Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Correction: Injectable hydrogels activated with copper sulfide nanoparticles for enhancing spatiotemporal sterilization and osteogenesis in periodontal therapy
Yuting Yanga,
Chunbin Xua,
Shengqian Xua,
Yan Lia,
Ke'er Chena,
Tao Yangb,
Jiaqi Baoa,
Yajing Xub,
Jingyao Chend,
Chuanbin Mao*c,
Lili Chen*a and
Weilian Sun*a
aDepartment of Periodontology, The Second Affiliated Hospital of Zhejiang University, School of Medicine, Hangzhou 310009, P.R. China. E-mail: weiliansun@zju.edu.cn; chenlili_1030@zju.edu.cn
bSchool of Materials Science and Engineering, Zhejiang University, Hangzhou, Zhejiang 310027, China
cDepartment of Biomedical Engineering, The Chinese University of Hong Kong, Hong Kong SAR, China. E-mail: maophage@gmail.com
dFacility for Histomorphology, Zhejiang University School of Medicine, Hangzhou, Zhejiang 310027, China
First published on 21st April 2026
Abstract
Correction for ‘Injectable hydrogels activated with copper sulfide nanoparticles for enhancing spatiotemporal sterilization and osteogenesis in periodontal therapy’ by Yuting Yang et al., Biomater. Sci., 2025, 13, 1434–1448, https://doi.org/10.1039/D3BM02134C.
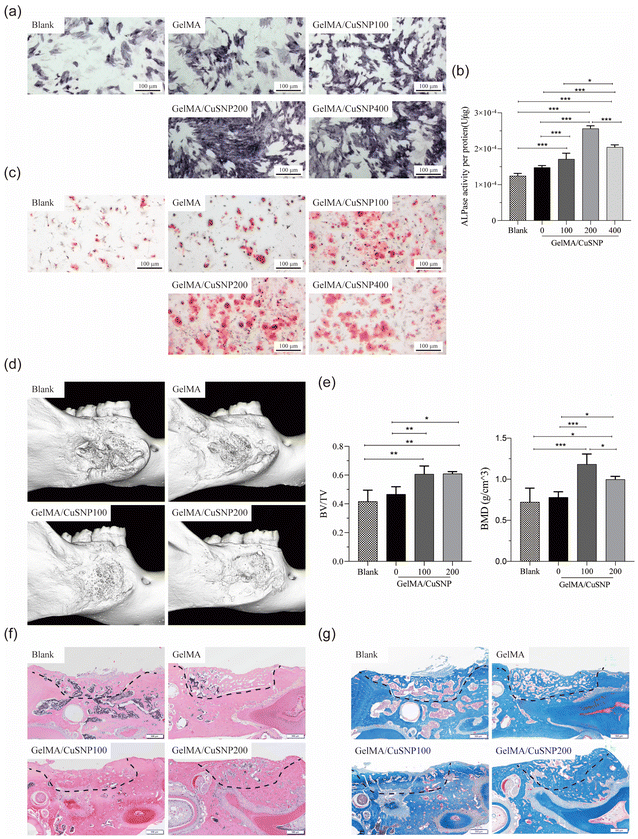
The authors regret the errors in Fig. 4, 6 and S7 in the original manuscript. The correct versions of Fig. 4, 6 and S7 are as shown below.
The Royal Society of Chemistry apologises for these errors and any consequent inconvenience to authors and readers.
| This journal is © The Royal Society of Chemistry 2026 |