Synthesis of a novel DOPO-based ionic liquid flame retardant and its application in epoxy resin†
Jinzhuo
Zhang‡
a,
Jiaming
Liang‡
 *b,
Jiapeng
Long
*a and
Bing
Liang
*a
*b,
Jiapeng
Long
*a and
Bing
Liang
*a
aSchool of Material Science and Technology, Shenyang University of Chemical Technology, Shenyang 110142, People's Republic of China. E-mail: liangbing@syuct.edu.cn; long2682@126.com
bDepartment of Applied Chemistry, School of Engineering, University of Toyama, Gofuku 3190, Toyama 930-8555, Japan. E-mail: jmliang@eng.u-toyama.ac.jp
First published on 2nd December 2024
Abstract
The low mechanical properties of flame-retardant epoxy resins restrict their applicability; therefore, the development of flame retardants designed to enhance the mechanical properties while maintaining the flame retardancy of epoxy resins presents significant potential for various applications. The present study introduces a novel fused-type ionic-liquid flame retardant, [DAmim]Ps, which is developed and applied to enhance the flame retardancy of EP. Unlike conventional additive flame retardants, [DAmim]Ps exhibits unique characteristics such as a melting point at 101 °C and active participation in the curing reaction of EP, thereby significantly facilitating the improvement of flame retardancy. Upon the incorporation of [DAmim]Ps, the EP composite system exhibited a [DAmim]Ps content of 6.8%, demonstrating exceptional performance with a remarkable limiting oxygen index (LOI) value of 32.9% and achieving a V-0 rating in a UL-94 vertical combustion test; meanwhile, the combustions of THR and HRR in EP-5 are 39.3% and 36.3% lower than those in EP-0, respectively, reflecting the excellent flame-retardant performance. Furthermore, the addition of [DAmim]Ps ionic liquid also promotes the mechanical properties of the EP system. The tensile strength, bending modulus, and impact strength increase by 51.14%, 57.7%, and 146.0%, respectively.
1. Introduction
Epoxy resin (EP), which has the advantages of low cost and chemical corrosion resistance,1–4 due to its excellent chemical resistance and thermal stability, has demonstrated a wide range of industrial applications in adhesives, electronics, aerospace, and marine systems.5–7 However, the poor flame retardancy of traditional epoxy resin is one of the main shortcomings, which greatly limits its application.8,9 Therefore, flame retardants were added to epoxy resins as additives or co-reactants to improve the flame retardancy, such as anhydrides and nanoparticles.10–12It is well-known that the combustion reaction requires the three elements of fuel, O2, and a certain temperature, all of which are indispensable.13,14 In this context, the mechanisms of flame retardant behaviors are to inhibit one or more elements when the material is burned, thus achieving the purpose of preventing or slowing down the combustion.15 The specific flame retardant mechanism of each flame retardant is different, but in general, it can be divided into physical effects and chemical effects. The physical effects include absorbing heat, diluting combustible substances, and isolating air, while the chemical effects include carbonization, free radical elimination, and phosphorylation.16–18 However, regardless of physical or chemical flame retardants, it usually requires a higher load rate, which greatly sacrifices some of the mechanical properties, glass transition temperature, and thermal stability of the crosslinked polymer. Under this background, how to improve the flame-retardant properties without affecting the mechanical properties of epoxy resin becomes a challenge.19
To achieve this target, a variety of flame retardants were introduced to the epoxy resin system to enhance the flame retardancy. In the previous studies, taking advantage of the surface reactions, additive flame retardants were employed for EP, and this kind of flame-retardant-modified composite exhibited better flame retardant effects compared with pure EP. The compound DOPO, which contains phosphorus, is widely recognized as an effective flame retardant due to its inherent properties. The flame retardancy mechanism of DOPO can be divided into two distinct parts. The first component functioned as a vapor phase flame retardant. Upon thermal decomposition of DOPO, it released free radicals such as PO˙ and ˙PO2, which effectively captured and quenched other free radicals including ˙H, ˙OH, and ˙O. Consequently, this process terminated the chain combustion reaction and exhibited its efficacy in suppressing flames. The second component was the coacervated flame retardant. Thermal decomposition of DOPO yielded phosphate ester and gas. The phosphate ester reacted with the combustible material to form a carbonized layer, effectively isolating oxygen and impeding flame propagation. Simultaneously, the resulting carbonized layer collaborated with the gas layer to further suppress flame spread.20 Several examples are presented in Table 1. Cheng et al. synthesized a VPD reactive flame retardant with 3,5-diamino-1,2,4-triazole and phenylphosphine dichloride as raw materials, and applied it to epoxy resin. The incorporation of 5 wt% VPD resulted in an increase in the limiting oxygen index (LOI) of the VPD/EP flame-retardant epoxy thermoset material from 24.7% to 26.9%, thereby achieving a V-2 classification in the UL-94 test. However, due to its high melting point, the limited surface reaction restricted its retardant properties. Meanwhile, the addition of solid particles reduced the mechanical properties of the epoxy resin system. As a result, the mechanical properties of VPD/EP flame-retardant epoxy thermosetting materials were lower than those of pure epoxy.21 A DOPO-derived flame retardant 6-((1H-benzo[d]imidazole-2-amino)dibenzooxy)phosphine hexaoxide (BADO) was successfully synthesized and incorporated into epoxy resin by Liu et al. The results demonstrated the exceptional flame retardancy of the modified epoxy resin. Upon the addition of 5 wt% BADO, the limiting oxygen index of the modified epoxy resin ranged from 19.8% to 28.3%, and it achieved a V-0 rating in the UL-94 test. Notably, BADO acted as a crosslinking agent at the crosslinking site; excessive usage might compromise its effectiveness, hindering the formation of a three-dimensional network structure and consequently reducing the mechanical properties of epoxy resin.22 Duan et al. successfully synthesized a novel phosphorus/nitrogen-containing polycarboxylic acid (TMD) and incorporated it into epoxy resin. The results indicated that the addition of 6.5 wt% TMD led to a slight increase in the limiting oxygen index of the modified epoxy resin to 25.0%, while its tensile strength had improved by 50%.23 Wang et al. synthesized a bisphenol flame retardant based on aromatic imide DOPO (DOPO-AI) and incorporated it into epoxy resin. The results indicated that the addition of 5 wt% DOPO-AI resulted in an increase in the limiting oxygen index of the modified epoxy resin from 27.5% to 32.7%, thereby achieving a V-0 rating in the UL-94 test. Furthermore, its mechanical properties were slightly enhanced, with tensile strength increasing by 0.2% and bending modulus improving by 1.37%.24 Consequently, developing a novel type of flame retardant which could better participate in the reaction to increase the flame retardancy of EP and take into account its mechanical properties has great prospects. Meanwhile, as future developments progress, the performance requirements for epoxy resins are expected to increase gradually.25
| Flame retardant | wt% | LOI | UL-94 | Tensile strengtha (%) | Impact strengtha (%) | Bending modulusa (%) |
|---|---|---|---|---|---|---|
| a Compared with epoxy resin without flame retardant (tensile strength, impact strength and bending modulus), “+” is improved, “−” is reduced. | ||||||
| VPD | 5.00 | 26.9 | V-2 | −13.75 | −22.96 | — |
| BADO | 5.00 | 28.3 | V-0 | −7.46 | −10.10 | — |
| TMD | 6.50 | 25.0 | NR | +50.00 | — | +0.30 |
| DOPO-AI | 5.00 | 32.7 | V-0 | +0.20 | — | +1.37 |
| [DAmim]Ps | 5.36 | 31.3 | V-0 | +51.14 | +146.00 | +57.70 |
Herein, leveraging the excellent flame retardancy of DOPO, a [DAmim]Ps ionic liquid incorporating phosphorus, sulfur, and nitrogen elements based on DOPO was successfully synthesized and added into the epoxy resin matrix. [DAmim]Ps could melt and react well with the epoxy resin at 200 °C. Simultaneously, the amino groups in the structure could effectively participate in the curing process of the epoxy resin. As a result, both the flame retardancy and mechanical properties of the [DAmim]Ps and epoxy resin composites were enhanced. With the modification of [DAmim]Ps, the EP-5 composite exhibited the highest limiting oxygen index (LOI) of 32.9% with a V-0 rating in UL-94 vertical combustion. Furthermore, the tensile strength, bending modulus, and impact strength increased by 51.14%, 57.7%, and 146.0%, respectively. Overall, our work provided a new approach to addressing high-fire risk scenarios with stringent material strength requirements.
2. Experimental
2.1. Experimental materials
Analytically pure DOPO, polyformaldehyde, propyl sulfonyl chloride, and 1–3 aminopropyl imidazole were provided by Shanghai Maclin Biochemical Technology Co., Ltd; analytically pure xylene, triethylamine, ethyl acetate, dichloromethane, anhydrous ethanol, acetonitrile, and 4,4-diaminodiphenylmethane (DDM) were provided by Sinopharm Chemical Reagent Co., Ltd; E-51 epoxy resin was provided by Nantong Star Synthetic Material Co., Ltd.2.2. Preparation of [DAmim]Ps
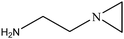
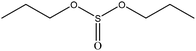
The synthesis roadmap of [DAmim]Ps is displayed in Fig. 1. First, xylene of 50 mL, DOPO of 21.6 g (0.1 mol), and paraformaldehyde of 3.0 g (0.1 mol) were added to the reaction bottle. In a N2 atmosphere, the mixture was magnetically stirred at 60 °C and reacted for 3 hours. After the reaction was completed, the white solid powder DOPO-OH of 22.1 g with an 89.9% yield was obtained by using xylene and distilled water to filter twice alternately, and then the product was vacuum dried at 80 °C for 24 hours.50 mL of dichloromethane, triethylamine of 10.3 g (0.1 mol), and DOPO-OH of 24.6 g (0.1 mol) were put into a three-mouth flask. After the DOPO-OH was dissolved, propyl sulfonyl chloride of 14.2 g (0.1 mol) was added to the liquid at the atmosphere pressure, followed by stirring at room temperature for 3 hours. After the reaction was completed, the dichloromethane was removed by vacuum distillation, and the residual product was then dispersed in ethyl acetate and extracted three times with distilled water to incorporate the organic layer. The organic layer was distilled under pressure again to remove ethyl acetate and dried under vacuum at 80 °C for 24 hours to obtain white solid powder DOPO-Ps of 16.4 g with a yield of 46.7%.
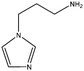
50 mL of acetonitrile and DOPO-Ps of 35.2 g (0.1 mol) were put into a three-mouth flask. After the DOPO-Ps was dissolved, 1–3 aminopropyl imidazole of 12.5 g (0.1 mol) was added, and then the mixture was stirred at 60 °C for 24 hours. After the reaction was completed, acetonitrile was removed by vacuum distillation. The residual product was dispersed in ethyl acetate after vacuum distillation and extracted three times with distilled water to incorporate the water phase. After the water phase was distilled again under 0.01 MPa pressure and the distilled water was removed, the light yellow powder [DAmim]Ps of 42.7 g was obtained by vacuum drying at 80 °C for 24 hours with a yield of 92.3%.
2.3. Preparation of [DAmim]Ps/DDM/EP composites
[DAmim]Ps, DOPO, and 4,4-diaminodiphenylmethane (DDM) were utilized to formulate a series of halogen-free flame-retardant epoxy resin systems with varying phosphorus, nitrogen, and sulfur contents, employing bisphenol a epoxy resin (E-51) as the matrix. The specific amounts added and the corresponding content calculation formulas are presented in Table S1 (ESI†).2.4. Characterization
Fourier transform infra-red spectroscopy (FTIR, NEXUS-470) was performed on samples using the KBr tablet method, ranging from 4000 to 400 cm−1. 1H NMR spectra (1H NMR, Bruker AscEndTM 500) and 13C NMR spectra (13C NMR, Burker AscEndTM 500) were used to further analyse the structure of [DAmim]Ps, and DMSO-d6 was used as the solvent. Mass spectrometry (MS, 6550 QTOF) was characterized by ESI† ion source with methanol as the solvent and positive spectrum test mode. The thermogravimetric analyses (TG) were conducted using a Pyris thermogravimetric analyzer (PerkinElmer TGA 4000) by heating the samples (approximately 10 mg) from room temperature to 800 °C at a rate of 10 °C min−1, while maintaining a continuous flow of nitrogen throughout the process. The differential scanning calorimeter (DSC, instrument TA Company, Q25) was the curing behavior of the epoxy system under the nitrogen atmosphere from 50 °C to 225 °C at different heating rates (2, 5, and 10 °C min−1). The sample mass was about 5.0 mg.The limiting oxygen index (LOI, Nanjing Jiangning Instrument Factory, JF-3) was the minimum oxygen concentration required for a material to flame in a mixture of oxygen and nitrogen under specified conditions. The procedure and dimensions (130.0 mm × 9.5 mm × 4.0 mm) were in accordance with GB/T2406-93 standards. Five samples were utilized for each set of tests. The UL-94 ratings were determined using the testing procedure specified in ASTM D 3801 (Nanjing Jiangning Instrument Factory, CZF-3), with a sample size of 130.0 mm × 9.5 mm × 4.0 mm. The burning grade of the sample was classified as V-0, V-1, V-2 or NR (unclassified), based on its combustion behavior.
The cone calorimeter test (CCT United Kingdom FTT0007) was measured by a cone calorimeter 6810. The sample size (100.0 mm × 100.0 mm × 3.0 mm) and heat flux (50 kW m−2) complied with the ISO 5660-1 protocol. A scanning electronic microscope (SEM, Hitachi S4800) test was used for testing and analyzing the morphology of the residual carbon. All samples were characterized after gold spraying treatment. The X-ray photoelectron spectroscopy (XPS, the ESCALAB 250Xi) analysis was conducted with a Thermo Scientific K-Alpha+ spectrometer with a mono AlKα excitation source. The layer present on top of the residual chars after the test was selected as the sample for testing. A Laser Raman spectrometer (Lab HORIBA JY) was used to test the char residue of the sample using the RAM HR reflection confocal.
The TGA Q500IR (TG-IR) thermogravimetric test was conducted using a TGA Q50IR thermogravimetric analyzer coupled with a Nicolet 6700 red external spectrometer. The sample, weighing approximately 5 mg, was placed in an oxygenating crucible under a high-purity nitrogen gas atmosphere for testing within a temperature range of 30 to 800 °C. The temperature ramp rate was set at 10 °C min−1, with a nitrogen flow rate of 50 mL min−1. Pyrolytic-gas chromatography-mass spectrometry (py-GC-MS) was used to analyze the pyrolysis behavior of EP samples by an Agilent 7890/5975 GC/MS, and the pyrolysis temperature of GC/MS was set at 400 °C.
The dynamic mechanical analysis (DMA, TA 850) was conducted in the three-point bending mode. The test temperature ranged from 10–250 °C at a rate of 10 °C min−1. The dimensions of the samples were 50.0 mm × 10.0 mm × 4.0 mm. Mechanical properties (Shenzhen Chuyinghao Technology Co., Ltd; CMT4202) were tested by the GT-7045-MDL impact testing machine and TCS-2000 tensile testing machine. All of the above results were the average of five samples.
3. Results and discussion
3.1. Structural analysis of [DAmim]Ps
The structure of the synthesized [DAmim]Ps ionic liquid was verified by FTIR, NMR, and mass spectrometry. Fig. 2a displays the FT-IR spectra of DOPO, DOPO-OH, DOPO-Ps, and [DAmim]Ps. The peaks that appeared at 831 cm−1, 932 cm−1, 1371 cm−1, 1380 cm−1, 1448 cm−1, 1645 cm−1, and 3230 cm−1 were attributed to the asymmetrical stretching vibration of S–C, the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration, the asymmetric stretching vibration of C–O–S, the S–O stretching vibration, the stretching vibration peak of the imidazole skeleton, the –NH2 bending vibration, and a kind of symmetric stretching vibration of the –OH bond, respectively.26,27 These stretching vibration peaks corresponded well with the expected product structures, which sufficiently indicated the successful synthesis of the four compounds.
O stretching vibration, the asymmetric stretching vibration of C–O–S, the S–O stretching vibration, the stretching vibration peak of the imidazole skeleton, the –NH2 bending vibration, and a kind of symmetric stretching vibration of the –OH bond, respectively.26,27 These stretching vibration peaks corresponded well with the expected product structures, which sufficiently indicated the successful synthesis of the four compounds.
 | ||
| Fig. 2 Structural analysis of [DAmim]Ps. (a) FTIR spectra of DOPO, DOPO-OH, DOPO-Ps and [DAmim]Ps. (b) 1H NMR spectra of [DAmim]Ps. (c) 13C NMR spectra of [DAmim]Ps. (d) Mass spectrum of [DAmim]Ps. | ||
The 1H NMR spectra were employed to determine the H atom microenvironment of DOPO, DOPO-OH, DOPO-Ps, and [DAmim]Ps (Fig. 2b and Fig. S1, ESI†). The signal of δ = 2.5 corresponded to the solvent peak of DMSO-d6. For DOPO, the resonance signal of all unsaturated H atoms occurred in the range of about 6.7 to 8.4 ppm, and there was a P–H bond observed at δ = 8.7. In DOPO-OH, the δ = 4.3 peak corresponded to the proton peak of the hydrogen atom on –CH2 on methylene, and δ = 3.8 was attributed to the hydrogen atoms attached to the –OH group on the methyl moiety. As for the DOPO-Ps, δ = 5.1 corresponded to proton peaks of hydrogen atoms on P–CH2–O, and δ = 3.2, 1.4, and 0.8 corresponded to hydrogen atoms on methyl and methylene groups attached to S atoms (Fig. S1, ESI†). Finally, in [DAmim]Ps, δ = 8.9–9.1 corresponded to the H atom on the imidazole ring, while δ = 2.0 corresponded to the proton peak of the hydrogen atom on –NH2, and δ = 4.0, 2.8, and 2.6 corresponded to the proton peak of the hydrogen atom on the propyl group attached to –NH2 (Fig. 2b).28 The above results were basically consistent with the design synthesis route of [DAmim]Ps (Fig. 1).
Furthermore, the microenvironment of C atoms in the [DAmim]Ps was detected by 13C NMR (Fig. 2c). The signal of δ = 53.9 corresponded to the C atom on –CH2 of methylene; the signals of δ = 29.4, 36.7, and 43.5 were the C atoms above the propyl group attached to –NH2, and the peaks of δ = 13.9, 18.9, and 60.2 corresponded to the methyl and methylene carbon atoms on the propyl group attached to the S atom.29 In addition, the resonance signals of all unsaturated C atoms occurred in the range of about 120 to 155 ppm, with a high chemical shift in the low NMR field. Among them, δ = 128.6, 128.9, and 142.1 corresponded to the C atom on the imidazole ring. 13C NMR spectra further confirmed that the structure of [DAmim]Ps was consistent with the target product.
The mass spectrum of [DAmim]Ps was depicted and summarized in Fig. 2d and Table S2 (ESI†). The mass-to-charge ratio of 353.25 was consistent with the cationic structure of [DAmim]Ps, while the mass-to-charge ratio of 123.07 corresponded to the anionic structure of [DAmim]Ps. Additionally, a mass-to-charge ratio equal to 601.44 represented [M + 2A]+, where M denoted the cationic structure and A signified the anionic structure of [DAmim]Ps. The results obtained from mass spectrometry also aligned with the theoretical relative molecular weight of [DAmim]Ps.
In conclusion, the synthesis path and the predicted structure of the monoamine-modified DOPO-based ionic liquid [DAmim]Ps were confirmed by characterizing the microenvironment of C and H atoms and the mass–charge ratios of the ionic liquid.
3.2. Curing behavior of [DAmim]Ps in the [DAmim]Ps/DDM/EP composite materials
The curing conditions of the composite system were investigated by differential scanning calorimeter (DSC). According to the results of DSC data, the curing process of the [DAmim]Ps/DDM/EP was ensured: 75 °C curing for 2 hours, then 115 °C curing for 4 hours, and finally 140 °C curing for 2 hours to obtain the flame-retardant epoxy resin composite (Fig. S2, ESI†).The melting point of [DAmim]Ps was initially determined to investigate the role of the designed ionic liquid in the composite curing process. As shown in Fig. 3a, the melting point of [DAmim]Ps was about 101.3 °C, indicating that it could melt adequately under the curing conditions of EP, which reflected the superiority of [DAmim]Ps in the curing and crosslinking reactions compared with the traditional additive flame retardants. Furthermore, the incorporation of molten flame retardants can effectively mitigate the formation of solvent-induced bubbles, thereby minimizing their detrimental impact on the mechanical properties of EP.
 | ||
| Fig. 3 The exploration of melting and curing characteristics of [DAmim]Ps. (a) Fusing point of [DAmim]Ps. (b) FTIR spectra of EP and [DAmim]Ps/EP. | ||
Subsequently, to clearly understand the curing effect of [DAmim]Ps ionic liquid, we performed FTIR spectra on pure EP and the [DAmim]Ps/EP composite, which were without curing agent. Obviously, compared with pure EP, the peaks at 3521 cm−1 and 3061 cm−1 appeared, which were attributed to the –OH peak and CO–NH peak, respectively, further proving that [DAmim]Ps participated in the curing reaction due to the presence of monoamine groups (Fig. 3b).30 The melting point test and FTIR spectroscopy sufficiently demonstrated the melting and curing characteristics of [DAmim]Ps ionic liquid, which could greatly improve the flame retardant and mechanical properties of EP.
3.3. Flame retardancy of [DAmim]Ps/DDM/EP composites
In the following, we employed a variety of techniques to comprehensively evaluate the flame retardancy of the [DAmim]Ps/DDM/EP composite. First, the EP composites were investigated using the LOI and UL-94 tests, which are two common methods for testing the flame-retardant properties of the materials. In the LOI test, the pure EP exhibited an LOI value of 24.7%. An increase in the concentration of [DAmim]Ps ionic liquid significantly enhanced the LOI, resulting in a value of 32.9% for EP-5 and achieving a V-0 rating without any dripping (Fig. 4).The time to ignition (TTI) is a crucial parameter for evaluating the fire resistance of materials, representing the time required for the surface of the material to ignite under constant thermal radiation conditions. As shown in Table 2, TTI decreased from 61 s (EP-0) to 51 s (EP-4) and 47 s (EP-5), respectively, as the amount of [DAmim]Ps increased, indicating that the overall ignition time of the EP composites decreased due to the premature decomposition of [DAmim]Ps, while the decomposed products had a flame retardant effect.31
| Samples | TTI (s) | PHRR (kW m−2) | THR (MJ m−2) | TSP (m2) | COP (g s−1) | FPI (s m2 kW−1) |
|---|---|---|---|---|---|---|
| EP-0 | 61 | 1446 | 168.7 | 61.3 | 0.052 | 0.042 |
| EP-4 | 51 | 1007.5 | 119.3 | 21.58 | 0.038 | 0.050 |
| EP-5 | 47 | 920.7 | 102.4 | 20.40 | 0.025 | 0.051 |
Furthermore, the term HRR is defined as the heat release per unit area upon ignition of the material under constant heat flow conditions. The maximum value of HRR was referred to as the peak heat release rate (PHRR), which represents the highest rate of heat release during continuous combustion of the material.32 The flame-retardant EP composite exhibited an earlier decomposition time compared to the pure EP, resulting in a lower PHRR for all flame-retardant epoxy resins. Specifically, the PHRR was decreased by 30.3% and 36.3% in EP-4 and EP-5, from 1446 kW m−2 to 1007.5 kW m−2 and 920.7 kW m−2, respectively, indicating an effective reduction in PHRR through the addition of [DAmim]Ps (Fig. 5a). The ratio of the TTI to PHRR (TTI/PHRR) mentioned above was denoted as fire performance (FPI) and was used to measure the flame propagation rate in a fire.32 Obviously, the FPI values of the EP composites were higher than that of pure EP (Table 2), illustrating that the addition of [DAmim]Ps increased the combustion performance of EP to a certain extent.
 | ||
| Fig. 5 Cone calorimetric test analysis. (a) HRR curves of [DAmim]Ps/DDM/EP composites. (b) THR curves of [DAmim]Ps/DDM/EP composites. | ||
THR (total heat release) was the sum of the heat released by the material from the beginning of combustion to the end of combustion under the irradiation of a constant heat flow.31–33 According to Fig. 5b, compared with the EP-0, the THR of the composites with [DAmim]Ps was distinctly reduced; among them, EP-4 and EP-5 decreased THR by 29.3% and 39.3%, respectively, demonstrating a weakening in flame intensity brought by [DAmim]Ps ionic liquid. The total amount of smoke released (TSP) was another critical factor in the fire evaluation, serving as a measure of the intensity of smoke emitted during combustion.31–33 As depicted in Table 2, EP-5 exhibited the lowest TSP value of 20.4 m2, which revealed that [DAmim]Ps effectively suppressed the smoke generation of EP.
Due to the poisoning and the threat to human health caused by the CO released, the CO yield of the composites during the combustion was also tested.33 The COP (CO production rate) data for EP-0 to EP-5 is displayed in Table 2. The CO yield of EP-0 was 0.052 g s−1, while the CO yields of EP-4 and EP-5 were 0.025 g s−1 and 0.038 g s−1, decreased by 26.9% and 51.9% compared to EP-0, respectively.
The above tests clearly illustrated that the addition of [DAmim]Ps ionic liquid could not only enhance the flame retardant performances of EP but also reduce the heat release and CO emission during the combustion process, which is of great significance for the application of EP in fire conditions.
3.4. Flame retardant mechanism of [DAmim]Ps and [DAmim]Ps/DDM/EP composites
In order to clearly explore the flame retardant mechanism of the [DAmim]Ps ionic liquid, the TG-IR analyses were performed on the [DAmim]Ps, EP-0, and EP-5 (Fig. 6 and Fig. S3, ESI†). For the [DAmim]Ps sample, as the temperature rose, [DAmim]Ps began to decompose, and characteristic peaks appeared in the FTIR spectra at the same time (Fig. S3, ESI†). The peak observed in the 2400–2200 cm−1 range corresponded to the formation of CO2, while the peak in the 1400–1300 cm−1 range was attributed to the SO2 formation. Additionally, peaks in the 1000–900 cm−1 range were attributed to NH3. Peaks associated with phosphorus groups appeared at 1177 cm−1 (P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O), suggesting the cleavages of the P–C bonds in DOPO. These phosphorous groups would further break into phosphorous radicals, which could synergize with non-combustible gases such as H2O and CO2, thereby enhancing the flame retardancy of the gas phase.34
O), suggesting the cleavages of the P–C bonds in DOPO. These phosphorous groups would further break into phosphorous radicals, which could synergize with non-combustible gases such as H2O and CO2, thereby enhancing the flame retardancy of the gas phase.34
The thermogravimetric analysis curves and their corresponding FTIR spectra of EP-0 and EP-5 are shown in Fig. 6a–d. Similarly, the characteristic absorption peaks that belonged to the EP matrix in EP-0 and EP-5 were observed at 2344 cm−1 (CO2), 1610–1510 cm−1 (aromatic compounds), and 1176 cm−1 (adipose ether).35 Moreover, due to the weak bonds of C–O, the peak of the adipose ether disappeared at 500 °C. These results indicated that the thermal oxidative degradation of EP/DDM thermoset materials was due to the fracture of O–H and C–O bonds in the skeleton. However, the differences between EP-0 and EP-5 were that the polyaromatic structure peaks of the EP-5 sample were more pronounced than those of the EP-0 sample in the range of 400 °C to 600 °C. Furthermore, the formations of SO2 and NH3 were detected in the range of 1400–1300 cm−1 and 1000–900 cm−1, respectively, in the EP-5 composite.36 In addition, absorption peaks associated with phosphorus groups were observed at 1177 cm−1 (P![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) (Fig. 6b and d). Whereas these peaks were almost not observed in the EP-0 sample (Fig. 6d). The observed phenomena provide strong evidence for the enhanced generation of these flame retardant groups and consequent improvement in the flame retardant properties of epoxy resin composites upon the addition of [DAmim]Ps.
O) (Fig. 6b and d). Whereas these peaks were almost not observed in the EP-0 sample (Fig. 6d). The observed phenomena provide strong evidence for the enhanced generation of these flame retardant groups and consequent improvement in the flame retardant properties of epoxy resin composites upon the addition of [DAmim]Ps.
To further understand the pyrolysis behavior of the [DAmim]Ps ionic liquid, the decomposition products of [DAmim]Ps were directly defined by Py-GC/MS analysis (Table 3). The pyrolysis products of the [DAmim]Ps sample contained abundant aniline and phenolic fragments, which were in good agreement with the results of TG-FTIR. Only a small fraction of the debris was observed before the residence time of 15 minutes, indicating that most of them remained in the condensed phase to form polyaromatic compounds during pyrolysis.37 Combining the above conclusions, it could be summarized that during the combustion process, the P-containing compounds generated by [DAmim]Ps facilitated the formation of heat-stable oxides in the condensed phase, thus facilitating the formation of N-based and S-based non-flammable gases.
3.5. Microstructure of char residue in [DAmim]Ps/DDM/EP composites
It is well-known that the char residue layer after combustion also plays a crucial role in flame retardants. Accordingly, the epoxy resin splines were burned, and their char residue was tested and analyzed. Spline diagrams displayed the burned epoxy resin composites from EP-0 to EP-5 (Fig. 7a). It was evident that the expanded carbon layer gradually formed with the addition of [DAmim]Ps, thus inhibiting combustion and promoting the flame-retardant properties of epoxy resin. Meanwhile, the density of the expanded carbon layer was enhanced correspondingly with the increase in [DAmim]Ps ionic liquid, as observed by SEM images (Fig. 7a).Raman spectroscopy is an effective method to explore the structure of carbon materials. As depicted in Fig. 7b, two distinct peaks at 1339 and 1586 cm−1 were observed, which were consistent with the disordered (D-band) and ordered (G-band) structures of the graphite layer, and the intensity ratios of the D-band to G-band (ID/IG) are utilized to determine the degree of disorder of residual carbon after combustion. Typically, a higher ID/IG value represents a higher degree of disorder.38,39 The ID/IG value for char residue after the combustion of the EP-5 composite was found to be lower than that of EP-0, indicating that a more ordered and dense carbon layer was formed, which was consistent with the SEM results (Fig. 7a). Furthermore, the char residue rate of the composite material significantly increased with the rise in phosphorus content (Fig. 7c). Under identical conditions, the char residue ratios of the EP-0 at 600 °C and 800 °C were recorded as 17.3% and 15.2%, respectively, while the ratios were improved to 26.2% and 21.7% in EP-5. Since the denser and thicker char residue layer could efficiently hinder the combustion of EP by providing a physical barrier effect, the characterizations of the char residue adequately illustrated the superiority of our designed [DAmim]Ps ionic liquid in the flame retardant of the EP system.
Moreover, XPS tests were performed on the combusted char residue to conduct a detailed analysis of the chemical elements. The carbon layer of EP-0, which was without [DAmim]Ps, consisted of only three elements: C, O, and N (Fig. 8a). However, in the total spectrum of residual carbon in EP-5 (Fig. 8b), the characteristic peaks of P and S elements were observed.40–42
 | ||
| Fig. 8 XPS spectra of EP-0 and EP-5. (a) Total char residue spectrum of EP-0. (b) Total char residue spectrum of EP-5. (c) C 1s spectra of EP-5 char residue. (d) C 1s spectra of EP-0 char residue. | ||
Meanwhile, in the C 1s spectra of EP-5, the absorption peaks at 284.8, 285.2, 286.5, and 288.2 eV were attributed to C–C, C–O–P, C–N, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C, respectively (Fig. 8c),43,44 while in the C 1s spectra of EP-0, the C–O–P bonds were not observed, which provided strong evidence for the combination of C with P and N elements during the formation of the carbon layers after [DAmim]Ps was added. Moreover, the existence of the C
C, respectively (Fig. 8c),43,44 while in the C 1s spectra of EP-0, the C–O–P bonds were not observed, which provided strong evidence for the combination of C with P and N elements during the formation of the carbon layers after [DAmim]Ps was added. Moreover, the existence of the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C absorption peak proved that the carbon layer could realize aromatization, and the aromatic structure could further improve the thermal stability of the carbon layer.43–45
C absorption peak proved that the carbon layer could realize aromatization, and the aromatic structure could further improve the thermal stability of the carbon layer.43–45
Therefore, combined with the above characterization results, we could conclude that, the improvements in flame-retardant performance could be attributed to the superior synergistic effect between P, N, and S as flame retardants. Specifically, phosphorus-based condensed-phase flame retardants could form a stable carbon layer containing phosphorus during combustion. Additionally, P in the composite material contributed to early-stage thermal decomposition.46 The strong acids, such as phosphoric acid and polyphosphate, generated in this process could facilitate the substrate's dehydration into carbon, thereby increasing both the quantity of char residue and the quality of the resulting carbon layer. Simultaneously, nitrogen gas produced during combustion could serve as a blowing agent for creating a foaming expanded porous carbon layer, effectively preventing combustible gases from entering the gas phase and isolating oxygen.47
3.6. Mechanical property analysis of [DAmim]Ps/DDM/EP composites
In addition to the flame-retardant abilities, mechanical properties are also very important indicators for the application of epoxy resins. However, the additive and solvent types of flame retardants in the previous studies still have great defects in the mechanical properties of EP. In this context, we also tested the mechanical properties of the EP composites, which were modified by [DAmim]Ps ionic liquid.The tensile strength, impact strength, and bending modulus of EP composites as classical mechanical performance indicators were evaluated. All of them exhibited an initial increase followed by a subsequent decrease with the augmentation of the [DAmim]Ps mass fraction in the ionic liquid (Fig. 9a–c). The mechanical properties of the sample attained their optimum level at the EP-4; however, an additional increase in the amount of [DAmim]Ps resulted in a decline in the mechanical properties of the materials. The reasons for this trend were as follows: first, the moderate crosslinking could improve the elasticity of the material. The modulus plateau appeared when the linear molecule was stretched to a certain extent, leading to slip between the chain segments. However, crosslinking could limit the movement of the chain and the distance between the chain segments, resulting in an improvement in the elasticity of the material.
Secondly, the incorporation of the ionic liquid as a plasticizer enhanced the intermolecular spacing between polymer chain segments, mitigating energy dissipation resulting from direct chain–chain contact, and finally enhancing the elasticity.48 The mechanical properties of EP-5 materials exhibited a tendency to decrease, which was potentially attributed to the reduction in crosslinking density due to the excessive introduction of ionic liquids.
The DMA curves of EP composites with varied contents of the ionic liquid [DAmim]Ps are presented in Fig. 10a. The energy storage modulus of different samples exhibited a trend of an initial increase followed by a subsequent decrease. Furthermore, the glass transition temperature of the sample corresponded to the highest point on the peak. A slight decrease in the glass transition temperature of the EP composite compared to EP-0 was observed (Fig. 10b). The crosslinking density of the epoxy system is shown in Table 4. This indicated that the presence of the ionic liquid exerted a plasticizing effect on the system and reduced the glass transition temperature of the composite.48 This was consistent with the mechanical results.
 | ||
| Fig. 10 DMA analysis of [DAmim]Ps/DDM/EP composites. (a) DMA test of the [DAmim]Ps/DDM/EP composite system. (b) Loss factor of the [DAmim]Ps/DDM/EP composite system. | ||
| Sample | EP-0 | EP-1 | EP-2 | EP-3 | EP-4 | EP-5 |
|---|---|---|---|---|---|---|
| Mc | 184.49 | 251.71 | 272.37 | 302.00 | 317.38 | 240.17 |
4. Conclusions
A novel DOPO-based ionic liquid, [DAmim]Ps, was successfully designed and employed in the flame-retardant system of epoxy resin. Various characterizations illustrated that the [DAmim]Ps could melt under the curing conditions and participate in the curing reaction of EP due to the existence of monoamine bonds. With this characteristic, [DAmim]Ps not only exhibited excellent flame retardancy but also significantly improved the mechanical properties of EP compared with the previously reported additive types of flame retardants. At a phosphorus content of 0.45%, the EP-5 achieved 32.9% in the LOI test, meeting the V-0 grade of UL-94. Additionally, in cone calorimeter tests, the PHRR, THR, and TSP of the modified epoxy thermoset resin decreased by 36.3%, 39.3%, and 66.7%, respectively, compared to the pure EP. At the same time, the addition of [DAmim]Ps ionic liquid also increased the tensile strength, bending modulus, and impact strength by 51.4%, 57.7%, and 146.0%, respectively.Furthermore, we conducted an analysis of the decomposition products of the flame-retardant system and char residue post-combustion to elucidate the mechanism underlying flame retardancy. The remarkable flame retardant properties observed in the EP composite can be attributed to the synergistic effect of nitrogen, phosphorus, and sulfur present in the [DAmim]Ps ionic liquid. In conclusion, our study not only presents a novel approach for simultaneously utilizing a well-designed ionic liquid as both a flame retardant and curing agent to enhance the flame-retardant and mechanical characteristics of EP systems, but also provides a promising strategy for addressing high-fire-risk scenarios with stringent material strength requirements.
Data availability
The data supporting this article have been included as part of the ESI.†Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work was supported by Department of Science and Technology of Liaoning province, Liaoning province unveils science and technology project (2021JH1/10400101); and the PhD Start-up Research Foundation of Department of Science and Technology of Liaoning Province (2021-BS-184).References
- J. Sun, B. Wang, Z. Guo, Z. Fang and J. Li, Flame retardant epoxy resin toughened and strengthened by a reactive compatibilizer, Polymer, 2022, 248, 124798 CrossRef CAS.
- Y. Qiu, L. Qian, H. Feng, S. Jin and J. Hao, Toughening effect and flame-retardant behaviors of phosphaphenanthrene/phenylsiloxane bigroup macromolecules in epoxy thermoset, Macromolecules, 2018, 51, 9992–10002 CrossRef CAS.
- H. Duan, Y. Chen, S. Ji and R. H. Hu, A novel phosphorus/nitrogen-containing polycarboxylic acid endowing epoxy resin with excellent flame retardance and mechanical properties, Chem. Eng. J., 2019, 375, 121916 CrossRef CAS.
- Y. Zhang, P. A. Song, S. Fu and F. Chen, Morphological structure and mechanical properties of epoxy/polysulfone/cellulose nanofiber ternary nanocomposites, Compos. Sci. Technol., 2015, 115, 66–71 CrossRef CAS.
- H. Gu, C. Ma, J. Gu, J. Guo, X. Yan, J. Huang, Q. Zhang and Z. Guo, An overview of multifunctional epoxy nanocomposites, J. Mater. Chem. C, 2016, 4(25), 5890–5906 RSC.
- J. Guo, H. Song, H. Liu, C. Luo, Y. Ren, T. Ding, M. Khan, D. Young, X. Liu, X. Zhang and J. Kong, Polypyrrole-interface-functionalized nano-magnetite epoxy nanocomposites as electromagnetic wave absorbers with enhanced flame retardancy, J. Mater. Chem. C, 2017, 5(22), 5334–5344 RSC.
- Y. Li, Y. Qian, Q. Jiang, A. Haruna, Y. Luo and J. Yang, Thermally conductive polymer-based composites: fundamentals, progress and flame retardancy/anti-electromagnetic interference design, J. Mater. Chem. C, 2022, 10(39), 14399–14430 RSC.
- J. C. Capricho, B. Fox and N. Hameed, Multifunctionality in epoxy resins, Polym. Rev., 2020, 60, 1–41 CrossRef CAS.
- A. Surnova, D. Balkaev, D. Musin, R. Amirov and A. M. Dimiev, Fully exfoliated graphene oxide accelerates epoxy resin curing, and results in dramatic improvement of the polymer mechanical properties, Composites, Part B, 2019, 162, 685–691 CrossRef CAS.
- Z. Yao, L. Qian, Y. Qiu, Y. Chen, B. Xu and J. Li, Flame retardant and toughening behaviors of bio-based DOPO-containing curing agent in epoxy thermoset, Polym. Adv. Technol., 2020, 31, 461–471 CrossRef CAS.
- Y. Chen, L. Xu, X. Wu and B. Xu, The influence of nano ZnO coated by phosphazene/triazine bi-group molecular on the flame retardant property and mechanical property of intumescent flame retardant poly (lactic acid) composites, Thermochim. Acta, 2019, 679, 178336 CrossRef CAS.
- M. Zhang, X. Ding, Y. Zhan, Y. Wang and X. Wang, Improving the flame retardancy of poly(lactic acid) using an efficient ternary hybrid flame retardant by dual modification of graphene oxide with phenylphosphinic acid and nano MOFs, J. Hazard. Mater., 2020, 384, 121260 CrossRef CAS.
- Y.-C. Zhang, G.-L. Xu, Y. Liang, J. Yang and J. Hu, Preparation of flame retarded epoxy resins containing DOPO group, Thermochim. Acta, 2016, 643, 33–40 CrossRef CAS.
- L. Long, J. Yin, W. He, Y. Xiang, S. Qin and J. Yu, Synergistic effect of different nanoparticles on flame retardant poly (lactic acid) with bridged DOPO derivative, Polym. Compos., 2019, 40, 1043–1052 CrossRef CAS.
- M. Xie, S. Zhang, Y. Ding, F. Wang, P. Liu, H. Tang, Y. Wang and M. Yang, Synthesis of a heat-resistant DOPO derivative and its application as flame-retardant in engineering plastics, J. Appl. Polym. Sci., 2017, 134, 44892–44903 CrossRef.
- J. Liu, J. Tang, X. Wang and D. Wu, Synthesis, characterization and curing properties of a novel cyclolinear phosphazene-based epoxy resin for halogen-free flame retardancy and high performance, RSC Adv., 2012, 2, 5789–5799 RSC.
- X. Qian, L. Song, Y. Bihe, B. Yu, Y. Shi, Y. Hu and R. K. Yuen, Organic/inorganic flame retardants containing phosphorus, nitrogen and silicon: Preparation and their performance on the flame retardancy of epoxy resins as a novel intumescent flame retardant system, Mater. Chem. Phys., 2014, 143, 1243–1252 CrossRef CAS.
- S. Liu, Z. Fang, H. Yan and H. Wang, Superior flame retardancy of epoxy resin by the combined addition of graphene nanosheets and DOPO, RSC Adv., 2016, 6, 5288–5295 RSC.
- S. Chen, J. Zhang, J. Zhou, D. Zhang and A. Zhang, Dramatic toughness enhancement of benzoxazine/epoxy thermosets with a novel hyperbranched polymeric ionic liquid, Chem. Eng. J., 2018, 334, 1371–1382 CrossRef CAS.
- M. Ciesielski, A. Schäfer and M. Döring, Novel efficient DOPO-based flame-retardants for PWB relevant epoxy resins with high glass transition temperatures, Polym. Adv. Technol., 2008, 19(6), 507–515 CrossRef CAS.
- H. Cheng, J. Guo, T. Zhao, Y. Ye, B. Yang, J. Cui and Y. Zhou, Molecular design and properties of a P–N synergistic flame retardant epoxy resin curing agent, J. Vinyl Addit. Technol., 2024, 300, 383–397 CrossRef.
- D. Liu, F. Jiang, T. Zhang, C. Yu, Z. Hu, L. Zhang and S. Shi, A DOPO-based reactive flame retardant containing benzimidazole groups: Synthesis and its flame-retardation on epoxy resin, J. Appl. Polym. Sci., 2024, 141(5), e54890 CrossRef CAS.
- H. Duan, Y. Chen, S. Ji, R. Hu and H. Ma, A novel phosphorus/nitrogen-containing polycarboxylic acid endowing epoxy resin with excellent flame retardance and mechanical properties, Chem. Eng. J., 2019, 375, 121916 CrossRef CAS.
- S. Wang, S. Lou, P. Fan, L. Ma, J. Liu and T. Tang, A novel aromatic imine-containing DOPO-based reactive flame retardant towards enhanced flame-retardant and mechanical properties of epoxy resin, Polym. Degrad. Stab., 2023, 213, 110364 CrossRef CAS.
- Y. Shi, B. Yu, Y. Zheng, J. Yang, Z. Duan and Y. Hu, Design of reduced graphene oxide decorated with DOPO-phosphanomidate for enhanced fire safety of epoxy resin, J. Colloid Interface Sci., 2018, 521, 160–171 CrossRef CAS.
- G. Jiang, Y. Xiao, Z. Qian, Y. Yang, P. Jia, L. Song, Y. Hu, C. Ma and Z. Gui, A novel phosphorus-, nitrogen-and sulfur-containing macromolecule flame retardant for constructing high-performance epoxy resin composites, Chem. Eng. J., 2023, 451, 137823 CrossRef CAS.
- P. Wang, L. Chen, H. Xiao and T. Zhan, Nitrogen/sulfur-containing DOPO based oligomer for highly efficient flame-retardant epoxy resin, Polym. Degrad. Stab., 2020, 171, 109023 CrossRef CAS.
- J. Qin, W. Zhang and R. Yang, Synthesis of novel phosphonium bromide-montmorillonite nanocompound and its performance in flame retardancy and dielectric properties of epoxy resins, Polym. Compos., 2021, 42, 362–374 CrossRef CAS.
- B. A. Howell and Y. G. Daniel, The impact of sulfur oxidation level on flame retardancy, J. Fire Sci., 2018, 36, 518–534 CrossRef CAS.
- A. Barth, The infrared absorption of amino acid side chains, Prog. Biophys. Mol. Biol., 2000, 74, 141–173 CrossRef CAS PubMed.
- Z. Sun, Y. Hou, Y. Hu and W. Hu, Effect of additive phosphorus-nitrogen containing flame retardant on char formation and flame retardancy of epoxy resin, Mater. Chem. Phys., 2018, 214, 154–164 CrossRef CAS.
- Y.-J. Xu, J. Wang, Y. Tan, M. Qi, L. Chen and Y.-Z. Wang, A novel and feasible approach for one-pack flame-retardant epoxy resin with long pot life and fast curing, Chem. Eng. J., 2018, 337, 30–39 CrossRef CAS.
- L. Chen, L. Song, P. Lv, G. Jie, Q. Tai, W. Xing. and Y. Hu, A new intumescent flame retardant containing phosphorus and nitrogen: Preparation, thermal properties and application to UV curable coating, Prog. Org. Coat., 2011, 70, 59–66 CrossRef CAS.
- S. Huo, Z. Zhou, J. Jiang, T. Sai, S. Ran, Z. Fang, P. Song and H. Wang, Flame-retardant, transparent, mechanically-strong and tough epoxy resin enabled by high-efficiency multifunctional boron-based polyphosphonamide, Chem. Eng. J., 2022, 427, 131578 CrossRef CAS.
- T. Zheng, W. Xia and J. Guo, Preparation of flame-retardant polyamide 6 by incorporating MgO combined with g-C3N4, Polym. Adv. Technol., 2020, 31, 1963–1971 CrossRef CAS.
- Z. Hou, C. Li, H. Wang, B. Li and H. Cai, The P/Si synergistic effect enduing epoxy resin with improved flame retardancy and outstanding mechanical properties, High Perform. Polym., 2022, 34, 545–559 CrossRef CAS.
- Z. B. Shao, M. X. Zhang, Y. Li, Y. Han, L. Ren and C. Deng, A novel multi-functional polymeric curing agent: synthesis, characterization, and its epoxy resin with simultaneous excellent flame retardance and transparency, Chem. Eng. J., 2018, 345, 471–482 CrossRef CAS.
- R. Umer, Y. Li, Y. Dong, H. Haroosh and K. Liao, The effect of graphene oxide (GO) nanoparticles on the processing of epoxy/glass fiber composites using resin infusion, Int. J. Adv. Manuf. Technol., 2015, 81, 2183–2192 CrossRef.
- J. Liang, J. Liu, L. Guo, W. Wang, C. Wang, W. Gao, X. Guo, Y. He, G. Yang, S. Yasuda, B. Liang and N. Tsubaki, CO2 hydrogenation over Fe–Co bimetallic catalysts with tunable selectivity through a graphene fencing approach, Nat. Commun., 2024, 15, 512 CrossRef CAS PubMed.
- S. D. Jiang, Z.-M. Bai, G. Tang, L. Song, A. A. Stec, T. R. Hull, J. Zhan and Y. Hu, Fabrication of Ce-doped MnO2 decorated graphene sheets for fire safety applications of epoxy composites: flame retardancy, smoke suppression and mechanism, J. Mater. Chem. A, 2014, 2, 17341–17351 RSC.
- Y. Guo, C. Bao, L. Song, B. Yuan and Y. Hu, In situ polymerization of graphene, graphite oxide, and functionalized graphite oxide into epoxy resin and comparison study of on-the-flame behavior, Ind. Eng. Chem. Res., 2011, 50, 7772–7783 CrossRef CAS.
- F. Ai, Y. Zhong, X. Hu and X. Yan, Characterization on the exfoliation degree of graphite oxide into graphene oxide by UV-visible spectroscopy, J. Wuhan Univ. Technol., 2016, 31, 515–518 CrossRef CAS.
- J.-E. An and Y. G. Jeong, Structure and electric heating performance of graphene/epoxy composite films, Eur. Polym. J., 2013, 49, 1322–1330 CrossRef CAS.
- W.-H. Xu, S.-J. Yan and J.-Q. Zhao, High performance flame-retardant organic–inorganic hybrid epoxy composites with POSS and DOPO-based co-curing agent, RSC Adv., 2022, 12, 8559–8568 RSC.
- S. K. Tiwari, A. Huczko, R. Oraon, A. De Adhikari and G. Nayak, A time efficient reduction strategy for bulk production of reduced graphene oxide using selenium powder as a reducing agent, J. Mater. Sci., 2016, 51, 6156–6165 CrossRef CAS.
- G. You, Z. Cheng, H. Peng and H. He, The synthesis and characterization of a novel phosphorus–nitrogen containing flame retardant and its application in epoxy resins, J. Appl. Polym. Sci., 2014, 131, 1–10 Search PubMed.
- H. Luo, W. Rao, P. Zhao, L. Wang, Y. Liu and C. Yu, An efficient organic/inorganic phosphorus–nitrogen–silicon flame retardant towards low-flammability epoxy resin, Polym. Degrad. Stab., 2020, 178, 109195 CrossRef CAS.
- Y. Zheng, S. Li, Z. Weng and C. Gao, Hyperbranched polymers: advances from synthesis to applications, Chem. Soc. Rev., 2015, 44, 4091–4130 RSC.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4tc03681f |
| ‡ These authors contributed equally: Jinzhuo Zhang, Jiaming Liang. |
| This journal is © The Royal Society of Chemistry 2025 |