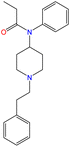
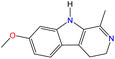
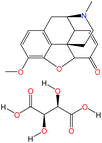
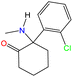
Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Rapid detection of psychoactive and new psychoactive substances using qualitative color tests: chemistry, techniques, and applications
Fabio Antonio
Cajamarca Suquila
 *a,
Andrés Fernando
Lancheros Sánchez
*a,
Andrés Fernando
Lancheros Sánchez
 *b and
Julie Gesselle
Benavides Melo
c
*b and
Julie Gesselle
Benavides Melo
c
aDepartment of Chemistry, National Pedagogic University (UPN), Bogotá, Colombia. E-mail: facajamarcas@upn.edu.co
bDepartment of Chemical Engineering and Bioprocesses, Pontificia Universidad Católica de Chile, Santiago, Chile. E-mail: aflancheros@uc.cl
cNational Pedagogic University (UPN), Bogotá, Colombia
First published on 5th May 2026
Abstract
This review may serve as a guide to develop technical and analytical guidelines for the selection and application of color test screening protocols for the detection of psychoactive substances (PSs), including new psychoactive substances (NPSs), to support their rapid identification or confirmation by competent authorities and users, particularly in the context of seizure and control procedures, thereby helping to mitigate the risks of intoxication and reduce the harm associated with their consumption. Given the global increase in the use of PSs and the rapid emergence of NPSs, these compounds pose critical challenges that require simple and rapid identification tools, such as colorimetric tests. These tests include Marquis, Mecke, Mandelin, Ehrlich, Duquenois–Levine, Zwikker, Froehde, Scott, nitric acid, ferric chloride, Simon, Dille–Koppanyi, Liebermann, Chen–Kao, Zimmermann, Hofmann, and Robadope. They operate through specific chemical reactions with key functional groups for presumptive identification. Each test is meticulously described, including its reagent formulations, preparation procedures, and applicability for detecting PSs and NPSs. The characteristic colors observed upon interaction with PSs or NPSs are also detailed, along with the underlying chemical reactions. However, the efficiency of this screening depends on the absence of adulterants with structures analogous to PSs and NPSs as well as on the absence of matrix effects in complex samples, where chromatic interferences can distort results and compromise analytical sensitivity. Given this complexity, integrating colorimetric testing into monitoring protocols optimizes regulatory control and facilitates access to chemical information. This transforms the analysis into an evidence-based care and prevention tool.
1. Introduction
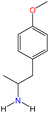
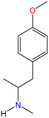
Psychoactive substances (PSs), more commonly known as psychoactive drugs, are defined by the World Health Organization (WHO) as substances that, when ingested or administered, impact mental processes such as perception, consciousness, cognition, mood, and emotions.1 They include a wide range of natural and synthetic substances that disrupt neurotransmitter systems by either mimicking (e.g., opioids, cannabinoids, and nicotine), stimulating (e.g., cocaine and amphetamines), or blocking (e.g., alcohol) neurotransmitter effects.2PSs can also be classified into seven broad categories based on their primary effects on the brain and body: stimulants, depressants, cannabinoids, psychedelics, opioids, dissociatives, and empathogens.3,4 Each category encompasses various substances with similar properties and effects. Stimulants increase activity in the central nervous system, leading to heightened alertness, euphoria, hyperactivity, and focus.5,6 Depressants, in contrast, slow down brain function, inducing relaxation, drowsiness, and impaired judgment.6 Cannabinoids are primarily associated with effects such as general pleasant feeling, mild euphoria, relaxation, time distortion, and appetite suppression, commonly found in substances derived from cannabis.7 Psychedelics profoundly alter perception, mood, and cognitive processes, often leading to hallucinations and sensory distortions.8 Opioids are potent pain-relievers that also produce feelings of euphoria but carry a high risk of dependence.9 Dissociatives distortions of perceptions of reality, pleasure, and excitement and can create feelings of detachment from the environment and the self.10 Lastly, empathogens enhance emotional communication and feelings of empathy, making users more sociable and emotionally open.3Fig. 1 highlights the main effects of each category and provides specific examples of substances included in these classifications.
 | ||
| Fig. 1 (a) Effects of psychoactive substances and (b) examples of psychoactive substances. Adapted from ref. 4. | ||
According to the World Drug Report 2024 of the United Nations Office on Drugs and Crime (UNODC), over the past decade, global drug use has increased by 20%, with 292 million people, or 5.6% of the worldwide population aged 15 to 64, having used drugs in 2022. Cannabis remains the most-used drug, with 228 million users, followed by opioids (60 million), amphetamine-type stimulants (30 million), cocaine (23 million), and ecstasy (20 million).11
Based on data collected between 2010 and 2023, with the most recent figures selected for each country, cannabis use is most widespread in North America and Western Europe. Opioid use is primarily concentrated in Eastern Europe and Central Asia, while amphetamines are most frequently used in North America and Oceania. Cocaine use is predominantly observed in North America, Western Europe, and Oceania. Ecstasy is particularly prevalent in Oceania and Europe, while opiate use is highest in Asia and Eastern Europe (Fig. 2).
Legal restrictions on traditional drugs have led to the creation of New Psychoactive Substances (NPSs).12 These substances are chemically modified versions of well-known drugs like cocaine, lysergic acid diethylamide (LSD), and cannabis, designed to provide users with a legal and recreational alternative.13,14 NPSs, also known as ‘legal highs,’ ‘bath salts,’ and ‘research chemicals,’ are defined by the UNODC as substances of abuse that are not controlled by international drug conventions but may pose public health risks.15 They have become a global issue, with 141 countries and territories reporting at least one NPS. By November 2023, 1.230 substances had been reported to the UNODC Early Warning Advisory. The term ‘new’ refers to their recent market availability, even though some were synthesized decades ago.16
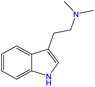
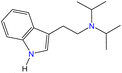
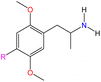
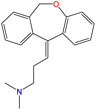
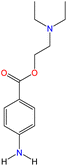
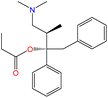
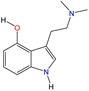
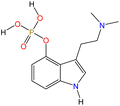
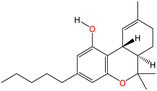
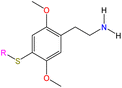
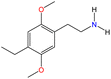
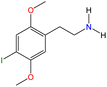
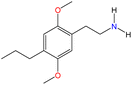
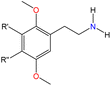
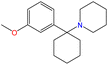
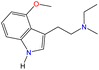
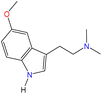
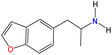
Although there are over 1.230 identified NPSs, most share a common structural feature: the presence of an amino group. However, a smaller subset consists primarily of polycyclic hydrocarbons, alcohols, and others. Fig. 3 provides a detailed chemical classification of the most prominent PSs and NPSs containing an amine functional group.17
 | ||
| Fig. 3 Chemical classification of amine-containing psychoactive and new psychoactive substances. Adapted from ref. 17. | ||
In 2022, an estimated 64 million individuals globally (approximately 1 in 81) were affected by drug use disorders, marking a 3% increase from 2018.11 This rise underscores the persistent and escalating burden of opioids, which continue to represent a major global health challenge and a significant factor in drug-related diseases.18 Opioids are involved in the majority of overdose deaths.19 In the United States alone, 915.515 drug overdose deaths occurred between 2000 and 2020, with opioid-related fatalities increasing by 350% over this period.20,21 Furthermore, the growing variety of available drugs has led to more complex patterns of use and a rising prevalence of polydrug consumption.11,22
Given the alarming increase in health issues and fatalities associated with drug use, the identification and quantification of NPSs have become increasingly critical. However, testing NPSs is challenging due to their vast variety, rapid global emergence, and intentional design to evade detection.13 Usually, NPS analysis relies on conventional techniques such as gas chromatography (GC) and liquid chromatography (LC) for screening and quantification. High-resolution mass spectrometry (HRMS) has recently gained popularity due to its ability to measure accurate masses.23 Chromatographic techniques offer excellent sensitivity and selectivity, but they have several drawbacks: they are expensive, demand highly trained chemists with specialized expertise for accurate operation,24,25 lack portability, require extensive and intrusive sample preparation,13 and rely on well-characterized reference materials, which are rarely available, for definitive NPS identification.26 However, the sophistication of adulteration methods and rapid metabolic elimination pose critical temporal and biological challenges for these advanced techniques. For example, substances such as GHB (gamma-hydroxybutyric acid) have an extremely short detection window due to their rapid elimination from the body (they disappear within a few hours), requiring immediate determination methods (point-of-care) before they degrade and the analytical signal decays significantly.25 Similarly, the vulnerability of traditional matrices, such as urine, to user manipulation and adulteration makes it necessary to explore less susceptible matrices and screening methods.27 This is why instrumental techniques are not readily available to mitigate toxicity risks and reduce harm. However, color tests are a quick, economical, and accessible alternative for preliminary detection in the field or in laboratories with limited resources. These methods allow for efficient screening and a timely response to cases of poisoning or substance adulteration.
Detection and screening tools for NPSs are essential across various settings, including seized-product analysis, adulterated beverage testing, clinical monitoring, post-mortem examinations, and health interventions. The challenge involves addressing the diverse chemical structures of NPSs and the wide range of biological matrices, with a growing focus on non-conventional matrices (e.g., dried blood spots, hair, and oral fluid) due to their detection window, minimally invasive collection methods, and reduced risk of adulteration.14 In forensic and clinical fields, accurate screening is essential because phenomena such as the endogenous and in vitro formation of certain substances in stored samples can lead to misinterpretations if adequate detection thresholds are not established.28 Additionally, the presence of highly toxic cutting agents, such as levamisole in cocaine samples, complicates the clinical picture by increasing the toxicity of the primary drug. This situation emphasizes the importance of screening not only to confirm the presence of a substance but also to serve as an early warning system for dangerous adulterants.29 From this perspective, color tests can alert to the presence of a substance because they provide valuable chemical information about functional groups, which can then be confirmed by other methods to rule out natural production.30
The increasing prevalence of health issues linked to PS and NPS use underscores the urgent need to develop and adopt more efficient rapid identification methods to mitigate associated risks and respond effectively to the crisis.31 In this context, this article proposes implementing color screening protocols to rapidly detect PSs and NPSs, thereby mitigating the risks of intoxication and reducing the harm associated with chemical toxicity and adulterants, strengthening informed decision-making and conscious consumption as fundamental public health strategies. Therefore, this review offers an overview of the primary color tests for detecting PSs and NPSs. It covers the preparation techniques for these methods, their specific applications for different PSs and NPSs, and the typical color changes associated with various substances. Furthermore, the review explains the chemical reactions responsible for the colorimetric changes observed when substances interact with test reagents.
2. Qualitative color tests for psychoactive substance identification
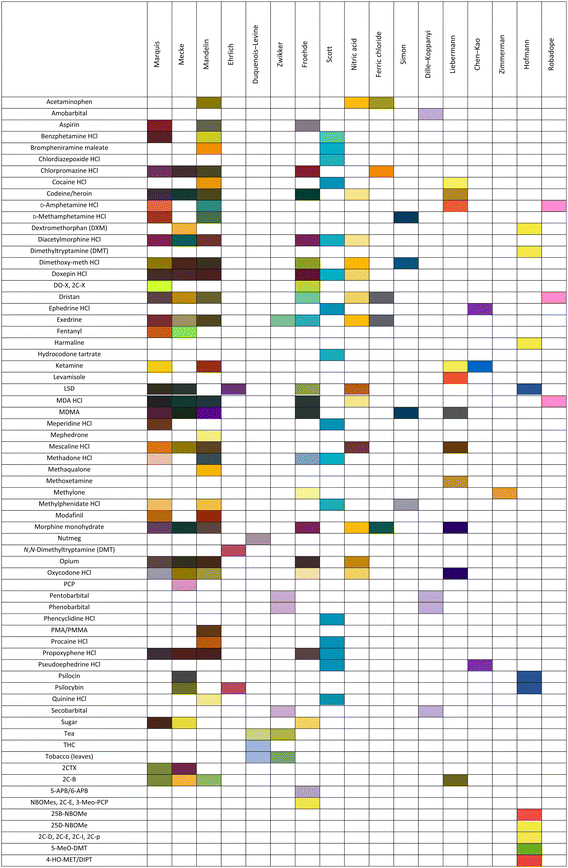
Colorimetric reactions are widely used in qualitative assays to determine the presence or absence of suspected PSs or NPSs. While these tests are practical for presumptive detection by color change, they do not provide quantitative data. The observed color changes result from specific interactions between the test reagents and functional groups within the chemical structures of the psychoactive compounds. The results should be interpreted within five minutes of the test to maintain accuracy. However, it should be noted that a positive colorimetric shift does not guarantee absolute purity. The presence of complex mixtures may interfere with the reagents by suppressing the primary reaction or producing overlapping colors (Section 4). The psychoactive substances detectable by the color tests discussed in this paper are outlined in Table 1, and their corresponding formulations are summarized in Table 2.| Color test | Reagent formulation | Preparation of the reagent | Solutions quantities | Targeted drugs | Ref. |
|---|---|---|---|---|---|
| Marquis reagent | 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 v/v formaldehyde, 40.0%: conc. sulfuric acid, (98.0%) 20 v/v formaldehyde, 40.0%: conc. sulfuric acid, (98.0%) |
1.0 mL of formaldehyde (CH2O, 30.03 g mol−1) solution is added to 20.0 mL of sulphuric acid (H2SO4, 98.03 g mol−1, ρ = 1.83 g cm−3) | 2–3 drops | Opiates, amphetamine-type compounds, and general screening agents for other drugs | 32–36 |
| Mecke reagent | 1.0% w/v selenious acid in conc. sulfuric acid (98.0%) | 0.25 g of selenious acid (H2SeO3, 128.97 g mol−1) were dissolved to 25.0 mL with sulfuric acid (H2SO4, 98.03 g mol−1, ρ = 1.83 g cm−3) | 2–3 drops | All forms of heroin | 32–36 |
| Mandelin reagent | 1.0% w/v ammonium vanadate in conc. sulfuric acid (98.0%) | 1.00 g of ammonium vanadate (NH4VO3, 116.98 g mol−1) were dissolved in 1.5 mL water and diluted to 100.0 mL with sulfuric acid (H2SO4, 98.03 g mol−1, ρ = 1.83 g cm−3) | 2–3 drops | Methadone, MDA, and MDMA | 32–36 |
| Ehrlich reagent | 5.0% w/v of p-DMAB in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v (mL) ethanol (95.0%) and conc. hydrochloric acid (37.0%) 1 v/v (mL) ethanol (95.0%) and conc. hydrochloric acid (37.0%) |
1.00 g of p-dimethylaminobenzaldehyde (C9H11NO, 149.19 g mol−1) is dissolved to 10.0 mL with methanol. Then, 10.0 mL of hydrochloric acid (HCl, 36.46 g mol−1, ρ = 1.19 g cm−3) is added | 1–2 drops | LSD, DMT and psilocybin | 32, 34 and 35 |
| Duquenois–Levine reagent | Solution (1): acetaldehyde, vanillin, and ethanol (95.0%) | Solution (1): 0.6 mL of acetaldehyde (C2H4O, 44.05 g mol−1, ρ = 0.88 g cm−3) and 0.5 g of vanillin (C8H8O3, 152.15 g mol−1, ρ = 1.06 g cm−3) are dissolved to 25.0 mL with 95.0% of ethanol (C2H6O, 46.8 g mol−1, ρ = 0.79 g cm−3) | Add 2.0 mL of solution (1) to the drug and wait for 1 min. Then, add 2.0 mL of reagent (1). Allow it to stand for 10 min. Finally, add 2.0 mL of reagent (2) | THC (marijuana) | 32–34 |
| Reagent (1): conc. hydrochloric acid | Hydrochloric acid (HCl, 36.45 g mol−1, ρ = 1.16 g cm−3) | ||||
| Reagent (3): chloroform | Chloroform (CHCl3, 119.38 g mol−1, ρ = 1.49 g cm−3) | ||||
| Zwikker reagent | Solution (1): 0.5% w/v copper(II) sulfate pentahydrate | Solution (1): 0.125 g copper(II) sulfate pentahydrate (CuSO4·5H2O) is dissolved to 25.0 mL with distilled water | Add 1 drop of solution (1) to the drug, followed by 1 drop of solution (2) | Barbiturates | 33–35 |
Solution (2): 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 19 v/v pyridine and chloroform 19 v/v pyridine and chloroform |
Solution (2): 1.0 mL pyridine (C5H5N, 79.10 g mol−1, ρ = 0.98 g cm−3) in 19.0 mL of chloroform (CHCl3, 119.38 g mol−1, ρ = 1.49 g cm−3) | ||||
| Froehde reagent | 5.0% w/v sodium molybdite in conc. sulfuric acid (98.0%) | 0.125 g sodium molybdite (Na2MoO4, 205.92 g mol−1) is dissolved to 25.0 mL with hot sulfuric acid (H2SO4, 98.03 g mol−1, ρ = 1.83 g cm−3) | 2–3 drops | Secondary reagent, effective when used in conjunction with the Marquis test | 32, 34 and 35 |
| Scott reagent | Solution (1): cobalt thiocyanate (2.0% w/v) diluted 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 with glycerine 1 with glycerine |
Solution (1): 0.20 g cobalt thiocyanate (C2CoN2S2, 175.10 g mol−1) is dissolved in 10.0 mL water and mixed with 10.0 mL 96.0% glycerin (C3H8O3, 92.09 g mol−1, ρ = 1.26 g cm−3) | Add 5 drops of solution (1) to the drug, followed by drops of reagent (1) until the precipitate disappears, and 10 drops of reagent (2) | Cocaine HCl | 32, 34 and 35 |
| Reagent (1): conc. hydrochloric acid | Hydrochloric acid (HCl, 36.45 g mol−1, ρ = 1.16 g cm−3) | ||||
| Reagent (2): chloroform | Chloroform (CHCl3, 119.38 g mol−1, ρ = 1.49 g cm−3) | ||||
| Nitric acid reagent | Conc. nitric acid (65.0%) | Nitric acid (HNO3, 63.01 g mol−1, ρ = 1.51 g cm−3) | 1–2 drops | Opiates, amphetamine-type compounds, and general screening agents for other drugs | 32–34 |
| Ferric chloride reagent | 5.0% w/v ferric chloride | 1.25 g of anhydrous ferric chloride (FeCl3, 162.20 g mol−1) or 2.08 g of ferric chloride hexahydrate (FeCl3·6H2O, 270.20 g mol−1) are dissolved to 25.0 mL with distilled water | 5 drops | Phenolic opioids | 32–35 |
| Simon reagent | Solution (1): 1.0% m/v of sodium nitroprusside | Solution (1): 0.50 g of sodium nitroprusside (Na2[Fe(CN)5NO], 261.92 g mol−1) are dissolved to 50.0 mL with distilled water | Add 1 drop of solution (1) to the drug, followed by 1 drop of solution (2) and 1 drop of solution (3) | Secondary amines | 32–35 |
| Solution (2): 4.0% v/v acetaldehyde | Solution (2): 0.5 mL of acetaldehyde (C2H4O, 44.05 g mol−1, ρ = 0.88 g cm−3) are dissolved to 25.0 mL with ethanol | ||||
| Solution (3): 2.0% m/v of sodium carbonate | Solution (3): 0.50 sodium carbonate (Na2CO3, 105.99 g mol−1) is dissolved to 25.0 mL with distilled water | ||||
| Dille–Koppanyi reagent | Solution (1): 0.1% m/v cobalt(II) acetate dihydrate in methanol and glacial acetic acid | Solution (1): 0.1 g of cobalt(II) acetate dihydrate (Co(C2H3O2)2·2H2O, 213.02 g mol−1) are dissolved to 100.0 mL with methanol and 0.2 mL of glacial acetic acid (CH3COOH, 60.05 g mol−1, ρ = 1.05 g cm−3) | Add 2 drops of solution (1) to the drug, followed by 1 drop of solution (2) | Barbiturates | 32–35 |
| Solution (2): 5.0% v/v of isopropylamine in methanol | Solution (2): 5.0 mL of isopropylamine (C3H9N, 59.11 g mol−1, ρ = 0.69 g cm−3) are dissolved to 95.0 mL with methanol | ||||
| Liebermann reagent | Solution (1): sodium nitrite in conc. sulfuric acid (98.0%) | Solution (1): 2.50 g of sodium nitrite (NaNO2, 68.99 g mol−1) are dissolved to 25.0 mL with sulphuric acid (H2SO4, 98.03 g mol−1, ρ = 1.83 g cm−3), cooling and swirling to absorb the brown fumes | Add 2 drops of solution (1) to the drug, followed by 2 drops of solution (2) | Cathinones | 32, 34 and 35 |
| Solution (2): sodium hydroxide | Solution (2): 2.00 g of sodium hydroxide (NaOH, 40,00 g mol−1) are dissolved to 25.0 mL with distilled water | Cocaine and levamisole exclusion test | |||
| Chen–Kao reagent | Solution (1): 1.0% v/v of acetic acid | Solution (1): 0.25 g of acetic acid (CH3COOH, 60.05 g mol−1, ρ = 1.05 g cm−3) are dissolved to 25.0 mL with distilled water | Add 2 drops of solution (1) to the drug, followed by 2 drops of solution (2) and 2 drops of solution (3) | Vicinal amino and hydroxyl groups | 32 and 35 |
| Solution (2): 1.0% w/v of copper sulfate | Solution (2): 0.25 g of anhydrous copper sulfate (CuSO4, 159.61 g mol−1) is dissolved to 25.0 mL with distilled water | ||||
| Solution (3): 8.0% w/v of sodium hydroxide | Solution (3): 2.00 g of sodium hydroxide (NaOH, 40.00 g mol−1) are dissolved to 25.0 mL with distilled water | ||||
| Zimmerman reagent | Solution (1): 1.0% w/v of m-dinitrobenzene in methanol | Solution (1): 0.25 g of m-dinitrobenzene (C6H4N2O4, 202.55 g mol−1) are dissolved to 25.0 mL with methanol | Add 2 drops of solution (1) to the drug and 2 drops of solution (2) | General screening test only | 34 |
| Solution (2): 15.0% w/v of potassium hydroxide | Solution (2): 3.75 g of potassium hydroxide (KOH, 56.11 g mol−1) are dissolved to 25.0 mL with distilled water | ||||
| Hofmann reagent | Solution (1): 2.0–5.0% w/v of p-DMAB in 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 v/v (mL) ethanol (95.0%) 1 v/v (mL) ethanol (95.0%) |
2.00 g of p-dimethylaminobenzaldehyde (C9H11NO, 149.19 g mol−1) is dissolved to 90.0 mL with ethanol. Next, slowly add 10.0 mL of concentrated H2SO4 while stirring constantly. Prepare in an amber glass jar and store at 4 °C | 1–2 drops | LSD and other tryptamines | 36 and 37 |
| Solution (2): conc. sulfuric acid (98.0%) | |||||
| Robadope reagent | Solution (1): 1.0% m/v of sodium nitroprusside | Solution (1): 0.50 g of sodium nitroprusside (Na2[Fe(CN)5NO], 261.92 g mol−1) are dissolved to 50.0 mL with distilled water | Add 1 drop of solution (1) to the drug, followed by 1 drop of solution (2) and 1 drop of solution (3) | Primary amines | 38 |
| Solution (2): 4.0% v/v acetone | Solution (2): 0.5 mL of acetone (C3H6O, 58.08 g mol−1, ρ = 0.78 g cm−3) are dissolved to 25.0 mL with ethanol | ||||
| Solution (3): 2.0% m/v sodium carbonate | Solution (3): 0.50 sodium carbonate (Na2CO3, 105.99 g mol−1) is dissolved to 25.0 mL with distilled water |
After presenting the reagents required to prepare the different tests, the substances that can be identified in each test and their respective colors and chemical reactions are shown below, along with their explanations.
2.1. Marquis test (Eduard Marquis)
The Marquis reagent is an effective tool for detecting and identifying a wide range of drugs, including: amphetamines, dimethyltryptamine (DMT), ketamine, LSD, 3,4-methylenedioxyamphetamine (MDA), 3,4-methylenedioxymethamphetamine (MDMA), heroin, mephedrone, methamphetamine, opioids,39 and a variety of synthetic compounds, such as those in the 4-bromo-2,5-dimethoxyphenethylamine (2C-B) and substituted dimethoxyphenethylamines (2C-X) families.34,35,40 The color change observed in the Marquis test may vary depending on the substance in the sample, as shown in Fig. 4. | ||
| Fig. 4 Colors produced by the reaction of PSs or NPSs with the reagents in the Marquis test. DO-X: substituted amphetamine derivatives. | ||
The Marquis reagent involves a reaction between formaldehyde and concentrated sulfuric acid (a dehydrating agent and catalyst) that interacts with functional groups in PSs and NPSs, forming carbocations. This highly reactive electrophile attacks the positions of the highest electron density on the aromatic ring. Meanwhile, another molecule of PS or NPS reacts with the intermediate product to form a methylene (–CH2) bridge between the two molecules. In an acidic medium, this system undergoes oxidation or protonation forming a halochromic cation with a conjugated structure. Due to extensive electron delocalization, this cation absorbs light in the visible range, producing the characteristic colors such as green, yellow, and red (Fig. 5a).41,42 For specific examples, in the case of methamphetamine (METH), the mechanism involves the formation of a benzyl carbocation and subsequent reactions leading to distinct color changes (Fig. 5b). For drugs like morphine and opium, a violet color is typically observed, which is believed to result from the dimerization of formaldehyde and opiate molecules, forming an oxonium ion.33,34,43
2.2. Mecke test
The Mecke test, often used in conjunction with the Marquis test for detecting psychoactive substances, is a highly effective reagent for detecting opium alkaloids and is especially valuable in differentiating heroin from fentanyl, as well as MDMA from dextromethorphan. Its strength lies in its ability to identify a wide range of substances, including those in the 2C-TX family (like 2C-T-2, 2C-T-7, 2C-T-4, and 2C-T-21), amphetamines, certain cathinones, cocaine, DMT, ketamine, LSD, MDA, MDMA, ibuprofen, and opioids such as morphine and codeine, as illustrated in Fig. 6.The Mecke test uses a reagent containing selenious acid and concentrated sulfuric acid to detect the presence of various substances by oxidizing the oxygen groups of the PSs or NPSs.44 The reaction is the same as in the Marquis test, except that the electrophile is a selenium species. Fig. 7 shows the chemical reaction of a morphine derivative with the Mecke reagent. In the test, the phenolic groups in opiates like morphine and codeine are oxidized, producing a dark bluish-green color. Amine groups in amphetamines and methamphetamines turn green upon oxidation, while the methylenedioxy groups in MDMA (ecstasy) yield a dark green to black color. Cocaine's ester groups typically produce an orange to pale green hue.
Due to the reagent's sensitivity to light and heat, proper storage is crucial for preserving its effectiveness.34,44 Although the Mecke test provides reliable identification across a broad spectrum of substances, it should be used alongside other reagent tests to ensure a more comprehensive and accurate analysis.45
2.3. Mandelin test (Karl Friedrich Mandelin)
The Mandelin test is a primary test for amphetamines, cocaine, and ketamine. It is also effective for detecting benzofurans, MDA, MDMA, phenethylamines, opioids, tryptamines, and certain antidepressants. Additionally, it can distinguish between NBOMe [N-(2-methoxybenzyl)] compounds and other phenethylamine derivatives. Fig. 8 shows the typical colors when the Mandelin reagent encounters a PS or NPS. | ||
| Fig. 8 Colors produced by the reaction of PSs or NPSs with the reagents in the Mandelin test. PMA: para-methoxyamphetamine; PMMA: para-methoxymethamphetamine. | ||
The Mandelin test is a secondary reagent, meaning it is most effective when used in conjunction with a primary reagent, such as the Marquis test or the Mecke test. This reagent has a yellow base color, so reactions start out yellow and progress to other colors. If the test remains the same yellow color, there is no reaction. This test combines concentrated sulfuric acid and ammonium metavanadate (NH4VO3) to oxidize specific functional groups in a substance, producing distinct color changes.46 Ammonium metavanadate in the presence of sulfuric acid is converted to VO2+, a very powerful oxidizing agent, which is subsequently reduced to VO2+ in the presence of alkaloid compounds. At the same time, sulfuric acid acts as a dehydrating agent, helping the molecules to condense (Fig. 9).
Examples of the Mandelin test include amphetamines and methamphetamines, which produce a green color due to the oxidation of amine groups. MDMA results in a dark green to black color from the oxidation of its methylenedioxy group. Opiates like morphine and codeine turn brown as their phenolic groups are oxidized, while cocaine typically gives an orange to pale green color due to oxidation of its ester group.
2.4. Ehrlich test
This test can be used for the identification of benzocaine, cocaine, DMT/DET (N,N-diethyltryptamine), LSD, psilocin, psilocybin, procaine, tetracaine, 5-HTP (L-5-hydroxytryptophan), 4-ACO-DMT [(4-acetoxy-N,N-dimethyltryptamine)], A-HO-MET (4-hydroxy-N-methyl-N-ethyltryptamine). Fig. 10 shows the typical colors obtained after the reaction between a PS or NPS and the Ehrlich reagent.The test uses p-dimethylaminobenzaldehyde (DMAB) as the primary reagent dissolved in a mixture of ethanol or methanol and concentrated HCl. The carbonyl group of DMAB becomes protonated in the presence of HCl, increasing its electrophilic character. The resulting electrophile then attacks the indole ring (a heterocyclic aromatic compound), usually at the C-2 and C-3 positions, forming a water molecule and a carbon bridge between the two aromatic systems. The final product is a highly conjugated iminium cation that absorbs light from the visible spectrum, specifically the yellow-green region. It emits the complementary color, violet or purple.46–48
Fig. 11 shows the general reaction between DMAB and the indole ring, present in several PSs and NPSs.46–48 Ehrlich reactions are typically slower than those with other reagents. It may take approximately 10 minutes to complete.
 | ||
| Fig. 11 Chemical reaction between the indole ring and DMAB/H+, producing a highly conjugated iminium cation. | ||
2.5. Duquenois–Levine test
The Duquenois–Levine test is a chemical spot test used to detect cannabinoids, mainly tetrahydrocannabinol (THC), cannabidiol (CBD), and cannabinol (CBN), by forming a purple-colored complex. That can be extracted with chloroform. The typical colors obtained in this test are shown in Fig. 12. | ||
| Fig. 12 Colors produced by the reaction of PSs or NPSs with the reagents of the Duquenois–Levine test. | ||
This test is specific to the ring structure present in these substances. Regarding the reaction mechanism, hydrochloric acid (HCl) protonates vanillin, forming a carbocation on the aldehyde carbon.49,50 This carbocation then forms a carbon–carbon bond with the resorcinol ring. The presence of acid and acetaldehyde leads to condensation and dehydration, producing a molecule with a conjugated double-bond system that allows electron delocalization and the absorption of visible light. The pH adjustment facilitates the formation of the chromophore. The initial color is blue or grayish but quickly changes to purple or intense violet. After the violet color appears, chloroform is added to form a new molecule with lower polarity. Because of this low polarity, the molecule migrates from the acid-methanol phase to the chloroform phase (Fig. 13).51–54
There is a modified version of the Duquenois–Levine test that is used in forensic laboratories. In this version, a preliminary extraction is performed with petroleum ether, followed by solvent evaporation to obtain the dry residue. Then, the reagents are added in the sequence described for this test. To avoid toxicity problems, chloroform can be replaced with methylene chloride or butyl chloride.55
2.6. Zwikker test (Cornelis Zwikker)
An aqueous copper sulfate-pyridine solution (Zwikker reagent) is used to identify barbiturates and other substances containing imidic nitrogen groups, such as sulfonamides. This test consists of a mixture of two different solutions. The first contains cupric sulfate, and the second is pyridine in chloroform.56 Phenobarbital, pentobarbital, and secobarbital turn light purple, while yellow-green is observed for Exedrine, tea, and tobacco (Fig. 14).The detection of barbiturates using the Zwikker test involves a three-step chemical mechanism: first, barbituric acids undergo enolization, forming negatively charged enolate ions. Second, copper sulfate reacts with pyridine to create a positively charged copper–pyridine complex. Finally, the enolate ion reacts with the copper–pyridine complex, forming a stable, purple-colored copper–barbiturate complex.57 This complex's coloration is the result of electronic transitions, such as charge transfer or d–d transitions, frequently due to the Jahn–Teller effect, in the orbitals of the central copper atom.58 The general chemical reaction is shown in Fig. 15. This reaction is influenced by the extent of enolization and the copper–pyridine ratio, making the process highly specific for detecting barbiturates.
 | ||
| Fig. 15 Chemical reaction between barbituric acids and the Zwikker reagent [copper(II) sulfate and pyridine] forms a distorted octahedral complex of copper(II) with barbiturates (copper–barbiturate). | ||
2.7. Froehde test
The Froehde test is best for aspirin, amphetamine, benzofurans, cocaine, DMT, ketamine, LSD, MDA, MDMA, mephedrone, methamphetamine, opioids, phenethylamines, tryptamines, and 2C-X. The characteristic colors produced by this test in some PSs and NPSs are shown in Fig. 16.The Froehde test is a secondary reagent. Therefore, it is most effective when used with the Marquis test. It is the longest-lasting reagent. It can be stored for up to 2 years when protected from light and heat. The Froehde reaction is a redox process that uses a reagent composed of sodium molybdate (Na2MoO4) dissolved in sulfuric acid and proceeds in two stages. In the first redox reaction, when an alkaloid such as morphine is present, the phenolic group reduces molybdic acid to molybdenum dioxide (MoO2), producing a violet color. The intermediate form is a pyrocatechol derivative of the alkaloid. In the second redox reaction, further oxidation leads to the formation of ortho-quinone (conjugated system) and hydrated molybdenum dioxide, resulting in a green color due to the mixture of yellow ortho-quinone and blue molybdate.59,60Fig. 17 illustrates the reaction between the Froehde reagent and an amine and an alcohol.
 | ||
| Fig. 17 Chemical reaction between the Froehde reagent (sodium molybdate and sulfuric acid) and an amine (a) or alcohol (b), forming nitrobenzene and benzaldehyde, respectively, and molybdate. | ||
2.8. Scott test (cobalt thiocyanate)
For presumptive identification of cocaine, ketamine, DCK (deschloroketamine), 2F-DCK (2-fluorodeschloroketamine), and derivatives, the Scott test is the best.The Scott test was developed and refined to distinguish between cocaine (in both base and salt forms) and other compounds that may yield false positives, such as procaine, lidocaine, and tetracaine, using Co(II) thiocyanate as the reagent.
In Scott's test, cocaine acts as a polydentate ligand that binds with the cobalt ion, forming an insoluble, deep blue complex, which can be extracted with chloroform. Upon the addition of hydrochloric acid, a pink intermediate is observed, indicating the release of the cobalt ion, which subsequently leads to the dissociation of the complex due to protonation. Finally, the addition of chloroform re-extracts the cocaine–cobalt complex, turning the solution blue again, confirming the presence of cocaine. This stepwise process enhances the test's specificity by differentiating cocaine from other local anesthetics and reducing false positives.61–63 The characteristic blue colors of this test are illustrated in Fig. 18 for various PSs and NPSs. Additionally, Fig. 19 depicts the chemical reaction between the Scott reagent and cocaine.
 | ||
| Fig. 19 Chemical reaction between cocaine (polydentate ligand) and cobalt thiocyanate, forming an insoluble complex. | ||
This test has been modified to improve its specificity for cocaine base (crack), which reacts differently from cocaine hydrochloride. In some cases, the modified Scott test uses a glycerin and sodium hydroxide (NaOH) mixture to detect ketamine hydrochloride64 and acetic acid (10%) to dissolve cocaine base. Another modification of this test involves changing the solvent. Chloroform can be replaced with methylene chloride,55 which has a similar partition coefficient for the blue lipophilic complex. This solvent is more effective for separating cocaine from lidocaine or procaine.
2.9. Nitric acid test
The nitric acid reagent is a secondary reagent used to confirm opiates (morphine, codeine, or heroin) and amphetamine-type compounds. The reaction of concentrated nitric acid with morphine, codeine, and diamorphine produces distinct nitro products, resulting in distinct color changes for each compound (Fig. 20). Morphine transitions from orange to red, then yellow; codeine changes from orange to yellow; and diamorphine changes from yellow to light green. O-Substituted derivatives like codeine and diamorphine require more nitric acid and longer reaction times to form visible nitro products compared to morphine.33,65Concentrated nitric acid (HNO3) self-ionizes to produce the nitronium ion (NO2+), which is a strong electrophile that attacks the aromatic ring to form nitro derivatives. Introducing the NO2 group into the ring extends the molecule's electronic conjugation and creates color. Fig. 21 illustrates that the nitro group in morphine forms a hydrogen bond with the hydroxyl group, resulting in a unique structural configuration. In contrast, O-substituted derivatives like codeine and heroin lack this hydrogen bond due to substitution, which leads to distinct chemical properties and color variations.33
2.10. Ferric chloride test
Ferric chloride is used to determine PSs or NPSs that have phenol in their structure, such as opioids. When ferric chloride interacts with the phenolic hydroxyl groups (–OH) in these opioids, a redox reaction occurs where the Fe3+ ion oxidizes the hydroxyl group, forming a quinone-like structure. This oxidized phenolic group then forms a complex with the Fe3+ ion, resulting in a characteristic color change, typically blue-green for morphine (Fig. 22 and 23). This reaction serves as a qualitative test for the presence of phenolic opioids, providing a quick screening method, though confirmatory tests are needed for precise identification and quantification.66 | ||
| Fig. 22 Colors produced by the reaction of PSs or NPSs with the reagents of the ferric chloride test. | ||
Because of the equilibrium between the phenol (Ar–OH) and the phenoxide ion (Ar–O–), the phenoxide ion acts as a ligand by donating its electrons to the Fe3+ ion. Depending on the phenol concentration, the Fe3+ ion can coordinate with up to six phenol molecules, resulting in octahedral geometry. The color of the complex is due to a ligand-to-metal charge transfer (LMCT), in which an electron from the oxygen p orbital of the phenol ligand is excited to the d orbital of the iron center.
2.11. Simon test
Sodium nitroprusside is a chemical reagent that detects methyl ketones and aldehydes. Thus, this test is suitable for amphetamines, cocaine, ketamine, MDA, MDMA, mephedrone, stimulants, and 2C-B. Typical colors produced by this reagent with some PSs and NPSs are shown in Fig. 24.The Simon test is used to differentiate between primary and secondary amines. It is most used to distinguish between MDMA (a secondary amine) and MDA (a primary amine), or between methamphetamine (a secondary amine) and amphetamine (a primary amine).67,68
Secondary amines, such as MDMA, are hydrolyzed in an alkaline medium (sodium carbonate solution). This process makes the amine nucleophilic, which attacks the carbonyl carbon of acetaldehyde. Water is eliminated, and an intermediate known as an enamine, is formed. Conversely, primary amines form an imine, also known as a Schiff base.
The enamine derived from the secondary amine reacts with the nitrosyl group (NO+) of nitroprussides to form a highly conjugated coordination complex known as the Simon–Awe complex (Fig. 25). This complex's electronic structure allows it to absorb light in the red region of the spectrum, producing an intense cobalt blue color. In contrast, the primary amine (e.g., amphetamine or MDA) forms a pink complex.
 | ||
| Fig. 25 Chemical reaction between a secondary amine and the Simon reagent (aldehyde and nitrosyl group – sodium nitroprusside), forming a Simon–Awe complex. | ||
2.12. Dille–Koppanyi test (James Madison Dille–Theodore Koppanyi)
The Dille–Koppanyi test is a chemical presumptive test, like the Zwikker test, used to detect barbiturates, such as amobarbital, pentobarbital, phenobarbital, and secobarbital. This test is based on the chemical reaction between Co2+ ions and barbiturate compounds, which forms a distinct violet-blue complex (Fig. 26). The Co2+ acetate is the source of Co2+ ions, and isopropylamine provides the necessary basic environment to facilitate complex formation. Barbiturates contain functional groups, such as carbonyl oxygens and nitrogen atoms, that act as ligands, coordinating with the Co2+ ions. This coordination leads to the formation of a cobalt–barbiturate complex, with the observed color change resulting from ligand-to-metal charge transfer (LMCT) or d–d electronic transitions within the complex, indicating a positive result for barbiturates.56,69,70 The complex reaction is shown in Fig. 27. | ||
| Fig. 26 Colors produced by the reaction of PSs or NPSs with the reagents of the Dille–Koppanyi test. | ||
 | ||
| Fig. 27 The chemical reaction between a barbiturate and the Dille–Koppanyi reagent [Co2+ ions and isopropylamine], producing a cobalt–barbiturate complex. | ||
2.13. Liebermann test (Leo Liebermann)
The Liebermann test detects amphetamine, cocaine, DMT, ketamine, LSD, MDA, MDMA, mephedrone, methamphetamine, opioids, and 2C-B. The Liebermann test uses a mixture of concentrated sulfuric acid and sodium nitrite to detect phenols, aromatic amines, and similar compounds by electrophilic aromatic substitution. Sulfuric acid acts as a strong dehydrating and oxidizing agent, creating an acidic environment that facilitates the formation of reactive intermediates. In this acidic medium, sodium nitrite produces nitrous acid, which generates nitrosonium ions (NO+). These ions react with aromatic amines or phenolic compounds, forming a highly conjugated indophenol cation. Phenols are oxidized to quinones or related structures, while aromatic amines undergo nitrosation, forming colored intermediates like diazonium salts. These color changes indicate the presence of specific functional groups, as shown in Fig. 28. Phenols typically produce a blue or green color, indicating oxidation to quinone structures. In contrast, aromatic amines often yield deep blue, violet, or red colors due to nitrosation and the formation of conjugated systems.35,71,72 Carbocations and N-nitrosamines are formed with primary and secondary amines, respectively, by the nitrous acid reagent. Chemical reactions occurring in the Liebermann test for phenols or aromatic amines are shown in Fig. 29. | ||
| Fig. 29 Chemical reactions between a phenol and the Liebermann reagent, resulting in a highly conjugated indophenol cation. | ||
2.14. Chen–Kao test (Ko Kuei Chen and Chung-Hsi Kao)
Phenylalkylamines, pseudoephedrine, ephedrine, norephedrine, cathinone, and methcathinone, all with vicinal amino and hydroxyl groups, are selective for the Chen–Kao reaction. Only ephedrine and pseudoephedrine produce a typical stable violet color, while other related compounds produce blue to greenish-blue precipitates. Keto-amines such as cathinone and methcathinone initially form blue complexes that transition to yellow and orange-brown, indicating instability under alkaline conditions (Fig. 31). The test demonstrates specificity within the ephedrine group but can produce similar blue copper complexes with other pharmaceuticals. The typical color changes develop slowly, requiring a few milligrams of the substance for optimal results (Fig. 30).73The Chen–Kao test involves mixing an acidic solution of ephedrine or pseudoephedrine with copper sulfate and sodium hydroxide, resulting in a violet color. Upon extraction with ether, the organic phase turns red/purple, while the aqueous phase becomes blue, indicating the formation of a coordination complex between two ephedrine molecules and a Cu2+ ion.
The amino (–NH2 or –NHR) and hydroxyl (–OH) groups on adjacent carbon atoms (in neighbouring positions) of PS or NPS molecules act as bidentate ligands. In an alkaline environment (e.g., NaOH), these groups become deprotonated and act as strong nucleophiles. In this reaction, two PS or NPS molecules coordinate with copper to form an intense violet or purple complex. Depending on the amine substitution, this complex can be either neutral or ionic. This color change is due to a charge transfer when nitrogen and oxygen coordinate with copper.74Fig. 31 illustrates the chemical reaction that occurs in this test.
 | ||
| Fig. 31 Chemical reaction between phenylalkylamines and the Chen–Kao reagent, forming an intense violet or purple complex in an alkaline medium. | ||
2.15. Zimmermann test (Robert Zimmermann)
The Zimmermann test is a colorimetric assay used to detect certain psychoactive substances, particularly compounds with active methylene (groups –CH2– adjacent to a carbonyl group C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), as occurs in benzodiazepines; however, amphetamines, cannabinoids, cathinones, clonazepam, cocaine, ketamine, MDA, MDMA, 4-MMC (4-methylmethcathinone), mephedrone, methamphetamine, opioids, and 2C-B can also be detected (Fig. 32).
O), as occurs in benzodiazepines; however, amphetamines, cannabinoids, cathinones, clonazepam, cocaine, ketamine, MDA, MDMA, 4-MMC (4-methylmethcathinone), mephedrone, methamphetamine, opioids, and 2C-B can also be detected (Fig. 32).
In this test, the hydrogen atom at the alpha position of the molecule's carbonyl group is removed by the strong base (–OH). The resulting carbanion (enolate) is a strong nucleophile that attacks the aromatic ring of m-dinitrobenzene forming a stable anionic intermediate, the Meisenheimer complex, in the first step of the reaction mechanism. Next, the complex is oxidized by an excess of m-dinitrobenzene, which acts as a hydride acceptor. This restores the planarity and aromaticity of the ring, which is known as Zimmermann anionization. The system is more efficient because aromaticity is regained and the nitro (–NO2) groups remain in resonance with the new substituent. Additionally, the system is highly conjugated, resulting in a color change indicative of the presence of the target substances (Fig. 33).75,76
 | ||
| Fig. 33 Chemical reaction between aryl ketoamines and the Zimmermann reagent (–OH and m-nitrobenzene), resulting in the Zimmermann anionization complex. | ||
2.16. Hofmann test (Albert Hofmann)
The Hofmann test is used to test for the presence of dextromethorphan, DMT, LSD, psilocin, psilocybin, NBOMe, and indole substances. The characteristic colors resulting from the reaction of PSs or NPSs with the Hofmann reagent are shown in Fig. 34.In this test, p-dimethylaminobenzaldehyde (DMAB) is used; as in the Ehrlich test, it is dissolved in a mixture of ethanol or methanol, with concentrated sulfuric acid. The carbonyl group of DMAB becomes protonated in the presence of H2SO4, increasing its electrophilic character. The electrophile attacks the indole ring, producing a water molecule and a carbon bridge between the two aromatic systems. The conjugated iminium cation (azomethine cation) obtained produces a purple/violet color.77 The reaction is the same as in the Ehrlich test (see Fig. 11).
The use of sulfuric acid rather than HCl, as in the Ehrlich test, defines the modern Hofmann method. This provides a more dehydrating medium, which accelerates the formation of the azomethine cation in bulky molecules, such as LSD, where steric hindrance (the molecule's complexity) prevents the reagent from attacking. The sulfuric acid medium better stabilizes the final complex charge, resulting in a sharper, more distinct blue color characteristic of LSD, as opposed to the pink or purple hues of simpler tryptamines, such as DMT. For this reason, the Hofmann test improves color discrimination among different indole families.78
2.17. Robadope test
Unlike the Simon test, which identifies secondary amines, the Robadope test identifies primary amines and alkaloids, such as amphetamine, cocaine, DO-X, ketamine, MDA, MDMA, mephedrone, methamphetamine, and 2C-B. The primary amine group produces a characteristic pink or red color, as illustrated in Fig. 35.In an alkaline medium containing sodium carbonate, the amine (R–NH2) present in PSs and NPSs is released, and the ketone–enolate equilibrium is favored simultaneously. The primary amine then reacts with the ketone to form an imine (Schiff base). The terminal carbon of the imine (nucleophile) attacks the nitrosyl group (NO) of the nitroprusside. This produces an electron displacement in which the nitrogen of the amine is oxidized to an iminium cation. At the same time, the oxygen of the nitrosyl group is reduced and acquires a negative charge, generating an iron complex with a highly conjugated structure.38,79 The iron complex has a zwitterionic structure, meaning it has one positive charge and one negative charge on different atoms. This charge separation promotes the formation of an extended electron cloud capable of absorbing visible radiation. In an alkaline environment, the positively charged nitrogen atom loses a proton to the surroundings, while the negatively charged oxygen atom can capture a proton or a sodium ion. The electronic delocalization arising from charge transfer between the iron center and the imine results in an intense violet or purple color (Fig. 36).79
 | ||
| Fig. 36 Chemical reaction between a primary amine and the Robadope reagent (ketone and nitrosyl group – sodium nitroprusside) forming an iron complex. | ||
As the evidence presented demonstrates, qualitative chemistry is fundamental to the analysis of PSs and NPSs, as it allows for the preliminary characterization of analytes through the identification of functional groups (assemblies of atoms), such as indoles, amines, carbonyl groups, and others, by means of color changes.30 During chemical reactions, the formation of new bonds allows for extensive delocalization of charge by resonance. This creates a chromophore that absorbs light in the visible spectrum, generating the characteristic color change used for the presumptive identification of the substance. These reactions establish the necessary chemical basis, which must be corroborated and quantified using sophisticated highly selective instrumental techniques.
These color tests are currently operational through pre-designed commercial kits that include standardized protocols for immediate field use. These kits have become essential technical tools in various preventive systems worldwide by integrating specific chemical reagents into portable, safe formats. Implementing them in harm reduction strategies enables the early detection of adulterants and highly toxic substances,37,80 transforming the rigor of coordination and organic chemistry into an efficient, practical mechanism for mitigating risk and protecting public health in non-laboratory settings.80,81
Table 3 presents the typical color changes observed for various drugs under different tests. This format provides a clear and practical overview, making it easier to identify which test is most effective for detecting a specific drug. Implementing a set of independent colorimetric tests establishes an analytical complementarity relationship, in which various reagents act synergistically when used simultaneously to enhance the assay's discriminatory power.45 This multi-reagent approach is essential for identifying masking effects or chemical interferences caused by adulterants. These agents often have structural analogues that can distort or inhibit the development of the main analyte's characteristic color. Integrating the color profiles obtained through different assays makes it easier to detect discrepancies in the chemical response. This allows us to distinguish between pure substances and complex mixtures that could otherwise generate false negatives or misinterpretations during initial screening.82
According to SWGDRUG standards,30 using multiple category C tests reinforces the consistency of the presumptive finding. An important example is the detection of cocaine adulterated with levamisole. While the Scott reagent may yield a positive result for the analyte of interest, complementary tests may reveal chromatic dissonance or an atypical coloration or variation in intensity, which indicates the presence of the cutting agent.
Therefore, creating a chemical profile using color tests significantly reduces the likelihood of false positives due to adulterants. This allows for more accurate guidance before moving on to instrumental confirmation techniques. The convergence of results reduces analytical uncertainty and, from a harm-reduction perspective, shields the protocol. Breaking through chemical masking provides more accurate information to users and healthcare personnel, enabling them to identify mixtures with higher toxicity profiles than the pure substance.62,82
3. Legal and ethical implications
After learning about the chemical basis of color tests, it is important to place these tools in their real-world context. The implementation of screening protocols for PSs and NPSs is not only focused on the scientific field but also linked to regulatory standards, accountability, and harm reduction.The United Nations Office on Drugs and Crime (UNODC) promotes the use of field color test kits as essential tools for the initial identification of controlled substances (CSs) and new psychoactive substances (NPSs) that are not yet subject to international control.71,76,81 Similarly, the WHO considers access to rapid diagnostics to be a health-related right. Implementing colorimetric tests can prevent overdoses and adverse reactions in vulnerable populations.83
In Colombia, Law 30 of 1986 (the National Narcotics Statute) states that color tests are presumptive, not definitive, expert evidence. These tests provide authorities with evidence for their investigations and help develop the principles of economy and procedural efficiency.80
However, the ethical use of color tests focuses on the researcher's responsibility and the user's well-being. A false negative could create a false sense of security in the presence of a lethal substance, while a false positive could lead to unfair legal or social consequences.84 These false results may be due to the presence of adulterants, which can be presumptively detected by color and confirmed by instrumental techniques.
According to Walker (2025),84 color tests are ethically permissible if they help prevent lethal harm from accidental exposure to adulterants and strengthen user autonomy by providing immediate access to technical information. Focusing on the analysis of the chemical safety and reliability of the substance avoids moralizing consumption, transforming screening into an act of care and scientific responsibility proportional to the current health emergency.
4. Qualitative color tests disadvantages
As the article shows, color tests are a viable, rapid, and economical way to identify PSs and NPSs. However, these tests have difficulty distinguishing the analyte signal (GHB) from natural degradation or matrix components in complex matrices, such as human hair or adulterated beverages. HPLC can accurately separate these components.25Such limitations occur because many reagents used in color tests are not selective and react with various functional groups. This can lead to false positives when adulterants are present in the sample (PSs), which are molecules with a chemical structure similar to that of the substance of interest that can react with the reagent, causing cross-interference and an overestimated reading. One critical case is levamisole in cocaine samples. The presence of this adulterant increases the mixture's toxicity and can cause cross-interference leading to erroneous or overestimated readings during initial screening. Such effects compromise the reliability of presumptive detection.29
Remember that manufacturers use adulterants to conceal degradation or actual purity. In analytical terms, if an adulterant has its own color, alters the sample's pH, or causes turbidity, it can shift the absorption spectrum. This results in a color change that masks the original color. Thus, the resulting color does not reflect the sample's actual concentration, but rather an unpredictable optical mixture.
5. Conclusion and perspectives
Color tests are a practical cost-effective method for rapid detection of PSs and NPSs in various forms, such as tablets, powders, and blotters. These tests are particularly valuable for preliminary screening, providing qualitative information about the presence of active substances. They are also essential for law enforcement agencies, enabling quick, efficient field screening of substances that are not legally permitted. However, factors such as sample adulteration, sample concentration, and reagent degradation can affect their accuracy. While these tests are quick and easy to use, they lack the precision needed for definitive identification. Therefore, results should be confirmed using more accurate techniques, such as LC-MS/MS or GC-MS/MS, which provide the necessary specificity and sensitivity.The increasing variety and complexity of PSs and NPSs pose significant challenges for public health and drug enforcement. When the proliferation of new psychoactive substances (NPSs) outpaces the updating of forensic laboratory databases, color tests provide an opportunity for immediate intervention. As Shafi et al. (2020)82 pointed out, clinical uncertainty in emergency departments and lack of confidence of health professionals in managing unknown NPS toxicities highlight the need for rapid screening tools. Therefore, integrating colorimetry into harm reduction protocols is an economical and innovative alternative that mitigates risks before a clinical emergency arises, thereby democratizing diagnosis.
In this context, colorimetry enables presumptive identification at the point of care, facilitating timely clinical decision-making and preventing the consumption of highly adulterated or lethal samples. Thus, these methods act as a critical bridge between exposure and specialized medical care, providing access to chemical information in settings with limited resources where advanced instrumentation is technically or economically infeasible.
While color tests offer a rapid, on-site solution, improving their selectivity and accuracy is crucial to reducing false positives and negatives. That is why future efforts should focus on integrating these tests with advanced portable technologies to enhance real-time drug detection. Developing more sophisticated screening protocols and fostering international collaboration will be essential to address the evolving landscape of psychoactive substances, improving both consumer safety and regulatory responses.
Conflicts of interest
There are no conflicts to declare.Data availability
All information contained in this minireview has been extracted from publicly accessible sources. The data and references used are available in the scientific literature and publicly accessible resources. No private or restricted data were used in the preparation of this manuscript.Acknowledgements
This research was supported by the FONDECYT Postdoctoral Grant No. 3240504 from the Agencia Nacional de Investigación y Desarrollo (ANID), Chile. The authors gratefully acknowledge this financial support.References
- World Health Organization, Neuroscience of Psychoactive Substance Use and Dependence, Geneva, 2004, ISBN 9241562358 Search PubMed.
- D. Dias da Silva, J. Silva, H. Carmo and F. Carvalho, Neurotoxicity of Psychoactive Substances: A Mechanistic Overview, Curr. Opin. Toxicol., 2021, 28, 76–83, DOI:10.1016/j.cotox.2021.10.002.
- M. Adley, G. Jones and F. Measham, Jump-Starting the Conversation about Harm Reduction: Making Sense of Drug Effects, Drugs: Education, Prevention and Policy, 2023, 30, 347–360, DOI:10.1080/09687637.2021.2013774.
- Alcohol and Drug Foundation Drug Facts, available online: https://adf.org.au/drug-facts/#wheel, accessed on 22 August 2024 Search PubMed.
- J. Docherty and H. Alsufyani, Pharmacology of Drugs Used as Stimulants, J. Clin. Pharmacol., 2021, 61, S53–S69, DOI:10.1002/jcph.1918.
- D. Ciucă Anghel, G. Niţescu, A. Tiron, C. Guţu and D. Baconi, Understanding the Mechanisms of Action and Effects of Drugs of Abuse, Molecules, 2023, 28, 4969, DOI:10.3390/molecules28134969.
- K. Cohen, A. Weizman and A. Weinstein, Positive and Negative Effects of Cannabis and Cannabinoids on Health, Clin. Pharmacol. Ther., 2019, 105, 1139–1147, DOI:10.1002/cpt.1381.
- F. Vollenweider, Brain Mechanisms of Hallucinogens and Entactogens, Dialogues Clin. Neurosci., 2001, 3, 265–279, DOI:10.31887/dcns.2001.3.4/fxvollenweider.
- A. Paul, C. Smith, M. Rahmatullah, V. Nissapatorn, P. Wilairatana, M. Spetea, N. Gueven and N. Dietis, Opioid Analgesia and Opioid-Induced Adverse Effects: A Review, Pharmaceuticals, 2021, 14, 1091, DOI:10.3390/ph14111091.
- M. Bates and K. Trujillo, Use and Abuse of Dissociative and Psychedelic Drugs in Adolescence, Pharmacol. Biochem. Behav., 2021, 203, 173129, DOI:10.1016/j.pbb.2021.173129.
- UNODC, World Drug Report 2024, United Nations publication, Vienna, 2024 Search PubMed.
- P. Reuter and B. Pardo, New Psychoactive Substances: Are There Any Good Options for Regulating New Psychoactive Substances?, International Journal of Drug Policy, 2017, 40, 117–122, DOI:10.1016/j.drugpo.2016.10.020.
- C. Awuchi, M. Aja, N. Mitaki, S. Morya, I. Amagwula, C. Echeta and V. Igwe, New Psychoactive Substances: Major Groups, Laboratory Testing Challenges, Public Health Concerns, and Community-Based Solutions, J. Chem., 2023, 2023, 1–36, DOI:10.1155/2023/5852315.
- S. Graziano, L. Anzillotti, G. Mannocchi, S. Pichini and F. Busardò, Screening Methods for Rapid Determination of New Psychoactive Substances (NPS) in Conventional and Non-Conventional Biological Matrices, J. Pharm. Biomed. Anal., 2019, 163, 170–179, DOI:10.1016/j.jpba.2018.10.011.
- A. Bruni, C. Rodrigues, C. dos Santos, J. de Castro, L. Mariotto and L. Sinhorini, Analytical Challenges for Identification of New Psychoactive Substances: A Literature-Based Study for Seized Drugs, BrJAC--Braz. J. Anal. Chem., 2021, 9, 52–78, DOI:10.30744/brjac.2179-3425.RV-41-2021.
- UNODC, UNODC Early Warning Advisory on New Psychoactive Substances, available online: https://www.unodc.org/LSS/Page/NPS Search PubMed.
- F. Zapata, J. M. Matey, G. Montalvo and C. García-Ruiz, Chemical Classification of New Psychoactive Substances (NPS), Microchem. J., 2021, 163, 105877, DOI:10.1016/j.microc.2020.105877.
- S. Colledge-Frisby, N. Jones, S. Larney, A. Peacock, D. Lewer, T. D. Brothers, M. Hickman, M. Farrell and L. Degenhardt, The Impact of Opioid Agonist Treatment on Hospitalisations for Injecting-Related Diseases among an Opioid Dependent Population: A Retrospective Data Linkage Study, Drug Alcohol Depend., 2022, 236, 109494, DOI:10.1016/j.drugalcdep.2022.109494.
- D. Judd, C. R. King and C. Galke, The Opioid Epidemic: A Review of the Contributing Factors, Negative Consequences, and Best Practices, Cureus, 2023, 15, e41621, DOI:10.7759/cureus.41621.
- C. Dennen, K. Blum, E. R. Braverman, A. Bowirrat, M. Gold, I. Elman, P. K. Thanos, D. Baron, A. Gupta, D. Edwards and R. Badgaiyan, How to Combat the Global Opioid Crisis, CPQ Neurology and Psychology, 2023, 5, 01–12 Search PubMed.
- M. Matero, S. Giorgi, B. Curtis, L. Ungar and H. Schwartz, Opioid Death Projections with AI-Based Forecasts Using Social Media Language, npj Digit. Med., 2023, 6, 35, DOI:10.1038/s41746-023-00776-0.
- K. Karjalainen, K. Kuussaari, K. Kataja, C. Tigerstedt and P. Hakkarainen, Measuring Concurrent Polydrug Use in General Populations: A Critical Assessment, Eur. Addctn. Res., 2017, 23, 163–169, DOI:10.1159/000477802.
- D. Pasin, A. Cawley, S. Bidny and S. Fu, Current Applications of High-Resolution Mass Spectrometry for the Analysis of New Psychoactive Substances: A Critical Review, Anal. Bioanal. Chem., 2017, 409, 5821–5836, DOI:10.1007/s00216-017-0441-4.
- B. Feigel, P. Adamowicz and S. Wybraniec, Recent Advances in Analysis of New Psychoactive Substances by Means of Liquid Chromatography Coupled with Low-Resolution Tandem Mass Spectrometry, Anal. Bioanal. Chem., 2024, 416, 107–124, DOI:10.1007/s00216-023-05057-z.
- F. Busardò, F. Vaiano, G. Mannocchi, E. Bertol, S. Zaami and E. Marinelli, Twelve Months Monitoring of Hair GHB Decay Following a Single Dose Administration in a Case of Facilitated Sexual Assault, Drug Test. Anal., 2017, 9, 953–956, DOI:10.1002/dta.2100.
- R. Archer, R. Treble and K. Williams, Reference Materials for New Psychoactive Substances, Drug Test. Anal., 2011, 3, 505–514, DOI:10.1002/dta.317.
- M. Arvidsson, S. Ullah, J. Franck, M. Dahl and O. Beck, Drug Abuse Screening with Exhaled Breath and Oral Fluid in Adults with Substance Use Disorder, Drug Test. Anal., 2018, 11, 27–32, DOI:10.1002/dta.2384.
- F. Busardò, E. Bertol, F. Vaiano, G. Baglio, A. Montana, N. Barbera, S. Zaami and G. Romano, Post Mortem Concentrations of Endogenous Gamma Hydroxybutyric Acid (GHB) and In Vitro Formation in Stored Blood and Urine Samples, Forensic Sci. Int., 2014, 243, 144–148, DOI:10.1016/j.forsciint.2014.07.019.
- S. Karch, F. Busardò, F. Vaiano, F. Portelli, S. Zaami and E. Bertol, Levamisole adulterated cocaine and pulmonary vasculitis: Presentation of two lethal cases and brief literature review, Forensic Sci. Int., 2016, 265, 96–102, DOI:10.1016/j.forsciint.2016.01.015.
- Scientific Working Group for the Analysis of Seized Drugs (SWGDRUG), SWGDRUG Recommendations, Version 8.2, available online: https://www.swgdrug.org/Documents/SWGDRUG%20Recommendations%20Version%208.2%20Final%20for%20posting.pdf, accessed on 12 March 2026 Search PubMed.
- V. Lukić, R. Micić, B. Arsić, B. Nedović and Ž. Radosavljević, Overview of the Major Classes of New Psychoactive Substances, Psychoactive Effects, Analytical Determination and Conformational Analysis of Selected Illegal Drugs, Open Chem., 2021, 19, 60–106, DOI:10.1515/chem-2021-0196.
- A. Fatah, Color Test Reagents/Kits for Preliminary Identification of Drugs of Abuse, available online: https://www.nist.gov/publications/color-test-reagentskits-preliminary-identification-drugs-abuse Search PubMed.
- K.-A. Kovar and M. Laudszun, Chemistry and Reaction Mechanisms of Rapid Tests for Drugs of Abuse and Precursors Chemicals, Tubingen, 1989 Search PubMed.
- M. Philp and S. Fu, A Review of Chemical' Spot' Tests: A Presumptive Illicit Drug Identification Technique, Drug Test. Anal., 2018, 10, 95–108, DOI:10.1002/dta.2300.
- K. Toole, S. Fu, R. Shimmon, N. Kraymen and S. Taflaga, Color Tests for the Preliminary Identification of Methcathinone and Analogues of Methcathinone, Microgram J., 2007, 9, 27–32 Search PubMed.
- H. Van Urk, New Sensitive Reaction for the Ergot Alkaloids, Ergotamine, Ergotoxine and Ergotinine and Its Adaptation to the Examination and Colorimetric Determination of Ergot Preparations, Pharm. Weekbl., 1929, 66, 473–481 CAS.
- D. Martins, M. Barratt, C. Pires, H. Carvalho, M. Vilamala, I. Espinosa and H. Valente, The Detection and Prevention of Unintentional Consumption of DOx and 25x-NBOMe at Portugal's Boom Festival, Hum. Psychopharmacol. Clin. Exp., 2017, 32, e2608, DOI:10.1002/hup.2608.
- R. Lancashire, Chemistry and Crime. Narcotics and Test Reagent Kits, available online: http://wwwchem.uwimona.edu.jm/courses/CHEM2402/Crime/Reagent_Kits.html Search PubMed.
- T. Pin, L. Chim, L. Hin and L. Lim, Presumptive Testing of Amphetamine-Type Stimulants via Colour Tests, ASEAN Journal on Science and Technology for Development, 2010, 27, 66–75 Search PubMed.
- L. Clancy, M. Philp, R. Shimmon and S. Fu, Development and Validation of a Color Spot Test Method for the Presumptive Detection of 25-NBOMe Compounds, Drug Test. Anal., 2021, 13, 929–943, DOI:10.1002/dta.2905.
- H. Auterhoff and D. Braun, Die Farbreaktion Des Morphins Nach E. MARQUIS, Arch. Pharm., 1973, 306, 866–872, DOI:10.1002/ardp.19733061109.
- K. Rehse and G. Kawerau, Mechanism of the Reaction of Aromatic Compounds with Formaldehyde in Concentrated Sulfuric Acid (Marquis Reagent), Arch. Pharmazie, 1974, 307, 934–942 CrossRef CAS PubMed.
- A. Choodum and N. NicDaeid, Quantitative Colorimetric Assays for Methamphetamine, Elsevier Inc., 2016, vol. 2, ISBN 9780128002124 Search PubMed.
- F. Sánchez-Viesca and R. Gómez, The Mechanism of Mecke's Test for Opioids, World Journal of Chemical and Pharmaceutical Sciences, 2023, 2, 023–027, DOI:10.53346/wjcps.2023.2.1.0014.
- C. O'Neal, D. Crouch and A. Fatah, Validation of Twelve Chemical Spot Tests for the Detection of Drugs of Abuse, Forensic Sci. Int., 2000, 109, 189–201, DOI:10.1016/S0379-0738(99)00235-2.
- A. Lamb, R. Federico-Perez and Z. Xue, Product in Indole Detection by Ehrlich's Reagent, Anal. Biochem., 2015, 484, 21–23, DOI:10.1016/j.ab.2015.04.033.
- Q. Jin, L. Shan, J. Yue and X. Wang, Spectrophotometric Determination of Total Serotonin Derivatives in the Safflower Seeds with Ehrlich's Reagent and the Underlying Color Reaction Mechanism, Food Chem., 2008, 108, 779–783, DOI:10.1016/j.foodchem.2007.11.022.
- P. Byrom, Spectrophotometric Determination of Indoles Using a Modification of the Ehrlich Colour Reaction, Talanta, 1963, 10, 1217–1221, DOI:10.1016/0039-9140(63)80180-0.
- R. A. Isaacs, Structure-Reactivity Relationship Driven Approach to the Identification of a Color Test Protocol for the Presumptive Indication of Synthetic Cannabimimetic Drugs of Abuse, Forensic Sci. Int., 2014, 242, 135–141, DOI:10.1016/j.forsciint.2014.06.027.
- C. Rubiano, J. Firmin and S. Rao, The Effect of Benzene Ring Substituents on the Mechanism of Duquenois Levine (DL) Test for Cannabinoid Detection, J. Chem. Pharm. Res., 2014, 6, 1261–1264 CAS.
- A. Jacobs and R. Steiner, Detection of the Duquenois–Levine Chromophore in a Marijuana Sample, Forensic Sci. Int., 2014, 239, 1–5, DOI:10.1016/j.forsciint.2014.02.031.
- J. Thornton and G. Nakamura, The Identification of Marijuana, J. Forensic Sci. Soc., 1972, 12, 461–519, DOI:10.1016/S0015-7368(72)70716-1.
- F. Fochtman and C. Winek, A Note on the Duquenois-Levine Test for Marijuana, Clin. Toxicol., 1971, 4, 287–289, DOI:10.3109/15563657108990968.
- J. Vargas, Reactivo de Duquenois: Química, Historia e Importancia, Revista Médica Basadrina, 2024, 18, 69–74, DOI:10.33326/26176068.2024.2.2251.
- A. Hanson, Specificity of the Duquenois–Levine and Cobalt Thiocyanate Tests Substituting Methylene Chloride or Butyl Chloride for Chloroform, available online: https://web.archive.org/web/20070828164524/http://www.usdoj.gov/dea/programs/forensicsci/microgram/journal_v3_num34/journal_v3_num34_pg9.html, accessed on 12 March 2026 Search PubMed.
- R. Das and J. Mishra, Comparative Spectroscopic Analysis between Dille- Koppanyi Test and Zwikker's Test for the Detection of Barbiturates in Illicit Cases, Eur. J. Mol. Clin. Med., 2019, 6, 35–49 Search PubMed.
- L. Levi and C. Hubley, Detection and Identification of Clinically Important Barbiturates, Anal. Chem., 1956, 58, 1591–1605 CrossRef.
- T. Mizokawa, Jahn–Teller Effects in Transition-Metal Compounds with Small Charge-Transfer Energy, J. Phys.:Conf. Ser., 2013, 428, 012020, DOI:10.1088/1742-6596/428/1/012020.
- F. Sánchez-Viesca and R. Gómez, On the Mechanism of the Froehde Reaction, World J. Org. Chem., 2019, 7, 1–4, DOI:10.12691/wjoc-7-1-1.
- F. Sánchez-Viesca and R. Gómez, Insight into the Mechanism of the Froehde Test for Morphine, World J. Adv. Res. Rev., 2022, 6, 001–004, DOI:10.30574/msarr.2022.6.2.0071.
- M. Marcelo, K. Mariotti, R. Ortiz, M. Ferrão and M. Anzanello, Scott Test Evaluation by Multivariate Image Analysis in Cocaine Samples, Microchem. J., 2016, 127, 87–93, DOI:10.1016/j.microc.2016.02.012.
- Y. Tsumura, T. Mitome and S. Kimoto, False Positives and False Negatives with a Cocaine-Specific Field Test and Modification of Test Protocol to Reduce False Decision, Forensic Sci. Int., 2005, 155, 158–164, DOI:10.1016/j.forsciint.2004.11.011.
- K. Oguri, S. Wada, S. Eto and H. Yamada, Specificity and Mechanism of the Color Reaction of Cocaine with Cobaltous Thiocyanate, Jpn. J. Toxicol. Environ. Health, 1995, 41, 274–279, DOI:10.1248/jhs1956.41.274.
- P. Dubey, S. Shukla and K. Gupta, Modified Scott's Test for Ketamine Hydrochloride, Aust. J. Forensic Sci., 2013, 45, 165–171, DOI:10.1080/00450618.2012.733026.
- A. Choodum and N. Nic Daeid, Rapid and Semi-Quantitative Presumptive Tests for Opiate Drugs, Talanta, 2011, 86, 284–292, DOI:10.1016/j.talanta.2011.09.015.
- S. Banerjee and B. Haldar, Constitution of Ferri-Phenol Complex in Solution, Nature, 1950, 165, 1012, DOI:10.1038/1651012b0.
- S. Prunty, D. Carmany, E. Dhummakupt and N. E. Manicke, Combining Presumptive Color Tests, Pressure-Sensitive Adhesive-Based Collection, and Paper Spray-Mass Spectrometry for Illicit Drug Detection, Analyst, 2023, 148, 3274–3284, 10.1039/D3AN00559C.
- K. Loach and M. Carvalho, A Modified Simon-Lewin Reaction: N-Nitrosamines as Intermediates in the Reaction of Secondary Amines with Nitroprusside and Acetaldehyde, Anal. Lett., 1973, 6, 25–29, DOI:10.1080/00032717308062165.
- M. de Faubert Maunder, An Improved Field Test for Barbiturates and Hydantoins with a Modified Cobalt(II) Thiocyanate Reagent, Analyst, 1975, 100, 878–883, 10.1039/an9750000878.
- E. Rhodes and J. Thornton, Microchemical Tests for Hydantoins and Barbiturate-Hydantoin Mixtures, Microchem. J., 1972, 17, 528–539, DOI:10.1016/0026-265X(72)90091-4.
- United Nations Office on Drugs and Crime, Rapid Testing Methods of Drugs of Abuse: Manual for Use by National Law Enforcement and Narcotics Laboratory Personnel, New York, 1994, available online: https://www.unodc.org/documents/scientific/Rapid_Testing_Methods_of_Drugs_of_Abuse_E.pdf, accessed on 12 March 2026 Search PubMed.
- M. Fraenkl, D. Svobodová and J. A. Gasparič, Critical Investigation of the Liebermann Colour Test: The Formation and Behaviour of Phenolindophenol in Strong Acid Media, Mikrochim. Acta, 1986, 90, 367–386, DOI:10.1007/BF01199279.
- G. Nagy, I. Szöllösi and K. Szendrei, Colour Tests for Precursor Chemicals of Amphetamine-Type Substances, 2005, available online: https://www.unodc.org/pdf/scientific/SCITEC20-fin.pdf, accessed on 12 March 2026 Search PubMed.
- K. Chen and C. Kao, Ephedrine and Pseudoephedrine, Their Isolation, Constitution, Isomerism, Properties, Derivatives and Synthesis. (with a Bibliography), J. Am. Pharm. Assoc., 1926, 15, 625–639, DOI:10.1002/jps.3080150804.
- C. Corker, J. Norymberski and R. Thow, Some Aspects of the Zimmermann Reaction, Biochem. J., 1962, 83, 583–588, DOI:10.1042/bj0830583.
- United Nations Office on Drugs and Crime, Recommended Methods for the Identification and Analysis of Synthetic Cathinones in Seized Materials, Vienna, 2020, available online: https://www.unodc.org/documents/scientific/Recommended_methods_for_the_Identification_and_Analysis_of_Synthetic_Cathinones_in_Seized_Materials-Rev..pdf, accessed on 12 March 2026 Search PubMed.
- J. Aubé, C. Fehl, R. Liu, M. McLeod and H. F. Motiwala, Hofmann Curtius, Schmidt Lossen and Related Reactions, in Comprehensive Organic Synthesis, 2nd edn, 2014, vol. 6, pp. 609–616, ISBN 9780080977430 Search PubMed.
- D. Ginsburg, Practical Considerations in Preparation of Amines, in Concerning Amines, Elsevier, 1967, pp. 17–62 Search PubMed.
- A. Hayatbakhsh Novel, Rapid and Sensitive Detection of Isomeric Fluorinated New Psychoactive Substance (s) and Their Principal Metabolites Using Analytical Techniques, Master thesis, Manchester Metropolitan University, 2018.
- Policía Nacional de Colombia—Dirección de Antinarcóticos, Nuevas Sustancias Psicoactivas y Drogas Emergentes, Centro Internacional de Estudios Estratégicos contra el Narcotráfico (CIENA), 2022, available online: https://www.policia.gov.co/sites/default/files/nuevas_sustancias_psicoactivas_0.pdf, accessed on 12 March 2026 Search PubMed.
- United Nations Office on Drugs and Crime (UNODC), Rapid Testing Methods of Drugs of Abuse, United Nations, New York, NY, USA, 2006, available online: https://www.unodc.org/documents/scientific/Rapid_Testing_Methods_of_Drugs_of_Abuse_S.pdf, accessed on 12 March 2026 Search PubMed.
- A. Shafi, A. Berry, H. Sumnall, D. Wood and D. Tracy, New Psychoactive Substances: A Review and Updates, Ther. Adv. Psychopharmacol., 2020, 10, 1–21, DOI:10.1177/2045125320967197.
- World Health Organization (WHO), Fortalecimiento de la Capacidad en Materia de Medios de Diagnóstico, 76a Asamblea Mundial de la Salud, available online: https://apps.who.int/gb/ebwha/pdf_files/WHA76/A76_R5-sp.pdf, accessed on 12 March 2026 Search PubMed.
- M. Walker, A Proportionality Approach to the Ethics of Drug Policy, International Journal of Drug Policy, 2025, 142, 104861, DOI:10.1016/j.drugpo.2025.104861.
| This journal is © The Royal Society of Chemistry 2026 |