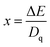Cr3+-doped CaMgGe2O6 phosphors: crystal field effects and the synergistic role of FIR and fluorescence lifetime in multi-mode optical thermometry†
Yosra
Bahrouni
a,
Ikhlas
Kachou
a,
Kamel
Saidi
 ab,
Christian
Hernández-Álvarez
cd,
Mohamed
Dammak
ab,
Christian
Hernández-Álvarez
cd,
Mohamed
Dammak
 *a and
Inocencio R
Martín
c
*a and
Inocencio R
Martín
c
aLaboratoire de Physique Appliquée, Faculté des Sciences de Sfax, Département de Physique, Université de Sfax, BP 1171, Sfax, Tunisia. E-mail: madidammak@yahoo.fr; mohamed.dammak@fss.usf.tn
bDepartment of Physics, Sfax Preparatory Engineering Institute, University of Sfax, 1172 – 3000, Sfax, Tunisia
cUniversidad de La Laguna, Departamento de Física, MALTA-Consolider Team, IMN and IUdEA Apdo. Correos 456, E-38206, San Cristóbal de La Laguna, Santa Cruz de Tenerife, Spain
dAdam Mickiewicz University, Faculty of Chemistry, Uniwersytetu Poznańskiego 8, 61-614, Poznań, Poland
First published on 4th June 2025
Abstract
Luminescence thermometry has attracted growing interest for its potential in remote and non-contact temperature sensing. Among luminescent ions, Cr3+ is widely studied for optical thermometry using fluorescence intensity ratio (FIR), but its fluorescence lifetime (FL) thermometric potential remains largely undiscovered. In this study, we present a detailed spectroscopic investigation of Cr3+-doped CaMgGe2O6, demonstrating the complementary advantages of FIR and FL-based thermometry. X-Ray diffraction (XRD) confirms the monoclinic phase with high crystallinity, while diffuse reflectance spectroscopy provides insight into the crystal field strength (Dq/B ≈ 1.81) and optical band gap (Eg). Under 405 nm excitation, we systematically analyze the photoluminescence and temperature-dependent luminescence behavior. The multi-mode thermal sensing approach reveals that FIR (I589/I771) achieves a remarkable maximum sensitivity of 1.4% K−1 at 390 K, with an exceptionally low temperature uncertainty (∼0.11 K at room temperature), establishing its reliability for precise temperature detection. Meanwhile, FL thermometry exhibits an even higher maximum sensitivity of 2.5% K−1 at 478 K, underscoring its strong potential as an alternative or complementary technique. By integrating both methods, we achieve enhanced accuracy, broader temperature coverage, and improved adaptability to various sensing environments. This work highlights the first comprehensive demonstration of FL-based thermometry in Cr3+-doped phosphors, paving the way for optimized multi-mode luminescent thermal sensors and reinforcing the critical role of both the host matrix and advanced spectroscopic characterization.
Introduction
Luminescence thermometry has received considerable attention recently and is becoming a rapidly growing field of nanotechnology, particularly for temperature measurement in remote sensing.1–3 This technique relies on the luminescence properties of phosphors, such as spectral shape, emission intensity, or the kinetics of excited states of incorporated dopants, enabling fast and accurate real-time temperature measurement with sub-micrometer spatial and millikelvin thermal resolution.4,5 These characteristics make luminescence thermometry a valuable complement to thermal imaging cameras, which only provide surface temperature maps.Among various luminescence thermometry approaches, the fluorescence intensity ratio (FIR) method is widely employed due to its self-referencing nature. Systems with two exciting states in thermal equilibrium (strongly thermodynamically coupled) offer high selectivity with respect to temperature. In such systems, the population distribution between thermally coupled states follows the Boltzmann law, ensuring a rapid renormalization of the emission intensity when external factors influence the overall quantum yield.6,7 For biological applications, an ideal FIR-based optical thermometer requires a single-doped material with two thermally coupled excited states that can be efficiently excited and emit within the biological optical windows, where tissue absorption and scattering are minimized.8,9 Another widely used luminescence thermometry technique is fluorescence lifetime (FL) thermometry, which is independent of external influences and relies on measuring the temperature-dependent luminescence lifetime of excited states.10,11 Combining FIR and FL methods enhances the accuracy and reliability of temperature measurements by providing complementary information: FIR ensures high sensitivity in a specific range, while FL extends the measurable temperature range and improves robustness against fluctuations in the excitation power or concentration.
For the development of highly sensitive phosphors, the choice of a stable host material and an appropriate activator with thermally coupled levels is crucial. While phosphate,12,13 borate,14,15 and silicate16–18 luminescent materials have been extensively studied, germanate hosts have received comparatively less attention. However, germanate compounds offer advantages such as high thermal and chemical stability, hardness, and broad transparency. Furthermore, they exhibit the ability to generate deep red emissions in the desired wavelength range under UV and blue excitation.19 The crystal structure of germanates varies despite their similar chemical formulas, primarily due to differences in the size of monovalent and divalent cations, which influence bond lengths and the crystal field strength.20–22
The performance of inorganic luminescent materials is determined by the combination of the host lattice and the activator ions. Rare earth and transition metal ions have been widely investigated as activators for near-infrared (NIR) emission. However, a major challenge in NIR-emitting materials is the enhancement of the photoluminescence quantum yield, particularly for 4f–4f transitions in lanthanide ions.23–25 Chromium ions (Cr3+), with a 3d3 valence electron configuration, are attractive alternatives due to their strong crystal field interactions and tunable luminescence properties.26 Unlike the 4f orbitals of lanthanides, the 3d electrons of Cr3+ interact strongly with the surrounding crystal field, leading to well-defined energy level splitting, including the 4A2g, 2E, 2T1g, and 4T2g states.27 The relative positioning of the 2E and 4T2 states is highly dependent on the crystal field strength, which directly affects the emission characteristics.1 Because of these properties, Cr3+ is commonly doped into various inorganic hosts for laser and luminescence applications.
Germanate-based host materials provide a weaker crystal field environment for Cr3+ ions compared to silicates, leading to red-shifted emission with potential applications in luminescence thermometry. In this study, CaMgGe2O6 was selected as a host matrix, with Cr3+ as the activator, to develop a novel series of CaMgGe2O6:0.01Cr3+ (CMGO:0.01Cr3+) luminescent materials.
In this work, we investigate the luminescence thermometric properties of Cr3+-doped CaMgGe2O6 (CMGO:Cr3+) as a potential dual-mode optical thermometer. The study explores the structural, optical, and thermal characteristics of the synthesized phosphors, focusing on their ability to function as a reliable temperature sensor. By combining fluorescence intensity ratio (FIR) and fluorescence lifetime (FL) techniques, we aim to assess the potential of CMGO:0.01Cr3+ for accurate and robust temperature measurements. The influence of the crystal field environment on the luminescence properties of Cr3+ ions is analyzed to understand its impact on thermometric performance. This work provides insights into the design of non-lanthanide-based luminescent thermometers and their applicability in advanced temperature sensing.
Experimental section
Sample preparation
The chemical composition of CMGO:0.01Cr3+ was successfully synthesized by a high temperature solid-state reaction method. The crude reagents CaCO3, GeO2, MgO and Cr2O3 were used without further purification. Stoichiometric amounts of the starting materials were first thoroughly mixed by grinding in an agate mortar and presintered at 800 °C for 8 h in air. After complete grinding, the presintered samples were then sintered at a higher temperature of 1200 °C for 5 hours in air. Finally, the samples were allowed to cool naturally to room temperature in an oven and then ground into powders for further characterization.Characterization
The X-ray diffraction (XRD) patterns of phosphorus were obtained at room temperature using a Bruker D8-Advance powder X-ray diffractometer. Monochromatic CuKa1 copper radiation (1.5406 Å) was used in the 2θ range (20–80°). The morphology of the samples was analyzed using a Zeiss Supra55VP FEG-SEM and a Bruker XFlash 5030. For the determination of the UV-vis-NIR absorption, a UV-vis-NIR spectrometer (PerkinElmer Lambda 365) was used.A high-resolution CCD detector (Avantes) was used to detect the emission spectra. Spectra Physics 3900S was used for 405 nm excitation and a Linkam (THMS600) for sample heating. Decay curves were obtained using a 200 MHz LeCroy WS424 oscilloscope, a Hamamatsu R928 photomultiplier tube (PMT), and a tunable EKSPLA/NT342/3/UVE 10 ns pulsed laser (optical parametric oscillator – OPO) operated at a repetition rate of 10 Hz as the excitation source.
Results and discussion
Structural and morphological analysis of the obtained materials
To confirm their composition and morphology, the synthesized particles were subjected to extensive analysis. X-Ray powder diffraction measurements showed that the synthesized sample was composed of a single phase, with no occurrence of appearance phases. The XRD patterns, both experimental and calculated by Rietveld refinement, are shown in Fig. 1a for CMGO:0.01Cr3+. The structural data given in ref. 28 were used as the starting model for the Rietveld calculations. There was good agreement between the experimental and calculated patterns, resulting in satisfactory fit factors. The monoclinic system and the space group C2/c were confirmed.29 Table S1 (ESI†) shows a summary of the structural parameters derived from the Rietveld refinement of the samples illustrated. Depending on these experimental results, the crystal structure of CaMgGe2O6 is depicted in Fig. 1b. It is made up of [CaO8] dodecahedrons, [MgO6] octahedrons and [GeO4] tetrahedrons. Each type of polyhedron is linked by common oxygen atoms to form an infinite chain parallel to the c-axis. Consider the ionic radii of Ca2+ (1.12 Å, coordination number (CN) = 8), Mg2+ (0.72 Å, CN = 6), Ge4+ (0.39 Å, CN = 4) and Cr3+ (0.615 Å, CN = 6).30 Since the appearance of the emission spectra of this transition metal ion is sensitive to changes in the crystal field strength, this can strongly influence the spectroscopic properties of the Cr3+ ions. In accordance with the above description, Cr3+ ions could be substitutes for Mg2+ ions (polyhedral representation), as shown in Fig. 1c. In addition, the energy difference between the electron configuration of the particular ligand field and the isotropic field is referred to as the crystal field stabilization energy (CFSE). The CFSE is large for ions with an electronic configuration of d3 and d8 ions in the octahedral ligand, so the Cr3+ site has a preference for octahedral sites. Thus, we suggest that Cr3+ ions substitute for Mg2+ to form [CrO6] octahedrons.31 | ||
| Fig. 1 (a) Rietveld refinement of XRD patterns. (b) Visual crystal structure. (c) The potential occupancies of doped ions in a polyhedron. (d) SEM image of the CMGO:0.01Cr3+ sample. | ||
The size of the individual particles formed is generally in the micrometer range (∼1 μm) as shown by the SEM images of the synthesized luminophores (Fig. 1d). The agglomerated morphology with a broad size distribution is achieved using solid-state synthesis.
Optical characterization
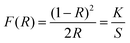 | (1) |
| αhv = c(hv − Eg)1/2 | (2) |
 | ||
| Fig. 2 (a) Excitation spectrum when monitoring the emission at 704 nm. (b) Emission spectrum upon excitation at 405 nm of CMGO:0.01Cr3+. | ||
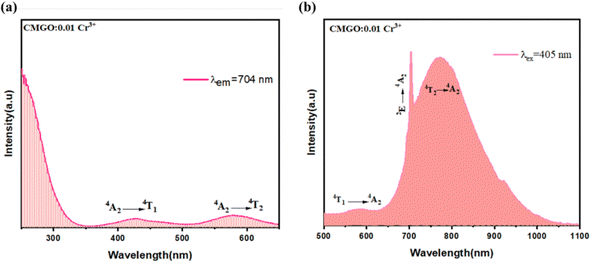
Furthermore, it is well known that the R line of the Cr3+ zero phonon is mainly affected by the side band vibration. It is noteworthy that the anti-Stokes (696 nm) disappears in the PL spectra, while the Stokes remains at 704 nm. The red shift of the PL is related to the variation of the crystal field according to previous work.41 This indicates that the crystal field was weakened by the stretching of the Cr–O bonds. The estimation of the crystal field (Dq) and the ray parameter (B), using the Tanabe–Sugano diagram shown in Fig. 3b, in the crystal conditions is given by using the following formula:42,43
| ΔE = E(4A2 → 4T1) − E(4A2 → 4T2) | (3) |
| ΔS = E(4A2 → 4T1) − E(4A2 → 4T2) | (4) |
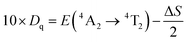 | (5) |
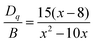 | (6) |
where B is the Racah parameter, Dq is the crystal field splitting energy, ΔE is the energy difference between 4A2 → 4T1 and 4A2 → 4T2 based on the PLE spectrum, and ΔS is the Stokes shift, which can be calculated from the excitation band of 4A2 → 4T2 and the emission band of 2E → 4A2. In agreement with previous relations are the estimated values of B = 685 cm−1, Dq = 1254 cm−1 and Dq/B = 1.84. Thus, the Cr3+ ions are located in a weak crystalline field in the CMGO host, a case where sharp and broader peaks coexist. If the Cr3+ ions were in a weak crystal field, the energy interval between the ground (4A2) and excited states (4T1 and 4T2) would be smaller, resulting in a red shift in the Cr3+ d–d transition. Indeed, the intensity of the crystal field is strongly influenced by variations of the cation–oxygen–anion distance caused by lattice vibrations, defects and local structure distortions.44,45
 | ||
| Fig. 3 (a) Schematic configurational coordinate diagram. (b) Tanabe–Sugano energy level diagram in which the straight lines indicate the crystal fields of the CMGO:0.01Cr3+ samples. | ||
To further study this mechanism, we explain thermal quenching on the basis of conformational coordinates as follows. The luminescence spectra of Cr3+-doped materials result from d–d electronic transitions; in the case of transition metals with the 3d3 electronic configuration observed for Cr3+, depending on the intensity of the crystal field of the host material, the spectrum is dominated either by a narrow 2E → 4A2 emission band (strong crystal field) or by a broad 4T2 → 4A2 emission band for the weak crystal field (Fig. 3a). Bearing in mind, the fact that the analyzed structures contain perfect octahedral sites of Mg2+/Ca2+ ions the observed changes in the shape of the emission spectra and the gradual increase in the intensity of the 4T2 → 4A2 band relative to the 2E → 4A2 band can be directly correlated with the change in the strength of the crystal field resulting from the extension of the Cr3+–O2− bond length.46,47
Temperature dependence of the PL spectra
The temperature-dependent PL emission spectra of the Cr3+-doped CMGO system were analyzed with the dual aim of analyzing the thermal response and investigating in detail the electronic configuration and the parameters describing the coupling with the phonon lattice. The spectra in Fig. 4a show the strong thermal quenching of the system with increasing temperature. The trend shows a strong decrease of the broad emission of 4T2 with respect to the spin-forbidden R-line emission with increasing temperature, The extinction temperature is in the range of 298–450 K according to the integrated PL intensity trend 4T2, suggesting a thermally activated process between the excited states.48 | ||
| Fig. 4 (a) Temperature dependence of the PL emission spectra from 298 to 450 K. (b) CIE chromaticity diagram of the CMGO:0.01Cr3+ phosphor excited at various temperatures. | ||
With a further increase in temperature from 298 K to 450 K, the emission intensity of CMGO:0.01Cr3+ gradually decreases for the levels (4T2 →4A2) but gradually increases for the levels (4T1 →4A2), showing an opposite temperature dependence. Moreover, the latter considers the background contribution to the signal from other emission processes. The trend shows a strong decrease of the broad emission of 4T2 with respect to the spin-forbidden R-line emission with increasing temperature, suggesting a thermally activated process between the excited states.49
As the light intensity of Cr3+ ions decreases with increasing temperature, the color of the CMGO luminophores is believed to change under UV excitation. In other words, the color of the luminophores can be modified by temperature control. In order to further study the color change visually, we note that when the temperature gradually increases from 298 K to 450 K, the color of our samples changes significantly from violet to red and the brightness continues to decrease. Reflecting the good stability of the Cr3+ ion, the results indicate that the temperature can be qualitatively determined by observation of the thermal discoloration behavior (Fig. 4b).
The important advantage of using the intensity ratio as a measure of the temperature of a thermally coupled system is that the population of each level is directly proportional to the total population. This makes the luminescence intensity ratio independent of the excitation intensity, since a change in the total population due to a change in excitation will affect the population of each level to the same extent. Eqn (7) was used for the fitting of the experimental results.50,51
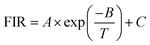 | (7) |
The thermal equilibrium for FIR (589/771) and FIR (704/771) thus suggests that the Cr3+ substitution in the Mg2+ octahedral site leads to the splitting of the 2E state into two R lines due to the low symmetry site. The solid line in Fig. 5 shows the fit using eqn (7). In particular, the energy gap ΔE was computed as 1814.4 cm−1 and 2123.7 cm−1. There was a good linear function between the two energy levels. These results strongly suggest that the sample of CMGO:0.01Cr3+ could be used to reflect the variation in temperature.
 | ||
| Fig. 5 FIR thermometry depending on CMGO:0.01Cr3+. (a) FIR (I589/771) and (b) FIR (I704/771) in the studied range of temperature (298–450 K). | ||
In order to estimate the thermometric sensitivity, the absolute sensitivity (Sa) and the relative sensitivity (Sr), which reflect the rate of variation of the FIR parameter with temperature, were quantified using the following equations:52,53
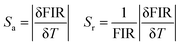 | (8) |
 | ||
| Fig. 6 The dependence of absolute sensitivity (Sa) and relative sensitivity (Sr) with temperatures of (a) I589/I771 and (b) I704/I771 for CMGO:0.01Cr3+. | ||
As can be seen in Fig. S3 (ESI†), the experimental temperature of the uncertainty was carried out by measuring the 100 spectra at 298 K under similar conditions, as the operation of the sensor is additionally dependent on the estimation of the temperature resolution. The temperature resolution values are generally distributed in a Gaussian pattern around room temperature. In this case, the standard deviation is 0.1 K. In order to evaluate the high temperature performance of different luminophores, we have summarized the maximum relative temperature sensitivities Sr at the highest temperature values for the luminescence thermometers, as shown in Table 1. These results highlight the potential of CMGO:0.01Cr3+ as an excellent candidate for optical temperature sensing applications, particularly in the context of multi-mode thermometry.
| Materials | Temperature range (K) | S r (% K−1) | T m (K) | Ref. |
|---|---|---|---|---|
| α Ga2O3:Cr3+ | 200–450 | 1.05 | 300 | 54 |
| Bi2Ga4O9:Cr3+ | 80–600 | 0.7 | 90 | 42 |
| MgSiO4:Cr3+ | 200–350 | 0.7 | 310 | 55 |
| Y3Al5O12:Cr3+ | 123–573 | 0.55 | 384 | 56 |
| LiGa5O8:Cr3+ | 200–600 | 0.83 | 447 | 57 |
| LiGa5O8:Cr3+ | 300–463 | 0.59 | 386 | 58 |
| Al2O3:Cr3+ | 292–923 | 0.9 | 450 | 59 |
| Ga2O3:Cr3+ | 298–563 | 0.46 | 370 | 60 |
| ZnGa2O3:Cr3+ | 298–563 | 0.58 | 420 | 60 |
| CaMgGe2O6:Cr3+ | 298–450 | 1.4 | 390 | This work |
Temperature-dependent decay
The decay curve of the CMGO:0.01Cr3+ phosphor was obtained by excitation at 405 nm and monitoring the emission wavelength at 704 nm (Fig. 7a). The double exponential fit (DEF) is often used when the emission from optically active ions is localized in two different crystallographic positions from the point of view of local point symmetry. In this case, the DEF was used with a good correlation between the experimental decay and the fit. The lifetime values of the fast (τ1) and slow (τ2) components were determined using the following fitting formula, which describes the double exponential function:61| I = I0 + A1e−(t−t0)τ1 + A2e−(t−t0)τ2 | (9) |
 | ||
| Fig. 7 (a) Decay profiles of CMGO:0.01Cr3+ measured in the range of temperature 298–578 K. (b) The average emission lifetime as a function of temperature. | ||
The Cr3+ excitation lifetime is an average lifetime value based on the 2E and 4T2 excitation state population. As the temperature increases, the electrons gain enough energy to repopulate the higher 4T2 state and the population of the 4T2 state starts to increase and at the same time the population of the 2E state starts to decrease. Thus, 4T2 → 4A2 is dominant over 2E → 4A2. The decay rate of the 2E state is slower than that of the 4T2 state. Consequently, the lifetime decreases with increasing temperature.62–64
The emission lifetime reflects the decay of the excited state population of Cr3+ ions and decreases with increasing temperature due to enhanced non-radiative relaxation mechanisms as shown in Fig. 7b.
Two different environments have been occupied by the doped ions. Ions with shorter lifetimes (τ1) are located at the surface or near the defect site and ions with longer lifetimes (τ2) are situated in the center of the crystallite site and far from the defect site. In this context, the average lifetime was estimated using the formula:65
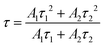 | (10) |
Since a low activation energy facilitates non-radiative quenching of the 2E state, the results obtained are more or less in line with the expectations. An increase in temperature results in a progressive shortening of the lifetime, as can be seen for CMGO:0.01Cr3+ microcrystals (see Fig. 8a for other luminophores). This dependence is an indication that the microcrystals can be used for non-contact temperature detection. In order to quantify the observed thermal changes, the relative sensitivities (Sr) of the investigated LTs were determined according to the following formula:66,67
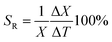 | (11) |
 | ||
| Fig. 8 (a) Thermal evolution of the mean decay times for Cr3+-doped CMGO. (b) Relative sensitivities as a function of temperature. | ||
To analyze the performance of a sensor, one important parameter is required in addition to the sensitivity, which is called the temperature uncertainty or the minimum thermal resolution. This parameter can be estimated using the equation:68
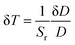 | (12) |
The work presented in this article demonstrates that the use of Cr3+ dopants, each having a different quenching rate and energy flowing in the electronic level with little interference with each other, is an attractive way to design luminescence thermometers that operate over a wide temperature range. Using this approach, a CMGO:0.01Cr3+ phosphor thermometer has been developed with an exceptional operating range of 298 → 578 K, with reasonable Sr values over this range while preserving reasonable thermal resolution.
Table 2 shows that the sensitivity of our samples is higher than the values reported so far in other host materials. It is evident from the comparative analysis, that the CMGO:0.01Cr3+ phosphor synthesized in this work exhibits unique thermometric properties in the temperature range of 298–578 K. In particular, it has an exceptionally high relative sensitivity (Sr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) max) of 2.5 K−1. This Sr
max) of 2.5 K−1. This Sr![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) max value is higher than that of any of the other materials in the comparison and makes it particularly promising for applications where high temperature resolution and accuracy are required. The combination of a moderate temperature range, improved relative sensitivity and stable luminescence intensity make CMGO:0.01Cr3+ a competitive candidate for advanced optical thermometry applications where high accuracy is critical.
max value is higher than that of any of the other materials in the comparison and makes it particularly promising for applications where high temperature resolution and accuracy are required. The combination of a moderate temperature range, improved relative sensitivity and stable luminescence intensity make CMGO:0.01Cr3+ a competitive candidate for advanced optical thermometry applications where high accuracy is critical.
| Luminescent thermometer |
S
r![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) max (% K−1) max (% K−1) |
T max (K) | λ ex (nm) | λ em (nm) | Ref. |
|---|---|---|---|---|---|
| GC CaF2:Cr3+ | 0.47 | 573 | 464 | 1000 | 71 |
| LaF3:Er3+, Yb3+/Ga2O3:Cr3+ | 0.66 | 500 | 400 | 720 | 72 |
| GC Ga2O3:Cr3+ | 0.46 | 370 | 406 | 687 | 73 |
| CaF2:Er3+/ZnAl2O4:Cr3+ | 0.67 | 535 | 396 | 690 | 74 |
| MgGa2O4:Cr3+ | 1.77 | 450 | 436 | 707 | 49 |
| ZnGa2O4:Cr3+ | 1.84 | 450 | 436 | 696 | 75 |
| Bi2Al4O9:Cr3+ | 2.13 | 550 | 600 | 705 | 66 |
| CaMgGe2O6:Cr3+ | 2.5 | 578 | 405 | 704 | This work |
Conclusion
The Cr3+-doped CaMgGe2O6 (CMGO:0.01Cr3+) phosphors were successfully synthesized using a microparticle-enhanced solid-state method. X-Ray diffraction (XRD) confirmed the formation of a phase-pure monoclinic structure with high crystallinity. The luminescence properties were found to be strongly temperature-dependent, primarily governed by the spin-forbidden 2Eg → 4A2g and spin-allowed 4T2g → 4A2g transitions of Cr3+, enabling highly precise optical thermometry. This study highlights the importance of a dual-mode thermometric approach, combining fluorescence intensity ratio (FIR) and fluorescence lifetime (FL) techniques to enhance calibration accuracy and measurement reliability.The self-referenced luminescence thermometry achieved by FIR (I589/I771) demonstrated a high relative sensitivity of 1.4% K−1 at 390 K, with an exceptionally low temperature uncertainty (δT = 0.11 K), making it a reliable method for precise temperature detection. More importantly, the integration of FL thermometry significantly improved the performance, achieving a maximum relative sensitivity of 2.5% K−1 at 578 K, along with the lowest temperature uncertainty of 1.3 K. The synergy between FIR and FL not only enhances sensitivity (Sr, Sa) but also refines the precision of the optical thermometer, addressing critical challenges in temperature sensing. These results establish Cr3+-doped CMGO as a promising alternative to lanthanide-based phosphors, providing an efficient and cost-effective solution for luminescence thermometry. By demonstrating the feasibility of Cr3+-activated phosphors for dual-mode thermometry, this work paves the way for further advances in transition metal-based optical temperature sensors and promotes their integration into next-generation high-precision thermal sensing applications.
Data availability
All data underlying the results are available as part of the article and no additional source data are required.Conflicts of interest
There are no conflicts to declare.References
- M. Dramićanin, Lanthanide and transition metal ion doped materials for luminescence temperature sensing, Luminescence Thermometry, Elsevier, 2018, pp. 113–157 Search PubMed.
- J. Xue, Z. Yu, H. M. Noh, B. R. Lee, B. C. Choi, S. H. Park, J. H. Jeong, P. Du and M. Song, Designing multi-mode optical thermometers via the thermochromic LaNbO4:Bi3+/Ln3+ (Ln = Eu, Tb, Dy, Sm) phosphors, Chem. Eng. J., 2021, 415, 128977 CrossRef CAS.
- P. Du, J. Tang, W. Li and L. Luo, Exploiting the diverse photoluminescence behaviors of NaLuF4:xEu3+ nanoparticles and g-C3N4 to realize versatile applications in white light-emitting diode and optical thermometer, Chem. Eng. J., 2021, 406, 127165 CrossRef CAS.
- D. Chrétien, P. Bénit, H.-H. Ha, S. Keipert, R. El-Khoury, Y.-T. Chang, M. Jastroch, H. T. Jacobs, P. Rustin, M. Rak and N. Lane, Mitochondria are physiologically maintained at close to 50 °C, PLoS Biol., 2018, 16(1), e2003992 CrossRef.
- S. Kiyonaka, R. Sakaguchi, I. Hamachi, T. Morii, T. Yoshizaki and Y. Mori, Validating subcellular thermal changes revealed by fluorescent thermosensors, Nat. Methods, 2015, 12(9), 801–802 CrossRef CAS PubMed.
- Y. Bahrouni, I. Kachou, K. Saidi, T. Kallel, M. Dammak and I. Mediavilla Jiménez, Enhancing Luminescence Intensity of Eu3+-Activated NaYb(MoO4)2 Phosphors through Bismuth Doping: Judd-Ofelt Analysis, Lighting, and Temperature-Sensing Applications, Mater. Adv., 2025, 6(4), 1307–1318 RSC.
- G. Wei, P. Li, R. Li, Y. Wang, S. He, J. Li, Y. Shi, H. Suo, Y. Yang and Z. Wang, How to Achieve Excellent Luminescence Properties of Cr Ion-Doped Near-Infrared Phosphors, Adv. Optical Mater., 2023, 11, 2301794 CrossRef CAS.
- N. Ahmed, H. Kraus, H. J. Kim, V. Mokina, V. Tsiumra, A. Wagner, Y. Zhydachevskyy and V. B. Mykhaylyk, Characterisation of tungstate and molybdate crystals ABO4 (A = Ca, Sr, Zn, Cd; B = W, Mo) for luminescence lifetime cryothermometry, Materialia, 2018, 4, 287–296 CrossRef CAS.
- X. F. Wang, Q. Liu, Y. Y. Bu, C. S. Liu, T. Liu and X. H. Yan, Optical temperature sensing of rare-earth ion doped phosphors, RSC Adv., 2015, 5, 86219–86236 RSC.
- M. D. Dramićanin, B. Milićević, V. Đorđević and Z. Ristić, Li2TiO3:Mn4+ Deep-Red Phosphor for the Lifetime-Based Luminescence Thermometry, ChemistrySelect, 2019, 4, 7067–7075 CrossRef.
- D. Chen, X. Chen, X. Li, H. Guo, S. Liu and X. Li, Cr3+-doped Bi2Ga4O9-Bi2Al4O9 solid-solution phosphors: crystal-field modulation and lifetime-based temperature sensing, Opt. Lett., 2017, 42, 4950–4953 CrossRef CAS.
- J. Zhang, Y. Zhang and X. Jiang, Investigations on upconversion luminescence of K3Y(PO4)2:Yb3+-Er3+/Ho3+/Tm3+ phosphors for optical temperature sensing, J. Alloys Compd., 2018, 748, 438–445 CrossRef CAS.
- M. Prasad and V. K. Rai, Thermally stable upconverting Na3Zr2(SiO4)2PO4:Er3+/Yb3+ phosphors for displays and optical thermometry, J. Alloys Compd., 2022, 911, 164968 CrossRef CAS.
- C. Li, B. Chen, D. Deng, M. Wu, H. Yu, H. Li, C. Shen, L. Wang and S. Xu, Ratiometric optical thermometer with high-sensitive temperature sensing based on synergetic luminescence of Ce3+-Eu2+ in LiSr4(BO3)3 phosphors, J. Alloys Compd., 2020, 838, 155675 CrossRef CAS.
- S. Zhu, D. Zhao, R. Zhang, Q. Yao and W. Liu, Study on energy transfer and temperature sensing properties of Pb2+/Sm3+ co-doped Ba2MgY2(BO3)4, Ceram. Int., 2023, 49, 18654–18661 CrossRef CAS.
- S. P. Singh, M. Kim, W. B. Park, J. W. Lee and K. S. Sohn, Discovery of a red-emitting Li3RbGe8O18:Mn4+ phosphor in the alkali-germanate system: structural determination and electronic calculations, Inorg. Chem., 2016, 55, 10310–10319 CrossRef CAS PubMed.
- Q. Huang, W. Ye, G. Hu and X. Liu, Strong red emission in Bi3+ and Mn4+ codoped Mg3.5Ge1.25O6 phosphors applied in optical agriculture, J. Lumin., 2019, 210, 89–95 CrossRef CAS.
- Q. Bai, Z. Wang, P. Li, T. Li, S. Xu and Z. Yang, Luminescence properties of novel deep-red-emitting Ca3Al2Ge2O10:Cr3+ phosphors, Luminescence, 2016, 31, 1277–1282 CrossRef CAS.
- K. Thejas, M. Abraham, A. Kunti, M. Tchernychev, S. Ahmad and S. Das, Review on deep red-emitting rare-earth-free germanates and their efficiency as well as adaptability for various applications, Appl. Mater. Today, 2021, 24, 101094 CrossRef.
- P. Li, L. Wondraczek, M. Peng, Q. Zhang and A. Setlur, Tuning Mn4+ red photoluminescence in (K,Rb)2Ge4O9:Mn4+ solid solutions by partial alkali substitution, J. Am. Ceram. Soc., 2016, 99, 3376–3381 CrossRef CAS.
- J. Kunitomo, R. Suzuki, Y. Takahashi, T. Miyazaki, N. Terakado and T. Fujiwara, Red-emissive Mn-doped Li2Ge4O9 phase synthesized via glass-ceramic route, J. Ceram. Soc. Jpn., 2014, 122, 725–727 CrossRef.
- J. Xue, W. Ran, H. M. Noh, B. C. Choi, S. H. Park, J. H. Jeong and J. H. Kim, Influence of alkaline ions on the luminescent properties of Mn4+-doped MGe4O9 (M = Li2, LiNa and K2) red-emitting phosphors, J. Lumin., 2017, 192, 1072–1083 CrossRef CAS.
- H. Zeng, T. Zhou, L. Wang and R.-J. Xie, Chem. Mater., 2019, 31, 5245 CrossRef CAS.
- J. Qiao, G. Zhou, Y. Zhou, Q. Zhang and Z. Xia, Nat. Commun., 2019, 10, 5267 CrossRef.
- W.-T. Huang, C.-L. Cheng, Z. Bao, C.-W. Yang, K.-M. Lu, C.-Y. Kang, C.-M. Lin and R.-S. Liu, Angew. Chem., Int. Ed., 2019, 58, 2069 CrossRef CAS.
- L. Chen, S. Yu, G. Shen, S. Tang, T. Zhang, J. Li and Q. Zhu, Large-scale irrigation of Cr3+ into different octahedra of zinc aluminate toward continual broadband near-infrared emission, Ceram. Int., 2024, 50, 1956–1969 CrossRef CAS.
- L. Chen, P. Li, G. Shen, X. Kang, S. Tang, T. Zhang and Q. Zhu, Effective electron trap regulation in near-infrared persistent phosphor of ZnAl2O4:Cr3+ for round-the-clock plant lighting, Mater. Today Chem., 2024, 40, 102204 CrossRef CAS.
- L. Fang, L. Zhang, H. Wu, H. Wu, G. Pan, Z. Hao, F. Liu and J. Zhang, Efficient Broadband Near-Infrared CaMgGe2O6:Cr3+ Phosphor for pc LED, Inorg. Chem., 2022, 61, 8815–8822 CrossRef CAS.
- L. Bellucci, M. Cassetta, H. Skogby and S. Nazzareni, Pure and Sc-Doped Diopside (CaMgSi2O6) Vibrational Spectra: Modelling and Experiments, Phys. Chem. Chem. Phys., 2024, 26, 4029–4038 RSC.
- R. D. Shannon, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta Crystallogr., Sect. A: Found. Crystallogr., 1976, 32, 751–767 CrossRef.
- M. Akasaka, Y. Takasu, M. Handa, M. Nagashima, M. Hamada and T. Ejima, Distribution of Cr3+ between octahedral and tetrahedral sites in synthetic blue and green (CaMgSi2O6)95 (CaCrAlSiO6)5 diopsides, Mineral. Mag., 2019, 83, 497–505 CrossRef CAS.
- Q. Wu, P. L. Li, Z. J. Ye, X. X. Huo, H. F. Yang, Y. Wang, D. W. Wang, J. X. Zhao, Z. P. Yang and Z. J. Wang, Near-infrared emitting phosphor LaMg0.5(SnGe)0.5O3:Cr3+ for plant growth applications: crystal structure, luminescence, and thermal stability, Inorg. Chem., 2021, 60, 16593–16603 CrossRef CAS.
- Q. Wang, S. W. Wang, T. Tan, J. T. Wang, R. Pang, D. Li, C. Y. Li and H. J. Zhang, Efficient Cr3+-activated NaInP2O7 phosphor for broadband near-infrared LED applications, Inorg. Chem. Front., 2022, 9, 3692–3701 RSC.
- K. Saidi, C. Hernández-Álvarez and M. Runowski, et al., Ultralow pressure sensing and luminescence thermometry based on the emissions of Er3+/Yb3+ codoped Y2Mo4O15 phosphors, Dalton Trans., 2023, 52, 14904–14916 RSC.
- M. Vasile, P. Vlazan and N. M. Avram, Characterization and optical properties of ZnGa2O4:Eu3+ nanophosphor grown by hydrothermal method, J. Alloys Compd., 2010, 500, 185–189 CrossRef CAS.
- L. Xi, L. Pan, Y. Wang and P. D. Townsend, The influence of doped Cr ions on the luminescence properties of infrared long persistent phosphor ZnAl2O4 with the substitution of Ge ions, J. Lumin., 2021, 233, 117941 CrossRef CAS.
- Y. Zhang, R. Huang, H. L. Li, D. J. Hou, Z. X. Lin, J. Song, Y. Z. Guo, H. H. Lin, C. Song, Z. W. Lin and J. Robertson, Germanium substitution endowing Cr3+-doped zinc aluminate phosphors with bright and super-long near-infrared persistent luminescence, Acta Mater., 2018, 155, 214–221 CrossRef CAS.
- A. Mondal and J. Manam, Structural, optical and temperature-dependent photoluminescence properties of Cr3+-activated LaGaO3 persistent phosphor for optical thermometry, Ceram. Int., 2020, 46, 25601 Search PubMed.
- N. Basavaraju, K. R. Priolkar, D. Gourier, S. K. Sharma, A. Bessière and B. Viana, The importance of inversion disorder in the visible light-induced persistent luminescence in Cr3+ doped AB2O4 (A = Zn or Mg and B = Ga or Al), Phys. Chem. Chem. Phys., 2015, 17, 1790–1799 RSC.
- Q. Zhu, J. Q. Xiahou, X. D. Li, X. D. Sun and J. G. Li, Defect cluster engineering in ZnGa2−x(Mg/Ge)xO4:Cr3+ nanoparticles for remarkably improved NIR persistent luminescence, J. Am. Ceram. Soc., 2021, 104, 4594–4605 CrossRef CAS.
- T. Narendrudu, S. Suresh, G. C. Ram, N. Veeraiah and D. K. Rao, Spectroscopic and structural properties of Cr3+ ions in lead niobium germanosilicate glasses, J. Lumin., 2017, 183, 172–179 CrossRef.
- M. Back, E. Trave, J. Ueda and S. Tanabe, Ratiometric optical thermometer based on dual near-infrared emission in Cr3+-doped bismuth-based gallate host, Chem. Mater., 2016, 28, 8347–8356 CrossRef CAS.
- C. X. Yuan, R. Y. Li, Y. F. Liu, L. L. Zhang, J. H. Zhang, G. Leniec, P. Sun, Z. H. Liu, Z. H. Luo, R. Dong and J. Jiang, Efficient and broadband LiGaP2O7:Cr3+ phosphors for smart near-infrared light-emitting diodes, Laser Photonics Rev., 2021, 15, 2100227 CrossRef CAS.
- J. Yang, Y. X. Liu, Y. Y. Zhao, Z. Gong, M. Zhang, D. T. Yan, H. C. Zhu, C. G. Liu, C. S. Xu and H. Zhang, Ratiometric afterglow nanothermometer for simultaneous in situ bioimaging and local tissue temperature sensing, Chem. Mater., 2017, 29, 8119–8131 CrossRef CAS.
- Y. Li, Y. Y. Li, R. C. Chen, K. Sharafudeen, S. F. Zhou, M. Gecevicius, H. H. Wang, G. P. Dong, Y. L. Wu, X. X. Qin and J. R. Qiu, Tailoring of the trap distribution and crystal field in Cr3+-doped non-gallate phosphors with near-infrared long-persistence phosphorescence, NPG Asia Mater., 2015, 7, e180 CrossRef.
- Y. Y. Zhan, Y. H. Jin, H. Y. Wu, L. F. Yuan, G. F. Ju, Y. Lv and Y. H. Hu, Cr3+-doped Mg4Ga4Ge3O16 near-infrared phosphor membrane for optical information storage and recording, J. Alloys Compd., 2019, 777, 991–1000 CrossRef CAS.
- H. H. Lin, G. X. Bai, T. Yu, M. K. Tsang, Q. Y. Zhang and J. H. Hao, Site occupancy and near-infrared luminescence in Ca3Ga2Ge3O12:Cr3+ persistent phosphor, Adv. Opt. Mater., 2017, 5, 1700227 CrossRef.
- V. Mykhaylyk, H. Kraus, Y. Zhydachevskyy, V. Tsiumra, A. Luchechko, A. Wagner and A. Suchocki, Multimodal Non-Contact Luminescence Thermometry with Cr-Doped Oxides, Sensors, 2020, 20, 5259 CrossRef CAS.
- A. Mondal and J. Manam, Investigations on spectroscopic properties and temperature-dependent photoluminescence of Cr3+ doped MgGa2O4 phosphor, Mater. Res. Express, 2019, 6, 095081 CrossRef CAS.
- L. Marciniak, M. Szalkowski, A. Bednarkiewicz and K. E. Piecka, A Cr3+ luminescence-based ratiometric optical laser power meter, J. Mater. Chem. C, 2022, 10, 11040–11047 RSC.
- M. Fhoula, K. Saidi, C. H. Álvarez, K. S. Carracedo, M. Dammak and I. R. Martín, Unlocking the luminescent potential of Pr3+/Yb3+ Co-doped Y2Mo4O15 for advanced thermometry applications, J. Alloys Compd., 2024, 979, 173537 CrossRef CAS.
- I. Kachou, M. Dammak, S. Auguste, F. Amiard and P. Daniel, A novel optical temperature sensor and energy transfer properties based on Tb3+/Sm3+ codoped SrY2(MoO4)4 phosphors, Dalton Trans., 2023, 52, 18233–18246 RSC.
- C. D. S. Brites, P. P. Lima, N. J. O. Silva, A. Millán, V. S. Amaral, F. Palacio and L. D. Carlos, Thermometry at the Nanoscale, Nanoscale, 2012, 4, 4799–4829 RSC.
- M. Back, E. Trave, J. Ueda and S. Tanabe, Chem. Mater., 2016, 28, 8347 CrossRef CAS.
- Z. Ristić, V. Đorđević, M. Medić, S. Kuzman, M. Sekulić, Ž. Antić and M. D. Dramićanin, Meas. Sci. Technol., 2021, 32, 054004 CrossRef.
- K. Elzbieciak and L. Marciniak, Front. Chem., 2018, 6, 424 CrossRef.
- X. Li, G. Jiang, S. Zhou, X. Wei, Y. Chen, C. K. Duan and M. Yin, Sens. Actuators, B, 2014, 202, 1065 CrossRef CAS.
- D. Chen, S. Liu, W. Xu and X. Li, J. Mater. Chem. C, 2017, 5, 11769 RSC.
- A. Ćirić, S. Stojadinović, Z. Ristić, Ž. Antić and M. D. Dramićanin, Sens. Actuators, A, 2021, 331, 112987 CrossRef.
- D. Chen, Z. Wan, Y. Zhou and Z. Ji, J. Eur. Ceram. Soc., 2015, 35, 4211 CrossRef CAS.
- K. E. Piecka, J. Drabik, D. Jaque and L. Marciniak, Cr3+-based nanocrystalline luminescent thermometers operating in a temporal domain, Phys. Chem. Chem. Phys., 2020, 22, 22555–22564 RSC.
- B. Malysa, A. Meijerink, W. Wu and T. Jüstel, On the influence of calcium substitution to the optical properties of Cr3+ doped SrSc2O4, J. Lumin., 2017, 190, 234–241 CrossRef CAS.
- M. Jong, L. Seijo, A. Meijerink and F. T. Rabouw, Resolving the ambiguity in the relation between Stokes shift and Huang-Rhys parameter, Phys. Chem. Chem. Phys., 2015, 17, 16959–16969 RSC.
- E. Glais, M. Pellerin, V. Castaing, D. Alloyeau, N. Touati, B. Viana and C. Chaneac, Luminescence properties of ZnGa2O4:Cr3+, Bi3+ nanophosphors for thermometry applications, RSC Adv., 2018, 8, 41767–41774 RSC.
- X. Xu, J. R. G. Chen, D. Kong, C. Gu, C. Chen and L. Kong, Bright green emission from the Mn2+-doped zinc gallogermanate phosphors, Opt. Mater. Express, 2013, 3, 1727–1732 CrossRef.
- D. Chen, X. Chen, X. Li, H. Guo, X. Liu and X. Li, Cr3+-doped Bi2Ga4O9-Bi2Al4O9 solid solution phosphors: crystal-field modulation and lifetime-based temperature sensing, Opt. Lett., 2017, 42, 4950–4953 CrossRef CAS.
- S. Gharouel, L. Labrador-Páez, P. Haro-González, K. Horchani-Naifer and M. Férid, Fluorescence Intensity Ratio and lifetime thermometry of praseodymium phosphates for temperature sensing, J. Lumin., 2018, 201, 372–383 CrossRef CAS.
- J. A. Mares, G. Boulon, A. Brenier, L. Lou and S. R. Rotman, Spectroscopy of multisites Cr3+ and Nd3+ in yttrium aluminum perovskite, Chem. Phys. Lett., 1994, 217, 105–111 CrossRef CAS.
- P. R. Nelson, A. Brenier, C. Pedrini, G. Boulon and J. A. Mares, Chromium Neodymium energy transfer in YAP, J. Phys. IV, 1991, 1, 257–262 CrossRef.
- N. L. Ross, J. Zhao and R. J. Angel, High-pressure single-crystal X-ray diffraction study of YAlO3 perovskite, J. Solid State Chem., 2004, 177, 1276–1284 CrossRef CAS.
- C. Wang, A. Wadhwa, S. Cui, R. Ma, X. Qiao, X. Fan and X. Zhang, Dual mode temperature sensing through luminescence lifetimes of F- and O-coordinated Cr3+ sites in fluorosilicate glass-ceramics, RSC Adv., 2017, 7, 52435–52441 RSC.
- Z. Fang, L. Zhao, Q. Yang, Z. Yang, Y. Cai, D. Zhou, X. Yu, J. Qiu and X. Xu, Optical thermometry properties of silicate glass ceramics with dual-phase for spatial isolation of Er3+ and Cr3+, J. Lumin., 2020, 219, 116861 CrossRef CAS.
- D. Chen, Z. Wan, Y. Zhou and Z. Ji, Cr3+-doped gallium-based transparent bulk glass ceramics for optical temperature sensing, J. Eur. Ceram. Soc., 2015, 35, 4211–4216 CrossRef CAS.
- A. Wadhwa, C. Wang, C. Wang and R. Ma, Multi-phase glass-ceramics containing CaF2:Er3+ and ZnAl2O4:Cr3+ nanocrystals for optical temperature sensing, J. Am. Ceram. Soc., 2019, 102, 2472–2481 CrossRef CAS.
- A. Mondal, S. Das and J. Manam, Investigation on spectroscopic properties and temperature-dependent photoluminescence of NIR emitting Cr3+ doped zinc gallate long persistent nanophosphor, Physica B, 2019, 569, 20–30 CrossRef CAS.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5tc01229e |
| This journal is © The Royal Society of Chemistry 2025 |