Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Correction: Dynamic conformational equilibria in the active states of KRAS and NRAS
Enrico Rennellab,
Chrystèle Henrya,
Callum J. Dicksonc,
Florian Georgescauldd,
Thomas E. Walesd,
Dirk Erdmanna,
Simona Cotestaa,
Michel Mairaa,
Richard Sedrania,
Saskia M. Brachmanna,
Nils Ostermanna,
John R. Engend,
Lewis E. Kayb,
Kim S. Beyer*a,
Rainer Wilcken*a and
Wolfgang Jahnke*a
aNovartis Biomedical Research, Basel, Switzerland. E-mail: wolfgang.jahnke@novartis.com; rwilcken@flaretx.com; kim.beyer@novartis.com
bDepartment of Biochemistry, University of Toronto, Toronto, Canada
cNovartis Biomedical Research, Cambridge, MA, USA
dDepartment of Chemistry and Chemical Biology, Northeastern University, Boston, MA, USA
First published on 5th May 2026
Abstract
Correction for ‘Dynamic conformational equilibria in the active states of KRAS and NRAS’ by Enrico Rennella et al., RSC Chem. Biol., 2025, 6, 106–118, https://doi.org/10.1039/D4CB00233D.
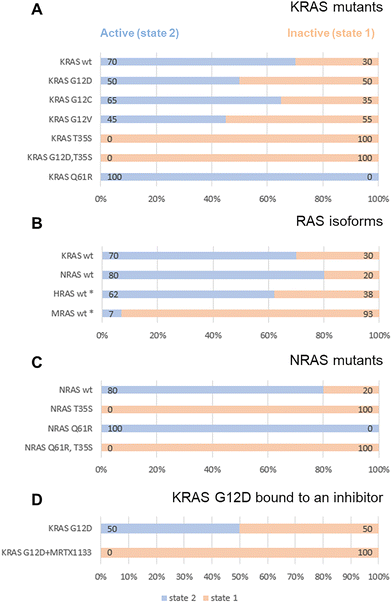
The authors regret two instances of incorrectly displayed data in the published article whereby the conformation states corresponding to inactive (state 1) and active (state 2) have been mislabelled and should be swapped.
One instance is in a line of text on page 113 under the subheading “Hydrogen/deuterium exchange mass spectrometry” and a second instance is in Fig. 2 in the originating manuscript. The corrected manuscript text, figure and accompanying text are below. The conclusions of the paper have not been affected.
“Again, this is in line with the observations from 31P NMR, although the extent to which these regions saw increases in deuterium incorporation in HDX-MS was surprisingly large, given the relatively small shift seen in 31P NMR from active state-2 to inactive state-1 (70/30 in wildtype to 50/50 in G12D).”
The Royal Society of Chemistry apologises for these errors and any consequent inconvenience to authors and readers.
| This journal is © The Royal Society of Chemistry 2026 |