 Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Waste to resource approach: CoFe2O4-modified spent tea waste for magnetic solid-phase extraction of toxic metal ions from water samples
Serdal
Seker
a,
Duygu
Ozdes
 *b and
Celal
Duran
*b and
Celal
Duran
 c
c
aGumushane University, Graduate School of Education, Gumushane, 29100, Türkiye
bGumushane University, Gumushane Vocational School, 29100, Gumushane, Türkiye. E-mail: duyguozdes@gumushane.edu.tr
cKaradeniz Technical University, Faculty of Sciences, Department of Chemistry, 61080, Trabzon, Türkiye
First published on 9th March 2026
Abstract
In this research, within the framework of a waste-to-resource conversion approach, biochar obtained from spent tea waste (STWB), a widely available and cost-free biomass source, was modified with magnetic CoFe2O4 nanoparticles to develop an environmentally friendly adsorbent (CoFe2O4/STWB). The applicability of this material for efficient separation and preconcentration of Cd(II), Pb(II), and Ni(II) ions from mining wastewater, stream water, and wastewater samples collected from an organized industrial zone was systematically investigated using the solid phase extraction (SPE) method. Optimization of the experimental variables was conducted to enhance the extraction performance of the target analyte ions. The fact that the adsorption and desorption equilibrium times occur in less than 1 min demonstrates the rapid and highly efficient extraction performance of the developed adsorbent. Langmuir isotherm model analysis revealed that CoFe2O4/STWB exhibits adsorption capacities of 69.4 mg g−1 for Cd(II), 185.2 mg g−1 for Pb(II), and 45.2 mg g−1 for Ni(II). In contrast, the corresponding adsorption capacities obtained for unmodified STWB were considerably lower, with values of 42.9, 75.8, and 16.8 mg g−1 for Cd(II), Pb(II), and Ni(II), respectively. These results clearly demonstrate that modification with CoFe2O4 significantly enhances the adsorption performance of STWB. Consequently, CoFe2O4-modified spent tea waste can be regarded as an effective, economical, and sustainable adsorbent for trace heavy metal analysis in environmental water samples, owing to its rapid extraction kinetics, high adsorption capacity, and low detection limits.
1 Introduction
Heavy metals (HMs) represent a major class of environmental contaminants owing to their pronounced toxicity, tendency to bioaccumulate, and resistance to degradation in environmental systems.1 Heavy metal ions such as cadmium (Cd), lead (Pb), and nickel (Ni) can enter aquatic environments through mining activities, metal plating industries, battery and accumulator production, paint and pigment manufacturing, fertilizer application, and the discharge of domestic and industrial wastewater.2 Even at low concentrations, these metals exert serious toxic effects on living organisms; Cd is associated with kidney and bone damage,3 Pb can cause disorders of the nervous system and developmental impairments,4 while Ni may induce allergic reactions, respiratory diseases, and an increased risk of cancer.5 Therefore, the precise and reliable determination of HM levels in drinking and surface waters is of great importance for environmental monitoring and the protection of public health.6In the analysis of HMs in environmental matrices, the presence of metal ions at very low concentrations, along with matrix interferences, often complicates analytical measurements. Therefore, the application of a preconcentration step prior to direct analysis is of great importance.7 Commonly employed preconcentration techniques include liquid–liquid extraction,8 cloud point extraction,9 solid-phase extraction,10 and coprecipitation.11 Among these methods, SPE has gained particular prominence due to its ability to provide high preconcentration factors, low solvent consumption, reusability of adsorbents, ease of automation, and environmentally friendly characteristics.12 SPE is an efficient preconcentration method based on the selective retention of target analytes on a suitable adsorbent, followed by their recovery using a small volume of eluent.13 When magnetic adsorbents are integrated into SPE systems, the need for centrifugation or filtration is eliminated, resulting in a significant reduction in processing time. Magnetic solid-phase extraction (MSPE) enables the improvement of detection limits, particularly in metal analyses performed using techniques with relatively low sensitivity, such as flame atomic absorption spectrometry (FAAS).14
In recent years, research has increasingly concentrated on the development of environmentally friendly, low-cost, and sustainable adsorbents that can be effectively employed in SPE techniques. Biochar is a carbonaceous substance obtained via pyrolysis of biomass-derived resources under oxygen-limited or oxygen-free conditions. Its production from agricultural and organic wastes, together with its high specific surface area and abundant surface functional groups, makes it a highly attractive adsorbent for environmental applications.15,16 Accordingly, the conversion of abundant biomass resources commonly regarded as waste—such as spent tea waste—into biochar provides significant advantages in terms of waste valorization and the generation of value-added materials.17 In the existing literature, biochar has been extensively used as a sorbent for the separation and preconcentration of organic contaminants using SPE. Yang et al. reported the use of wolfberry biomass waste derived biochar as a SPE sorbent, enabling efficient extraction of 26 antibiotics from aquatic products.18 Niu et al. developed a SPE method utilizing eco-friendly coconut shell biochar as the adsorbent for the extraction and quantification of phenoxyacetic acid herbicides in environmental matrices.19
Surface modification has been widely applied in numerous studies to enhance the adsorption capacity and separation efficiency of biochars. In particular, modifying biochar with magnetic nanoparticles provides significant advantages by improving adsorption properties as well as enabling rapid and efficient phase separation with the assistance of an external magnetic field.20–22 Among various magnetic materials, spinel-structured CoFe2O4 magnetic nanoparticles are frequently preferred for adsorbent modification owing to their high specific surface area, well-developed porosity, excellent sorption capability, non-toxic nature, insolubility in water, cost-effectiveness, excellent phase stability, and strong resistance to corrosion.23,24 When biochar-based structures are modified with CoFe2O4, they can establish strong interactions with heavy metal ions via mechanisms including electrostatic attraction, surface complexation, and ion exchange, thereby acting as efficient adsorbents in analytical applications.25,26 Shabelskaya et al. reported a one-step synthesis of CoFe2O4/biochar composites derived from agricultural waste, demonstrating high adsorption efficiency toward Cr(VI) removal from aqueous solutions.27 Ozdes and Duran developed a CoFe2O4-modified melon peel biochar for MSPE, achieving rapid and sensitive FAAS analysis of Cu(II), Cd(II), and Pb(II) in environmental and food samples.17
In this study, biochar obtained from spent tea waste was modified with CoFe2O4 magnetic nanoparticles, and the resulting CoFe2O4-modified biochar was employed, for the first time, as an adsorbent for the preconcentration of Cd(II), Pb(II), and Ni(II) ions from aqueous media via MSPE. In the context of this research, experimental parameters affecting adsorption and desorption efficiencies were optimized, and the obtained data were evaluated using various adsorption isotherm models. Accuracy of the developed procedure was verified through spike-recovery experiments by adding known amounts of analytes to water sample matrices. According to the literature, there is no report on the application of biochar derived from spent tea waste modified with magnetic nanoparticles (CoFe2O4) for the recovery of heavy metals via the SPE method. In this respect, the present study offers an environmentally friendly approach by developing a novel adsorbent system and converting waste biomass into high value-added products.
2 Materials and methods
2.1 Chemicals and instrumentation
All reagents employed throughout the experimental procedures, including nitric acid (HNO3), sodium hydroxide (NaOH), hydrochloric acid (HCl), iron(III) chloride hexahydrate (FeCl3·6H2O), cobalt(II) nitrate hexahydrate (Co(NO3)2·6H2O), and the salts employed in interference studies (NaCl, KCl, Ca(NO3)2, Mg(NO3)2, Na2CO3, NaNO3, Na3PO4, Na2SO4, and NH4Cl), were of analytical grade and purchased from Sigma-Aldrich (Shanghai, China) and Merck (Darmstadt, Germany). The reagents were used without further purification. Cd(II), Pb(II), and Ni(II) stock solutions (1000 mg L−1) were prepared in HNO3, and subsequent working solutions were obtained by diluting them with distilled water. Before use, all glassware was immersed in 5% (v/v) HNO3 overnight and subsequently rinsed with tap water and distilled water to remove any residual contaminants.Scanning electron microscopy coupled with energy-dispersive X-ray spectroscopy (SEM-EDX) analysis (ZEISS SIGMA 300) was performed to assess the surface morphology and elemental composition of STWB and CoFe2O4/STWB. Fourier-transform infrared (FTIR) spectroscopy was implemented through a PerkinElmer Spectrum Two device to characterize the surface functional groups. XRD patterns of the adsorbents were obtained employing Cu Kα radiation (λ = 1.5406 Å) over 2θ = 5–80° on a PANalytical X'Pert3 powder diffractometer. N2 adsorption–desorption isotherms at 77 K were used to determine the specific surface area and porosity of the adsorbents via the BET method.
Determination of metal ion concentrations was conducted through a flame atomic absorption spectrometer (PerkinElmer AAnalyst 400). A WTW inoLab pH 7110 benchtop pH meter was used for pH adjustment and measurement. Adsorption experiments were conducted using a BOECO PSU-15i mechanical shaker, and a NUVE NF 200 centrifuge was employed for phase separation. An analytical balance (KERN ABJ 220-4NM) was used for weighing the adsorbents and chemicals, while magnetic stirring was carried out using an IKA RCT Basic stirrer. A Weightlab WF-UD6 ultrasonic bath was used during the synthesis of CoFe2O4, and calcination was performed in a Protech muffle furnace. Drying of the adsorbents was carried out in a Santen SE 125 drying oven.
2.2 Preparation of CoFe2O4 modified spent tea waste-derived biochar (CoFe2O4/STWB)
In order to obtain biochar from spent tea waste, the tea residue was first dried in an oven at 105 °C and then placed in a lidded crucible inside a muffle furnace. Subsequently, the material was heated to 500 °C under an oxygen-limited atmosphere, and biochar (STWB) was produced through a pyrolysis process maintained at this temperature for 2 h.The modification of spent tea waste biochar with CoFe2O4 magnetic nanoparticles was carried out based on the co-precipitation method proposed by Lai et al. with slight modifications.28 Briefly, Fe3+ and Co2+ ions were weighed from FeCl3·6H2O and Co(NO3)2·6H2O salts at a molar ratio of 2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and mixed with 7.0 g of STWB. Subsequently, 250 mL of deionized water was added, and the resulting suspension was stirred on a magnetic stirrer at 35–40 °C for 30 min. After magnetic stirring, the suspension was placed in an ultrasonic bath and maintained at 80 °C for 2 h. To adjust the pH in the range of 10–11, a dropwise addition of preheated 3 M NaOH solution (80 °C) was carried out into the suspension. Following NaOH addition, the system was kept at 80 °C for a further 30 min. The resulting precipitate was cooled to room temperature and isolated by filtration. The precipitate was repeatedly rinsed with deionized water until the pH approached neutrality. After washing, the magnetic biochar was dried in an oven at 105 °C for 6 h and subsequently calcined at 400 °C for 4 h. Finally, the obtained magnetic biochar was ground into a fine powder using a porcelain mortar and stored in plastic containers.
1 and mixed with 7.0 g of STWB. Subsequently, 250 mL of deionized water was added, and the resulting suspension was stirred on a magnetic stirrer at 35–40 °C for 30 min. After magnetic stirring, the suspension was placed in an ultrasonic bath and maintained at 80 °C for 2 h. To adjust the pH in the range of 10–11, a dropwise addition of preheated 3 M NaOH solution (80 °C) was carried out into the suspension. Following NaOH addition, the system was kept at 80 °C for a further 30 min. The resulting precipitate was cooled to room temperature and isolated by filtration. The precipitate was repeatedly rinsed with deionized water until the pH approached neutrality. After washing, the magnetic biochar was dried in an oven at 105 °C for 6 h and subsequently calcined at 400 °C for 4 h. Finally, the obtained magnetic biochar was ground into a fine powder using a porcelain mortar and stored in plastic containers.
2.3 Adsorption and desorption of analyte ions
The SPE experiments were carried out using a batch method. In this procedure, 10 mL aqueous solutions containing 5.0 µg of Cd(II), 20.0 µg of Pb(II), and 10.0 µg of Ni(II) ions, along with 25 mg of the adsorbent, were transferred into centrifuge tubes. The pH of the solutions was adjusted to 6.0 using dilute HCl and NaOH. The prepared mixtures were shaken on a mechanical shaker for 1 min and then either centrifuged at 3000 rpm for 2 min or subjected to an external magnetic field to settle the CoFe2O4/STWB adsorbent, which had adsorbed the analyte ions, at the bottom of the tube. The supernatant was removed by decantation. For desorption of the analyte ions, the adsorbent collected at the bottom of the tube was treated with 5.0 mL of 0.2 M HCl solution for 1 min. Following this step, the mixture was centrifuged again under the same conditions. The concentrations of Cd(II), Pb(II), and Ni(II) ions transferred into the aqueous phase were then determined by FAAS.2.4 Preparation of water samples
The CoFe2O4/STWB-based SPE method was employed for the analysis of mining wastewater, stream water (Harşit Stream, Gümüşhane), and wastewater collected from an organized industrial zone. The water samples were filtered through a cellulose nitrate membrane (0.45 µm) immediately after collection, acidified with 1% (v/v) HNO3 to inhibit microbial activity, and stored in polyethylene bottles until analysis.3 Results and discussion
3.1 Adsorbent synthesis and characterization
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching of aromatic rings, while the band at 1410 cm−1 corresponds to symmetric carboxylate stretching or C
C stretching of aromatic rings, while the band at 1410 cm−1 corresponds to symmetric carboxylate stretching or C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C vibrations of aromatic structures.29,30 The band observed around 1325 cm−1 is related to C–H bending vibrations, indicating the presence of aliphatic or aromatic hydrocarbon bonds in the organic structure.31 The band at 1134 cm−1 arises from C–O stretching vibrations in compounds such as ethers or phenols.32 In addition, the sharp bands detected at 875 cm−1, 810 cm−1, and 751 cm−1 can be assigned to out-of-plane bending vibrations of aromatic C–H bonds in benzene derivatives.33
C vibrations of aromatic structures.29,30 The band observed around 1325 cm−1 is related to C–H bending vibrations, indicating the presence of aliphatic or aromatic hydrocarbon bonds in the organic structure.31 The band at 1134 cm−1 arises from C–O stretching vibrations in compounds such as ethers or phenols.32 In addition, the sharp bands detected at 875 cm−1, 810 cm−1, and 751 cm−1 can be assigned to out-of-plane bending vibrations of aromatic C–H bonds in benzene derivatives.33
As shown in Fig. 1, the FTIR spectra of STWB and CoFe2O4/STWB reveal noticeable changes in both band positions and intensities following the modification process. Slight shifts in the positions of several characteristic bands, along with significant variations in band intensities, were observed, which can be attributed to the interactions between CoFe2O4 nanoparticles and the functional groups present on the biochar surface. In particular, the distinct band appearing at 567 cm−1, assigned to Fe–O stretching vibrations, confirms the successful loading of CoFe2O4 nanoparticles onto the biochar surface and verifies the effective modification process.34
According to the EDX analysis results of STWB presented in Fig. 3(a), the material is mainly composed of carbon (69 wt% C) and oxygen (22 wt% O), which is consistent with its lignocellulosic origin. In addition, nitrogen (5 wt% N) and sulfur (5 wt% S) were detected at lower proportions in the biochar structure. The presence of these heteroatoms indicates that the biochar surface contains various functional groups that may contribute to metal ion binding. In the EDX spectrum shown in Fig. 3(b), the detected cobalt (8 wt% Co) and iron (21 wt% Fe) contents clearly demonstrate the successful incorporation of CoFe2O4 components onto the biochar surface. In parallel with the presence of the metal oxide structure, the relative surface oxygen content increased to 51 wt%, while the carbon content decreased to approximately 10 wt%, owing to surface coverage by magnetic nanoparticles. These findings collectively confirm that the biochar surface was effectively and successfully modified with magnetic CoFe2O4 nanoparticles.
