Ultrathin plasmonic PtCu nanosheets as an efficient peroxidase-like nanozyme for colorimetric detection of H2O2
Zhi
Cao
a,
Fei
Mu
b,
Qianshi
Shang
b,
Yi
Chen
b,
Peiyao
Zhang
b,
Feiyu
Chen
b,
Chuang
Shen
b,
Faisal
Saleem
*c,
Zhimin
Luo
 *b and
Ying
Zhang
*b
*b and
Ying
Zhang
*b
aSchool of Food and Bioengineering, Fujian Polytechnic Normal University, Fujian Universities and Colleges Engineering Research Center of Modern Facility Agriculture, Fuqing 350300, China
bState Key Laboratory of Flexible Electronics (LoFE), Jiangsu Key Laboratory of Smart Biomaterials and Theranostic Technology, Institute of Advanced Materials (IAM), College of Electronic and Optical Engineering & College of Flexible Electronics (Future Technology), School of Chemistry and Life Sciences, Nanjing University of Posts and Telecommunications, 9 Wenyuan Road, Nanjing 210023, China. E-mail: iamzmluo@njupt.edu.cn; yingzhang@njupt.edu.cn
cKey Laboratory of Flexible Electronics (KLOFE), Institute of Advanced Materials (IAM), Nanjing Tech University (NanjingTech), 30 South Puzhu Road, Nanjing 211816, China. E-mail: iamfaisalsaleem@njtech.edu.cn
First published on 3rd December 2025
Abstract
Bimetallic PtCu nanosheets were synthesized as a nanozyme with remarkable peroxidase-like catalytic activity for colorimetric detection of H2O2 in milk. In an analytical application, they demonstrated a wide linear range (0.001–5 mM) and a low limit of detection (0.58 µM), as well as a good anti-interference performance.
Hydrogen peroxide (H2O2) is a strong oxidant and is used in the food industry as a bleaching agent and disinfectant.1 It is often utilized as a preservative to prevent spoilage in foods such as milk, cheeses, and fresh-cut fruits.2 H2O2 as an additive can extend the shelf life of foods by reducing the entry of pathogens and killing harmful microorganisms.3 However, excessive levels of residual H2O2 in food can cause an imbalance in the cellular redox environment of human body, which in turn can lead to gastric disorders, cardiovascular diseases, depression, diabetes, Alzheimer's disease, and even cancers.4
The food and drug administration (FDA) of the United States stipulates that the maximum residual limit of H2O2 in finished packaged food is 0.5 ppm.5 The maximum level of H2O2 allowed in food according to the Chinese National Standard (GB5009.226-2016) is 3 mg kg−1.6 Unregulated uses of H2O2 in the food industry always result in substantial food safety problems. Therefore, the development of a cost-effective tool to sensitively, selectively, and rapidly detect H2O2 levels in food is urgently needed.
Nanozyme-enabled H2O2 biosensors possess the significant advantages of peroxidase-like nanozymes, such as high surface-to-volume ratio, catalytic stability, low production cost, and performance tunability.7–9 Peroxidase-like nanozymes can catalyze the H2O2 oxidation of 3,3′,5,5′-tetramethylbenzidine (TMB) into a typical blue product for colorimetric detection of H2O2.10–12 Various nanomaterials have been explored for the construction of H2O2 biosensors, such as metal,13 metal oxide,14 metal sulfide,15 carbon nanomaterial,16 and metal organic framework.17
Noble monometallic nanomaterials have been reported as promising alternatives of natural enzymes to fabricate colorimetric biosensors for the detection of H2O2.4,18–20 However, noble monometallic nanomaterials demonstrate relatively low peroxidase-like activity. An alloy strategy can create abundant active sites for catalytic reactions due to the surface and lattice strain in the alloy nanostructures.13,21–26 Compared with single noble-metal nanostructures, noble-metal alloy nanostructures exhibit enhanced peroxidase-like activity because of their coordination effect, and therefore, they are promising as a peroxidase mimic for constructing highly sensitive nanozyme-enabled H2O2 biosensors (Scheme 1).13,20,21,25
 | ||
| Scheme 1 Solvothermal synthesis of ultrathin PtCu nanosheets as a peroxidase-like nanozyme to construct a colorimetric biosensor for the detection of H2O2 in milk. | ||
In this work, two-dimensional PtCu nanosheets were prepared by a solvothermal method with copper(II) acetylacetonate and platinum(II) acetylacetonate as precursors. PtCu nanosheets can rapidly catalyze the H2O2 oxidation of TMB with a Michaelis constant (Km) and maximum reaction rate (Vmax) of 0.44 mM and 10.21 µM min−1, respectively. Based on the intrinsic peroxidase-like activity of PtCu nanosheets, a visual colorimetric biosensor for sensitive and accurate detection of H2O2 was constructed, which was able to function in the high-performance analysis of H2O2 in milk, indicating that PtCu nanosheets can be a promising nanozyme to fabricate a prospective colorimetric biosensor for the detection of H2O2 in food.
As shown from the transmission electron microscopy (TEM) images in Fig. 1a and b and their corresponding lateral size distribution in Fig. S1, our prepared PtCu nanosheets display a lateral size distribution of approximately 8 nm, with two-dimensional morphology. The high-resolution TEM (HRTEM) images in Fig. 1c and d show crystal lattices with an interplanar spacing of 0.219 nm, which is ascribed to the (111) plane of the PtCu alloy nanostructure.27 The energy-dispersive X-ray spectroscopy (EDS) elemental mapping images in Fig. 1e–h confirm the compositions of Pt and Cu in the product. The X-ray diffraction (XRD) spectrum in Fig. 2a shows typical diffraction peaks of 40.5 °C and 47.8 °C, which correspond to the (111) and (200) planes of the PtCu alloy nanostructure, respectively.27,28
 | ||
| Fig. 1 (a and b) TEM, (c and d) HRTEM, and (e–h) corresponding EDS mapping images of PtCu nanosheets. | ||
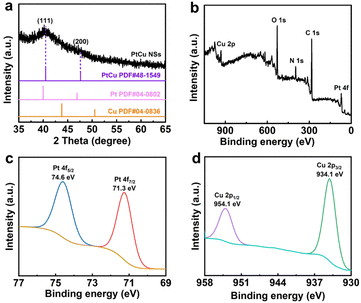 | ||
| Fig. 2 (a) XRD and (b) survey XPS spectra of PtCu nanosheets. High-resolution XPS spectra of (c) Pt 4f and (d) Cu 2p in PtCu nanosheets. | ||
The survey X-ray photoelectron spectroscopy (XPS) spectrum in Fig. 2b presents binding energy peaks of Pt 4f and Cu 2p, confirming the elemental compositions of the formed alloy nanostructure. The high-resolution XPS spectra of Pt 4f (Fig. 2c) and Cu 2p (Fig. 2d) show the binding energies of Pt 4f7/2 (70.7 eV) and Pt 4f5/2 (74.1 eV) of zero-valent Pt, and Cu 2p3/2 (931.8 eV) and Cu 2p1/2 (951.3 eV) of zero-valent Cu, further proving the formation of PtCu alloy nanosheets.29
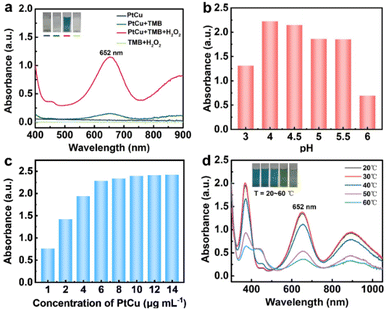
To investigate the intrinsic peroxidase-like activity of the PtCu nanosheets, experiments were performed through the catalytic oxidation reaction of TMB in the presence of H2O2 and PtCu nanosheets. As shown in Fig. 3a, a characteristic absorption peak ascribed to oxidized TMB appears at 652 nm in the reaction system containing TMB, H2O2, and PtCu nanosheets. The blue color of the reaction system was quickly observed after PtCu nanosheets were added to a solution containing TMB and H2O2. However, the reaction systems of PtCu nanosheets, PtCu nanosheets + TMB, and TMB + H2O2 showed no evident absorption at 652 nm with color changes. This indicates that the PtCu nanosheets can catalyze the oxidation of TMB in the presence of H2O2, confirming the peroxidase-like catalytic activity of PtCu nanosheets.
The influences of pH values, concentrations of PtCu nanosheets, and reaction temperatures on the peroxidase-like catalytic reaction of PtCu nanosheets were further explored. As shown in Fig. 3b–d, the catalytic activity of PtCu nanosheets increases with the rise of pH values from 3 to 4, and decreases after the pH value is greater than 4.0. The catalytic reaction rate increases with increasing concentration of PtCu nanosheets, and reaches the maximum after the concentration of PtCu nanosheets is greater than 10 µg mL−1. The catalytic activity of PtCu nanosheets increases as the temperature rises from 20 to 30 °C and is inhibited when the incubation temperature exceeds 30 °C. The experimental results in Fig. 3b–d confirm that the optimum conditions of the catalytic reaction are the pH value of 4.0, 10 µg mL−1 PtCu nanosheets, and a reaction temperature of 30 °C.
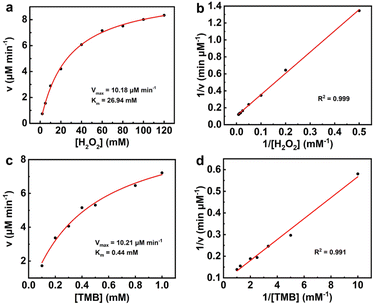
The steady-state kinetics of the peroxidase-like catalytic reaction were studied by changing the concentration of H2O2 or TMB while keeping the other constant. The experimental results were measured by a UV-Vis spectrophotometer at selected time intervals to obtain the typical Michaelis–Menten curves for H2O2 or TMB. Fig. 4 shows that the peroxidase-like catalytic reaction of PtCu nanosheets towards H2O2 and TMB follows the Michaelis–Menten mechanism.
As shown in Fig. 4a and b, the Km and Vmax for H2O2 substrate were calculated to be 26.94 mM and 10.18 µM min−1, respectively, according to the Lineweaver–Burk plot. Fig. 4c and d demonstrate that the Km and Vmax for TMB substrate are 0.44 mM and 10.21 µM min−1, respectively, which are comparable to those of natural horseradish peroxidase (HRP). Moreover, PtCu nanosheets present a lower Km value and higher Vmax value than other reported nanozymes in Table S1,20,21,23,26,30–33 suggesting the increased affinity and peroxidase-like catalytic activity of PtCu nanosheets. It is indicated that PtCu nanosheets with excellent peroxidase-like activity can serve as a promising alternative to HRP in the biosensing field.
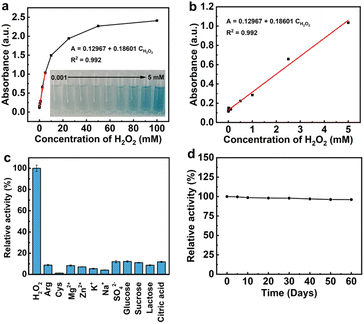
A colorimetric biosensor based on the peroxidase-like activity of PtCu nanosheets was constructed to detect H2O2. Fig. 5a and b show that the absorbance of oxidized TMB at 652 nm is linear with H2O2 concentrations from 0.001 to 5 mM. The limit of detection (LOD) of the PtCu nanosheet-enabled colorimetric biosensor was calculated to be 0.58 µM according to the formula of LOD = 3s/k, where s and k denote the relative standard deviation of parallel controlled measurements and the slope of the linear calibration plots, respectively. In this formula, s = 0.34 × 10−4, k = 1.8601 × 10−4, and LOD = 3s/k = 0.58 µM. Compared with the biosensors based on other nanozymes such as Fe3O4-Ag,34 Pt-Pd/MoS2,25 F-MoS2-FePt NCs,24 NiV2O6,33 PAnFc/Pt foil,35 PAA(DS)SPCE,14 and H@M,36 there is a wider linear range and lower LOD for the PtCu nanosheet-enabled colorimetric biosensor (Table S2).
To investigate the selectivity of the PtCu nanosheet-enabled colorimetric biosensor for H2O2 detection, arginine, cysteine, Mg2+, Zn2+, K+, Na+, SO42−, glucose, sucrose, lactose, and citric acid were used as controls for H2O2 determination experiments. As shown in Fig. 5c, the impact level of all interfering agents against H2O2 detection is less than 14%, suggesting that there is high selectivity of the PtCu nanosheet-enabled colorimetric biosensor for H2O2 detection.
The stability of our constructed colorimetric biosensor was measured by detecting its peroxidase-like catalytic activity after different storage times (5, 10, 20, 30, 40, 50, and 60 days). The experimental results in Fig. 5d show that the peroxidase-like catalytic activity of the PtCu nanosheet-enabled colorimetric biosensor remains at 95% over 60 days. All the above analytical results demonstrate that PtCu nanosheets with remarkable peroxidase-like catalytic activity can offer a sensitive, selective, and convenient colorimetric sensing strategy for H2O2 detection.
It is well known that H2O2 is often used as a stabilizer or preservative agent in commercial milk. Because residual H2O2 in milk may be harmful to human health, it is of significance to accurately detect H2O2 in milk. Encouraged by the satisfactory analytical performance of the PtCu nanosheet-enabled colorimetric biosensor, we used our proposed biosensor to determine the content of H2O2 in sour milk. Experiments were carried out by adding different concentrations of H2O2 to the pretreated milk sample, and then, the mixed sample was added to the reaction system containing the PtCu nanosheets and TMB.
As shown in Table 1, the content of H2O2 in 20-fold diluted sour milk is determined to be 17.128 µM according to the linear equation, and the concentration of H2O2 in the sour milk is calculated to be 342.56 µM, which is nearly that of the measured result (340.98 µM) by the iodometric standard method,37 indicating the accuracy of the PtCu nanosheet-enabled colorimetric biosensor. The recovery for detecting H2O2 in three milk samples ranges from 96.8% to 103.0%, with a relative standard deviation (RSD) of 3.25%, which indicates that our constructed biosensor is promising for detecting H2O2 in real samples.
| Added H2O2 (µM) | Found H2O2 (µM) | RSD (%) | Recovery (%) |
|---|---|---|---|
| 0 | <1 | 0 | 0 |
| 5 | 4.838 | 5.6 | 96.8 |
| 10 | 10.304 | 6.5 | 103.0 |
| 20 | 19.641 | 3.1 | 98.2 |
| Sour milk diluted 20-fold | 17.128 | — | — |
In summary, PtCu nanosheets are prepared as a peroxidase-like mimic to construct a sensitive and convenient colorimetric biosensor for the detection of H2O2. The experimental results show that PtCu nanosheets exhibit remarkable peroxidase-like catalytic activity and can efficiently catalyze the oxidation reaction of TMB in the presence of H2O2. The PtCu nanosheet-enabled colorimetric biosensor demonstrates a satisfactory analytical performance with a wide linear range (0.001–5 mM) and a low limit of detection (0.58 µM), as well as a good anti-interference performance, which can be applied to accurately detect H2O2 in milk samples. Our work facilitates the practical applications of PtCu nanosheets in food additive detection.
Author contributions
Z. Cao, F. Saleem, Z. Luo, and Y. Zhang conceived the project, designed the studies, and analyzed the results. Z. Cao, F. Mu, Q. Shang, Y. Chen, P. Zhang, F. Chen, and C. Shen carried out the experiments. Z. Cao, Z. Luo, and Y. Zhang wrote the manuscript. F. Saleem, Z. Luo, and Y. Zhang supervised and revised the project.Conflicts of interest
There are no conflicts to declare.Data availability
All relevant data are available from the corresponding author upon reasonable request.Supplementary information (SI): materials, methods, characterizations. See DOI: https://doi.org/10.1039/d5ay01909e.
Acknowledgements
We acknowledge the National Natural Science Foundation of China (22275096, 62288102, 52002396, 22473060), the Natural Science Foundation of Jiangsu Province-Major Project (BK20212012), the Natural Science Key Fund for Universities in Jiangsu Province (22KJA430007), the Key Research and Development Program of Jiangsu (BE2018732), the China Postdoctoral Science Foundation (2022M711691), and the General Project of Natural Science Research in Colleges and Universities of Jiangsu Province (22KJB350001).Notes and references
- Y. Zhang, G. Ma, L. Li, X. Li, M. Wang, R. Liu and J. Qu, Food Chem., 2025, 489, 144998 CrossRef CAS PubMed.
- M. E. Abbas, W. Luo, L. Zhu, J. Zou and H. Tang, Food Chem., 2010, 120, 327–331 CrossRef CAS.
- S. Aliakbarpour, M. Amjadi and T. Hallaj, Food Chem., 2024, 432, 137273 CrossRef CAS PubMed.
- D. Li, R. Tian, S. Kang, X. Chu, D. Ge and X. Chen, Food Chem., 2022, 393, 133386 CrossRef CAS PubMed.
- Y. Li, S. Wu, H. Lu and S. Xu, Food Chem., 2025, 469, 142577 CrossRef CAS PubMed.
- Y. Zhang, H. Zhang, C. Ma, Y. Yang, X. Mo, Y. Shi, Z. Yin, H. Li and L. Sun, Food Chem., 2025, 492, 145351 CrossRef CAS PubMed.
- R. Zhang, X. Yan and K. Fan, Acc. Mater. Res., 2021, 2, 534–547 CrossRef CAS.
- Q. Wang, H. Wei, Z. Zhang, E. Wang and S. Dong, Trac-trend in Anal. Chem., 2018, 105, 218–224 CrossRef CAS.
- X. Zhang, D. Zhu, X. Yang, C. Man, Y. Jiang, Q. Zhao and X. Zhang, Trends Food Sci. Tech., 2024, 148, 104486 CrossRef CAS.
- J. Guo, Z. Yang, X. Liu, L. Zhang, W. Guo, J. Zhang and L. Ding, Rare Metals, 2023, 42, 797–805 CrossRef CAS.
- S. Li, F. Zhao, H. Yu, Z. Xu, Z. Ali, W. Li, Y. Ying, L. Qiao, J. Zheng, J. Li, S. Che and J. Yu, Rare Metals, 2025, 44, 6375–6387 CrossRef CAS.
- Q. Yang, Y. Mao, Q. Liu and W. He, Rare Metals, 2023, 42, 2928–2948 CrossRef CAS.
- Y. Ding, B. Yang, H. Liu, Z. Liu, X. Zhang, X. Zheng and Q. Liu, Sensor. Actuat. B-Chem., 2018, 259, 775–783 CrossRef CAS.
- Y. Ma, Z. Zhang, C. Ren, G. Liu and X. Chen, Analyst, 2012, 137, 485–489 RSC.
- W. Zhang, X. Li, T. Cui, S. Li, Y. Qian, Y. Yue, W. Zhong, B. Xu and W. Yue, Microchim. Acta, 2021, 188, 174 CrossRef CAS PubMed.
- X. Wang, L. Qin, M. Lin, H. Xing and H. Wei, Anal. Chem., 2019, 91, 10648–10656 CrossRef CAS PubMed.
- J. Chen, Y. Shu, H. Li, Q. Xu and X. Hu, Talanta, 2018, 189, 254–261 CrossRef CAS PubMed.
- X. Liu, Z. Ding, C. Xu, J. Zhang, Y. Liu, T. Chen, S. Dai, X. Bao, M. Hu and Z. Liu, Adv. Sci., 2025, 12, e03237 CrossRef CAS.
- Y. Fan, X. Gan, H. Zhao, Z. Zeng, W. You and X. Quan, Chem. Eng. J., 2022, 427, 131572 CrossRef CAS.
- B. Xu, M. Cui, Z. Luo and L. Wang, Chinese Chem. Lett., 2025, 487, 111227 CrossRef.
- Z. Xiao, J. Cao, J. Liu, Z. Song, T. Du and X. Du, Adv. Sci., 2025, 10, e12875 CrossRef PubMed.
- T. Zhang, Y. Xing, Y. Song, Y. Gu, X. Yan, N. Lu, H. Liu, Z. Xu, H. Xu, Z. Zhang and M. Yang, Anal. Chem., 2019, 91, 10589–10595 CrossRef CAS PubMed.
- Z. Li, B. Ding, J. Li, H. Chen, J. Zhang, J. Tan, X. Ma, D. Han, P. Ma and J. Lin, Angew. Chem., Int. Ed., 2024, 64, e202413661 CrossRef PubMed.
- Z. Hu, Z. Dai, X. Hu, B. Yang, Q. Liu, C. Gao, X. Zheng and Y. Yu, J. Nanobiotechnol., 2019, 17, 38 CrossRef PubMed.
- R. Sha, N. Vishnu and S. Badhulika, Microchim. Acta, 2018, 185, 399 CrossRef PubMed.
- X. Tong, G. Li, Q. Guo, J. Hu, B. Zhang, S. Liu, J. Guo and L. Zhang, Microchim. Acta, 2024, 192, 20 CrossRef.
- F. Saleem, Z. Zhang, B. Xu, X. Xu, P. He and X. Wang, J. Am. Chem. Soc., 2013, 135, 18304–18307 CrossRef CAS PubMed.
- Z. Zhang, Z. Luo, B. Chen, C. Wei, J. Zhao, J. Chen, X. Zhang, Z. Lai, Z. Fan and C. Tan, Adv. Mater., 2016, 28, 8712–8717 CrossRef CAS PubMed.
- Y. Zhang, C. Shen, J. Zhang, Q. Shen, F. Xu, S. Wang, J. Hu, F. Saleem, F. Huang and Z. Luo, Chinese Chem. Lett., 2025, 36, 111059 CrossRef CAS.
- W. Xu, L. Jiao, H. Yan, Y. Wu, L. Chen, W. Gu, D. Du, Y. Lin and C. Zhu, ACS Appl. Mater. Interfaces, 2019, 11, 22096–22101 CrossRef CAS PubMed.
- L. Su, W. Qin, H. Zhang, Z. Rahman, C. Ren, S. Ma and X. Chen, Biosens. Bioelectron., 2015, 63, 384–391 CrossRef CAS PubMed.
- L. Gao, J. Zhuang, L. Nie, J. Zhang, Y. Zhang, N. Gu, T. Wang, J. Feng, D. Yang, S. Perrett and X. Yan, Nat. Nanotechnol., 2007, 2, 577–583 CrossRef CAS PubMed.
- Q. Zhu, J. Yang, Z. Peng, Z. He, W. Chen, H. Tang and Y. Li, Talanta, 2021, 234, 122645 CrossRef CAS.
- Z. Liu, B. Zhao, Y. Shi, C. Guo, H. Yang and Z. Li, Talanta, 2010, 81, 1650–1654 CrossRef CAS PubMed.
- Y. Yang and S. Mu, Biosens. Bioelectron., 2005, 21, 74–78 CrossRef CAS PubMed.
- F. Qin, S. Jia, F. Wang, S. Wu, J. Song and Y. Liu, Catal. Sci. Technol., 2013, 3, 2761–2768 RSC.
- R. J. Kieber and G. R. Helz, Anal. Chem., 1986, 58, 2312–2315 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |