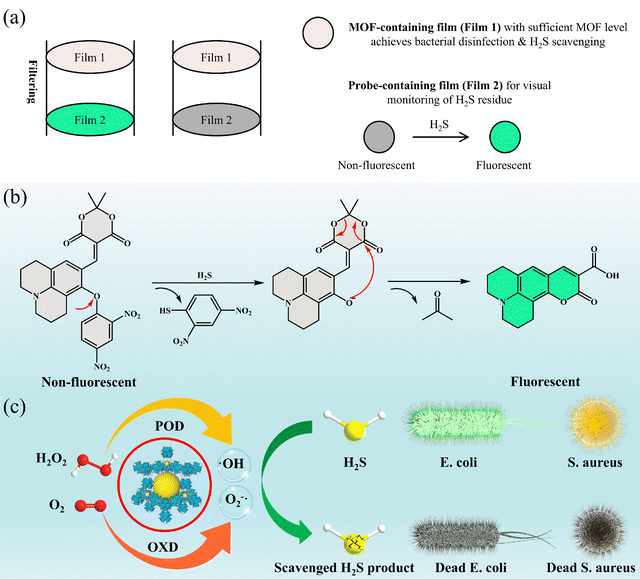
A dual-film platform for simultaneous bacterial disinfection and H2S scavenging with integrated fluorescent sensing for real-time visual monitoring
Xiangshan
Lei
,
Fang
Zeng
 * and
Shuizhu
Wu
* and
Shuizhu
Wu
 *
*
State Key Laboratory of Luminescent Materials and Devices, Guangdong Provincial Key Laboratory of Luminescence from Molecular Aggregates, School of Materials Science and Engineering, South China University of Technology, Wushan Road 381, Guangzhou, 510640, China. E-mail: mcfzeng@scut.edu.cn; shzhwu@scut.edu.cn
First published on 30th September 2025
Abstract
Slaughterhouse and meat-processing wastewater contains a complex mixture of organic pollutants, pathogenic bacteria, and toxic hydrogen sulfide (H2S), posing serious risks to both public health and the environment. To address these challenges, we have developed a dual-film platform comprising: (1) a cellulose acetate (CA) film loaded with a cerium-based metal–organic framework (Ce-MOF) for efficient bacterial disinfection and H2S scavenging, and (2) a CA film embedded with a fluorescent probe (JM-NO2) for real-time visual monitoring of H2S. The incorporation of JM-NO2 into the CA matrix enables both naked-eye visualization and smartphone-based quantification of H2S. The Ce-MOF film exhibits robust reactive oxygen species (ROS)-generating catalytic activity under mild conditions, enabling rapid microbial inactivation and effective H2S elimination. In a dual-film configuration, sequential filtration through the Ce-MOF and JM-NO2 films allows for simultaneous disinfection, H2S removal, and immediate visual detection of residual H2S. The system demonstrates excellent stability and sensitivity under environmentally relevant conditions. This dual-film platform offers a scalable and practical approach to wastewater treatment, integrating contaminant removal with real-time monitoring, and holds significant potential for environmental remediation and sustainable water management.
1. Introduction
Wastewater generated from slaughterhouses and meat processing plants poses a significant environmental burden due to its complex composition, which includes high levels of organic matter, nutrients, fats, oils, and a range of hazardous contaminants.1–3 These effluents originate from multiple stages of meat processing—including slaughtering, cleaning, deboning, and packaging—contributing to a substantial pollutant load.4,5 Among these contaminants, hydrogen sulfide (H2S) stands out as a particularly harmful pollutant.6,7 As a colorless gas which is highly soluble in water with a characteristic foul odor, H2S is highly toxic and corrosive, posing serious environmental, infrastructural, and public health concerns.8,9 Its release into the environment can lead to air and water pollution, corrosion of wastewater infrastructure, and oxygen depletion in aquatic ecosystems.10,11 Therefore, the efficient and sustainable removal of H2S from wastewater has become a pressing goal in modern environmental management.In addition to chemical pollutants, slaughterhouse wastewater frequently contains high concentrations of pathogenic bacteria,12 which can contaminate downstream water bodies, promote the spread of waterborne diseases, and disrupt aquatic ecosystems.13 Accordingly, effective strategies that can simultaneously eliminate H2S and inactivate microbial contaminants are essential for ensuring safe wastewater discharge and regulatory compliance.
Conventional methods for H2S detection and removal often suffer from significant drawbacks, including high operational costs and a lack of real-time monitoring capabilities.14–18 In recent years, fluorescent probes have become powerful tools for chemical sensing and bioimaging, offering excellent sensitivity, high selectivity and rapid response time. These advantages make them superior to conventional detection methods, particularly real-time monitoring applications.19–23 And fluorescent probes have been designed for the detection of H2S.24–28 Among them, film-based fluorescent sensors are particularly attractive due to their portability, reusability, and ease of integration into treatment systems. These films enable simple visual readouts without the need for complex instrumentation, making them ideal for on-site monitoring applications.29–33
Simultaneously, nanozymes—particularly metal–organic frameworks (MOFs)—have gained attention as promising agents for environmental remediation.34–39 MOFs are crystalline, porous materials constructed from metal ions coordinated with organic ligands.40–42 Compared to conventional nanozymes, MOFs offer well-defined structures and tunable properties.43–45 By modulating metal valence states (e.g., Ce(III)/Ce(IV) and Fe(II)/Fe(III)) and ligand functionalities (e.g., porphyrins, imidazoles), MOFs can mimic natural oxidase (OXD) and peroxidase (POD) activity.46–49 These enzyme-mimicking activities catalyze the generation of reactive oxygen species (ROS), including superoxide anions (˙O2−) and hydroxyl radicals (˙OH), which can both oxidize H2S and disinfect bacterial pathogens.50–55 Therefore, MOF-based nanozymes provide dual functions for simultaneous pollutant scavenging and antimicrobial activity.
In this study, we report a dual-film system that integrates chemical sensing, pollutant removal, and microbial disinfection into a single platform (Scheme 1). The first film consists of CA embedded with a cerium-based MOF (Ce-MOF), designed for the oxidative removal of H2S and antibacterial treatment. The Ce-MOF demonstrates both OXD-like and POD-like catalytic activities across a broad pH range, promoting robust ROS generation. These ROS serve a dual role by (1) oxidizing and neutralizing H2S and (2) effectively inactivating bacterial contaminants. The second film is composed of cellulose acetate (CA) doped with a small-molecule fluorescent probe, JM-NO2. This film is non-emissive under normal conditions but becomes highly fluorescent upon reacting with H2S, enabling rapid, visual, and quantitative detection of H2S in aqueous environments. JM-NO2 exhibits excellent sensitivity (detection limit: 23.8 nM), high selectivity and strong anti-interference capacity, making it suitable for real-world wastewater analysis.
Together, this dual-film system offers a multifunctional, low-cost, and environmentally friendly solution for slaughterhouse wastewater treatment. It enables (1) effective H2S scavenging, (2) broad-spectrum bacterial disinfection, and (3) real-time, visual monitoring of H2S levels. This integrated approach holds significant promise for advancing wastewater management practices in meat processing and other high-impact industries.
2. Results and discussion
2.1. Synthesis and optical response of JM-NO2 toward H2S
The fluorescent probe JM-NO2 was synthesized as outlined in Scheme S1. Its structure was fully characterized by 1H/13C NMR spectroscopy and high-resolution mass spectrometry (as shown in Fig. S1–S5). The probe's optical response to H2S was evaluated using NaHS as the H2S donor. As shown in Fig. 1a, JM-NO2 (20 μM) in phosphate buffer (PB) containing 2% DMSO exhibits negligible fluorescence in the absence of H2S. Upon addition of H2S, the fluorescence intensity at 487 nm (λex = 425 nm) increases progressively, reaching a plateau at ∼25 μM H2S. The fluorescence response is rapid, with equilibrium achieves within 25 minutes (Fig. S6), indicating the probe's high sensitivity and fast kinetics.A linear correlation between H2S concentration and fluorescence intensity can be observed (Fig. 1b). The limit of detection (LOD), calculated via the 3σ/k method, is 23.8 nM—well below the WHO limit for H2S in drinking water (1.47 μM).56 Compared with reported probes for H2S (Table S1), JM-NO2 offers key advantages, including a turn-on fluorescence response, lower detection limit, and the ability to detect H2S in water. The absorption maximum of JM-NO2 at 495 nm exhibits a blue shift to 422 nm in the presence of H2S, accompanied by a visible color change from brown to light yellowish green. UV-vis absorption titration confirmed this shift, with a decreasing 495 nm band and emerging 422 nm peak (Fig. 1c and Fig. S7).
JM-NO2 exhibits stable and robust fluorescence responses to H2S across a broad pH range (6.0–10.0), supporting its utility in environmental samples (Fig. 1d). To evaluate the selectivity and anti-interference capability of JM-NO2, a series of substances (cations, anions, and biothiols) were added to the JM-NO2 solution, including cations (Na+, Ca2+, K+, Al3+, Zn2+, Mg2+, Fe3+, Cu2+, Ba2+), anions (CO32−, SO42−, PO43−, H2PO4−, HPO42−, Cl−, F−), small-molecule biological thiols (Cys, GSH and HCy), inorganic sulfur species such as thiosulfate (S2O32−), bisulfite (HSO3−), and sulfite (SO32−). As shown in Fig. 1e, compared to H2S, the fluorescence at 487 nm shows almost no change for the probe in the presence of the other substances. Furthermore, the presence of other substances has no obvious effect on the detection of H2S (Fig. 1f). The above results demonstrate that JM-NO2 possesses strong selectivity and anti-interference ability, confirming the probe's high specificity for H2S even in complex matrices.
2.2. Mechanism of JM-NO2 response to H2S
The reaction mechanism was elucidated by high-resolution mass spectrometry. Upon addition of NaHS, peaks corresponding to 2,4-dinitrobenzenethiol (m/z = 198.9812, negative ion mode) and coumarin 343 (m/z = 308.0891, positive ion mode) were observed (Fig. 2a, b and Fig. S8, S9). The initially weak fluorescence of JM-NO2 is due to photoinduced electron transfer (PET). Upon H2S-triggered cleavage of the ether bond, PET is disrupted, enabling a nucleophilic attack by the resulting phenol on the intramolecular ester, forming highly fluorescent, water-soluble coumarin 343 via acetone elimination and proton transfer (Fig. 2c), which emits bright green fluorescence.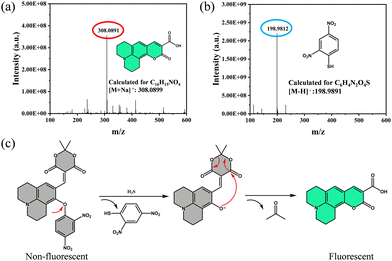 | ||
| Fig. 2 (a) HRMS spectrum of JM-NO2 after addition of H2S (positive ion mode). (b) HRMS spectrum (negative ion mode). (c) Schematic illustration of the recognition mechanism of JM-NO2 toward H2S. | ||
2.3. Development of a JM-NO2-doped CA film for H2S detection
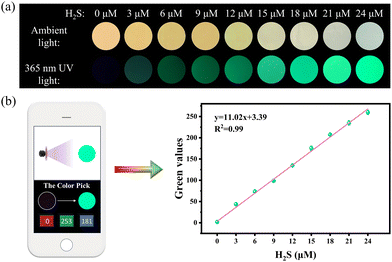
To facilitate practical application, JM-NO2 was embedded into a cellulose acetate (CA) film. Immersion in H2S-containing solution causes a visible color change (brown to pale yellowish green) under ambient light and a concentration-dependent increase in green fluorescence under 365 nm UV illumination (Fig. 3a). Fluorescence changes are perceptible to the naked eye above 3 μM H2S. For field analysis, fluorescence images are captured with a smartphone, and the green-channel (G-value) intensity is quantified via a color analysis app. A strong linear relationship (R2 = 0.99) is observed between G-value and H2S concentration (0–24 μM, Fig. 3b), highlighting the film's utility for rapid, visual, and quantitative detection.2.4. Synthesis and characterization of Ce-MOF
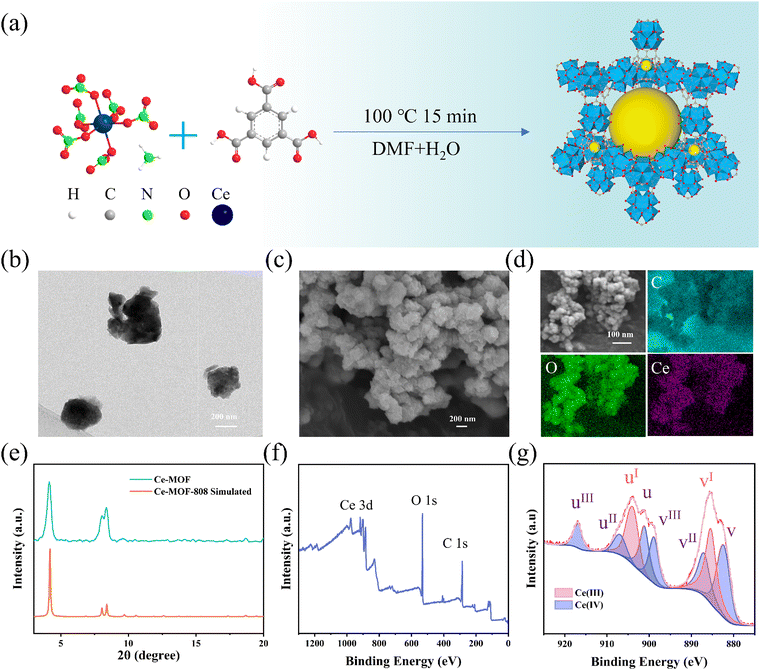
Cerium metal–organic frameworks (Ce-MOF) are considered promising candidates for developing novel biomimetic oxidase and peroxidase nanozymes due to the Ce(III)/Ce(IV) redox pair's properties, offering potential for H2S removal and disinfection to ensure quality of wastewater treatment and discharge. To enable catalytic disinfection and H2S scavenging, a Ce-MOF was synthesized using (NH4)2Ce(NO3)6 and 1,3,5-benzenetricarboxylic acid (Fig. 4a). Transmission electron microscopy (TEM) and scanning electron microscopy (SEM) images reveal that Ce-MOF exhibits predominantly spherical morphology with diameters ranging 300–400 nm (Fig. 4b and c). Dynamic light scattering (DLS) analysis indicates a slightly larger hydrodynamic diameter of 531 nm (Fig. S10). EDS mapping indicates that C, O, and Ce are uniformly distributed on the Ce-MOF surface (Fig. 4d). The structure of Ce-MOF was confirmed by X-ray diffraction (XRD) analysis, with the XRD pattern of Ce-MOF showing good agreement with the calculated Ce-MOF-808 curve (Fig. 4e). The survey spectrum of X-ray photoelectron spectroscopy (XPS) analysis confirms the presence of C, O, and Ce (Fig. 4f), further verifying successful Ce-MOF synthesis. High-resolution Ce 3d analysis of Ce-MOF (Fig. 4g) indicates mixed-valence Ce(III)/Ce(IV) states. The characteristic Ce(III) signature includes the 903.91 eV peak corresponding to the Ce 3d3/2 component (labeled as uI) and the 885.40 eV peak corresponding to the Ce 3d5/2 component (labeled as vI) (Table S2). While Ce(IV) exhibits six distinct subpeaks: 916.93, 906.95 and 901.15 eV belonging to the Ce 3d3/2 component (uIII, uII, and u), as well as 898.87, 887.04, and 882.37 eV corresponding to the Ce 3d5/2 component (vIII, vII, and v).57 Quantitative analysis (peak fitting and peak area integration calculations) shows Ce(IV) dominates the surface composition which was determined to be 63.64%. These results demonstrate the successful synthesis of mix-valence Ce-MOF.2.5. Ce-MOF's oxidase-like and peroxidase-like activities
The catalytic (nanozyme) activities of Ce-MOF were assessed using 3,3′,5,5′-tetramethylbenzidine (TMB) oxidation assays. Ce-MOF demonstrates both oxidase-like (OXD-like) and peroxidase-like (POD-like) activities. OXD-like activity converts O2 into ˙O2−, while POD-like activity decomposes H2O2 into ˙OH (Fig. 5a). The ˙O2− generated by OXD-like activity and the ˙OH generated by POD-like activity can catalyze the oxidation of 3,3′,5,5′-tetramethylbenzidine (TMB) into blue oxidized TMB (oxTMB) in the absence and presence of H2O2, respectively, which exhibits characteristic absorption at 652 nm. Therefore, colorimetric determination using TMB was employed to validate the Ce-MOF's OXD-like and POD-like activities. As shown in Fig. 5b, TMB + Ce-MOF and H2O2 + TMB + Ce-MOF exhibit distinct UV absorption peaks at 652 nm, whereas no corresponding absorption peaks can be observed in the control groups TMB + H2O2 and H2O2+ Ce-MOF. This indicates that Ce-MOF possesses OXD-like and POD-like activities. The effect of pH on Ce-MOF's OXD-like and POD-like activities was then investigated. Ce-MOF exhibits high OXD-like and POD-like activities over a broad pH range (Fig. S11a and b). Notably, while most reported nanozymes exhibit enhanced activity in acidic conditions, Ce-MOF maintains high activity across a broad pH range. Even at pH = 7.4, its OXD-like and POD-like activities remain exceptionally prominent, a characteristic that significantly outperforms many reported nanozymes (Tables S3 and S4). At pH = 7.4, as shown in Fig. 5c, d, f and g, Ce-MOF's steady-state kinetic parameters follow the classical Michaelis–Menten equation and Lineweaver–Burk model. The Michaelis constant (Km) represents the enzyme–substrate binding affinity, where lower values indicate stronger binding interactions. The maximum reaction rate (Vmax) corresponds to the enzyme's maximum catalytic efficiency (conversion efficiency at substrate saturation). For OXD-like activity (TMB substrate), we obtain Km = 0.041 mM and Vmax = 2.58 × 10−7 M s−1, respectively (as shown in Fig. 5c and d), indicating exceptionally strong substrate affinity and efficient catalytic conversion. Similarly, POD-like activity (H2O2 substrate) shows Km = 1.339 mM and Vmax = 2.48 × 10−7 M s−1, respectively (as shown in Fig. 5f and g). Notably, these kinetic parameters outperform most reported nanozymes (Tables S3 and S4), especially the Km value, which is reduced by an order of magnitude compared to most reported values. These values indicate high catalytic efficiency and substrate affinity of the Ce-MOF.To further validate the enzymatic mechanisms (Ce-MOF's POD-like and OXD-like activities), we employed electron paramagnetic resonance (EPR) spectroscopy with 5,5-dimethyl-1-pyrroline-N-oxide (DMPO) as a spin trap. DMPO specifically captures ˙O2−, forming the stable DMPO–OOH adduct that can be detected by EPR. As shown in Fig. 5e, the EPR spectrum exhibits a six-line signal composed of four main peaks and two minor peaks, with the main peak intensities being nearly 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1
1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 characteristic of ˙O2−. The control group lacks this signal, confirming OXD-like activity. When verifying POD-like activity, using DMPO as a spin trap (which captures ˙OH, forming DMPO–OH), we detect ˙OH generation through the characteristic 1
1 characteristic of ˙O2−. The control group lacks this signal, confirming OXD-like activity. When verifying POD-like activity, using DMPO as a spin trap (which captures ˙OH, forming DMPO–OH), we detect ˙OH generation through the characteristic 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2
2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 four-line signal observed in the EPR spectrum (Fig. 5h). The absence of this signal in control experiments confirms the ˙OH production originates specifically from the POD-like activity of Ce-MOF.
1 four-line signal observed in the EPR spectrum (Fig. 5h). The absence of this signal in control experiments confirms the ˙OH production originates specifically from the POD-like activity of Ce-MOF.
2.6. H2S scavenging by Ce-MOF monitored via JM-NO2
To verify that the ˙O2− generated by Ce-MOF's OXD-like can undergo redox reactions with H2S to remove H2S, we first investigated the H2S removal efficiency of Ce-MOF in aqueous solutions, using JM-NO2 as a fluorescent indicator of H2S level. As shown in Fig. 6a, when no H2S is present, adding Ce-MOF to the JM-NO2 system results in little change in fluorescence. However, when H2S is present, a concentration-dependent fluorescence quenching at 487 nm with increasing Ce-MOF amounts can be observed (Fig. 6b and c). When the Ce-MOF amount reaches 120 μg mL−1, the fluorescence intensity is nearly quenched, indicating that ˙O2− can efficiently reacts with H2S and effectively removes H2S. The final oxidation products of H2S were detected using ion chromatography (IC), as shown in Fig. 6d. Compared with the sulfate standard solution, the reaction system exhibits a peak at 5.0 min, indicating that the ˙O2− generated by Ce-MOF's OXD-like activity react with H2S and ultimately form SO42−, resulting in the removal of H2S. Additionally, the peak at 7.5 minutes corresponds to phosphates in the PB, and the peak at 3.4 minutes may be an intermediate product of H2S oxidation.582.7. Ce-MOF's antibacterial properties
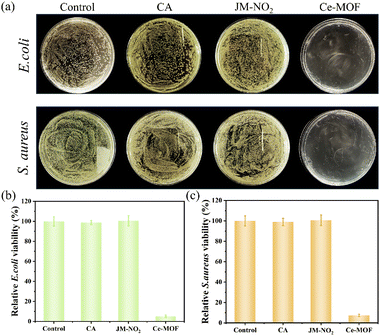
Ce-MOF's nanozyme activities of can generate ROS to achieve efficient bactericidal effects. However, POD-like activity requires the addition of exogenous hydrogen peroxide as a catalytic substrate, which greatly limits its practical application. Therefore, our subsequent studies utilized the Ce-MOF's OXD-like activity to eliminate bacteria in wastewater by the ˙O2− generated by the MOF. We evaluated the antimicrobial efficacy of Ce-MOF against representative Gram-negative (Escherichia coli, E. coli) and Gram-positive (Staphylococcus aureus, S. aureus) strains using standard plate count assays. As demonstrated in Fig. 7a, neither the cellulose acetate (CA) film nor the JM-NO2 probe alone exhibits significant antibacterial effects compared to untreated controls. In striking contrast, Ce-MOF treatment significantly reduces colonies counts of both bacteria strains. Quantitative results of the plate counts are presented in Fig. 7b (E. coli) and Fig. 7c (S. aureus), respectively. Ce-MOF (at 1 mg mL−1) achieves inactivation rates of 94.6% and 92.7% for E. coli and S. aureus, respectively. Thus, due to its OXD-like activity, Ce-MOF nanozyme can serve as potent and broad-spectrum bactericides.2.8. Application of the dual-film system in slaughterhouse and meat-processing wastewater treatment
The last step in wastewater treatment is disinfection. This process involves killing or removing harmful microorganisms to ensure the water is safe for reuse or discharge. Hence, we prepared the dual-film system for the last-step treatment of slaughterhouse and meat-processing wastewater, so as to achieve simultaneous H2S scavenging, bacterial disinfection, and real-time, visual monitoring of H2S levels before discharge. The schematic illustration of the dual-film platform for final- stage treatment of slaughterhouse wastewater is shown in Fig. 8a. Film 1 (the Ce-MOF-loaded CA film) and film 2 (JM-NO2-loaded CA film) are mounted on the filtering cylinder, In the last-step treatment, the slaughter wastewater was filtered through film 1 and film 2 sequentially. Film 1 removes bacteria and H2S, while film 2 enables visual monitoring of residual H2S. As shown in Fig. 8a, water passes sequentially through film 1 (with varying Ce-MOF contents) and film 2. As shown in Fig. 8b, when water passes through film 1 containing different amounts of Ce-MOF (0–10 wt%) and then film 2, the color of film 2 clearly changes from pale green to brown under ambient light. While under 365 nm UV light irradiation, the green fluorescence intensity of film 2 gradually decreases, as the Ce-MOF content in film 1 increases. When the Ce-MOF content in film 1 reaches 10 wt%, the green fluorescence intensity of film 1 becomes almost invisible, indicating that the H2S content in the treated water is extremely low. With 2.5 wt% of Ce-MOF in film 1, film 1 can achieve 99.7% bactericidal rate (Fig. 8c and e). This indicates that Ce-MOF has excellent bactericidal properties. Ce-MOF loading of 10 wt% on the cellulose acetate membrane simultaneously achieves high efficiency in both H2S removal and a bactericidal rate of 99.8%. As shown in Fig. 8d, we used a smartphone to capture fluorescent color images of film 2 and digitized the images using a color scanning application. By analyzing the green-channel intensity (G-value) of the images, we could accurately reflect the extent of H2S removal at different Ce-MOF concentrations in film 1. The stability of the films was evaluated under ambient storage conditions. For the JM-NO2 films, stability was assessed by periodically testing their H2S detection performance. Each film exhibits bright and comparable fluorescence after reaction with H2S (quantified by G-value analysis in Fig. S13), confirming excellent stability for H2S sensing. For the Ce-MOF films, a dual-film testing approach was employed: NaHS solution (as H2S donor) was first passed through the Ce-MOF film, and the residual H2S was quantified downstream using a JM-NO2 film. The consistently weak fluorescence of the JM-NO2 films (Fig. S14) indicate stable and effective H2S removal by the Ce-MOF films over time. These experimental results clearly demonstrate that this dual-film system provides effective bacterial disinfection, efficient H2S removal, and real-time, visual feedback on effluent quality. It offers significant potential for deployment in industrial, municipal, and domestic wastewater treatment scenarios.3. Conclusions
In summary, we have developed a cellulose acetate-based dual-film platform that simultaneously achieves bacterial disinfection, H2S scavenging, and real-time fluorescent monitoring of H2S in aqueous environments. The Ce-MOF-loaded film (film 1) exhibits potent peroxidase- and oxidase-like catalytic properties, enabling efficient ROS generation for broad-spectrum antibacterial action and oxidative degradation of H2S. The JM-NO2-doped film (film 2) provides sensitive and selective turn-on fluorescence and visible color changes in response to residual H2S, allowing intuitive on-site monitoring. This integrated system demonstrates excellent performance under environmentally relevant conditions, with robust antibacterial efficacy and H2S detection in complex media such as slaughterhouse wastewater. The modularity, scalability, and dual functionality of the platform make it a promising candidate for practical application in wastewater treatment and environmental monitoring, particularly in the final disinfection and decontamination stages before effluent discharge.Author contributions
Xiangshan Lei: conceptualization, data curation, formal analysis, methodology, writing – original draft. Fang Zeng: conceptualization, formal analysis, supervision, funding acquisition, writing – reviewing and editing. Shuizhu Wu: conceptualization, resources, formal analysis, funding acquisition, supervision, writing – reviewing and editing.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Additional experimental procedures, scheme for synthetic route, 1H/13C NMR and HR mass spectra, time-dependent fluorescent intensity, absorption spectra, size distribution, and the comparison table. See DOI: https://doi.org/10.1039/d5tc03098f.Acknowledgements
This work was supported by NSFC (52373209 and 22274057), the Fund of Guangdong Provincial Key Laboratory of Luminescence from Molecular Aggregates (2023B1212060003).Notes and references
- U. Javourez, L. Tiruta-Barna, M. Pizzol and L. Hamelin, Nat. Sustainability, 2025, 8, 385–395 CrossRef.
- J. Sun, Y. Feng, R. Zheng, Y. Mao, Q. Zhu, X. Wu, L. Kong, K. Zhang and S. Liu, Nat. Sustainability, 2025, 8, 1048–1057 CrossRef.
- M. Priyadarshini, I. Das, M. M. Ghangrekar and L. Blaney, J. Environ. Manage., 2022, 316, 115295 CrossRef CAS PubMed.
- S. Wang, Z. Wei and L. Wang, Renewable Sustainable Energy Rev., 2024, 189, 113881 CrossRef CAS.
- K. J. Haselroth, P. Wilke, I. M. Dalla Costa, V. M. L. Rotta, A. F. Rosado and E. Hermes, J. Cleaner Prod., 2021, 287, 125533 CrossRef CAS.
- B. Hao, Y. Ahmadi, J. Szulejko, T. Zhang, Z. Lu and K. H. Kim, Chin. J. Catal., 2025, 68, 282–299 CrossRef.
- L. Yan, J. Ye, P. Zhang, D. Xu, Y. Wu, J. Liu, H. Zhang, W. Fang, B. Wang and G. Zeng, Bioresour. Technol., 2018, 259, 67–74 CrossRef CAS PubMed.
- Z. Chen, Y. Chen, X. Hao, Q. Wen, X. Wang, A. Liu, D. Gao, K. Hu, N. A. Samak, M. Yang and J. Xing, Chem. Eng. J., 2025, 508, 160632 CrossRef CAS.
- G. Prabakaran and H. Xiong, Food Chem., 2024, 464, 141809 CrossRef PubMed.
- Y. Li, Y. He, S. Dai, H. Guo, J. Hou, P. Zhang, Y. Wang, T. Zhu, B. Ni and Y. Liu, Chem. Eng. J., 2025, 506, 160227 CrossRef CAS.
- L. Chen, R. Delatolla, P. M. D’Aoust, R. Wang, F. Pick, A. Poulain and C. D. Rennie, Environ. Technol., 2019, 40, 642–653 CrossRef CAS PubMed.
- E. Ntakiyisumba, S. Lee and G. Won, Food Res. Int., 2023, 170, 112999 CrossRef PubMed.
- H. Yin, H. Zhao, L. Sun, J. Jia, Z. Zhang and Y. Zhang, Process Saf. Environ., 2024, 191, 1784–1792 CrossRef CAS.
- K. Wang, C. Bi, L. Zelenkov, X. Liu, M. Song, W. Wang, S. Makarov and W. Yin, ACS Sens., 2024, 9, 5708–5727 CrossRef CAS PubMed.
- Y. Xu, Z. Yin, Q. Long, H. Xie, X. Yan, F. Liao, H. Cui and H. Fan, Food Chem., 2025, 484, 144373 CrossRef CAS PubMed.
- J. Yang, S. Cui, F. Zhao, F. Wang, J. Feng, P. Ning and L. Jia, Environ. Sci. Technol., 2024, 58, 20267–20276 CrossRef CAS PubMed.
- J. Bao, X. Sun, P. Ning, K. Li, J. Yang, F. Wang, L. Shi and M. Fan, Green Energy Environ., 2025, 10, 34–83 CrossRef CAS.
- M. Yin, S. C. Pillai, N. Bolan, H. Wang, H. Yin, Z. Wu, Y. Zheng, L. Zhao, F. Fan and H. Hou, Chem. Eng. J., 2024, 479, 147854 CrossRef CAS.
- J. Ouyang, L. Sun, Z. She, R. Li, F. Zeng, Z. Yao and S. Wu, ACS Appl. Mater. Interfaces, 2024, 16, 20315–20328 CAS.
- R. Li, F. Zeng and S. Wu, Anal. Chem., 2025, 97, 13681–13690 CrossRef CAS PubMed.
- Z. She, R. Li, F. Zeng and S. Wu, Adv. Healthcare Mater., 2024, 13, 2400791 CrossRef CAS PubMed.
- Y. Zhang, F. Zeng and S. Wu, J. Mater. Chem. B, 2025 10.1039/D5TB01208B.
- C. Zeng, Y. Tan, L. Sun, Y. Long, F. Zeng and S. Wu, ACS Appl. Mater. Interfaces, 2023, 15, 17664 CrossRef CAS PubMed.
- Q. Zong, J. Li, Q. Xu, Y. Liu, K. Wang and Y. Yuan, Nat. Commun., 2024, 15, 7558 CrossRef CAS PubMed.
- K. G. Fosnacht and M. D. Pluth, Chem. Rev., 2024, 124, 4124–4257 CrossRef CAS PubMed.
- N. Y. Kim and M. H. Lee, Sens. Actuators, B, 2025, 422, 136567 CrossRef CAS.
- J. Chen, M. Wang, X. Yang, M. Yuan, H. Liu, P. Cheng and K. Xu, Sens. Actuators, B, 2024, 405, 135313 CrossRef CAS.
- X. Yang, S. Zhang, F. Luo, Y. Zhang, D. Yan, M. Lai, Y. Ye, K. Sun and X. Ji, Biosens. Bioelectron., 2025, 267, 116798 CrossRef CAS PubMed.
- J. Cao, Z. Liu, Y. Zhang, X. Su, C. Yang and H. Yan, Carbohydr. Polym., 2025, 353, 123275 CrossRef CAS PubMed.
- R. Huang, T. Liu, H. Peng, J. Liu, X. Liu, L. Ding and Y. Fang, Chem. Soc. Rev., 2024, 53, 6960 RSC.
- M. Chen, R. Chu, M. Babazadeh, J. C. M. Kistemaker, P. L. Burn, I. R. Gentle and P. E. Shaw, Adv. Funct. Mater., 2024, 34, 2311328 CrossRef CAS.
- L. Zhang, K. Song, G. Liu, X. Li, Y. He, Y. Meng, Y. Yang, T. Gong, H. Wang, K. Sun, L. Zeng, S. Yan and H. Ma, Coord. Chem. Rev., 2025, 540, 216766 CrossRef CAS.
- X. Yuan, N. Qu, M. Xu, L. Liu, Y. Lin, L. Xie, X. Chai, K. Xu, G. Du and L. Zhang, Int. J. Biol. Macromol., 2024, 265, 131111 CrossRef CAS PubMed.
- G. Wu, S. Li, L. Luo, Y. Li, W. Zhang, H. Wang, S. Liu, C. Du, J. Wang, J. Cheng, Y. Wu and Y. Shen, Nano-Micro Lett., 2025, 17, 1–33 CrossRef PubMed.
- X. Liao, X. Wang, B. Li, L. Wang and Y. Chen, Coord. Chem. Rev., 2025, 541, 216807 CrossRef CAS.
- J. Li, X. Cai, P. Jiang, H. Wang, S. Zhang, T. Sun, C. Chen and K. Fan, Adv. Mater., 2024, 36, 2307337 CrossRef CAS PubMed.
