A CO-detecting torch using carbon nanodots†
Shrodha
Mondal
and
Prithidipa
Sahoo
 *
*
Department of Chemistry, Visva-Bharati University, Santiniketan-731235, India. E-mail: prithidipa.sahoo@visva-bharati.ac.in
First published on 15th August 2025
Abstract
Carbon monoxide (CO), a common environmental contaminant, is responsible for a significant proportion of accidental poisonings and fatalities reported globally each year. Accurate detection and regulation of carbon monoxide in both residential and industrial settings are critical to mitigate the risk of severe health complications in humans due to its hazardous features and widespread sources. CO is odorless, colorless, tasteless, and non-irritating, which makes it particularly challenging to identify without a specialized tool. In this study, we have designed a portable sensing device utilizing N-CQDs for the real-time detection of atmospheric CO with fluorescence quenching and visual detection. In this detection process, we elucidated the fluorescence quenching mechanism of N-CQDs upon binding with CO (binding affinity: 9.2 × 10−4 M−1) with a detection limit of 0.07 μM (linear range between 4.77 and 41.23 μM). Several techniques, including HRTEM, DLS, EDX, FT-IR, XPS, UV-vis spectroscopy, fluorescence spectroscopy, and fluorescence lifetime decay, have also been employed to elucidate the quenching mechanism.
Introduction
Carbon monoxide (CO), often referred to as the “silent killer”, can be fatal as people are unaware of its presence due to its lack of sensory warning characteristics, leading to an untimely death. It is highly toxic and poses significant health risks, making its control and management critical in environmental protection efforts. CO poisoning affects approximately 50![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 people annually in the United States. With a 1–3% fatality rate, its clinical symptoms vary greatly,from weakness, confusion, nausea, disorientation, visual disturbances, dizziness, and headaches to coma and death. Many survivors of CO poisoning experience long-term neurological and emotional aftereffects. CO is discharged into the air primarily through the incomplete combustion of carbon-containing materials. Sources of carbon monoxide are numerous, making it, aside from carbon dioxide (CO2), one of the most prevalent pollutants in the lower atmosphere. This gas is produced by various activities, including vehicle emissions, industrial processes, and residential heating systems that burn fossil fuels.1–12 A study conducted at the All India Institute of Medical Sciences, New Delhi, examined the pattern of fatal CO poisoning between 2010 and 2015, involving 40 cases. Approximately 80% of these incidents occurred during the winter months. In all but one case, the individuals died in proximity to a CO source and were in a closed, unventilated space. The primary sources of CO were fire pots, chum tents, hearths, and electric room heaters, with some cases resulting from CO buildup inside cars with running engines and AC, and most of the fatalities were accidental.13 CO is odorless, tasteless, colorless, and non-irritating, making it extremely difficult to detect without specialized equipment. This stealthy nature allows CO to be a silent killer, as individuals in affected environments may not realize they are inhaling a toxic gas. In our recent research, a novel approach involving carbon quantum dot sensors was utilized to detect the presence of CO. The primary objective is to develop a device that detects the presence of carbon monoxide and provides timely alerts. This alert system is crucial as it allows individuals to take immediate action to safeguard themselves against the potentially lethal effects of CO poisoning. This technology will also enhance the support of public health efforts to manage and prevent CO-related incidents.14
000 people annually in the United States. With a 1–3% fatality rate, its clinical symptoms vary greatly,from weakness, confusion, nausea, disorientation, visual disturbances, dizziness, and headaches to coma and death. Many survivors of CO poisoning experience long-term neurological and emotional aftereffects. CO is discharged into the air primarily through the incomplete combustion of carbon-containing materials. Sources of carbon monoxide are numerous, making it, aside from carbon dioxide (CO2), one of the most prevalent pollutants in the lower atmosphere. This gas is produced by various activities, including vehicle emissions, industrial processes, and residential heating systems that burn fossil fuels.1–12 A study conducted at the All India Institute of Medical Sciences, New Delhi, examined the pattern of fatal CO poisoning between 2010 and 2015, involving 40 cases. Approximately 80% of these incidents occurred during the winter months. In all but one case, the individuals died in proximity to a CO source and were in a closed, unventilated space. The primary sources of CO were fire pots, chum tents, hearths, and electric room heaters, with some cases resulting from CO buildup inside cars with running engines and AC, and most of the fatalities were accidental.13 CO is odorless, tasteless, colorless, and non-irritating, making it extremely difficult to detect without specialized equipment. This stealthy nature allows CO to be a silent killer, as individuals in affected environments may not realize they are inhaling a toxic gas. In our recent research, a novel approach involving carbon quantum dot sensors was utilized to detect the presence of CO. The primary objective is to develop a device that detects the presence of carbon monoxide and provides timely alerts. This alert system is crucial as it allows individuals to take immediate action to safeguard themselves against the potentially lethal effects of CO poisoning. This technology will also enhance the support of public health efforts to manage and prevent CO-related incidents.14
Unlike bulk carbon with low solubility in water and weak fluorescence, CQDs exhibit good solubility and strong luminescence. This combination of properties has earned them the nickname “carbon nanolights”, highlighting their potential for applications in areas such as bioimaging, sensing, and optoelectronics. Photoluminescent carbon-based quantum dots offer several advantages over conventional semiconductor quantum dots and organic dyes, including strong chemical inertness, high aqueous solubility, ease of surface modification, and superior resistance to photobleaching. The exceptional electronic properties of carbon-based quantum dots (CQDs) enable them to function as both electron donors and acceptors, facilitating chemiluminescence and electrochemiluminescence. These attributes open up a wide range of applications in fields such as sensor technologies, optoelectronics, and catalysis.15 Herein, we synthesized these greenish-yellow-fluorescent nitrogen-doped carbon quantum dots (NCQDs) using a straightforward microwave-assisted method that included o-phenylenediamine and tris(2-aminoethyl)amine. Interestingly, the N-CQDs have remarkable CO detection capabilities through both fluorescence quenching and visual detection, where the absorption of CO causes the yellow solution of N-CQDs to turn brown (naked eye detection). Table S1, provides performance parameters for CO detection, comparing our proposed method with previously published methodologies. From those studies, we have perceived that the detection of atmospheric CO is a difficult one. This research presents an innovative and environmentally sustainable method for the synthesis of nitrogen-doped carbon quantum dots (N-CQDs), which exhibit remarkable fluorescence quenching characteristics, specifically for the selective detection of carbon monoxide (CO) at ambient temperature. Unlike previous sensors that rely on metallic oxides or polymers, the proposed approach is characterized by its cost-effectiveness and sustainability. Furthermore, it facilitates the creation of a portable and non-invasive CO detection device, which provides considerable enhancements in sensitivity, user-friendliness, and overall practicality. The detecting torch has been engineered to be quick and easy to use, making it ideal for monitoring CO levels in various settings such as cars, homes, or public spaces with the naked eye. As far as we are concerned, this is the first report to employ nanodots to recognize airborne CO to reduce life-threatening risks significantly.
Experimental section
Materials and methods
Nicotine, carbon monoxide, methanol, ethanol, hydrogen peroxide, o-phenylenediamine, tris(2-aminoethyl)amine, and all other analytes were sourced from TCI India and Sigma-Aldrich and used as received without further purification. Double-distilled water was employed as the solvent in the study.Preparation and characterization of N-CQDs
In this study, nitrogen-doped carbon quantum dots (N-CQDs) were synthesized using a household microwave oven. The starting materials for this process were o-phenylenediamine and tris(2-aminoethyl)amine, chosen for their high carbon and nitrogen content (see Scheme 1). Detailed information on the synthesis procedure, including specific conditions and procedures, can be found in the SI. In addition, a comprehensive suite of spectroscopic and microscopic techniques was utilized to characterize the N-CQDs thoroughly. The morphology and size distribution were analyzed using dynamic light scattering (DLS) and transmission electron microscopy (TEM). X-ray diffraction (XRD) analysis was employed to study the structure and crystallinity. Surface functionalities and chemical structures were examined using Fourier transform infrared (FT-IR) spectroscopy and energy dispersive X-ray spectroscopy (EDX). The optical properties, including UV absorption and fluorescence, were explored using a UV-visible spectrophotometer and a fluorimeter. Fluorescence lifetimes were determined through time-correlated single-photon counting (TCSPC). X-ray photoelectron spectroscopy (XPS) was conducted to investigate the elemental composition. Additional details are available in the SI. | ||
| Scheme 1 Schematic illustration of the preparation of N-CQDs using o-phenylenediamine and tris(2-aminoethyl)amine via a household microwave approach. | ||
Detection of CO
To prepare the N-CQD solution, 200 μL of N-CQDs were mixed with 1.8 mL of deionized water in a quartz cuvette. The solution was then combined with different concentrations of CO from a 10−4 M stock solution. Fluorescence spectra were recorded using an excitation wavelength of 400 nm.Determination of the quantum yield (QY)
The following equation16 was used to estimate the quantum yield (QY) of N-CQDs, with quinine sulfate serving as the reference (QY = 0.54):| Qx = Qst(Ix/Ist)(Ast/Ax)(nx2/nst2) | (1) |
Results and discussion
Morphological analysis
Fig. 1A displays the TEM images of the N-CQD sample, clearly showing that the synthesized N-CQDs exhibit a spherical nanoparticle morphology with uniformly distributed particles and no signs of agglomeration. The size distribution of the N-CQDs ranges from 1.7 to 3.6 nm, with an average diameter of approximately 2.3 nm (Fig. 1B) (corroborated by the DLS results). The HRTEM image (inset in Fig. 1A) reveals distinct lattice fringes in most particles, and the inset shows that the N-CQDs have a crystalline nature (with a lattice spacing of 0.1090 nm), which is substantiated by the circular ring pattern in the SAED image (Fig. 1C).17XRD was employed to analyze the phase structure and crystallinity of the produced N-CQDs. The prominent diffraction peak in the XRD pattern, centered around 2θ = 24.5°, indicates that the N-CQDs are mainly amorphous and possess a disordered carbon structure. This broad peak distinguishes the N-CQDs from highly crystalline materials like graphite or graphene, which typically show sharper and more intense peaks in the same region (2θ = 26.05°) (Fig. S1).
FTIR analysis was conducted to examine the surface functional groups of the N-CQDs, as shown in Fig. 1D. The broad absorption bands in the range of 3100–3500 cm−1 were attributed to n(N–H) vibrations. The stretching vibration of C–H and the asymmetric and symmetric stretching modes of the –CH2– groups were identified as the sources of the absorption bands at 2938 and 2844 cm−1. Additionally, the bands between 1640 and 1550 cm−1 were associated with δ(N–H), indicating the presence of amino-containing functional groups on the surface of the N-CQDs. The distinctive peak at 1471 cm−1, which corresponds to C–N, verifies the nitrogen doping on the N-CQDs. Moreover, the absorption peaks of N-CQDs in the 1000–1300 cm−1 range are attributed to C–N and N–H bonds.18–23 The further studies of the N-CQDs/CO complex clarified the interaction between CO and the surface groups of N-CQDs. The generation of a new peak at 1972 cm−1 denotes the CO stretching frequency (Fig. S2). The shifts in absorption peaks compared to the dots indicate the formation of the complex between CO and the functional groups of N-CQDs, which likely contributes to the fluorescence quenching mechanism. Also, the variations in peak intensity suggest alterations in the vibrational modes, potentially due to CO adsorption or bonding.
XPS and EDX analyses were employed to explore the structural details and elemental composition of the N-CQDs. The EDX spectra (Fig. 1E) revealed the presence of carbon (C) and nitrogen (N) atoms, aligning with the results from the XPS study (Fig. 1F). The main peaks in the XPS survey spectrum of the N-CQDs correspond to the binding energies of C1s and N1s at 284 and 398 eV, respectively (Fig. 1F). Three distinct peaks at 283.6, 284.4, and 286.2 eV, corresponding to sp2C in graphene, C–C/C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, and C–N/C–O, were identified in the scrupulously examined high-resolution XPS spectra of C1s (Fig. 1G). Pyridinic N (C–N–C, C
C, and C–N/C–O, were identified in the scrupulously examined high-resolution XPS spectra of C1s (Fig. 1G). Pyridinic N (C–N–C, C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), pyrrolic N (C3–N), and N–H, which verify the nitrogen-doping of CDs, were clearly represented by peaks that could be seen at 398.1, 398.7, and 400.09 eV in the N1s spectra (Fig. 1H).24–27
N), pyrrolic N (C3–N), and N–H, which verify the nitrogen-doping of CDs, were clearly represented by peaks that could be seen at 398.1, 398.7, and 400.09 eV in the N1s spectra (Fig. 1H).24–27
Optical properties of N-CQDs
The UV-visible absorption spectra of N-CQDs, as depicted in Fig. 2A, exhibit a characteristic peak at 415 nm. The surface state of the N-CQDs is responsible for the absorption band at 415 nm.28 The N-CQDs exhibited maximal fluorescence emission at 530 nm (Emax: 400 nm) (Fig. 2B) and showed high solubility and vivid greenish-yellow emission when irradiated with a UV lamp (inset of Fig. 2B). The highest fluorescence intensity of N-CQDs appeared at an excitation wavelength of 400 nm, and the intensity gradually decreased with a redshift as seen in Fig. 2C. The fluorescence quantum yield (FLQY) of the N-CQDs was estimated to be approximately 35% (Fig. S3). A pH investigation of N-CQDs was carried out with and without CO, and the fluorescence intensity remained constant within the pH range of 7–9 in both cases (Fig. 2D). The photostability of N-CQDs was rigorously assessed by exposing them to continuous UV light at 365 nm. Notably, after 240 minutes of enlightenment, the fluorescence intensity remained virtually unchanged as depicted in Fig. S4. This underscores the superior photostability and resistance to photodegradation of N-CQDs under protracted UV irradiation. Furthermore, even after a storage period of a few months, the fluorescence intensity remained consistent when re-exposed to UV light (365 nm). Even after 24-hour exposure to ambient air, N-CQDs maintained consistent performance within the temperature range of 16–40 °C and under varying humidity conditions, exhibiting a stable fluorescence response and no sensitivity loss, with a response time of approximately 30 seconds.Fluorescence detection of N-CQDs in the presence of CO
To evaluate the potential of N-CQDs as a CO sensor, CO solution (10−4 M) was introduced into the N-CQD solution, and the fluorescence spectra were recorded. A progressive decrease in fluorescence intensity was observed (Fig. 3A) with the increase of the volume of CO from 100 to 1400 μL, confirming the successful quenching of N-CQDs. The changes in the fluorescence intensity of N-CQDs with different CO concentrations under a UV lamp have also been recorded (Fig. S5). Furthermore, the UV-vis titration shows that when the CO concentration increases, the intensity of N-CQDs consistently increases (Fig. 3B). This demonstrates the ability of N-CQDs to detect CO through changes in fluorescence intensity in addition to UV intensity. Regression analysis was used to quantify the binding affinity of N-CQDs for CO, and the value obtained was 9.2 × 104 M−1 (Fig. S6). With a correlation coefficient (R2) of 0.99695, Fig. S7, shows an excellent linear relationship between fluorescence intensity and CO concentration over the 4.77–41.23 μM range. With the use of the formulas 3σ/m and 10σ/m, where m is the curve's slope and σ is the standard deviation, the limit of detection (LOD) and limit of quantification (LOQ) of N-CQDs for CO were determined to be 0.07 μM and 0.235 μM, respectively (Table S2). Comprehensive selectivity tests were further conducted (Fig. S8) to reveal the fluorescence quenching of N-CQDs in the presence of CO. Other analytes, when tested, showed minimal to no effect on the fluorescence, underscoring the high selectivity of N-CQDs for CO detection. Furthermore, to assess the selectivity of the N-CQDs under real-world conditions, we tested their response to CO alongside potential interfering substances like NO, CO2, SO2, and common VOCs such as methanol, ethanol, benzene, and acetone. As shown in Fig. S9, the fluorescence intensity remains significantly quenched with CO in all cases. This slight increase compared to CO alone may result from competitive binding, partial inhibition, or matrix effects. Importantly, no interfering species eliminate or hide the CO-induced quenching, confirming the high selectivity of this nanosensor for CO even in complex environmental mixtures. Fluorescence spectroscopy combined with UV-vis absorption provides preliminary insights into the interaction mechanisms, while FT-IR, DLS, and XPS offer more detailed information on specific chemical interactions and the functional groups involved. | ||
| Fig. 3 (A) Fluorescence emission spectra of N-CQDs upon addition of CO (10−4 M). (B) UV-vis absorption spectra of N-CQDs upon addition of CO (10−4 M). | ||
Probable quenching mechanism discussion
Several studies were conducted to comprehend the quenching process of N-CQDs caused by CO. Numerous processes, including static quenching, dynamic quenching, and combinations of the above two are reasons for the quenching of fluorescence. To clarify the quenching principle of N-CQDs by CO, the Stern–Volmer equation23,29 was utilized (eqn (2)). A plot of F0/F versus [Q] was generated, where [Q] represents the CO concentration, F0 and F are the fluorescence intensities of N-CQDs in the absence and presence of CO (the quencher), respectively, and K denotes the Stern–Volmer quenching constant. A linear curve is anticipated in the case of exclusive static or dynamic quenching as observed in our experiment.| F0/F = 1 + K[Q] | (2) |
In the case of combined dynamic and static quenching, the Stern–Volmer equation can be expressed as follows:
| F0/F = (1 + KD[Q])(1 + KS[Q]) | (3) |
This implies that the plot of F0/F versus [Q] should show an upward curvature, which did not occur in our results (Fig. 4A). Thus, combined mechanisms were ruled out.
Fluorescence lifetime studies were conducted to understand the quenching concept comprehensively. In the absence and presence of CO, the average fluorescence lifetimes of the N-CQDs were determined to be 0.84 and 0.87 ns, respectively (Table S3). Fig. 4C and Table S3, showed no discernible changes upon adding CO, suggesting the ground state interaction of N-CQDs and CO through static quenching and ruling out the prospect of dynamic quenching.30 Additionally, UV-vis spectroscopy evaluations were carried out to verify static quenching by complex formation in the ground state. Fig. 3B shows a shift in the absorption peak of N-CQDs following the addition of CO, corroborating the ground state complex formation.
Additionally, a modified Stern–Volmer equation was employed to ensure the formation of the ground-state complex by static quenching:
| Kapp = ((F0/F − 1)/[Q]) = (KS + KD) + KSKD[Q] | (4) |
Plotting (F0/F − 1)/[Q] vs. [Q] most likely results in a straight line, indicating the quenching was likely facilitated by the development of a ground-state complex, which was seen in this instance (Fig. 4B).31
Conversely, the fluorescence emission and/or excitation spectra of the fluorophore (N-CQDs) overlap with the quencher's (CO) absorption band to indicate the inner filter effect (IFE), which was also observed in our scenario (Fig. 4D).32
Therefore, it can be deduced that the quenching mechanism of the N-CQDs is likely related to the static quenching process through ground-state complex formation and IFE (Fig. 5).
 | ||
| Fig. 5 Diagrammatic representation of combined static quenching and IFE of the N-CQDs via the quencher (Q) CO. | ||
Development of the carbon monoxide detection device
To evaluate the suitability of N-CQDs for detecting atmospheric CO, we assembled the apparatus as illustrated in Fig. 6A. | ||
| Fig. 6 (A) Experimental setup for CO detection from partially combusted cigarettes. (B) Fluorescence emission spectra of N-CQDs in different concentrations of CO developed from the experiment. | ||
Since carbon monoxide (CO) is not well soluble in water, ethanol was placed in the round-bottom flask kept on the left side, while the right side contained partially combusted cigarettes as a CO source. Ethanol served as the medium to absorb the smoke produced, capturing the CO. Fluorescence measurements of the N-CQDs with the seized CO revealed a consistent decrease in intensity (Fig. 6B), confirming the capability of N-CQDs to bind CO. Aiming to these findings, we developed a portable sensing device (Scheme 2) designed for real-time CO monitoring in various environments with naked eye detection. Given that CO can be produced in settings such as vehicles with air conditioning, room heaters, hearths, oven tents, or from incomplete combustion of wood, its detection is critical due to its colorless and odorless nature. Our nanodot-based device offers a simple yet highly effective sensing detector of CO, providing an essential tool for monitoring this hazardous gas in unpredictable scenarios.
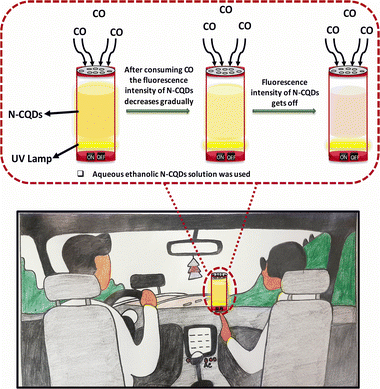 | ||
| Scheme 2 Diagrammatic representation of the device designed for real-time CO detection across different environments. | ||
Conclusions
The undetectable properties of CO cause it to be a significant threat to human health, as it can silently accumulate to hazardous levels, leading to severe poisoning or fatal outcomes. The significance of CO detectors lies in their ability to save lives by providing early warnings of emergent CO. In this research, we have established a highly water-soluble, photostable, exceptionally biocompatible, and non-toxic nanosensor (N-CQDs) that demonstrates a superior ability to detect and bind with carbon monoxide through a fluorescence quenching mechanism, offering a reliable approach for CO sensing. Employing this mechanism, we have developed a CO-detecting torch that could serve as an indicator for measuring atmospheric CO. Such nanosensors represent an important technological advancement in gas detection, providing a sensitive and effective means to enhance public safety.Author contributions
S. M.: analysis, investigation, data collection, experiment, visualization, validation, methodology, original draft writing, and editing. P. S.: conceptualization, design of experiments, data curation, script reviewing & editing, visualization and supervision of the investigation, and funding acquisition.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the SI. See DOI: https://doi.org/10.1039/d5tc02382cAcknowledgements
P. S. acknowledges the SERB, India, for awarding her SERB power grant [project file no. SPG/2020/000713]. The authors sincerely acknowledge the Department of Chemistry, Visva-Bharati, Santiniketan, West Bengal, India. The authors also acknowledge Supriya Bhunia, Department of Microbiology, University of Calcutta, Kolkata, India, for helping with the XRD analysis. The authors thank the Department of ISERC, Visva-Bharati University, India for the assistance with FT-IR, funded by the DST FIST (ref. no. SR/FST/CS-I/2017/21).References
- S. A. Henn, J. L. Bell, A. L. Sussell and S. Konda, Am. J. Ind. Med., 2013, 56, 1280–1289 CrossRef CAS PubMed.
- C. H. Cho, N. C. Chiu, C. S. Ho and C. C. Peng, Pediatr Neonatol., 2008, 49, 121–125 CrossRef PubMed.
- N. B. Hampson, Undersea Hyperbaric Med., 2018, 45(2), 165–171 CrossRef.
- K. Otterness and C. Ahn, Emerg. Med. Pract., 2018, 20(3), 1–24 Search PubMed.
- J. B. Buboltz and M. Robins, StatPearls [Internet], StatPearls Publishing, Treasure Island (FL), 2023. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470531/ Search PubMed.
- G. Reumuth, Z. Alharbi, K. S. Houschyar, B. S. Kim, F. Siemers, P. C. Fuchs and G. Grieb, Burns, 2019, 45(3), 526–530 CrossRef PubMed.
- N. B. Hampson, J. Emerg. Med., 1998, 16, 695–698 CrossRef CAS PubMed.
- N. B. Hampson and L. K. Weaver, Undersea Hyperbaric Med., 2007, 34, 163–168 CAS.
- A. Vaidyanathan, J. Malilay, P. Schramm and S. Saha, Centers for Disease Control and Prevention (CDC). Carbon monoxide-related deaths-United States, 1999–2004, MMWR Morb. Mortal. Wkly. Rep., 2007, 56, 1309–1312 Search PubMed.
- N. B. Hampson, Ann. Am. Thorac. Soc., 2016, 13, 1768–1774 Search PubMed.
- N. B. Hampson and D. Bodwin, J. Emerg. Med., 2013, 44, 625–630 CrossRef PubMed.
- J. A. Mott, M. I. Wolfe, C. J. Alverson, S. C. Macdonald, C. R. Bailey, L. B. Ball, J. E. Moorman, J. H. Somers, D. M. Mannino and S. C. Redd, JAMA, 2002, 288, 988–995 CrossRef PubMed.
- A. K. Sikary, S. Dixit and O. P. Murty, J. Family Med. Prim. Care, 2017, 6(4), 791–794 CrossRef PubMed.
- M. F. Hashim, N. Sazali, Z. Harun, N. Sazali, W. N. W. Salleh and T. Gunawan, Mater. Today: Proc., 2023, 2214–7853 Search PubMed.
- Y. Wang and A. Hu, J. Mater. Chem. C, 2014, 2, 6921 RSC.
- Sk. Md Palashuddin and A. Chattopadhyay, RSC Adv., 2014, 4, 31994–31999 RSC.
- W. Peng, L. Wei, W. Qiong, L. Yushan and L. Shouxin, RSC Adv., 2017, 7, 44144–44153 RSC.
- Y. Zheng, D. Yang, X. Wu, H. Yan, Y. Zhao, B. Feng, K. Duan, J. Weng and J. Wang, RSC Adv., 2015, 5, 90245–90254 RSC.
- J. Shen, S. Shang, X. Chen, D. Wang and Y. Cai, Sens. Actuators, B, 2017, 248, 92–100 CrossRef CAS.
- S. Cai, Q. Houjuan, M. Rongxiu, S. Zhe, X. Lidong, W. Guangbiao, H. Zhanhua, L. Shouxin, L. Jian, D. Mengyao, F. Jincheng and G. Zhanhu, Mater. Sci. Eng. C, 2019, 105, 110132 CrossRef PubMed.
- K. G. Nguyen, I.-A. Baragau, R. Gromicova, A. Nicolaev, S. A. J. Thomson, A. Rennie, N. P. Power, M. T. Sajjad and S. Kellici, Sci. Rep., 2022, 12, 13806 CrossRef CAS PubMed.
- F. Jie, Z. Xiaoran, B. Wei and T. Xinjing, Mater. Chem. Front., 2019, 3, 2751–2758 RSC.
- L. Wenjing, J. Yuan, G. Yifang, Q. Jie, M. Maedeh, S. Shaomin, D. Chuan and Z. Chen, ACS Appl. Mater. Interfaces, 2018, 10(49), 42915–42924 CrossRef PubMed.
- S. Mondal, O. Sarkar, J. Raut, S. M. Mandal, A. Chattopadhyay and P. Sahoo, ACS Appl. Bio Mater., 2024, 7(7), 4690–4701 CrossRef CAS PubMed.
- Y. M. AlZahrani, S. Alharthi, H. A. AlGhamdi, A. O. Youssef, S. S. Ahmed, E. H. Mohamed, S. A. Mahmoud and M. S. Attia, RSC Adv., 2023, 13, 21318 RSC.
- S. Mondal, O. Sarkar, S. K. Jana, A. Chattopadhyay, S. Mandal and P. Sahoo, ACS Sustainable Chem. Eng., 2025, 13(7), 2998–3009 CrossRef CAS.
- C. Shi, H. Qi, R. Ma, Z. Sun, L. Xiao, G. Wei, Z. Huang, S. Liu, J. Li, M. Dong, J. Fan and Z. Guo, Mater. Sci. Eng. C, 2019, 105, 110132 CrossRef CAS PubMed.
- W. Lu, Y. Jiao, Y. Gao, J. Qiao, M. Mozneb, S. Shuang, C. Dong and C. Li, ACS Appl. Mater. Interfaces, 2018, 10, 42915–42924 CrossRef CAS PubMed.
- J. Raut, R. D. Sherpa, S. K. Jana, S. M. Mandal, S. Mandal, S. P. Hui and P. Sahoo, ACS Appl. Nano Mater., 2023, 6, 23611–23619 CrossRef CAS.
- R. L. Joseph, Principles of Fluorescence Spectroscopy, Kluwer Academic/Plenum Publishers, New York, 2nd edn, 1999 Search PubMed.
- S. Mondal, J. Raut, O. Sarkar, S. M. Mandal, A. Chattopadhyay and P. Sahoo, New J. Chem., 2023, 47, 20859–20865 RSC.
- S. A. Tanwar, R. Parui, R. Garai, A. M. Chanu and K. P. Iyer, ACS Meas. Sci. Au, 2022, 2, 23–30 CrossRef PubMed.
Footnote |
| † A tribute to the late Prof. Amitabha Sarkar on his 75th birthday. |
| This journal is © The Royal Society of Chemistry 2025 |



