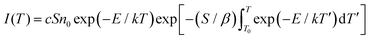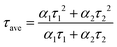Mn2+-doped Cs3Cu2I5 halides and their application in high resolution X-ray imaging and thermal neutron detection
Yuzhu
Li
 ab,
Jun
Chen
ac,
Xunsheng
Zhou
a,
Qi
Wu
ad,
Lu
Yao
*ad and
Cailin
Wang
ab,
Jun
Chen
ac,
Xunsheng
Zhou
a,
Qi
Wu
ad,
Lu
Yao
*ad and
Cailin
Wang
 *ad
*ad
aNeutron Science-Imaging Science and Technology, Songshan Lake Materials Laboratory, Dongguan, Guangdong 523808, China. E-mail: yaolu@sslab.org.cn; clwang@sslab.org.cn
bSchool of Materials Science and Engineering, Wuhan University of Technology, Wuhan 430070, China
cSchool of Materials Science & Engineering, Beijing Institute of Technology, Beijing 100081, China
dQuantum Science Center of Guangdong-Hongkong-Macao Greater Bay Area, Shenzhen, Guangdong 518045, China
First published on 3rd September 2025
Abstract
In this study, Mn2+-doped Cs3Cu2I5 microcrystalline powders were synthesized via a solution-based method, and their potential applications in X-ray and thermal neutron detection were explored. By optimizing the trace doping concentration, it was found that the incorporation of Mn2+ effectively modifies defect states, enhances photoluminescence (PL) intensity, and increases the Stokes shift, thereby significantly reducing self-absorption. In X-ray imaging, the Mn-doped samples achieved a maximum spatial resolution of 16.5 lp mm−1, surpassing most known halide materials. Additionally, by incorporating this material with 6LiF to fabricate thin films for thermal neutron detection, experimental results demonstrated that at a Mn-doping concentration of 50 ppm, a light yield of approximately 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 photons per thermal neutron, nearly 3.5 times that of the commercial GS20 (7000 photons per thermal neutron), was obtained and clear discrimination between neutron and gamma signals from the same source was achieved. These findings highlight the significant potential of Mn2+-doped Cs3Cu2I5 for multimodal radiation detection.
500 photons per thermal neutron, nearly 3.5 times that of the commercial GS20 (7000 photons per thermal neutron), was obtained and clear discrimination between neutron and gamma signals from the same source was achieved. These findings highlight the significant potential of Mn2+-doped Cs3Cu2I5 for multimodal radiation detection.
Introduction
Over the past decade, lead-based perovskites have demonstrated tremendous potential in the field of non-destructive testing, due to their high energy conversion efficiency, tunable wavelength, adjustable response time, and low manufacturing cost.1–3 Generally, different materials convert high-energy radiation into signals via distinct mechanisms.4 In contrast to direct detection, which converts high-energy radiation directly into electrical signals, indirect detection relies on scintillators to absorb radiation and emit visible light, which is subsequently detected by photodetector arrays to produce clear digital images.5,6 Indirect detection offers advantages such as lower costs, greater flexibility, larger-area coverage, and improved compatibility, making it more suitable for large-scale industrial applications. However, lead-based scintillators exhibit challenges, including limited stability against moisture and oxygen, photon self-absorption, and the inherent toxicity of lead.7 Consequently, there is growing interest in developing lead-free metal halide scintillators with enhanced stability, high light yield, and large Stokes shifts.8Copper-based halides have garnered significant attention due to their inherent non-toxicity, low self-absorption, and excellent chemical stability.9 Among new materials, Cs3Cu2I5 stands out for its high density and effective atomic number, which contribute to efficient radiation absorption and enhanced detection performance. Previous studies have demonstrated that Cs3Cu2I5 exhibits strong resistance to moisture-induced degradation, remains insensitive to air, humidity, and light, and can be rapidly synthesized into powder using a straightforward method.10–12 These attributes underscore its great potential for a variety of radiation detection applications.
To enhance material performance, researchers have proposed various approaches, with doping being one of the cost-effective methods.13–15 Incorporating one or more elements into a material can effectively modify its internal defects or surface properties, thereby enhancing its overall performance. Researchers have explored doping Cs3Cu2I5 with various ions, including alkali metals (e.g., Na+ and K+),16,17 transition metals (e.g., In+ and Cd2+),18,19 and halogen substitutions (e.g., Br− and Cl−)20,21 to improve luminescence efficiency and stability. Among these, Mn2+ doping is particularly noteworthy. Due to its magnetic properties, Mn2+ can interact with neighboring electronic states through exchange coupling or spin–orbit interactions, leading to spin-polarized electrons with altered spin orientations. As a radiative recombination center, Mn2+ ions are excited via energy transfer from the lattice, enhancing light yield and shifting the emission wavelength toward the longer-wavelength region.22,23 This increases the Stokes shift, significantly reducing self-absorption. However, it is worth noting that previously reported doped-scintillators often involved heavy doping, which typically introduces additional luminescence centers and significantly prolongs decay times. To mitigate this disadvantage, we propose a micro-doping strategy. By introducing trace amounts of magnetic ions, significant improvements can be achieved under high-energy radiation excitation, while avoiding the inclusion of slow decay components.
In this study, we developed Mn-doped Cs3Cu2I5 powders with a doping level of parts per million (ppm) using a solution method. The results demonstrate that this doping approach effectively modifies defects, leading to improved luminescence efficiency. Experimental data showed that at a Mn-doping concentration of 20 ppm, photoluminescence quantum efficiency peaked at 68%. For X-ray imaging, the maximum spatial resolution reached 16.5 lp mm−1. We fabricated a micro-composite scintillator using 6LiF and PMMA for thermal neutron detection. The light yield has been increased up to 3.5 times that of commercial GS20, a 6Li-containing neutron scintillator. Furthermore, it enabled clear discrimination between neutron and gamma signals. These findings confirm the material's significant potential for application in mixed field high-energy radiation detection.
Results and discussion
To optimize the micro-doping concentration, we prepared a series of samples with a concentration gradient. The SEM images and particle size distribution of the powders are shown in Fig. S1 and S2. At ppm-level doping concentrations, the average grain size is approximately 1.09–1.29 μm. When the doping concentration increases to 1%, the grain size increases significantly to about 2.7 μm. Apart from a few irregular particles, most grains exhibit a well-defined hexagonal prismatic morphology. As shown in Fig. 1(a) from XRD analysis, the peak positions and intensities of the prepared samples exhibit a high degree of correspondence with the standard reference data, indicating that the powder materials synthesized using the solution method possess good precision. Furthermore, the doping of Mn did not significantly affect the crystal structure. Upon detailed examination of the data, it was observed that a slight leftward shift in the peak positions occurred only when the Mn doping concentration reached 1%, suggesting that higher doping concentrations result in the lattice expansion of the crystals.To verify the successful doping of Mn2+ ions, X-ray photoelectron spectroscopy (XPS) measurements were performed. The full-spectrum data (Fig. S3) confirm the presence of Mn elements, while the analysis of characteristic peaks (Fig. 1b) reveals the Mn 2p3/2 peak at 642 eV and the Mn 2p1/2 peak at 652 eV, further validating the incorporation of Mn2+ dopants.24 Subsequently, the optical band gap of the samples was measured and analyzed. Fig. 1(c) and (d) show that the transmittance in the 360–500 nm range generally decreases initially and then increases with Mn doping. This trend correlates with changes in the optical band gap, which is influenced by the introduction of impurity levels resulting from Mn doping.
As shown in Fig. 2(a) and Fig. S4, photoluminescence (PL) and photoluminescence excitation (PLE) spectra were recorded for the synthesized powders, revealing broad excitation and emission bands with peaks at 310 nm and 440 nm, respectively. A large Stokes shift of 130 nm was observed between PLE and PL, consistent with previous reports. This characteristic is advantageous for subsequent applications. Notably, compared to pure Cs3Cu2I5 powder, the micro-doped samples exhibit significantly enhanced PL intensity at room temperature, which initially increases and then decreases with doping concentration. This behavior indicates the presence of an intriguing photophysical mechanism. Unlike samples with higher doping levels, trace amounts of Mn2+ do not form new luminescent centers at room temperature but significantly enhance the stress-related emission. The sample doped with 15 ppm Mn2+ achieves a photoluminescence quantum yield (PLQY) of up to 68.9% (Fig. 2b).
To investigate the reason why Mn2+ does not exhibit photoluminescence (PL) at room temperature, low-temperature PL measurements were performed. As shown in Fig. 2(d), for the sample doped with 15 ppm, a pronounced emission appears at 580 nm below 154 K, in contrast to the undoped sample shown in Fig. 2(c).25 Studies indicate that this peak originates from an increased concentration of point defects at low temperatures.23 This behavior is likely related to the low Mn2+ doping concentration and the one-dimensional lattice structure of Cs3Cu2I5. After Mn2+ incorporation, the enhanced emission is predominantly governed by increased radiative recombination, which contributes to increased light output and improved detector signal intensity. Moreover, the two samples exhibit completely opposite photoluminescence trends upon decreasing temperature.
To further investigate the trend variation and elucidate the emission mechanism of the doped sample, we performed a fitting of the full width at half maximum (FWHM) as a function of temperature using the following equation:26
To further explore the mechanism by which ppm-level doping affects the optical properties of the sample, we first measured its fluorescence lifetime under 310 nm excitation, as shown in Fig. 3(g). Furthermore, the fluorescence lifetime was analyzed by fitting the decay curves using a single-exponential decay function, as summarized in Table 1. The fitting was performed using the standard form
| y(t) = y0 + αe−t/τ |
| Mn2+ | τ (ns) |
|---|---|
| 0% | 1129.3 |
| 7.5 ppm | 1092.5 |
| 15 ppm | 1100.2 |
| 20 ppm | 2226.0 |
| 50 ppm | 1074.6 |
| 1% | 1075.3 |
Additionally, thermoluminescence (TL) analyses were performed on both doped and undoped samples (Fig. 2h and i). We employ the TL first-order kinetic model for fitting:
| Sample | E trap (eV) | n 0 | Peak position (K) |
|---|---|---|---|
| Cs3Cu2I5 | 1.03 | 8.54 × 105 | 505 |
| 0.67 | 8.62 × 104 | 388 | |
| 0.39 | 4.15 × 104 | 306 | |
| 0.24 | 3.28 × 104 | 231 | |
| 0.12 | 1.01 × 106 | 143 | |
| Mn2+ 15 ppm | 0.145 | 4.24 × 105 | 167 |
| 0.139 | 4.31 × 106 | 140 |
The above study demonstrates that the incorporation of trace amounts of Mn2+ influences defect states in the energy levels due to its magnetic properties, resulting in significantly improved optical performance. As shown in Fig. 3(a), a comparison with conventional Si-based and Pb-based materials reveals that Cs3Cu2I5 exhibits superior and reliable X-ray absorption capability. To investigate this further, radioluminescence (RL) tests were conducted in detail on powder samples with varying doping concentrations, as presented in Fig. 3(b), to evaluate the effect of dopant levels on the luminescence response under X-ray excitation.
Based on these results, along with previously evaluated lifetime and other fundamental properties, the 15 ppm-doped sample was selected for subsequent tests and applications. As shown in Fig. 3(c), we obtained the variation in luminescence intensity under different irradiation doses by adjusting the irradiation dose. Additionally, we compared the Na-doped sample, with the corresponding relationship presented in Fig. S5. A linear relationship was established between the luminescence intensity and irradiation dose, yielding a high linear fitting coefficient (R2 = 0.9977). Since the slope reflects the variation in luminescence intensity with respect to irradiation dose, it can be observed that the sensitivity of 15 ppm Mn-doped Cs3Cu2I5 is significantly higher than that of the reference sample.
The formulated solution was blended with a defined ratio of PMMA and subsequently spin-coated onto quartz substrates to yield crystalline films, which were employed for subsequent X-ray imaging evaluation, and the comprehensive experimental details are available in the SI. To evaluate the spatial resolution of X-ray imaging, a standard test pattern plate (0.03 mm Pb, CN82670), as depicted in Fig. 3(d), was used as the imaging target, and the resulting image resolution was subsequently analyzed. At certain dose rates, the detection limit was determined based on an SNR of 3. The sample achieved a resolution of up to 16 lp mm−1, as shown in Fig. 3(e), surpassing most currently known halide materials. To provide a more precise and objective quantification of the imaging system's spatial resolution, modulation transfer function (MTF) measurements were further conducted (in Fig. S9). As a result, the resolution was accurately determined to reach 16.5 lp mm−1, representing a significant breakthrough. Imaging tests also successfully resolved the structure of a spring inside a capsule (Fig. 3f), with clear and well-defined contours. These findings suggest that Cs3Cu2I5 is a promising material for X-ray detection application. To evaluate the broad applicability of the sample in X-ray imaging, we further conducted imaging tests on chicken wing joints and microchips, as shown in Fig. 3(g) and (h). The results demonstrate that the sample enables clear differentiation of internal structures and provides high-resolution visualization of fine structural details.
At the same time, the excellent optical properties of the material highlighted its potential application in neutron detection. Unlike X-rays, neutrons do not undergo electromagnetic interactions with matter, making direct detection extremely challenging. Typically, neutrons need to be converted into detectable secondary charged particles. Neutrons can be classified based on their energy, as shown in Fig. 4(a). Only a limited number of isotopes can react with neutrons to produce detectable secondary products within specific energy ranges. To address this, our team prepared thin films by combining the powder with 6LiF using PMMA as a matrix and tested them with a 252Cf radiation source. As illustrated in the schematic in Fig. 4(b), both gamma and neutron signals coexist in the 252Cf source. However, due to differing interaction principles with the material, the addition of 6LiF enables the simultaneous absorption of both signals, which are then transmitted via a PMT to a computer or oscilloscope for subsequent signal analysis. The experimental image of the fabricated composite film is shown in Fig. S6. Supplementary transmittance measurements were also conducted, as shown in Fig. S7, which may represent a factor influencing the experimental results. Further investigation will be conducted in future studies.
The critical metric of light yield was measured and compared to the conventional commercial material GS20. As shown in Fig. 4(c), at a doping concentration of 50 ppm, the light yield is approximately 3.5 times higher than that of GS20, with peak channel numbers of around 910 and 3200, respectively. Taking the commercial GS20 light yield of approximately 7000 photons per thermal neutron as a reference, the thin film achieves a light yield of 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 photons per thermal neutron. Although the thermal neutron counting rate of the Cs3Cu2I5–Mn/6LiF/PMMA film is lower than that of commercial GS20, likely due to the lower transmittance of the Cs3Cu2I5–Mn/6LiF/PMMA film compared to GS20, it remains a highly promising material for thermal neutron detection. To further investigate the neutron-related performance, we conducted decay curves measurements of the samples using an oscilloscope (Fig. 4d). Furthermore, we employed a biexponential decay model for fitting, and the results are presented in Table 3. The mathematical expression of this model is given by:
500 photons per thermal neutron. Although the thermal neutron counting rate of the Cs3Cu2I5–Mn/6LiF/PMMA film is lower than that of commercial GS20, likely due to the lower transmittance of the Cs3Cu2I5–Mn/6LiF/PMMA film compared to GS20, it remains a highly promising material for thermal neutron detection. To further investigate the neutron-related performance, we conducted decay curves measurements of the samples using an oscilloscope (Fig. 4d). Furthermore, we employed a biexponential decay model for fitting, and the results are presented in Table 3. The mathematical expression of this model is given by:
| I(t) = α1e−t/τ1 + α2e−t/τ2 |
| Mn2+ | α 1 | τ 1 (ns) | α 2 | τ 2 (ns) | τ ave (ns) |
|---|---|---|---|---|---|
| 0% | 6.96 | 3.48 | 12.77 | 189.22 | 187.4 |
| 7.5 ppm | 6.54 | 4.75 | 75.55 | 302.50 | 302.1 |
| 15 ppm | 6.77 | 5.75 | 83.44 | 308.39 | 307.9 |
| 20 ppm | 7.04 | 6.78 | 96.33 | 331.55 | 331.1 |
| 50 ppm | 6.97 | 6.10 | 90.51 | 322.44 | 321.9 |
| 1% | 7.85 | 8.31 | 59.58 | 190.25 | 189.2 |
The fitting results reveal that unlike the stability observed in fluorescence lifetime, the lifetime of Mn-doped samples increases significantly under high-energy irradiation. This phenomenon may be attributed to the influence of high-energy irradiation on the excitation of magnetic Mn ions. The excited electrons may interact with magnetic ions through exchange interactions or spin–orbit coupling, potentially affecting the formation of triplet exciton states.
As shown in Fig. 4(b), the decay dynamics of scintillation induced by neutrons and gamma rays exhibit distinct characteristics, which directly impact the neutron-gamma discrimination (NGD) capability. NGD based on pulse shape differences utilizes the conventional dual-feature S1–S2 plot (Fig. 4(e)), where S1 and S2 represent the integrated pulse values within the lifetime windows of [−50, 0] ns and [0, 50] ns, respectively. In the two-dimensional S1–S2 scatter plot, a red boundary line can be applied to effectively separate most neutron events from gamma-ray events, demonstrating a more pronounced separation effect compared to previous studies. To further investigate the neutron detection efficiency, we established a classification boundary, where events within the boundary are categorized as neutron events, while those outside are identified as gamma-ray events. A fitting analysis was conducted to derive the efficiency contour plots for neutron and gamma-ray detection, as shown in the inset. The thermal neutron detection efficiency was determined to be 7% relative to the GS20 detector. After evaluating the gamma-detection efficiency using the defined boundary, as shown in Fig. 4(e), we found that the NGD ratio (neutron-gamma efficiency ratio) reached 2 × 105. Further improvements in neutron detection efficiency and NGD ratio could be made by increasing the 6LiF fraction in the microcomposite scintillator, and/or by using better but more complicated NGD algorithms.30
Conclusion
This study reports the synthesis of Mn2+-doped Cs3Cu2I5 powders with doping levels in ppm via a simple solution-based method and investigates their performance in both X-ray and thermal neutron detection. The results reveal that the doping strategy effectively passivates intrinsic defects, leading to enhanced photoluminescence efficiency. In X-ray imaging, the material achieved a maximum spatial resolution of 16.5 lp mm−1. Under thermal neutron irradiation, it demonstrated an outstanding light yield of approximately 24![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 photons per neutron and enabled clear discrimination between neutron and gamma signals originating from a single source. These findings underscore the considerable potential of this material for mixed-field, high-energy radiation detection applications.
500 photons per neutron and enabled clear discrimination between neutron and gamma signals originating from a single source. These findings underscore the considerable potential of this material for mixed-field, high-energy radiation detection applications.
Author contributions
Yuzhu Li contributed to conceptualization, data curation, and formal analysis; Yuzhu Li drafted the original manuscript and participated in review and editing; Jun Chen, Qi Wu, and Xunsheng Zhou conducted formal analysis and investigation; Cailin Wang acquired funding; Lu Yao led conceptualization, funding acquisition, supervision, and validation; Lu Yao participated in review and editing. All authors discussed and commented on the manuscript.Conflicts of interest
The authors declare no conflicts of interestData availability
The data that support the findings of this study are available from the corresponding authors upon reasonable request. All key datasets generated and analyzed during the current study are either included in this published article and its supplementary information files or can be obtained from the authors upon request.The supplementary information contains additional experimental detail. See DOI: https://doi.org/10.1039/d5tc02682b.
Acknowledgements
This work was supported by the National Natural Science Foundation of China (no. 12305203), the National Natural Science Foundation of China Major Instrument Project (no. 52127817), Guangdong provincial Quantum science strategic initiative GDZX23032006, and open research fund of Songshan Lake Materials Laboratory 2023SLABFN31.References
- W. Li, M. Li, Y. He, J. Song, K. Guo, W. Pan and H. Wei, Adv. Mater., 2024, 36, 2309588 CrossRef CAS PubMed.
- Y. Wu, J. Feng, Z. Yang, Y. Liu and S. Liu, Adv. Sci., 2023, 10, 2205536 CrossRef CAS PubMed.
- B. An, Y. Deng, Z. Jin and S. Sun, Adv. Funct. Mater., 2024, 2422522 Search PubMed.
- G. Kakavelakis, M. Gedda, A. Panagiotopoulos, E. Kymakis, T. D. Anthopoulos and K. Petridis, Adv. Sci., 2020, 7, 2002098 CrossRef CAS PubMed.
- A. Jana, S. Cho, S. A. Patil, A. Meena, Y. Jo, V. G. Sree, Y. Park, H. Kim, H. Im and R. A. Taylor, Mater. Today, 2022, 55, 110–136 CrossRef CAS.
- O. Balitskii, M. Sytnyk and W. Heiss, Adv. Mater. Technol., 2024, 9, 2400150 CrossRef CAS.
- S. Bhandari, S. Pramanik, M. Manna, S. Singha and F. Akhtar, Chem. Commun., 2025, 61, 817–840 RSC.
- Q. Zhou, W. Li, J. Xiao, A. Li and X. Han, Adv. Funct. Mater., 2024, 34, 2402902 CrossRef CAS.
- J. Wen, K. Rong, L. Jiang, C. Wen, B. Wu, B. Sa, Y. Qiu and R. Ahuja, Nano Energy, 2024, 109802 CrossRef CAS.
- F. Gao, X. Zhu, Q. Feng, W. Zhong, W. Liu, H. Xu and Y. Liu, Nano Energy, 2022, 98, 107270 CrossRef CAS.
- H. Chen, J. M. Pina, F. Yuan, A. Johnston, D. Ma, B. Chen, Z. Li, A. Dumont, X. Li and Y. Liu, J. Phys. Chem. Lett., 2020, 11, 4326–4330 CrossRef CAS PubMed.
- X. Huang, Q. Sun and B. Devakumar, Mater. Today Chem., 2020, 17, 100288 Search PubMed.
- J. T.-W. Wang, Z. Wang, S. Pathak, W. Zhang, D. W. de Quilettes, F. Wisnivesky-Rocca-Rivarola, J. Huang, P. K. Nayak, J. B. Patel and H. A. M. Yusof, Energy Environ. Sci., 2016, 9, 2892–2901 RSC.
- T. Yang, Y. K. Wang and L. S. Liao, Adv. Funct. Mater., 2024, 2420021 Search PubMed.
- Q. Cheng, W. Chen, Y. Li and Y. Li, Adv. Sci., 2024, 11, 2307152 CrossRef CAS PubMed.
- T. Wu, Y. Shi, H. Wu, M. Chen, H. Ran, J. Zheng, X. Li, J. Chen and Y. Tang, J. Mater. Chem. C, 2024, 12, 1002–1011 RSC.
- L. Xie, B. Chen, F. Zhang, Z. Zhao, T. Jiang, M. Wang, Y. Wu, L. Huang, W. Song and Y. Liu, J. Lumin., 2021, 239, 118333 CrossRef CAS.
- Q. Wang, Q. Zhou, M. Nikl, J. Xiao, R. Kucerkova, A. Beitlerova, V. Babin, P. Prusa, V. Linhart and J. Wang, Adv. Opt. Mater., 2022, 10, 2200304 CrossRef CAS.
- J. Qu, S. Xu, Y. Cui and C. Wang, Chem. Eur. J., 2022, 28, e202200877 CrossRef CAS PubMed.
- T. Haposan, A. Arramel, P. Y. D. Maulida, S. Hartati, A. A. Afkauni, M. H. Mahyuddin, L. Zhang, D. Kowal, M. E. Witkowski and K. J. Drozdowski, J. Mater. Chem. C, 2024, 12, 2398–2409 RSC.
- J. Zhao, B. Han, J. Wang, L. Chen, L. Xu, X. Wang and J. Song, Adv. Opt. Mater., 2024, 12, 2400277 CrossRef CAS.
- Q. Yao, J. Li, X. Li, Y. Ma, H. Song, Z. Li, Z. Wang and X. Tao, Adv. Mater., 2023, 35, 2304938 CAS.
- J. Zhang, Y. Han, L. Zhang, H. Zhu, T. Song, D. Yan, M. Zhang, C. Wang and X. Zhang, Mater. Res. Bull., 2023, 164, 112241 CrossRef CAS.
- L. Garvie and A. Craven, Phys. Chem. Miner., 1994, 21, 191–206 CrossRef CAS.
- F. Akman and H. Oğul, Macromol. Mater. Eng., 2024, 2400326 Search PubMed.
- Y. Wang, G. Liu and Z. Xia, Laser Photon. Rev., 2024, 18, 2300717 CrossRef CAS.
- C. Emary, L. A. Clark, M. Kataoka and N. Johnson, Phys. Rev. B, 2019, 99, 045306 CrossRef.
- J. Qin, C. Hu, B. Lei, J. Li, Y. Liu, S. Ye and M. Pan, J. Mater. Sci. Technol., 2014, 30, 290–294 CrossRef CAS.
- T. Hofbeck, U. Monkowius and H. Yersin, J. Am. Chem. Soc., 2015, 137, 399–404 CrossRef CAS PubMed.
- C. Wang, Rev. Sci. Instrum., 2021, 92, 10 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |