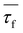Open Access Article
Open Access ArticleDigital fluorescent pH probes: polymer-based design, fluorescence response, mechanism, functional tuning and application to logic operation in live cells
Seiichi
Uchiyama
 *,
Eiko
Hamada
*,
Eiko
Hamada
 ,
Masaharu
Takarada
,
Masaharu
Takarada
 and
Kohki
Okabe
and
Kohki
Okabe

Graduate School of Pharmaceutical Sciences, The University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo 113-0033, Japan. E-mail: seiichi@g.ecc.u-tokyo.ac.jp
First published on 12th February 2026
Abstract
We report fluorescent digital pH probes based on a polymeric design comprising thermoresponsive, proton-binding and environment-sensitive fluorescent units. At selected temperatures, fluorescence switching was nearly complete, with a slight pH variation of <1 unit. The fluorescence-switching mechanism involved a proton-induced three-dimensional structural change in the polymer, which altered local hydrophobicity near the environment-sensitive fluorescent units. Based on the change in local hydrophobicity during operation, extremely sharp responses were produced because of the variation in proton-binding abilities of the probes. The polymeric design ensured functional flexibility, including a pH-responsive region and a direction of fluorescence switching. As an application of fluorescent digital pH probes, intracellular logic operations were performed by varying the pH and temperature. By distinguishing the minute pH and temperature difference inside mammalian cells, a specific condition—high pH and temperature—generated strong fluorescence with an extended fluorescence lifetime, as confirmed via fluorescence-lifetime imaging microscopy.
Introduction
Fluorescent probes change their fluorescence characteristics such as fluorescence intensity and lifetime, and maximum emission wavelength in response to concentrations of chemical species (i.e., ions or molecules) or variations in physical quantities (e.g., temperature and viscosity).1–3 To date, various fluorescent ion probes have been developed. Among them, fluorescent pH probes are used in specialized research areas and even daily life,4 as aqueous solutions always contain protons, and solution pH predominantly influences chemical reactions5 and bioenergetically represented biological events.6 Fluorescence-imaging microscopy is a practical application of fluorescent pH probes, with a significant impact on biology.7As a general principle of fluorescent ion probes, the function of fluorescent pH probes relies on the equilibrating complexation of the receptor (e.g., an amine) and target ion (i.e., the proton in this case) (eqn (1)).
| Free probe + H+ ⇄ Protonated probe | (1) |
The difference between free and protonated receptors in their effects on the electric properties of the fluorophore is the basis of the fluorescence off–on switching mechanism. The control of internal charge transfer (ICT) and photoinduced electron transfer (PET) efficiency is involved in fluorescent pH probes.8 The definition of the acidity constant (Ka) for a fluorescent pH probe is expressed as eqn (2).
| Ka = ([Free probe][H+])/[Protonated probe] | (2) |
The following eqn (3) should always be effective in an aqueous solution.
| −log ([Protonated probe]/[Free probe]) = pH − pKa | (3) |
Eqn (3) indicates that the operational pH width of probes is fundamentally constant (two pH units are necessary for the variation from [Protonated probe]/[Free probe] = 0.1 to [Protonated probe]/[Free probe] = 10), whereas the operational pH range shifts according to the pKa value of the probe.
Although the relation in eqn (3) is universal for fluorescent pH probes, unusually sharp responses to pH variation in a narrow pH width appear in considerably limited cases, involving inner filter effects,9 micellization of block copolymers,10–12 deoxyribonucleic acid (DNA) i-motifs13 and sequential protonation of multiple receptors in a molecule.14–16 Such digital responses of fluorescent pH probes are beneficial for highly sensitive pH monitoring and molecular logic operation,17–20 where fluorescence switching should be completed by a minor variation in proton concentrations (a small pH change). A noteworthy application of a digital fluorescent pH-sensing system is highly selective in vivo tumor imaging in mice.21
In 2009, we unexpectedly discovered the digital behavior of a few fluorescent polymeric pH probes and reported four examples in a preliminary communication.22 Our detailed findings are mentioned here again. (i) Poly(N-isopropylacrylamide) (PNIPAM) is a thermoresponsive polymer with a lower critical solution temperature (LCST)23,24 and forms a flexible structure upon hydration between the amide groups of NIPAM units and solvent water molecules in cold aqueous solutions. In contrast, it becomes rigid and globular by the breaking of hydrogen bonds and the accompanying entropy gain due to the released water molecules when the temperature of the solution is >31 °C (i.e., the LCST of PNIPAM).25 The hydrophobic interaction between isopropyl moieties in side chains is dominant, forming relatively large interpolymeric agglomerates. Thus, the solution becomes colloidal when the temperature is >31 °C (Fig. 1a).23 (ii) When ion-binding units are incorporated into PNIPAM, the copolymer responds to ions and temperature changes. The LCST of the copolymer containing the benzo-18-crown-6 moiety depends on the K+ concentration (Fig. 1b).26 At high K+ concentrations, benzo-18-crown-6 forms an ionic K+ complex, decreasing the hydrophobicity of the side chain and shifting the LCST to a high temperature. Thus, the copolymer comprising benzo-18-crown-6 is considered a polymeric K+ probe in aqueous solution when the temperature is fixed between the two LCSTs. (iii) A small amount of an environment-sensitive (polarity- and hydrogen-bonding-sensitive) fluorophore is incorporated into PNIPAM to form a copolymer, which functions as a fluorescent thermometer. The copolymer comprising NIPAM and N-{2-[(7-N,N-dimethylaminosulfonyl)-2,1,3-benzoxadiazol-4-yl](methyl)amino}ethyl-N-methylacrylamide (DBD-AA) [poly(NIPAM-co-DBD-AA)] emits strong fluorescence when the solution temperature is increased beyond the LCST (>31 °C) (Fig. 1c).27 Fluorescent polymeric thermometers developed based on this strategy play a crucial role in monitoring intracellular temperature.28 Additionally, the concept of thermal signaling29 has been proposed by using fluorescent polymeric thermometers in cellular biology. (iv) We predicted a fluorescent polymeric pH probe when proton-binding and environment-sensitive fluorescent units were incorporated into PNIPAM. The copolymer comprising NIPAM, N-[3-(dimethylamino)propyl]acrylamide (DMAPAM) and DBD-AA units [poly(NIPAM-co-DMAPAM-co-DBD-AA), 1; Fig. 1d] showed pH-dependent fluorescence characteristics, and for some reason, the response was much sharper than that of conventional fluorescent pH probes.22
 | ||
| Fig. 1 Thermoresponsive-PNIPAM-based functional polymers. (a) PNIPAM. Chemical structure (left), temperature-dependent transmittance of the aqueous solution (1 wt%) (middle), and operational mechanism (right). (b) Poly(NIPAM-co-B18C6AM). Chemical structure (left), [K+]-dependent transmittance of the aqueous solution (1 wt%) ([K+] = 0 or 0.105 mol L−1) (middle), and operational mechanism (right). (c) Poly(NIPAM-co-DBD-AA). Chemical structure (left) and temperature-dependent fluorescence intensity of the heated aqueous solution (0.01 w/v%) (right, measured using the procedure described in ref. 27). The excitation wavelength is 456 nm. (d) Chemical structure of poly(NIPAM-co-DMAPAM-co-DBD-AA) (copolymer 1). Transmittance changes in panels (a) and (b) are adapted with permission from ref. 23 and 26, respectively. | ||
This article is a full paper version based on the previous work.22 In the beginning, the fluorescence characteristics of 1 are described for the second time to demonstrate its unusually sharp response to a pH variation. Then, we elucidate the functional mechanism underlying high sensitivity to small pH variations. The structural flexibility and limitations of digital fluorescent pH probes for tuning the operational pH range (pKa of the probe) and the switching direction (i.e., on–off vs. off–on) are discussed using new and reported polymers.22 Finally, the digital fluorescent pH probes are applied to molecular logic operations that can simultaneously sense multiple chemical and physical factors in live cells. Extremely sharp responses to pH and temperature enable the discrimination of a specific cellular status, with slight pH and temperature differences.
Experimental
Materials
N-(3-Aminopropyl)acrylamide (APAM),30 DBD-AA,27 4-(2′-N,N-dimethylaminoethyl)amino-7-N,N-dimethylaminosulfonyl-2,1,3-benzoxadiazole (DBD-DMAEA),31N,2-dimethyl-N-(2-{methyl[7-(dimethylsulfamoyl)-2,1,3-benzoxadiazol-4-yl]amino}ethyl)propanamide (DBD-IA),32N-[3-(diethylamino)propyl]acrylamide (DEAPAM),22N-(3-morpholin-4-ylpropyl)acrylamide (MPAM),22N-n-propylacrylamide (NNPAM),33 and 4-vinylbenzoic acid (VBA)34 were prepared using reported methods. NIPAM was obtained from Wako Pure Chemicals (currently, FUJIFILM Wako Pure Chemical Corporation) and purified via recrystallization from n-hexane. DMAPAM was obtained from Wako Pure Chemicals. HPTS was purchased from Invitrogen (currently, Thermo Fisher Scientific, Inc.). N-tert-Butylacrylamide (NTBAM) was obtained from Sigma-Aldrich. Water was purified using a Milli-Q® reagent system (Direct-Q 3 UV, Millipore). All other reagents were of guaranteed reagent grade and used without further purification.Synthesis of dimethyl 2,2′-((3-acrylamidopropyl)azanediyl)diacetate (DMAcAPAM)
APAM (as hydrochloric acid salt, 165 mg, 1 mmol) and potassium carbonate (742 mg) were suspended in acetonitrile (10 mL). After the addition of methyl bromoacetate (918 mg, 6 mmol), the mixture was stirred at 70 °C for 6 h. After the reaction, the solution was filtered and evaporated under reduced pressure. The residue was chromatographically separated on a silica gel column using dichloromethane–methanol (100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, v/v) as an eluent to produce DMAcAPAM as a colorless oil (48.4 mg, 17.8%); 1H NMR (400 MHz, CDCl3, δ): 7.68 (br, 1H), 6.28–6.33 (ss, 1H, J = 2.6, 17.0 Hz), 6.20–6.26 (ss, 1H, J = 9.4, 17.0 Hz), 5.58–5.61 (d, 1H, J = 2.6, 9.4 Hz), 3.72 (s, 6H), 3.47–3.52 (m, 6H), 2.78 (t, 2H, J = 5.8 Hz), 1.63–1.69 (m, 2H); 13C NMR (100 MHz, CDCl3, δ): 171.9, 165.6, 131.5, 125.5, 54.7, 52.2, 51.8, 37.8, 25.9; HRMS-ESI (m/z): [M + Na+] calcd for C12H20N2NaO5+: 295.1264; found: 295.1264.
1, v/v) as an eluent to produce DMAcAPAM as a colorless oil (48.4 mg, 17.8%); 1H NMR (400 MHz, CDCl3, δ): 7.68 (br, 1H), 6.28–6.33 (ss, 1H, J = 2.6, 17.0 Hz), 6.20–6.26 (ss, 1H, J = 9.4, 17.0 Hz), 5.58–5.61 (d, 1H, J = 2.6, 9.4 Hz), 3.72 (s, 6H), 3.47–3.52 (m, 6H), 2.78 (t, 2H, J = 5.8 Hz), 1.63–1.69 (m, 2H); 13C NMR (100 MHz, CDCl3, δ): 171.9, 165.6, 131.5, 125.5, 54.7, 52.2, 51.8, 37.8, 25.9; HRMS-ESI (m/z): [M + Na+] calcd for C12H20N2NaO5+: 295.1264; found: 295.1264.
Preparation of copolymers
| No. | Thermo-responsive unit | Proton-binding unit | Feed ratioa | Yield (%) | Actual unit ratiob | M w | M n | M w/Mn |
|---|---|---|---|---|---|---|---|---|
| a Ratio of monomers for thermoresponsive, proton-binding and fluorescent DBD-AA units. b Ratio of thermoresponsive, proton-binding and fluorescent DBD-AA units. c Weight-average molecular weight. d Number-average molecular weight. e Originally reported in ref. 22. | ||||||||
| 1e | NIPAM | DMAPAM | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
83 | 87.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12.7 12.7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.038 0.038 |
120![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
3.01 |
| 2e | NIPAM | DEAPAM | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
71 | 92.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.8 7.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.028 0.028 |
79![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
39![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
2.03 |
| 3e | NIPAM | MPAM | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
82 | 90.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.2 9.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.066 0.066 |
70![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
23![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
3.03 |
| 4 | NIPAM | DMAcAPAM | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
67 | 92.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5 7.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.092 0.092 |
82![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
2.44 |
| 5e | NIPAM | AA | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
83 | 90.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.1 9.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.079 0.079 |
139![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
56![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
2.45 |
| 6 | NIPAM | VBA | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
56 | 86.1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 13.9 13.9![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.091 0.091 |
16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
1.46 |
| 7 | NNPAM | DMAPAM | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
63 | 90.7![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.3 9.3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.054 0.054 |
108![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
47![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
2.29 |
| 8 | NTBAM | DMAPAM | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.1 0.1 |
62 | 90.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 9.8 9.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.068 0.068 |
78![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
2.27 |
| 9 | NIPAM | MPAM | 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 1 |
63 | 91.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8.8 8.8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.77 0.77 |
118![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
55![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
2.12 |
Characterization of copolymers
The thermoresponsive and proton-binding units in the copolymers, except for copolymer 5, were characterized by 1H-nuclear magnetic resonance (1H-NMR) spectra recorded using Bruker Avance III HD 400 in deuterated chloroform (CDCl3). The content of AA units in 5 was determined by an acid–base titration. The proportions of DBD-AA units in the copolymers were determined from the absorbance in methanol, which was compared with that of the model fluorophore DBD-IA [molar absorption coefficient (ε) = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 M−1 cm−1 at 444 nm].32
800 M−1 cm−1 at 444 nm].32
The molecular weights of the copolymers were determined via gel permeation chromatography (GPC), comprising a JASCO PU-4580 pump, a JASCO RI-4030 refractive index detector, a JASCO CO-4060 column thermostat (set at 40 °C) and a Shodex GPC KD-806M column. A calibration curve was generated using a polystyrene standard, and 1-methyl-2-pyrrolidinone containing lithium bromide (LiBr; 20 mM) was used as the eluent.
Fluorescence measurements of copolymers
The fluorescence spectra of the copolymers were recorded using a JASCO FP-6500 spectrofluorometer—equipped with a Hamamatsu R-7029 optional photomultiplier tube (operative range: 200–850 nm)—or a JASCO FP-8500 spectrofluorometer—equipped with a Hamamatsu R928 optional photomultiplier tube (operative range: 200–850 nm)—in Britton–Robinson buffer solutions35 with excitation at 450 nm, and corrected using a JASCO ESC-333 standard light source. Borate (0.1 M),36 phosphate (0.2 M)37 and citrate (0.2 M)38 buffer solutions were also used for evaluating the functions of 1, 3 and 5, respectively. After adjusting the pH at 20 °C, the sample was equilibrated at a specific temperature for at least 60 s. The sample temperature was controlled using a JASCO ETC-273T temperature controller. The fluorescence quantum yields (Φf) were determined using a JASCO FP-8500 spectrofluorometer with a JASCO ILF-835 integrating sphere unit.The fluorescence lifetimes were measured using a Horiba Jobin Yvon FluoroCube 3000U. The samples were excited at 456 nm with an LED (NanoLED-460, Horiba) at a repetition rate of 1 MHz. The temperature of the sample was controlled using a JASCO ETC-273T temperature controller. The obtained fluorescence decay curve was fitted by a double-exponential function using eqn (4).
I(t) = A1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ1) + A2 exp(−t/τ1) + A2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(−t/τ2) exp(−t/τ2) | (4) |
Fractional contributions (P1 and P2 for τ1 and τ2, respectively) and average fluorescence lifetimes were calculated using the following equations:
| P1 = A1τ1/(A1τ1 + A2τ2) × 100 | (5) |
| P2 = A2τ2/(A1τ1 + A2τ2) × 100 | (6) |
| τf = (A1τ12 + A2τ22)/(A1τ1 + A2τ2) | (7) |
Determination of pKa for the conjugate acid of the dimethylmonoalkylamino moiety
The pKa value of the conjugate acid of the dimethylmonoalkylamino moiety was determined via pH titration of the model fluorescent pH probe, DBD-DMAEA, exhibiting PET control-based fluorescence off–on switching. The apparent pKa values (apppKa) of DBD-DMAEA were determined in pure water, mixtures of water and methanol (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 w/w), and mixtures of water and ethanol (4
4 w/w), and mixtures of water and ethanol (4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 4 w/w) by fitting the pH values measured using a glass electrode (9618S-10D, Horiba)-equipped pH meter (LAQUA F-73, Horiba) and pH-dependent fluorescence intensity data using eqn (8).
4 w/w) by fitting the pH values measured using a glass electrode (9618S-10D, Horiba)-equipped pH meter (LAQUA F-73, Horiba) and pH-dependent fluorescence intensity data using eqn (8).| −log[(FImax − FI)/(FI − FImin)] = pH − apppKa | (8) |
| Solvent | D | apppKab | δ | pKa |
|---|---|---|---|---|
| a Dielectric constant.76 b Measured using a glass electrode pH meter standardized as per usual using aqueous buffer solutions. c Correction constant for a specific composition of a solvent medium.40 | ||||
| Water | 78.5 | 8.35 | — | 8.35 |
Water–methanol (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, w/w) 2, w/w) |
69.2 | 8.01 | 0.01 | 8.00 |
Water–methanol (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, w/w) 5, w/w) |
54.9 | 7.52 | 0.13 | 7.39 |
Water–methanol (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8, w/w) 8, w/w) |
40.1 | 6.84 | −0.06 | 6.90 |
Water–ethanol (8![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, w/w) 2, w/w) |
67.0 | 7.98 | −0.03 | 8.01 |
Water–ethanol (5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, w/w) 5, w/w) |
49.0 | 7.25 | 0.17 | 7.08 |
Water–ethanol (2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 8, w/w) 8, w/w) |
32.8 | 6.72 | 0.11 | 6.61 |
Logic operation in live HeLa cells
Fluorescence lifetime imaging was performed using a Leica TCS SP8-FALCON confocal laser scanning microscope with a time-correlated single photon counting (TCSPC) module. Cultured HeLa cells were washed with phosphate-buffered saline (PBS) and then treated with a 5% glucose solution containing 0.05 w/v% of 9. After incubation at room temperature for 10 min, the cells were washed twice with PBS and immersed in 50 mM phosphate buffer solutions (pH 5.5–8.0) containing 80 mM potassium chloride, 1 mM magnesium chloride and 20 μM nigericin.41 The treated HeLa cells were allowed to stand at room temperature for 5 min and observed at fixed temperatures under excitation by a PicoQuant PDL 800-B pulsed laser at 470 nm with a repetition rate of 20 MHz. The temperature of the microscope stage was controlled using Tokai Hit STXG-WSKMX-SET. Fluorescence emitted in the 500–700 nm range was captured, using an HC PL APO 63 × 1.40 NA oil CS2 objective lens (Leica) in 512 × 512 pixel format, with a pinhole size of 3 units. Samples were scanned at 400 Hz and fluorescence lifetime images were acquired by accumulating 50 times. To achieve a photon count rate of 0.5%–2% of the pulse count rate (2 × 107 Hz), the laser power was controlled. Fluorescence lifetime images were obtained using Leica Application Suite X in a fast FLIM mode.Results and discussion
Digital fluorescence responses of 1 to pH variations
First, the function of 1 as a fluorescent pH probe were demonstrated. The copolymers studied herein were prepared via random polymerization using AIBN. The characterization of unit compositions and molecular weights of the as-prepared copolymers are summarized in Table 1. The unit ratios of thermoresponsive, proton-binding and fluorescent units in the copolymers were equivalent to the corresponding monomer ratios in the feed. Fig. 2a shows the relation between the fluorescence intensity of 1 and temperature in Britton–Robinson buffer solutions at pH 2 and 12.42 The DMAPAM units in 1 are expected to be protonated at pH 2 and deprotonated at pH 12. Fig. 2a (closed circle) shows NIPAM units-induced thermoresponsive behavior of 1 at pH 12. In contrast, this behavior disappeared at pH 2 (open circle), as protonated 1 is too hydrophilic to be subjected to a heat-induced phase transition. The orange region shown in Fig. 2a suggests that the fluorescence off–on switching of 1 is induced by changing pH values in the range of approximately 45 °C–60 °C.Fig. 2b displays the fluorescence response of 1 to a pH variation. When the solution temperature was 50 °C (closed circle), the fluorescence intensity was pH dependent. The corresponding pH-dependent fluorescence spectra at 50 °C are shown in Fig. 2c. However, no fluorescence switching was observed when the solution temperature was 20 °C (open circle, Fig. 2b). Fig. 2a clarifies the difference between the fluorescence-switching behavior at 20 °C and 50 °C. Notably, the response curve of 1 at 50 °C to pH variation was remarkably sharp. Fluorescence switching of 1 was completed in a narrower pH range than those of conventional organic fluorescent pH probes. The response curve of 8-hydroxypyrene-1,3,6-trisulfonate (HPTS)43 (Fig. S1) is a representative of the latter pH probes.
To analyze the unusual pH-dependent fluorescence behavior of 1, we introduced an extended relation (eqn (9)), based on the Henderson–Hasselbalch equation.44
| −log[(FImax − FI)/(FI − FImin)] = a (pH − pKa) | (9) |
The fluorescence lifetime is a valuable detection parameter, especially in bioimaging, because it is independent of the fluorophore concentration and excitation source strength.46Fig. 2d shows the pH-dependent fluorescence lifetime of 1 at 50 °C, calculated based on the corresponding fluorescence decay curve fitted using a double-exponential function (Table S1). A related study using poly(NIPAM-co-DBD-AA)32 suggests that double components in the fluorescence decay curves of 1 correspond to the fluorophore (DBD-AA units) with and without hydrogen bonding to water molecules. Similar to fluorescence intensity (Fig. 2b), the fluorescence lifetime of 1 at 50 °C sharply responded to a pH variation in a narrow range, yielding a = 3.12 and apparent pKa = 8.47 according to eqn (9), where the fluorescence intensity data are replaced by the average fluorescence lifetime data (Fig. 2d). The pKa (8.47) of copolymer 1 calculated based on fluorescence lifetime measurements is slightly lower than the pKa (8.68) obtained by measuring the fluorescence intensity because of the mathematical weighing of components in obtaining these values. The fluorescence properties of 1 in the fluorescence “off” and “on” states are summarized in Table 3.
| Copolymer | State | pH | T (°C) |
λ
em![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) /nm
/nm |
Φ f |
τ
f![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) /ns
/ns |
k f/107 s−1 | k nr/107 s−1 |
|---|---|---|---|---|---|---|---|---|
| a Excited at 450 nm and corrected. b Average fluorescence lifetimes at λem. Calculated using double-exponential components (Table S4) and eqn (7). | ||||||||
| 1 | Off | 7 | 50 | 605 | 0.046 | 3.3 | 1.4 | 29.2 |
| On | 11 | 50 | 574 | 0.20 | 9.8 | 2.1 | 8.2 | |
| 2 | Off | 7 | 40 | 600 | 0.033 | 3.2 | 1.0 | 30.1 |
| On | 11 | 40 | 570 | 0.20 | 10.4 | 1.9 | 7.7 | |
| 3 | Off | 4 | 50 | 601 | 0.084 | 3.0 | 2.8 | 30.9 |
| On | 8 | 50 | 570 | 0.28 | 9.8 | 2.9 | 7.4 | |
| 4 | Off | 2 | 40 | 602 | 0.057 | 3.0 | 1.9 | 31.6 |
| On | 6 | 40 | 571 | 0.30 | 10.1 | 3.0 | 6.9 | |
| 5 | Off | 6 | 45 | 600 | 0.042 | 2.9 | 1.5 | 33.3 |
| On | 2 | 45 | 560 | 0.34 | 12.8 | 2.7 | 5.1 | |
| 6 | Off | 6 | 20 | 601 | 0.044 | 8.5 | 0.5 | 11.2 |
| On | 2 | 20 | 553 | 0.37 | 18.2 | 2.0 | 3.5 | |
| 7 | Off | 7 | 37 | 600 | 0.086 | 3.7 | 2.3 | 24.7 |
| On | 11 | 37 | 568 | 0.25 | 10.2 | 2.4 | 7.4 | |
Mechanism of 1 for producing sharp fluorescence responses to a pH variation
The mechanism of exceptional digital-type responses to a pH variation was investigated. 1 functions as a fluorescent pH probe because of the three-dimensional structural change that occurs owing to the variation in hydrophobicity/hydrophilicity of proton-binding units. This is entirely different from that of conventional small organic fluorescent pH probes, which are based on the electronic effects of an acid–base equilibrium on a fluorophore. Thus, we hypothesized that the simultaneous two physicochemical variations according to the pH-dependent polymeric structural change in 1 were involved in the digital-type responses: (i) microenvironmental polarity near the main chain of 1 and (ii) basicity of amino moieties in DMAPAM units in 1. Therefore, we experimentally investigated these physicochemical variations during the operation of 1.Fig. 3a displays the pH-dependent maximum emission wavelength (left axis) of 1 at 50 °C. Because the maximum emission wavelength of DBD-IA (Fig. 3b), a model of a DBD-AA unit, correlates well with the dielectric constant (D) of solvent (Table S2 and Fig. S2),47 the pH-dependent microenvironmental polarity near 1 during its function can be evaluated based on the maximum emission wavelength as a local D (right axis; Fig. 3a). Thus, the local D values near 1 are 58 and 7 in acidic and basic pH regions, respectively.
The basicity of the dimethylalkylamino group in DMAPAM units in 1 under high or low polarity was assessed using DBD-DMAEA31 (Fig. 3c) as a fluorescent pH probe bearing the identical dimethylalkylamino moiety. The fluorescence off–on switching principle of DBD-DMAEA is based on the control of PET,48 and the pKa value of the conjugate acid of DBD-DMAEA in water can be determined via fluorescence titration with varying pH (Fig. S3). Similarly, the pKa values of the conjugate acid of DBD-DMAEA in various media with different polarities were obtained using mixtures of water–methanol and water–ethanol (Table 2 and Fig. 3d). Fig. 3d shows that the basicity of the dimethylalkylamino moiety in DBD-DMAEA is reduced when the environmental polarity decreases. Limited reports on the decrease in pKa values for amines with decreasing environmental polarity are found for aniline,49 pyridine,49 1-benzylimidazole50 and propranolol.50
Fig. 3e explains the sharp fluorescence responses of 1 to pH variations. In an acidic solution (low pH), 1 is extended by protonated DMAPAM units, where local polarity is relatively high near the copolymer, and the fluorescence response follows the rightmost (blue) curve with a relatively high pKa. With increasing pH, protonated DMAPAM units are gradually neutralized, shrinking 1 and reducing the microenvironmental polarity near it. The fluorescence response curve is shifted leftward (purple line), accompanying a moderate pKa. In an alkaline (high pH) solution, the basicity of the dimethylalkylamino moiety in DMAPAM units further decreases because of the low microenvironmental polarity, and the fluorescence response curve shifts further left (red line). Thus, the digital fluorescence response curve of 1 is produced by moving on these shifting response curves with the pH-dependent pKa values of proton receptors (I → II → III, Fig. 3e).
Modulation of the operational pH range and switching direction of digital fluorescent polymeric pH probes
The elucidated operational mechanism of 1 enables the development of digital fluorescent polymeric pH probes that operate in various pH ranges by replacing DMAPAM units with different proton-binding units. Copolymers 2–4 contain amino moieties with different basicities: DEAPAM units in 2, MPAM units in 3 and DMAcAPAM units in 4 in the decreasing order of basicity of an amino group (Fig. 4a–c). The pKa values of conjugate acids of DEAPAM and MPAM in water are 10.3 and 7.0, respectively.51 Due to the electron-withdrawing CH2COOMe group of DMAcAPAM units, the pKa value of the conjugated acid of DMAcAPAM is further reduced.52Fig. 4a–c show the fluorescence responses of 2–4 to pH variations in Britton–Robinson buffer solutions at the predetermined fixed functional temperatures (Fig. S4a–c). Ideally, 2 (a = 4.15 ± 0.59; pKa = 9.13 ± 0.04), 3 (a = 2.54 ± 0.04; pKa = 6.16 ± 0.03), and 4 (a = 3.95 ± 0.29; pKa = 3.03 ± 0.01) displayed sharp digital responses in different operational pH ranges, depending on amino group basicity in proton-binding units.53 The tunable operational pH range is a distinct advantage of our polymeric design for digital fluorescent pH probes.The fluorescence off–on switching direction can also be reversed by replacing the proton-binding amino units in digital fluorescent polymeric pH probes with carboxylic acids. Copolymer 5 contains AA units as proton-releasing structures. After evaluating its functional temperature range (Fig. S4d), 5 showed a sharp fluorescence response to pH variation (|a| = 4.29 ± 0.38; pKa = 5.16 ± 0.14) in Britton–Robinson buffer solutions at 45 °C (Fig. 4d).54 Strong fluorescence appeared in the low pH region (in this switching direction, a in eqn (9) is negative). In contrast to proton-binding amino units, AA units in 5 are less polar in a high-acidic pH range to take the shrinking polymeric structure. Similar to the cause of the extreme sharp fluorescence responses of 1, the polarity-dependent acidity of the carboxylic group in AA units55 is likely a secret of the digital-type fluorescence responses of 5.
Copolymer 6 with VBA units also displayed digital fluorescence responses to pH variation (|a| = 5.58 ± 0.98; pKa = 5.09 ± 0.05) in Britton–Robinson buffer solutions at 20 °C (Fig. 4e and S4e). These results indicate the generality of the operational mechanism of digital fluorescent polymeric pH probes with acidic or basic structures as proton-releasing or receiving moieties. The fluorescence properties of copolymers 2–6 are summarized in Table 3.
Modulation of the operational temperature of digital fluorescent polymeric pH probes
Next, we investigated the effect of thermoresponsive units on the copolymer's function to tune the operational temperature range. We prepared copolymers 7 and 8, containing NNPAM and NTBAM units (Fig. 5), respectively (Table 1). The phase transition temperatures of NIPAM and NNPAM homopolymers in water are 31 °C and 22 °C, respectively, while the NTBAM homopolymer is too hydrophobic to dissolve in water, even at 0 °C.56Fig. 6a and b display the detailed temperature-dependent fluorescence intensity of 1 and 7 at pH 2 and 12, respectively. The fluorescence enhancement factors of 1 and 7 (Fig. 6c) indicate that the operational temperature range of 7 is lower than that of 1. Despite changing the thermoresponsive units, the digital behavior of the fluorescence response to pH variation was maintained in 7 (a = 2.78 ± 0.39; pKa = 8.58 ± 0.02) in Britton–Robinson buffer solutions at 37 °C (Fig. 6d). This result suggests that 7 is more suitable for experiments with mammalian cells than 1 in terms of an operational temperature range, although further tuning might be required for the function in the physiological pH range. The fluorescence properties of 7 are summarized in Table 3. However, the highly hydrophobic NTBAM prevents 8 from adopting an open form, even at pH 2 (Fig. S5). Nevertheless, moderate fluorescence switching can be observed by varying the pH from 2 to 12 at a low temperature (5 °C). The phase transition temperature of a thermoresponsive polymer can be more precisely tuned by mixing two thermoresponsive units.57 Thus, the functional temperature range of a digital fluorescent polymeric pH probe can be arbitrarily tuned by choosing appropriate thermoresponsive units.Additional relations between the chemical structures of components and the function of fluorescent copolymers
Apart from the fluorescent copolymers described herein, we synthesized additional fluorescent copolymers comprising various ratio of different units. Most of the experimental procedures and functional data of these copolymers are summarized in the SI.The effect of the unit ratio (thermoresponsive NIPAM unit![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) proton-binding DMAPAM unit) in poly(NIPAM-co-DMAPAM-co-DBD-AA) was investigated. 1 and copolymers S1–S6 were prepared with different NIPAM
proton-binding DMAPAM unit) in poly(NIPAM-co-DMAPAM-co-DBD-AA) was investigated. 1 and copolymers S1–S6 were prepared with different NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMAPAM ratios in the feed solution (Table S3). The actual composition ratios of NIPAM and DMAPAM units in S1–S6 were essentially the same as those of the corresponding monomers in the feed. The temperature-dependent fluorescence intensity of 1 and S1–S6 at pH 2 and 12 (Fig. 2a and S6) indicates that the optimized NIPAM
DMAPAM ratios in the feed solution (Table S3). The actual composition ratios of NIPAM and DMAPAM units in S1–S6 were essentially the same as those of the corresponding monomers in the feed. The temperature-dependent fluorescence intensity of 1 and S1–S6 at pH 2 and 12 (Fig. 2a and S6) indicates that the optimized NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) DMAPAM ratio in the feed is 90
DMAPAM ratio in the feed is 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10, resulting in a wide functional temperature range where significant fluorescence enhancement is observed owing to pH variations. This was also confirmed for poly(NIPAM-co-MPAM-co-DBD-AA) by preparing S7 with an 80
10, resulting in a wide functional temperature range where significant fluorescence enhancement is observed owing to pH variations. This was also confirmed for poly(NIPAM-co-MPAM-co-DBD-AA) by preparing S7 with an 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 NIPAM
20 NIPAM![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) MPAM ratio in the feed and comparing its fluorescence response with 2 (Fig. 4b and S7). Therefore, we fixed the monomer ratio in the feed for thermoresponsive and proton-binding units as 90
MPAM ratio in the feed and comparing its fluorescence response with 2 (Fig. 4b and S7). Therefore, we fixed the monomer ratio in the feed for thermoresponsive and proton-binding units as 90![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 herein.
10 herein.
Next, we investigated the effects of the length of a methylene chain of proton-binding units. Copolymers S8 and S9 comprising N-[2-(diethylamino)ethyl]acrylamide (DEAEAM) and N-(2-morpholinoethyl)acrylamide (MEAM) units (Fig. 5) with ethylene chains instead of propylene chains of DEAPAM and MPAM units in 2 and 3, respectively, were prepared (Table S3). The fluorescence responses of S8 and S9 (Fig. S8) are not significantly different from those of 2 and 3, indicating that the methylene chain length in pH-responsive units does not have a remarkable effect on the function of digital fluorescent pH probes.
Finally, for practical applications using digital fluorescent pH probes, the limitations of our polymeric design were investigated. Copolymers S10 and S11 comprising APAM and N-{3-[bis(2-hydroxyethyl)amino]propyl}acrylamide (HEAPAM) units (Fig. 5), respectively, were prepared (Table S3). The fluorescence responses of S10 and S11 (Fig. S9) indicated that they failed to function as digital fluorescent polymeric pH probes. S10 exhibits heat-induced fluorescence enhancement, even at pH 2, likely because of the insufficient hydrophilicity of the protonated monoalkyl amino group in the APAM units. At a selected temperature (40 °C), although moderate pH-dependent fluorescence switching occurs, it was not as sharp as that of digital fluorescent pH probes (Fig. S9a; a = 0.82; pKa = 9.32). Additionally, S11 did not exhibit a digital-type response (a = 0.99 ± 0.03; pKa = 7.40 ± 0.08), although fluorescence off–on switching occurred by varying pH at 50 °C (Fig. S9b). The operational behavior of S11 is likely because of hydroxy groups in HEAPAM units, which increase local hydrophilicity and disable the operational mechanism of digital fluorescent polymeric pH probes.
Molecular logic operation using a digital fluorescent pH probe in an aqueous solution and live cells
Considering that the developed digital fluorescent polymeric pH probes have the sensing ability to temperature variation, their function is utilized as a fluorescent logic gate,17–20 which can simultaneously sense two or more chemical/physical factors and distinguish complex environmental conditions. The practical applications of molecular logic gates have been extended to the molecular computational identification of small objects58 and medical science (diagnostics59 and therapy60,61). As an application of the as-prepared probe, an intracellular logic operation using copolymer 9 (Fig. 7a) comprising thermoresponsive NIPAM, proton-binding MPAM and fluorescent DBD-AA units was explored. Compared with 3 comprising identical units, 9 possessed substantially more DBD-AA units for live-cell imaging via fluorescence microscopy. | ||
| Fig. 7 AND logic operation using 9 in Britton–Robinson buffer solutions. (a) Chemical structure of 9. (b and c) Fluorescence spectra (0.001 w/v%, excited at 450 nm) (b), and fluorescence decay curves (0.02 w/v%, excited at 456 nm and emission collected over 500 nm) (c) of 9 under four different pH and temperature conditions (I–IV, see Table 4). | ||
The function of 9 as a fluorescent polymeric logic gate was evaluated in Britton–Robinson buffer solutions under four different pH and temperature conditions using a spectrofluorometer (Fig. 7b) and a TCSPC fluorescence lifetime spectrometer (Fig. 7c): pH (input1) was set at 2 (low, input1 = 0) or 12 (high, input1 = 1), and the temperature (input2) was set at 20 °C (low, input2 = 0) or 40 °C (high, input2 = 1). A strong fluorescence with long fluorescence decay occurs only when pH and temperature are high. As summarized in a truth table showing the input–output pattern (Table 4), the outputs of 9 (fluorescence intensity and lifetime) are switched on when both inputs are high and switched off under the other conditions. The response pattern of 9 corresponds to an AND logic gate. Notably, the extremely sharp responses of 9 to pH and temperature variations enable effective logic operations because such distinguishable output states with slight input differences are crucial for accurate performance, especially in decision making in medical applications62 and sequential input–output communication between multiple molecules.63,64
| Condition | Input1 (pH) | Input2 (T) | Output (RFI, τf![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a) a) |
λ
em![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) /nm
/nm |
Φ f | k f/107 s−1 | k nr/107 s−1 |
|---|---|---|---|---|---|---|---|
| a Average fluorescence lifetimes at λem. Calculated using double-exponential components (Table S4) and eqn (7). b Excited at 450 nm and corrected. | |||||||
| I | 0 (2) | 0 (20 °C) | 0 (0.18, 2.7 ns) | 604 | 0.046 | 1.7 | 35.3 |
| II | 1 (12) | 0 (20 °C) | 0 (0.19, 3.2 ns) | 604 | 0.054 | 1.7 | 30.0 |
| III | 0 (2) | 1 (40 °C) | 0 (0.19, 2.7 ns) | 604 | 0.050 | 1.9 | 34.9 |
| IV | 1 (12) | 1 (40 °C) | 1 (1.0, 8.3 ns) | 580 | 0.21 | 2.6 | 9.5 |
Finally, we used 9 to perform a logic operation inside live cells. Although intracellular logic operation is an appealing application of fluorescent logic gates, its realization has been limited for a long time.65,66 Nevertheless, the number of reports on intracellular logic operations has increased significantly within the past five years,67,68 primarily because of their importance, especially in medicine.69,70 Multiplex sensory molecules that can simultaneously monitor multiple chemical/physical parameters in different output modes31,71 are accelerating this trend. The inputs (pH and temperature) of 9 are endogenous chemical and physical parameters of live cells. The fluorescent polymeric logic gate 9 was smoothly incorporated into live HeLa cells from a culture medium by treatment at room temperature for 10 min. Cationizable MPAM units facilitated the spontaneous entry of 9 into living cells through the plasma membrane.72 After the incorporation, 9 remained in the cytoplasm without leaking. To evaluate the logic operation of 9 in live cells, bright-field (BF) and fluorescence lifetime images of live HeLa cells with 9 were recorded under four different pH (5.5 (low) and 8.0 (high)) and temperature (22 °C (low) and 37 °C (high)) conditions (Fig. 8). In these cellular experiments, intracellular pH and temperature were controlled by nigericin-containing phosphate buffer solutions41 and a stage heater, respectively. In live cell imaging, the exact intracellular concentration of a fluorescent probe cannot be determined because the cell size varies. Therefore, probe concentration–independent fluorescence lifetime is a more suitable detection parameter than fluorescence intensity in fluorescence cell imaging.73
Among four different conditions, an extended fluorescence lifetime (>7 ns) was observed at high pH (8.0) and high temperature (37 °C), as summarized in the truth table for 9 in HeLa cells (Table 5). The three-dimensional structural change in 9 to hydrophobic globules during the operation at high pH and temperature inside HeLa cells with high ionic strength was verified based on moderate aggregation of 9 (recognized as dots in the fluorescence lifetime image in case IV; Fig. 8) and extension of the fluorescence lifetime. As confirmed in Fig. S10, the AND function of 9 within cells was achieved through small variations in intracellular pH and temperature. Thus, the digital polymeric pH probe-based logic gate 9 is superior to conventional small molecule-based AND logic gates because it can produce a definite output response with slight input variations. Such efficient switching is a significant advantage of our polymeric design of a fluorescent logic gate, which enables solid molecular computation, even within biological milieus, where environmental fluctuations are restricted. Our results pave the way for the next stage of intracellular molecular computation, where the intelligence of chemical logic operations is combined with the elaborateness of biological fields to create new science.
Conclusions
We designed fluorescent polymeric digital pH probes comprising thermoresponsive, proton-binding, and environment-sensitive fluorescent units. The ultimate response efficiency of digital pH probes based on fluorescence switching achieved by a slight difference in proton concentrations was enabled using a polymeric design involving local hydrophobicity variation during operation. Similar sharp responses were observed in the hydrophobic effect-induced cooperativity of protein–ligand complexation.74,75 Thus, we assumed that macromolecules in an aqueous solution could exploit the change in local hydrophobicity to acquire indispensable sensitivity and selectivity in molecular recognition. However, their synthetic costs are significantly higher than those for small molecules. As the next steps for fluorescent digital probes, we will perform highly sensitive pH monitoring of real samples (water and biological cells) and extend the detection target to alkaline (Na+ or K+) ions, exploiting the flexibility of polymeric design.Author contributions
S. U.: writing – original draft, visualization, project administration, investigation, funding acquisition, data curation, conceptualization. E. H.: investigation. M. T.: investigation. K. O.: writing – review & editing, investigation.Conflicts of interest
There are no conflicts to declare.Data availability
Data for this study are available from UTokyo Repository at https://r.dl.itc.u-tokyo.ac.jp/esploro/outputs/9913110009301.Supplementary information (SI) is available. See DOI: https://doi.org/10.1039/d5an01187f.
Acknowledgements
We dedicate this paper to Prof. Amilra Prasanna de Silva in sincere appreciation for his pioneering work in establishing molecular logic gates and for his continuous, engaging discussions, even after the closure of his laboratory at Queen's University Belfast in 2022. We thank Dr M. Shibata and Ms. M. Okada (HORIBA, Ltd) for valuable instruction in pH measurements of mixed solvents. S. U. thanks Japan Society for the Promotion of Science (Grant-in-Aid for Challenging Exploratory Research, No. 24655059), Foundation for Promotion of Material Science and Technology of Japan, Ogasawara Foundation for the Promotion of Science and Engineering, and Iketani Science and Technology Foundation (No. 0251023-A) for financial support.References
- A. P. de Silva, H. Q. N. Gunaratne, T. Gunnlaugsson, A. J. M. Huxley, C. P. McCoy, J. T. Rademacher and T. E. Rice, Signaling recognition events with fluorescent sensors and switches, Chem. Rev., 1997, 97, 1515–1566 CrossRef PubMed.
- K. P. Carter, A. M. Young and A. E. Palmer, Fluorescent sensors for measuring metal ions in living systems, Chem. Rev., 2014, 114, 4564–4601 CrossRef PubMed.
- S.-H. Park, N. Kwon, J.-H. Lee, J. Yoon and I. Shin, Synthetic ratiometric fluorescent probes for detection of ions, Chem. Soc. Rev., 2020, 49, 143–179 RSC.
- J. Han and K. Burgess, Fluorescent indicators for intracellular pH, Chem. Rev., 2010, 110, 2709–2728 CrossRef PubMed.
- R. P. Bell, The proton in chemistry, Cornell University Press, New York, 1959 Search PubMed.
- F. M. Harold, The vital force: a study of bioenergetics, Freeman, New York, 1986 Search PubMed.
- J.-T. Hou, W. X. Ren, K. Li, J. Seo, A. Sharma, X.-Q. Yu and J. S. Kim, Fluorescent bioimaging of pH: from design to applications, Chem. Soc. Rev., 2017, 46, 2076–2090 RSC.
- The change in ICT characteristics is also involved in ratiometric fluorescent probes such as seminaphtorhodafluor-1 (SNARF-1). See CrossRef PubMed; O. Seksek, N. Henry-Toulmé, F. Sureau and J. Bolard, SNARF-1 as an intracellular pH indicator in laser microspectrofluorometry: a critical assessment, Anal. Biochem., 1991, 193, 49–54 CrossRef PubMed.
- G. Gabor and D. R. Walt, Sensitivity enhancement of fluorescent pH indicators by inner filter effects, Anal. Chem., 1991, 63, 793–796 CrossRef.
- K. Zhou, Y. Wang, X. Huang, K. Luby-Phelps, B. D. Sumer and J. Gao, Tunable, ultrasensitive pH-responsive nanoparticles targeting specific endocytic organelles in living cells, Angew. Chem., Int. Ed., 2011, 50, 6109–6114 CrossRef PubMed.
- K. Zhou, H. Liu, S. Zhang, X. Huang, Y. Wang, G. Huang, B. D. Sumer and J. Gao, Multicolored pH-tunable and activatable fluorescence nanoplatform responsive to physiologic pH stimuli, J. Am. Chem. Soc., 2012, 134, 7803–7811 CrossRef PubMed.
- X. Ma, Y. Wang, T. Zhao, Y. Li, L.-C. Su, Z. Wang, G. Huang, B. D. Sumer and J. Gao, Ultra-pH-sensitive nanoprobe library with broad pH tunability and fluorescence emissions, J. Am. Chem. Soc., 2014, 136, 11085–11092 CrossRef PubMed.
- I. V. Nesterova and E. E. Nesterov, Rational design of highly responsive pH sensors based on DNA i-motif, J. Am. Chem. Soc., 2014, 136, 8843–8846 CrossRef PubMed.
- X. Luo, H. Yang, H. Wang, Z. Ye, Z. Zhou, L. Gu, J. Chen, Y. Xiao, X. Liang, X. Qian and Y. Yang, Highly sensitive Hill-type small-molecule pH probe that recognizes the reversed pH gradient of cancer cells, Anal. Chem., 2018, 90, 5803–5809 CrossRef PubMed.
- Y. Xiao, F. Hu, X. Luo, M. Zhao, Z. Sun, X. Qian and Y. Yang, Modulating the pKa values of Hill-type pH probes for biorelevant acidic pH range, ACS Appl. Bio Mater., 2021, 4, 2097–2103 CrossRef PubMed.
- F. Hu, Y. Huang, Y. Xiao, Y. Li, X. Luo, X. Qian and Y. Yang, A dual-channel Hill-type small-molecule pH probe, Anal. Methods, 2021, 13, 3012–3016 RSC.
- A. P. de Silva and S. Uchiyama, Molecular logic and computing, Nat. Nanotechnol., 2007, 2, 399–410 Search PubMed.
- A. P. de Silva, Molecular logic-based computation, Royal Society of Chemistry, Cambridge, 2013 Search PubMed.
- S. Erbas-Cakmak, S. Kolemen, A. C. Sedgwick, T. Gunnlaugsson, T. D. James, J. Yoon and E. U. Akkaya, Molecular logic gates: the past, present and future, Chem. Soc. Rev., 2018, 47, 2228–2248 Search PubMed.
- C.-Y. Yao, H.-Y. Lin, H. S. N. Crory and A. P. de Silva, Supra-molecular agents running tasks intelligently (SMARTI): recent developments in molecular logic-based computation, Mol. Syst. Des. Eng., 2020, 5, 1325–1353 RSC.
- Y. Wang, K. Zhou, G. Huang, C. Hensley, X. Huang, X. Ma, T. Zhao, B. D. Sumer, R. J. DeBerardinis and J. Gao, A nanoparticle-based strategy for the imaging of a broad range of tumours by nonlinear amplification of microenvironment signals, Nat. Mater., 2014, 13, 204–212 Search PubMed.
- S. Uchiyama and Y. Makino, Digital fluorescent pH sensors, Chem. Commun., 2009, 2646–2648 RSC.
- S. Fujishige, K. Kubota and I. Ando, Phase transition of aqueous solutions of poly(N-isopropylacrylamide) and poly(N-isopropylmethacrylamide), J. Phys. Chem., 1989, 93, 3311–3313 CrossRef.
- H. G. Schild, Poly(N-isopropylacrylamide): experiment, theory and application, Prog. Polym. Sci., 1992, 17, 163–249 Search PubMed.
- N. A. Shaibie, N. A. Ramli, N. D. F. M. Faizal, T. Srichana and M. C. I. M. Amin, Poly(N-isopropylacrylamide)-based polymers: recent overview for the development of temperature-responsive drug delivery and biomedical applications, Macromol. Chem. Phys., 2023, 224, 2300157 Search PubMed.
- M. Irie, Y. Misumi and T. Tanaka, Stimuli-responsive polymers: chemical induced reversible phase separation of an aqueous solution of poly(N-isopropylacrylamide) with pendent crown ether groups, Polymer, 1993, 34, 4531–4535 CrossRef.
- C. Gota, S. Uchiyama and T. Ohwada, Accurate fluorescent polymeric thermometers containing an ionic component, Analyst, 2007, 132, 121–126 Search PubMed.
- S. Uchiyama, Intracellular thermometry with fluorescent molecular thermometers, Wiley-VCH, Weinheim, 2024 Search PubMed.
- K. Okabe and S. Uchiyama, Intracellular thermometry uncovers spontaneous thermogenesis and associated thermal signaling, Commun. Biol., 2021, 4, 1377 CrossRef PubMed.
- Y. Akiyama, K. Yoshizako, Y. Hasegawa and T. Okano, Temperature-responsive polymer compound and process for producing the same, International PatentWO 00/44800, Aug 3, 2000 Search PubMed.
- S. Uchiyama, K. Iwai and A. P. de Silva, Multiplexing sensory molecules map protons near micellar membranes, Angew. Chem., Int. Ed., 2008, 47, 4667–4669 Search PubMed.
- C. Gota, S. Uchiyama, T. Yoshihara, S. Tobita and T. Ohwada, Temperature-dependent fluorescence lifetime of a fluorescent polymeric thermometer, poly(N-isopropylacrylamide), labeled by polarity and hydrogen bonding sensitive 4-sulfamoyl-7-aminobenzofurazan, J. Phys. Chem. B, 2008, 112, 2829–2836 CrossRef PubMed.
- Y. Maeda, T. Nakamura and I. Ikeda, Changes in the hydration states of poly(N-alkylacrylamide)s during their phase transitions in water observed by FTIR spectroscopy, Macromolecules, 2001, 34, 1391–1399 CrossRef.
- N. A. A. El-Ghany, M. S. A. Aziz, M. M. Abdel-Aziz and Z. Mahmoud, Antimicrobial and swelling behaviors of novel biodegradable corn starch grafted/poly(4-acrylamidobenzoic acid) copolymers, Int. J. Biol. Macromol., 2019, 134, 912–920 CrossRef PubMed.
- H. T. S. Britton and R. A. Robinson, Universal buffer solutions and the dissociation constant of veronal, J. Chem. Soc., 1931, 1456–1462 RSC.
- V. E. Bower and R. G. Bates, pH values of the Clark and Lubs buffer solutions at 25 °C, J. Res. Natl. Bur. Stand., 1955, 55, 197–200 CrossRef.
- G. Gomori, Preparation of buffers for use in enzyme studies, Methods Enzymol., 1955, 1, 138–146 Search PubMed.
- G. Gottschalk, Tartrat- und citratpuffer, Fresenius‘ Z. Anal. Chem., 1961, 183, 420–426 CrossRef.
- R. G. Bates, M. Paabo and R. A. Robinson, Interpretation of pH measurements in alcohol-water solvents, J. Phys. Chem., 1963, 67, 1833–1838 CrossRef.
- R. G. Bates, Determination of pH. Theory and practice, John Wiley & Sons, New York, 1973, pp. 243–249 Search PubMed.
- B. A. Webb, F. M. Aloisio, R. A. Charafeddine, J. Cook, T. Wittmann and D. L. Barber, pHLARE: a new biosensor reveals decreased lysosome pH in cancer cells, Mol. Biol. Cell, 2021, 32, 131–142 CrossRef PubMed.
- The Britton–Robinson buffer was primarily used in this study due to its universal characteristic for a wide pH range from 2 to 12. The pH values indicated in this paper were all measured at 20 °C and uncorrected. The pH values of the Britton–Robinson buffer solutions are temperature-dependent, especially in the alkaline region (i.e., 12.0 and 11.1 at 20 and 60 °C, respectively). Nevertheless, the fluorescence properties of our copolymers were not influenced by this temperature-dependent pH variation because the functional pH ranges of our copolymers were out of the region.
- O. S. Wolfbeis, E. Fürlinger, H. Kroneis and H. Marsoner, Fluorimetric analysis 1. A study on fluorescent indicators for measuring near neutral (“physiological”) pH-values, Fresenius‘ Z. Anal. Chem., 1983, 314, 119–124 CrossRef.
- H. N. Po and N. M. Senozan, The Henderson-Hasselbalch equation: its history and limitations, J. Chem. Educ., 2001, 78, 1499–1503 CrossRef.
- The sharp fluorescence response of 1 to pH variation was also confirmed in 0.1 M borate buffer solutions (a = 2.76, pKa = 8.41). See Fig. S11a.
- M. Y. Berezin and S. Achilefu, Fluorescence lifetime measurements and biological imaging, Chem. Rev., 2010, 110, 2641–2684 CrossRef PubMed.
- S. Uchiyama, K. Kimura, C. Gota, K. Okabe, K. Kawamoto, N. Inada, T. Yoshihara and S. Tobita, Environment-sensitive fluorophores with benzothiadiazole and benzoselenadiazole structures as candidate components of a fluorescent polymeric thermometer, Chem. – Eur. J., 2012, 18, 9552–9563 CrossRef PubMed.
- A. P. de Silva, T. S. Moody and G. D. Wright, Fluorescent PET (Photoinduced Electron Transfer) sensors as potent analytical tools, Analyst, 2009, 134, 2385–2393 RSC.
- M. S. K. Niazi and J. Mollin, Dissociation constants of some amino acid and pyridinecarboxylic acids in ethanol-H2O mixtures, Bull. Chem. Soc. Jpn., 1987, 60, 2605–2610 CrossRef.
- A. Avdeef, J. E. A. Comer and S. Thomson, pH-Metric log P. 3. Glass electrode calibration in methanol–water, applied to pKa determination of water-insoluble substances, Anal. Chem., 1993, 65, 42–49 CrossRef.
- M. Chiari, C. Ettori and P. G. Righetti, Capillary zone electrophoresis of acrylamido buffers for isoelectric focusing in immobilized pH gradients, J. Chromatogr., 1991, 559, 119–131 CrossRef.
- The pKa value of the conjugate acid of DMAcAPAM was calculated to be 3.4 by ChemDraw® Professional ver. 16.
- The sharp fluorescence response of 3 to pH variation was also confirmed in 0.2 M phosphate buffer solutions (a = 2.34, pKa = 5.97). See Fig. S11b.
- The sharp fluorescence response of 5 to pH variation was also confirmed in 0.2 M citrate buffer solutions (|a| = 2.29, pKa = 5.40). See Fig. S11c.
- E. Rossini, A. D. Bochevarov and E. W. Knapp, Empirical conversion of pKa values between different solvents and interpretation of the parameters: application to water, acetonitrile, dimethyl sulfoxide, and methanol, ACS Omega, 2018, 3, 1653–1662 Search PubMed.
- S. Ito, Phase transition of aqueous solution of poly(N-alkylacrylamide) derivatives. Effects of side chain structure, Kobunshi Ronbunshu, 1989, 46, 437–443 CrossRef.
- S. Uchiyama, Y. Matsumura, A. P. de Silva and K. Iwai, Modulation of the sensitive temperature range of fluorescent molecular thermometers based on thermoresponsive polymers, Anal. Chem., 2004, 76, 1793–1798 CrossRef PubMed.
- A. P. de Silva, M. R. James, B. O. F. McKinney, D. A. Pears and S. M. Weir, Molecular computational elements encode large populations of small objects, Nat. Mater., 2006, 5, 787–790 CrossRef PubMed.
- T. Konry and D. R. Walt, Intelligent medical diagnostics via molecular logic, J. Am. Chem. Soc., 2009, 131, 13232–13233 CrossRef PubMed.
- R. J. Amir, M. Popkov, R. A. Lerner, C. F. Barbas III and D. Shabat, Prodrug activation gated by a molecular “OR” logic trigger, Angew. Chem., Int. Ed., 2005, 44, 4378–4381 CrossRef PubMed.
- S. Ozlem and E. U. Akkaya, Thinking outside the silicon box: molecular AND logic as an additional layer of selectivity in singlet oxygen generation for photodynamic therapy, J. Am. Chem. Soc., 2009, 131, 48–49 CrossRef PubMed.
- J. Halámek, J. R. Windmiller, J. Zhou, M.-C. Chuang, P. Santhosh, G. Strack, M. A. Arugula, S. Chinnapareddy, V. Bocharova, J. Wang and E. Katz, Multiplexing of injury codes for the parallel operation of enzyme logic gates, Analyst, 2010, 135, 2249–2259 RSC.
- S. Silvi, E. C. Constable, C. E. Housecroft, J. E. Beves, E. L. Dunphy, M. Tomasulo, F. M. Raymo and A. Credi, All-optical integrated logic operations based on chemical communication between molecular switches, Chem. – Eur. J., 2009, 15, 178–185 CrossRef PubMed.
- E. T. Ecik, A. Atilgan, R. Guliyev, T. B. Uyar, A. Gumus and E. U. Akkaya, Modular logic gates: cascading independent logic gates via metal ion signals, Dalton Trans., 2014, 43, 67–70 RSC.
- D. P. Murale, H. Liew, Y.-H. Suh and D. G. Churchill, Mercuric-triggered hydrogen peroxide “turn-on” fluorescence detection in neuronal cells with novel fluorescein-based probe obtained in one pot, Anal. Methods, 2013, 5, 2650–2652 RSC.
- I. Takashima, R. Kawagoe, I. Hamachi and A. Ojida, Development of an AND logic-gate-type fluorescent probe for ratiometric imaging of autolysosome in cell autophagy, Chem. – Eur. J., 2015, 21, 2038–2044 CrossRef PubMed.
- J. Zheng, Q. Wang, L. Shi, P. Peng, L. Shi and T. Li, Logic-gated proximity aptasensing for cell-surface real-time monitoring of apoptosis, Angew. Chem., Int. Ed., 2021, 60, 20858–20864 CrossRef PubMed.
- Y. Shen, W. Li, Z. Zhou, J. Xu, Y. Li, H. Li, X. Zheng, S. Liu, X.-B. Zhang and L. Yuan, Dual-locked fluorescent probes activated by aminopeptidase N and the tumor redox environment for high-precision imaging of tumor boundaries, Angew. Chem., Int. Ed., 2024, 63, e202406332 CrossRef PubMed.
- L. Wu, J. Huang, K. Pu and T. D. James, Dual-locked spectroscopic probes for sensing and therapy, Nat. Rev. Chem., 2021, 5, 406–421 CrossRef PubMed.
- X. Zhang, T. Xiu, H. Wang, H. Wang, P. Li and B. Tang, Recent progress in the development of small-molecule double-locked logic gate fluorescence probes, Chem. Commun., 2023, 59, 11017–11027 RSC.
- L. Wu, J. Liu, X. Tian, R. R. Groleau, B. Feng, Y. Yang, A. C. Sedgwick, H.-H. Han, Y. Wang, H.-M. Wang, F. Huang, S. D. Bull, H. Zhang, C. Huang, Y. Zang, J. Li, X.-P. He, P. Li, B. Tang, T. D. James and J. L. Sessler, Dual-channel fluorescent probe for the simultaneous monitoring of peroxynitrite and adenosine-5′-triphosphate in cellular applications, J. Am. Chem. Soc., 2022, 144, 174–183 CrossRef PubMed.
- T. Tsuji, S. Yoshida, A. Yoshida and S. Uchiyama, Cationic fluorescent polymeric thermometers with the ability to enter yeast and mammalian cells for practical intracellular temperature measurements, Anal. Chem., 2013, 85, 9815–9823 CrossRef PubMed.
- W. Becker, Fluorescence lifetime imaging – techniques and applications, J. Microsc., 2012, 247, 119–136 CrossRef PubMed.
- D. H. Williams and B. Bardsley, Estimating binding constants – The hydrophobic effect and cooperativity, Perspect. Drug Discovery Des., 1999, 17, 43–59 CrossRef.
- L. Jiang, S. Cao, P. P.-H. Cheung, X. Zheng, C. W. T. Leung, Q. Peng, Z. Shuai, B. Z. Tang, S. Yao and X. Huang, Realtime monitoring of hydrophobic aggregation reveals a critical role of cooperativity in hydrophobic effect, Nat. Commun., 2017, 8, 15639 Search PubMed.
- G. Åkerlöf, Dielectric constants of some organic solvent-water mixtures at various temperatures, J. Am. Chem. Soc., 1932, 54, 4125–4139 CrossRef.
| This journal is © The Royal Society of Chemistry 2026 |