Star block copolymer POEGMA-b-P3HT enables tunable charge transport in organic field-effect transistors
Pallavi
Kumari
 *a,
Barbara
Hajduk
*a,
Barbara
Hajduk
 a,
Anna
Celny
a,
Anna
Celny
 a,
Agnieszka
Kowalczuk
a,
Agnieszka
Kowalczuk
 a,
Sonia
Kotowicz
a,
Sonia
Kotowicz
 b,
Pawel
Jarka
b,
Pawel
Jarka
 c,
Małgorzata
Łazarska
c,
Małgorzata
Łazarska
 d,
Marcin
Godzierz
d,
Marcin
Godzierz
 a,
Marta
Musioł
a,
Marta
Musioł
 a and
Anastasiia
Kobyliukh
a and
Anastasiia
Kobyliukh
 a
a
aCentre of Polymer and Carbon Materials, Polish Academy of Sciences, 34 Marie Curie-Skłodowska Str., 41-819 Zabrze, Poland. E-mail: pkumari@cmpw-pan.pl
bInstitute of Chemistry, University of Silesia, 9 Szkolna Str., 40 006 Katowice, Poland
cDepartment of Engineering Materials and Biomaterials, Silesian University of Technology, 18a Konarskiego Str., 41-100 Gliwice, Poland
dFaculty of Materials Engineering, Kazimierz Wielki University, Chodkiewicza 30, 85-064 Bydgoszcz, Poland
First published on 7th October 2025
Abstract
Conjugated polymers are widely recognized for their potential in advanced optoelectronic applications due to their tunable electrical and optical properties. Integrating a conjugated polymer with a star-shaped, non-conducting polymer matrix introduces unique physical and optoelectronic characteristics driven by architectural control. Based on this concept, we synthesized and explored star-shaped conjugated block copolymers composed of four-arm poly(oligo (ethylene glycol) methacrylate) (POEGMA) and linear poly(3-hexylthiophene) (P3HT). The star-shaped POEGMA cores were synthesized via atom transfer radical polymerization using a tetrafunctional initiator, pentaerythritol tetrakis(2-bromoisobutyrate). Separately, the alkyne-terminated P3HT was prepared through Kumada catalyst-transfer polymerization. The two polymer segments were then conjugated via copper(I)-catalyzed azide–alkyne cycloaddition, affording well-defined POEGMA-b-P3HT block copolymers with star-like architecture. Comprehensive structural characterization was conducted using nuclear magnetic resonance spectroscopy, gel permeation chromatography, and Fourier-transform infrared spectroscopy. The optoelectronic properties and surface morphology of the resulting materials were systematically investigated in the solid state. The star block copolymer POEGMA-b-P3HT exhibited isotropic behavior in thin films, along with a well-defined spike-like morphology. The resulted optical behavior and morphology is associated with ambipolar transistor characteristics, indicating that the P3HT segment retained its properties effectively within the star-shaped architecture. These findings demonstrate that the star-shaped molecular design significantly influences the optical properties, underscoring its potential for application in ambipolar organic field-effect transistors.
1. Introduction
Organic field-effect transistors (OFETs) have emerged as promising components in flexible, lightweight, and low-cost electronic devices, including sensors, displays, and wearable electronics.1–3 Conjugated polymers (CPs), in particular, are widely used as the semiconducting active layers in OFETs due to their excellent optoelectronic properties, mechanical flexibility, and easy of solution processing.4,5 To further tune the OFET performance, significant research has focused on designing and synthesizing novel CP-based systems with advanced molecular architectures and tunable functionalities.6,7 A crucial factor influencing OFET performance is the nanoscale morphology of the semiconducting layer.8 The incorporation of block copolymers (BCPs) is a promising strategy to achieve such defined nanostructures and to enhance the overall functionality of CPs.9,10 BCPs are macromolecules consisting of two or more chemically distinct polymer segments covalently linked together.11 Their intrinsic ability to self-assemble into well-defined nanoscale morphologies makes BCPs, both solution and solid states, attractive for advanced electronic applications.9,12 When BCPs incorporate flexible amorphous blocks with rigid semicrystalline segments, they can form robust semicrystalline nanostructures with improved mechanical strength and structural integrity.13 This synergy of distinct structural domains provides a versatile platform for tuning both functional and mechanical properties, enabling the development of materials with tailored performance across multiple length scales, particularly beneficial for applications in stretchable organic electronics.14,15Among BCP architectures, star-shaped BCPs represent an advanced subclass consisting of multiple linear di-block copolymer arms radiating outward from a central core, where all the chains are tethered to a single junction point.16 The compact architecture with high segment density influences material properties like enhanced solubility, improved mechanical strength, and greater processibility compared to their linear counterparts.17 These advantages tune star-shaped BCPs performance across diverse applications such as nanotechnology, optoelectronics, and biomedical engineering.18,19 Furthermore, the development of star-shaped conjugated BCP using CP with combining star-shaped non-conjugated polymers20 has led to create hybrid systems that combine the electronic properties of CP with the structural and processing advantages of star-shaped non-conjugated polymer.21
Within the broad spectrum of CPs investigated for optoelectronic applications, regioregular poly(3-hexylthiophene) (P3HT), CP, have been extensively studied owing to its superior solubility, excellent chemical stability, film forming ability and favourable charge transport characteristics.22,23 Numerous studies have focused on the design and synthesis of P3HT-based linear BCPs, including P3HT-b-PS,24 P3HT-b-PEG,25 P3HT-b-PNIPAM,26 P3HT-b-PMMA27 and more to further tailor its physical and electrical properties for advanced functional applications. These studies have demonstrated that, relative to pristine P3HT, the resulting BCPs exhibit superior physical attributes and improved electrical performance. This improvement primarily due to changes in molecular orientation and nanoscale morphology. In recent studies on the transistor performance of P3HT-based BCPs have shown that these materials demonstrate promising characteristics, including high charge carrier mobility, excellent on/off current ratios, and improved stability while retaining the intrinsic p-type semiconducting behaviour of P3HT14,15,28–30 (Table 1). Building on this strategy, some studies have also explored the synthesis of P3HT-based star-shaped BCPs with tunable arm numbers and molecular weights, employing click chemistry in combination with living polymerization techniques to construct well-defined macromolecular architectures.20,31,32 However, despite these synthetic advancements, the comprehensive studies on the optoelectronic properties and transistor characteristics of star-shaped P3HT-based BCP remains notably limited.
| Devices | Hole mobility (μh) | I on/off | Drain voltage | Type | Ref. |
|---|---|---|---|---|---|
| P3HT-b-PBA | 6.1 × 10−2 | ∼106 | NA | p | Wang, et al.14 |
| P3HT-b-PDL | 1.5 × 10−3 | 1.4 × 103 | NA | p | Hsu, et al.15 |
| PDL3-b-P3HT-b-PDL3 | 1.4 × 10−3 | 5.5 × 103 | NA | p | Hsu, et al.15 |
| PCL-b-P3HT-b-PCL | 4.6 × 10−4 | ∼2 × 104 | −60 V | p | Surin, et al.28 |
| PCL-b-P3HT | 1.1 × 10−5 | ∼1 × 103 | −60 V | p | Surin, et al.28 |
| PS-b-P3HT | 8.3 × 10−2 | 105–106 | −50 V | p | Yu, et al.29 |
| P3HT-b-PMMA | 1.5 × 10−2 | NA | NA | p | Lee, et al.30 |
| Star POEGMA-b-P3HT (non-annealed) | 1.6 × 10−3 (μh) | 0.7× 102 | –10 V | Ambipolar | This work |
| 0.25 × 10−3 (μe) | 1.1 × 101 | 10 V | |||
| (annealed) | 1.7 × 10−3 (μh) | 1.6 × 102 | −10 V | Ambipolar | This work |
| 3.09 × 10−3 (μe) (μe electron mobility) | ∼10 | 10 V |
To address this, a novel P3HT-based 4-arm star-shaped conjugated BCP, poly(oligo (ethylene glycol) methacrylate (POEGMA)-b-P3HT, was synthesized by coupling star-shaped POEGMA with alkyne functionalized linear P3HT via click chemistry. The star-shaped POEGMA was prepared using atom transfer radical polymerization (ATRP), while the linear P3HT segment was synthesized through the Kumada catalyst-transfer polymerization (KCTP) method. POEGMA was strategically selected for its intrinsic water solubility and biocompatibility, arising from its short ethylene glycol side chains, which facilitate solution processing and make it a promising material for bioelectronics and flexible, stretchable electronic devices.33–35 Following synthesis, both the homopolymers and the resulting BCP were subjected to comprehensive structural characterization using advanced spectroscopic and chromatographic techniques. The optical properties, morphology, crystallinity, and OFET performance of the star-shaped BCP were systematically investigated to understand the impact star architecture on physical and electrical behaviour, particularly in comparison to linear P3HT. These results demonstrate the functional advantages of the star-shaped architecture for tuning electrical performance in future optoelectronic applications in biomedical field.
2. Materials and methods
2.1. Materials
1,2-Dichlorobenzene (99%), 1,1,4,7,10,10-hexamethyltriethylenetetramine (HMTETA, 97%), copper(I) bromide (CuBr, 99.999%), copper(II) bromide (CuBr2, 99%), p-xylene (≥99%), and pentaerythritol tetrakis(2-bromoisobutyrate) (4F-BiB, 97%) tetrabutylammonium hexafluorophosphate (Bu4NPF6, ≥99.0%) were purchased from Sigma-Aldrich and used without further purification. Hydroxyl-terminated oligo (ethylene glycol) methacrylate (OEGMA-OH, Mn ≈ 360 g mol−1) was also obtained from Sigma-Aldrich and purified according to a previously reported procedure. Dowex Marathon MSC ion-exchange resin (Sigma-Aldrich) was converted to the H+ form using 1.6 M HNO3. High-purity water (H2O, 99.9%) and acetonitrile (ACN, 99.9%) for HPLC super-gradient applications were purchased from POCH and used as received. Tetrahydrofuran (THF, pure p.a.) was obtained from POCH and distilled prior to use. Deuterated chloroform (CDCl3, 99.8%) and tetramethylsilane (TMS, 99.9%) were purchased from Chemat and used as received. Anhydrous solvents, including toluene and THF, along with 2-bromo-5-iodo-3-hexylthiophene, isopropylmagnesium chloride (2.0 M in THF), ethynylmagnesium bromide (0.5 M in THF), [1,3-bis(diphenylphosphino)propane]dichloronickel(II), copper bromide, and N,N,N′,N′′,N′′-pentamethyldiethylenetriamine (PMDETA), and acetonitrile were purchased from Merck (Poland) and used without further purification.2.2. Synthesis of azide modified star poly-oligo (ethylene glycol) methacrylate (star POEGMA-N3)
Star polymers of OEGMA-OH with 4F-BiB as a core were synthesized according to the ATRP method and via core-first technique.36,37 For this purpose, 20 mg (2.73 × 10−5 mol) 4F-BiB (initiator, core), 8 mg (5.58 × 10−5 mol) CuBr and 0.6 mg (2.69 × 10−6 mol) CuBr2 were dissolved in 1,2-dichlorobenzene (monomer to solvent ratio was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 v/v) in a Schlenk flask under nitrogen with a magnetic stirrer and degassed using freeze–pump–thaw cycles. Next, 30 μL (1.10 × 10−4 mol) HMTETA and 3.6 mL OEGMA-OH (1.11 × 10−2 mol) were added to the solution, and the mixture was degassed. The ratio of 4F-BiB
3 v/v) in a Schlenk flask under nitrogen with a magnetic stirrer and degassed using freeze–pump–thaw cycles. Next, 30 μL (1.10 × 10−4 mol) HMTETA and 3.6 mL OEGMA-OH (1.11 × 10−2 mol) were added to the solution, and the mixture was degassed. The ratio of 4F-BiB![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CuBr
CuBr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) CuBr2
CuBr2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) HMTETA was 1
HMTETA was 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.5
0.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 0.025
0.025![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1. The mixture in the flask was placed in an oil bath at 30 °C and left on a magnetic stirrer. The synthesis was stopped when the appropriate conversion was reached (approximately 40% for all samples). Next, the mixture was diluted with acetone (5 mL) and passed through a DOWEX-MSC-1 ion exchange resin column to remove all the copper. The resulting solution was dialyzed in acetone (Spectra Por membrane with MWCO 1000 g mol−1) and dried under vacuum resulting a 1.58 g yield. The obtained star POEGMA-Br was analyzed through 1H NMR spectroscopy (Fig. S1).
1. The mixture in the flask was placed in an oil bath at 30 °C and left on a magnetic stirrer. The synthesis was stopped when the appropriate conversion was reached (approximately 40% for all samples). Next, the mixture was diluted with acetone (5 mL) and passed through a DOWEX-MSC-1 ion exchange resin column to remove all the copper. The resulting solution was dialyzed in acetone (Spectra Por membrane with MWCO 1000 g mol−1) and dried under vacuum resulting a 1.58 g yield. The obtained star POEGMA-Br was analyzed through 1H NMR spectroscopy (Fig. S1).
Further for azide end group modification, we followed the procedure described by Parrillo, et al.,38 20 mL solution of the star POEGMA-Br in acetone (1% w/w) was prepared. The solution was transferred to a round-bottom flask and heated in an oil bath at 60 °C under reflux. Sodium azide (30 mg) was added in a 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio with respect to the repeating units of the polymer. The reaction mixture was reflux for three days and subsequently allowed to cool to room temperature, followed by filteration using a 0.2 μm syringe filter to remove undissolved sodium azide. The resulting clear solution was used directly for FT-IR analysis. The final product was dried, yielding 1.5 g.
1 molar ratio with respect to the repeating units of the polymer. The reaction mixture was reflux for three days and subsequently allowed to cool to room temperature, followed by filteration using a 0.2 μm syringe filter to remove undissolved sodium azide. The resulting clear solution was used directly for FT-IR analysis. The final product was dried, yielding 1.5 g.
2.3. Synthesis of alkyne functionalized regioregular poly-3-hexyl thiophene (linear P3HT)
Alkyne functionalized P3HT was prepared following the methods described by Kumari, et al.26 A three-neck round-bottom flask was charged with 2-bromo-5-iodo-3-hexylthiophene (1 g, 2.68 mmol) under an argon atmosphere and evacuated under reduced pressure to remove residual moisture and oxygen. Anhydrous THF (20 mL) was added via syringe, and the resulting solution was stirred and cooled to 0 °C. A solution of isopropylmagnesium chloride in THF (1.34 mL, 2.68 mmol) was then added dropwise, and the mixture was stirred at 0 °C for 2 h. The reaction mixture was then diluted to 15 mL with THF, followed by the addition of Ni(dppp)Cl2 (32 mg, 0.06 mmol). The solution was heated to 35 °C and stirred for 30 min and then the reaction mixture was cooled at 0 °C. Subsequently, a 0.5 M solution of ethynyl magnesium bromide (2.68 mL, 1.34 mmol) was added, and the reaction was stirred for an additional 10 min at room temperature. The reaction was then quenched with methanol, yielding a dark-purple solid. The solid was filtered and washed thoroughly with excess methanol. To remove oligomers and low-molecular-weight fractions, the crude product was reprecipitated in hexane, filtered, and washed repeatedly until the filtrate ran clear. The resulting alkyne functionalized linear P3HT polymer was dried under vacuum, yielding 0.58 g (58%) of polymer material.2.4. Synthesis of star POEGMA-b-P3HT
The 4-arm star BCP, POEGMA-b-P3HT, was prepared via the click reaction of azide-terminated star-shaped POEGMA with an alkynyl-functionalized linear P3HT.31,39 In typical preparation, 4-arm azide-terminated star POEGMA, alkynyl-functionalized linear P3HT, CuBr, and PMDETA were used in the ratio of (1.2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1:10
1:10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10). The 4-arm star POEGMA (2.22 μmol) and linear P3HT (1.85 μmol) were added in THF (20.0 mL) and successively transfer into a 50-mL flask after that the CuBr (18.5 μmol) and PMDETA (18.5 μmol) were added. The reaction mixture was degassed by three pump-thaw cycles, back-filled with nitrogen, and maintained in an oil bath at 60 °C for 72 h. Upon completion, the mixture was diluted with THF and passed through a neutral alumina column to remove residual copper salts. The product was then precipitated into excess methanol, where unreacted star-shaped POEGMA dissolved and was removed. This precipitation and purification step was repeated three times to ensure complete removal of the unreacted star POEGMA. The solvent was subsequently evaporated, and the resulting material was dried overnight in a vacuum oven at room temperature, resulting a dark orange solid with an 85% (95 mg) yield.
10). The 4-arm star POEGMA (2.22 μmol) and linear P3HT (1.85 μmol) were added in THF (20.0 mL) and successively transfer into a 50-mL flask after that the CuBr (18.5 μmol) and PMDETA (18.5 μmol) were added. The reaction mixture was degassed by three pump-thaw cycles, back-filled with nitrogen, and maintained in an oil bath at 60 °C for 72 h. Upon completion, the mixture was diluted with THF and passed through a neutral alumina column to remove residual copper salts. The product was then precipitated into excess methanol, where unreacted star-shaped POEGMA dissolved and was removed. This precipitation and purification step was repeated three times to ensure complete removal of the unreacted star POEGMA. The solvent was subsequently evaporated, and the resulting material was dried overnight in a vacuum oven at room temperature, resulting a dark orange solid with an 85% (95 mg) yield.
2.5. Characterization methods
Based on the shadow patterns obtained from individual segments, a heightmap was generated, representing the surface topology. In the next step, roughness measurements were carried out using the 3D Roughness Reconstruction application. The procedure involved defining measurement lines on the reconstructed heightmap, followed by the automatic calculation of topographic profiles for each of them. The obtained profiles enabled a quantitative characterization of roughness parameters.
| HOMO = −(Eox + 4.8) eV |
| LUMO = −(Ered + 4.8) eV |
2.6. OFET characterization
The characteristics of OFET were evaluated using a bottom-gate and bottom-contact geometry with prefabricated high-density platinum OFET chips obtained from Ossila. These chips consisted of 20 devices, each with a constant channel width of 2.5 mm and variable channel lengths ranging from 2 to 10 μm. The prefabricated substrate consists of highly doped p-type Si with a 300 nm silicon oxide (SiO2) gate dielectric layer grown on both sides, serving as insulation. The source and drain contacts were fabricated with a 5 nm titanium adhesion layer beneath a 100 nm platinum layer. Additionally, a platinum gate contact was deposited along one edge of each substrate. Solutions of Linear P3HT, and Star POEGMA-b-P3HT were prepared at a concentration of 5 mg mL−1 in chloroform. These solutions were gently deposited on the substrate using the drop-casting method at room temperature. The output (IDSversus VDS) and transfer characteristics (IDSversus VG) of the devices, with a channel length of 2 μm, were measured using an Keithley Semiconductor Characterization Analyser (2636B) with the source continuously grounded under atmospheric conditions. The field-effect mobility of thin films was calculated from a plot of square root of the drain current in the saturation regime using the equation (eqn (1)) where the condition for the saturation regime (VDS > VGS − VTH) is satisfied. IDS is drain–source current, μ is the field-effect mobility, W and L are the channel width and length, Ci is the capacitance per unit area of the gate insulator (Ci = 1.09 × 10−8 F cm−2), VG is the gate voltage and VTH is threshold voltage, was extracted from the x-intercept of the linear fit.40 | (1) |
3. Result and discussion
A four-arm star-shaped BCP, consisting of a star-shaped POEGMA core and linear P3HT corona, was synthesized via a click reaction between azide-functionalized four-arm POEGMA and alkyne-functionalized linear P3HT shown in Scheme 1. Four-arm star-shaped POEGMA with bromide end groups was synthesized via ATRP, using tetrafunctional pentaerythritol as the initiator. The bromine termini were subsequently converted to azide groups through reaction with sodium azide in acetone. Whereas alkyne-functionalized linear P3HT was prepared using the Kumada catalyst-transfer polymerization method. The reaction scheme is well established, as it has been extensively studied and validated.31,41 The resulting homopolymers and the star-shaped BCP, POEGMA-b-P3HT were characterized by GPC in chloroform (Fig. 1(a)). All samples exhibited monomodal molecular weight distributions, indicating well-controlled polymerization. The number-average molecular weights (Mn) and polydispersity indices (Đ) were determined to be 13 kDa and 1.30 for linear P3HT, 36 kDa and 1.16 for star POEGMA, and 82 kDa and 1.14 for the star POEGMA-b-P3HT. Further we analyzed the structural characteristics of homopolymers and BCP using 1H NMR and FT-IR.25,33Fig. 1(b) and (c) shows the comparison graph of 1H NMR and FT-IR spectra of linear P3HT, star POEGMA and star POEGMA-b-P3HT. | ||
| Scheme 1 Synthetic mechanism of star poly-oligo (ethylene glycol) methacrylate-b-poly-3 hexyl thiophene (POEGMA-b-P3HT) block copolymer. | ||
From (Fig. 1(b)) All characteristic peaks corresponding to both alkyne functionalized linear P3HT and azide modified star-shaped POEGMA are distinctly observed in the spectrum of the star POEGMA-b-P3HT. Notably, the alkyne proton signal at δ 3.52 ppm, which is present in the spectrum of linear P3HT, completely disappears in the spectrum of the star POEGMA-b-P3HT and a new peak emerges at δ 7.48 ppm, attributed to the triazole proton formed via the click reaction between the alkyne-functionalized linear P3HT and the azide-terminated star-shaped POEGMA. This spectral change provides strong evidence for the successful synthesis of BCP. Furthermore, FT-IR analysis provides additional confirmation of the successful BCP formation (Fig. 1(c)). The characteristic azide stretching vibration at 2112 cm−1, observed in the spectrum of star POEGMA, is absent in the star BCP spectrum, indicating the complete reaction between azido group of star POEGMA and linear P3HT during the click reaction. In addition, noticeable changes in the C–H stretching region (2800–3100 cm−1) of the BCP spectrum, compared to those of the individual components, further support the formation of a new polymeric architecture.
3.1. Optical analysis
We measured the optical spectra of thin films deposited onto quartz substrates using ellipsometry in transmission mode. The corresponding absorption spectra was calculated using the eqn (2):| α = A/d | (2) |
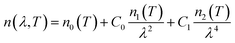 | (3) |
 | (4) |
 | ||
| Fig. 3 Ellipsometric models and refractive indexes of star poly-oligo (ethylene glycol) methacrylate (POEGMA) (a, b), linear poly(3-hexylthiophene) (P3HT) (c, d), and star POEGMA-b-P3HT (e, f). | ||
The thin film of star POEGMA-b-P3HT was modeled using an Effective Medium Approximation (EMA) layer based on the Bruggeman model, which accounts for small inclusions dispersed within the polymer matrix45 (Fig. 3(e) and (f)). In this case we have set the star POEGMA model as the inclusion material and the linear P3HT model as the host material. In this case, the content of both components is described by the so-called fraction of inclusion coefficient. The Bruggeman model for two-content case is the following relation: where:
 | (5) |
3.2. Electrochemical properties
The electronic state (HOMO/LUMO levels) of the polymer and BCP thin films was investigated using CV. The HOMO and LUMO energy levels were estimated from the onset potentials of the oxidation and reduction processes. Representative voltammograms are shown in Fig. 2(c) and (d), and the corresponding electrochemical parameters are summarized in Table S1. The linear P3HT exhibits an oxidation onset potential of +0.25 V, which corresponds to a HOMO level of −5.05 eV (Fig. 2(c)). This value is consistent with P3HT's well-established role as a p-type semiconductor, which predominantly supports hole transport.46 On the other hand, the reduction onset potential for linear P3HT appears at −1.48 V, indicating a LUMO level of −3.32 eV (Fig. 2(d)). The relatively low LUMO energy reflects its limited ability to transport electrons, reaffirming the hole-conducting nature of the polymer. In contrast, the star-shaped POEGMA-b-P3HT copolymer shows a deeper HOMO level (−5.17 eV) and a more stabilized LUMO level (−3.62 eV), derived from oxidation and reduction onset potentials of +0.37 V and −1.18 V, respectively. These shifts result in a narrower electrochemical bandgap, which implies a more balanced capability for both hole and electron transport.47 The resulted electrochemical properties of star BCP are due to its architecture, which allows for precise tuning of the conjugated P3HT backbone. This structural refinement enhances orbital alignment and promotes stronger π–π stacking interaction resulted stabilized HOMO and LUMO energy levels and more balanced charge transport characteristics.3.3. Phase behaviour and crystallinity
DSC was conducted to investigate the phase behavior of the homopolymers and the star BCP (SI). The glass transition temperature (Tg) of linear P3HT was observed at 22.9 °C (Fig. S2), which is attributed to its rigid chain structure and semi-crystalline nature which is comparable with earlier report.48 In contrast, star POEGMA exhibited a Tg at −51 °C (Fig. S3), consistent with its amorphous character and the flexibility imparted by the ethylene glycol side chains.49 For the star BCP, two distinct Tg were observed at −41 °C and 20.66 °C (Fig. S3), corresponding to the star POEGMA and P3HT segments, respectively. Studies have shown that incorporation of the low-Tg polymer into BCP enhances the flexibility, stretchability, and processability without significantly compromising device stability.14,15 The Tg of star-POEGMA in the BCP shifted slightly higher than the pure homopolymer, likely due to interactions with the P3HT domains. Similarly, the Tg of the P3HT segment shifted slightly lower than that of the linear P3HT, possibly influenced by the branched architecture by star-POEGMA. No distinct melting point was observed for either the homopolymers or the star BCP within the temperature range of −60 to 200 °C. The observation of two separate Tg values confirms the microphase-separated nanostructure of the star BCP.50 This result is further supported by AFM, which provided morphological evidence of distinct phase domains.Further to analyze the crystallinity and molecular orientation of linear P3HT and star-shaped POEGMA-b-P3HT in the thin film were investigated using XRD (Fig. 4(a)). Linear P3HT displays four distinct diffraction peaks at 2θ = 5.3°, 7.38°, 10.7°, and 16.1°, corresponding to the (100), (200), (300), and (400) planes, respectively, which are indicative of the lamellar stacking of hexyl side chains. A broad peak observed at 2θ = 23.8° corresponds to the (010) plane with d spacing 3.7 Å, signifying π–π stacking between the conjugated P3HT backbones. The high intensity of the (100) reflection, with a calculated d-spacing of 16.5 Å, suggests a well-ordered lamellar structure, which is consistent with AFM observations. The star-shaped POEGMA-b-P3HT copolymer exhibits all the characteristic diffraction peaks of P3HT, except for the (400) reflection. As shown in the graph, the (300) and (400) diffraction peaks have merged and appear broadened due to the presence of the amorphous halo structure attributed from POEGMA block, which imparts an overall amorphous character in the XRD pattern (Fig. S4). Moreover, it can be noticed that the diffraction pattern of the star-shaped BCP closely resembles that of linear P3HT.51 However, the star BCP shows a lower intensity in the (100) reflection and slightly increased d-spacings for both the (100) and (010) planes, measuring 16.7 Å and 3.8 Å, respectively. These changes indicate slightly reduced crystallinity, likely due to the substantial presence of the flexible ethylene glycol side chains in the amorphous POEGMA block, which interrupt the regular lamellar packing of the P3HT hexyl side chains as illustrated in the schematic diagram of material orientation in the solid state (Fig. 4(b)). Notably, the (010) peak is more pronounced in the star-shaped BCP, suggesting enhanced π–π stacking order along the (010) direction. This may be attributed to the spatial arrangement of the P3HT arms within the star-block architecture, which promotes better stacking between conjugated backbones.
3.4. Surface morphology
To analyze the surface morphology of the thin films of star-shaped POEGMA, linear P3HT, and star-shaped POEGMA-b-P3HT were examined using SEM (Fig. 5) and AFM (Fig. 6 and 7). SEM analysis (Fig. 5) revealed that while none of the thin films exhibited a well-defined structure, however their surface textures differed noticeably from one another. Star-shaped POEGMA showed a highly granular and irregular surface with pronounced voids. Linear P3HT also displayed an uneven morphology, but with a more continuous and cohesive texture. In contrast, the star-shaped POEGMA-b-P3HT film exhibited a significantly more uniform and densely packed surface, with fewer sharp boundaries, indicating improved molecular packing and higher structural order.Surface roughness profiles were further quantified using Phenom ProSuite software software (Table 2). The star-shaped POEGMA-b-P3HT exhibited the highest surface roughness (Rz = 3.65 μm), compared to linear P3HT (Rz = 2.13 μm) and star-shaped POEGMA (Rz = 1.97 μm). This increased roughness is attributed to the formation of phase-separated nanostructures or nano/micro-domains on the surface, likely induced by the unique architecture of the star-shaped BCP, as observed in the AFM images (Fig. 6(c)).
| Thin films | SEM | AFM (before annealing) | AFM (after annealing) | |||
|---|---|---|---|---|---|---|
| R z (μm) | R a (μm) | R q (nm) | R a (nm) | R q (nm) | R a (nm) | |
| Star POEGMA | 1.97 | 0.936 | 15.34 | 7.33 | 14.37 | 6.28 |
| Linear P3HT | 2.13 | 0.956 | 6.835 | 3.93 | 1.25 | 0.87 |
| Star POEGMA-b-P3HT | 3.15 | 1.86 | 27.34 | 21.53 | 4.40 | 3.39 |
AFM analysis were conducted for both before annealing and after annealing at 140 °C under inert atmosphere of thin films. To obtain comprehensive insights into the nano- and micro-scale organization of the materials on the surface, scans were conducted at large (20 × 20 μm2) (Fig. 6) and small (2 × 2 μm2) (Fig. 7) area at different regions of each samples. For the non-annealed star-POEGMA homopolymer (Fig. 6(a) and 7(a)), the AFM images revealed relatively small and large spherical or semi-spherical nanostructures throughout the scanned area, These features appeared well-dispersed with moderate aggregation, the big spherical structure which can be attributed to the clustering of the star polymer chains during the film casting process.52 The roughness in star POEGMA is higher due to its branched architecture and phase incompatibility between the POEGMA arms with the substrate, which promotes the formation of pronounced surface features. After annealing (Fig. 6(b) and 7(b)), no major changes in overall surface morphology were observed, although a slight reduction in surface roughness was noted. In contrast, the linear P3HT homopolymer displayed nanofibrillar structures at larger scan areas (Fig. 6(c)) with moderate roughness value, while smaller scan (Fig. 7(c)) regions revealed a lamellar morphology.53 This reflects the inherent crystallinity and π–π stacking of P3HT chains, which facilitate the formation of well-ordered domains, as confirmed by XRD analysis.
After annealing the P3HT film showed more uniform and continuous polymer network nanofibrillar structure with lower surface roughness, likely due to enhanced crystallinity and polymer chain reorganization54 (Fig. 6(d) and 7(d)). The star-shaped POEGMA-b-P3HT BCP films showed a completely different surface morphology compared to the individual homopolymers. The AFM images of non-annealed film revealed a densely packed and highly textured nanoscale surface (Fig. 6(e) and 7(e)). Unlike the pure star-POEGMA, the star BCP formed a surface with pronounced cylindrical spike-like features with moderate roughness value, showing a uniform array of nanoscale domains tightly packed together, creating a continuous and consistent film.41,55 After annealing the surface roughness value is much lower and morphology looks granular with localized areas of aggregation. In both images (Fig. 6(f) and 7(f)) clear phase separation between the POEGMA and P3HT blocks was evident, highlighting the microphase-separated nature of the BCP. The formation of these phase-separated structures in BCP thin films is primarily driven by the thermodynamic incompatibility between chemically distinct polymer blocks, which promotes microphase separation to minimize unfavourable interfacial interactions. These morphological features suggest that the presence of the rigid structure of linear P3HT block with π-conjugated structure also plays a dominant role in organizing the polymer into a well-ordered nanostructure while the star-shaped POEGMA-b-P3HT architecture enhances interchain interactions, reinforcing the nanostructured morphology.
3.5. Transistor characteristics
The electrical characterization of OFET devices was performed using both annealed and non-annealed thin films of P3HT (Fig. 8) as well as the star-shaped POEGMA-b-P3HT copolymer (Fig. 9). The extracted device parameters including μ, Ion/off ratio, and VTH are presented in Table S2. These parameters were extracted from the transfer characteristics (IDSversus VG) of devices. For the linear P3HT OFET device output characteristics were measured by sweeping the gate voltage from 0 to −30 V in −5 V steps, while the drain voltage was scanned from 0 to −30 V (Fig. 8(a) and (b)). The device exhibited typical p-type transistor behavior, with a pronounced saturation region in the drain current. The drain current increases with increasing the negative voltage, confirming hole-dominated charge transport. Transfer characteristics were measured at a drain voltage of −20 V, and the gate voltage was swept from +10 to –30 V (Fig. 8(c) and (d)). Hole mobilities were extracted from linear region in the vs. VGS plot. For the non-annealed P3HT films, the mobility was relatively low at 1 × 10−3 cm2 V−1 s−1. In contrast, annealed P3HT films exhibited a higher mobility of 3 × 10−3 cm2 V−1 s−1. This significant improvement in charge carrier transport and the shift of threshold voltage toward more negative values upon annealing is attributed to enhanced molecular ordering, reduced trap density and increased crystallinity within the P3HT chains.56 In contrast, to the unipolar p-type transport observed in linear P3HT, the star-shaped POEGMA-b-P3HT copolymer exhibited distinct ambipolar behavior, enabling conduction of both holes and electrons.57,58 The output characteristics were measured by sweeping the drain voltage from 0 to +30 V in +5 V increments, and the gate voltage from 0 to −30 V in −5 V steps under both p and n mode (Fig. 9(a), (b), (e) and (f)). Transfer characteristics were recorded at a fixed drain voltage of −20 V and +20 V (Fig. 9(c), (d), (g) and (h)). The non-annealed star POEGMA-b-P3HT device showed a hole mobility of 1.6 × 10−3 cm2 V−1 s−1 which is higher to linear P3HT and an electron mobility of 0.53 × 10−3 cm2 V−1 s−1, indicating ambipolar characteristics. However, annealing leads to overall improvement in charge transport, with hole mobility reaching to 1.7 × 10−3 cm2 V−1 s−1 and electron mobility increasing more substantially to 3. 09 × 10−3 cm2 V−1 s−1. This pronounced enhancement suggests that annealing more effectively benefits n-type conduction, possibly by reducing trap states and promoting molecular packing, which together enable more efficient electron hopping between localized states in the disordered BCP matrix. Furthermore, the on/off current ratio increased notably from 70 to 160, reflecting enhanced switching behavior, particularly for the holes. Interestingly, the subthreshold slope increased from 7.2 to 9.5 V decade−1, which may indicate, increased interfacial traps post-annealing. The observed ambipolar characteristics can be attributed to the structural and morphological modifications introduced by the star POEGMA side chains. These side chains interact with the hexyl groups of the polythiophene backbone, modifying the local dielectric environment and inducing significant changes in surface morphology. This interaction promotes the formation of microphase-separated nanostructures, resulting in well-defined domain. These nanoscale domains create efficient percolation pathways for both electron and hole transport, enhancing charge balance within the device. AFM analysis confirms improved morphology after annealing, correlating with increased charge carrier mobility.56 From the cyclic voltammetry results, the deeper HOMO level and elevated LUMO facilitate which support ambipolar transport However, further tuning of parameters such as the on/off current ratio and threshold voltage is necessary to achieve balanced and high-performance ambipolar OFETs.
vs. VGS plot. For the non-annealed P3HT films, the mobility was relatively low at 1 × 10−3 cm2 V−1 s−1. In contrast, annealed P3HT films exhibited a higher mobility of 3 × 10−3 cm2 V−1 s−1. This significant improvement in charge carrier transport and the shift of threshold voltage toward more negative values upon annealing is attributed to enhanced molecular ordering, reduced trap density and increased crystallinity within the P3HT chains.56 In contrast, to the unipolar p-type transport observed in linear P3HT, the star-shaped POEGMA-b-P3HT copolymer exhibited distinct ambipolar behavior, enabling conduction of both holes and electrons.57,58 The output characteristics were measured by sweeping the drain voltage from 0 to +30 V in +5 V increments, and the gate voltage from 0 to −30 V in −5 V steps under both p and n mode (Fig. 9(a), (b), (e) and (f)). Transfer characteristics were recorded at a fixed drain voltage of −20 V and +20 V (Fig. 9(c), (d), (g) and (h)). The non-annealed star POEGMA-b-P3HT device showed a hole mobility of 1.6 × 10−3 cm2 V−1 s−1 which is higher to linear P3HT and an electron mobility of 0.53 × 10−3 cm2 V−1 s−1, indicating ambipolar characteristics. However, annealing leads to overall improvement in charge transport, with hole mobility reaching to 1.7 × 10−3 cm2 V−1 s−1 and electron mobility increasing more substantially to 3. 09 × 10−3 cm2 V−1 s−1. This pronounced enhancement suggests that annealing more effectively benefits n-type conduction, possibly by reducing trap states and promoting molecular packing, which together enable more efficient electron hopping between localized states in the disordered BCP matrix. Furthermore, the on/off current ratio increased notably from 70 to 160, reflecting enhanced switching behavior, particularly for the holes. Interestingly, the subthreshold slope increased from 7.2 to 9.5 V decade−1, which may indicate, increased interfacial traps post-annealing. The observed ambipolar characteristics can be attributed to the structural and morphological modifications introduced by the star POEGMA side chains. These side chains interact with the hexyl groups of the polythiophene backbone, modifying the local dielectric environment and inducing significant changes in surface morphology. This interaction promotes the formation of microphase-separated nanostructures, resulting in well-defined domain. These nanoscale domains create efficient percolation pathways for both electron and hole transport, enhancing charge balance within the device. AFM analysis confirms improved morphology after annealing, correlating with increased charge carrier mobility.56 From the cyclic voltammetry results, the deeper HOMO level and elevated LUMO facilitate which support ambipolar transport However, further tuning of parameters such as the on/off current ratio and threshold voltage is necessary to achieve balanced and high-performance ambipolar OFETs.
 | ||
| Fig. 8 I–V characteristics of linear poly(3-hexylthiophene). (a) Output and (c) transfer characteristics before annealing; and (b) output and (d) transfer characteristics after annealing. | ||
4. Conclusions
In this study, we successfully synthesized a four-arm star-shaped BCP, POEGMA-b-P3HT, using click chemistry. The structure and successful formation of the BCP were confirmed through 1H NMR and FT-IR spectroscopy, while GPC analysis demonstrated a monomodal distribution, indicating complete coupling between the star-shaped POEGMA and linear P3HT chains. Optical characterization using Variable Angle Spectroscopic Ellipsometry revealed that linear P3HT exhibits anisotropic optical behavior in the solid state, whereas the star-shaped POEGMA-b-P3HT copolymer displays isotropic characteristics. This isotropy is attributed to the presence of localized ordered P3HT domains that are partially shielded by the surrounding amorphous POEGMA coils. Notably, the optical bandgap of the star copolymer was measured at 1.66 eV slightly lower than that of pure P3HT. CV measurements also demonstrated that the star-shaped BCP thin film exhibits a deeper HOMO and more stabilized LUMO than linear P3HT, leading to a narrower electrochemical bandgap, which enhances both hole and electron transport. Morphological studies using AFM and SEM revealed that the star-shaped architecture promotes the formation of a more uniform, densely packed film with a distinctive cone-like surface morphology. The phase behaviour and crystallinity via DSC and XRD confirmed a reduction in overall crystallinity due to the insulating nature of POEGMA compared to linear P3HT. However, π–π stacking interactions with an edge-on orientation were maintained in star BCP, which are essential for charge transport. The transistor measurements demonstrated that the linear P3HT showed a typical p-type semiconductor with a hole mobility of 1 × 10−3 cm2 V−1 s−1. Annealing improves its hole mobility to 3.1 × 10−3 cm2 V−1 s−1 and shifts the threshold voltage more negative, whereas the star-shaped POEGMA-b-P3HT exhibits ambipolar behavior with hole and electron mobilities of 1.6 × 10−3 and 0.53 × 10−3 cm2 V−1 s−1, respectively. Upon annealing, both mobilities increase, specifically electron mobility. The annealing process enhances the mobility of all devices and improved the morphology. These all results suggesting that the incorporation of non-conductive star POEGMA segments does not hinder, the electronic properties of the materials, Instead, it effectively tunes the characteristics, shifting the transistor behavior from unipolar p-type to ambipolar. This transition, driven by changes in internal molecular ordering, is attributed to the integration of star POEGMA blocks into the linear P3HT architecture. These findings highlight the potential of the star-shaped POEGMA-b-P3HT copolymer as a promising candidate for next-generation stretchable and biocompatible electronic devices, where structural versatility, mechanical flexibility, and balanced charge transport are essential. These parameters can be further optimized by tuning the copolymer architecture, molecular weight of the homopolymers, and processing methods.Author contributions
Pallavi Kumari – conceptualization, data curation, formal analysis, investigation, methodology, resources, supervision, validation, visualization, writing – original draft, writing – review and editing; funding; Barbara Hajduk: data curation, investigation, methodology, resources, writing – original draft, writing – review and editing; Anna celny–methodology and investigation; Agnieszka Kowalczuk– conceptualization, investigation, methodology, resources, supervision, writing – review and editing; Pawel Jarka – data curation, investigation, writing – review and editing; Sonia Kotowicz, Małgorzata Łazarska, Marcin Godzierz, Marta Musioł, and Anastasia Kobyliukh – data curation and investigation.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information is available. See DOI: https://doi.org/10.1039/d5tc03064a.Acknowledgements
This researcrh received no external funding.The authors gratefully acknowledges the institutional support and laboratory facilities provided by Centre of Polymer and Carbon Materials, Polish Academy of Sciences, Zabrze, Poland.References
- K. Liu, B. Ouyang, X. Guo, Y. Guo and Y. Liu, npj Flexible Electron., 2022, 6, 1 CrossRef CAS.
- M. Ashizawa, Y. Zheng, H. Tran and Z. Bao, Prog. Polym. Sci., 2020, 100, 101181 CrossRef CAS.
- X. Xu, Y. Zhao and Y. Liu, Small, 2023, 19, 2206309 CrossRef CAS PubMed.
- S. Chung, S. H. Kim, E. Ok, B. J. Kim, B. Kang and K. Cho, Chem. Mater., 2023, 36, 74–98 CrossRef.
- S. A. Jenekhe and D. Zhu, Polym. Chem., 2013, 4, 5142–5143 RSC.
- R. Liu, W. Yang, W. Xu, J. Deng, C. Ding, Y. Guo, L. Zheng, J. Sun and M. Li, ACS Appl. Polym. Mater., 2022, 4, 2233–2250 CrossRef CAS.
- K. Lee, M.-K. Jeong, E. H. Suh, W. Jeong, J. G. Oh, J. Jang and I. H. Jung, Adv. Electro. Mater., 2022, 8, 2101105 CrossRef CAS.
- H. N. Tsao, D. Cho, J. W. Andreasen, A. Rouhanipour, D. W. Breiby, W. Pisula and K. Müllen, Adv. Mater., 2009, 21, 209–212 CrossRef CAS.
- S. Kang, G.-H. Kim and S.-J. Park, Acc. Chem. Res., 2022, 55, 2224–2234 CrossRef CAS PubMed.
- J. Peng and Y. Han, Giant, 2020, 4, 100039 CrossRef.
- S. B. Darling, Prog. Polym. Sci., 2007, 32, 1152–1204 CrossRef CAS.
- A. F. Putranto, G. Fleury, C. Wulandari, A. Muslihati, Y. T. Amrillah, B. Yuliarto, M. Kogelschatz, F. A. A. Nugroho, H. S. Wasisto and M. Zelsmann, ACS Appl. Polym. Mater., 2024, 6, 14970–15001 CrossRef CAS.
- H. J. Kim, J.-H. Kim, J.-H. Ryu, Y. Kim, H. Kang, W. B. Lee, T.-S. Kim and B. J. Kim, ACS Nano, 2014, 8, 10461–10470 CrossRef CAS PubMed.
- J.-T. Wang, S. Takshima, H.-C. Wu, C.-C. Shih, T. Isono, T. Kakuchi, T. Satoh and W.-C. Chen, Macromolecules, 2017, 50, 1442–1452 CrossRef CAS.
- L.-C. Hsu, S. Kobayashi, T. Isono, Y.-C. Chiang, B. J. Ree, T. Satoh and W.-C. Chen, Macromolecules, 2020, 53, 7496–7510 CrossRef CAS.
- Y. Zhang, T. Guan, G. Han, T. Guo and W. Zhang, Macromolecules, 2019, 52, 718–728 CrossRef CAS.
- K. I. Aniagbaoso, M. Król, J. Ruokolainen, A. Bousquet, M. Save and L. Rubatat, ACS Appl. Mater. Interfaces, 2023, 15, 15998–16008 CrossRef CAS PubMed.
- A. S. R. Oliveira, P. V. Mendonça, S. Simões, A. C. Serra and J. F. J. Coelho, J. Polym. Sci., 2021, 59, 211–229 CrossRef CAS.
- P. P. Angelopoulou, I. Moutsios, G.-M. Manesi, D. A. Ivanov, G. Sakellariou and A. Avgeropoulos, Prog. Polym. Sci., 2022, 135, 101625 CrossRef CAS.
- D. Han, X. Tong and Y. Zhao, J. Polym. Sci., Part A: Polym. Chem., 2012, 50, 4198–4205 CrossRef CAS.
- T. Higashihara, S. Ito, S. Fukuta, T. Koganezawa, M. Ueda, T. Ishizone and A. Hirao, Macromolecules, 2015, 48, 245–255 CrossRef CAS.
- Y. Kim, H. Park, J. S. Park, J.-W. Lee, F. S. Kim, H. J. Kim and B. J. Kim, J. Mater. Chem. A, 2022, 10, 2672–2696 RSC.
- I. A. Aziz, G. Tullii, M. R. Antognazza and M. Criado-Gonzalez, Mater. Horiz., 2025, 12, 5570–5593 RSC.
- A. D. Nega, E. K. Pefkianakis, G. C. Vougioukalakis, E. Glynos and G. Sakellariou, Eur. Polym. J., 2016, 83, 148–160 CrossRef CAS.
- P. Kumari, B. Hajduk, P. Jarka, H. Bednarski, H. Janeczek, M. Łapkowski and S. Waśkiewicz, ACS Omega, 2024, 9, 39023–39032 CrossRef CAS PubMed.
- P. Kumari, M. K. Bera, S. Malik and B. K. Kuila, ACS Appl. Mater. Interfaces, 2015, 7, 12348–12354 CrossRef CAS PubMed.
- E. Ji, V. Pellerin, L. Rubatat, E. Grelet, A. Bousquet and L. Billon, Macromolecules, 2017, 50, 235–243 CrossRef CAS.
- M. Surin, O. Coulembier, K. Tran, J. D. Winter, P. Leclère, P. Gerbaux, R. Lazzaroni and P. Dubois, Org. Electron., 2010, 11, 767–774 CrossRef CAS.
- X. Yu, K. Xiao, J. Chen, N. V. Lavrik, K. Hong, B. G. Sumpter and D. B. Geohegan, ACS Nano, 2011, 5, 3559–3567 CrossRef CAS PubMed.
- Y. J. Lee, S. H. Kim, H. Yang, M. Jang, S. S. Hwang, H. S. Lee and K.-Y. Baek, J. Phys. Chem. C, 2011, 115, 4228–4234 CrossRef CAS.
- X. Pang, L. Zhao, C. Feng and Z. Lin, Macromolecules, 2011, 44, 7176–7183 CrossRef CAS.
- X. Pang, C. Feng, H. Xu, W. Han, X. Xin, H. Xia, F. Qiu and Z. Lin, Polym. Chem., 2014, 5, 2747–2755 RSC.
- C.-g Mu, X.-d Fan, W. Tian, Y. Bai and X. Zhou, Polym. Chem., 2012, 3, 1137–1149 RSC.
- A. Singh, N. Dowdall and T. Hoare, Biomacromolecules, 2025, 26(7), 3929–3973 CrossRef CAS PubMed.
- K. Sano, K. Umemoto, H. Miura, S. Ohno, K. Iwata, R. Kawakami, M. Munekane, T. Yamasaki, D. Citterio, Y. Hiruta and T. Mukai, ACS Appl. Polym. Mater., 2022, 4, 4734–4740 CrossRef CAS.
- B. L. Buss, L. R. Beck and G. M. Miyake, Polym. Chem., 2018, 9, 1658–1665 RSC.
- B. Mendrek, A. Fus-Kujawa, K. Klarzynska, A. Sieroń, S. Mario, A. Kowalczuk and A. Dworak, Polymers, 2018, 10, 1255 CrossRef PubMed.
- V. Parrillo, A. de los Santos Pereira, T. Riedel and C. Rodriguez-Emmenegger, Anal. Chim. Acta, 2017, 971, 78–87 CrossRef CAS PubMed.
- H. Gao and K. Matyjaszewski, Macromolecules, 2006, 39, 4960–4965 CrossRef CAS.
- Y. Zhou, X. Wang and A. Dodabalapur, Adv. Electron. Mater., 2023, 9, 2200786 CrossRef CAS.
- G. Yuan, T. Liang, Y. Liang, X. Pang and Z. Jia, Chem. Commun., 2021, 57, 1250–1253 RSC.
- J. Park, H. C. Moon and J. K. Kim, J. Polym. Sci., Part A: Polym. Chem., 2013, 51, 2225–2232 CrossRef CAS.
- B. Hajduk, P. Jarka, H. Bednarski, M. Godzierz, T. Tański, M. Staszuk, P. Nitschke, B. Jarząbek, M. Fijalkowski and K. Mazik, Sci. Rep., 2024, 14, 66 CrossRef CAS PubMed.
- A. J. Morfa, T. M. Barnes, A. J. Ferguson, D. H. Levi, G. Rumbles, K. L. Rowlen and J. van de Lagemaat, J. Polym. Sci., Part A: Polym. Chem., 2011, 49, 186–194 CrossRef CAS.
- P. Kumari, B. Hajduk, H. Bednarski, P. Jarka, H. Janeczek and M. Łapkowski, Nanomaterials, 2023, 13, 2918 CrossRef CAS PubMed.
- P. Kumari, K. Khawas, S. Hazra and B. K. Kuila, J. Polym. Sci., Part A: Polym. Chem., 2016, 54, 1785–1794 CrossRef CAS.
- J. H. Lee, D. H. Lee, H. J. Kim, S. Choi, G. E. Park, M. J. Cho and D. H. Choi, J. Polym. Sci., Part A: Polym. Chem., 2017, 55, 3223–3235 CrossRef CAS.
- R. Xie, Y. Lee, M. P. Aplan, N. J. Caggiano, C. Müller, R. H. Colby and E. D. Gomez, Macromolecules, 2017, 50, 5146–5154 CrossRef CAS.
- O. Vassiliadou, V. Chrysostomou, S. Pispas, P. A. Klonos and A. Kyritsis, Soft Matter, 2021, 17, 1284–1298 RSC.
- T.-Y. Wang, R. C.-C. Tsiang, J.-S. Liou, J. Wu and H.-C. Sheu, J. Appl. Polym. Sci., 2001, 79, 1838–1846 CrossRef CAS.
- F. Wang, R. D. Rauh and T. L. Rose, J. Am. Chem. Soc., 1997, 119, 11106–11107 CrossRef CAS.
- A. Deffieux, M. Schappacher, A. Hirao and T. Watanabe, J. Am. Chem. Soc., 2008, 130, 5670–5672 CrossRef CAS PubMed.
- N. Rujisamphan, R. E. Murray, F. Deng, C. Ni and S. I. Shah, ACS Appl. Mater. Interfaces, 2014, 6, 11965–11972 CrossRef CAS PubMed.
- S. Brixi, O. A. Melville, N. T. Boileau and B. H. Lessard, J. Mater. Chem. C, 2018, 6, 11972–11979 RSC.
- R. Goseki, A. Hirao, M.-A. Kakimoto and T. Hayakawa, ACS Macro Lett., 2013, 2, 625–629 CrossRef CAS PubMed.
- A. Zen, J. Pflaum, S. Hirschmann, W. Zhuang, F. Jaiser, U. Asawapirom, J. P. Rabe, U. Scherf and D. Neher, Adv. Funct. Mater., 2004, 14, 757–764 CrossRef CAS.
- Y. Ren, X. Yang, L. Zhou, J.-Y. Mao, S.-T. Han and Y. Zhou, Adv. Funct. Mater., 2019, 29, 1902105 CrossRef.
- C. Lu, J. Wang, H.-C. Chang, Y.-C. Chiu, H.-Y. Chen, H.-C. Wu, T. Higashihara and W.-C. Chen, J. Mater. Chem. C, 2014, 2(36), 7489–7493 RSC.
| This journal is © The Royal Society of Chemistry 2025 |







