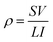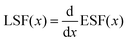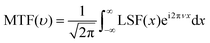Enhanced X-ray detection of one-dimensional DABCO–NH4I3 perovskites via anisotropic carrier transmission study
Long
Zhou
,
Meng
Xu
,
Jiahao
Geng
,
Yiran
Shi
,
Zhaolin
Song
,
Yuxuan
Liu
,
Hui
Yu
and
Menghua
Zhu
 *
*
State Key Laboratory of Solidification Processing, and School of Materials Science and Engineering, Northwestern Polytechnical University, Xi’an 710072, China. E-mail: mhzhu@nwpu.edu.cn
First published on 8th August 2025
Abstract
Metal-free perovskites exhibit significant potential for wearable X-ray detectors due to their non-toxic nature, degradability, aqueous solution processability and superior optoelectronic properties. DABCO–NH4I3 (DABCO denoted as N,N′-diazabicyclo[2.2.2]octonium) is a promising metal-free perovskite due to its advantages of reduced exciton binding energy, enhanced mobility–lifetime product and superior absorption coefficients. However, the undesirable X-ray detection performance of the device restricts its application. Reducing the dimensions of perovskite materials and adjusting the crystal orientation are effective methods for improving optoelectronic properties and enhancing detection performance. Herein, a high quality one-dimensional DABCO–NH4I3 single crystal is synthesized and in contrast to the conventional X-ray photoconductivity fitting approach, the time-of-flight (TOF) method using alpha particle-induced pulses is first employed to study the differences in single carrier transport performance between the [001] and [010] directions. The fitting results show that the hole mobility–lifetime products (μτ) for the [001] and [010] directions are 1.1 × 10−3 cm2 V−1 and 2.9 × 10−4 cm2 V−1 and hole mobilities (μ) are 55.53 cm2 V−1 s−1 and 4.63 cm2 V−1 s−1, respectively. Furthermore, the X-ray response results demonstrate a higher sensitivity of 62.1 μC Gy−1 cm−2 with a lower detection limit of 0.16 μGy s−1 for the [001] direction at 70 V (a sensitivity of 9.2 μC Gy−1 cm−2 with a detection limit of 0.16 μGy s−1 for the [010] direction). Hence, enhanced performances are successfully obtained by investigating the transport anisotropy of the DABCO–NH4I3 perovskite which also provides a feasible route to improving the detection performance of metal-free perovskites and promoting its applications.
Introduction
X-ray detectors are widely applied in various fields, including medical diagnostics, industrial testing, scientific research and security screening.1–3 Currently, direct X-ray detection predominantly relies on metal-based semiconductor materials.4,5 In addition to the existing detector materials such as silicon (Si), cadmium telluride (CdTe), and germanium (Ge), perovskite materials with superior optoelectronic properties have garnered significant interest.6–10 Compared to lead-based detectors, metal-free perovskite X-ray detectors characterized by their non-toxic and lightweight nature exhibit great suitability for wearable detection devices, favorable mobility–lifetime products and wide bandgaps, highlighting the significant potential for application in next-generation X-ray detectors.11–13As a representative one-dimensional metal-free perovskite, low cost DABCO–NH4I3 with quantum confinement effects and anisotropic characteristics shows great competitiveness for X-ray detection owing to the reduced exciton binding energy, prolonged carrier diffusion length and enhanced mobility–lifetime product.14 Additionally, the iodide ions contribute to superior absorption coefficients which leads to higher sensitivity and a lower limit of detection.15 Compared to conventional organic semiconductors (P3HT: 0.38 W m−1 K−1, MEH-PPV: 0.37 W m−1 K−1, and PBTTT-C14:0.39 W m−1 K−1), DABCO–NH4I3 with enhanced thermal conductivity (≈1.06 W m−1 K−1) can effectively mitigate thermal-induced degradation through improved heat dissipation and ensuring operational stability in X-ray detection.16,17 Although the optoelectronic properties of DABCO–NH4I3 are superior, enhancing the charged particle and X-ray detection performance remains a great challenge and shows great significance in promoting its application.
