In situ degradation and radiation-responsive behavior of metal organic frameworks under high-energy irradiation: implications for sensing and shielding
Godwin A.
Udourioh
 a,
Samuel E.
Egga
a,
Samuel E.
Egga
 b and
Moses M.
Solomon
b and
Moses M.
Solomon
 *c
*c
aAnalytical/Industrial/Material Chemistry Research Group, Department of Pure and Applied Chemistry, Veritas University Abuja, P.O. Box 6523, Garki, Abuja, Nigeria
bDepartment of Chemistry, Faculty of Natural Sciences, University of Jos, P.M.B. 2084, Jos, Plateau State, Nigeria
cFaculty of Science and Engineering, University of Nottingham Ningbo China, Ningbo 315100, Zhejiang Province, China. E-mail: moses.solomon@nottingham.edu.cn
First published on 13th November 2025
Abstract
This review examines the promise and challenges of metal–organic frameworks (MOFs) in high-radiation environments, focusing on their sensing and shielding applications. In radiation detection, specific MOF architectures, such as Tb–Cu4I4 clusters, achieve high light yields of ∼29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 photons MeV−1 and spatial resolutions of 12.6 lp mm−1, enabling low detection limits as sensitive as 23 nGy s−1 in flexible composite screens. Conversely, for dosimetry, actinide-based systems like U-Cbdcp show linear luminescence quenching across a wide dynamic range from 10 to 4700 Gy (R2 = 0.999). The structural resilience of MOFs under irradiation is a key differentiator. Frameworks such as MIL-100(Fe/Cr) retain crystallinity and over 95% of their original surface area after 1 MGy γ-dose, while thorium-based TOF-16 withstands doses up to 4 MGy. However, stability is not uniform; ZIF-8 suffers a ∼55% BET surface area loss at 1.75 MGy, highlighting a strong dependence on metal-node and linker chemistry. Despite these advances, critical knowledge gaps exist and could impede practical deployment. A systematic, mechanistic understanding of in situ degradation pathways, such as linker scission and defect accumulation, and their quantitative impact on functional performance is lacking. Furthermore, radiation shielding efficacy demands not only high attenuation but also mechanical integrity, a domain where MOF composites are underexplored. This review provides a design roadmap to address these challenges, advocating for linker engineering with radiation-resistant π-systems, post-synthetic defect healing, and the creation of hybrid MOF-composite architectures. By integrating advanced in situ characterization with predictive computational models, this review charts a course for engineering next-generation, radiation-hardened MOFs, transforming them from laboratory curiosities into robust materials for nuclear, medical, and aerospace applications.
400 photons MeV−1 and spatial resolutions of 12.6 lp mm−1, enabling low detection limits as sensitive as 23 nGy s−1 in flexible composite screens. Conversely, for dosimetry, actinide-based systems like U-Cbdcp show linear luminescence quenching across a wide dynamic range from 10 to 4700 Gy (R2 = 0.999). The structural resilience of MOFs under irradiation is a key differentiator. Frameworks such as MIL-100(Fe/Cr) retain crystallinity and over 95% of their original surface area after 1 MGy γ-dose, while thorium-based TOF-16 withstands doses up to 4 MGy. However, stability is not uniform; ZIF-8 suffers a ∼55% BET surface area loss at 1.75 MGy, highlighting a strong dependence on metal-node and linker chemistry. Despite these advances, critical knowledge gaps exist and could impede practical deployment. A systematic, mechanistic understanding of in situ degradation pathways, such as linker scission and defect accumulation, and their quantitative impact on functional performance is lacking. Furthermore, radiation shielding efficacy demands not only high attenuation but also mechanical integrity, a domain where MOF composites are underexplored. This review provides a design roadmap to address these challenges, advocating for linker engineering with radiation-resistant π-systems, post-synthetic defect healing, and the creation of hybrid MOF-composite architectures. By integrating advanced in situ characterization with predictive computational models, this review charts a course for engineering next-generation, radiation-hardened MOFs, transforming them from laboratory curiosities into robust materials for nuclear, medical, and aerospace applications.
1. Introduction
Metal–organic frameworks (MOFs) are a versatile class of porous crystalline materials constructed from inorganic secondary building units (SBUs) and organic linkers, enabling fine-tunability of topology, porosity, and function.1–3 Their modularity has made them highly attractive for gas storage, separation, catalysis, and sensing.1,4–6 In the context of extreme environments, particularly ionizing radiation, MOFs present dual potential: as functional materials for radiation detection and as structurally engineered systems for radiation shielding.MOFs’ high porosity and chemical diversity facilitate the integration of active functionalities, including scintillation centers and conductive pathways. For instance, the scintillating high-Z metal-centered MOF, [Pb4(2,6-ndc)3Cl2]n (SMOF-4), where 2,6-ndc2− = 2,6-naphthalene dicarboxylate synthesized by Lu et al.7 was reported to exhibit a calculated theoretical linear attenuation coefficient of 9.27 cm−1 at 100 keV based on XCOM simulations,8,9 surpassing traditional detectors like Thallium-doped Cesium Iodide (CsI:Tl) and amorphous phase (non-crystalline, glassy) selenium (α-Se), which offer approximately 2.1 and 0.537 cm−2 g−1, respectively9 (Fig. 1). Although this simulation data represents an upper bound for optimized dense MOF monoliths, such architectures offer opportunities for lightweight, solution-processable X-ray detection platforms. Evidence of MOF resilience under irradiation has emerged from the current demonstrations of radiation stability. For example, a chromium-based MOF, MIL-101(Cr), showed slight chemical changes up to 30 kGy of γ-radiation by FTIR analysis.10 A thorium-containing MOF (TOF-16) maintained bulk crystallinity up to 4 MGy of γ-rays and began to degrade only under 5 MeV He2+ ion irradiation at approximately 15 MGy.11 Prototypical MOFs, including ZIF-8, UiO-66, and HKUST-1, have also displayed notable radiation resistance,12,13 challenging previous assumptions of MOF fragility. While these studies highlight MOFs’ promise, many questions remain. They are limited to selected frameworks, radiation types, and conditions. The systematic understanding of in situ structural changes, dose-dependent degradation mechanisms, and functional outcomes such as luminescence modulation, conductivity changes is lacking. Moreover, practical radiation shielding demands not only immediate stability but also mechanical robustness and sustained attenuation. These are domains where MOFs are yet to be benchmarked against industrial standards.
 | ||
| Fig. 1 An analysis of MOF attenuation for X-ray detection. (a) High-throughput screening of a MOF database identifies candidates for strong attenuation, plotted by atomic number, density, and surface area. (b) Linear attenuation coefficients of MOFs and commercial materials across X-ray energies from 10 to 500 keV. (c) Density and attenuation length of MOFs versus commercial materials for 100% attenuation at 100 keV. Reproduced with permission from Salway et al.9 © The Royal Society of Chemistry 2025. | ||
Despite rapid progress, critical knowledge gaps remain at the interface of fundamental radiation chemistry and the practical deployment of MOFs in radiation technologies. Current studies rarely provide comparative insight into how diverse secondary building units and framework topologies evolve under high-energy irradiation, particularly with respect to bond scission, defect accumulation, and amorphization. Moreover, the connection between these degradation pathways and functional outcomes such as scintillation quenching, conductivity changes, or shielding performance has not been systematically established under operationally relevant doses. Also, rational design strategies that could enhance radiation tolerance, including linker optimization, defect engineering, and composite integration, remain underexplored. This review examines the structural radiation responses and emerging application scenarios in sensing and shielding, while articulating clear routes for engineering next-generation MOFs. We aim to provide a design roadmap rooted in mechanistic understanding, supported by quantitative analyses and comparative data across MOF families.
2. Fundamentals of radiation–MOF interactions
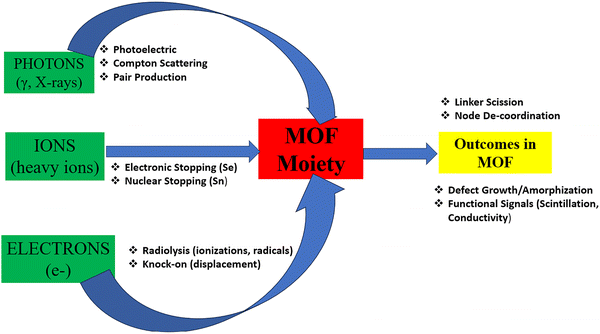
The interaction of ionizing radiation with MOFs is governed by how photons, electrons, and ions deposit energy into their hybrid inorganic–organic lattices.14,15 Unlike dense ceramics or metals, MOFs possess high porosity, long-range order, and chemically diverse linkers, producing distinct pathways for energy transfer, defect formation, and amorphization. Upon irradiation, energy deposition triggers electronic excitations, ionization cascades, and localized heating, which can induce bond scission, defect growth, or complete amorphization, depending on the dose and radiation type.16 Understanding these processes is central to correlating structural stability with functional performance, particularly when MOFs are deployed as scintillators, dosimeters, or lightweight radiation shields. Fig. 2 schematically maps the principal photon, electron, and ion interaction channels and their outcomes (linker scission, defect growth, functional signal, etc.).2.1. Photons, electrons, and ions: energy deposition in porous frameworks
Ionizing radiation interacts with matter through processes whose probabilities are energy/composition dependent. For photons (X/γ), the dominant processes across 1 keV–20 MeV are the photoelectric effect, Compton scattering, and pair production. Their relative weights scale strongly with atomic number (Z) and photon energy (E), which is quantified by the mass attenuation coefficient (µ/ρ).8,17 At E ≤ 100 keV, the photoelectric effect (∝Zn/E3, n ∼ 4–5) favours high Z-nodes (e.g., Bi, Th, Pb, Hf), while Compton scattering dominates in the 0.1–5 MeV window. At E ≥ 1.022 MeV, pair production contributes increasingly.8,17Primarily, electrons transfer energy through inelastic interactions, producing secondary electrons and radicals (radiolysis), with knock-on atomic displacements possible above material-specific thresholds.18–20 In porous, often insulating MOFs, radiolysis frequently dominates over knock-on under typical transmission electron microscopy (TEM) voltages (60–200 kV), though conductive 2D MOFs can suppress radiolysis, revealing knock-on limits.18,21
In MOF research, stopping and range of ions in matter (SRIM) is applied to model how protons or heavy ions deposit energy in porous frameworks, helping to estimate track formation, amorphization thresholds, and defect densities under irradiation.22 SRIM provides benchmark stopping powers and ranges, widely used to model dose profiles and displacement cascades in crystalline/porous solids.22 Ions (α, heavy ions) lose energy through electronic stopping power (Se) and nuclear stopping power (Sn).
The key governing relations include:
(i) Beer–Lambert attenuation for photons:
| I(E, x) = I0(E) exp[−µ(E)·x] | (1) |
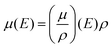 | (1a) |
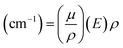 , where
, where 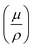 is the mass attenuation coefficient (cm2 g−1) and ρ is the density. The expression quantifies exponential photon attenuation. In any case, high-Z nodes in MOFs increase
is the mass attenuation coefficient (cm2 g−1) and ρ is the density. The expression quantifies exponential photon attenuation. In any case, high-Z nodes in MOFs increase 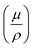 , improving attenuation and scintillation efficiency.17,18
, improving attenuation and scintillation efficiency.17,18
(ii) Absorbed dose (D) in Gray (Gy):
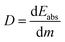 | (2) |
(iii) Ion stopping (electronic and nuclear):
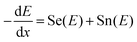 | (3) |
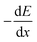 is the stopping power (KeV µm−1), Se(E) – electronic stopping (energy loss to electrons), Sn(E) – nuclear stopping (elastic collision with nuclei). The expression describes how ions lose energy in matter. In MOFs, high Se produces an ionization track, driving amorphization; Sn leads to atom displacement and cascades.15,18 The overall implication for MOFs utilization is that high-Z clusters (e.g., Hf6, Th-oxo) amplify photoelectric absorption below ∼100 keV, increasing both attenuation (shielding) and scintillation yields when coupled to emissive linkers. Conversely, organic linkers and porosity govern radical chemistry and defect formation under electrons/gamma-rays (See Sections 2.3 and 2.4 for more explanation).15 The interaction regimes and MOFs’ relevant consequences are presented in Table 1.
is the stopping power (KeV µm−1), Se(E) – electronic stopping (energy loss to electrons), Sn(E) – nuclear stopping (elastic collision with nuclei). The expression describes how ions lose energy in matter. In MOFs, high Se produces an ionization track, driving amorphization; Sn leads to atom displacement and cascades.15,18 The overall implication for MOFs utilization is that high-Z clusters (e.g., Hf6, Th-oxo) amplify photoelectric absorption below ∼100 keV, increasing both attenuation (shielding) and scintillation yields when coupled to emissive linkers. Conversely, organic linkers and porosity govern radical chemistry and defect formation under electrons/gamma-rays (See Sections 2.3 and 2.4 for more explanation).15 The interaction regimes and MOFs’ relevant consequences are presented in Table 1.
| Radiation | Typical energy window | Dominant interaction (s) | Key material parameters | Primary products/effects | MOF-relevant outcomes | Ref. |
|---|---|---|---|---|---|---|
| X/γ photons | 1 keV–20 MeV | Photoelectric (low E), Compton (mid E), pair production (high E) | µ/ρ(E), Z, ρ | Fast electrons, secondary photons, radical formation (indirect), heat | Attenuation/scintillation; indirect radiolysis; degradation depends on dose-rate | 17 |
| Electrons (TEM/e− beam) | 60–300 kV | Inelastic ionization, knock-on | Conductivity, bond energies, threshold Ed | Secondary e−, radicals, atomic displacements | Radiolysis-driven amorphization unless conductive; imaging artifacts | 18 |
| Ions (α, heavy ions) | 100 keV–MeV | Se, Sn stopping cascade | Se, Sn (SRIM), density | Dense ionization tracks, cascades, and sputtering | Localized damage; color centers; shielding vs. α negligible without encapsulation | 22 |
2.2. Influence of energy, dose, dose-rate, and sample form
The radiation response of MOFs is a complex and multivariate property that extends far beyond the simplistic metric of total absorbed dose, being critically governed by a synergistic interplay of dose rate, energy spectrum, incident particle type, and the physical sample form, each parameter capable of dramatically altering the ultimate material stability23 and thus its viability for applications. The foundational principle is that radiation-induced damage arises when the energy of the incoming radiation exceeds a critical value, such that the metal nodes or organic linkers within the MOFs undergo ionization of their outermost electrons or π-electrons, resulting in the generation of electron–hole pairs and excited states that can break chemical bonds,23 a process whose net effect is influenced by the kinetics of damage creation versus repair.23 The dose rate, for instance, is a pivotal factor; a low dose rate allows for the dissipation of localized heat and the recombination of radiolytically generated radicals or the diffusion of defects to sinks, thereby mitigating cumulative damage,24,25 whereas a high dose rate can overwhelm these innate healing mechanisms, leading to a concentrated burst of reactive species that can cause catastrophic and irreversible framework collapse.25 Ghosh et al.26 performed electron diffraction on ZIF-8 using the FEI Technai G2 F30(S) TEM equipped with a Schottky FEG and operated at 300 kV. Acquisition of the diffraction patterns was at dose rates of 2.91 e− Å−2 s−1, 1.14 e− Å−2 s−1, and 0.34 e− Å−2 s−1. It was found that at 300 kV, the MOF degraded with radiolysis being the dominant mechanism for damage. Zhu et al.25 in their work examining the ZIF-8 crystals' size (∼85 ± 15 nm) and shape also noted that the crystals were highly vulnerable to electron beam illumination, with their crystallinity quickly lost when the accumulated electron dose reaches ∼25 e− Å−2 s−1. The authors recommended that, to prevent structural damage during sample searching, a dose rate as low as ∼1 e− Å−2 s−1 should be chosen, and immediate image acquisition upon initial inspection is required. This corroborates the work of Banerjee et al.24 Banerjee et al. examined the dose-rate dependence of the stability of ZIF-8 (average size ∼ 160 nm) using a dose rate of 0.1 e− Å−2 s−1, 1 e− Å−2 s−1, and 6.6 e− Å−2 s−1. Time series diffraction pattern was recorded in FEI Tecnai G2 microscope (LaB6, 200 kV) with an exposure time of 1 s. Fig. 3(a) shows the bright field TEM image of the ZIF-8 and the corresponding diffraction pattern (Fig. 3(b)) with the radial average of the intensity of the Bragg rings plotted in Fig. 2(c). The time dependence of the relative intensity of the {431} Bragg spot is plotted in Fig. 2(d). However, Hanna et al.27 found that a chronic, low-radiation environment is more detrimental to Zr MOF stability than acute, high-radiation conditions. In their study, NU-1000 and UiO-66 were exposed to high (423.3 Gy min−1, 23 min, and 37 s) and low (0.78 Gy min−1, 4320 min) dose rates of 60Co gamma irradiation. NU-1000 was found to display insignificant radiation damage under both dose rates due to its high linker connectivity, low node density, and low node connectivity. The low radiation dose rates were found to cause considerable damage to UiO-66, a framework with lower aromaticity and smaller inter-ligand separation.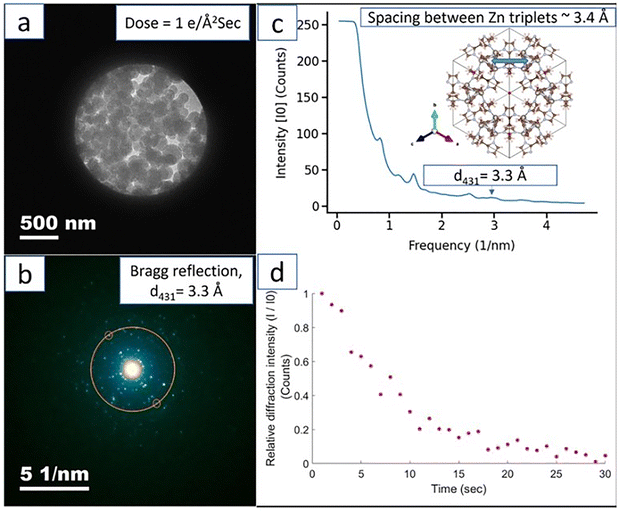 | ||
| Fig. 3 (a) Bright field image of ZIF-8 MOF taken with an electron dose rate of 1 e− Å−2 s−1. (b) Ring diffraction pattern showing the {431} Bragg reflection at t = 1 s (first acquisition). (c) Radial average of the Bragg reflections. Inset showing schematic of ZIF-8 unit cell along [111] direction (d). Relative intensity plot of 431 Bragg reflection with time (I0 = intensity at t = 1 s, I = intensity at t s). Reproduced from Banerjee et al.24 | ||
Equally critical is the energy spectrum of the radiation, as lower-energy photons or particles (e.g., soft X-rays versus hard gamma rays from Co-60) exhibit a higher linear energy transfer (LET), depositing energy more densely along their trajectory and creating localized hot spots of severe damage, such as displacement cascades, compared to the more sparse ionization events from high-energy radiation, which can lead to a qualitatively different degradation profile even for the same total dose. The particle type itself (be it gamma photons, beta electrons, alpha particles, or heavy ions) introduces another layer of complexity, as each interacts with matter through distinct mechanisms; low-LET gamma radiation primarily causes ionization and electronic excitation through Compton scattering and the photoelectric effect, while high-LET particles like alpha particles or fission fragments cause intense, localized damage via direct atomic displacements and dense ionization tracks, making the extrapolation of stability from one radiation type to another highly non-trivial. Ding et al.28 studied the effects of β-ray irradiation on MIL-101 and UiO-66 with various organic groups. Group-free and single-group MOFs exhibited poor stability, especially for MIL-101-SO3H, UiO-66-NO2, and UiO-66-OH, where the porosity and crystallinity were largely destroyed. The authors found that the dual-group UiO-66-(OH)2 and UiO-66-(COOH)2 had no significant structural changes even under 2000 kGy of β-ray irradiation. It was also found that the irradiated UiO-66-(COOH)2 showed consistent adsorption amounts for UO22+ ion with the pristine sample. Gilson et al.29 reported that thorium-based MOF (TOF-16) can withstand γ radiation up to a dose of 4 MGy and α particles up to a dose of 25.5 MGy. MIL-100(Fe/Cr) was also found to remain largely intact at a γ dose of ∼4 MGy, with MIL-101(Cr) degrading beyond ∼4 MGy, which indicated topology/node differences and a dose-threshold behavior.16,30
Furthermore, the physical sample form and the nature of the metal ion dramatically influence radiation tolerance, as powders benefit from a high surface-area-to-volume ratio that facilitates the escape of volatile radiolysis products (e.g., hydrogen gas, carbon dioxide) and minimizes internal pressure buildup, whereas dense pellets or thick films can trap these species, leading to prolonged exposure, internal stress, and accelerated degradation, an effect compounded in composites where the matrix material (e.g., a polymer) may itself radiolyze and release damaging species that attack the MOF crystallites. Volkringer et al.31 examined the effect of γ irradiation on various MOFs to determine their resistance under strong ionizing radiation. ZIF-8,15 HKUST-1,16 MILs,17, and UiO-6618, which involve different metals and oxidation states (Zn2+, Cu2+, Al3+, and Zr4+) were selected and irradiated with doses up to 1.75 MGy. Volkringer et al. found that MOFs constructed with transition metals (Cu2+, Zn2+, and Zr4+) degraded, whereas those with Al remained intact, a stability that was linked to the lower cross-section of Al that limited the effect of γ rays. This concept of a material-specific “dose-threshold” behaviour, beyond which irreversible amorphization occurs, was further explored in a high-dose investigation extending to 5 MGy on the mesoporous MIL-100 and MIL-101 families by Ma et al.32 Despite sharing similar trimeric metal-carboxylate building units (M(III) = Fe or Cr with µ3-oxo cores), MIL-100(Fe/Cr) demonstrated superior robustness, remaining largely structurally intact up to approximately 4 MGy, while the isoreticular MIL-101(Cr) degraded beyond this threshold, indicating that even within a closely related chemical family, topological differences (specifically, the larger pore size and window dimensions in MIL-101) can render a framework more susceptible to radiation-induced collapse, likely due to reduced structural stiffness and a lower activation barrier for bond rotation or linker displacement under radiolytic stress.
Finally, the particle-type-specific robustness and its direct implication for practical applications were demonstrated by the performance of the pillared-layer framework SIFSIX-3-Cu (Cu(pyrazine)2(SiF6)),33 which not only preserved its crystalline structure and gate-opening adsorption behaviour under irradiation with high-energy (1.5 MeV) electrons to a dose of 50 kGy but also showed exceptional resilience in mixed gamma/beta (γ/β) fields relevant to the capture and separation of radioactive gases like 85Kr and 133Xe in nuclear fuel reprocessing streams, underscoring how a specific and robust chemistry (in this case, the strong, charge-assisted coordination of the inorganic SiF62− pillars to the Cu2+ centers, creating a rigid and stable framework) can confer remarkable resistance to certain types of radiation damage.
2.3. Electron-beam interactions and the radiolysis–knock-on continuum
Transmission and scanning electron microscopy (TEM/SEM) analyses inherently impose in situ electron irradiation, which consistently functions as both an analytical probe and a significant source of structural perturbation.18 For the vast majority of metal–organic frameworks (MOFs), which are characterized by their organic ligands and coordination bonds, the primary degradation mechanism under the conventional 60–200 kV accelerating voltages is radiolysis, a process driven by inelastic scattering events that deposit energy, break chemical bonds, and generate free radicals, leading to rapid amorphization and collapse.18 A common strategy to mitigate the alternative damage mechanism of knock-on displacement (the physical ejection of atoms by elastic collisions) is to lower the incident electron voltage; however, this well-intentioned approach can paradoxically worsen radiolysis damage for many MOFs. This counterintuitive outcome arises because lower-energy electrons exhibit a higher cross-section for inelastic scattering, thereby increasing the rate of energy deposition per unit volume, while simultaneously reducing the beam's penetration power, which confines this intensified damage to a smaller, more superficial region of the specimen and can obscure the underlying, intact structure. This phenomenon, drawing analogies from the more extensively studied zeolite systems, highlights a fundamental trade-off where a tactic to suppress one damage pathway inadvertently exacerbates another, complicating the acquisition of representative microstructural data. Conversely, the recent investigation of highly conductive two-dimensional MOFs, such as Cu3(BHT) [BHT = benzenehexathiol], reveals a dramatically different and more resilient behavior, wherein their extended π-conjugation and metallic-like electronic structure facilitate the rapid dissipation of the energy deposited by the electron beam, effectively suppressing the radiolysis process.21 In these robust materials, any remaining observable damage is predominantly attributable to knock-on displacement, a threshold-based mechanism that becomes significant only above specific voltage thresholds. This stark contrast in degradation pathways not only enables clearer, higher-resolution imaging of these conductive MOFs but also serves to clarify the mechanistic crossover in beam-induced damage, illustrating that the dominant degradation mechanism is not a universal constant for MOFs but is intrinsically tied to the material's electronic structure and electrical conductivity, thereby providing a crucial design principle for developing next-generation MOFs with enhanced stability for in situ electron microscopy studies and applications in demanding environments.2.4. Radiation chemistry in and around MOFs: from water radiolysis to linker scission
The fundamental challenge of electron irradiation in the characterization of metal–organic frameworks (MOFs) lies in the profound chemical consequences of energy deposition, a process that generates dense ionization tracks and a cascade of highly reactive radical species. This is particularly catastrophic in hydrated or ambiently exposed samples, where the radiolysis of water molecules by the incident beam produces a predictable and potent cocktail of primary radicals with well-established G-values—the yield per unit of absorbed energy—on the order of 0.28, 0.28, and 0.062 micromoles per joule for the hydroxyl radical (˙OH), the hydrated electron (e−(aq)), and the hydrogen atom (˙H), respectively, at room temperature.34 These primary radiolytic products, especially the powerfully oxidizing ˙OH and the strongly reducing e−(aq), do not remain inert; they rapidly and indiscriminately attack the very building blocks of the MOF, targeting both the organic linkers and the inorganic metal clusters. The existence of these quantifiable yields directly rationalizes the empirically observed, accelerated degradation of MOFs irradiated in humid air compared to under high vacuum conditions, as the presence of water provides a continuous source for generating these destructive agents. Once initiated, the radiolytic damage propagates through the framework via several distinct but often concurrent pathways: firstly, through direct linker bond scission, where the energy transfer leads to the fragmentation of carboxylate groups or the aromatic rings themselves, dismantling the molecular struts of the structure; secondly, through node–linker bond cleavage, severing the critical coordination bonds (M–O) that connect the organic and inorganic components, thereby dissolving the network's connectivity; and thirdly, through a more insidious, radical-mediated defect growth, where initial damage sites act as sinks, propagating further reactions and leading to a cascading failure that culminates in the complete amorphization or de-coordination of the entire network. The susceptibility to these pathways is not uniform across all MOFs, and comparative gamma radiation studies, which provide a bulk analog to localized electron beam effects, reveal a clear hierarchy of stability: Al-oxo clusters are most vulnerable, followed by Cr/Fe-oxo clusters, then the more robust Zr-oxo clusters, with Cu/Zn paddlewheel structures demonstrating superior resistance.12,16,30 This distinct trend is not arbitrary but serves as a powerful indicator that the inherent chemistry, coordination geometry, and bond strength of the metal-node itself are critical factors that either buffer against or dramatically amplify these radiolytic pathways. A more robust node, such as the highly connected Zr6 cluster, possesses a greater density of strong bonds and a different electronic structure that may better dissipate energy or resist radical attack, whereas less stable nodes like Al-based clusters offer fewer defenses, making the rational selection of metal cluster a paramount consideration for designing MOFs intended for use in radiologically harsh environments, from nuclear waste processing to aerospace applications.2.5. Linking energy deposition to functional response: detection vs. shielding
The seemingly destructive interactions between high-energy radiation and metal–organic frameworks (MOFs)—often leading to radiolytic degradation—can, through careful chemical and structural design, be co-opted into useful functionalities such as radiation detection and shielding, showcasing a fascinating duality in which the same processes act as either liabilities or assets. In radiation detection, for instance, hafnium-cluster MOFs incorporating rigid, conjugated, and emissive linkers demonstrate that precise control of electronic structure and connectivity can transform radiolytic excitation into luminescence. Hf-based frameworks such as Hf-DBD and Hf-anthracene MOFs display fast scintillation and fluorescence yields approaching 40%,15 enabling sensitive time-resolved detection of noble-gas radionuclides (e.g., xenon and krypton) via coincidence counting of subsequent decay events. Here, the high atomic number (Z) of hafnium boosts the mass attenuation coefficient (µ/ρ), ensuring efficient interaction with incident photons, while the rigid, highly ordered coordination environment confines excitons—bound electron–hole pairs—directing them toward emissive sites and minimizing nonradiative decay. This synergistic interplay between heavy-atom nodes and π-conjugated linkers underlies the emergence of MOF scintillators that combine high light yield, rapid response, and structural stability under exposure. Beyond detection, similar design strategies confer resilience under ionizing radiation in process environments. For example, the ultramicroporous SIFSIX-3-Cu framework retains its selectivity for Kr over Xe and its pore structure even after significant β and γ irradiation at reprocessing-relevant doses, demonstrating that proper electronic and topological design can preserve function under extreme conditions.33Conversely, in radiation shielding, the same high-Z metal nodes (Hf, Bi, Pb, Gd) that enhance detection also strengthen photon attenuation through the photoelectric effect. Yet a trade-off arises because the intrinsic low bulk density and porosity of pristine MOF powders reduce macroscopic shielding efficiency, even when intrinsic µ/ρ values are high. This limitation has spurred composite strategies in which high-Z MOFs are embedded within dense polymers, ceramics, or other inorganic matrices to increase overall density and mechanical integrity without sacrificing the tunable chemistry and porosity that define MOFs.35,36 Such composites—Hf-MOF/epoxy, Bi-MOF/polyimide, and Gd-MOF/PEEK hybrids—achieve lightweight, flexible shielding while maintaining energy-selective attenuation, thus reconciling microscopic attenuation efficiency with bulk protection.34 The determinants of resilience across these applications lie primarily in linker chemistry and framework dimensionality. Aromatic, π-conjugated, and electron-deficient linkers delocalize excitation energy, raising the bond-breaking threshold and allowing nonradiative relaxation that preserves the metal–ligand coordination sphere. By contrast, aliphatic or heavily substituted linkers localize charge and feature weak C–X bonds that are prone to scission, promoting amorphization under radiation. Consequently, frameworks built from rigid, planar linkers—such as terephthalates, pyrenes, or anthracenes—tolerate doses into the MGy regime and sustain optoelectronic function, as demonstrated in anthracene-Ln MOFs that exhibit strong radioluminescence even after repeated exposure.37
Dimensionality further governs radiation endurance. Three-dimensional, high-connectivity networks (e.g., UiO-66 and MIL-101 with Zr6 or Cr3O clusters) distribute radiolytic and mechanical stresses over multiple bonds, resisting collapse and maintaining porosity at high doses. In contrast, large-pore or low-connectivity frameworks like NU-1000 deform at much lower thresholds, highlighting how node connectivity and topology modulate radiation tolerance.27 Two-dimensional conductive MOFs add another dimension to resilience: delocalized charge and heat flow suppress radiolysis and shift damage mechanisms to higher-energy knock-on processes, making them attractive as detector substrates even though their small thickness limits bulk attenuation.
The interplay between heavy-atom density and emissive efficiency defines a core trade-off between shielding and scintillation. Maximizing heavy-metal content increases µ/ρ and improves shielding but can polarize bonds and quench luminescence by facilitating nonradiative pathways, while optimizing for scintillation demands rigid linkers, defect-free structures, and sufficient porosity to allow exciton migration—conditions that typically reduce density and thus shielding.32 Rational MOF design must therefore balance these opposing goals through hierarchical structuring and composite integration. Embedding emissive high-Z clusters in robust matrices, protecting linkers with radiolabile capping ligands, or incorporating sacrificial scavengers for radicals produced by water radiolysis all extend operational lifetimes and allow materials to perform dual roles as both scintillators and shields.38 Ultimately, MOF functionality under radiation is an exercise in balancing electronic delocalization, atomic weight, and dimensional topology: choosing heavy, high-connectivity nodes for attenuation while maintaining conjugated linkers for emission defines the design space for next-generation multifunctional materials that co-localize shielding and sensing within a single crystalline architecture.
3. In situ degradation mechanisms under high-energy irradiation
Understanding how MOFs degrade under different forms of ionizing radiation is central to both predicting their performance and designing radiation-hardened systems. Unlike conventional oxides or halides, MOFs combine inorganic clusters with organic linkers, making them particularly sensitive to bond scission, radical formation, and defect accumulation. The following sections analyze degradation pathways under photons, electrons, protons, and mixed-field exposures, emphasizing in situ studies that correlate structural evolution with functional outcomes.3.1. Photon Irradiation (γ-rays, X-rays)
MOFs exposed to ionizing photons (γ-rays and hard X-rays) frequently undergo severe structural degradation, manifesting as significant porosity loss, crystallographic phase transformation, and ultimately, complete framework collapse, with the specific degradation pathway being highly dependent on the intrinsic chemical stability of the metal-node and organic linker. Volkringer et al.12 report on γ-irradiation revealed that transition-metal MOFs are typically far more susceptible to framework destruction than Al-based materials.12 Volkringer et al. reported progressive loss of crystallinity for archetypal transition-metal MOFs under cumulative γ doses up to 1.75 MGy, while Al-MOFs remained comparatively intact.12 As seen in Fig. 4(a), higher irradiation doses (between 1.25 and 1.75 MGy) resulted in broader Bragg peaks with lower intensities, indicating an important loss of crystallinity. A similar observation was reported by Hastings et al.39 More so, the nitrogen sorption at 77 K shows an important loss of porosity (Fig. 4(b and c)). The irradiation process induces a lower porosity when increasing the gamma dose. For example, a dose of 1.75 MGy, irradiated ZIF-8 characterized by a BET surface of 598 m2 g−1 (microporous volume of 0.198 cm3 g−1) which is approximately half as much as the non-irradiated solid (BET of 1328 m2 g−1 and microporous volume of 0.545 cm3 g−1). In contrast, Al-MOFs retained BET surface area and diffraction integrity at comparable doses, highlighting node chemistry and connectivity as stabilizing factors.12 Comparative studies further show MIL-100(Cr) frameworks withstand up to ∼4 MGy, with amorphization onset emerging beyond that threshold, whereas Cu-paddlewheel frameworks degrade substantially at lower doses (∼100–200 kGy).16 These differences emphasize the importance of the coordination environment and the need for systematic dose–response mapping across MOF families.12,16 | ||
| Fig. 4 (a) Evolution of powder X-ray diffraction patterns of non-irradiated and gamma irradiated zinc-based compound MOF ZIF-8 (copper wavelength) at different doses (0–1.75 MGy). Reproduced with permission from Volkringer et al.12 Copyright 2016 © The Royal Society of Chemistry. Isotherm curves for the adsorption (solid) and desorption (dash) of nitrogen (77 K) in irradiated and non-irradiated (b) ZIF-8 and (c) MIL-120. Reproduced with permission from Volkringer et al.12 Copyright 2016 © The Royal Society of Chemistry. | ||
The mechanism involves the absorbed photons producing secondary electrons that deposit energy locally and form radical species (˙OH, e−(aq)) that preferentially attack organic linkers. Studies on azobenzene-containing MOFs and azobenzene-functionalized Zr-MOFs demonstrated light- and ionizing-radiation-sensitive azo (N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) units where scission or isomerization (trans to cis) changes linker geometry and triggers pore-volume and connectivity changes (e.g., mechanized UiO-68-azo showed photo-triggered cargo release linked to azobenzene switching.40 In contrast, ultramicroporous frameworks with strongly bound, low-polarizability linkers and high node connectivity (such as SIFSIX-3-Cu) exhibit markedly higher resilience.33 SIFSIX-3-Cu retained its original crystal structure after both β and γ exposure to 50 kGy (and other SIFSIX variants decomposed at much lower doses, e.g., Co-containing analogues near ∼10 kGy), demonstrating that narrow pore topology and strong M–N bonding can extend useful lifetime in radioactive environments. The complementary work by Ma et al.32 and by Pacific Northwest National Laboratory (PNNL)41 documented that both linker aromaticity32 and metal choice41 control the damage threshold. For Ma et al., Al-MOFs (e.g., MIL-100(Al)) are more tolerant at the MGy scale while typical Cu- and Zr-based MOFs showed progressive pore loss and amorphization at lower doses. The PNNL engineered-particle study likewise showed that composite (polymer-bound) CuBTC and UiO-67 beads retained long-range order after exposures up to 1000 kGy (1 MGy) in that specific engineered form, underscoring that formulation (composite vs powder) and temperature/atmosphere moderate the apparent dose–response.
N) units where scission or isomerization (trans to cis) changes linker geometry and triggers pore-volume and connectivity changes (e.g., mechanized UiO-68-azo showed photo-triggered cargo release linked to azobenzene switching.40 In contrast, ultramicroporous frameworks with strongly bound, low-polarizability linkers and high node connectivity (such as SIFSIX-3-Cu) exhibit markedly higher resilience.33 SIFSIX-3-Cu retained its original crystal structure after both β and γ exposure to 50 kGy (and other SIFSIX variants decomposed at much lower doses, e.g., Co-containing analogues near ∼10 kGy), demonstrating that narrow pore topology and strong M–N bonding can extend useful lifetime in radioactive environments. The complementary work by Ma et al.32 and by Pacific Northwest National Laboratory (PNNL)41 documented that both linker aromaticity32 and metal choice41 control the damage threshold. For Ma et al., Al-MOFs (e.g., MIL-100(Al)) are more tolerant at the MGy scale while typical Cu- and Zr-based MOFs showed progressive pore loss and amorphization at lower doses. The PNNL engineered-particle study likewise showed that composite (polymer-bound) CuBTC and UiO-67 beads retained long-range order after exposures up to 1000 kGy (1 MGy) in that specific engineered form, underscoring that formulation (composite vs powder) and temperature/atmosphere moderate the apparent dose–response.
Beyond these general trends are striking counter-examples that elucidate mechanisms: Gilson et al.29 reported a thorium-binaphthol MOF that withstood γ doses up to ∼4 MGy (and extreme α exposures) without catastrophic collapse, attributing robustness to actinide-center chemistry and unusually radiation-hard bonding motifs, while Hastings et al.39 showed that substitution of the metal node (Zr, Hf, Th, Pu, Ce) produces large differences in both primary radiolytic sensitivity and self-irradiation accumulation (Pu–UiO-66 samples accumulated tens of MGy equivalent self-dose), demonstrating the central role of metal selection in setting both the initial and cumulative degradation kinetics. However, Hanna et al.27 observed dose-rate effects (chronic low dose can be more damaging than an equivalent high-rate acute dose for some Zr-MOFs), signifying that dynamic defect annealing and radical recombination pathways compete with damage accumulation.
Hanna et al.27 observed two-stage degradation pathways: an initial radiation-triggered structural rearrangement or single-crystal-to-single-crystal (SCSC) transformation (driven, for example, by ligand scission or conformational switching that contracts unit cells and lowers pore volumes) can precede final global amorphization once a critical concentration of broken linkers or displaced nodes is reached. Frameworks that show an SCSC intermediate might transiently retain crystallinity (potentially allowing in situ operations such as separations or sensing immediately post-irradiation), but the metastable state commonly has reduced porosity and chemical stability so functional performance (adsorption capacity, selectivity) can degrade well before full amorphization. Therefore, for Cu-propeller-type linkers, the presence of azo groups suggests design strategies: replace radiation-labile moieties, increase node connectivity, or employ sacrificial, radiolabile capping molecules to protect structural integrity. The quantified dose thresholds (tens of kGy to several MGy), metal- and linker-specific mechanisms, dose-rate and formulation effects, and mitigation strategies (node choice, composite formulations, sacrificial linkers) must be considered if MOFs are to be deployed in radiation-rich environments.
3.2. Electron irradiation – (S)TEM beam effects
Electron irradiation from (S)TEM probes degrades MOFs through a well-defined set of in situ mechanisms whose relative importance depends on accelerating voltage, dose (e− Å−2), dose-rate, local chemistry, and temperature. The primary ionization (radiolysis) and secondary electron chemistry (inelastic scattering) break organic linkers and create radical species, elastic collisions (knock-on) displace light atoms, and sputter surface species at high voltages.18 Local heating and charging drive bond rearrangements and phase segregation, and these primary events cascade into common observable outcomes such as linker scission, node reduction, and metal-nanoparticle nucleation, vacancy formation, and progressive loss of long-range order (amorphization) or densification of the lattice with pore collapse. Zheng et al.42 reported the onset of visible damage for common MOFs in the low tens of electrons per Å2 (e.g., UiO-66 shows lattice degradation at ∼10–20 e− Å−2 and MIL-101(Cr) near ∼16 e− Å−2, while ZIF-8 is somewhat more robust at ∼25 e− Å−2 under typical 200–300 kV imaging conditions), establishing a pragmatic “safe window” for in situ imaging and motivating low-dose strategies. The radiolysis mechanism dominates at lower beam energies and for hydrogen-rich, π-conjugated linkers. In situ TEM studies repeatedly observe the disappearance of diffraction spots and loss of contrast corresponding to organic linker loss long before any large-scale atomic displacements are evident, consistent with ionization-driven bond scission forming carbon-center radicals that either recombine into cross-linked amorphous carbon or desorb as volatile fragments.43 Knock-on displacements become important at higher accelerating voltages for light atoms (H, C, N) and explain why lowering the voltage or using cryo-conditions can suppress certain damage modes.18,21 Recently, Xu et al.44 reported non-classical sequences where initial radiolysis increases local lattice flexibility and enhances subsequent knock-on susceptibility (a radiolysis-enhanced knock-on pathway).One frequent, practically important outcome under focused probes is metal-cluster nucleation. Several TEM and in situ experiments on copper- and zinc-based MOFs (e.g., HKUST-1/CuBTC, MOF-5) show electron-beam-induced reduction of Cu2+ or Zn2+ with concomitant growth of metallic nanoparticles within minutes of sustained exposure, producing dark contrast and catalysing framework collapse—a pathway exploited intentionally in electron-beam synthesis of metal nanoparticles from MOF precursors but problematic when the goal is a careful structural characterization.45 Microcrystal electron diffraction (MicroED) and cryo-TEM offer partial remedies. MicroED has yielded sub-Å structural determinations for beam-sensitive frameworks (ZIF-8 refined to 0.87 Å resolution in cryo-MicroED experiments), because continuous-rotation diffraction collects crystallographic signal at extremely low cumulative dose per reciprocal-space wedge; similarly, cryo-FIB specimen preparation and cryo-TEM imaging reduce radiolysis and volatile desorption, preserving host–guest interactions and enabling otherwise impossible atomic models. Recent advances in detector technology and phase-contrast modalities have further shifted the practical limits. Electron ptychography and optimized 4D-STEM protocols have produced near-atomic reconstructions of MOFs at doses as low as ∼100 e− Å−2, while modern direct electron detectors and integrated differential phase contrast (iDPC-STEM) permit higher information yield per electron and allow discernment of metal clusters and even benzene rings at doses that formerly caused rapid damage. Li et al.46 demonstrated the implementation of near-atomic-resolution (∼2 Å) electron ptychography reconstruction at electron doses as low as ∼100 e− Å−2 for MOF Zr-BTB (Fig. 5).
 | ||
Fig. 5 (a) Ptychography phase image reconstructed from an experimental 4D-STEM dataset acquired at conditions of 10-mrad convergence semi-angle, 1.05 Å scan step, and an electron dose of 115 e− Å−2. (b) The power spectrum of (a), demonstrating information transfer up to 2.25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Å. (c) Enlarged image from the square-marked area in (a). (d) Structural model of MOF Zr-BTB, projected along the [100] axis. Green Zr, red O, gray C, pink H. The high consistency between (c) and (d) indicates the accuracy of ptychography reconstruction at high resolution. Reproduced with permission from Li et al.46 © The Author(s) 2025. Å. (c) Enlarged image from the square-marked area in (a). (d) Structural model of MOF Zr-BTB, projected along the [100] axis. Green Zr, red O, gray C, pink H. The high consistency between (c) and (d) indicates the accuracy of ptychography reconstruction at high resolution. Reproduced with permission from Li et al.46 © The Author(s) 2025. | ||
Crucially, in situ STEM experiments have documented sequence-specific damage signatures that can be diagnostic of mechanism: (i) immediate loss of organic linker contrast with retained metal-cluster diffraction indicating radiolytic linker cleavage; (ii) progressive spot broadening and unit-cell contraction consistent with selective linker loss and densification; and (iii) sudden appearance of polycrystalline metal rings in SAED patterns marking reduction and nanoparticle formation—each stage has been correlated with specific dose ranges in reported studies, enabling dose-budgeting for live experiments.43 For practical imaging and study of in situ behaviour, dose management and sample engineering are decisive. Lowering accelerating voltage (below knock-on thresholds), cryogenic cooling, sparse-sampling acquisition, beam-blanking, direct-detector counting, ptychographic phase retrieval, and embedding MOF microcrystals in protective matrices or operating in liquid or gas environmental TEM reduce net damage and can allow observation of true dynamic processes rather than beam-driven artefacts.42 More so, controlled harnessing of beam effects has become a research tool in its own right. By understanding threshold doses and kinetics, researchers deliberately trigger linker cleavage, metal reduction, or densification to study subsequent catalytic activity, nanoparticle formation, or defect chemistry.47
3.3. Proton and heavy-ion irradiation
Proton and heavy-ion irradiation interact with MOFs quite differently from photons. Energetic ions deposit energy very densely along straight trajectories, producing narrow electronic tracks and displacement cascades whose local energy density can be orders of magnitude higher than the average dose delivered by γ-rays. This concentrated energy drives rapid local heating, massive electronic excitation, and primary knock-on events that nucleate nanometric damaged cylinders (ion tracks) typically a few nanometres in radius, and the resulting damage morphology and kinetics depend sensitively on electronic stopping power (Se), ion energy, and the chemical/thermal transport properties of the target.48 Modeling tools such as SRIM and the inelastic-thermal-spike (i-TS) framework are used to estimate local Se and the temperature transients around an ion path and typically predict track radii on the order of 1–5 nm for many inorganic and polymeric targets at MeV–GeV energies. The predictions have been borne out by experiment in oxides and semiconductors and are directly applicable to MOF components (linkers and SBUs) for estimating local deposited energy and peak transient temperatures.48 Ishikawa et al.49 work on UO2 irradiated with various ions with the specific energy in the order of 1 MeV u−1 demonstrated that ion tracks of only a few-nanometre radius produce measurable amorphization or phase changes at low fluences once track overlap becomes significant. In UO2 and related actinide/oxide systems, heavy ions with sufficiently large Se produce continuous amorphous tracks (track radii ≈3–5 nm) with macroscopic effects (swelling, loss of crystallinity) at fluences that are orders of magnitude lower than those needed to produce comparable effects with γ-irradiation.49 Translating those findings to MOFs, Fairley et al.50 ion studies showed that ion irradiation can induce localized, near-surface structural breakdown even for frameworks that are gamma-tolerant. Fairley et al.50 noted that isoreticular UiO derivatives show markedly different He-ion tolerance depending on linker chemistry (NU-403 with aliphatic rings showed significant diffraction changes after ∼6–10 MGy He-ion exposures, while aromatic UiO-66 derivatives retained diffraction features up to tens of MGy). This indicates that Se-localized chemistry strongly targets labile linker-node connections. Similarly, Gilson et al.11 observed that TOF-16, a thorium–binaphthol framework, retained bulk X-ray crystallinity after 4 MGy of Co-60 γ-irradiation but exhibited an onset of crystallinity loss under 5 MeV He2+ irradiation at ≈15 MGy and completed loss by ≈25 MGy. This implies that the highly localized energy deposition of He ions produced near-surface track damage and defect accumulation even though bulk photon tolerance was high.Mechanistically, ion tracks in MOFs may combine (a) ultra-local radiolytic chemistry that cleaves or oxidises organic linkers, (b) transient thermal spikes that cause linker desorption or node restructuring, and (c) knock-on cascades that directly displace light atoms (H, C, N) and create vacancies at the metal–ligand interface.51 These processes can produce missing-linker defects, reduced metal clusters, and densified, amorphous surface shells that evolve with fluence into deeply modified near-surface regions while the un-irradiated bulk remains crystalline.51 Studies using combined ion irradiation and synchrotron-based XRD/XAS showed that, for MOFs and related coordination solids, damage first appears as peak broadening and small-angle scattering features (evidence of nanometric heterogeneity and defect clusters) before full loss of Bragg intensity, consistent with an ion-track nucleation/overlap pathway.52 These observations imply that (1) MOF screening for ionizing environments cannot rely solely on bulk γ-dose tests—ion-beam testing (protons, He, SHI) is essential to reveal surface/near-surface vulnerability; (2) linker chemistry (aromatic vs aliphatic, electron-donating substituents), node identity (Zr/Th vs. Cu), and microstructure (particle size, porosity) dominate ion track susceptibility; and (3) mitigation strategies (surface passivation, sacrificial capping ligands, dense encapsulation or composite architectures) should be prioritized when near-surface integrity is critical.
3.4. Mixed-field and space environment conditions
Real-world applications in nuclear or cosmic environments involve combined stressors, including radiation, thermal cycling, vacuum, and low-energy particle exposure. A study on MOFs under simulated space conditions (UV, protons, thermal vacuum cycling) found that UiO-66-based materials can degrade via ligand desorption and node hydrolysis, especially under repeated cooling–heating and UV flux, even when X-ray resistance is adequate.53 Although no MOFs have yet been tested under full mixed neutron/ion/vacuum irradiation, the literature—especially from irradiated polymers—suggests synergistic damage that accelerates amorphization and mechanical failure.54,55 This gap highlights the need for integrated environmental studies.3.5. Experimental approaches for real-time radiation studies
The emergence of in situ characterization tools is transforming how degradation is understood. X-ray absorption spectroscopy (XAS) provides oxidation state changes at metal nodes, while Raman and FTIR track linker bond scission during irradiation. For instance, in situ Raman of UiO-66 under γ-rays showed the gradual disappearance of C–C stretching modes above 200 kGy.56–58 Time-resolved X-ray absorption spectroscopy (XAS) can track oxidation state and coordination changes in metal nodes under irradiation; for example, Ti-MOFs show Ti3+ formation under electron beam, indicating linker reduction.59 Raman and FTIR under γ/X-ray exposure can capture linker breakage or bond formation dynamics in real time, as demonstrated in UiO-66 during in-beam FTIR, revealing loss of carboxylate features at cumulative doses of ∼100 kGy.56 Synchrotron PXRD and total scattering techniques reveal amorphization kinetics. Amorphization in ZIF-8 was observed in situ at ∼700 kGy, evidenced by peak broadening and loss of long-range order.60,61 Pair distribution function analysis captures local connectivity loss before full crystallinity collapse. Furthermore, low-dose cryo-TEM combined with 4D-STEM enables nanoscale mapping of radiation-induced disorder.20,62 Real-time studies on HKUST-1 revealed preferential collapse of Cu–O clusters, correlating with conductivity loss.62,63 Such techniques are invaluable for quantifying threshold doses for defect formation in specific SBUs. Table 2 presents the comparative summary of in situ degradation data for representative MOFs (UiO-66, MIL-100, ZIF-8, Cu-paddlewheel), showing radiation tolerance for selected MOFs: dose thresholds, BET change, and crystallinity evolution (PXRD/FWHM).| MOF (SBU/topology) | Radiation (energy) & atmosphere | Dose and rate | BET surface area change | Crystallinity change (PXRD/FWHM) | Notes | Ref. |
|---|---|---|---|---|---|---|
| MIL-100(Fe) (Fe–O trimer; mtn-a) | γ-rays (Co-60; 1.17 and 1.33 MeV); air | 0 → 5 MGy (stepwise) | 1574 → 1507 m2 g−1 (≈–4% at 5 MGy) | Small FWHM fluctuation; crystallinity retained to 5 MGy | Minimal morphology changes by SEM; FT-IR largely unchanged | 16 |
| MIL-100(Cr) (Cr–O trimer; mtn-a) | γ-rays (Co-60; 1.17 & 1.33 MeV); air | 0 → 5 MGy (stepwise) | 1862 → 1802 (1 MGy, −3.2%); 1744 (2 MGy, −6.3%); 1660 (3 MGy, −10.8%); 1600 (4 MGy, −14.1%); 1510 (5 MGy, −18.9%) m2 g−1 | PXRD pattern maintained; FWHM shows limited change to 5 MGy ⇒ good crystallinity retention | Micropore volume declines with dose; XPS indicates partial linker–cluster bond weakening | 16 |
| MIL-101(Cr) (Cr–O trimer; mtq) | γ-rays (Co-60; 1.17 & 1.33 MeV); air | 0 → 5 MGy (stepwise) | 2203 → 2099 m2 g−1 at 4 MGy (–4.7%); 1762 m2 g−1 at 5 MGy (–20.0%) | At 5 MGy, FWHM ↑ ∼22% ⇒ significant loss of crystallinity/onset of decomposition | Micropore volume –30% at 5 MGy (1.0715 → 0.7508 cm3 g−1) | 16 |
| Al-fumarate (AlFu) (Al-oxo chain) | γ-rays (Co-60; 1.17 & 1.33 MeV); air | 0 → 2 MGy | Surface area decreases; pore structure collapses by ∼2 MGy | Peak broadening with dose; crystallinity degrades after ∼1 MGy, pore features vanish by 2 MGy | Lower stability linked to non-aromatic linker; contrasts with MIL-100/101 | 16 |
| UiO-66(Zr) (Zr6O4(OH)4; fcu) | γ-rays (up to 1.75 MGy) | Up to 1.75 MGy | Not reported (in this study) | Framework destruction reported for transition-metal MOFs, including UiO-66, at high doses | Comparative γ-stability screen across archetypal MOFs; Al-based frameworks remained intact | 12 |
| ZIF-8(Zn) (Zn–imidazolate; sod) | γ-rays (up to 1.75 MGy) | Up to 1.75 MGy | Not reported (in this study) | Framework destruction at high dose (loss of long-range order) | Highlights the vulnerability of some Zn–Zn-imidazolate frameworks under γ-fields | 12 |
| HKUST-1(Cu) (Cu2 paddlewheel; tbo) | Electron beam (200–300 keV, TEM/STEM) | Dose-rate dependent (in situ EM) | BET not applicable in EM; pronounced signal loss under the beam | Rapid amorphization via radiolysis, Cu–O bond cleavage; knock-on is less dominant for organics | Low-dose/cryo mitigations recommended; 4D-STEM maps dose thresholds | 12 |
For MIL-100/101 and Al-fumarate, explicit BET trajectories, micropore-volume changes, and PXRD/FWHM trends with γ-dose are extracted from Ma et al.,16 which irradiated MOFs to 5 MGy using a Co-60 source and reported quantitative adsorption/diffraction metrics.16 The broader multi-MOF γ-stability screening by Volkringer et al.12 established relative trends up to 1.75 MGy for UiO-66, ZIF-8, and HKUST-1, but does not tabulate BET values.12 We therefore report its crystallinity outcomes qualitatively. For electron-beam sensitivity (TEM/STEM), is used to summarize knock-on vs. radiolysis mechanisms and practical dose-mitigation strategies relevant to in situ/operando electron microscopy (EM) (i.e. using electron microscopy techniques (TEM, STEM, SEM) while a material is actively functioning under realistic operating conditions such as applied electrical bias, gas exposure, illumination, heating, or irradiation); quantitative BET is not meaningful under EM conditions.18,19
4. Application of MOFs for sensing and radiation shielding
4.1. MOFs for radiation sensing
MOFs have emerged as highly tunable materials for radiation sensing owing to their hybrid composition, ordered porosity, and structural diversity, which enable both scintillation-based photon detection and radiation-induced charge or luminescence modulation. In MOF-based radiation sensors, X- or γ-ray photons interact with the high-Z metal centers to produce energetic photoelectrons that propagate through the lattice, exciting organic linkers or generating charge carriers.64 These excited states or carriers subsequently decay radiatively,63 leading to scintillation or radioluminescence,65 or are modulated under sustained irradiation to produce measurable luminescent quenching or current signals. The performance of these MOF systems is typically assessed in terms of light yield (photons MeV−1), spatial resolution (lp mm−1), detection limit (Gy or nGy s−1), linear dose range, response time, and stability across repeated exposure cycles.63–65 These characteristics determine the suitability of MOFs for diagnostic imaging, environmental dosimetry, and radiation detection in both medical and industrial settings.66–68A major advance in this area has come from lanthanide-based MOF micro-scintillators, which demonstrate efficient energy conversion due to their heavy nodes and photophysically active linkers. In such systems, the high atomic number of lanthanide centers (e.g., Tb3+, Eu3+, Hf4+) enhances photoelectric absorption, while the organic linkers mediate energy transfer via singlet–triplet exciton pathways.69Fig. 6 shows the X-ray-excited radioluminescence in lanthanide metal–organic frameworks. Upon X-ray exposure, primary photoelectrons are ejected from inner shells of the heavy atoms in the MOF, and the subsequent relaxation processes produce low-energy secondary photons. These photons excite the organic linkers, creating a distribution of singlet and triplet excitons (in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 ratio). The energy stored in the triplet states is then efficiently transferred to the neighboring lanthanide ions through triplet-to-4f energy transfer, generating lanthanide-activated radioluminescence. Concurrently, the secondary photons can also excite the MOF lattice to produce free charge carriers that may recombine at lanthanide sites, further contributing to radioluminescent emission. Such mechanisms have been extensively studied in lanthanide MOFs such as Tb-MOF-76, where theoretical calculations of valence and conduction band states have revealed that the lanthanide 4f orbitals align favorably with the host's electronic bands to facilitate efficient energy transfer and minimize the non-radiative decay channel.69 The combination of heavy-metal absorption and triplet-state energy relay endows these frameworks with strong, persistent light output and high tolerance to repeated radiation exposure. To enhance performance in practical devices, polymer-embedded Ln-MOF micro-phosphors have been fabricated to create flexible scintillating screens.69–71 These composites maintain the intrinsic high light yield of the Ln-MOFs while adding mechanical flexibility and environmental stability, enabling low detection limits and long-term photostability under continuous exposure. Such systems demonstrate that integrating MOFs with polymer matrices not only enhances durability but also provides versatile platforms for flexible X-ray detectors and portable imaging screens. Zhang et al.69 demonstrated that a solution-processable lanthanide-based MOF micro-scintillators coupled with elastic, transparent polymer matrices to form flexible X-ray detectors can achieve a detection limit of 23 nGy s−1 and a high resolution of ∼16.6 line-pairs mm−1. In another work, the team embedded an aggregation-induced emission luminogen-functionalized MOF (Y-PCN-94) into a PDMS polymer matrix to fabricate a flexible scintillating membrane.70 The composite achieved a low X-ray detection limit (1.6 µGy s−1) and high imaging resolution >14.3 lp mm−1. In the work of Wang et al.,71 a Tb-MOF (Tb-MOF-1) was used to fabricate a flexible scintillation screen which showed excellent photoluminescence and radioluminescence signals, a low detection limit of 1.71 µGy s−1, and a high spatial resolution of ∼7.7 lp mm−1 for flexible X-ray imaging. The molecular-level design of these MOFs, combining high-Z lanthanide clusters, rigid linkers, and efficient energy transfer pathways, sets the foundation for optimizing light yield and spatial resolution in radiation-sensing applications.
3 ratio). The energy stored in the triplet states is then efficiently transferred to the neighboring lanthanide ions through triplet-to-4f energy transfer, generating lanthanide-activated radioluminescence. Concurrently, the secondary photons can also excite the MOF lattice to produce free charge carriers that may recombine at lanthanide sites, further contributing to radioluminescent emission. Such mechanisms have been extensively studied in lanthanide MOFs such as Tb-MOF-76, where theoretical calculations of valence and conduction band states have revealed that the lanthanide 4f orbitals align favorably with the host's electronic bands to facilitate efficient energy transfer and minimize the non-radiative decay channel.69 The combination of heavy-metal absorption and triplet-state energy relay endows these frameworks with strong, persistent light output and high tolerance to repeated radiation exposure. To enhance performance in practical devices, polymer-embedded Ln-MOF micro-phosphors have been fabricated to create flexible scintillating screens.69–71 These composites maintain the intrinsic high light yield of the Ln-MOFs while adding mechanical flexibility and environmental stability, enabling low detection limits and long-term photostability under continuous exposure. Such systems demonstrate that integrating MOFs with polymer matrices not only enhances durability but also provides versatile platforms for flexible X-ray detectors and portable imaging screens. Zhang et al.69 demonstrated that a solution-processable lanthanide-based MOF micro-scintillators coupled with elastic, transparent polymer matrices to form flexible X-ray detectors can achieve a detection limit of 23 nGy s−1 and a high resolution of ∼16.6 line-pairs mm−1. In another work, the team embedded an aggregation-induced emission luminogen-functionalized MOF (Y-PCN-94) into a PDMS polymer matrix to fabricate a flexible scintillating membrane.70 The composite achieved a low X-ray detection limit (1.6 µGy s−1) and high imaging resolution >14.3 lp mm−1. In the work of Wang et al.,71 a Tb-MOF (Tb-MOF-1) was used to fabricate a flexible scintillation screen which showed excellent photoluminescence and radioluminescence signals, a low detection limit of 1.71 µGy s−1, and a high spatial resolution of ∼7.7 lp mm−1 for flexible X-ray imaging. The molecular-level design of these MOFs, combining high-Z lanthanide clusters, rigid linkers, and efficient energy transfer pathways, sets the foundation for optimizing light yield and spatial resolution in radiation-sensing applications.
 | ||
| Fig. 6 X-ray-excited radioluminescence in lanthanide metal–organic frameworks. (a) Mechanistic diagram of the scintillation process in Ln-MOFs. (b) Calculated energies of the lanthanide 4f orbitals (in red) with respect to the host bands. (c) Calculated partial charge densities of Tb-MOF-76 microcrystals. Reproduced with permission from Zhang et al.69 Copyright © 2023. Advanced Science published by Wiley-VCH GmbH. ISC – intersystem crossing, ET – energy transfer, S – singlet, T – triplet, VBM – valence band maximum, CBM – conduction band minimum. | ||
Beyond single-component systems, cluster-sensitized MOFs have expanded the design landscape for scintillation. In an antenna-type configuration, Tb–Cu4I4 MOF scintillators have demonstrated remarkable performance, with light yields of approximately 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 ± 3000 photons MeV−1 and spatial resolution of 12.6 lp mm−1 under diagnostic X-ray conditions (30–120 keV).72 These materials also exhibit linear dose-rate responses and stability across multiple on–off X-ray cycles totaling ∼4.5 Gy_air, maintaining radioluminescence intensity over extended operation.72 The light yield and imaging resolution achieved by these MOFs approach or even rival those of conventional copper-iodide cluster scintillators (>30 lp mm−1), while providing the structural tunability and chemical modularity characteristic of framework materials. Mechanistic analyses reveal that in Ln–Cu4I4 MOFs, the [Cu4I4] clusters act as sensitizers, efficiently harvesting X-ray energy and transferring it to the Tb3+ centers via intersystem crossing and energy transfer channels. Such cluster-sensitized architectures underscore the importance of rational structural engineering (high-Z nodes, rigid linkers with large oscillator strengths, and minimized self-absorption) to preserve modulation transfer function (MTF) and enhance spatial imaging fidelity at high frequencies. The schematic representation of the rational design and the relative XEL processes of Ln–Cu4I4 MOFs, with a perspective view of the 3D crystal structure of Tb–Cu4I4 MOFs, is presented in Fig. 7. The design rules emerging from these studies suggest that high-Z metallic nodes such as Hf, Pb, Sr, or Tb significantly enhance X-ray absorption and conversion efficiency, while rigid aromatic linkers help to maintain structural stability under irradiation. Furthermore, the suppression of self-absorption through careful linker selection and framework topology optimization leads to improved transparency and light out-coupling.
400 ± 3000 photons MeV−1 and spatial resolution of 12.6 lp mm−1 under diagnostic X-ray conditions (30–120 keV).72 These materials also exhibit linear dose-rate responses and stability across multiple on–off X-ray cycles totaling ∼4.5 Gy_air, maintaining radioluminescence intensity over extended operation.72 The light yield and imaging resolution achieved by these MOFs approach or even rival those of conventional copper-iodide cluster scintillators (>30 lp mm−1), while providing the structural tunability and chemical modularity characteristic of framework materials. Mechanistic analyses reveal that in Ln–Cu4I4 MOFs, the [Cu4I4] clusters act as sensitizers, efficiently harvesting X-ray energy and transferring it to the Tb3+ centers via intersystem crossing and energy transfer channels. Such cluster-sensitized architectures underscore the importance of rational structural engineering (high-Z nodes, rigid linkers with large oscillator strengths, and minimized self-absorption) to preserve modulation transfer function (MTF) and enhance spatial imaging fidelity at high frequencies. The schematic representation of the rational design and the relative XEL processes of Ln–Cu4I4 MOFs, with a perspective view of the 3D crystal structure of Tb–Cu4I4 MOFs, is presented in Fig. 7. The design rules emerging from these studies suggest that high-Z metallic nodes such as Hf, Pb, Sr, or Tb significantly enhance X-ray absorption and conversion efficiency, while rigid aromatic linkers help to maintain structural stability under irradiation. Furthermore, the suppression of self-absorption through careful linker selection and framework topology optimization leads to improved transparency and light out-coupling.
 | ||
| Fig. 7 (a) A schematic representation of the rational design and the relative XEL processes of Ln–Cu4I4 MOFs. (b) Perspective view of the 3D crystal structure of Tb–Cu4I4 MOFs, (c) asymmetric unit of Tb–Cu4I4 MOFs, (d) connection mode of cubic [Cu4I4] center, and (e) connection mode of Ln–O centers. Reproduced with permission from Liu et al.72 Copyright © 2023. Advanced Materials published by Wiley-VCH GmbH. | ||
Beyond radioluminescent scintillators, other MOF systems demonstrate dose-dependent luminescence quenching, providing a basis for dosimetric applications. For instance, Zheng et al.73 reported that an actinide-based coordination polymer, U-Cbdcp, exhibited linear luminescence quenching from 10 to 4700 Gy (R2 = 0.999), with a limit of detection of 0.093 Gy and near-complete quenching (∼90%) at 4.7 kGy. Notably, both PXRD and SCXRD analyses confirmed that the U-Cbdcp structure remained intact up to 5 kGy of X-ray exposure and under γ/β irradiation at 11.8–150 kGy h−1, highlighting its exceptional radiolytic stability. The quenching behavior was attributed to radical-mediated pathways, in which radiation-generated defects or trapped charge centers interact with luminescent sites, reducing emission intensity proportionally to absorbed dose. Such characteristics make this material promising for high-dose dosimetry applications, including radiation processing and sterilization monitoring, where robust and linear responses are required over kilogray-level exposures.
In parallel, semi-conductive MOFs have been investigated for direct electronic readout of radiation signals, expanding the functionality of MOF-based detectors beyond optical emission. In these systems, X-ray illumination generates measurable photocurrents due to intrinsic band-like charge transport across the crystalline framework,74 similar to behavior observed in wide-bandgap semiconductors.75 Millimeter-scale single-crystal devices have demonstrated reproducible current responses under variable dose rates, offering new opportunities to link radiation-induced defect formation with charge mobility and trap density in conductive 2D and 3D MOF architectures.74,76 While comprehensive radiation-hardness mapping of conductive MOFs remains limited, these results suggest that with careful control of electronic structure and defect engineering, MOFs can serve as both scintillating and direct-detection platforms, unifying optical and electrical readout schemes in a single, modular material system. Table 3 presents some MOF scintillators and luminescent dosimeter systems with their characteristic mechanisms and performance metrics.
| MOF/system | Mechanism | Key numeric metrics | Radiation range (used in study) | Main finding | Ref. |
|---|---|---|---|---|---|
| Notes: ph MeV−1 = photons per MeV (light yield); LOD = limit of detection; µτ = mobility–lifetime product (cm2 V−1); RL = Radioluminescence; PLQY = Photoluminescence Quantum Yield; LY = Light Yield; and NR = Not reported. | |||||
| Tb-MOF-76 (polymer-embedded microscintillator) | Triplet-mediated ligand → Tb3+ radioluminescence | LOD = 23 nGy s−1; spatial resolution >16.6 lp mm−1; PLQY: NR; light yield: NR (high RL reported) | Diagnostic X-rays (30–120 keV) | Flexible polymer-MOF screens, low detection limit and high imaging resolution | 38 |
| Tb–Cu4I4 cluster MOF | Cluster antenna sensitizer → Tb3+ emission | Light yield = 29![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 379 ± 3000 ph MeV−1; spatial resolution 12.6 lp mm−1; dose-rate linearity reported; LOD: NR 379 ± 3000 ph MeV−1; spatial resolution 12.6 lp mm−1; dose-rate linearity reported; LOD: NR |
Diagnostic X-rays (30–120 keV) | High light yield, robust cycling stability; rivals Cu-iodide cluster scintillators | 72 |
| SCU-15 (millimeter single-crystal semiconductive MOF) | Direct photoconductive detection (photocurrent) | µτ = 2.18 × 10−3 cm2 V−1; carrier diffusion length > 70 µm; reproducible photocurrent imaging | X-rays (imaging tests) | Demonstrates direct detection, links transport metrics to X-ray sensitivity | 74 |
| U-Cbdcp (uranyl coordination polymer) | Radical-mediated luminescence quenching (dosimetry) | Linear quenching 10–4700 Gy (R2 = 0.999); LOD = 0.093 Gy; ∼90% quenching at 4.7 kGy; PXRD unchanged to ∼5 kGy | X/γ (10–4700 Gy) | Quantitative, linear dosimeter for kGy regime; high radiolytic stability before structural change | 77 |
| TOF-16 (Th-binaphthol MOF) | Structural radiation tolerance | Survives 4 MGy γ (bulk crystallinity); surface track damage under 5 MeV He2+ (∼15 MGy local ion dose reported in tests) | γ and He-ion tests | Exceptional γ stability; ion beams produce localized near-surface damage | 29 |
| UiO-series (Zr/Hf MOFs) | Stable scaffold for sensing/scintillator hosts | Reported retention of long-range order up to MGy in some experiments (framework-dependent); numeric light yields vary by study → PLQY/LY: NR (varies by derivative) | Photon irradiation (high γ doses) | High-valence nodes confer better photon tolerance; used as robust hosts | 33,39 |
| Eu/Tb hetero-ligand MOFs (hetero-antenna strategy) | Enhanced triplet sensitization → lanthanide emission | PLQY improvement reported; decay times in µs range typical for 4f emissions; specific LY: NR (depends on compound) | X-ray radioluminescence tests | Hetero-ligand designs reduce self-absorption and boost RL output | 78 |
| [Bmmim]2MnCl4 (Bmmim = 1-butyl-2,3-dimethylimidazolium)/printable inks | Cluster emission (Mn–I) → XEL | Reported LY 3.63 times of the commercial Bi4Ge3O12 (BGO) (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 photons MeV−1) in some ink systems (∼36 000 photons MeV−1) in some ink systems (∼36![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 ph MeV−1 estimated when BGO = 10 300 ph MeV−1 estimated when BGO = 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 ph MeV−1 in that study)—varies with composition; LOD: NR 000 ph MeV−1 in that study)—varies with composition; LOD: NR |
Diagnostic X-ray imaging | Printable scintillator inks with high output and recyclability | 79 |
| Eu-doped hybrid MOF films | Lanthanide radioluminescence in film form | High RL intensity reported; quantitative LY/LOD: NR (in SI for particular samples) | X-ray imaging | Film processing improves mechanical robustness for devices | 69 |
| HKUST-1 (CuBTC) | Beam-stimulated emission; beam sensitivity | Rapid degradation under humid or beam conditions; LY/LOD: NR (not targeted as robust scintillator) | Electron/X-ray beam studies | Demonstrates beam-driven emission but poor long-term stability without protection | 80 |
| MIL-101/MIL-100 family (Cr) | Host/target for radionuclide chemistry and RL tests | Structural robustness used in irradiation experiments; LY/LOD: NR | Neutron/γ irradiation studies | Useful for radiochemistry; stability quantified in specific reports (see refs) | 81 |
| Gd/Bi hybrid MOFs (shielding/sensing hybrids) | High-Z absorption + neutron capture (Gd) | µ/ρ enhancement reported vs organic matrices; LY/LOD: NR | X-ray/γ/mixed fields | Multifunctional attenuation + sensing elements; used in composites | 9 |
| Thorium/actinide coordination polymers (radioluminescent dosimetric studies) | Actinide-centered luminescence/dosimetry | Linear quenching (U-CP example above): LOD = 0.093 Gy; structural stability to several kGy—metrics are system dependent | High doses (kGy–MGy) | Actinide centers give strong RL/unique signatures and high radiolytic stability | 77 |
| Multi-cluster sensitized MOFs (cluster + lanthanide) | Cluster harvesting → ligand → 4f emission | Reported LY improvements; exact ph MeV−1 depends on system (NR) | X-ray imaging | Combining sensitizer clusters and lanthanide centers enhances absorption and transfer. | 82 |
| Cu2I2(TPPA) (CuI-TPPA, TPPA = tris[4-(pyridine-4-yl)phenyl]amine) | Amorphous/continuous MOF-based films for direct detection or scintillator layers | Reports: device sensitivity of 73.1 µC Gyair−1 cm−2; LODs vary | X-ray imaging | Continuous films enable device integration and improved stability | 83 |
| SIFSIX-3-Cu and ultramicroporous frameworks (radiation tests) | High chemical stability and some radiation resilience | Retained diffraction after moderate doses (tens of kGy in some tests); LY/LOD: NR | β/γ irradiation studies (low to mid kGy) | Ultramicroporous topology confers stability in some gas-separation/radiation tests | 33 |
| Inorganic microcrystals (Tb-doped oxides) (Benchmark) | Inorganic RL | Reported LY values e.g., 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 ph MeV−1 for some inorganic hosts 500 ph MeV−1 for some inorganic hosts |
X-ray imaging | Serve as performance targets for MOF scintillators | 84 |
4.2. MOFs for radiation shielding
MOFs, due to their tunable compositions, structural diversity, and ability to incorporate high-Z elements within their porous frameworks, are equally promising materials for radiation shielding. Shielding effectiveness against ionizing radiation, particularly X-rays and γ-rays, depends primarily on the material's mass attenuation coefficient (µ/ρ), density, and thickness, factors that govern photon attenuation through photoelectric absorption, Compton scattering, and pair production.17 Traditional shielding materials, such as Pb or W provide excellent attenuation due to their high density and atomic number, but suffer from drawbacks such as toxicity, heaviness, and poor mechanical flexibility. In contrast, MOFs offer structural versatility and the possibility to integrate high-Z metals (e.g., Pb, Bi, W, Hf, Gd) within low-density matrices, thus creating materials that balance attenuation performance with processability and environmental safety.85,86 Furthermore, the modularity of MOFs allows densification and composite fabrication with polymers, ceramics, or carbonaceous matrices to enhance shielding efficiency while maintaining lightweight and flexible characteristics.85,86A key strategy in designing MOF-based radiation shields is the incorporation of high-Z metal nodes that maximize the probability of photon absorption. Lead-based MOFs were among the earliest examples to demonstrate substantial radiopacity due to the proximity of the Pb K-edge (∼88 keV) to diagnostic X-ray energies, resulting in short attenuation lengths in the 20–100 keV range.87 Similarly, Bi-containing MOFs such as Bi-NU-901 and Bi-MOF-74 have been studied as both CT contrast agents and potential shielding materials, exhibiting strong µ/ρ values compared to organic or polymer matrices.38 According to Liu et al.,88 Bi and W exhibit complementary absorption edges between 70 and 90 keV, a feature that can be exploited through edge-engineered MOF composites containing both metals to enhance broadband attenuation. This synergistic absorption behavior enables Bi/W-based MOFs to outperform Pb at selected photon energies, offering an environmentally benign, lead-free alternative for diagnostic and industrial applications.
Despite the inherent porosity of MOFs limiting their bulk density, significant progress has been made through composite engineering. MOF–polymer or MOF–ceramic hybrids have demonstrated improved attenuation while retaining processability and mechanical integrity. For instance, W/Bi-loaded polymer multilayers achieved lower transmittance than 1 mm Pb across 45–90 keV energy windows while being approximately 30% lighter (7.81 g cm−3vs. 11.3 g cm−3).88 The linear attenuation coefficient and mass attenuation coefficient, both of which were found to be significantly enhanced, with a similar trend observed with an increase in the metal filler content (Fig. 8). Translating this concept to MOFs, W- or Bi-rich MOFs dispersed within flexible elastomers or thermoplastics have been shown to provide uniform filler dispersion and effective energy absorption via the organic–inorganic interface. Bi2O3/PMMA composites routinely matched or exceeded the shielding effectiveness of Pb aprons at tube voltages between 60 and 120 kVp, demonstrating their practicality for medical radiation protection.38,89,90 In these systems, the combination of heavy-metal oxides or MOF domains with lightweight polymer matrices allows the formation of flexible, non-toxic, and high-performance shielding films suitable for wearable or portable protection applications.
 | ||
| Fig. 8 (a) Dose ratio (I/I0) of X-ray passed through the two composites (W/POE and Bi/POE); (b) linear attenuation coefficient versus the amount of metal content; (c) mass attenuation coefficient versus the amount of metal content; (d) digital images of the W/POE and Bi/POE composites. Reproduced with permission from Li et al.88 © The Author(s) 2021. | ||
Further innovations involve hybrid hierarchical composites, such as Gd2O(CO3)2@Bi-MOF/graphene systems, which integrate multiple functional components for synergistic attenuation and enhanced conductivity. Gadolinium (Gd) offers strong neutron absorption capability due to its high neutron-capture cross-section, while Bi-MOFs contribute to γ-ray attenuation, and graphene improves structural integrity and electron transport.91 These multifunctional composites demonstrate superior shielding efficiency at 59.5 keV γ-radiation and can be extended to mixed-field environments involving γ, neutron, and charged-particle exposure.92 Moreover, the inclusion of graphene or carbon nanotubes helps to reinforce mechanical stability and mitigate brittleness in MOF-based composites, ensuring durability during repeated radiation exposure or physical stress.91
For neutron or charged-particle environments, epoxy-based composites incorporating Gd-MOFs represent an emerging class of hybrid shields. Gd-MOFs provide effective neutron capture while the hydrogenous epoxy matrix moderates incoming fast neutrons through elastic scattering.87 The combination of moderation, absorption, and mechanical toughness offers an attractive alternative to traditional boron-based polymers or heavy concretes, especially in aerospace and nuclear instrumentation applications where weight reduction is critical. The ability to tailor the composite structure at the molecular level through MOF chemistry further allows optimization of both attenuation and mechanical performance.
In evaluating performance benchmarks, Pb remains the reference standard for photon shielding due to its high density (11.3 g cm−3) and continuous spectrum of attenuation interactions.93 However, edge-engineered, lead-free MOF composites can rival or even surpass Pb performance in the 45–90 keV range relevant to diagnostic imaging, particularly when leveraging Bi/W edge complementarity.88 For higher γ-ray energies (e.g., 662 keV from 137Cs), dense W/epoxy composites with high filler loadings can approach the attenuation of concrete, though pure MOF fillers alone remain less competitive without densification or oxide conversion.94 Nevertheless, the lightweight, flexible, and non-toxic nature of MOF-based composites provides a critical advantage for wearable shielding, radiation dosimetry housings, and mobile protective barriers, areas where Pb's toxicity and rigidity are major limitations.95
The path forward for MOF-based radiation shielding would involve the integration of material design and processing strategies. Techniques such as high-pressure compaction, in situ polymerization, and thermal densification can increase MOF density without collapsing porosity, enhancing attenuation while preserving chemical tunability. Additionally, computational simulations using Monte Carlo and SRIM models aid in predicting mass attenuation coefficients and optimizing element composition for specific radiation energies.96,97 The incorporation of MOFs into multifunctional composites that combine shielding, sensing, and structural properties could enable smart protective materials capable of self-diagnosis and radiation monitoring. Overall, by coupling high-Z metal content with lightweight frameworks and hierarchical structuring, MOF-based materials are poised to provide a new generation of eco-friendly, flexible, and high-performance radiation shields that balance efficiency, processability, and safety for diverse technological and biomedical applications. Table 4 presents the attenuation metrics for high-Z MOF composites vs conventional fillers. The table shows MOF identity, matrix, loading (vol%), density, µ/ρ at 60/80/120 kVp or 662 keV, HVL (mm), flexural modulus/strain, and processing route.
| MOF/Composite | Mechanism | Performance metric reported (numeric values & test conditions) | Radiation range/conditions | Main findings | Ref. |
|---|---|---|---|---|---|
| µ/ρ = mass attenuation coefficient; µ = linear attenuation coefficient; HVL = half-value layer; I/I0 = transmitted intensity fraction; HU = Hounsfield units (CT contrast); GLAC = geometric half-value layer; POE = ethylene-octene copolymer; RL = radioluminescence intensity; dose = gamma dose in gray (Gy) or mega-gray (MGy); detection limit = minimum dose rate detectable | |||||
| Bi-NU-901 (Bi-MOF) | High-Z Bi nodes → strong X-ray attenuation/CT contrast | Quantitative CT contrast: at 30 mM (Bi or equivalent element concentration) and 30 kV: ≈14× better attenuation vs iodixanol; at 70 kV: ≈7× better than iodixanol | CT at 35, 50, 70 kV; concentrations 3.25–30 mM | Bi-NU-901 gives much higher CT contrast per attenuating-element concentration vs. Zr analogue and vs commercial iodinated agent | 38 |
| Tb-MOF-76 (lanthanide MOF microscintillator) | Tb3+ centers → radioluminescence (scintillation) | Detection limit (radioluminescent signal): 23 nGy s−1 (3σ/slope method). Spatial resolution (film detector): >16.6 line-pairs mm−1 | X-ray dose rates from nGy s−1 to µGy s−1; lab X-ray sources used (imaging demos) | Tb-MOF-76 microscintillators show very low detection limit and high spatial resolution for flexible X-ray detectors—excellent for low-dose imaging | 69 |
| Eu@UiO-66-(COOH)2, Tb@UiO-66-(COOH)2 (lanthanide-chelated UiO-66) | Lanthanide doping in robust Zr-MOF → X-ray scintillation/radioluminescence | Radioluminescence spectra/RL intensity reported; no µ/ρ/HVL reported—imaging/scintillation metrics only. | X-ray excitation (laboratory sources) | Post-synthetic metalation produces bright RL. It demonstrated that the UiO-66 platform can be converted into scintillators. | 98 |
| MIL-100 (Fe), MIL-100 (Cr), MIL-101 (Cr), and AlFu MOFs (radiation-stability studies) | Stability under γ irradiation (important for shielding materials) | MIL-101 (Cr): stable up to ∼4 MGy (Co-60 γ); Al-fum/Al-based MOFs began decomposing at ∼1 MGy | Co-60 γ up to multiple MGy (megagray) exposures | Some MOFs (MIL-100/101, certain UiO variants) are surprisingly radiation-tolerant to MGy doses; others decompose at much lower doses | 32 |
| ZIF-8 & BDC-Cu | Common MOFs studied for stability & potential hosts for heavy atoms | At 1.333 MeV, BDC-Cu GLAC reaches 0.106 cm−1, ∼17% of Pb shielding | 137Cs (0.662 MeV), 133Ba (0.356 MeV), and 60Co (1.173 and 1.333 MeV) sources | The GLAC/LACPb values at 1.333 MeV were found to be 15% for ZIF-8 and 17% for BDC-Cu, which indicated that BDC-Cu exhibited superior attenuation characteristics | 99 |
| Gd-MOF/polyimide (PI) films (Gd-MOF/PI) | Gd high thermal-neutron capture cross-section → neutron shielding | Simulated thermal neutron permeability I/I0 (SuperMC) for 1 wt% and 3 wt% Gd-MOF films: At 0.2 cm thickness 3 wt% film I/I0 ≈ 0.9% (thermal neutrons) | Neutron (thermal & fast) simulations and experiments (thickness varied 0–0.2 cm for thermal data; fast neutron tests up to 5 mm thickness) | Increase in Gd-MOF content improves thermal neutron attenuation significantly; performance competes with/exceeds B4C/PI and borated polyethylene for thin films | 87 |
| W/Bi multilayer polymer composite | Layered heavy-metal-polymer architecture | The multilayered polymer composites with a layer thickness ratio (POE/tungsten: POE/bismuth)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) = = ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 and 6 layers had the best X-ray shielding ability 3 and 6 layers had the best X-ray shielding ability |
70–90 keV range | The density of the multilayered polymer composites with 6 layers (7.81 g cm−3) was about 30% lighter than that of traditional lead materials (11.3 g cm−3). X-ray shielding ability can be enhanced by increasing the number of layers and by the synergistic effect of multi-layered interfaces | 88 |
| Bi2O3/Polymethylmethaacrylate (PMMA) composite | Gd-based neutron capture + hydrogenous matrix moderation | High thermal neutron capture; improved moderation combined with polymer | 60–120 keV; (<59 keV to 1332 keV) | Easy processing, lighter. Highly effective, lead-free, safe, and lightweight shielding materials for X- and gamma rays | 94 |
4.3. Engineering MOFs for real-world long-term sensing and shielding applications
The long-term stability and mechanical robustness of MOFs are decisive factors determining their transition from laboratory-scale porous crystals to durable components in sensing and radiation-shielding devices. Over the past decade, advances in structural chemistry, defect control, and composite engineering have converged to establish clear principles for designing MOFs that can sustain long operational lifetimes under combined mechanical, chemical, and radiative stresses.Intrinsic chemical robustness (resistance to hydrolysis, oxidation, and ligand displacement) forms the baseline for durability. High-valence, high-connectivity node frameworks such as Zr4+-, Hf4+-, and Th4+-based systems (e.g., the UiO and MIL series) exhibit exceptional chemical and thermal stability compared to more labile Cu- or Zn-carboxylates. For instance, UiO-66 maintains crystallinity and porosity across prolonged solvent exposure and thermal cycling, whereas HKUST-1 degrades rapidly in humid conditions.100 However, even chemically stable MOFs can exhibit mechanical fragility depending on defect concentration and node chemistry. The Ce-UiO analogues, for example, display reduced bulk moduli compared to Zr-UiO due to partial linker lability.101,102 Son et al.102 observed a trend of decreasing bulk moduli with decreasing coordination strength when UiO-66(Zr) and UiO-66(Hf) were compared to UiO-66(Ce), revealing a distinctive behaviour of UiO-66 in response to pressures. Thus, chemical endurance must be integrated with mechanical metrics to predict service reliability.
The mechanical response of MOFs arises from a complex interplay between topology, pore fraction, and linker rigidity. Nanoindentation and in situ high-pressure X-ray diffraction reveal moduli spanning several orders of magnitude (from ∼2 GPa for flexible, open frameworks to >20 GPa for dense Zr/Hf systems.103,104 Interestingly, flexible “breathing” frameworks such as MIL-53(Al) can better resist amorphization under pressure by accommodating strain through reversible linker rotation, whereas overly rigid lattices may fracture under similar stress. This demonstrates that optimal mechanical design requires a balance between flexibility and stiffness.
Defect chemistry further governs long-term mechanical stability. Missing-linker or missing-cluster defects often lower stiffness and promote hydrolytic degradation, but controlled defect densities can enhance ductility and delay fracture. For instance, partially defective UiO-66 samples exhibited higher strain tolerance under uniaxial compression without significant loss of adsorption performance.105 Mechanical optimization, therefore, hinges on tuning defect populations to balance flexibility and fatigue resistance.
Shaping and composite integration strongly influence mechanical performance. Pelletization, monolith formation, and mixed-matrix membrane fabrication enable scalable device architectures but may introduce intergranular stress. Embedding MOFs in polymer matrices or coating them with covalently grafted polymers has proven to be an effective means of combining mechanical integrity and environmental resistance. Polymer-encapsulated UiO-66 films demonstrated over 50-fold improved tensile strength while sustaining gas selectivity.106 Similarly, polymer/MOF nanocomposites offer superior crack resistance and flexibility,107 highlighting the mechanical synergy between inorganic frameworks and polymeric elasticity.
Under ionizing radiation, mechanical robustness becomes a more complex challenge. Photon (γ/X-ray) exposure primarily induces linker scission and partial amorphization, while heavy ions and electrons create localized defect clusters that act as crack nucleation sites. Zr- and Th-based MOFs exhibit the highest radiation tolerance, retaining crystallinity up to MGy doses before surface degradation appears.33 However, radiolytic removal of pore-filling guests can lead to embrittlement, coupling chemical deterioration with stress-induced failure. Reliable lifetime modeling must therefore integrate radiation dose, defect evolution, and fatigue kinetics.
Several strategies have been validated for reinforcing MOFs mechanically. Selecting rigid aromatic linkers coordinated to high-valence metal clusters ensures uniform stress distribution, as in UiO-66 and MIL-53(Al), which sustain pressures of several hundred MPa.108 Controlled defect engineering enhances ductility without sacrificing porosity. Densification through extraction or sol–gel methods yields monolithic MOFs with improved load transfer,109 and post-synthetic crosslinking or metal exchange stiffens the coordination network, extending fatigue life under cyclic stress.33 Hydrophobic surface treatments such as PDMS coatings further protect against moisture-driven cracking.
These insights emphasize that achieving practical reliability in MOF-based sensing and shielding materials requires a holistic co-design approach. Accelerated qualification protocols that couple mechanical cycling, humidity and temperature variation, and photon/ion irradiation while tracking both structural and functional performance, provide the most realistic durability assessments. This requires the convergence of chemistry, morphology, and protective engineering to transform MOFs from fragile powders into robust, multifunctional materials for long-term technological deployment.
5. Challenges, opportunities, and prospects
The deployment of MOFs in high-energy radiation environments is not without significant hurdles. However, these challenges also present clear pathways for innovation and material advancement. The primary obstacles hindering the widespread adoption of MOFs in radiological applications stem from methodological inconsistencies, environmental vulnerabilities, and analytical limitations.(iv) Lack of standardized irradiation protocols and comparative stability metrics: a critical barrier to progress is the absence of standardized testing. Studies employ vastly different radiation sources (gamma, X-ray, neutrons, electrons), dose rates (Gy s−1 to MGy h−1), and total absorbed doses, making direct comparison of MOF stability nearly impossible.57,110 Furthermore, there is no consensus on the metrics for “stability”, which can be measured by crystallinity loss, porosity reduction, ligand degradation, or changes in sorption capacity.
(v) Sensitivity to combined stressors (thermal cycling, vacuum, humidity): real-world environments, such as nuclear reactors or space, subject materials to simultaneous stressors. While a MOF may exhibit radiation resistance under inert conditions, its performance can degrade rapidly when irradiation is coupled with thermal cycling, vacuum (which can accelerate ligand loss), or humidity (where radiolysis of water within pores generates highly damaging secondary radicals like ˙OH).32 This synergistic effect is often overlooked in preliminary studies.
(vi) Limited in situ tools for capturing ultrafast radiation-induced processes: The initial events following radiation absorption—such as exciton formation, energy transfer, bond cleavage, and electron–hole pair generation—occur on picosecond to femtosecond timescales. Most analytical techniques (e.g., PXRD, gas sorption) are ex situ and only provide a “before and after” snapshot, missing the transient species and mechanisms responsible for degradation or resistance.111,112 Developing and utilizing in situ characterization under beam conditions is essential but technically challenging.
To overcome these challenges, researchers are pursuing sophisticated material design strategies to engineer MOFs with inherent radiation hardness.
(i) Linker engineering with radiation-resistant π-conjugated ligands: a promising approach involves designing linkers with extended aromaticity and π-conjugation. These systems can efficiently dissipate absorbed radiation energy as heat or fluorescence without undergoing permanent bond scission. Ligands like pyrene, porphyrins, and other polycyclic aromatic hydrocarbons have shown enhanced radiation resistance compared to simpler carboxylates like terephthalate (BDC).32,110
(ii) Post-synthetic modifications for defect healing and guest insertion: MOFs' synthetic tunability allows for treatments after initial synthesis. Post-synthetic modification (PSM) can be used to “heal” radiation-induced defects or install functional groups that act as radical scavengers. Similarly, post-synthetic infusion of radiation-protective guest molecules, such as iodine or hydrogen-rich molecules, into the MOF pores can significantly enhance stability by absorbing energy and mitigating damage to the framework.113
(iii) MOF–nanocarbon or MOF–metal hybrid architectures for combined detection and shielding: creating composites leverages the strengths of different materials. Integrating MOFs with carbon nanotubes or graphene can create a conductive network for enhanced radiation-induced electrical signal changes (sensing) while simultaneously improving mechanical strength and radiation shielding through their high-Z carbon content.114 Similarly, embedding nanoparticles of high-Z metals (e.g., Bi, W) within a MOF matrix can create a hybrid material that excels at both shielding gamma rays and sensing neutron radiation via the MOF's luminescent response.
The future of MOFs in high-energy fields is exceptionally bright, contingent upon a coordinated effort between synthesis, characterization, and theory.
(i) Outlook on their integration into next-generation systems: MOFs are poised for integration into multifunctional systems for nuclear facilities (e.g., as smart coatings that sense and capture radionuclides), in medical imaging and radiotherapy (e.g., as responsive dosimeters or drug carriers), and for space missions (e.g., lightweight radiation shielding and gas sensing in habitats).115 Their multifunctionality is their key advantage over conventional materials.
(ii) Design roadmap for MOFs in high-radiation environments: a systematic roadmap is needed. This involves: (1) high-throughput computational screening to identify promising building blocks a priori; (2) standardized irradiation testing protocols with a focus on combined environmental stressors; and (3) a focus on scalability and processing of MOFs into practical forms like coatings, membranes, or monoliths.
(iii) Need for predictive computational models: empirical testing is slow and costly. The development of predictive models is crucial. Density Functional Theory can calculate bond dissociation energies and simulate the behaviour of excited states upon radiation exposure. Classical or ab initio molecular dynamics simulations can model the cascading effects of primary knock-on atoms and predict damage propagation within the framework over time, guiding the synthesis of the most robust materials.
6. Conclusions
Metal–organic frameworks represent a paradigm shift in materials science for radiation environments. Their inherent structural and functional tunability offer unparalleled opportunities to create devices that simultaneously sense, shield, and even separate radioactive species. While significant challenges remain—particularly in standardizing stability assessment and understanding degradation mechanisms under complex conditions—the targeted strategies of linker engineering, composite formation, and post-synthetic modification provide a clear path forward. The convergence of advanced in situ characterization, predictive computational modeling, and innovative synthesis will be instrumental in unlocking the full potential of MOFs, paving the way for their deployment in the demanding high-radiation environments of the future.Conflicts of interest
There are no conflicts to declare.Data availability
No primary research results, software, or code have been included, and no new data were generated or analysed as part of this review.References
- A. Schoedel and O. M. Yaghi, Macrocyclic and Supramolecular Chemistry: How Izatt–Christensen Award Winners Shaped the Field, 2016 Search PubMed.
- G. A. Udourioh, M. M. Solomon and E. I. Epelle, Cell. Mol. Bioeng., 2021, 14, 535–553 CrossRef CAS.
- G. A. Udourioh, M. M. Solomon, C. O. Matthews-Amune, E. I. Epelle, J. A. Okolie, V. E. Agbazue and U. Onyenze, React. Chem. Eng., 2023, 8, 278–310 RSC.
- L. Longley, C. Calahoo, R. Limbach, Y. Xia, J. M. Tuffnell, A. F. Sapnik, M. F. Thorne, D. S. Keeble, D. A. Keen, L. Wondraczek and T. D. Bennett, Nat. Commun., 2020, 11, 5800 CrossRef CAS PubMed.
- K. J. Wu, C. Wu, M. Fang, B. Ding, P. P. Liu, M. X. Zhou, Z. Y. Gong, D. L. Ma and C. H. Leung, TrAC, Trends Anal. Chem., 2021, 143, 116384 CrossRef CAS.
- M. Ahmadipour, P. Damacet, C. Xiang, K. A. Mirica and R. Montazami, ACS Appl. Mater. Interfaces, 2025, 17, 12425–12439 CrossRef CAS.
- J. Lu, X. H. Xin, Y. J. Lin, S. H. Wang, J. G. Xu, F. K. Zheng and G. C. Guo, Dalton Trans., 2019, 48, 1722–1731 RSC.
- M. J. Berger, J. H. Hubbell, S. M. Seltzer, J. Chang, J. S. Coursey, R. Sukumar, D. S. Zucker and K. Olsen, NIST Standard Reference Database 8 (XGAM), 1998.
- H. Salway, X. W. Chua and M. Anaya, Mater. Horiz., 2025, 12, 8989–9008 RSC.
- A. G. Al Lafi, B. Assfour and T. Assaad, J. Mater. Sci., 2021, 56, 12154–12170 CrossRef CAS.
- S. E. Gilson, M. Fairley, P. Julien, A. G. Oliver, S. L. Hanna, G. Arntz, O. K. Farha, J. A. Laverne and P. C. Burns, J. Am. Chem. Soc., 2020, 142, 13299–13304 CrossRef CAS.
- C. Volkringer, C. Falaise, P. Devaux, R. Giovine, V. Stevenson, F. Pourpoint, O. Lafon, M. Osmond, C. Jeanjacques, B. Marcillaud, J. C. Sabroux and T. Loiseau, Chem. Commun., 2016, 52, 12502–12505 RSC.
- S. L. Hanna, D. X. Rademacher, D. J. Hanson, T. Islamoglu, A. K. Olszewski, T. M. Nenoff and O. K. Farha, Ind. Eng. Chem. Res., 2020, 59, 7520–7526 CrossRef CAS.
- S. Mauree, Scintillating Metal Organic Frameworks (MOFs) for radioactive gas detection, Material chemistry, Thesis, Université Paris-Saclay, 2023, English. NNT: 2023UPAST151.
- M. Orfano, J. Perego, F. Cova, C. X. Bezuidenhout, S. Piva, C. Dujardin, B. Sabot, S. Pierre, P. Mai, C. Daniel, S. Bracco, A. Vedda, A. Comotti and A. Monguzzi, Nat. Photonics, 2023, 17, 672–678 CrossRef CAS.
- C. Ma, H. Liu, H. T. Wolterbeek, A. G. Denkova and P. S. Crespo, Langmuir, 2022, 38, 8928–8933 CrossRef CAS.
- J. H. Hubbell and S. M. Seltzer, NIST Standard Reference Database 126: X-Ray Mass Attenuation Coefficients, 1996.
- O. Ugurlu, J. Haus, A. A. Gunawan, M. G. Thomas, S. Maheshwari, M. Tsapatsis and K. A. Mkhoyan, Phys. Rev. B: Condens. Matter Mater. Phys., 2011, 83, 113408 CrossRef.
- S. Ghosh, P. Kumar, S. Conrad, M. Tsapatsis and K. A. Mkhoyan, Microsc. Microanal., 2019, 25, 1704–1705 CrossRef.
- K. Gnanasekaran, R. dos Reis and N. Gianneschi, Microsc. Microanal., 2022, 28, 2170–2172 CrossRef.
- D. Mücke, I. Cooley, B. Liang, Z. Wang, S. W. Park, R. Dong, X. Feng, H. Qi, E. Besley and U. Kaiser, Nano Lett., 2024, 24, 3014–3020 CrossRef PubMed.
- J. F. Ziegler, M. D. Ziegler and J. P. Biersack, Nucl. Instrum. Methods Phys. Res., Sect. B, 2010, 268, 1818–1823 CrossRef CAS.
- B. Chen, J. Wang, L. Peng, Q. Wang, Y. Wang and X. Xu, Adv. Funct. Mater., 2024, 34, 2310270 CrossRef CAS.
- P. Banerjee, K. L. Kollmannsberger, R. A. Fischer and J. R. Jinschek, Microsc. Microanal., 2023, 29, 1253–1254 CrossRef.
- Y. Zhu, J. Ciston, B. Zheng, X. Miao, C. Czarnik, Y. Pan, R. Sougrat, Z. Lai, C. E. Hsiung, K. Yao, I. Pinnau, M. Pan and Y. Han, Nat. Mater., 2017, 16, 532–536 CrossRef CAS PubMed.
- S. Ghosh, P. Kumar, S. Conrad, M. Tsapatsis and K. A. Mkhoyan, Microsc. Microanal., 2019, 25, 1704–1705 CrossRef.
- S. L. Hanna, D. X. Rademacher, D. J. Hanson, T. Islamoglu, A. K. Olszewski, T. M. Nenoff and O. K. Farha, Ind. Eng. Chem. Res., 2020, 59, 7520–7526 CrossRef CAS.
- X. Ding, S. Xiao, T. Wang, Z. Zeng, X. Zhao and Q. Yang, Microporous Mesoporous Mater., 2023, 354, 112533 CrossRef CAS.
- S. E. Gilson, M. Fairley, P. Julien, A. G. Oliver, S. L. Hanna, G. Arntz, O. K. Farha, J. A. Laverne and P. C. Burns, J. Am. Chem. Soc., 2020, 142, 13299–13304 CrossRef CAS PubMed.
- S. Mauree, V. Villemot, M. Hamel, B. Sabot, S. Pierre, C. Dujardin, F. Belloni, A. Comotti, S. Bracco, J. Perego and G. H. V. Bertrand, Adv. Funct. Mater., 2023, 33, 2302877 CrossRef CAS.
- C. Volkringer, C. Falaise, P. Devaux, R. Giovine, V. Stevenson, F. Pourpoint, O. Lafon, M. Osmond, C. Jeanjacques, B. Marcillaud, J. C. Sabroux and T. Loiseau, Chem. Commun., 2016, 52, 12502–12505 RSC.
- C. Ma, H. Liu, H. T. Wolterbeek, A. G. Denkova and P. S. Crespo, Langmuir, 2022, 38, 8928–8933 CrossRef CAS PubMed.
- S. K. Elsaidi, M. H. Mohamed, A. S. Helal, M. Galanek, T. Pham, S. Suepaul, B. Space, D. Hopkinson, P. K. Thallapally and J. Li, Nat. Commun., 2020, 11, 1–8 CrossRef.
- G. Buxton, Radiat. Chem., 2021, 3–16 Search PubMed.
- J. Liu, Y. Fang, Y. Ou, X. Shi, Y. Zhang, Q. Chen, L. Li, F. Zhou and W. Liu, J. Mater. Sci. Technol., 2025, 220, 140–149 CrossRef CAS.
- J. Liu, Y. Qian, D. Li, W. Wu, M. Zhang, J. Yan, B. Li and F. Zhou, Friction, 2024, 12, 1499–1511 CrossRef CAS.
- L. Robison, L. Zhang, R. J. Drout, P. Li, C. R. Haney, A. Brikha, H. Noh, B. L. Mehdi, N. D. Browning, V. P. Dravid, Q. Cui, T. Islamoglu and O. K. Farha, ACS Appl. Bio Mater., 2019, 2, 1197–1203 CrossRef CAS PubMed.
- A. M. Hastings, M. Fairley, M. C. Wasson, D. Campisi, A. Sarkar, Z. C. Emory, K. Brunson, D. B. Fast, T. Islamoglu, M. Nyman, P. C. Burns, L. Gagliardi, O. K. Farha, A. E. Hixon and J. A. Laverne, Chem. Mater., 2022, 34, 8403–8417 CrossRef CAS.
- X. Meng, B. Gui, D. Yuan, M. Zeller and C. Wang, Sci. Adv., 2016, 2, e1600480 CrossRef PubMed.
- P. Thallapally, A. J. Robinson, J. Crum, M. S. Murphy and M. R. Lish DOI:10.2172/1889731.
- A. Zheng, K. Yin, R. Pan, M. Zhu, Y. Xiong and L. Sun, Nanomaterials, 2023, 13, 1742 CrossRef CAS PubMed.
- D. Zhang, Y. Zhu, L. Liu, X. Ying, C. E. Hsiung, R. Sougrat, K. Li and Y. Han, Science, 2018, 359, 675–679 CrossRef CAS PubMed.
- X. Xu, L. Xia, C. Zheng, Y. Liu, D. Yu, J. Li, S. Zhong, C. Li, H. Song, Y. Liu, T. Sun, Y. Li, Y. Han, J. Zhao, Q. Lin, X. Li and Y. Zhu, Nat. Commun., 2025, 16, 1–15 Search PubMed.
- B. W. Jacobs, R. J. T. Houk, B. M. Wong, A. A. Talin and M. D. Allendorf, Nanotechnology, 2011, 22, 375601 CrossRef.
- G. Li, M. Xu, W. Q. Tang, Y. Liu, C. Chen, D. Zhang, L. Liu, S. Ning, H. Zhang, Z. Y. Gu, Z. Lai, D. A. Muller and Y. Han, Nat. Commun., 2025, 16, 1–8 Search PubMed.
- F. Banihashemi, G. Bu, A. Thaker, D. Williams, J. Y. S. Lin and B. L. Nannenga, Ultramicroscopy, 2020, 216, 113048 CrossRef CAS PubMed.
- J. F. Ziegler, M. D. Ziegler and J. P. Biersack, Nucl. Instrum. Methods Phys. Res., Sect. B, 2010, 268, 1818–1823 CrossRef CAS.
- N. Ishikawa, T. Sonoda, T. Sawabe, H. Sugai and M. Sataka, Nucl. Instrum. Methods Phys. Res., Sect. B, 2013, 314, 180–184 CrossRef CAS.
- M. Fairley, S. E. Gilson, S. L. Hanna, A. Mishra, J. G. Knapp, K. B. Idrees, S. Chheda, H. Traustason, T. Islamoglu, P. C. Burns, L. Gagliardi, O. K. Farha and J. A. Laverne, Chem. Mater., 2021, 33, 9285–9294 CrossRef CAS.
- R. A. Rymzhanov, N. Medvedev, J. H. O’Connell, A. Janse van Vuuren, V. A. Skuratov and A. E. Volkov, Sci. Rep., 2019, 9, 1–10 CrossRef CAS PubMed.
- M. Lang, C. L. Tracy, R. I. Palomares, F. Zhang, D. Severin, M. Bender, C. Trautmann, C. Park, V. B. Prakapenka, V. A. Skuratov and R. C. Ewing, J. Mater. Res., 2015, 30, 1366–1379 CrossRef CAS.
- Y. L. Wang, S. Zhang, Y. F. Zhao, J. Bedia, J. J. Rodriguez and C. Belver, J. Environ. Chem. Eng., 2021, 9, 106087 CrossRef CAS.
- A. Ashfaq, M. C. Clochard, X. Coqueret, C. Dispenza, M. S. Driscoll, P. Ulański and M. Al-Sheikhly, Polymers, 2020, 12, 2877 CrossRef CAS PubMed.
- T. Zaharescu and M. Mariş, J. Compos. Sci., 2022, 6, 109 CrossRef CAS.
- V. V. Butova, V. R. Zdravkova, O. A. Burachevskaia, A. A. Tereshchenko, P. S. Shestakova and K. I. Hadjiivanov, Nanomaterials, 2023, 13, 1675 CrossRef CAS.
- T. He, Y. Zhou, P. Yuan, J. Yin, L. Gutiérrez-Arzaluz, S. Chen, J. X. Wang, S. Thomas, H. N. Alshareef, O. M. Bakr and O. F. Mohammed, ACS Energy Lett., 2023, 8, 1362–1370 CrossRef CAS.
- H. He, X. Wang, Q. Yu, W. Wu, X. Feng, D. Kong, X. Ren and J. Gao, Catalysts, 2023, 13, 876 CrossRef CAS.
- J. Xu, H. Li, H. Feng, Y. Ma, D. Wang and J. Zhu, J. Alloys Compd., 2024, 976, 173347 CrossRef CAS.
- S. Chen, X. Li, E. Dong, H. Lv, X. Yang, R. Liu and B. Liu, J. Phys. Chem. C, 2019, 123, 29693–29707 CrossRef CAS.
- A. R. Dok, S. Radhakrishnan, F. de Jong, E. Becquevort, O. Deschaume, V. C. Chandran, Y. de Coene, C. Bartic, M. Van der Auweraer, W. Thiele-mans, C. Kirschhock, M. A. van der Veen,, T. Verbiest, E. Breynaert and S. Van Cleuvenbergen, Amorphous-to-crystalline transformation: how cluster aggregation drives the multi-step nucleation of ZIF-8, J. Am. Chem. Soc., 2025, 147, 8455–8466 CrossRef CAS PubMed.
- X. Xu, L. Xia, C. Zheng, Y. Liu, D. Yu, J. Li, S. Zhong, C. Li, H. Song, Y. Liu, T. Sun, Y. Li, Y. Han, J. Zhao, Q. Lin, X. Li and Y. Zhu, Nat. Commun., 2025, 16, 261 CrossRef CAS PubMed.
- Y. Wang, X. Liu, X. Li, F. Zhai, S. Yan, N. Liu, Z. Chai, Y. Xu, X. Ouyang and S. Wang, J. Am. Chem. Soc., 2019, 141, 8030–8034 CrossRef CAS PubMed.
- J. Perego, I. Villa, A. Pedrini, E. C. Padovani, R. Crapanzano, A. Vedda, C. Dujardin, C. X. Bezuidenhout, S. Bracco, P. E. Sozzani, A. Comotti, L. Gironi, M. Beretta, M. Salomoni, N. Kratochwil, S. Gundacker, E. Auffray, F. Meinardi and A. Monguzzi, Composite fast scintillators based on high-Z fluorescent metal–organic framework nanocrystals, Nat. Photonics, 2021, 15, 393–400 CrossRef CAS.
- H. T. Sun and N. Shirahata, Nat. Photonics, 2023, 17, 646–647 CrossRef CAS.
- M. Ahmadipour, P. Damacet, C. Xiang, K. A. Mirica and R. Montazami, ACS Appl. Mater. Interfaces, 2025, 17, 12425–12439 CrossRef CAS PubMed.
- Z. Li, L. Tian, W. Wu, L. Feng, B. Khaniyev, Y. Mukhametkarimov, M. Ibraimov, F. Zhou, W. Liu and J. Liu, Small Methods, 2025, 9, 2401485 CrossRef CAS PubMed.
- M. Xu, J. Liu, W. Wu, Y. Chen, D. Ma, S. Chen, W. Jie, M. Zhu and Y. Xu, Nano Res., 2024, 17, 2988–2993 CrossRef CAS.
- X. Zhang, H. Qiu, W. Luo, K. Huang, Y. Chen, J. Zhang, B. Wang, D. Peng, Y. Wang and K. Zheng, Adv. Sci., 2023, 10, 2207004 CrossRef CAS PubMed.
- L. Zhang, X. Wang, X. Wang, X. Wang, Y. Luo, C. Tan, L. Jiang, Y. Wang and W. Liu, Inorg. Chem., 2023, 62, 6421–6427 CrossRef CAS PubMed.
- P. K. Wang, W. F. Wang, B. Y. Li, R. X. Qian, Z. X. Gao, S. H. Wang, F. K. Zheng and G. C. Guo, Inorg. Chem. Front., 2025, 12, 1040–1048 RSC.
- X. Liu, R. Li, X. Xu, Y. Jiang, W. Zhu, Y. Yao, F. Li, X. Tao, S. Liu, W. Huang, Q. Zhao, X. Liu, R. Li, X. Xu, Y. Jiang, Y. Yao, F. Li, X. Tao, S. Liu, W. Huang, Q. Zhao and W. Zhu, Adv. Mater., 2023, 35, 2206741 CrossRef CAS PubMed.
- Z. Zheng, J. Qiu, H. Lu, J. Q. Wang and J. Lin, RSC Adv., 2022, 12, 12878–12881 RSC.
- L. Cheng, C. Liang, B. Li, H. Qin, P. Mi, B. Chen, Y. Yan, X. Dai, C. Zhang, Y. Wang, Y. Wang and S. Wang, Cell Rep. Phys. Sci., 2022, 3, 101004 CrossRef CAS.
- L. Zhou, J. Huang, L. Windgaetter, C. S. Ong, X. Zhao, C. Zhang, M. Tang, Z. Li, C. Qiu, S. Latini, Y. Lu, D. Wu, H. Gou, A. T. S. Wee, H. Hosono, S. G. Louie, P. Tang, A. Rubio and H. Yuan, Nat. Mater., 2022, 21, 773–778 CrossRef CAS PubMed.
- X. Zhu, T. He, X. Song, O. Shekhah, S. Thomas, H. Jiang, W. Wu, T. He, V. Guillerm, A. Shkurenko, J. X. Wang, H. N. Alshareef, O. M. Bakr, M. Eddaoudi and O. F. Mohammed, Adv. Mater., 2024, 36, 2412432 CrossRef CAS PubMed.
- Z. Zheng, J. Qiu, H. Lu, J. Q. Wang and J. Lin, RSC Adv., 2022, 12, 12878–12881 RSC.
- M. Orfano, J. Perego, C. X. Bezuidenhout, I. Villa, R. Lorenzi, B. Sabot, S. Pierre, S. Bracco, S. Piva, A. Comotti and A. Monguzzi, Adv. Funct. Mater., 2024, 34, 2404480 CrossRef CAS.
- T. Wang, X. Xu, S. Yao, J. Yu, Z. Yang, J. Qiu, Y. Yang and X. Yu, Adv. Opt. Mater., 2024, 12, 2302203 CrossRef CAS.
- S. N. Mane, S. K. Kolekar, K. B. Sapnar and S. D. Dhole, J. Radioanal. Nucl. Chem., 2025, 2025, 1–11 Search PubMed.
- C. Ma, I. Sánchez-García, R. Wang, H. Galán, A. G. Denkova and P. Serra Crespo, Sep. Purif. Technol., 2025, 352, 128212 CrossRef CAS.
- J. X. Wang, O. Shekhah, O. M. Bakr, M. Eddaoudi and O. F. Mohammed, Chem, 2025, 11, 102273 CAS.
- J. Mi, Q. Li, B. Li, W. Wang, S. Wang, F. Zheng and G. Guo, ACS Appl. Mater. Interfaces, 2024, 16, 9002–9011 CrossRef CAS PubMed.
- C. Q. Lin, J. Li, J. Shi, J. R. Wang, R. L. Bo, L. K. Gong, C. Y. Pan, X. W. Lei and Z. Gong, Inorg. Chem., 2025, 64, 18490–18498 CrossRef CAS PubMed.
- X. Cao, H. Li, J. Yi, F. Zhao and Y. Hao, J. Phys. Conf. Ser., 2023, 2478, 062029 CrossRef.
- B. Ortaç, S. Mutlu, A. Hakan Yilmaz, N. Arsu and S. Savaskan Yilmaz, Transition Metals – From Fundamentals to Frontiers, IntechOpen, 2025 Search PubMed.
- C. Hu, Q. Huang and Y. Zhai, RSC Adv., 2021, 11, 40148–40158 RSC.
- Z. Li, W. Zhou, X. Zhang, Y. Gao and S. Guo, Sci. Rep., 2021, 11, 1–13 CrossRef PubMed.
- E. O. Echeweozo, M. Kırkbınar, S. Alomairy and M. S. Al-Buriahi, J. Radiat. Res. Appl. Sci., 2025, 18, 101247 CAS.
- D. Cao, M. Bourham and G. Yang, ACS Appl. Eng. Mater., 2024, 2, 1966–1975 CrossRef CAS.
- J. Chen, X. Zhang, W. Xiao, D. Pan and W. Wu, Front. Mater., 2025, 12, 1561198 CrossRef.
- S. Tan, P. Jiang, J. Liu, X. Yang, Y. Lai, X. Hu, G. Peng and G. Wang, Polym. Compos., 2025, 46, 5618–5630 CrossRef CAS.
- J. P. McCaffrey, H. Shen, B. Downton and E. Mainegra-Hing, Med. Phys., 2007, 34, 530–537 CrossRef CAS PubMed.
- A. H. Alsaab and S. Zeghib, Polymers, 2023, 15, 2142 CrossRef CAS PubMed.
- S. Wu, W. Zhang and Y. Yang, Polymers, 2024, 16, 3274 CrossRef CAS PubMed.
- P. Ma, X. Wu, Y. Zhang, D. Wang, W. Tian, S. Qiu and G. Su, Nucl. Eng. Technol., 2025, 57, 103346 CrossRef CAS.
- Q. Ma, Y. Wang, X. Zhang, Q. Zhao, J. guo, J. Guo, X. Ren, J. Huang, Y. Zhang, Y. Xie and J. Hao, Mater. Sci. Eng., B, 2025, 311, 117765 CrossRef CAS.
- Z. Ali, F. Liu, Y. Wang, H. G. Rasool, F. Wang and M. Haseeb, Advancements in primary radiation damage models and SRIM simulations: A review of radiation damage predictions, Nuclear Eng. Technol., 2025, 57, 103570 CrossRef CAS.
- T. A. Hamdalla, S. A. M. Issa, H. M. H. Zakaly and A. M. A. Mostafa, Appl. Radiat. Isot., 2025, 226, 112149 CrossRef CAS PubMed.
- J. H. Cavka, S. Jakobsen, U. Olsbye, N. Guillou, C. Lamberti, S. Bordiga and K. P. Lillerud, J. Am. Chem. Soc., 2008, 130, 13850–13851 CrossRef PubMed.
- L. R. Redfern, M. Ducamp, M. C. Wasson, L. Robison, F. A. Son, F. X. Coudert and O. K. Farha, Chem. Mater., 2020, 32, 5864–5871 CrossRef CAS.
- F. A. Son, K. M. Fahy, M. A. Gaidimas, C. S. Smoljan, M. C. Wasson and O. K. Farha, Commun. Chem., 2023, 6, 1–8 CrossRef PubMed.
- L. R. Redfern and O. K. Farha, Chem. Sci., 2019, 10, 10666–10679 RSC.
- L. R. Redfern and O. K. Farha, Chem. Sci., 2019, 10, 10666–10679 RSC.
- W. Xiang, Y. Zhang, Y. Chen, C. J. Liu and X. Tu, J. Mater. Chem. A, 2020, 8, 21526–21546 RSC.
- G. E. B. Redwine, W. A. Braunecker and T. Gennett, ACS Mater. Au, 2024, 5, 268–298 CrossRef PubMed.
- D. Giliopoulos, A. Zamboulis, D. Giannakoudakis, D. Bikiaris and K. Triantafyllidis, Molecules, 2020, 25, 185 CrossRef CAS.
- F. Ahmadijokani, R. Mohammadkhani, S. Ahmadipouya, A. Shokrgozar, M. Rezakazemi, H. Molavi, T. M. Aminabhavi and M. Arjmand, Chem. Eng. J., 2020, 399, 125346 CrossRef CAS.
- B. Li, Y. Wang, B. Guo, D. Shi and S. Han, Braz. J. Chem. Eng., 2025, 1–27 Search PubMed.
- M. Panahi-Sarmad, S. Samsami, A. Ghaffarkhah, S. A. Hashemi, S. Ghasemi, M. Amini, S. Wuttke, O. Rojas, K. C. Tam, F. Jiang, M. Arjmand, F. Ahmadijokani and M. Kamkar, Adv. Funct. Mater., 2024, 34, 2304473 CrossRef CAS.
- T. Iuchi, Y. Yamada, M. Sugiura and A. Torao, Exp. Methods Phys. Sci., 2010, 43, 217–277 CAS.
- E. E. Khozemy, E. F. Salem and A. El-Hag Ali, Radiat. Phys. Chem., 2022, 193, 109976 CrossRef CAS.
- M. Hefayathullah, S. Singh, V. Ganesan and G. Maduraiveeran, Adv. Colloid Interface Sci., 2024, 331, 103210 CrossRef CAS PubMed.
- K. Jayaramulu, S. Mukherjee, D. M. Morales, D. P. Dubal, A. K. Nanjundan, A. Schneemann, J. Masa, S. Kment, W. Schuhmann, M. Otyepka, R. Zbořil and R. A. Fischer, Chem. Rev., 2022, 122, 17241–17338 CrossRef CAS PubMed.
- D. Li, A. Yadav, H. Zhou, K. Roy, P. Thanasekaran and C. Lee, Global Challenges, 2024, 8, 2300244 CrossRef.
- L. Jiao, J. Y. R. Seow, W. S. Skinner, Z. U. Wang and H. L. Jiang, Mater. Today, 2019, 27, 43–68 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |