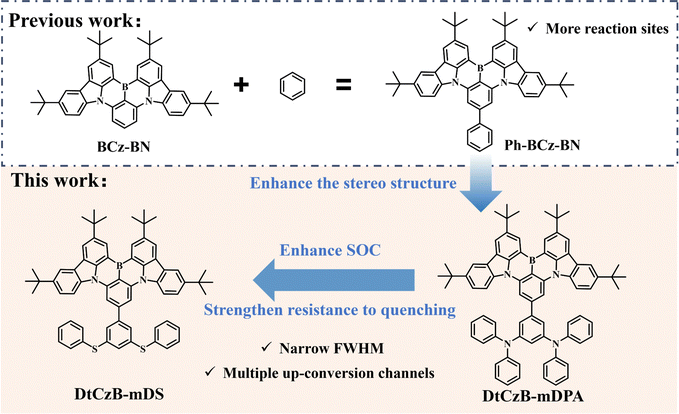
Enhancing the stereo structure via bulky peripheral groups to improve resistance to concentration quenching in OLEDs†
Haotian
Yue
a,
Yongxing
Tian
a,
Shan
Huang
a,
Hanrui
Su
a,
Hongbo
Shao
a,
Qiang
Zhang
a,
Runda
Guo
 *a and
Lei
Wang
*a and
Lei
Wang
 ab
ab
aWuhan National Laboratory for Optoelectronics, School of Optical and Electronic Information, Huazhong University of Science and Technology, Wuhan, 430074, People's Republic of China. E-mail: runda_guo@hust.edu.cn
bResearch Institute of Huazhong University of Science and Technology in Shenzhen, Shenzhen, 518038, People's Republic of China
First published on 3rd January 2025
Abstract
Multi-resonant thermally activated delayed fluorescence (MR-TADF) materials have attracted widespread attention from researchers in the field of organic light-emitting diodes (OLEDs). However, there remains significant room for improvement regarding the concentration quenching issue in OLEDs. Herein, we report two MR-TADF emitters, DtCzB-mDPA and DtCzB-mDS, based on the boron–nitrogen MR core, which were connected to bulky groups on the periphery of the framework. This strategy successfully enhanced the stereo structure in the molecule. For DtCzB-mDS, the introduction of the methyl(phenyl)sulfane group resulted in a larger dihedral angle and thus stronger concentration quenching resistance, as well as stronger spin–orbit coupling (SOC) compared to DtCzB-mDPA. Finally, OLEDs using DtCzB-mDS as a dopant achieved a 25.4% maximum external quantum efficiency (EQEmax) and a full width at half maximum (FWHM) of 27 nm, while the EQEmax of the DtCzB-mDPA-based device was 23.4%. As the doping concentration increased, the EQEmax of the DtCzB-mDS-based device remained around 24%, while the DtCzB-mDPA-based device saw a significant decrease in EQEmax. This work broadened the MR-TADF material design strategy and provided new ideas for the development of high-performance OLEDs.
Introduction
In the development of organic light-emitting materials, multi-resonant thermally activated delayed fluorescence (MR-TADF) materials based on polycyclic aromatic hydrocarbons (PAHs) with embedded heteroatoms exhibiting outstanding optoelectronic properties are relatively prevalent.1–18 In particular, PAHs with alternating boron and nitrogen atoms exhibited narrower full width at half maxima (FWHM) than traditional donor–acceptor type thermally activated delayed fluorescence (TADF) materials, resulting from the electron-withdrawing boron and electron-donating nitrogen atoms achieving frontier molecular orbital (FMO) separation at the atomic scale, which reduced vibrational relaxation.3,19–24 In recent years, the classic MR-TADF material boron–nitrogen–carbazole resonance core BCz-BN was widely used to construct blue materials.25,26 In terms of molecular structure, it had a peripheral tert-butyl coating that reduced π–π stacking,27 but its strong planar rigidity still led to significant concentration quenching. Therefore, to solve this problem, connecting peripheral bulky groups to BCz-BN and increasing the dihedral angle with the MR framework was an effective way to enhance molecular distortion and construct the stereo structure.28 However, directly introducing groups at the para position on the MR framework enhanced the charge transfer (CT) state, leading to spectral broadening. With fewer reaction sites at the para position, this limited molecular design strategies and hindered the development of new advanced materials, hence, addressing the issues of wide FWHM in BCz-BN and the lack of reaction sites was crucial. One approach was to introduce peripheral benzene at the para position of the B-substituted phenyl in BCz-BN, achieving a narrow FWHM and providing more reaction sites. This structure, known as Ph-BCz-BN,29 was commonly used for constructing MR-TADF materials with narrow FWHM.26,30,31 In addition, from the perspective of exciton dynamics, because the extent of spatial separation between the highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) of MR-TADF materials was always not as high as that of TADF materials,24,32,33 the rate of reverse intersystem crossing (kRISC) of the MR-TADF molecule was lower than that of TADF. This was another important issue that needed to be taken into account and addressed.In this work, we designed and synthesized two boron–nitrogen MR-TADF materials based on the Ph-BCz-BN core, namely DtCzB-mDPA and DtCzB-mDS. Electron-donating groups N-methyl-N-phenylaniline (DPA) and methyl(phenyl)sulfane (DS) were introduced into the periphery of the Ph-BCz-BN framework. The bulky peripheral groups were introduced to enhance the resistance to concentration quenching. The molecular design strategies are shown in Scheme 1. Theoretical calculations showed that DtCzB-mDS exhibited an even larger dihedral angle, leading to stronger resistance to concentration quenching. Moreover, due to the heavy atom effect induced by sulfur, the spin–orbit coupling (SOC) and kRISC values of DtCzB-mDS also improved. The prepared OLEDs using DtCzB-mDPA and DtCzB-mDS as dopants exhibited maximum external quantum efficiency (EQEmax) values of 23.4% and 25.4%, respectively, and their FWHM was 27 nm. Additionally, the DtCzB-mDS-based devices demonstrated better resistance to concentration quenching. DtCzB-mDS-based devices maintained an external quantum efficiency (EQE) of approximately 24% despite an increase in doping concentration, whereas DtCzB-mDPA-based devices exhibited significant EQE decreases with doping concentration increases. These results indicated that the strategy of linking peripheral groups to enhance the molecular stereo structure effectively improved resistance to quenching. It also highlighted that incorporating sulfur atoms into the molecule was an effective approach to enhance the exciton utilization.
Results and discussion
Molecular synthesis and thermal properties
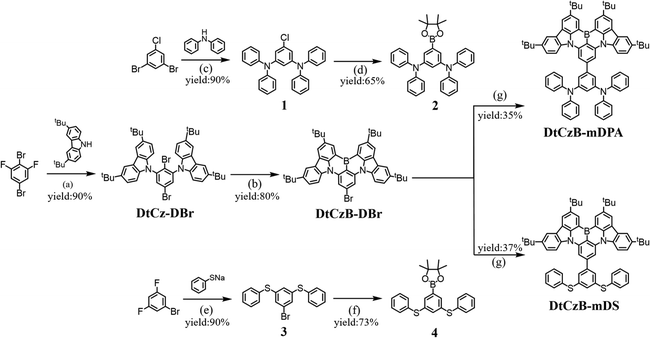
The synthesis routes and molecular structures of the two target materials are shown in Scheme 2, with the boronation yields of both target products exceeding 35%. The nuclear magnetic resonance (NMR) and mass spectrometry (MS) analysis of the two target materials and the NMR analysis of their intermediates are provided in Fig. S1–S12 (ESI†). The synthesis methods of the two target materials are provided with subsequent experimental details. The synthesis methods of their intermediates are provided in the ESI.† To investigate the thermal stability and morphological stability of DtCzB-mDPA and DtCzB-mDS, thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) analyses were performed, with the corresponding curves shown in Fig. S13 (ESI†) and the relevant data listed in Table 1. Both DtCzB-mDPA and DtCzB-mDS exhibited high thermal decomposition temperatures (Td), which were 473 °C and 487 °C, respectively. According to DSC results, there were no significant glass transition temperatures (Tg) observed in both materials. This was likely due to the unique rigid conjugated structure of MR-TADF emitters, which restricted the free rotation and movement of the molecules. To sum up, both DtCzB-mDPA and DtCzB-mDS demonstrated good thermal stability, which was beneficial for maintaining stability during vacuum deposition processes.| Compound | T g/Tda (°C) | λ abs (nm) | λ em (nm) | HOMOc/LUMOd (eV) | E S/ETe (eV) | PLQYf (%) | τ PF (ns) | τ DF (μs) | FWHMh (nm) |
|---|---|---|---|---|---|---|---|---|---|
| a T d was measured by TGA (corresponding to 5% weight loss) and Tg was measured by DSC. b The peak wavelength of the lowest-energy absorption band and the peak wavelength of the PL spectrum in toluene. c HOMO was estimated from the CV. d LUMO = HOMO + Eg. e The singlet and triplet energy values were calculated from the peak wavelength of fluorescence (Fl, 77 K) and the phosphorescence (Phos., 77 K) spectra in toluene. f Absolute photoluminescence quantum yields in the thin film. g Lifetimes were calculated from the transient fluorescence curve and delayed fluorescence curve in the thin film. h Full width at half maxima in toluene. | |||||||||
| DtCzB-mDPA | NA/473 | 473 | 492 | −5.13/−2.61 | 2.48/2.36 | 82.9 | 9.13 | 86.27 | 22 |
| DtCzB-mDS | NA/487 | 472 | 494 | −5.21/−2.70 | 2.46/2.36 | 92.5 | 9.25 | 56.73 | 21 |
Theoretical calculations
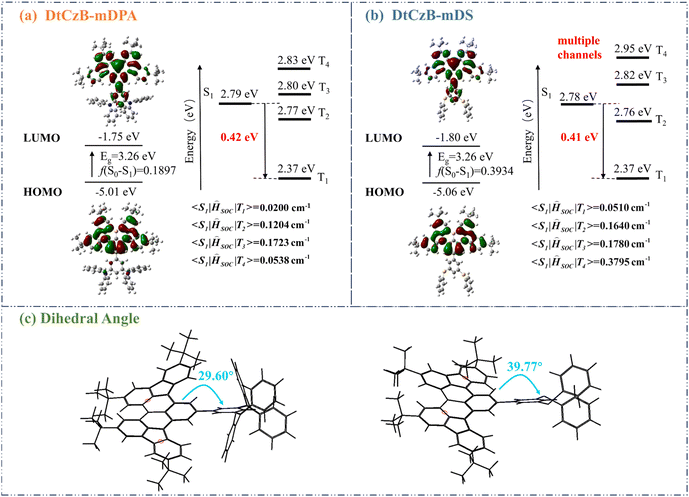
Theoretical calculations using the B3LYP/6-31G(d) method were performed to examine the influence of peripheral groups on FMO distribution and excited state energy levels of DtCzB-mDPA and DtCzB-mDS. As shown in Fig. 1, the HOMO and LUMO of both molecules were distributed in the MR framework, which all conformed to the multiple resonance characteristics of the boron–nitrogen MR framework. The energy gap (Eg) of both materials was 3.26 eV, predicting nearly identical emission spectra. DtCzB-mDS had a higher oscillator strength (0.3934) than DtCzB-mDPA (0.1897), predicting better utilization of triplet exciton and device performance. To investigate the excited state transitions of DtCzB-mDPA and DtCzB-mDS, energy levels and spin–orbit coupling matrix elements (SOCME) were calculated. The SOCME values were calculated using the ORCA program and also under the TD-DFT method, and it was carried out with the B3LYP hybrid functional at the basis set level of def2-TZVP.34 The two materials exhibited energy gaps of 0.42 eV and 0.41 eV between T1 and S1, respectively, and both showed a gap of 0.02 eV between S1 and T2. The high SOCME values were observed between higher triplet states and S1, suggesting multiple RISC channels.35DtCzB-mDS exhibited generally higher SOCME due to the involvement of sulfur, which promoted exciton spin–flip,36 allowing us to predict that DtCzB-mDS would have a better device performance. To evaluate the quenching resistance characteristics of the two molecules, we calculated the dihedral angle between the MR framework and the peripheral connecting groups. In DtCzB-mDS, this angle was 39.77°, while in DtCzB-mDPA, it was 29.60°. This difference predicted that DtCzB-mDS exhibited superior resistance to concentration quenching in the device, as reflected in the subsequent device data.The natural transition orbital (NTO) distribution for both compounds was computed simultaneously, as shown in Fig. S14 (ESI†). The S1 and T1 states of DtCzB-mDPA and DtCzB-mDS exhibited identical “hole” and “particle” attributes, both being locally excited (LE) states. The T2 and T4 states of both molecules were hybridized local and charge transfer (HLCT) states. The difference lay in T3, which was the CT state in DtCzB-mDPA, whereas for DtCzB-mDS, the T3 state was the HLCT state. Furthermore, the interfragment charge transfer (IFCT) method in Multiwfn was used to calculate the LE percentage of the energy levels with HLCT states. As further shown in Fig. S14 (ESI†), the LE percentages of T2 and T4 for DtCzB-mDPA were 75.2% and 91.1%, respectively, while the LE percentages of T2, T3, and T4 for DtCzB-mDS were 94.0%, 92.8%, and 77.2%, respectively. Next, it was well established that introducing sulfur atoms was essential in facilitating exciton spin-flipping. As a result, DtCzB-mDS, which contained sulfur atoms, generally exhibited higher SOCME than DtCzB-mDPA. In particular, there was pronounced hole occupation on the sulfur atoms in the T4 state of DtCzB-mDS, which contributed to its generally stronger SOC.35
Photophysical properties
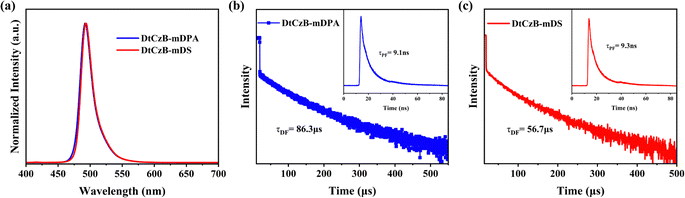
To assess the photophysical performance of the molecules, ultraviolet-visible light (UV-Vis) absorption spectra and fluorescence spectra of DtCzB-mDPA and DtCzB-mDS were measured in dilute toluene (1.0 × 10−5 M). As shown in Fig. S15 (ESI†), due to their similar structures, the absorption spectra of DtCzB-mDPA and DtCzB-mDS were nearly identical. Absorption peaks at 310–330 nm corresponded to the π–π* transition of the DPA and DS groups, while the 380 nm peak was attributed to the n–π* transition in both compounds. The CT absorption peaks were at 473 nm and 472 nm, with Eg values of 2.52 eV and 2.51 eV, consistent with theoretical calculations, indicating similar emissive colors. In terms of emission in solution, as shown in Fig. 2(a), DtCzB-mDPA and DtCzB-mDS exhibited fluorescence emission at 492 nm and 494 nm in dilute toluene solution, with corresponding FWHM of 22 nm and 21 nm, respectively. Furthermore, the UV absorption and PL spectra of DtCzB-mDPA and DtCzB-mDS with host materials in solid film were shown in Fig. S16 (ESI†). Compared with their properties in toluene solution, the absorption and emission characteristics of the two materials in the solid film exhibited no significant changes. This indicated that the intermolecular interactions of these materials in the film were relatively weak, causing no notable alterations in molecular energy levels. Notably, due to the high rigidity of the MR framework, the intramolecular charge transfer in MR-TADF was mainly confined between atoms with different resonance effects, significantly reducing energy loss during the charge transfer process. Additionally, the vibrational relaxation of the molecules was suppressed, resulting in minimal Stokes shifts of 19 nm and 22 nm for DtCzB-mDPA and DtCzB-mDS, respectively.Furthermore, as shown in Fig. S17 (ESI†), when the solvent changed from cyclohexane to dichloromethane, both DtCzB-mDPA and DtCzB-mDS maintained narrow emission, but the emissive color red shifted and the spectra broadened during increasing solvent polarity. The red-shift and spectra broadening occurred because the dipole moment of TADF molecules caused rearrangement in polar solvents, reducing the Coulomb energy of the CT state, and the emission of the two molecules in various solvents is shown in Table S1 (ESI†). When solvent molecules were frozen and unable to rearrange, the intrinsic emission peak could be observed. As shown in Fig. S18 (ESI†), the low-temperature fluorescence and phosphorescence spectra of DtCzB-mDPA and DtCzB-mDS were measured at 77 K, revealing ES levels of 2.48 eV and 2.46 eV, and ET levels of 2.36 eV for both compounds. The corresponding ΔEST values were 0.12 eV for DtCzB-mDPA and 0.10 eV for DtCzB-mDS. Notably, DtCzB-mDS exhibits a smaller ΔEST than DtCzB-mDPA, consistent with the trend predicted with theoretical calculations, suggesting that DtCzB-mDS may possess a faster RISC process.
To further explore the TADF characteristics of DtCzB-mDPA and DtCzB-mDS, we performed time-resolved photoluminescence (TRPL) testing. As shown in Fig. 2(b) and (c), using single exponential fitting, the transient fluorescence lifetimes (τPF) of DtCzB-mDPA and DtCzB-mDS were found to be 9.1 ns and 9.3 ns, and the delayed fluorescence lifetimes (τDF) were 86.3 μs and 56.7 μs, respectively. The delayed fluorescence lifetime result indicated that DtCzB-mDS exhibited better TADF performance. Additionally, the PLQYs of DtCzB-mDPA and DtCzB-mDS in thin films were 82.9% and 92.5%, respectively, consistent with the oscillator strength results. According to the principle that triplet excitons could be quenched under oxygen conditions, the transient photoluminescence quantum yield (ΦPF) and delayed photoluminescence quantum yield (ΦDF) of DtCzB-mDPA and DtCzB-mDS were calculated to be 75.1% and 7.8%, and 74.8% and 17.7%, respectively. According to the known PLQY and fitted lifetime data, the emissive kinetics constants of the two compounds were calculated based on eqn (S1)–(S6) (ESI†), and specific data shown in Table S2 (ESI†). The kRISC values were 1.3 × 104 s−1 for DtCzB-mDPA and 2.2 × 104 s−1 for DtCzB-mDS, suggesting that DtCzB-mDS would likely perform better in the device.
Electrochemical properties
Cyclic voltammetry (CV) based on a traditional three-electrode system was employed to study the electrochemical properties of DtCzB-mDPA and DtCzB-mDS. As shown in Fig. S19 (ESI†), ferrocene solution was used as the reference electrode to calibrate the HOMO level. Based on the CV curves, the HOMO values of DtCzB-mDPA and DtCzB-mDS were determined to be −5.1 eV and −5.2 eV, respectively. Additionally, the LUMO levels were calculated using the Eg obtained from UV-Vis absorption spectra and the aforementioned HOMO levels, resulting in values of −2.6 eV for DtCzB-mDPA and −2.7 eV for DtCzB-mDS. The relevant data are summarized in Table 1.Electroluminescence properties
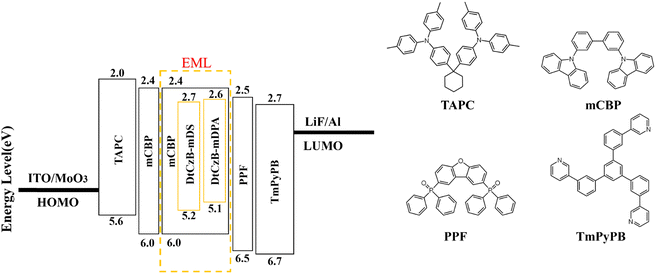
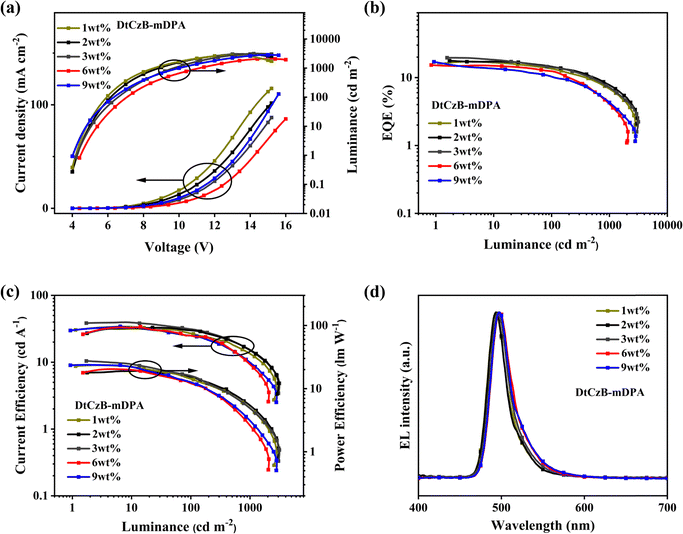
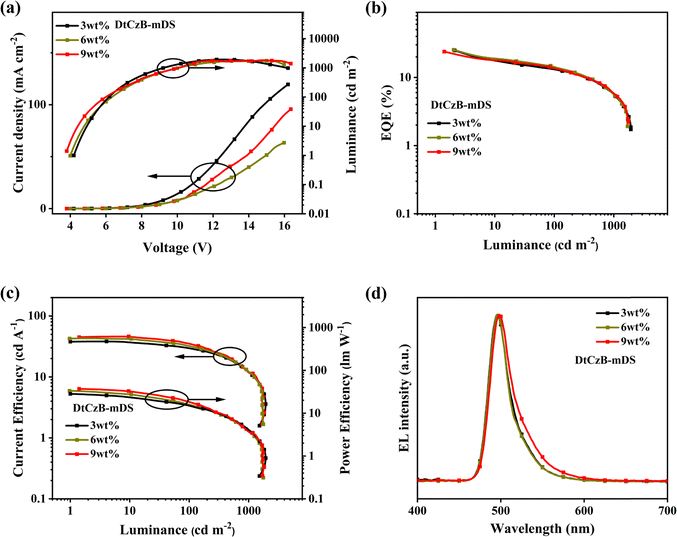
OLEDs were fabricated to evaluate the EL performance of the materials. As shown in Fig. 3, we fabricated devices with the following structure: ITO/MoO3 (5 nm)/TAPC (30 nm)/mCBP (10 nm)/mCBP: x wt% emitters (for DtCzB-mDPAx = 1, 2, 3, 6, 9 or for DtCzB-mDSx = 3, 6, 9) (20 nm)/PPF (10 nm)/TmPyPB (30 nm)/LiF (1 nm)/Al (100 nm). In this structure, LiF and MoO3 served as the electron injection layer and hole injection layer, respectively. 1,3,5-Tri(m-pyrid-3-yl-phenyl) benzene (TmPyPB) and 1,1-bis[(di-4-tolylamino) phenyl] cyclohexane (TAPC) acted as the electron transport layer and hole transport layer, respectively. 3,3-Di(9H-carbazol-9-yl) biphenyl (mCBP) functioned as the electron blocking layer, exciton blocking layer, and host, while 2,8-bis(diphenylphosphoryl)dibenzo[b,d]furan (PPF) served as the hole blocking layer and exciton blocking layer. Data for devices with varying doping concentrations are shown in Table S3 (ESI†), while the corresponding EL characteristics are illustrated in Fig. 4 and 5. Optimal device data for DtCzB-mDPA and DtCzB-mDS as dopants are presented in Table 2. The EL spectra of the optimal devices at different voltages are illustrated in Fig. S20 (ESI†), indicating that both DtCzB-mDPA and DtCzB-mDS exhibited excellent spectral stability. As the doping concentration increased, the turn-on voltage of both devices decreased. This was primarily due to the shortening of the triplet exciton diffusion path with increasing dopant concentration, allowing more excitons to participate in the effective thermally activated delayed fluorescence process. The optimal devices using DtCzB-mDPA and DtCzB-mDS as dopants exhibited EQEmax values of 23.4% and 25.4%, respectively, along with narrow FWHM of 27 nm for both materials. It could be seen that the device using DtCzB-mDS as a dopant exhibited a higher EQE, which could be attributed to its higher kRISC and SOCME. The device incorporating DtCzB-mDS as a dopant achieved a significantly higher EQEmax than DtCzB-mDPA, primarily due to its superior exciton utilization efficiency. This improvement stems from the higher kRISC and the stronger SOCME of DtCzB-mDS, which enabled more efficient triplet exciton harvesting. Compared with DtCzB-mDPA, DtCzB-mDS exhibited a markedly enhanced capacity to convert excitons into radiative emissions, highlighting its outstanding potential to maximize exciton utilization. Moreover, the EQEmax of both molecules was higher than that of BCz-BN.14 This was attributed to their high horizontal dipole orientation (Θ‖), which exceeded 80% (Fig. S21, ESI†) and enhanced light extraction efficiency (LEE), thereby improving device performance. The morphology of EML films at the optimal doping concentration of the dopant molecules was characterized using an atomic force microscope (AFM), as shown in Fig. S22 (ESI†). The root mean square roughness (RMS) of the EML films for DtCzB-mDPA and DtCzB-mDS was 1.79 nm and 1.18 nm, respectively, indicating a more uniform film formation for DtCzB-mDS. In terms of resistance to concentration quenching, DtCzB-mDS-based devices consistently maintained an EQE of around 24% as the doping concentration increased. In contrast, DtCzB-mDPA-based devices exhibited a significant decline in EQE, dropping to below 20% with the rise in doping concentration. This result was consistent with the theoretical calculation outcomes. In summary, the device performance results indicated that the molecular design strategies were highly effective.| Compound | V on (V) | L max (cd m−2) | CEmaxb (cd A−1) | PEmaxb (lm W−1) | EQEmaxb (%) | λ EL /FWHMc (nm) | CIEd (x, y) |
|---|---|---|---|---|---|---|---|
| a Voltage at 1 cd m−2 (V). b Maximum luminance (cd m−2), maximum current efficiency (cd A−1), maximum power efficiency (lm W−1), and maximum external quantum efficiency (%). c The peak of the EL spectrum recorded at 4 V; full width at half maxima of the EL spectrum recorded at 4 V. d Commission Internationale de l’Eclairage coordinates. | |||||||
| DtCzB-mDPA (3 wt%) | 4.1 | 3144 | 39.9 | 27.4 | 23.4 | 495/27 | (0.104, 0.526) |
| DtCzB-mDS (6 wt%) | 3.8 | 1764 | 42.9 | 33.4 | 25.4 | 496/27 | (0.117, 0.540) |
Conclusions
In summary, based on the idea of introducing para-positioned benzene to achieve a narrow FWHM and incorporating bulky peripheral groups to enhance the stereo structure, thereby improving resistance to quenching, two MR-TADF molecules, DtCzB-mDPA and DtCzB-mDS, were designed and synthesized. DtCzB-mDS exhibited larger SOCME values than DtCzB-mDPA. The kRISC of DtCzB-mDS was twice that of DtCzB-mDPA. OLEDs using DtCzB-mDPA and DtCzB-mDS as dopants achieved EQEmax values of 23.4% and 25.4%, respectively, accompanied by a narrow FWHM of 27 nm. Furthermore, DtCzB-mDS-based devices maintained a high EQE of around 24% when increasing doping concentrations attributed to the introduction of bulky peripheral groups successfully increased the dihedral angle between the MR framework and peripheral groups, thus increasing the resistance to quenching. This work provides guidance for the design of MR-TADF molecules with improved performance.Experimental details
Preparation of compounds
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) eluent to obtain 1.3 g of yellow solid, with a yield of 35%. 1H NMR (600 MHz, CD2Cl2) δ[ppm]: 9.11 (s, 2H), 8.51 (s, 2H), 8.33 (d, J = 35.6 Hz, 4H), 8.07 (d, J = 8.7 Hz, 2H), 7.61 (d, J = 8.7 Hz, 2H), 7.46–7.21 (m, 18H), 7.13 (t, J = 7.5 Hz, 4H), 6.98 (s, 1H), 1.66 (s, 18H), 1.52 (s, 18H). 13C NMR (151 MHz, CD2Cl2) δ [ppm]: 149.52, 147.60, 145.45, 145.30, 144.92, 144.52, 142.51, 141.59, 138.15, 129.78, 129.35, 126.99, 124.75, 124.72, 123.60, 123.18, 120.89, 117.90, 117.38, 116.40, 113.91, 106.30, 35.08, 34.72, 31.89, 31.75, 31.59. HRMS (APCI): theoretical value: C76H71BN4, 1051.2420; experimental value: 1052.2316, [M + H]+.
1) eluent to obtain 1.3 g of yellow solid, with a yield of 35%. 1H NMR (600 MHz, CD2Cl2) δ[ppm]: 9.11 (s, 2H), 8.51 (s, 2H), 8.33 (d, J = 35.6 Hz, 4H), 8.07 (d, J = 8.7 Hz, 2H), 7.61 (d, J = 8.7 Hz, 2H), 7.46–7.21 (m, 18H), 7.13 (t, J = 7.5 Hz, 4H), 6.98 (s, 1H), 1.66 (s, 18H), 1.52 (s, 18H). 13C NMR (151 MHz, CD2Cl2) δ [ppm]: 149.52, 147.60, 145.45, 145.30, 144.92, 144.52, 142.51, 141.59, 138.15, 129.78, 129.35, 126.99, 124.75, 124.72, 123.60, 123.18, 120.89, 117.90, 117.38, 116.40, 113.91, 106.30, 35.08, 34.72, 31.89, 31.75, 31.59. HRMS (APCI): theoretical value: C76H71BN4, 1051.2420; experimental value: 1052.2316, [M + H]+.
Synthesis of compound DtCzB-mDS
The synthesis route to compound DtCzB-mDS is the same as that to compound DtCzB-mDPA. The experimental procedure and post-treatment are identical, using dichloromethane (10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) as the eluent for column chromatography to obtain 1.2 g of yellow solid with a yield of 37%. 1H NMR (600 MHz, CD2Cl2)δ [ppm]: 9.02 (d, J = 2.1 Hz, 2H), 8.52–8.44 (m, 2H), 8.34–8.21 (m, 4H), 8.13 (d, J = 8.7 Hz, 2H), 7.72–7.50 (m, 8H), 7.43 (t, J = 7.5 Hz, 4H), 7.37 (t, J = 7.6 Hz, 2H), 7.32 (s, 1H), 1.65 (s, 18H), 1.56 (s, 18H).13C NMR (151 MHz, CD2Cl2)δ[ppm]: 145.49, 144.84, 144.53, 144.41, 143.24, 141.56, 139.02, 138.10, 134.07, 132.47, 129.67, 129.65, 129.56, 128.04, 127.03, 126.80, 124.67, 123.57, 121.44, 120.94, 117.42, 114.05, 106.65, 35.06, 34.73, 31.89, 31.72, 31.58, 30.06, 29.69. HRMS (APCI): theoretical value: C64H61BN2S2, 933.1360; experimental value: 934.1345, [M + H]+.
1) as the eluent for column chromatography to obtain 1.2 g of yellow solid with a yield of 37%. 1H NMR (600 MHz, CD2Cl2)δ [ppm]: 9.02 (d, J = 2.1 Hz, 2H), 8.52–8.44 (m, 2H), 8.34–8.21 (m, 4H), 8.13 (d, J = 8.7 Hz, 2H), 7.72–7.50 (m, 8H), 7.43 (t, J = 7.5 Hz, 4H), 7.37 (t, J = 7.6 Hz, 2H), 7.32 (s, 1H), 1.65 (s, 18H), 1.56 (s, 18H).13C NMR (151 MHz, CD2Cl2)δ[ppm]: 145.49, 144.84, 144.53, 144.41, 143.24, 141.56, 139.02, 138.10, 134.07, 132.47, 129.67, 129.65, 129.56, 128.04, 127.03, 126.80, 124.67, 123.57, 121.44, 120.94, 117.42, 114.05, 106.65, 35.06, 34.73, 31.89, 31.72, 31.58, 30.06, 29.69. HRMS (APCI): theoretical value: C64H61BN2S2, 933.1360; experimental value: 934.1345, [M + H]+.
Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
The authors declare no conflicts of interest.Acknowledgements
This work was supported by the National Natural Science Foundation of China (52473175), the Science and Technology Department of Hubei Province (2024BAB014, 2022BAA015), and the Science, Technology and Innovation Commission of Shenzhen Municipality (JCYJ20220530161208020). Thanks to SCTS/CGCL HPCC of Huazhong University of Science and Technology (HUST) for providing computing resources and technical support. The Analytical and Testing Center at HUST is acknowledged for the characterization of new compounds.References
- Y. Sun, H. Wang, S. Liu, X. Lu, Z. Feng, D. Zhong, D. Jia, X. Yang, B. Su, Y. Sun, B. Jiao and G. Zhou, Innovative Mater., 2023, 1, 100028 CrossRef.
- P. Liu, X.-Y. Chen, J. Cao, L. Ruppenthal, J. M. Gottfried, K. Müllen and X.-Y. Wang, J. Am. Chem. Soc., 2021, 143, 5314–5318 CrossRef.
- S. M. Suresh, D. Hall, D. Beljonne, Y. Olivier and E. Zysman-Colman, Adv. Funct. Mater., 2020, 30 Search PubMed.
- M. Stepien, E. Gonka, M. Zyla and N. Sprutta, Chem. Rev., 2017, 117, 3479–3716 CrossRef PubMed.
- J. Kang, S. O. Jeon, H. L. Lee, J. Lim, U. Jo and J. Y. Lee, Mater. Today, 2023, 69, 88–96 CrossRef.
- J. M. Ha, S. H. Hur, A. Pathak, J.-E. Jeong and H. Y. Woo, NPG Asia Mater., 2021, 13, 53 CrossRef.
- W. Zhang, H. Zhuang, S. Chen, S. Hu, Y. Qian, Z. Wu, Z. Yang, J. Chen, Y. Xin and X. Ban, Chem. Eng. J., 2024, 498, 155350 CrossRef.
- P. Palanisamy, O. P. Kumar, H. U. Kim, K. R. Naveen, J.-Y. Kim, J.-H. Baek, M. Y. Chae and J. H. Kwon, Chem. Eng. J., 2024, 481, 148781 CrossRef.
- K. Kumada, H. Sasabe, M. Matsuya, N. Yoshida, K. Hoshi, T. Nakamura, H. Nemma and J. Kido, J. Mater. Chem. C, 2023, 11, 13782–13787 RSC.
- H. Wu, Y.-Z. Shi, M.-Y. Li, X.-C. Fan, F. Huang, K. Wang, J. Yu and X.-H. Zhang, Chem. Eng. J., 2024, 480, 147977 CrossRef.
- L. Yan, B. Chen, D. Wang, N. Su, L. Zhao, S. Wang and J. Ding, J. Mater. Chem. C, 2024, 12, 16827–16833 RSC.
- B. Du, K. Zhang, P. Wang, X. Wang, S. Wang, S. Shao and L. Wang, J. Mater. Chem. C, 2023, 11, 9578–9585 RSC.
- X. Huang, Y. Xu, J. Miao, Y.-Y. Jing, S. Wang, Z. Ye, Z. Huang, X. Cao and C. Yang, J. Mater. Chem. C, 2023, 11, 11885–11894 RSC.
- H. Li, H. Yan, L. Meng, X. Zhang, C. Kuang, Z. Meng, Y. He, H. Xu, X. Zhang, Y. Zheng, C. Yan and H. Meng, J. Mater. Chem. C, 2023, 11, 15548–15554 RSC.
- J.-J. Hu, X.-F. Luo, Y.-P. Zhang, M.-X. Mao, H.-X. Ni, X. Liang and Y.-X. Zheng, J. Mater. Chem. C, 2022, 10, 768–773 RSC.
- L. Chen, X. Tang, R. Liu, Q. Zheng, Y. Yu, D. Zhou, S. Yang, C. Adachi, L. Liao and Z. Jiang, Innovative Mater., 2023, 1, 100026 CrossRef.
- R. Jiang, N. Qiu, J. Li, X. Wu, N. Zheng, B. Z. Tang and Z. Zhao, Innovative Mater., 2023, 1, 100012 CrossRef.
- H.-X. Ni, W. Sun, X.-F. Luo, L. Yuan, X. Liang, X.-J. Liao, L. Zhou and Y.-X. Zheng, Innovative Mater., 2023, 1, 100041 CrossRef.
- D. Y. Chen, W. Liu, C. J. Zheng, K. Wang, F. Li, S. L. Tao, X. M. Ou and X. H. Zhang, ACS Appl. Mater. Interfaces, 2016, 8, 16791–16798 CrossRef.
- R. Ansari, W. H. Shao, S. J. Yoon, J. Kim and J. Kieffer, ACS Appl. Mater. Interfaces, 2021, 13, 28529–28537 CrossRef.
- Y. W. Zhang, D. D. Zhang, T. Y. Huang, A. J. Gillett, Y. Liu, D. P. Hu, L. S. Cui, Z. Y. Bin, G. M. Li, J. B. Wei and L. Duan, Angew. Chem., Int. Ed., 2021, 60, 20498–20503 CrossRef.
- Z. G. Wu, Y. Y. Xin, C. W. Lu, W. C. Huang, H. J. Xu, X. Liang, X. D. Cao, C. Li, D. D. Zhang, Y. W. Zhang and L. Duan, Angew. Chem., Int. Ed., 2024, e202318742 Search PubMed.
- J. Bian, S. Chen, L. Qiu, R. Tian, Y. Man, Y. Wang, S. Chen, J. Zhang, C. Duan, C. Han and H. Xu, Adv. Mater., 2022, 34, e2110547 CrossRef PubMed.
- T. Hatakeyama, K. Shiren, K. Nakajima, S. Nomura, S. Nakatsuka, K. Kinoshita, J. Ni, Y. Ono and T. Ikuta, Adv. Mater., 2016, 28, 2777–2781 CrossRef PubMed.
- S. Xu, Q. Q. Yang, Y. Zhang, H. Li, Q. Xue, G. H. Xie, M. Z. Gu, J. B. Jin, L. Huang and R. F. Chen, Chin. Chem. Lett., 2021, 32, 1372–1376 CrossRef.
- F. Liu, Z. Cheng, L. Wan, Z. Feng, H. Liu, H. Jin, L. Gao, P. Lu and W. Yang, Small, 2022, 18, 2106462 CrossRef CAS.
- Y. Wang, Y. Duan, R. Guo, S. Ye, K. Di, W. Zhang, S. Zhuang and L. Wang, Org. Electron., 2021, 97, 106275 CrossRef.
- Q. Wu, J. Y. Li, D. Liu, Y. Q. Mei, B. T. Liu, J. H. Wang, M. Xu and Y. X. Li, Dyes Pigm., 2023, 217, 111421 CrossRef.
- T. Fan, Y. Zhang, D. Zhang and L. Duan, Chem. – Eur. J., 2022, 28, e202104624 CrossRef PubMed.
- Y. Zhang, J. Wei, D. Zhang, C. Yin, G. Li, Z. Liu, X. Jia, J. Qiao and L. Duan, Angew. Chem., Int. Ed., 2022, 61, e202113206 CrossRef PubMed.
- P. Jiang, J. Miao, X. Cao, H. Xia, K. Pan, T. Hua, X. Lv, Z. Huang, Y. Zou and C. Yang, Adv. Mater., 2022, 34, 2106954 CrossRef.
- H. Hirai, K. Nakajima, S. Nakatsuka, K. Shiren, J. Ni, S. Nomura, T. Ikuta and T. Hatakeyama, Angew. Chem., Int. Ed., 2015, 54, 13581–13585 CrossRef PubMed.
- N. Ikeda, S. Oda, R. Matsumoto, M. Yoshioka, D. Fukushima, K. Yoshiura, N. Yasuda and T. Hatakeyama, Adv. Mater., 2020, 32, 2004072 CrossRef PubMed.
- F. Neese, WIREs Comput. Mol. Sci., 2012, 2, 73–78 CrossRef.
- F. Huang, Y.-C. Cheng, H. Wu, X. Xiong, J. Yu, X.-C. Fan, K. Wang and X.-H. Zhang, Chem. Eng. J., 2023, 465, 142900 CrossRef.
- W. Zhao, Z. He and B. Z. Tang, Nat. Rev. Mater., 2020, 5, 869–885 CrossRef.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4tc04656k |
| This journal is © The Royal Society of Chemistry 2025 |