- J. Zhou, D. Xiong, H. Zhang, J. Xiao, R. Huang, Z. Qiao, Z. Yang and Z. Zhang, Environ. Sci. Technol., 2025, 59, 1844–1853 CrossRef CAS PubMed.
- Y. Bai, S. Nie, W. Gao, N. Li, P. Zhu, L. Zhang and J. Yu, Adv. Funct. Mater., 2025, 35, 2419499 CrossRef CAS.
- X. Liao, B. Li, L. Wang and Y. Chen, Biosens. Bioelectron., 2024, 264, 116637 CrossRef CAS PubMed.
- Y. Wang, Y. Yang, J. Liu, X. Zi, H. Zhu, X. Sun, Y. Miao and Y. Fu, Chem. Eng. J., 2024, 500, 156850 CrossRef CAS.
- M. Wang, H. Zhang, S. Yan, Y. Zhou, X. Guo, D. An, W. Zhong and Y. Zhang, Coord. Chem. Rev., 2025, 532, 216506 CrossRef CAS.
- Y. Cai, Y. Wu, Y. Tang, W. Xu, Y. Chen, R. Su, Y. Fan, W. Jiang, Y. Wen, W. Gu, H. Sun and C. Zhu, Small, 2024, 20, 2403354 CrossRef CAS PubMed.
- J. Liu, X. Yu, Y. Zhou, L. Sun, Y. Liu and J. Li, Biosens. Bioelectron., 2025, 280, 117441 CrossRef CAS PubMed.
- X. Wang, K. Jiao, Y. Huang and Z. Wang, J. Colloid Interface Sci., 2025, 695, 137784 CrossRef CAS PubMed.
- Y. Xu, A. Huang, W. Yi, G. Chen, S. Huang and G. Ouyang, Coord. Chem. Rev., 2024, 500, 215517 CrossRef CAS.
- H. Miao, T. Ye, M. Liu, J. Gao, L. Tan, Q. Dong, Q. Lv, Q. Ye and Y. Wang, Adv. Funct. Mater., 2025, 2508310 CrossRef.
- J. Qin, J. Zhou, J. Ma, S. Li, A. Zhou, L. Xie, Y. Dou and Y. Zhang, Angew. Chem., Int. Ed., 2025, 137, e202505786 CrossRef.
- Q. Shi, Y. Zhao, M. Liu, F. Shi, L. Chen, X. Xu, J. Gao, H. Zhao, F. Lu, Y. Qin, Z. Zhang and M. Lian, Small, 2024, 20, 2309366 CrossRef CAS PubMed.
- S. Sun, X. Liu, X. Meng, Z. Yang, X. Zhang and H. Dong, ACS Nano, 2025, 19, 15109–15119 CrossRef CAS PubMed.
- C. Yu, Q. Lu, Y. Wang, Z. Liu, S. Gnanasekar, U. D’Amora, E. Kang, L. Xu, J. Xu and X. Rao, ACS Appl. Mater. Interfaces, 2025, 17, 31993–32012 CrossRef CAS PubMed.
- Y. Gong, W. Qi, W. Lu, Q. Chang, Y. Xie, J. Wang and X. Deng, J. Colloid Interface Sci., 2025, 689, 137247 CrossRef CAS PubMed.
- H. Xiu, K. Yang, L. Dong, H. Lai, Z. Zhu, D. Jiang, J. Yan, C. Shi, S. Pan, Z. Yin, L. Yuwen and B. Liang, Adv. Healthcare Mater., 2025, 14, 2403205 CrossRef CAS PubMed.
- J. Zou, W. Ding, Y. Chen, X. Pan, X. Fu, S. Zheng, Y. Lin, C. Xia, Y. Ma, X. Zhang, S. Wu and F. Gao, Carbohydr. Polym., 2025, 361, 123501 CrossRef CAS PubMed.
- Y. Li, W. Chen, Y. Qi, S. Wang, L. Li, W. Li, T. Xie, H. Zhu, Z. Tang and M. Zhou, Small, 2020, 16, 2001356 CrossRef CAS PubMed.
- Y. Ma, Y. Tu, Y. Chen, X. Chen, X. Pan, M. Sun, X. Fu, J. Zou and F. Gao, ACS Appl. Mater. Interfaces, 2024, 17, 617–631 CrossRef PubMed.
- K. Zhong, X. Hu, S. Zhou, X. Liu, X. Gao, L. Tang and X. Yan, J. Agric. Food Chem., 2021, 69, 4628–4634 CrossRef CAS PubMed.
- H. Li, J. Zhang, X. Deng, Y. Wang, G. Meng, R. Liu, J. Huang, M. Tu, C. Xu, Y. Peng, B. Wang and Y. Hou, Angew. Chem., Int. Ed., 2024, 63, e202316384 CrossRef CAS PubMed.
- G. H. Kelsall and I. Thompson, J. Appl. Electrochem., 1993, 23, 287–295 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |