3D printable collagen-like protein hydrogels via dynamic covalent assembly for soft tissue engineering
Mercyjayapriya
Jebakumar†
 abc,
Mohandass
Pachaiyappan†
abc,
Mohandass
Pachaiyappan†
 ab,
Chayla L.
Reeves
ab,
Chayla L.
Reeves
 c,
Kate
Fox
c,
Kate
Fox
 d,
Amy
Gelmi
d,
Amy
Gelmi
 *c and
Niraikulam
Ayyadurai
*c and
Niraikulam
Ayyadurai
 *ab
*ab
aDepartment of Biochemistry and Biotechnology, Council of Scientific and Industrial Research (CSIR) Central Leather Research Institute (CLRI), Chennai, Tamil Nadu 600020, India
bAcademy of Scientific and Innovative Research (AcSIR), Ghaziabad 201002, India. E-mail: ayyadurai@clri.res.in
cSchool of Science, RMIT University, VIC 3000, Australia. E-mail: amy.gelmi@rmit.edu.au
dSchool of Engineering, RMIT University, VIC 3000, Australia
First published on 21st November 2025
Abstract
This study presents a novel 3D collagen-like protein/aldehyde-functionalized dextran (CLP–AD) hydrogel designed for tissue engineering. Developed under physiological conditions, the hydrogel forms without harsh crosslinkers or external triggers, relying on Schiff base reactions between aldehyde groups of oxidized dextran and amino groups of recombinant collagen-like protein. This dynamic covalent bonding enables a sol-to-gel transition ideal for 3D bioprinting. Comprehensive characterization confirmed its favorable gelation, swelling, degradation, and cytocompatibility profiles. Rheological analysis revealed viscoelastic and self-healing properties, making it suitable for dynamic tissue environments. Co-culture with human endothelial cells demonstrated high cell viability and migration, comparable to collagen hydrogels (RC-AD). Evaluation as a cell adhesion substrate for umbilical cord-derived mesenchymal stem cells (UC-hMSCs) revealed less spreading and fewer focal adhesions compared to the stiffer tissue culture polystyrene (TCPS) substrate, observed through immunostaining. Cell encapsulation studies with UC-hMSCs demonstrated high cell viability within the 3D matrix, supporting the hydrogel's suitability for cell-laden bioprinting applications. These findings suggest that the CLP–AD hydrogel maintains a morphology conducive to soft tissue engineering applications. This CLP overcomes the limitations associated with animal-derived collagen, such as risk of disease transmission and batch-to-batch variability. Overall, this study presents a straightforward approach for fabricating tunable and 3D-printable hydrogels, highlighting their potential for developing materials for soft tissue repair and regeneration.
1. Introduction
Protein-based hydrogels have garnered considerable attention in tissue engineering and regenerative medicine due to their ability to closely replicate the natural extracellular matrix (ECM) environment, thereby supporting critical cellular functions such as proliferation, nutrient diffusion, migration, and tissue remodeling.1–3 Among these, collagen, the most abundant structural protein in the ECM, has been extensively employed to create hydrogels for biomedical applications. However, collagen derived from animal sources is associated with several drawbacks, including batch-to-batch variability, potential immunogenic responses, the risk of zoonotic disease transmission, and ethical concerns related to animal use.4,5 In line with this, the implementation of EU Directive 2010/63/EU, which promotes the reduction of animal testing, has further accelerated the demand for non-animal-derived biomaterials for in vitro models and biomedical applications.6 These challenges highlight the need for alternative, more controlled, and safer collagen-mimetic materials.Collagen-like proteins (CLPs) have emerged as a promising class of recombinant biomaterials that structurally and functionally mimic native collagen while offering greater control over production and functionalization. Recent genomic and proteomic studies have identified over a hundred putative bacterial CLPs, and several of these, particularly those from Escherichia coli expression systems, have demonstrated the ability to form stable triple helices with melting temperatures (Tm) comparable to mammalian collagens (35–39 °C).7,8 Notably, Streptococcus pyogenes–derived Scl1 and Scl2 proteins have been widely studied.9–12 Scl2 features a distinct architecture with an N-terminal globular trimerization domain, and a collagen-like domain composed of 79 Gly-Xaa-Yaa repeats, enabling stable triple helix formation. The recombinant production of Scl2 in microbial systems offers distinct advantages, such as scalability, reproducibility, and free from animal-derived contaminants, making it an attractive platform for biomedical material development. Moreover, Scl2 proteins are inherently non-cytotoxic, non-thrombogenic, and non-immunogenic, further supporting their suitability for clinical applications.13
One of the unique characteristics of bacterial CLPs, compared to native collagens, is their inherent bioinertness, earning them the designation of “blank-state collagens.” This lack of intrinsic bioactivity presents an opportunity for precise bioengineering, allowing the incorporation of functional peptide motifs or degradation sequences to tailor biological responses. Several successful examples include the insertion of the GFOGER motif to enhance integrin-mediated cell adhesion,14,15 the incorporation of heparin-binding domains to modulate growth factor interactions, and matrix metalloproteinase (MMP)-sensitive sequences to enable enzyme-responsive degradation.16 This tunability renders CLPs a state-of-the-art collagen-mimetic platform, enabling precise molecular customization to accommodate patient-specific therapeutic needs or targeted functional applications in regenerative medicine.15 These advancements have significantly broadened the applicability of CLPs in tissue engineering and regenerative medicine. For instance, Scl2 variants were engineered with GFPGER integrin-binding motifs and photo-crosslinking sites, and their incorporation into 3D PEG hydrogels was successfully applied in multilayer vascular grafts.17 In another application, Scl2 were engineered with integrin, heparin, and hyaluronic acid binding motifs for articular cartilage tissue engineering. These modified CLPs were crosslinked into hydrogels incorporating MMP-7 and aggrecanase-cleavable peptides and encapsulated with human mesenchymal stem cells, leading to controlled degradation, enhanced cell differentiation, and promotion of neo-cartilage matrix formation.18 Similarly, Scl2 variants were modified with peptide sequences that specifically bind hyaluronic acid and chondroitin sulfate, leading to the development of collagen-mimetic foams that improved chondrogenesis of human mesenchymal stem cells (hMSCs) compared to type I collagen-based foams.19 Despite these promising features, constructing CLP-based 3D scaffolds with suitable mechanical integrity and biocompatibility remains a major hurdle. Traditional methods often rely on chemical crosslinking strategies to assemble CLPs into hydrogels. Crosslinkers such as glutaraldehyde,20 EDC/NHS,21–23 and the naturally derived genipin6 have been employed to stabilize CLP networks, improving mechanical strength and durability. However, these methods can involve harsh chemical conditions or residual toxicity, potentially compromising cell viability and limiting the clinical applicability of the resulting materials. Photo-crosslinking using photoinitiators (PIs) has emerged as an alternative approach that enables rapid and spatially controlled gelation under mild conditions. However, the free radicals generated from PIs during light exposure can cause cellular damage to membranes, proteins, and DNA, which negatively impacts cell viability24
In response to these challenges, we developed a novel 3D-printable hydrogel system by combining a recombinant collagen-like protein (CLP) with aldehyde-functionalized dextran (AD). The hydrogel is formed via a dynamic covalent Schiff base reaction between the primary amines of the CLP and the aldehyde groups of oxidized dextran under physiological conditions (pH 7.4), avoiding the need for cytotoxic chemical crosslinkers. The resulting CLP–AD hydrogel exhibits tunable viscoelasticity, self-healing capabilities, and controlled degradation, making it particularly suitable for extrusion-based bioprinting. This CLP–AD ink offers a good combination of biocompatibility, tunability and cell loading. Systematic characterization confirmed its cytocompatibility, with hydrogel extracts promoting high viability and enhanced migration of human endothelial cells. Additionally, umbilical cord-derived human mesenchymal stem cells (UC-hMSCs) cultured on the hydrogel surface showed a morphology conducive to soft tissue engineering, with pronounced filopodia and limited focal adhesion formation, suggesting a favourable microenvironment for stem cell-based regenerative applications. Furthermore, cell encapsulation studies demonstrated high UC-hMSC viability within the 3D hydrogel matrix, supporting its applicability for cell-laden bioprinting approaches. This initial assessment using UC-hMSCs represents an important first step toward establishing the CLP–AD hydrogel's suitability for stem cell viability, attachment, and tissue-regenerative potential. Overall, this study presents a simple and versatile approach to engineer safe, animal-free, and customizable protein-based hydrogels, offering significant potential for soft tissue repair and regenerative medicine applications.
2. Materials and methods
2.1 Materials
Dextran (Mr ∼ 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000), ethylene glycol, sodium periodate, dialysis tubing (MWCO: 10 kDa), Dulbecco's Modified Eagle's Medium (DMEM), penicillin–streptomycin solution, trypsin-EDTA, fetal bovine serum (FBS), fluorescein diacetate (FDA) and propidium iodide (PI), bovine serum albumin, Alexa Fluor 488 Phalloidin, DAPI, were purchased from Sigma-Aldrich, USA. CLP-pQE80L constructs were synthesized and procured from Invitrogen, USA. Nickel nitrilotriacetic acid (Ni-NTA) was acquired from Cytiva Life Sciences, USA. Isopropyl β-D-1-thiogalactopyranoside (IPTG), ampicillin, Luria-Bertani (LB) broth, acetic acid and imidazole were purchased from Hi-Media Limited, India.
000), ethylene glycol, sodium periodate, dialysis tubing (MWCO: 10 kDa), Dulbecco's Modified Eagle's Medium (DMEM), penicillin–streptomycin solution, trypsin-EDTA, fetal bovine serum (FBS), fluorescein diacetate (FDA) and propidium iodide (PI), bovine serum albumin, Alexa Fluor 488 Phalloidin, DAPI, were purchased from Sigma-Aldrich, USA. CLP-pQE80L constructs were synthesized and procured from Invitrogen, USA. Nickel nitrilotriacetic acid (Ni-NTA) was acquired from Cytiva Life Sciences, USA. Isopropyl β-D-1-thiogalactopyranoside (IPTG), ampicillin, Luria-Bertani (LB) broth, acetic acid and imidazole were purchased from Hi-Media Limited, India.
2.2 Production and purification of CLP
CLP proteins were produced and purified by following previous protocols.25 Briefly, the construct CLP-pQE80L was transformed to the expression strain of E. coli BL21 (DE3). Transformed colonies were grown in LB broth containing 100 µg mL−1 ampicillin. CLP production was induced with 1 mM IPTG when it reached the mid-log phase and incubated at 37 °C for 6 hours. The culture was centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 rpm for 10 minutes to separate the pellets. Harvested cell pellets were incubated in lysis buffer (20 mM of Tris Hcl, 500 mM of NaCl, 20 mM of Imidazole, pH 7.4) containing 2 mg mL−1 of lysozyme for 2 h at 37 °C, sonicated, and centrifuged to collect the supernatant. Ni-NTA affinity column connected to an AKTA Avant FPLC system (Cytiva, USA) was used to purify the bound protein by gradient elution with 1 M imidazole. The eluted proteins were desalted using a 10 kDa dialysis membrane with 10 mM phosphate. The purity of the protein fractions was analyzed using SDS-PAGE analysis. Desalted proteins were freeze-dried using a lyophilizer (Southern Scientific Pvt. Ltd) and further used for hydrogel preparations.
000 rpm for 10 minutes to separate the pellets. Harvested cell pellets were incubated in lysis buffer (20 mM of Tris Hcl, 500 mM of NaCl, 20 mM of Imidazole, pH 7.4) containing 2 mg mL−1 of lysozyme for 2 h at 37 °C, sonicated, and centrifuged to collect the supernatant. Ni-NTA affinity column connected to an AKTA Avant FPLC system (Cytiva, USA) was used to purify the bound protein by gradient elution with 1 M imidazole. The eluted proteins were desalted using a 10 kDa dialysis membrane with 10 mM phosphate. The purity of the protein fractions was analyzed using SDS-PAGE analysis. Desalted proteins were freeze-dried using a lyophilizer (Southern Scientific Pvt. Ltd) and further used for hydrogel preparations.
2.3 Synthesis of aldehyde functionalized dextran (AD)
The aldehyde functionalization of dextran was carried out following standard periodate method.26 Briefly, 10 g of dextran was dissolved in 100 ml of distilled water on a magnetic stirrer at room temperature. Then, 6.34 g of sodium periodate dissolved in 100 mL of distilled water was added dropwise to oxidize the dextran solution. The reaction was carried out in the dark for 6 h at room temperature. After adding 2 ml of ethylene glycol to terminate oxidation reaction, the resulting solution was purified by dialysis for 3 days against water and finally freeze-dried to obtain pure aldehyde-functionalized dextran (AD). The lyophilized sample was weighed to determine the percentage yield of AD (86 ± 4% yield). | (1) |
2.4 Fabrication of CLP–AD hydrogel
The CLP–AD hydrogel was fabricated using AD and CLP stock solutions. In brief, AD was dissolved in distilled water at a concentration of 20% (200 mg mL−1), while CLP was dissolved in phosphate-buffered saline (PBS) at a concentration of 2% (20 mg mL−1). Various CLP–AD hydrogels were formed by mixing the respective stock solutions of AD and CLP at different concentrations. The solutions were completely mixed to achieve a homogenous state and then left at room temperature for gelation. This study used a hydrogel formed from rat tail-derived collagen (RC) and AD (RC-AD) as a positive control. The critical gelation concentration (CGC) of CLP and AD time (tgel), and temperature (Tgel) required for hydrogel formation was finalized using the conventional vial inversion method. To assess the influence of buffer type and pH on the gelation properties of the hydrogel, CLP stock solutions were prepared in different buffers and across a range of pH values. The time required for gelation was subsequently monitored.| SR = (Wt − W0)/W0 | (2) |
| Weight loss (%) = [(W0 − Wt)/W0] × 100 | (3) |
 | (4) |
 | (5) |
2.5 3D printability
The 3D printability of the CLP–AD hydrogel was tested using a pneumatic extrusion-based 3D printer (NBIL-TRIVIMA, Bangalore). After printing, filament extrusion was performed to assess the hydrogel's ability to maintain shape fidelity in a 3D grid. Various 3D printing configurations were used, including single (20 G and 22 G) and coaxial printing (inner core – 20 G; outer shell – 18 G), to create different 3D designs such as grids and circles. The 3D designs were generated using CAD software before printing. A cartridge containing 3 mL of CLP–AD hydrogel was loaded, and printing was initiated. The pressure (0.8–1 bar) needed for the hydrogel to extrude as a continuous filament through the nozzle was applied during the process. This occurred at a feed rate of 400 mm s−1 for printing various geometric shapes using single and coaxial bioprinting configurations. Post-printing, the images were compared to the original CAD designs for analysis.2.6 In vitro biological study
Endothelial cells (EA. hy926) purchased from ATCC were cultured and maintained at 37 °C in a humidified atmosphere with 5% CO2 in high-glucose DMEM supplemented with 10% FBS and 1% penicillin–streptomycin until reaching 80% confluency.Umbilical cord-derived human mesenchymal stem cells (UC-hMSCs) purchased from RoosterBio, Lot no. 310357 (USA), were cultured at 37 °C in a humidified atmosphere with 5% CO2 in alpha minimal essential medium (αMEM) supplemented with 10% FBS and 1% penicillin–streptomycin (Gibco, USA) until they reached 80% confluency. Experiments were conducted using UC-hMSCs between passages 4 and 6.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5, CLP–AD 1
2.5, CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, CLP–AD 1
5, CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5, and CLP–AD 1
7.5, and CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) was evaluated using an MTT assay with EA.hy926 cells. A total of 1 × 104 cells per well were seeded into 96-well plates and maintained at 37 °C in a humidified incubator with 5% CO2 for 24 hours to allow cell adhesion. Meanwhile, 0.04 g of each CLP–AD hydrogel was immersed in 5 mL of high-glucose DMEM supplemented with 10% FBS and incubated for 24 hours at 37 °C. After this period, the hydrogels were removed from the medium to obtain the extracts. EA.hy926 cells were then treated with CLP–AD hydrogel extracts and incubated for 3 days. Untreated EA.hy926 cells were used as a control, while cells treated with RC-AD as a positive control. The MTT assay was performed daily to evaluate cell growth. After 24, 48, and 72 hours, the medium was removed from each well and replaced with media containing 1 mg mL−1 of MTT. The cells were incubated with this solution for 3 hours at 37 °C with 5% CO2. The formed formazan crystals were dissolved in DMSO, and the absorbance was measured at 570 nm using an Epoch2 microplate reader. The % of cell viability was calculated using the following formula:
10) was evaluated using an MTT assay with EA.hy926 cells. A total of 1 × 104 cells per well were seeded into 96-well plates and maintained at 37 °C in a humidified incubator with 5% CO2 for 24 hours to allow cell adhesion. Meanwhile, 0.04 g of each CLP–AD hydrogel was immersed in 5 mL of high-glucose DMEM supplemented with 10% FBS and incubated for 24 hours at 37 °C. After this period, the hydrogels were removed from the medium to obtain the extracts. EA.hy926 cells were then treated with CLP–AD hydrogel extracts and incubated for 3 days. Untreated EA.hy926 cells were used as a control, while cells treated with RC-AD as a positive control. The MTT assay was performed daily to evaluate cell growth. After 24, 48, and 72 hours, the medium was removed from each well and replaced with media containing 1 mg mL−1 of MTT. The cells were incubated with this solution for 3 hours at 37 °C with 5% CO2. The formed formazan crystals were dissolved in DMSO, and the absorbance was measured at 570 nm using an Epoch2 microplate reader. The % of cell viability was calculated using the following formula: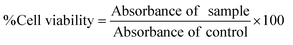 | (6) |
Cell viability was further assessed with fluorescence-based dual-color live/dead cell staining using fluorescein diacetate (green, Ex/Em = 495/515 nm) and propidium iodide (red, Ex/Em = 535/615 nm). Images of the stained cells were captured using a fluorescence microscope (Leica Microsystems DMi8, Germany).
2.7 Human mesenchymal stem cell culture
To each well of a 24-well plate, 0.1 mL of CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 solution was added. The plates were sterilized under UV light for 15 minutes and then incubated at 37 °C overnight to facilitate gelation. The formed hydrogel was thoroughly washed with PBS and preconditioned with medium for 2 hours before cell seeding. Subsequently, 3000 cells per cm2 UC-hMSCs were carefully added on top of the formed CLP–AD hydrogel. Cells seeded on RC-AD hydrogel were used as the control group. The samples were incubated at 37 °C in a 5% CO2 atmosphere, and resazurin assays were conducted at 1 and 3 days to assess cell viability on the CLP–AD hydrogel. For qualitative analysis, a fluorescence-based dual-color live/dead cell staining was performed using NucBlue™ Live ReadyProbes™ Reagent and using NucGreen™ Live ReadyProbes™ Reagent (Invitrogen, USA). Images were captured using EVOS fluorescence imaging system (Thermo Fisher Scientific, USA).
2.5 solution was added. The plates were sterilized under UV light for 15 minutes and then incubated at 37 °C overnight to facilitate gelation. The formed hydrogel was thoroughly washed with PBS and preconditioned with medium for 2 hours before cell seeding. Subsequently, 3000 cells per cm2 UC-hMSCs were carefully added on top of the formed CLP–AD hydrogel. Cells seeded on RC-AD hydrogel were used as the control group. The samples were incubated at 37 °C in a 5% CO2 atmosphere, and resazurin assays were conducted at 1 and 3 days to assess cell viability on the CLP–AD hydrogel. For qualitative analysis, a fluorescence-based dual-color live/dead cell staining was performed using NucBlue™ Live ReadyProbes™ Reagent and using NucGreen™ Live ReadyProbes™ Reagent (Invitrogen, USA). Images were captured using EVOS fluorescence imaging system (Thermo Fisher Scientific, USA).
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) solution was added. The plates were sterilized under UV light for 15 minutes and then incubated at 37 °C for 4 hours to facilitate partial gelation. After partial gelation, UC-hMSCs were seeded at a concentration of 5 × 104 cells per well, mixed gently with the semi-solid matrix, and incubated for 20 minutes at 37 °C to allow complete gelation in the presence of cells. After complete gelation, 100 µL of complete culture medium was added to each well, and the plates incubated at 37 °C in a 5% CO2 atmosphere. Cell viability was evaluated on days 1, 3, and 7 using a fluorescence-based dual-staining live/dead assay. For this, NucBlue™ Live ReadyProbes™ Reagent and NucGreen™ Dead ReadyProbes™ Reagent were added according to the manufacturer's instructions. Fluorescence images were captured using EVOS fluorescence imaging system. Cell viability was quantified by analysing the fluorescence images using Fiji (ImageJ software). For each sample, three replicates were analyzed. The total number of cells (NucBlue-stained) and the number of dead cells (NucGreen-stained) were counted from the images, and the percentage of viable cells was calculated using the following formula:
2.5) solution was added. The plates were sterilized under UV light for 15 minutes and then incubated at 37 °C for 4 hours to facilitate partial gelation. After partial gelation, UC-hMSCs were seeded at a concentration of 5 × 104 cells per well, mixed gently with the semi-solid matrix, and incubated for 20 minutes at 37 °C to allow complete gelation in the presence of cells. After complete gelation, 100 µL of complete culture medium was added to each well, and the plates incubated at 37 °C in a 5% CO2 atmosphere. Cell viability was evaluated on days 1, 3, and 7 using a fluorescence-based dual-staining live/dead assay. For this, NucBlue™ Live ReadyProbes™ Reagent and NucGreen™ Dead ReadyProbes™ Reagent were added according to the manufacturer's instructions. Fluorescence images were captured using EVOS fluorescence imaging system. Cell viability was quantified by analysing the fluorescence images using Fiji (ImageJ software). For each sample, three replicates were analyzed. The total number of cells (NucBlue-stained) and the number of dead cells (NucGreen-stained) were counted from the images, and the percentage of viable cells was calculated using the following formula: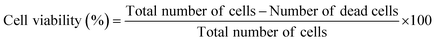 | (7) |
All Fluorescence images of F-actin, DAPI, and Vinculin channels were processed using Fiji to enhance visualization and ensure consistency across all samples. Background noise was minimized using the “Subtract Background” function for each channel. To improve local contrast and feature definition, Contrast Limited Adaptive Histogram Equalization (CLAHE) was applied uniformly across all channels. For the Vinculin channel, an additional contrast enhancement step was carried out using the “Enhance Contrast” function with 0.05% saturated pixels. This further enhancement was specifically applied to better visualize vinculin-rich regions (focal adhesion plaques). The enhanced vinculin image was used for generating merged composite images alongside F-actin and DAPI channels. For single-channel vinculin representation, only the CLAHE-processed version was used to preserve the original contrast profile for qualitative assessment.
2.8 Statistical analysis
The data, obtained from at least three independent experiments, are presented as mean ± SD. Statistical analysis was performed using one-way ANOVA followed by Duncan's test in SPSS software. Significance levels are indicated as NS (no significant difference), *p < 0.05, **p < 0.01, and ***p < 0.001.3. Results and discussion
Schiff base linkage is highly regarded in biomaterials for its capacity to create reversible and stable networks, facilitating the development of self-healing and responsive materials.28 In this study, a CLP–AD hydrogel was fabricated using this Schiff base linkage, resulting in a gel with self-healing properties and excellent printability. This approach eliminates the need for post-gelation procedures or harsh crosslinkers during preparation, underscoring the hydrogel's potential for soft tissue engineering applications, including stem cell culture.3.1 Collagen like protein
The recombinant collagen-like protein (CLP) used for hydrogel preparation was expressed in E. coli BL21[DE3] and purified using Ni-NTA affinity chromatography, as previously described.25 The purity and molecular weight (∼35 kDa) of the protein were confirmed by SDS-PAGE (Fig. S1). The average yield was ∼30 mg L−1, consistent with earlier reports. Structural characterization of the CLP protein using MALDI-TOF and circular dichroism (CD) spectroscopy has been extensively reported by our lab, and the lyophilized protein was used for hydrogel fabrication.293.2 Synthesis and characterization of aldehyde functionalized dextran
Dextran is a polysaccharide synthesized by Leuconostoc mesenteroides, primarily consisting of α-1,6-glycosidic linkages with minor branching at α-1,2-, α-1,3-, and α-1,4-positions.30 It contains vicinal diols, which, upon reaction with periodate ions, lead to C–C bond cleavage (C3–C4 or C2–C3), forming aldehyde groups that can undergo further oxidation reaction. The oxidized dextran was prepared according to the literature with little modification.26 The synthesis and characterization of the AD is shown in Fig. S2, which was then used to form a hydrogel with Collagen-like protein (CLP). Initially, we used NMR to verify the formation of the desired AD structure. Fig. S2B presents the 1H NMR spectra for both pristine dextran and oxidized dextran. Both spectra exhibit peaks corresponding to the protons in the glucose units (1, 2, 3, 4, 5, and 6). However, several additional peaks between 4.2 and 5.8 ppm were observed in the aldehyde-functionalized dextran, which are attributed to the protons from the hemiacetal groups. This confirmed that the oxidation of dextran was successful and that aldehyde groups were incorporated into the dextran molecular chains.31 FTIR analysis was also performed to confirm the presence of the aldehyde group on the functionalized dextran. Fig. S2C shows the changes in the FTIR spectra after oxidation, with an absorbance peak at around 1733 cm−1, which corresponds to the aldehyde group. The oxidation degree of AD was quantified using the iodometry method, which measures the concentration of unconsumed periodate after the oxidation reaction. Based on the average results from three oxidation experiments, the oxidation degree of dextran was determined to be 48 ± 1%, which is slightly higher than reported values.26 This difference arises because that study used hydroxylamine titration, which quantifies oxidation by measuring the formed aldehydes, whereas iodometry determines the oxidation degree by measuring the unreacted sodium periodate, assuming that all consumed periodate contributed to oxidation. A slight overestimation may occur if sodium periodate reacts with functional groups other than dextran hydroxyls.3.3 Preparation of the CLP–AD hydrogel
For the gelation test, stock solutions of 20% AD in distilled water and 20 mg mL−1 CLP in various buffers (such as 1X PBS at pH 7.4, 5 mM acetic acid, and 0.5 mM acetic acid at pH 6.5) were prepared. From these stock solutions, 10 mg mL−1 CLP and 10% AD were mixed, collectively termed as CLP/AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10. The 10% AD concentration was initially chosen to examine the effect of higher aldehyde content on crosslinking efficiency. The mixture in vials was incubated at room temperature, then inverted at predefined time points to determine gelation time under different buffer conditions. The CLP–AD mixture underwent rapid gelation, confirming hydrogel formation. Schiff base bonds are generally formed within a pH range of 4–7, are stable at pH above 7, and readily hydrolyse below pH 5.32 The possible crosslinking mechanism between the amino group of CLP and the aldehyde group of AD is illustrated in Scheme 1.
10. The 10% AD concentration was initially chosen to examine the effect of higher aldehyde content on crosslinking efficiency. The mixture in vials was incubated at room temperature, then inverted at predefined time points to determine gelation time under different buffer conditions. The CLP–AD mixture underwent rapid gelation, confirming hydrogel formation. Schiff base bonds are generally formed within a pH range of 4–7, are stable at pH above 7, and readily hydrolyse below pH 5.32 The possible crosslinking mechanism between the amino group of CLP and the aldehyde group of AD is illustrated in Scheme 1.
The Schiff base reaction is a condensation process where a primary amine (–NH2) reacts with an aldehyde or ketone (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O) to form an imine (–C
O) to form an imine (–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) NR) with the elimination of water. The mechanism involves three key steps: nucleophilic attack, where the amine attacks the carbonyl carbon to form a carbinolamine intermediate; proton transfer, which stabilizes the intermediate; and water elimination, leading to imine formation.33 In our observations, the CLP–AD 1
NR) with the elimination of water. The mechanism involves three key steps: nucleophilic attack, where the amine attacks the carbonyl carbon to form a carbinolamine intermediate; proton transfer, which stabilizes the intermediate; and water elimination, leading to imine formation.33 In our observations, the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 mixture in 5 mM and 0.5 mM acetic acid (pH 6.5) took 60 and 120 minutes, respectively, for gelation, while the CLP–AD 1
10 mixture in 5 mM and 0.5 mM acetic acid (pH 6.5) took 60 and 120 minutes, respectively, for gelation, while the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 in 1X PBS (pH 7.4) took only 40 minutes (Fig. S3). This is because, at physiological pH, the amine groups are deprotonated and highly nucleophilic, making them more reactive with aldehydes. In contrast, under acidic conditions, Schiff base formation is slower because the amino groups are protonated, reducing their reactivity. However, hydrogel formation still occurs, possibly because the acetic acid improves the solubility of CLP. Despite this, gelation was 33.33% faster in the physiological condition (pH 7.4) compared to the 5 mM acetic acid condition, and 66.67% faster compared to the 0.5 mM acetic acid condition. To maintain positive control, collagen derived from rat tails was dissolved in 5 mM acetic acid at a concentration of 10 mg mL−1 and mixed with 10% AD (RC-AD) to check the gelation time. The gel formed within 10 minutes. However, the CLP–AD hydrogel can be prepared directly in 1X PBS, whereas rat tail collagen requires dissolution in acetic acid followed by a neutralization step with sodium hydroxide, as reported in previous studies.31 The preparation process of CLP–AD allows direct use of 1X PBS, eliminating the need for acetic acid and subsequent neutralization with sodium hydroxide. Overall, the CLP-based gel provides a more efficient, safer, and reliable alternative for hydrogel preparation, making it a preferable choice in tissue engineering applications.
10 in 1X PBS (pH 7.4) took only 40 minutes (Fig. S3). This is because, at physiological pH, the amine groups are deprotonated and highly nucleophilic, making them more reactive with aldehydes. In contrast, under acidic conditions, Schiff base formation is slower because the amino groups are protonated, reducing their reactivity. However, hydrogel formation still occurs, possibly because the acetic acid improves the solubility of CLP. Despite this, gelation was 33.33% faster in the physiological condition (pH 7.4) compared to the 5 mM acetic acid condition, and 66.67% faster compared to the 0.5 mM acetic acid condition. To maintain positive control, collagen derived from rat tails was dissolved in 5 mM acetic acid at a concentration of 10 mg mL−1 and mixed with 10% AD (RC-AD) to check the gelation time. The gel formed within 10 minutes. However, the CLP–AD hydrogel can be prepared directly in 1X PBS, whereas rat tail collagen requires dissolution in acetic acid followed by a neutralization step with sodium hydroxide, as reported in previous studies.31 The preparation process of CLP–AD allows direct use of 1X PBS, eliminating the need for acetic acid and subsequent neutralization with sodium hydroxide. Overall, the CLP-based gel provides a more efficient, safer, and reliable alternative for hydrogel preparation, making it a preferable choice in tissue engineering applications.
Further, the gelation behavior of CLP–AD hydrogel system was quantitatively characterized using time sweep rheological analysis and compared with the qualitative vial inversion method (Table S2). The gel point was determined as the crossover point of the storage modulus (G′) and loss modulus (G″), representing the sol–gel transition where the elastic behavior begins to dominate over the viscous response.34 For CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) formulation, rheological measurements showed that G′ surpassed G″ at approximately 120 minutes (∼2 hours), indicating the onset of network formation between CLP and AD (Fig. S4). The pre-gel solution was mixed and loaded onto the rheometer after one hour of incubation, specifically for CLP–AD (1
2.5) formulation, rheological measurements showed that G′ surpassed G″ at approximately 120 minutes (∼2 hours), indicating the onset of network formation between CLP and AD (Fig. S4). The pre-gel solution was mixed and loaded onto the rheometer after one hour of incubation, specifically for CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) sample. Similar trends were observed for other formulations, with gelation times of approximately 100 minutes for CLP–AD (1
2.5) sample. Similar trends were observed for other formulations, with gelation times of approximately 100 minutes for CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), 50 minutes for CLP–AD (1
5), 50 minutes for CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5), and 40 minutes for CLP–AD (1
7.5), and 40 minutes for CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10). These results exhibited good agreement with the vial inversion method, confirming consistency between the qualitative and quantitative assessments of gelation kinetics. These results indicate that the gelation kinetics of the CLP–AD system are composition-dependent and can be fine-tuned to achieve desired network characteristics, making this hydrogel a promising candidate for biomedical applications.
10). These results exhibited good agreement with the vial inversion method, confirming consistency between the qualitative and quantitative assessments of gelation kinetics. These results indicate that the gelation kinetics of the CLP–AD system are composition-dependent and can be fine-tuned to achieve desired network characteristics, making this hydrogel a promising candidate for biomedical applications.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel exhibited the highest equilibrium swelling ratio of 23.31 ± 0.97, achieved within 2 hours. This high swelling ratio indicates that the hydrogel can absorb and retain a substantial amount of water, creating a hydrated environment crucial for tissue engineering applications. Fig. S5 shows the gel images before and after swelling. The stability of the swelling ratio over time further underscores the efficiency of crosslinking within this hydrogel formulation, as shown in Fig. 2(G). The study observed that the swelling ratios for CLP–AD 1
2.5 hydrogel exhibited the highest equilibrium swelling ratio of 23.31 ± 0.97, achieved within 2 hours. This high swelling ratio indicates that the hydrogel can absorb and retain a substantial amount of water, creating a hydrated environment crucial for tissue engineering applications. Fig. S5 shows the gel images before and after swelling. The stability of the swelling ratio over time further underscores the efficiency of crosslinking within this hydrogel formulation, as shown in Fig. 2(G). The study observed that the swelling ratios for CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, 1
5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5, and 1
7.5, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 hydrogels were lower than that of CLP–AD 1
10 hydrogels were lower than that of CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5, with values of 14.0 ± 0.5, 9.5 ± 1.5, and 6.2 ± 0.2, respectively. These hydrogels reached their equilibrium swelling ratios within 1 hour, but the ratios began to decrease thereafter. This reduction in swelling behaviour is attributed to the excessive solid content of AD in these formulations, leading to partial dissolution of the hydrogel in water. The CLP–AD 1
2.5, with values of 14.0 ± 0.5, 9.5 ± 1.5, and 6.2 ± 0.2, respectively. These hydrogels reached their equilibrium swelling ratios within 1 hour, but the ratios began to decrease thereafter. This reduction in swelling behaviour is attributed to the excessive solid content of AD in these formulations, leading to partial dissolution of the hydrogel in water. The CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel demonstrated superior crosslinking efficiency, as evidenced by its ability to maintain a high swelling ratio without dissolving. SEM analysis revealed that this hydrogel had larger, uniform pores, facilitating greater water retention. This observation is consistent with the results reported by Shubin et al., where single crosslinked network hydrogels with larger pores were found to hold more moisture compared to double crosslinked network hydrogels with denser pores.36 However, in this study, the reduction in swelling for CLP–AD 1
2.5 hydrogel demonstrated superior crosslinking efficiency, as evidenced by its ability to maintain a high swelling ratio without dissolving. SEM analysis revealed that this hydrogel had larger, uniform pores, facilitating greater water retention. This observation is consistent with the results reported by Shubin et al., where single crosslinked network hydrogels with larger pores were found to hold more moisture compared to double crosslinked network hydrogels with denser pores.36 However, in this study, the reduction in swelling for CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, 1
5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5, and 1
7.5, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 was not due to higher crosslinking efficiency but rather the high solid content leading to dissolution. The comparison with RC-AD hydrogels further emphasized the superior performance of the CLP–AD formulations. All CLP–AD hydrogels exhibited higher swelling ratios than the RC-AD hydrogels, demonstrating their enhanced water retention capabilities.
10 was not due to higher crosslinking efficiency but rather the high solid content leading to dissolution. The comparison with RC-AD hydrogels further emphasized the superior performance of the CLP–AD formulations. All CLP–AD hydrogels exhibited higher swelling ratios than the RC-AD hydrogels, demonstrating their enhanced water retention capabilities.
Further, the crosslinking efficiency of CLP hydrogels was analyzed using the TNBS assay, as shown in Fig. 2(J). TNBS assay quantifies the remaining free amino groups after gelation, and a lower percentage of free amines indicates higher crosslinking efficiency.6 Among the formulations tested, the CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) hydrogel exhibited the highest crosslinking efficiency (72%) compared to CLP–AD (1
2.5) hydrogel exhibited the highest crosslinking efficiency (72%) compared to CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), CLP–AD (1
5), CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5), and CLP–AD (1
7.5), and CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10). Although higher oxidized dextran content theoretically provides more aldehyde groups to react with CLP amines, excess AD can hinder efficient crosslinking due to steric effects and restricted chain mobility.37 Moreover, excessive aldehyde groups may remain unreacted or form unstable imine linkages under hydrated conditions.38 Therefore, the CLP–AD (1
10). Although higher oxidized dextran content theoretically provides more aldehyde groups to react with CLP amines, excess AD can hinder efficient crosslinking due to steric effects and restricted chain mobility.37 Moreover, excessive aldehyde groups may remain unreacted or form unstable imine linkages under hydrated conditions.38 Therefore, the CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) hydrogel, with an optimal balance of amine and aldehyde groups, exhibits maximal imine crosslinking and a stable, high swelling ratio, making it an ideal scaffold for creating hydrated environments that support cell growth and differentiation.
2.5) hydrogel, with an optimal balance of amine and aldehyde groups, exhibits maximal imine crosslinking and a stable, high swelling ratio, making it an ideal scaffold for creating hydrated environments that support cell growth and differentiation.
Among the formulations, CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 exhibited the slowest degradation rate, with approximately 31% degradation observed at day 10, as illustrated in Fig. 2(H). This suggests that the crosslinking efficiency significantly enhances the hydrogel's resistance to degradation. The degradation rates of the hydrogels followed the order: CLP–AD 1
2.5 exhibited the slowest degradation rate, with approximately 31% degradation observed at day 10, as illustrated in Fig. 2(H). This suggests that the crosslinking efficiency significantly enhances the hydrogel's resistance to degradation. The degradation rates of the hydrogels followed the order: CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 > CLP–AD 1
10 > CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5 > CLP–AD 1
7.5 > CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5. These findings indicate that, except for CLP–AD 1
5. These findings indicate that, except for CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5, the higher solid content of AD in the other formulations results in increased degradation rates. The stability of CLP–AD hydrogels under non-aqueous conditions was assessed by storing the gels in sealed vials at 37 °C. CLP–AD (1
2.5, the higher solid content of AD in the other formulations results in increased degradation rates. The stability of CLP–AD hydrogels under non-aqueous conditions was assessed by storing the gels in sealed vials at 37 °C. CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5), (1
2.5), (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5), (1
5), (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5), and (1
7.5), and (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10) maintained their structural integrity for 5 days without visible liquefaction, demonstrating good dry-state stability. Upon refrigeration at 4 °C, the hydrogels remained stable for up to two months, consistent with the findings of Ilamaran et al., who also reported that lower storage temperatures significantly improved CLP hydrogel stability compared to room-temperature storage.6 Furthermore, the Young's modulus of CLP–AD hydrogels was evaluated using compression tests. Among the formulations, CLP–AD 1
10) maintained their structural integrity for 5 days without visible liquefaction, demonstrating good dry-state stability. Upon refrigeration at 4 °C, the hydrogels remained stable for up to two months, consistent with the findings of Ilamaran et al., who also reported that lower storage temperatures significantly improved CLP hydrogel stability compared to room-temperature storage.6 Furthermore, the Young's modulus of CLP–AD hydrogels was evaluated using compression tests. Among the formulations, CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 exhibited the highest modulus (282.5 ± 7.6 kPa) compared to CLP–AD 1
2.5 exhibited the highest modulus (282.5 ± 7.6 kPa) compared to CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5 (212.3 ± 12.3 kPa), 1
5 (212.3 ± 12.3 kPa), 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5 (101.1 ± 3.4 kPa), and 1
7.5 (101.1 ± 3.4 kPa), and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 (166 ± 5 kPa), as shown in Fig. 2(I), consistent with the crosslinking results from the TNBS assay.
10 (166 ± 5 kPa), as shown in Fig. 2(I), consistent with the crosslinking results from the TNBS assay.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 ratio was selected for subsequent experiments based on its superior physicochemical and morphological characteristics. This formulation exhibited the most efficient crosslinking behavior, with highest swelling ratio among the tested combinations (1
2.5 ratio was selected for subsequent experiments based on its superior physicochemical and morphological characteristics. This formulation exhibited the most efficient crosslinking behavior, with highest swelling ratio among the tested combinations (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, 1
5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5, and 1
7.5, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10), indicating improved water uptake. SEM analysis revealed that 1
10), indicating improved water uptake. SEM analysis revealed that 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel displayed a more open and interconnected porous architecture, which is favorable for cellular attachment and infiltration. Furthermore, the 1
2.5 hydrogel displayed a more open and interconnected porous architecture, which is favorable for cellular attachment and infiltration. Furthermore, the 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel showed minimal degradation over 10 days, suggesting improved structural stability and sustained support for tissue regeneration. Although other formulations can be tuned by varying the CLP and AD concentrations, this study aims to elucidate the mechanism of hydrogel formation between CLP and AD at this optimal ratio and to assess its efficacy as a biomimetic scaffold for regenerative applications.
2.5 hydrogel showed minimal degradation over 10 days, suggesting improved structural stability and sustained support for tissue regeneration. Although other formulations can be tuned by varying the CLP and AD concentrations, this study aims to elucidate the mechanism of hydrogel formation between CLP and AD at this optimal ratio and to assess its efficacy as a biomimetic scaffold for regenerative applications.
The CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel, aged overnight, was analyzed for its viscoelastic properties using a rheometer. For viscoelastic materials, a solid-like gel is characterized by the storage modulus (G′) being greater than the loss modulus (G″), while a liquid-like material has G′ < G″. Fig. 3(A) shows the strain-sweep results of CLP–AD 1
2.5 hydrogel, aged overnight, was analyzed for its viscoelastic properties using a rheometer. For viscoelastic materials, a solid-like gel is characterized by the storage modulus (G′) being greater than the loss modulus (G″), while a liquid-like material has G′ < G″. Fig. 3(A) shows the strain-sweep results of CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel at a constant angular frequency (1 rad s−1), when increasing strain from 0.1% to 1000%, the G′ of the as prepared CLP–AD 1
2.5 hydrogel at a constant angular frequency (1 rad s−1), when increasing strain from 0.1% to 1000%, the G′ of the as prepared CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 significantly descended from 85.6 Pa to 1.69 Pa whereas the corresponding G″ ascended from 5.6 Pa to 18.05 Pa. When the strain rose from 0.1% to 50% the G′ maintained at high values, which was significantly higher than the corresponding G″ values indicating elastic-like gel behavior of the associated or crosslinked polysaccharides. Further rising strain, G′ value began to decrease, meanwhile, G″ value increased with strain growth, and the intersection points of G′ and G″ values of the hydrogel sample appeared. At this point, the mechanical property of the samples was in the state of gel-to-sol transition, owing to the damage of some reversible imine bonds of polymer networks under relatively high strain. Moreover, when strain continued to rise from 50% to 1000%, most dynamic imine bonds were broken under high strain, leading to translation from the state of gel to sol with the drop of both G′ and G″ values. This liquid-like behavior is derived from the disassociation of polysaccharide chains under high strains. From that, the linear viscoelastic range (LVR) of CLP–AD 1
2.5 significantly descended from 85.6 Pa to 1.69 Pa whereas the corresponding G″ ascended from 5.6 Pa to 18.05 Pa. When the strain rose from 0.1% to 50% the G′ maintained at high values, which was significantly higher than the corresponding G″ values indicating elastic-like gel behavior of the associated or crosslinked polysaccharides. Further rising strain, G′ value began to decrease, meanwhile, G″ value increased with strain growth, and the intersection points of G′ and G″ values of the hydrogel sample appeared. At this point, the mechanical property of the samples was in the state of gel-to-sol transition, owing to the damage of some reversible imine bonds of polymer networks under relatively high strain. Moreover, when strain continued to rise from 50% to 1000%, most dynamic imine bonds were broken under high strain, leading to translation from the state of gel to sol with the drop of both G′ and G″ values. This liquid-like behavior is derived from the disassociation of polysaccharide chains under high strains. From that, the linear viscoelastic range (LVR) of CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 was found to be around 50% strain, indicating that the hydrogel maintains its viscoelastic properties up to this strain. Beyond this point, the hydrogel begins to exhibit liquid-like behavior as G″ becomes greater than G′. This LVR region was then used for subsequent rheological measurements to ensure the viscoelastic nature of the hydrogel was maintained.
2.5 was found to be around 50% strain, indicating that the hydrogel maintains its viscoelastic properties up to this strain. Beyond this point, the hydrogel begins to exhibit liquid-like behavior as G″ becomes greater than G′. This LVR region was then used for subsequent rheological measurements to ensure the viscoelastic nature of the hydrogel was maintained.
For frequency-sweep measurement, a strain of 0.5% was selected as CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 exhibited gel-like behaviour (G′ > G″) at this strain (Fig. 3(B)). The results confirm that the CLP–AD 1
2.5 exhibited gel-like behaviour (G′ > G″) at this strain (Fig. 3(B)). The results confirm that the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 gel had a strong viscoelastic nature in the frequency range of 0.1–100 rad s−1. Additionally, the storage modulus (G′) of the CLP–AD 1
2.5 gel had a strong viscoelastic nature in the frequency range of 0.1–100 rad s−1. Additionally, the storage modulus (G′) of the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel was higher than that of the reported CLP gel crosslinked using genipin.6 This higher storage modulus indicates a greater ability to store elastic energy, suggesting improved mechanical stability of the CLP–AD 1
2.5 hydrogel was higher than that of the reported CLP gel crosslinked using genipin.6 This higher storage modulus indicates a greater ability to store elastic energy, suggesting improved mechanical stability of the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel. The difference in G′ between the CLP–AD 1
2.5 hydrogel. The difference in G′ between the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 gel and the genipin crosslinked CLP gel can be attributed to the crosslinking chemistry. The CLP–AD hydrogel was crosslinked with oxidized dextran, which likely formed strong dynamic covalent Schiff base bonds.31 This crosslinking process enhances the viscoelastic properties of the hydrogel. These findings demonstrate that the CLP–AD 1
2.5 gel and the genipin crosslinked CLP gel can be attributed to the crosslinking chemistry. The CLP–AD hydrogel was crosslinked with oxidized dextran, which likely formed strong dynamic covalent Schiff base bonds.31 This crosslinking process enhances the viscoelastic properties of the hydrogel. These findings demonstrate that the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel possesses superior mechanical properties over genipin crosslinked CLP gel, making it a promising material for applications requiring robust viscoelastic performance.
2.5 hydrogel possesses superior mechanical properties over genipin crosslinked CLP gel, making it a promising material for applications requiring robust viscoelastic performance.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel, we performed a macro self-healing test. Initially, the hydrogel was sliced into two pieces (Fig. 3(E)). The cut sections were then pressed together and left at room temperature without any additional stimuli. After 3 hours, the CLP–AD 1
2.5 hydrogel, we performed a macro self-healing test. Initially, the hydrogel was sliced into two pieces (Fig. 3(E)). The cut sections were then pressed together and left at room temperature without any additional stimuli. After 3 hours, the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel demonstrated self-healing, as it was able to support its own weight again. This self-healing ability is likely due to the reformation of dynamic imine bonds at the cut interface. The self-healing property of the CLP–AD 1
2.5 hydrogel demonstrated self-healing, as it was able to support its own weight again. This self-healing ability is likely due to the reformation of dynamic imine bonds at the cut interface. The self-healing property of the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel could be attributed to the optimal ratio of oxidized dextran (AD) and CLP. When more AD is added, fewer free amino groups remain on the interfaces, reducing the likelihood of forming Schiff base linkages.
2.5 hydrogel could be attributed to the optimal ratio of oxidized dextran (AD) and CLP. When more AD is added, fewer free amino groups remain on the interfaces, reducing the likelihood of forming Schiff base linkages.
Additionally, a higher concentration of AD results in a more rigid network, which restricts the mobility of the polymer chains. This rigid network has been observed in scanning electron microscopy (SEM) images when increasing the concentration of AD. Consequently, CLP hydrogels with higher concentrations of AD may not exhibit self-healing properties due to these limitations. Although we have not conducted self-healing experiments on CLP hydrogels with higher AD content, it has been reported that high AD content can hinder self-healing due to these reasons.41
The self-healing ability of the hydrogel was quantitatively tested by rheological measurement. The samples were tested by alternating low 1% and high 300% shear strain at a constant angular frequency of 1 rad s−1 with 1 minute intervals (Fig. 3(C)). When the shear strain rose from 1% to 300% the G′ of CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel decreased dramatically an order of magnitude due to shear thinning. As shown in Fig. 3(C), the G′ value decreased from 64.6 Pa to 8.9 Pa and was lower than the G″ (13.1 Pa) under a high amplitude force (strain = 300%, angular frequency 1 rad s−1) suggesting the hydrogel network collapsed and turned into sol state. This phenomenon demonstrates that the network structure of the dynamic hydrogels was destroyed under large strain, and the samples underwent a gel–sol transition. The dissociation of imide bonds among polysaccharide chains is believed to contribute to fast stress relaxation of the dynamic covalently crosslinked hydrogel. On the other hand, when the shear strain declined from 300% to 1% the G′ value almost fully restored to the initial level of gel state instantaneously, as associations or recovery of dissociated dynamic polymer networks. The results demonstrated that the CLP–AD 1
2.5 hydrogel decreased dramatically an order of magnitude due to shear thinning. As shown in Fig. 3(C), the G′ value decreased from 64.6 Pa to 8.9 Pa and was lower than the G″ (13.1 Pa) under a high amplitude force (strain = 300%, angular frequency 1 rad s−1) suggesting the hydrogel network collapsed and turned into sol state. This phenomenon demonstrates that the network structure of the dynamic hydrogels was destroyed under large strain, and the samples underwent a gel–sol transition. The dissociation of imide bonds among polysaccharide chains is believed to contribute to fast stress relaxation of the dynamic covalently crosslinked hydrogel. On the other hand, when the shear strain declined from 300% to 1% the G′ value almost fully restored to the initial level of gel state instantaneously, as associations or recovery of dissociated dynamic polymer networks. The results demonstrated that the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel possessed excellent fast relaxation and self-recoverable property.
2.5 hydrogel possessed excellent fast relaxation and self-recoverable property.
3.4 3D printability of the hydrogel
3D bioprinting enables the precise placement of cells and materials, resulting in structures that closely mimic natural tissues.42 Our CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 demonstrated excellent printability for 3D bioprinting applications. Interestingly, CLP–AD 1
2.5 demonstrated excellent printability for 3D bioprinting applications. Interestingly, CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 exhibited a decrease in viscosity with increasing shear rates, as observed in the shear rate ramp test in rheology (Fig. 3(D)). This shear-thinning behavior enables the material to be extruded easily during printing. The printability of the CLP–AD 1
2.5 exhibited a decrease in viscosity with increasing shear rates, as observed in the shear rate ramp test in rheology (Fig. 3(D)). This shear-thinning behavior enables the material to be extruded easily during printing. The printability of the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 was demonstrated in Fig. 3(E). The hydrogel was able to extrude as a continuous filament through both single and core–shell nozzles, with a pressure range of 0.8 to 1.0 bar. This flexibility allowed the ink to print various geometric shapes, including grids and core–shell circle structures, as shown in Fig. 3(F). For these experiments, only a single layer was printed to demonstrate the printability of the material. The circle shape was printed using a core–shell nozzle, with crystal violet added to the inner core CLP–AD before printing, and the outer shell consisting of CLP–AD without crystal violet. This visually differentiates the core from the shell, demonstrating the hydrogel's capability to print complex core–shell structures, as shown in Fig. 3(F), highlighting its potential for advanced tissue engineering applications.
2.5 was demonstrated in Fig. 3(E). The hydrogel was able to extrude as a continuous filament through both single and core–shell nozzles, with a pressure range of 0.8 to 1.0 bar. This flexibility allowed the ink to print various geometric shapes, including grids and core–shell circle structures, as shown in Fig. 3(F). For these experiments, only a single layer was printed to demonstrate the printability of the material. The circle shape was printed using a core–shell nozzle, with crystal violet added to the inner core CLP–AD before printing, and the outer shell consisting of CLP–AD without crystal violet. This visually differentiates the core from the shell, demonstrating the hydrogel's capability to print complex core–shell structures, as shown in Fig. 3(F), highlighting its potential for advanced tissue engineering applications.
3.5 Biocompatibility of CLP–AD hydrogel
The cytotoxicity of the CLP–AD hydrogel was evaluated to determine whether its leachants cause cell death, even in minimal quantities. Extracts were collected after incubating the hydrogel samples in a cell culture medium at 37 °C for 24 hours and then exposed to endothelial cells for 3 days. As per ISO 10993 part-5, the viability of cells reduced to <70% of control (TCPS) by the treatment of material extract represents its cytotoxic potential.43 The MTT assay, which measures cell activity proportional to the number of living cells, was used to assess this. The results, shown in Fig. S6, indicate that cell activity for endothelial cells treated with extracts of CLP–AD hydrogels at ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5, 1
2.5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 5, 1
5, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 7.5, and 1
7.5, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 were 108.28 ± 8.9%, 99.8 ± 2.3%, 97.5 ± 0.5%, and 105.6 ± 4.5%, respectively, compared to control cells. These findings confirm the non-cytotoxic nature of the CLP–AD hydrogel, suggesting that it is suitable for cell culture studies, like naturally derived collagen hydrogel. However, at day 3, the cell viability is significantly reduced in all groups except CLP 1
10 were 108.28 ± 8.9%, 99.8 ± 2.3%, 97.5 ± 0.5%, and 105.6 ± 4.5%, respectively, compared to control cells. These findings confirm the non-cytotoxic nature of the CLP–AD hydrogel, suggesting that it is suitable for cell culture studies, like naturally derived collagen hydrogel. However, at day 3, the cell viability is significantly reduced in all groups except CLP 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5. This might be due to toxicity caused by the presence of excess AD in other groups that might have hampered cell responses. Cell viability was also reduced in cells treated with RC-AD hydrogel extracts. We speculate that this reduction in viability might be due to the acetic acid required to dissolve the collagen in the RC-AD hydrogel, which could cause toxicity. This proves that proper crosslinking of CLP with AD are necessary for cell viability when selecting CLP hydrogels, ensuring the material supports cell growth and function. Corresponding fluorescence microscopic images demonstrate that almost all hydrogel extract-treated cells are alive, as shown in Fig. S6A. This further supports the biocompatibility and suitability of CLP–AD 1
2.5. This might be due to toxicity caused by the presence of excess AD in other groups that might have hampered cell responses. Cell viability was also reduced in cells treated with RC-AD hydrogel extracts. We speculate that this reduction in viability might be due to the acetic acid required to dissolve the collagen in the RC-AD hydrogel, which could cause toxicity. This proves that proper crosslinking of CLP with AD are necessary for cell viability when selecting CLP hydrogels, ensuring the material supports cell growth and function. Corresponding fluorescence microscopic images demonstrate that almost all hydrogel extract-treated cells are alive, as shown in Fig. S6A. This further supports the biocompatibility and suitability of CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel and further chosen for other analysis.
2.5 hydrogel and further chosen for other analysis.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel exhibited similar cell migration ability to the RC-AD hydrogel, with wound confluence percentages of 95% for the control, 93% for RC-AD, and 89% for CLP–AD 1
2.5 hydrogel exhibited similar cell migration ability to the RC-AD hydrogel, with wound confluence percentages of 95% for the control, 93% for RC-AD, and 89% for CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 at 23 hours (Fig. 4(A)). Initially, the CLP–AD 1
2.5 at 23 hours (Fig. 4(A)). Initially, the CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 showed similar migration trends to the control and RC-AD, but after 10 hours, its migration potential decreased, taking longer to achieve wound confluence (Fig. 4(B)). These findings align with previous studies where genipin crosslinked CLP alone showed delayed migration, with 16% of the wound area remaining after 15 hours. Further quantitative analysis utilizing Juli stage analysis software provided insights into wound area, cell confluence, wound width and relative wound density, shown in Fig. S7. These results highlight the potential of CLP–AD hydrogels in tissue engineering applications, suggesting that further functionalization could enhance their effectiveness in promoting cell migration.29
2.5 showed similar migration trends to the control and RC-AD, but after 10 hours, its migration potential decreased, taking longer to achieve wound confluence (Fig. 4(B)). These findings align with previous studies where genipin crosslinked CLP alone showed delayed migration, with 16% of the wound area remaining after 15 hours. Further quantitative analysis utilizing Juli stage analysis software provided insights into wound area, cell confluence, wound width and relative wound density, shown in Fig. S7. These results highlight the potential of CLP–AD hydrogels in tissue engineering applications, suggesting that further functionalization could enhance their effectiveness in promoting cell migration.29
3.6 Engineered collagen like protein hydrogel (CLP–AD) as a substrate for hMSCs culture
Stem cells are a cornerstone of regenerative medicine due to their unique abilities to self-renew and differentiate into various specialized cell types.44 Designing suitable substrates for stem cell culture is fundamental to harnessing the full potential of stem cells in regenerative medicine. It ensures that stem cells maintain their unique properties, supports their growth and differentiation, and enhances the reproducibility and scalability of stem cell-based therapies.45–47 In this study, CLP–AD was evaluated as a substrate for the culture of umbilical cord-derived human mesenchymal stem cells (UC-hMSC). The results showed that the cell viability of UC-hMSC cultured on CLP–AD 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5 hydrogel was lower than that on RC-AD and the 2D tissue culture plate control (TCPS) after 3 days (Fig. S8). This lower viability was also confirmed through live/dead staining (Fig. S8). The low cell viability observed on the CLP–AD hydrogel is likely due to the absence of hydroxyproline in the CLP protein used in this study. Supporting this explanation, a previous study comparing cell proliferation between CLP and CLPTHP congener protein engineered to incorporate hydroxyproline via genetic code expansion demonstrated significantly higher proliferation on CLPTHP. This highlights the critical role of hydroxyproline in facilitating cell binding and viability.4 Further, UC-hMSCs were encapsulated within the CLP–AD hydrogel formulation to evaluate the suitability of the hydrogel for cell encapsulation and 3D bioprinting of cell-laden constructs. Live/dead staining revealed high cell viability, with more than 90% of cells viable, comparable to the control RC-AD group. No significant differences in cell viability were observed between the CLP–AD and RC-AD groups at both Day 1 and Day 7 time points (Fig. 5(D)). Representative fluorescence images in Fig. 5(C) show the distribution of viable cells within the CLP–AD hydrogels, confirming the cytocompatibility of the formulation. Encapsulated cells in the CLP–AD hydrogel exhibited larger spheroid-like aggregate formation by day 7, reflecting the hydrogel's ability to support cell–cell interactions and promote local cellular organization. For instance, Kripamol et al. demonstrated that allylated gelatin (GAG) has a remarkable ability to induce encapsulated cells to self-organize into spheroids. This behavior is attributed to the non-cell-adhesive property of GAG, which serves primarily as a matrix, allowing cells to cluster into round spheroids rather than spreading.43 Similarly, the lack of binding sites in CLP likely promotes cell–cell interactions, ultimately leading to spheroid-like aggregation.
2.5 hydrogel was lower than that on RC-AD and the 2D tissue culture plate control (TCPS) after 3 days (Fig. S8). This lower viability was also confirmed through live/dead staining (Fig. S8). The low cell viability observed on the CLP–AD hydrogel is likely due to the absence of hydroxyproline in the CLP protein used in this study. Supporting this explanation, a previous study comparing cell proliferation between CLP and CLPTHP congener protein engineered to incorporate hydroxyproline via genetic code expansion demonstrated significantly higher proliferation on CLPTHP. This highlights the critical role of hydroxyproline in facilitating cell binding and viability.4 Further, UC-hMSCs were encapsulated within the CLP–AD hydrogel formulation to evaluate the suitability of the hydrogel for cell encapsulation and 3D bioprinting of cell-laden constructs. Live/dead staining revealed high cell viability, with more than 90% of cells viable, comparable to the control RC-AD group. No significant differences in cell viability were observed between the CLP–AD and RC-AD groups at both Day 1 and Day 7 time points (Fig. 5(D)). Representative fluorescence images in Fig. 5(C) show the distribution of viable cells within the CLP–AD hydrogels, confirming the cytocompatibility of the formulation. Encapsulated cells in the CLP–AD hydrogel exhibited larger spheroid-like aggregate formation by day 7, reflecting the hydrogel's ability to support cell–cell interactions and promote local cellular organization. For instance, Kripamol et al. demonstrated that allylated gelatin (GAG) has a remarkable ability to induce encapsulated cells to self-organize into spheroids. This behavior is attributed to the non-cell-adhesive property of GAG, which serves primarily as a matrix, allowing cells to cluster into round spheroids rather than spreading.43 Similarly, the lack of binding sites in CLP likely promotes cell–cell interactions, ultimately leading to spheroid-like aggregation.
Moreover, while natural collagen contains more than 40 biologically active binding sites for interaction with functional molecules, CLP is a “blank slate” collagen-like protein that lacks these domains.29 A similar reduction (20–30%) in cell viability on CLP compared to natural collagen was also reported by Ramshaw et al.5,7 To examine the influence of substrate characteristics of CLP hydrogel on cell morphology, the cell area was quantified. After 3 days of culturing on the CLP–AD hydrogel and control, cells were immunostained for vinculin and F-actin to view the adhesion on the substrate (Fig. 5(A)). It was observed that the cells on the CLP–AD gel had less spreading, fewer stress fibers, and long filopodial structure. Cells cultured on the CLP–AD hydrogel showed no distinct vinculin localization, reflecting the limited integrin-mediated adhesion typically observed on compliant substrates with low stiffness.48 In contrast, cells on tissue culture polystyrene (TCPS) displayed vinculin accumulation at the cell periphery (shown in red in Fig. 5(B)) with well-organized actin stress fibres, indicative of focal adhesion formation. Vinculin was also detected in the RC-AD hydrogel but was less pronounced compared to TCPS.
The highest degree of cell spreading was observed on TCPS, with a mean projected area of ∼8500 µm2. In contrast, cells cultured on CLP–AD gel showed a markedly smaller spreading area of ∼ 4450 µm2, which is about 1.93 times less than that on TCPS. This reduction aligns with previous studies demonstrating that softer substrates limit cell spreading.49,50 Cells on RC-AD surfaces showed intermediate spreading of about 6470 µm2, representing a 1.3-fold decrease compared to TCPS and a 1.4-fold increase relative to CLP–AD (Fig. 5(E)). Notably, cells on RC-AD displayed well-organized actin microfilaments as seen in Fig. 5(B). In the case of CLP–AD, elongated filopodial extensions were observed, possibly indicating early mechanosensitive differentiation in response to substrate compliance. Interestingly, this behaviour parallels that of neuron-like cells, which exert minimal traction forces and exhibit enhanced branching on softer matrices due to a more pliable, less structured cytoskeleton.51,52 The reduced integrity of the actin cytoskeleton on CLP–AD may be attributed to its low stiffness, with a storage modulus of approximately 100 pascals.
These results are consistent with previous studies that have shown that mesenchymal stem cell differentiation is responsive to important stimuli in the cellular microenvironment which is substrate stiffness.53–55 For instance, a study by Caliari and Burdick highlighted that hydrogels with lower stiffness better mimic the natural extracellular matrix (ECM) of soft tissues, supporting cell viability and proliferation while maintaining stemness.3 Additionally, research by Na et al. indicated that stiffer substrates promote differentiation, particularly osteogenic differentiation, due to increased cytoskeletal tension and chromatin compaction.56
Overall, the CLP–AD hydrogel's properties align well with the requirements for a soft substrate in stem cell culture, promoting cell adhesion and growth of umbilical cord-derived mesenchymal stem cells, while also demonstrating suitability for 3D bioprinting applications. These characteristics make it a valuable tool for soft tissue engineering applications, where maintaining the undifferentiated state of stem cells is crucial for successful tissue regeneration. In the future, hydrogels incorporating CLP with hydroxyproline or adhesion peptides will provide valuable data to further optimize this hydrogel for stem cell culture and differentiation. This study on the CLP–AD hydrogel serves as a preliminary investigation, laying the groundwork for future advancements in this field.
4. Conclusion
In this study, we successfully engineered a three-dimensional hydrogel by crosslinking recombinant collagen-like protein (CLP) with an aldehyde-functionalized dextran (AD) macromolecular crosslinker under physiological conditions. This green, mild gelation approach, operating at neutral pH in 1X PBS eliminates the need for chemical crosslinking agents or external triggers, offering a sustainable and cytocompatible alternative to conventional collagen-based materials derived from animal sources. Hydrogel formation was found to be concentration-dependent, with stable gelation achieved at a minimum CLP concentration of 10 mg mL−1. Rheological analysis confirmed the viscoelastic nature and self-healing capacity of the resulting CLP–AD hydrogels. Among the various formulations, the CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) hydrogel exhibited superior in vitro stability and degradation resistance. Additionally, its excellent printability enables the fabrication of tissue-mimetic structures, highlighting its potential for bio fabrication applications. Biocompatibility studies using EA. hy926 endothelial cells revealed that the CLP–AD (1
2.5) hydrogel exhibited superior in vitro stability and degradation resistance. Additionally, its excellent printability enables the fabrication of tissue-mimetic structures, highlighting its potential for bio fabrication applications. Biocompatibility studies using EA. hy926 endothelial cells revealed that the CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) hydrogel supports cell viability and migration comparable to cell controls. Moreover, when evaluated as a cell substrate, the thin coating of CLP–AD (1
2.5) hydrogel supports cell viability and migration comparable to cell controls. Moreover, when evaluated as a cell substrate, the thin coating of CLP–AD (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2.5) hydrogel supported the attachment, spreading, and viability of umbilical cord-derived mesenchymal stem cells (UC-MSCs), demonstrating its potential as a soft substrate for culturing cells relevant to soft tissue engineering. Taken together with previous findings on CLP-based hydrogels,6,13,15,57–59 our results demonstrate that recombinant CLP, when crosslinked using green polysaccharide-based crosslinking strategies, provides a versatile and sustainable platform for the development of next-generation biomaterials. With the successful development of the CLP–AD hydrogel, future studies could focus on elucidating how stem cell fate can be precisely regulated on this biomaterial to achieve more consistent and stable outcomes for tissue engineering applications. Additionally, the CLP hydrogel could be further optimized for rapid gelation, enhancing its potential as a bioink platform for 3D bioprinting and regenerative medicine.
2.5) hydrogel supported the attachment, spreading, and viability of umbilical cord-derived mesenchymal stem cells (UC-MSCs), demonstrating its potential as a soft substrate for culturing cells relevant to soft tissue engineering. Taken together with previous findings on CLP-based hydrogels,6,13,15,57–59 our results demonstrate that recombinant CLP, when crosslinked using green polysaccharide-based crosslinking strategies, provides a versatile and sustainable platform for the development of next-generation biomaterials. With the successful development of the CLP–AD hydrogel, future studies could focus on elucidating how stem cell fate can be precisely regulated on this biomaterial to achieve more consistent and stable outcomes for tissue engineering applications. Additionally, the CLP hydrogel could be further optimized for rapid gelation, enhancing its potential as a bioink platform for 3D bioprinting and regenerative medicine.
Author contributions
Mercyjayapriya Jebakumar: writing – review & editing, writing – original draft, visualization, validation, methodology, investigation, formal analysis, data curation, conceptualization. Mohandass Pachaiyappan – methodology, investigation, formal analysis, data curation. Chayla Reeves: methodology, formal analysis. Kate Fox: supervision, validation. Amy Gelmi: writing – review & editing, validation, supervision, formal analysis, data curation, conceptualization, funding acquisition. Niraikulam Ayyadurai: writing – review & editing, validation, supervision, conceptualization, resources, funding acquisition.Conflicts of interest
The authors declare no conflict of interest.Data availability
The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: expression and purification of collagen like protein (Fig. S1); characterization of synthesised oxidised dextran (Fig. S2); photographs demonstrated sol to gel transformation of CLP–AD (Fig. S3). time sweep experiment of CLP–AD hydrogel (Fig. S4). Swelling images of CLP–AD hydrogels (Fig. S5). In vitro cytotoxicity assay by extract method using endothelial cells (Fig. S6); cell migration potential of the CLP–AD hydrogel (Fig. S7); live/dead images of UC-hMSCs cells cultured on CLP–AD hydrogel and In vitro cytotoxicity test by direct culture of umbilical cord derived stem cells (UC-MSCs) on hydrogel surface for 3 days (Fig. S8); average pore size of CLP–AD hydrogels (Table S1). Summary of hydrogel optimization with different concentrations of CLP and AD (Table S2). See DOI: https://doi.org/10.1039/d5tb01716e.Acknowledgements
The authors gratefully acknowledge the grants received from the Department of Biotechnology (BT/PR50611/MED/32/985/2023), India, and CSIR-CLRI, ‘Collagen Biomaterial Theme’ (OLP 2403), for funding this research. AG and KF acknowledge the support of the Australian Government through the Australian Research Council Discovery Project (DP200100612) for this work. MJ gratefully acknowledges the Department of Science and Technology, India, for the award of the Inspire Research Fellowship (IF190444) and RMIT University for the Research Stipend Scholarship. MP acknowledges CSIR, India, for the award of Senior Research Fellowship.References
- R. Lev and D. Seliktar, Hydrogel biomaterials and their therapeutic potential for muscle injuries and muscular dystrophies, J. R. Soc., Interface, 2018, 15(138), 20170380 CrossRef PubMed
.
- J. L. Drury and D. J. Mooney, Hydrogels for tissue engineering: scaffold design variables and applications, Biomaterials, 2003, 24(24), 4337–4351 CrossRef CAS PubMed
.
- S. R. Caliari and J. A. Burdick, A practical guide to hydrogels for cell culture, Nat. Methods, 2016, 13(5), 405–414 CrossRef CAS PubMed
.
- J. Hu, J. Wang, X. Zhu, R. S. Tu, V. Nanda and F. Xu, Design Strategies to Tune the Structural and Mechanical Properties of Synthetic Collagen Hydrogels, Biomacromolecules, 2021, 22(8), 3440–3450 CrossRef CAS PubMed
.
- J. A. Werkmeister and J. A. Ramshaw, Recombinant protein scaffolds for tissue engineering, Biomed. Mater., 2012, 7(1), 012002 CrossRef PubMed
.
- I. Meganathan, A. Sundarapandian, G. Shanmugam and N. Ayyadurai, Three-dimensional tailor-made collagen-like proteins hydrogel for tissue engineering applications, Biomater. Adv., 2022, 139, 212997 CrossRef CAS PubMed
.
- Z. Yu, B. An, J. A. M. Ramshaw and B. Brodsky, Bacterial collagen-like proteins that form triple-helical structures, J. Struct. Biol., 2014, 186(3), 451–461 CrossRef CAS PubMed
.
- Z. Yu, B. Brodsky and M. Inouye, Dissecting a bacterial collagen domain from Streptococcus pyogenes: sequence and length-dependent variations in triple helix stability and folding, J. Biol. Chem., 2011, 286(21), 18960–18968 CrossRef CAS PubMed
.
- C. C. Caswell, M. Barczyk, D. R. Keene, E. Lukomska, D. E. Gullberg and S. Lukomski, Identification of the first prokaryotic collagen sequence motif that mediates binding to human collagen receptors, integrins α 2β1 and α11β1, J. Biol. Chem., 2008, 283(52), 36168–36175 CrossRef CAS PubMed
.
- S. Lukomski, K. Nakashima, I. Abdi, V. J. Cipriano, R. M. Ireland and S. D. Reid,
et al., Identification and characterization of the scl gene encoding a group A Streptococcus extracellular protein virulence factor with similarity to human collagen, Infect. Immun., 2000, 68(12), 6542–6553 CrossRef CAS PubMed
.
- Y. Xu, D. R. Keene, J. M. Bujnicki, M. Höök and S. Lukomski, Streptococcal Scl1 and Scl2 proteins form collagen-like triple helices, J. Biol. Chem., 2002, 277(30), 27312–27318 CrossRef CAS
.
- A. Mohs, T. Silva, T. Yoshida, R. Amin, S. Lukomski and M. Inouye,
et al., Mechanism of stabilization of a bacterial collagen triple helix in the absence of hydroxyproline, J. Biol. Chem., 2007, 282(41), 29757–29765 CrossRef CAS PubMed
.
- Y. Y. Peng, A. Yoshizumi, S. J. Danon, V. Glattauer, O. Prokopenko and O. Mirochnitchenko,
et al., A Streptococcus pyogenes derived collagen-like protein as a non-cytotoxic and non-immunogenic cross-linkable biomaterial, Biomaterials, 2010, 31(10), 2755–2761 CrossRef CAS PubMed
.
- W. M. Zhang, J. Käpylä, J. S. Puranen, C. G. Knight, C. F. Tiger and O. T. Pentikäinen,
et al., α11β1 integrin recognizes the GFOGER sequence in interstitial collagens, J. Biol. Chem., 2003, 278(9), 7270–7277 CrossRef CAS PubMed
.
- M. Aarthy, T. Hemalatha, P. Suryalakshmi, V. Vinoth, J. Mercyjayapriya and G. Shanmugam,
et al., Biomimetic design of fibril-forming non-immunogenic collagen like proteins for tissue engineering, Int. J. Biol. Macromol., 2024, 266(Pt 1), 130999 CrossRef CAS
.
- I. Meganathan, M. Pachaiyappan, M. Aarthy, J. Radhakrishnan, S. Mukherjee and G. Shanmugam,
et al., Recombinant and genetic code expanded collagen-like protein as a tailorable biomaterial, Mater. Horiz., 2022, 9(11), 2698–2721 RSC
.
- M. B. Browning, D. Dempsey, V. Guiza, S. Becerra, J. Rivera and B. Russell,
et al., Multilayer vascular grafts based on collagen-mimetic proteins, Acta Biomater., 2012, 8(3), 1010–1021 CrossRef CAS PubMed
.
- P. A. Parmar, S. C. Skaalure, L. W. Chow, J. P. St-Pierre, V. Stoichevska and Y. Y. Peng,
et al., Temporally degradable collagen-mimetic hydrogels tuned to chondrogenesis of human mesenchymal stem cells, Biomaterials, 2016, 99, 56–71 CrossRef CAS PubMed
.
- P. A. Parmar, J. P. St-Pierre, L. W. Chow, J. L. Puetzer, V. Stoichevska and Y. Y. Peng,
et al., Harnessing the Versatility of Bacterial Collagen to Improve the Chondrogenic Potential of Porous Collagen Scaffolds, Adv. Healthcare Mater., 2016, 5(13), 1656–1666 CrossRef CAS PubMed
.
- B. Depalle, Z. Qin, S. J. Shefelbine and M. J. Buehler, Influence of cross-link structure, density and mechanical properties in the mesoscale deformation mechanisms of collagen fibrils, J. Mech. Behav. Biomed. Mater., 2015, 52, 1–13 CrossRef
.
- M. Haagdorens, E. Edin, P. Fagerholm, M. Groleau, Z. Shtein and A. Ulčinas,
et al., Plant Recombinant Human Collagen Type I Hydrogels for Corneal Regeneration, Regener. Eng. Transl. Med., 2022, 8(2), 269–283 CrossRef CAS
.
- Y. Yang, A. Campbell Ritchie and N. M. Everitt, Recombinant human collagen/chitosan-based soft hydrogels as biomaterials for soft tissue engineering, Mater. Sci. Eng., C, 2021, 121, 111846 CrossRef CAS PubMed
.
- S. McLaughlin, B. McNeill, J. Podrebarac, K. Hosoyama, V. Sedlakova and G. Cron,
et al., Injectable human recombinant collagen matrices limit adverse remodeling and improve cardiac function after myocardial infarction, Nat. Commun., 2019, 10(1), 4866 CrossRef CAS PubMed
.
- S. J. Bryant, C. R. Nuttelman and K. S. Anseth, Cytocompatibility of UV and visible light photoinitiating systems on cultured NIH/3T3 fibroblasts in vitro, J. Biomater. Sci., Polym. Ed., 2000, 11(5), 439–457 CrossRef CAS PubMed
.
- M. Ilamaran, A. Janeena, S. Valappil, K. N. Ramudu, G. Shanmugam and A. Niraikulam, A self-assembly and higher order structure forming triple helical protein as a novel biomaterial for cell proliferation, Biomater. Sci., 2019, 7(5), 2191–2199 RSC
.
- J. F. Pan, L. Yuan, C. A. Guo, X. H. Geng, T. Fei and W. S. Fan,
et al., Fabrication of modified dextran-gelatin in situ forming hydrogel and application in cartilage tissue engineering, J. Mater. Chem. B, 2014, 2(47), 8346–8360 RSC
.
- A. Mousavi, S. Mashayekhan, N. Baheiraei and A. Pourjavadi, Biohybrid oxidized alginate/myocardial extracellular matrix injectable hydrogels with improved electromechanical properties for cardiac tissue engineering, Int. J. Biol. Macromol., 2021, 180, 692–708 CrossRef CAS PubMed
.
- K. Sahajpal, S. Shekhar, A. Kumar, B. Sharma, M. K. Meena and A. K. Bhagi,
et al., Dynamic protein and polypeptide hydrogels based on Schiff base co-assembly for biomedicine, J. Mater. Chem. B, 2022, 10(17), 3173–3198 RSC
.
- M. Ilamaran, A. Sundarapandian, M. Aarthy, G. Shanmugam, G. Ponesakki and K. N. Ramudu,
et al., Growth factor-mimicking 3,4-dihydroxyphenylalanine-encoded bioartificial extracellular matrix like protein promotes wound closure and angiogenesis, Biomater. Sci., 2020, 8(23), 6773–6785 RSC
.
- Q. Yang, J. Peng, H. Xiao, X. Xu and Z. Qian, Polysaccharide hydrogels: Functionalization, construction and served as scaffold for tissue engineering, Carbohydr. Polym., 2022, 278, 118952 CrossRef CAS
.
- X. Zhang, Y. Yang, J. Yao, Z. Shao and X. Chen, Strong Collagen Hydrogels by Oxidized Dextran Modification, ACS Sustainable Chem. Eng., 2014, 2(5), 1318–1324 CrossRef CAS
.
- F. S. Sarvestani, A. M. Tamaddon, R. Yaghoobi, B. Geramizadeh and N. Azarpira, Biocompatible scaffolds based on collagen and oxidized dextran for endothelial
cell survival and function in tissue engineering, Eng. Life Sci., 2023, 23(7), 2200140 CrossRef CAS PubMed
.
- S. Li, M. Pei, T. Wan, H. Yang, S. Gu and Y. Tao,
et al., Self-healing hyaluronic acid hydrogels based on dynamic Schiff base linkages as biomaterials, Carbohydr. Polym., 2020, 250, 116922 CrossRef CAS PubMed
.
- G. Stojkov, Z. Niyazov, F. Picchioni and R. K. Bose, Relationship between Structure and Rheology of Hydrogels for Various Applications, Gels, 2021, 7(4), 255 CrossRef CAS PubMed
.
- E. Y. Xi Loh, M. B. Fauzi, M. H. Ng, P. Y. Ng, S. F. Ng and H. Ariffin,
et al., Cellular and Molecular Interaction of Human Dermal Fibroblasts with Bacterial Nanocellulose Composite Hydrogel for Tissue Regeneration, ACS Appl. Mater. Interfaces, 2018, 10(46), 39532–39543 CrossRef CAS PubMed
.
- S. Li, J. Yi, X. Yu, H. Shi, J. Zhu and L. Wang, Preparation and Characterization of Acid Resistant Double Cross-Linked Hydrogel for Potential Biomedical Applications, ACS Biomater. Sci. Eng., 2018, 4(3), 872–883 CrossRef CAS PubMed
.
- M. M. H. Rumon, M. S. Rahman, A. A. Akib, M. S. Sohag, M. R. A. Rakib and M. A. R. Khan,
et al., Progress in hydrogel toughening: addressing structural and crosslinking challenges for biomedical applications, Discover Mater., 2025, 5(1), 5–29 CrossRef CAS
.
- W. Hu, Z. Wang, Y. Xiao, S. Zhang and J. Wang, Advances in crosslinking strategies of biomedical hydrogels, Biomater. Sci., 2019, 7(3), 843–855 RSC
.
- A. K. Gaharwar, I. Singh and A. Khademhosseini, Engineered biomaterials for in situ tissue regeneration, Nat. Rev. Mater., 2020, 5(9), 686–705 CrossRef CAS
.
- F. Ding, X. Shi, S. Wu, X. Liu, H. Deng and Y. Du,
et al., Flexible Polysaccharide Hydrogel with pH-Regulated Recovery of Self-Healing and Mechanical Properties, Macromol. Mater. Eng., 2017, 302(11), 1700221 CrossRef
.
- F. Ding, S. Wu, S. Wang, Y. Xiong, Y. Li and B. Li,
et al., A dynamic and self-crosslinked polysaccharide hydrogel with autonomous self-healing ability, Soft Matter, 2015, 11(20), 3971–3976 RSC
.
- T. Li, J. Chang, Y. Zhu and C. Wu, 3D Printing of Bioinspired Biomaterials for Tissue Regeneration, Adv. Healthcare Mater., 2020, 9(23), e2000208 CrossRef PubMed
.
- R. Kripamol, S. Velayudhan and P. R. Anil Kumar, Evaluation of allylated gelatin as a bioink supporting spontaneous spheroid formation of HepG2 cells, Int. J. Biol. Macromol., 2024, 274(Pt 1), 133259 CrossRef CAS PubMed
.
- Y. Jin, S. Li, Q. Yu, T. Chen and D. Liu, Application of stem cells in regeneration medicine, MedComm, 2023, 4(4), e291 CrossRef CAS PubMed
.
- M. Jarrige, E. Frank, E. Herardot, S. Martineau, A. Darle and M. Benabides,
et al., The Future of Regenerative Medicine: Cell Therapy Using Pluripotent Stem Cells and Acellular Therapies Based on Extracellular Vesicles, Cells, 2021, 10(2), 240 CrossRef CAS PubMed
.
- J. Huard, B. Gharaibeh and A. Usas, Regenerative medicine based on muscle-derived stem cells, Ope.r Tech. Orthop., 2010, 20(2), 119–126 CrossRef
.
- D. M. Hoang, P. T. Pham, T. Q. Bach, A. T. L. Ngo, Q. T. Nguyen and T. T. K. Phan,
et al., Stem cell-based therapy for human diseases, Signal Transduction Targeted Ther., 2022, 7(1), 272 CrossRef PubMed
.
- M. Valeo, S. Marie, M. Rémy, T. Menguy, C. Le Coz and M. Molinari,
et al., Bioactive hydrogels based on lysine dendrigrafts as crosslinkers: tailoring elastic properties to influence hMSC osteogenic differentiation, J. Mater. Chem. B, 2024, 12(48), 12508–12522 RSC
.
- A. Engler, L. Bacakova, C. Newman, A. Hategan, M. Griffin and D. Discher, Substrate Compliance versus Ligand Density in Cell on Gel Responses, Biophys. J., 2004, 86(1 I), 617–628 CrossRef CAS PubMed
.
- D. E. Discher, P. Janmey and Y. L. Wang, Tissue cells feel and respond to the stiffness of their substrate, Science, 2005, 310(5751), 1139–1143 CrossRef CAS PubMed
.
- L. A. Flanagan, Y. E. Ju, B. Marg, M. Osterfield and P. A. Janmey, Neurite branching on deformable substrates, NeuroReport, 2002, 13(18), 2411–2415 CrossRef
.
- P. C. Bridgman, S. Dave, C. F. Asnes, A. N. Tullio and R. S. Adelstein, Myosin IIB is required for growth cone motility, J. Neurosci., 2001, 21(16), 6159–6169 CrossRef CAS PubMed
.
- J. S. Park, J. S. Chu, A. D. Tsou, R. Diop, Z. Tang and A. Wang,
et al., The effect of matrix stiffness on the differentiation of mesenchymal stem cells in response to TGF-beta, Biomaterials, 2011, 32(16), 3921–3930 CrossRef CAS PubMed
.
- A. J. Engler, S. Sen, H. L. Sweeney and D. E. Discher, Matrix elasticity directs stem cell lineage specification, Cell, 2006, 126(4), 677–689 CrossRef CAS PubMed
.
- W. S. Leong, C. Y. Tay, H. Yu, A. Li, S. C. Wu and D. H. Duc,
et al., Thickness sensing of hMSCs on collagen gel directs stem cell fate, Biochem. Biophys. Res. Commun., 2010, 401(2), 287–292 CrossRef CAS PubMed
.
- J. Na, C. Tai, Z. Wang, Z. Yang, X. Chen and J. Zhang,
et al., Stiff extracellular matrix drives the differentiation of mesenchymal stem cells toward osteogenesis by the multiscale 3D genome reorganization, Biomaterials, 2025, 312, 122715 CrossRef CAS PubMed
.
- S. Jia, J. Wang, X. Wang, X. Liu, S. Li and Y. Li,
et al., Genetically encoded in situ gelation redox-responsive collagen-like protein hydrogel for accelerating diabetic wound healing, Biomater. Sci., 2023, 11(24), 7748–7758 RSC
.
- J. Wang, J. Hu, X. Yuan, Y. Li, L. Song and F. Xu, Recombinant collagen hydrogels induced by disulfide bonds, J. Biomed. Mater. Res., Part A, 2022, 110(11), 1774–1785 CrossRef CAS
.
- B. An, D. L. Kaplan and B. Brodsky, Engineered recombinant bacterial collagen as an alternative collagen-based biomaterial for tissue engineering, Front. Chem., 2014, 2, 40 Search PubMed
.
Footnote |
| † Equally contributed. |
| This journal is © The Royal Society of Chemistry 2025 |






