Carbon dot engineered trimetallic hydroxyl carbonates: a strategy for enhanced redox-diffusion coupled charge storage†
Tanzeem
Fatema
 ,
Gaurav
Singh
,
Gaurav
Singh
 and
Darshna
Potphode
and
Darshna
Potphode
 *
*
Department of Chemistry, Sunandan Divatia School of Science, SVKM's Narsee Monjee Institute of Management Studies (NMIMS) Deemed-to-be University, Mumbai 400 056, Maharashtra, India. E-mail: drdarshnapotphode@gmail.com; Tel: +91 2242330364
First published on 14th July 2025
Abstract
Transition metal oxides have long been vital in energy storage due to their high theoretical capacitance and synergistic redox mechanisms. This study introduces a unique, binder-free synthetic approach to synthesize nickel–cobalt–zinc metal hydroxyl carbonate (MHC) with a flower-like morphology, ensuring efficient electrolyte access. These MHCs achieved an impressive specific capacitance of 1304 C g−1 (3261 F g−1) at 5 A g−1, attributed to trimetallic synergy. Incorporating carbon dots derived from ajwain leaves further boosted the specific capacitance by 9.3%, as these dots acted as efficient conductive channels. A hybrid asymmetric device, pairing the carbon dot-incorporated MHC as a positive electrode with activated carbon as a negative electrode, delivered 93 F g−1 at 1 A g−1, alongside excellent energy and power densities of 33.5 W h kg−1 and 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 W kg−1, respectively. This device demonstrated remarkable stability, retaining 77% capacitance with 100% coulombic efficiency over 5000 cycles at 5 A g−1. Dunn's method confirmed diffusion-controlled charge storage as predominant. These findings highlight carbon dot-modified MHCs as highly promising electrodes for future energy storage applications.
200 W kg−1, respectively. This device demonstrated remarkable stability, retaining 77% capacitance with 100% coulombic efficiency over 5000 cycles at 5 A g−1. Dunn's method confirmed diffusion-controlled charge storage as predominant. These findings highlight carbon dot-modified MHCs as highly promising electrodes for future energy storage applications.
1. Introduction
Energy demand has risen sharply with development and continues to grow steadily as modernization advances. Currently, fossil fuels, including coal, oil, and natural gas, are the primary sources of energy. Our reliance on non-renewable energy makes it essential to discover lasting alternatives. Though renewable sources like solar, wind, and tidal energy hold significant potential, their intermittent nature means that effective energy storage technologies are crucial for their widespread adoption.1,2 To be ideal, an energy storage device needs a high capacity, durability, long shelf-life, and rapid charging and discharging. Supercapacitors are a promising candidate because they’re compact and lightweight, and also offer quick charge and discharge, as well as being inexpensive. There are three different types of supercapacitors classified based on their working principle: electric double-layer capacitors (EDLCs), pseudocapacitors, and hybrid supercapacitors.3 EDLCs typically use carbon-based electrodes. They store energy by physically separating charges at the electrode–electrolyte interface, which creates a potential difference. This method allows for high power density due to its rapid charge separation but often results in lower energy density because of limited capacitance.4,5 In contrast, pseudocapacitors store energy through fast, reversible redox reactions where electrons are exchanged at the electrode surface. These often use transition metals with varying valence states or conducting polymers in their electrodes, leading to high energy density and specific capacitance. However, the electron exchange process gives them lower power density compared to EDLCs, and bulk phase reactions in metal oxides can impact their stability.5 Hybrid supercapacitors combine the best of both EDLCs and pseudocapacitors. They feature a carbon-based negative electrode and a positive electrode made from materials like metal oxides, metal hydroxides, or composites of metal oxide/polymer with carbon. This design delivers both high power density and high energy density, and excellent stability.3 Recently scientists have explored battery-type behaviour in supercapacitors. This involves electrolyte ion intercalation into the electrode material followed by electron exchange. These electrode materials primarily store charge through a diffusion-controlled process.6 Transition metals like nickel, cobalt, and zinc are effective in energy storage due to their variable oxidation states and excellent conductivity, making them ideal for cathodic electrode materials in supercapacitor devices.7,8 For the positive electrode, ternary metal composites demonstrate enhanced charge storage capacity compared to binary and single metal ion counterparts due to synergistic effects. Several studies have highlighted the effectiveness of these materials: Manikandan et al. synthesized a mixed metal oxide of nickel–cobalt–zinc on nickel foam (NF), achieving a specific capacity of 596 C g−1 at 1 A g−1.9 Akram and Mahmood et al. created 3D nanowires of zinc–nickel–cobalt ternary oxide, resulting in a specific capacitance of 2082.21 F g−1 at 1 A g−1.10 In another study, Wu and Jia et al. synthesized mesoporous nanosheets of cobalt–nickel–zinc ternary oxide, showing a specific capacity of 1172.2 C g−1 at 1 A g−1.11 Gaur et al. developed copper-doped zinc–cobalt oxide nanostructures with a specific capacitance of 1425 F g−1 at 1 A g−1.12 In metal oxide electrodes, the movement of counter ions during charge–discharge cycles leads to undesirable expansion and compression, which degrades the material's performance. This occurs because counter ions, being oppositely charged, must move near the electrode surface to maintain electrical neutrality during electron exchange. This limitation has been overcome by using metal hydroxides and metal hydroxyl carbonates, which exhibit superior specific capacitance compared to metal oxides, attributed to the presence of hydroxyl and carbonate groups within their structure. For example, Shim et al. synthesized a ternary composite of nickel–cobalt–manganese hydroxyl carbonate with a specific capacitance of 4635 F g−1 at 1 A g−1.7 Kim and Yun et al. created a binder-free nickel–cobalt hydroxyl carbonate with a specific capacity of 1288.2 mA h g−1 at 3 A g−1, and Ma et al. synthesized monolayer nickel–cobalt hydroxyl carbonate with a specific capacitance of 2266 F g−1 at 0.5 A g−1.13 Carbonate and hydroxy ions act as counter ions, enhancing electrode performance. However, metal hydroxy carbonates typically exhibit poor high-rate performance; to overcome this limitation, researchers have combined metal oxides and hydroxides with carbon-based materials like graphene, carbon nanotubes, and activated carbon. These carbon materials enhance the electrochemical properties by providing conductive channels for capacitive charge storage. Recent studies have explored incorporating small-scale carbon-based materials, like CDs and graphene quantum dots, into electrode materials. These CDs or quantum dots are typically derived from graphene or other chemical precursors.14–17 However, in a notable study, Satpati et al. used CDs derived hydrothermally from lemon juice and incorporated in nickel sulfide electrodes, achieving a specific capacitance of 880 F g−1 at 2 A g−1.18 Hence, there is a need to enhance the high-rate performance nature of metal hydroxyl carbonate by incorporating small scale carbon derived from natural precursors without the use of harsh chemicals or high temperature reactions.In this study, we derived carbon dots (CDs) from dried ajwain leaves using a straightforward method involving torrefaction and ultrasonication. Typically, carbon black is added to the electrode materials to improve ion conduction. However, we used these CDs, which are 1–100 nm, quasi-spherical carbon nanoparticles with a sp2 hybridized carbon core and sp3 hybridized defects, heteroatoms, and surface functional groups.19 CDs offer several advantages due to their large surface area, tuneable band gaps, and excellent wetting properties.20 Their small size and ability to be shaped make them ideal for flexible and micro-scale supercapacitors by shortening ion-diffusion pathways.18 We incorporated these CDs into a cobalt–nickel–zinc metal hydroxyl carbonate (MHC) composite. The CDs were synthesized without harsh chemicals, and both the CDs and MHC electrode material were prepared using a hydrothermal technique. This method allowed for the direct growth of the electrode material onto the current collector, eliminating the need for hydrophobic binders. This is crucial because binders can increase resistance and weight, reducing the active surface area and hindering performance. In our research, the CDs actually served the function of facilitating high-rate performance. The hydrothermal method also gave us precise control over the material's growth and deposition on the nickel foam (NF) substrate by allowing us to adjust time and temperature parameters. We then evaluated this novel electrode material for its energy storage capabilities. The theoretical specific capacitance values for cobalt hydroxyl carbonate, nickel hydroxyl carbonate, and zinc hydroxyl carbonate from the literature are 2732, 2297, and 2240 F g−1, respectively.7,21 In our study, we combined the synergistic effects of cobalt–nickel–zinc metal hydroxyl carbonates with the efficient conductive pathways provided by CDs to achieve an enhanced specific capacity of 1304 C g−1 (3261 F g−1) at 5 A g−1, leading to enhanced potential for efficient energy storage and application in energy storage devices.
2. Experimental section
2.1. Materials
All chemicals used were of analytical grade and were employed without further purification. Cobalt(II) nitrate hexahydrate [Co(NO3)2·6H2O, 98%] was purchased from Loba Chemie Pvt. Ltd, zinc nitrate hexahydrate [Zn(NO3)2·6H2O] was supplied from Sigma Aldrich, potassium hydroxide (KOH, 85%) and L-(+)-ascorbic acid (L-AA, 99%) were purchased from Research-Lab Fine Chem Industries. Ethanol (99.9%) was purchased from Changshu Hongsheng Fine Chemical Co., Ltd; NF (1.6 mm thickness) was purchased from Global Nanotech. The NF was cut to 5 × 1 cm2, cleaned using 3 M hydrochloric acid and acetone, and then dried at 60 °C in a vacuum oven for 12 hours.2.2. CDs from agrowaste
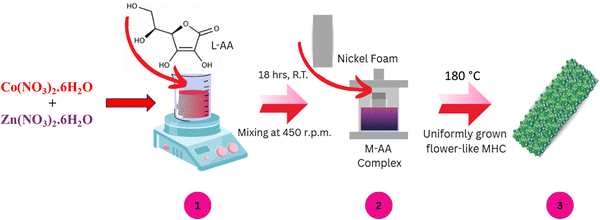
The CDs were synthesized from dried Trachyspermum ammi (ajwain) leaves as shown in Scheme 1. The dried leaves were crushed and torrefied in a tube furnace at 300 °C for 2 hours under an inert atmosphere (N2 gas). The carbonized product was then crushed, sonicated in DI water (10 mL per 100 mg of product) for 30 minutes, filtered through a 0.22 μm syringe filter, freeze-dried and stored for further use. Since the CDs are of small size, they usually exhibit the property of photoluminescence under UV irradiation.22 The photoluminescence under UV light and its UV-Vis spectrum can be seen in Fig. S1 (ESI†), which confirms the formation of CDs.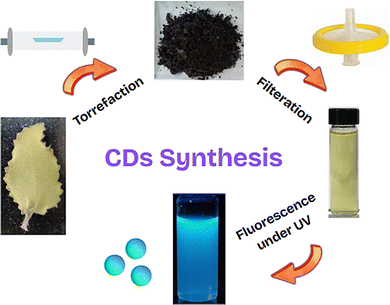 | ||
| Scheme 1 Schematic diagram for the synthesis process of CDs derived from Trachyspermum ammi (ajwain) leaves. | ||
2.3. Synthesis of carbon dot incorporated metal hydroxyl carbonate (CDMHC) on NF
Scheme 2 illustrates the stepwise process for the synthesis of CDMHC on NF. CDs were dispersed in 10 mL DI water, then mixed with 2 mM Zn(NO3)2·6H2O and 8 mM Co(NO3)2·6H2O and diluted to 60 mL with DI water. Separately, 120 mg L-AA was dissolved in 20 mL DI water and added slowly to the solution with continuous stirring [step 1]. After 30 minutes of stirring at room temperature, 0.3 g KOH dissolved in 20 mL DI water was added dropwise over 30 minutes. The mixture was stirred for 18 hours, then transferred to a 100 mL Teflon-lined hydrothermal autoclave containing a 5 × 1 cm2 NF. The autoclave was heated at 180 °C for 12 hours [step 2], cooled to room temperature, and the deposited foam was washed by dipping in DI water three times, then dried in a vacuum oven at 60 °C for 12 hours [step 3].To evaluate the impact of CDs on supercapacitor performance, MHC was synthesized separately. Furthermore, various amounts of CDs (2.5 mg, 5 mg, and 10 mg) were added to optimize performance, resulting in samples denoted as 2.5CDMHC, 5CDMHC, and 10CDMHC, respectively. This methodology was chosen for its simplicity and eco-friendliness, utilizing a one-step reaction with mild precursors and green reagents such as L-AA. The positive electrode fabrication was done using hydrothermal synthesis which grows material directly on NF, eliminating the need for hydrophobic binders that block active sites and reduce electrode performance due to an unwanted increase in resistance and weight of the electrode, enhancing efficiency. The amount of active material deposition per cm2 was found to be in the range of 1.5 ± 0.2 mg.
2.4. Characterization and measurements
Structural properties of the synthesized material were explored using Fourier-transform infrared spectroscopy (FT-IR, IRAffinity-1S), and chemical composition was further confirmed using a Raman Spectrometer (Thermo Fisher Scientific) with an excitation wavelength of 532 nm. The phase and crystallinity changes were discovered using X-ray diffraction (XRD, MiniFlex 300/600), which was carried out at a 2θ range of 5 to 65°. The surface area of the active electrode material was investigated by studying adsorption–desorption isotherms achieved from Brunauer–Emmett–Teller (BET) analysis using a Microtrac Belsorp MiniX instrument. Morphological examination and elemental composition were studied using field emission scanning electron microscopy (FE-SEM, Carl Zeiss Model Supra 55) and high-resolution transmission electron microscopy (HR-TEM, FEI Tecnai G2, F30) with an acceleration voltage of 300 kV. Surface elemental composition was analyzed through X-ray photoelectron spectroscopy (XPS, Omicron ESCA, Oxford Instrument Germany) using monochromatic Al Kα radiation.2.5. Fabrication of the supercapacitor device
The positive electrode was fabricated by hydrothermal growth of CDMHC on an NF substrate. The negative electrode was developed by depositing activated carbon (AC) onto NF. The mass of the electrode material for the negative electrode was calculated and optimized using the mass law equation (eqn (S1), ESI†).7 Initially, AC (85%) and 10% conducting carbon was dispersed in isopropyl alcohol, and 5% PTFE binder was added. The resulting mixture was drop-cast onto the NF and was further dried in a vacuum oven at 60 °C for 12 h. A device was fabricated by assembling 1 cm2 areas of the positive and negative electrodes, separated by a separator.2.6. Electrochemical characterization
Electrochemical analysis such as cyclic voltammetry (CV), galvanostatic charge discharge (GCD) and electrochemical impedance spectroscopy (EIS) was carried out using a Metrohm Autolab electrochemical workstation PGSTAT204. To examine the electrochemical properties and redox behavior of the synthesized positive electrode material, a three-electrode system was assembled. The working electrode was MHC or CDMHC grown on NF, with Ag/AgCl as the reference electrode and a platinum pan as the counter electrode. All electrochemical analyses, including CV, GCD, and EIS, were performed in 6 M KOH electrolyte.3. Results and discussion
3.1 Plausible mechanism for the formation of CDMHC
The electrode material was synthesized via the hydrothermal technique. L-AA acts as a morphology-directing agent, first undergoing oxidative degradation to form dehydro-L-AA (eqn (1)). At higher temperatures, Ni2+ ions detach from the NF and enter the reaction medium (eqn (2)). These further form complexes with metal precursors to form a metal-L-ascorbate chelate (eqn (3)),7 lowering the pH of the reaction medium. Some dehydro-L-AA decomposes into diketogulonic acid (eqn (4)), which further breaks down into erythrose and oxalate ions (eqn (5)). These compounds, in an alkaline medium and at high temperatures, decompose to release carbonate (CO32−) ions. The metal ions combine with hydroxide and carbonate ions to form respective metal hydroxyl carbonate (eqn (6)). Over time, flower-like structures of CDMHC grow on the NF through coalescence and Ostwald ripening (eqn (1)–(4)).7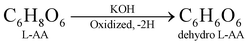 | (1) |
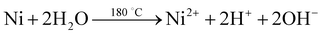 | (2) |
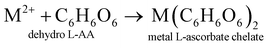 | (3) |
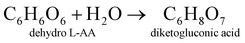 | (4) |
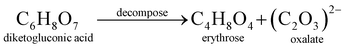 | (5) |
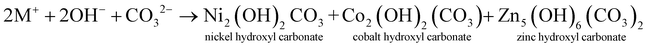 | (6) |
3.2. Chemical structure and morphology
Initially, FT-IR analysis was conducted to examine the functional groups associated with the synthesized material and confirm composite formation (Fig. 1(a)). A broad peak at around 3483 cm−1 was observed, indicating the presence of –OH stretching vibration. This peak is attributed to the hydroxyl groups in both MHC and CDMHC. A similar peak was also observed for CDs, suggesting the presence of OH functionality in the CD structure as well. This –OH functionality was responsible for imparting a hydrophilic nature to the CDMHC electrode material.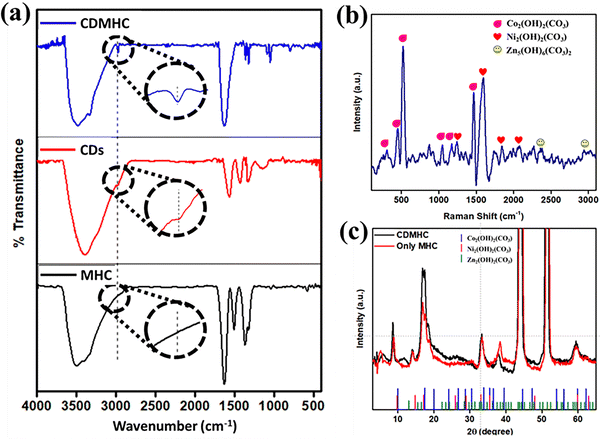 | ||
| Fig. 1 (a) Comparative FT-IR spectra of (i) CDMHC, (ii) CDs and (iii) MHC, (b) Raman spectrum of CDMHC and (c) comparative XRD diffraction pattern for MHC and CDMHC at a 2θ range of 5 to 65°. | ||
The major peak at 1625 cm−1 corresponds to the bending vibration of the –CO functional group from the CO32− ion. Peaks at 1359 and 1049 cm−1 are attributed to the stretching vibration modes of CO32−. The peaks at 798 and 640 cm−1 indicate the out-of-plane and in-plane bending vibrations of CO32−, respectively. An additional peak at 2979 cm−1, present in both CDMHC and CDs, confirms the incorporation of CDs into MHC. This peak is attributed to sp3 hybridized C–H in CDs due to its thymol content, a major constituent of ajwain leaves, the source of the CDs.23 Peaks at 486, 470, and 490 cm−1 indicate the presence of Zn2+, Co2+, and Ni2+, respectively.13,24,25
Raman analysis, a precise and non-destructive technique, was employed to gain deeper insights into the chemical composition, crystal structure, and vibrational properties of the synthesized CDMHC (Fig. 1(b)). The prominent peaks observed in the Raman spectra confirm the presence of Co2(OH)2(CO3), Ni2(OH)2(CO3), and Zn5(OH)6(CO3)2, validating the chemical composition of the material. Low intensity bands for Zn5(OH)6(CO3)2 are observed since a very low amount of Zn precursor was added.
The Raman spectrum shows bands at 445 cm−1 and 523 cm−1 corresponding to Co–O stretching vibrations, while the band at 1046 cm−1 indicates hydroxyl deformation (out of plane bending mode) in cobalt hydroxyl carbonate (Co2(OH)2CO3). An intense band at 1469 cm−1 is attributed to the asymmetric stretching C–O vibration mode of CO32− ions having B1g symmetry. Other Raman active modes observed for carbonate ions are near 737 (B3g) and 794 (Ag) belonging to its bending mode. Additional unidentified bands at 309 cm−1 and 1167 cm−1 are also associated with Co2(OH)2CO3 vibrations.26–28 For nickel hydroxyl carbonate (Ni2(OH)2CO3), the Raman band at 1589 cm−1 corresponds to asymmetric stretching vibrations of CO32− ions, with other bands observed at 1243 cm−1, 1843 cm−1, and 2081 cm−1.25 Finally, shifts at 2366 cm−1 and 2932 cm−1 are characteristic of zinc hydroxyl carbonate (Zn5(CO3)2(OH)6), confirming its presence in the sample.29 The absence of a pronounced carbon peak can be attributed to the lower amount of CDs added (2.1 weight%).
XRD analysis was utilized to assess the crystallinity and phases present in the synthesized composite material, and to verify the incorporation of CDs into the MHC material. Fig. 1(c) illustrates the comparative XRD diffractograms of MHC and CDMHC over a 2θ range of 5–65° at a scan rate of 5° min−1. By matching the corresponding JCPDS numbers, it was established that both samples contain four distinct phases: Co2(OH)2CO3 – JCPDS-00-048-0083, Ni2(OH)2CO3 – JCPDS-00-038-0714, Zn5(CO3)2(OH)6 – JCPDS-00-019-1458, and characteristic peaks of the NF substrate/current collector. This confirmation validates the anticipated chemical composition of the synthesized material.
The orthorhombic crystal system of Co2(OH)2CO3 exhibits diffraction peaks at 18.56°, 33.82°, and 38.51°, corresponding to the (020), (221), and (412) planes, respectively. The peak at 14.08° can be attributed to the Ni2(OH)2CO3 plane. Additionally, the monoclinic crystal system of Zn5(CO3)2(OH)6, belonging to space group C2/m (space group no. 12), shows peaks at 16.92° (001 plane) and 38.32° (−511 plane). This confirms the mixed phase of MHC. Upon comparison, slight peak shifts were observed in CDMHC, with a new peak emerging at 11.34°.30 This peak corresponds to the characteristic XRD pattern of CDs, derived from dead-dry ajwain leaves with a high thymol content, which remains stable through processes like partial carbonization or torrefaction. This validates the presence of CDs in CDMHC. Moreover, the observed shifts towards lower angles in CDMHC suggest an expansion in crystallite size following CD incorporation. The expansion/increase in size is also confirmed from calculating the average crystallite size using the Scherrer formula as follows:
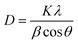 | (7) |
The crystallite size of MHC was determined to be 9.13 nm, while that of CDMHC measured 11.78 nm. Furthermore, an increase in intensity was observed for the major peaks corresponding to the (100), (001), and (020) planes. This enhancement suggests effective crystal growth in these phases, likely induced by the incorporation of CDs during hydrothermal synthesis. The CDs obtained exhibit blue-range photoluminescence and are estimated to have a diameter of 2–3 nm.22 The broad, weak peaks observed in the XRD pattern indicate low crystallinity and a high degree of amorphousness in the synthesized material, which hinders long-range order. This amorphous nature is due to the presence of carbonate and hydroxyl ions in the electrode material. These characteristics contribute to isotropic conduction, enhanced reversible specific capacity, electrochemical activity improved electrical and ionic conductivity, a higher number of active site defects, and increased electrochemical stability. Together, these attributes enhance the overall electrochemical performance of the electrode material for energy storage applications.31
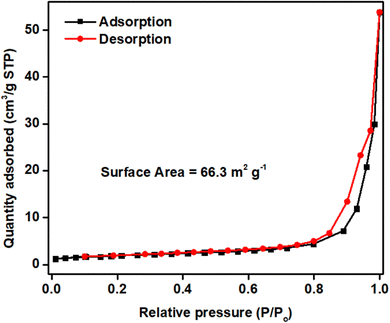
Brunauer–Emmett–Teller (BET) analysis was performed to analyze the specific surface area and porous architecture of the electrode material (Fig. 2). A hysteresis loop was observed in between 0.5 to 1.0 P/P0 indicating mesoporous nature of the electrode material with a surface area of 66.3 m2 g−1. This high surface area is attributed to the presence of carbonate and hydroxyl ions in the electrode material.
Surface morphology was analyzed with FE-SEM. Fig. 3(a) shows CDMHC grown on nickel foam at low magnification. Further magnifying at higher resolution of it revealed a porous flower-like structure with a single flower having a diameter of ∼2.642 μm (Fig. 3(b)). This morphology enhances the surface area and provides three-dimensional conducting channels, effectively shortening ion-diffusion pathways.
 | ||
| Fig. 3 FE-SEM analysis of CDMHC hydrothermally grown on NF (a) at lower magnification of 1 μm and (b) higher magnification of 200 nm. | ||
This morphology facilitates efficient ion transport and rapid diffusion of counter ions due to easy electrolyte penetration, promoting a fast charge–discharge process. It also enhances electrical conductivity with optimal incorporation of conductive materials, such as CDs. The abundant channels and pores support high-performance maintenance at elevated current densities, as they allow for sufficient ion diffusion and transport. Additionally, the structure mitigates mechanical stress, resulting in high cycling stability and prolonged lifespan.32 Consequently, this morphology significantly improves the material's overall charge storage capacity.
HR-TEM analysis was conducted to examine the detailed atomic structure of CDMHC. Fig. 4(a) presents a low magnification TEM image, showing the flower-like arrangement of petals. At a higher magnification of 50 nm (Fig. 4(b)), individual petals are observed. The petals contain evenly distributed pores, which enhance charge storage capacity by facilitating ion transport. Fig. 4(c) shows an HR-TEM image with dark spherical spots (2–3 nm), marked in yellow, indicating the presence of CDs. Separate HR-TEM images for CDs can be seen in Fig. S2 (ESI†). The CDs are synthesized of almost equal size where its average size lies between 2–3 nm. Fig. 4(d) displays lattice fringes with spacings of 0.15 nm, 0.27 nm, and 0.51 nm, corresponding to the (020), (221), and (412) planes of Co2(OH)2CO3, Ni2(OH)2CO3, and Zn5(CO3)2(OH)6, respectively.7 These measurements are consistent with the JCPDS data obtained from the XRD analysis (Co2(OH)2CO3) – 00-048-0083, Ni2(OH)2CO3 – 00-038-0714, (Zn5(CO3)2(OH)6 – 00-019-1458), thereby confirming the results. Fig. 4(e) presents a high-angle annular dark field (HAADF) image of a selected area of the CDMHC flower. Elemental mapping was performed on this area using scanning transmission electron microscope-energy dispersive spectroscopy (STEM-EDS) to analyze the distribution pattern of the elements. The mapping confirmed the presence of C, O, Co, Ni, and Zn, represented in red, yellow, orange, olive green, and dark green, respectively, as shown in Fig. 4(f)–(j), with uniform distribution. This analysis confirmed a flower-like morphology featuring a well-defined 3D charge-conducting pathway and uniformly distributed porosity, the incorporation of CDs, and matched lattice fringes with the expected MHC composition, and verified the presence and uniform distribution of the desired elements.
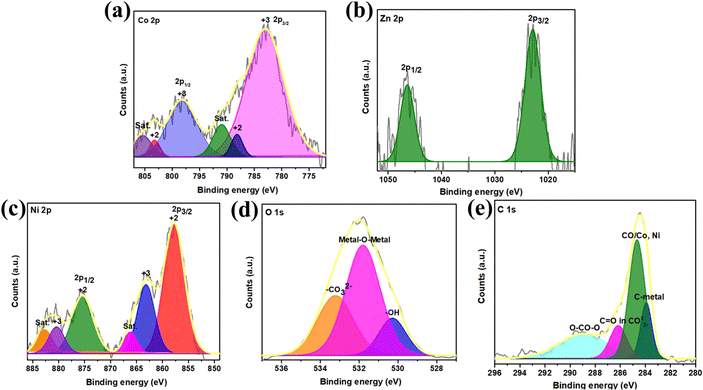
Following validation of the structural, chemical and morphological analysis of the electrode material, surface elemental constituents were examined using X-ray photoelectron spectroscopy (XPS) (Fig. 5). An overall survey of the surface elements is depicted in Fig. S3 (ESI†), confirming the presence of the desired elemental composition: Co 2p, Zn 2p, Ni 2p, O 1s, and C 1s. The Gaussian fitting method with Shirley background correction was employed to deconvolute all the peaks.
Fig. 5(a) displays the XPS spectra of Co 2p, with fitted peaks for Co 2p3/2 and Co 2p1/2 at 783 eV and 798 eV, respectively. These peaks represent Co in +2 and +3 oxidation states, with the +3 state being predominant. Satellite peaks appear at binding energies of 791 eV and 805 eV.33 In Fig. 5(b), the deconvoluted spectra of Zn 2p show peaks at 1022 eV and 1046 eV, corresponding to Zn 2p3/2 and Zn 2p1/2. Fig. 5(c) illustrates the Ni 2p emission spectrum, highlighting Ni 2p3/2 and Ni 2p1/2 at 857 eV and 875 eV, predominantly in the +2 state, with satellite peaks at 866 eV and 882 eV. The Gaussian-fitted spectrum of O 1s in Fig. 5(d) confirms –OH, –CO32−, and metal–O–metal bonds at 530 eV, 531 eV, and 533 eV, respectively. The C 1s spectrum in Fig. 5(e) indicates the presence of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in CO32− at 288 eV and C–metal bonds at 285 eV.7 Overall, the XPS analysis verifies the expected elemental composition, confirming the successful formation of metal hydroxyl carbonate.
O in CO32− at 288 eV and C–metal bonds at 285 eV.7 Overall, the XPS analysis verifies the expected elemental composition, confirming the successful formation of metal hydroxyl carbonate.
Thus, this comprehensive analysis substantiates the structural integrity and elemental composition of the CDMHC flower-like morphology, confirming the successful synthesis and uniform distribution of key components essential for enhanced charge storage capacity.
3.3. Electrochemical investigation of CDMHC
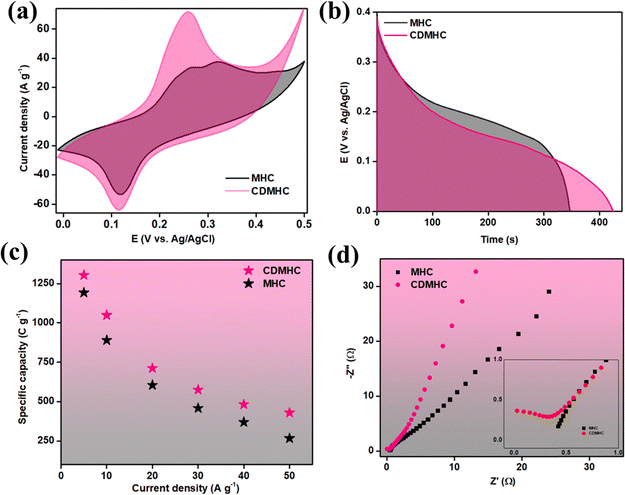
To optimize CDs incorporation, the weight ratio of CDs was varied and tested, with 5 mg of CDs being identified as optimal based on the electrochemical performance of the individual nanocomposites. The performance of four different compositions was compared using CV, GCD, and EIS methods (Fig. S4, ESI†). Fig. S4(a) (ESI†) shows comparative CV curves of different CDMHC compositions at a scan rate of 10 mV s−1. The MHC alone displayed a good area under the CV curve, indicating significant specific capacity. However, with the incorporation of 2.5 mg of CDs, the area decreased. Incorporation of 5 mg of CDs significantly increased the area under the curve compared to that of MHC alone, which then decreased with the addition of 10 mg of CDs, confirming the best performance with 5 mg of CDs (5CDMHC). Further analysis using the GCD technique at a constant current density of 5 A g−1 (Fig. S4(b), ESI†) yielded results that aligned well with the CV analysis. Finally, Fig. S4(c) (ESI†) presents the comparative Nyquist plot from EIS analysis, indicating different slopes in the low-frequency regime for the various compositions. These slopes reflect the Warburg resistance, which is the resistance faced by the electrolyte during diffusion through the electrode's porous structure.34 The most vertical slope in the Nyquist plot is observed for 5CDMHC, indicating the lowest resistance to electrolyte ion diffusion. This efficiency is due to optimal incorporation of CDs acting as connecting channels within the electrode material's microstructure. However, increasing the amount of CDs leads to higher electrode–electrolyte contact resistance, thereby diminishing the electrode material's electrochemical performance.35Fig. 6 compares the electrochemical performance of MHC alone and 5CDMHC, with 5CDMHC showing superior results. The enhanced area under both the CV and GCD plots (Fig. 6(a) and (b)) demonstrates an increase in the overall capacitive nature of the electrode material due to the incorporation of the optimized amount of CDs. The specific capacity was determined by calculating the area under the discharge profile of the GCD curve using the following formula:7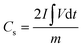 | (8) |
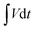 = integration area under the discharge curve, and m = mass of active material. The highest specific capacities recorded at 5 A g−1 for MHC and CDMHC were 1193 C g−1 and 1304 C g−1, respectively.
= integration area under the discharge curve, and m = mass of active material. The highest specific capacities recorded at 5 A g−1 for MHC and CDMHC were 1193 C g−1 and 1304 C g−1, respectively.
Fig. 6(c) shows the comparative specific capacity as a function of current density for MHC and 5CDMHC. Based on these results, 5CDMHC was identified as the optimal composition with the highest charge storage capacity. Hereafter, 5CDMHC is symbolized as CDMHC. Fig. 6(d) shows the comparative Nyquist plots for MHC and CDMHC. The appearance of a partial small semi-circle, characteristic of carbon-based materials at high frequencies, indicates the incorporation of CDs. This incorporation reduces both solution and diffusion resistance.
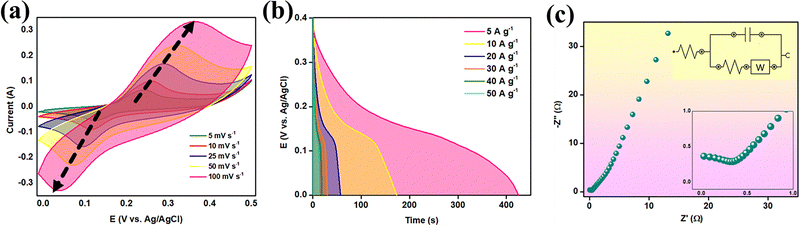
Fig. 7 illustrates the overall electrochemical performance of CDMHC. Fig. 7(a) displays the CV plot at scan rates of 5, 10, 25, 50, and 100 mV s−1. The presence of distinct anodic and cathodic peaks at all scan rates suggests a reversible faradaic process. At lower scan rates, there is enough time for the electrochemical redox process, which offers well-defined redox peaks. The persistence of these prominent peaks at the higher scan rate of 100 mV s−1 indicates the material's excellent redox reversibility even at elevated scan rates.36 As the scan rate increases, the cathodic peak shifts to a higher potential, while the anodic peak shifts to a lower potential. This shift in peak potential is related to the charge storage mechanism during the electrochemical process. At higher scan rates, the reduced time for electrode–electrolyte interaction limits electrolyte ion diffusion through the electrode material, leading to surface charge storage and causing the observed shift in redox peak potential.37 The electrochemical reaction involved can be written as follows:7
| MHC + 4OH− ⇌ 2MOOH + CO32− + 2H2O + 2e− | (9) |
Fig. 7(b) displays the discharge profiles from GCD scans at current densities of 5, 10, 20, 30, 40, and 50 A g−1. The specific capacities calculated from these profiles were 1304.4, 1048.4, 711.6, 574.0, 482.0, and 429.2 C g−1, respectively. The material maintains a notable specific capacity of 429.2 C g−1 even at a high current density of 50 A g−1, indicating excellent rate capability. This performance is attributed to the efficient ion diffusion pathways provided by the electrode material. Fig. 7(c) shows the EIS plot with the equivalent circuit diagram, demonstrating the supercapacitive behavior of the electrode material. The inset shows a partial semi-circle emerging in the high frequency region showing the property of the carbon-based material, and it can be witnessed that the incorporation of CDs improved the materials conductivity by dropping the internal resistance.
3.4. Investigation of the charge storage mechanism
To assess the ratio of capacitive and diffusion contribution of the CDMHC electrode, a thorough investigation of the charge storage mechanism was done.38 According to the power law equation,| i = aνb | (10) |
log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) i = log i = log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) a + b a + b![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) log log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ν ν | (11) |
This equation helps us to understand the relationship between the peak current (i) and the scan rate (v). In the equation, ‘a’ and ‘b’ are constants, with ‘b’ specifically being the power law exponent.
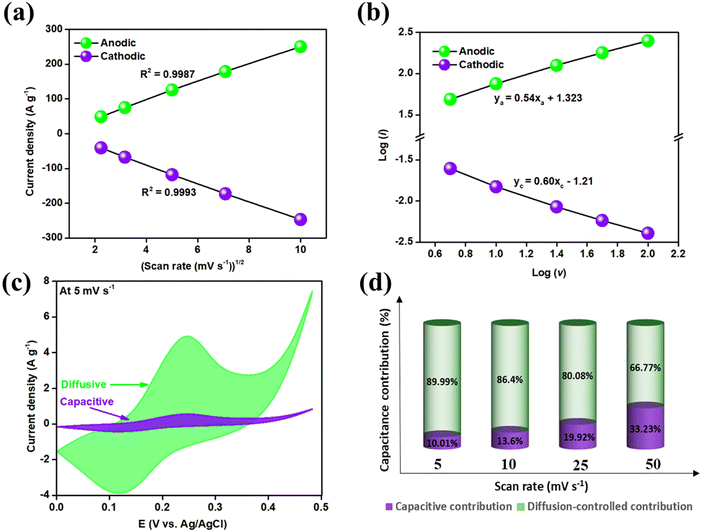
Fig. 8(a) plots the current density versus the square root of the scan rate, showing linear relationships for both cathodic and anodic peak currents. This linearity indicates that the electrochemical process of the electrode material is primarily diffusion-controlled.
Fig. 8(b) presents linearly fitted curves of log(i) versus log(v), with equations for anodic and cathodic processes given as ya = 0.54xa + 1.323 and yc = 0.6xc − 1.21, respectively. The power law exponents, ‘b’, are 0.54 for the anodic process and 0.6 for the cathodic process. These values, close to 0.5, suggest that the material predominantly exhibits diffusion-controlled or battery-type behavior. Although the process is mainly diffusion-controlled, there are also some capacitive contributions. The extent of these contributions is quantified using the following formula:39
| i(V) = k1ν + k2ν1/2 | (12) |
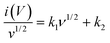 | (13) |
3.5. Electrochemical investigation of the CDMHC//AC hybrid device
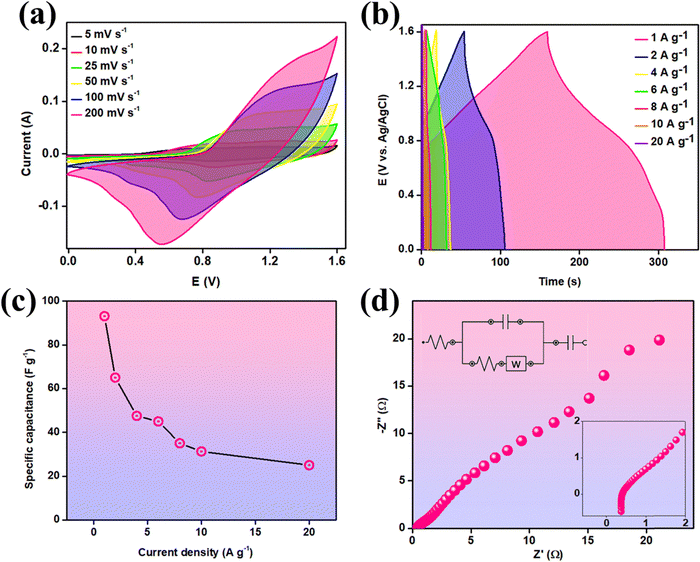
To assess the practical potential of the CDMHC electrode material, it was incorporated into a hybrid device as the positive electrode and AC deposited on NF as the negative electrode, referred to as CDMHC//AC. The AC electrode achieved a maximum specific capacitance of 200 F g−1 at 1 A g−1 (Fig. S5, ESI†). Based on the positive electrode's performance, the AC mass loading was optimized using eqn (S1) (ESI†). Separate CV for AC and CDMHC can be seen in Fig. S6(a) (ESI†). After device assembly and fabrication of the device, the potential window was slowly increased from 1.2 to 1.6 V for covering the maximum area under the CV curve (Fig. S6(b), ESI†). The electrochemical performance of the device was evaluated using CV, GCD, and EIS techniques, with 6 M KOH as the electrolyte. CV measurements were conducted at scan rates of 5, 10, 25, 50, 100, and 200 mV s−1 (Fig. 9(a)). The device, combining metal-based and carbon-based materials, demonstrated promising performance within a potential window of 0 to 1.6 V. As the scan rate increased, the area under the CV curve grew as expected, maintaining this trend up to 200 mV s−1 without altering the curve's shape, indicating stable performance during rapid scans. The CV curves deviated from a rectangular shape, reflecting the diffusion-controlled or battery-type charge storage mechanism of the material.37GCD curves were recorded at current densities ranging from 1 to 20 A g−1 within a potential window of 0–1.6 V (Fig. 9(b)). The highest specific capacitance achieved was 93 F g−1 at 1 A g−1, with a minimal potential drop. Even at 20 A g−1, the device maintained a specific capacitance of 25 F g−1. The charge and discharge profiles at various current densities were nearly symmetrical, demonstrating excellent electrochemical reversibility and coulombic efficiency. Fig. 9(c) shows the variation in specific capacitance with increasing current density. The impedance of the hybrid device is illustrated in Fig. 9(d), with an Rs value of 0.34 Ω (inset).
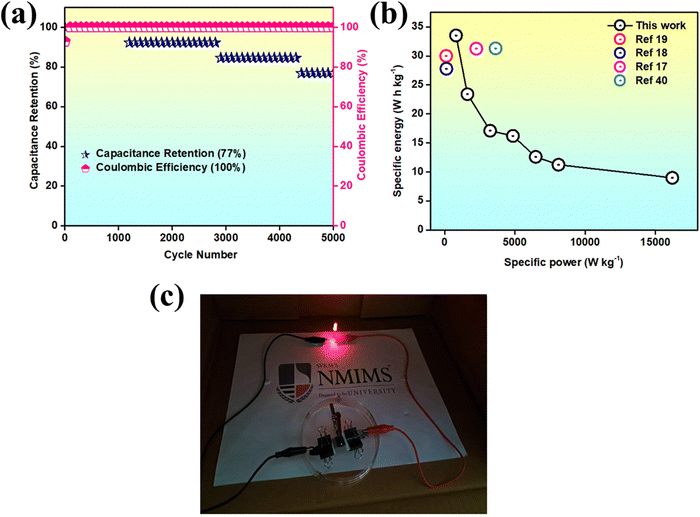
To assess the cycling stability, the device was subjected to 5000 continuous charge–discharge cycles at a current density of 5 A g−1 (Fig. 10(a)). It demonstrated commendable stability, with 77% capacitance retention and 100% coulombic efficiency. This occurs because, at lower current densities, the prolonged interaction between the electrode and electrolyte imposes greater stress on the electrode material, accelerating its degradation. This enhanced performance is largely attributed to the incorporation of CDs, which create conductive pathways that facilitate efficient and quick counter-ion diffusion. The specific power and energy were calculated, with the device achieving a maximum energy density of 33.5 W h kg−1 at a power density of 810 W kg−1, and a peak specific power of 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 W kg−1 at an energy density of 9 W h kg−1. These values were determined using the following formula:43
200 W kg−1 at an energy density of 9 W h kg−1. These values were determined using the following formula:43
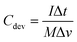 | (14) |
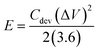 | (15) |
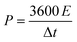 | (16) |
Fig. 10(b) presents the specific energy vs. specific power plot, which, along with results from related literature, highlights the device's exemplary performance and potential. This remarkable performance can be attributed to several factors:
(i) Binder-free electrode fabrication for significantly improved electrical conductivity.
(ii) The flower-like morphology increases the electrochemical accessible surface area for efficient interactions between the electrode and electrolyte, enhancing overall performance.
(iii) CDs derived from dried ajwain leaves create effective conducting pathways within the electrode material. These pathways improved the charge conduction, leading to enhanced electrochemical performance.
(iv) The inclusion of metal ions (Co, Ni and Zn) in the electrode material facilitates rapid redox reactions. These ions work together to improve the efficiency of charge storage.
(v) The carbonate (CO32−) and hydroxide (OH−) ions are easily available within the material's bulk. This availability supports efficient redox processes, which is crucial for enhanced material's performance in electrochemical applications.
Fig. 10(c) shows a prototype of the device illuminating an LED bulb. This is accomplished by connecting two hybrid devices in series. The bulb remained illuminated for a total duration of 5 minutes. The cost-effective and simple synthesis of the CDs and its integration with MHC contribute to the device's performance, highlighting its potential as a supercapacitor with battery-type charge storage capabilities.
3.6. Post cycling analysis
After subjecting the device to 5000 consecutive charge–discharge cycles at 5 A g−1, the positive electrode material (CDMHC) was analyzed to investigate any resulting changes on the electrode surface. Based on the stability test results, it was observed that the electrode material exhibited a capacitance retention of only 77%, indicating phase change in the material. This change could be attributed to several factors. The sustained operation of the nickel foam electrode leads to a decline in its charge storage capability, primarily stemming from alterations in the material's chemical makeup. Within the NF's porous architecture, continuous cycling converts hydroxyl carbonates into stable, unreactive oxides, and leaves some active material in a partially charged state, fostering detrimental faradaic side reactions.44 Furthermore, prolonged exposure to elevated current densities at the electrode surface can induce structural damage.45 A contributing factor is also the electrolyte concentration; in this instance, the 6 M KOH solution, whose high concentration causes electrolytic counter ions to promote irreversible chemical transformations in the active material. The chemical changes can be observed in comparative FT-IR analysis as shown in Fig. 11(a). A prominent peak around 3400 cm−1 observed initially disappeared after the stability test. This disappearance can be attributed to the consumption of hydroxyl ions within the bulk electrode materials during the electrochemical process, leading to the formation of metal oxides. The presence of these metal oxides is confirmed by new peaks observed for post cycling analysis. New distinct peaks were identified: a peak at 1007 cm−1 corresponding to nickel oxide, and another at 832 cm−1 indicating nickel hydroxide. Additionally, peaks at 663 cm−1 and 702 cm−1 were observed, indicating the presence of cobalt oxide and zinc oxide, respectively.46,47In Fig. 11(b), the comparative XRD diffraction pattern of the electrode material is presented. Most peaks exhibit similarities, such as those at 2θ angles around 33.82°, 14.08°, and 38.32° for Co2(OH)2CO3, Ni2(OH)2CO3, and Zn5(CO3)2(OH)6, respectively. However, several new peaks were also observed, notably at 2θ angles of 22°, 30.7°, and 36.3°, corresponding to nickel hydroxide, zinc oxide, and cobalt oxide, respectively.48–50 Prolonged exposure to 6 M KOH electrolytic solution and vigorous electrochemical reactions caused the phase change and ultimately resulted in performance fading.
The overall reaction leading to the conversion of MHC to metal oxides can be represented as follows:
| MHC + 4OH− ⇌ 2MOOH + CO32− + 2H2O + 2e− | (17) |
| MOOH + 4OH− → MO + 2H2O + e− | (18) |
A comparative Nyquist plot for before and after the stability tests is shown in Fig. 11(c). The Rs values before and after the stability test were found to be 0.34 and 0.45 Ω, respectively. This is due to exposure of the electrode material for the long term-charge discharge process, which leads to an increase in resistance of the diffusion of ions across the electrode material.51
4. Conclusions
In this study, CDs were successfully produced from dry ajwain leaves using a straightforward torrefaction and ultrasonic method. These CDs were then effectively integrated into MHC and verified for their composition and successful incorporation. The unique flower-like structure of the electrode material notably expanded its surface area, enhancing its electrochemical performance. Electrochemical tests using a three-electrode system showed that including CDs in MHC raised the specific capacity to 1304.4 C g−1 (3261 F g−1), an increase of 111.2 C g−1 (278 F g−1) compared to MHC alone, which was due to the conductive pathways established by the CDs. A device was constructed with CDMHC as the cathode and AC as the anode, achieving a peak specific capacitance of 93 F g−1 at 1 A g−1. This device maintained 77% retention and demonstrated 100% coulombic efficiency at 5 A g−1. The CDMHC//AC hybrid device delivered a specific energy of 33.5 W h kg−1 and a specific power of 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 W kg−1. Future work on material synthesis and manufacturing scaling promises to extend the applications of these materials beyond traditional electronics to areas like renewable energy, consumer devices, electric vehicles, defence, and medical technologies. This research underscores the promise of CD-incorporated MHC composites for advancing energy storage and addressing global energy challenges.
200 W kg−1. Future work on material synthesis and manufacturing scaling promises to extend the applications of these materials beyond traditional electronics to areas like renewable energy, consumer devices, electric vehicles, defence, and medical technologies. This research underscores the promise of CD-incorporated MHC composites for advancing energy storage and addressing global energy challenges.
Author contributions
Tanzeem Fatema: conceptualization, data curation, formal analysis, investigation, methodology, software, writing – original draft; Gaurav Singh – visualization, writing – review & editing, validation; Darshna Potphode – conceptualization, data curation, formal analysis, funding acquisition, methodology, project administration, resources, supervision, validation.Conflicts of interest
The authors have no competing interests to declare.Data availability
All data generated or analyzed during this study will be made available upon request.Acknowledgements
This work was supported by the NMIMS University SEED Grant (I/O no. 403080) research fund. We would like to thank Dr Pravin Tirmali from IR Technology Services Pvt. Ltd., Navi Mumbai, Maharashtra for providing the XRD data measurement and analysis of the CDMHC sample.References
- A. A. Kebede, T. Kalogiannis, J. Van Mierlo and M. Berecibar, Renewable Sustainable Energy Rev., 2022, 159, 112213 CrossRef CAS.
- P. Simon and Y. Gogotsi, Nat. Mater., 2008, 7, 845–854 CrossRef CAS PubMed.
- M. Pershaanaa, S. Bashir, S. Ramesh and K. Ramesh, J. Energy Storage, 2022, 50, 104599 CrossRef.
- P. Simon and Y. Gogotsi, Nat. Mater., 2020, 19, 1151–1163 CrossRef CAS PubMed.
- G. Wang, L. Zhang and J. Zhang, Chem. Soc. Rev., 2012, 41, 797–828 RSC.
- S. Subhadarshini, N. K. Peyada, D. K. Goswami and N. Ch. Das, Energy Fuels, 2024, 38, 11455–11493 CrossRef CAS.
- D. Potphode, M. S. Sayed, T. Lama Tamang and J. J. Shim, Chem. Eng. J., 2019, 378, 122129 CrossRef CAS.
- X. Qiu, N. Wang, Z. Wang, F. Wang and Y. Wang, Angew. Chem., 2021, 133, 9696–9703 CrossRef.
- M. Manikandan, E. Manikandan, V. Swetha, S. Kurpaa, S. Vijay and V. Kiruthika, Sci. Rep., 2024, 14, 10821 CrossRef CAS PubMed.
- M. T. Ahsan, M. Usman, Z. Ali, S. Javed, R. Ali, M. U. Farooq, M. A. Akram and A. Mahmood, Front. Chem., 2020, 8, 487 CrossRef CAS PubMed.
- C. Wu, L. Chen, X. Lou, M. Ding and C. Jia, Front. Chem., 2018, 6, 597 CrossRef CAS PubMed.
- M. Sharma and A. Gaur, J. Phys. Chem. C, 2020, 124, 9–16 CrossRef CAS.
- Y. Zhao, H. Ma, S. Huang, X. Zhang, M. Xia, Y. Tang and Z. F. Ma, ACS Appl. Mater. Interfaces, 2016, 8, 22997–23005 CrossRef CAS PubMed.
- H. Lv, X. Gao, Q. Xu, H. Liu, Y. G. Wang and Y. Xia, ACS Appl. Mater. Interfaces, 2017, 9, 40394–40403 CrossRef CAS PubMed.
- G. Wei, X. Xu, J. Liu, K. Du, J. Du, S. Zhang, C. An, J. Zhang and Z. Wang, Mater. Lett., 2017, 186, 131–134 CrossRef CAS.
- A. B. Ganganboina, E. Y. Park and R. A. Doong, Nanoscale, 2020, 12, 16944–16955 RSC.
- Y. Zhu, Z. Wu, M. Jing, H. Hou, Y. Yang, Y. Zhang, X. Yang, W. Song, X. Jia and X. Ji, J. Mater. Chem. A, 2015, 3, 866–877 RSC.
- S. Sahoo, A. K. Satpati, P. K. Sahoo and P. D. Naik, ACS Omega, 2018, 3, 17936–17946 CrossRef CAS PubMed.
- S. Zhu, Y. Song, X. Zhao, J. Shao, J. Zhang and B. Yang, Nano Res., 2015, 8, 355–381 CrossRef CAS.
- V. C. Hoang, K. Dave and V. G. Gomes, Nano Energy, 2019, 66, 104093 CrossRef CAS.
- Q. Zhang, H. Liu, Y. Xu and L. Wang, J. Solid State Chem., 2018, 267, 76–84 CrossRef CAS.
- A. Gaurav, A. Jain and S. K. Tripathi, Materials, 2022, 15, 7888 CrossRef CAS PubMed.
- S. Kumari, R. V. Kumaraswamy, R. C. Choudhary, S. S. Sharma, A. Pal, R. Raliya, P. Biswas and V. Saharan, Sci. Rep., 2018, 8, 6650 CrossRef PubMed.
- J. Winiarski, W. Tylus, K. Winiarska, I. Szczygieł and B. Szczygieł, J. Spectrosc., 2018, 2018, 2079278 Search PubMed.
- L. A. Saghatforoush, M. Hasanzadeh, S. Sanati and R. Mehdizadeh, Bull. Korean Chem. Soc., 2012, 33, 2613–2618 CrossRef CAS.
- J. Yang, H. Cheng and R. L. Frost, Spectrochim. Acta, Part A, 2011, 78, 420–428 CrossRef PubMed.
- J. F. Alves, H. G. M. Edwards, A. Korsakov and L. F. C. deOliveira, Minerals, 2023, 13, 1358 CrossRef CAS.
- R. L. Frost, W. N. Martens, L. Rintoul, E. Mahmutagic and J. T. Kloprogge, J. Raman Spectrosc., 2002, 33, 252–259 CrossRef CAS.
- M. C. Hales and R. L. Frost, Polyhedron, 2007, 26, 4955–4962 CrossRef CAS.
- M. K. Trivedi, S. Patil, R. K. Mishra and S. Jana, J. Mol. Pharm. Org. Process Res., 2015, 3, 127 CAS.
- Z. Zhao, H. Chen, W. Zhang, S. Yi, H. Chen, Z. Su, B. Niu, Y. Zhang and D. Long, Mater. Adv., 2023, 4, 835–867 RSC.
- A. Mukhopadhyay and B. W. Sheldon, Prog. Mater. Sci., 2014, 63, 58–116 CrossRef CAS.
- S. A. Khayatian, A. Kompany, N. Shahtahmassebi and A. Khorsand Zak, J. Inorg. Organomet. Polym. Mater., 2018, 28, 2677–2688 CrossRef CAS.
- A. C. Lazanas and M. I. Prodromidis, ACS Meas. Sci. Au, 2023, 3, 162–193 CrossRef CAS PubMed.
- O. Sangabathula, M. Kandasamy, B. Chakraborty and C. S. Sharma, J. Energy Storage, 2023, 65, 107274 CrossRef.
- I. Shaheen, K. S. Ahmad, C. Zequine, R. K. Gupta, A. G. Thomas and M. A. Malik, RSC Adv., 2021, 11, 23374–23384 RSC.
- L. Zhang, K. N. Hui, K. S. Hui, X. Chen, R. Chen and H. Lee, Int. J. Hydrogen Energy, 2016, 41, 9443–9453 CrossRef CAS.
- F. H. Gourji, T. Rajaramanan, A. Kishore, M. Heggertveit and D. Velauthapillai, ACS Omega, 2023, 8, 23446–23456 CrossRef CAS PubMed.
- F. Fei, H. Zhou, J. Lin and M. Kang, Mater. Today Commun., 2023, 36, 106797 CrossRef CAS.
- S. Kumar, B. K. Satpathy and D. Pradhan, Mater. Adv., 2024, 5, 2271 RSC.
- N. Poompiew, P. Pattananuwat and P. Potiyaraj, ACS Omega, 2021, 6, 25138–25150 CrossRef CAS PubMed.
- C. Li, G. Ren, Z. Zhang, L. Guan and J. Tao, Mater. Res. Bull., 2024, 179, 112969 CrossRef CAS.
- B. Xiao, W. Zhu, Z. Li, J. Zhu, X. Zhu and G. Pezzotti, R. Soc. Open Sci., 2018, 5, 180867 CrossRef PubMed.
- W. Shang, W. Yu, X. Xiao, Y. Ma, Y. He, Z. Zhao and P. Tan, Adv. Power Mater., 2023, 2, 100075 Search PubMed.
- X. Y. Yao and M. G. Pecht, IEEE Access, 2019, 7, 24082–24095 Search PubMed.
- S. Joghee, P. Ganeshan, A. Vincent and S. I. Hong, Bionanoscience, 2019, 9, 141–154 CrossRef.
- A. N. Naveen and S. Selladurai, Electrochim. Acta, 2014, 125, 404–414 CrossRef CAS.
- H. Cai, Q. Wei, H. Xiao, H. Liu and J. Wang, J. Mater. Sci.: Mater. Electron., 2020, 31, 7606–7615 CrossRef CAS.
- A. Kaleva, T. Tassaing, V. Saarimaa, G. Le Bourdon, P. Väisänen, A. Markkula and E. Levänen, Corros. Sci., 2020, 174, 108850 CrossRef CAS.
- V. N. Kalpana, B. A. S. Kataru, N. Sravani, T. Vigneshwari, A. Panneerselvam and V. Devi Rajeswari, OpenNano, 2018, 3, 48–55 CrossRef.
- Q. Wang, X. Tian, Q. Gao, X. Zhang, K. Rong, M. Cao, L. Han and W. Fan, J. Mater. Res. Technol., 2023, 24, 909–917 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5tc02199e |
| This journal is © The Royal Society of Chemistry 2025 |