DOI:
10.1039/D5TB01302J
(Paper)
J. Mater. Chem. B, 2025,
13, 12640-12652
Impact of pyrazine doping on the electrical conductance and antimicrobial behaviour of novel one-pot synthesized polyaniline-co-pyrazine: DFT and molecular docking
Received
1st June 2025
, Accepted 1st September 2025
First published on 16th September 2025
Abstract
The present investigation employs the chemical oxidative polymerization approach to synthesize co-doped polyaniline (PANI) and polyaniline-co-pyrazine (PANI-co-Pyr) in a single pot. UV-visible spectroscopy, FTIR spectroscopy, TGA, viscosity, BET, and conductivity analyses were conducted to characterize the structural, thermal, and electrical properties of the synthesised compounds. The conductivities of the fabricated PANI and PANI-co-Pyr were depicted in a range of semiconductors due to their smaller band gaps. The stability and reactivity of PANI and PANI-co-Pyr are supported by DFT calculations, providing essential insights into their structural and energetic characteristics. Furthermore, the antibacterial properties of PANI-co-Pyr were tested against S. aureus, E. coli, B. subtilis, and P. aeruginosa, and their antifungal activity against A. niger and P. chrysogenum was measured in terms of IC50 and minimum inhibitory concentration (MIC). Molecular docking was used to investigate the bonding interactions of the Pyr ring with the surrounding residues of 1IX1 and 1UKC. PANI-co-Pyr shows higher binding affinity towards bacterial and fungal strains and exhibits significant antibacterial and antifungal activity.
1. Introduction
One of the significant, intrinsically conducting polymers with potential applications in organic electronic and optoelectronic devices is polyaniline (PANI), and it has garnered considerable interest due to its excellent thermal and environmental stability, tunable electrical conductivity, and ease of preparation from chemical oxidative polymerization.1,2 PANI's oxidation and protonation states significantly impact its characteristics; in the emeraldine state, a broad range of electrical conductivity can be attained using straightforward doping/dedoping chemistry based on acidic/basic processes.2,3 Adding an oxidant, such as ammonium peroxydisulfate (APS), initiates the traditional chemical oxidative polymerization of aniline in an acidic solution.2–4 PANI, known initially as black aniline, comes in various forms depending on its degree of oxidation. It is well-recognized as it is easy to use, is environmentally stable, and can be doped with protonic acids. Aniline monomers are linked via 1,4-coupling to form PANI. PANI's unique qualities, including its remarkable electrical properties, reasonable environmental and chemical stability, simple fabrication procedure, and low cost, make it a promising material for use in various disciplines, such as biomedical applications, anti-corrosion materials, and organic electronics.5 Photovoltaic cells,6 gas separation membranes,7 corrosion sensors,8 and chemical sensors9 are just a few of their applications. PANI's antimicrobial properties have been investigated using several bacterial strains for use in medical equipment and food packaging.10 PANI has been shown to exhibit intense antibacterial action against S. aureus and E. coli under visible light and dark conditions.11 Various concentrations of PANI dissolved in dimethyl sulfoxide have demonstrated antibacterial activity against a range of Gram-positive and Gram-negative bacterial strains.12 PANI's insoluble nature in aqueous solutions extends its antibacterial action time, allowing for the reuse of coatings based on it, which is a significant benefit over previously proposed antibacterial agents.13 Additionally, the addition of functional groups to PANI's aromatic ring can increase the polymer's solubility in organic solvents, making it easier to produce thin films based on it.13,14 Antibacterial activity and high solubility in organic solvents can therefore be combined to create a superior coating with outstanding utility.
Maruthapandi et al. studied the antimicrobial properties of the PANI composites against two common Gram-negative pathogens, P. aeruginosa and K. pneumoniae. The negative groups of bacterial cell walls were tightly attached by all the composites, inhibiting their normal functions and ultimately causing cell lysis and damage to the cell wall envelope.15 The in situ inverse microemulsion approach was used to synthesize polyaniline/Cu0.05Zn0.95O (PANI/CZO) nanocomposites. The powder inhibition zone, MIC, and MBC were used to investigate the antimicrobial activity of the sample against S. aureus, E. coli, and C. albicans. The findings demonstrated that PANI/CZO nanocomposites showed outstanding antibacterial activity against the bacterial and fungal strains.16 PANI was doped with nitro compounds such as picric acid, 3,5-dinitrobenzoic acid, and hydrochloric acid in the emeraldine salt. Polyaniline chloride was dedoped to prepare the polyaniline emeraldine base (PANIEB). Doping PANI with nitro compounds increases its crystallinity. Using the agar well diffusion method, the in vitro antibacterial capabilities of PANI against a range of bacterial strains and the fungus Candida albicans were evaluated. The MIC values and zone of inhibition diameter were used to determine the antibacterial effects. In comparison to PANIEB, the test outcomes showed that the doped PANIs had enhanced antibacterial efficacy.17 Chaubisa et al. compared the PANI–pyrazole composite to other synthetic composites; they observed that it had a remarkable MIC for antibacterial activity against the pathologically essential bacterial strains S. aureus and S. pyogenes. The antifungal activity of the PANI–tetrazole composite was better than that of individual PANI against A. niger and A. clavatus. The PANI–tetrazole and PANI–pyrazole composites exhibited a higher and more positive MIC, confirming their potential use as beneficial antibacterial and antifungal agents.18 The conducting and antibacterial properties of the dye-substituted PANI copolymer were examined by Jangid et al. The antifungal activity was investigated against C. albicans, A. niger, and A. clavatus, whereas the antibacterial activity was studied against E. coli, P. aeruginosa, S. aureus, and S. pyogenes. When dye-substituted polyanilines are compared to PANI and dyes alone, their antimicrobial qualities demonstrate good results.19 Chaubisa et al. synthesised the copolymers of PANI and PANI–indole. They investigated the antifungal activity of their compounds against A. niger, A. clavatus, and C. albicans, as well as their antibacterial activity against two Gram-positive bacteria, S. pyogenes and S. aureus, and two Gram-negative bacteria, P. aeruginosa and E. coli.20 Despite the abundance of research on the antimicrobial properties of PANIs and PANI-based copolymers, a literature review has revealed a scarcity of studies on PANIs doped with organic acids (dual doping/co-doping). Kumari et al. studied co-doped one-pot synthesised PANI-substituted imidazole copolymers and their molecular docking. The conductivity measurements were analyzed by the two-probe method. Additionally, PANI–imidazole antibacterial activity was evaluated against S. aureus and E. coli, and the results were expressed in terms of MIC, minimum bactericidal concentration (MBC), and IC50. They observed that against S. aureus in particular, PANI–imidazole exhibited more antimicrobial activity than PANI.21 According to Quan et al., bromine-benzyl-disubstituted PANI exhibits antibacterial properties against both Gram-positive B. subtilis and Gram-negative E. coli. They found that bromine-benzyl-disubstituted PANI has more potent antibacterial activity than PANI and benzyl-substituted PANI.22 In 2014, Jangid et al. investigated the antibacterial activities of dye-substituted PANIs. The dyes had superior antibacterial properties when tested against E. coli, S. aureus, and S. pyogenes.23 According to Cai et al., bromo-substituted PANI exhibits antibacterial activity against both Gram-positive B. subtilis and Gram-negative E. coli. With increased bromine concentration, the bromo-substituted PANI demonstrated a remarkable antibacterial property.24 Pyrazine exhibits a broad spectrum of biological activities, including antimicrobial, antifungal, antiviral, anticancer, and antioxidant properties. Their biological efficiency stems from their aromatic, nitrogen-containing heterocyclic structure, which enables strong interactions with biomolecules, including DNA, proteins, and microbial cell membranes. Substitution on the pyrazine ring can further enhance lipophilicity, binding affinity, and selectivity, thereby improving antimicrobial and antifungal efficiency. The need for new antimicrobial materials is urgent due to the increasing number of microorganisms that are resistant to antibiotics. Combining Pyr units with the distinct structural and electrical characteristics of conductive polymers, such as PANI, offers an efficient antibacterial and antifungal agent.
In this work, the chemical oxidative polymerisation method (COPM) was used to synthesize co-doped PANI and PANI-co-Pyr. PANI and PANI-co-Pyr were doped with hydrochloric acid and co-doped with trifluoroacetic acid to increase the processability and mobility of π-electrons. By combining the heteroring Pyr with PANI, the antibacterial and antifungal properties of PANI were improved. PANI and PANI-co-Pyr were characterized using various techniques, including UV-visible spectroscopy, FTIR spectroscopy, TGA, viscosity measurements, and conductivity measurements. Their antibacterial properties were studied against S. aureus, B. subtilis, E. coli, and P. aeruginosa, and the antifungal activity was studied against A. niger and P. chrysogenum. The synthesized copolymers demonstrated strong antibacterial and antifungal activity, indicating their potential for use in biological applications.
2. Experimental
2.1. Materials
The chemicals utilized for the synthesis of PANI and its copolymer are aniline (C6H5NH2, 99% pure), hydrochloric acid (HCl, 98% pure), trifluoroacetic acid (CF3CO2H, 99.5% pure), ethanol (99.9% pure), ammonium persulfate (APS, (NH4)2S2O8, 98% pure), and the pyrazine heterocyclic moiety (C4H4N2, 99% pure). We used all the chemicals from SRL exactly as supplied for the synthesis. Gram-positive and Gram-negative bacteria (S. aureus MTCC3160, B. subtilis MTCC441, E. coli MTCC1652, P. aeruginosa MTCC741) and fungal strains (A. niger MTCC282, P. chrysogenum MTCC2552) were obtained from MTCC, Chandigarh.
2.2. Synthesis of co-doped PANI
An acidic medium containing 20 mL of hydrochloric acid (1.0 M) in distilled water as a dopant was mixed with 6 mL of aniline and stirred for 1 h at 800 rpm to create a homogeneous solution. Following that, 20 mL of trifluoroacetic acid (0.5 M) was added to the solution as a co-dopant, and the mixture was stirred at 800 rpm for at least 1 h at 0 °C in an ice bath. For the polymerization, 100 mL of ammonium persulfate (1.0 M) was added dropwise as an oxidant, while the mixture was continuously stirred for 4 h at 0 °C. After polymerization, the filtrate was separated and then washed with ethanol and distilled water to remove all impurities. The residue was oven-dried for 24 hours at 50–60 °C to obtain a free-flowing powder of emeraldine salt of PANI, with a yield of 79%. The resulting pure PANI was found to be dark green after drying. The synthesized emeraldine salt of PANI was kept in ammonium hydroxide solution overnight to form the PANI emeraldine base. Scheme 1 illustrates the synthesized emeraldine base of PANI. The COPM was used to synthesize PANI.25
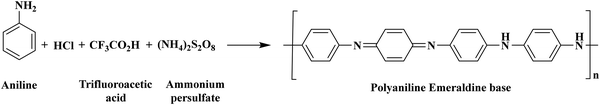 |
| | Scheme 1 Synthesis of co-doped PANI. | |
2.3. Synthesis of co-doped polyaniline-co-pyrazine
Using aniline-to-pyrazine ratios (i.e., 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1
2, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), the COPM was used to synthesize co-doped PANI-co-Pyr. Here, 20 mL of hydrochloric acid (1.0 M) and 20 mL of trifluoroacetic acid (0.5 M) were added to an aqueous acidic medium containing 6 mL of aniline (0.0658 mol) and 5.16 g of pyrazine (0.644 mol). The solution was stirred at 800 rpm for 2 h at 0 °C in an ice bath. For the polymerization process, 100 mL of ammonium persulfate (1.0 M) was added dropwise to the solution at 0 °C, and the solution was stirred continuously at 800 rpm for 4 h. After 7 h, a dark green precipitate formed. The filtrate was separated by filtration and then washed with ethanol and distilled water to remove all impurities. The residue was oven-dried for 24 h at 50–60 °C to obtain a free-flowing powder of the PANI-co-Pyr. The resulting pure PANI-co-Pyr was found to be dark green after drying, with a yield of 73%. PANI-co-Pyr ratios of 1
3), the COPM was used to synthesize co-doped PANI-co-Pyr. Here, 20 mL of hydrochloric acid (1.0 M) and 20 mL of trifluoroacetic acid (0.5 M) were added to an aqueous acidic medium containing 6 mL of aniline (0.0658 mol) and 5.16 g of pyrazine (0.644 mol). The solution was stirred at 800 rpm for 2 h at 0 °C in an ice bath. For the polymerization process, 100 mL of ammonium persulfate (1.0 M) was added dropwise to the solution at 0 °C, and the solution was stirred continuously at 800 rpm for 4 h. After 7 h, a dark green precipitate formed. The filtrate was separated by filtration and then washed with ethanol and distilled water to remove all impurities. The residue was oven-dried for 24 h at 50–60 °C to obtain a free-flowing powder of the PANI-co-Pyr. The resulting pure PANI-co-Pyr was found to be dark green after drying, with a yield of 73%. PANI-co-Pyr ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 1
2 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 were also synthesized under similar circumstances. The synthesis of PANI-co-Pyr is shown in Scheme 2. The chemical oxidative polymerization process that converts PANI to PANI-co-Pyr is illustrated in Scheme S1.
3 were also synthesized under similar circumstances. The synthesis of PANI-co-Pyr is shown in Scheme 2. The chemical oxidative polymerization process that converts PANI to PANI-co-Pyr is illustrated in Scheme S1.
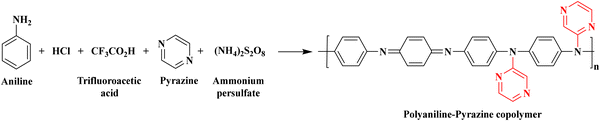 |
| | Scheme 2 One-pot synthesis of co-doped PANI-co-Pyr. | |
2.4. Characterization of synthesized polyaniline and polyaniline-co-pyrazine
The synthesized PANI and PANI-co-Pyr were analyzed using various techniques, including UV-visible spectroscopy, FTIR spectroscopy, TGA, and viscosity and conductivity measurements. The synthesized PANI and PANI-co-Pyr absorption spectra between 200–800 nm were obtained using a UV-visible spectrophotometer, SHIMADZU UV-1900. FTIR spectra in the 4000–600 cm−1 range were obtained using a Bruker ALPHA FTIR spectrometer. The TGA thermal analysis technique was used to investigate the synthesized copolymer's thermal properties. The thermal stabilities of PANI-co-Pyr copolymers were investigated using TGA methods in a nitrogen environment at a heating rate of 10 °C min−1. A temperature range of 33 to 1000 °C was covered. The intrinsic viscosity [η] and viscosity average molar mass (Mv) of the PANI and PANI-co-Pyr were determined using viscosity measurements. The viscosities of homopolymers and copolymers were measured at room temperature (34 °C) using an Ostwald viscometer, which is a U-shaped glass tube with a bulb and a capillary tube. As the liquid flows, the bulb helps retain hydrostatic pressure; because of its weight, the liquid moves through the capillary tube. The Brunauer–Emmett–Teller (BET) method was used to calculate the surface area, pore size, and total pore volume using a Quantachrome Autosorb automated gas sorption system at 77 K. Measurements of temperature-dependent conductivity were performed between 30 °C and 75 °C. A Keithley electrometer 6517-A (India) was used to measure the conductivities of various PANIs using two-probe measurements. A silver paste was used to create electrical connections. The electrometer has two spring-loaded contact probes, and Teflon washers provide insulation for these probes, which move in a pipe. The sample plate and resistance temperature detector (RTD) sensor are also held in place by the appropriate stand on which this probe setup is installed. Additionally, the stand serves as the oven's lid for proportional-integral-derivative (PID) control. The Digital Pico ammeter, DPM-111, and the high-voltage power supply, EHT-11, are connected via Teflon-coated leads. The setup's maximum voltage was 1000 V, and the maximum current was 100 × 10−12 A.
Theoretical analyses of geometrical and energetic molecular properties, including total energy, dipole moment, bond lengths, bond angles, and dihedral angles of the adsorbents, were performed using the Gaussian 16 package. All calculations were performed using density functional theory at the B3LYP/6-311g(d) level of theory. The absence of any imaginary frequency confirmed the local minima of optimization. The calculations were also performed for important electronic parameters such as the energy gap (ΔE), the highest occupied molecular orbital energy (EH), and the lowest unoccupied molecular orbital energy (EL). Furthermore, reactivity descriptors, such as the electrophilicity index, energy gap, chemical hardness, and softness, have been calculated from HOMO–LUMO data. The interaction and reactivity of the molecule are understood with the help of frontier molecular orbital (FMO) theory. The stability and reactivity of the complexes' structures are explained by the energy gap (ΔE), which has essential chemical implications. Different physicochemical characteristics were obtained using the HOMO and LUMO energy values. The chemical hardness of a molecule, which is directly connected to the HOMO–LUMO energy gap, can be used to explain its reactivity. A molecule is less reactive if its E gap or chemical hardness is more significant.26–28
By the microbroth dilution method, the antibacterial properties of PANI, PANI-co-Pyr, and amoxicillin were examined against bacteria, including S. aureus, B. subtilis, E. coli, and P. aeruginosa. After 24-hour incubation, the minimum inhibitory concentration (MIC) and inhibitory concentration 50 (IC50) were determined. The antifungal properties of PANI, PANI-co-Pyr, and ketoconazole were studied against A. niger and P. chrysogenum by the same method. After 48–72 h of incubation, the MIC and IC50 were calculated. Optical density was taken at 600 nm. The experiments were conducted in triplicate to ensure accuracy. To scan the geometrically and energetically stable conformations of PANI and PANI-co-Pyr, the molecular docking of these probes was in the microenvironment of 1IX1 and 1UKC. The crystal structure of proteins was retrieved from the Protein Data Bank. The PANI and PANI-co-Pyr DFT-optimized three-dimensional (3D) structures were used as input for molecular docking. Before docking, suitable binding sites for inserting the ligands into the active sites of proteins were determined by the site finder, and a total of 11 binding sites in 1IX1 and 18 binding sites for 1UKC were discovered for both ligands in the process. Finally, the ligands were docked to the favourable sites of proteins determined by MOE's site finder using the triangle matcher placement approach, and the best-fitting domain was chosen for the docking research.
3. Results and discussion
3.1. UV-visible spectroscopic analysis
The synthesized copolymers were examined using a UV-visible spectrophotometer in the wavelength range of 200–800 nm. The UV-visible spectrum of PANI in DMSO showed two major peaks, one at 320 nm and another at 618 nm. The band at 320 nm in the UV region was responsible for π–π* transitions in benzenoid rings, and the transition at 618 nm in the visible region was associated with the polaronic transitions. Similarly, the UV-visible spectrum of PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) showed a peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the peak at 617 nm in the visible region corresponded to polaronic transitions or indicated extended conjugation.29 Further, the UV-visible spectra of PANI-co-Pyr (1
1) showed a peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the peak at 617 nm in the visible region corresponded to polaronic transitions or indicated extended conjugation.29 Further, the UV-visible spectra of PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) and PANI-co-Pyr (1
2) and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) showed a significant peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the second peak at 584 nm in the visible region corresponded to polaronic transitions, as shown in Fig. 1. The UV-Vis spectra of PANI and PANI-co-Pyr also exhibited distinct changes in intensity. A hyperchromic shift was observed in PANI because PANI shows a high-intensity peak due to doping and conjugation. A slight hyperchromic shift was observed in the polaronic transition bands for the PANI-co-Pyr (1
3) showed a significant peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the second peak at 584 nm in the visible region corresponded to polaronic transitions, as shown in Fig. 1. The UV-Vis spectra of PANI and PANI-co-Pyr also exhibited distinct changes in intensity. A hyperchromic shift was observed in PANI because PANI shows a high-intensity peak due to doping and conjugation. A slight hyperchromic shift was observed in the polaronic transition bands for the PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), indicating enhanced conjugation due to the incorporation of Pyr into the PANI backbone. However, with increasing the Pyr ratio (1
2), indicating enhanced conjugation due to the incorporation of Pyr into the PANI backbone. However, with increasing the Pyr ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), a hypochromic effect was observed, suggesting a reduction in polaron concentration and disrupted π-conjugation. This trend is consistent with the observed decrease in electrical conductivity and suggests that excess pyrazine interferes with the optimal electronic structure of PANI, as shown in Fig. 1.
3), a hypochromic effect was observed, suggesting a reduction in polaron concentration and disrupted π-conjugation. This trend is consistent with the observed decrease in electrical conductivity and suggests that excess pyrazine interferes with the optimal electronic structure of PANI, as shown in Fig. 1.
 |
| | Fig. 1 UV-visible spectra of polyaniline and polyaniline-co-pyrazine. | |
3.2. FTIR analysis
FTIR spectra of the synthesized PANI and PANI-co-Pyr copolymers were obtained in the 4000–600 cm−1 range. The FTIR spectra of the PANI-co-Pyr revealed many shifted bands, indicating that pure PANI has been doped. The efficient substitution of Pyr on the backbone of PANI was validated by the FTIR spectra, which displayed the unique peaks of both PANI and PANI-co-Pyr (Fig. 2). Para-substitution was observed at 794, 798, 797, and 797 cm−1, confirming benzene ring substitution from PANI to PANI-co-Pyr.21 The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations of the aromatic ring were observed at 1484, 1490, 1490, and 1489 cm−1 for PANI, PANI-co-Pyr (1
C stretching vibrations of the aromatic ring were observed at 1484, 1490, 1490, and 1489 cm−1 for PANI, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), PANI-co-Pyr (1
1), PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and PANI-co-Pyr (1
2), and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. The C
3), respectively. The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations of the benzenoid ring were observed at 1564, 1571, 1571, and 1571 cm−1 for PANI, PANI-co-Pyr (1
C stretching vibrations of the benzenoid ring were observed at 1564, 1571, 1571, and 1571 cm−1 for PANI, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), PANI-co-Pyr (1
1), PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and PANI-co-Pyr (1
2), and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. Pyr shifted the aromatic and benzenoid peaks, indicating altered conjugation (minor shift due to substitution effect). The stretching vibrations of C–N resulted in strong peaks at 1295, 1296, and 1294 cm−1 for PANI-co-Pyr ratios (1
3), respectively. Pyr shifted the aromatic and benzenoid peaks, indicating altered conjugation (minor shift due to substitution effect). The stretching vibrations of C–N resulted in strong peaks at 1295, 1296, and 1294 cm−1 for PANI-co-Pyr ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1
2, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. The stretching vibrations of the N–H bands were observed at 3220 cm−1 for PANI. The Pyr nitrogen atoms influence hydrogen bonding, slightly shifting or broadening this peak. The observed peak of N–H also indicates that Pyr polymerizes through C with the N of PANI for PANI-co-Pyr ratios 1
3), respectively. The stretching vibrations of the N–H bands were observed at 3220 cm−1 for PANI. The Pyr nitrogen atoms influence hydrogen bonding, slightly shifting or broadening this peak. The observed peak of N–H also indicates that Pyr polymerizes through C with the N of PANI for PANI-co-Pyr ratios 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1
2, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, respectively. The FTIR results of PANI and PANI-co-Pyr are shown in Table 1 and Fig. 2.
3, respectively. The FTIR results of PANI and PANI-co-Pyr are shown in Table 1 and Fig. 2.
 |
| | Fig. 2 FTIR spectra of (a) polyaniline, (b) polyaniline-co-pyrazine (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), (c) polyaniline-co-pyrazine (1 1), (c) polyaniline-co-pyrazine (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and (d) polyaniline-co-pyrazine (1 2), and (d) polyaniline-co-pyrazine (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). 3). | |
Table 1 FTIR data of PANI and PANI-co-Pyr
| S. no. |
Vibrational modes in copolymers |
IR frequency (cm−1) |
| PANI |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
| 1 |
Para-substitution |
794 |
798 |
797 |
797 |
| 2 |
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C Stretching (aromatic ring) C Stretching (aromatic ring) |
1484 |
1490 |
1490 |
1489 |
| 3 |
C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C Stretching (quinoid ring) C Stretching (quinoid ring) |
1564 |
1571 |
1571 |
1571 |
| 4 |
C–H Stretching (aromatic ring) |
— |
— |
2903 |
2946 |
| 5 |
N–H Stretching (PANI) |
3220 |
— |
— |
— |
| 6 |
C–N Stretching |
— |
1295 |
1296 |
1294 |
| 7 |
Protonation band |
1122 |
1127 |
1128 |
1128 |
3.3. Thermogravimetric analysis (TGA)
The thermal stabilities of the PANI and PANI-co-Pyr were studied using the TGA technique. The synthesized PANI and PANI-co-Pyr have different weight losses with temperature. A TGA analysis revealed four stages of weight loss as a function of temperature. The first weight losses of 7.11%, 7.27%, 7.64%, and 5.9% for PANI, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), PANI-co-Pyr (1
1), PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and PANI-co-Pyr (1
2), and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) were observed between 20–130 °C, 17–126 °C, 17–152 °C, and 22–116 °C due to vaporizing H2O molecules. The second weight losses of 19.42%, 20.84%, 18.86%, and 18.3% were observed between 130–394 °C, 126–444 °C, 152–431 °C, and 116–391 °C, respectively, and are related to the loss of dopants. The third weight losses of 23.13%, 22.49%, 22.46%, and 27.49% occurred between 394–596 °C, 444–679 °C, 431–648 °C, and 391–671 °C due to the thermal breakdown of PANI-co-Pyr. The fourth weight losses of 12.85%, 7.8%, 10.33%, and 8.6% were observed at 596–996 °C, 980 °C, 982 °C, and 979 °C due to the continuous disintegration of the PANI-co-Pyr. Thermal behaviour supports the presence of Pyr in the copolymer.20,21 The graph indicates that PANI-co-Pyr is more thermally stable than pure PANI, as shown in Table 2 & Fig. 3.
3) were observed between 20–130 °C, 17–126 °C, 17–152 °C, and 22–116 °C due to vaporizing H2O molecules. The second weight losses of 19.42%, 20.84%, 18.86%, and 18.3% were observed between 130–394 °C, 126–444 °C, 152–431 °C, and 116–391 °C, respectively, and are related to the loss of dopants. The third weight losses of 23.13%, 22.49%, 22.46%, and 27.49% occurred between 394–596 °C, 444–679 °C, 431–648 °C, and 391–671 °C due to the thermal breakdown of PANI-co-Pyr. The fourth weight losses of 12.85%, 7.8%, 10.33%, and 8.6% were observed at 596–996 °C, 980 °C, 982 °C, and 979 °C due to the continuous disintegration of the PANI-co-Pyr. Thermal behaviour supports the presence of Pyr in the copolymer.20,21 The graph indicates that PANI-co-Pyr is more thermally stable than pure PANI, as shown in Table 2 & Fig. 3.
Table 2 TGA data of polyaniline and polyaniline-co-pyrazine
| S. no. |
Synthesized copolymers |
Losses in weight (%) at various temperatures (°C) |
| 50 |
100 |
200 |
300 |
400 |
500 |
600 |
700 |
800 |
900 |
1000 |
| 1 |
PANI |
98 |
93 |
91 |
81 |
73 |
62 |
50 |
46 |
42 |
39 |
37 |
| 2 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
98 |
94 |
91 |
84 |
75 |
66 |
53 |
49 |
45 |
43 |
41 |
| 3 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
98 |
94 |
91 |
84 |
75 |
67 |
53 |
49 |
45 |
42 |
40 |
| 4 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
98 |
94 |
92 |
84 |
74 |
65 |
51 |
47 |
44 |
41 |
39 |
 |
| | Fig. 3 TGA graphs of polyaniline and polyaniline-co-pyrazine. | |
3.4. Viscosity measurement
Solutions were prepared in dimethyl sulfoxide (DMSO) at 1 mg mL−1 for the viscosity determination of PANI and PANI-co-Pyr, and then sonicated at 40 °C for 1 h. Next, 13 ml of the PANI and PANI-co-Pyr solutions were pipetted into the Ostwald viscometer. The viscosities of PANI and PANI-co-Pyr solutions were determined at room temperature (34 °C). The densities of solutions were measured using a Gay-Lussac-type pycnometer.
The Mark–Houwink equation [η] = K Mva was used to determine the intrinsic viscosities. In this equation, η stands for intrinsic viscosity ([η] = 1.95 × 10−6Mv1.36), Mv for viscosity average molar mass, and K and a are Mark–Houwink constants. The viscosity-average molar mass was calculated using an intrinsic viscosity of PANI and PANI-co-Pyr.29 Intrinsic viscosities and the viscosity average molar mass of synthesized PANI and PANI-co-Pyr are shown in Table 3. PANI-co-Pyr has a higher viscosity average molar mass than pure PANI due to the extent of conjugation and polymerization. Table 3 indicates that incorporating Pyr into the PANI backbone generally increases the viscosity-average molar mass, suggesting enhanced polymer chain growth and conjugation due to copolymerization. Among the copolymers, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) exhibited the highest intrinsic viscosity (2.36 dL g−1) and molar mass (9.48 × 104 g mol−1), followed by PANI-co-Pyr (1
2) exhibited the highest intrinsic viscosity (2.36 dL g−1) and molar mass (9.48 × 104 g mol−1), followed by PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and (1
1) and (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). Interestingly, the PANI-co-Pyr (1
3). Interestingly, the PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) composition showed a slightly lower molar mass compared to the (1
3) composition showed a slightly lower molar mass compared to the (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and (1
1) and (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). This reduction may be attributed to a less optimal ratio for chain extension, possibly resulting in fewer oligomer units linking effectively, and thus yielding a lower overall molar mass.
2). This reduction may be attributed to a less optimal ratio for chain extension, possibly resulting in fewer oligomer units linking effectively, and thus yielding a lower overall molar mass.
Table 3 Intrinsic viscosities and viscosity average molar masses of synthesized PANI and PANI-co-Pyr
| S. no. |
Synthesized copolymers |
Intrinsic viscosity [η] (dL g−1) |
Viscosity Average Molar Mass (Mv) (g mol−1) |
| 1 |
PANI |
1.92 |
2.50 × 104 |
| 2 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
2.02 |
2.66 × 104 |
| 3 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
2.36 |
3.39 × 104 |
| 4 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
1.98 |
2.62 × 104 |
3.5. Brunauer–Emmett–Teller analysis (BET)
Nitrogen adsorption–desorption analysis was employed to obtain information about the specific surface area, pore size, and the total pore volume of the PANI and PANI-co-Pyr copolymers.30 The surface area was found to be 34.63 m2 g−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 33.19 m2 g−1 for PANI-co-Pyr (1
1), 33.19 m2 g−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and 33.10 m2 g−1 for PANI-co-Pyr (1
2), and 33.10 m2 g−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. However, the surface area of the PANI was found to be smaller (24.86 m2 g−1), as shown in Table 4. The surface area was increased by the presence of micropores in the PANI-co-Pyr. The nitrogen adsorption–desorption isotherms of polyaniline and polyaniline-co-pyrazine are shown in Fig. S1. Surface area increased when pyrazine was added to the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). The highest surface area represents a good sorption ability of materials.
3), respectively. However, the surface area of the PANI was found to be smaller (24.86 m2 g−1), as shown in Table 4. The surface area was increased by the presence of micropores in the PANI-co-Pyr. The nitrogen adsorption–desorption isotherms of polyaniline and polyaniline-co-pyrazine are shown in Fig. S1. Surface area increased when pyrazine was added to the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). The highest surface area represents a good sorption ability of materials.
Table 4 Pore size, surface area, and pore volume of synthesized PANI and PANI-co-Pyr copolymers
| S. no. |
Synthesized copolymers |
Pore size (Å) |
Surface area (m2 g−1) |
Total pore volume (cm3 g−1) |
| 1 |
PANI |
2.16 |
24.86 |
2.69 |
| 2 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
1.98 |
34.63 |
3.44 |
| 3 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
1.97 |
33.19 |
3.28 |
| 4 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
2.01 |
33.10 |
3.33 |
3.6. Conductivity measurements
Pyr-substituted PANI comprises Pyr molecules that have been substituted on the PANI backbone. Structural studies indicate that interactions between the PANI and Pyr heterorings occur at the N atom of imine groups on the backbones of polymeric chains via enhanced conjugated electron delocalization.23 Conductivity is improved by substituting the Pyr heteroring on the backbone of PANI. Electrical conductivity increased when pyrazine was added to the PANI backbone (from 4.2 × 10−3 S cm−1 to 5.3 × 10−3 S cm−1), suggesting that charge transport is favoured by the structural and electronic changes generated by copolymerization. Plotted graphs of resistivity (a) log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ρ vs. 1000/T, and conductivity (b) log10
ρ vs. 1000/T, and conductivity (b) log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) σ vs. 1000/T of PANI and PANI-co-Pyr are shown in Fig. 4. The slope, band gap, and conductivity of PANI and PANI-co-Pyr are illustrated in Table 5.
σ vs. 1000/T of PANI and PANI-co-Pyr are shown in Fig. 4. The slope, band gap, and conductivity of PANI and PANI-co-Pyr are illustrated in Table 5.
 |
| | Fig. 4 Plots of (a) log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ρ vs. 1000/T, and (b) log10 ρ vs. 1000/T, and (b) log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) σ vs. 1000/T for polyaniline and polyaniline-co-pyrazine. σ vs. 1000/T for polyaniline and polyaniline-co-pyrazine. | |
Table 5 Slopes, band gaps, and conductivities of the copolymers
| S. no. |
Synthesized copolymers |
Slope (m) |
Band gap (Eg; eV) |
Conductivity (S cm−1) |
| 1 |
PANI |
1.95 |
3.36 |
4.20 × 10−3 |
| 2 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
1.33 |
2.29 |
5.14 × 10−3 |
| 3 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
1.22 |
2.10 |
5.38 × 10−3 |
| 4 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
1.42 |
2.44 |
4.41 × 10−3 |
3.7. Density functional theory (DFT)
The optimized geometries of PANI and PANI-co-Pyr at the B3LYP/6-311g(d) level of theory are presented in Fig. 5. It is noteworthy that the PANI-co-Pyr was found to be more stable than PANI by −28.6 keV. This stabilization was derived from the dipolar interactions, which was endorsed by the increase in the dipole moment value from 1.4387 Debye (PANI) to 2.1032 Debye (PANI-co-Pyr); this can be visualized through the donor nature of the Pyr unit in frontier molecular orbitals. The HOMO and LUMO energies are typically employed as reactivity indicators to determine a molecule's propensity to donate and receive electrons under favourable circumstances, which in turn determines its antioxidant and associated properties. The HOMO and LUMO of compounds have been investigated and are reported in Fig. 5.
 |
| | Fig. 5 The optimized geometries of (a) PANI and (b) PANI-co-Pyr. (c–f) 3D HOMO–LUMO pictorial illustration depicting the PANI and PANI-co-Pyr energy gap. | |
The stability of PANI and PANI-co-Pyr is supported by the positive values of ΔE and the negative eigenvalues of the HOMO and LUMO (Table S1). An electron's excitation energies from the HOMO to the LUMO also decrease when the HOMO–LUMO energy gap (ΔE) values decrease, which facilitates the charge transfer. The HOMO–LUMO energy gap for PANI was calculated to be 2.11 eV, while for PANI-co-Pyr, it was calculated to be 1.85 eV, indicating that PANI-co-Pyr exhibits higher polarizability and reactivity. Frontier molecular orbital topology revealed that HOMO is dispersed over the entire molecular unit, whereas LUMO is primarily spread across the centred aniline unit of PANI and PANI-co-Pyr.
Furthermore, reduced ionization potential (IP) and energy gap ΔE values enhance the PANI-co-Pyr electron-donating capacity, facilitating active engagement in biological activities. The obtained IP values are displayed in Table S1 and Fig. 6 in the following order for both ΔE and ionization potentials: PANI > PANI-co-Pyr. PANI-co-Pyr will thus demonstrate the superior electron-donating ability and associated biological activities. The positive electronegativity (χ) and the negative chemical potential (μ) showed that the compounds could accept electrons from the environment, thereby decreasing their energy. Moreover, the negative value of chemical potential (−3.54 eV for PANI and −3.64 eV for PANI-co-Pyr) supports the stability of the compound and its resistance to decomposition into elemental forms. Additionally, the relatively high chemical softness (0.54) and low chemical hardness (0.93) of PANI-co-Pyr make it a promising candidate for biological applications. Using η and χ, the global electrophilicity index (ω) was computed, which measures the energy stabilization in the molecule. According to Parr et al., a molecule's electrophilic power can be calculated by dividing its chemical potential squared by its hardness.31 The electrophilicity index of PANI-co-Pyr was calculated to be 7.20, which is significantly higher than that of PANI (5.92), indicating the enhanced biological activity of PANI-co-Pyr. As a result, the determined electrophilicity index value supported the stability of PANI-co-Pyr in 1IX1 and 1UKC, as discussed in the molecular docking section.
 |
| | Fig. 6 Graphical representation of FMO, ΔE, IP, and ω. | |
3.8. Antibacterial activity
The antibacterial activity of PANI and PANI-co-Pyr copolymers was investigated against both Gram-positive bacteria S. aureus and B. subtilis, and Gram-negative bacteria E. coli and P. aeruginosa, at various concentrations (150, 130, 100, 80, 50, 30, 10, 1, 0.50, and 0.25 μg mL−1). The percentage of activity was measured, and based on this, IC50 values and MIC values were calculated. The antibacterial properties of PANI, PANI-co-Pyr, and amoxicillin were calculated in terms of MIC and IC50. The MIC of PANI was found to be 0.5 μg mL−1, and the MICs of PANI-co-Pyr and amoxicillin were found to be 0.25 μg mL−1 against S. aureus, B. subtilis, E. coli, and P. aeruginosa, respectively, as shown in Fig. S2. In addition, PANI and PANI-co-Pyr showed significant activity against E. coli with IC50 values of 221.56 μg mL−1 for PANI, 120.56 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 162.46 μg mL−1 for PANI-co-Pyr (1
1), 162.46 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and 155.97 μg mL−1 for PANI-co-Pyr (1
2), and 155.97 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). The standard drug amoxicillin shows excellent activity against B. subtilis with an IC50 value of 24.50 μg mL−1. In sum, from a comparison of the activities among different copolymers of PANI, PANI-co-Pyr (1
3). The standard drug amoxicillin shows excellent activity against B. subtilis with an IC50 value of 24.50 μg mL−1. In sum, from a comparison of the activities among different copolymers of PANI, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) indicated the most vigorous activity against Gram-negative bacteria E. coli. The results of the antibacterial properties of PANI and PANI-co-Pyr against S. aureus, B. subtilis, E. coli, and P. aeruginosa are illustrated in Table 6 & Fig. 7, respectively. The surface area of PANI and PANI-co-Pyr strongly influences their antibacterial activity. A higher BET surface area provides more active sites and greater surface contact between the copolymer and microbial cell walls. This enhanced interaction facilitates stronger binding, penetration, and disruption of microbial membranes, thereby improving antibacterial and antifungal activity. Thus, the observed increase in the antimicrobial performance of PANI-co-Pyr can be directly correlated with its higher BET surface area compared to pure PANI. Remarkably, PANI-co-Pyr copolymers showed greater antibacterial activity than PANI.
1) indicated the most vigorous activity against Gram-negative bacteria E. coli. The results of the antibacterial properties of PANI and PANI-co-Pyr against S. aureus, B. subtilis, E. coli, and P. aeruginosa are illustrated in Table 6 & Fig. 7, respectively. The surface area of PANI and PANI-co-Pyr strongly influences their antibacterial activity. A higher BET surface area provides more active sites and greater surface contact between the copolymer and microbial cell walls. This enhanced interaction facilitates stronger binding, penetration, and disruption of microbial membranes, thereby improving antibacterial and antifungal activity. Thus, the observed increase in the antimicrobial performance of PANI-co-Pyr can be directly correlated with its higher BET surface area compared to pure PANI. Remarkably, PANI-co-Pyr copolymers showed greater antibacterial activity than PANI.
Table 6 Antibacterial activities of PANI, PANI-co-pyrazine copolymers, and the standard drug amoxicillin against S. aureus, B. subtilis, E. coli, and P. aeruginosa
| S. no. |
Synthesized copolymers |
S. aureus
|
B. subtilis
|
E. coli
|
P. aeruginosa
|
| MIC (μg mL−1) |
IC50 (μg mL−1) |
MIC (μg mL−1) |
IC50 (μg mL−1) |
MIC (μg mL−1) |
IC50 (μg mL−1) |
MIC (μg mL)−1 |
IC50 (μg mL−1) |
| 1 |
PANI |
0.50 |
287.00 ± 18.87 |
0.50 |
286.74 ± 21.73 |
0.50 |
221.56 ± 7.11 |
0.50 |
382.72 ± 7.04 |
| 2 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
0.25 |
225.47 ± 17.01 |
0.25 |
233.07 ± 30.19 |
0.25 |
120.56 ± 9.01 |
0.25 |
220.09 ± 5.91 |
| 3 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
0.25 |
256.05 ± 53.04 |
0.25 |
218.77 ± 5.25 |
0.25 |
162.46 ± 10.61 |
0.25 |
241.81 ± 12.58 |
| 4 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
0.50 |
254.64 ± 34.81 |
0.50 |
250.79 ± 30.29 |
0.50 |
155.97 ± 13.51 |
0.25 |
191.73 ± 0.63 |
| 5 |
Amoxicillin |
0.25 |
58.25 ± 0.28 |
0.25 |
24.50 ± 2.55 |
0.25 |
69.10 ± 0.28 |
0.25 |
93.77 ± 2.28 |
 |
| | Fig. 7 Antibacterial activities of polyaniline and polyaniline-co-pyrazine copolymers and amoxicillin against S. aureus, B. subtilis, E. coli, and P. aeruginosa. | |
3.9. Antifungal activity
The antifungal activities of PANI and PANI-co-Pyr copolymers were investigated against A. niger, and P. chrysogenum at various concentrations (150, 130, 100, 80, 50, 30, 10, 1, 0.50, and 0.25 μg mL−1). The antifungal activities of the PANI, PANI-co-Pyr, and ketoconazole were calculated in terms of MIC and IC50. The MICs of PANI and PANI-co-Pyr and ketoconazole were found to be 0.5, 0.25, and 0.25 μg mL−1 against A. niger and P. chrysogenum, respectively, as shown in Fig. S3. In addition, the IC50 for PANI against A. niger were found to be 332.33 μg mL−1, 215.47 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 200.70 μg mL−1 for PANI-co-Pyr (1
1), 200.70 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), 202.31 μg mL−1 for PANI-co-Pyr (1
2), 202.31 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and 89.03 μg mL−1 for ketoconazole respectively, whereas the IC50 for PANI against P. chrysogenum was 395.76 μg mL−1, 230.78 μg mL−1 for PANI-co-Pyr (1
3), and 89.03 μg mL−1 for ketoconazole respectively, whereas the IC50 for PANI against P. chrysogenum was 395.76 μg mL−1, 230.78 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 207.89 μg mL−1 for PANI-co-Pyr (1
1), 207.89 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), 215.54 μg mL−1 for PANI-co-Pyr (1
2), 215.54 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and 117.45 μg mL−1 for ketoconazole. PANI-co-Pyr exhibited more significant antifungal activity than PANI due to the substitution of Pyr on the backbone of PANI. The antifungal activities of PANI and PANI-co-Pyr against A. niger, and P. chrysogenum are shown in Table 7 & Fig. 8, respectively. Notably, PANI-co-Pyr copolymers show greater antifungal activity than PANI. Statistical analysis of the results, which used ANOVA ONE-WAY, showed that there were highly significant differences observed in the MIC and IC50 values for antibacterial and antifungal activity, as shown in Tables S2 & S3.
3), and 117.45 μg mL−1 for ketoconazole. PANI-co-Pyr exhibited more significant antifungal activity than PANI due to the substitution of Pyr on the backbone of PANI. The antifungal activities of PANI and PANI-co-Pyr against A. niger, and P. chrysogenum are shown in Table 7 & Fig. 8, respectively. Notably, PANI-co-Pyr copolymers show greater antifungal activity than PANI. Statistical analysis of the results, which used ANOVA ONE-WAY, showed that there were highly significant differences observed in the MIC and IC50 values for antibacterial and antifungal activity, as shown in Tables S2 & S3.
Table 7 Antifungal activities of PANI, PANI-co-Pyr copolymers, and the standard drug ketoconazole against A. niger and P. chrysogenum
| S. no. |
Synthesized copolymers |
A. niger
|
P. chrysogenum
|
| MIC (μg mL−1) |
IC50 (μg mL−1) |
MIC (μg mL−1) |
IC50 (μg mL−1) |
| 1 |
PANI |
0.50 |
332.33 ± 6.64 |
0.50 |
395.76 ± 22.24 |
| 2 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) 1) |
0.25 |
215.47 ± 35.40 |
0.25 |
230.78 ± 4.97 |
| 3 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) 2) |
0.25 |
200.70 ± 9.05 |
0.25 |
207.89 ± 13.56 |
| 4 |
PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) 3) |
0.25 |
202.31 ± 1.93 |
0.25 |
215.54 ± 8.80 |
| 5 |
Ketoconazole |
0.25 |
91.04 ± 2.84 |
0.25 |
121.64 ± 5.92 |
 |
| | Fig. 8 Antifungal activities of polyaniline and polyaniline-co-pyrazine copolymers and ketoconazole against A. niger, and P. chrysogenum. | |
3.10. Molecular docking
To identify the most probable binding site, a molecular-docking study of PANI and PANI-co-Pyr with 1IX1 and 1UKC was performed using a molecular-docking technique. Notably, PANI and PANI-co-Pyr exist in linear form upon optimization. With docking in 1IX1 and 1UKC, they underwent a significant conformational change from the linear to the angular form. The docking structure (Fig. S4) of PANI in 1IX1 reveals that the ligand is surrounded by residues such as His134, Glu135, Cys131, Gly44, Gly46, Pro43, Ile45, Glu8, Ile6, Asn5, Leu4, Ile3, Glu41, Pro96, Gly97, Tyr99, Leu93, Gly91, Cys92. The free energy of the PANI-1IX1 complex is −7.39 kcal mol−1. The terminal amine of PANI forms two hydrogen bonds with His134 and Glu135 with interaction distances/energy of 3.46 Å/−1.6 kcal mol−1 and 3.15 Å/1.9 kcal mol−1, respectively. Also, the π cloud of PANI interacted through π-H interactions with a distance/energy of 4.13 Å/−0.5 kcal mol−1.
On the other hand, PANI-co-Pyr in 1IX1 reveals that the ligand is surrounded by residues such as Glu90, Gly44, Ile45, Leu93, Pro96, Pro43, Gly97, Ala42, Asn5, Gly8, His164, Ile6, Leu7, Ala168, Gln167, His170, NME171, Cys131, Tyr99, Val130, Cys92, Glu135, Gly91, and His134. The free energy of the PANI-co-Pyr-1IX1 complex is −9.32 kcal mol−1, as shown in Fig. 9.
 |
| | Fig. 9 Docked pose of PANI-co-Pyr in a 1IX1 cavity with the ligand surface. The proximity of amino acid residues in the 3D and 2D presentations (left to right). | |
On the other hand, PANI and PANI-co-Pyr in 1UKC revealed that the ligand is surrounded by residues, as shown in Fig. 10 and 11. Furthermore, the PANI-co-Pyr copolymer is stabilized by hydrogen bonding interactions, as listed in the table below. The PANI and PANI-co-Pyr copolymers’ binding energies, surrounding residues, and hydrogen bonding interactions for protein 1IX1 and 1UKC are shown in Table 8. In both cases (1IX1 and 1UKC), PANI-co-Pyr exhibited greater stability compared to PANI. This enhanced stability is primarily due to the presence of additional hydrogen bonding in the PANI-co-Pyr complexes docked with 1IX1 and 1UKC. Notably, the pyrazine unit within PANI-co-Pyr also engaged in intermolecular hydrogen bonding, which played a significant role in molecular stabilization and further contributed to increased biological activity.
 |
| | Fig. 10 Docked pose of PANI in a 1UKC cavity with the ligand surface. The proximity of amino acid residues in the 3D and 2D presentations (left to right). | |
 |
| | Fig. 11 Docked pose of PANI-co-Pyr in a 1UKC cavity with the ligand surface. The proximity of amino acid residues in the 3D and 2D presentation (left to right). | |
Table 8 PANI and PANI-co-Pyr binding energies, residues, and interactions with proteins 1IX1 and 1UKC
| Protein ID |
Synthesized copolymers |
Binding energy (kcal mol−1)/RMSD |
Surrounding residues (in the proximity of 4.5 Å) |
Hydrogen bonding interaction |
|
1IX1
|
PANI |
−7.39/2.246 |
His134, Glu135, Cys131, Gly44, Gly46, Pro43, Ile45, Glu8, Ile6, Asn5, Leu4, Ile3, Glu41, Pro96, Gly97, Tyr99, Leu93, Gly91, and Cys92 |
N28⋯GLU135 [3.15 Å; −1.9 kcal mol−1] |
| N28⋯HIS134 [3.46 Å; −1.6 kcal mol−1] |
| 6-π⋯ASN5 [4.13 Å; −0.5 kcal mol−1] |
| PANI-co-Pyr |
−9.32/1.350 |
Glu90, Gly44, Ile45, Leu93, Pro96, Pro43, Gly97, Ala42, Asn5, Gly8, His164, Ile6, Leu7, Ala168, Gln167, His170, NME171, Cys131, Tyr99, Val130, Cys92, Glu135, Gly91, and His134 |
C55⋯HIS164 [3.89 Å; −0.5 kcal mol−1] |
| 6π⋯ILE45 [4.65 Å; −0.5 kcal mol−1] |
| 6π⋯ALA168 [4.59 Å; −0.5 kcal mol−1] |
| 6π⋯HIS170 [4.64 Å; −0.7 kcal mol−1] |
|
1UKC
|
PANI |
−7.08/2.102 |
Thr401, Asp335, Thr336, Arg423, Ser521, Asp334, Leu381, Gly389, Asp337, Lys379, Ala393, Ala390, Val374, Pro376, Gly378, Tyr375, Arg377, Asp525, Asp397, Ser522, and Arg505 |
6π⋯THR336 [4.23 Å; −0.6 kcal mol−1] |
| PANI-co-Pyr |
−9.36/1.425 |
Gln509, Glu516, Arg423, Thr336, Pro519, Ser521, Asp397, Ser340, Arg507, Asn425, Asp337, Ile436, Ser392, Gly437, Leu381, Gly435, Ala393, Gly378, Lys379, Gly389, Pro376, Val374, Tyr375, Asp525, Ser522, Thr401, Arg505, and Asp335 |
6π⋯LYS379 [3.40 Å; −0.6 kcal mol−1] |
| 6π⋯ARG423 [3.23 Å; −0.7 kcal mol−1] |
4. Conclusion
PANI and PANI-co-Pyr copolymers co-doped with dual acids were synthesized using the COPM in a single pot. The structural changes in synthesized PANI and PANI-co-Pyr were validated by UV-visible and FTIR spectroscopy. TGA analysis was used to investigate the thermal stability of the synthesized PANI and PANI-co-Pyr at temperatures ranging from 33 °C to 1000 °C. The viscosity average molar mass was studied by viscosity measurements and found to be higher for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) with 9.48 × 104 g mol−1 than for pure PANI (7.66 × 104 g mol−1). By BET analysis, we found that the surface area increased when pyrazine was incorporated into the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). Conductivity measurements were studied using the two-probe method, and it was found that PANI-co-Pyr (1
2) with 9.48 × 104 g mol−1 than for pure PANI (7.66 × 104 g mol−1). By BET analysis, we found that the surface area increased when pyrazine was incorporated into the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). Conductivity measurements were studied using the two-probe method, and it was found that PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) has a higher conductivity of 5.387 × 10−3 S cm−1 than pure PANI (4.2016 × 10−3 S cm−1). According to DFT, the stabilities of PANI and PANI-co-Pyr are supported by the positive values of ΔE and the negative eigenvalues of HOMO and LUMO. The band gaps of PANI and PANI-co-Pyr were obtained experimentally (3.36 & 2.10 eV) by conductance measurements and theoretically (2.11 & 1.85 eV), as confirmed by DFT. The observed results follow the same trends and show that PANI-co-Pyr has high electrical conductance due to a smaller band gap (2.11 & 1.85 eV) with an increase in conjugation as compared to PANI (3.36 & 2.11 eV). The MIC and IC50 of PANI were found to be 0.5 and 221.56 ± 7.11 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1
2) has a higher conductivity of 5.387 × 10−3 S cm−1 than pure PANI (4.2016 × 10−3 S cm−1). According to DFT, the stabilities of PANI and PANI-co-Pyr are supported by the positive values of ΔE and the negative eigenvalues of HOMO and LUMO. The band gaps of PANI and PANI-co-Pyr were obtained experimentally (3.36 & 2.10 eV) by conductance measurements and theoretically (2.11 & 1.85 eV), as confirmed by DFT. The observed results follow the same trends and show that PANI-co-Pyr has high electrical conductance due to a smaller band gap (2.11 & 1.85 eV) with an increase in conjugation as compared to PANI (3.36 & 2.11 eV). The MIC and IC50 of PANI were found to be 0.5 and 221.56 ± 7.11 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were found to be 0.25 and 120.56 ± 9.01 μg mL−1 against E. coli, showing significant activity against E. coli. The MIC and IC50 of PANI were found to be 0.5 and 332.33 ± 6.64 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1
1) were found to be 0.25 and 120.56 ± 9.01 μg mL−1 against E. coli, showing significant activity against E. coli. The MIC and IC50 of PANI were found to be 0.5 and 332.33 ± 6.64 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) were found as 0.25 and 200.70 ± 9.05 μg mL−1, showing significant activity against A. niger. The results suggest that the synthesized co-doped PANI-co-Pyr exhibits more antifungal and antibacterial activity than pure PANI. According to molecular docking, the PANI-co-Pyr copolymer had a greater affinity for binding (-9.36 kcal mol−1) to a protein and showed superior antibacterial and antifungal activity compared to PANI (-7.08 kcal mol−1). Herein, we have presented a one-pot synthesis of PANI-co-Pyr, highlighting its antifungal and antibacterial properties.
2) were found as 0.25 and 200.70 ± 9.05 μg mL−1, showing significant activity against A. niger. The results suggest that the synthesized co-doped PANI-co-Pyr exhibits more antifungal and antibacterial activity than pure PANI. According to molecular docking, the PANI-co-Pyr copolymer had a greater affinity for binding (-9.36 kcal mol−1) to a protein and showed superior antibacterial and antifungal activity compared to PANI (-7.08 kcal mol−1). Herein, we have presented a one-pot synthesis of PANI-co-Pyr, highlighting its antifungal and antibacterial properties.
Author contributions
Chetna Kumari: Writing – original draft, Investigation, Gulshan Kumar: Analysis, Sapana Jadoun: Writing – review & editing, Nirmala Kumari Jangid: Writing – review & editing, Supervision, and Conceptualization
Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Data availability
The data supporting this article have been included as part of the SI. Supporting file includes mechanism of polymerization, data of DFT and molecular docking, and figure of BET, MIC values and molecular docking. See DOI: https://doi.org/10.1039/d5tb01302j.
Acknowledgements
The authors acknowledge Prof. Ina Aditya Shastri, Vice-Chancellor, Banasthali Vidyapith, Rajasthan, India, and DST-FIST India (Order Number SR/FST/2022/252), for providing all the necessary facilities for the successful accomplishment of the present work.
References
- S. Bhadra, D. Khastgir, N. K. Singha and J. H. Lee, Progress in preparation, processing, and applications of polyaniline, Prog. Polym. Sci., 2009, 34(8), 783–810, DOI:10.1016/j.progpolymsci.2009.04.003.
- I. Sapurina and J. Stejskal, The mechanism of the oxidative polymerization of aniline and the formation of supramolecular polyaniline structures, Polym. Int., 2008, 57(12), 1295–1325, DOI:10.1002/pi.
- S. J. Tang, K. Y. Lin, Y. R. Zhu, H. Y. Huang, W. F. Ji, C. C. Yang and K. C. Chiu, Structural and electrical characterization of polyanilines synthesized from chemical oxidative polymerization via doping/de-doping/re-doping processes, J. Phys. D: Appl. Phys., 2013, 46(50), 505301, DOI:10.1088/0022-3727/46/50/505301.
- L. Zhang, Z. D. Zujovic, H. Peng, G. A. Bowmaker, P. A. Kilmartin and J. Travas-Sejdic, Structural characteristics of polyaniline nanotubes synthesized from different buffer solutions, Macromolecules, 2008, 41(22), 8877–8884, DOI:10.1021/ma801728j.
- G. Liao, Q. Li and Z. Xu, The chemical modification of polyaniline with enhanced properties: A review, Prog. Org. Coat., 2019, 126, 35–43, DOI:10.1016/j.porgcoat.2018.10.018.
- H. Bejbouji, L. Vignau, J. L. Miane, M. T. Dang, E. M. Oualim, M. Harmouchi and A. Mouhsen, Polyaniline as a hole injection layer on organic photovoltaic cells, Sol. Energy Mater. Sol. Cells, 2010, 94(2), 176–181, DOI:10.1016/j.solmat.2009.08.018.
- G. Illing, K. Hellgardt, R. J. Wakeman and A. Jungbauer, Preparation and characterization of polyaniline-based membranes for gas separation, J. Membr. Sci., 2001, 184(1), 69–78, DOI:10.1016/S0376-7388(00)00606-2.
- T. Schauer, A. Joos, L. Dulog and C. D. Eisenbach, Protection of iron against corrosion with polyaniline primers, Prog. Org. Coat., 1998, 33(1), 20–27, DOI:10.1016/S0300-9440(97)00123-9.
- P. Stamenov, R. Madathil and J. M. D. Coey, Dynamic response of ammonia sensors constructed from polyaniline nanofiber films with varying morphology, Sens. Actuators, B, 2012, 161(1), 989–999, DOI:10.1016/j.snb.2011.11.082.
- M. R. Gizdavic-Nikolaidis, J. R. Bennett, S. Swift, A. J. Easteal and M. Ambrose, Broad-spectrum antimicrobial activity of functionalized polyanilines, Acta Biomater., 2011, 7(12), 4204–4209, DOI:10.1016/j.actbio.2011.07.018.
- N. Shi, X. Guo, H. Jing, J. Gong, C. Sun and K. Yang, Antibacterial Effect of the Conducting Polyaniline, J. Mater. Sci. Technol., 2006, 22(03), 289 CAS.
- R. G. S. V. Prasad, K. S. V. Chaitanya, M. Tejoram, D. Basavaraju, K. N. Rao, R. R. Kumar, S. Sreenivasan and A. R. Phani, Antibacterial properties of nanofiber structured conducting polyaniline synthesized by cost effective wet chemical process, J. Pharm. Res., 2012, 5(1), 370–373 Search PubMed.
- A. N. Andriianova, Y. N. Biglova and A. G. Mustafin, Effect of structural factors on the physicochemical properties of functionalized polyanilines, RSC Adv., 2020, 10(13), 7468–7491, 10.1039/C9RA08644G.
- L. R. Latypova, A. N. Andriianova, S. M. Salikhov, I. N. Mullagaliev, R. B. Salikhov, I. B. Abdrakhmanov and A. G. Mustafin, Synthesis and physicochemical properties of poly [2-(2-chloro-1-methylbut-2-en-1-yl) aniline] obtained with various dopants, Polym. Int., 2020, 69(9), 804–812, DOI:10.1002/pi.6016.
- M. Maruthapandi, A. Saravanan, J. H. Luong and A. Gedanken, Antimicrobial properties of the polyaniline composites against Pseudomonas aeruginosa and Klebsiella pneumoniae, J. Funct. Biomater., 2020, 11(3), 59, DOI:10.3390/jfb11030059.
- X. Liang, M. Sun, L. Li, R. Qiao, K. Chen, Q. Xiao and F. Xu, Preparation and antibacterial activities of polyaniline/Cu 0.05 Zn 0.95 O nanocomposites, Dalton Trans., 2012, 41(9), 2804–2811, 10.1039/c2dt11823h.
- C. Dhivya, S. A. A. Vandarkuzhali and N. Radha, Antimicrobial activities of nanostructured polyanilines doped with aromatic nitro compounds, Arabian J. Chem., 2019, 12(8), 3785–3798, DOI:10.1016/j.arabjc.2015.12.005.
- P. Chaubisa, D. Dharmendra, Y. Vyas, P. Chundawat, C. Paliwal and C. Ameta, Biological activity of PANI-tetrazole and PANI-pyrazole composites against microbial strains, Discover Chem., 2025, 2(1), 1–13, DOI:10.1007/s44371-025-00127-w.
- N. K. Jangid, N. P. S. Chauhan, K. Meghwal, R. Ameta and P. B. Punjabi, Synthesis of dye-substituted polyanilines and study of their conducting and antimicrobial behavior, Cogent Chem., 2015, 1(1), 1084666, DOI:10.1080/23312009.2015.1084666.
- P. Chaubisa, D. Dharmendra, Y. Vyas, P. Chundawat, N. K. Jangid and C. Ameta, Synthesis and characterization of PANI and PANI-indole copolymer and study of their antimalarial and antituberculosis activity, Polym. Bull., 2024, 81(4), 3333–3353, DOI:10.1007/s00289-023-04873-8.
- C. Kumari, A. Singh, N. K. Jangid, J. Dwivedi and S. Sharma, Molecular docking and antibacterial activity of a one-pot synthesized novel polyaniline substituted imidazole (PANI–imidazole) copolymer, New J. Chem., 2025, 49(11), 4658–4667, 10.1039/D4NJ04331F.
- X. Quan, J. Wang, T. Souleyman, W. Cai, S. Zhao and Z. Wang, Antibacterial and antifouling performance of bisphenol-A/Poly (ethylene glycol) binary epoxy coatings containing bromine-benzyl-disubstituted polyaniline, Prog. Org. Coat., 2018, 124, 61–70, DOI:10.1016/j.porgcoat.2018.08.010.
- N. K. Jangid, N. P. S. Chauhan and P. B. Punjabi, Novel dye-substituted polyanilines: conducting and antimicrobial properties, Polym. Bull., 2014, 71, 2611–2630, DOI:10.1007/s00289-014-1210-6.
- W. Cai, J. Wang, X. Quan and Z. Wang, Preparation of bromo-substituted polyaniline with excellent antibacterial activity, J. Appl. Polym. Sci., 2018, 135(1), 45657, DOI:10.1002/app.45657.
- N. P. Chauhan, N. K. Jangid and P. B. Punjabi, Synthesis and characterization of conducting polyanilines via catalytic oxidative polymerization, Int. J. Polym. Mater. Polym. Biomater., 2013, 62(10), 550–555, DOI:10.1080/00914037.2012.761625.
- J. Muscat, A. Wander and N. M. Harrison, On the prediction of band gaps from hybrid functional theory, Chem. Phys. Lett., 2001, 342(3–4), 397–401, DOI:10.1016/S0009-2614(01)00616-9.
- J. C. Rienstra-Kiracofe, C. J. Barden, S. T. Brown and H. F. Schaefer, Electron affinities of polycyclic aromatic hydrocarbons, J. Phys. Chem. A, 2001, 105(3), 524–528, DOI:10.1021/jp003196y.
- A. Vektariene, G. Vektaris and J. Svoboda, A theoretical approach to the nucleophilic behavior of benzofused thieno [3, 2-b] furans using DFT and HF-based reactivity descriptors, Arkivoc, 2009, 7, 311–329 Search PubMed.
- U. Riaz, S. M. Ashraf, S. Aleem, V. Budhiraja and S. Jadoun, Microwave-assisted green synthesis of some nanoconjugated copolymers: characterization and fluorescence quenching studies with bovine serum albumin, New J. Chem., 2016, 40(5), 4643–4653, 10.1039/C5NJ02513C.
- M. Ayad and S. Zaghlol, Nanostructured crosslinked polyaniline with high surface area: Synthesis, characterization and adsorption for organic dye, Chem. Eng. J., 2012, 204, 79–86 CrossRef.
- R. G. Parr, L. V. Szentpály and S. Liu, Electrophilicity index, J. Am. Chem. Soc., 1999, 121(9), 1922–1924, DOI:10.1021/ja983494x.
|
| This journal is © The Royal Society of Chemistry 2025 |
Click here to see how this site uses Cookies. View our privacy policy here.  a,
Sapana
Jadoun
b and
Nirmala Kumari
Jangid
a,
Sapana
Jadoun
b and
Nirmala Kumari
Jangid
 *a
*a
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1
2, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), the COPM was used to synthesize co-doped PANI-co-Pyr. Here, 20 mL of hydrochloric acid (1.0 M) and 20 mL of trifluoroacetic acid (0.5 M) were added to an aqueous acidic medium containing 6 mL of aniline (0.0658 mol) and 5.16 g of pyrazine (0.644 mol). The solution was stirred at 800 rpm for 2 h at 0 °C in an ice bath. For the polymerization process, 100 mL of ammonium persulfate (1.0 M) was added dropwise to the solution at 0 °C, and the solution was stirred continuously at 800 rpm for 4 h. After 7 h, a dark green precipitate formed. The filtrate was separated by filtration and then washed with ethanol and distilled water to remove all impurities. The residue was oven-dried for 24 h at 50–60 °C to obtain a free-flowing powder of the PANI-co-Pyr. The resulting pure PANI-co-Pyr was found to be dark green after drying, with a yield of 73%. PANI-co-Pyr ratios of 1
3), the COPM was used to synthesize co-doped PANI-co-Pyr. Here, 20 mL of hydrochloric acid (1.0 M) and 20 mL of trifluoroacetic acid (0.5 M) were added to an aqueous acidic medium containing 6 mL of aniline (0.0658 mol) and 5.16 g of pyrazine (0.644 mol). The solution was stirred at 800 rpm for 2 h at 0 °C in an ice bath. For the polymerization process, 100 mL of ammonium persulfate (1.0 M) was added dropwise to the solution at 0 °C, and the solution was stirred continuously at 800 rpm for 4 h. After 7 h, a dark green precipitate formed. The filtrate was separated by filtration and then washed with ethanol and distilled water to remove all impurities. The residue was oven-dried for 24 h at 50–60 °C to obtain a free-flowing powder of the PANI-co-Pyr. The resulting pure PANI-co-Pyr was found to be dark green after drying, with a yield of 73%. PANI-co-Pyr ratios of 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2 and 1
2 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3 were also synthesized under similar circumstances. The synthesis of PANI-co-Pyr is shown in Scheme 2. The chemical oxidative polymerization process that converts PANI to PANI-co-Pyr is illustrated in Scheme S1.
3 were also synthesized under similar circumstances. The synthesis of PANI-co-Pyr is shown in Scheme 2. The chemical oxidative polymerization process that converts PANI to PANI-co-Pyr is illustrated in Scheme S1.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) showed a peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the peak at 617 nm in the visible region corresponded to polaronic transitions or indicated extended conjugation.29 Further, the UV-visible spectra of PANI-co-Pyr (1
1) showed a peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the peak at 617 nm in the visible region corresponded to polaronic transitions or indicated extended conjugation.29 Further, the UV-visible spectra of PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) and PANI-co-Pyr (1
2) and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) showed a significant peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the second peak at 584 nm in the visible region corresponded to polaronic transitions, as shown in Fig. 1. The UV-Vis spectra of PANI and PANI-co-Pyr also exhibited distinct changes in intensity. A hyperchromic shift was observed in PANI because PANI shows a high-intensity peak due to doping and conjugation. A slight hyperchromic shift was observed in the polaronic transition bands for the PANI-co-Pyr (1
3) showed a significant peak at 330 nm in the UV range correlated to the π–π* transitions. In comparison, the second peak at 584 nm in the visible region corresponded to polaronic transitions, as shown in Fig. 1. The UV-Vis spectra of PANI and PANI-co-Pyr also exhibited distinct changes in intensity. A hyperchromic shift was observed in PANI because PANI shows a high-intensity peak due to doping and conjugation. A slight hyperchromic shift was observed in the polaronic transition bands for the PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 and 1
1 and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), indicating enhanced conjugation due to the incorporation of Pyr into the PANI backbone. However, with increasing the Pyr ratio (1
2), indicating enhanced conjugation due to the incorporation of Pyr into the PANI backbone. However, with increasing the Pyr ratio (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), a hypochromic effect was observed, suggesting a reduction in polaron concentration and disrupted π-conjugation. This trend is consistent with the observed decrease in electrical conductivity and suggests that excess pyrazine interferes with the optimal electronic structure of PANI, as shown in Fig. 1.
3), a hypochromic effect was observed, suggesting a reduction in polaron concentration and disrupted π-conjugation. This trend is consistent with the observed decrease in electrical conductivity and suggests that excess pyrazine interferes with the optimal electronic structure of PANI, as shown in Fig. 1.
![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations of the aromatic ring were observed at 1484, 1490, 1490, and 1489 cm−1 for PANI, PANI-co-Pyr (1
C stretching vibrations of the aromatic ring were observed at 1484, 1490, 1490, and 1489 cm−1 for PANI, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), PANI-co-Pyr (1
1), PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and PANI-co-Pyr (1
2), and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. The C
3), respectively. The C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching vibrations of the benzenoid ring were observed at 1564, 1571, 1571, and 1571 cm−1 for PANI, PANI-co-Pyr (1
C stretching vibrations of the benzenoid ring were observed at 1564, 1571, 1571, and 1571 cm−1 for PANI, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), PANI-co-Pyr (1
1), PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and PANI-co-Pyr (1
2), and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. Pyr shifted the aromatic and benzenoid peaks, indicating altered conjugation (minor shift due to substitution effect). The stretching vibrations of C–N resulted in strong peaks at 1295, 1296, and 1294 cm−1 for PANI-co-Pyr ratios (1
3), respectively. Pyr shifted the aromatic and benzenoid peaks, indicating altered conjugation (minor shift due to substitution effect). The stretching vibrations of C–N resulted in strong peaks at 1295, 1296, and 1294 cm−1 for PANI-co-Pyr ratios (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1
2, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. The stretching vibrations of the N–H bands were observed at 3220 cm−1 for PANI. The Pyr nitrogen atoms influence hydrogen bonding, slightly shifting or broadening this peak. The observed peak of N–H also indicates that Pyr polymerizes through C with the N of PANI for PANI-co-Pyr ratios 1
3), respectively. The stretching vibrations of the N–H bands were observed at 3220 cm−1 for PANI. The Pyr nitrogen atoms influence hydrogen bonding, slightly shifting or broadening this peak. The observed peak of N–H also indicates that Pyr polymerizes through C with the N of PANI for PANI-co-Pyr ratios 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1, 1
1, 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, and 1
2, and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, respectively. The FTIR results of PANI and PANI-co-Pyr are shown in Table 1 and Fig. 2.
3, respectively. The FTIR results of PANI and PANI-co-Pyr are shown in Table 1 and Fig. 2.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), (c) polyaniline-co-pyrazine (1
1), (c) polyaniline-co-pyrazine (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and (d) polyaniline-co-pyrazine (1
2), and (d) polyaniline-co-pyrazine (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3).
3).![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)
2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3)
3)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C Stretching (aromatic ring)
C Stretching (aromatic ring)![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C Stretching (quinoid ring)
C Stretching (quinoid ring)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), PANI-co-Pyr (1
1), PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and PANI-co-Pyr (1
2), and PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) were observed between 20–130 °C, 17–126 °C, 17–152 °C, and 22–116 °C due to vaporizing H2O molecules. The second weight losses of 19.42%, 20.84%, 18.86%, and 18.3% were observed between 130–394 °C, 126–444 °C, 152–431 °C, and 116–391 °C, respectively, and are related to the loss of dopants. The third weight losses of 23.13%, 22.49%, 22.46%, and 27.49% occurred between 394–596 °C, 444–679 °C, 431–648 °C, and 391–671 °C due to the thermal breakdown of PANI-co-Pyr. The fourth weight losses of 12.85%, 7.8%, 10.33%, and 8.6% were observed at 596–996 °C, 980 °C, 982 °C, and 979 °C due to the continuous disintegration of the PANI-co-Pyr. Thermal behaviour supports the presence of Pyr in the copolymer.20,21 The graph indicates that PANI-co-Pyr is more thermally stable than pure PANI, as shown in Table 2 & Fig. 3.
3) were observed between 20–130 °C, 17–126 °C, 17–152 °C, and 22–116 °C due to vaporizing H2O molecules. The second weight losses of 19.42%, 20.84%, 18.86%, and 18.3% were observed between 130–394 °C, 126–444 °C, 152–431 °C, and 116–391 °C, respectively, and are related to the loss of dopants. The third weight losses of 23.13%, 22.49%, 22.46%, and 27.49% occurred between 394–596 °C, 444–679 °C, 431–648 °C, and 391–671 °C due to the thermal breakdown of PANI-co-Pyr. The fourth weight losses of 12.85%, 7.8%, 10.33%, and 8.6% were observed at 596–996 °C, 980 °C, 982 °C, and 979 °C due to the continuous disintegration of the PANI-co-Pyr. Thermal behaviour supports the presence of Pyr in the copolymer.20,21 The graph indicates that PANI-co-Pyr is more thermally stable than pure PANI, as shown in Table 2 & Fig. 3.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)
2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3)
3)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) exhibited the highest intrinsic viscosity (2.36 dL g−1) and molar mass (9.48 × 104 g mol−1), followed by PANI-co-Pyr (1
2) exhibited the highest intrinsic viscosity (2.36 dL g−1) and molar mass (9.48 × 104 g mol−1), followed by PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and (1
1) and (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). Interestingly, the PANI-co-Pyr (1
3). Interestingly, the PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3) composition showed a slightly lower molar mass compared to the (1
3) composition showed a slightly lower molar mass compared to the (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) and (1
1) and (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2). This reduction may be attributed to a less optimal ratio for chain extension, possibly resulting in fewer oligomer units linking effectively, and thus yielding a lower overall molar mass.
2). This reduction may be attributed to a less optimal ratio for chain extension, possibly resulting in fewer oligomer units linking effectively, and thus yielding a lower overall molar mass.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)
2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3)
3)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 33.19 m2 g−1 for PANI-co-Pyr (1
1), 33.19 m2 g−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and 33.10 m2 g−1 for PANI-co-Pyr (1
2), and 33.10 m2 g−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), respectively. However, the surface area of the PANI was found to be smaller (24.86 m2 g−1), as shown in Table 4. The surface area was increased by the presence of micropores in the PANI-co-Pyr. The nitrogen adsorption–desorption isotherms of polyaniline and polyaniline-co-pyrazine are shown in Fig. S1. Surface area increased when pyrazine was added to the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). The highest surface area represents a good sorption ability of materials.
3), respectively. However, the surface area of the PANI was found to be smaller (24.86 m2 g−1), as shown in Table 4. The surface area was increased by the presence of micropores in the PANI-co-Pyr. The nitrogen adsorption–desorption isotherms of polyaniline and polyaniline-co-pyrazine are shown in Fig. S1. Surface area increased when pyrazine was added to the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). The highest surface area represents a good sorption ability of materials.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)
2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3)
3)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ρ vs. 1000/T, and conductivity (b) log10
ρ vs. 1000/T, and conductivity (b) log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) σ vs. 1000/T of PANI and PANI-co-Pyr are shown in Fig. 4. The slope, band gap, and conductivity of PANI and PANI-co-Pyr are illustrated in Table 5.
σ vs. 1000/T of PANI and PANI-co-Pyr are shown in Fig. 4. The slope, band gap, and conductivity of PANI and PANI-co-Pyr are illustrated in Table 5.

![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ρ vs. 1000/T, and (b) log10
ρ vs. 1000/T, and (b) log10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) σ vs. 1000/T for polyaniline and polyaniline-co-pyrazine.
σ vs. 1000/T for polyaniline and polyaniline-co-pyrazine.![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)
2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3)
3)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 162.46 μg mL−1 for PANI-co-Pyr (1
1), 162.46 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), and 155.97 μg mL−1 for PANI-co-Pyr (1
2), and 155.97 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). The standard drug amoxicillin shows excellent activity against B. subtilis with an IC50 value of 24.50 μg mL−1. In sum, from a comparison of the activities among different copolymers of PANI, PANI-co-Pyr (1
3). The standard drug amoxicillin shows excellent activity against B. subtilis with an IC50 value of 24.50 μg mL−1. In sum, from a comparison of the activities among different copolymers of PANI, PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) indicated the most vigorous activity against Gram-negative bacteria E. coli. The results of the antibacterial properties of PANI and PANI-co-Pyr against S. aureus, B. subtilis, E. coli, and P. aeruginosa are illustrated in Table 6 & Fig. 7, respectively. The surface area of PANI and PANI-co-Pyr strongly influences their antibacterial activity. A higher BET surface area provides more active sites and greater surface contact between the copolymer and microbial cell walls. This enhanced interaction facilitates stronger binding, penetration, and disruption of microbial membranes, thereby improving antibacterial and antifungal activity. Thus, the observed increase in the antimicrobial performance of PANI-co-Pyr can be directly correlated with its higher BET surface area compared to pure PANI. Remarkably, PANI-co-Pyr copolymers showed greater antibacterial activity than PANI.
1) indicated the most vigorous activity against Gram-negative bacteria E. coli. The results of the antibacterial properties of PANI and PANI-co-Pyr against S. aureus, B. subtilis, E. coli, and P. aeruginosa are illustrated in Table 6 & Fig. 7, respectively. The surface area of PANI and PANI-co-Pyr strongly influences their antibacterial activity. A higher BET surface area provides more active sites and greater surface contact between the copolymer and microbial cell walls. This enhanced interaction facilitates stronger binding, penetration, and disruption of microbial membranes, thereby improving antibacterial and antifungal activity. Thus, the observed increase in the antimicrobial performance of PANI-co-Pyr can be directly correlated with its higher BET surface area compared to pure PANI. Remarkably, PANI-co-Pyr copolymers showed greater antibacterial activity than PANI.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)
2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3)
3)
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 200.70 μg mL−1 for PANI-co-Pyr (1
1), 200.70 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), 202.31 μg mL−1 for PANI-co-Pyr (1
2), 202.31 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and 89.03 μg mL−1 for ketoconazole respectively, whereas the IC50 for PANI against P. chrysogenum was 395.76 μg mL−1, 230.78 μg mL−1 for PANI-co-Pyr (1
3), and 89.03 μg mL−1 for ketoconazole respectively, whereas the IC50 for PANI against P. chrysogenum was 395.76 μg mL−1, 230.78 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1), 207.89 μg mL−1 for PANI-co-Pyr (1
1), 207.89 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2), 215.54 μg mL−1 for PANI-co-Pyr (1
2), 215.54 μg mL−1 for PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3), and 117.45 μg mL−1 for ketoconazole. PANI-co-Pyr exhibited more significant antifungal activity than PANI due to the substitution of Pyr on the backbone of PANI. The antifungal activities of PANI and PANI-co-Pyr against A. niger, and P. chrysogenum are shown in Table 7 & Fig. 8, respectively. Notably, PANI-co-Pyr copolymers show greater antifungal activity than PANI. Statistical analysis of the results, which used ANOVA ONE-WAY, showed that there were highly significant differences observed in the MIC and IC50 values for antibacterial and antifungal activity, as shown in Tables S2 & S3.
3), and 117.45 μg mL−1 for ketoconazole. PANI-co-Pyr exhibited more significant antifungal activity than PANI due to the substitution of Pyr on the backbone of PANI. The antifungal activities of PANI and PANI-co-Pyr against A. niger, and P. chrysogenum are shown in Table 7 & Fig. 8, respectively. Notably, PANI-co-Pyr copolymers show greater antifungal activity than PANI. Statistical analysis of the results, which used ANOVA ONE-WAY, showed that there were highly significant differences observed in the MIC and IC50 values for antibacterial and antifungal activity, as shown in Tables S2 & S3.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1)
1)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2)
2)![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3)
3)



![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) with 9.48 × 104 g mol−1 than for pure PANI (7.66 × 104 g mol−1). By BET analysis, we found that the surface area increased when pyrazine was incorporated into the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). Conductivity measurements were studied using the two-probe method, and it was found that PANI-co-Pyr (1
2) with 9.48 × 104 g mol−1 than for pure PANI (7.66 × 104 g mol−1). By BET analysis, we found that the surface area increased when pyrazine was incorporated into the PANI backbone (from 24.86 m2 g−1 to 34.63 m2 g−1). Conductivity measurements were studied using the two-probe method, and it was found that PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) has a higher conductivity of 5.387 × 10−3 S cm−1 than pure PANI (4.2016 × 10−3 S cm−1). According to DFT, the stabilities of PANI and PANI-co-Pyr are supported by the positive values of ΔE and the negative eigenvalues of HOMO and LUMO. The band gaps of PANI and PANI-co-Pyr were obtained experimentally (3.36 & 2.10 eV) by conductance measurements and theoretically (2.11 & 1.85 eV), as confirmed by DFT. The observed results follow the same trends and show that PANI-co-Pyr has high electrical conductance due to a smaller band gap (2.11 & 1.85 eV) with an increase in conjugation as compared to PANI (3.36 & 2.11 eV). The MIC and IC50 of PANI were found to be 0.5 and 221.56 ± 7.11 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1
2) has a higher conductivity of 5.387 × 10−3 S cm−1 than pure PANI (4.2016 × 10−3 S cm−1). According to DFT, the stabilities of PANI and PANI-co-Pyr are supported by the positive values of ΔE and the negative eigenvalues of HOMO and LUMO. The band gaps of PANI and PANI-co-Pyr were obtained experimentally (3.36 & 2.10 eV) by conductance measurements and theoretically (2.11 & 1.85 eV), as confirmed by DFT. The observed results follow the same trends and show that PANI-co-Pyr has high electrical conductance due to a smaller band gap (2.11 & 1.85 eV) with an increase in conjugation as compared to PANI (3.36 & 2.11 eV). The MIC and IC50 of PANI were found to be 0.5 and 221.56 ± 7.11 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1) were found to be 0.25 and 120.56 ± 9.01 μg mL−1 against E. coli, showing significant activity against E. coli. The MIC and IC50 of PANI were found to be 0.5 and 332.33 ± 6.64 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1
1) were found to be 0.25 and 120.56 ± 9.01 μg mL−1 against E. coli, showing significant activity against E. coli. The MIC and IC50 of PANI were found to be 0.5 and 332.33 ± 6.64 μg mL−1, and the MIC and IC50 of PANI-co-Pyr (1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2) were found as 0.25 and 200.70 ± 9.05 μg mL−1, showing significant activity against A. niger. The results suggest that the synthesized co-doped PANI-co-Pyr exhibits more antifungal and antibacterial activity than pure PANI. According to molecular docking, the PANI-co-Pyr copolymer had a greater affinity for binding (-9.36 kcal mol−1) to a protein and showed superior antibacterial and antifungal activity compared to PANI (-7.08 kcal mol−1). Herein, we have presented a one-pot synthesis of PANI-co-Pyr, highlighting its antifungal and antibacterial properties.
2) were found as 0.25 and 200.70 ± 9.05 μg mL−1, showing significant activity against A. niger. The results suggest that the synthesized co-doped PANI-co-Pyr exhibits more antifungal and antibacterial activity than pure PANI. According to molecular docking, the PANI-co-Pyr copolymer had a greater affinity for binding (-9.36 kcal mol−1) to a protein and showed superior antibacterial and antifungal activity compared to PANI (-7.08 kcal mol−1). Herein, we have presented a one-pot synthesis of PANI-co-Pyr, highlighting its antifungal and antibacterial properties.





