Investigating the angle-dependent emission of dye-doped 3D luminescent opals: optical property insights†
Weronika
Zając
ab,
Maciej
Czajkowski
 *a,
Bartłomiej
Potaniec
*a,
Bartłomiej
Potaniec
 a,
Maria
Zdończyk
ab and
Joanna
Cybińska
*ab
a,
Maria
Zdończyk
ab and
Joanna
Cybińska
*ab
aŁukasiewicz Research Network – PORT Polish Center for Technology Development, ul. Stabłowicka 147, 54-066 Wrocław, Poland. E-mail: maciej.czajkowski@port.lukasiewicz.gov.pl
bUniversity of Wrocław, Faculty of Chemistry, ul. Joliot-Curie 14, 50-383 Wrocław, Poland. E-mail: joanna.cybinska@uwr.edu.pl
First published on 16th May 2025
Abstract
This study explores the synthesis protocol and examines the optical properties of dye-doped poly(methyl methacrylate) (PMMA) spheres, self-assembled into 3D colloidal photonic crystals. Utilizing a variety of luminescent dyes (POPOP, coumarin 6, DPP derivative, and DCM), emitting across the visible spectrum, this research proposes the incorporation of these dyes into PMMA spheres at the stage of the synthesis and use of the latter to produce luminescent 3D opals. The results of the study, which include comprehensive photoluminescence characterization, including quantum yield (QY) determination and angle-dependent emission analysis, provide a deep understanding of the integration process and the resulting photophysical properties. In addition, the effect of the dye addition on the size of the spheres and their monodispersity was evaluated. A careful choice of dyes and synthesis conditions in this study resulted in obtaining the materials that exhibit angle-dependent emission in different spectral ranges, demonstrating the versatility of the synthesis approach. Adjusting the sphere size to match specific dye emission wavelengths enabled the effective alignment of photonic band gap positions with the emission spectra, resulting in emission color shifts with the viewing angle. These findings significantly contribute to the advancement of the design of functional materials with tailored optical properties. Moreover, the results highlight the potential of these materials for advanced optical applications, particularly in anti-counterfeiting material technologies, and provide a versatile approach to designing materials with tunable optical properties.
1. Introduction
Photonic crystals (PCs) are a remarkably interesting and deeply studied group of modern optical materials due to their fascinating optical properties, including opalescence. One method of their preparation involves the self-assembly of uniformly sized spheres from colloids. The resulting structures, which exhibit a periodic refractive index distribution, are known as colloidal crystals or artificial opals.1The synthesis of monodisperse polymers or inorganic spheres of controlled size is a relatively easy process, and it can be performed by the use of monomers, such as styrene,2,3 methyl methacrylate,4–6 or oxide precursors, including silica.7,8 Such a method, by modifying the size of the spheres during synthesis, allows the position of the photonic band gap to be adjusted.4,7 Furthermore, doping PCs with luminescent materials enables the fabrication of functional materials with distinctive optical properties.9–11
The spatial pattern of the structure of colloidal crystals enables them to modulate the emission characteristics of their dopants in a manner that is distinct from other materials. An illustrative example of a phenomenon is the dependence of the observed emission color on the viewing angle,2,3,6,12,13 which is related to the angle dependence of the photonic band gap position of the PC.
Doping of colloidal crystals with an organic dye can be achieved in two different ways: firstly, by soaking of a synthesized opal in a dye solution14,15 and secondly, by introducing a dopant during the sphere synthesis.5,6,12,16 According to the literature, the second method has more advantages.12 Ensuring good miscibility of the dye with the matrix of the spheres and its precursor can help to avoid dye aggregation, which causes fluorescence quenching in many organic dyes. However, it should be emphasized that the effect of the presence of the dye during the synthesis of the spheres on the parameters obtained, such as their size, monodispersity or zeta potential, can also be unpredictable. Therefore, it is important to carry out further empirical investigations into the fabrication processes of the dye-doped polymeric spheres.
Many organic luminescent dyes, which possess high Quantum Yield (QY) in solution, such as rhodamine 6G (R6G), exhibiting the QY of 95% in ethanol solution, show very low QY in the solid state. The question arises about their properties and QY values in polymeric spheres. It has been demonstrated that the QY of the dye-doped opals can be strongly dependent on the dye concentration.17 The observed effect can be explained in terms of the form of the luminescent dye within the opal. The molecules can be either isolated, aggregated, or a mixture of both forms. In the latter case, values of QYs are expected to be in the intermediate range, as shown in ref. 17.
Dye-doped photonic crystals were already prepared using rhodamine 6G (R6G),14 rhodamine B,15,18 sulforhodamine B,19 and Kiton Red S,20 as well as using dyes from other groups, like fluorescein isothiocyanate,2 DCJTB,21 perylene diimides,12,22 coumarin 6,19 coumarin 334,23 pyrene,23 Nile Red,24 tetraphenylporphyrin,24 ethyl eosin,17 NFTPE and TPE.6 Unfortunately, despite the numerous reports, the measurements of the photoluminescence QY of the dyes in photonic crystals under investigation have received only limited attention in the literature.
The potential of PCs to be applied in a variety of areas is significant due to their interesting optical properties and wide availability. For instance, the use of PCs in the fabrication of security features has already been demonstrated in several novel approaches.25–27 However, the possibility of modifying the optical properties of opals during their preparation allows the design of completely new ones, including angle-dependent emission properties. The doping of these materials has already allowed the fabrication of a new type of ink,28 a low-threshold laser,29 a component of a photoelectric device, and a gas17,30 or an ionic sensor.31
This paper presents a step-by-step method for the synthesis of monodisperse luminescent dye-doped poly(methyl methacrylate) (PMMA) spheres and the preparation of 3D luminescent colloidal crystals thereof. The dyes have been selected to ensure that their emission spectra cover different regions of the visible spectrum. A comprehensive photoluminescence characterization, including absolute QY determination, has been performed to study the form that the dye dopants take in the PMMA spheres and their performance as light emitters. Then, a significant emphasis was placed on the adjustment of the size of the dye-doped spheres to achieve the angle-dependent emission phenomenon. Part of the obtained materials, besides efficient photoluminescence, exhibited distinct angle-dependent colors under UV excitation. Proposed materials can be applied as main components of authentication marks, where their unique visual properties can be easily verified. The investigations contribute to the materials science of luminescent dye-doped colloidal photonic crystals, which have the potential to be used in anti-counterfeiting solutions and advanced optical devices.
2. Experimental
2.1. Materials
3-(2-Benzothiazolyl)-7-(diethylamino)coumarin (coumarin 6, 98%), 4-(dicyanomethylene)-2-methyl-6-(4-dimethylaminostyryl)-4H-pyran (DCM, 98%), 1,4-bis(5-phenyl-2-oxazolyl)benzene (POPOP, 98%), and 2,2′-azobis(2-methylpropionamidine) dihydrochloride (AAPH, 97%) were obtained from Sigma-Aldrich. Methyl methacrylate (MMA, 98%) was purchased from Acros Organics. All the reagents were used without further purification. Deionized water from a water deionizer, HLP5UV (Hydrolab), having conductivity not higher than 0.1 μS cm−1 was used.The syntheses of (E)-3-(4-(dimethylamino)phenyl)-1-(2-hydroxyphenyl)prop-2-en-1-one) (2′OH4NDMe-chalcone) and 1,4-diketo-2,5-diethyl-3,6-diphenylpyrrolo-[3,4-c]-pyrrole (DPP) were performed according to the modified procedure of another dye32–34 and are described in the ESI.† The structures of the compounds were confirmed by NMR and MS spectroscopies (Fig. S1–S4, ESI†).
2.2. Methods
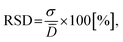 | (1) |
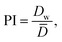 | (2) |
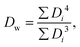 | (3) |
![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) is the average sphere diameter and Di is the diameter of the i-th sphere. As a rule, samples with RSD < 20%4 and with PI close to 1.035 are considered to be monodisperse.
is the average sphere diameter and Di is the diameter of the i-th sphere. As a rule, samples with RSD < 20%4 and with PI close to 1.035 are considered to be monodisperse.
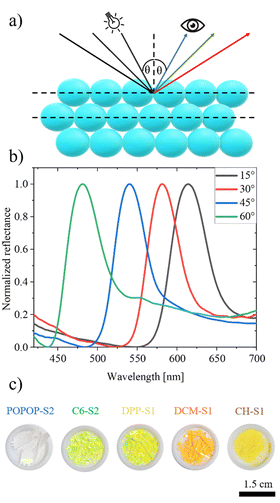
The system consisted also of a height-adjustable stage, where the samples of the 3D colloidal crystals were placed. Nearly plane-parallel samples of the fabricated 3D opals were placed with the position of their top adjusted to the axis of rotation of the arms. Thus, the plane (111) of the colloidal crystal was in the plane of the sample. During the measurements, the angular positions of the detector and the light source were changed in a range from 10° to 70°, symmetrically, with respect to the normal to the sample and the spectra were collected. A distinct spectral band shifting with the angle of the incidence/detection indicated the Bragg reflection.
The emission and excitation spectra of the dye solutions and dye-doped spheres were measured using the FLS980 spectrofluorimeter equipped with a xenon lamp Xe1 (Edinburgh Instruments). Ethyl acetate (EA) was chosen as a solvent for dye solutions, because of good or sufficient solubility of the selected dyes. MMA was not used for this purpose, due to its potentially destructive effect on cuvettes and components of measuring apparatus. The QYs of the dye-doped spheres and the studied dyes in the solid state were measured in the integrating sphere assembly F-M01 (Edinburgh Instruments) dedicated to the FLS980 spectrofluorimeter. Decay time measurements were conducted using the FLS980 spectrofluorometer (Edinburgh Instruments) equipped with 360 nm and 450 nm laser diodes.
The observed emission spectra were measured at different detection angles of the fabricated 3D dye-doped PMMA colloidal crystals in a modified self-made setup. This setup was adjusted from the one used for Optical Bragg reflection measurements. A CCD camera, QE65 Pro (Ocean Optics), equipped with an optical fiber was used as a detector and mounted on the rotating arm. The light source of the UV lamp VL-6.LC (Vilber Lourmat) with a short pass filter cutting off the spectrum below the wavelength λ = 395 nm was used to excite the samples containing POPOP dye. The Nova Pro diode laser (RGB Lasersystems GmbH) was used to excite coumarin 6, DPP and DCM samples with a light output power of 75 mW at λ = 405 nm.
Additional information on the measurements can be found in the ESI.†
3. Results and discussion
3.1. Synthesis and characterization of PMMA spheres and 3D colloidal crystals
Initially, the syntheses of the PMMA particles with varying average size (samples S1–S4) were performed utilizing a methodology documented in the literature.36Next, a similar series of colloidal crystals doped with selected luminescent dyes (Fig. 1) were prepared. The selected dyes exhibited good solubility in the monomer. Moreover, the primary criterion for the selection of the POPOP, coumarin 6, DPP, and DCM dyes was high photoluminescence QY. The exception was made for 2′OH4NDMe-chalcone, which was chosen because of the aggregation induced emission (AIE) phenomenon. It has a higher quantum yield in the solid state – equal to 18.5%37 – than in solvents – in the case of which it is less than 1% – in various solvents.38 The dye was chosen for a purpose to act as a direct visual indicator of the aggregated or isolated form of the dye in the PMMA matrix. Additionally, the dyes were selected to cover different spectral ranges, and the size of the spheres was modified to achieve unique optical effects.
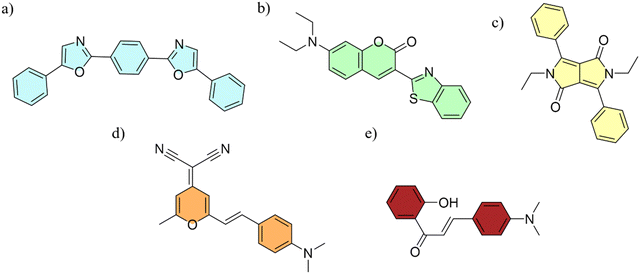 | ||
| Fig. 1 Chemical structures of the selected dyes used as dopants in the synthesized PMMA spheres: (a) POPOP, (b) coumarin 6, (c) DPP, (d) DCM, and (e) 2′OH4NDMe-chalcone. | ||
The obtained colloidal photonic crystal samples doped with the dyes and possessing varying average size were named as follows: POPOP-S1 – POPOP-S3 (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) = 134, 169, and 210 nm) – for the POPOP samples, C6-S1 and C6-S2 (
= 134, 169, and 210 nm) – for the POPOP samples, C6-S1 and C6-S2 (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) = 195 and 274 nm) – for the coumarin 6 samples, DPP-S1 (
= 195 and 274 nm) – for the coumarin 6 samples, DPP-S1 (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) = 267 nm) – for the DPP sample, DCM-S1 and DCM-S2 (
= 267 nm) – for the DPP sample, DCM-S1 and DCM-S2 (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) = 276 and 330 nm) – for the DCM samples and CH-S1 (
= 276 and 330 nm) – for the DCM samples and CH-S1 (![[D with combining macron]](https://www.rsc.org/images/entities/i_char_0044_0304.gif) = 288 nm) – for the 2′OH4NDMe-chalcone sample. The parameters of the syntheses are presented in Table S1 (ESI†).
= 288 nm) – for the 2′OH4NDMe-chalcone sample. The parameters of the syntheses are presented in Table S1 (ESI†).
The SEM results showed that the synthesized particles have a spherical shape (Fig. 2). The majority of the PCs obtained exhibited a hexagonal lattice ordering (Fig. 2a). However, the DPP-S1 and DCM-S1 samples exhibited also a square lattice ordering in certain areas (see for example in the inset in Fig. 2b). The average diameter of the undoped PMMA spheres was in the range of 215–320 nm (S1–S4), whereas in the case of the spheres doped with the luminescent dyes – in the range of 135–330 nm. Standard deviation and RSD and PI values of the spheres’ diameter measurements for all of the syntheses are given in Table S2 (ESI†). Furthermore, the size distribution of the obtained spheres is depicted by a series of histograms presented in Fig. S5 (ESI†).
Optical Bragg reflectance measurements were carried out for a vast majority of the samples in the form of plane-parallel pieces of 3D colloidal crystals, with an exception of the sample POPOP-S1, omitted due to a limited spectral range of the spectrophotometer. According to the literature, modified Bragg's equation could be used for experimental determination of the average diameter of the spheres and the average refractive index of the hexagonally-packed 3D opal (navg). This can be expressed by means of the following equation (eqn (4)):4
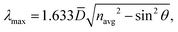 | (4) |
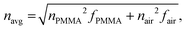 | (5) |
An example of a series of reflection spectra measured according to the scheme shown in Fig. 3a and used to determine the size of the spheres in an example sample of 3D opal is shown in Fig. 3b. In the obtained spectra, it is apparent that as the value of the incidence angle θ increases, the reflection band shifts toward lower wavelengths.
In addition, as the diameter of the spheres forming the opal increases, the reflection band shifts toward a higher wavelength. Furthermore, the corresponding macroscopic photographs of the obtained dye-doped photonic crystal flakes are shown in Fig. 3c.
The obtained results allowed plotting λmax2versus sin2![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) θ dependence, related to the modified form of Bragg's formula (eqn (4)), for the studied samples (Fig. S6, ESI†). Based on linear fit parameters it was possible to calculate the average diameter of the spheres and the average refractive index of the opal. For each of the syntheses, nearly linear dependence was found. The SEM observations confirmed that the prepared spheres possess low polydispersity in size, making it possible to form a regular crystal lattice in close-packed 3D colloidal crystals. Almost all of the average refractive index values determined by the optical method were close with respect to that expected for perfect close-packed hexagonal fcc 3D colloidal crystals. Moreover, the obtained statistical results illustrate that the doping of the spheres at the stage of their synthesis did not affect the obtained quality of spheres in terms of RSD parameter. The only exception where RSD > 20% is the DCM-S2 sample. However, the opal based on these spheres still had a lattice arrangement, which allowed observation of the Bragg reflection band.
θ dependence, related to the modified form of Bragg's formula (eqn (4)), for the studied samples (Fig. S6, ESI†). Based on linear fit parameters it was possible to calculate the average diameter of the spheres and the average refractive index of the opal. For each of the syntheses, nearly linear dependence was found. The SEM observations confirmed that the prepared spheres possess low polydispersity in size, making it possible to form a regular crystal lattice in close-packed 3D colloidal crystals. Almost all of the average refractive index values determined by the optical method were close with respect to that expected for perfect close-packed hexagonal fcc 3D colloidal crystals. Moreover, the obtained statistical results illustrate that the doping of the spheres at the stage of their synthesis did not affect the obtained quality of spheres in terms of RSD parameter. The only exception where RSD > 20% is the DCM-S2 sample. However, the opal based on these spheres still had a lattice arrangement, which allowed observation of the Bragg reflection band.
Moreover, the results of the sphere size measurements by the position of the Bragg reflection band correlated well with the size obtained by the SEM method. The analysis of all the samples revealed that they exhibited a hexagonal arrangement. Observed slight deviations between the results may have originated from a number of different factors. These may include the non-uniform orientation of the domains, which can occur when the domains are of limited size; the loose packing of the spheres in the structure; any defects in the 3D opal structure; and the presence of any substance filling the air voids between spheres, such as water. The polydispersity in sphere size may influence most of the above examples.
Thus, one may expect that the first method of the spheres’ diameter determination can be successfully used for this purpose in close-packed 3D colloidal crystals. Compared to SEM, it is less expensive and does not require use of sophisticated equipment. Further detailed results from the performed measurements, as well as a comparison with the SEM results, can be found in Fig. S7 and Table S2 (ESI†), respectively.
The zeta potential (ζ) of the spheres was also determined. This value can be used to assess the stability of the obtained sphere dispersions. An absolute value of the zeta potential |ζ| higher than 20–30 mV is typically sufficient to ensure good dispersion of particles and formation of an ordered lattice during the drying process. Furthermore, a higher potential increases repulsive electrostatic forces between the spheres, which may enhance the quality of the resulting crystalline arrangement. It is also important to note that the force in discussion is not the only one acting between the spheres, as there are also attractive gravitational interactions that can influence crystallization. The zeta potential for the S3 material is 48.0 mV, which is consistent with the result reported in the literature for PMMA spheres, synthesized with the same polymerization initiator.39 In the case of spheres doped with luminescent dyes, ζ takes the respective values, given in milivolts: 49.5 for POPOP-S3, 50.7 for C6-S2, 45.4 for DPP-S1, 44.0 for DCM-S1, and 46.2 for CH-S1. These results demonstrate that the zeta potential remains largely unaffected by the doping. The results and associated standard deviations are summarized in Table S3 (ESI†).
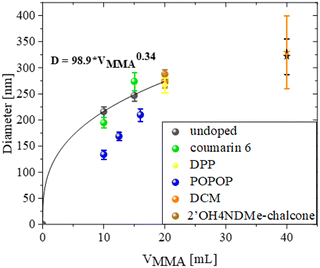
This research indicated that the diameter of the spheres is directly correlated with the quantity of the monomer used during the synthesis process. For both undoped and dye-doped opals, it can be seen that as the amount of the monomer increases, the average diameter of the resulting spheres increases. The figure illustrates that the increase in the spheres’ diameter can be characterized through an exponential function. The exponent value determined by fitting to be 0.34 for the undoped spheres indicates the dependency of the sphere diameter on the cube root of the monomer volume. This shows that the size of the spheres increases in a predictable manner and that the number of spheres remains constant between the syntheses. Such an insight holds potential for refining future methodologies to synthesize spheres with the designed diameter.
The diameter of the spheres is different when comparing undoped to doped ones. The biggest noticeable differences are visible in the case of the colloidal crystals doped with POPOP. The dye has lower solubility in the MMA monomer than other dyes shown in these studies, which may have influenced this result.
Despite these differences, the quality of the dye-doped spheres in terms of, e.g., spherical shape, monodispersity of size and absence of other particles after purification is high.
3.2. Photoluminescence properties of the dye-doped PMMA spheres and reference dye systems
Research in the following subsections was devoted to evaluating the form of the dyes in the spheres obtained using photoluminescence properties. The second objective was to assess the performance of these luminescent spheres to serve as anti-counterfeiting materials.The miscibility of the dye with the matrix seems to be one of the most critical parameters influencing the photoluminescence properties of the resulting colloidal PC. Doping of the spheres with an amount of the dye that exceeds the miscibility limit with the polymer is expected to result in aggregation of the dye, but some part of the dye may still be present in the isolated state. Then, complex systems might be formed, showing different shapes of the absorption, excitation and emission spectra, as well as quenching of the photoluminescence QY.
In our recent studies,17 we demonstrated that the employment of an ethyl eosin dye at a concentration of 0.05 wt% enabled the production of materials with a QY that was comparable to that of the dye in solution, reaching approximately 65%. However, an increase in the concentration to 1 wt% resulted in a reduction in QY to around 30%. Conversely, in the context of visual applications such as anti-counterfeiting materials, the intensity of photoluminescence plays the most important role in making the feature visible. Therefore, not only QY, but also concentration of the dye influences the brightness of the photoluminescent material.
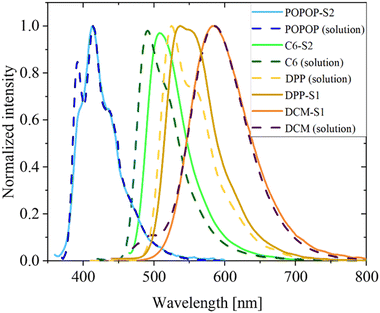
The photoluminescence spectra of the solutions are in good agreement with those reported in the literature.40,41 The selected dyes have wide emission with the following FWHM parameter: 3708 cm−1 for POPOP, 2539 cm−1 for coumarin 6, 2599 cm−1 for DPP and 2677 cm−1 for DCM. The spectra of the dye solutions are presented in Fig. S19 (ESI†) (excitation and emission) and S10 (absorption). 2′OH4NDMe-chalcone was not characterized here, because of very low emission efficiency in EA.
A comparison of the emission spectra of the dissolved dyes and the PMMA dye-doped spheres is presented in Fig. 5. There is no significant difference in the position of the band maximum for the DCM dye-doped spheres. However, for the coumarin 6- and DPP-doped spheres, a slight shift of the emission band maximum towards a higher wavelength and change of the shape are observed. Moreover, a lack of one of the maxima at the low wavelength side of the band was found for the POPOP-doped spheres. Both effects may potentially have an origin in the aggregation of the dyes or interaction between PMMA and dye molecules. However, they may also be caused by reabsorption of the light emitted by the dye. Based on further QY results, this may be the case for the coumarin 6- and DPP-doped spheres.
The morphological form of the dye can also influence the QY and photoluminescence decay time when present in different matrices. Consequently, these parameters may serve as potential indicators of the form in which the dye occurs in the PCs. Therefore, they were chosen for characterization of the PMMA spheres obtained.
A comparison of the QY values of the fabricated dye-doped PMMA spheres with the dyes in solution and in the solid (powdered) form is presented in Fig. 6. The QY values of the dyes in the respective solutions were taken from the literature: POPOP (EtOH),40 coumarin 6 (EtOH),46 DCM (DMSO),47 and 2′OH4NDMe-chalcone (ACN).38 Furthermore, the precise results, with a comparison to existing literature values, are presented in Table S4 (ESI†). The literature data includes also acetonitrile and cyclohexane coumarin 6 solutions.48
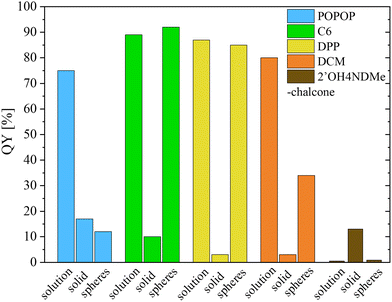 | ||
| Fig. 6 Comparison of the QY results for the investigated dyes in different matrices – solutions, solid state (powders), and PMMA spheres. | ||
POPOP-doped PMMA spheres have a QY value similar to the dye crystals. Most probably, a large part of the dye is in an aggregated form, which reduces the QY. This high content of the aggregated form of the dye may be connected with the worst solubility in MMA among the studied dyes.
Coumarin 6-doped spheres have a QY value much higher than that of the dye crystals and even slightly higher than that of the solution, suggesting that the dye is dispersed well in the PMMA matrix and occurs in the isolated form. The observed slight enhancement in the QY value relative to the dye in the solution may indicate the dye's presence in a different state. This alternative state could potentially be attributed to specific interactions occurring between the dye and the PMMA matrix.
The QY value for the spheres doped with the DPP dye of 86% is very similar to that observed for the EA solution of this dye. However, the QY value for the dye in the solid is significantly lower, equal to 3%. These observations suggest that the dye is well dispersed in the polymer and do not form aggregates.
In the case of the DCM-doped spheres, the QY value is higher than that for the dye powder, but lower than that in the solution. Previously published results on DCM-doped PMMA thin films have shown a QY of 33%,49 which is very close to the result obtained by us. According to the literature, there are two different forms in which this luminescent dye can be found in PMMA.47 It is likely that in the studied case the dye is partly aggregated and partly isolated.
As mentioned above, the 2′OH4NDMe-chalcone exhibits the AIE behavior. This fact made it possible to determine the form of occurrence of the dye in the PMMA matrix just by direct observation of the opal under UV light. Very low photoluminescence was observed, which was confirmed by the very low QY (below 1%). A similarly low value determined in the solution indicates most probably the isolated form of this dye in the PMMA spheres.
In comparison to the dyes present in the solid, the QY value has increased for the coumarin 6, DPP and DCM doped opals. In the case of the 2′OH4NDMe-chalcone the QY decreased, but the dye shows an inverse behavior for this parameter with respect to the other studied dyes. These changes are notable and indicate that these dyes in the PMMA matrix are not in the solid form. The only exception is POPOP, which exhibits low emission efficiency in the PMMA colloidal crystals.
Among the studied dyes in the obtained luminescent colloidal crystals at 0.5 wt% dye content the highest QYs are shown by coumarin 6 and DPP, which most probably indicates their occurrence in the isolated state and that their miscibility limit in PMMA has not been exceeded yet.
In the next step, the luminescence decay time was determined for the dye-doped PCs, dyes in solution and in solid form. The literature offers limited information regarding the fluorescence lifetime of dyes doped within polymeric or silica spheres. However, observations of varied decay time in dye-doped colloidal crystals, distinguished by different sphere diameters, have been documented.8,50,51 Nevertheless, the literature indicates that for high QY emitters, non-radiative decay paths are limited and the observed decay rate is governed by the radiative decay rate; thus, inhibited photon emission in the stopband results in the observed extended lifetime.52 As observed in the experimental results, an increase in luminescence decay time was found to occur for each dye-doped PMMA sphere with respect to the solutions. On the other hand various results were found by comparing the fabricated spheres to the dye powders. Typically, the decay time was lower for the PMMA spheres, with an exception of the DPP dye. Moreover, when analyzing the relation between the QY and decay life time results for the samples in different forms we were unable to find a clear correlation between the two. Experimental decay curves are shown in Fig. S11 in the ESI.† Furthermore, Table S4 (ESI†) presents the comparison of the results of the luminescence decay time for the dyes in the synthesized PMMA spheres with the dyes in EA solutions and dyes in the solid state.
The observed fluorescence emission spectra, QY and decay time data indicate that the morphological form of dyes within the PMMA spheres varies according to the specific dye used. Moreover, it should be taken into account that there may be a possibility of the presence of multiple forms of the dyes in the PMMA matrix. They can exist in other forms such as aggregates, e.g., dimers or nanocrystals, which are different from the aggregated form in the studied microcrystal powders. Furthermore, the latter do not show monoexponential decays, which makes the analysis more complex.
On the other hand, one cannot neglect any chemical reactions between the dyes and e.g. monomer during the sphere synthesis. The limited possibility to predict clearly the results of the synthesis raises the need to extend experimental studies on fabrication of highly efficient dye-doped polymeric spheres.
At the end of this section, it should be underlined that one of essential features of the luminescent materials for anticounterfeiting markings, as also for other applications, is their high emission efficiency. Determined QY values characterize the properties of the dyes in a given matrix, such as the tendency for dyes to show aggregation and luminescence quenching. Consequently, they facilitate the selection of appropriate dyes for a specific matrix. However, in general, due to differences in the amount of light absorbed by different materials, even those with similar QYs and spectral emission ranges, the intensity of the light emitted can vary. As a result, the light intensity perceived by the eye or measured by a detector may be different. For the purpose of comparing the appearance of the samples, their photographs were taken in the dark using a UV lamp (with emission maximum at λ = 365 nm).
Fig. 7 presents the results for the selected dyes dissolved in EA, dyes in the solid, and dye-doped PMMA spheres. The photographs demonstrate that the selected dissolved dyes exhibit strong emission, with the exception of the 2′OH4NDMe-chalcone solution, which displays AIE behavior. This dye shows a strong emission when undissolved, whilst the emission of the other dyes is weakened when they are in the solid. In the case of coumarin-6, DPP, DCM and 2′OH4NDMe-chalcone doped PMMA spheres, their visible emission resembles the emission of dyes dissolved in EA. Conversely, in the case of POPOP-S2, the observed emission decreased, and the intensity is even lower than that of the solid POPOP sample. The dye-doped opals, when compared to the solid state samples of these dyes, show visibly more intense emission, demonstrating that incorporation of these dyes into the PMMA matrix has enhanced their brightness under such excitation conditions. The strongest emission among the opals is observed for coumarin 6-doped ones. High intensity is also observed for the DPP- and DCM-doped opals. This paves the way for these dye-doped PMMA spheres to be used in applications, in which efficient light conversion is desired, such as luminescent powder additives for inks or paints, light amplification systems and originality protections. However, it must be noted, that the results may change with a different excitation source. Moreover, the coumarin 6 and DPP dyes exhibited very high QY in the opals, meaning good compatibility with the matrix, so there is a possibility that increasing their concentration may lead to increased brightness in the emission. However, the studies described in this paper were focused more on obtaining an efficient angle-dependent effect, which is described in the following section.
Moreover, the studies have shown that using the AIE type dye that disperses well in the PMMA matrix resulted in fluorescence quenching, so such dyes should be used rather in non-compatible matrices, in which they agglomerate.
The aim of this stage was evaluation of angle-dependent properties of the fabricated dye-doped colloidal crystals, which are introduced by these novel materials. The angle-dependent emission effect is observed for luminescent light emitted from the inside of the photonic crystal. The effect is related to a decrease in the intensity of the emitted light in the spectral region corresponding to the photonic band gap (PBG) position, which is described by eqn (4). The latter depends on the angle with respect to the crystallographic plane. It therefore also affects the emission spectrum in an angular way.
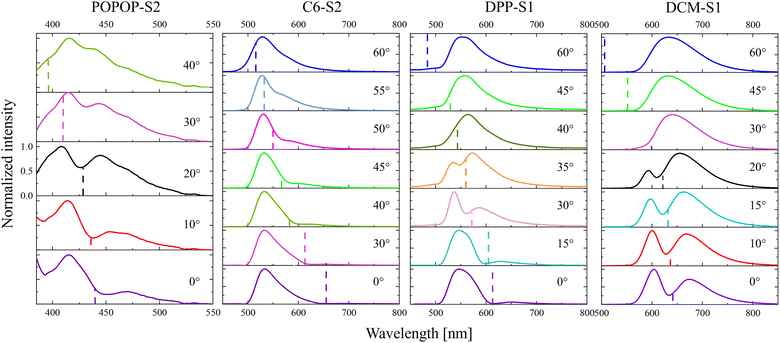
The first task of this stage of the studies was optimization of the diameter of the spheres to obtain angle-dependent emission of the fabricated 3D dye-doped PMMA opals. Table S2 (ESI†) presents the list of the synthesized dye-doped colloidal crystals of various sizes and emitting blue, green, yellow and red light. The size of the spheres of each dye was optimized, so that the emission range of the dye falls within the range of the occurrence of Bragg reflection band of the photonic crystal. Fig. 8 illustrates the emission spectra of the four produced optimized-in-size three-dimensional dye-doped PMMA colloidal crystals with respect to the observation angle. The observation angle is defined as the angle with respect to the normal to the sample plane parallel to the (111) crystallographic plane of the fcc colloidal crystal. The positions of the optical Bragg reflection band maxima (λmax) at certain angles have been previously determined (in the configuration for the sphere size measurements) and are indicated in Fig. 8 by dashed lines. For comparison, the emission spectra of the samples with non-optimized sphere diameters are shown in Fig. S12 (ESI†).
In the case of opals whose diameter has been optimized on the spectra, it can be seen that as the angle on the emission spectrum changes, the shape of the spectrum and the position of the emission maximum changes. The spectral range of the emission and reflection bands of all presented samples coincided. This resulted in decreased emission at a position of the photonic band gap, which was changing continuously with the observation angle. There was even a local minimum in the emission spectrum, which was most pronounced for the DPP and DCM samples.
In the case of the DPP-S1 opal, a local minimum shifted from about 610 nm to about 550 nm, when changing the angle from 0° to 35°. In the case of the DCM-S1 sample, the minimum shifted from about 640 nm to about 610 nm with the angle changing from 0° to 20°. Moreover, when changing the angle from 30° to about 45° for the DPP-S1 opal, it affected strongly the emission maximum position, which was originally lying at around 540 nm. The high change of the maximum was related to the continuous angle-dependent change of the shape of the emission spectrum. The effect was also clearly evident for the DCM sample. Moreover, a continuous angle-dependent change of the shape of the emission spectrum was also clearly observed for the C6-S2 and POPOP-S2 samples.
In this work, the size of the spheres was optimized and it was found that for the observation of angle-dependent emission the relationship between the optimized diameter of the spheres and position of the emission band maximum ranged from 0.4 to 0.52. This observation suggests that in the future it may be used to predict the desired size of spheres to obtain luminescent dye-doped colloidal crystals that exhibit angle-dependent emission.
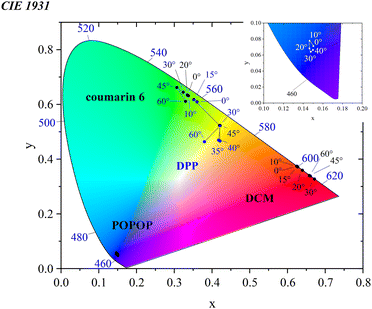
As in the case of nanoparticles, chromaticity diagrams are a commonly used tool for determining changes in emission;53,54 similarly, in the case of 3D luminescent colloidal crystals, they allow for the straightforward characterization of materials in terms of emission color changes depending on the viewing angle. In order to facilitate a comparison of the magnitude of the color changes, it was necessary to determine the chromaticity coordinates of the emission. The results are presented in the form of CIE 1931 chromaticity diagram in Fig. 9.
The diagram shows that for the opal containing DPP, as the observation angle increases in the range of 0–30°, the emission color changes from green toward yellow. As the angle of incidence is further increased, the observable color of the emission undergoes changes, becoming orange, then yellowish green at 60°. What is interesting is that non-monotonic changes of the chromaticity coordinates with the detection angle are observed, which are most distinct for this dye, but also noticed in the other samples. For the coumarin 6 opal, as the observation angle increases in the range of 0–45°, the emission color changes from green towards bluish green, while as the angle increases further from 45° to 60° the color returns roughly. For the opal containing DCM, the diagram shows that initially, as the angle increases in the range of 0–30°, the emission color shifts from orange toward red, then slightly returns at 60°. Despite very high changes in the emission spectrum, in the case of the sample containing the POPOP dye the changes in the emission color coordinates are not as big as the changes in the coordinates of the three other studied dyes (see the inset in Fig. 9, which shows an enlarged fragment of the diagram). It may be connected with the fact that a large part of the emission spectrum including the emission maximum of POPOP is lying below 450 nm, for which range the color changes perceived visually by an eye is not as big as in the emission range of the other three studied dyes.
Finally, the appearance of the samples with the highest color-dependent emission, namely the DCM- and DPP-doped opals, was studied at varied viewing angles (Fig. 10). The samples were placed on the UV lamp, and the photographs were taken from the top of the samples. Angle-dependent color behavior was found in chosen flakes with an optimal thickness.
 | ||
| Fig. 10 Photographs of the dye-doped PMMA colloidal crystals in the form of flakes excited by UV light at different observation angles – samples (a) DPP-S1 and (b) DCM-S1. | ||
The investigation confirmed the results obtained in the spectra and chromaticity diagram. Distinct color changes – from green to orange – were confirmed in the case of the DPP sample (Fig. 10a). The DCM opal also showed easy to observe changes in color, however slightly lower – from orange to red-orange (Fig. 10b).
The optimization process resulted in PCs that exhibit angle-dependent emission spectra in four spectral regions. This trait, when combined with the inherent properties of the samples, shows potential as a means of ensuring authenticity. Among the materials studied, the highest change in the emission chromaticity coordinates with respect to the viewing angle is the best for the DPP-doped sample. This, combined with the high brightness of the material observed under UV light (Fig. 7), makes this material the most very promising for applications. As the effect of changing color could be checked visually, the verification of the originality of a secured product is very easy. It introduces much novelty, with respect to the known PC materials for this application. However, further steps in the development of the opals for the security features should include their integration with the object to secure. In addition, using other methods, such as dip-coating, for producing these opals, should give better control of the properties and reproducibility of the samples.
Moreover, the presence of the reflection band of opals within the range of emission bands of the dyes in the prepared optimized luminescent colloidal crystals makes them suitable for modern applications,55 like generation of the light amplification effect56,57 and the development of biosensor technologies.58
4. Conclusions
A step-by-step preparation method of synthetic dye-doped 3D photonic crystals for anti-counterfeiting purposes was presented. Spectroscopic characterization of the PMMA colloidal crystals doped with efficient luminescent dyes emitting blue, green, yellow and red light from different groups was performed. Doping of the spheres with the chosen dyes with high solubility in the monomer during the synthesis did not affect the quality of the obtained spheres and 3D colloidal crystals. The polydispersity index of the size of the synthesized doped spheres remained low in most cases. The influence of the presence of the dye on the average size of the synthesized spheres was observed.Parameters such as QY and angle-dependent photoluminescence, which characterize the visual appearance and spectroscopic properties of the materials, were determined. It was found that the doping of dyes with high solubility in the monomer, at the level of 0.5 wt%, may help to minimize or even prevent the effect of the aggregation of the dye-dopants and the resulting low QY. However, the studies have shown that the solubility, which is necessary, may not be always sufficient to obtain a high QY.
The form of occurrence of the studied dyes in the PMMA spheres depends on type of the dye. Taking into account the QY results, coumarin 6, DPP and 2′OH4NDMe-chalcone dyes occur in the PMMA matrix in the isolated form, DCM – partly in the isolated or an aggregated form, which shows an emission, but different than the form occurring in the solid state and POPOP – mostly in an aggregated form. The variety of the obtained results for different dyes in the PMMA matrix shows that there is still a need to extend experimental research in this area, both in terms of the type of the dye as well as the type of the polymer.
The appearance of the dye-doped opals under arbitrary excitation conditions under UV light was compared to characterize better the photoluminescence effect, which could be noticed by the eye. The brightest luminescence was observed in C6-, DPP- and DCM-doped opals. Optimization of the size of the dye-doped spheres allowed obtaining four luminescent 3D opal materials, showing an angle-dependent emission spectrum in four different spectral ranges. The highest change of the color was noticed for the DPP-doped opal, which was confirmed in the photographs. Use of these materials or the method of the synthesis allows for the design of completely new and difficult to counterfeit security features. This method opens a way for the fabrication of opals that can be further integrated into products or documents, thereby providing an effective authentication layer. The results can also be utilized in the application of light amplification systems.
Author contributions
Weronika Zając – conceptualization, data curation, formal analysis, investigation, methodology, validation, visualization, writing – original draft; Maciej Czajkowski – conceptualization, formal analysis, funding acquisition, investigation, methodology, project administration, validation, writing – original draft; Bartłomiej Potaniec – investigation, writing – original draft, Maria Zdończyk – investigation; and Joanna Cybińska – conceptualization, formal analysis, funding acquisition, investigation, methodology, supervision, validation, writing – original draft.Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
These studies were supported by the (Poland) within the framework of the 12th edition of the LIDER programme – project contract no. LIDER/39/0203/L-12/20/NCBR/2021. WZ would like to thank the Ministry of Science and Higher Education in Poland for Grant No. DWD/6/0316/2022. in the frame of Implementation of Doctorate Programme. The authors would like to thank Dr Wojciech Gil and Dr Krzysztof Rola for execution of the SEM measurements.References
- Z. Gu, A. Fujishima and O. Sato, Fabrication of High-Quality Opal Films with Controllable Thickness, Chem. Mater., 2002, 14, 760–765 CrossRef CAS.
- Y. Li, T. Kunitake, S. Fujikawa and K. Ozasa, Photoluminescence Modification in 3D-Ordered Films of Fluorescent Microspheres, Langmuir, 2007, 23, 9109–9113 CrossRef CAS PubMed.
- T. Yamasaki and T. Tsutsui, Spontaneous emission from fluorescent molecules embedded in photonic crystals consisting of polystyrene microspheres, Appl. Phys. Lett., 1998, 72, 1957–1960 CrossRef CAS.
- G. I. N. Waterhouse and M. R. Waterland, Opal and inverse opal photonic crystals: Fabrication and characterization, Polyhedron, 2007, 26, 356–368 CrossRef CAS.
- R. De Angelis, I. Venditti, I. Fratoddi, F. De Matteis, P. Prosposito, I. Cacciotti, L. D’Amico, F. Nanni, A. Yadav, M. Casalboni and M. V. Russo, From spheres to microribbons: Self-assembled Eosin Y doped PMMA nanoparticles as photonic crystals, J. Colloid Interface Sci., 2014, 414, 24–32 CrossRef CAS PubMed.
- H. Wang, X. Gu, R. Hu, J. W. Y. Lam, D. Zhang and B. Z. Tang, Luminescent Photonic Crystals with Multi-functionality and Tunability, Chem. Sci., 2016, 7, 5692–5698 RSC.
- D. W. McComb, B. M. Treble, C. J. Smith, R. M. De La Rue and N. P. Johnson, Synthesis and characterisation of photonic crystals, J. Mater. Chem., 2001, 11, 142–148 RSC.
- M. Megens, J. E. G. J. Wijnhoven, A. Lagendijk and W. L. Vos, Fluorescence lifetimes and linewidths of dye in photonic crystals, Phys. Rev. A: At., Mol., Opt. Phys., 1999, 59, 4727–4731 CrossRef CAS.
- S. Wu, H. Xia, J. Xu, X. Sun and X. Liu, Manipulating Luminescence of Light Emitters by Photonic Crystals, Adv. Mater., 2018, 30, 1803362 CrossRef CAS.
- Y. Song, N. Li, D. Chen, Q. Xu, H. Li, J. He and J. Lu, 3D ordered MoP inverse opals deposited with CdS quantum dots for enhanced visible light photocatalytic activity, Appl. Catal., B, 2018, 238, 255–262 CrossRef CAS.
- L. Tao, W. Xu, Y. Zhu, L. Xu, H. Zhu, Y. Liu, S. Xu, P. Zhou and H. Song, Modulation of upconversion luminescence in Er3+, Yb3+-codoped lanthanide oxyfluoride (YOF, GdOF, LaOF) inverse opals, J. Mater. Chem. C, 2014, 2, 4186–4195 RSC.
- A. Diacon, A. Mocanu, C. Boscornea and P. Hudhomme, Monomer type emission of perylenediimide derivatives doped polymer particles, Colloids Surf., 2012, 407, 9–15 CrossRef CAS.
- M. Barth, A. Gruber and F. Cichos, Spectral and angular redistribution of photoluminescence near a photonic stop band, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 72, 085129 CrossRef.
- L. Bechger, P. Lodahl and W. L. Vos, Directional Fluorescence Spectra of Laser Dye in Opal and Inverse Opal Photonic Crystals, J. Phys. Chem. B, 2005, 109, 9980–9989 CrossRef CAS.
- Y. Q. Zhang, J. X. Wang, Z. Y. Ji, W. P. Hu, L. Jiang, Y. L. Song and D. B. Zhu, Solid-state fluorescence enhancement of organic dyes by photonic crystals, J. Mater. Chem., 2007, 17, 90–94 RSC.
- X. Wang, S. Xu and W. Xu, Luminescent properties of dye–PMMA composite spheres, Phys. Chem. Chem. Phys., 2011, 13, 1560–1567 RSC.
- W. Zając, M. Czajkowski and J. Cybińska, Chameleon photonic crystals: Acid-induced emission and absorption shifts for sensor applications, Mater. Today Chem., 2024, 40, 102195 CrossRef.
- R. V. Nair, R. Vijaya, K. Kurodoka and K. Sakoda, Emission studies on photonic crystals fabricated using dyed polystyrene colloids, J. Appl. Phys., 2007, 102, 123106 CrossRef.
- X. Chi, N. Sui, Y. Wang, L. Zou, C. Qian and H. Zhang, Emission and Energy Transfer Characteristics of Coumarin 6 Molecules Doped in Opal Polymer Photonic Crystal, Chem. Res. Chin. Univ., 2015, 31, 466–470 CrossRef CAS.
- B. Y. Tong, P. K. John, Y.-T. Zhu, Y. S. Liu, S. K. Wong and W. R. Ware, Fluorescence-lifetime measurements in monodispersed suspensions of polystyrene particles, J. Opt. Soc. Am. B, 1993, 10, 356–359 CrossRef CAS.
- J. Feng, S. Bian, Y. Long, H. Yuan, Q. Liao, H. Cai, H. Huang, K. Song and G. Yang, Low threshold photonic crystal lasing from a dye with high emission quantum yield and weak self-quenching, J. Mater. Chem. C, 2013, 1, 6157–6162 RSC.
- A. Diacon, E. Rusen, A. Mocanu and P. Hudhomme, Fluorescence Properties of Photonic Crystals Doped with Perylenediimide, Langmuir, 2011, 27, 51–62 CrossRef.
- M. Müller, R. Zentel, T. Maka, S. G. Romanov and C. M. Sotomayor Torres, Dye-Containing Polymer Beads as Photonic Crystals, Chem. Mater., 2000, 12, 2508–2512 CrossRef.
- C. Wu, Y. Zheng, C. Szymanski and J. McNeill, Energy Transfer in a Nanoscale Multichromophoric System:
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) Fluorescent Dye-Doped Conjugated Polymer Nanoparticles, J. Phys. Chem. C, 2008, 112, 1772–1781 CrossRef CAS PubMed.
Fluorescent Dye-Doped Conjugated Polymer Nanoparticles, J. Phys. Chem. C, 2008, 112, 1772–1781 CrossRef CAS PubMed. - C. Xu, C. Huang, D. Yang, L. Luo and S. Huang, Photo-Luminescent Photonic Crystals for Anti-Counterfeiting, ACS Omega, 2022, 7, 7320–7326 CrossRef CAS PubMed.
- D. Yang, C. Ouyang, Y. Zhang, D. Ma, Y. Ye, D. Bu and S. Huang, Simple and efficient fabrication of multi-stage color-changeable photonic prints as anti-counterfeit labels, J. Colloid Interface Sci., 2021, 590, 134–143 CrossRef CAS PubMed.
- H. S. Lee, T. S. Shim, H. Hwang, S.-M. Yang and S.-H. Kim, Colloidal Photonic Crystals toward Structural Color Palettes for Security Materials, Chem. Mater., 2013, 25, 2684–2690 CrossRef CAS.
- L. Bai, Z. Xie, W. Wang, C. Yuan, Y. Zhao, Z. Mu, Q. Zhong and Z. Gu, Bio-Inspired Vapor-Responsive Colloidal Photonic Crystal Patterns by Inkjet Printing, ACS Nano, 2014, 8, 11094–11100 CrossRef CAS PubMed.
- M. Loncan, T. Yoshie, A. Scherer, P. Gogna and Y. Qiu, Low-threshold photonic crystal laser, Appl. Phys. Lett., 2002, 81, 2680–2682 CrossRef.
- C. Liu, G. Gao, Y. Zhang, L. Wang, J. Wang and Y. Song, The Naked-Eye Detection of NH3–HCl by Polyaniline-Infiltrated TiO2 Inverse Opal Photonic Crystals, Macromol. Rapid Commun., 2012, 33, 380–385 CrossRef CAS.
- A. V. Goponenko and S. A. Asher, Modeling of Stimulated Hydrogel Volume Changes in Photonic Crystal Pb2+ Sensing Materials, J. Am. Chem. Soc., 2005, 127, 10753–10759 CrossRef CAS.
- Ł. Duda, M. Czajkowski, B. Potaniec and P. Vankatova, Helical twisting power and compatibility in twisted nematic phase of new chiral liquid crystalline dopants with various liquid crystalline matrices, Liq. Cryst., 2019, 46, 1769–1779 CrossRef.
- M. Zdończyk, B. Potaniec, M. Skoreński and J. Cybińska, Development of Efficient One-Pot Methods for the Synthesis of Luminescent Dyes and Sol–Gel Hybrid Materials, Materials, 2021, 15, 203 CrossRef.
- B. Potaniec, M. Zdończyk and J. Cybińska, Controlled Synthesis of Luminescent Xanthene Dyes and Use of Ionic Liquid in Thermochromic Reaction, Molecules, 2022, 27, 3092 CrossRef CAS PubMed.
- D.-H. Lee, J.-W. Kim and K.-D. Suh, Monodisperse micron-sized polymethylmethacrylate particles having a crosslinked network structure. V, J. Mater. Sci., 2000, 35, 6181–6188 CrossRef CAS.
- R. C. Schroden, M. A.-Daos, C. F. Blanford and A. Stein, Optical Properties of Inverse Opal Photonic Crystals, Chem. Mater., 2002, 14, 3305–3315 CrossRef CAS.
- Z. Song, R. T. K. Kwok, E. Zhao, Z. He, Y. Hong, J. W. Y. Lam, B. Liu and B. Z. Tang, A Ratiometric Fluorescent Probe Based on ESIPT and AIE Processes for Alkaline Phosphatase Activity Assay and Visualization in Living Cells, ACS Appl. Mater. Interfaces, 2014, 6, 17245–17254 CrossRef CAS.
- H. Song, Z. Kuang, X. Wang, Y. Guo, Q. Guo, H. Zhang and A. Xia, Solvent Polarity Dependent Excited State Dynamics of 2′-Hydroxychalcone Derivatives, J. Phys. Chem. C, 2018, 122, 15108–15117 CrossRef CAS.
- M. Matamoros-Ambrocio, E. Sánchez-Mora, E. Gómez-Barojas and J. A. Luna-López, Synthesis and Study of the Optical Properties of PMMA Microspheres and Opals, Polymers, 2021, 13, 2171 CrossRef CAS.
- S. A. El-Daly, S. A. El-Azim, F. M. Elmekawey, B. Y. Elbaradei, S. A. Shama and A. M. Asir, Photophysical Parameters, Excitation Energy Transfer, and Photoreactivity of 1,4-Bis(5-phenyl-2-oxazolyl)benzene (POPOP) Laser Dye, Int. J. Photoenergy, 2012, 2012, 458126 CrossRef.
- U. S. Raikar, C. G. Renuka, Y. F. Nadaf, B. G. Mulimani, A. M. Karguppikar and M. K. Soudagar, Solvent effects on the absorption and fluorescence spectra of coumarins 6 and 7 molecules: Determination of ground and excited state dipole moment, Spectrochim. Acta, Part A, 2006, 65, 673–677 CrossRef CAS PubMed.
- J. Gierschner, J. Shi, B. Milián-Medina, D. Roca-Sanjuán, S. Varghese and S. Park, Luminescence in Crystalline Organic Materials: From Molecules to Molecular Solids, Adv. Opt. Mater., 2021, 9, 2002251 CrossRef CAS.
- J. R. Lakowicz, Principles of Fluorescence Spectroscopy, Springer, 3rd edn, 2006 Search PubMed.
- A. Reisch and A. S. Klymchenko, Fluorescent Polymer Nanoparticles Based on Dyes: Seeking Brighter Tools for Bioimaging, Small, 2016, 12, 1968–1992 CrossRef CAS PubMed.
- S. Doose, H. Neuweiler and M. Sauer, A Close Look at Fluorescence Quenching of Organic Dyes by Tryptophan, ChemPhysChem, 2005, 6, 2277–2285 CrossRef CAS.
- A. N. Fletcher and D. E. Bliss, Laser Dye Stability. Part 5 Effect of Chemical Substituents of Bicyclic Dyes Upon Photodegradation Parameters, Appl. Phys., 1978, 16, 289–295 CAS.
- S. L. Bondarev, V. N. Knyukshto, V. I. Stepuro, A. P. Stupak and A. A. Turban, Fluorescence and electronic structure of the laser dye DCM in solutions and in polymethylmethacrylate, J. Appl. Spectrosc., 2004, 71, 194–201 CrossRef CAS.
- G. Jones, W. R. Jackson, C. Choi and W. R. Bergman, Solvent Effects on Emission Yield and Lifetime for Coumarin Laser Dyes. Requirements for a Rotatory Decay Mechanism, J. Phys. Chem. C, 1985, 89, 294–300 CrossRef CAS.
- A. P. Green and A. R. Buckley, Solid state concentration quenching of organic fluorophores in PMMA, Phys. Chem. Chem. Phys., 2015, 17, 1435–1440 RSC.
- J. Martorell and N. M. Lawandy, Observation of Inhibited Spontaneous Emission in a Periodic Dielectric Structure, Phys. Rev. Lett., 1990, 65, 1877–1880 CrossRef CAS PubMed.
- B. Y. Tong, P. K. John, Y.-T. Zhu, Y. S. Liu, S. K. Wong and W. R. Ware, Fluorescence-lifetime measurements in monodispersed suspensions of polystyrene particles, J. Opt. Soc. Am., 1993, 10, 356–359 CrossRef.
- H. W. Jones, Y. Bandera and S. H. Foulger, Probing Light-Matter Interactions: Fluorescence Lifetime Manipulation in a Crystalline Colloidal Array, ACS Omega, 2024, 9, 48524–48535 CrossRef CAS PubMed.
- A. Rai, S. Bhaskar, K. M. Ganesh and S. S. Ramamurthy, Cellphone-based attomolar tyrosine sensing based on Kollidon-mediated bimetallic nanorod in plasmon-coupled directional and polarized emission architecture, Mater. Chem. Phys., 2022, 285, 126129 CrossRef CAS.
- K. M. Ganesh, S. Bhaskar, V. S. K. Cheeral, P. Battampara, R. Reddy, S. C. Neelakantan, N. Reddy and S. S. Ramamurthy, Review of Gold Nanoparticles in Surface Plasmon-Coupled Emission Technology: Effect of Shape, Hollow Nanostructures, Nano-Assembly, Metal–Dielectric and Heterometallic Nanohybrids, Nanomaterials, 2024, 14, 111 CrossRef CAS.
- Y. Xiong, S. Shepherd, J. Tibbs, A. Bacon, W. Liu, L. D. Akin, T. Ayupova, S. Bhaskar and B. T. Cunningham, Photonic Crystal Enhanced Fluorescence: A Review on Design Strategies and Applications, Micromachines, 2023, 14, 668 CrossRef.
- S. V. Frolov, Z. V. Vardeny, A. A. Zakhidov and R. H. Baughman, Laser-like emission in opal photonic crystals, Opt. Commun., 1999, 162, 241–246 CrossRef CAS.
- L. Shi, M. Zheng, F. Jin, X. Dong, W. Qiang, Z. Zhao and Z. Duan, Transition of lasing modes in polymeric opal photonic crystal resonating cavity, Appl. Opt., 2016, 5, 4759–4761 CrossRef PubMed.
- S. Bhaskar, W. Liu, J. Tibbs and B. T. Cunningham, Photonic crystal-coupled enhanced steering emission: A prism-free, objective-free, and metal-free loss-less approach for biosensing, Appl. Phys. Lett., 2024, 124, 161102 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5tc01577d |
| This journal is © The Royal Society of Chemistry 2025 |