Provenance and firing temperatures of Shang dynasty (1600–1300 BC) pottery from the Shuyuanjie site, China: an integrated study using EDXRF, XRD, FTIR and thermal dilatometry (TD)
Pengcheng
Li
a,
Haiwang
Liu
c,
Bing
Wang
a,
Tianxing
Cui
b,
Yajie
Zhang
a,
Weijuan
Zhao
 *ab and
Haizhou
Zhao
*b
*ab and
Haizhou
Zhao
*b
aSchool of Physics and Zhongyuan Zhiguang Laboratory, Zhengzhou University, Zhengzhou 450001, China. E-mail: zwj@zzu.edu.cn
bSchool of Archaeology and Cultural Heritage, Zhengzhou University, Zhengzhou 450001, China. E-mail: zhaohaizhou77@163.com
cHenan Provincial Institute of Cultural Heritage and Archaeology, Zhengzhou, 450000, China
First published on 10th December 2025
Abstract
This study employed a combined analytical approach utilizing EDXRF, XRD, FTIR, and thermal dilatometry (TD) to comprehensively analyze the raw material characteristics, provenance, mineral composition, and firing temperatures of Shang dynasty grey pottery excavated from the Shuyuanjie (SYJ) site. Based on significant differences in the SiO2, K2O, and CaO content, the grey pottery bodies can be classified into three distinct raw material types. The chemical compositions of pottery from the SYJ and Xiaoshuangqiao (XSQ) sites are highly similar, suggesting a possible common raw material source. The principal phases within the grey pottery bodies are quartz and microcline, with some samples containing trace amounts of cristobalite and rutile. XRD and FTIR analyses confirmed that this batch of grey pottery underwent high-temperature firing, with firing temperatures all exceeding 750 °C. Thermal dilatometry analysis (using a single heating cycle combined with the first derivative curve) determined that the firing temperatures of the grey pottery range between 870 °C and 990 °C; all samples are underfired bodies. For pottery of unknown firing histories, water absorption and apparent porosity cannot serve as reliable proxies for determining the high or low firing temperature of the body.
1 Introduction
The Shangcheng site, situated in Zhengzhou city, Henan province, China, represents an early royal capital of the Shang dynasty (circa 1600–1300 BC). This site encompasses both inner and outer city areas. The Shuyuanjie (SYJ) site is located in the south–western corner of the inner city of the Zhengzhou Shangcheng site. This area falls within the boundaries of the residential area of the early Shang dynasty.1 In 2019, to support the construction of the first phase of the SYJ environmental renovation project in Zhengzhou city, the Henan provincial institute of cultural relics and archaeology undertook continuous collaborative archaeological excavations at the SYJ site.2 During the excavation process, a significant number of pottery artifacts from the early Shang dynasty were uncovered. The pottery materials primarily comprised two categories: clay and sand pottery. Manufacturing techniques included molding, hand-forming, and wheel-throwing. Some surfaces exhibited polishing, while impressed patterns predominantly featured rope marks, square grids, and circular motifs. As a fundamental craft product in ancient societies, pottery functioned both practically and ritually, acting as a critical medium for studying craft technology, regional interaction, and cultural transmission. The pottery samples excavated from the SYJ site exhibit fine paste and a hard texture, reflecting the advanced firing technology of the early Shang dynasty and providing valuable materials for investigating contemporary social life.In archaeology, scholars primarily rely on pottery typology (form and decoration) and petrographic analysis to infer provenance. However, typological characteristics are susceptible to cultural transmission and imitation, making precise provenance determination challenging. To address this, energy-dispersive X-ray fluorescence (EDXRF) has been introduced for non-destructive chemical composition analysis, combined with multivariate statistics to investigate provenance. For example, Li Hongfei et al.'s EDXRF analysis of raw material characteristics of Yueshi and Erligang cultural-style pottery unearthed from the Xiaoshuangqiao (XSQ) site demonstrated a Zhengzhou origin for the Yueshi-style pottery.3 Lü Liangbo et al. employed EDXRF to analyze the raw material characteristics of Shang dynasty pottery unearthed from the Moyishan site in Zengcheng, Guangdong province, demonstrating that the pottery was locally produced.4 Beyond typology and petrography, firing temperature serves as a critical technical parameter, closely related to clay mineral composition, kiln structure, and technological traditions. Regional variations in raw materials and kiln types result in geographically distinct firing temperature ranges and control patterns among potters from different production centers. Zhang Yajie et al. analyzed the chemical composition and firing temperature of Song-Jin period celadon from the SYJ site using EDXRF and thermal dilatometry (TD), revealing that the celadon originated from the Donggou kiln and Yaozhou kiln, with significant differences in firing temperatures between the two kilns, thus providing key indicators for provenance determination.5 Consequently, firing temperature can be considered an effective ‘technical fingerprint’. However, when the original firing temperature is lower than the vitrification temperature of the pottery body, the shrinkage inflection observed in the thermal expansion curve is often caused by the vitrification process rather than reflecting the original firing temperature of the pottery.6 Zhang et al. pointed out that for pottery with an original firing temperature significantly below 800 °C, a repeated stepwise heating protocol with holding stages should be applied in thermal expansion analysis to avoid interference caused by vitrification during measurement.7 Li et al. experimentally validated the applicability of thermal expansion analysis for determining the firing temperature of pottery by testing laboratory-prepared samples fired at different temperatures.8 Their results demonstrated that when the firing temperature reaches approximately 800 °C or higher, a clear correlation exists between the firing temperature and the inflection point in the thermal expansion curve.8 Furthermore, variations in the flux content within the ceramic paste across different regions may lead to differences in the vitrification temperature. Therefore, pre-characterization techniques such as XRD and FTIR are recommended to estimate the firing temperature range prior to thermal expansion measurements. For samples predicted to be low-fired (<<800 °C), a more complex testing protocol involving repeated heating cycles with temperature holds is necessary to minimize measurement artifacts induced by vitrification. In contrast, for high-fired samples (≈800 °C or ≥800 °C), a simpler single-round heating procedure can be reliably employed.
Extensive analyses of the chemical composition of pottery bodies and firing temperatures have enabled scholars to reconstruct regionally distinct pottery production techniques. However, as a core area reflecting the pottery production level of the Shang dynasty royal capital settlement, the SYJ site has not yet been subjected to systematic scientific analysis of its excavated pottery. Therefore, this study focuses on fine-paste grey pottery from the early Shang period at the SYJ site. Representative samples, whose typical morphology is shown in Fig. 1, were analyzed using EDXRF to determine their chemical composition. Combined with multivariate statistics, the raw material characteristics and potential provenance of these artifacts were investigated, providing scientific insights into the production and circulation of pottery within the SYJ site. XRD and FTIR were employed to characterize the mineral composition and suggest firing temperature ranges of the grey pottery. TD with controlled heating rates determined precise firing temperatures, providing accurate results and scientific insights into the firing technology of Shang dynasty grey pottery from the SYJ site.
2 Experimental samples and methods
2.1 Experimental preprocessing
The grey pottery samples analyzed in this study were provided by the Henan Provincial Institute of Cultural Heritage and Archaeology, excavated from the Shang dynasty cultural layer at the Zhengzhou SYJ site in 2022. Prior to any destructive analysis, the surfaces of the intact samples were cleaned with acetone and anhydrous ethanol for non-destructive EDXRF analysis.Following EDXRF analysis, two rectangular blocks with parallel ends were sectioned from the cleaned pottery body using a 0.35 mm diamond wire saw. These blocks underwent 2–3 cycles of ultrasonic cleaning in deionized water to remove impurities completely, followed by drying at 120 °C to eliminate moisture and residual organic compounds. One block was ground in an agate mortar to a fine powder, sieved through a 200-mesh sieve, and retained for XRD and FTIR analysis; the other block was reserved for thermal dilatometry (TD).
2.2 EDXRF experiment
The chemical composition of each grey pottery body was quantitatively analyzed non-destructively using energy-dispersive X-ray fluorescence (EDXRF) spectroscopy. Precise positioning at the fixed focal point of the analysis system was ensured by high/low magnification CCD cameras and a 3-axis stepper motor, maintaining fixed distances between the detector, collimator, and sample surface. The EDXRF operating conditions were set at 40 kV/250 µA with a beam spot diameter of Φ = 1 mm, an energy resolution of 160.3 eV (Mn Kα), and a 300 s measurement time. Data processing utilized the integrated VISION32 software, applying the fundamental parameter method for quantification.2.3 XRD experiment
The mineral composition of the pottery body was analyzed using a Rigaku SmartLab X-ray diffractometer (Rigaku, Japan). Scans were performed across the 10–80° (2θ) range at a rate of 10° min−1 with a step size of 0.01° under operating conditions of 40 kV and 30 mA. The XRD patterns were interpreted using the Jade software coupled with the ICDD database.2.4 FTIR experiment
Structural characterization of the pottery body was performed using an IS5 Fourier transform infrared (FTIR) spectrometer (Thermo Fisher Scientific, USA). Spectra were collected over the 4000–400 cm−1 range with 16 scans at 4 cm−1 resolution, requiring approximately 30 s per measurement. Data analysis utilized Thermo Scientific OMNIC software. Samples were prepared by homogeneously grinding the body with KBr at a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 150 mass ratio. Triplicate measurements were conducted for each sample to ensure spectral reproducibility.
150 mass ratio. Triplicate measurements were conducted for each sample to ensure spectral reproducibility.
2.5 TD experiments and physical properties
Thermal dilatometry (TD) analysis was conducted on a DIL 402 dilatometer (Netzsch, Germany) equipped with an LVDT sensor. The instrument was calibrated prior to measurement using an Al2O3 reference standard. Measurements were performed on rectangular samples with parallel faces at a heating/cooling rate of 5 °C min−1 over a temperature range of 50–1200 °C. Each sample was secured in the tubular furnace with a push rod, and its length variation was monitored as a function of temperature. For the Archimedes method, the dry, water-saturated, and immersed weights of the pottery samples were measured using an electronic balance (model LT502E, 500 g capacity, ±0.1 g accuracy). Based on these measurements, physical parameters—including water absorption, apparent porosity, and bulk density—were calculated according to the Chinese national standard GB/T 2997-2015.3 Result and discussion
3.1 Analysis of raw material characteristics of grey pottery
Table 1 presents the chemical composition of the grey pottery bodies from the SYJ site, as determined by EDXRF. Hierarchical cluster analysis (HCA) was conducted on the oxide compositions (SiO2, Al2O3, Na2O, K2O, CaO, MgO, Fe2O3, TiO2) in pottery body using SPSS software. Prior to clustering, the oxide content data were standardized using Z-scores to eliminate scale differences. Ward's linkage method was employed with squared Euclidean distance as the distance measure. The results are shown in Fig. 2(a). At a threshold of λ = 44, the compositions were classified into three groups: group I (10 samples: SYJT20–SYJT35), group II (3 samples: SYJT27–SYJT34), and group III (17 samples: SYJT22–SYJT41). As shown in Fig. 2(a), when the threshold value λ is below 44, although the systematic distances among the chemical compositions of the grey pottery bodies are relatively small, certain differences in the raw materials persist. These variations reflect potential distinctions in the raw material sources of the SYJ grey pottery, which may arise from natural geochemical differences due to varying strata or depths within the same clay deposit, diverse processing techniques applied to different types of pottery bodies, or the intentional addition of tempering materials in different proportions. To verify this inference, principal component analysis (PCA) was performed on eight oxides in the pottery bodies for dimensionality reduction. Prior to analysis, the oxide content data were standardized using Z-scores. The first two principal components were extracted, accounting for 88.8% of the total variance. The factor score function is as follows:| Factor 1 = −0.050 Na2O − 0.058 MgO − 0.079 Al2O3 + 0.918 SiO2 − 0.123 K2O − 0.330 CaO + 0.010 TiO2 − 0.146 Fe2O3 |
| Factor 2 = 0.177 Na2O − 0.107 MgO − 0.719 Al2O3 + 0.046 SiO2 + 0.643 K2O + 0.104 CaO − 0.020 TiO2 − 0.115 Fe2O3 |
| Sample number | Na2O | MgO | Al2O3 | SiO2 | P2O5 | K2O | CaO | TiO2 | Fe2O3 | MnO |
|---|---|---|---|---|---|---|---|---|---|---|
| SYJT20 | 0.83 | 1.59 | 18.95 | 54.78 | 2.07 | 3.08 | 7.16 | 0.73 | 10.46 | 0.07 |
| SYJT21 | 1.90 | 1.98 | 17.50 | 58.51 | 0.39 | 3.79 | 4.57 | 0.93 | 10.02 | 0.17 |
| SYJT22 | 0.25 | 1.74 | 19.74 | 60.29 | 0.00 | 3.71 | 2.24 | 0.95 | 10.82 | 0.07 |
| SYJT23 | 1.63 | 2.55 | 17.48 | 56.61 | 0.32 | 4.22 | 4.52 | 0.87 | 11.48 | 0.13 |
| SYJT24 | 0.49 | 0.92 | 18.42 | 64.07 | 0.00 | 3.89 | 1.11 | 1.17 | 9.63 | 0.10 |
| SYJT25 | 0.45 | 1.02 | 17.13 | 65.43 | 0.00 | 3.73 | 1.60 | 0.86 | 9.52 | 0.10 |
| SYJT26 | 2.05 | 1.08 | 16.03 | 50.86 | 0.20 | 8.17 | 8.83 | 0.67 | 10.16 | 0.15 |
| SYJT27 | 1.19 | 0.92 | 14.80 | 60.09 | 0.06 | 10.27 | 2.22 | 0.74 | 9.37 | 0.06 |
| SYJT28 | 0.40 | 0.93 | 18.08 | 63.10 | 0.00 | 3.94 | 1.25 | 1.04 | 10.97 | 0.07 |
| SYJT29 | 2.52 | 0.95 | 11.31 | 63.94 | 0.11 | 8.26 | 4.04 | 0.70 | 7.81 | 0.06 |
| SYJT30 | 0.72 | 0.92 | 18.47 | 64.25 | 0.00 | 4.62 | 1.10 | 0.96 | 8.55 | 0.12 |
| SYJT31 | 0.38 | 1.12 | 19.17 | 64.10 | 0.00 | 4.33 | 1.13 | 0.75 | 8.80 | 0.14 |
| SYJT32 | 0.64 | 1.74 | 17.65 | 62.04 | 0.82 | 3.88 | 1.74 | 0.88 | 9.52 | 0.46 |
| SYJT33 | 0.98 | 1.21 | 15.10 | 52.92 | 0.00 | 3.98 | 9.10 | 0.71 | 9.65 | 0.15 |
| SYJT34 | 2.51 | 0.85 | 12.25 | 58.80 | 0.67 | 10.94 | 2.80 | 0.76 | 9.99 | 0.18 |
| SYJT35 | 0.41 | 4.84 | 17.07 | 55.99 | 3.65 | 3.55 | 4.00 | 0.73 | 9.19 | 0.07 |
| SYJT36 | 0.12 | 1.04 | 13.94 | 72.89 | 0.00 | 2.40 | 0.71 | 0.60 | 8.13 | 0.05 |
| SYJT37 | 0.37 | 0.91 | 14.92 | 71.52 | 0.00 | 2.71 | 0.95 | 0.80 | 7.65 | 0.03 |
| SYJT38 | 0.64 | 1.50 | 19.53 | 59.93 | 0.00 | 5.67 | 1.70 | 0.92 | 9.01 | 0.07 |
| SYJT39 | 0.66 | 1.01 | 15.53 | 67.86 | 0.01 | 3.85 | 1.73 | 0.78 | 8.37 | 0.06 |
| SYJT40 | 0.35 | 1.30 | 17.63 | 61.65 | 0.14 | 4.23 | 3.14 | 0.68 | 10.67 | 0.05 |
| SYJT41 | 0.77 | 1.03 | 14.77 | 70.65 | 0.11 | 3.16 | 1.41 | 1.85 | 5.99 | 0.07 |
| SYJT42 | 0.46 | 1.15 | 16.00 | 62.05 | 0.90 | 4.95 | 2.53 | 0.80 | 10.90 | 0.07 |
| SYJT43 | 0.34 | 0.96 | 13.95 | 67.06 | 0.73 | 3.32 | 2.22 | 0.75 | 10.40 | 0.07 |
| SYJT44 | 0.34 | 1.24 | 17.35 | 64.57 | 0.19 | 3.96 | 2.19 | 1.31 | 8.33 | 0.07 |
| SYJT45 | 0.32 | 1.14 | 15.72 | 68.54 | 0.29 | 3.46 | 2.12 | 0.69 | 7.40 | 0.07 |
| SYJT46 | 0.75 | 2.33 | 17.51 | 58.28 | 1.60 | 4.25 | 6.40 | 0.86 | 7.66 | 0.09 |
| SYJT47 | 0.57 | 2.02 | 16.12 | 54.69 | 1.34 | 4.44 | 6.12 | 0.81 | 13.50 | 0.16 |
| SYJT48 | 1.26 | 1.79 | 17.35 | 59.21 | 0.00 | 4.60 | 4.10 | 1.24 | 9.70 | 0.12 |
| SYJT49 | 1.04 | 1.88 | 17.10 | 58.82 | 0.54 | 4.99 | 5.18 | 0.91 | 9.18 | 0.12 |
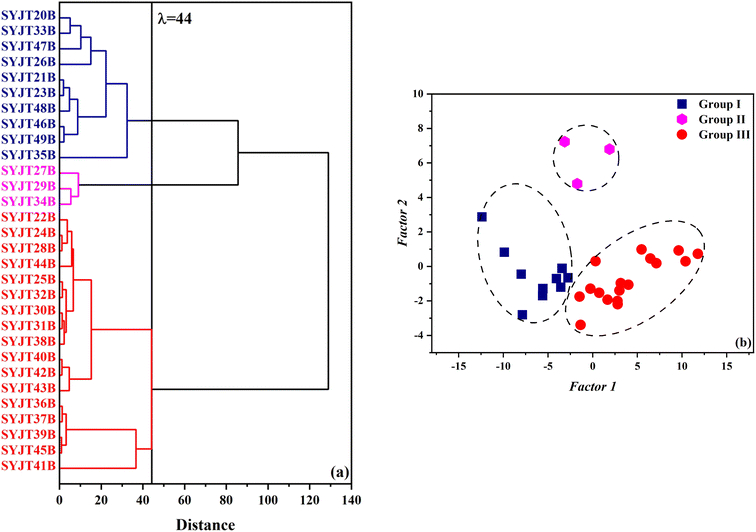 | ||
| Fig. 2 (a) Hierarchical clustering dendrogram and (b) factor loading scatter plot of chemical compositions in grey pottery bodies. | ||
Scatter plots of the grey pottery bodies were constructed with factor 1 and factor 2 as the x- and y-axes, respectively, as depicted in Fig. 2(b). The chemical compositions of the SYJ grey pottery remained classifiable into three groups, consistent with the clustering results in Fig. 2(a). In Fig. 2(b), compositional characteristics exhibited greater dispersion and variability, which accords with the HCA outcome.
The scatter plots of SiO2–Al2O3, K2O–CaO, Na2O–MgO, and Fe2O3–TiO2 oxide contents in the grey pottery bodies from the SYJ site are presented in Fig. 3. In Fig. 3(a), groups I and III exhibit relatively high SiO2 content with similar Al2O3 levels, suggesting that these two groups may have employed raw materials rich in silica—either through the selection of highly weathered clay or the intentional addition of siliceous temper such as quartz sand. In Fig. 3(b), group III shows notably elevated K2O content, indicating the possible use of raw materials enriched in potassium feldspar minerals or the incorporation of potassium-based flux in the manufacturing process. Meanwhile, group I displays higher CaO content compared to groups II and III, implying the addition of calcareous materials such as crushed shell, calcite, or calcium-rich clay as flux. As shown in Fig. 3(c), the Na2O and MgO contents in group II are relatively consistent and clustered, while those in group I are more dispersed. In Fig. 3(d), with the exception of samples SYJT47 and SYJT41, the Fe2O3 and TiO2 contents—which are associated with coloring agents—are generally similar across all three groups, indicating a stable source of iron and titanium impurities in the raw materials of most specimens.
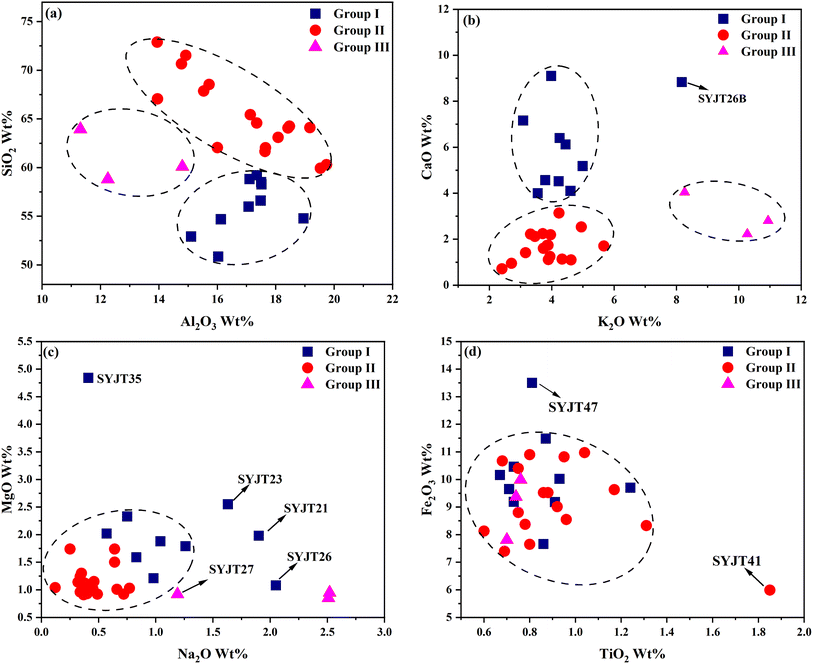 | ||
| Fig. 3 Binary scatter plots of chemical compositions in grey pottery bodies: (a) Al2O3–SiO2; (b) K2O–CaO; (c) Na2O–MgO; (d) TiO2–Fe2O3. | ||
In summary, significant differences in SiO2, K2O, and CaO contents serve as key indicators for distinguishing the chemical characteristics of the three groups of grey pottery from the SYJ site. These compositional variations reflect differences in raw material recipes, which may have resulted from multiple factors. For instance, the discrepancies could be attributed to the use of clays from different sources, or natural geochemical variability within different strata or depths of the same clay deposit. Additionally, ancient potters may have intentionally added various types or proportions of temper—such as silica-rich sand, calcareous shell, or potassium-rich plant ash—to improve the properties of the clay paste. Such adjustments likely aimed to meet the functional requirements of specific vessel types (e.g., cooking, storage, or ritual ware), including thermal shock resistance, controlled porosity, or enhanced mechanical strength.
3.2 Provenance analysis of the grey pottery
To ensure the plasticity of pottery bodies, ancient potters processed natural clay through levigation, screening, and the addition of temper materials, which modified the original chemical composition of the body. Therefore, for pottery provenance studies, clay sediments and unfired clay objects from nearby kilns should serve as appropriate reference materials. However, due to the limited availability of such archaeological remains, research often relies on pottery fragments from workshops or pottery samples of known origin as proxies. Previous studies have demonstrated that the chemical composition of pottery bodies remains largely unchanged before and after firing, and that potters consistently used the same clay recipes over certain periods.9,10 Thus, pottery from the same site or region can be attributed to respective production units, thereby facilitating the reconstruction of the provenance of pottery excavated within the site.As the early Shang capital, Zhengzhou was surrounded by numerous contemporaneous sites, forming a royal hinterland network. The XSQ site is located in Xiaoshuangqiao Village, Guangwu Town, Xingyang, approximately 20 km northwest of the Zhengzhou Shangcheng site. As the largest and highest-ranking capital-scale settlement during the Baijiazhuang phase of Shang culture, the XSQ site has been subject to scholarly analysis from the perspectives of typology and raw material characteristics.3 Previous research suggests that some of the pottery unearthed at the site may have been produced by potters familiar with Yueshi pottery traditions, working within the Zhengzhou region.3 The Erlitou site (ELT), located in Yanshi district, Luoyang city, Henan province, is approximately 80 km in straight-line distance from the Zhengzhou Shangcheng site and represents a critical site for exploring the Xia–Shang cultural transition. The Yanshi Shangcheng (YSS) site, situated in Shangcheng Subdistrict, Yanshi city, Henan province, borders the Mang mountains to the north and the Luo River to the south. It is an important urban center of the early Shang period, also located about 80 km from the Zhengzhou Shangcheng site. Given early artisans' predominant use of locally available raw materials in pottery production, chemical compositions of pottery bodies from the Xiaoshuangqiao (XSQ), Erlitou (ELT), and Yanshi Shangcheng (YSS) sites were referenced to investigate provenance characteristics of Shang-period grey pottery unearthed at the SYJ site.3,11–13 Principal Component Analysis (PCA) was applied to six oxide contents (Al2O3, SiO2, K2O, CaO, TiO2, Fe2O3) in pottery bodies from SYJ, XSQ, ELT, and YSS for dimensionality reduction. Prior to analysis, the oxide content data were standardized using Z-scores. Two principal components explaining 91.22% of the total variance were extracted, with factor score functions as follows:
| Factor 3 = 0.038 Al2O3 + 0.886 SiO2 − 0.104 K2O − 0.289 CaO − 0.005 TiO2 − 0.343 Fe2O3 |
| Factor 4 = 0.886 Al2O3 − 0.229 SiO2 − 0.096 K2O − 0.105 CaO − 0.004 TiO2 − 0.376 Fe2O3 |
Using factor 3 and factor 4 as the x- and y-axes respectively, a two-dimensional principal component (PC) score plot was constructed to visualize the chemical compositions of pottery from Shuyuanjie (SYJ), Xiaoshuangqiao (XSQ), Yanshi Shangcheng (YSS), and Erlitou (ELT), as shown in Fig. 4. The SYJ grey pottery and XSQ samples form a distinct cluster, indicating a similar or identical raw material provenance. Although pottery from ELT and YSS—both located in the modern Luoyang region—show some compositional similarities, they constitute a separate cluster that is distinctly different from the SYJ-XSQ group.
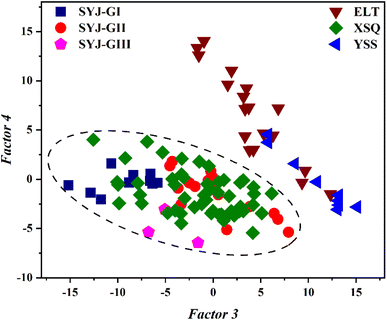 | ||
| Fig. 4 Principal component (PC) score plot for six oxides (Al2O3, SiO2, K2O, CaO, TiO2, Fe2O3) in pottery bodies from the SYJ, XSQ, ELT, and YSS sites. | ||
During the Shang culture period, pottery workshops served as crucial sites for manufacturing both tools and daily utensils, and were commonly present in various settlements.14 Previous research on the classification of handicraft industries at the Shangcheng site has indicated that pottery production there was characterized by local procurement of raw materials, local production, and local distribution.15 For instance, the pottery workshop, located west of present-day Minggong Road in Zhengzhou, measured approximately 800 meters in length and 150 meters in width, covering a total area of 120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 square meters. Within this area, 15 kilns and 4 workshop floors have been identified.16 Extensive remains, including complete vessels, pottery sherds, raw clay, fired products, and dedicated pits for discarding defective pottery (“pottery burial pits”), have been unearthed, reflecting a relatively large-scale and mature production system.15 A trench, approximately 36 meters long, 8–9 meters wide, and 4–6.5 meters deep, oriented northwest–southeast, was found adjacent to the workshop and is believed to have resulted from clay extraction by the potters.15 As a large, high-ranking ritual zone affiliated with the Zhengzhou Shangcheng site, the XSQ site has yielded pottery exhibiting Yueshi cultural stylistic features. Archaeometric and typological analyses suggest that these vessels share morphological and chemical characteristics with those unearthed from the Zhengzhou Shangcheng site, indicating that they may have been produced by artisans familiar with Yueshi pottery traditions within the Zhengzhou area.3,11,17
000 square meters. Within this area, 15 kilns and 4 workshop floors have been identified.16 Extensive remains, including complete vessels, pottery sherds, raw clay, fired products, and dedicated pits for discarding defective pottery (“pottery burial pits”), have been unearthed, reflecting a relatively large-scale and mature production system.15 A trench, approximately 36 meters long, 8–9 meters wide, and 4–6.5 meters deep, oriented northwest–southeast, was found adjacent to the workshop and is believed to have resulted from clay extraction by the potters.15 As a large, high-ranking ritual zone affiliated with the Zhengzhou Shangcheng site, the XSQ site has yielded pottery exhibiting Yueshi cultural stylistic features. Archaeometric and typological analyses suggest that these vessels share morphological and chemical characteristics with those unearthed from the Zhengzhou Shangcheng site, indicating that they may have been produced by artisans familiar with Yueshi pottery traditions within the Zhengzhou area.3,11,17
In summary, although some variation is observed in the raw material characteristics of the grey pottery fragments excavated from the SYJ site, their overall chemical composition is highly consistent with that of ceramics from XSQ. Given that both sites belonged to the settlement and ritual zones within the broader Shangcheng site complex, it can be concluded that the pottery from SYJ was likely produced in or around Zhengzhou Shangcheng site and may have shared similar raw material sources and manufacturing technology with pottery from the XSQ site.
3.3 Analysis of the firing temperature range for grey pottery
Fig. 5 presents the XRD patterns of the grey pottery bodies. With the exception of sample SYJT23, the bodies are predominantly composed of quartz and microcline. Notably, minor cristobalite was detected in samples SYJT22, SYJT23, and SYJT28. Sample SYJT23 also contains spinel and anorthite, while a small amount of rutile was identified in SYJT45. Quartz primarily exists in the [SiO4]4− tetrahedral structure, and its crystal phase transforms with temperature. Cristobalite, a high-temperature polymorph of quartz, typically begins to form slowly at around 1200 °C and develops rapidly at approximately 1400 °C.18 Consequently, it is inferred that samples SYJT22, SYJT23, and SYJT28 were fired at relatively high temperatures. However, the presence of feldspar minerals in the body likely acted as a flux, potentially lowering the crystallization temperature of cristobalite. Furthermore, microcline, a triclinic feldspar, forms from the structural rearrangement of orthoclase (KAlSi3O8) between 410 and 470 °C, indicating that the firing temperature for all samples exceeded 470 °C. However, due to the complexity of phase transitions with temperature, determining the precise firing temperature range for some samples based solely on XRD analysis remains challenging. Therefore, infrared spectroscopy was employed for further investigation.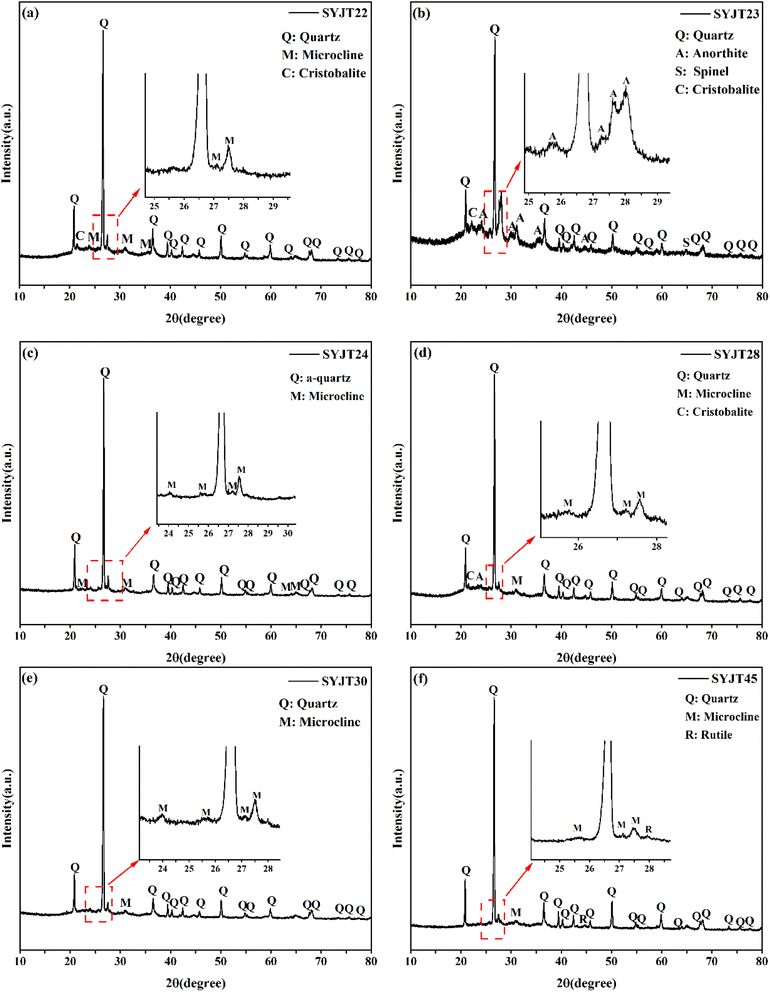 | ||
| Fig. 5 XRD patterns of the pottery bodies of samples (a) SYJT22, (b) SYJT23, (c) SYJT24, (d) SYJT28, (e) SYJT30, and (f) SYJT45. | ||
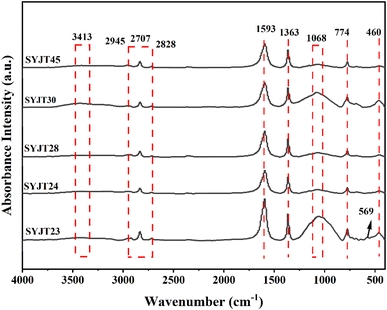
Fourier-transform infrared (FTIR) spectra of the SYJ grey pottery bodies are presented in Fig. 6. Because pottery bodies are composed of various clay minerals, temperature variations during firing directly affect their structure, thereby altering the infrared absorption bands. In Fig. 6, the infrared spectral absorption peaks of different pottery bodies exhibit similar wavenumbers, located near 460 cm−1, 774 cm−1, 1068 cm−1, 1363 cm−1, 1593 cm−1, 2707 cm−1, 2828 cm−1, 2945 cm−1, and 3413 cm−1. During pottery production, the addition of appropriate amounts of organic matter enhances the plasticity of the clay body. The absorption peaks near 2828 cm−1 and 2945 cm−1 originate from the C–H stretching vibrations of organic compounds within the pottery body.19 Absorption peaks near 460 cm−1 and 774 cm−1 correspond to Si–O bond bending and symmetric stretching vibrations, confirming the presence of quartz within the body.20,21 The absorption peak near 3413 cm−1 is attributed to hydrogen bonding.22 Due to the relatively intense dehydroxylation of the SYJ grey pottery body during firing as temperature increased, no absorption peak associated with hydroxyl groups bridging the lattice Al and Mg structures was observed near 3620 cm−1.23,24 This indicates that the firing temperature exceeded 550 °C. The Si–O–Si stretching vibration peak is located near 1030 cm−1 at lower temperatures, gradually shifting toward higher wavenumbers with increasing temperature.25,26 When the Si–O–Si absorption peak is observed at 1068 cm−1, and the absorption bands near 1030 cm−1 and 3620 cm−1 disappear, a firing temperature above 750 °C is indicated.24 The FTIR spectrum of sample SYJT23 shows an absorption peak at 569 cm−1, which is attributed to the dehydroxylation of clay minerals upon heating. During this process, the Al–O octahedral structure gradually breaks down and transforms into an Al–O tetrahedral structure. This absorption peak disappears when the temperature reaches approximately 1050 °C.24 The presence of the absorption peak at 569 cm−1 indicates that the firing temperature of SYJT23 was below 1050 °C. Based on the analysis of XRD and FTIR results, the body of the SYJ pottery exhibits characteristics indicative of a high firing temperature. Therefore, the thermal expansion method can be applied using a single heating cycle to determine its firing temperature, as the inflection point in the thermal expansion curve corresponds reliably to the firing temperature.
3.4 Analysis of firing temperature and physical properties of grey pottery
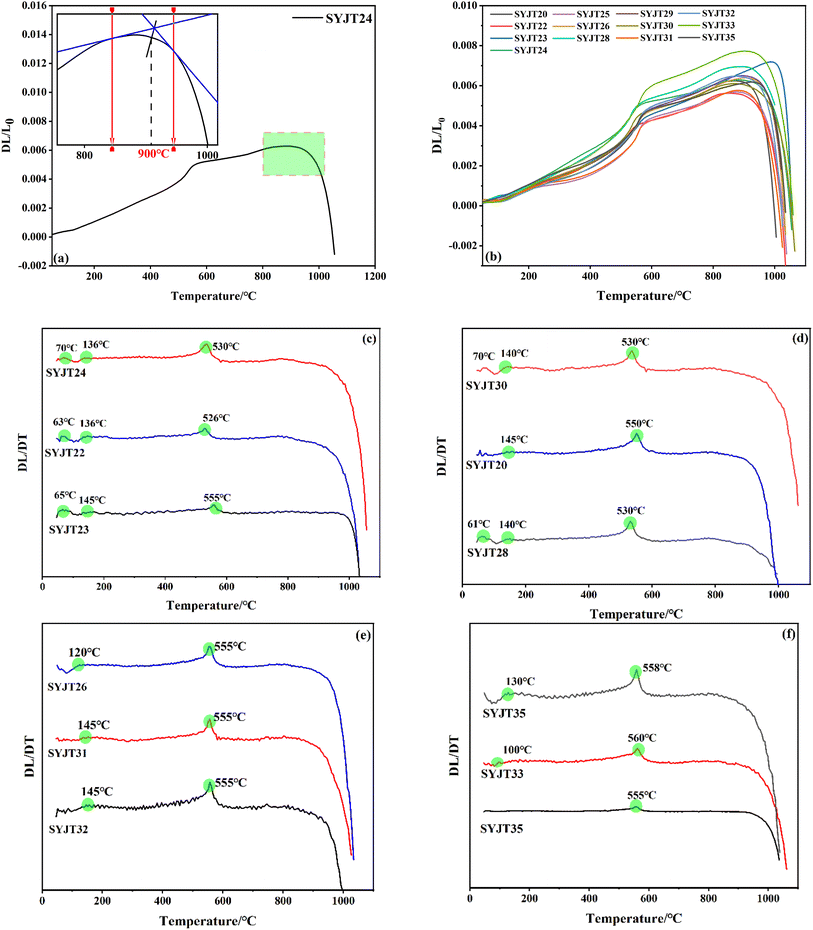
Fig. 7 presents thermal expansion and first derivative curves for bodies of SYJ grey pottery. The thermal expansion curve reflects the magnitude of displacement changes during the heating process of the pottery body, while the first derivative curve indicates the rate of displacement change. The onset temperature of the shrinkage effect on the thermal expansion curve, which corresponds to the original firing temperature, was determined using the tangent method, with an estimated error of ±20 °C.27 As shown in Fig. 7(a) and (b), the volume of the pottery body expands with increasing temperature until a certain point is reached, whereupon rapid contraction occurs, indicated by a sharp decrease in ΔL/L0. This shrinkage behavior indicates that all the grey pottery bodies from the SYJ site were underfired (incompletely sintered). Among these, SYJT23 exhibits the highest firing temperature of 990 °C, while the firing temperatures of the remaining grey pottery range between 870–940 °C.Abrupt changes in the thermal expansion coefficient, caused by phase transformations during sintering, are reflected in the derivative curves (i.e., the rate of change) presented in Fig. 7(c)–(f). Below 100 °C, the pottery bodies undergo slight shrinkage due primarily to the removal of physically adsorbed water, which brings clay particles closer together. During heating, residual moisture is expelled as the temperature rises, causing a discernible change (or: deviation) in the thermal expansion curve between 130 and 145 °C. As one of the most common mineral constituents in ceramic bodies, quartz exhibits multiple polymorphic variants under different temperatures and conditions. Upon heating, α-quartz (the low-temperature form) transforms reversibly to β-quartz at 573 °C, a transition that is accompanied by a distinct volume expansion.28 The presence of feldspathic fluxing minerals within the body can broaden and lower the temperature range over which this phase transition occurs. Consequently, a shift in the thermal expansion curve is observed around 530–560 °C, which is attributed primarily to the α-to β-quartz phase transition. As the temperature approaches the original firing temperature, the rate of contraction decreases dramatically, manifesting as a sharp trough in the first derivative curve. This result is consistent with the firing temperature determined from the onset of shrinkage on the expansion curve.
Table 2 presents the firing temperatures, water absorption, and apparent porosity of the SYJ grey pottery bodies. Water absorption ranges from 10.5% to 14.74%, and apparent porosity ranges from 24.28% to 31.29%. Before a ceramic body reaches its sintering temperature, its apparent porosity and water absorption decrease with increasing temperature.29 Therefore, these two properties generally exhibit a negative correlation with the firing temperature. Fig. 8 presents a two-dimensional scatter plot illustrating the relationship between the firing temperature, water absorption, and apparent porosity of the SYJ pottery. As the firing temperature increases, both the water absorption and apparent porosity of the pottery body exhibit limited variation. Notably, sample SYJT23, which has the highest firing temperature, also demonstrates the highest values of water absorption and apparent porosity. Under well-controlled laboratory conditions–where parameters such as raw material, forming method, firing duration, atmosphere, and particle size are kept consistent—an increase in firing temperature generally promotes a higher degree of vitrification, leading to a significant reduction in both apparent porosity and water absorption. However, in the uncontrolled context of ancient pottery production, the compactness of pottery bodies is considerably lower than that of porcelain. For individual archaeological samples with unknown firing histories, water absorption and apparent porosity are influenced by multiple factors, including the forming technique, chemical composition, firing duration, atmosphere, and the size of clay particles. Thus, the dispersed distribution of data points in Fig. 8 indicates that there is no strict negative correlation between firing temperature and the physical properties of water absorption and apparent porosity. This can be primarily attributed to the fact that, in real ancient pottery-making environments, variations in raw materials and forming methods often overshadow the dominant effect of temperature. Consequently, for pottery samples of unknown firing background, water absorption and apparent porosity are unreliable for determining the level (or range) of firing temperature.