3.2. Adsorption study
| Concentration (M) | Recovery (%) | |||||
|---|---|---|---|---|---|---|
| Cd(II) | Ni(II) | Pb(II) | ||||
| HCl | HNO3 | HCl | HNO3 | HCl | HNO3 | |
| 0.001 | 0.1 | 0.1 | 0.1 | 4.5 | 0.1 | 0.1 |
| 0.005 | 82.4 | 90.2 | 69.9 | 75.5 | 0.1 | 4.9 |
| 0.01 | 98.8 | 97.7 | 86.9 | 88.4 | 47.5 | 43.3 |
| 0.05 | 99.9 | 99.5 | 93.7 | 90.7 | 94.3 | 92.5 |
| 0.10 | 101.3 | 99.5 | 96.1 | 94.5 | 99.9 | 98.3 |
| 0.20 | 99.7 | 98.5 | 97.3 | 95.7 | 100.3 | 98.1 |
| 0.30 | 98.0 | 99.7 | 99.2 | 97.6 | 100.2 | 99.4 |
Optimizing the desorption solution volume is essential for achieving a high preconcentration factor (PF), a key performance indicator of the method. An insufficient eluent volume may hinder the complete transfer of analyte ions from the adsorbent surface to the solution phase, resulting in low recovery, whereas excessive eluent volumes lead to dilution of the analytes and a consequent decrease in the PF. Therefore, in this study, the effect of the volume of the 0.2 M HCl solution, determined as the eluent, on the recovery of the analyte ions was tested over the volume range of 2.5–10.0 mL. When the eluent volume was increased from 2.5 to 5.0 mL, quantitative recovery of the analyte ions was achieved, and no significant improvement was observed at higher eluent volumes. Therefore, an eluent volume of 5.0 mL was selected for all subsequent SPE experiments.
The contact time required for the desorption of analyte ions from the CoFe2O4/STWB surface was investigated over a time range of 10–600 s. For this purpose, solutions adjusted to pH 6.0 and containing 5.0 µg of Cd(II), 20.0 µg of Pb(II), and 10.0 µg of Ni(II) ions were treated with a CoFe2O4/STWB suspension at a concentration of 2.0 g L−1 for 1 min. After the adsorption step, CoFe2O4/STWB was separated from the medium, and the desorption process was carried out by shaking the adsorbent with 5.0 mL of 0.2 M HCl solution for contact times ranging from 10 to 600 s. Following each contact time, CoFe2O4/STWB was separated from the solution, and the concentrations of Cd(II), Pb(II), and Ni(II) in the desorption solutions were determined by FAAS. The obtained results demonstrated that desorption occurred within a very short time of approximately 40 s; therefore, a desorption equilibrium time of 1 min was selected for subsequent experiments (Fig. 8).
| Ions | Conc. (mg L−1) | Added as | Cd(II) (recovery%) | Pb(II) (recovery%) | Ni(II) (recovery%) |
|---|---|---|---|---|---|
| Na+ | 5000 | NaCl | 93.0 ± 3.1 | 93.6 ± 1.5 | 95.7 ± 0.4 |
| K+ | 250 | KCl | 98.8 ± 3.5 | 97.8 ± 1.6 | 97.2 ± 4.5 |
| Ca2+ | 250 | Ca(NO3)2 | 96.1 ± 0.4 | 97.8 ± 1.9 | 97.3 ± 2.4 |
| Mg2+ | 250 | Mg(NO3)2 | 97.2 ± 0.9 | 96.5 ± 2.5 | 96.7 ± 0.2 |
| CO32− | 250 | Na2CO3 | 93.9 ± 0.1 | 93.5 ± 0.1 | 92.3 ± 3.3 |
| NO3− | 250 | NaNO3 | 98.8 ± 5.3 | 98.9 ± 2.2 | 98.4 ± 2.6 |
| PO43− | 250 | Na3PO4 | 95.6 ± 3.6 | 90.6 ± 0.4 | 95.6 ± 1.1 |
| SO42− | 250 | Na2SO4 | 94.7 ± 1.7 | 90.2 ± 2.0 | 91.4 ± 0.6 |
| NH4+ | 250 | NH4Cl | 96.3 ± 0.3 | 94.1 ± 2.3 | 94.5 ± 1.3 |
| Fe(II), Mn(II), Zn(II), Hg(II) | 25 | * | 99.2 ± 1.8 | 100.1 ± 1.6 | 101.4 ± 0.7 |
 | ||
| Fig. 9 Impact of initial metal ion concentration on the adsorption of (a) Cd(II), (b) Pb(II) and (c) Ni(II). | ||
In order to elucidate the adsorbent–adsorbate interactions during the adsorption of analyte ions onto CoFe2O4/STWB and to gain insight into the surface characteristics of the adsorbent, the experimental data were fitted to the Langmuir and Freundlich isotherm models. The Langmuir isotherm model is based on the assumption that adsorption occurs as a monolayer on a homogeneous adsorbent surface possessing energetically equivalent binding sites, with no interactions between the adsorbed species.49 In contrast, the Freundlich isotherm model was developed for adsorbents with heterogeneous surfaces and assumes that adsorption takes place in a multilayer manner on active binding sites with different adsorption energies.50 The linearized equations of the Langmuir and Freundlich isotherm models are given in eqn (1) and (2), respectively.
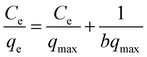 | (1) |
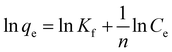 | (2) |
In these equations, qe denotes the amount of metal ions adsorbed per unit mass of CoFe2O4/STWB (mg g−1), Ce refers to the metal ion concentration at equilibrium (mg L−1), qmax represents the maximum adsorption capacity of CoFe2O4/STWB (mg g−1), b is related to the free adsorption energy (L mg−1), Kf is the Freundlich constant related to adsorption capacity (mg g−1), and n is a dimensionless constant related to adsorption intensity. The Langmuir constants b and qmax are determined from the linear plot of Ce/qeversus Ce, where the slope and intercept correspond to qmax and b, respectively. For the determination of Freundlich constants Kf and n, a linear plot of ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qeversus ln
qeversus ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Ce is constructed, where the slope and intercept are used to calculate n and Kf, respectively.
Ce is constructed, where the slope and intercept are used to calculate n and Kf, respectively.
Fig. 10 presents a comparison of the experimental adsorption data with the Langmuir and Freundlich isotherm models for the adsorption of Cd(II), Pb(II), and Ni(II) ions onto CoFe2O4/STWB. For the adsorption of all three metal ions, the correlation coefficients obtained from the Langmuir isotherm model were higher than those of the Freundlich isotherm model (>0.95), indicating that the adsorption process fits the Langmuir model well. This suggests that the metal ions are adsorbed as a monolayer onto the homogeneous surface of CoFe2O4/STWB. Using the Langmuir isotherm model, the adsorption capacities of CoFe2O4/STWB for Cd(II), Pb(II), and Ni(II) ions were calculated to be 69.4 mg g−1, 185.2 mg g−1, and 45.2 mg g−1, respectively. In order to evaluate the effect of CoFe2O4 modification on the adsorption capacity of STWB, the adsorption capacities of pristine STWB for the metal ions were also determined using the Langmuir isotherm model. According to the obtained results, the adsorption capacities of pristine STWB for Cd(II), Pb(II), and Ni(II) ions were found to be 42.9 mg g−1, 75.8 mg g−1, and 16.8 mg g−1, respectively (Table 3). These findings clearly demonstrate that modification with CoFe2O4 significantly enhances the adsorption capacity of STWB. Moreover, the adsorption capacity of CoFe2O4/STWB was found to be higher than that of many adsorbents previously reported in the literature for SPE applications (Table 4).13,51–56
| Cd(II) | Pb(II) | Ni(II) | |
|---|---|---|---|
| Langmuir isotherm model | |||
| q max | 69.4 | 185.2 | 45.2 |
| b | 0.043 | 0.141 | 0.041 |
| R 2 | 0.9847 | 0.9977 | 0.9715 |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) |
|||
| Freundlich isotherm model | |||
| K f | 7.73 | 33.5 | 8.0 |
| n | 2.59 | 2.71 | 3.48 |
| R 2 | 0.9456 | 0.8638 | 0.9092 |
| Adsorbent | Analyte | Method | Analysis technique | PF | LOD (µg L−1) | RSD (%) | Ads. capacity (mg g−1) | Ref. |
|---|---|---|---|---|---|---|---|---|
| a Fe3O4 nanoparticles coated with (3-aminopropyl)triethoxysilane and polyethylene glycol tert-octylphenyl ether. b Metal organic framework. c Cetyltrimethylammonium bromide. d Graphene oxide. e Magnetic solid phase extraction. f Micro solid phase extraction. g Dispersive solid phase extraction. | ||||||||
| Fe3O4@APTES@TX-114a | Cd(II) | MSPEe | ICP-OES | 21 | 0.889 | 1.4 | 9.9 | 51 |
| MOFb (ZIF-8)@ alginate nanocomposite | Cd(II) | µSPEf | FAAS | 60 | 3.87 | <5 | 9.1 | 52 |
| Ni(II) | 7.47 | 9.4 | ||||||
| Magnetic Fe3O4Alnus glutinosa sawdust biochar/SiO2/CTABc | Cd(II) | MSPE | FAAS | 100 | 0.62 | 3.69 | 80 | 13 |
| Pb(II) | 1.55 | 3.12 | 118.5 | |||||
| Silica nanoparticle-covered GOd | Ni(II) | dSPEg | FAAS | 10 | 13.5 | 4.0 | — | 53 |
| Molybdenum disulfide nanosheets | Cd(II) | SPE | FAAS | 71.4 | 0.37 | 3.0 | 10.32 | 54 |
| Pb(II) | 1.09 | 4.8 | 15.04 | |||||
| Ni(II) | 0.97 | 4.9 | 7.40 | |||||
| Chitosan/thiol functionalized MOFb | Pb(II) | SPE | GFAAS | 25 | 0.033 | 3.9 | 425.3 | 55 |
| Cd(II) | 0.008 | 4.1 | 109.9 | |||||
| E. profundum loaded onto Amberlite XAD-4 | Pb(II) | SPE | ICP-OES | 80 | 0.043 | 4.68 | 49.83 | 56 |
| Ni(II) | 0.042 | 4.39 | 64.35 | |||||
| CoFe2O4/STWB | Cd(II) | MSPE | FAAS | 40 | 0.35 | 1.98 | 69.4 | This work |
| Pb(II) | 2.67 | 3.31 | 185.2 | |||||
| Ni(II) | 1.83 | 2.67 | 45.2 | |||||
To evaluate the suitability of the developed adsorption systems, the dimensionless separation factor, RL, derived from the Langmuir isotherm model and the Freundlich isotherm constant n values can be considered. RL values in the range of 0–1 and n values between 1 and 10 suggest favorable adsorption behavior.57 According to the obtained results, the RL values for Cd(II), Pb(II), and Ni(II) ions in the concentration range of 50–500 mg L−1 were found to be 0.32–0.04, 0.12–0.01, and 0.33–0.05, respectively, while the n constants were determined to be 2.59, 2.71, and 3.48, respectively. These results demonstrate that the adsorption of Cd(II), Pb(II), and Ni(II) ions onto CoFe2O4/STWB is favorable.
The pronounced pH dependence, with high adsorption efficiencies observed at pH 6.0, indicates that partial deprotonation of surface functional groups increases the negative surface charge, thereby enhancing electrostatic interactions with metal ions. Furthermore, the rapid quantitative desorption achieved using 0.2 M HCl suggests that proton competition weakens metal–surface interactions. These findings support a predominantly reversible adsorption process governed by ionic and coordination-type bonding sensitive to pH conditions.
These interaction pathways are likely to remain operative in complex environmental water matrices. The high analytical recoveries obtained from real water samples indicate that the proposed adsorption mechanism remains effective even in the presence of competing ions and dissolved constituents.
3.3 Method validation and applications
| Performance parameters | Analyte ions | ||
|---|---|---|---|
| Cd(II) | Pb(II) | Ni(II) | |
| LOD (µg L−1) | 0.35 | 2.67 | 1.83 |
| LOQ (µg L−1) | 1.05 | 8.02 | 5.50 |
| RSD (%) | 1.98 | 3.31 | 2.67 |
| PF | 40 | 40 | 40 |
| Linear range (mg L−1) | 0.04–2.0 | 0.30–10.0 | 0.20–5.0 |
| Correlation coefficient | 0.9997 | 0.9998 | 0.9998 |
| Element | Added (µg) | Mining wastewater | Stream water | Organized industrial zone wastewater (OIZW) | |||
|---|---|---|---|---|---|---|---|
| Found (µg) | Recovery (%) | Found (µg) | Recovery (%) | Found (µg) | Recovery (%) | ||
| a Not detected. | |||||||
| Pb(II) | 0 | NDa | — | ND | — | ND | — |
| 50.0 | 46.6 ± 0.1 | 93.2 | 47.8 ± 0.5 | 95.5 | 44.6 ± 1.1 | 89.2 | |
| 100.0 | 99.5 ± 0.7 | 99.5 | 103.5 ± 0.9 | 103.5 | 87.4 ± 0.5 | 87.4 | |
| Cd(II) | 0 | ND | — | ND | — | ND | — |
| 10.0 | 9.72 ± 0.22 | 97.2 | 10.1 ± 0.4 | 101.0 | 9.21 ± 0.15 | 92.1 | |
| 20.0 | 20.5 ± 0.4 | 102.5 | 18.9 ± 0.3 | 94.5 | 17.6 ± 0.1 | 88.1 | |
| Ni(II) | 0 | ND | — | ND | — | ND | — |
| 10.0 | 10.3 ± 0.6 | 103.0 | 10.1 ± 0.4 | 101.0 | 8.81 ± 0.20 | 88.1 | |
| 20.0 | 17.6 ± 0.5 | 88.0 | 19.0 ± 0.6 | 95.0 | 18.3 ± 0.7 | 91.5 | |
| Element | Mining wastewater (µg L−1) | Stream water (µg L−1) | OIZW (µg L−1) |
|---|---|---|---|
| Pb(II) | GSA | 20.9 ± 0.8 | GSA |
| Cd(II) | 2.81 ± 0.02 | GSA | GSA |
| Ni(II) | 8.32 ± 0.30 | 7.21 ± 0.18 | 6.19 ± 0.14 |
4 Conclusion
In this study, spent tea waste biochar was magnetically modified with CoFe2O4 and the performance of the adsorbent for the separation and preconcentration of Cd(II), Pb(II), and Ni(II) ions from mining wastewater, stream water, and wastewater samples collected from an organized industrial zone was comprehensively evaluated using the SPE method. The findings reveal that magnetic modification of spent tea waste with CoFe2O4 markedly enhances the performance characteristics of the adsorbent. The incorporation of CoFe2O4 promotes more effective binding mechanisms for heavy metal ions, leading to high adsorption efficiencies for Cd(II), Pb(II), and Ni(II). In particular, the pronounced increase in adsorption capacity for Pb(II) clearly indicates that CoFe2O4 modification strengthens heavy metal–surface interactions. The developed SPE method is distinguished by extremely short equilibrium times, rendering it highly advantageous in terms of time and energy efficiency, as both adsorption and desorption processes are completed within seconds. Furthermore, the magnetic properties of the adsorbent enable its rapid and facile separation from the medium, representing a significant advantage that enhances the practicality and ease of application of the method. From an analytical perspective, the developed method provides a reliable and robust approach for trace level heavy metal determination, characterized by low LODs, high PF, and satisfactory precision. The effective performance of the method in complex water matrices demonstrates its robustness against matrix effects and confirms its suitability for real sample analysis.In conclusion, this study demonstrates that a widely available and cost-effective waste material can be successfully transformed into a high-value adsorbent material, thereby providing a meaningful contribution to environmental sustainability. With its economical, environmentally friendly, and high-performance characteristics, CoFe2O4-modified spent tea waste can be regarded as a viable alternative adsorbent for heavy metal monitoring in environmental water samples.
Author contributions
SS: design of methodologies, investigation, data curation and analysis. DO: supervision, resources, design of methodologies, writing – review & editing, conceptualization. CD: writing – review & editing, methodology, formal analysis, conceptualization.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data that support the findings are within the paper and additional data are available from the corresponding author upon reasonable request.Acknowledgements
This work was supported by Gümüşhane University, Scientific Research Projects Coordination Department (Project Number: 23.E0116.07.01). The authors would also like to thank Dr Yunus Önal for his valuable assistance in carrying out the BET surface area analysis measurements.References
- H. Ali, E. Khan and I. Ilahi, J. Chem., 2019, 2019, 6730305 Search PubMed.
- R. M. Madjar and G. Vasile Scăețeanu, Sustainability, 2025, 17(16), 7368 CrossRef CAS.
- A. Bernard, Indian J. Med. Res., 2008, 128(4), 557–564 CAS.
- C. Gundacker, M. Forsthuber, T. Szigeti, R. Kakucs, V. Mustieles, M. F. Fernandez, E. Bengtsen, U. Vogel, K. S. Hougaard and A. T. Saber, Int. J. Hyg. Environ. Health, 2021, 238, 113855 CrossRef CAS PubMed.
- G. Genchi, A. Carocci, G. Lauria, M. S. Sinicropi and A. Catalano, Int. J. Environ. Res. Public Health, 2020, 17(3), 679 CrossRef CAS PubMed.
- L. D. Nguyen, C. T. G. Hua, T. X. H. Dao and P. H. Tran, Microchem. J., 2025, 215, 114530 CrossRef CAS.
- J. Ruenjaiman, D. Sroichot, K. Kaewjua, W. Siangproh and K. Songsrirote, Microchem. J., 2026, 220, 116610 CrossRef CAS.
- A. A. Zendehdel, S. M. Sorouraddin and M. A. Farajzadeh, Anal. Methods, 2024, 16(11), 1593–1602 RSC.
- Y. Liu, L. Xu, J. Liu and X. Liu, Anal. Methods, 2015, 7(24), 10151–10161 RSC.
- M.-M. Zhao, H.-Z. Wu, X.-K. Deng, R.-N. Yi and Y. Yang, Anal. Methods, 2023, 16(3), 333–343 RSC.
- C. Özdemir, S. Saçmaci and Ş. Kartal, Anal. Methods, 2013, 5(16), 3977–3983 RSC.
- R. R. Pasupuleti and Y.-L. Huang, J. Chin. Chem. Soc., 2023, 70(6), 1326 CrossRef CAS.
- C. Duran and D. Ozdes, Int. J. Environ. Anal. Chem., 2023, 103(16), 4857–4875 CrossRef CAS.
- P. Montoro-Leal, J. C. García-Mesa, M. T. Siles Cordero, M. M. López Guerrero and E. Vereda Alonso, Microchem. J., 2020, 155, 104796 CrossRef CAS.
- R. N. Avcı, T. Oymak and E. Bağda, Anal. Lett., 2022, 55(16), 2495–2506 CrossRef.
- B.-M. Jun, J.-Y. Jung, M. Oh, H.-C. Eun, S. Moon, H. Rho, K. Chon, S.-N. Nam and Y. Yoon, J. Water Process Eng., 2025, 75, 107956 CrossRef.
- D. Ozdes and C. Duran, Environ. Monit. Assess., 2021, 193, 642 CrossRef CAS PubMed.
- G. Yang, J. Zhang, J. Zhang, P. Wang, W. Xia, J. Wang, X. Shen and C. Kong, Food Chem., 2025, 492, 145390 CrossRef CAS.
- Z. Niu, Y. Ma, M. Ma, Z. Tang and Y. Wen, Microchem. J., 2025, 212, 113275 CrossRef CAS.
- C. Duran, S. T. Ozeken, S. Seker and D. Ozdes, Gels, 2025, 11, 628 CrossRef CAS.
- S. Gitipour, M. Sanaei, R. Lak and A. Karbassi, Sci. Rep., 2025, 15, 7912 CrossRef CAS.
- J. Li, M. Zhang, Q. Kong, T. Zeng, Y. Mao, J. Liu and S. Xie, J. Taiwan Inst. Chem. Eng., 2025, 166(2), 105532 CrossRef CAS.
- S. I. S. Al-Hawary, A. Kamel, S. S. Abdullaev, A. K. Kareem, K. A. Alkhuzai, R. M. Romero-Parra, A. H. Amini, T. Alawsi, M. Abosaooda and M. Dejaverdi, Alex. Eng. J., 2023, 74, 737–749 CrossRef.
- F. Lin, J. Zeng, J. Chen, S. Li and W. Lin, Int. J. Biol. Macromol., 2025, 325, 147071 CrossRef CAS PubMed.
- M. H. Ullah and M. J. Rahman, Heliyon, 2024, 10(17), e36869 CrossRef CAS PubMed.
- G. Yin, X. Chen, B. Sarkar, N. S. Bolan, T. Wei, H. Zhou and H. Wang, Chem. Eng. J., 2023, 466, 143199 CrossRef CAS.
- N. Shabelskaya, M. Egorova, A. Radjabov, M. Burachevskaya, I. Lobzenko, T. Minkina and S. Sushkova, Water, 2023, 15(1), 93 CrossRef CAS.
- C. Lai, F. Huang, G. Zeng, D. Huang, L. Qin, M. Cheng, C. Zhang, B. Li, H. Yi, S. Liu, L. Li and L. Chen, Chemosphere, 2019, 224, 910–921 CrossRef CAS PubMed.
- Y. I. E. Aboulsoud, Environ. Dev. Sustain., 2024, 27(7), 16451–16468 CrossRef.
- S. Fan and L. Zhang, Biomass Convers. Biorefin., 2021, 11(5), 1719–1732 CrossRef CAS.
- N. Zhang, F. Reguyal, S. Praneeth and A. K. Sarmah, Sci. Total Environ., 2023, 886, 163923 CrossRef CAS PubMed.
- S. Fan, J. Tang, Y. Wang, H. Li, H. Zhang, J. Tang, Z. Wang and X. Li, J. Mol. Liq., 2016, 220, 432–441 CrossRef CAS.
- T. N. Mashoene, A. L. Taka, S. O. Akpotu, I. A. Lawal and M. J. Klink, Appl. Sci., 2023, 13(13), 7493 CrossRef CAS.
- D. H. K. Reddy and S.-M. Lee, Colloids Surf., A, 2014, 454, 96–103 CrossRef.
- E. Parandi, M. Shirani, Z. S. Korrani, M. Teimouri, M. A. Poor, H. Sadatfaraji, Z. Sarlak and S. Shafiei, Emergent Mater., 2025, 8, 4321–4335 CrossRef CAS.
- L. Suo, X. Dong, X. Gao, J. Xu, Z. Huang, J. Ye, X. Lu and L. Zhao, Microchem. J., 2019, 149, 104039 CrossRef CAS.
- M. Rajabi, S. Arghavani-Beydokhti, B. Barfi and A. Asghari, Anal. Chim. Acta, 2017, 957, 1–9 CrossRef CAS PubMed.
- S. M. M. Attaf, R. Alotaibi, A. A. Alotaibi and S. M. A. Azeem, J. Food Compos. Anal., 2025, 137, 106883 CrossRef CAS.
- M. S. Yalçın, S. Özdemir, E. Kılınç and M. Soylak, J. Food Compos. Anal., 2024, 134, 106561 CrossRef.
- H. E. H. Ahmed, A. M. A. Mohammed and M. Soylak, Water, Air, Soil Pollut., 2026, 237, 261 CrossRef CAS.
- H. Akbiyik, A. Girgin, B. T. Zaman, A. Atakol, N. San and S. Bakirdere, J. Anal. Sci. Technol., 2025, 16, 23 Search PubMed.
- H. E. H. Ahmed, O. Ozalp and M. Soylak, J. Food Compos. Anal., 2023, 118, 105163 CrossRef CAS.
- M. B. Arain and M. Soylak, Food Chem., 2025, 471, 142766 CrossRef CAS PubMed.
- M. Soylak, A. A. Isak and O. Ozalp, J. Food Compos. Anal., 2025, 148(3), 108401 CrossRef CAS.
- A. Türker and O. Acar, Food Anal. Methods, 2025, 18, 2106–2119 Search PubMed.
- M. Ahmad, A. Badshah, H. Li, M. Asad, M. A. Ibrahim, E. A. Ali, A. A. Shahat, U. Nishan, M. Khan and W. Sun, J. Sep. Sci., 2024, 47, e70004 Search PubMed.
- X. Tan, Y. Liu, G. Zeng, X. Wang, X. Hu, Y. Gu and Z. Yang, Chemosphere, 2015, 125, 70–85 CrossRef CAS.
- M. Suppapruek, P. Threepopnatkul, A. Sittattrakul and W. Lerdwijitjarud, E3S Web of Conferences, 2021, vol. 302, p. 02020 Search PubMed.
- I. Langmuir, J. Am. Chem. Soc., 1918, 40, 1361–1403 CrossRef CAS.
- H. M. F. Freundlich, Z. Phys. Chem., 1906, 57, 385–470 CAS.
- C. Caner, M. Yogurtcuoglu Ciftci, S. Tabassum, H. Altundag and E. Bulut, J. Mol. Struct., 2025, 1321(1), 139836 CrossRef CAS.
- M. Soylak, M. Alparslan and O. Ozalp, Anal. Lett., 2026, 59(6), 993–1011 CrossRef CAS.
- K. Seval and A. Akdoğan, Int. J. Environ. Anal. Chem., 2022, 102(19), 8402–8418 CrossRef CAS.
- Z. Tamoradi Babaei, A. Larki and K. Ghanemi, J. Iran. Chem. Soc., 2022, 19, 95–107 CrossRef.
- L. Huang, W. Huang, R. Shen and Q. Shuai, Food Chem., 2020, 330, 127212 CrossRef CAS PubMed.
- S. Ozdemir, E. Kılınç, Ö. Acer and M. Soylak, Microchem. J., 2021, 171, 106758 CrossRef CAS.
- S. T. Ozeken, D. Ozdes and C. Duran, J. Anal. Sci. Technol., 2025, 16, 22 CrossRef CAS.
- B. Zhao, X. Xu, R. Zhang and M. Cui, Environ. Sci. Pollut., 2021, 28, 16408–16419 CrossRef CAS PubMed.
- Z. Abbas, S. Ali, M. Rizwan, I. E. Zaheer, A. Malik, M. A. Riaz, M. R. Shahid, M. Z. U. Rehman and M. I. Al-Wabel, Arab. J. Geosci., 2018, 11(16), 448 CrossRef.
- H. Chakhtouna, H. Benzeid, N. Zari, A. E. K. Qaiss and R. Bouhfid, Sep. Purif. Technol., 2021, 266, 118592 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |










