructures and morphologies, such as 2D few/mono layered thin films, triangular nanoflakes, nanosheet clusters, or quantum dots, can be synthesized by a wide variety of approaches, including, mechanical/liquid-phase exfoliation,72,73 chemical vapor deposition (CVD),74 solvo/hydrothermal synthesis,75,76etc. The versatility of TMD synthesis gives rise to a wide range of possible architectures for TMD-based heterostructures, whether via in situ assembly or ex situ incorporation. In summary, fabrication strategies for TMD-based heterostructure photoanodes can be broadly categorized into two approaches: top-down and bottom-up methods, shown in Fig. 4a.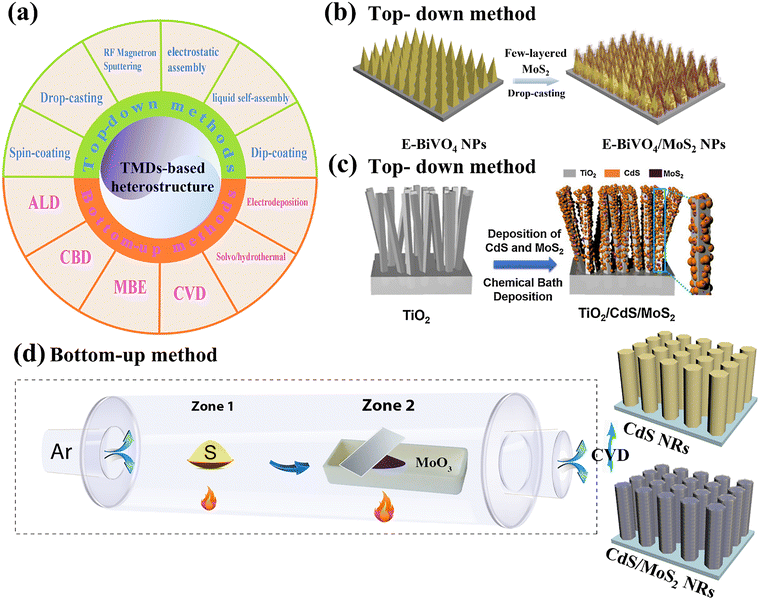 | ||
| Fig. 4 (a) The general synthetic strategies for TMDs-based heterostructure photoanodes categorized by: top-down and bottom-up methods; (b and c) representative top-down methods of drop-casting and CBD; reprinted from ref. 77 and 78 with permission. Copyright 2023 Elsevier and Copyright 2019 Elsevier. (d) Representative bottom-up route of CVD. Reprinted from ref. 32 with permission. Copyright 2023 Elsevier. | ||
For example, Siaj's group successfully fabricated a highly-oriented E-BiVO4/MoS2 heterojunction photoanode via directly drop-casting exfoliated few-layered MoS2 nanosheets on electrochemically-treated BiVO4 nanopyramids, followed by further heat treatment at 400 °C for 1 h under an Ar atmosphere, as shown in Fig. 4b.77 In addition, Jiang et al. demonstrated the fabrication of a heterojunction photoanode comprising 2D MoS2 nanosheet-modified 1D TiO2 nanorods (NRs) integrated with 0D CdS nanocrystals.78 This architecture was realized via in situ chemical bath deposition, achieved by immersing the TiO2 NR film into a CdS precursor solution containing exfoliated MoS2 nanosheets, as illustrated in Fig. 4c. As mentioned above, TMDs are generally employed as cocatalysts rather than primary photoactive materials because of their limited intrinsic photoactivity. Consequently, top-down methods typically start with high-purity exfoliated 2D TMDs, which can be readily incorporated onto semiconductor scaffolds without requiring in situ growth. This approach offers clear advantages in terms of cost-effectiveness and scalability, making it particularly suitable for large-area deposition and roll-to-roll manufacturing. However, the thickness, lateral size, and uniformity of exfoliated TMD flakes are inherently difficult to regulate, often resulting in limited control over flake distribution, orientation, and interfacial junction quality within the assembled heterostructures. Such limitations may lead to flake agglomeration, structural heterogeneity, and increased interfacial defect density. Moreover, many top-down processed films require subsequent high-temperature annealing to improve interfacial adhesion or crystallinity, which may unintentionally introduce impurity states, alter energy-level alignment, or even induce undesirable phase transitions.
4.2. Band engineering for optimized photoexcition separation and injection
Except for the selection of materials and synthetic methods, another consideration is involved regarding the heterostructure types. In semiconductor-based photoanodes, photoexcited charge carriers often undergo recombination before reaching the photoelectrode surface and participating in photoreaction. To promote the efficient charge separation and injection, heterojunction fabrication by the incorporation of TMDs as the hole transporting layers (HTLs) is essential. To direct charge migration effectively, it is essential to establish well-matched band alignment with favourable electron transfer energetics. Generally speaking, based on the distinct charge transfer mechanism, the TMD-based heterojunctions can be categorized into the following configurations: conventional heterostructures (types-I and II), Z-scheme architectures, p–n junctions, and Schottky junctions. A detailed discussion of these classifications is provided in the following section. Meanwhile, a comprehensive summary and comparison of their PEC performances and photoconversion efficiencies are presented in Table 1.| Charge transfer model | Photocatalysts | Light source and intensity (mV cm−2) | Electrolyte | Photocurrent density at 1.23 VRHE (mA cm−2) | Onset potential (VRHE) | IPCE | ABPE | Ref. |
|---|---|---|---|---|---|---|---|---|
| Type-I | CdS/MoS2 | AM 1.5G 100 | 10% lactic acid | 1.9 (at 0.8 VRHE) | −0.1 | 45% at 450 nm | N.A. | 33 |
| Type-I | MoS2/CdS | AM 1.5G 100 | 0.25 M NaS2 + 0.35 M Na2SO3 | 2.14 (at 1 VRHE) | −0.14 | 28.1% at 340 nm | N.A. | 32 |
| Type-I | MoS2/TTZ-COF | AM 1.5G 100 | 0.1 M Na2SO4 | 2.5 µA cm−2 | ∼0.6 | 3.1% at 350 nm | N.A. | 85 |
| Type-II/Type-I | TiO2/CdS/MoS2 | AM 1.5G 100 | 0.25 M NaS2 + 0.35 M Na2SO3 | ∼3.45 (at 0.9 VRHE) | −0.09 | 45% at 475 nm | N.A. | 78 |
| Type-II | MoS2/BL-BiVO4 | λ ≥ 420 nm 300 W Xe lamp | PPCPs sewage | 2.21 (at 1 VAg/AgCl) | 0 VAg/AgCl | N.A. | N.A. | 81 |
| Type-II | E-BiVO4/MoS2 | AM 1.5G 100 | 0.5 M Na2SO4 + 0.1 M PBS | 2.11 | 0.36 | 40.9% at 340 nm | 0.53% at 0.77 VRHE | 77 |
| Type-II | W:α-Fe2O3/MoS2 | 300 W Xe lamp 100 | 1 M NaOH | 1.83 | ∼0.7 | 38.7% at 325 nm | 0.26% at 0.96 VRHE | 86 |
| Type-II | MoS2/WS2 | AM 1.5G 100 | 0.5 M NaClO4 | 0.45 | ∼0.65 | ∼0.1% at 600 nm | N.A. | 87 |
| Type-II | MoS2/rGO/Zn-CuInS2 | AM 1.5G 100 | 0.25 M NaS2 + 0.35 M Na2SO3 | 0.44 (at 0.93 VRHE) | ∼0.15 | ∼6.5% at 375 nm | N.A. | 88 |
| Type-II | MoS2/WO3 | AM 1.5G 100 | 0.50 M H2SO4 | 0.96 | ∼0.3 | ∼45% at 320 nm | N.A. | 89 |
| Type-II | ZnO/MoS2 | AM 1.5G 100 | 0.5 M Na2SO4 | 2.04 | 0.37 | N.A. | 0.67% at ∼0.76 VRHE | 93 |
| Type-II | MoS2/WO3 | AM 1.5G 100 | 0.3 M KH2PO4 | 1.12 (at 0.8 VSCE) | ∼0 VSCE | N.A. | 0.52% at 0.8 VRHE | 90 |
| Type-II | ZnO/MoS2 | AM 1.5G 100 | 0.5 M Na2SO4 | 0.61 | ∼0.5 | N.A. | 0.21% at 0.72 VRHE | 94 |
| Type-II | ZnO/MoS2 | 150 W Xe lamp 100 | 0.1 M Na2SO4 | 1.28 (at 1.65 VRHE) | N.A. | N.A. | 0.39% at 1.36 VRHE | 95 |
| Type-II | MoS2/a-ZrO2/(010)-BiVO4 | AM 1.5G 100 | 0.5 M Na2SO4 | 0.6 (at 1.2 VRHE) | ∼0.3 | N.A. | N.A. | 82 |
| Type-II | F−Fe2O3−x/MoS2 | AM 1.5G 100 | 1 M NaOH | 2.2 | ∼0.8 | 30.3% at 360 nm | 0.21% at 1.04 VRHE | 98 |
| Type-II | MoS2/ZnO/graphene/NF | AM 1.5G 100 | 0.5 M Na2SO3 | 0.25 A cm−2 (at 1 VAg/AgCl) | 0.3 | N.A. | 5.5% at 0.09 VAg/AgCl | 99 |
| Type-II | TiO2−x-MoS2 | AM 1.5G 100 | 0.5 M Na2SO4 + 1 M KOH | 2.6 | ∼0.55 | N.A. | N.A. | 100 |
| Type-II | Ti-doped WO3–MoS2 | 150 W Xe lamp | 0.5 M Na2SO4 | 1.15 mA cm−2 (at 1.6 VAg/AgCl) | ∼0.8 | 0.62% at 400 nm | N.A. | 91 |
| Type-II | MoS2/GQD/ZnO | AM 1.5G 100 | 0.5 M Na2SO4 | 4.36 (at 1.2 VRHE) | 0.19 | N.A. | 0.5% at 0.97 VRHE | 102 |
| Type-II | Ag/MoS2/ZnO | AM 1.5G 100 | 0.5 M Na2SO4 | 0.7 (at 0 VAg/AgCl) | 0.3 | N.A. | 1.45% at 0.79 VRHE | 96 |
| Type-II | In2S3/MoS2 | AM 1.5G 100 | 0.3 M KH2PO4 | 1.28 | ∼0.1 | ∼18% at 440 nm | 0.75% at 1.23 VRHE | 97 |
| Type-II | MoS2/TiO2/CSF | AM 1.5G 100 | 0.5 M Na2SO4 | 33.81 | N.A. | N.A. | 6.97% at 0.87 VRHE | 121 |
| Type-II | MoS2/TiO2/Au | AM 1.5G 100 | 0.5 M NaOH | 16.3 µA cm−2 | N.A. | N.A. | 0.34% | 101 |
| Type-II | TiO2 NFs/MoSe2 | AM 1.5G | 1 M NaOH | 1.4 (at 1.2 VSCE) | N.A. | N.A. | N.A. | 118 |
| Type-II | MoSe2/p+-n Si | AM 1.5G 100 | 1 M HBr | 30 (at 0.8 VRHE) | 0.3 | ∼75% at 620 nm | 13.8% at ∼0.5 VRHE | 83 |
| Type-II | WO3/WS2 | AM 1.5G 100 | 0.5 M Na2SO4 | 5.6 (at 1.23 VAg/AgCl) | ∼0 VAg/AgCl | 55% at 350 nm | 2.26% at 0.75 VAg/AgCl | 103 |
| Type-II | Fe2O3/WS2 | AM 1.5G 100 | 0.5 M NaOH | 0.52 | 0.3 | ∼12.5% at ∼420 nm | ∼0.15% at ∼0.8 VRHE | 104 |
| Type-II | CdS/WS2 | AM 1.5G 100 | 0.1 M NaS2 + 0.2 M Na2SO3 | 3.35 (at 0.4 VSCE) | ∼−1.05 | N.A. | ∼2.64% at ∼0 VSCE | 105 |
| Type-II | WS2/TiO2 | AM 1.5G 100 | 0.1 M Na2SO4 | 2.07 (at 0.7 VSCE) | ∼−0.2 VSCE | N.A. | 1.52% at 0.32 VSCE | 106 |
| Type-II | WO3@WS2 | AM 1.5G | 1 M NaOH | 0.91 | ∼0.6 | N.A. | N.A. | 107 |
| Ionic Z-scheme | SnSe2/WSe2 | 150 W Xe lamp | O.5 M NaOH + 0.05 M KI | 0.164 (at 0 VAg/AgCl) | 0 VAg/AgCl | N.A. | N.A. | 108 |
| All solid-state Z-scheme | Ag-MoS2/RGO/NiWO4 | AM 1.5G 100 | 0.5 M Na2SO4 | 3.5 | 0.61 | N.A. | 0.52% at ∼1.02 VRHE | 109 |
| Direct Z-scheme | MoOx/MoS2 | AM 1.5G 150 | 0.5 M H2SO4 | 2.08 (at 0.9 VRHE) | N.A. | N.A. | 0.81% at ∼0.6 VRHE | 80 |
| Direct Z-scheme | MoSe2–CdS–ZnO | AM 1.5G 100 | 0.55 M NaS2 + 0.127 M Na2SO3 | 2.4 (at 0.3 VRHE) | 0 | 52% at 460 nm | N.A. | 111 |
| Type II/Z-scheme | MoS2/Bi2S3/Bi2WO6 | AM 1.5G | 0.5 M PBS + 1 M Na2SO3 | 3.2 | ∼0.34 | 16.1% at 432 nm | 0.93% at 0.81 VRHE | 110 |
| p–n junction | FeOOH@1T-MoS2@BiVO4 | AM 1.5G 100 | 0.1 M KH2PO4 | 4.02 | ∼0.1 | ∼78% at 420 nm | 1.28% at ∼0.62 VRHE | 122 |
| p–n junction | MoSX/BP/BiVO4 | AM 1.5G 100 | 0.2 M Na2SO4 | 1.2 | N.A. | N.A. | N.A. | 112 |
| p–n junction | WO3/MoS2-MoOX | AM 1.5G 100 | 0.5 M Na2SO4 | 2.15 | ∼0.9 | 27.5% at 375 nm | 0.18% at 1 VRHE | 114 |
| p–n junction | MoS2/CdS | AM 1.5G 80 | 0.1 M NaS2 + 0.01 M Na2SO3 | 1.6 (at 0.2 VSCE) | N.A. | 11% at 440 nm | N.A. | 79 |
| p–n junction | α-Fe2O3/MoS2 | AM 1.5G 100 | 1 M NaOH | 1.52 mA cm−2 (at 0.6 VAg/AgCl) | 0.1 VAg/AgCl | 7.6% at 325 nm | 0.132% at 0.97 VRHE | 115 |
| p–n junction | MoS2/NiFe LDH | AM 1.5G 100 | 1 M NaOH | 2.6 | 1.08 | 26.8% at 420 nm | N.A. | 116 |
| p–n junction | a-Fe2O3/WS2/WOx | AM 1.5G 100 | 1 M NaOH | 2.1 | 0.47 | 15.5% at 325 nm | 0.194% at 0.9 VRHE | 117 |
| Type-II/p–n junction | α-Fe2O3/BiVO4/MoS2 | AM 1.5G 100 | 0.5 M Na2SO4 | 1.67 | ∼0.3 | 22% at 325 nm | 0.28% at 0.85 VRHE | 92 |
| Schottky junction | TNTAs/(BPEI–MoS2 QDs)n | AM 1.5G 100 | 0.5 M Na2SO4 | ∼0.3 (at 0.8 VRHE) | ∼0.2 | 20.7% at 300 nm | 0.16% at ∼0.6 VRHE | 120 |
| Schottky junction | N,S-GQDs/MoSe2 | AM 1.5G 100 | 10% lactic acid | 1.8 (at 0.8 VRHE) | ∼0.09 | 7.62% at 420 nm | N.A. | 123 |
| Schottky junction | MoS2/Au@CFC | AM 1.5G 100 | 0.1 M KH2PO4 | 10 | 0.32 | ∼12% at 530 nm | 1.27% at ∼0.9 VRHE | 119 |
| Schottky junction | Au-WS2 | 300 W Xe lamp | 1 M Na2SO4 | 23 (at 1 VSCE) | 0 VSCE | N.A. | N.A. | 124 |
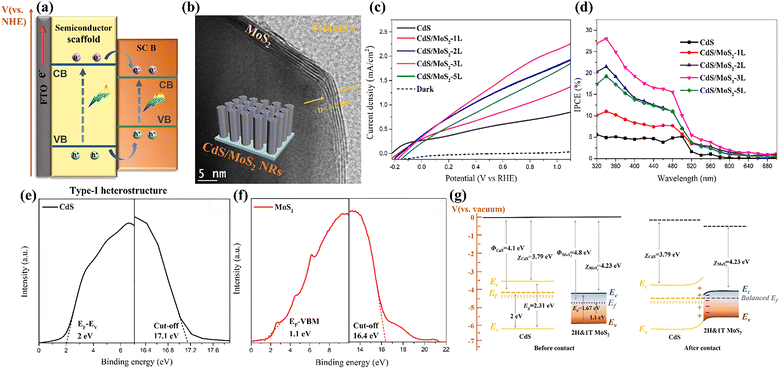 | ||
| Fig. 5 (a) Type-I heterostructure with straddling bandgap alignment. (b) HRTEM image of the CdS/MoS2 nanorod with distinct core-shell structural feature as the insert; (c) LSV curves measured at Na2SO3/Na2S mixture using a three-electrode setup and (d) IPCE plots of the pristine CdS and CdS/MoS2 heterojunction photoanodes; (e and f) UPS spectra of the bare CdS and MoS2 nanoflakes. ECut-off denotes the spectrum edge of secondary electrons. EF − EV means the valence band energies relative to the Fermi levels. (g) The resulting type-I energy band alignment of MoS2 and CdS before and after interface contact at thermal equilibrium in darkness. Φ, Ef, EC, and EV represent work function, Fermi level, conduction band edge, and valence band edge, respectively. Reprinted from ref. 32 with permission. Copyright 2023 Elsevier. | ||
Jang et al. demonstrated the formation of a type I heterojunction photoanode, composed of 2D MoS2 nanosheet modified 1D TiO2 NRs/0D CdS nanocrystals prepared by the all solution process (Fig. S3a), for PEC hydrogen evolution applications.78 Compared to the pristine TiO2/CdS, this heterojunction photoanode exhibits a significantly higher photocurrent density of 3.25 mA cm−2 at 0.9 VRHE and IPCE enhancement. Through a series of electrochemical (EC), PEC, and TRPL analysis, it is concluded that the introduction of MoS2 as a type I heterojunction greatly facilitates the interfacial hole capturing, extends the electron lifetime, and optimizes the charge transfer efficiency, thereby resulting in this PEC enhancement. The straddling/staggered energy band alignment and type II/type I charge transport pathway is displayed in Fig. S3b. Note that photoexcited electrons are injected into TiO2 from CdS under thermodynamic driving forces due to the formation of type II heterojunctions. The higher VB edge of MoS2 facilitates the efficient transfer of photogenerated holes from CdS VB to MoS2, leading to the reduced charge recombination loss. Shen. et al. also synthesized an efficient 2D–2D type I heterojunction photoanode (Fig. S3c and d) through decorating 2D COFs with MoS2 for PEC water oxidation.85 The CB and VB values of triazine-based COF and MoS2 are determined using Mott–Schottky measurements and VB XPS spectra analysis, confirming the formation of straddling energy band alignment. The fabrication of MoS2/TTZ-COF type I heterojunctions can not only accelerate charge separation and transfer, but also enhance water oxidation kinetics, thus leading to the improved photocurrent density (Fig. S3e), negatively-shifted onset potential, and higher IPCE values. Although these previous studies highlight the effectiveness of type-I heterojunctions in enhancing PEC performance, the accumulation of photoexcited electrons and holes within the same semiconductor in the absence of an external bias often leads to severe exciton recombination and reduced quantum efficiency. Moreover, the interfacial transfer of holes from a higher to a lower VBM potential inevitably leads to a pronounced reduction in oxidation capability. These constraints explain the relatively limited number of reports on TMD-based type-I heterojunctions in recent years, which greatly limit its further advancements.
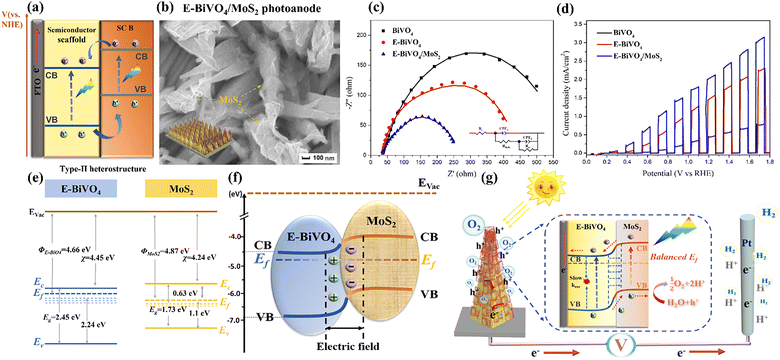 | ||
| Fig. 6 (a) Type II heterostructure with staggered bandgap junctions; (b) top-view SEM image of E-BiVO4/MoS2 nanopyramid arrays with the corresponding structural diagram as the inset; (c) Nyquist plots under sunlight irradiation and (d) chopped LSV curves over the pristine BiVO4, E-BiVO4, and E-BiVO4/MoS2 heterojunction photoanode measured at a mixture of 0.5 M Na2SO4 and 0.1 M PBS solution with a typical three-electrode setup; (e) the detailed energy band information of E-BiVO4 and MoS2 nanosheets; Φ, Ef, EC, and EV represent work function, Fermi level, conduction band edge, and valence band edge, respectively; (f) the energy band alignment at E-BiVO4/MoS2 heterojunction interface and (g) the possible type II charge transfer pathway over this heterojunction photoanode system. Reprinted from ref. 77 with permission. Copyright 2023 Elsevier. | ||
In recent years, a variety of TMD-based type-II heterostructures with diverse nanoarchitectures have been investigated, including stacked planar films, core–shell nanorods, and quantum-dot-decorated nanowires. Compared with single-component photoabsorbers, these TMD-based type-II heterostructure photoanodes, including MoS2@BL-BiVO4,81 E-BiVO4/MoS2,77 W:α-Fe2O3/MoS2,86 MoS2/WS2,87 TiO2/MoS2-rGO/Zn-CuInS2,88 MoS2/WO3,89–91 α-Fe2O3/BiVO4/MoS2,92 ZnO/MoS2,93–96 MoS2/In2S3,97 MoS2@(010)-BiVO4,82 F-Fe2O3−x/MoS2,98 MoS2/ZnO/graphene/NF,99 TiO2−x-MoS2,100 Au/TiO2/MoS2,101 MoS2/GQD/ZnO,102 W/WO3/WS2,103 Fe2O3/WS2,104 CdS/WS2,105 WS2/TiO2,106 WO3@WS2,107etc., all demonstrated higher PEC performance and solar conversion efficiencies. Taking one of our studies as an example, an innovative highly-oriented E-BiVO4/MoS2 type-II heterojunction photoanode was prepared through drop-casting exfoliated few-layered MoS2 nanosheets onto oxygen-vacancy-rich BiVO4 nanopyramid arrays (Fig. 6b).77 As shown in Fig. 6c and Fig. S4a, EIS analysis and efficiency calculation provide the powerful evidence that encapsulating MoS2 as a type-II heterojunction with the built-in electrical field can greatly boost the hole extraction from the E-BiVO4 host and enhance the overall charge injection efficiency. Simultaneously, the electrochemical surface-active areas (ECSA, Fig. S4b) and DRS analysis indicate the improved surface reaction action sites and light harvesting after MoS2 decoration. Accordingly, the engineered E-BiVO4/MoS2 type-II heterojunction photoanode delivers the optimum photocurrent density of 2.11 mA cm−2 at 1.23 VRHE (Fig. 6d) and IPCE value of 40.6%, representing enhancements of approximately 4.6- and 4.9-fold over pristine BiVO4, together with improved OER kinetics and superior photostability. Based on the relevant Tauc plots and UPS analysis, the detailed energy band distribution of E-BiVO4 and MoS2, and the formation of the heterojunction interface with staggered bandgap configuration are confirmed, as shown in Fig. 6e and f. Upon the direct contact of E-BiVO4 and MoS2, a charge rectification process will occur to achieve the thermodynamic equilibrium of Fermi level. The higher work function of E-BiVO4 (4.66 eV) can induce the rapid electron transfer from the E-BiVO4 to MoS2 shell, forming an upward band bending near the E-BiVO4 interface and built-in electric field. Thus, a plausible type II charge transfer route and operation mechanism are proposed. As displayed in Fig. 6g, under solar illumination, both semiconductors can be photoexcited to generate charge carriers due to their suitable bandgaps. Electrochemically induced oxygen vacancies help suppress bulk charge carrier recombination (Krec), facilitating the release and migration of photogenerated holes to the photoanode surface. Further modification with MoS2 nanosheets to construct a type-II heterojunction establishes unidirectional charge-transfer pathways, facilitating the rapid extraction of surface-accumulated holes from E-BiVO4. These holes are driven into the MoS2 valence band by the synergistic effects of the built-in electric field and electrostatic interactions, then participate in the water oxidation reaction. Meanwhile, under an applied bias, photoexcited electrons in the CB of MoS2 migrate toward the CB of E-BiVO4 and flow together to the Pt electrode for hydrogen evolution. The well-separated photogenerated electron–hole pairs finally contribute to the enhanced photoresponse.
Roy's group reported the fabrication of a Fe2O3/WS2 type-II heterojunction photoanode (Fig. S5a) through drop-casting WS2 nanosheets on Fe2O3 nanoflake arrays, followed by annealing in a N2 atmosphere.104 This heterojunction presents significantly improved optical absorption, photoresponse, ABPE and IPCE values, and obtains a synergistic photocurrent density of 0.52 mA cm−2 at 1.3 VRHE, 2.23 times higher than the pristine Fe2O3. The staggered energy band alignment (Fig. S5c) and charge transfer mechanism are confirmed by the DFT method and XPS valence band spectroscopy. The Density of States plots reveal both higher CBM and VBM positions of WS2 than those of Fe2O3, conforming to the type-II energy band distribution. Bader charge analysis with charge density difference plots, presented in Fig. S5b, further verifies the occurrence of charge redistribution with a net charge flow of 4.8 × 10−4 e/interfacial unit from WS2 to the Fe2O3 side. This charge transfer results in the formation of a charge accumulation/depletion layer and a weak built-in electric field at the heterogeneous interface, helping the rapid charge separation and hindering charge carrier recombination. Lee et al. prepared a unique W:α-Fe2O3/MoS2 heterojunction photoanode through directly drop-casting exfoliated MoS2 nanosheets on the W-doped α-Fe2O3 surface (Fig. S5d).86 The optimum 0.5 W: the α-Fe2O3/MoS2 photoanode achieves the highest photocurrent density of 1.83 mA cm−2, IPCE value of 37%, and ABPE value of 26%, which are 26, 5.2, and 13 times higher than those of pure α-Fe2O3, respectively. The UPS spectra are used to analyse the relevant energy band information, wherein the VBM positions of W:α-Fe2O3 and MoS2 are determined to be 6.49 and 5.27 eV through subtracting the UPS spectrum width from the excitation energy. Their work functions are estimated to be 5.05 and 4.34 eV. When W:α-Fe2O3 and MoS2 are in contact, the type-II heterojunction with band bending near the interface is established, as shown in Fig. S5e. The collaborative approach of heterojunction fabrication and atomic doping co-contributes to increasing carrier density, reducing the space charge layer, and decreasing flat band potential, thereby resulting in this remarkably PEC performance enhancement. However, the type-II heterostructure shares a similar drawback with the type I junction, where the charge carrier separation certainly brings a decrease in redox capability. Besides, the presence of electrostatic repulsion will also lead to a charge barrier and loss. Therefore, more types of heterostructures are developed for optimizing charge separation.
(I) Ionic Z-scheme heterostructures. The concept of the ionic-state Z-scheme heterojunction was first proposed and experimentally demonstrated in 1979. In this configuration (Fig. 7a), the two semiconductors are spatially separated without direct physical contact. Charge transfer between them is mediated by redox shuttle ions, which function as electron mediators. Specifically, the conduction band (CB) electrons of one semiconductor reduce the electron acceptor species, while the valence band (VB) holes of the other semiconductor oxidize the electron donor species, thereby facilitating an indirect recombination process. Consequently, the remaining photogenerated electrons and holes with stronger redox potentials in their respective semiconductors are preserved to participate in water photolysis. Representative redox shuttle couples employed in this system include Fe3+/Fe2+, NO3−/NO2−, IO3−/I−, [Co(phen)3]3+/2+, [Co(bpy)3]3+/2+, etc.
