Open Access Article
Open Access ArticleMOF-based arginine nanocarriers for coordinated immunometabolic and antitumor modulation in triple negative breast cancer
Amir M.
Alsharabasy
 *a,
Aibhe
Boran
*a,
Aibhe
Boran
 b,
Roberto
González-Gómez
b,
Harry
Revill
a,
Abhay
Pandit
a and
Pau
Farràs
*ab
b,
Roberto
González-Gómez
b,
Harry
Revill
a,
Abhay
Pandit
a and
Pau
Farràs
*ab
aCÚRAM, Research Ireland Centre for Medical Devices, University of Galway, H91 W2TY, Ireland. E-mail: amir.abdo@universityofgalway.ie; pau.farras@universityofgalway.ie
bSchool of Biological and Chemical Sciences, Ryan Institute, University of Galway, H91 TK33, Ireland
First published on 21st April 2026
Abstract
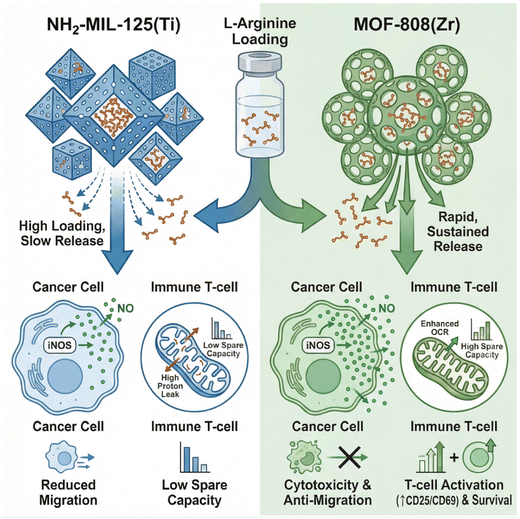
L-Arginine (L-Arg) is a key immunometabolite and nitric oxide (˙NO) precursor with therapeutic potential in cancer and immunotherapy. However, its clinical application is hindered by poor bioavailability and uncontrolled dosing. Here, two distinct metal–organic frameworks (MOFs), NH2-MIL-125(Ti) and MOF-808(Zr), were engineered as nanocarriers for L-Arg to enable coordinated tumour–immune modulation in triple-negative breast cancer (TNBC). L-Arg loading and release were systematically characterized, followed by Seahorse metabolic flux, flow cytometry, live-cell imaging, and wound healing assays to evaluate biological effects in activated human T cells and inducible nitric oxide synthase (iNOS)-transduced MDA-MB-231 cells. Both MOFs demonstrated successful L-Arg encapsulation with distinct release kinetics. In activated CD4+ T cells, Arg-loaded MOFs induced profound metabolic reprogramming independent of detectable ˙NO production. MOF-808-Arg enhanced oxidative phosphorylation and preserved spare respiratory capacity, while NH2-MIL-125-Arg triggered hypermetabolism characterized by elevated proton leak and loss of respiratory reserve, mimicking high-dose L-Arg stress. In contrast, in iNOS-expressing MDA-MB-231 cells, both MOFs increased intracellular ˙NO levels, resulting in reduced viability and inhibited migration. These findings demonstrate that controlled arginine delivery exerts dual and context-dependent effects, coupling ˙NO-mediated tumour cytotoxicity with ˙NO-independent enhancement of T-cell metabolic fitness. Overall, this work establishes MOF-based nutrient delivery as a strategy that integrates redox-based gasotransmitter therapy with immunometabolic reprogramming, highlighting the importance of carrier-dependent release kinetics in shaping both tumour and immune cell responses in metabolically hostile cancers such as TNBC.
1. Introduction
Nitric oxide (˙NO) is a key endogenous gasotransmitter produced by nitric oxide synthases (NOS), with established roles in vasodilation, neurotransmission, and immune defence.1,2 In cancer, however, dysregulated ˙NO signalling can promote tumour progression, angiogenesis, and metastatic dissemination.3–5 In triple-negative breast cancer (TNBC), inducible NOS (iNOS) is often overexpressed, and elevated ˙NO has been linked to activation of pro-tumour pathways, including epidermal growth factor receptor/extracellular signal-regulated kinase (EGFR/ERK) and nuclear factor kappa B (NF-κB), thereby promoting invasion and metastasis.6–8 Sustained or spatially uncontrolled ˙NO can also disrupt protein function and promote epithelial–mesenchymal transition,9 At the same time, ˙NO may exert anti-tumour effects under specific conditions, including suppression of cell motility and normalization of tumour vasculature.10,11 This concentration- and context-dependent duality highlights the need for controlled, localized strategies for modulating ˙NO levels within the tumour microenvironment (TME).12 To achieve this, diverse ˙NO-delivery strategies have been explored (e.g. diazeniumdiolates, nitrosothiols, metal–nitrosyls, photoactive hydrogels) that release ˙NO in the tumour microenvironment. However, many synthetic ˙NO donors (e.g. diazeniumdiolates or nitrosothiols) suffer from burst release kinetics, systemic off-target effects, and limited responsiveness to tumour-specific cues.13,14As an alternative, L-arginine (L-Arg), the endogenous substrate of NOS, enables enzyme-mediated and context-dependent ˙NO generation. Supplementation with L-Arg can markedly elevate intracellular ˙NO levels, a phenomenon termed the “L-arginine paradox”, even when extracellular arginine appears sufficient.15–17 In arginine-deprived tumours, particularly those enriched in myeloid arginase activity, restoring L-Arg availability can enhance ˙NO production and sensitize tumour cells to oxidative stress.18,19 By restoring arginine levels, one can not only fuel ˙NO generation but also reinvigorate tumour-infiltrating T cells, improving their survival and anti-tumour activity.20 Hence, L-Arg delivery allows more controlled, enzyme-mediated ˙NO production in situ.21
Beyond conventional drug delivery, growing evidence shows that biomaterials can actively reprogram T-cell metabolism. Amino acid availability, particularly arginine, strongly influences T-cell mitochondrial function, survival, and effector differentiation. Elevating intracellular L-Arg enhances oxidative phosphorylation and spare respiratory capacity in activated T cells, thereby improving persistence and antitumour function. Importantly, these metabolic effects can occur without measurable increases in ˙NO production, indicating that arginine-mediated T-cell reprogramming may proceed largely independently of canonical NO signalling.20 Thus, arginine delivery may exert dual effects within tumours: enhancing ˙NO-driven cytotoxicity in iNOS-expressing cancer cells while simultaneously promoting metabolic fitness in T cells.
Metal–organic frameworks (MOFs) are attractive candidates for such applications because of their high porosity, tuneable surface chemistry, and controllable cargo release.22,23 Different MOF-based systems have been engineered for chemotherapeutic delivery, induction of immunogenic cell death, and tumour microenvironment modulation. For example, transferrin-functionalized Zn-MOFs improved tumour targeting in TNBC models,24 while pH-sensitive zeolitic imidazolate framework-8 (ZIF-8) composites co-delivered chemotherapeutic and epigenetic agents to convert immune-“cold” tumours into inflamed phenotypes.25 MOFs have also been explored as immunomodulatory platforms for delivering antigens, adjuvants, and metabolic regulators to shape antitumour immunity. However, direct enhancement of T-cell metabolic fitness through MOF-mediated nutrient delivery remains largely unexplored.
Among these platforms, zirconium- and titanium-based MOFs, including MOF-808 and NH2-MIL-125(Ti), are particularly attractive because of their structural stability, biocompatibility, and tunable defect chemistry. MOF-808, constructed from Zr6-oxo clusters, contains large hexagonal pores (∼1.8 nm) and shows high defect tolerance, enabling substantial cargo loading.26,27 In parallel, NH2-MIL-125(Ti), built from Ti8O clusters, exhibits a cage-like porous architecture with high surface area (>1500 m2 g−1) and pendant amine groups that facilitate hydrogen bonding, electrostatic interactions, and pH-responsive release.28,29 Despite these favourable properties, a systematic comparison of these frameworks as arginine delivery platforms for linked redox-based tumour targeting and T-cell metabolic modulation has not been reported.
Here, NH2-MIL-125(Ti) and MOF-808(Zr) were developed and compared as tuneable nanocarriers for localized L-Arg delivery in TNBC. We hypothesize that controlled arginine release can simultaneously (i) amplify intracellular ˙NO generation in iNOS-expressing TNBC cells to induce cytotoxic and anti-migratory effects, and (ii) enhance mitochondrial respiration and metabolic fitness in activated human T cells. By integrating materials characterization, release kinetics, redox biology, and Seahorse metabolic profiling, we establish a dual-function framework that couples tumour-directed gas-based therapy with immune metabolic support. This work positions MOF-mediated arginine delivery not merely as a ˙NO amplification strategy, but as a platform for coordinated tumour–immune metabolic modulation within the TNBC microenvironment. An overview of the study design is presented in Scheme 1.
2. Materials and methods
2.1. Materials and reagents
Titanium isopropoxide (TTIP), 2-aminoterephthalic acid (NH2-BDC), zirconyl chloride octahydrate (ZrOCl2·8H2O), 1,3,5-benzenetricarboxylic acid (trimesic acid, BTC), formic acid, L-Arg, ninhydrin (25 mg mL−1), hydrindantin, equine heart myoglobin, anhydrous dimethyl sulphide (DMSO), calibration standards for Ti (#43843) and Zr (#51244), BODIPY™ 581/591 C11 dye, phosphate-buffered saline (PBS), RPMI-1640 medium, L-glutamine, penicillin/streptomycin, poly-L-lysine (PLL), fetal bovine serum (FBS) were all purchased from Sigma-Aldrich. N,N-Dimethylformamide (DMF), methanol, hydrochloric acid, dichloromethane (DCM), sodium hydroxide, Cell-Quant alamarBlue™ Cell Viability Reagent, Invitrogen Griess reagent kit, Invitrogen 4-Amino-5-ethylamino-2′,7′-difluorofluorescein diacetate (DAF-FM), human interleukin-2 (IL-2), anti-CD3/CD28 Dynabeads, human interferon gamma (IFN-É£) Uncoated ELISA Kit (#88-7316-88), and Nunc™ 96-well optical-bottom black microplates were obtained from Fisher Scientific. Lymphoprep™ (#18061) and SepMate™ (#85450) were from STEMCELL, Germany, CD4+ T Cell Isolation Kit, (human, #130-096-533) was from Miltenyi Biotec, Germany. Zombie Red™ Fixable Viability kit (#423109) was from BioLegend. Seahorse XF Cell Mito Stress Test Kits, XFp cell culture microplates, and sensor cartridges were purchased from Agilent Technologies.2.2. Materials synthesis and physicochemical characterisation
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 10 min) and subsequent activation under dynamic vacuum at 150 °C overnight. L-Arg loading was performed using a solution-based impregnation method.32 Typically, 30 mg of activated MOFs was dispersed in 30 mL of deionized water and sonicated for 30 min to form a homogeneous suspension. A 7.5 mL aliquot of a freshly prepared L-Arg aqueous solution (2 mg mL−1) was added dropwise to the dispersion of the nanoparticles. The pH of the mixture was adjusted to 5.5 and stirred overnight at room temperature to facilitate adsorption. The product was then collected by centrifugation (10
000 rpm, 10 min) and subsequent activation under dynamic vacuum at 150 °C overnight. L-Arg loading was performed using a solution-based impregnation method.32 Typically, 30 mg of activated MOFs was dispersed in 30 mL of deionized water and sonicated for 30 min to form a homogeneous suspension. A 7.5 mL aliquot of a freshly prepared L-Arg aqueous solution (2 mg mL−1) was added dropwise to the dispersion of the nanoparticles. The pH of the mixture was adjusted to 5.5 and stirred overnight at room temperature to facilitate adsorption. The product was then collected by centrifugation (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm, 10 min) and thoroughly washed with deionized water to remove unbound amino acids. The final L-Arg-loaded MOFs were obtained as dry powders after freeze-drying for 24 h. For comparison, L-Arg was loaded into nonactivated, as-synthesized MOFs following the same procedure.
000 rpm, 10 min) and thoroughly washed with deionized water to remove unbound amino acids. The final L-Arg-loaded MOFs were obtained as dry powders after freeze-drying for 24 h. For comparison, L-Arg was loaded into nonactivated, as-synthesized MOFs following the same procedure.
Hydrodynamic particle size and zeta potential were measured by dynamic light scattering (DLS) using a Litesizer™ 500 (Anton Paar) at 25 °C. Samples were dispersed in PBS or PBS supplemented with 10% FBS at a concentration of 100 µg mL−1, probe-sonicated for 20 s, and analysed using appropriate cuvettes (refractive index 1.37 for size measurements and 1.33 for zeta potential). Results are reported as the mean of five independent replicates, with 60 runs per measurement.
For degradation studies, MOF nanoparticles were suspended in PBS at 100 µg mL−1, briefly sonicated, and incubated under shaking conditions at 37 °C for up to 36 h. At predefined time points, aliquots were collected, centrifuged at 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000g for 20 min, and the supernatants were diluted in 2% nitric acid. The concentrations of Zr and Ti released from the respective MOFs were quantified using an Agilent 5110 inductively coupled plasma–optical emission spectrometer (ICP–OES), with data processed using ICP Expert software (version 7.4.1.10449).
000g for 20 min, and the supernatants were diluted in 2% nitric acid. The concentrations of Zr and Ti released from the respective MOFs were quantified using an Agilent 5110 inductively coupled plasma–optical emission spectrometer (ICP–OES), with data processed using ICP Expert software (version 7.4.1.10449).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) FBS/PBS mixture, briefly sonicated (20 s), and maintained at 37 °C under gentle agitation. At predetermined time points (1, 2, 4, 8, 12, 24, and 48 h), 70 µL aliquots were withdrawn and immediately centrifuged (8000g, 10 min) to remove particulates. Arginine concentrations in the supernatants were determined using the optimized ninhydrin protocol described before.33 The reagent was freshly prepared and contained ninhydrin (25 mg mL−1) and hydrindantin (1 mg mL−1) in a 1
1 (v/v) FBS/PBS mixture, briefly sonicated (20 s), and maintained at 37 °C under gentle agitation. At predetermined time points (1, 2, 4, 8, 12, 24, and 48 h), 70 µL aliquots were withdrawn and immediately centrifuged (8000g, 10 min) to remove particulates. Arginine concentrations in the supernatants were determined using the optimized ninhydrin protocol described before.33 The reagent was freshly prepared and contained ninhydrin (25 mg mL−1) and hydrindantin (1 mg mL−1) in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (v/v) mixture of DMSO and aqueous acetate buffer (2 M acetic acid, 1 M potassium acetate). For each measurement, 50 µL of sample was mixed with 200 µL of reagent, incubated at 90 °C for 45 min, cooled for 15 min, diluted with 1250 µL of 2-propanol/water (1
1 (v/v) mixture of DMSO and aqueous acetate buffer (2 M acetic acid, 1 M potassium acetate). For each measurement, 50 µL of sample was mixed with 200 µL of reagent, incubated at 90 °C for 45 min, cooled for 15 min, diluted with 1250 µL of 2-propanol/water (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v), and the absorbance was measured at 570 nm using a microplate reader (Varioskan Flash, Thermo Fisher Scientific, Finland). Aliquots collected from PBS release studies were diluted and analysed using the ninhydrin assay under identical aqueous reaction conditions as the calibration standards. Background absorbance from PBS was corrected using corresponding blank controls. A small but consistent baseline difference between PBS and water was observed and accounted for during data processing, ensuring accurate quantification of L-Arg.
1, v/v), and the absorbance was measured at 570 nm using a microplate reader (Varioskan Flash, Thermo Fisher Scientific, Finland). Aliquots collected from PBS release studies were diluted and analysed using the ninhydrin assay under identical aqueous reaction conditions as the calibration standards. Background absorbance from PBS was corrected using corresponding blank controls. A small but consistent baseline difference between PBS and water was observed and accounted for during data processing, ensuring accurate quantification of L-Arg.
2.3. T-Cell isolation and functional assays
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2. Cells were maintained at 37 °C in a humidified atmosphere containing 5% CO2 for 72 h prior to downstream treatments.
2. Cells were maintained at 37 °C in a humidified atmosphere containing 5% CO2 for 72 h prior to downstream treatments.
2.4. In vitro cancer cell studies (iNOS-MDA-MB-231)
2.5. Statistical analysis
All experiments were performed with at least three independent replicates unless otherwise specified, and the results are expressed as the mean ± standard deviation (SD) or standard error of means (SEM). For T-cell experiments, biological replicates (n) refer to independent human donors, as indicated in the figure legends. All measurements were performed using cells derived from these donors. For cancer cell assays, n represents independent experiments performed on separate days. Data were analysed using Microsoft Excel and OriginPro software for validation and graphical representation of the results. Statistical significance between groups was determined using a two-tailed Student's t-test for pairwise comparisons as specified in the figure legends. A p value of <0.05 was considered statistically significant.3. Results and discussion
3.1. Characterization of synthesized MOF nanoparticles
The synthesized NH2-MIL-125 and MOF-808 nanoparticles were first characterized to confirm framework formation, morphology, and colloidal behaviour prior to biological evaluation. SEM, EDX, and PXRD analyses verified successful synthesis and preservation of the expected structural features, while DLS and ζ-potential measurements revealed framework-dependent differences in dispersion and serum stability. Detailed physicochemical characterization data are provided in the SI (Fig. S1 and S2).The colloidal properties of the MOFs were evaluated by dynamic light scattering and ζ-potential measurements under different conditions. Both frameworks exhibited changes in hydrodynamic diameter and surface charge upon activation and arginine loading, reflecting alterations in surface chemistry and interparticle interactions. Notably, MOF-808 showed improved dispersion in serum-containing media, whereas NH2-MIL-125 displayed a greater tendency toward aggregation. These observations indicate that framework composition and surface chemistry influence colloidal stability in biologically relevant environments. Full datasets and statistical analyses are presented in the SI (Table S1). Collectively, these results confirm the successful synthesis and functionalisation of both MOF platforms, enabling subsequent evaluation of their arginine delivery performance.
3.2 Arginine loading and surface modification
L-Arg was employed as a ˙NO precursor, exploiting its role as a substrate for iNOS37,38 to enhance intracellular ˙NO production in TNBC cells. The controlled delivery of L-Arg via MOF carriers was therefore designed to enable localised and sustained ˙NO generation, amplifying cytotoxic and anti-migratory effects while minimising systemic exposure. The successful incorporation into both MOF frameworks was confirmed by complementary spectroscopic and elemental analyses, without evidence of framework collapse or loss of crystallinity. Changes in surface charge further supported arginine adsorption, indicating interactions between the cationic amino acid and negatively charged framework sites. Detailed FTIR, PXRD, and SEM–EDX data are provided in the SI (Fig. S3, S5 and S6).Quantitative ninhydrin analysis confirmed these trends, showing that NH2-MIL-125 exhibited a significantly higher apparent arginine loading capacity (25.32 ± 2.43%) and loading efficiency (50.65 ± 4.87%) than MOF-808 (11.84 ± 1.13% and 23.7 ± 2.26%, respectively). The calibration curve used for arginine quantification is shown in Fig. S7. These results indicate that the Ti-based framework provides a greater number of accessible binding sites for L-Arg under the loading conditions used, whereas the lower loading capacity of MOF-808 is consistent with its comparatively limited structural perturbation following arginine incorporation. Together, these findings show that framework chemistry and activation state critically govern amino-acid loading behaviour, with NH2-MIL-125 favouring higher arginine uptake and MOF-808 showing improved colloidal stability in biologically relevant media.
To assess framework stability following arginine incorporation, metal ion release was quantified under physiological conditions. The ICP-OES analysis revealed marked differences in titanium release kinetics between bare and Arg-loaded NH2-MIL-125. Sampling-corrected cumulative release calculations showed minimal Ti leaching at early timepoints (≤10 h) for both materials; however, bare NH2-MIL-125 exhibited a pronounced Ti burst at 36 h, corresponding to a cumulative release of 29 µg Ti (equivalent to 96 µg mg−1 MOF) (Fig. 1A). In contrast, Arg-loaded NH2-MIL-125 displayed substantially suppressed Ti release over the entire incubation period, reaching only 4.1 µg Ti at 36 h (13.7 µg mg−1 MOF). These results indicate that L-Arg loading significantly stabilizes the Ti-oxo framework against hydrolytic degradation, an effect that is not detectable by PXRD alone but becomes evident through solution-phase metal release analysis. However, the cumulative zirconium release profiles revealed markedly different degradation behaviours between unloaded and Arg-loaded MOF-808 nanoparticles (Fig. 1B). Arg-loaded MOF-808 exhibited a pronounced early Zr release, reaching a maximum at the intermediate timepoints, followed by a partial decline and subsequent stabilization at later times. In contrast, unloaded MOF-808 showed consistently low and nearly time-independent Zr release throughout the incubation period, indicating high intrinsic stability of the framework under these conditions. The transient Zr release observed for the Arg-loaded material suggests that Arg incorporation enhances the initial lability of surface-accessible Zr6-oxo clusters, potentially through competitive coordination or ligand exchange, without inducing sustained framework collapse.
The release behaviour of activated NH2-MIL-125(Ti)-Arg and MOF-808(Zr)-Arg was next evaluated to determine how framework composition influences arginine retention and diffusion. The two carriers displayed markedly distinct release profiles (Fig. 1C). MOF-808-Arg showed a pronounced initial burst, releasing approximately 19–23 µg mL−1 of arginine within the first hour and approaching a plateau of 25–26 µg mL−1 by 24 h. In contrast, NH2-MIL-125-Arg exhibited a slower and more progressive release profile, increasing from 4–6 µg mL−1 at 1 h to 17–18 µg mL−1 after 24 h, consistent with sustained release. These contrasting release kinetics reflect the distinct structural and chemical features of the two frameworks.
In brief, MOF-808, built from Zr6O4(OH)4 clusters bridged by 1,3,5-benzenetricarboxylate linkers, contains large adamantane-like cages (∼1.8 nm) and a high internal surface area (>2000 m2 g−1), which favour rapid diffusion of small, hydrophilic, positively charged L-Arg molecules.39 Together with the strong hydrolytic stability conferred by Zr–carboxylate coordination bonds,40,41 this suggests that arginine release from MOF-808 is governed mainly by pore diffusion, with only limited and transient perturbation of surface-accessible Zr6 clusters rather than bulk framework degradation. In contrast, NH2-MIL-125(Ti) possesses narrower pore apertures (∼0.6 nm) and Ti–oxo clusters that can engage in hydrogen-bonding and electrostatic interactions with the guanidinium group of L-Arg.42 These stronger host–guest interactions restrict molecular mobility, resulting in a diffusion-limited release profile without a pronounced initial burst. In addition, the absence of competing anions such as phosphate in the release medium minimises disruption of Ti–carboxylate coordination, further preserving framework integrity and slowing arginine liberation.43 Accordingly, arginine release from NH2-MIL-125 proceeds in a more sustained manner through its compact pore network, consistent with the near-linear cumulative release observed over the first 24 h. Collectively, these results indicate that framework-dependent differences in loading capacity and structural lability directly govern arginine release kinetics and provide a mechanistic basis for the distinct biological responses observed for the two MOF systems.
3.3. Immunomodulatory effects of Arg-releasing MOFs on T Cells
Arg-loaded MOFs induced marked increases in T-cell mitochondrial respiration (Fig. 2A). Basal OCR was significantly elevated in cells treated with Arg-loaded MIL-125 or MOF-808 compared with both untreated cells and cells exposed to unloaded MOFs (Fig. 2B). MIL-125-Arg increased basal respiration by ∼4.5-fold relative to untreated T cells and by ∼35-fold relative to empty MIL-125, while MOF-808-Arg increased basal OCR by ∼3.3-fold versus untreated cells. Notably, MIL-125-Arg produced a higher basal OCR than MOF-808-Arg, consistent with the greater L-Arg loading capacity of the Ti-based carrier. In contrast, unloaded MOFs severely suppressed respiration, with basal OCR reduced to 13 ± 2 pmol min−1 for MIL-125 and 4 ± 1 pmol min−1 for MOF-808, compared with 102 ± 5 pmol min−1 in untreated controls. This pronounced impairment is consistent with the known dependence of T-cell bioenergetics on arginine availability.44,45
The increase in basal respiration in the Arg-loaded groups translated into substantially higher ATP-linked respiration (Fig. 2C). Following oligomycin injection, both MIL-125-Arg and MOF-808-Arg exhibited ∼3.4-fold greater ATP-linked OCR than untreated cells, whereas ATP-linked respiration in the unloaded MOF groups was nearly abolished. Although both Arg-loaded formulations strongly enhanced maximal respiration (Fig. S9B), their respiratory reserve differed markedly: MOF-808-Arg retained substantial spare respiratory capacity, whereas MIL-125-Arg showed essentially none (Fig. 2D). This indicates that MIL-125-Arg-treated cells were operating close to their maximal respiratory rate under basal conditions.
These differences were reinforced by the proton leak data (Fig. S9C). Oligomycin-resistant OCR rose to 212 ± 25 pmol min−1 in MIL-125-Arg-treated cells, compared with 93.6 ± 10 pmol min−1 for MOF-808-Arg and 29.6 ± 2 pmol min−1 in untreated controls. Thus, although both carriers enhanced mitochondrial activity, MIL-125-Arg induced more than double the proton leak observed with MOF-808-Arg, indicating lower coupling efficiency and a greater fraction of respiration uncoupled from ATP synthesis. This pattern suggests that the sustained arginine exposure associated with MIL-125-Arg drives a hypermetabolic state accompanied by mitochondrial stress, whereas the more rapid release profile of MOF-808-Arg supports enhanced respiration with comparatively lower metabolic inefficiency.
Non-mitochondrial oxygen consumption was also elevated, particularly in the MIL-125-Arg group (Fig. S9D), suggesting activation of extra-mitochondrial oxygen-consuming processes. Parallel nitrite quantification by Griess assay showed no detectable accumulation of NO-derived metabolites across conditions, indicating that ˙NO production is unlikely to be a dominant driver of the observed metabolic changes. This is consistent with previous reports showing that exogenous L-Arg can enhance T-cell metabolic fitness and survival without measurable ˙NO production, supporting an immunometabolic rather than ˙NO-dependent mechanism.20 The elevated non-mitochondrial OCR therefore likely reflects arginine-driven metabolic rewiring, potentially involving arginine-sensing pathways, enhanced biosynthetic activity, and increased demand from other oxygen-consuming processes.
More broadly, the two Arg-loaded MOFs produced metabolic phenotypes resembling different soluble arginine exposure states, highlighting the importance of delivery kinetics in shaping T-cell responses. MOF-808-Arg promoted a more balanced bioenergetic profile, with enhanced respiration but preserved reserve capacity, whereas MIL-125-Arg induced stronger basal activation at the expense of coupling efficiency and respiratory flexibility. These differences are consistent with the faster release profile of MOF-808 and the higher loading capacity and more sustained arginine exposure associated with NH2-MIL-125, indicating that the immunometabolic effects of MOF-delivered arginine depend not only on total cargo release but also on the extent and duration of cellular exposure.20,46
Following 72 h of treatment, T-cell viability was assessed across all conditions. Untreated cells maintained high viability (∼96%), and soluble L-Arg supplementation (0.5–2 mM) had no adverse effect (94–96%; Fig. S10A). In contrast, direct exposure to empty MOF-808 significantly reduced viability to ∼82% (p < 0.05 vs. control), whereas separation by transwell largely mitigated this effect (∼91%), indicating that direct particle–cell interaction contributes substantially to toxicity (Fig. 3A). Empty NH2-MIL-125 showed only a mild, non-significant effect on viability. Importantly, arginine loading improved the tolerability of both carriers, with Arg-loaded MOF-808 restoring viability toward control levels and Arg-loaded NH2-MIL-125 showing no evident cytotoxicity. This protective effect was less pronounced in the transwell setup (∼84% viability), indicating that while arginine release is beneficial, diffusible nanoparticle components may still exert minor negative effects. Arg-loaded MIL-125 did not impair viability. Immunophenotyping revealed a consistently predominant CD4+ population (∼98–99% of live cells) across all conditions, with no treatment altering the CD4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CD8 ratio (Fig. S11A). This confirms that subsequent functional differences are attributable to changes in activation state, not subset composition. The improved cytocompatibility of MOF-808 formulations may also be attributed to its greater colloidal stability in serum-containing media, which reduces aggregation and limits direct particle–cell interactions. In contrast, the comparatively lower stability of NH2-MIL-125 may promote particle aggregation, contributing to the observed differences in cellular responses.
CD8 ratio (Fig. S11A). This confirms that subsequent functional differences are attributable to changes in activation state, not subset composition. The improved cytocompatibility of MOF-808 formulations may also be attributed to its greater colloidal stability in serum-containing media, which reduces aggregation and limits direct particle–cell interactions. In contrast, the comparatively lower stability of NH2-MIL-125 may promote particle aggregation, contributing to the observed differences in cellular responses.
T-cell activation was further evaluated using the markers CD25 and CD69. Untreated cells were already highly activated, and soluble L-Arg modestly increased CD25 expression. Empty MOF-808 reduced CD25 levels and showed donor-dependent suppression of activation, whereas arginine loading reversed this effect and increased CD25 expression to levels comparable to or greater than control (Fig. 3B). A similar pattern was observed for CD69, with Arg-loaded MOF-808 producing the clearest increase over untreated cells (Fig. 3C). Together, these data indicate that arginine delivery not only improves T-cell viability but also promotes an activated phenotype, particularly in the MOF-808 formulation where loading rescues the adverse effects of the empty carrier.
The exhaustion-associated markers TIM-3 and PD-1 revealed a more nuanced phenotypic response. Soluble L-Arg reduced TIM-3 expression relative to untreated cells, consistent with delayed terminal exhaustion (Fig. 3D). Empty MOF-808 increased TIM-3 in one donor and contributed to a higher group mean, whereas Arg-loaded formulations generally showed lower TIM-3 than their unloaded counterparts. PD-1 displayed a different pattern, with Arg-loaded MOFs often associated with increased expression (Fig. S11B), likely reflecting activation-associated checkpoint induction rather than overt dysfunction. Taken together, these results suggest that arginine delivery promotes activation while limiting later exhaustion-associated features, rather than simply driving uncontrolled T-cell stimulation.
These phenotypic findings are consistent with the Seahorse analysis, which showed that Arg-loaded MOFs restore oxidative metabolism in activated T cells, whereas empty carriers suppress mitochondrial function. Thus, the improved viability and activation phenotype observed with arginine-loaded formulations is accompanied by enhanced bioenergetic capacity, linking carrier-mediated arginine delivery to both metabolic and functional reprogramming of T cells.
Overall, empty MOF-808 exerted the most pronounced negative effects on T-cell viability and activation, likely due to direct particle–cell interactions and impaired mitochondrial function. Arginine loading largely mitigated these effects and converted the formulation into a metabolically supportive and functionally activating carrier. NH2-MIL-125 displayed better intrinsic biocompatibility and higher arginine loading capacity, but its Arg-loaded form induced a more metabolically intense response. Together, these findings show that MOF-mediated arginine delivery can reshape T-cell state in a formulation-dependent manner, with MOF-808 favouring a more balanced metabolic phenotype and NH2-MIL-125 supporting stronger but potentially less restrained activation.
To further assess whether arginine delivery influenced T-cell phenotype beyond the activation markers analysed above, C–C chemokine receptor type 7 (CCR7) expression was examined in a follow-up experiment using an independent donor set and focusing on untreated control, soluble L-arginine, Arg-loaded NH2-MIL-125, and Arg-loaded MOF-808 seeded to transwells. Across all groups, CCR7 expression remained consistently high, indicating that neither soluble arginine nor arginine-loaded MOFs caused a substantial loss of this marker (Fig. 4A). The highest and most uniform CCR7 levels were observed in the soluble arginine group, while both Arg-loaded MOF formulations also maintained CCR7 within a similarly high range, but with no significant differences between conditions. As CCR7 is a hallmark of central-memory and lymphoid-homing T cells, its preservation is generally associated with maintenance of a less differentiated phenotype rather than terminal effector polarization, as established in foundational studies of T-cell memory and more recent work showing that elevated intracellular arginine promotes a central-memory-like state with retention of CCR7 expression.20,47 Thus, these findings suggest that arginine delivery, either in free form or via MOFs, does not adversely affect this memory-associated marker.
To complement this phenotypic analysis, IFN-γ secretion was quantified by ELISA under corresponding transwell conditions. In contrast to the relatively stable CCR7 profile, IFN-γ release showed clearer formulation-dependent differences. Arg-loaded NH2-MIL-125 produced the most consistent increase in IFN-γ secretion across donors, while Arg-loaded MOF-808 also enhanced IFN-γ in two donors but showed greater donor-to-donor variability (Fig. 4B). However, no significant differences were detected between these conditions. Soluble 500 µM arginine increased IFN-γ relative to untreated controls in two donors, although this effect was less consistent overall. These findings are in line with the broader literature showing that arginine availability supports T-cell bioenergetics, survival, and activation, and can enhance effector responses including cytokine production, while the magnitude of this effect depends on the metabolic state of the cells and the mode of arginine delivery.20,48 Taken together, these data indicate that arginine delivery can preserve CCR7 expression while modulating effector cytokine output in a carrier-dependent manner, with NH2-MIL-125-based arginine delivery showing the most consistent association with preserved CCR7 expression and enhanced IFN-γ secretion across donors.
The metabolic effects observed following MOF-mediated arginine delivery are likely to reflect integration of nutrient-sensing and downstream functional pathways in activated T cells. Although mTOR signalling and other arginine-sensing mechanisms were not directly investigated in the present study, they represent plausible upstream regulators of the enhanced oxidative metabolism observed here. Importantly, the Seahorse findings were accompanied by changes in T-cell viability, activation-associated phenotypic markers, nitrite production, and IFN-γ secretion, indicating that arginine delivery influences not only mitochondrial respiration but also broader aspects of T-cell functional state. The nitrite data further suggest coupling between arginine availability and downstream nitrogen metabolism, although the present study does not establish whether the metabolic phenotype is directly dependent on ˙NO production. Future work should therefore examine mTOR-associated signalling, arginine transport/sensing pathways, and the causal contribution of ˙NO to the immunometabolic effects of these MOF formulations.
3.4. Modulation of iNOS-expressing MDA-MB-231 cells by Arg-releasing MOFs
To mimic the pathophysiological microenvironment of breast cancer tissue, which is characterised by elevated iNOS expression and excessive ˙NO generation,8,49 iNOS-transduced MDA-MB-231 cells were employed as previously described.34 First, the metabolic activity of cells was assessed following exposure to free L-Arg in serum-free and serum-containing media to decouple the intrinsic biological effects of L-Arg from MOF-mediated delivery. Free L-Arg induced only modest and serum-dependent changes in metabolic activity, with attenuated effects observed in the presence of FBS, likely due to reduced cellular uptake and altered extracellular availability (Fig. S12). These findings highlight the limitations of soluble L-Arg administration and underscore the need for carrier-mediated delivery to enhance intracellular substrate availability for ˙NO production.
Next, to enable fluorescence-based tracking of nanoparticle internalisation, NH2-MIL-125(Ti) and MOF-808(Zr) were loaded with the lipophilic fluorescent probe BODIPY™ 581/591 C11. Fluorescence emission was recorded between 591 and 599 nm following excitation at 581 nm to identify the optimal detection wavelength. Based on maximal fluorescence intensity and linearity of the standard curve, an emission wavelength of 595 nm was selected for all subsequent quantitative measurements. Fig. S13 shows the corresponding standard curve. Both frameworks exhibited comparable apparent loading capacities (0.69 ± 0.03% for BODIPY-NH2-MIL-125 and 0.76 ± 0.01% for BODIPY–MOF-808) and high loading efficiencies (63.17 ± 2.62% and 69.34 ± 0.61%, respectively), confirming effective dye incorporation into both MOF carriers. Minimal dye release was observed upon suspension in PBS for 1 h, with no detectable BODIPY release from BODIPY–MIL-125 and only a trace amount released from BODIPY–MOF-808 (0.25 ± 0.02 µg), indicating stable probe retention under physiological conditions. These results confirm that the fluorescent signal used for subsequent uptake studies predominantly reflects nanoparticle-associated BODIPY rather than free dye, thereby providing a reliable basis for evaluating cellular internalisation.
Next, the cellular uptake of both BODIPY-labelled NH2-MIL-125 and BODIPY-labelled MOF-808 particles was studied in FBS-free medium to maximize nanoparticle uptake and simplify fluorescence readouts. We also performed parallel tests in FBS-containing medium (data not shown), which as expected yielded lower uptake; this approach follows common practice since serum proteins bind nanoparticles and reduce apparent internalization.50 For example, others have pre-incubated cells in FBS-free medium before applying MOFs and then measured uptake by flow cytometry.51 In line with these reports, we found that adding FBS at 10% dramatically decreased the internalized signal. Prior studies similarly show that serum (FBS) induces MOF aggregation and protein corona formation; for instance, Hidalgo et al. reported that nanoMOFs in FBS-supplemented medium swelled in size and aggregated, altering their cellular uptake.52 Likewise, Gan et al. observed that once a protein corona was formed (≥10% FBS), further increases in serum (to 20%) did not significantly change uptake.53 Hence, focusing on FBS-free conditions aligns with the literature and allows us to isolate the intrinsic uptake behaviour of our BODIPY-labelled MOFs.
To account for potential spectral crosstalk from green fluorescent protein (GFP)-expressing MDA-MB-231 cells, we implemented rigorous fluorescence compensation and controls. Cells, treated with BODIPY™ 581/591 C11 alone at varying concentrations and in different media conditions, were used to establish compensation matrices by tracking red and green fluorescence over time (Fig. S14A, B and Videos S1–S3). Additional controls included untreated GFP-positive cells (to establish baseline autofluorescence; Video S4) and MOF samples in medium without cells (to account for extracellular fluorescence or surface-adsorbed signal). These data informed a 3% spillover correction from green to red, which was systematically applied to all subsequent analyses.
Fig. S15 shows the red fluorescence signal from BODIPY™ 581/591-loaded NH2-MIL-125 and MOF-808 nanoparticles incubated in FBS-free medium without cells over 24 hours. For MIL-125, red fluorescence intensity steadily declined across all concentrations, likely reflecting gradual dye release and quenching in the extracellular environment (Fig. S15A and Videos S5–S7). In contrast, BODIPY-MOF-808 samples showed relatively stable or slightly increasing red signal over time, especially at higher concentrations, suggesting stronger dye retention or slower release from MOF-808 (Fig. S15B and Videos S8–S10). These measurements establish baseline dye behaviour in cell-free conditions, critical for interpreting cellular uptake experiments.
Upon incubation with iNOS-transduced MDA-MB-231 cells, distinct patterns emerged. For cells treated with BODIPY-loaded NH2-MIL-125 nanoparticles, the red fluorescence (Fig. 5A and Videos S11–S13), which corresponds to the reduced, unoxidized form of BODIPY, decreased sharply within the first few hours, particularly at higher nanoparticle concentrations, suggesting rapid cellular uptake followed by intracellular oxidation of the probe. Concurrently, the green fluorescence (Fig. 5B), indicative of oxidized BODIPY, increased in a dose-dependent manner, peaking at 8 hours post-treatment and stabilizing thereafter. The temporal inverse trend of red and green signals confirms successful internalization of NH2-MIL-125 and oxidation of the encapsulated probe, consistent with intracellular reactive oxygen species (ROS) and reactive nitrogen species (RNS) activity. These data support the use of BODIPY-loaded NH2-MIL-125 as a reporter for oxidative processes in engineered tumour cells and demonstrate its responsiveness to redox conditions in live-cell assays.
A more pronounced response was observed with BODIPY-loaded MOF-808 nanoparticles. The red fluorescence (Fig. 5E and Videos S14–S16), showed an initial rise followed by a gradual decline, with peak intensities increasing in a concentration-dependent manner, indicating nanoparticle uptake and intracellular delivery of the dye. The green fluorescence (Fig. 5F), increased progressively over time in treated cells, most notably at the highest concentration, suggesting active intracellular oxidation likely mediated by ˙NO or ROS/RNS activity. While the green signal increase was moderate compared to that observed with MIL-125, it remained above control throughout, confirming redox-responsive oxidation. Together, these results confirm cellular uptake of MOF-808 and release of the encapsulated probe, albeit with slightly delayed or less intense oxidation kinetics than MIL-125, possibly due to differences in particle internalization rate or redox reactivity.
In parallel, the concentration-dependent effects of NH2-MIL-125(Ti) and MOF-808(Zr) on the metabolic activity of iNOS-expressing MDA-MB-231 cells were evaluated in both serum-free and serum-containing media. The two frameworks exhibited distinct cytocompatibility profiles, which were strongly influenced by medium composition (Fig. S16A and B). Furthermore, metal release/degradation measurements provide an initial indication of framework lability and potential exposure to Ti- or Zr-containing species (Fig. 1A and B). Under serum-free conditions, NH2-MIL-125 caused a greater reduction in metabolic activity compared with MOF-808, consistent with previous reports indicating that zirconium–carboxylate frameworks such as MOF-808 generally display higher biocompatibility,54 whereas titanium-based MOFs may exert moderate cytotoxic effects at elevated concentrations.55 Importantly, no interference of the MOF nanoparticles with the AlamarBlue™ assay was detected under the experimental conditions used.
The inclusion of 10% FBS markedly attenuated the cytotoxic effects of both MOFs. This effect is attributed to protein corona formation, whereby adsorbed serum proteins alter nanoparticle surface properties, reduce cellular uptake, and limit direct membrane interactions.56,57 This serum-dependent modulation highlights the critical role of nanoparticle–biomolecule interactions in governing cellular responses. Consequently, subsequent functional assays were conducted under serum-free conditions to minimise protein corona formation and enable direct evaluation of the biological effects arising from L-Arg release from the MOF carriers. This experimental design is consistent with standard protocols used for intracellular ˙NO measurements and migration assays.
Both NH2-MIL-125(Ti) and MOF-808(Zr) nanoparticles exhibited a significant reduction in the metabolic activity of MDA-MB-231(iNOS) cells upon L-Arg loading compared with their unloaded counterparts (Fig. 5C and G). In iNOS-overexpressing cells, MOF-mediated delivery of L-Arg supplies excess substrate for ˙NO synthesis, thereby amplifying intracellular ˙NO production. Elevated ˙NO levels are known to promote apoptotic signalling and suppress proliferation in cancer cells,58,59 consistent with the pronounced decrease in metabolic activity observed following treatment with Arg-loaded MOFs. Sonication further enhanced the cytotoxic efficacy of both MOF systems. Sonicated nanoparticles (Son) induced a significantly stronger reduction in metabolic activity than their non-sonicated counterparts (ϕ), as shown in Fig. 5D and H. This enhancement is attributed to improved dispersion and reduced aggregation following sonication, resulting in increased colloidal stability and more efficient cellular uptake.60 Consequently, intracellular delivery of encapsulated L-Arg was enhanced, leading to greater suppression of cellular metabolic activity.
Together, these data support the short-term in vitro tolerability of the platforms under the tested conditions, while also showing that safety is material- and context-dependent. However, the identity and biological effects of long-term degradation products, as well as the consequences of chronic metal ion exposure, remain to be established in dedicated long-term and in vivo studies.
Sonication further amplified the intracellular ˙NO response, particularly for Arg–MIL-125, which exhibited the earliest and most pronounced ˙NO burst. At 4 h, fluorescence in cells treated with sonicated Arg–MIL-125 was approximately 3.2-fold higher than that of sonicated unloaded MIL-125, while the difference between Arg-loaded and unloaded MOF-808 was around 1.4-fold. Under non-sonication conditions, Arg–MOF-808 outperformed Arg–MIL-125. Over prolonged incubation (12–24 h), MOF-808 sustained higher intracellular ˙NO levels; at 12 h, fluorescence in Arg–MIL-125-treated cells reached 16 ± 1.7 arbitrary units (a.u). versus 5 ± 0.2 a.u. for the unloaded framework, whereas Arg–MOF-808 achieved 2.5 ± 0.3 a.u. compared to 1.0 ± 0.2 a.u. for its unloaded form. Videos S17–24 show the time-lapse fluorescence images of ˙NO generation in response to different nanoparticles, compared to the untreated cells.
Overall, Arg–MOF-808 generated significantly more sustained ˙NO than MIL-125 (p < 0.01, 6–12 h), whereas sonication primarily accelerated the early release peak without altering the total cumulative ˙NO output. Although sonicated MOF-808 nanoparticles appeared to cause minor interference in fluorescence readings, the substantial increase observed with Arg–MOF-808 confirms that the enhanced signal reflects genuine intracellular ˙NO production rather than optical artifacts. These ˙NO-release profiles closely aligned with the arginine-release kinetics, wherein MIL-125 exhibited a slower, diffusion-controlled release, whereas MOF-808 enabled faster and more sustained substrate delivery. Collectively, DAF-FM data demonstrated that Arg loading markedly enhanced iNOS-dependent ˙NO generation in cancer cells, with MOF-808 providing a more stable and prolonged intracellular ˙NO signal, especially following sonication. The time course ˙NO generation in L-Arg-treated cells is shown in Fig. S17. These results indicate that the Arg-loaded MOFs, provides significantly more potent and sustained ˙NO delivery than free L-Arg, especially when sonicated. This sustained intracellular ˙NO is crucial for therapeutic applications, and DAF-FM imaging here effectively captures the differences in kinetic profile and delivery performance between the two approaches.
This pronounced inhibition by Arg-loaded MOFs is consistent with the established cytostatic and antimigratory effects of high ˙NO concentrations. L-Arg serves as a substrate for iNOS, and its excess drives elevated ˙NO generation. While low ˙NO levels (nanomolar range) promote tumour cell survival and migration, high ˙NO fluxes (micromolar range) induce oxidative and nitrosative stress, DNA damage, and apoptosis.6,7 Comparable observations have been reported in other cancer models as well. For instance, it was demonstrated that the overexpression of argininosuccinate synthetase in hepatocellular carcinoma cells, leading to elevated intracellular arginine levels, suppressed wound healing and metastasis.61 Similarly, nanoscale ˙NO delivery via lipid–PLGA nanoparticles reprograms the tumour microenvironment toward an immunostimulatory phenotype, inhibiting metastasis in vivo.62
Our findings align with these reports: Arg-loaded MOFs likely provided a burst of substrate to iNOS, generating a transient but intense ˙NO flux that suppressed migration. The concentration-dependent behaviour of ˙NO explains this effect: low ˙NO supports PI3K/Akt and HIF-1α pathways to promote survival and migration, whereas sustained high ˙NO in serum-free conditions overwhelms these signals, leading to cytostasis. In our serum-free assay setting, rapid arginine-release probably triggered a high ˙NO peak that transiently arrested cell motility, consistent with previous findings where fast-degrading ˙NO donors inhibited migration in FBS-free media.63 The MOF scaffolds may have contributed by locally concentrating arginine and facilitating localized ˙NO generation. Moreover, MOFs at the nanoscale can enhance intracellular uptake and, under certain conditions, generate reactive species,64 which might further potentiate this effect, although this mechanism was not explored in this study.
Together, these data show that NH2-MIL-125 and MOF-808 act as active arginine carriers in iNOS-expressing TNBC cells, enhancing intracellular ˙NO-associated responses and reducing migration, while also modulating T-cell metabolic fitness. Viewed alongside the T-cell findings, this supports a dual biological role for MOF-mediated arginine delivery, in which the overall outcome depends not only on cargo identity but also on carrier-dependent release behaviour and the metabolic context of the target cell. The two frameworks therefore provide distinct yet complementary delivery profiles that influence both immune and tumour-cell behaviour within the TNBC microenvironment. As these findings are based on in vitro models, future studies will assess pharmacokinetics, biodistribution, and safety in orthotopic TNBC models to establish translational relevance.
4. Conclusion
This study establishes NH2-MIL-125(Ti) and MOF-808(Zr) as tunable arginine nanocarriers for coordinated modulation of tumour and immune-cell behaviour in TNBC. The two frameworks exhibited distinct physicochemical properties, with NH2-MIL-125 showing higher arginine loading capacity and more sustained release, while MOF-808 displayed faster release and greater colloidal stability in biologically relevant media. These delivery differences translated into markedly different biological responses. In activated human T cells, Arg-loaded MOFs restored mitochondrial respiration relative to unloaded carriers and induced formulation-dependent immunometabolic phenotypes. NH2-MIL-125-Arg drove a stronger but less regulated respiratory response characterized by elevated proton leak and loss of spare respiratory capacity, whereas MOF-808-Arg supported a more balanced bioenergetic profile. Flow-cytometry, follow-up CCR7 analysis, and IFN-γ measurements further showed that arginine delivery influenced T-cell viability, activation state, and cytokine output in a carrier-dependent manner. In parallel, both Arg-loaded MOFs enhanced intracellular ˙NO-associated responses in iNOS-expressing MDA-MB-231 cells and inhibited tumour-cell migration, supporting their role as functional substrate-delivery systems in the TNBC setting. These differences are summarized in Scheme 2. Collectively, these findings show that MOF-mediated arginine delivery is not merely a strategy for amplifying tumour-cell ˙NO production, but a broader platform for coordinated tumour–immune metabolic modulation. By linking carrier chemistry to release kinetics, T-cell bioenergetics, and tumour-cell behaviour, this work provides a foundation for future development of nutrient-delivery biomaterials for metabolically hostile cancers. Further studies should define the underlying arginine-sensing pathways in immune cells, clarify long-term degradation and safety profiles, and evaluate therapeutic efficacy in vivo.Author contributions
Amir Alsharabasy: data curation (lead), formal analysis (lead), investigation (lead), methodology (lead), validation (lead), writing – original draft (lead), writing – review & editing (supporting). Aibhe Boran: data curation (supporting), methodology (supporting), formal analysis (supporting), investigation (supporting). Roberto González-Gómez: data curation (supporting), methodology (supporting), formal analysis (supporting), investigation (supporting), writing – original draft (supporting). Harry Revill: data curation (supporting), methodology (supporting), formal analysis (supporting). Abhay Pandit: conceptualization (equal), funding acquisition (lead), supervision (lead), writing – review & editing (lead). Pau Farràs: conceptualization (equal), funding acquisition (lead); supervision (lead), writing – review & editing (lead).Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d6bm00195e.Acknowledgements
We acknowledge the research grant from Research Ireland co-funded under the European Regional Development Fund (Grant number 13/RC/2073_P2). A. A. acknowledges funding support from the Irish Research Council (Grant number GOIPD/2023/1640). A. B. acknowledges funding support from the Irish Research Council (GOIPG/2025/8895). H. R. acknowledges MedDevDoc, Co-funded by the European Union's Marie Skłodowska-Curie Actions (MSCA) programme under Grant Agreement 101126640.References
- C. Nathan and Q.-w. Xie, Cell, 1994, 78, 915–918 CrossRef CAS PubMed.
- S. W. Ryter and A. M. K. Choi, Am. J. Respir. Cell Mol. Biol., 2009, 41, 251–260 CrossRef CAS PubMed.
- D. Fukumura, S. Kashiwagi and R. K. Jain, Nat. Rev. Cancer, 2006, 6, 521–534 CrossRef CAS PubMed.
- H. Cheng, L. Wang, M. Mollica, A. T. Re, S. Wu and L. Zuo, Cancer Lett., 2014, 353, 1–7 CrossRef CAS PubMed.
- P. Fagone, E. Mazzon, P. Bramanti, K. Bendtzen and F. Nicoletti, Eur. J. Pharmacol., 2018, 834, 92–102 CrossRef CAS PubMed.
- A. J. Burke, F. J. Sullivan, F. J. Giles and S. A. Glynn, Carcinogenesis, 2013, 34, 503–512 CrossRef CAS PubMed.
- A. M. Alsharabasy, S. A. Glynn and A. Pandit, Biochem. Soc. Trans., 2020, 48, 2539–2555 CrossRef CAS PubMed.
- P. Garrido, A. Shalaby, E. M. Walsh, N. Keane, M. Webber, M. M. Keane and F. J. Sullivan, et al. , Oncotarget, 2017, 8, 80568–80588 CrossRef PubMed.
- A. M. Alsharabasy, A. Aljaabary, R. Bohara, P. Farràs, S. A. Glynn and A. Pandit, ACS Pharmacol. Transl. Sci., 2023, 6, 1416–1432 CrossRef CAS PubMed.
- M. Lahiri and J. H. J. Martin, Oncol. Rep., 2009, 21, 275–281 CAS.
- S. Kashiwagi, K. Tsukada, L. Xu, J. Miyazaki, S. V. Kozin, J. A. Tyrrell and W. C. Sessa, et al. , Nat. Med., 2008, 14, 255–257 CrossRef CAS PubMed.
- K. M. Miranda, L. A. Ridnour, C. L. McGinity, D. Bhattacharyya and D. A. Wink, Inorg. Chem., 2021, 60, 15941–15947 CrossRef CAS PubMed.
- K.-D. Kröncke and C. V. Suschek, J. Invest. Dermatol., 2008, 128, 258–260 CrossRef PubMed.
- J. A. Hrabie and L. K. Keefer, Chem. Rev., 2002, 102, 1135–1154 CrossRef CAS PubMed.
- S. Bodeboger, F. Scalera and L. Ignarro, Pharmacol. Ther., 2007, 114, 295–306 CrossRef PubMed.
- K. Zhang, H. Xu, X. Jia, Y. Chen, M. Ma, L. Sun and H. Chen, ACS Nano, 2016, 10, 10816–10828 CrossRef CAS PubMed.
- S.-S. Wan, J.-Y. Zeng, H. Cheng and X.-Z. Zhang, Biomaterials, 2018, 185, 51–62 CrossRef CAS PubMed.
- V. Bronte and P. Zanovello, Nat. Rev. Immunol., 2005, 5, 641–654 CrossRef CAS PubMed.
- P. C. Rodriguez, D. G. Quiceno and A. C. Ochoa, Blood, 2007, 109, 1568–1573 CrossRef CAS PubMed.
- R. Geiger, J. C. Rieckmann, T. Wolf, C. Basso, Y. Feng, T. Fuhrer, M. Kogadeeva, P. Picotti, F. Meissner, M. Mann, N. Zamboni, F. Sallusto and A. Lanzavecchia, Cell, 2016, 167, 829–842 CrossRef CAS PubMed.
- H. B. Ji, S.-N. Kim, C. R. Kim, C. H. Min, J. H. Han, M. J. Kim, C. Lee and Y. B. Choy, Biomater. Adv., 2023, 145, 213268 CrossRef CAS PubMed.
- R. Chaudhari, V. Patel and A. Kumar, Nanoscale Adv., 2024, 6, 2270–2286 RSC.
- X.-R. Jiang, J. Mi, Y. Wang, M. Yin, Y. Tong and Y. Zhu, Int. J. Nanomed., 2025, 20, 10127–10149 CrossRef CAS PubMed.
- S. Kulkarni, S. Soman, A. Pandey and S. Mutalik, Int. J. Biol. Macromol., 2025, 322, 146857 CrossRef CAS PubMed.
- S. Zhou, Q. Shang, J. Ji and Y. Luan, ACS Appl. Mater. Interfaces, 2021, 13, 47407–47417 CrossRef CAS PubMed.
- F. D. Duman, A. Monaco, R. Foulkes, C. R. Becer and R. S. Forgan, ACS Appl. Nano Mater., 2022, 5, 13862–13873 CrossRef PubMed.
- A. Kazemi, H. A. M. Aliabadi, M. H. Afshari, M. Tamtaji, H. Baesmat, S. Keshavarz and F. Zeinali, et al. , ACS Appl. Bio Mater., 2025, 8, 7830–7841 CrossRef CAS PubMed.
- A. Rengaraj, P. Puthiaraj, N.-S. Heo, H. Lee, S. K. Hwang, S. Kwon, W.-S. Ahn and Y.-S. Huh, Colloids Surf., B, 2017, 160, 1–10 CrossRef CAS PubMed.
- M. Bagherzadeh, M. Safarkhani, M. Kiani, F. Radmanesh, H. Daneshgar, A. M. Ghadiri and F. Taghavimandi, et al. , Sci. Rep., 2022, 12, 12105 CrossRef CAS PubMed.
- G. Wen and Z. Guo, Colloids Surf., A, 2018, 541, 58–67 CrossRef CAS.
- J. Y. Oh, E. Choi, B. Jana, E. M. Go, E. Jin, S. Jin and J. Lee, et al. , Small, 2023, 19, 2300218 CrossRef CAS PubMed.
- X. Yin, S. Xue, J. Ji, M. Zhao, M. Shao, L. Yin and S. Zhang, et al. , Adv. Compos. Hybrid Mater., 2025, 8, 332 CrossRef CAS.
- A. C. Stauß, C. Fuchs, P. Jansen, S. Repert, K. Alcock, S. Ludewig and W. Rozhon, Molecules, 2024, 29, 3262 CrossRef.
- A. M. Alsharabasy, D. Cherukaraveedu, J. Warneke, Z. Warneke, J. R. Galán-Mascarós, S. A. Glynn, P. Farràs and A. Pandit, Small Sci., 2024, 4, 2400237 CrossRef CAS PubMed.
- A. Ahmed, C. G. Efthymiou, R. Sanii, E. Patyk-Kazmierczak, A. M. Alsharabasy, M. Winterlich and N. Kumar, et al. , J. Mater. Chem. B, 2022, 10, 1378–1385 RSC.
- B. Möller, M. Glaß, D. Misiak and S. Posch, J. Open Res. Softw., 2016, 4, 17 CrossRef.
- M. Rath, I. Müller, P. Kropf, E. I. Closs and M. Munder, Front. Immunol., 2014, 5, 532 Search PubMed.
- Z. V. Kuropteva, L. M. Baider, L. G. Nagler, T. N. Bogatyrenko and O. L. Belaia, Russ. Chem. Bull., 2019, 68, 174–180 CrossRef CAS.
- H. Furukawa, F. Gándara, Y.-B. Zhang, J. Jiang, W. L. Queen, M. R. Hudson and O. M. Yaghi, J. Am. Chem. Soc., 2014, 136, 4369–4381 CrossRef CAS PubMed.
- J. H. Cavka, S. Jakobsen, U. Olsbye, N. Guillou, C. Lamberti, S. Bordiga and K. P. Lillerud, J. Am. Chem. Soc., 2008, 130, 13850–13851 CrossRef PubMed.
- S. Yuan, J. Peng, Y. Zhang and Y. Shao-Horn, J. Phys. Chem. C, 2019, 123, 28266–28274 CrossRef CAS.
- C. Păun, L. Motelică, D. Ficai, A. Ficai and E. Andronescu, Materials, 2023, 16, 6143 CrossRef PubMed.
- S. Vilela, P. Salcedo-Abraira, I. Colinet, F. Salles, M. De Koning, M. Joosen, C. Serre and P. Horcajada, Nanomaterials, 2017, 7, 321 CrossRef PubMed.
- P. C. Rodriguez, D. G. Quiceno, J. Zabaleta, B. Ortiz, A. H. Zea, M. B. Piazuelo, A. Delgado, P. Correa, J. Brayer, E. M. Sotomayor, S. Antonia, J. B. Ochoa and A. C. Ochoa, Cancer Res., 2004, 64, 5839–5849 CrossRef CAS PubMed.
- A. H. Zea, P. C. Rodriguez, K. S. Culotta, C. P. Hernandez, J. DeSalvo, J. B. Ochoa, H.-J. Park, J. Zabaleta and A. C. Ochoa, Cell. Immunol., 2004, 232, 21–31 CrossRef CAS PubMed.
- A.-A. M. Líndez, I. Dunand-Sauthier, M. Conti, F. Gobet, N. Núñez, J. T. Hannich, H. Riezman, R. Geiger, A. Piersigilli, K. Hahn, S. Lemeille, B. Becher, T. De Smedt, S. Hugues and W. Reith, JCI Insight, 2019, 4, e132975 CrossRef PubMed.
- F. Sallusto, D. Lenig, R. Förster, M. Lipp and A. Lanzavecchia, Nature, 1999, 402, 34–38 CrossRef.
- H. Shi, N. M. Chapman, J. Wen, C. Guy, L. Long, Y. Dhungana, S. Rankin, S. Pelletier, P. Vogel, H. Wang, J. Peng, K.-L. Guan and H. Chi, Immunity, 2019, 51, 1012–1027 CrossRef CAS PubMed.
- W. Tschugguel, C. Schneeberger, G. Unfried, K. Czerwenka, W. Weninger, M. Mildner and D. M. Gruber, et al. , Breast Cancer Res. Treat., 1999, 56, 143–149 CrossRef PubMed.
- E. Linnane, S. Haddad, F. Melle, Z. Mei and D. Fairen-Jimenez, Chem. Soc. Rev., 2022, 51, 6065–6086 RSC.
- Z. He, Z. Liu, Y. Zhang, T. Guo and N. Feng, J. Nanobiotechnol., 2025, 23, 118 CrossRef CAS PubMed.
- T. Hidalgo, M. Alonso-Nocelo, B. L. Bouzo, S. Reimondez-Troitiño, C. Abuin-Redondo, M. de la Fuente and P. Horcajada, Nanoscale, 2020, 12, 4839–4845 RSC.
- N. Gan, Q. Sun, L. Zhao, P. Tang, Z. Suo, S. Zhang, Y. Zhang, M. Zhang, W. Wang and H. Li, Int. J. Biol. Macromol., 2019, 140, 709–718 CrossRef CAS PubMed.
- P. Wiśniewska, J. Haponiuk, M. R. Saeb, N. Rabiee and S. A. Bencherif, Chem. Eng. J., 2023, 471, 144400 CrossRef PubMed.
- M. Safarkhani, A. Ojaghi, S. M. Nezhad, H. Daneshgar, A. C. Paiva-Santos, F. Radmanesh and M. Bagherzadeh, et al. , Adv. Compos. Hybrid Mater., 2024, 7, 18 CrossRef CAS.
- G. Duan, S.-g. Kang, X. Tian, J. A. Garate, L. Zhao, C. Ge and R. Zhou, Nanoscale, 2015, 7, 15214–15224 RSC.
- D. P. Mitev, A. M. Alsharabasy, L. Morrison, S. Wittig, C. Diener and A. Pandit, Front. Bioeng. Biotechnol., 2021, 9, 637587 CrossRef PubMed.
- R. Singh, S. Pervin, A. Karimi, S. Cederbaum and G. Chaudhuri, Cancer Res., 2000, 60, 3305–3311 CAS.
- J. L. Gordon, K. J. Hinsen, M. M. Reynolds and M. A. Brown, Diseases, 2021, 9, 82 CrossRef CAS PubMed.
- Y. Sun, L. Zheng, Y. Yang, X. Qian, T. Fu, X. Li and Z. Yang, et al. , Nano-Micro Lett., 2020, 12, 103 CrossRef CAS PubMed.
- H. S. Eun, J. Liver Cancer, 2025, 25, 1–3 CrossRef PubMed.
- Y.-C. Sung, P.-R. Jin, L.-A. Chu, F.-F. Hsu, M.-R. Wang, C.-C. Chang and S.-J. Chiou, et al. , Nat. Nanotechnol., 2019, 14, 1160–1169 CrossRef CAS PubMed.
- A. M. Alsharabasy, S. Glynn, P. Farràs and A. Pandit, Biomacromolecules, 2022, 23, 3621–3647 CrossRef CAS PubMed.
- K. Ni, G. Lan and W. Lin, ACS Cent. Sci., 2020, 6, 861–868 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2026 |