Cationic disorder effect on the structural and magnetic properties in SmCr1−xFexTiO5†
Christine
Martin
 *a,
Juan P.
Bolletta
a,
Balazs
Kobzi
b,
François
Fauth
*a,
Juan P.
Bolletta
a,
Balazs
Kobzi
b,
François
Fauth
 c,
Vivian
Nassif
de,
Emmanuelle
Suard
c,
Vivian
Nassif
de,
Emmanuelle
Suard
 d,
Alexander I.
Kurbakov
f,
Samuel
Jouen
b,
Virginie
Nachbaur
b and
Antoine
Maignan
d,
Alexander I.
Kurbakov
f,
Samuel
Jouen
b,
Virginie
Nachbaur
b and
Antoine
Maignan
 a
a
aLaboratoire CRISMAT, Normandie Université, ENSICAEN, UNICAEN, CNRS, 14050 Caen, France. E-mail: christine.martin@ensicaen.fr
bGroupe de Physique des Matériaux, Normandie Université, INSA Rouen, UNIROUEN, CNRS, 76000 Rouen, France
cALBA-CELLS, BP1413, 08290 Cerdanyola del Vallés, Barcelona, Spain
dInstitut Laue Langevin (ILL), 38042 Grenoble, France
eUniv. Grenoble Alpes, CNRS, Institut Néel, 38000 Grenoble, France
fNRC “Kurchatov Institute” – PNPI, 188300 Gatchina, Russia
First published on 28th April 2025
Abstract
Combining synchrotron X-ray and neutron powder diffraction with Mössbauer and Raman spectroscopy and magnetization measurements, a comprehensive experimental study of the SmCr1−xFexTiO5 series (x = 0, 0.25, 0.50, 0.75 and 1) is presented. The compounds which exhibit a structure derived from mullite crystallize in the orthorhombic Pbam space group in the entire series, and from room temperature down to 2 K. The transition metals occupy two crystallographic sites, one octahedron (MoctO6) and one square-based pyramid (MpyrO5), in a non-random fashion depending on x. Accordingly, different magnetic behaviours are generated from long range antiferromagnetism to spin glass. This study demonstrates that the impact of the Fe for Cr substitution on the magnetic properties is mainly due to the distribution of Ti4+ (d0), Cr3+ (d3) and Fe3+ (d5) over the M-cationic framework rather than the induced structural effect.
1. Introduction
Transition metal oxides are known as a fantastic class of materials for the huge variety of properties they exhibit in link with the plethora of structures in which they crystallize. Most of them, as perovskites, spinels, pyrochlores, Ruddlesden–Popper phases, etc. are highly flexible and can adapt various substitutions and non-stoichiometry. This means that the oxidation state of the transition metal (M) can be changed via the oxygen content and/or playing with counter cations of different valence. The lattices adapt by varying interatomic distances and angles, i.e., distortion and rotation of the elementary structural units, depending mainly on the ionic radius and the electronic configuration of the cations. Since the relationships between structures and physical properties are intertwined, the control of the structural parameters is crucial to monitor the properties. It is why solid solutions are studied, as in the well-known case of manganites that exhibit colossal magnetoresistance. Indeed, depending on the occupation of the A-site of the AMnO3 perovskite (i.e., nature and ratio of the A3+, A′2+ cations), very rich phase diagrams (from Mn3+ to Mn4+) can be drawn.1 In the same way, substitutions of Mn by other transition metals M′ provide a plethora of magnetic behaviours.2 In that manganite case, the strong impact of substitutions on the properties is linked to the cooperative distortions induced by the Jahn–Teller character of Mn3+. On the other hand, transition metal oxides, as numerous materials, may also present a disordered component from different nature (like composition, atomic displacement, charges, etc.) that is more often not random but correlated.3 For instance, the compositional flexibility of transition metal oxides can lead to the coexistence of orders at different scales, and these order/disorder phenomena appear then as a means of obtaining new properties. The current research on this topic, including both experimental and fundamental studies, testifies to the interest of this approach to design new materials, as recent works devoted to high-entropy oxides.4,5 Numerous structural types have already been studied from this angle, like pyrochlores, perovskites, or spinels,6 this approach concerning thus different types of properties3 and consequently different research fields, such as catalysis,7 batteries,8 and of course magnetism. This latter is particularly impacted in the case of the substitution of magnetic cations by non-magnetic ones that affects magnetic properties so much that the dilution can lead to a spin glass behaviour.9,10In this context, the mullite system appears as a powerful platform since this structure is able to incorporate a large variety of elements.11,12 For that matter, the formula of mullite itself already indicates a chemical complexity as described by the general formula Al4+2xSi2−2xO10−x. In addition, the robustness of this structure is not to be overlooked, as it enables applications in harsh conditions (high temperature or working conditions for catalysis applications)12,13 which is rarely the case with oxides. Several polymorphs exist in this family of aluminosilicates, and we are interested here in the AMM′O5 compounds crystallizing in the Pbam space group. In this structure, the eightfold coordinated A cation is a lanthanide (or Y) and the M and M′ cations occupy two crystallographic sites of different symmetry (octahedron and square pyramid). The MO6 octahedra share edges to form infinite chains, that are linked by pairs of M′O5 pyramids having a common edge (Fig. 1). These structural features explain for instance the interesting catalytic properties of SmMn2O5 (i.e., A = Sm and M = M′ = Mn) in connexion with the two types of crystal fields (Mn4+O6 and Mn3+O5) and the lattice oxygen lability.13,14 Other Mn-based compounds were studied for their magnetic and dielectric properties that depend on A (Y, Gd, Tb, Ho, Er, Tm) in AMn2O5.15,16 Numerous investigations were also performed in the AMTiO5 system, motivated by the multiferroic properties of NdCrTiO5.17–19 The richness of this system was first demonstrated by Buisson,20 who determined the stability of the phases as a function of the composition [A (La to Tb), M (Cr, Mn, Fe)] and the synthesis temperatures; the structural study of 15 compounds was carried out including the magnetic structure of two of them. From several publications that succeeded this pioneer work, it appeared that the Sm-based compounds behave differently.21,22 It was confirmed recently for SmCrTiO5 described as alternating Heisenberg antiferromagnetic spin-3/2 chains and compared with SmCrGeO5 for which a spin gap was evidenced.23,24 Starting from these results, it is relevant to study the impact of the gradual replacement of chromium by iron in SmCrTiO5 on structural and magnetic behaviours. It is what we are reporting in this article, supported by a comprehensive discussion of cation distribution, structural features, and magnetic properties in the SmCr1−xFexTiO5 series (0 ≤ x ≤ 1). By combining neutron and synchrotron X-ray diffraction, together with Mössbauer and Raman spectroscopies, and magnetic measurements, we assigned the source of most structural and property changes to the non-random distribution of the transition-metal cations towards the two available crystallographic sites, Moct and Mpyr.
2. Experimental procedure
2.1. Sample preparation
SmCr1−xFexTiO5 (x = 0, 0.25, 0.50, 0.75 and 1) samples were prepared by solid state reaction at high temperature. Mixtures of stoichiometric amounts of Sm2O3 (99.9%, Alfa Aesar), Cr2O3 (99%, Alfa Aesar), Fe2O3 (>99%, Sigma Alrich), and TiO2 (99.8%, Alfa Aesar) were ground in an agate mortar. The powders were then pressed into bars or pellets to be heated in air. The synthesis temperature (from 1200 °C to 1250 °C) and the duration (between 12 h and 48 h) were adapted case by case. Several successive thermal treatments were applied when necessary to decrease the amount of impurities (below 5% in mass) that appeared in the form of Sm2M2O7 pyrochlores and/or SmMO3 perovskites. Indeed, additional thermal treatments with intermediate grindings usually decrease the amount of impurities, in agreement with the domains of stability, depending on the choice of the cations and the synthesis temperature, already reported.20,23 This method has resulted in high quality samples with colours ranging from green (x = 0) to dark red (x = 1), via brown for mixed Cr/Fe samples (Fig. SI1, ESI†).To increase sensitivity to Mössbauer spectroscopy for the compound with the lower iron content, 57Fe2O3 (>99%, BuyIsotope) was used to prepared a specific sample (SmCr0.7557Fe0.25TiO5) following the procedure described above. Since natural Sm is unsuitable for diffraction with thermal neutrons due to its very high absorption cross-section, a special sample 152SmCr0.5Fe0.5TiO5 was also synthetized for neutron powder diffraction experiments using the same process but with 152Sm2O3 (Isotope JSC – ROSATOM). The SmCrTiO5 samples (x = 0) used for this study are those characterized in details in.23
2.2. Powder diffraction
The samples were systematically characterized by X-ray powder diffraction (XRPD) at room temperature (RT), by means a PANalytical X’Pert Pro diffractometer equipped with a PIXcel detector and a Co source in Bragg–Brentano geometry. For a deeper structural analysis, synchrotron X-ray powder diffraction (SXRPD) experiments were performed on the BL04-MSPD beamline at the ALBA synchrotron (Barcelona area)25 on sieved powders enclosed in capillaries (0.3 mm outer diameter). Data were recorded with λ = 0.3869 Å wavelength in the 0.3° to 54.0° 2θ-range using the high angular resolution Multi Analyzer Detection (MAD) setup. The low-temperature measurements were performed at constant temperatures between 10 K and RT in a DynaFlow lHe flow cryostat.26 Neutron powder diffraction (NPD) patterns of 152SmCr0.5Fe0.5TiO5 were obtained at the high flux D1B diffractometer at the ILL (Grenoble) with powder enclosed in a 6 mm vanadium can and using a standard orange cryostat for temperature control. Data were collected at 2 and 50 K and in dynamic conditions from 165 to 2 K with λ = 2.52 Å in the 0.8–128° 2θ-range. A RT pattern was also collected at higher angular resolution using the D2B diffractometer (λ = 1.59 Å and 0–160° 2θ-range). Diffraction data were analysed by the Rietveld method using the FULLPROF software.272.3. Mössbauer and Raman spectroscopy
Mössbauer spectroscopy analyses were carried out with a 57Co radioactive source in a Rh matrix. The powdered samples were measured in transmission mode using a constant acceleration spectrometer. For this purpose, the samples were mechanically crushed and mixed with boron nitride to achieve 10 mg Fe cm−2. To probe the SmCr0.7557Fe0.25TiO5 sample the iron density is decreased to 1 mg Fe cm−2. Isomer shifts are reported relative to metallic iron at RT. Raman spectroscopy was performed with a Horiba Jobin–Yvon LabRam Aramis Raman spectrometer, using a laser of wavelength 632.8 nm and a power of 0.2 mW. The laser beam was focused on the sample using a 100× objective giving a spatial resolution in the order of 1 μm.2.4. Magnetic characterization
Magnetic moment measurements were performed on small pieces of bars (of the order of a few mm3) using a commercial magnetic property measurement system (MPMS-5T) superconducting quantum interference device magnetometer (SQUID), warming from 5 K to 400 K in zero-field cooling and field cooling (ZFC-FC) modes with μ0H = 1 T. The magnetic susceptibility (χ) was then obtained from the magnetic moment (m) [χ = m/(magnetic field × sample mass)] for the five SmCr1−xFexTiO5 samples. The AC-option of the MPMS was also activated to measure AC-magnetic susceptibility, data were recorded increasing the temperature in 10 Oe and with frequencies of 102, 103 and 104 Hz. For isothermal magnetization loops, the AC measurement system option of a physical property measurement system (PPMS-14T) was used.3. Results and discussion
3.1. RT structural characterization
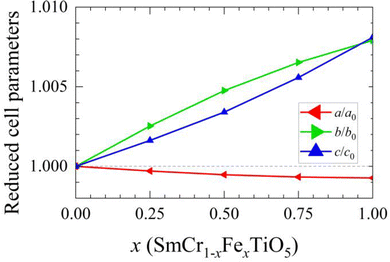
The crystalline structure of the SmCr1−xFexTiO5 samples (with x varying from 0 to 1 by 0.25 steps) were studied by means of SXRPD. A RT pattern of SmCr0.75Fe0.25TiO5 is shown as an example in Fig. 2, together with the result of its Rietveld refinement within the expected orthorhombic Pbam space group. The unit cell parameters (a, b and c) of the five samples given in Table 1 are in agreement with the reports on SmCrTiO5 and SmFeTiO5.20,23,28 The unit cell volume increases with x but this evolution is anisotropic, as shown by the plot of the reduced unit cell parameters (Fig. 3). Both the b and c parameters expand as Cr3+ is replaced by larger Fe3+ cations (rCr3+ = 0.615 Å vs. rFe3+ = 0.645 Å for coordination six29). In contrast, the a parameter shows a very slight decrease with x, of about one order of magnitude lower than the expansion of the other two cell parameters, which renders a almost constant in comparison. The dependence of each parameter is given in (Fig. SI2, ESI†). Anisotropic expansion of the unit cell with substitution by larger cations was also observed in the isostructural BiMn2−xTixO5, where Ti4+ replaces the smaller Mn4+ in the octahedral sites inducing longer a and c parameters while b stays approximately constant.30 The structurally-related aluminosilicate sillimanites, consisting on chains of edge-sharing octahedra connected by pairs of tetrahedra instead of square pyramids, also display anisotropic expansion linked to site-selective substitutions, with different changes in cell parameters depending on whether substituting cations locate on octahedral or tetrahedral sites.31 With this in mind, it becomes imperative to accurately determine the distribution of the cations (Cr3+, Fe3+ and Ti4+) in SmCr1−xFexTiO5 across the two available sites (Moct and Mpyr) using the multi-technique approach, mixing Mössbauer and Raman with SXRPD and NPD.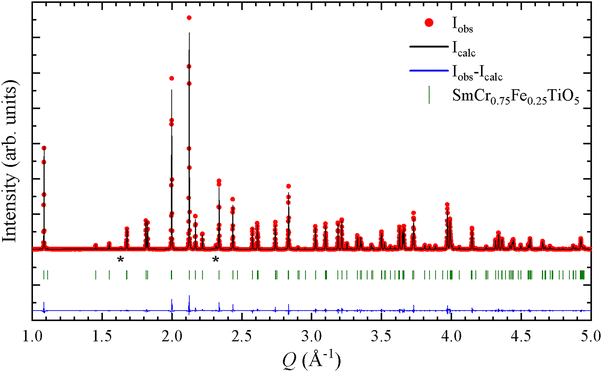 | ||
| Fig. 2 RT SXRPD pattern of SmCr0.75Fe0.25TiO5. The asterisks indicate peaks from an impurity with an orthorhombic perovskite-type structure (<1.5% in mass). | ||
| x (Fe3+) | a (Å) | b (Å) | c (Å) | V (Å3) | Formula |
|---|---|---|---|---|---|
| 0.00 | 7.50414(2) | 8.61579(3) | 5.78412(2) | 373.967(4) | Sm(Cr0.94Ti0.06)oct(Cr0.06Ti0.94)pyrO5 |
| 0.25 | 7.50193(2) | 8.63760(2) | 5.79356(2) | 375.415(3) | Sm(Cr0.66Fe0.10Ti0.24)oct(Cr0.09Fe0.15Ti0.76)pyrO5 |
| 0.50 | 7.50022(2) | 8.65677(2) | 5.80380(2) | 376.827(3) | Sm(Cr0.39Fe0.26Ti0.35)oct(Cr0.11Fe0.24Ti0.65)pyrO5 |
| 0.75 | 7.49912(2) | 8.67199(2) | 5.81639(2) | 378.253(3) | Sm(Cr0.11Fe0.43Ti0.46)oct(Cr0.14Fe0.32Ti0.54)pyrO5 |
| 1.00 | 7.49867(2) | 8.68400(2) | 5.83097(1) | 379.703(3) | Sm(Fe0.59Ti0.41)oct(Fe0.41Ti0.59)pyrO5 |
3.2. Cation distribution
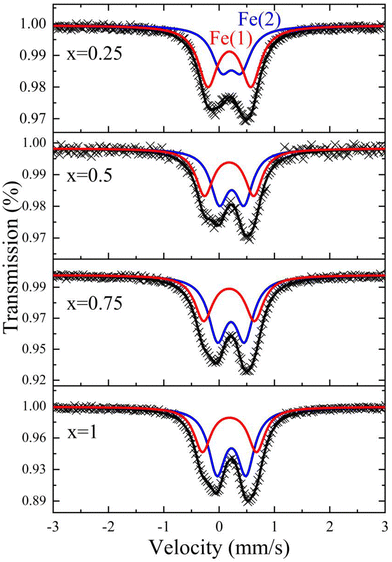
The 293 K Mössbauer spectra of the SmCr1−xFexTiO5 series are shown in Fig. 4. They are very similar to each other and are fitted by superposition of two quadrupolar doublets characteristic of paramagnetic behaviour. The need to use two quadrupolar components, labelled Fe(1) and Fe(2), indicates that iron is present in two chemical environments, even for the lowest amount of iron. The hyperfine Mössbauer parameters of the Fe(1) and Fe(2) components are listed in Table 2, namely A (area of the component), IS (isomer shift), QS (quadrupole splitting). The isomer shift depends on the local electron density at the nucleus and thus informs on the oxidation state. For iron, IS is known to increase when the density of s-electrons at the nucleus decreases, which occurs for a same valence state when the coordination number increases.32 Depending on the iron content, IS(1) varies between 0.305–0.313 mm s−1 and IS(2) between 0.340–0.351 mm s−1. All these IS values are characteristic of a high-spin Fe3+ valence state, excluding the presence of other oxidation state. The difference in IS between the two components Fe(1) and Fe(2) is significant enough to assign each of them to a crystallographic site. The paramagnetic doublet with the lowest isomer shift, Fe(1), is assigned to Fe3+ in the O5 square pyramidal sites, while the second one, Fe(2), with the highest isomer shift, is assigned to Fe3+ in the O6 octahedral sites. The QS value is proportional to the electric field gradient (EFG) at the iron nucleus due to a non-spherical charge density distribution in the nearest neighbours of the probing nucleus. The largest value of the quadrupole splitting QS(1) [0.78–0.98] mm s−1, compared to QS(2) [0.35–0.52] mm s−1, indicates the lowest symmetry in coordination and charge distribution for the environment (1), consistent with the assignment of Fe(1) and Fe(2) to square pyramidal and octahedral sites, respectively. The presence of Fe3+ only and its distribution on both sites were also observed in AFeTiO5, AMn0.95Fe0.05TiO5 and ACr0.95Fe0.05TiO5 (A = Pr, Nd, Sm, Eu, Gd)33 and in YbFeMnO5.34 The Fe3+ distribution in both sites is determined from the Mössbauer sub-spectrum relative area (Table 2) assuming that Lamb–Mössbauer factors of iron are equal in both environments. The proportion of iron in the octahedral site increases with the iron content. For x = 0.25 the proportion of iron is larger in the pyramids (60%) than in the octahedra whereas in SmCr0.25Fe0.75TiO5 the opposite situation is observed with 43% in the pyramids.| x | Site | A (%) | IS (mm s−1) | QS (mm s−1) |
|---|---|---|---|---|
| 0.25 | Pyramidal | 60.2 ± 0.1 | 0.308 ± 0.002 | 0.78 ± 0.01 |
| Octahedral | 39.8 ± 0.1 | 0.343 ± 0.002 | 0.35 ± 0.01 | |
| 0.5 | Pyramidal | 47.7 ± 0.1 | 0.306 ± 0.008 | 0.89 ± 0.03 |
| Octahedral | 52.3 ± 0.1 | 0.351 ± 0.008 | 0.45 ± 0.03 | |
| 0.75 | Pyramidal | 42.7 ± 0.1 | 0.305 ± 0.005 | 0.92 ± 0.02 |
| Octahedral | 57.3 ± 0.1 | 0.340 ± 0.005 | 0.49 ± 0.02 | |
| 1 | Pyramidal | 41.3 ± 0.1 | 0.313 ± 0.002 | 0.98 ± 0.01 |
| Octahedral | 58.7 ± 0.1 | 0.348 ± 0.002 | 0.52 ± 0.01 |
The Raman spectra of these samples, shown in Fig. SI3 (ESI†), are consistent with the results of the Mössbauer study. The broadening of peaks in the medium frequency range corresponds to a mixed occupation of the octahedral and pyramidal sites and their shifts toward the lower frequencies correlate with a higher amount of iron in the two sites as x increases. The evolution of the peak intensity at 820 cm−1, attributed to internal vibrations in TiO5 polyhedra, that decreases as the iron content increases, confirms the presence of iron into the square-pyramidal site with titanium.
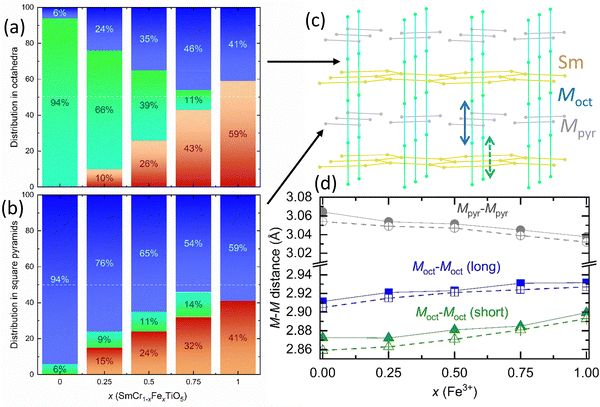
Given the small differences of electronic densities and scattering factors between the M3+ (i.e., d5 Fe3+ and d3 Cr3+) cations and d0 Ti4+, only an estimation of their distribution over the two available crystallographic sites is possible from SXRPD data. This was made without differentiating Cr and Fe (M), due to the difference of 2 electrons only, and was incorporated into the Rietveld analysis as variable fractional occupations (M and Ti) of the crystallographic sites, constrained in a way that the total stoichiometry is kept constant throughout the calculations. By combining this distribution of M3+ and Ti4+ with the one of Fe3+ obtained from Mössbauer spectroscopy, a contribution of each cation to each crystallographic site can be proposed leading to the structural formulae given in Table 1 and illustrated in Fig. 5. The cationic distributions are also supported by the average M–O distances that will be discussed later in Section 3.3.
The distribution of cations was further confirmed by the use of neutron powder diffraction for the two compounds, 152SmCrTiO5 and 152SmCr0.5Fe0.5TiO5. The scattering lengths for Cr (3.635 fm), Fe (9.45 fm), and Ti (−3.438 fm)35 are sufficiently different that their respective occupations can be considered separately in the Rietveld analysis. The methodological approach is similar to that used for the analysis of SXRPD data, with some constrains to properly calculate occupations when three cations are involved in two available sites, using the Ti4+ distribution obtained from SXRPD as a starting point of the analysis. The obtained formulae are in perfect agreement with the results obtained from the combination of Mössbauer spectroscopy and SXRPD within the margin of error. More details are given for the x = 0 composition in.23
The occupation of both sites can be explained by the different electronic configurations of the cations, as high-spin Fe3+ is a spherical d5 cation with a zero crystal field stabilization energy (CFSE) in the octahedral site, while Cr3+ is a d3 cation that has a large CFSE in octahedral site.20 In parallel, Ti4+ can adapt both octahedral and pyramidal environments, included in a same structure, as in zorite mineral (Na6Ti(Ti,Nb)4(Si6O17)2(OH,O)4(OH)(H2O)10.5),36 even if the 5-coordination is less common and occurs mainly in titanosilicates.37
It follows that the effect of substitution of transition metals in these materials is twofold, due to the identity of the M cations (oxidation state and size) and their amount, but also to the disorder they induce (or not) by their distribution in pyramids and octahedra. Nevertheless, the distribution of transition metals in AMTiO5 is not always addressed in detail in the available literature. For ACrTiO5 materials, the neutron powder diffraction experiments performed by Buisson revealed a strong preference of the Cr3+ cation towards the octahedral sites at 95%.20 Same observation was done in the isostructural ACrGeO5 materials, with little to no signs of disorder between the two sites.38 However, for AFeTiO5, Fe3+ is more evenly distributed over octahedra and square pyramids, but there is also a small effect of A on the distribution.20 In addition, for a same compound, namely NdFeTiO5, differences have been reported with an occupation of pyr-43%/oct-57% by20 and of pyr-60%/oct-40% by.28 These discrepancies are probably assigned to different synthesis process, and in any case, the cationic order is stronger for the Cr-side (near 100%) than for the Fe-side (close to a random distribution).
3.3. Structural analysis vs. x in SmCr1−xFexTiO5
The level of substitution (x) and the distribution of the transition metal between the two available crystallographic sites lead to small structural changes. The three types of polyhedra present in SmCr1−xFexTiO5 change in size as composition varies, as illustrated by the Fig. 6 in which the volume of each polyhedron has been divided by the volume observed for x = 0. The SmO8 unit shows the least change across the series, staying within 1% of its original volume. The MO6 octahedron displays small variations, with some contraction and expansion that end up cancelling each other to produce almost no net change. In stark contrast, the volume of the MO5 square pyramid markedly increases as the Fe content increases, with constant expansion that reaches almost 6% for complete substitution. The uneven changes in volumes between the polyhedra may be the source of the anisotropic change in the cell parameters with respect to x, as the expansion of the square pyramids is not accompanied by the other polyhedra, which behave as relatively rigid units and adapt by rotations or small displacements rather than changes in distances and volumes.Taking into account the cationic formula (Table 1) and the ionic radii of the Shannon tables,29 the replacement of Cr3+ by Fe3+ leads to an increase of the average radius of each site, larger in the square pyramid than in the octahedron. Indeed, the increase due to the larger amount of Fe3+ (rFe3+ > rCr3+) is counter balanced by the concomitant increase of Ti4+ (rCr3+ > rTi4+) amount in the octahedron leading to a charge increase. In contrast the changes in the pyramid involve an increase of the radius and correspond to an overall decrease of the charge (q). Both q and r parameters can be combined using the q/r ratio for each site, highlighting the opposite trend for the pyramid and the octahedron. This anisotropic expansion of the volume of the octahedral and the pyramidal polyhedra is confirmed by the evolution of the quadrupole splitting which increases with x. This corresponds to an increase of the EFG at the iron nucleus linked to the coordination symmetry and charge distribution around iron in both sites.
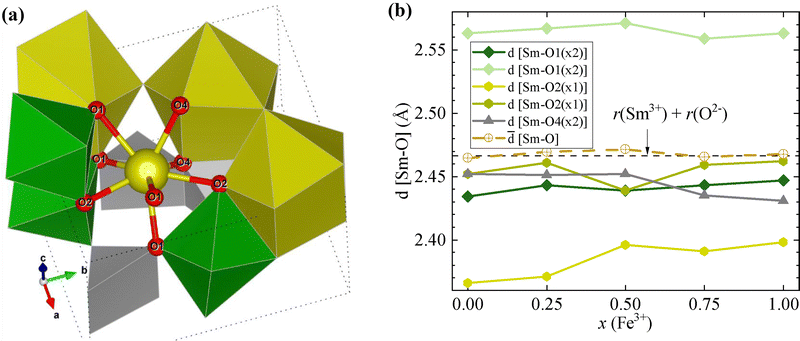
Regardless of composition, the Sm–O distances stay approximately constant, in the range of expected lengths,29 suggesting that the SmO8 units remain as mostly rigid, limiting the displacement of the O bonded to Sm, whatever identity and distribution of the M cations (Fig. 7). It is in agreement with the Raman spectra that indicate no shift of the peaks characteristic of the SmO8 polyhedron along the series. Similar observations for AO8 units in ACrMnO5 and NdMnTiO539,40 suggest that this polyhedron behaves rigidly, even in the presence of Jahn–Teller-active Mn3+, demonstrating thus the particular structural role of the A3+.
The M–O distances in the octahedra are drawn in Fig. 8. There are three sets of two distances, i.e. with O1, O2 and O3. M–O2 is the shortest and is the only one that has a clear expansion when increasing Fe content, more pronounced in the right part, i.e. for x > 0.5. This distance, shorter than the expected one obtained from the sum of ionic radii,29 involves O2 that is linked to two Moct and to two Sm. These O2 belong to one of the common edges of the MO6 octaedra that form chains running along the c-axis. The two M linked by O2 correspond to the shorter distance M–M along the chain, that also increases with x (Fig. 5(d)). The distance between M and O1, that is common to one M-octahedron, the basal plane of one M-pyramid and two lanthanide polyhedra, remains approximately constant vs. x. In contrast, the bond with O3, that is shared between two MO6 octahedra and is also the apical oxygen of the square pyramid, is smaller in SmFeTiO5 than in SmCrTiO5, it first contracts and then expands slightly vs. x. The two M linked by two O3 correspond to the longer M–M distance of the chain, that surprisingly increases with x.
When analysing the M–O bonds that compose the pyramid, as detailed in Fig. 9, there is an overall expansion across the series. The apex distance of the pyramid (O3) shorter than the four distances of the basal plane (O1 and O4) is common to TiO5 units, and is often referred as titanyl bond to evidence a more covalent character for this axial bond compared to the four others. This specific arrangement plays an important role in the chemistry of Ti-based oxides and is at the origin of functional properties as photoluminescence in titanosilicates for instance.37 This apical distance is the one that varies most with x, in agreement with the decrease of its titanyl character since the content of Ti in the pyramid decreases. The O3 anion implied in this short distance is involved in a rather flexible M–O bond of the octahedra (Fig. 8) and is not bonded to a Sm, making it the more mobile of the O. The Mpyr–Mpyr distance that corresponds to two M linked by O4 (with dM-O4 > dM-O1) decreases with x, even if the M–O distances increases.
The Mössbauer parameters of the Fepyr and Feoct sub-spectra could reflect the structural modifications along the series, particularly the isomer shift, which is primarily influenced by changes in the interatomic distances and the covalent character of the Fe3+–O2− bond. An increase in the Fe–O distance or a decrease of the covalent character in the bond lead to the lowering of the 4s-electron density at the iron nucleus, thereby increasing the IS values.32 This is not what is observed since IS values appear relatively constant for both sites regardless to the iron content (Table 2). Even though small variations in Fe–O distances have been established, the covalent character of the bond is more difficult to assess, as it can be influenced by the electronegativity of the neighboring cation M that shares an oxygen with iron. According to Portier et al.,41 cations rank in ascending order of electronegativity as Ti4+ < Fe3+(high spin) for the pyramidal site and Ti4+ < Cr3+ < Fe3+ (high spin) for the octahedral site. As x increase, the substitution of Ti4+ in MO5 units by the more electronegative cation Fe3+ contribute to lowering the degree of covalency of the Fepyr–O bond. Regarding the MO6 units, substituting Cr3+ with Ti4+ (less electronegative) and Fe3+ (more electronegative) has opposing effects, preventing a clear trend on the covalent character of the Feoct–O bond with increasing iron content. Since octahedral and pyramidal sites are interconnected, the substitution of chromium and titanium by iron affects the covalence of Fe–O bonds at each site differently, complicating the interpretation of the IS evolution vs. x.
3.4. Structural evolution vs. temperature
To go further, a structural study was performed at low temperature for the five samples. Whatever x, no structural transition is observed from SRXPD measurements within the 10 K – RT range. The Pbam structure is preserved in all cases and the lattice volume of the samples decrease with decreasing temperature (Fig. 10). Nevertheless, SmCrTiO5 differs from other samples, displaying negative thermal expansion along the a axis23 whereas the rest of the cell parameters as well as the other materials contract progressively with decreasing temperatures. | ||
| Fig. 10 Variation of cell parameters and unit cell volume with temperature for SmFe1−xCrxTiO5 obtained from SXRPD data. | ||
The lack of structural transition is confirmed by the NPD study of 152SmCr0.5Fe0.5TiO5 that also indicates no long range magnetic ordering down to 2 K, since no new Bragg peaks nor extra intensity are observed (Fig. 11). In addition, even at the lower temperatures there is no change in the background of the NPD patterns which could have been an indication of short range magnetic ordering.
 | ||
| Fig. 11 Experimental NPD patterns of 152SmCr0.5Fe0.5TiO5 recorded at 2 and 50 K (blue and pink lines, respectively). The 50 K pattern is shifted up to avoid the superposition of the two patterns. The row of vertical ticks corresponds to the Bragg positions (using RT parameters given in Table 1 and Table SI1, ESI†). | ||
At 10 K, the dependence of the M–M distances vs. x follows the same trend than at RT (Fig. 5(d)), i.e. Mpyr–Mpyr decreases whereas long and short Moct–Moct increase with x, like the larger Mpyr–Moct distances (≈3.5 Å). The distortion of the lattice is thus similar at RT and 10 K whatever x; the alternating Heisenberg character with the alternation of short and long Moct–Moct distances along the c axis is maintained all along the series, but the difference between them is smaller on the Fe-rich side.
3.5. Magnetic properties
The temperature dependence of the magnetic susceptibility (χ) collected in 1 T is plotted in Fig. 12(a) for the five SmCr1−xFexTiO5 samples. For SmFeTiO5, a clear change of slope is observed at 53 K which corresponds to the TN = 52.8 K determined by Mössbauer.33 In order to study the paramagnetic regime, a first attempt was made to fit the χ(T) curve beyond 100 K using a Curie–Weiss law, χ = χ0 + C/(T − Θp). Despite a good match, the obtained μeff value of 2.86μB is much smaller than the expected one, μeff = 5.92μB for high-spin Fe3+. Even performing measurements up to 950 K within the same applied external magnetic field, the curve χ−1(T) does not reach a linear paramagnetic regime (Fig. SI4, ESI†), suggesting that local magnetic interactions are still at play in such temperature range. Their origin is still unclear; it could be attributed to cationic disorder or to a lifting in the orbital degeneracy due to spin orbit coupling which makes the local magnetic moment temperature-dependent.In contrast to the clear signature of TN on the χ(T) curve of SmFeTiO5, the antiferromagnetic transition that occurs at TN = 11 K33 for SmCrTiO5 could be hardly detected in the χ(T) curve, only a small downturn is observed.23 In addition, the quasi T-independent χ(T) curve in the 30 to 300 K range was interpreted as the signature of a spin gap opening by similarity with the isotructural germanate SmCrGeO5.24 Accordingly, the observed magnetic susceptibility values were much smaller than the calculated ones considering independent Cr3+ paramagnetic cations.24
The transition from one magnetic behaviour, SmCrTiO5 (TN = 11 K), to another, SmFeTiO5 (TN = 53 K), is gradual, as shown by the progressive changes vs. x in the χ(T) curves, whose magnitude increases with the iron content (Fig. 12(a)). Such an evolution is in agreement with the M(H) curves registered at 5 K in the −14 T to +14 T range (Fig. SI5, ESI†). The maximal magnetization value is reached for SmFeTiO5 with 0.8μB f.u.−1 at 5 K in 14 T, which is far from the complete ferromagnetic alignment of the Fe3+ spins, that would be 5μB f.u.−1 The χ(T) curves of the mixed Cr/Fe compounds are enlarged below 50 K in Fig. 12(b) showing at first glance magnetic-like transitions. As the NPD patterns of SmCr0.5Fe0.5TiO5 do not evidence magnetic ordering, even at 2 K (Fig. 11), AC-magnetic susceptibility measurements versus temperature were recorded at different frequencies f (Fig. 13(a)). The corresponding χ′(T) curves exhibit a cusp at 18.3 K for 0.1 kHz that is shifted up by 1.0 K for 10 kHz. Such a frequency dependence at the χ maximum is consistent with a temperature Tf of spin freezing characteristic of spin glasses which frequency dependence can be quantified by K,42K = ΔTf/[TfΔlogf]. From the Tf = 19.3 K and ΔTf = 1.0 K values for two frequency decades, one obtains K = 0.026 which makes SmCr0.5Fe0.5TiO5 enters the category of the spin glasses for which K values are in between 0.005 and 0.08.42 For this composition also, the Curie–Weiss fitting gives an unrealistic μeff value of 2.43μB instead of a calculated μeff = [0.5 × 3.872 + 0.5 × 5.922]1/2 = 5.00μB. Clearly, the magnetic susceptibility is much smaller than the calculated one considering μeff = 5.00μB for free paramagnetic Fe3+ and Cr3+ cations: at 300 K, χ300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K = C/T300
K = C/T300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K = (μeff2/8)/300 = 0.01042 emu mol−1, against an experimental value χ300
K = (μeff2/8)/300 = 0.01042 emu mol−1, against an experimental value χ300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) K − χ0 of 0.00241 emu mol−1. Similarly, SmCr0.75Fe0.25TiO5 exhibits also a spin glass behaviour (Fig. 13(b)), with a lower Tf (= 9.8 K) and ΔTf = 0.74 K for the same two decades, leading to K = 0.038. On the other side, the χ(T) curve of SmCr0.25Fe0.75TiO5 shows a shape close to that of SmFeTiO5 and a transition at ≈36 K (Fig. 12) but without frequency dependence in χ′(T) measurements (not shown). The magnetic measurements reveal thus various behaviours depending on x, such as antiferromagnetism or spin glass, whose origins are discussed below.
K − χ0 of 0.00241 emu mol−1. Similarly, SmCr0.75Fe0.25TiO5 exhibits also a spin glass behaviour (Fig. 13(b)), with a lower Tf (= 9.8 K) and ΔTf = 0.74 K for the same two decades, leading to K = 0.038. On the other side, the χ(T) curve of SmCr0.25Fe0.75TiO5 shows a shape close to that of SmFeTiO5 and a transition at ≈36 K (Fig. 12) but without frequency dependence in χ′(T) measurements (not shown). The magnetic measurements reveal thus various behaviours depending on x, such as antiferromagnetism or spin glass, whose origins are discussed below.
 | ||
| Fig. 13 AC-magnetization curves χ′(T) of SmCr0.5Fe0.5TiO5 (a) and SmCr0.75Fe0.25TiO5 (b) recorded within 10 Oe AC-field and frequencies of 102, 103 and 104 Hz. | ||
3.6. Discussion
Even if it does not take into account the M–O distances and M–O–M angles, the cationic network drawn in Fig. 5(c) can be used in a rough analysis of the magnetic properties. Two magnetic sub lattices can be considered, one made of octahedra sharing edges to form files running along the c axis and Mpyr2O8 units which link the chains (Fig. 1(a) and 8(a)). In the chains of MoctO6, two Moct–Moct distances alternate along c, they increase both with the iron substitution (from 2.8727 Å and 2.9114 Å in SmCrTiO5 to 2.8995 Å and 2.9315 Å in SmFeTiO5, Fig. 5(d)). In contrast, the Mpyr–Mpyr distance (>Moct–Moct) decreases with x, even if the Mpyr–O distances increase (Fig. 5(d) and 9). It is like a scissors effect, in link with the opposite evolution of the a and b parameters vs. x (Fig. 3). For the same reason, the distorted HC-like Sm network, with 4 long and 2 short Sm–Sm distances in the ab plane, shows an anisotropic evolution with x, the long one increases of 0.2% whereas the short one decreases of 0.9% although the SmO8 polyhedra are described as rather rigid. The lanthanide has an important role because it controls the short Moct–Moct distance in the chain (Fig. 5(c)) and steric effects influence probably the distribution of cations. Cr3+ prefers the octahedral sites (due to the CFSE); as soon as it is replaced, Fe3+ distributes between the octahedra and the square pyramids. This means that the smaller Ti4+ is displaced from the square pyramids into the octahedra, moderating the increase in the average radius, while also increasing the charge in the site, two factors that may counteract to hinder distances extension. This cationic distribution appears thus as a way to minimize the constraints generated by the rather rigid Sm3+ polyhedra. Consequently, the cations are subtly distributed over the three intricate frameworks (Sm, Moct and Mpyr), showing different structural evolutions vs. x that remain rather limited. This is in agreement with the different domains of stability of the structure observed depending on the lanthanide discussed in the first paper of Buisson.20 The analysis of the SXRPD data recorded at 10 K leads to similar observations (versus composition) (Fig. 5(d)) because the structural evolution vs. temperature is rather small for the five samples.The impact of the substitution on the magnetic properties is thus mainly due to the distribution of Ti4+ (d0), Cr3+ (d3: t32ge0g) and Fe3+ (d5: t32ge2g) over the M-cationic framework rather than the induced structural effect (Fig. 5) even if the structure monitors the distribution. Whatever x, the pyramid is mainly occupied by titanium, i.e. the nonmagnetic cation (Ti > 50%), and the octahedra by the magnetic cations (Ti < 50%). Going from SmCrTiO5 to SmFeTiO5, the amount of Ti increases in the octahedra inducing magnetic dilution in the chains. At the same time, the Ti content decreases in the pyramids, involving magnetic cations that favor magnetic interactions between the chains and thus altering the 1D character of magnetism reported for SmCrTiO5 in which magnetic chains appear rather isolated. The nearly equal distribution of Ti over pyramids and octahedra in SmFeTiO5 explains its low TN, compared to the strong antiferromagnetic coupling usually observed in trivalent iron oxides.43 For the mixed Fe/Cr samples, the disorder is increased with two magnetic cations in addition to Ti at each M-site. The lack of long range magnetic ordering in SmCr0.5Fe0.5TiO5 results from the weakening of the magnetic interactions in the chains, with roughly 1/3 Fe, 1/3 Cr and 1/3Ti, and their magnetic disconnection, via the square pyramids occupied by 2/3 Ti (Fig. 5). In SmCr0.75Fe0.25TiO5, the substitution of 1/3 Cr by (Ti and Fe) in the chains most probably hinders the spin gap formation and the occupation of the connecting square pyramids by Ti (3/4) limits the 3D magnetic coupling, which also results in spin-glass behaviour. On the other side, the close magnetic behaviour of SmCr0.25Fe0.75TiO5 and SmFeTiO5 is in agreement with their close magnetic cation distributions at both crystallographic sites (Fig. 5) supporting long range antiferromagnetism.
It is rather difficult to apply the usual Goodenough rules in this structure due to the values of the angles involved in the super exchange along the chains, i.e. Cr–O2–Cr and Cr–O3–Cr with angles ≈96 and 93° for the Cr-compound. In the Fe-sample, since Fe occupy both sites, the magnetic pathway implies the super exchange in the chains too (with Fe–O2–Fe and Fe–O2–Fe angles of ≈96 and 94°) but also the coupling between octahedra and pyramids, i.e. Fe–O1–Fe (≈ 128°), Fe–O3–Fe (≈132°) and Fe–O4–Fe (≈100°). The interpretation of these intermediate cation–oxygen–cation angles is rather tricky, even without speaking of the dilution by Ti. This complexity can be compared with the case of spinels: the dominant interactions correspond to ≈125° angles and there is a critical angle between 135 and 150° for which the d3–d5 coupling changes from ferro (larger angle) to antiferromagnetic (lower angle).44 The difficulty of interpreting the 90° interaction between cations in octahedral sites is also illustrated in the Kanamori paper.45
Modelling of the magnetic interactions is complex since the structure implies J1 and J2 along the chains (short and long Moct–Moct distances, respectively), J3 and J4 (between Moct and Mpyrvia O), J5 in the Mpyr2O8 units and J6 for the Moct–O–Mpyr–O–Moct super-superexchange coupling. In addition to these magnetic pathways, we need to consider several pairs of cations, such as Cr3+–Cr3+, Cr3+–Fe3+, Cr3+–Ti4+, Fe3+–Fe3+, Fe3+–Ti4+. And finally, a cluster model must also probably be taken into account in link with an inhomogeneous distribution of the spins, as proposed to explain the magnetic interactions at high temperature in AFeTiO533 as previously done for the spinel Fe2−2xMg1+xSnxO4 solid solution.46 In any case, this series allows the transition from one antiferromagnetic compound to another one, via intermediates that belong to the category of spin glasses, deemed to be useless but that are of great fundamental interest.47 On the other hand, no ferromagnetic order is observed whatever x, in contrast to the ferromagnetic and antiferromagnetic ground states reported in Cr-based pyroxenes despite structural similarities.48,49
The impact of the disorder induced by substitution is confirmed by the spin glass behaviour evidenced for SmFe0.5Mn0.5TiO5 (Fig. 14(a)) for which the Mössbauer analysis lead to 47.3% of iron in pyramids [IS = 0.304 mm s−1 and SQ = 0.92 mm s−1] and 52.7% in octahedra [IS = 0.335 mm s−1 and QS = 0.51 mm s−1], i.e., close to the Fe distribution in SmCr0.5Fe0.5TiO5. In the same way, the f-dependence of the χ′(T) (Fig. 14(b)) of SmCr0.5Mn0.5TiO5 would therefore mean that chromium, manganese and titanium are all three spread in the pyramids and octahedra.
 | ||
| Fig. 14 AC-magnetization curves χ′(T) of SmFe0.5Mn0.5TiO5 (a) and SmCr0.5Mn0.5TiO5 (b) recorded with a 10 Oe AC-field and frequencies of 102, 103 and 104 Hz. | ||
4. Conclusion
Only a multi-technique approach could provide a detailed description of the SmCr1−xFexTiO5 system and thus enable us to understand the effects of the substitution. The combination of Mössbauer with SXRPD and NPD techniques proved invaluable for the structural study, highlighting the absence of structural transition as a function of x and identifying the occupancy of each crystallographic site in the five studied compounds. The temperature dependent SXRPD and NPD measurements also show no structural transition but susceptibility characterizations reveal different magnetic behaviours vs. x.Despite a linear dependence of the lattice volume vs. x characteristic of a Végard law, an anisotropic evolution of the cell parameters is observed, with non-monotonous behaviours. As x increases in SmCr1−xFexTiO5, Fe3+ does not simply replace Cr3+ but is distributed in octahedra and square pyramids, with a scattering of Ti4+ over both sites. The disorder induced by the cation and charge redistribution on the two sites affects mainly the magnetic properties. At x = 0.5, the disorder is maximal with an occupation of the octahedra close to random (≈1/3 of each cation) that hinders the setting of a long range magnetic order in the chains, inducing a spin glass behaviour, as observed in SmFe0.5Mn0.5TiO5 and SmCr0.5Mn0.5TiO5. Depending on the occupation of each sub network (chains of octahedra sharing edges and pairs of square-based pyramids with a common edge), the magnetic ground state may vary from uni- to tri-dimensional. Since the cation distribution over the crystallographic sites appears as a key parameter, modelling using Monte Carlo and DFT approaches would be necessary to go further in the prediction of the site occupations.
Coming back to the applications in catalysis for instance, the present study shows that the interpretations of the chemical reactions with such catalysts containing several cations sitting in several crystallographic sites cannot avoid taking into account the local cationic distribution. In that respect, synthesizing compounds of the same nominal formula by different routes would be important in order to test if the site occupations can be changed. Similarly, if we could control the cation distribution, it would be highly interesting to substitute selectively octahedra or pyramids to affect only the spin chains or their coupling, respectively.
Such a class of materials stable up to high temperature and exhibiting versatile compositions and properties might provide a good playground for modelling. With the current development of artificial intelligence in material sciences, its investigation would be relevant since the cation distribution is difficult to predict, even though it plays a major role in the properties.
Data availability
The data supporting this article have been included as part of the ESI.† ILL data {https://doi.ill.fr/10.5291/ILL-DATA.EASY-1300 and https://doi.ill.fr/10.5291/ILL-DATA.5-31-2815}. ALBA synchrotron data {ID 2021095438}.Conflicts of interest
There are no conflicts to declare.Acknowledgements
JPB, BK, SJ, VN, AM and CM acknowledge the financial support of the French Agence Nationale de la Recherche LabEx EMC3 through the Project MaPhoOBi (grant no. ANR-10-LABX-0009), and the Normandy Region (Réseau d’Intérêt Normand – Label d’excellence), as well as the helpful assistance of Sophie Dufourd and Fabien Veillon with synthesis and physical measurements. A. I. K. thanks the Russian Science Foundation (RSF) for grant no. 24–12–00217. All authors are thankful to the ILL {https://doi.ill.fr/10.5291/ILL-DATA.EASY-1300 and https://doi.ill.fr/10.5291/ILL-DATA.5-31-2815} and ALBA synchrotron {ID 2021095438} for the allocated beam time, and particularly for the thorough assistance of the staff during remote experiments.References
- Y. Tokura and Y. Tomioka, Colossal magnetoresistive manganites, J. Magn. Magn. Mater., 1999, 200, 1–23, DOI:10.1016/S0304-8853(99)00352-2.
- A. Maignan, F. Damay, A. Barnabé, C. Martin, M. Hervieu and B. Raveau, The effect of Mn-site doping on the magneto transport properties of CMR manganites, Philos. Trans. R. Soc., A, 1998, 356, 1635–1659, DOI:10.1098/rsta.1998.0239.
- A. Simonov and A. L. Goodwin, Designing disorder into crystalline materials, Nat. Rev. Chem., 2020, 4, 657–673, DOI:10.1038/s41570-020-00228-3.
- K. Bhaskar, V. Nallathambi and R. Kumar, Critical role of cationic local stresses on the stabilization of entropy-stabilized transition metal oxides, J. Am. Ceram. Soc., 2020, 103, 3416–3424, DOI:10.1111/jace.17029.
- N. Kotsonis, S. S. I. Almishal, F. M. dos Santos Vieira, V. H. Crespi, I. Dabo, C. M. Rost and J.-P. Maria, High-entropy oxides: Harnessing crystalline disorder for emergent functionality, J. Am. Ceram. Soc., 2023, 106, 5587–5611, DOI:10.1111/jace.19252.
- T. Subramani, A. Voskanyan, K. Jayanthi, M. Abramchuk and A. Navrotsky, A Comparison of order-disorder in several families of cubic oxides, Front. Chem., 2021, 9, 719169, DOI:10.3389/fchem.2021.719169.
- Y. Zhu, Z. Tang, L. Yuan, B. Li, Z. Shao and W. Guo, Beyond conventional structures: emerging complex metal oxides for efficient oxygen and hydrogen electrocatalysis, Chem. Soc. Rev., 2025, 54, 1027–1092, 10.1039/d3cs01020a.
- A. Urban, A. Abdellahi, S. Dacek, N. Artrith and G. Ceder, Electronic-structure origin of cation disorder in transition-metal oxides, Phys. Rev. Lett., 2017, 119, 176402, DOI:10.1103/PhysRevLett.119.176402.
- K. Binder and A. P. Young, Spin glasses: experimental facts, theoretical concepts, and open questions, Rev. Mod. Phys., 1986, 58, 801, DOI:10.1103/RevModPhys.58.801.
- M. Songvilay, S. Petit, V. Hardy, J. P. Castellan, G. André, C. Martin and F. Damay, Random dilution effects in the frustrated spin chain β-CaCr2−xScxO4, Phys. Rev. B: Condens. Matter Mater. Phys., 2015, 91, 054408, DOI:10.1103/PhysRevB.91.054408.
- A. Kirsch, E. D. Bøjesen, N. Lefeld, R. Larsen, J. K. Mathiesen, S. L. Skjærvø, R. K. Pittkowski, D. Sheptyakov and K. M. Ø. Jensen, High-Entropy Oxides in the Mullite-Type Structure, Chem. Mater., 2023, 35, 8664–8674, DOI:10.1021/acs.chemmater.3c01830?urlappend=%3Fref%3DPDF&jav=VoR&rel=cite-as.
- L. Wang, H. Li, J. Liu, X. Langa and W. Wang, Labile oxygen participant adsorbate evolving mechanism to enhance oxygen reduction in SmMn2O5 with double-coordinated crystal fields, J. Mater. Chem. A, 2021, 9, 380–389, 10.1039/d0ta09537k.
- S. Thampy, N. Ashburn, K. Cho and J. W. P. Hsu, Earth-Abundant Transition Metal-Based Mullite-Type Oxide Catalysts for Heterogeneous Oxidation Reactions, Adv. Energy Sustainability Res, 2021, 2, 2000075, DOI:10.1002/aesr.202000075.
- Y. Zheng, S. Thampy, N. Ashburn, S. Dillon, L. Wang, Y. Jangjou, K. Tan, F. Kong, Y. Nie, M. J. Kim, W. S. Epling, Y. J. Chabal, J. W. P. Hsu and K. Cho, Stable and active oxidation catalysis by cooperative lattice oxygen redox on SmMn2O5 mullite surface, J. Am. Chem. Soc., 2019, 141, 10722–10728, DOI:10.1021/jacs.9b03334.
- A. Inomata and K. Kohn, Pyroelectric effect and possible ferroelectric transition of helimagnetic GdMn2O5, TbMn2O5 and YMn2O5, J. Phys.: Condens. Matter, 1996, 8, 2673–2678, DOI:10.1088/0953-8984/8/15/016.
- Y. Noda, H. Kimura, M. Fukunaga, S. Kobayashi, I. Kagomiya and K. Kohn, Magnetic and ferroelectric properties of multiferroic RMn2O5, J. Phys.: Condens. Matter, 2008, 20, 434206, DOI:10.1088/0953-8984/20/43/434206.
- J. Hwang, E. S. Choi, H. D. Zhou, J. Lu and P. Schlottmann, Magnetoelectric effect in NdCrTiO5, Phys. Rev. B: Condens. Matter Mater. Phys., 2012, 85, 024415, DOI:10.1103/PhysRevB.85.024415.
- J. Saha, G. Sharma and S. Patnaik, Evidence for multiferroic characteristics in NdCrTiO5, J. Magn. Magn. Mater., 2014, 360, 34–37, DOI:10.1016/j.jmmm.2014.02.008.
- K. Gautam, A. Ahad, K. Dey, S. S. Majid, S. Francoual, V. G. Sathe, I. da Silva and D. K. Shukla, Symmetry breaking and spin lattice coupling in NdCrTiO5, Phys. Rev. B, 2019, 100, 104106, DOI:10.1103/PhysRevB.100.104106.
- G. Buisson, Etude par rayons X et neutrons de la série isomorphe ATiTO5 (A = Cr, Mn, Fe, T = Terres Rares), J. Phys. Chem. Solids, 1970, 3, 1171–1183, DOI:10.1016/0022-3697(70)90326-4.
- S. Kori, T. Okamura, R. Okazaki and I. Terasaki, Anomalous magnetic order in the magnetoelectric oxide NdCrTiO5 revealed by impurity effects, Phys. Rev. B: Condens. Matter Mater. Phys., 2015, 91, 144403, DOI:10.1103/PhysRevB.91.144403.
- X. L. Qian, Y. F. Fang, J. Kang, S. X. Cao and J. C. Zhang, Influence of Sm3+ substitution on the mutiferroic effect in NdCrTiO5, Phys. B, 2016, 495, 1–3, DOI:10.1016/j.physb.2016.04.041.
- A. E. Susloparova, J. P. Bolletta, B. Kobzi, A. A. Paecklar, S. Jouen, F. Fauth, V. Nachbaur, V. Nassif, E. Suard, D. Sedmidubsky, A. I. Kurbakov, A. Maignan and C. Martin, Structural and magnetic properties of SmCrTiO5, Phys. Rev. B, 2024, 110, 224429, DOI:10.1103/PhysRevB.00.004400.
- M. Hase, M. Soda, T. Masuda, D. Kawana, T. Yokoo, S. Itoh, A. Matsuo, K. Kindo and M. Kohno, Experimental confirmation of spin gap in antiferromagnetic alternating spin-3/2 chain substances RCrGeO5 (R = Y or 154Sm) by inelastic neutron scattering experiments, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 90, 024416, DOI:10.1103/PhysRevB.90.024416.
- F. Fauth, R. Boer, F. Gil-Ortiz, C. Popescu, O. Vallcorba, I. Peral, D. Fulla, J. Benach and J. Juanhuix, The crystallography stations at the Alba synchrotron, Eur. Phys. J. Plus, 2015, 130, 160, DOI:10.1140/epjp/i2015-15160-y.
- J. E. M. van der Linden, M. M. Sala, C. Henriquet, M. Rossi, K. Ohgushi, F. Fauth, L. Simonelli, C. Marini, E. Fraga, C. Murray, J. Potter and M. Krisch, A compact and versatile dynamic flow cryostat for photon science, Rev. Sci. Instrum., 2016, 87, 115103, DOI:10.1063/1.4966270.
- J. Rodríguez-Carvajal, Recent advances in magnetic structure determination by neutron powder diffraction, Phys. B, 1993, 192, 55, DOI:10.1016/0921-4526(93)90108-I.
- P. J. Schurer and A. H. Morrish, Mössbauer study of ATiNdO5 (A = Cr, Mn, Fe), Physica, 1977, 86–88B, 925–926, DOI:10.1016/0022-3697(85)90001-0.
- R. D. Shannon, Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides, Acta Crystallogr., Sect. A, 1976, 32, 751, DOI:10.1107/S0567739476001551.
- D. K. Shukla, S. Mollah, R. Kumar, P. Thakur, K. H. Chae, W. K. Choi and A. Banerjee, Effect of Ti substitution on multiferroic properties of BiMn2O5, J. Appl. Phys., 2008, 104, 033707, DOI:10.1063/1.2964072.
- H. Schneider, J. Schreuer and B. Hildmann, Structure and properties of mullite - A review, J. Eur. Ceram. Soc., 2008, 28, 329–344, DOI:10.1016/j.jeurceramsoc.2007.03.017.
- F. Menil, Systematic trends of the 57Fe Mössbauer isomer shifts in (FeOn) and (FeFn) polyhedra. Evidence of a new correlation between the isomer shift and the inductive effect of the competing bond T-X (→ Fe) (where X is O or F and T any element with a formal positive charge), J. Phys. Chem. Solids, 1985, 46, 763–789, DOI:10.1016/0022-3697(85)90001-0.
- P. J. Schurer and A. H. Morrish, 57Fe Mössbauer study of ATiRO5 compounds (A = Cr, Mn, Fe, and R = Pr, Nd, Sm, Eu, Gd), Phys. Rev. B, 1977, 16, 951–959, DOI:10.1103/PhysRevB.16.951.
- M. J. Martínez-Lope, M. Retuerto, J. A. Alonso, M. García-Hernández, K. Krezhov, I. Spirov, T. Ruskov and M. T. Fernández-Díaz, Crystal structure and magnetism of YbFeMnO5: A neutron diffraction and Mössbauer spectroscopy study, Solid State Commun., 2009, 149, 540–545, DOI:10.1016/j.ssc.2009.01.009.
- V. F. Sears, Neutron Scattering Lengths and Cross Sections, Neutron News, 1992, 3, 26, DOI:10.1080/10448639208218770.
- E. Sokolova and F. C. Hawthorne, The crystal chemistry of silicate minerals with chains of (TiO6) octahedra, Can. Mineral., 2004, 42, 807–824, DOI:10.2113/gscanmin.42.3.807.
- N. V. Chukanov, Ol. N. Kazheva, R. X. Fischer and S. M. Aksenov, Refinement of the crystal structure of fresnoite, Ba2TiSi2O8, from Löhley (Eifel district, Germany); Gladstone–Dale compatibility, electronic polarizability and vibrational spectroscopy of minerals and inorganic compounds with pentacoordinated TiIV and a titanyl bond, Acta Cryst., 2023, B79, 184–194, DOI:10.1107/S2052520622012045.
- R. V. Shpanchenko, A. A. Tsirlin, E. S. Kondakova, E. V. Antipov, C. Bougerol, J. Hadermann, G. van Tendeloo, H. Sakurai and E. Takayama-Muromachi, New germanates RCrGeO5 (R = Nd-Er, Y): Synthesis, structure, and properties, J. Solid State Chem., 2008, 181, 2433–2441, DOI:10.1016/j.jssc.2008.05.043.
- F. Pomiro, J. Lohr, E. V. Pannunzio Miner, V. Nassif, R. D. Sánchez and R. E. Carbonio, Crystal Structure, Magnetic and Electrical Properties of Compounds in the RCrMnO5 Family (R = Sm, Eu, Gd,Tb, Ho and Er) Synthesized Under High Oxygen Pressure, Eur. J. Inorg. Chem., 2015, 4737–4749, DOI:10.1002/ejic.201500662.
- K. Ghosh, M. M. Murshed, T. Frederichs, N. K. C. Muniraju and T. M. Gesing, Structural, vibrational, thermal, and magnetic properties of mullite-type NdMnTiO5 ceramic, J. Am. Ceram. Soc., 2022, 105, 2702–2712, DOI:10.1111/jace.18261.
- J. Portier, G. Campet, J. Etourneau and B. Tanguy, A simple model for the estimation of electronegativities of cations in different electronic states and coordinations, J. Alloys Compd., 1994, 209, 285–289, DOI:10.1016/0925-8388(94)91115-0.
- J. A. Mydosh, Spin glasses: an experimental introduction, Taylor & Francis, London Wahington DC, 1993 DOI:10.1201/9781482295191.
- D. Treves, M. Eibschutz and P. Coppens, Dependence of superexchange interactions on Fe3+–O2−–Fe3+ linkage angle, Phys. Lett., 1965, 18, 216 CrossRef CAS.
- J. B. Goodenough, Magnetism and the chemical bond, Interscience publishers, John Wiley & Sons, New York-London, 1963 Search PubMed.
- J. Kanamori, Superexchange interaction and symmetry properties of electron orbitals, J. Phys. Chem. Solids, 1959, 10, 87–98 CrossRef CAS.
- F. Basile and P. Poix, Effet Mössbauer et mesures magnétiques de la solution solide Fe2–2xMg1+xSnxO4, Phys. Status Solidi A, 1976, 35, 153, DOI:10.1002/pssa.2210350117.
- M. Mézard, Spin glasses and optimization in complex systems, Europhys. News, 2022, 53, 10, DOI:10.1051/epn/2022103.
- A. N. Vasiliev, O. L. Ignatchik, A. N. Sokolov, Z. Hiroi, M. Isobe and Y. Ueda, Long-range magnetic order in quasi-one-dimensional chromium-based (S = 3/2) pyroxenes (Li,Na)Cr(Si,Ge)2O6, Phys. Rev. B: Condens. Matter Mater. Phys., 2005, 72, 012412, DOI:10.1103/PhysRevB.72.012412.
- J. O’Connell, X. Xu, L. Jin and R. J. Cava, The ferromagnetic to antiferromagnetic crossover in chromium-based pyroxenes tuned via the Ge to Si ratio, J. Solid State Chem., 2024, 339, 124919, DOI:10.1016/j.jssc.2024.124919.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5tc01022e |
| This journal is © The Royal Society of Chemistry 2025 |