Open Access Article
Open Access ArticleFluorescent cationic fluorinated oxazoliniums for cysteine bioconjugation via an SNAr reaction
Karen Ka-Yan
Kung†
ab,
Yosephine Tania
Limanto†
a,
Ajcharapan
Tantipanjaporn
a,
Jie-Ren
Deng
c,
Lai-Yi
Tsang
c and
Man-Kin
Wong
 *ab
*ab
aResearch Institute for Future Food, Department of Food Science and Nutrition, The Hong Kong Polytechnic University, Hung Hom, Hong Kong, China. E-mail: mankin.wong@polyu.edu.hk
bCentre for Eye and Vision Research (CEVR), 17W Hong Kong Science Park, Hong Kong, China
cDepartment of Applied Biology and Chemical Technology, The Hong Kong Polytechnic University, Hung Hom, Hong Kong, China
First published on 20th February 2026
Abstract
Using cationic fluorinated oxazolinium compounds, fluorescent cysteine-selective SNAr bioconjugation proceeds under mild conditions, resulting in fluorescent-labelled peptides and proteins with moderate to excellent conversions of up to 99%. Live cell imaging studies reveal good compatibility of these oxazoliniums as fluorescent dyes for mitochondrial targeting.
Introduction
Fluorescent labelling allows the attachment of fluorophores to biomolecules, resulting in fluorescent bioconjugates with excellent selectivity and functional diversity for visualizing and tracking biological activities.1–3 Cysteine is commonly used as a handle in bioconjugation due to its high nucleophilicity and relatively low abundance.1,4 Therefore, different classes of electrophiles, including α,β-unsaturated carbonyls, maleimides and hypervalent iodine compounds, have been developed for cysteine-based modification of peptides and proteins.Cysteine arylation by fluorinated aromatic compounds via nucleophilic aromatic substitution (SNAr) has demonstrated advantages over metal- or photo-catalyzed arylation.5,6 Since 2013, Pentelute et al.,7,8 Derda et al.,9 Cobb et al.,10 Wu et al.11 and Harran et al.12 have developed polyfluoroaromatic compounds for cysteine-selective bioconjugation and Cys–Cys stapling of native peptides (Fig. 1a). Recently, pyridinium salts, such as N-methyl-o-fluoropyridinium iodide (CAP1), showed excellent reactivity due to the cationic nature, water solubility and highly polarized C–F bonds towards rapid thiol arylation (Fig. 1b).13
Various advancements have been made in fluorescence analysis using small molecule fluorescent probes including the incorporation of fluorine to fluorophores to improve imaging capabilities, targeting functionalities and overall biological activities.14–16 To achieve fluorescent cysteine bioconjugation, attachment of a fluorescent dye into the bioconjugation reagents is necessary, requiring additional synthetic effort. Thus, it is interesting to develop a fluorescent bioconjugation reagent without the need for addition of a separate fluorescent dye. Notably, Zhang et al. introduced a series of fluorescent polyfluoroporphyrins for the labelling of cysteine-containing peptides and proteins via SNAr reactions, while preserving water solubility, cationic nature and tunable fluorescence (Fig. 1c).17
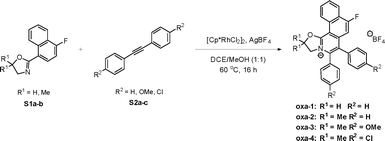
Although many cationic polycyclic heteroaromatics, such as isoquinoliniums, quinoliziniums, and pyridiniums, have been reported as water-soluble fluorophores, very few reports are on the synthesis and applications of oxazoliniums.18–20 Here, we report a series of fluorescent cationic fluorinated oxazoliniums for cysteine bioconjugation via an SNAr reaction (Fig. 1d). By using rhodium (Rh)-catalyzed reaction, a series of fluorinated oxazolinium compounds were prepared using a modular approach.21
Investigation of the photophysical properties and applications of the fluorinated oxazoliniums for cysteine bioconjugation and live cell imaging was performed.
Results and discussion
Design and synthesis of oxazoliniums
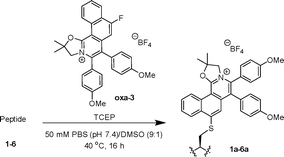
Based on our previous study on the development of polycyclic quinoliziniums22–24 and oxazoliniums,25 oxazolinium oxa-1 was first synthesized via a Rh-catalyzed C–H annulation between oxazoline S1a and diphenylacetylene S2a in the presence of silver salt (Scheme 1). Oxazolinium oxa-1 was characterized by NMR and mass spectrometry (SI).We initiated the study through the reaction of oxa-1 (10 equiv.) with cysteine-containing peptide 1 (STSSSCNLSK) (0.1 mM, 1 equiv.) for 18 h at room temperature (Scheme S1, SI). By utilization of LC-MS to determine the conversion, we found that reaction at room temperature led to a moderate conversion of 61% of modified peptide 1a (Fig. S13, SI). To improve the conversion, the reaction mixture was heated at 40 °C for 18 h. Unexpectedly, 75% of peptide 1 was converted to the hydrolysed product 1a′ (Fig. S15, SI). In addition, dimerization of the native peptide 1 increased from 9% to 23% at room temperature. As the C2 carbon atom of the oxazoline is susceptible to nucleophilic attack,26oxa-2–4 were designed with geminal dimethyl groups to overcome the hydrolysis of the target product.
Optimization of the peptide modification reaction conditions
Treatment of the peptide 1 with oxa-2 (10 equiv.) at room temperature gave a moderate conversion of 66% and 10% dimerization (Table 1, entry 1). Upon heating of the reaction mixture at 40 °C, a significant increase to 86% conversion was observed (entry 2). As dimerization of the peptide was a competing reaction to the cysteine arylation, TCEP (a reducing agent) was added to inhibit the dimerization (entry 3). Optimization of the reaction conditions was conducted by screening different conditions and varying the amount of oxa-2 and TCEP (entries 4–9). To our delight, up to 96% conversion was attained when only 5 equivalents of oxa-2 and 1 equivalent of TCEP were utilized under heating at 40 °C (entry 7).| Entry | Compound (equiv.) | TCEP (equiv.) | Temp. (°C) | Time (h) | Product | Conv.b (%) |
|---|---|---|---|---|---|---|
a Reaction conditions for modification: Treatment of peptide 1 (0.1 mM) with oxa-2 in the presence of TCEP, in 50 mM PBS buffer (pH 7.4)/DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1).
b Conversion of the modification was determined by LC-MS/MS analysis.
c Optimized conditions following time-course study, oxa-3, oxa-4 and oxazoline S1b were utilized. 1).
b Conversion of the modification was determined by LC-MS/MS analysis.
c Optimized conditions following time-course study, oxa-3, oxa-4 and oxazoline S1b were utilized.
|
||||||
| 1 | oxa-2 (10) | 0 | rt | 18 | 1b | 66 |
| 2 | oxa-2 (10) | 0 | 40 | 18 | 1b | 86 |
| 3 | oxa-2 (10) | 10 | rt | 18 | 1b | 64 |
| 4 | oxa-2 (10) | 10 | 40 | 18 | 1b | 94 |
| 5 | oxa-2 (5) | 5 | 40 | 18 | 1b | 95 |
| 6 | oxa-2 (5) | 2 | 40 | 18 | 1b | 96 |
| 7 | oxa-2 (5) | 1 | 40 | 18 | 1b | 96 |
| 8 | oxa-2 (3) | 1 | 40 | 18 | 1b | 91 |
| 9 | oxa-2 (1) | 1 | 40 | 18 | 1b | 89 |
| 10c | oxa-3 (5) | 1 | 40 | 16 | 1c | 90 |
| 11c | oxa-4 (5) | 1 | 40 | 16 | 1d | 99 |
| 12c | S1b (control) (5) | 1 | 40 | 16 | 1e | 0 |
With the optimized conditions in hand, a time course study was conducted. Up to 95% conversion was obtained within 16 h of reaction before reaching a plateau (Fig. S1, SI). Peptide 1 was then treated with electron-donating substituted oxa-3 and electron-withdrawing substituted oxa-4, leading to 90% and 99% conversion respectively (entries 10 and 11). The higher conversion obtained from the electron-withdrawing oxa-4 compared to oxa-2 and oxa-3 is possibly due to the higher electrophilicity of the cation due to the –Cl substituent. Furthermore, as a proof of concept, no target product was observed when peptide 1 was treated with oxazoline S1b as a control (entry 12). This observation indicated that cysteine arylation is enhanced by the cationic nature of the oxazolinium electrophiles and highly polarized C–F bonds.13
Model reactions of peptide modifications
Model reactions were conducted by treatment of oxa-2–4 (0.1 mmol, 1 equiv.) and N-Boc cysteine methyl ester (1.5 equiv.) at room temperature for 16 h to give oxa-5–7 (Scheme 2). By using mass spectrometry, no starting oxa-2–4 were observed in the crude mixture, indicating complete consumption of the starting oxazolinium compounds. However, difficulties in purification of the resulting products led to low isolated yields (accounting only for the purest fraction collected). Yet, the cysteine-arylated products oxa-5–7 were successfully characterized by NMR and mass spectrometry (SI).Evaluation of the photophysical properties and DFT calculations
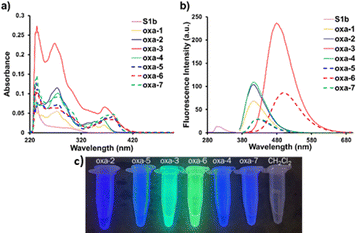
As depicted in Fig. 2 and Table 2, the photophysical properties of oxa-1–7 and S1b were studied in CH2Cl2. Oxazolinium oxa-1 (R1 = H) caused a red-shift of the absorption and emission, higher molar absorptivity (ε), and greater fluorescence quantum yield (ΦF) compared to the non-cationic S1b (entries 1 and 2). Comparing oxa-1 (R1 = H) and oxa-2 (R1 = CH3), the geminal methyl substituents of oxa-2 demonstrated a negligible effect on the photophysical properties (entries 2 and 3). The electron donating –OMe group on R2 of oxa-3 gave red-shifted emission (480 nm), a larger Stoke shifts, and the highest ΦF (0.51) (entry 4), while an electron withdrawing –Cl group on R2 of oxa-4 showed similar photophysical properties to oxa-2 (entry 5 vs. entry 3). Thus, the substituents on R2 showed significant effects on ε, emission, and ΦF. In addition, oxa-5–7 resulting from the substitution of N-Boc cysteine methyl ester with oxa-2–4 exhibited red-shifted absorption and emission (entries 6–8). Notably, the bright fluorescence images of oxa-3 and oxa-6 imply the potential application of oxa-3 in fluorescent labelling and bioimaging (Fig. 2c). Moreover, the photophysical properties of oxa-1–7 and S1b studied in 50 mM PBS (pH 7.4)/DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) also gave comparable results when using CH2Cl2 as a solvent (Fig. S2–S9 and Table S2, SI).
1) also gave comparable results when using CH2Cl2 as a solvent (Fig. S2–S9 and Table S2, SI).
| Entry | Compound | Max. abs (nm) | Max. Em (nm) | Stokes shift (nm) | ε (M−1 cm−1) | λ ex (nm) | Φ F |
|---|---|---|---|---|---|---|---|
| a Quantum yields were measured using coumarin 153 (ΦF = 0.54 in ethanol) as the standard reference. | |||||||
| 1 | S1b | 295 | 310 | 15 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
270 | 0.06 |
| 2 | oxa-1 | 363 | 413 | 50 | 22![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
360 | 0.35 |
| 3 | oxa-2 | 363 | 413 | 50 | 36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
360 | 0.33 |
| 4 | oxa-3 | 365 | 480 | 115 | 68![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
365 | 0.51 |
| 5 | oxa-4 | 363 | 413 | 50 | 35![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
360 | 0.34 |
| 6 | oxa-5 | 380 | 425 | 45 | 42![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
370 | 0.09 |
| 7 | oxa-6 | 380 | 500 | 132 | 30![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
365 | 0.40 |
| 8 | oxa-7 | 380 | 430 | 50 | 46![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
365 | 0.07 |
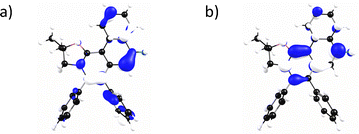
Based on the TD-DFT calculated electronic transitions, we simulated the absorption spectrum (Fig. S10a, red line, SI) for oxa-2. The simulated spectrum resembles the measured spectrum. TD-DFT calculations reveal that the lowest energy absorption band (λabs,cal = 341 nm) originated from the HOMO → LUMO transition. The HOMO is mainly contributed by the π orbital of oxazolinium and the phenyl rings, whereas the LUMO is composed of the π* of the oxazolinium ring (Fig. 3). We have also simulated the emission spectrum (Fig. S10b, red line, SI) for oxa-2, where the calculated emission maximum (S1 → S0 transition, λem,cal = 436 nm) is close to the measured value (λem = 413 nm).
Scope study for peptide modifications
A substrate scope study was conducted utilizing oxa-3 due to its possession of the highest quantum yield among other analogues. Various cysteine-containing peptides were treated with oxa-3 under the optimized conditions (Table 3). Up to 90% conversion was observed when various cysteine-containing peptides were treated (entries 1–7). No conversion was observed when peptide 7 (without cysteine) was employed (entry 7), indicating excellent cysteine selectivity.| Entry | Peptide | Amino acid sequence | Product | Conv.b (%) |
|---|---|---|---|---|
a Reaction conditions for modification: treatment of peptide (0.1 mM) with oxa-3 (5 equiv.) in the presence of TCEP (1 equiv.) in 50 mM PBS buffer (pH 7.4)/DMSO (9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) at 40 °C for 16 h.
b Conversion was determined by LC-MS/MS analysis. 1) at 40 °C for 16 h.
b Conversion was determined by LC-MS/MS analysis.
|
||||
| 1 | 1 | STSSS![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) NLSK NLSK |
1c | 90 |
| 2 | 2 | AYEMW![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) FSQR FSQR |
2a | 69 |
| 3 | 3 | KSTF![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) |
3a | 72 |
| 4 | 4 | ![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) SKFR SKFR |
4a | 89 |
| 5 | 5 | ![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) DPGYIGSR DPGYIGSR |
5a | 83 |
| 6 | 6 | AYEMW![[C with combining low line]](https://www.rsc.org/images/entities/char_0043_0332.gif) FHQR FHQR |
6a | 71 |
| 7 | 7 | YTSSSKNVVR | 7a | 0 |
Modification of proteins by fluorescent oxa-3
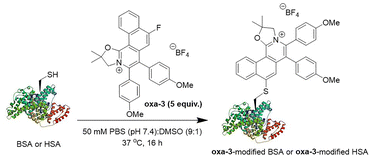
Next, oxa-3 was utilized for protein modification of bovine serum albumin (BSA; PDB ID: 4F5S) and human serum albumin (HSA; PDB ID: 1AO6) with a single free cysteine residue at position 34 (Cys-34), without TCEP to maintain the structural integrity of proteins (Scheme 3). By LC-MS analysis, 60% conversion of oxa-3-modified BSA (Fig. S53 and S54, SI) and 70% conversion of oxa-3-modified HSA (Fig. S55 and S56, SI) were achieved after 16 h. Conversely, lysozyme (PDB ID: 3LYZ) remained intact after the reaction (Fig. S57 and S58, SI).Analysis of protease-digested oxa-3-modified proteins showed that the reaction only occurred at Cys-34 of the targeted peptide fragments, while other amino acid residues remained intact (Fig. S59–S64, SI). SDS-PAGE analysis revealed a strong blue fluorescent signal for oxa-3-modified proteins while no signal was observed for native BSA and HSA at UV 365 nm (Table 4). Coomassie blue staining on the same gel gave deep blue colour signals of all native and modified proteins, indicating successful cysteine arylation of proteins by oxa-3. Both native and treated lysozyme had no fluorescence, stipulating excellent site-selectivity for peptide and protein modification.
Live cell imaging
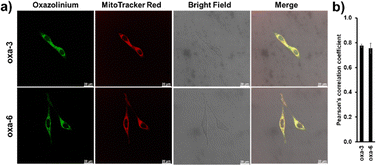
Following the success of oxa-3 in the provision of visual indication in protein modification, we sought to explore its applicability for cell imaging in biological systems. The cytotoxicity of oxa-3 and oxa-6 was evaluated at 24 h, showing moderate cytotoxicity to HeLa cells (IC50 = 16.11 and 14.54 µM, respectively, Fig. S66 and S67, SI). Then, the colocalization subcellular imaging of oxa-3 and oxa-6 was investigated at a non-cytotoxic concentration. HeLa cells were incubated with oxazoliniums (5 µM) in the presence of MitoTracker Red (MTR, 500 nM) for 15 min and washed with DPBS before the confocal imaging. As shown in Fig. 4, oxa-3 and oxa-6 showed remarkable cell permeability, intense green fluorescence, and good overlapping with MTR, with a Pearson's correlation coefficient value close to 0.8, indicating that oxa-3 and oxa-6 localized in the mitochondria.Due to the high concentrations of glutathione (GSH, 1–10 mM) present in the cells,27 we studied the reactivity of oxa-3 with GSH by incubating oxa-3 (1 mM) with GSH (1 mM) for 15 min. Our results showed that 31% conversion was observed, indicating that the oxazoliniums have reactivity towards –SH containing biomolecules in cells (Fig. S69–S71, SI).
Conclusions
In conclusion, we have developed a series of fluorescent cationic fluorinated oxazoliniums for cysteine-selective arylation via an SNAr reaction. Measurements of the photophysical properties and quantum yields presented oxa-3, with the electron-donating –OMe group, with a high quantum yield of 0.51. Under the optimized conditions, modifications of cysteine-containing peptides afforded up to 99% conversion. Finally, the applicability of oxa-3 to provide visual indication for fluorescent labelling of cysteine-containing proteins and cell imaging was successfully demonstrated. This work highlights the potential of these fluorescent oxazoliniums for broad applications in chemical biology and biological studies.Author contributions
K. K.-Y. K. and Y. T. L.: conceptualization, investigation, data curation, writing – original draft, and writing – review and editing; A. T.: investigation, formal analysis, and writing – original draft; J.-R. D.: formal analysis and writing – original draft; L.-Y. T.: investigation; M.-K. W.: conceptualization, funding acquisition, supervision, and writing – review and editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: details of experimental procedures, LC-MS analyses, DFT data and NMR data. See DOI: https://doi.org/10.1039/d5cb00289c.Supplementary data have been deposited with the Protein Data Bank.28–30
Acknowledgements
We gratefully acknowledge the financial support from the Research Grants Council of the Hong Kong Special Administrative Region, China (PolyU15300520), the PolyU Postdoc Matching Fund Scheme (P0043412: W262), the InnoHK initiative of the Innovation and Technology Commission of the Hong Kong Special Administrative Region Government, the State Key Laboratory of Chemical Biology and Drug Discovery, The Hong Kong Polytechnic University (P0053072: ZZV2), the University Research Facility in Life Sciences (ULS) and the University Research Facility in Chemical and Environmental Analysis (UCEA) of PolyU. We would like to thank Prof. F. K.-C. Leung from Department of Applied Biology and Chemical Technology (ABCT) of The Hong Kong Polytechnic University for hardware support during the in vitro and cell imaging experiments.References
- C. D. Spicer and B. G. Davis, Nat. Commun., 2014, 5, 4740 CrossRef CAS PubMed.
- N. Stephanopoulos and M. B. Francis, Nat. Chem. Biol., 2011, 7, 876–884 CrossRef CAS PubMed.
- L. R. Malins, Curr. Opin. Chem. Biol., 2018, 46, 25–32 CrossRef CAS PubMed.
- S. B. Gunnoo and A. Madder, ChemBioChem, 2016, 17, 529–553 CrossRef CAS PubMed.
- C. Zhang, E. V. Vinogradova, A. M. Spokoyny, S. L. Buchwald and B. L. Pentelute, Angew. Chem., Int. Ed., 2019, 58, 4810–4839 CrossRef CAS PubMed.
- W. Lin, X. Ding, J.-W. Han, L.-S. Yu and F.-J. Chen, Org. Chem. Front., 2025, 12, 2777–2789 RSC.
- A. M. Spokoyny, Y. Zou, J. J. Ling, H. Yu, Y.-S. Lin and B. L. Pentelute, J. Am. Chem. Soc., 2013, 135, 5946–5949 CrossRef CAS PubMed.
- Y. Zou, A. M. Spokoyny, C. Zhang, M. D. Simon, H. Yu, Y.-S. Lin and B. L. Pentelute, Org. Biomol. Chem., 2014, 12, 566–573 RSC.
- S. Kalhor-Monfared, M. Jafari, J. Patterson, P. Kitov, J. Dwyer, J. Nuss and R. Derda, Chem. Sci., 2016, 7, 3785–3790 RSC.
- D. Gimenez, C. A. Mooney, A. Dose, G. Sandford, C. R. Coxon and S. L. Cobb, Org. Biomol. Chem., 2017, 15, 4086–4095 RSC.
- W. Liu, Y. Zheng, X. Kong, C. Heinis, Y. Zhao and C. Wu, Angew. Chem., Int. Ed., 2017, 56, 4458–4463 CrossRef CAS PubMed.
- T. Tsunemi, S. J. Bernardino, A. Mendoza, C. G. Jones and P. G. Harran, Angew. Chem., Int. Ed., 2020, 59, 674–678 CrossRef CAS PubMed.
- B. M. Lipka, V. M. Betti, D. S. Honeycutt, D. L. Zelmanovich, M. Adamczyk, R. Wu, H. S. Blume, C. A. Mendina, J. M. Goldberg and F. Wang, Bioconjugate Chem., 2022, 33, 2189–2196 CrossRef CAS PubMed.
- S. Casa and M. Henary, Molecules, 2021, 26, 1160 CrossRef CAS PubMed.
- S. Zeng, X. Liu, Y. S. Kafuti, H. Kim, J. Wang, X. Peng, H. Li and J. Yoon, Chem. Soc. Rev., 2023, 52, 5607–5651 RSC.
- F. de Moliner, F. Nadal-Bufi and M. Vendrell, Curr. Opin. Chem. Biol., 2024, 80, 102458 CrossRef CAS PubMed.
- G.-Q. Jin, J.-X. Wang, J. Lu, H. Zhang, Y. Yao, Y. Ning, H. Lu, S. Gao and J.-L. Zhang, Chem. Sci., 2023, 14, 2070–2081 RSC.
- P. Gandeepan and C. H. Cheng, Chem. – Asian J., 2016, 11, 448–460 CrossRef CAS PubMed.
- P. Karak, S. S. Rana and J. Choudhury, Chem. Commun., 2022, 58, 133–154 RSC.
- R. Sanjana, J. Jayakumar and K. Parthasarathy, Asian J. Org. Chem., 2025, e202500177 CrossRef CAS.
- C. Z. Luo, P. Gandeepan, J. Jayakumar, K. Parthasarathy, Y. W. Chang and C. H. Cheng, Chem. – Eur. J., 2013, 19, 14181–14186 CrossRef CAS PubMed.
- J.-R. Deng, W.-C. Chan, N. C.-H. Lai, B. Yang, C.-S. Tsang, B. C.-B. Ko, S. L.-F. Chan and M.-K. Wong, Chem. Sci., 2017, 8, 7537–7544 RSC.
- W.-M. Yip, Q. Yu, A. Tantipanjaporn, W.-C. Chan, J.-R. Deng, B. C.-B. Ko and M.-K. Wong, Org. Biomol. Chem., 2021, 19, 8507–8515 RSC.
- A. Tantipanjaporn, K.-Y. K. Kung, J.-R. Deng and M.-K. Wong, Spectrochim. Acta, Part A, 2024, 319, 124524 CrossRef CAS PubMed.
- L.-Y. Tsang, A. Tantipanjaporn, K. K.-Y. Kung, H.-Y. Sit, W. Y. O, A. K.-H. Chan, N. C.-H. Lai and M.-K. Wong, Adv. Synth. Catal., 2025, 367, e70023 CrossRef CAS.
- M. N. Holerca and V. Percec, Eur. J. Org. Chem., 2000, 2257–2263 CrossRef CAS.
- C.-Y. Cui, B. Li and X.-C. Su, ACS Cent. Sci., 2023, 9, 1623–1632 CrossRef CAS PubMed.
- A. Bujacz, Structures of bovine, equine and leporine serum albumin, Acta Crystallogr., Sect. D: Biol. Crystallogr., 2012, 68, 1278–1289 DOI:10.1107/S0907444912027047.
- S. Sugio, A. Kashima, S. Mochizuki, M. Noda and K. Kobayashi, Crystal structure of human serum albumin at 2.5 A resolution, Protein Eng., 1999, 12, 439–446 DOI:10.1093/protein/12.6.439.
- R. Diamond, Real-space refinement of the structure of hen egg-white lysozyme, J. Mol. Biol., 1974, 82, 371–391 DOI:10.1016/0022-2836(74)90598-1.
Footnote |
| † These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2026 |