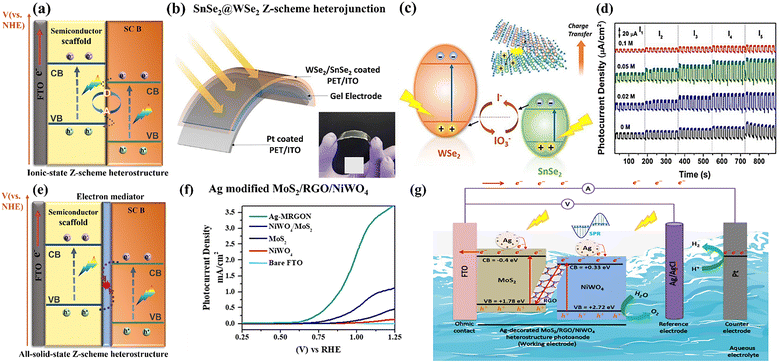 | ||
| Fig. 7 (a) Schematic illustration of ionic state Z-scheme heterostructure; (b) schematic illustration of the prepared flexible SnSe2/WSe2 photodetector with the real digital image as the inset; (c) the possible charge transfer model and (d) on/off photoresponse switching behaviour over SnSe2/WSe2 ionic state Z-scheme heterostructure photoanode system in 0.5 M KOH with different molar concentrations of KI; herein, the IO3−/I− redox couple accepts the photoexcited electrons from the CB of SnSe2 and holes from VB of WSe2, thus promoting the efficient spatial charge separation across the heterojunction. Reprinted from ref. 108 with permission. Copyright 2021 American Chemical Society. (e) All-solid-state Z-scheme heterostructure model. (f) LSV curves measured in 0.5 M Na2SO4 with a standard three-electrode configuration and (g) the proposed charge transfer pathway over the Ag-MRGON all-solid-state Z-scheme photoanode system. The rGO conductive matrix facilitates the recombination between NiWO4 CB electrons and MoS2 VB holes, thereby optimizing the Z-scheme charge-transfer pathway. Reprinted from ref. 109 with permission. Copyright 2020 American Chemical Society. | ||
As a typical TMD material, WSe2 is recognized as a p-type semiconductor with high hole mobility and a relatively narrow bandgap of 1.27 eV. In contrast, SnSe2 displays intrinsic n-type conductivity, attributed to its strong electron affinity and the positioning of its Fermi level close to the conduction band minimum. When combined, the SnSe2/WSe2 heterojunction exhibits an almost broken-gap band alignment, which is particularly advantageous for constructing Z-scheme heterostructures. For example, Chauhan et al. reported a flexible liquid-exfoliated nanocrystal-based SnSe2@WSe2 Z-scheme heterojunction photoanode for self-powered PEC conversion, displayed in Fig. 7b and c.108 This prepared heterojunction photoanode exhibits a considerably improved self-powered photoresponse, 3 times higher than that of individual materials, which predominantly results from their superior visible-light absorption ability and the rapid Z-scheme charge transfer mechanism. According to the provided energy band alignment, the IO3−/I− redox couple exhibits a suitable redox potential of −4.7 eV vs. vacuum level, more negative than the CB of SnSe2 and positive than the VB of WSe2, enabling it to be feasible as an electron mediator and effectively enhancing the Z-scheme charge transfer between SnSe2 and WSe2 nanocrystals. Under light irradiation, I− ions readily scavenge photogenerated holes from the VB of WSe2, being oxidized to IO−3. Simultaneously, IO3− species are reduced back to I− by accepting photoexcited electrons from the CB of SnSe2, thereby completing the redox cycle. This reversible IO3−/I− interconversion promotes efficient charge carrier separation across the heterojunction, accounting for the markedly improved photoelectrochemical performance observed in Fig. 7d.
However, the incorporation of an acceptor–donor pair generally introduces several inherent drawbacks: (i) carrier migration is hindered due to the involvement of redox mediator diffusion ion pairs; (ii) the mediator itself can interfere with light absorption; (iii) commonly used redox mediators, such as IO3−/I−, exhibit strong pH sensitivity, thereby narrowing the operational pH window; (iv) numerous irreversible side reactions occur during electron transfer through the mediator; and (v) the limited stability of the mediator further constrains the overall reaction rate. To address these limitations, an improved Z-scheme heterostructure of all-solid-state is employed and replaces its function.
(II) All-solid-state Z-scheme heterostructures. A representative all-solid-state Z-scheme heterojunction is typically composed of two semiconductor photoabsorbers with appropriately aligned energy bands, bridged by a metallic mediator (Fig. 7e). In this configuration, metallic nanoparticles, such as Au, Ag, Pt, Cu, W, Cd, or conductive materials like reduced graphene oxide (rGO), establish ohmic contacts that are essential for efficient charge transfer. These electron mediators act as recombination centers, enabling the direct annihilation of photogenerated electrons and holes originating from different semiconductors, thereby promoting selective charge separation. Recent studies have highlighted the promising performance of TMD-based all-solid-state Z-scheme heterostructures for PEC water-splitting, demonstrating their potential to significantly enhance energy conversion efficiency. For example, Hendi et al. reported the preparation of Ag-modified MoS2/RGO/NiWO4 (Ag-MRGON) all-solid-state Z-scheme heterojunction photoanodes through all hydrothermal processes.109 Ag nanoparticles are used as the optical property enhancer to improve the overall photo absorption and rGO serves as the electron mediator to optimize the Z-scheme charge transport routes, suppressing the charge carrier recombination (Fig. 7g). The detailed energy band structures are evaluated by VB-XPS analysis. Under simulated sunlight illumination, photoexcited electrons in CB of NiWO4 transfer and recombine with the VB holes of MoS2 through the rGO conductive medium, thus efficiently separating and preserving the photogenerated electrons and holes with strong redox capabilities. As a result, the formed Ag-MRGON heterojunction photoanode demonstrates an improved PEC water splitting performance and ABPE conversion efficiency of 0.52%, ∼17.3 and ∼4.3 times better than those of pure MoS2 and MoS2/NiWO4 photoanodes, together with an enhanced photochemical stability of ∼2 h, as shown in Fig. 7f and Fig. S6a and b. More importantly, the fabrication of the all-solid-state Z-scheme heterojunction via the rGO mediator greatly enhances the interfacial water oxidation kinetics, responsible for the cathodically shifted onset potential from 0.73 VRHE (MoS2/NiWO4 heterojunction) to 0.61 VRHE (Ag-MRGON). The strategy of all-solid-state Z-scheme heterojunction is effective, but also suffers from the light-shielding effect of the electron mediator and poor long-term stability. Thus, further development is necessary.
(III) Direct Z-scheme heterostructures. To advance the practical application of Z-scheme heterojunctions, the concept of the direct Z-scheme system, inspired by natural photosynthesis, has been developed (Fig. 8a). In this configuration, two semiconductors with distinct energy levels are brought into intimate contact, allowing electron redistribution until their Fermi levels are equilibrated. This process establishes a space charge region at the interface, characterized by band bending and an internal electric field. Upon solar irradiation, the built-in electric field facilitates the recombination of conduction band electrons and valence band holes with weaker redox potentials, while the carriers exhibiting stronger redox potentials remain available to drive the respective water oxidation and reduction half-reactions. Unlike ionic- or all-solid-state-mediated systems, the direct Z-scheme heterojunction eliminates the need for an external mediator, enabling interfacial charge transfer with reduced diffusion distance, enhanced recombination kinetics, and an increased built-in photovoltage. Consequently, this architecture effectively addresses the intrinsic limitations associated with conventional Z-scheme configurations.
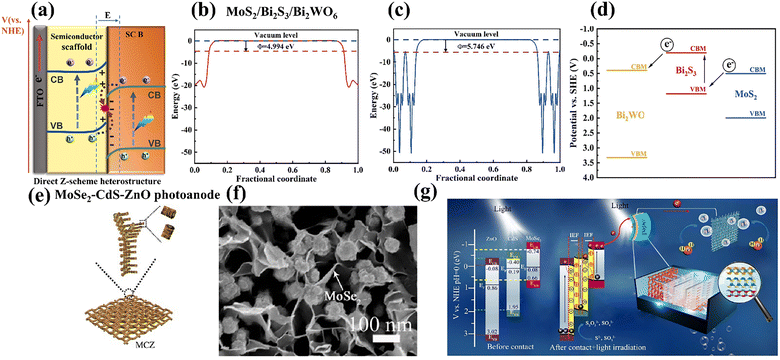 | ||
| Fig. 8 (a) Schematic illustration of direct Z-scheme heterostructure; (b and c) DFT-based work functions of Bi2S3 and MoS2, and (d) photoexcited charge transfer direction over the reported type II/Z-scheme MoS2/Bi2S3/Bi2WO6 heterojunction photoanode; Herein, the photogenerated electrons in CBM of MoS2 trend to recombine with the VBM holes of Bi2S3, forming direct Z-scheme charge transfer pathway. The synergistic effect of type II heterojunction further drives the remaining CBM electrons of Bi2S3 to Bi2WO4 for PEC H2 evolution. Reprinted from ref. 110 with permission. Copyright 2025 Elsevier. (e) Fabrication process and (f) SEM image of MoSe2–CdS–ZnO flexible photoanode paper chip; (g) DFT-calculated band structures and PEC reaction mechanism over the previous reported built-in tandem Z-scheme MoSe2–CdS–ZnO photoanode system. Two parallel internal electric fields (from MoSe2 to CdS and CdS to ZnO) with electron depletion/accumulation layer drive the CB photoexcited electrons of both ZnO and CdS to the VB of CdS and MoSe2, respectively. Reprinted from ref. 111 with permission. Copyright 2021 Elsevier. | ||
The construction of direct Z-scheme heterojunctions requires intimate interfacial contact to ensure efficient charge transfer with minimal resistance, thereby imposing stringent demands on material dimensional control and synthetic methodologies. In recent years, increasing research efforts have been directed toward the development of high-performance TMD-based direct Z-scheme heterojunction photoanodes for PEC hydrogen evolution, surpassing the focus on their ionic or all-solid-state counterparts. For example, Wen et al. created a type II/Z-scheme MoS2/Bi2S3/Bi2WO6 heterojunction photoanode through the hydrothermal process.110 As illustrated in Fig. 8b and c, DFT calculation reveals the higher work function of MoS2 (5.746 eV) than Bi2S3 (4.994 eV), and confirms the formation of direct Z-scheme heterojunctions (Fig. 8d). In the presence of irradiation, the photogenerated electrons in the CBM of MoS2 tend to recombine with the VBM holes of Bi2S3. Meanwhile, the synergistic effect of type II heterojunctions further drives the remaining CBM electrons of Bi2S3 to Bi2WO4, resulting in a significantly extended electron lifetime and the inhibited reassembling of photogenerated charge carriers. As a result, the ternary MoS2/Bi2S3/Bi2WO6 composite photoanode presents excellent PEC performance, achieving a great photocurrent density at around 3.2 mA cm−2, 17.78 times as high as that of pure Bi2WO6. Through connecting with a MoS2-Ni foam photocathode in series, this tandem PEC cell with dual-photoelectrodes manifested an efficient solar-driven hydrogen production with an output of 18.32 µmol cm−2 h−1. Li et al. reported a programmable flexible heterojunction photoanode with a built-in tandem Z-scheme configuration, fabricated by in situ growing a MoSe2–CdS–ZnO array on Au paper with nanosheet-interlaced-nanorod architectures (Fig. 8e and f).111 DFT calculation and UPS analysis revealed the relevant bandgap values and double staggered energy band alignment, displayed in Fig. 8g. Photocatalytic degradation of terephthalic acid (TA) and RhB further corroborates the direct Z-scheme charge transport mechanism within the interfaces. Through aligning the Fermi levels of ZnO (0.86 eV), CdS (0.19 eV), and MoSe2 (0.08 eV), two parallel internal electric fields (from MoSe2 to CdS and CdS to ZnO) with an electron depletion/accumulation layer near their interface are established, which drive the CB electrons of both ZnO and CdS to the VB of CdS and MoSe2, respectively. Consequently, electrons and holes enrich the CB of MoSe2 and the VB of ZnO with a better redox ability for further PEC reactions. Benefiting from the construction of tandem Z-scheme interfaces (CdS-ZnO and MoSe2-CdS), powerful driving forces are formed to promote rapid charge separation and transfer spatially. Combining the strengthened optical absorption, photothermal effects, and unique 3D cross-linked network structure, this fabricated MoSe2–CdS–ZnO Z-scheme heterostructure photoanode exhibits an admirable PEC HER property (39.7 µmol cm−2 h−1). What's more, another convincing case involves the MoOx/MoS2/FTO Z-scheme heterostructure photoanode reported by Ghosh et al., which is prepared through the facile solvothermal approach, followed by a two-step annealing technique.80 The experimental results show that the formed Z-scheme heterostructure photoanode exhibits a higher photoresponse and lower charge transfer resistance compared to the pristine MoOx. All these examples illustrate the appealing superiorities of TMD-based Z-scheme heterostructure photoanodes in increasing the overall quantum yield and efficiency. However, the construction and characterization of TMD-based direct Z-scheme heterojunction photoanodes are still challenging and need further exploration.
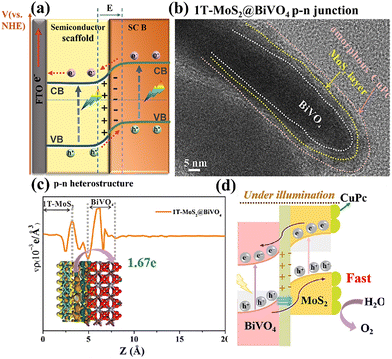 | ||
| Fig. 9 (a) Schematic illustration of p–n heterostructure; (b) HRTEM image of the CuPc@1T-MoS2@BiVO4 sample; (c) planar averaged charge density difference Δρ along the Z direction for the 1T-MoS2@BiVO4 heterostructure; (d) the relevant energy band structure distribution after contact under illumination and the possible charge transfer pathway analysis over the 1T-MoS2@BiVO4 p–n heterostructure photoanode. Reprinted from ref. 113 with permission. Copyright 2023 Elsevier. | ||
Recently, TMD-based Schottky junction photoanodes have also demonstrated their potential for PEC HER applications. For instance, Xu et al. reported the design of a novel Au/MoS2 photoanode for PEC water splitting, which is prepared by first ion sputtering the Au layer on carbon fibre cloth (CFC) substrate, followed by hydrothermal growth of MoS2 nanosheets (Fig. 10a).119 Through comparing the onset potentials observed from their linear sweep J–V curves (Fig. S7), it is inferred that the photoexcitation of Au plasmons can efficiently modulate the interfacial Schottky barrier. As illustrated in Fig. 10b and c, under illumination, the free electrons of Au oscillating into the SPR state exhibits a higher energy level than the CB of MoS2, which could rebalance the Fermi levels and change the band bending directions, thus inducing a transition from an n-type to a p-type Schottky junction at the interface. This change could greatly promote the electron transfer dynamics, benefiting charge separation and output. As a result, with the embedded Au layer, the prepared MoS2/Au@CFC photoanode exhibits a photocurrent density of 10 mA cm−2 at 1.23 VRHE and a maximum ABPE of around 1.27% (Fig. 10d), exceeding a 5-times increase relative to the MoS2@CFC counterpart. A similar result is displayed in the TiO2/(BPEI/MoS2 QDs)n photoanode reported by Lu et al.,120 which is composed of branched polyethylenimine (BPEI) and MoS2 quantum dots (QDs) electrostatically assembled onto the TiO2 nanotube array in a layer-to-layer stacking fashion, as shown in Fig. S8a. Systematic analysis proves that the introduced MoS2 QDs as a Schottky junction build the cascade electron transfer channel and branched BPEI serves as a directional hole transport channel, synergistically contributing to the accelerated charge separation and prolonged electron lifetimes, then causing the enhanced PEC water splitting (Fig. S8b). The relevant PEC water splitting mechanism over the reported TiO2/(BPEI/MoS2 QDs)n photoanode system is schematically illustrated in Fig. S8c.
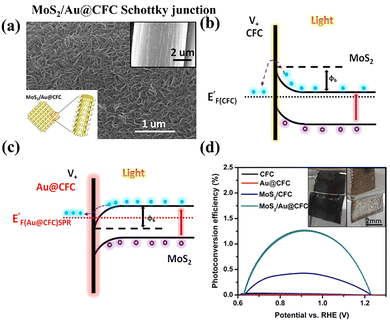 | ||
| Fig. 10 (a) SEM image of the MoS2/Au@CFC photoanode; (b and c) interface energy band structure with charge transfer pathway of the n-type MoS2/carbon fiber cloth (CFC) Schottky junction and p-type MoS2/Au@CFC Schottky junction; (d) the calculated ABPE photoconversion efficiencies (%) with a digital photo of PEC O2 production as the inset, over the previous reported CFC, Au@CFC, MoS2/CFC, and MoS2/Au@CFC Schottky junction photoanode systems; Reprinted from ref. 119 with permission. Copyright 2017 American Chemical Society. | ||
As summarized in the above literature and in Table 1, integrating TMDs into type-I, type-II, Z-scheme, p–n, and Schottky heterojunctions through rational structural design and interfacial engineering can effectively enhance PEC HER performance by promoting charge separation and transport. Among these configurations, the type-II heterojunction is the most widely explored due to its conceptual simplicity and reliable charge-separation capability. However, achieving an optimal balance between efficient carrier transfer dynamics and the preservation of strong redox potentials is still a critical challenge. In contrast, direct Z-scheme heterostructures, although less extensively studied, offer greater architecture innovation in charge transfer channels that eliminates the need for electron mediators and enables superior retention of photogenerated redox power, demonstrating significant promise for practical PEC applications. Nevertheless, the development of scalable synthetic routes and the unambiguous verification of Z-scheme charge-transfer pathways remain major obstacles. Therefore, further intensive research into TMD-based direct Z-scheme heterostructures is urgently required in the future. What's more, beyond charge transfer pathways, TMD-based heterostructures can also be categorized according to their dimensionality, such as 0D–2D, 1D–2D, 2D–2D, and 3D–2D etc. The focus here is on low-dimensional TMD materials, particularly their 2D structures, which feature weak interlayer van der Waals interactions and dangling-bond-free surfaces. These characteristics provide significant structural flexibility, allowing free junctions with other semiconductor scaffolds without the constraint of lattice mismatches. Therefore, even when composed of identical individual components, TMD-based heterojunction photoanodes with different dimensional configurations can also exhibit markedly divergent PEC performances. But, in total, the superiority of TMD-based heterostructures and the positive effects of incorporating TMD materials, such as enhanced light absorption, established charge transport channels, improved charge separation and transfer, and reinforced photostability, have been well demonstrated in the PEC water-splitting applications analysed above.
5. Conclusion and outlook
PEC water splitting for hydrogen evolution provides a promising approach for directly converting renewable solar energy into green chemical fuels. The current major bottleneck still remains the design and development of high performing semiconductor photoanodes with simultaneously sufficient spectral absorption, quantum efficiency, and long-term durability. Low dimensional TMD photoactive materials, featuring narrow bandgaps, large surface-to-volume atomic ratios, stackability, high charge mobility, short carrier diffusion distances, and excellent photoelectronic properties, are feasible for PEC-HER, targeting scale-up in industrial realization. To address intrinsic limitations such as poor solar utilization and severe carrier recombination, the rational integration of TMDs with complementary semiconductor photoabsorbers to construct heterojunction photoanodes is the focus of current research. This feature article has reviewed the most recent advancements and our contributions in designing and constructing TMD-based heterostructure photoanodes for optimized PEC hydrogen generation. The unique advantages of TMD utilization, general heterojunction synthetic strategies (i.e., top-down and bottom-up methods), TMD-based heterostructure types categorized by charge transfer pathways (including conventional heterostructures, Z-scheme architectures, p–n and Schottky junctions) and distinct dimensionality are summarized in detail. Particular emphasis is placed on the multifaceted roles of TMDs, as photosensitizers, electron-transport mediators, passivation layers, cocatalysts, and protective coatings in optimizing light harvesting, modulating charge-transfer pathways, enhancing charge separation and transport, and improving long-term photostability, within diverse TMDs/SC heterostructure PEC systems, which are discussed separately.Despite notable progress and increasing research interest, the development of TMD heterostructure photoanodes for PEC water splitting remains at an early stage and has yet to meet the performance benchmarks required for practical industrial application. Considerable efforts and broader design considerations are still required to realize a reliable alternative PEC device. Firstly, achieving a well-defined heterostructure with a clear structure–activity relationship is essential, as the thickness of TMDs affects both their energy band structure and charge diffusion distance, which determines their suitability for high-performance PEC applications. Concurrently, the development of robust synthetic strategies enabling the controllable fabrication of TMD-based heterostructures with scalability, high crystallinity, and cost-effectiveness is crucial for their commercialization. Secondly, strengthened fundamental research on the interfacial connections and properties of low-dimensional TMD-based heterostructures is of great importance. To deepen understanding, model systems with atomic-scale precision and atomically sharp interfaces based on advanced spectroscopic techniques and DFT computations are highly required, enabling elucidation of interfacial mechanisms, optimization of heterostructure design, and enhancement of device performance. Thirdly, although 1T-phase TMDs typically exhibit superior catalytic activity compared to their 2H-phase counterparts, they suffer from inherent instability under ambient conditions and during photoreaction processes. Therefore, future research should prioritize strategies to stabilize the 1T phase, such as electron-donor doping (Re, V), intercalation, noble-metal mediated SPR hot-electron injection, and other advanced phase-engineering approaches.125–128 These efforts will be crucial for fully exploiting the catalytic potential of 1T-phase TMDs in PEC applications. Fourth, interfacial adhesion between TMDs and semiconductor scaffolds could be further enhanced through substrate surface activation (plasma/UV-ozone) or nanoscale texturing, interlayer engineering (ultrathin oxides or functional polymer) and chemical functionalization for strengthening the junction chemical bonding, reducing interface defects, and promoting coherent heterojunction formation. Meanwhile, employing conformal encapsulation of OER cocatalysts, such as transition metal-based oxides, MOFs, phosphates or supramolecular cocatalysts, to isolate TMDs from electrolytes, avoid intercalation, and optimize hole extraction is critical to mitigating degradation and photocorrosion, thereby improving their photostability.129–132 Or, further exploration of suitable organic sacrificial agents, as alternatives to conventional hole scavengers (e.g., Na2S/Na2SO3), is feasible to rationally utilize surface holes for producing high-value-added chemicals while synchronously accomplishing stable hydrogen evolution. Finally, future efforts should also focus on the development of TMD-based heterostructure photoanode–photocathode configurations or photovoltaic–photoelectrochemical (PV–PEC) tandem architectures to enable unassisted solar water splitting. Integrating these photoelectrodes with state-of-the-art photovoltaic technologies, such as TOPC on Si, perovskites, and CZTS, in dual- or multi-junction layouts, or designing monolithically integrated tandem devices with sufficiently high STH efficiencies and long-term operational stability,133 represents a promising and essential direction for advancing practical PEC systems, which is also a key focus of our future research. In summary, our efforts will continue in developing highly efficient TMD-based and other heterostructure photoanodes with enhanced solar harvesting, optimized charge dynamics, and prolonged stability. We hope this feature article could inspire further innovation in the design of advanced TMD-based van der Waals heterojunction photoelectrodes for next-generation PEC catalysis.
Author contributions
Zhiyuan Peng: conceptualization, investigation, and writing – original draft. Yilu Su: conceptualization and writing – original draft. Imane En-Naji: investigation and visualization. Amir Khojastehnezhad: visualization. Mohamed Siaj: funding acquisition, resources, supervision, and writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software or code have been included and no new data were generated or analysed as part of this review.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5cc05030h.
Acknowledgements
We thank the Natural Sciences and Engineering Research Council of Canada (NSERC), the Canada Research Chairs program (CRC) and the Canada Foundation for Innovation (CFI). We thank the China Scholarship Council (CSC Scholarship). The NanoQAM center at UQAM is gratefully acknowledged for all the characterization experiments. We thank the Quebec Centre for Advanced Materials (QCAM).References
- J. H. Kim, D. Hansora, P. Sharma, J.-W. Jang and J. S. Lee, Chem. Soc. Rev., 2019, 48, 1908–1971 RSC.
- S. E. Hosseini and M. A. Wahid, Renewable Sustainable Energy Rev., 2016, 57, 850–866 CrossRef CAS.
- M. Bullock and K. More, Basic energy sciences roundtable: foundational science for carbon-neutral hydrogen technologies, USDOE Office of Science (SC), Basic Energy Sciences (BES), 2021 Search PubMed.
- M. Grätzel, Nature, 2001, 414, 338–344 CrossRef PubMed.
- H. Song, S. Luo, H. Huang, B. Deng and J. Ye, ACS Energy Lett., 2022, 7, 1043–1065 Search PubMed.
- H. Wu, L. Zhang, A. Du, R. Irani, R. van de Krol, F. F. Abdi and Y. H. Ng, Nat. Commun., 2022, 13, 6231 Search PubMed.
- T.-G. Vo, C.-C. Kao, J.-L. Kuo, C.-C. Chiu and C.-Y. Chiang, Appl. Catal., B, 2020, 278, 119303 Search PubMed.
- D. Voiry, J. Yang and M. Chhowalla, Adv. Mater., 2016, 28, 6197–6206 CrossRef CAS PubMed.
- M. Chhowalla, H. S. Shin, G. Eda, L.-J. Li, K. P. Loh and H. Zhang, Nat. Chem., 2013, 5, 263–275 CrossRef PubMed.
- X. Yu, N. Guijarro, M. Johnson and K. Sivula, Nano Lett., 2018, 18, 215–222 CrossRef CAS PubMed.
- M. Xu, T. Liang, M. Shi and H. Chen, Chem. Rev., 2013, 113, 3766–3798 CrossRef CAS PubMed.
- X. Huang, Z. Zeng and H. Zhang, Chem. Soc. Rev., 2013, 42, 1934–1946 RSC.
- V. H. Nguyen, T. P. Nguyen, T. H. Le, D. V. N. Vo, D. L. Nguyen, Q. T. Trinh, I. T. Kim and Q. V. Le, J. Chem. Technol. Biotechnol., 2020, 95, 2597–2607 Search PubMed.
- A. Hannan, A. Khalil, N. Arshad, M. B. Tahir, A. G. Wattoo and M. K. Shahzad, Energy Technol., 2022, 10, 2200546 CrossRef CAS.
- X. Zheng, Z. Guo, G. Zhang, H. Li, J. Zhang and Q. Xu, J. Mater. Chem. A, 2019, 7, 19922–19928 RSC.
- M. Faraji, M. Yousefi, S. Yousefzadeh, M. Zirak, N. Naseri, T. H. Jeon, W. Choi and A. Z. Moshfegh, Energy Environ. Sci., 2019, 12, 59–95 RSC.
- C. Li, Q. Cao, F. Wang, Y. Xiao, Y. Li, J.-J. Delaunay and H. Zhu, Chem. Soc. Rev., 2018, 47, 4981–5037 RSC.
- J. Liu, Z. Luo, X. Mao, Y. Dong, L. Peng, D. Sun-Waterhouse, J. V. Kennedy and G. I. Waterhouse, Small, 2022, 18, 2204553 CrossRef CAS PubMed.
- M. G. Walter, E. L. Warren, J. R. McKone, S. W. Boettcher, Q. Mi, E. A. Santori and N. S. Lewis, Chem. Rev., 2010, 110, 6446–6473 CrossRef CAS PubMed.
- L. Clarizia, M. N. Nadagouda and D. D. Dionysiou, Curr. Opin. Green Sustainable Chem., 2023, 41, 100825 CrossRef CAS PubMed.
- J. Jia, L. C. Seitz, J. D. Benck, Y. Huo, Y. Chen, J. W. D. Ng, T. Bilir, J. S. Harris and T. F. Jaramillo, Nat. Commun., 2016, 7, 13237 CrossRef CAS PubMed.
- W. Yang, R. R. Prabhakar, J. Tan, S. D. Tilley and J. Moon, Chem. Soc. Rev., 2019, 48, 4979–5015 RSC.
- J. Fu, Z. Fan, M. Nakabayashi, H. Ju, N. Pastukhova, Y. Xiao, C. Feng, N. Shibata, K. Domen and Y. Li, Nat. Commun., 2022, 13, 729 CrossRef CAS PubMed.
- E. Nurlaela, Y. Sasaki, M. Nakabayashi, N. Shibata, T. Yamada and K. Domen, J. Mater. Chem. A, 2018, 6, 15265–15273 RSC.
- H. Hajibabaei, D. J. Little, A. Pandey, D. Wang, Z. Mi and T. W. Hamann, ACS Appl. Mater. Interfaces, 2019, 11, 15457–15466 CrossRef CAS PubMed.
- P. Boddy, J. Electrochem. Soc., 1968, 115, 199 CrossRef CAS.
- A. Fujishima and K. Honda, Nature, 1972, 238, 37–38 Search PubMed.
- W.-H. Cheng, M. H. Richter, M. M. May, J. Ohlmann, D. Lackner, F. Dimroth, T. Hannappel, H. A. Atwater and H.-J. Lewerenz, ACS Energy Lett., 2018, 3, 1795–1800 Search PubMed.
- Y. Tachibana, L. Vayssieres and J. R. Durrant, Nat. Photonics, 2012, 6, 511–518 CrossRef CAS.
- M. T. Mayer, Curr. Opin. Electrochem., 2017, 2, 104–110 CrossRef CAS.
- K. Peng, X. Wang and S.-T. Lee, Appl. Phys. Lett., 2008, 92 Search PubMed.
- Z. Peng, Y. Su, M. Jafari and M. Siaj, J. Mater. Sci. Technol., 2023, 167, 107–118 Search PubMed.
- T. A. Ho, C. Bae, J. Joe, H. Yang, S. Kim, J. H. Park and H. Shin, ACS Appl. Mater. Interfaces, 2019, 11, 37586–37594 Search PubMed.
- L. Clarizia, D. Spasiano, I. Di Somma, R. Marotta, R. Andreozzi and D. D. Dionysiou, Int. J. Hydrogen Energy, 2014, 39, 16812–16831 CrossRef CAS.
- C. Jiang, S. J. Moniz, A. Wang, T. Zhang and J. Tang, Chem. Soc. Rev., 2017, 46, 4645–4660 RSC.
- Y. Zhao, Z. Niu, J. Zhao, L. Xue, X. Fu and J. Long, Electrochem. Energy Rev., 2023, 6, 14 Search PubMed.
- H. Zhang, D. Li, W. J. Byun, X. Wang, T. J. Shin, H. Y. Jeong, H. Han, C. Li and J. S. Lee, Nat. Commun., 2020, 11, 4622 CrossRef PubMed.
- V. K. Khanna, Eur. J. Phys., 2004, 25, 221 CrossRef CAS.
- C. Xu, H. Wang, H. Guo, K. Liang, Y. Zhang, W. Li, J. Chen, J. S. Lee and H. Zhang, Nat. Commun., 2024, 15, 9712 CrossRef CAS PubMed.
- K. Kang, C. Tang, J. H. Kim, W. J. Byun, J. H. Lee, M. H. Lee, H. Zhang and J. S. Lee, ACS Catal., 2023, 13, 7002–7012 CrossRef CAS.
- K. Sivula, F. L. Formal and M. Gratzel, Chem. Mater., 2009, 21, 2862–2867 CrossRef CAS.
- L. M. Peter, K. U. Wijayantha and A. A. Tahir, Faraday Discuss., 2012, 155, 309–322 Search PubMed.
- W. W. Gärtner, Phys. Rev., 1959, 116, 84–87 Search PubMed.
- L. K. Dhandole, H. H. Lee, W.-S. Chae, J. S. Jang and J. S. Lee, J. Energy Chem., 2022, 65, 415–423 CrossRef.
- K. Sivula, R. Zboril, F. Le Formal, R. Robert, A. Weidenkaff, J. Tucek, J. Frydrych and M. Gratzel, J. Am. Chem. Soc., 2010, 132, 7436–7444 Search PubMed.
- Y. Zhang, Y. Li, D. Ni, Z. Chen, X. Wang, Y. Bu and J. P. Ao, Adv. Funct. Mater., 2019, 29, 1902101 CrossRef.
- Y. Su, Z. Peng, E.-N. Imane, P. Liu, A. Khojastehnezhad, J. Claverie and M. Siaj, ACS Appl. Nano Mater., 2025, 8, 14689–14702 Search PubMed.
- P. Liu, A. Dorfler, A. A. Tabrizi, L. Skokan, D. Rawach, P. Wang, Z. Peng, J. Zhang, A. P. Ruediger and J. P. Claverie, ACS Appl. Mater. Interfaces, 2023, 15, 27832–27844 CrossRef CAS PubMed.
- J. Gangwar, B. K. Gupta, S. K. Tripathi and A. K. Srivastava, Nanoscale, 2015, 7, 13313–13344 Search PubMed.
- Z. Peng, J. Zhang, P. Liu, J. Claverie and M. Siaj, ACS Appl. Mater. Interfaces, 2021, 13, 34658–34670 CrossRef CAS PubMed.
- Z. Peng, Y. Su and M. Siaj, Appl. Catal., B, 2023, 330, 122614 Search PubMed.
- X. Zhou, J. Wu, Y. Xiao, Y. Jiang, W. Zhang, Y. Liu, Z. Liu and J. Zhang, Sep. Purif. Technol., 2023, 311, 123243 Search PubMed.
- Y. Xiao, Y. Jiang, E. Zhou, W. Zhang, Y. Liu, J. Zhang, X. Wu, Q. Qi and Z. Liu, J. Mater. Sci. Technol., 2023, 153, 205–218 CrossRef CAS.
- F. Chen, Z. Deng, X. Li, J. Zhang and J. Zhao, Chem. Phys. Lett., 2005, 415, 85–88 CrossRef CAS.
- H. Wang, L. Zhang, Z. Chen, J. Hu, S. Li, Z. Wang, J. Liu and X. Wang, Chem. Soc. Rev., 2014, 43, 5234–5244 RSC.
- D. Li, R. Wei, H. Yin, H. Zhang, X. Wang and C. Li, Chem. Sci., 2023, 14, 1861–1870 RSC.
- M. G. Lee, J. W. Yang, I. J. Park, T. H. Lee, H. Park, W. S. Cheon, S. A. Lee, H. Lee, S. G. Ji and J. M. Suh, Carbon Energy, 2023, 5, e321 CrossRef CAS.
- Q. Cao, J. Yu, K. Yuan, M. Zhong and J.-J. Delaunay, ACS Appl. Mater. Interfaces, 2017, 9, 19507–19512 Search PubMed.
- S. Zhao, B. Liu, K. Li, S. Wang, G. Zhang, Z.-J. Zhao, T. Wang and J. Gong, Nat. Commun., 2024, 15, 2970 CrossRef CAS PubMed.
- D. Voiry, A. Mohite and M. Chhowalla, Chem. Soc. Rev., 2015, 44, 2702–2712 RSC.
- S. Shi, Z. Sun and Y. H. Hu, J. Mater. Chem. A, 2018, 6, 23932–23977 RSC.
- J. Kang, S. Tongay, J. Zhou, J. Li and J. Wu, Appl. Phys. Lett., 2013, 102 Search PubMed.
- R. J. Toh, Z. Sofer, J. Luxa, D. Sedmidubský and M. Pumera, Chem. Commun., 2017, 53, 3054–3057 Search PubMed.
- J. Xie, H. Zhang, S. Li, R. Wang, X. Sun, M. Zhou, J. Zhou, X. W. Lou and Y. Xie, Adv. Mater., 2013, 25, 5807–5813 Search PubMed.
- J. N. Coleman, M. Lotya, A. O’Neill, S. D. Bergin, P. J. King, U. Khan, K. Young, A. Gaucher, S. De and R. J. Smith, Science, 2011, 331, 568–571 Search PubMed.
- N. Martín, N. Tagmatarchis, Q. H. Wang and X. Zhang, Chem. – Eur. J., 2020, 26, 6292–6295 Search PubMed.
- A. R. Brill, E. Koren and G. de Ruiter, J. Mater. Chem. C, 2021, 9, 11569–11587 Search PubMed.
- S. Manzeli, A. Allain, A. Ghadimi and A. Kis, Nano Lett., 2015, 15, 5330–5335 Search PubMed.
- Y. Gong, Z. Liu, A. R. Lupini, G. Shi, J. Lin, S. Najmaei, Z. Lin, A. L. Elías, A. Berkdemir and G. You, Nano Lett., 2014, 14, 442–449 CrossRef CAS PubMed.
- B. Radisavljevic, A. Radenovic, J. Brivio, V. Giacometti and A. Kis, Nat. Nanotechnol., 2011, 6, 147–150 CrossRef CAS PubMed.
- H. Abdullah, H. Shuwanto, J. Lie and M. Sillanpää, J. Environ. Chem. Eng., 2023, 11, 109356 Search PubMed.
- H. Li, J. Wu, Z. Yin and H. Zhang, Acc. Chem. Res., 2014, 47, 1067–1075 Search PubMed.
- M. Wang, S. Osella, R. Brescia, Z. Liu, J. Gallego, M. Cattelan, M. Crisci, S. Agnoli and T. Gatti, Nanoscale, 2023, 15, 522–531 Search PubMed.
- D. B. Seo, J. Kim, Y. M. Jo, D. I. Kim, T. G. Lim, S. Kang, S. Yim, S. S. Lee, E. T. Kim and K. S. An, Energy Environ. Mater., 2025, 8, e70055 Search PubMed.
- K. Roy, S. Maitra, D. Ghosh, P. Kumar and P. Devi, Chem. Eng. J., 2022, 435, 134963 CrossRef CAS.
- J. B. Naceur, R. B. Zaghouani, M. Amlouk, M. A. Zaabi and R. Chtourou, Inorg. Chem. Commun., 2023, 147, 110270 Search PubMed.
- Z. Peng, Y. Su, I. Ennaji, A. Khojastehnezhad and M. Siaj, Chem. Eng. J., 2023, 477, 147082 CrossRef CAS.
- S. S. Bhat, S. A. Pawar, D. Potphode, C.-K. Moon, J. M. Suh, C. Kim, S. Choi, D. S. Patil, J.-J. Kim and J. C. Shin, Appl. Catal., B, 2019, 259, 118102 CrossRef CAS.
- A. Pareek, H. G. Kim, P. Paik and P. H. Borse, J. Mater. Chem. A, 2017, 5, 1541–1547 RSC.
- D. Ghosh, P. Devi, O. S. Kushwaha, R. Kumar and P. Kumar, ACS Appl. Energy Mater., 2020, 3, 6834–6844 Search PubMed.
- Z. Zheng and I. M. Lo, Appl. Catal., B, 2021, 299, 120636 Search PubMed.
- H. Zhao, X. Wei, R. Gu, M. Xie and W. Han, Appl. Phys. Lett., 2024, 125, 183901 CrossRef CAS.
- S. Vanka, Y. Wang, P. Ghamari, S. Chu, A. Pandey, P. Bhattacharya, I. Shih and Z. Mi, Sol. RRL, 2018, 2, 1800113 CrossRef.
- J. Wang, Y. Peng, T. Zhou, J. Fu, W. Quan, Y. Cheng, H. Ding and Y. Zhang, Small, 2025, 2407233 CrossRef PubMed.
- Z. Zhou, P. Li, X. Gao, J. Chen, K. Akhtar, E. M. Bakhsh, S. B. Khan, Y. Shen and M. Wang, J. Environ. Chem. Eng., 2022, 10, 106900 CrossRef CAS.
- Z. Masoumi, M. Tayebi, M. Kolaei, A. Tayyebi, H. Ryu, J. I. Jang and B.-K. Lee, ACS Appl. Mater. Interfaces, 2021, 13, 39215–39229 CrossRef CAS PubMed.
- F. M. Pesci, M. S. Sokolikova, C. Grotta, P. C. Sherrell, F. Reale, K. Sharda, N. Ni, P. Palczynski and C. Mattevi, ACS Catal., 2017, 7, 4990–4998 Search PubMed.
- F. Li, D. Benetti, M. Zhang, L. Shi, J. Feng, Q. Wei and F. Rosei, ACS Appl. Mater. Interfaces, 2022, 14, 54790–54802 CrossRef CAS PubMed.
- Y.-H. Xiao and W.-D. Zhang, Electrochim. Acta, 2017, 252, 416–423 CrossRef CAS.
- D.-B. Seo, V. Dongquoc, R. A. Jayarathna, S. Lee, J.-H. Lee and E.-T. Kim, J. Alloys Compd., 2022, 911, 165090 CrossRef CAS.
- J. Yadav, A. Phutela, J. A. Khan, S. Bhattacharya and J. Singh, Int. J. Hydrogen Energy, 2024, 79, 826–838 Search PubMed.
- Z. Masoumi, M. Tayebi, M. Kolaei and B.-K. Lee, J. Alloys Compd., 2022, 890, 161802 Search PubMed.
- K. Karmakar, D. Maity, D. Pal, K. Mandal and G. G. Khan, ACS Appl. Nano Mater., 2020, 3, 1223–1231 CrossRef CAS.
- A. R. Fareza, L. Roza, F. A. A. Nugroho and V. Fauzia, Surf. Interfaces, 2023, 37, 102663 Search PubMed.
- P. Shinde, Y. Hase, V. Doiphode, B. R. Bade, D. Kale, S. Rahane, J. Thombare, D. Borkar, S. R. Rondiya and M. Prasad, ACS Appl. Energy Mater., 2025, 8, 935–948 Search PubMed.
- M. Chia, W. Chiu, P. Khiew, N. Chanlek, H. Lee, C. Haw, R. Abd-Shukor and M. A. A. Hamid, CrystEngComm, 2025, 27, 749–761 Search PubMed.
- R. A. Jayarathna, J.-H. Heo and E.-T. Kim, Nanomaterials, 2024, 14, 1628 CrossRef CAS PubMed.
- J. Wu, M. Meng, X.-D. Du, M. Li, L. Jin and W. Liu, Inorg. Chem., 2024, 63, 6192–6201 Search PubMed.
- N. N. Rosman, N. R. A. M. Shah, S. N. F. Moridon, K. Arifin, L. J. Minggu, N. A. Ludin and R. M. Yunus, Int. J. Hydrogen Energy, 2025, 104, 324–335 CrossRef CAS.
- M. Rezaei, A. A. Ensafi and E. Heydari-Bafrooei, J. Ind. Eng. Chem., 2025, 146, 589–602 CrossRef CAS.
- H.-B. Do, L. D. Pham, T. X. Phan, W.-Z. Chen, L.-W. Lan, H.-J. Lin, V.-H. Nguyen, C.-L. Dong, A. S. Kumar and A. F. El-Mahdy, Nanotechnology, 2024, 35, 385703 CrossRef PubMed.
- F. Khodabandeh, H. Abdizadeh and M. R. Golobostanfard, ACS Appl. Energy Mater., 2024, 8, 170–180 Search PubMed.
- M. Tayebi, Z. Masoumi and B.-K. Lee, Ultrason. Sonochem., 2021, 70, 105339 Search PubMed.
- G. C. Behera, B. Biswal, J. K. Bidika, B. R. K. Nanda, S. Alwarappan, J. K. Rath and S. C. Roy, Small, 2025, 21, 2406715 CrossRef CAS PubMed.
- S. Ladhane, S. Shah, V. Doiphode, P. Shinde, D. Kale, S. Rahane, J. Thombare, M. Ingole, P. Vairale and Y. Hase, J. Power Sources, 2025, 656, 238056 CrossRef CAS.
- S. Ladhane, S. Shah, V. Doiphode, P. Shinde, A. Punde, D. Kale, S. Rahane, J. Thombare, Y. Hase and A. Waghmare, Mater. Chem. Phys., 2025, 334, 130440 Search PubMed.
- J. Lai, B. Wang, Y. Gong, C. Sun, W. Wang and W. Yang, RSC Adv., 2023, 13, 4150–4155 RSC.
- P. Chauhan, A. B. Patel, G. Solanki, H. K. Machhi, C. Sumesh, S. S. Soni, V. Patel and V. Pathak, J. Phys. Chem. C, 2021, 125, 14729–14740 Search PubMed.
- A. H. Hendi, A. M. Osman, I. Khan, T. A. Saleh, T. A. Kandiel, T. F. Qahtan and M. K. Hossain, ACS Omega, 2020, 5, 31644–31656 Search PubMed.
- A. Wan, Z. Ren, Y. He, Y. Zhang, S. Zhang, M. Tu, J. Li and M. Qiu, Appl. Surf. Sci., 2025, 690, 162607 CrossRef CAS.
- L. Li, H. Shi, H. Yu, X. Tan, Y. Wang, S. Ge, A. Wang, K. Cui, L. Zhang and J. Yu, Appl. Catal., B, 2021, 292, 120184 Search PubMed.
- Y. Xu, X. Zhang, Z. Chen, K. Kempa, X. Wang and L. Shui, J. Mater. Sci. Technol., 2021, 68, 1–7 Search PubMed.
- L. Yu, K. Xue, H. Luo, C. Liu, H. Liu, H. Zhu and Y. Zhang, Chem. Eng. J., 2023, 472, 144965 CrossRef CAS.
- M. Tayebi, Z. Masoumi, M. Kolaei, A. Tayyebi, M. Tayebi, B. Seo, C.-S. Lim, H.-G. Kim and B.-K. Lee, Chem. Eng. J., 2022, 446, 136830 CrossRef CAS.
- Z. Masoumi, M. Tayebi and B.-K. Lee, Ultrason. Sonochem., 2021, 72, 105403 Search PubMed.
- X. Yang, J. Sun, H. Fu, J. Chen, J. Fan, X. Tong, X. Ran and X. An, Powder Technol., 2024, 431, 119051 Search PubMed.
- Z. Masoumi, M. Tayebi, M. Kolaei and B.-K. Lee, Appl. Catal., B, 2022, 313, 121447 CrossRef CAS.
- H. Li, C. Yang, X. Wang, J. Zhang, J. Xi, G. Du and Z. Ji, J. Am. Ceram. Soc., 2020, 103, 1187–1196 CrossRef CAS.
- X. Xu, G. Zhou, X. Dong and J. Hu, ACS Sustainable Chem. Eng., 2017, 5, 3829–3836 CrossRef CAS.
- Z.-Q. Wei, X.-C. Dai, S. Hou, Y.-B. Li, M.-H. Huang, T. Li, S. Xu and F.-X. Xiao, J. Mater. Chem. A, 2020, 8, 177–189 Search PubMed.
- H. Cho, J.-Y. Kim, D. S. Shin, J. S. Lee, J. Shim, I.-H. Lee, W. K. Choi, N. Kwon, S. Park and M. S. Yazici, Adv. Sensor Energy Mater., 2024, 3, 100088 Search PubMed.
- L. Yu, K. Xue, H. Luo, C. Liu, H. Liu, H. Zhu and Y. Zhang, Chem. Eng. J., 2023, 472, 144965 Search PubMed.
- L. Zhang, J. Sun, M. Zhao, Y. Wei, T. Luo, Z. Zhao and Y. Yan, Molecules, 2024, 29, 1070 CrossRef CAS PubMed.
- G. Bharath, K. Rambabu, B. Alqassem, P. P. Morajkar, M. A. Haija, A. K. Nadda, V. K. Gupta and F. Banat, Chem. Eng. J., 2023, 456, 141062 CrossRef CAS.
- Q. Liu, X. Li, Q. He, A. Khalil, D. Liu, T. Xiang, X. Wu and L. Song, Small, 2015, 11, 5556–5564 CrossRef CAS PubMed.
- A. N. Enyashin, L. Yadgarov, L. Houben, I. Popov, M. Weidenbach, R. Tenne, M. Bar-Sadan and G. Seifert, J. Phys. Chem. C, 2011, 115, 24586–24591 CrossRef CAS.
- X. Sun, J. Dai, Y. Guo, C. Wu, F. Hu, J. Zhao, X. Zeng and Y. Xie, Nanoscale, 2014, 6, 8359–8367 RSC.
- Y. Liu, W. Huang, W. Chen, X. Wang, J. Guo, H. Tian, H. Zhang, Y. Wang, B. Yu and T.-L. Ren, Appl. Surf. Sci., 2019, 481, 1127–1132 CrossRef CAS.
- L. Liardet, J. E. Katz, J. Luo, M. Grätzel and X. Hu, J. Mater. Chem. A, 2019, 7, 6012–6020 RSC.
- J. B. Pan, B. H. Wang, J. B. Wang, H. Z. Ding, W. Zhou, X. Liu, J. R. Zhang, S. Shen, J. K. Guo and L. Chen, Angew. Chem., Int. Ed., 2021, 60, 1433–1440 CrossRef CAS PubMed.
- G. Liu, Y. Zhao, R. Yao, N. Li, M. Wang, H. Ren, J. Li and C. Zhao, Chem. Eng. J., 2019, 355, 49–57 CrossRef CAS.
- J. Miao, C. Lin, X. Yuan, Y. An, Y. Yang, Z. Li and K. Zhang, Nat. Commun., 2024, 15, 2023 CrossRef CAS PubMed.
- G. Jung, C. Moon, F. Martinho, Y. Jung, J. Chu, H. Park, A. Hajijafarassar, R. Nielsen, J. Schou and J. Park, Adv. Energy Mater., 2023, 13, 2301235 CrossRef CAS.
Footnote |
| † These authors contributed equally. |
| This journal is © The Royal Society of Chemistry 2026 |