| Sample number | Firing temperature/°C | Apparent porosity % | Water absorption % |
|---|---|---|---|
| SYJT20 | 880 | 26.49 | 13.87 |
| SYJT21 | 870 | 20.49 | 10.05 |
| SYJT22 | 900 | 26.21 | 13.96 |
| SYJT23 | 990 | 31.29 | 17.32 |
| SYJT24 | 900 | 26.31 | 13.91 |
| SYJT25 | 910 | 27.27 | 14.49 |
| SYJT26 | 900 | 27.82 | 14.74 |
| SYJT27 | 910 | 27.32 | 14.45 |
| SYJT28 | 895 | 26.81 | 14.04 |
| SYJT29 | 890 | 27.35 | 14.56 |
| SYJT30 | 880 | 24.28 | 14.57 |
| SYJT31 | 920 | 27.48 | 14.64 |
| SYJT32 | 940 | 26.38 | 13.87 |
| Mean | 917 | 26.57 | 14.19 |
| SD | 29.83 | 2.30 | 1.47 |
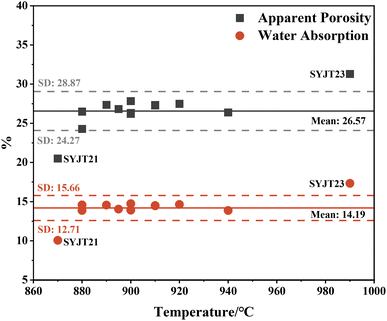 | ||
| Fig. 8 Relationship between firing temperature and the physical properties (apparent porosity and water absorption) of the grey pottery bodies. | ||
4 Conclusion
This study employed a combination of modern analytical techniques, including EDXRF, XRD, FTIR, and thermal dilatometry (TD), to systematically investigate the raw material characteristics, provenance, and firing temperatures of Shang dynasty grey pottery excavated from the SYJ site. Analyses were conducted from four perspectives: chemical composition, phase analysis, infrared spectral features, and thermal expansion curve. Significant differences in the contents of SiO2, K2O, and CaO were observed in the pottery bodies, indicating three distinct chemical compositional groups. The chemical composition of the SYJ pottery is similar to that of artifacts unearthed from the XSQ site, suggesting a common production origin likely within the Zhengzhou Shangcheng site and its vicinity. Phase composition revealed that the pottery bodies are primarily composed of quartz and microcline, with minor amounts of cristobalite and rutile detected in some samples. The firing temperature is estimated to have exceeded 750 °C. Based on complementary XRD and FTIR results, TD measurements further refined the firing temperature range to between 870 and 990 °C, indicating that all samples were underfired. For pottery samples with unknown firing histories, the correlation between firing temperature and both water absorption and apparent porosity is relatively weak, rendering it impossible to directly predict the firing temperature level (high or low) of the pottery body based on the magnitudes of water absorption and apparent porosity.Author contributions
Pengcheng Li: conceptualization, methodology, data curation, formal analysis, visualization, writing – original draft; Haiwang Liu: writing – review & editing; Bing Wang: project administration; Tianxing Cui: investigation; Yajie Zhang: validation; Weijuan Zhao: funding acquisition, supervision, resources, writing – review & editing; Haizhou Zhao: supervision, resources, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The authors confirm that the data supporting the findings of this study are available within the article.Acknowledgements
This research was supported by the National Natural Science Foundation of China (Grant No. 11975210) and the Henan Science and Technology Archaeology Enhancement Project.References
- Y. Zhou, master thesis, Sichuan Agricultural University, 2022.
- S. Yang, L. Hong and W. Hou, Huaxia Archaeol., 2022, 16–36 Search PubMed.
- H. Li, S. Li, J. Cui, N. Wang, X. Zeng and G. Song, Cultural Relics of Central China, 2015, pp. 116–121 Search PubMed.
- L. Lv, Q. Li and X. Zhang, J. Archaeol. Mus., 2024, 40–50 Search PubMed.
- Y. Zhang, W. Zhao, H. Zhao, T. Cui, H. Liu, B. Wang, X. Feng, L. Li and P. Li, Anal. Methods, 2025, 17, 4206–4215 RSC.
- M. S. Tite, Archaeometry, 1969, 11, 131–143 CrossRef CAS.
- Y. Zhang, J. Zhu, T. Wang, C. Zhao, J. Yu and C. Wang, Cultural Relics in Southern China, 2012, pp. 140–146 Search PubMed.
- Y. Li, C. Wang and K. Zheng, J. Ceram., 2012, 33, 215–221 Search PubMed.
- H. Mommsen, Archaeometry, 2004, 46, 267–271 CrossRef CAS.
- J. Cogswell, H. Neff and M. Glascock, J. Archaeol. Sci, 1996, 23, 283–287 Search PubMed.
- S. Song, S. Li and G. Song, Huaxia Archaeol., 2023, 147–156 CAS.
- Y. Yang, L. Zeng, G. Chen, B. Sun, X. Gong and X. Zhang, Huaxia Archaeol., 2024, 29–37 Search PubMed.
- X. Lu, W. Li, H. Luo, H. Xu, H. Zhao and J. Yuan, Archaeology, 2012, 89–96 CAS.
- S. He, Master Degree thesis, Zhengzhou University, 2019.
- C. Chen, Doctoral Dissertation, Zhengzhou University, 2004 Search PubMed.
- X. Han, Cultural Relics of Central China, 2009, 39–43, p. 80 Search PubMed.
- M. Wang, Oriental Collection, 2024, pp. 116–118 Search PubMed.
- R. Zhang, H. Wang and H. Xu, Ceramic Technology, Chemical Industry Press, Beijing, 2013 Search PubMed.
- L. He, S. Yao, Z. Sun, J. Shao, N. Di and T. Li, J. Archaeol. Sci. Rep., 2023, 48, 103844 Search PubMed.
- J. A. Kittrick, Soil Sci. Soc. Am. J., 1968, 32, 4 CrossRef.
- S. Shoval and Y. Paz, Period. Mineral., 2015, 84, 213–231 Search PubMed.
- V. Lorenzelli and G. Busca, Mater. Chem. Phys., 1985, 13, 261–281 Search PubMed.
- C. Lu, Soils, 1986, 94–100+113 CAS.
- S. Liu, X. Gong and B. Yan, Spectrosc. Spectral Anal., 2023, 43, 1495–1500 CAS.
- K. Ramaswamy and M. Kamalakkannan, J. Therm. Anal., 1995, 44, 629–638 CrossRef CAS.
- S. Shoval, J. Therm. Anal., 1994, 42, 175–185 CrossRef CAS.
- X. Lu, C. Xu and W. Li, Sci. Conserv. Archaeol., 2020, 32, 70–80 Search PubMed.
- P. W. J. Glover, P. Baud, M. Darot, P. G. Meredith, S. A. Boon, M. LeRavalec, S. Zoussi and T. Reuschlé, Geophys. J. Int., 1995, 120, 775–782 CrossRef.
- J. Li, A History of Science and Technology in China (Ceramics), Science Press, 1998 Search PubMed.
| This journal is © The Royal Society of Chemistry 2026 |