Study of the long-term stability conditions of cyanide ion standard solutions
Tomohiro
Narukawa
 *,
Ayaka
Wada
and
Chikako
Cheong
*,
Ayaka
Wada
and
Chikako
Cheong

National Metrology Institute of Japan, National Institute of Advanced Industrial Science and Technology, 1–1–1 Umezono, Tsukuba, Ibaraki 305–8563, Japan. E-mail: tomohiro-narukawa@aist.go.jp
First published on 15th December 2025
Abstract
Cyanide ions (CN−) are highly toxic and pose significant environmental and health risks. This study quantitatively evaluates the temperature-dependent decomposition kinetics of cyanide ion standard solutions under long-term storage conditions and explores implications for analytical calibration. The decomposition rate constants were determined at temperatures ranging from −18 °C to 25 °C, revealing a strong correlation between temperature and degradation rate. Freezing (−18 °C) maintained stability for approximately one year (rate change: −0.0005%), whereas at 25 °C, daily degradation reached −0.1032%. A predictive equation for estimating original cyanide mass fractions based on storage time and temperature was developed (r2 > 0.999). And, this is the first time that it has been demonstrated that a cyanide ion standard solution is stable for more than one year when stored in a refrigerator. Additionally, effects of mass fraction and pH conditions of the cyanide ion standard solution were assessed. These findings provide guidance for preparing and storing traceable cyanide solutions and suggest future research on other toxic anions such as thiocyanate and cyanate. Additionally, preliminary tests on environmental water matrices were conducted.
1 Introduction
Cyanide is extremely toxic and has severe effects on human health. The maximum contaminant level for cyanide set by the United States Environmental Protection Agency in drinking water is 200 µg L−1.1 The European Union has set an even lower limit of 50 µg L−1.2 Codex Alimentarius states that the cyanide content of mineral water should not exceed 0.07 mg L−1, which is the same as the drinking water limit set by the World Health Organization.3 However, cyanide continues to be used in various industries, including electroplating, metallurgy, organic chemical and plastic manufacturing, analytical reagents (e.g., masking agents), and insect specimens. In some parts of Asia, high-concentration sodium cyanide (NaCN) solutions are used in fishing, raising concerns about environmental pollution.Cyanide is produced by certain bacteria, fungi, and algae, and some foods and plants contain cyanide. For example, apple seeds4,5 and almonds6 contain small amounts of cyanide. Cyano groups exist as cyanogenic glycosides bound to carbohydrate molecules in plants, and the roots of cassava, a staple food in tropical regions, contain cyanogenic glycosides.7
Although cyanide ions are toxic, they are found in the environment and are also used industrially. Recently, they have also attracted attention in the field of biology, such as the enzyme's critical function in cyanide detoxification and tracking of intracellular levels employing biocompatible branching molecular architectures.8,9
According to Japan's Water Pollution Control Law, the maximum concentrations of cyanide in wastewater and tap water should be 1 mg L−1 or less and 0.01 mg L−1 or less, respectively. Furthermore, the soil elution criteria require that no cyanide be detected in groundwater or test solutions.10–12
Methods for measuring cyanide concentrations include titration,13,14 flow injection,15 ion chromatography,16 colorimetry,17–19 and capillary electrophoresis.4,20 Ion chromatography with post column-adsorption photometry allows for the simultaneous measurement of cyanide ions and cyanogen chloride.21 The technique allows the measurement of cyanide ions at the 1 µg L−1 level with a relative standard deviation (RSD) of approximately 5% for repeated analyses.22 Also, the determination techniques of atomic absorption spectrometry, electrochemical methods, mass spectrometry, and gas chromatography had been reported.23 In addition, the multi-functional captor/sensor microstructures was developed for a sensitive detection and selective removal approaches for ultra-trace concentrations of cyanide ion.24
However, even if an analytical method is useful, the most important factor in validating it is the standard solution used for quantification. However, metrological traceability only guarantees the quality of cyanide ion standard solutions for around two months. Therefore, the stability of cyanide ions has been investigated for some time.
The stability of cyanide ions varies significantly depending on environmental and chemical conditions. The decomposition rate of cyanide ions (CN−) is relatively fast under acidic conditions. For example, cyanide ions transform into hydrogen cyanide (HCN) in sulfuric and acidic solutions. In contrast, the decomposition rate of cyanide ions is slow under alkaline conditions and relatively stable under higher pH conditions. The decomposition rate of cyanide ions depends on temperature; the decomposition rate of cyanide ions increases with increasing temperature.
Suzuki et al. used a cyanide ion standard solution to investigate the storage stability of cyanide ions at different potassium hydroxide concentrations and storage temperatures (8 °C and 25 °C). According to the results, a higher potassium hydroxide concentration was more stable at the same temperature, and the cyanide mass fraction changed by approximately 10 times more when stored at 25 °C than at 8 °C.25 Shikakume et al. investigated the relationship between cyanide ion concentration and storage stability at 5 °C to 40 °C and reported that the cyanide ion concentration decreases as cyanide decomposes into ammonia and formic acid.26
The aim of this study was to establish stability conditions for long-term storage of cyanide ions. Therefore, we investigated the changes in mass fraction of cyanide ion standard solutions with respect to storage temperature and time and demonstrated that long-term storage is possible through frozen storage. Also, it was discussed the temperature-dependent decomposition rate of cyanide ions.
2 Experimental
2.1. Instrumentation
An automatic titrator (AT-420win, Kyoto Electronics Manufacturing Co., Ltd, Kyoto, Japan) was used for photometric titration. The piston burette was calibrated using the discharged mass of pure water, and a 562 nm interference filter was used. An automatic density meter (DMA™ 4100M, Anton Paar GmbH, Graz, Austria) was used to measure the solution density.Ion chromatography with post column-adsorption photometry (hereafter, the IC-post column method) was performed using a Prominence (Shimadzu Co., Kyoto, Japan).21 A Shim-pack Amino-Na column (filler particle size 5 µm, ID 6.0 mm × 100 mm, Shimadzu) was used with a mobile phase containing 10 mM sodium tartrate (KANTO Chemical Co., Inc., Tokyo, Japan) at a flow rate of 0.6 mL min−1. N,N-Dimethylformamide (FUJIFILM Wako Pure Chemical Co., Osaka, Japan) containing 28.7 mM 3-methyl-1-phenyl-5-pyrazolone (FUJIFILM Wako) + 96.5 mM sodium 4-pyridinecarboxylate (FUJIFILM Wako) were added online for the color reaction. The measurements were then performed at 638 nm (ultraviolet-visible). The detection limits (3σ) of cyanide and cyanogen chloride ions under the conditions were calculated based on their peak areas of 200 µL injection, and the detection limits were 0.03 and 0.05 ng g−1, respectively.
A Seven compact pH/Ion S220 meter (Mettler Toledo AG, Schwerzenbach, Switzerland) was used for the pH measurement.
2.2. Standards and reagents
NMIJ CRM 3809 Cyanide Ion Standard Solution CN− (1000) (ca. 1000 mg kg−1) was used as the calibrant for the titration process. The Japan calibration service system (JCSS) standard solutions of cyanide ion standard solution (ca. 1000 mg L−1, Kanto) were employed as calibration standards for the IC-post column method. The cyanide ion solution was prepared by weighing out approximately 2.5 g of high-purity potassium cyanide (purity 98%<, FUJIFILM Wako) and dissolving it in approximately 1 kg of 0.5 mol L−1 potassium hydroxide (KOH, FUJIFILM Wako).The nickel standard titration solution (0.02 mol L−1) was prepared as follows. Approximately 1 g of high-purity nickel metal (Sumitomo Metal Mining Co., Ltd, Tokyo, Japan, purity 99.99%<) was washed with 3 mol L−1 hydrochloric acid, pure water, and ethanol and dried at 55 °C for 30 min. After cooling in a silica gel desiccator for 30 min, the sample was weighed and dissolved by heating 30 mL of 6.6 mol L−1 HNO3. After cooling, the sample was neutralized with 0.2 mol L−1 ammonia solution and diluted to 1 kg with water.
The other reagents used in the experiments were of ultrapure grade (FUJIFILM Wako). Ultrapure water used throughout the experiments was generated using a Milli-Q Labo filter (Nippon Millipore Ltd, Tokyo, Japan).
2.3. Material
Well water, river water, and spring water (Ibaraki Prefecture, Japan) were collected as environmental water samples. The water samples were filtered through a 0.45 µm membrane filter and collected in high-density polyethylene containers. The containers were transported in a dry-ice box. The transported containers (∼2 h) were immediately transferred to a freezer (−18 °C ± 2 °C) and stored frozen.2.4. Procedure
A gravimetric preparation method was used to prepare all samples in this study for SI traceability.Approximately 100 mL of a cyanide ion solution with a mass fraction of 1000 mg kg−1 was placed in an aluminium laminate bag in a high-density polyethylene container, capped, and sealed. Some samples were stored in a freezer (−18 °C ± 2 °C), a refrigerator (4.1 °C ± 1.2 °C, 5.7 °C ± 1.8 °C, and 7.3 °C ± 1.2 °C), and at room temperature (25 °C ± 2 °C). Each solution was periodically removed, held at room temperature (25 °C ± 2 °C), and then immediately quantified via titration using the complex formation reaction between nickel and cyanide ions.
In addition, we measured both diluted cyanide ion solutions and environmental water samples (well water, river water, and spring water) using the IC-post column method.21 These samples were analyzed either directly or after approximately 10 ng g−1 of the cyanide ion solution was added. Furthermore, the well water sample containing approximately 10 ng g−1 of the cyanide ion solution was stored at −18 °C ± 2 °C for 5 days and then measured using the IC-post column method based on the JCSS cyanide ion standard solution.
3 Results and discussion
3.1. Influence of storage temperature of cyanide ions
The prepared cyanide ions were stored for a long period (maximum storage time: 1 year) at −18 °C ± 2 °C, 4.1 °C ± 1.2 °C, 5.7 °C ± 1.8 °C, 7.3 °C ± 1.2 °C, and 25 °C ± 2 °C. The sample solutions were periodically removed and allowed to reach equilibrium with room temperature. Approximately 20 g of each solution was weighed and mixed with 60 g of pure water, 10 mL of 0.02 mol L−1 ammonium chloride (NH4Cl)–12.5% ammonium hydroxide (NH3H2O), and 0.1 g of 0.2% composite preparation of ammonium purpurate and potassium sulfate (MX) powder. The cyanide ion mass fraction was determined within 8 h via titration using nickel as the titrant. Titration method is the primary standard measurement method. The RSD of the repeated titration analysis (n = 3) in this experiment was less than 0.02%.The results are presented in Fig. 1. The mass fraction of cyanide ions decreased with increasing storage time. Furthermore, the rate of decrease increased with increasing storage temperature. The rate of decrease in the cyanide ion mass fraction at each storage temperature was converted to a daily rate at −0.0021% at 4.1 °C ± 1.2 °C, −0.0028% at 5.7 °C ± 1.8 °C, −0.0044% at 7.3 °C ± 1.2 °C, and −0.1032% at 25 °C ± 2 °C. These values are almost consistent with previous results reported.
In contrast, when frozen at −18 °C ± 2 °C, which was examined for the first time in this experiment, the change in the mass fraction of cyanide ions after storage for approximately 1 year was −0.0005%, and the daily decrease rate of the cyanide ion mass fraction was −0.000001%. The rate of change over 1 year (−0.0005%) is within the range of measurement uncertainty. In other words, no change was observed. This result shows for the first time that frozen storage suppresses cyanide ion decomposition and allows for stable storage.
3.2. Decomposition rate of cyanide ions
Based on the results presented in Fig. 1, the dependence of the decomposition rate of cyanide ions on storage temperature is shown in Fig. 2 and 3. | ||
| Fig. 2 Decomposition rate of cyanide ions at different storage temperatures. Temperature range: solid line indicates 0–25 °C; the dotted line indicates 4–25 °C. | ||
Fig. 2 shows the rate of change in the mass fraction due to the decomposition rate (v), based on the change in the mass fraction of cyanide ions at each storage temperature and the number of storage days. Note that at a storage temperature of −18 °C ± 2 °C, even if the actual storage temperature is −18 °C ± 2 °C, the solution freezes from less than 0 °C, indicating no difference in temperature conditions below freezing.
Therefore, the temperature dependence was confirmed at temperatures of 4.1 °C ± 1.2 °C, 5.7 °C ± 1.8 °C, 7.3 °C ± 1.2 °C, and 25 °C ± 2 °C. Consequently, a quadratic equation holds between the mass fraction change rate and storage temperature (°C), and the change rate at storage temperature (°C) is expressed by the quadratic function of eqn (1-1), with a correlation coefficient (r2) of 1.0000. Furthermore, when the storage temperature of −18 °C is set to the freezing temperature of the aqueous solution of 0 °C, the change rate at storage temperature is expressed by eqn (1-2), with a r2 value of 0.9993.
| y = −0.00018x2 + 0.00119x − 0.007151 | (1-1) |
| y = −0.00014x2 + 0.00019x − 0.00061 | (1-2) |
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) k) calculated using the logarithm of the decomposition rate (v) (Fig. 3).
k) calculated using the logarithm of the decomposition rate (v) (Fig. 3).
Given that Celsius (°C) is the more common temperature scale in daily use, the storage temperature is °C, the decomposition rate of cyanide ions per day at the storage temperature (expressed in °C) is calculated using eqn (2) for temperatures ranging from 4 °C to 25 °C.
| v(%/day) = e(0.1373×storage temperature (°C)−5.8093) | (3) |
These results demonstrate that the decomposition rate of cyanide ions is highly and strictly dependent on temperature and time. Therefore, using eqn (3), the mass fraction of the original cyanide ions was calculated from the decomposition rate of cyanide ions depending on the storage temperature (°C) and storage time (tstorage days, day) at storage temperatures ranging from 4 °C to 25 °C.
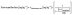 | (4) |
The prepared cyanide ion solution (initial mass fraction: 992.8) was used to measure the actual stored solution, and the initial mass fraction was calculated using eqn (3). The degree of concordance was approximately 100% (Table 1).
| Cyanide ion (mg kg−1)* | Storage temperature (°C) | Storage time (day) | Measurement result (mg kg−1, n = 3)** | Correction value (mg kg−1)*** | Degree of concordance (%)**** |
|---|---|---|---|---|---|
| a *Mass fraction of cyanide ion (preparation value); **titration method; ***calculation results using eqn (3); ****correction value/992.8 × 100. | |||||
| 992.8 | 4.1 | 121 | 986.1 | 992.5 | 100.0 |
| 992.8 | 5.7 | 121 | 984.0 | 991.9 | 99.9 |
| 992.8 | 7.3 | 90 | 985.4 | 992.8 | 100.0 |
| 992.8 | 25.0 | 90 | 909.8 | 992.7 | 100.0 |
From another perspective, a cyanide ion standard solution, whose mass fraction or concentration had been fixed at the production, the amount of cyanide ions at the time of use can be accurately calculated from the storage temperature and storage time. When the NMIJ 3809 Cyanide Ion Standard Solution (certified value: 993 mg kg−1 ± 11 mg kg−1, k = 2) was stored at 4 °C for 90 days, the mass fraction of cyanide ions after certification can predict to be 988.50 mg kg−1 at 4 °C. When actually measured 90 days later, the mass fraction of cyanide ions was 988.60 mg kg−1, and the degree of agreement was 100.01%. Furthermore, the result obtained was calculated by eqn (3), the initial mass fraction was calculated to be 993.31 mg kg−1, and the degree of agreement was 100.03%.
3.3. Effect of mass fraction of cyanide ions and pH conditions
Cyanide ion standard solutions are generally used for calibration curves in IC and other methods. The mass fraction of cyanide ions used in these methods is lower than that used in titration. Therefore, we evaluated the stability of the standard solutions at lower mass fractions using IC.The dependence of the mass fraction of cyanide ions and the influence of pH was examined. Solutions (50 g) containing cyanide ion mass fractions of 500 mg kg−1, 5 mg kg−1, and 0.5 mg kg−1 were prepared by diluting the cyanide ion solution with water and then were frozen for 60 days. Therefore, 500 mg kg−1, 5 mg kg−1, and 0.5 mg kg−1 cyanide ion solutions contain 0.25 mol L−1 KOH, 0.0026 mol L−1 KOH, and 0.00026 mol L−1 KOH, respectively. Furthermore, cyanide ion solutions with constant KOH concentrations of 0.5 mol L−1 were prepared.
Following the JCSS cyanide ion standard solution, the mass fraction of cyanide ions was determined as a calibration curve using the IC-post column method after diluting each solution to approximately 10 ng g−1 cyanide ion. The results are presented in Table 2.
| Cyanide ion (mg kg−1)* | Storage temperature (°C) | KOH (mol L−1) | pH | Storage time (day) | Measurement result (mg kg−1, n = 3) | Recovery (%)** |
|---|---|---|---|---|---|---|
| a *Prepared mass fraction; **measurement result/prepared mass fraction × 100. | ||||||
| 995.2 | −18 ± 2 | 0.50 | 13.54 | 60 | 995.1 ± 0.5 | 100.0 |
| 504.7 | −18 ± 2 | 0.50 | 13.54 | 60 | 508.4 ± 12.0 | 100.7 |
| 0.25 | 13.34 | 60 | 513.5 ± 20.7 | 101.8 | ||
| 5.234 | −18 ± 2 | 0.50 | 13.54 | 60 | 5.247 ± 0.237 | 100.2 |
| 0.0026 | 10.99 | 60 | 5.221 ± 0.292 | 99.7 | ||
| 0.5189 | −18 ± 2 | 0.50 | 13.54 | 60 | 0.5150 ± 0.0047 | 99.2 |
| 0.00026 | 9.35 | 60 | 0.4833 ± 0.0063 | 93.1 | ||
Although the decomposition rate depends on the element or ion, it changes depending on the mass fraction in many cases, and the lower the mass fraction or concentration, the greater the chemical change. At KOH concentrations above 0.0026 mol L−1 (pH 11), no mass fraction dependence of cyanide ions was observed even after 60 days of storage, meaning that no change was observed and a nearly 100% recovery rate was obtained. However, at a KOH concentration of 0.00026 mol L−1 (pH 9.35), the recovery rate dropped to approximately 93%.
This result shows that the stability of cyanide ions depends not only on temperature but also on the pH of the solution. In other words, if the solution is not sufficiently alkaline, it is difficult to maintain the stability of cyanide ions.
3.4. Determination of free cyanide ion in environmental waters
Although the titration method allows for precise measurements, the presence of coexisting substances can affect the chemical reactions and results. Also, measuring small amounts of cyanide ions is difficult. The actual environmental samples were analyzed using the IC-post column method, because environmental water contains only a trace amount of cyanide ions. The obtained chromatograms and the calibration curves of cyanide ion are shown in Fig. 4(a) and Fig. 5. The results obtained are shown in Table 3. According to the results, no cyanide or cyanogen chloride ions were detected in the well, river, or spring water samples used in the experiment.| Added (ng g−1) | Storage time (day) | Measurement result (ng g−1, n = 3) | pH | Recovery (%)a | |
|---|---|---|---|---|---|
| a Measurement result/added × 100. | |||||
| Well water | — | 0 | ND | 7.01 | — |
| 10.1027 | 0 | 10.520 ± 0.177 | 104.1 | ||
| 10.1027 | 5 | 10.030 ± 0.013 | 99.3 | ||
| River water | — | 0 | ND | 6.72 | — |
| 10.1246 | 0 | 10.290 ± 0.232 | 101.6 | ||
| 10.1246 | 5 | 10.538 ± 0.043 | 104.1 | ||
| Spring water | — | 0 | ND | 7.57 | — |
| 10.0866 | 0 | 9.871 ± 0.218 | 97.9 | ||
| 10.0866 | 5 | 10.239 ± 0.269 | 101.5 | ||
Therefore, an approximately 10 ng g−1 cyanide ion standard solution was added to well, river, and spring water samples. According to the results, the added cyanide ions were easily detected in the well, river, and spring water samples with a recovery rate of 101% ± 3%.
In addition, the water samples containing 10 ng g−1 cyanide ions were stored for 5 days at −18 °C ± 2 °C, after which the water samples were also investigated. According to the results presented in Fig. 4(b) and Table 3, although the pH levels of the samples were neutral, the recovery rates of cyanide ions were 102% ± 2%. Therefore, it was considered that changes in cyanide ions were suppressed for a short period without an alkaline solution by frozen storage. However, when these solutions were stored for 60 days by frozen storage, the recoveries of the cyanide ion were approximately 84%. This is thought to be due to the low alkalinity of the sample.
Therefore, the pH of the same environmental water was adjusted to approximately 11 using KOH and stored in a refrigerator for 60 days. As a result, the recovery rate was nearly 100%. The environmental water used in this study contained only a small amount of coexisting substances, and no precipitate was observed even when KOH was added. However, many metal elements form hydroxide precipitates in the strongly alkaline range. Therefore, samples with many coexisting substances are difficult to store and measure under alkaline conditions.
Therefore, freezing is effective for the stability of cyanide ions for actual samples, but it was clear that measurements must be made within about 5 days.
On the other hand, calibration curves were created for these actual samples using a cyanide ion standard solution prepared and used immediately, a cyanide ion standard solution stored in a refrigerator for one year, and a cyanide ion standard solution stored at 4.1 ± 1.2 °C for 250 days. As is clear from the above results, the slope of the calibration curve was lower only for the standard solution stored at 4.1 ± 1.2 °C, resulting in higher quantification results for the actual samples. However, by correcting for the mass fraction of the standard solution using eqn (3), the quantification result for the actual sample agreed with the other two results within a range of about ± 2%.
4 Conclusions
In this study, we investigated the storage stability of cyanide ions at temperatures between −18 °C and 25 °C that the maximum storage time period was 1 year. Although the temperature dependence of cyanide ion is known, this is the first time that their stability for one year when stored frozen has been demonstrated.The mass fraction of cyanide ions was measured rigorously via titration. According to the results, the decomposition rate of cyanide ions was strictly temperature dependent. In addition, a clear correlation was observed between the decomposition rate constant and storage temperature. The predictive equation developed enables accurate correction for storage-induced changes. This allows the mass fraction of the standard solution to be clearly calculated from the storage temperature and time, making it possible to apply this to ensuring traceability.
Considering the results of this study, anion standard solutions, such as a nitrite ion, which are particularly susceptible to instability depending on storage conditions, may be able to maintain their stability for longer by freezing them. Thus, these findings support best practices for preparing and storing traceable cyanide solutions etc. and open avenues for studying other toxic anions under variable environmental conditions.
Author contributions
T. Narukawa: conceptualization, methodology, validation, formal analysis, investigation, resources, data curation, writing – original draft, writing – review and editing. A. Wada: validation, formal analysis, investigation, resources, data curation, writing – review and editing. C. Cheong: writing – review and editing.Conflicts of interest
There are no conflicts declare.Data availability
All data are contained within the manuscript.Acknowledgements
This work had been supported by Dr Toshihiro Suzuki (ex- National Metrology Institute of Japan, National Institute of Advanced Industrial Science and Technology) and is thanked for his contributions.References
- EPA (United States Environmental Protection Agency), Methods for chemical analysis of water and wastes, Environmental Monitoring and Support Laboratory, Cincinnati, OH, 1983, https://www.epa.gov/ogwdw000/contaminants/basicinformation/cyanide.html Search PubMed.
- EU, Official Journal of the European Union, Commission Directive 1998/83/EC, 1998, pp. L30–42, https://eur-lex.europa.eu/LexUriServ/LexUriServ.do?uri=OJ:L:1998:330:0032:0054:EN:PDF Search PubMed.
- WHO (World Health Organization), Guidelines for Drinking-Water Quality, Geneva, 3rd edn, 2008, p. 188, https://www.who.int/watersanitationhealth/dwq/fulltext.pdf Search PubMed.
- D. B. Craig and M. S. Guimond, Food Chem., 2022, 370, 131377, DOI:10.1016/j.foodchem.2021.131377.
- U. R. Gandra, R. Lo, P. B. Managutti, A. M. Butt, P. S. Reddy, A. U. H. Qurashi, S. Mohamed and M. I. H. Mohideen, Analyst, 2025, 150, 489–497, 10.1039/d4an01265h.
- E. Doria, L. Lerno, M.-A. Chen, J. Lee, G. Huang and A. E. Mitchell, J. Agric. Food Chem., 2025, 73, 5500–5510, DOI:10.1021/acs.jafc.4c08437.
- N. D. Ndubuisi and A. C. U. Chidiebere, Int. J. Geom. Data Min., 2018, 2, 118, DOI:10.29011/2577-0616.000118.
- O. E. Agboola, Z. A. Ayinla, B. O. Itakorode, P. O. Akinsanya, R. E. Okonji, O. B. Odeghe, S. S. Agboola, O. E. Oluranti, F. O. Olojo and B. E. Oyinloye, Toxics, 2025, 13, 750, DOI:10.3390/toxics13090750.
- I. M. El-Sewify, M. A. Shenashen, R. F. El-Agamy, M. S. Selim, N. F. Alqahtani, A. Elmarakbi, M. Ebara, M. M. Selim, M. M. H. Khalil and S. A. El-Safty, J. Hazard. Mater., 2024, 465, 133271, DOI:10.1016/j.jhazmat.2023.133271.
- J. Vetter, Toxicon, 2000, 38, 11–36 CrossRef CAS PubMed.
- Ministry of Health, Labour and Welfare, Japan, https://www.mhlw.go.jp/index.html Search PubMed.
- Ministry of the Environment, Japan, https://www.env.go.jp.
- T. Suzuki, A. Hioki and M. Kurahashi, Anal. Chim. Acta, 2003, 476, 159–165, DOI:10.1016/S0003-2670(02)01362-4.
- P. L. Breuer, C. A. Sutcliffe and R. L. Meakin, Hydrometallurgy, 2011, 106, 135–140, DOI:10.1016/j.hydromet.2010.12.008.
- A. V. López Gómeza and J. M. Calatayud, Analyst, 1998, 123, 2103–2107, 10.1039/A803439G.
- T. T. Christison and J. S. Rohre, J. Chromatogr., A, 2007, 1155, 31–39, DOI:10.1016/j.chroma.2007.02.083.
- S. Bhardwaj and A. K. Singh, J. Hazard. Mater., 2015, 296, 54–60, DOI:10.1016/j.jhazmat.2015.04.043.
- I. J. Kim, M. Ramalingam and Y.-A. Son, Sens. Actuators, B, 2017, 246, 319–326, DOI:10.1016/j.snb.2017.02.015.
- P. Gholamzadeh, G. M. Ziarani, N. Lashgari, A. Badiei, A. Shayesteh and M. Jafari, J. Fluoresc., 2016, 26, 1857–1864, DOI:10.1007/s10895-016-1879-z.
- Q. Zhang, N. Maddukuri and M. Gong, J. Chromatogr., A, 2015, 1414, 158–162, DOI:10.1016/j.chroma.2015.08.050.
- Ministry of the Environment, Japan, notification No. 261, https://www.env.go.jp/content/900547779.pdf Search PubMed.
- Ministry of Health, Labour and Welfare, Japan, Ministry of Health, Labor and Welfare notification No. 261, https://www.mhlw.go.jp/web/t_doc?dataId=79aa5142%26dataType=0%26pageNo=1.
- J. Ma and P. K. Dasgupta, Anal. Chim. Acta, 2010, 673, 117–125, DOI:10.1016/j.aca.2010.05.042.
- I. M. El-Sewify, M. A. Shenashen, H. Gomaa, M. S. Selim, N. Akhtar, A. Azzam, M. Mekawy, M. Y. Emran, M. Khairy, A. Shahat, M. Hasan, A. Elmarakbi and S. A. El-Safty, Environ. Pollut., 2025, 385, 127094, DOI:10.1016/j.envpol.2025.127094.
- T. Suzuki, A. Hioki and M. Kurahashi, Bunseki Kagaku, 2003, 52, 51–54 CrossRef CAS.
- K. Shikakume and H. Sato, Bunseki Kagaku, 2003, 52, 611–618 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2026 |




