Polypyrrole (PPy) coating concentration mediates electromagnetic coupling and oxidative protection for iron oxide core particles in biomedical applications
Shahemi Nur
Hidayah
 a,
Kasri Mohamad
Arif
a,
Kasri Mohamad
Arif
 c,
Ab Rahim
Sharaniza
b,
Amir Muhammad
Abid
b,
Sikiru
Surajudeen
c,
Ab Rahim
Sharaniza
b,
Amir Muhammad
Abid
b,
Sikiru
Surajudeen
 *a and
Mahat Mohd
Muzamir
*a and
Mahat Mohd
Muzamir
 *a
*a
aFaculty of Applied Sciences, Universiti Teknologi MARA, Shah Alam, 40450, Malaysia. E-mail: mmuzamir@uitm.edu.my
bFaculty of Medicine, Universiti Teknologi MARA, Sungai Buloh Campus, Sungai Buloh, 47000, Malaysia
cKuliyyah of Sciences, International Islamic University Malaysia, Kuantan, 25200, Malaysia
First published on 1st October 2025
Abstract
While polypyrrole (PPy) coatings are known to enhance iron oxide nanoparticles' (IONPs') functional properties, the concentration-dependent interplay between electromagnetic coupling and oxidative protection remains challenging, due to their opposing dependence. In this study, we report a concentration-dependent strategy using PPy coatings to mediate this dual functionality in IONPs. Through micellar polymerisation, IONPs (50–100 nm) were coated with PPy at 10–50 mM concentrations. At 40 mM, conductivity peaked at 456 μS cm−1, a 99% increase compared to that of bare IONPs. Electromagnetic characterization via vector network analysis (VNA) revealed maximum shielding effectiveness (SET = 1.36 dB) at the same concentration, primarily through the reflection mechanism. Cyclic voltammetry (CV) curves showed suppressed core oxidation for >30 mM coating concentration, due to the formation of a redox-buffering interface that reduced Fenton-like reactivity under acidic conditions. This oxidative shielding effect, coupled with maintained cytocompatibility (>80% cell viability at ≤2% w/v), provides critical insight into redox-safe design. Despite ∼25% reduction in saturation magnetisation, this reflects the intrinsic trade-off associated with surface functionalization, yet retaining applicability for magnetically guided biomedical applications. These findings provide a concentration-dependent framework for designing multifunctional fillers, which could be aimed at promoting cell alignment and bioelectronic responsiveness in ferromagnetic hydrogel systems, potentially for biomedical applications.
1. Introduction
Iron oxide nanoparticles (IONPs) have attracted considerable attention for biomedical applications such as magnetic resonance imaging (MRI), targeted drug delivery, and magnetic hyperthermia,1–5 owing to their intrinsic magnetic properties, biocompatibility, and tunable surface properties. Among the most studied types of IONPs are magnetite (Fe3O4) and maghemite (γ-Fe2O3), which exhibit superparamagnetic behaviour due to the presence of mixed-valence Fe2+ and Fe3+ ions. They are also known as superparamagnetic iron oxide nanoparticles (SPIONs). SPIONs' magnetic properties are critically influenced by their size, morphology, and surface chemistry for targeted biomedical applications. For instance, in drug delivery applications, spherical SPIONs with a size less than 30 nm displayed superior cellular uptake and retention in the transport mechanism of blood, as they can penetrate tissues and circulate intravenously,6 while <100 nm IONPs are widely regarded as optimal for intravenous applications. However, their limited magneto-mechanical response to external magnetic fields due to their small magnetic volume may not be sufficient for the targeted use in magnetic actuation, hyperthermia, and scaffold-guided regeneration.2–7 To overcome this, recent advances in tissue engineering highlight the therapeutic benefits of the micron to the submicron scale of iron oxide clusters for magnetic actuation and hyperthermia applications.7 Moreover, in this micron size range, they can also be cleared by the immune system through phagocytosis.6,7 This supports our approach using PPy-coated IONPs with a hydrodynamic size of 150–800 nm, which not only offer collective dipole interactions but also aim to facilitate scaffold integration when subjected to magnetic actuation.The surface chemistry of IONPs also plays a crucial role in dictating the colloidal stability and interaction with biological systems, which leads to biocompatibility.8 Their high surface-to-volume ratio leads to strong interparticle magnetic, electrostatic, and van der Waals interactions, causing them to aggregate in physiological environments.9 The aggregated IONPs allow interactions with plasma proteins, leading to opsonisation and rapid clearance.10,11 This limits their effective surface area and bioavailability. In addition, due to their amphoteric surface behaviour, IONPs can undergo a shift in surface charge, often becoming more positively charged (Fe2+ to Fe3+).12 This not only promotes aggregation but also further reduces the stability of uncoated IONPs. Hence, these unprotected particles will be susceptible to oxidative degradation, which can trigger cytotoxic effects through Fenton-like reactions.8,13 These challenges limit their long-term performance, which can compromise the safe use of IONPs in biomedical applications.
The surface modification of IONPs has emerged as a critical strategy to overcome the inherent limitations of bare IONPs in biomedical applications, including poor colloidal stability, aggregation in a physiological environment, rapid clearance by the immune system, and vulnerability to oxidative degradation, particularly under physiological conditions.3,14,15 Conventional surface coating techniques, such as co-precipitation, electrochemical polymerisation, and chemical grafting, have been employed to achieve surface passivation and offer scalability.3,14,16 However, these methods often lack precise control over coating thickness and uniformity and typically require a metallic material as a conductive substrate,17–19 which can be harmful to biological systems. Additionally, radio frequency plasma treatment has been used to precisely modify nanoparticle surfaces.20 Yet the complex setup and harsh processing environment limit its usage for biomaterials. Among the available surface functionalization strategies, micellar polymerisation has emerged as a versatile and biologically compatible technique for the localised in situ coating of nanoparticles with functional polymers.21–23 Micellar polymerisation enables localised coating of nanoparticles under mild aqueous conditions using surfactant-stabilized micelles, facilitating the controlled polymerisation of monomer units directly at the nanoparticle interface.23–25 This enables the formation of uniform, tunable coating layers while preserving the integrity of both the core nanoparticle and the surrounding biological medium.21,22
Building on this coating strategy, a wide range of polymers can be employed. Typically, hydrophilic polymers such as polyethylene glycol (PEG),26–28 polyvinyl alcohol (PVA),29–31 polyvinylpyrrolidone (PVP), and natural polysaccharides like dextran32,33 or chitosan34,35 are commonly used in previous studies for biomedical applications. This is due to the capability of hydrophilic polymers to prevent aggregation, enhance colloidal stability, and prolong circulation time in vivo.27,30,33,35 Hydrophilic polymer coatings also reduce protein adsorption and immune recognition, making them suitable for systemic delivery. However, their lack of conductivity and passive nature limit their functional role, including protection against redox-driven ion release and minimal control over the magnetic or electrical response of the nanoparticle system. To overcome these limitations, this study focuses on hydrophobic, electroconductive polymer coatings, specifically polypyrrole (PPy).
Conductive polymers such as PPy, polyaniline (PANI), and PEDOT are becoming alternative conducting materials due to their π-electron system along the conjugated polymer backbone, which supports redox activity and electrical conductivity.18,36–38 PPy stands out for its environmental stability, ease of processing, and potential to impart simultaneous protective and functional electroactive characteristics.39–41 PPy is also particularly attractive for biomedical use due to its chemical stability and redox buffering capability. Unlike inert polymer coatings, PPy can form a physical barrier against oxidative degradation while also enabling electromagnetic (EM) interaction through its conductive network.14,42 As the increasing demand for electronic–biological integration advances, such as in neural interfaces and bioelectronic sensors,43–45 achieving stable electromagnetic coupling under physiological conditions becomes increasingly critical. In this context, the interaction between a magnetic nanoparticle core and its surrounding polymer coating, particularly PPy, plays a decisive role in facilitating efficient charge transport, interfacial polarisation, and field responsiveness. Previous studies23 have shown that the conductivity improvement is highly dependent on the PPy coating's thickness, while other studies46,47 have independently reported increases in electromagnetic shielding with a thicker PPy coating layer. However, the structural irregularities and increased electrical impedance with a thicker non-magnetic PPy coating layer, which may disrupt charge flow and field penetration, have not been thoroughly investigated.
Given the contrasting roles that PPy coatings can play, there is a critical need to delineate the structural and functional thresholds at which this transition occurs. This study addresses a key gap in the literature by systematically investigating the influence of varying PPy coating concentrations on the electromagnetic behaviour of IONPs. By correlating coating morphology with charge percolation and field interaction, this study aims to define the critical percolation point that governs the switch from effective electromagnetic coupling to shielding-dominated attenuation. Establishing this relationship is essential for the rational design of multifunctional nanomaterials with precisely tuned electromagnetic properties. Such insight is particularly valuable for advancing biomedical platforms that rely on localised magnetic actuation, oxidative protection, and electrical conductivity, such as injectable conductive scaffolds, magneto-electroactive hydrogels, and stimuli-responsive regenerative systems.
A simple micellar coating technique was employed, in which sodium dodecyl sulphate (SDS) micelles served as a polymerisation template for pyrrole monomers and guided polymer growth directly at the nanoparticle interface. This approach allows localised in situ polymerisation under mild, biologically compatible conditions.21 The resulting PPy-coated IO nanoparticles were characterised for their chemical, physical, and electromagnetic properties. The concentration-dependent behaviour of the surface-modified IO nanoparticles was analysed using vector network analysis and cyclic voltammetry to understand their roles in preserving redox stability and promoting effective electromagnetic coupling. We also examine the protective role of the PPy coating against nanoparticle oxidation under both neutral and acidic conditions, simulating physiological environments during normal and inflammatory states. This work aims to establish a dual-functional platform that integrates oxidative protection with enhanced EM responsiveness, offering new insights into the rational design of nanomaterials for advanced biomedical applications.
2. Experimental section
2.1. Materials
Bare IONPs, comprising both Fe3+ and Fe2+ species, with a molecular weight of 231.53 g mol−1, particle size 50–100 nm (SEM), and 6–8 m2 g−1 surface area, as specified by the manufacturer (Sigma Aldrich, USA), were used as the core particle in this study. The iron oxide powder (0.5 g) was dispersed in sodium dodecyl sulfate (SDS, 99.9% purity, Fisher, USA), which served as the polymerisation template at the nanoparticle interface. To facilitate protonation, 0.1 M hydrochloric acid (37 wt% HCl, Fisher, USA) was added, adjusting the pH to 5.5. Subsequently, the pyrrole monomer (Py, 98% purity, Sigma Aldrich, USA) was introduced as the primary coating material, while ammonium persulfate (C12H25NaSO4, Sigma Aldrich, USA) was employed as the oxidising agent to initiate the polymerisation of PPy. The detailed procedure of the surface coating process is described in the following section.2.2. Surface coating of PPy on IO
5 wt% of Fe3O4 (IO) particles were dispersed in the 8 mM SDS solution under ultrasonication at room temperature for 10 min. Then, the pH of the solution was adjusted to 5.5 by adding 0.1 M HCl and the solution was further ultrasonicated for 24 h at 35 °C for the equilibrium adsorption step and to allow admicelle formation on the IO surfaces. After cooling to room temperature, pyrrole monomers were added to the aqueous medium at concentrations of 10 mM, 20 mM, 30 mM, 40 mM, and 50 mM, respectively, into each separate container and left to be continuously ultrasonicated for 2 h to allow adsolubilisation into the admicelle. APS was added as an oxidant to initiate the polymerisation at 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 wt% to the added pyrrole monomer.23 The polymerisation proceeded at 60 °C for 2 h to obtain IO-PPy. The coated IO was collected by magnetic separation and then washed three times with distilled water to remove excess unreacted pyrrole, surfactant, and persulfate. The final IO-PPy was dried under a vacuum for 12 h at 50 °C. The procedure is illustrated as shown in Fig. 1.
1 wt% to the added pyrrole monomer.23 The polymerisation proceeded at 60 °C for 2 h to obtain IO-PPy. The coated IO was collected by magnetic separation and then washed three times with distilled water to remove excess unreacted pyrrole, surfactant, and persulfate. The final IO-PPy was dried under a vacuum for 12 h at 50 °C. The procedure is illustrated as shown in Fig. 1.
 | ||
| Fig. 1 Schematic illustration of the PPy coating procedure via micellar polymerisation on iron oxide nanoparticles. | ||
2.3. Microstructure characterization
The chemical interactions between the surface coating and iron oxide core were characterised using Fourier transform infrared (FTIR, Nicolet, iS10, USA) spectra, which were recorded in transmission mode in the 1600–500 cm−1 range for PPy, bare IO, and IO-PPy.Particle size distributions were measured by using the images collected from a field emission electron microscope (FESEM). There are four distinct segments to ensure accuracy and to avoid repeated measurements of the same particle, which accounts for at least 100 measurements of each micrograph by ImageJ analysis. The particle size measurement is also evaluated using the dynamic light scattering (DLS) technique, where the IO was suspended in anhydrous ethanol for dispersion. The samples were previously sonicated for 15 min, and the measurements were taken 5 times. The variability of these two methods of particle size measurements was discussed. The surface morphology of the bare IO and coated IO at varied PPy concentrations was investigated using a field emission scanning electron microscope (FESEM, JSM-7600F, JEOL, Japan) operating at an acceleration voltage of 3 kV, at a magnification of ×100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 to ×200
000 to ×200![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000. The samples were prepared by spreading a thin layer on a conductive carbon tape for surface analysis.
000. The samples were prepared by spreading a thin layer on a conductive carbon tape for surface analysis.
The crystalline structure of pure PPy, bare IO, and IO-PPy has been investigated using X-ray diffraction (XRD) patterns on a Malvern PanAnalytical, NL, using CuKα radiation (λ = 1.546 Å) in the scattering range of 10°–90° with the scan rate of 4° min−1. The crystallinity index was quantitatively measured using the following formula:48
 | (1) |
2.4. Performance measurements
The electrical resistivity of pressed pellets of pure PPy, bare IO, and coated IO was measured using a four-point probe setup (CCS-01 programmable current source and Jandel Model RM3-AR, UK) at room temperature.The magnetic properties were determined using a vibrating sample magnetometer (VSM, Lakeshore 7400, USA) at room temperature. The VSM procedure was performed by varying the magnetic field density from −14 kOe to +14 kOe with a step size of 1 kOe.
The prepared IO and IO-PPy powder at various concentrations (10, 20, 30, 40, and 50 mM) were mixed with an epoxy polymer matrix consisting of epoxy resin (Araldite 506, Sigma Aldrich) and triethylenetetramine (TETA) (Sigma Aldrich) as a curing agent in a 15%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 85% ratio. The respective epoxy mixture was poured into a rectangular sheet mould of 10 mm × 23 mm × 1 mm and left for 24 hours to dry for 8–12 GHz frequency range analysis (X-band). The dielectric properties and electromagnetic shielding effectiveness (EMI) were measured using a vector network analyser (Agilent PNA, N5227A) using the transmission reflection line method through a parallel plate WR90 waveguide. A two-port setup was used as a simulation method of the S-parameters to test the electromagnetic attenuation response of the specimens.
85% ratio. The respective epoxy mixture was poured into a rectangular sheet mould of 10 mm × 23 mm × 1 mm and left for 24 hours to dry for 8–12 GHz frequency range analysis (X-band). The dielectric properties and electromagnetic shielding effectiveness (EMI) were measured using a vector network analyser (Agilent PNA, N5227A) using the transmission reflection line method through a parallel plate WR90 waveguide. A two-port setup was used as a simulation method of the S-parameters to test the electromagnetic attenuation response of the specimens.
Electrochemical measurements were performed using a Multi Autolab M204 (Metrohm Autolab, USA) using three electrode assemblies consisting of a platinum electrode as a counter electrode, a standard calomel electrode (SCE) as a reference electrode (Ag/AgCl), and IO-PPy as the working electrode. The electrochemical output was evaluated using cyclic voltammetry with the potential range between −1.0 V and 1.0 V, 5 times scan cycle in PBS solution at pH 5.8 and 7.4.
2.5. Biological evaluation through cell viability response
The in vitro biocompatibility of IO and IO-PPy was investigated by evaluating cell viability of human fibroblast cells in high glucose DMEM culture medium (Sigma Aldrich, USA), supplemented with 10% v/v fetal bovine serum (FBS) (Sigma Aldrich, USA) and antibiotics (1% solution of penicillin–streptomycin 100 μg mL−1) (Sigma Aldrich, USA) and incubated at 37 °C, 5% (v/v) CO2, and 95% relative humidity overnight before the nanoparticle treatment. These cells were seeded with 5 × 104 cells per well into 24-well plates in a 0.8 mL complete medium per well and left to settle in an incubator for 24 h (Cellbound, Corning Inc, USA). Then, the 40 mM PPy-IOs were added with target concentrations of 2, 4, 6, 8, and 10% w/v. The even dispersion of the nanoparticles was ensured by diluting each concentration in 200 μL medium before adding it to the cells. Trypan blue solution was applied to detect cell viability after culture for 1, 3, and 5 days using an automated cell counter (Invitrogen Countess, Thermo Fisher, UK).3. Results and discussion
3.1. FTIR analysis
The iron oxide (IO) particle surface was coated as previously shown in Fig. 1. In order to verify the successful surface coating formation, control samples of polypyrrole (PPy) and IO particles were characterised and compared with the surface-coated IO particle (IO-PPy) using FTIR analysis. The characteristics of the IR spectra of pure PPy and bare IO are shown in Fig. 2(a) in the range between 4000 cm−1 and 500 cm−1. In contrast, Fig. 2(b) displays the IR spectra of the surface-modified IO at various PPy concentrations: 10 mM, 20 mM, 30 mM, 40 mM, and 50 mM, detected in the range of 3500 cm−1 to 580 cm−1. The band at 608 cm−1 was associated with the Fe–O stretching vibration for the IO.23,49 Additionally, bands at 1609 cm−1 and 3856 cm−1 represented the OH group of ν(Fe–OH) on the surface of the IO.49,50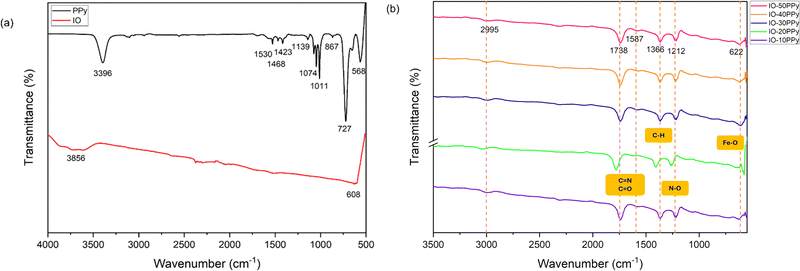 | ||
| Fig. 2 FTIR spectra of (a) pure PPy and bare IO and (b) IO–PPy at various concentrations (10 mM, 20 mM, 30 mM, 40 mM, and 50 mM). | ||
In the PPy's IR spectra, the peak at 3396 cm−1 corresponds to the N–H stretching vibration of Py units, with possible contribution from the OH group, as it was tested in liquid form.40,51–54 Furthermore, the characteristics of the PPy peak can be observed at 1530 cm−1, 1468 cm−1, and 1423 cm−1, associated with the asymmetric and symmetric vibration of the pyrrole ring.17,40,49,51,52,55,56 Transmission peaks at 1139 cm−1, 1074 cm−1, 1011 cm−1, and 867 cm−1 indicated C–H in-plane,3,57 C–N stretching,51 ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H in-plane deformation,50,51 and
C–H in-plane deformation,50,51 and ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C–H out-plane23,40,55 deformation, respectively. Additionally, the peak observed at 727 cm−1 is consistent with pyrrole ring deformation or C–H out-of-plane vibrations, in agreement with a previous report.54 In contrast, the 568 cm−1 transmission peak is less commonly discussed for PPy vibrations. However, similar to this earlier study,53 this peak is attributed to low-frequency pyrrole skeletal or ring deformation modes of the pyrrole backbone.
C–H out-plane23,40,55 deformation, respectively. Additionally, the peak observed at 727 cm−1 is consistent with pyrrole ring deformation or C–H out-of-plane vibrations, in agreement with a previous report.54 In contrast, the 568 cm−1 transmission peak is less commonly discussed for PPy vibrations. However, similar to this earlier study,53 this peak is attributed to low-frequency pyrrole skeletal or ring deformation modes of the pyrrole backbone.
Meanwhile, unlike the individual absorbance peaks of bare IO and pure PPy, the surface-modified IO-PPy exhibited newly distinct absorbance peaks, as shown in Fig. 2(b). The Fe–O stretching vibration shifted from 608 cm−1 to 622 cm−1, indicating the presence of Fe3O4 and vibrational interference due to the interaction with PPy. The peaks at 1212 cm−1, 1366 cm−1, 1587 cm−1, and 1738 cm−1 were assigned to the pyrrole C–H in-plane vibration, C–N stretching, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in-plane stretching, and C
O in-plane stretching, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N stretching, respectively.17,49,58 The peak observed at 2995 cm−1 may also suggest surface interactions between IO and PPy. These findings confirmed the formation of a PPy coating on the IO surface, as both Fe–O and pyrrole-related functional groups, such as C
N stretching, respectively.17,49,58 The peak observed at 2995 cm−1 may also suggest surface interactions between IO and PPy. These findings confirmed the formation of a PPy coating on the IO surface, as both Fe–O and pyrrole-related functional groups, such as C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, C
N, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, and N–O, were detected, creating a distinct FTIR spectral fingerprint.
O, and N–O, were detected, creating a distinct FTIR spectral fingerprint.
3.2. Morphological and particle size analyses
Scanning electron microscopy (SEM) revealed that the IO-PPy particles with various coating concentrations (10 mM to 50 mM) retained a spherical morphology as compared to the bare IO particles, as shown in Fig. 3. In the SEM analysis, which was performed on dry sample powder, particle size distributions were measured across four distinct segments using ImageJ, with over 100 measurements per micrograph to ensure accuracy. The results indicated no significant difference (p > 0.05) in particle size with increasing PPy concentration from 0 mM to 10 mM and 20 mM, yielding sizes of 150.13 nm ± 1.60, 166.44 nm ± 1.36, and 185.63 nm ± 1.29, respectively. However, at PPy concentrations of 30 mM to 50 mM, a significant increase (p < 0.05) in particle size compared to bare IO particles can be observed. The particle sizes of IO-30PPy, IO-40PPy, and IO-50PPy were calculated to be 190.99 nm ± 1.27, 205.82 nm ± 1.31, and 210.29 nm ± 1.33, respectively, suggesting a significant thickening of the PPy layer. Apart from that, particle aggregation was noticeable at higher concentrations (30 mM, 40 mM, and 50 mM), as highlighted in the red-dotted outline box in Fig. 3(d–f). To further assess the impact of PPy coating on particle size and aggregation tendency, dynamic light scattering (DLS) analysis was conducted.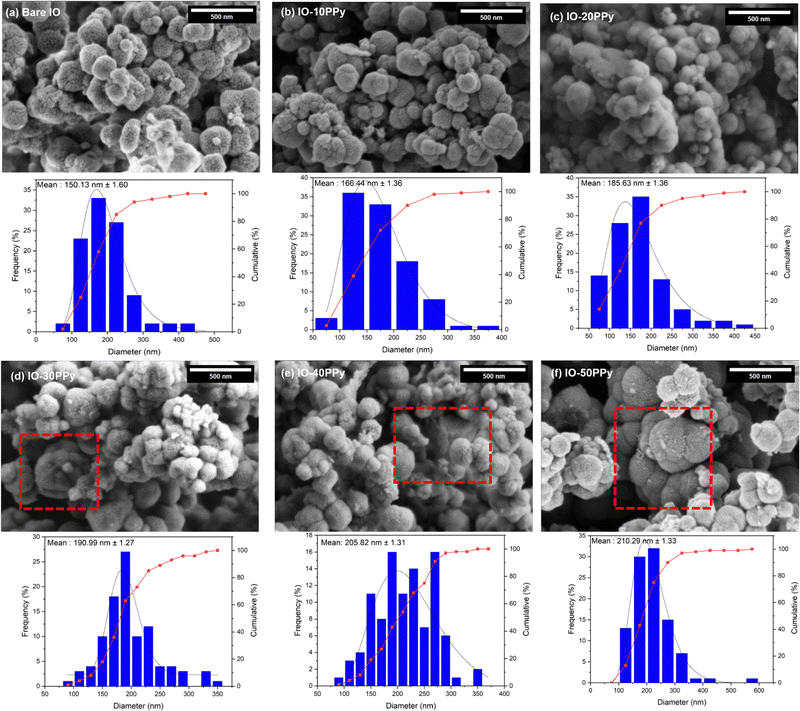 | ||
| Fig. 3 Morphological analysis and particle size distribution of (a) bare IO, (b) IO-10PPy, (c) IO-20PPy, (d) IO-30PPy, (e) IO-40PPy, and (f) IO-50PPy using ImageJ evaluation and statistical analysis. | ||
As shown in Fig. 4, DLS finding reveals a significant increase in hydrodynamic size at higher PPy concentrations, with sizes notably larger than those observed in SEM measurements. This discrepancy arises because DLS measures the hydrodynamic radius of particles in suspension, accounting for both the hydration layer and the ionic corona surrounding the particles.59,60 The polydispersity index (PdI) values further support this observation, showing values between 0.124 and 0.179 for PPy concentrations between 10 mM and 40 mM. While these values indicate good homogeneity and minimal aggregation, a slight tendency for surface-modified particles to aggregate is still observed, as shown in Fig. 3(d–f). However, at 50 mM PPy, the PdI value exceeds 0.3, indicating increased surface variability and pronounced aggregation.
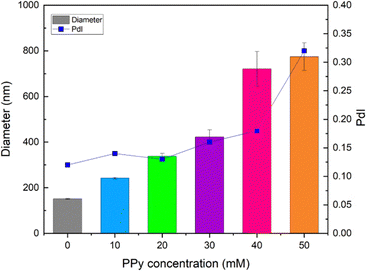 | ||
| Fig. 4 Hydrodynamic particle size and polydispersity index (PdI) of IO surface coatings at different PPy concentrations using DLS. | ||
This can be attributed to the reduced electrostatic and steric stabilisation caused by a high polymer-to-surfactant molar ratio (P/N ratio), where P is the concentration of pyrrole matrix chains and N corresponds to the grafted chains of the surfactant. This behaviour is consistent with literature findings,14,61 which highlight the critical role of polymer-to-surfactant ratios in governing the aggregation behaviour. In line with this, the calculated P/N molar ratio values for IO-10PPy, IO-20PPy, IO-30PPy, IO-40PPy, and IO-50PPy are 1.25, 2.50, 3.75, 5.0, and 6.25, respectively, further supporting the observed aggregation pattern. A P/N ratio threshold of 5.0 serves as a critical point. At a lower P/N ratio (<5.0),14,61 SDS provides sufficient electrostatic stabilisation and steric hindrance to maintain particle dispersion, reducing aggregation tendencies. Conversely, at a higher P/N ratio (>5.0), the dominance of matrix chains (PPy) relative to stabilizing surfactant chains leads to insufficient stabilisation. This imbalance promotes clustering and increases surface variability, as reflected in the higher PdI (>0.3) value62 and larger hydrodynamic size. Hence, the correlation between the PdI and P/N molar ratio demonstrates that increasing PPy concentrations beyond a threshold value (>40 mM) could compromise particle stabilisation, favouring aggregation and surface heterogeneity. These findings from SEM and DLS are summarised in Table 1.
| Sample | Particle size using SEM (nm) | Hydrodynamic size, d, (nm) | Polydispersity index (PdI) |
|---|---|---|---|
| Bare IO | 150.13 ± 1.60 | 151.23 ± 1.90 | 0.124 |
| IO-10PPy | 166.44 ± 1.36 | 242.25 ± 3.13 | 0.141 |
| IO-20PPy | 185.63 ± 1.29 | 338.36 ± 12.52 | 0.138 |
| IO-30PPy | 190.99 ± 1.27 | 422.96 ± 30.98 | 0.162 |
| IO-40PPy | 205.82 ± 1.31 | 720.73 ± 76.37 | 0.179 |
| IO-50PPy | 210.29 ± 1.33 | 774.31 ± 60.89 | 0.322 |
On the other hand, the destabilisation of inter-particle interactions has significant implications for the structural organization of the PPy coating layer. At lower PPy concentrations (10 mM to 40 mM), where the P/N ratio is ≤5.0,61 the particles remain relatively well dispersed, which corresponds to a more amorphous state. In this state, the uniform spacing between the PPy chains minimises opportunities for close packing and the formation of highly ordered crystalline regions. Conversely, as the P/N ratio increases with higher PPy concentrations, it enhances inter-particle interactions and aggregation. This aggregation promotes localised PPy chain densification, favouring the development of crystalline domains within the PPy coating.14,61 The XRD findings presented below further validate the transition from a predominantly amorphous to a more crystalline structure at increasing P/N ratios.
3.3. XRD analysis
Fig. 5 displays the diffraction patterns of bare IO and IO-PPy at various concentrations. The diffraction peaks of the surface-modified IO appear at the same angles as those of the bare IO, suggesting that the IO structure remains unchanged after the surface modification with the PPy coating layer. Specifically, the main peaks were observed at 30.0°, 35.5°, 43.1°, 53.5°, 57.0°, and 62.6°, corresponding to the (220), (311), (400), (422), (333), and (440) planes,11,23,50,55 in line with JCPDS 89-4319,51,63 confirming that the surface-modified IO retains its inverse spinel structure. Furthermore, an increase in diffraction peak intensity with higher PPy concentrations indicates enhanced crystallinity of the IO surface. This observation is quantitatively supported by calculating the crystallinity index,48 defined as the area under the crystalline peaks, divided by the total area under the XRD curve.17 The results indicate a progressive increase in crystallinity percentage with higher PPy coating concentrations, with values of 24.89%, 27.16%, 28.92%, 29.51%, 30.08%, and 31.98% for bare IO, IO-10PPy, IO-20PPy, IO-30PPy, IO-40PPy, and IO-50PPy, respectively.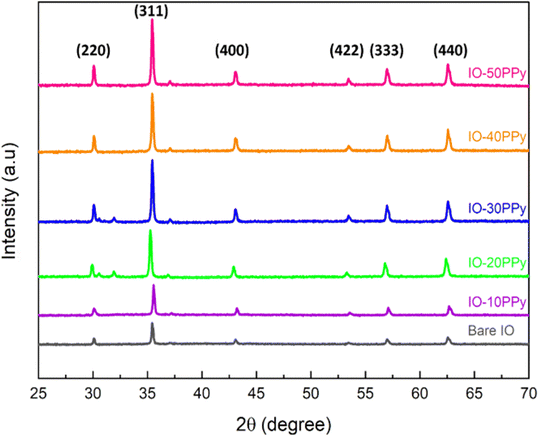 | ||
| Fig. 5 XRD diffraction angle on bare IO and coated IO surfaces with various PPy concentrations (10 mM, 20 mM, 30 mM, 40 mM, and 50 mM). | ||
As previously discussed, this effect can be attributed to the increasing P/N ratio, which promotes localised densification of the PPy layer on the iron oxide surface, leading to a more ordered structure and higher crystallinity. At higher P/N ratios, increased clustering facilitates closer chain alignment within the PPy layer, fostering the formation of crystalline regions.45,51,64,65 Conversely, at lower P/N ratios, the PPy coating remains amorphous, as evidenced by broader and less intense XRD peaks. This suggests that reduced aggregation at lower concentrations inhibits effective PPy chain alignment and packing, thereby maintaining a disordered structure. These findings highlight the dual influence of PPy concentration on both particle dispersion and surface structural organization. Table 2 summarizes the crystallinity index of the IO-PPy with its respective P/N molar ratios.
| Sample | P/N molar ratio | Crystallinity index (%) |
|---|---|---|
| Bare IO | N/A | 24.89 |
| IO-10PPy | 1.25 | 27.16 |
| IO-20PPy | 2.50 | 28.92 |
| IO-30PPy | 3.75 | 29.51 |
| IO-40PPy | 5.00 | 30.08 |
| IO-50PPy | 6.25 | 31.98 |
Meanwhile, the crystallite size was measured to further study the effect of surface coating on the iron oxide core properties. According to LaMer's model for nucleation and growth, exposure to high concentrations of surfactant can significantly reduce nanoparticle crystallite size.16,57 This reduction is attributed to the formation of stable monomers, which slows the nucleation rate.9,16 However, a fixed SDS concentration of 8 mM was used while varying concentrations of Py monomer were applied. Thus, it was hypothesised that higher Py monomer concentrations lead to increased reactivity, potentially influencing crystallite size during surface modification. The crystallite sizes of the specimens were calculated using the Scherrer equation below:1,50,66,67
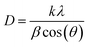 | (2) |
| Sample | Crystallite size (nm) | Std dev. |
|---|---|---|
| Bare IO | 44.086 | 6.600 |
| IO-10PPy | 44.038 | 3.600 |
| IO-20PPy | 43.808 | 5.057 |
| IO-30PPy | 42.971 | 6.879 |
| IO-40PPy | 39.764 | 3.520 |
| IO-50PPy | 41.916 | 6.071 |
The results show no significant differences in the crystallite size between the samples (p > 0.05), suggesting that the surface modification of IO with PPy did not significantly affect the crystallite size. This indicates that the core structural properties of IO remain unchanged following surface modification. With the successful establishment of the chemical interaction between PPy and IO, it is essential to investigate the effect of the surface coating on the physical characteristics of the particles, including their conductivity, magnetic properties, and oxidation properties.
Hence, two main findings were observed from the XRD analysis. Firstly, at high P/N ratios (>5.0), the lower stabilisation of the particles tends to result in increased aggregation. This aggregation can promote the formation of crystalline regions within the PPy coating, leading to more prominent crystalline peaks in the XRD analysis, indicating higher crystallinity. Secondly, the PPy coating does not influence the crystallite size of the underlying iron oxide core, indicating that the core's intrinsic crystal structure remains unchanged despite its surface modifications, which is significant to ensure that the PPy coating can be enhanced without compromising the core's inherent qualities, like its magnetic properties. Additionally, since the ordering of their crystalline structure often influences the electrical conductivity of materials, conductivity measurements were subsequently performed to assess the impact of crystallinity on electron transport.
3.4. Electrical conductivity property analysis
Fig. 6 illustrates the conductivity properties of IO-PPy, which gradually increase with higher concentrations of PPy, reaching a maximum value of 456 μS cm−1 at 40 mM PPy. From 0 mM to 20 mM PPy, the conductivity rises steadily from 3.39 μS cm−1 to 35.25 μS cm−1, then to 66.77 μS cm−1, respectively. However, at 30 mM PPy, there is a sharp increase in conductivity to 242.34 μS cm−1. This significant rise may indicate the formation of a more continuous and efficient conductive network at higher PPy concentrations, enhancing electron mobility.38In this IO-PPy system, the conductive pathways within the iron oxide matrix become increasingly interconnected as the PPy concentration rises.23 At lower concentrations (0 to 20 mM), PPy may not form a continuous network, resulting in only a gradual increase in conductivity. Meanwhile, at 30 mM, the concentration likely surpasses the percolation threshold, creating a continuous conductive network and a sharp increase in conductivity.68,69 Beyond 30 mM, the conductivity continues to increase, reaching a maximum at the PPy concentration of 40 mM. The thickened PPy coating contributes to a more uniform and continuous conductive network on the iron oxide surface. This enhanced uniformity and coverage not only improve conductivity but also enhance the material's crystallinity, as demonstrated by our XRD results. The thickened PPy layer allows for better alignment of the polymer chains, reducing amorphous regions and promoting more ordered crystalline structures.38,68,70,71 This improved crystallinity likely optimizes the electron transfer properties, further supporting the observed conductivity enhancements. However, at 50 mM PPy, the conductivity slightly decreases to 422.59 μS cm−1 (p > 0.05). At this concentration, the coating may become excessively thick, leading to intralayer aggregation and structural disorder, which disrupts the conductive pathways.23,64,72
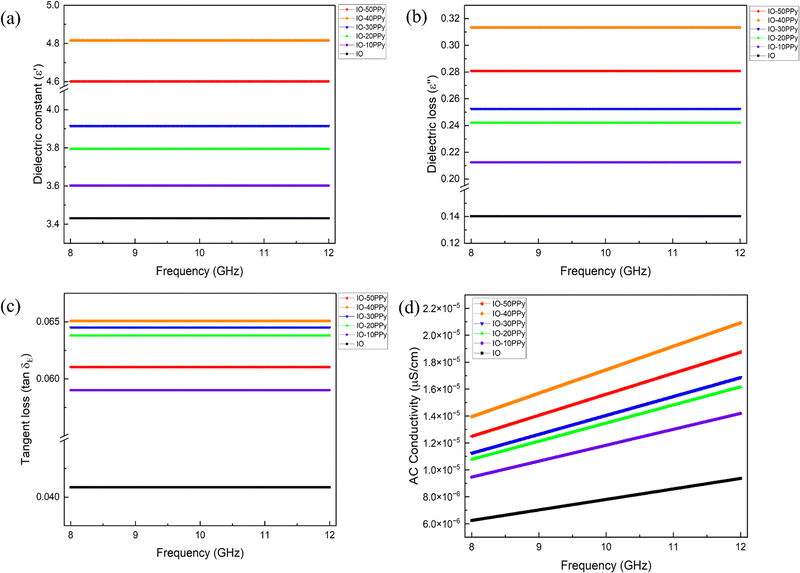
This can be further supported by the dielectric properties of the sample through a vector network analyser (VNA). A VNA is a tool to measure electrical properties by analysing the propagation of electromagnetic signals over a range of frequencies. In this study, X-band frequencies (8 GHz–12 GHz) were used to measure the complex permittivity (ε′, ε′′). The real permittivity (ε′), also known as the dielectric constant, represents the material's capacity to store electrical energy by polarizing charges in response to an applied field. Meanwhile, the imaginary permittivity (ε′′), or dielectric loss, shows the capacity of the dissipation of electromagnetic energy as heat. Through the complex permittivity, the dissipation factor (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ) and AC (σAC) conductivity can be calculated using the respective equations:73,74
δ) and AC (σAC) conductivity can be calculated using the respective equations:73,74
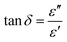 | (3) |
σAC = ωε0ε′tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ or σAC = ωε0ε′′ δ or σAC = ωε0ε′′ | (4) |
3.5. Dielectric property analysis
Fig. 7 depicts the electroconductive characteristics of a material throughout a frequency spectrum of 8–12 GHz, which is important for biomedical applications, including electromagnetic shielding, sensing, and tissue engineering.47,75 The dielectric constant (ε′) denotes a material's capacity to store electrical energy. The consistent results across the frequency spectrum indicate that the material has steady permittivity, which is essential for biomedical applications necessitating reliable electrical performance, such as bioelectronic interfaces.38,75 The IO-40PPy sample exhibits the highest performance, with ε′ and ε′′ peaks at 4.819 F m−1 and 0.3134 F m−1, respectively, indicating enhanced polarisation effects at optimal PPy coating concentrations. Dielectric loss (ε′′) denotes the energy expended as heat when a substance is subjected to an alternating electromagnetic field. The observed increases in ε′′ with higher PPy coating concentration are consistent with trends reported in previous studies, which attributed such behaviour to two complementary mechanisms: (i) enhanced dipolar polarisation due to PPy's conductive domains55,58,73 and (ii) strengthened interfacial polarisation from charge accumulation at IO-PPy boundaries.47,73,76 Our results further validate these mechanisms by demonstrating that the systematic increase in ε′′ with progressive PPy loading follows the same trend, thereby reinforcing the role of both dipolar and interfacial polarisation in governing the dielectric response of the IO-PPy system. This makes them advantageous for applications like electromagnetic shielding or regulated heating in biological therapies.77 Furthermore, the tangent loss (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δ), which quantifies the efficiency of energy dissipation to storage capacity, follows a similar increasing trend with PPy coating concentration, reflecting improved polarisation relaxation, which is advantageous for electromagnetic interference (EMI) shielding applications and bioelectronic implants requiring regulated conductivity.11,77
δ), which quantifies the efficiency of energy dissipation to storage capacity, follows a similar increasing trend with PPy coating concentration, reflecting improved polarisation relaxation, which is advantageous for electromagnetic interference (EMI) shielding applications and bioelectronic implants requiring regulated conductivity.11,77
The rise of AC conductivity exhibits frequency-dependent enhancement that scales with PPy coating concentration, demonstrating the formation of efficient charge transport pathways through PPy's hopping conduction mechanism.23 The 40 mM PPy concentration represents an optimal balance, where the extended conjugated polymer network facilitates charge delocalisation without inducing excessive aggregation. This improvement in charge transport can be attributed to the formation of a denser PPy shell around the IO core, creating an expanded interfacial region that supports effective charge separation and interfacial polarisation. The well-defined dipole alignment under an applied electric field promotes charge mobility and retention, optimising charge transport pathways. Beyond this threshold (50 mM PPy), we observed a 4.5% reduction in charge storage capacity (ε′ = 4.601 F m−1), accompanied by decreased heat dissipation (ε′′ = 0.2808 F m−1). This is likely due to excessive PPy deposition, which leads to uneven coating thickness and particle aggregation. Such structural inhomogeneity introduces charge trapping sites and increases scattering effects, impeding the continuous flow of charge carriers.64
As previously discussed in Fig. 6, the same aggregation-induced structural disorder had also contributed to a decline in DC conductivity, further reinforcing the impact of coating uniformity on charge transport efficiency. Notably, IO-40PPy exhibited the highest dissipation factor due to greater dielectric loss relative to its dielectric constant. This suggests that at 40 mM PPy, more energy is being lost as heat due to free charge movement or conduction loss rather than being stored as polarisation. This can stem from the optimized Maxwell–Wagner interfacial polarisation, where charge separation at the IO–PPy interface enhances dielectric response.78,79 This is prominently seen in heterogeneous composites such as IO-PPy, where charge deposition at the interfaces between insulating and conducting areas augments dielectric characteristics. The ionic polarisation also entails the migration of ions inside the substance, which is especially pertinent in polymer-based composites.58,75,77 These characteristics render the material appropriate for biomedical applications, including neural regeneration scaffolds and bioelectronic implants that could benefit from stable dielectric properties and controlled energy dissipation.11,77 Moreover, the material's ability to maintain synaptic transmission fidelity while minimizing signal attenuation makes it especially promising for spinal cord injury repair applications.39,80–82 Having established the influences of PPy coating concentration on both conductivity and dielectric behaviour, the next section evaluates its impact on magnetic properties using VSM and VNA-derived complex permeability.
3.6. Magnetic property analysis using VSM
The hysteresis loop for the bare and PPy-coated iron oxide particles was measured at room temperature under a maximum applied field of 14 kG, as shown in Fig. 8(a). The inset of Fig. 8(a) illustrates the relationship between the coercivity (Hc) and squareness ratio (Mr/Ms) with increasing PPy coating concentrations. As the PPy concentration increased from 0 mM to 50 mM, coercivity decreased from 130 Oe to 97 Oe, indicating reduced interparticle magnetic interactions. This trend demonstrates that the non-magnetic PPy coating effectively separates the IO particles, weakening direct magnetic coupling while maintaining the material's soft properties,50,67 as supported by the consistent squareness ratio (Mr/Ms ≈ 0.2) across all samples. The preservation of this soft magnetic character after surface modification is particularly advantageous for biomedical applications requiring reversible magnetisation, like drug delivery or MRI imaging.4The progressive decline in both saturation magnetisation (Ms) and remanent magnetisation (Mr) with increasing PPy concentration (Fig. 8b and c) reflects the fundamental change in material composition.66 However, unlike the simple dilution effect, the conformal PPy coating creates core–shell structures where each magnetic particle becomes surrounded by an increasingly thick non-magnetic layer.3,4,50,67 This architecture simultaneously replaces magnetically active material with diamagnetic polymer at the surface, maintains physical separation between cores to prevent magnetic coupling, and preserves the intrinsic soft magnetic properties of individual particles while reducing the net magnetic response per unit mass, as also similarly observed in previous studies.11,58,66,67 These modified magnetic properties offer significant benefits for biomedical applications, as the lower coercivity minimises the energy required for demagnetisation, while the reduced remanence decreases risks of particle aggregation and localised heating after field removal.3 These characteristics are especially valuable for magnetic drug delivery systems, where controlled actuation and minimal residual magnetisation are critical for safe interactions in biological environments.
To complement the DC magnetic properties obtained from VSM, the complex permeability was characterised using VNA to evaluate high-frequency magnetic behaviour, as shown in Fig. 9. The real permeability (μ′) represents the material's magnetic energy storage capacity, while the imaginary component (μ′′) quantifies energy dissipation through eddy currents and hysteresis losses. These measurements provide crucial insights into how the PPy coating influences the material's response to alternating magnetic fields, which is particularly relevant for specific applications such as magnetic hyperthermia or RF-based medical devices where high-frequency performance is essential.4 The combination of VSM and VNA analyses offers a comprehensive understanding of how PPy modification tailors both static and dynamic magnetic properties for biomedical applications.
3.7. Magnetic property analysis using VNA
The complex permeability analysis reveals significant concentration coating-dependent magnetic behaviour in the IO-PPy system. As shown in Fig. 9(a), the real permeability (μ′) exhibits a gradual decline from 1.18 H m−1 (bare IO) to 1.01 H m−1 (IO-50PPy) with increasing PPy concentration. This reduction in magnetic energy storage capacity (μ′) directly correlates with the decreasing saturation magnetisation observed in VSM measurements, confirming that the growing PPy coating thickness forms increasingly effective non-magnetic barriers around the IO cores. These conformal polymer layers weaken dipolar interactions between particles while reducing the net magnetic dipole density per unit volume, explaining the systematic μ′ reduction.50,67Interestingly, the magnetic loss characteristics (μ′′) show more complex behaviour, as evidenced in Fig. 9(b-c). At lower PPy concentrations (0–30 mM), both the μ′′ and magnetic loss tangent (tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δM) increase, reaching a maximum at 30 mM. This enhancement is potentially derived from the improved conductivity of the PPy coating, which promotes eddy current formation and associated resistive heating under alternating electromagnetic fields.50 However, beyond 30 mM, a dramatic reversal occurs where both μ′′ and tan
δM) increase, reaching a maximum at 30 mM. This enhancement is potentially derived from the improved conductivity of the PPy coating, which promotes eddy current formation and associated resistive heating under alternating electromagnetic fields.50 However, beyond 30 mM, a dramatic reversal occurs where both μ′′ and tan![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) δM values drop below those of bare IO particles. This unexpected transition suggests that the PPy coating reaches a critical thickness where it may begin functioning as an electromagnetic shield, attenuating field penetration to the magnetic core particles. The DC conductivity plateau observed at 40–50 mM PPy (Fig. 6) supports this interpretation, indicating that while conductivity remains elevated, excessive polymer deposition introduces charge-scattering centres that disrupt efficient eddy current pathways.
δM values drop below those of bare IO particles. This unexpected transition suggests that the PPy coating reaches a critical thickness where it may begin functioning as an electromagnetic shield, attenuating field penetration to the magnetic core particles. The DC conductivity plateau observed at 40–50 mM PPy (Fig. 6) supports this interpretation, indicating that while conductivity remains elevated, excessive polymer deposition introduces charge-scattering centres that disrupt efficient eddy current pathways.
Further, since magnetic losses are associated with the eddy current effect, these findings can be validated through the expression below:76,83
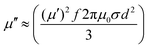 | (5) |
On the other hand, the term of 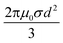 can be expressed as the eddy current coefficient, C0, which equals μ′′(μ′)−2f−1 as below:
can be expressed as the eddy current coefficient, C0, which equals μ′′(μ′)−2f−1 as below:
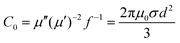 | (6) |
A key parameter in characterising eddy current dissipation is the eddy current coefficient (C0), which is typically assumed to be frequency-independent for conventional metallic and ferrite materials, where magnetic and dielectric losses remain relatively stable across a broad frequency range. However, in the present study, the observed frequency dependence of the C0 reveals a thickness-mediated transition in dissipation mechanisms, as mentioned in the previous studies.68,83
As shown in Fig. 9(d), at lower PPy concentrations (10–30 mM), enhanced C0 values reflect efficient eddy current formation facilitated by continuous conductive pathways in the thin PPy coating, where charge transport occurs primarily through polaron hopping.1 However, beyond 30 mM (inset Fig. 9d), the observed C0 reduction indicates a shielding-dominated regime, where excessive PPy thickness (>40 mM) introduces structural defects that disrupt charge percolation while simultaneously attenuating electromagnetic field penetration. This dual effect (morphological degradation and wave impedance mismatch) explains the non-monotonic behaviour. While moderate coatings optimize eddy current generation through interfacial polarisation and Joule heating, thicker coatings (>40 mM) act as lossy electromagnetic barriers, effectively decoupling the applied field from the magnetic IO cores. The frequency dependence further suggests competing low-frequency (hopping conduction) and high-frequency (dielectric relaxation) loss mechanisms, with the transition threshold dictated by PPy's critical percolation thickness. To conclude, the frequency-dependent eddy current coefficient (C0) shows that thinner PPy coatings (10–30 mM) enhance electromagnetic coupling, while thicker coatings (40–50 mM) induce shielding effects. This can be further explained through the electromagnetic interference (EMI) shielding effect in the next section.
3.8. Electromagnetic interference shielding effect
EMI shielding effectiveness reflects the ability of a material to attenuate electromagnetic interference through reflection (SER), absorption (SEA), and transmission (SET) mechanisms. Specifically, SER is associated with reflected energy at the material surface, SEA accounts for the internal energy dissipation within the material, and SET encompasses the total attenuation, defined as the sum of SER and SEA. Fig. 10(a–d) presents the shielding effectiveness by SER, SEA, and SET, and overall shielding efficiency (%) for bare IO and PPy-coated IO samples (IO-10PPy to IO-50PPy). These shielding components are quantitatively evaluated using S-parameters (S11 and S21) in response to incident signals using the equations below:46,50,58SER = −10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log(1 − |S11|2) log(1 − |S11|2) | (7) |
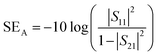 | (8) |
SET = −20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log|S21| log|S21| | (9) |
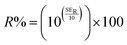 | (10) |
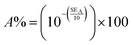 | (11) |
Hence, the percentage of electromagnetic waves blocked by the shielding material (SE%) can be expressed by:
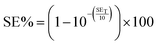 | (12) |
Through these measurements, the average SER value for bare IO is 0.44 dB, with incremental increases observed for the coated samples: 0.48 dB (IO-10PPy), 0.56 dB (IO-20PPy), and 0.59 dB (IO-30PPy), peaking at 0.97 dB (IO-40PPy), before slightly dropping to 0.86 dB (IO-50PPy). The average SET values, which quantify the total electromagnetic wave attenuation, also show positive correlation with PPy coating concentration: 0.61 dB (bare IO), 0.88 dB (IO-10PPy and IO-20PPy), 1.01 dB (IO-30PPy), and reaching 1.36 dB and 1.35 dB for IO-40PPy and IO-50PPy, respectively. Meanwhile, for SEA, a notable increase is observed with higher PPy content up to 30PPy; 0.17 dB (bare IO), rising to 0.40 dB (IO-10PPy), 0.42 dB (IO-20PPy), and 0.49 dB (IO-30PPy). However, a reduction is seen at higher concentrations, dropping to 0.39 dB (IO-40PPy) and 0.32 dB (IO-50PPy). This trend suggests that while PPy enhances both reflection and absorption initially, excessive coating may lead to saturation or morphological effects such as aggregation, reducing effective internal dissipation.15,58,72
To highlight, IO-40PPy achieves the highest total shielding effectiveness (SET = 1.36 dB) and shielding efficiency (SE% = 31.4%), primarily dominated by reflection (SER = 0.97 dB). In contrast, IO–30PPy exhibits the highest absorption contribution (SEA = 0.49 dB), with a more balanced interplay between reflection and absorption, leading to a moderate yet consistent SE% of 25.2%. Although the difference in shielding efficiency between the samples is relatively small (∼6%), distinct shielding mechanisms can be observed. These findings suggest that even with comparable overall efficiency, the dominant shielding mechanism can shift significantly depending on the particles’ coating thickness and composition. This distinction also indicates that a PPy coating at critical thickness (≥40 mM) can be strategically utilised in applications requiring high overall shielding effectiveness, such as in enclosures for electronic devices to prevent signal leakage.58,78 Conversely, lower PPy concentration (30 mM), with its higher absorption contribution, is more advantageous in environments where internal dissipation of electromagnetic waves is preferred to minimise secondary reflections, such as in aerospace or medical device shielding.1,4,84 The proposed electromagnetic wave interaction mechanisms are illustrated in Fig. 11.
 | ||
| Fig. 11 Illustration of proposed electromagnetic wave interaction mechanisms at various PPy coating concentrations (30 mM and 40 mM). | ||
While the shielding efficiency analysis highlighted the thickness-dependent electromagnetic behaviour of PPy coatings, these coating layers also play a crucial role in enhancing electrochemical stability. To further explore this function, the next section presents cyclic voltammetry (CV) results obtained under acidic (pH 5.8) and neutral (pH 7) conditions, aimed at evaluating the coatings’ reactive oxygen species (ROS) scavenging capacity, which is a critical property for neural interfaces, as oxidative stress dictates device longevity.
3.9. Electrochemical behaviour at various pH values
Cyclic voltammetry was used to evaluate the influence of the PPy coating on IO's electrochemical behaviour at a linear potential sweep rate of 50 mV s−1, between −1.0 and 1.0 V vs. Ag/AgCl during 5 cycles at pH 7.4 and pH 5.8. This study was conducted under neutral and acidic conditions to mimic physiological environments, which can be applied to normal tissue conditions and inflammed sites, such as injury regions.85 While tissue acidity in injury sites primarily results from lactic acid accumulation due to anaerobic metabolism,86 sulfuric acid was used to adjust the pH in this study. Although not exactly replicating the biological environment, the sulfuric acid-based electrolyte allows for precise pH control, providing a consistent experimental setup.87As shown in Fig. 12(I), at pH 7.4, the oxidation of bare IO occurred at 0.21 mA. With increasing PPy coating concentration from 10 mM, 20 mM, and 30 mM, the oxidation current exhibited a notable rise to 0.40, 0.59, and 0.62 mA, respectively. This trend suggests that the PPy coating enhances electron transfer, likely due to its intrinsic redox activity and interaction with the IO core surface. Additionally, since PPy was doped with APS, the anion exchange process between sulphate (SO42−) dopant ions and phosphate (PO43−) ions from the PBS electrolyte may also contribute to improved charge transport. This ion exchange modulates the doping level of PPy, enhancing its ionic and electrical conductivity, which in turn facilitates higher oxidation currents at 10 mM, 20 mM, and 30 mM PPy coating concentrations.
However, at 40 mM and 50 mM of PPy, no oxidation peaks can be observed. This absence is likely due to the formation of a thicker and denser coating layer acting as an insulating barrier rather than a conductive pathway. The iron oxide core surface can be fully encapsulated at this point, preventing direct electron transfer to the electrolyte, thereby explaining the absence of the oxidation peaks. Furthermore, at these coating concentrations (40 mM and 50 mM), the voltammetry scans predominantly capture the redox activity of PPy itself rather than the IO core. This suggests that PPy functions as a buffer oxidation layer, effectively shielding the IO core from oxidation. Thus, while PPy coatings with lower concentrations (≤30 mM) enhance electron transfer and oxidation current, those with higher concentrations (≥40 mM) not only enhance conductivity (as supported by the previous 4-point probe measurements) but also act as effective shielding layers against the IO core oxidation. The interplay between PPy redox activity and electrolyte interactions further stabilizes the system, making it a promising strategy for improving electrochemical stability and long-term performance in a physiological environment (pH 7.4). Subsequently, the presence of dopants like APS in PPy may contribute to further acidification in the local environment due to interactions with H+ ions, providing insight into the electrochemical stability of the material under acidic conditions over time.
Interestingly, under lower pH conditions, as shown in Fig. 12(II), no oxidation peaks were observed in any of the samples, suggesting suppressed oxidation kinetics in the low pH environment. However, the bare IO sample exhibited two distinct reduction peaks, indicating sequential redox transitions. The first reduction peak at +0.68 V likely corresponds to the reduction of (i) Fe3+ to Fe2+. Meanwhile, the second reduction peak at −0.56V is attributed to (ii) Fe2+ undergoing further reduction to form partially metallic (Fe0). This indicates that under acidic conditions, it enhances Fe ion solubility and thermodynamic tendencies for reduction. Notably, these reactions could pose cytotoxicity risks, as Fe may act as a Fenton-like catalyst, promoting the formation of reactive oxygen species (ROS), such as hydroxyl radicals, posing potential cytotoxic effects in biological systems. These highly reactive ROS can induce oxidative stress, leading to cell membrane damage and triggering inflammatory responses, as expected from mechanisms proposed in the literature.8,13 The reduction reactions observed can be summarised as follows:
| Fe3+ + e− → Fe2+ (+0.68 V, high reduction potential) | (i) |
| Fe2+ + 2e− → Fe (−0.56 V, low reduction potential) | (ii) |
On the other hand, the potential of Fe to act as a Fenton-like catalyst at low pH, forming ROS, can be summarised as:
| Fe + O2 + H2O → Fe2+ + H2O2 | (iii) |
| Fe2+ + H2O2 → Fe3+ + OH− + ˙OH | (iv) |
However, in a physiological environment, full reduction to metallic iron (Fe) is unlikely, particularly in the presence of oxygen. This is due to the relatively low electrochemical potential required for the reduction, which makes the complete reduction of iron oxide to its metallic form less favourable.13 Nonetheless, the dissolution and release of Fe2+ and Fe3+ ions under acidic conditions could still contribute to oxidative stress and cytotoxicity, primarily through ROS formation, even without full reduction to metallic Fe.8
On the other hand, after surface coating with PPy, only one reduction peak was detected at a much lower potential range of −0.75 to −0.81 V. This suggests that the reduction of Fe3+ to Fe2+, which previously occurred at +0.68 V, is either suppressed or shifted to a significantly lower potential. This shift may be due to the PPy coating acting as a physical and electrochemical barrier, restricting electron transfer and trapping Fe3+ within the PPy layer, thereby limiting its direct participation in electrochemical reduction. Consequently, a much lower potential is required for the reduction to occur. This indicates that the PPy coating acts as a more effective barrier to electron transfer in a lower pH environment.
Moreover, the possibility of ion exchange between the pre-existing sulphate dopants and newly introduced H+ ions in the acidic environment may further modify the redox behaviour of PPy. If H+ ions replace some of the sulphate dopant ions from the PPy backbone, it could alter the local charge distribution and electrostatic interactions within the polymer network, indirectly affecting the redox potential of Fe3+. However, since the CV data do not show any additional reduction peak corresponding to PPy, this suggests that its primary role at pH 5.8 is to enhance electron transport rather than undergo significant redox transitions. Therefore, the observed shift in Fe3+ to a much lower reduction potential is likely a combined effect of the PPy barrier properties and the modified electrochemical environment induced by protonation. As the PPy concentration increases, its influence on electron transfer becomes more pronounced. At 30 mM and 40 mM PPy, the reduction peaks broaden, suggesting multiple overlapping reduction processes, likely due to variations in local charge distribution and enhanced protonation effects at pH 5.8. At 50 mM PPy, a new reduction peak emerges at −0.45 V, indicating a possible shift in Fe3+ reduction dynamics. This could be attributed to the increment of PPy redox activity, where protonation-induced charge delocalisation facilitates an alternative reduction pathway. These combined effects result in a more complex reduction behaviour than simply suppressing Fe3+ reduction. The proposed mechanism of possible electrochemical interactions of the PPy layer at different pH levels is summarised in Fig. 13.
 | ||
| Fig. 13 Proposed mechanism of ROS scavenging behaviour of the PPy coating at pH 7.4 and pH 5.8 according to the voltammetry curve analysis. | ||
3.10. Cell viability
Cell viability and cytotoxicity assessments are critical benchmarks in evaluating the biocompatibility of biomaterials for tissue engineering applications. In this study, we evaluated the biocompatibility of IO-PPy particles using the Trypan blue exclusion assay, which differentiates viable (membrane-intact) from non-viable (membrane-compromised) cells. Human fetal osteoblast cells (HFOB) were exposed to IO-PPy concentrations ranging from 2% to 10% w/v (100–500 mg mL−1) over 1, 3, and 5 days (Fig. 14).The applied concentration range is at least 2 orders of magnitude higher than that typically applied in iron oxide nanoparticle cytotoxicity studies (≤500 μg mL−1),88–91 which is driven by the requirement to impart sufficient ferromagnetic responsiveness when used as a hydrogel filler. Notably, at 100 mg mL−1 (2% w/v), the IO-PPy will constitute approximately 10% of the hydrogel's total mass, a threshold necessary to generate coercive fields exceeding 10 mT for effective magnetic alignment,43,45,92 an essential feature for promoting axonal guidance in neural regeneration.
As shown in Fig. 14, cell viability was observed to have a dose-dependent behaviour, where it decreases with increasing IO-PPy concentrations across all time points (1,3, and 5 days). Concentrations below 2% w/v (100 mg mL−1) showed consistent viability above 80%, suggesting acceptable cytocompatibility. Two-way ANOVA revealed statistically significant effects of both exposure duration (p = 8.12 × 10−12) and IO-PPy concentration (p = 1.97 × 10−20), along with a significant interaction between the two variables, which highlights the compounded influence of dose and time on cellular response.
Interestingly, although PPy is widely recognised for its electrochemical stability and potential to mitigate oxidative stress, the observed dose-dependent cytotoxicity suggests that its protective effects may be limited at higher IO-PPy concentrations. While CV data confirm enhanced redox activity in PPy-coated particles, this enhancement does not appear to translate into proportional biological compatibility at elevated doses. This discrepancy likely arises because the cytotoxic effects observed at ≥2% w/v are not purely chemical (i.e., ROS-mediated), but also physical. At such high filler concentrations, factors like particle aggregation, sedimentation, and mechanical crowding may dominate the cellular response,12,13,93 overwhelming any mitigating role of the PPy layer. Thus, the results do not negate PPy's functional contribution, but rather highlight the complexity of microparticle-cell interactions at high loading levels. These findings reinforce the need to optimize IO-PPy dosage not limited for ferromagnetic performance but also to maintain a biologically permissive environment within the hydrogel.
Moreover, it is important to note the limitations of the Trypan blue exclusion assay as well. While it offers a quick and accessible measure of membrane integrity, it does not capture sublethal cellular stress, such as mitochondrial dysfunction or oxidative damage.94,95 Therefore, future studies will incorporate metabolic (MTT), membrane damage (LDH), and apoptosis-specific assays (Annexin V/PI flow cytometry) to yield a more nuanced understanding of IO-PPy's cytological impact. Ultimately, the high concentrations of IO-PPy in this study accommodate a functionality-driven design, which prioritizes magneto-electrical performance while maintaining biocompatibility within an acceptable range. These insights are critical for advancing the development of multifunctional hydrogel scaffolds in tissue engineering, where both structural and directional cues are essential for regenerative success.
4. Conclusions
This study successfully demonstrates the dual functionality of PPy coatings in enhancing the oxidative stability and electromagnetic performance of iron oxide particles, suited for biomedical applications. By employing a facile micellar polymerisation method using SDS as a soft template, we achieved a uniform and tunable PPy coating on the dry IO surface (150–210 nm) and a hydrodynamic size of 150–800 nm. The coating thickness, controlled by varying PPy concentrations (10–50 mM), was systematically characterised using FESEM, DLS, FTIR, and XRD, confirming successful surface modification.Our key finding was the concentration-dependent enhancement in conductivity, peaking at 456 μS cm−1 (99% increase over bare IO) at 40 mM PPy, as measured by four-point probe analysis. Despite an ∼25% reduction in saturation magnetisation (between 40 mM PPy and bare IO) due to thicker coatings, the PPy layer significantly improved electromagnetic shielding effectiveness, with SET = 1.36 dB (31.4% shielding efficiency at 40 mM), primarily through reflection mechanisms (SER = 0.97 dB). This finding suggests that PPy-coated IO particles can retain sufficient magnetic responsiveness while gaining improved electromagnetic coupling, which is crucial for applications like hyperthermia and magnetic manipulation.
Another significant finding from CV analysis revealed that PPy coatings can effectively suppress oxidative degradation of the IO core, particularly at higher concentrations (≥30 mM), where oxidation currents were entirely inhibited; hence, the amphoteric surface behaviour of iron oxide was suppressed. Under acidic conditions (simulating injury states), bare IO exhibited cytotoxic two-step reduction behaviour linked to Fenton-like reactions, whereas PPy-coated samples mitigated these effects, highlighting their protective role. Cytocompatibility assays demonstrated that the 40 mM PPy coating is non-toxic, with cell viability dictated by the particle concentration. While uncoated IO particles are typically tested at lower doses ≤500 μg mL−1, PPy-coated particles retained >80% viability even at 2% (w/v) (100 mg mL−1), underscoring their enhanced biocompatibility.
By correlating electromagnetic performance with redox stability, this work provides novel insights into the dual-functionality of PPy coatings, which enhances electromagnetic properties while offering oxidative degradation protection in physiologically relevant environments. Future works should focus on integrating these PPy-coated IO particles into hydrogel systems to evaluate their functional behaviour in 3D environments, while exploring intracellular oxidation dynamics, dynamic structural responses to stimuli, and tissue-specific responses.
Author contributions
The original draft of this publication was conceptualised, synthesised, characterised, and written in part by Shahemi Nur Hidayah. Kasri Mohd Arif partly helped with cyclic voltammetry experimentation. Dr Ab Rahim Sharaniza and Prof. Amir Muhammad Abid helped in supervising, validating, and formal analysis related to cell work activities, while Dr Sikiru Surajudeen and Dr Mahat Mohd Muzamir helped with the final reviewing, supervising, editing, and formal analysis.Conflicts of interest
There are no conflicts to declareData availability
All essential data supporting the findings of this study are included in the manuscript. Any additional data not included in the manuscript will be made available by the corresponding author upon reasonable request.Acknowledgements
The authors gratefully acknowledge Dr Reusmaazran bin Yusof from the Malaysia Nuclear Agency for his assistance in sterilizing the samples via gamma irradiation for cell viability studies. The research studies were not supported by any grants.References
- R. V. Stigliano, F. Shubitidze, J. D. Petryk, L. Shoshiashvili, A. A. Petryk and P. J. Hoopes, Mitigation of eddy current heating during magnetic nanoparticle hyperthermia therapy, Int. J. Hyperthermia, 2016, 32(7), 735–748 CrossRef PubMed.
- K. Y. Qian, Y. Song, X. Yan, L. Dong, J. Xue and Y. Xu, et al., Injectable ferrimagnetic silk fibroin hydrogel for magnetic hyperthermia ablation of deep tumor, Biomaterials, 2020, 259, 120299 Search PubMed.
- J. Santos, J. C. Silva, M. A. Valente, T. Vieira and P. I. P. Soares, Enhancing magnetic hyperthermia: Investigating iron oxide nanoparticle coating and stability, Next Nanotechnol., 2025, 7, 100141 Search PubMed.
- C. Martins, C. Rolo, V. R. G. Cacho, L. C. J. Pereira, J. P. Borges and J. C. Silva, et al., Enhancing the magnetic properties of superparamagnetic iron oxide nanoparticles using hydrothermal treatment for magnetic hyperthermia application, Mater. Adv., 2025, 6(5), 1726–1743 RSC.
- C. Pucci, A. Degl’Innocenti, M. Belenli Gümüş and G. Ciofani, Superparamagnetic iron oxide nanoparticles for magnetic hyperthermia: recent advancements, molecular effects, and future directions in the omics era, Biomater. Sci., 2022, 10(9), 2103–2121 Search PubMed.
- S. Sindhwani, A. M. Syed, J. Ngai, B. R. Kingston, L. Maiorino and J. Rothschild, et al., The entry of nanoparticles into solid tumours, Nat. Mater., 2020, 19(5), 566–575 Search PubMed.
- C. Naud, C. Thébault, M. Carrière, Y. Hou, R. Morel and F. Berger, et al., Cancer treatment by magneto-mechanical effect of particles, a review, Nanoscale Adv., 2020, 2(9), 3632–3655 RSC.
- M. A. Siddiqui, R. Wahab, Q. Saquib, J. Ahmad, N. N. Farshori and E. S. Al-Sheddi, et al., Iron oxide nanoparticles induced cytotoxicity, oxidative stress, cell cycle arrest, and DNA damage in human umbilical vein endothelial cells, J. Trace Elem. Med. Biol., 2023, 80, 127302 CrossRef PubMed.
- J. E. Ogbezode, U. S. Ezealigo, A. Bello, V. C. Anye and A. P. Onwualu, A narrative review of the synthesis, characterization, and applications of iron oxide nanoparticles, Discover Nano, 2023, 18(1), 125 Search PubMed.
- S. J. H. Soenen, U. Himmelreich, N. Nuytten and M. De Cuyper, Cytotoxic effects of iron oxide nanoparticles and implications for safety in cell labelling, Biomaterials, 2011, 32(1), 195–205 CrossRef PubMed.
- L. E. Low, H. P. Lim, Y. S. Ong, S. P. Siva, C. S. Sia and B. H. Goh, et al., Stimuli-controllable iron oxide nanoparticle assemblies: Design, manipulation and bio-applications, J. Controlled Release, 2022, 345, 231–274 CrossRef PubMed.
- M. G. Montiel Schneider, M. J. Martín, J. Otarola, E. Vakarelska, V. Simeonov and V. Lassalle, et al., Biomedical Applications of Iron Oxide Nanoparticles: Current Insights Progress and Perspectives, Pharmaceutics, 2022, 14(1), 204 Search PubMed.
- A. Kessler, J. Hedberg, E. Blomberg and I. Odnevall, Reactive Oxygen Species Formed by Metal and Metal Oxide Nanoparticles in Physiological Media—A Review of Reactions of Importance to Nanotoxicity and Proposal for Categorization, Nanomaterials, 2022, 12(11), 1922 CrossRef PubMed.
- Q. Yang, J. Zhao, A. Muhammad, L. Tian, Y. Liu and L. Chen, et al., Biopolymer coating for particle surface engineering and their biomedical applications, Mater. Today Bio, 2022, 16, 100407 Search PubMed.
- R. Wirecka, M. M. Marzec, M. Marciszko-Wiąckowska, M. Lis, M. Gajewska and E. Trynkiewicz, et al., The effect of shell modification in iron oxide nanoparticles on electrical conductivity in polythiophene-based nanocomposites, J. Mater. Chem. C, 2021, 9(32), 10453–10461 RSC.
- H. Sharifi Dehsari, A. Halda Ribeiro, B. Ersöz, W. Tremel, G. Jakob and K. Asadi, Effect of precursor concentration on size evolution of iron oxide nanoparticles, CrystEngComm, 2017, 19(44), 6694–6702 RSC.
- C. Wu, Z. Pei, M. Lv, D. Huang, Y. Wang and S. Yuan, Polypyrrole-Coated Low-Crystallinity Iron Oxide Grown on Carbon Cloth Enabling Enhanced Electrochemical Supercapacitor Performance, Molecules, 2023, 28(1), 434 Search PubMed.
- T. H. Le, Y. Kim and H. Yoon, Electrical and Electrochemical Properties of Conducting Polymers, Polymers, 2017, 9(12), 150 CrossRef PubMed.
- S. A. A. Shah, M. Firlak, S. R. Berrow, N. R. Halcovitch, S. J. Baldock and B. M. Yousafzai, et al., Electrochemically enhanced drug delivery using polypyrrole films, Materials, 2018, 11(7), 1123 Search PubMed.
- B. Mol, A. E. Beeran, P. S. Jayaram, P. Prakash, R. S. Jayasree and S. Thomas, et al., Radio frequency plasma assisted surface modification of Fe3O4 nanoparticles using polyaniline/polypyrrole for bioimaging and magnetic hyperthermia applications, J. Mater. Sci.: Mater. Med., 2021, 32(9), 108 CAS.
- H. Li, H. Wen, H. Zhang, J. Li, X. Cao and J. Zhang, et al., Polymeric micelle-hydrogel composites design for biomedical applications, Chin. Chem. Lett., 2025, 36(5), 110072 CAS.
- D. Uppalapati, M. Sharma, Z. Aqrawe, F. Coutinho, I. D. Rupenthal and B. J. Boyd, et al., Micelle directed chemical polymerization of polypyrrole particles for the electrically triggered release of dexamethasone base and dexamethasone phosphate, Int. J. Pharm., 2018, 543(1–2), 38–45 CrossRef CAS.
- P. Kongsat, E. A. O’Rear and T. Pongprayoon, Conductivity improvement of magnetite and hematite nanoparticles via admicellar polymerization of polypyrrole, J. Ind. Eng. Chem., 2022, 113, 540–552 CrossRef CAS.
- S. K. Fanourakis, S. Q. Barroga, J. V. D. Perez, L. He and D. F. Rodrigues, In Situ Polymerization of Polypyrrole and Polyaniline on the Surface of Magnetic Molybdenum Trioxide Nanoparticles: Implications for Water Treatment, ACS Appl. Nano Mater., 2021, 4(11), 12415–12428 CrossRef CAS.
- K. J. Ahn, Y. Lee, H. Choi, M. S. Kim, K. Im and S. Noh, et al., Surfactant-Templated Synthesis of Polypyrrole Nanocages as Redox Mediators for Efficient Energy Storage, Sci. Rep., 2015, 5(1), 14097 CrossRef CAS PubMed.
- K. H. Chan, E. T. Wong, M. Irfan, A. Idris and N. M. Yusof, Enhanced Cu(II) rejection and fouling reduction through fabrication of PEG-PES nanocomposite ultrafiltration membrane with PEG-coated cobalt doped iron oxide nanoparticle, J. Taiwan Inst. Chem. Eng., 2015, 47, 50–58 CAS.
- A. Lazaro-Carrillo, M. Filice, M. J. Guillén, R. Amaro, M. Viñambres and A. Tabero, et al., Tailor-made PEG coated iron oxide nanoparticles as contrast agents for long lasting magnetic resonance molecular imaging of solid cancers, Mater. Sci. Eng., C, 2020, 107, 110262 CrossRef CAS PubMed.
- O. Karaagac and H. Köçkar, Improvement of the saturation magnetization of PEG coated superparamagnetic iron oxide nanoparticles, J. Magn. Magn. Mater., 2022, 551, 169140 CrossRef CAS.
- S. Kayal and R. V. Ramanujan, Doxorubicin loaded PVA coated iron oxide nanoparticles for targeted drug delivery, Mater. Sci. Eng., C, 2010, 30(3), 484–490 CrossRef CAS.
- J. Lu, M. Wu, L. Luo, R. Lu, J. Zhu and Y. Li, et al., Incorporating iron oxide nanoparticles in polyvinyl alcohol/starch hydrogel membrane with biochar for enhanced slow-release properties of compound fertilizers, Carbohydr. Polym., 2025, 348, 122834 CrossRef CAS.
- A. Al Rashid, N. Al-Maslamani, A. Abutaha, M. Hossain and M. Koç, Cobalt iron oxide (CoFe2O4) reinforced polyvinyl alcohol (PVA) based magnetoactive polymer nanocomposites for remote actuation, Mater. Sci. Eng., B, 2025, 311, 117838 CrossRef CAS.
- S. Sadeghi, A. Esmaeili and A. Zarrabi, Dextran-coated iron oxide nanoparticles in combination with ginger extract without NGF promote neurite outgrowth and PC12 cell branching, Environ. Res., 2023, 232, 116302 CAS.
- I. Rosca, I. A. Turin-Moleavin, A. Sarghi, A. L. Lungoci, C. D. Varganici and A. R. Petrovici, et al., Dextran coated iron oxide nanoparticles loaded with protocatechuic acid as multifunctional therapeutic agents, Int. J. Biol. Macromol., 2024, 256, 128314 CAS.
- H. M. Meena, S. Kukreti and P. S. Jassal, Synthesis, and characterization of novel chitosan coated superparamagnetic iron oxide nanoparticles to optimization for adsorption of mercury from industrial effluent wastewater, Int. J. Biol. Macromol., 2025, 306, 141403 CAS.
- U. Zaman, K. Rehman, F. Zouidi, I. Zghab, A. K. Hajri and M. Alissa, et al., Immobilization of phytase on chitosan-coated iron oxide nanoparticles (Fe3O4 NPs) and its application for the reduction of soymilk phytic acid, J. Ind. Eng. Chem., 2025, 147, 545–554 CAS.
- S. Jadoun, U. Riaz and V. Budhiraja, Biodegradable conducting polymeric materials for biomedical applications: a review, Med. Dev. Sens., 2021, 4(1), e10141 CrossRef CAS.
- K. Namsheer and C. S. Rout, Conducting polymers: a comprehensive review on recent advances in synthesis, properties and applications, RSC Adv., 2021, 11(10), 5659–5697 Search PubMed.
- R. A. Nasser, S. S. Arya, K. H. Alshehhi, J. C. M. Teo and C. Pitsalidis, Conducting polymer scaffolds: a new frontier in bioelectronics and bioengineering, Trends Biotechnol., 2024, 42(6), 760–779 CAS.
- Y. Liang and J. C. H. Goh, Polypyrrole-Incorporated Conducting Constructs for Tissue Engineering Applications: A Review, Bioelectricity, 2020, 2(2), 101–119 CrossRef.
- R. Gholipur, Polypyrrole-based NanoComposite with excellent absorption properties by facile approach, Opt. Mater., 2021, 112, 110479 CrossRef.
- A. Manzari-Tavakoli, R. Tarasi, R. Sedghi, A. Moghimi and H. Niknejad, Fabrication of nanochitosan incorporated polypyrrole/alginate conducting scaffold for neural tissue engineering, Sci. Rep., 2020, 10, 22012 CrossRef CAS PubMed.
- A. Orlowska, P. T. Perera, M. A. Al Kobaisi, A. Dias, H. K. D. Nguyen and S. Ghanaati, et al., The effect of coatings and nerve growth factor on attachment and differentiation of Pheochromocytoma Cells, Materials, 2017, 11(1), 60 CrossRef PubMed.
- J. L. Funnell, B. Balouch and R. J. Gilbert, Magnetic Composite Biomaterials for Neural Regeneration, Front. Bioeng. Biotechnol., 2019, 7, 179 CrossRef.
- K. I. Ritzau-Reid, C. D. Spicer, A. Gelmi, C. L. Grigsby, J. F. Ponder and V. Bemmer, et al., An Electroactive Oligo-EDOT Platform for Neural Tissue Engineering, Adv. Funct. Mater., 2020, 30(42), 2003710 CrossRef CAS.
- J. P. M. Sousa, C. F. Monteiro, I. A. Deus, A. Completo, E. Stratakis and J. F. Mano, et al., Magnetoresponsive Anisotropic Fiber-Integrating Hydrogels for Neural Tissue Regeneration, Small Struct., 2024, 5(11), 2400213 CrossRef CAS.
- H. Kaftelen-Odabaşı, F. Ruiz-Perez, A. Odabaşı, S. Helhel, S. M. López-Estrada and F. Caballero-Briones, EMI-shielding response of GO/Fe3O4/polypyrrole(PPy)/thermoplastic polyurethane (TPU) composites, Eng., Sci. Technol., 2024, 55, 101753 Search PubMed.
- R. Gahlawat and R. Shukla, Enhanced EMI shielding performance of CoFe2-xDyxO4/polypyrrole nanocomposites in Ku-band, Surf. Interfaces, 2025, 56, 105629 CrossRef CAS.
- E. Tarani, I. Arvanitidis, D. Christofilos, D. N. Bikiaris, K. Chrissafis and G. Vourlias, Calculation of the degree of crystallinity of HDPE/GNPs nanocomposites by using various experimental techniques: a comparative study, J. Mater. Sci., 2023, 58(4), 1621–1639 CrossRef CAS.
- N. Guo, F. Cang, Z. Wang, T. T. Zhao, X. R. Song and S. Farris, et al., Magnetism and NIR dual-response polypyrrole-coated Fe3O4 nanoparticles for bacteria removal and inactivation, Mater. Sci. Eng., C, 2021, 126, 112143 CrossRef CAS PubMed.
- N. Janem, Z. S. Azizi and M. M. Tehranchi, Microwave absorption and magnetic properties of thin-film Fe3O4 @polypyrrole nanocomposites: The synthesis method effect, Synth. Met., 2021, 282, 116948 CrossRef.
- L. K. Dhugga, H. B. Baskey, K. K. Gaur and D. P. Singh, Absorption dominant electromagnetic interference shielding in the novel polypyrrole-CaCu3Ti4O12-CoFe2O4 nanocomposites, Compos. Sci. Technol., 2023, 240, 110067 CrossRef.
- S. Z. Yow, T. H. Lim, E. K. F. Yim, C. T. Lim and K. W. Leong, A 3D Electroactive Polypyrrole-Collagen Fibrous Scaffold for Tissue Engineering, Polymers, 2011, 3(1), 527–544 CrossRef.
- B. Alawa, A. Srivastava, J. K. Srivstava and J. Palsania, Adsorption of Heavy Metal Pb (II) from Synthetic Waste Water by Polypyrrole composites. ∼ 4 ∼, Int. J. Chem. Stud., 2015, 3(1), 04–08 Search PubMed.
- S. Krawczyk, S. Golba, C. Neves and J. Tedim, Chlorpromazine–Polypyrrole Drug Delivery System Tailored for Neurological Application, Molecules, 2024, 29(7), 1531 Search PubMed.
- T. Bashir, A. Shakoor, E. Ahmed, N. A. Niaz, M. S. Akhtar and M. R. Raza, et al., Polypyrrole-Fe2O3 Nanocomposites with High Dielectric Constant: In Situ Chemical Polymerisation, Polym. Polym. Compos., 2018, 26(3), 233–241 Search PubMed.
- S. Wang, J. Lei, X. Yi, L. Yuan, L. Ge and D. Li, et al., Fabrication of Polypyrrole-Grafted Gelatin-Based Hydrogel with Conductive, Self-Healing, and Injectable Properties, ACS Appl. Polym. Mater., 2020, 2(7), 3016–3023 CrossRef.
- C. Boyer, M. R. Whittaker, V. Bulmus, J. Liu and T. P. Davis, The design and utility of polymer-stabilized iron-oxide nanoparticles for nanomedicine applications, NPG Asia Mater., 2010, 2(1), 23–30 Search PubMed.
- R. Kanwal, M. F. Maqsood, M. A. Raza, A. Inam, M. Waris and Z. U. Rehman, et al., Polypyrrole coated carbon fiber/magnetite/graphene oxide reinforced hybrid epoxy composites for high strength and electromagnetic interference shielding, Mater. Today Commun., 2024, 38, 107684 CrossRef.
- G. Rischitor, M. Parracino, R. La Spina, P. Urbán, I. Ojea-Jiménez and E. Bellido, et al., Quantification of the cellular dose and characterization of nanoparticle transport during in vitro testing, Part. Fibre Toxicol., 2015, 13(1), 47 CrossRef PubMed.
- Z. Varga, B. Fehér, D. Kitka, A. Wacha, A. Bóta and S. Berényi, et al., Size Measurement of Extracellular Vesicles and Synthetic Liposomes: The Impact of the Hydration Shell and the Protein Corona, Colloids Surf., B, 2020, 192, 111053 Search PubMed.
- X. Wen, Y. Su, G. Liu, S. Li, A. J. Müller and S. K. Kumar, et al., Direct Relationship between Dispersion and Crystallization Behavior in Poly(ethylene oxide)/Poly(ethylene glycol)- g -Silica Nanocomposites, Macromolecules, 2021, 54(4), 1870–1880 CrossRef.
- S. K. Filippov, R. Khusnutdinov, A. Murmiliuk, W. Inam, L. Y. Zakharova and H. Zhang, et al., Dynamic light scattering and transmission electron microscopy in drug delivery: a roadmap for correct characterization of nanoparticles and interpretation of results, Mater. Horiz., 2023, 10(12), 5354–5370 RSC.
- N. Velhal, N. D. Patil, G. Kulkarni, S. K. Shinde, N. J. Valekar and H. C. Barshilia, et al., Electromagnetic shielding, magnetic and microwave absorbing properties of Polypyrrole/Ba0.6Sr0.4Fe12O19 composite synthesized via in-situ polymerization technique, J. Alloys Compd., 2019, 777, 627–637 CrossRef CAS.
- H. Ali and A. M. Ismail, Fabrication of Magnetic Fe3O4/Polypyrrole/Carbon Black Nanocomposite for Effective Uptake of Congo Red and Methylene Blue Dye: Adsorption Investigation and Mechanism, J. Polym. Environ., 2023, 31(3), 976–998 CrossRef CAS.
- K. Ahmed, F. Kanwal, S. Ramay, S. Atiq, R. Rehman and S. Ali, et al., Synthesis and Characterization of BaTiO3/Polypyrrole Composites with Exceptional Dielectric Behaviour, Polymers, 2018, 10(11), 1273 CrossRef PubMed.
- T. Köhler, A. Feoktystov, O. Petracic, E. Kentzinger, T. Bhatnagar-Schöffmann and M. Feygenson, et al., Mechanism of magnetization reduction in iron oxide nanoparticles, Nanoscale, 2021, 13(14), 6965–6976 RSC.
- M. Abdolrahimi, M. Vasilakaki, S. Slimani, N. Ntallis, G. Varvaro and S. Laureti, et al., Magnetism of Nanoparticles: Effect of the Organic Coating, Nanomaterials, 2021, 11(7), 1787 CrossRef CAS PubMed.
- S. Gupta, U. Acharya, H. Pištěková, O. Taboubi, Z. Morávková and M. Kašparová, et al., Tuning the Conductivity, Morphology, and Capacitance with Enhanced Antibacterial Properties of Polypyrrole by Acriflavine Hydrochloride, ACS Appl. Polym. Mater., 2021, 3(12), 6063–6069 CAS.
- A. Dianatdar, M. Miola, O. De Luca, P. Rudolf, F. Picchioni and R. K. All-dry Bose, one-step synthesis, doping and film formation of conductive polypyrrole, J. Mater. Chem. C, 2022, 10(2), 557–570 RSC.
- L. Mahelová, P. Slobodian, K. Kocourková, A. Minařík, R. Moučka and M. Trchová, et al., Method for in situ polypyrrole coating, and the example of its use for functionalization of polyurethane anisotropic electrospun mats, Heliyon, 2024, 10(6), e27883 CrossRef PubMed.
- S. A. Fraser and W. E. van Zyl, In situ polymerization and electrical conductivity of polypyrrole/cellulose nanocomposites using Schweizer's reagent, RSC Adv., 2022, 12(34), 22031–22043 RSC.
- M. A. Ashraf, W. Peng, Y. Zare and K. Y. Rhee, Effects of Size and Aggregation/Agglomeration of Nanoparticles on the Interfacial/Interphase Properties and Tensile Strength of Polymer Nanocomposites, Nanoscale Res. Lett., 2018, 13, 214 CrossRef.
- U. Anwar, M. Rafi, N. A. Noor, S. Nazir, S. Mumtaz and I. M. Moussa, Innovative multi-layered Fe 3 O 4 -Gr/carbon/polypyrrole nanofiber composite: a new frontier in dielectric enhancement and EMI shielding, RSC Adv., 2024, 14(50), 37483–37497 RSC.
- X. Xie, Z. Z. He, X. D. Qi, J. H. Yang, Y. Z. Lei and Y. Wang, Achieving high performance poly(vinylidene fluoride) dielectric composites via in situ polymerization of polypyrrole nanoparticles on hydroxylated BaTiO 3 particles, Chem. Sci., 2019, 10(35), 8224–8235 RSC.
- D. D. L. Chung, Materials for electromagnetic interference shielding, Mater. Chem. Phys., 2020, 255, 123587 CrossRef CAS.
- N. A. A. Apandi, N. B. Ibrahim, Z. I. Khan and R. S. Azis, Microwave absorption properties of (1 − x)La 0.85 Ag 0.15 MnO3/x graphene (x = 0, 3, and 5 wt%) composites, Int. J. Appl. Ceram. Technol., 2023, 20(5), 3148–3162 CrossRef CAS.
- R. P. Gambhir, S. S. Rohiwal and A. P. Tiwari, Multifunctional surface functionalized magnetic iron oxide nanoparticles for biomedical applications: A review, Appl. Surf. Sci. Adv., 2022, 11, 100303 CrossRef.
- H. V. Hussain, M. Ahmad, M. T. Ansar, G. M. Mustafa, S. Ishaq and S. Naseem, et al., Polymer based nickel ferrite as dielectric composite for energy storage applications, Synth. Met., 2020, 268, 116507 CrossRef.
- L. Ge, X. Liu, H. Feng, H. Jiang, T. Zhou and H. Chu, et al., The interaction between microwave and coal: A discussion on the state-of-the-art, Fuel, 2022, 314, 123140 Search PubMed.
- A. Casella, A. Panitch and J. K. Leach, Endogenous Electric Signaling as a Blueprint for Conductive Materials in Tissue Engineering, Bioelectricity, 2021, 3(1), 27–41 CrossRef PubMed.
- C. H. Ho, R. J. Triolo, A. L. Elias, K. L. Kilgore, A. F. DiMarco, K. Bogie, et al., Functional electrical stimulation and Spinal Cord Injury, Physical Medicine and Rehabilitation Clinics of North America, W.B. Saunders, 2014, vol. 25, pp. 631–654 Search PubMed.
- F. Torregrossa, M. Sallì and G. Grasso, Emerging Therapeutic Strategies for Traumatic Spinal Cord Injury, World Neurosurg., 2020, 140, 591–601 Search PubMed.
- A. Hernando, F. Giacomone, J. Viñolas, M. A. García, F. Galvez and A. Castellanos, et al., Detecting Magnetic Permeability and Electrical Conductivity Fluctuations in Metallic Ferromagnetic Sheets through the Shielding Effect, Phys. Status Solidi B, 2022, 259(6), 2100446 Search PubMed.
- N. Lee, Y. Choi, Y. Lee, M. Park, W. K. Moon and S. H. Choi, et al., Water-dispersible ferrimagnetic iron oxide nanocubes with extremely high r 2 relaxivity for highly sensitive in vivo MRI of tumors, Nano Lett., 2012, 12(6), 3127–3131 Search PubMed.
- K. Xi, Y. Gu, J. Tang, H. Chen, Y. Xu and L. Wu, et al., Microenvironment-responsive immunoregulatory electrospun fibers for promoting nerve function recovery, Nat. Commun., 2020, 11(1), 4504 CrossRef PubMed.
- S. Schindler and T. Bechtold, Mechanistic insights into the electrochemical oxidation of dopamine by cyclic voltammetry, J. Electroanal. Chem., 2019, 836, 94–101 CrossRef.
- K. Kremser, M. Maltschnig, H. Schön, A. Jandric, M. Gajdosik and T. Vaculovic, et al., Optimized biogenic sulfuric acid production and application in the treatment of waste incineration residues, Waste Manage., 2022, 144, 182–190 CrossRef PubMed.
- A. Nagaraj, O. S. N. Ghosh, H. K. Ghneim, Y. A. AlSheikh, K. Mohammed and S. Poda, et al., Fe2O3-type iron oxide nanoparticles from Aerva lanata leaf extract exhibit antibiofilm, discriminatory toxicity in cancer cells, and theranostic against oxidative stress in zebrafish, Chem. Phys. Impact, 2025, 10, 100849 Search PubMed.
- Z. Cheng, D. Liu, J. Y. Park, X. Meng, Y. Yang and M. Dang, et al., Evaluation of the management of rotator cuff injuries utilising superparamagnetic iron oxide tracking stem cells, Tissue Cell, 2025, 95, 102836 CrossRef PubMed.
- Z. Derikvand, A. Tahmourespour, N. Akbari, G. R. Amiri and M. Fesharaki, Early apoptosis induction in MCF-7 breast cancer cells by bacterial exopolysaccharide-coated magnetic iron oxide nanoparticles, Int. J. Biol. Macromol., 2025, 306, 141605 CrossRef PubMed.
- M. M. Fathy, O. A. Saad and H. M. Fahmy, Radiosensitization impact assessment of silica-layered iron oxide nanocomposites with various shell thickness, Arch. Biochem. Biophys., 2025, 764, 110257 CAS.
- L. Xue and J. Sun, Magnetic hydrogels with ordered structure for biomedical applications, Front. Chem., 2022, 10, 1040492 CAS.
- L. E. Low, H. P. Lim, Y. S. Ong, S. P. Siva, C. S. Sia and B. H. Goh, et al., Stimuli-controllable iron oxide nanoparticle assemblies: Design, manipulation and bio-applications, J. Controlled Release, 2022, 345, 231–274 CAS.
- A. Cruz-Gregorio, A. K. Aranda-Rivera, O. E. Aparicio-Trejo, O. N. Medina-Campos, E. Sciutto and G. Fragoso, et al., GK-1 Induces Oxidative Stress, Mitochondrial Dysfunction, Decreased Membrane Potential, and Impaired Autophagy Flux in a Mouse Model of Breast Cancer, Antioxidants, 2022, 12(1), 56 Search PubMed.
- A. J. Dominijanni, M. Devarasetty, S. D. Forsythe, K. I. Votanopoulos and S. Soker, Cell Viability Assays in Three-Dimensional Hydrogels: A Comparative Study of Accuracy, Tissue Eng., Part C, 2021, 27(7), 401–410 Search PubMed.
| This journal is © The Royal Society of Chemistry 2025 |