Dimensionality reduction and hydrogen bond strengthening serve as main strategies to enhance the performance of metal-free perovskites where dimension engineering is more efficient with a facile process.18–20 DABCO–NH4X3 crystals from molecular self-assembly at room temperature were prepared in previous research. The dimensionality of the crystal structure is determined by halogen atoms and results in different detection properties. The one-dimensional iodine crystal exhibited superior X-ray response with a hole mobility of 110 cm2 V−1 s−1, a hole migration distance of 90 μm and a μτ value of 10−3.21
Recently, carrier transport anisotropy of metal-free perovskites was found to have a significant effect on X-ray detection capabilities. The X-ray detection performance of large-sized CsPbBr3 single crystals across [100], [010], and [001] crystallographic orientations has been compared in previous work.22 The [010] orientation exhibits a higher activation energy and ion diffusion barrier, which effectively inhibited ion migration, resulting in a maximum sensitivity of 34![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 449 μC Gyair−1 cm2 and a limit of detection of 52.6 nGyair s−1. Therefore, acquiring DABCO–NH4I3 perovskites with a suitable crystalline orientation represents a promising strategy to enhance the optoelectronic performance.
449 μC Gyair−1 cm2 and a limit of detection of 52.6 nGyair s−1. Therefore, acquiring DABCO–NH4I3 perovskites with a suitable crystalline orientation represents a promising strategy to enhance the optoelectronic performance.
In this work, by reducing the material dimensions and regulating carrier transport anisotropy, high quality DABCO–NH4I3 crystals with a large size of 45 mm3 exhibit enhanced optoelectronic properties and detection performances. The electric field direction in devices with sandwich and coplanar structures aligns with the [010] and [001] crystal orientations, respectively, and exhibits different hole mobility (μ) and mobility-lifetime product (μτ) as measured by the time-of-flight (TOF) method using alpha particle-induced current pulses. The anisotropy of carrier transport also leads to the difference of X-ray detection performance. The coplanar device in [001] direction achieves a mobility–lifetime product (μτ) of 1.1 × 10−3 cm2 V−1, a hole mobility (μ) of 55.53 cm2 V−1 s−1, and a sensitivity of 62.1 μC Gyair−1 cm−2, representing a 4×, 12×, and 7× enhancement compared to other crystallographic orientations, respectively. Hence, by exploring different dimensions and transport anisotropy of DABCO–NH4I3 perovskite materials, enhanced detection performances are obtained, indicating that the oriented crystal growth of metal-free perovskites is an effective method to obtain optimal detection performance which then promotes the potential application in X-ray detection.23
Results and discussion
The aqueous solution was slowly evaporated at 35 °C, resulting in the formation of high-quality and transparent DABCO–NH4I3 crystals with dimensions of 9 × 3 × 2 mm (Fig. 1a and b). The morphology of the crystals is a hexagonal prism with pointed tips, as observed through scanning electron microscopy, as depicted in Fig. 1c. The utilization of water as a solvent aligned with principles of environmental sustainability and non-toxicity. In comparison to crystal growth at room temperature (25 °C), the occurrence of liquid-phase encapsulation defects was markedly diminished (Fig. S1a and b). The elevated temperature accelerated the deposition of solute molecules onto the crystal surface. This enhancement in growth rates effectively impeded the formation of liquid inclusions.24 Although increased temperature may lead to the development of numerous steps during crystal growth, these steps can be effectively eliminated through grinding and polishing, ensuring that excellent quality crystals are still obtained (Fig. S1c).The X-ray diffraction spectrum exhibits sharp peaks at 2θ = 21°, 32°, 43°, and 54°, which correspond to the (200), (300), (400), and (500) crystal planes, respectively (Fig. 1d). This observation suggests that the surface crystal plane is {100}. The UV-visible transmission spectrum presented in Fig. 1e indicates that the crystal demonstrates a transmittance of approximately 80% within the visible light spectrum. The absorption coefficient was calculated, and the direct bandgap Tauc plot was constructed as shown in Fig. 1f, revealing that the bandgap of the crystal is approximately 4.91 eV. This large bandgap (>4 eV) contributes to the suppressed dark current,25 while the relatively smaller bandgap achieved by iodine substitution (compared to DABCO–NH4Cl3 and DABCO–NH4Br3)21 facilitates more efficient carrier excitation. This optimal balance between bandgap reduction and dark current suppression ultimately enhances the X-ray detection sensitivity.26
DABCO–NH4I3 exhibits a one-dimensional hexagonal perovskite structure, characteristic of the classic ABX3 type. The NH4+ ion located at the B-site forms a twisted octahedron with six adjacent iodine atoms, forming a linear arrangement of the [(NH4)I6] octahedron through coplanar connections along the [001] direction. The A-site is occupied by the DABCO2+ cation, due to its non-spherical geometric configuration and hydrogen bonding interactions with iodine atoms which cannot be accommodated within the octahedral framework.20,27 Consequently, it is dispersed between the chains, leading to the formation of a one-dimensional structure, as illustrated in Fig. 2a (left). In this study, the [001] direction is designated as the c-axis. The one-dimensional nature of the crystal structure ensures that it maintains the same structural characteristics along both the [100] and [010] directions, which are uniformly defined as the ab-axis direction, as depicted in Fig. 2a (right). Following the grinding and polishing of the crystal, detection devices were fabricated with a sandwich structure that the electric field is oriented perpendicular to the c-axis, and a coplanar structure which is aligned parallel to the c-axis. The crystal surface was coated with sputtered gold electrodes and carbon glue, resulting in the formation of an Au–DABCO–NH4I3–Au device structure. Fig. 2b compares the resistivity in the two directions. The dark current I–V curve exhibited a resistivity of 5.64 × 108 Ω cm in the ab-axis direction and 6.55 × 109 Ω cm in the c-axis direction. The latter being an order of magnitude higher than the former, suggesting that the c-axis orientation may be more effective in minimizing the dark current under X-ray irradiation to achieve a lower limit of detection and an enhanced signal-to-noise ratio.28 At the same time, the charge transfer pathway in the c-axis direction demonstrates a pronounced quantum confinement effect due to the one-dimensional structure, resulting in more uniform and less obstructed carrier transfer.
The radioactive source in this study is an uncollimated 241Am@5.49 MeV α source, which was housed in a copper enclosure with the crystal positioned 1.5 cm from the crystal surface and the entire setup maintained at room temperature. The energy attenuation of α-particles at this specified distance has been modeled utilizing the SRIM software package. The α-particles incident on the crystal exhibits a mean energy of 3.99 MeV, penetrating to a depth of 19.9 μm, as illustrated in Fig. S2. To exclude the effects of gamma radiation emitted by the 241Am source, a sufficiently thick layer of tape was employed to encase the α source, obtaining a background curve that confirms the absence of gamma contributions to the full energy peak, as shown in Fig. S3. The α-particles interact with the crystal surface, generating electron–hole pairs. The surface electrons are rapidly collected at the anode, while the holes migrate towards the cathode under the external electric field. The pulse signal generated by the charge carrier drift is amplified using a preamplifier and then analyzed using a multi-channel analyzer to produce the pulse height spectrum, as shown in Fig. 3a. In the case of coplanar structure detectors, a sheet of paper is utilized to cover the region surrounding the anode which can characterize the transport properties of individual charge carriers.
Fig. 3b presents the pulse-height spectra of the DABCO–NH4I3 crystal along the c-axis and ab-axis over a wide bias voltage range (600–1200 V), demonstrating anisotropic charge transport behavior in the perovskite lattice. As the electric field intensifies, an increased collection of charge carriers is observed, which is reflected in the enhanced amplitudes of the signals from both the preamplifier and the main amplifier. This phenomenon results in a higher full energy peak position (channel address) and an enhanced energy spectrum as recorded using the multi-channel analyzer, thereby improving the charge collection efficiency (CCE). Although increasing the voltage enhances CCE and subsequently improves energy resolution, it also leads to an increased leakage current and degraded resolution. Consequently, there exists an optimal voltage at which resolution is optimal. The energy resolution (ER) for both the c-axis and ab-axis directions peaks at 800 V, achieving values of 51% and 62% (ER = FWHM/Epeak × 100%, where FWHM is full width at half maximum and Epeak is the peak position), respectively. At elevated bias voltages, the Joule heating effect generates significant thermal energy; yet, the crystal maintains a commendable spectrum response and resolution under these conditions. It can be attributed to the heat dissipation from the hydrogen bond network in the crystal lattice which leads to exceptionally high thermal conductivity (1.06 W m−1 K−1).17 The penetration depth is limited, allowing them to be treated as a single carrier transport phenomenon. The CCE can be characterized using a simplified Hecht equation:
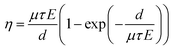 | (1) |
Data on forty fall times of holes were obtained using an oscilloscope across various voltage levels (Fig. 3d), and the pulse times (measured from 10% to 90% of the full amplitude) were subjected to histogram analysis. Typical results for a bias voltage of 1000 V are presented in Fig. 3e. It was observed that as the voltage increased, the drift time of holes decreased, resulting in an increase in drift velocity. The mobility (μ) of the photon-generated carriers throughout the entire thickness can be determined using the following equation:
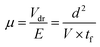 | (2) |
High-energy radiation was employed to assess the performance differences between the c-axis and ab-axis orientations. A comparative analysis of the X-ray absorption capabilities of various materials reveals that DABCO–NH4I3 demonstrates relatively high absorption coefficients within a photon energy range of 0.001 to 10 MeV, surpassing those of 4HCB and Si (Fig. 4a).33 This enhanced X-ray attenuation originates from the high atomic number of iodine in DABCO–NH4I3, aligning with the X-ray absorption relationship α ∝ Z4/E3, where Z is the atomic number and E is the X-ray photon energy. The radiation dose rates achieved by modulating the tube currents from 0.3 to 0.9 mA at a fixed tube voltage of 50 kVp were recorded as 0.86, 1.23, 1.6, 1.97, 2.34, 2.71 and 3.06 mGyair s−1. Current–time (I–t) curves were measured over a wide range of bias voltages (5–300 V). The photocurrent increases monotonically with voltage due to the enhanced directional drift of photogenerated carriers. With elevating voltages, the coplanar device exhibits stable photocurrent and dark current with a negligible drift (Fig. 4b), the sensitivity also increases with the increase of voltage (Fig. 4c).
In contrast, measurements along the ab-axis (using the same methodology) reveal a significant dark current drift and photocurrent instability at high voltages, as shown in Fig. S4. This behavior originates from the lower resistivity and non-uniform carrier transport characteristics along the ab-axis direction. Although the severe dark current drift in the ab-axis leads to elevated photocurrent levels, the maximum sensitivity reaches only 9.2 μC Gy−1 cm−2 at 70 V substantially lower than 62.1 μC Gy−1 cm−2 recorded for the c-axis at the same voltage (Fig. 4e). This discrepancy may be linked to the inhibitory effects of low-dimensional structures on the migration of iodide ions.34 According to the guidelines established by the International Union of Pure and Applied Chemistry (IUPAC), the minimum detection limit of a detector is defined as the X-ray dose rate that corresponds to a signal-to-noise ratio (SNR) of 3.35,36 The signal-to-noise ratio is defined as the ratio of photocurrent to noise current, with its calculation formula being:
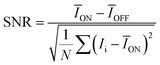 | (3) |
To further evaluate the stability of the DABCO–NH4I3 detector, the coplanar device underwent cycle performance and voltage stability testing after 120 days placed at room temperature and standard atmosphere. After 90 switching cycles under a bias of 30 V and a dose rate of 9.2 mGy s−1, the coplanar device exhibited steady photocurrent and dark current with negligible fluctuation (σph = 2.1 pA, σdark = 0.47 pA, and σ is the standard deviation) over a 5600-second continuous operation, demonstrating high photocurrent repeatability, as quantified and shown in Fig. 5a. In contrast, both the dark current and photocurrent of the sandwich device demonstrated a significant drift over time (σph = 4.4 pA and σdark = 5.9 pA) at the same voltage as shown in Fig. S5a. Additionally, I–t curves were evaluated at varying voltages under a dose rate of 9.2 mGy s−1. The results indicated that the photocurrent of the coplanar device increased with increasing voltage while maintaining a notably suppressed dark current as shown in Fig. 5b. Conversely, the sandwich device exhibited a marked increase in dark current as shown in Fig. S5b.
The coplanar device exhibits superior cycling stability and voltage stability, further substantiating the efficacy of one-dimensional structures in mitigating the dark current drift. Beyond high sensitivity and low limit of detection, X-ray detectors also need to possess high spatial resolution (defined as the minimum distinguishable distance between two points) for clinical imaging applications.38 Imaging characterization was performed using a line pair phantom with 0.2 mm step resolution under 200 V bias, achieving distinguishable line pairs at a dose rate of 9.2 mGy s−1 (Fig. 5c). The Boltzmann function was employed to fit the relationship between photocurrent and position to acquire the edge spread function (ESF) (Fig. S6a). Fourier transform was applied to the line spread function (LSF) (Fig. S6b) which is the first derivative of the ESF to establish the correlation between the modulation transfer function (MTF) and spatial frequency (Fig. 5d). The MTF represents the capability to achieve high imaging contrast at elevated resolutions. When the MTF reaches 20%, it corresponds to a spatial resolution of 1.9 Lp mm−1 for the DABCO–NH4I3 coplanar device.
Conclusions
In conclusion, to enhance the X-ray detection performance of metal-free DABCO–NH4I3 perovskites, high-quality one-dimensional DABCO–NH4I3 crystals are synthesized and the anisotropic property effects on carrier transport performance are explored. Radiation detection devices in coplanar and sandwich structures are fabricated along the [001] and [010] crystallographic directions, respectively. By using time-of-flight (TOF) methods, the charged alpha particle spectrum is measured for the first time. The carrier transport performance along the [001] direction (μτ = 1.1 × 10−3 cm2 V−1) is higher than that along the [010] and [100] directions (μτ = 2.9 × 10−4 cm2 V−1), with mobilities of 55.53 cm2 V−1 s−1 in the [001] direction and 4.63 cm2 V−1 s−1 in other directions. Compared to the [010] and [100] directions, the X-ray response of the device along [001] demonstrates more stable and lower dark current, indicating high sensitivity and low detection limit. Furthermore, [001] achieves a spatial imaging resolution of 1.9 Lp mm−1. It is demonstrated that an enhanced carrier transport performance and a repression effect on the dark current for DABCO–NH4I3 perovskites can be acquired by controlling the one-dimensional crystal direction, providing an effective strategy to enhance the detection performance of metal-free perovskites.Experimental
Materials
N,N′-Diazabicyclo[2.2.2]octane (DABCO), ammonium iodide (NH4I), hydroiodic acid (HI) (57%, stabilized with 1.5% hypophosphorous acid) and hypophosphoric acid (H3PO2) (50%) were purchased from Aladdin Reagent. All the reagents and chemicals were used as received without further purification.DABCO–NH4I3 crystal growth
DABCO (2.24 g) and NH4I (2.9 g) were dissolved in 5.3 mL HI (57%) and 30 mL H2O, and 3 mL H3PO2 was added as a stabilizer to avoid reduction to elemental iodine. The transparent solution was stirred at room temperature for 2 hours, and the filtered solution was slowly evaporated in a constant 35 °C water bath. After a few days, high-quality transparent crystals were obtained.Detection device fabrication
The grown single crystals were polished with sandpaper of 1000, 2000, 3000 and 4000 mesh in sequence, and then polished with nanosilica powder on a polishing cloth to reduce the surface roughness. Au/DABCO–NH4I3/Au devices with coplanar and sandwich structures were prepared by thermal evaporation (SBC-2-1 vacuum coating machine) of gold on the crystal surface. The circular gold electrode of the sandwich device has a diameter of 2 mm and a rectangular electrode of the coplanar device has a 1.2 × 2.5 mm scale; the center distance of the electrode was the same and the thickness of the electrode was approximately 100 nm.Scanning electron microscopy (SEM)
A field emission SEM (JSM-7500F) was used at an electron beam capability of 10 keV. The working distance was 11.0 mm.X-ray diffraction (XRD)
This work used a D8 Discovery A25 X-ray diffractometer. The basic experimental parameters were 1 mm slit, scanning start and end angles of 15–60°, and staying for 3 seconds every 0.05°, and the anode target material was a Co target.Optical measurements
UV-Visible transmission spectra were acquired using a UV-3150 Vis-NIR spectrophotometer operating in the 200 to 800 nm region.Current–voltage analysis
An Agilent 4155C semiconductor parameter analyzer was used at 100 V in the dark. The resistivity can be calculated using the following formula:where I is the current, V is the applied voltage, L is the distance between electrodes and S is the electrode area.
Alpha particle detection experiments
Using 241Am as the alpha source at room temperature, the sample was placed together with the device in a copper box at a distance of 1.5 cm and tested under a bias voltage of 600 to 1200 V. The induced current generated by the hole drift process can be integrated through the RC circuit of the preamplifier (Imdetek MA-01A) to obtain a charge related voltage signal, usually in the form of rising (or falling) steps. The time taken for the 10% to 90% rise in amplitude between the upper and lower platforms was recorded and an oscilloscope was used to calculate the carrier transport time of the detector. The main amplifier (RTEC 673) further amplified the signal output using the preamplifier and converted it into a Gaussian peak shape. Finally, a digital multi-channel analyzer and GammaAnt V2 software identified the height of each waveform and counting analysis was performed to form an energy spectrum. In this work, the maximum channel of the energy spectrum was set to 2048.X-ray detection and imaging experiments
This work used a LUX-50PC1-D X-ray source with a tube voltage range of 10 to 50 kV and a tube current range of 0.3 to 1 mA. Carriers were directed towards the two electrodes under the action of an external electric field to generate photocurrent, which was collected using a Keithley 6517B testing system. Different I–V curves were obtained by adjusting the tube current and bias voltage. A Geiger counter and a thermoluminescence dosimeter were used to calibrate X-ray doses. Single pixel imaging was performed by placing a line pair card on a 3D moving platform between an X-ray source and a detector. Different electrical signal intensities indicated the degree to which X-rays are obstructed by objects, and a Keithley 6517B electrometer was used to record the current signal of the detector at each position.Calculation process of the MTF
The measured photocurrent curve was fitted with the Boltzmann function to obtain the edge spread function (ESF(x)). The line spread function (LSF(x)) can be calculated using the following equation:The LSF(x) was fitted by the Gaussian function, and performing fast Fourier transform on LSF(x) can obtain the modulation transfer function (MTF(υ)):
Conflicts of interest
The authors declare no competing financial interest.Data availability
The data supporting this article have been included as part of the SI: additional experimental details, materials, tests, and methods, including photographs of the experimental setup. See DOI: https://doi.org/10.1039/d5tc02629fAcknowledgements
This work was supported by the National Natural Science Foundation of China (No. 62104194) and the Research Fund of the State Key Laboratory of Solidification Processing (NPU), China (No. 2024-TS-09 and 2025-QZ-05).References
- B. Wang, X. Yang, S. Chen, S. Lu, S. Zhao, Q. Qian, W. Cai, S. Wang and Z. Zang, iScience, 2022, 25, 26 Search PubMed.
- R. Liu, F. Li, F. Zeng, R. Zhao and R. Zheng, Appl. Phys. Rev., 2024, 11, 37 Search PubMed.
- M. H. Miah, M. U. Khandaker, M. A. Islam, M. Nur-E-Alam, H. Osman and M. H. Ullah, RSC Adv., 2024, 14, 6656–6698 Search PubMed.
- J. Yan, Q. Zheng, S. Wang, Y. Tian, W. Gong, F. Gao, J. Qiu, L. Li, S. Yang and M. Cao, Adv. Mater., 2023, 35, 14 Search PubMed.
- J. Yan, F. Gao, Y. Tian, Y. Li, W. Gong, S. Wang, H. Zhu and L. Li, Adv. Opt. Mater., 2022, 10, 10 Search PubMed.
- T. Jing, C. A. Goodman, J. Drewery, G. Cho, W. S. Hong, H. Lee, S. N. Kaplan, V. Perez-Mendez and D. Wildermuth, Nucl. Instrum. Methods Phys. Res., 1996, 368, 757–764 Search PubMed.
- O. Grimm, M. Bednarzik, G. Birrer and V. Commichau, Nucl. Instrum. Methods Phys. Res., Sect. A, 2020, 972, 164116 Search PubMed.
- L. Manzanillas, S. Aplin, A. Balerna, P. Bell, J. Casas, M. Cascella, S. Chatterji, C. Cohen, G. Dennis, P. Fajardo, H. Graafsma, H. Hirsemann, F. J. Iguaz, K. Klementiev, T. Kołodziej, T. Martin, R. Menk, F. Orsini, M. Porro, M. Quispe, B. Schmitt, N. Tartoni, M. Turcato, C. Ward and E. Welter, Nucl. Instrum. Methods Phys. Res., Sect. A, 2023, 1047, 167904 CrossRef CAS.
- A. Jana, S. Cho, S. A. Patil, A. Meena, Y. Jo, V. G. Sree, Y. Park, H. Kim, H. Im and R. A. Taylor, Mater. Today, 2022, 55, 110–136 Search PubMed.
- K. Sakhatskyi, A. Bhardwaj, G. J. Matt, S. Yakunin and M. V. Kovalenko, Adv. Mater., 2025, 37, 21 CrossRef PubMed.
- Z. Hao, Y. Li, Y. Deng, Z. Chen, J. Liang, X. Lu and J. Zhang, Org. Electron., 2024, 131, 107088 CrossRef CAS.
- P. Deng, H. Wang, X. Yang, H. Ren and Q. Jiao, J. Alloy. Compd., 2020, 827, 7 CrossRef.
- H. Ye, Y. Tang, P. Li, W. Liao, J. Gao, X. Hua, H. Cai, P. Shi, Y. You and R. Xiong, Science, 2018, 361, 151–155 CrossRef CAS PubMed.
- Z. Chu, X. Chu, Y. Zhao, Q. Ye, J. Jiang, X. Zhang and J. You, Small Struct., 2021, 2, 21 Search PubMed.
- Y. Morizet, V. Jolivet, N. Trcera, T. Suzuki-Muresan and J. Hamon, J. Nucl. Mater., 2021, 553, 153050 CrossRef CAS.
- H. Ushirokita and H. Tada, Chem. Lett., 2016, 45, 735–737 CrossRef CAS.
- J. Lee, S. Jung, G. Kim, J. Park, K. Park, C. Yang and J. Lee, Adv. Mater., 2025, 37, 10 Search PubMed.
- P. Dong, C. Lin, N. Ye and M. Luo, Angew. Chem., Int. Ed., 2024, 63, 9 Search PubMed.
- P. Liu, Y. Xian, W. Yuan, Y. Long, K. Liu, N. U. Rahman, W. Li and J. Fan, Adv. Energy Mater., 2020, 10, 10 Search PubMed.
- Z. Li, G. Peng, Z. Li, Y. Xu, T. Wang, H. Wang, Z. Liu, G. Wang, L. Ding and Z. Jin, Angew. Chem., Int. Ed., 2023, 62, e202218349 Search PubMed.
- Q. Cui, X. Song, Y. Liu, Z. Xu, H. Ye, Z. Yang, K. Zhao and S. F. Liu, Matter, 2021, 4, 2490–2507 Search PubMed.
- Y. Hua, X. Sun, X. Li, F. Cui, Z. Yue, J. Liu, H. Liu, G. Zhang and X. Tao, J. Mater. Chem. C, 2023, 11, 9153–9160 Search PubMed.
- R. Su, S. Ghosh, T. C. H. Liew and Q. Xiong, Sci. Adv., 2021, 7, 8 Search PubMed.
- M. Li, C. Zhang, M. Li, F. Liu, L. Zhou, Z. Gao, J. Sun, D. Han and J. Gong, Chem. Eng. J., 2022, 429, 132450 Search PubMed.
- Z. Li, G. Peng, C. Shi, H. Wang, S. Ding, Q. Wang, Z. Liu and Z. Jin, Adv. Mater., 2023, 35, 2300480 Search PubMed.
- R. Devanathan, L. R. Corrales, F. Gao and W. J. Weber, Nucl. Instrum. Methods Phys. Res., Sect. A, 2006, 565, 637–649 Search PubMed.
- X. Xu and S. Yang, Matter, 2021, 4, 3067 Search PubMed.
- X. Song, H. Cohen, J. Yin, H. Li, J. Wang, Y. Yuan, R. Huang, Q. Cui, C. Ma, S. F. Liu, G. Hodes and K. Zhao, Small, 2023, 19, 2300892 CrossRef CAS PubMed.
- A. Bolognesi, A. Di Carlo, P. Lugli and G. Conte, Synth. Met., 2003, 138, 95–100 CrossRef CAS.
- X. Song, Q. Cui, Y. Liu, Z. Xu, H. Cohen, C. Ma, Y. Fan, Y. Zhang, H. Ye, Z. Peng, R. Li, Y. Chen, J. Wang, H. Sun, Z. Yang, Z. Liu, Z. Yang, W. Huang, G. Hodes, S. F. Liu and K. Zhao, Adv. Mater., 2020, 32, 2003353 CrossRef CAS PubMed.
- Y. Zhang, F. Liu, C. Jiang, F. Tang and X. Zhang, J. Mater. Chem. C, 2021, 9, 3470–3476 Search PubMed.
- Z. Yuan, C. Zhou, Y. Tian, Y. Shu, J. Messier, J. C. Wang, L. J. van de Burgt, K. Kountouriotis, Y. Xin, E. Holt, K. Schanze, R. Clark, T. Siegrist and B. Ma, Nat. Commun., 2017, 8, 7 Search PubMed.
- National Institute of Standards and Technology (NIST). XCOM: Photon Cross Sections Database [Online]. Available: https://physics.nist.gov/PhysRefData/Xcom/html/xcom1.html (accessed Jan. 12, 2025).
- H. Yang, Z. Cui, W. Li, X. Guo, C. Lu, H. Yuan, Y. Hu, W. Zhang, X. Li and J. Fang, Adv. Funct. Mater., 2024, 34, 8 Search PubMed.
- W. Zhu, W. Ma, Y. Su, Z. Chen, X. Chen, Y. Ma, L. Bai, W. Xiao, T. Liu, H. Zhu, X. Liu, H. Liu, X. Liu and Y. M. Yang, Light: Sci. Appl., 2020, 9, 10 Search PubMed.
- M. Chen, Z. Zhang, B. Wen, R. Zhan, K. Wang, S. Deng, J. Xie and J. Chen, J. Alloy. Compd., 2023, 941, 9 Search PubMed.
- J. Yuan, Q. Peng, J. Fu, Q. Yang, Z. Gao, Z. Wang, K. Li, S. Zang and B. Z. Tang, J. Am. Chem. Soc., 2023, 145, 27095–27102 Search PubMed.
- J. Yu, Y. Luo, N. Tian, Y. Liu, Z. Yang, J. Pi, L. Li, R. Zheng, C. Wang and S. F. Liu, Adv. Mater., 2025, 37, 9 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |