Synthesis of π-extended CinNapht fluorophores via late-stage Pd-catalyzed C–N and C–C bond formation and application for selective imaging of lipid droplets in living cells
Eléonore
Tacke
 ,
Philippe
Durand
,
Philippe
Durand
 and
Arnaud
Chevalier
and
Arnaud
Chevalier
 *
*
Université Paris-Saclay, CNRS, Institut de Chimie des Substances Naturelles, UPR 2301, 91198 Gif-sur-Yvette, France. E-mail: arnaud.chevalier@cnrs.fr
First published on 16th September 2025
Abstract
We report the efficient synthesis of a 4′-brominated CinNapht scaffold, enabling late-stage functionalization via palladium-catalyzed cross-coupling reactions. This strategy granted access to 17 novel derivatives, including the first N-acetyl, N-carbamoyl, and N-aryl analogues, as well as entirely new C–C bonded structures via Suzuki–Miyaura, Heck, and Sonogashira couplings. These unprecedented modifications expand the π-conjugated system and tune the emission properties. Two of these fluorophores were successfully applied as selective probes for lipid droplet imaging in live cells, demonstrating the potential of this approach for biological sensing.
Introduction
For decades, the organic synthesis of fluorophores has driven the expansion of their applications across various fields. However, their preparation often relies on labor-intensive, multi-step synthetic routes that hinder the widespread development of more sophisticated compounds. To address these challenges, late-stage functionalization methods have emerged as powerful alternatives, enabling the facile introduction of molecular diversity and the fine-tuning of photophysical properties in a more streamlined manner. Among these strategies, palladium-catalyzed cross-coupling reactions have been the most extensively explored. In 2010, Luke Lavis's group made a landmark contribution in this field using Buchwald–Hartwig coupling on a fluorescein di-triflate scaffold to introduce a broad range of amines, thereby expanding the library of rhodamine dyes.1 Since then, this approach has been successfully applied to various fluorophore families,2–4 aiming either at biological applications5–8 or photophysical properties optimizations.9–12 More recently, aromatic nucleophilic substitution (SNAr) has been proposed as an alternative late-stage functionalization strategy.13–15 However, this method remains more constrained, primarily allowing the formation of C–N or C–S bonds. Building upon these advances, our team has developed a family of red-emitting hybrid Naphthalimide/Cinnolines fluorophores that exhibit significant Stokes shifts.16,17 Our extensive work on CinNapht fluorophores has demonstrated their strong potential, particularly for fluorescence microscopy. We have employed late-stage functionalization strategies to introduce amine functionalities, either selectively at the 4′ position18 or simultaneously at both the 4′ and 5′ positions.19 However, the scope of this approach has remained limited to the incorporation of aliphatic and aromatic amines, underscoring the need for further methodological advancements to expand the chemical space accessible for CinNapht derivatives (Fig. 1). | ||
| Fig. 1 Interest of Pd-catalyzed late-stage cross-coupling functionalization on CinNapht structural diversification. | ||
To date, no examples of late-stage functionalization of CinNaphts using palladium-catalyzed couplings have been reported in the literature. This study presents an efficient, scalable synthesis of a 4′-brominated CinNapht chromophore, which demonstrates long-term stability under various storage conditions, making it readily available for further applications. It was successfully engaged in various of Pd-catalyzed transformations, enabling both C–C and C–N bond formation, thereby demonstrating the versatility of this late-stage functionalization strategy synthesizing structurally diverse CinNapht derivatives. In this study, we investigate the photophysical characteristics of these newly developed π-extended analogues and present a first demonstration of their potential utility by demonstrating their suitability as fluorescent probes for selective visualization of lipid droplets in living cells.
Results and discussions
Synthesis of brominated CinNapht and use in palladium catalysed coupling reactions
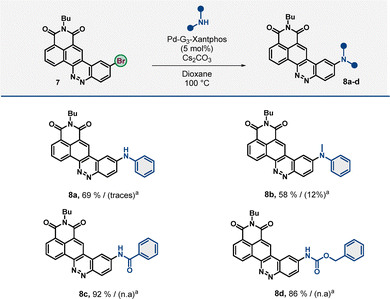
To enable this, we designed a synthetic route for the selective bromination of CinNapht at the 4′-position (Scheme 1). Initially, ortho-iodination of aniline 1 followed by a reaction with n-butylamine in refluxing ethanol afforded ortho-iodinated naphthalimide 3 with 93% yield. In parallel, we successfully carried out the bromination of 2-aminophenylboronic acid pinacol ester 4 with NBS at 0 °C in DMF, obtaining the para-brominated derivative 5 in 89% yield. We then leveraged the higher reactivity of iodine over the bromine atom in the Suzuki reaction to efficiently obtain the bis-aryl adduct 6 in 90% yield. A subsequent oxidation step of this dianiline with tert-butyl nitrite, followed by an intramolecular Mills reaction, successfully yielded the brominated CinNapht 7 in 77% yield.This five-step synthesis can be performed on a gram scale, providing a stable brominated CinNapht 7 that can be stored for extended periods without special storage conditions. As a result, this chromophore can be readily utilized in subsequent palladium-catalyzed cross-coupling reactions for the preparation of multiple new chromophores. To further explore the versatility of this brominated CinNapht, we first examined its potential in Buchwald–Hartwig amination reactions (Scheme 2).
We achieved promising results using Pd-G3-Xantphos, obtaining derivatives 8a–d in satisfactory yields ranging from 58% to 92%. Notably, access to compound 8b, which previously yielded only 12% via SNAr, showed a significant improvement to 58% under these conditions. More importantly, this strategy unlocked access to N-phenyl CinNapht (8a) as well as N-acylated CinNapht (8c) and N-carbamoyl CinNapht (8d) derivatives, with remarkable yields of 69%, 92% and 86%, respectively. These results highlight the effectiveness of this approach in overcoming the key limitations of the SNAr strategy,18 reinforcing its potential as a powerful tool for expanding the structural diversity of CinNapht derivatives. These initial results prompted us to explore the functionalization of the 4′-position via C–C bond formation, a transformation previously unreported for this scaffold. Several Pd-catalyzed coupling strategies could be employed using CinNapht 7. We started by implementing the Suzuki coupling reaction, leveraging its efficiency and versatility to introduce diverse aryl groups onto the CinNapht core.
Compound 7 was reacted with a selection aryl pinacolboronates (Scheme 3). After testing several catalytic systems (see Table S1), the best results were achieved using palladium tetrakis Pd(PPh3)4, leading to the efficient synthesis of aryl derivatives 9a–e with excellent yields ranging from 68% (9e) to 99% (9a). Notably, the incorporation of a triphenylamine unit resulted in CinNapht 9c with a remarkable 92% yield, demonstrating the efficiency of this approach. Furthermore, we successfully coupled CinNapht 7 with a tetraphenylethylene (TPE) unit via C–C bond formation (aryl–aryl), yielding a novel CinNapht-TPE hybrid 9e compound in 68%. We then explored the synthesis of styryl derivatives of CinNaphts using chromophore 7 as a key substrate for Heck reactions (Scheme 4). After optimizing the reaction conditions (Table S2), palladium acetate combined with tri-ortho-tolylphosphine emerged as the most effective catalytic system here. This strategy enabled the synthesis of five styryl derivatives with yields ranging from 45% to 74%. We successfully generated a CinNapht-π-TPE hybrid analogue (10d) via ethylene linkage, achieving an impressive 74% yield.
 | ||
| Scheme 3 Use of CinNapht 7 for Suzuki coupling reaction to synthesise of 4′-aryl substituted CinNaphts dyes 9a–e. | ||
 | ||
| Scheme 4 Use of CinNapht 7 for Heck cross-coupling reactions: synthesis of 4′-Stiryl substituted CinNaphts dyes 10a–e. | ||
Expanding on this π-conjugation approach, we introduced additional olefin partners to construct A–π–D type fluorophores, where CinNapht serves as the electron acceptor. This led to the synthesis of dimethylaniline (10b) and methoxyphenyl (10c) derivatives with 62% and 45% yield respectively. Finally, the incorporation of a 7-diethyl-aminocoumarin (DEAC) unit yielded the CinNapht-π-DEAC hybrid fluorophore (10e), marking the first reported example of a Coumarin/CinNapht hybrid in the literature. Finally, we explored the extension of the conjugated π-system by introducing an acetylenic function. For this, compound 7 was subjected to Sonogashira cross-coupling with various substrates (Scheme 5). This approach led to synthesizing of three additional fluorophores, including the model compound phenylacetylene (11a), obtained in an excellent 95% yield. Furthermore, we applied this strategy to the development of an A–π–D fluorophore (11b), analogous to compound 10b, where the ethylene linker was replaced by an acetylenic one.
 | ||
| Scheme 5 Use of CinNapht 7 for Sonogashira cross-coupling reactions: synthesis of 4′-Stiryl substituted CinNaphts dyes 11a–c. | ||
Finally, we successfully synthesized a novel CinNapht-TPE hybrid fluorophore (11c) with an excellent 87% yield. In total, this strategy enabled the one-step synthesis of 17 original CinNapht-derived chromophores from brominated precursor 7.
Evaluation of photophysical properties
To evaluate the relevance of this approach from a photophysical perspective, we conducted a comprehensive study of the fluorescence properties of the compounds described in this work. Due to their limited solubility in polar solvents, all photophysical characterizations were performed in toluene ensuring adequate compounds solubilisation. Absorption, excitation, and emission spectra were recorded at 25 °C, allowing determination of the absorption and emission maxima (λmax Em and λmax Abs), molar extinction coefficient (εmax) and the fluorescence quantum yields (QYFl) as summarized in Table 1 (All data are accessible in Fig. S3 and S4). Among the synthesized CinNapht derivatives, only those bearing electron-donating amino substituents exhibited fluorescence. In contrast, compounds lacking such groups showed negligible or no emission, consistent with the absence of intramolecular charge transfer (ICT). Interestingly, although CinNapht-TPE compounds 9e, 10d, and 11c were non-emissive in solution, they demonstrated an orange emission in the solid state (Fig. S5). Although not thoroughly investigated here, this phenomenon opens the door to further studies focusing on their solid-state photophysical behavior. No significant emission was observed in solution for the N-acylated compounds 8c and 8d. The lack of fluorescence is most likely due to the suppression of internal charge transfer (ICT) caused by acylation of the nitrogen atom. On the other hand, fluorescent properties in solution were measurable for compounds 8a, 8b, 9b, 9c, 9d, 10b, 10e, and 11b. The non-extended derivatives 8a and 8b exhibit photophysical properties typical of the CinNapht family, with absorption maxima centered around 470 nm and emission in the green-yellow region in toluene. Their fluorescence quantum yields (QYFl = 0.05 and 0.08, respectively) are comparable to the reference CinNapht-diMe fluorophore. In contrast, extension of the π-conjugated system via C–C bond formation and introduction of multiple π-links leads to markedly different properties. While absorption maxima remain largely unaffected, emission wavelengths are significantly red-shifted, consistently exceeding 600 nm.| Dye | λ max Absa (nm) | ε max (M−1 cm−1) | λ max Em (nm) | Stokes shift (cm−1) | QYFlc | ||
|---|---|---|---|---|---|---|---|
| a Values corresponding to the S0–S1 transition, (see Fig. S3). b Molar extinction coefficient related to the S0–S1 transition (see Fig. S4). c Relative fluorescence QY determined at 25 °C using 4-(dicyanomethylene)-2-methyl-6-(4-dimethylaminostyryl)-4H-pyran, “DCM” (QY = 0.43 in EtOH).20 d Data reproduced from previous study.17 | |||||||
| CinNapht-Br | 7 | 364 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
— | — | — | |
|
–NHPh | 8a | 460 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
547 | 3458 | 0.05 |
| –NMePh | 8b | 471 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
582 | 4049 | 0.08 | |
| –NH(CO)Ph | 8c | 382 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
— | — | — | |
| –NH(CO)OPh | 8d | 384 | 8500 | — | — | — | |
|
–H | 9a | 375 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
— | — | — |
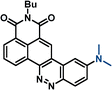
| –NMe2 | 9b | 471 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
642 | 5655 | 0.47 | |
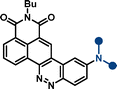
| –NPh2 | 9c | 468 | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
614 | 5081 | 0.36 | |
| –N(p-OMePh)2 | 9d | 493 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 500 |
683 | 5643 | <0.01 | |
| –TPE | 9e | 404 | 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 300 300 |
— | — | — | |

|
–H | 10a | 404 | 18![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
— | — | — |
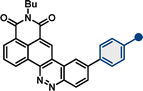
| –NMe2 | 10b | 493 | 11![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 600 600 |
673 | 5425 | 0.50 | |
| –OMe | 10c | 427 | 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
— | — | — | |
| –TPE | 10d | 404 | 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 800 |
— | — | — | |
| –DEAC | 10e | 486 | 27![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 700 700 |
619 | 4421 | 0.55 | |
|
–H | 11a | 385 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 200 200 |
— | — | — |
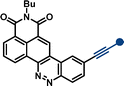
| –NMe2 | 11b | 469 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 900 900 |
636 | 5599 | 0.45 | |
| –TPE | 11c | 406 | 19![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 400 400 |
— | — | — | |
|
–CH3 | CinNapht-diMe | 480d | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000d 000d |
550d | 2652d | 0.09d |
In particular, compounds 9b, 10b, and 11b bearing a dimethylamino auxochromic group display far-red fluorescence, with emission maxima in toluene at 642 nm, 673 nm, and 636 nm, respectively, representing bathochromic shifts of approximately +100 nm compared to the extended reference compound CinNapht-diMe. As a result, these fluorophores also show significant Stokes shifts exceeding 5000 cm−1 while maintaining high fluorescence quantum yields of 0.47, 0.50, and 0.45, respectively. The favourable near-infrared emission characteristics motivated further evaluation of these compounds in fluorescence imaging by confocal microscopy.
Imaging lipid droplets in living cells using π-extended CinNapht fluorophores
Prior to the microscopy experiments, a preliminary solvatochromic study was performed on compounds 9b, 10b, and 11b (Fig. 2a). Note that we observed protonation of the cinnoline moiety leading to fluorescence quenching (Fig. S8). Unfortunately, due to both weak solubility and the lack of emission in aqueous media, we could not determine the pKa of the protonated CinNaphts with precision. Nevertheless, our data suggest that quenching protonation occurs only at very low pH values (likely below ∼5, as observed with other cinnoline-based dyes),21 thereby ensuring stable fluorescence across the physiological pH range. Unlike CinNapht-diMe, these extended structures are highly sensitive to solvent polarity, with a complete quenching of fluorescence observed in solvents with ET30 values exceeding 36 kcal mol−1 (Fig. 2b and Fig. S6). Such high selectivity for lipophilic environment make these fluorophores well suited for selective imaging of lipophilic cellular compartments, particularly lipid droplets (LDs).22,23 To evaluate their potential, we investigated the behavior of compounds 9b, 10b, and 11b in live A549 cells.Unfortunately, CinNapht 9b exhibited insufficient solubility in culture medium, preventing its use in cell imaging experiments. Only compounds 10b and 11b demonstrated adequate solubility and could be successfully employed for this study. The effect of Cinnapht 10b and 11b on cell viability was evaluated. To this end, the corresponding dyes were incubated with A549 cells over an increasing concentration range from 0.5 μM to 2 μM for 48 h, and cell viability was quantified using a Celltiter-Glo® assay (Fig. S9). The results revealed no effect at the tested concentrations, supporting their suitability for microscopy experiments. CinNapht 10b and 11b were thus incubated at 1 μM in living A549 cells and subsequently imaged by confocal microscopy. Initial observations revealed a green fluorescence localized in circular vesicles strongly morphologically similar to lipid droplets. The emission spectra of compounds 10b and 11b, recorded by confocal microscopy within these vesicles, exhibited maxima at approximately 570 nm and 520 nm, respectively, thereby confirming green emission. Furthermore, the overlay of the emission spectra obtained in various solvents with those recorded in the vesicles showed a strong correspondence with the properties measured in cyclohexane, thus confirming the highly apolar nature of the observed vesicles. Additional photophysical measurements in viscous media did not provide a better correlation (Fig. S10). We therefore assume that the selectivity of the signal observed in lipid droplets arises primarily from the polarity of the medium rather than its viscosity. To further validate that these vesicles were indeed lipid droplets, a colocalization study was performed. As shown in Fig. 2c, a strong correlation was observed between the green fluorescence of CinNaphts 10b and 11b and the red fluorescence of the commercial lipid droplet marker (LipidSpot 610), yielding a yellow signal in the merged image. These findings were corroborated both by the high Pearson's correlation coefficients (PCC) of 0.79 and 0.81 respectively, and by plot profile analyses demonstrating a clear overlap of the two fluorophore signals. Altogether, these results confirm the accumulation of CinNaphts 10b and 11b within lipid droplets. This first proof-of-concept application highlights the practical utility of π-system extension in CinNapht fluorophores via C–C bond formation, enabled by this novel late-stage palladium-catalyzed functionalization strategy.
Conclusions
In conclusion, we report the synthesis of the first 4′-bromo-CinNapht via an efficient and scalable synthetic route. We demonstrated the versatility of this chromophore in various palladium-catalyzed cross-coupling reactions, including Buchwald–Hartwig, Suzuki–Miyaura, Sonogashira, and Heck couplings. This strategy enabled access to previously unattainable N-acetyl and N-carbamate derivatives. Moreover, we leveraged the Buchwald conditions' efficiency to improve yields significantly for N-arylated compounds. More importantly, this methodology provides a powerful tool for π-extension via C–C bond formation, unlocking a library of novel CinNapht-based fluorophores. These new structures exhibit far-red emission in nonpolar solvents and show remarkable selectivity toward apolar environments. This property was successfully exploited for live-cell imaging of lipid droplets, marking a first application of late-stage C–C bond functionalization in this context. We believe this synthetic approach holds great promise for the future development of diverse CinNapht derivatives tailored to a wide range of applications.Author contributions
Chemical synthesis and characterization of undescribed compounds as well as photophysical studies were performed by E. T. Cell culture and confocal microscopy experiments were performed by A. C. A. C. supervised the project and wrote the manuscript, with proof reading and suggestion given by E. T. and P. D. All authors have given corrections and approval to the final version of the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: absorbance, fluorescence, NMR and MS spectra. See DOI: https://doi.org/10.1039/d5tb01877c.Acknowledgements
This project has received funding by the French National Research Agency under the program CHARMMMAT ANR-11-LABX-0039-grant. We thank Romain Le Bars and Sandrine Lecart from Imagerie Gif light facility for their kind help in microscopy experiments. The present work has benefited from Imagerie-Gif light microscopy core facility supported by l’Agence Nationale de la Recherche (ANR-11-EQPX-0029/Morphoscope, ANR-10-INBS-04/FranceBioImaging; ANR-11-IDEX-0003-02/Saclay Plant Sciences). The University Paris-Saclay and the CNRS are acknowledged for funding.Notes and references
- J. B. Grimm and L. D. Lavis, Org. Lett., 2011, 13, 6354–6357 CrossRef CAS PubMed.
- T. Peng and D. Yang, Org. Lett., 2010, 12, 496–499 CrossRef CAS PubMed.
- K. N. Hearn, T. D. Nalder, R. P. Cox, H. D. Maynard, T. D. M. Bell, F. M. Pfeffer and T. D. Ashton, Chem. Commun., 2017, 53, 12298–12301 RSC.
- J. B. Grimm, A. J. Sung, W. R. Legant, P. Hulamm, S. M. Matlosz, E. Betzig and L. D. Lavis, ACS Chem. Biol., 2013, 8, 1303–1310 CrossRef CAS PubMed.
- T. Takagi, T. Ueno, K. Ikawa, D. Asanuma, Y. Nomura, S.-N. Uno, T. Komatsu, M. Kamiya, K. Hanaoka, C. Okimura, Y. Iwadate, K. Hirose, T. Nagano, K. Sugimura and Y. Urano, Sci. Adv., 2021, 7, eabg8585 CrossRef CAS PubMed.
- J. M. Meinig, L. Fu and B. R. Peterson, Angew. Chem., Int. Ed., 2015, 54, 9696–9699 CrossRef CAS PubMed.
- C. Deo, A. S. Abdelfattah, H. K. Bhargava, A. J. Berro, N. Falco, H. Farrants, B. Moeyaert, M. Chupanova, L. D. Lavis and E. R. Schreiter, Nat. Chem. Biol., 2021, 17, 718–723 CrossRef CAS PubMed.
- J. Bucevičius, G. Kostiuk, R. Gerasimaitė, T. Gilat and G. Lukinavičius, Chem. Sci., 2020, 11, 7313–7323 RSC.
- J. B. Grimm, L. Xie, J. C. Casler, R. Patel, A. N. Tkachuk, N. Falco, H. Choi, J. Lippincott-Schwartz, T. A. Brown, B. S. Glick, Z. Liu and L. D. Lavis, JACS Au, 2021, 1, 690–696 CrossRef CAS PubMed.
- J. B. Grimm, A. K. Muthusamy, Y. Liang, T. A. Brown, W. C. Lemon, R. Patel, R. Lu, J. J. Macklin, P. J. Keller, N. Ji and L. D. Lavis, Nat. Methods, 2017, 14, 987–994 CrossRef CAS PubMed.
- A. N. Butkevich, M. L. Bossi, G. Lukinavičius and S. W. Hell, J. Am. Chem. Soc., 2019, 141, 981–989 CrossRef CAS PubMed.
- G. Zhang, M. Wang, P. Bobadova-Parvanova, F. R. Fronczek, K. M. Smith and M. G. H. Vicente, Chem. – Eur. J., 2022, 28, e202200421 CrossRef CAS PubMed.
- G. Vives, C. Giansante, R. Bofinger, G. Raffy, A. D. Guerzo, B. Kauffmann, P. Batat, G. Jonusauskas and N. D. McClenaghan, Chem. Commun., 2011, 47, 10425–10427 RSC.
- W. Shi, J. Li, X. He, S. Zhou, H. Sun and H. Wu, Org. Lett., 2022, 24, 3368–3372 CrossRef CAS PubMed.
- A. Chevalier, K. Renault, F. Boschetti, P.-Y. Renard and A. Romieu, Eur. J. Org. Chem., 2015, 152–165 CrossRef CAS.
- M.-D. Hoang, F. Savina, P. Durand, P. R. Meallet-Renault, G. Clavier and A. Chevalier, ChemPhotoChem, 2022, 6, e202200138 CrossRef CAS.
- M.-D. Hoang, J.-B. Bodin, F. Savina, V. Steinmetz, J. Bignon, P. Durand, G. Clavier, R. Méallet-Renault and A. Chevalier, RSC Adv., 2021, 11, 30088–30092 RSC.
- E. Tacke, M.-D. Hoang, K. Tatoueix, B. Keromnes, E. Van Eslande, P. Durand, G. Pieters and A. Chevalier, Chem. Sci., 2023, 14, 6000–6010 RSC.
- E. Tacke, L. Estaque, M.-D. Hoang, P. Durand, G. Clavier, G. Pieters and A. Chevalier, Chem. – Eur. J., 2025, 31, e202403684 CrossRef CAS PubMed.
- K. Rurack and M. Spieles, Anal. Chem., 2011, 83, 1232–1242 CrossRef CAS PubMed.
- M. Coehlo, G. Clavier, R. Méallet, G. Pieters and A. Chevalier, Chem. Commun., 2025, 61, 12785–12788 RSC.
- L. Michel, P. Durand and A. Chevalier, ChemBioChem, 2024, 25(19), e202400270 CrossRef CAS PubMed.
- N. Aknine and A. S. Klymchenko, Anal. Chem., 2024, 96(32), 13242–13251 CrossRef CAS PubMed.
| This journal is © The Royal Society of Chemistry 2025 |



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/i_char_2009.gif)
