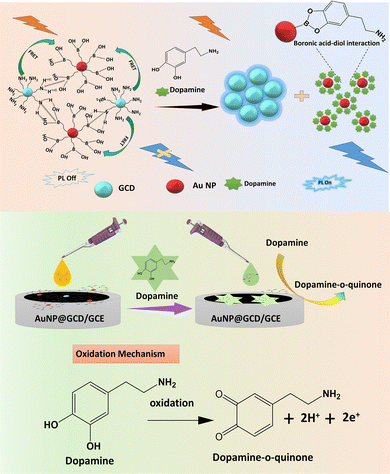
Hybrid AuNP@GCD nanosensor for dual-mode dopamine detection and imaging in the Drosophila brain†
Lingaraj
Behera
a,
Lopamudra
Mishra
b,
Monalisa
Mishra
bc and
Sasmita
Mohapatra
 *ac
*ac
aDepartment of Chemistry, National Institute of Technology Rourkela, Odisha 769008, India. E-mail: sasmitam@nitrkl.ac.in
bDepartment of Life Science, National Institute of Technology, Rourkela, 769008, India
cCentre for Nanomaterials, National Institute of Technology, Rourkela, 769008, Odisha, India
First published on 25th April 2025
Abstract
In this work, we constructed a surface passivated green carbon dot (GCD) integrated with a boronic acid functionalized gold nanoparticle (AuNP@GCD) FRET nanoprobe for the selective detection of dopamine through fluorescence and electrochemical dual methods. The intense blue FL emission of the GCD was decreased after integration with the AuNPs. The feeble FL emission of AuNP@GCDs gradually increased when it came in contact with dopamine owing to the strong diol–boronic acid interaction and restriction of energy transfer. Additionally, there was an 18-fold increment in the current response of the AuNP@GCD deposited electrode towards dopamine as compared to the bare glassy carbon electrode (GCE). The remarkable integration of conductance and fluorescence properties makes our probe suitable for the dual sensing of dopamine in human blood samples. The detection limits for the fluorescence and electrochemical detection are 0.0025 and 0.00026 μM, respectively. Furthermore, a detailed analysis of the toxicity induced by the probe and imaging properties in a fruit fly model (Drosophila melanogaster) suggests that the AuNP@GCD probe is safe and may further find application for dopamine imaging in the brain.
1. Introduction
Dopamine (DA) is a crucial neurotransmitter within the catecholamine family.1 It plays a significant role in many behavioural responses and brain activities and in the regulation of biological functions.2–4 Any disruption in the level of DA in the human brain can lead to various neurological diseases, including depression,5 Alzheimer's disease,6 Huntington's disease,7 Parkinson's disease,8 and schizophrenia.9 Meanwhile, the severity of these diseases is directly related to the extent of impairment in the DA system.10 The DA concentration in biological systems is typically 0–25 nM.11 To date, a number of methods, such as liquid chromatography (LC),12 mass spectrometry (MS),13 fluorescence,14 colorimetric analysis,15 and electrochemical techniques, have been developed to monitor the DA level in serum.16 However, these methods are not practical for the real-time monitoring of dopamine levels owing to the tedious sampling and time-consuming process.17 Significant advancements in nanomaterial research have led to the development of numerous nanoprobes that exhibit remarkable potential in diagnosing diseases at both the cellular and molecular levels.18 In contrast to other methods, electrochemical (EC) and fluorescence (FL) techniques have emerged as the frontrunners in chemosensing because of their affordability, ease of use, non-destructive nature, and quick response in measuring the analyte levels in a biological environment. The fluorescence method can provide a visual and real-time monitoring of dopamine. Electrochemical techniques also allow for the real-time recording of dopamine dynamics.19 In recent times, with the advancements in carbon dot (CD) research, a few FL and EC nanosensors have been reported. In such nanosensors, CDs are integrated with a semiconductor quantum dot,20 organic substances,21 MOFs,22 or conjugated polymers23 for the detection of dopamine, where the interaction of the CD-based probe and dopamine is built on distinct photophysical processes such as FL quenching, aggregation-induced emission (AIE), the inner filter effect (IFE), and fluorescence resonance energy transfer (FRET).24 While there have been a substantial number of works published on quantum dot (QD)-based FL methods for dopamine detection, most of these studies are associated with limitations in the detection range.25–27 Furthermore, most of the sensors are based on only either fluorescence or electrochemical sensing. In contrast, a dual-mode sensor that is fabricated by integrating different sensors into a single platform offers accuracy in identification over a wide concentration range. However, the main challenge to fabricating such a dual-mode sensor is (i) the feasibility of the process to simultaneously fabricate the two types of sensors, and (ii) to maintain the structural integrity while keeping the individual sensor's function intact. On the other side, based on the big Stokes shift (250–700 nm), gold nanoparticles (AuNPs) have been extensively used in biosensing. More specifically, boronic acid-functionalized gold nanocluster (BA-AuNC),28 AuNP@QD,29 and AuNP supported multiwalled CNT30 have been examined for the FL signal off–on detection of dopamine. AuNPs are excellent oxidising materials for the electrochemical detection of various ions due to their high specific surface area, superior electron conductivity, and excellent catalytic properties.31 It plays a crucial role in the electron transfer ability and in enhancing the voltammetric detection of analytes. The synergistic cooperation of AuNP and CDs may lead to the sensitive and selective electrochemical sensing of dopamine.32,33 Relying on the spectral overlap of CD with the absorption spectrum of AuNP and the ability of AuNP to oxidize dopamine, a multipurpose sensor probe can be constructed by integrating AuNP and carbon dot through appropriate surface modulations.In this work, a feeble blue fluorescent AuNP@GCD has been designed by integrating an EDEA-passivated carbon dot with 3-aminophenyl boronic acid modified on gold nanoparticles. When it comes in contact with dopamine, the FL intensity of the carbon dot appreciably increases linearly in a concentration-dependent manner in the range of 5 to 660 nM. At the same time, the AuNP@GCD nanoprobe exhibits a quick electrochemical response towards dopamine. Interestingly, the probe can be applied for the visualization of dopamine in the brain of a model fruit fly Drosophila through FL bioimaging. The detection of dopamine in both EC and FL modes using a single probe, along with the ex vivo imaging in a model organism, is a promising development that could have been possible for the conducting and luminescent features of the AuNP@GCD probe. It is the first paper to report on the dual-mode detection of dopamine and successful demonstration of dopamine imaging ex vivo; hence, this study may further attract research interest in in vivo neurotransmitter imaging.
2. Experimental
2.1. Reagents and materials
Chloroauric acid (HAuCl4·4H2O), 3-aminophenyl boronic acid (3-APBA), and dopamine (DA) were brought from Sigma Aldrich. The red cabbage was brought from the daily market of Rourkela. Glycine (Gly), methionine (Met), tryptophan (Try), threonine (Thr), aspartic acid (Asp), alanine (Ala), valine (Val), cysteine (Cys), arginine (Arg), histamine (His), serine (Ser), and glutamic acid (Glu) were procured from Thermo Scientific Pvt. Ltd. 2,2′-(Ethylenedioxy) bis(ethylamine) (EDEA) was obtained from Merck. Serum samples were collected from Ispat General Hospital, Rourkela.2.2. Synthesis of AuNP@GCD
The synthesis procedure of the green carbon dot (GCD), AuNP and 3-APBA modified AuNP has been described in the ESI.† 20 mg of each of the EDEA-modified GCD was taken in 20 mL Millipore water. After being stirred for 10 min, 5 mL of the suspension of GCD was withdrawn from it. A certain volume of boronic acid functionalized AuNPs was added dropwise into the GCD solution to obtain a turbid solution. Turbidity appears because of the instant agglomeration due to the lowering of the pH condition. However, after the addition of 0.1 M NaOH, there is stabilization of the carbon dot due to the conversion of the surface groups to –COO– and –NH2 groups. The solution was stirred at 30 °C in the dark for 120 minutes. The synthesized AuNP@GCD probe was then purified by a porous dialysis membrane filter (0.22 μm), and kept at 4 °C for further experiments (Scheme 1).2.3. Detection of dopamine by fluorescence
A standard solution with a conc. of 2 μg mL−1 was utilized for all sensing experiments. Each vial was filled with 3 mL of the probe solution containing certain dopamine concentrations. The mixtures were thoroughly stirred and shaken for 15 minutes at a temperature of 25 °C. The emission wavelength for each solution was measured (λex = 360 nm). To investigate the selectivity of dopamine at 25 °C, a freshly synthesized AuNP@GCD solution was mixed with various amino acids (arginine, methionine, histamine, glycine, alanine, serine, tyrosine, aspartic acid, lysine, valine, epinephrine, norepinephrine, and threonine), carbohydrates (fructose, glucose, cellulose, starch, sucrose, cations (Cu2+, Pb2+, Cd2+, Fe3+, Hg2+, Na+, K+)) and anions (SO42−, Cl−, Br−, PO43−, HCO3−). Dopamine detection in a serum sample is described in the ESI.†2.4. Electrochemical dopamine sensing
The EC detection of glycine, cysteine, dopamine, histamine, alanine, threonine, arginine, methionine, serine, tyrosine, epinephrin, norepine–phrine, aspartic acid and lysine on the AuNP@GCD/GCE electrode was examined by analyzing DPV curves. All studies were conducted using the PGSTAT 302N electrochemical workstation, AUT 8650A (Metrohm, Autolab). A three-electrode system was employed, consisting of a Ag(s)/AgCl(s)/Cl−1 (aq.) in 0.1 M PBS at pH 7 as the reference electrode, a glassy carbon electrode (GCE) as the working electrode, and platinum wire as the counter electrode. DPV was examined in the potential range from 0 to 0.8 V with the following conditions: the amplitude range of 10–100 mV and a pulse width range of 20–200 ms.2.5. Ex vivo dopamine imaging in a Drosophila brain
All physical behavior studies of the Oregon R strain of Drosophila melanogaster were examined before checking the dopamine levels in the brain (ESI†). The brain was isolated and fixed with PFA for 30 min. It was then treated with DMSO for 5 min, and kept in 4% PBST-BSA overnight in a shaker at 4 °C. The next day, PBST-BSA was removed and the sample was stained with 2 mg mL−1 AuNP@GCD solution for 1 h under dark conditions. It was then rinsed with 1X PBS and mounted on a clean slide with glycerol. Brain images were obtained by using a fluorescence confocal microscopy.3. Results and discussion
3.1. Structural analysis of AuNP@GCD
The morphology and microstructure of the AuNPs, GCD, and AuNP@GCD nanohybrid were characterized by transmission electron microscopy (TEM). The TEM image displays the uniform distribution of GCD with an average particle size of 3–5 nm (Fig. S1, ESI†). The corresponding high-resolution TEM image shows a lattice fringe of 0.22 nm indexed to the (002) plane of GCD, as reported previously.34 Furthermore, the XRD spectra of GCD clearly evidenced the formation of the (002) plane from the broad peak formed at 23.7° (Fig. S2, ESI†).35 A dynamic light scattering (DLS) study was conducted at room temperature (RT), which revealed that the average hydrodynamic diameters (HD) of GCD and AuNP@GCD were about 29 and 102 nm, respectively (Fig. S3a and b, ESI†). In Fig. S4a (ESI†), TEM images show the uniform solution of AuNPs within a size range of 2–10 nm. The high-resolution TEM image of AuNPs (inserts in Fig. S4a, ESI†) reveal that it shows crystal planes with a lattice spacing of 0.23 nm, consistent with Au (111). The TEM image shows the formation of the AuNP@GCD probe over a large dimension range of 10–30 nm (Fig. S4b, ESI†). The HRTEM image of AuNP@GCD confirms that the lattice spacing was 0.22 nm and 0.23 nm, which correspond to the (002) lattice plane of the carbon dot and (111) plane of AuNP, respectively (Fig. S4c and d, ESI†). The XRD pattern of the AuNP@GCD hybrid (Fig. S2, ESI†) reveals a peak at about 23.7°, referred to the graphitic plane of GCD (JCPDS No. 01-0646) and the peaks at 2θ = 39.2° and 45.9°, which are attributed to the (111), (200) planes of Au, respectively (JCPDS No. 04-0783). Addition of AuNP induces particle aggregation, which is evidenced from the particle size measurement using dynamic light scattering. When the concentration of AuNP increases from 0 to 25 μM, the AuNP@GCD aggregate size increases from 29 nm to 437 nm. From the AFM image, the topographic height of AuNP@GCD has been measured as 43.4 nm (Fig. S4e and f, ESI†), which complements the TEM result. The existence of elements N, C, O, B, Au in EDS in a reasonable amount, as well as in the elemental mapping, shows the formation of the AuNP@GCD (Fig. S5a and b, ESI†). A broad absorption in the range from 3100 cm−1 to 3500 cm−1 is ascribed to the stretching vibrations of the –N–H and O–H bonds. The symmetric and asymmetric H–C–H stretching appears at 2880 and 2956 cm−1 in both GCD and AuNP@GCD conjugate due to the presence of EDEA. In both cases, the peak that appears in the range between 1575 cm−1 and 1780 cm−1 is ascribed to the –NH bending vibration. Aromatic C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C and overtones are found in the frequency region of 1300 to 1565 cm−1. The B–OH bending peak also falls within the same region. From FTIR, it is not possible to conclude any particular interaction among GCD and AuNP (Fig. S6, ESI†). XPS gives further insight into the formation of the AuNP@GCD integrated structure. The XPS survey spectrum was examined to analyze the elemental composition of the AuNP@GCD composite (Fig. S7a, ESI†). The XPS survey peak shows the existence of C 1s, O 1s, N 1s, B 1s, and Au 4f peaks at 284.7, 531.9, 399.8, and 162.7, 84.5 eV, respectively. The high-resolution C 1s scan can be deconvoluted into four peaks centred at 288.3, 286.4, 284.2, and 282.6, indexed to C
C and overtones are found in the frequency region of 1300 to 1565 cm−1. The B–OH bending peak also falls within the same region. From FTIR, it is not possible to conclude any particular interaction among GCD and AuNP (Fig. S6, ESI†). XPS gives further insight into the formation of the AuNP@GCD integrated structure. The XPS survey spectrum was examined to analyze the elemental composition of the AuNP@GCD composite (Fig. S7a, ESI†). The XPS survey peak shows the existence of C 1s, O 1s, N 1s, B 1s, and Au 4f peaks at 284.7, 531.9, 399.8, and 162.7, 84.5 eV, respectively. The high-resolution C 1s scan can be deconvoluted into four peaks centred at 288.3, 286.4, 284.2, and 282.6, indexed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, C–OH, C–N/C–O, and C
O, C–OH, C–N/C–O, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/C–C, respectively (Fig. S7b, ESI†).36 The high-resolution O 1s spectrum shows four peaks at 533.6, 533.2, 531.9, and 531.2 eV, which are indexed to O–B, C–O–C, C–OH, and C
C/C–C, respectively (Fig. S7b, ESI†).36 The high-resolution O 1s spectrum shows four peaks at 533.6, 533.2, 531.9, and 531.2 eV, which are indexed to O–B, C–O–C, C–OH, and C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O, respectively (Fig. S7c, ESI†).37 The N 1s high-resolution scan reveals five peaks at 403.5, 402.6, 399.9, 398.2, and 397.1 eV ascribed to the protonated amine (N–H+, N⋯B, graphitic, pyrrrolic, and pyridinic nitrogens) (Fig. S7d, ESI†).38 The B 1s scan shows two main peak at 190.7 and 192.5 eV indexed to B–O–H and B⋯N, respectively (Fig. S7e, ESI†). The region for Au 4f can be deconvoluted into 88.6 and 83.2 eV (Fig. S7f, ESI†) peaks, which are attributed to Au 4f5/2 and Au 4f7/2, respectively.39 From the analysis of the N 1s region, the development of two peaks at higher binding energies of 403.5 eV and 402.6 eV, clearly shows the presence of protonated amines, as well as amine bonded to boron through N and B interaction.
O, respectively (Fig. S7c, ESI†).37 The N 1s high-resolution scan reveals five peaks at 403.5, 402.6, 399.9, 398.2, and 397.1 eV ascribed to the protonated amine (N–H+, N⋯B, graphitic, pyrrrolic, and pyridinic nitrogens) (Fig. S7d, ESI†).38 The B 1s scan shows two main peak at 190.7 and 192.5 eV indexed to B–O–H and B⋯N, respectively (Fig. S7e, ESI†). The region for Au 4f can be deconvoluted into 88.6 and 83.2 eV (Fig. S7f, ESI†) peaks, which are attributed to Au 4f5/2 and Au 4f7/2, respectively.39 From the analysis of the N 1s region, the development of two peaks at higher binding energies of 403.5 eV and 402.6 eV, clearly shows the presence of protonated amines, as well as amine bonded to boron through N and B interaction.
3.2. Optical properties of GCD and AuNP@GCD
UV-visible absorption and PL spectroscopy were used to analyse the optical properties of GCD and AuNP@GCD. The absorption spectrum of GCD shows two bands at 240 nm and 280 nm assigned to the π–π* and n–π* transitions, respectively (Fig. S8a, ESI†). GCD shows the maximum emission intensity at 430 nm when excited at 360 nm with a PL quantum yield of 16.34%, and the solution shows blue FL that is visible to the bare eye under a UV lamp (Fig. S8b, ESI†). After the modification of EDEA, the fluorescence intensity of GCD increases due to the surface passivation (Fig. S8c, ESI†). The ability of boronic acid functionalised AuNP to change the FL properties of GCD is clear from the overlap of the emission spectrum of GCD and the absorption spectrum of AuNP. Consequently, the FL emission of GCD at 430 nm decreased rapidly with the increment of the AuNP concentration until 19 μM, and then remained flat with increasing AuNP up to 25 μM (Fig. S8d, ESI†). It should be noted that AuNPs are stable for up to 3 h as evident from the UV absorption peak taken at different time intervals and the synthesized AuNP@GCD exhibited robust fluorescence stability for a duration of up to 10 days. (Fig. S9a and b, ESI†). All the sensing parameters are optimised as per the procedure and results are mention in the Fig. S10 (ESI†).3.3. Fluorescence sensing of dopamine
It was observed that with continuously increasing dopamine concentrations, the FL intensity of AuNP@GCD rapidly increased at 430 nm (Fig. 1a and b), indicating the recognition of the AuNP@GCD probe towards dopamine. These results confirmed the interaction of dopamine with AuNP@GCD, which is believed to form dopaquinone. As the linear regression between the relative FL intensity (F/F0) and the dopamine concentration was shown to be within the concentration range of 5 nM to 660 nM. The limit of detection (LOD) was determined by analysing the standard deviation from three consecutive blank readings. In response to dopamine ions, the FL emission intensity of AuNP@GCD shows a linear relationship between 5 nM and 310 nM, with a detection limit of 0.0025 μM, which is lower than previously reported literature. The practical applicability of the sensor is entirely hinged on its selectivity and sensitivity. The FL response of AuNP@GCD was investigated with other molecules like amino acids (AA) (Ala, Asp, Glu, Lys, Met, Try, His, Ser, Gly, Epi, Nepi, H2O2, glucose, fructose, ions (including Ca2+, Na+, K+, HCO3−, Mg2+)) under the same conditions to examine its selectivity for dopamine. FL intensity of AuNP@GCD in response to dopamine at 430 nm was unchanged despite the existence of other AA (Fig. 1c and d). These results confirmed the high selectivity and interference of AuNP@GCD for dopamine. The reproducibility of the system was examined by using the relative standard deviation (RSD) of several measurements. To evaluate the repeatability, a series of three solutions of AuNP@GCD with a constant concentration of dopamine (50 μM) were prepared, and detection was carried out on the same day. The calculated RSD% value was 0.51%, indicating good reproducibility of the assay using the AuNP@GCD nanoprobe. In addition, the stability of the proposed measurement was investigated over seven days of use for a particular concentration of dopamine (70 nM) solution. The calculated RSD% value was 1.23%, which demonstrates the stability of the proposed sensing protocol (Fig. 2).3.4. Fluorescence mechanism
To investigate the mechanism, the FL properties of GCDs and AuNPs were monitored. As illustrated in Fig. S11a (ESI†), the SPR absorption spectrum of AuNPs overlaps with the emission spectra of the GCD in 400–500 nm range, indicating the possibility of fluorescence resonance energy transfer (FRET) between GCD and AuNP. FRET takes place when the donor and acceptor are separated by a distance of less than 10 nm.40 In our case, the donor (GCD) and acceptor (AuNPs) are associated via several noncovalent interactions, as shown in Scheme 2. After construction of the AuNP@GCD conjugate, the distance between GCD and AuNPs is close enough to perform FRET, resulting in the quenching of the FL intensity of GCD. The amine group of aminophenyl boronic acid binds to surface Au atoms of AuNP, resulting in boronic acid functionalized AuNPs that bind with EDEA-functionalized GCD through hydrogen bonding and B–N interaction as evidenced from high resolution scan of N 1s and B 1s binding energy regions. When dopamine is added to the above FRET construct, dopamine molecules binds onto the surface of AuNPs through boronic acid–diol interaction, thus disintegrating AuNP@GCD and releasing GCD into the sensing medium, hence recovery of FL intensity of GCD is achieved. The formation and dissociation of AuNP@GCD is also evident from the hydrodynamic size measurement. The hydrodynamic (HD) size of GCD was increased from 29 nm to 437 nm after the addition of boronic acid functionalised AuNP. However, after the addition of dopamine in the concentration range of 5 nM to 310 nM, the HD size of the AuNP@GCD was decreased to 112 nm, which shows the disintegration of AuNP and GCD (Fig. S11b and c and Tables S1 and S2, ESI†). The fluorescence lifetime of GCDs and AuNP@GCD was examined to gain a deeper insight into the fluorescence quenching mechanism. As shown in Fig. S11d (ESI†), the FL lifetime of GCD was 7.2 ns, which decreased to 5.9 ns after the addition of AuNP. However, after the addition of dopamine, the FL life time increased to 6.3 ns, indicating the restriction of energy transfer. The details of the fluorescence life-time calculation has been explained in ESI.† The FRET efficiency (E) was calculated to be 24.7% using the following equation:| E = 1 − (td–a/td) | (1) |
t d–a = FL life time of AuNP@GCD (donor–acceptor)
We measured the donor–acceptor distance (r) to better understand the FRET mechanism. This was done by using FRET efficiency (E) and the Förster distance (R0) with the following equation.
| E = 1/[ 1 + (r/R0)6] (in Å unit) | (2) |
3.5. Application in real sample
The practical importance of this developed nanoprobe was verified in real blood serum samples through the fluorescence recovery test. The FL spectra of the AuNP@GCD solutions were recorded after adding human serum samples spiked with variable concentrations of dopamine. The amount of dopamine in real samples can be determined by utilising the linear correlation between F/F0 and dopamine. AuNP@GCD showed excellent detection recoveries between 100.09–101.43%, proving its effectiveness for detecting dopamine in real samples (Table S4, ESI†). The consistency of dopamine sensing method in blood serum was evaluated by subjecting it to freeze–thaw cycles at −70 °C for up to three cycles at 100, 150, and 200 nM. Additionally, instant stability was tested at ambient conditions. Before the experiment of real sample stability of the serum sample was analysed at room temperature (Table S5, ESI†).3.6. Electrochemical sensing of dopamine
The differential pulse voltammetry (DPV) method was used for the voltammetric detection of DA. The conductive properties of the AuNP led us to electrochemically monitor dopamine using AuNP@GCD.42 The initial study (Fig. S12, ESI†) shows one clear oxidation peak at 0.35 V for dopamine. The DPV profiles for DA using AuNP@GCD/GCE indicated 18 times increase in current response compared to bare GCE, GCD/GCE, and AuNP/GCE (Fig. 3a). It is anticipated that DA will participate in the redox reaction on the surface of AuNP@GCD via the established oxidation mechanism involving two electrons and two protons, as illustrated in Scheme 2.43 The increment in current for DA on AuNP@GCD/GCE may be assigned to synergistic effects of AuNP and GCDs due to the structural changes that happened during the preparation of AuNP@GCD and the consequent increment in the conducting and catalytic property of the AuNP@GCD. Thus, the increment in the current of AuNP@GCD/GCE toward dopamine can be ascribed to synergistic conductive properties of AuNP and enhanced interaction of the AuNP@GCD electrode towards dopamine through the π–π stacking interaction of GCD and the aromatic rings of dopamine. AuNPs serve as electrocatalytic materials that enhance the electron transfer efficiency and boost electrochemical performance.44 Moreover, GCDs contain numerous carboxyl groups, contributing to the material stability and facilitating interactions with DA via π–π stacking and electrostatic forces.45 The improved and selective detection of dopamine by AuNP@GCD was attributed to the synergistic effect of the conductive properties of AuNP and strong attraction of dopamine towards GCD. The DPV showed a steady rise in the current as the dopamine concentration increased, confirming that the AuNP@GCD probe is suitable for the quantitative response of dopamine (Fig. 3b). The graph depicting the relationship between the peak current and concentration at 0.35 V reveals a strong linear correlation with the DA concentration between 10−12 M to 9 × 10−2 M with a R2 of 0.998 (Fig. 3c). The robust linear regression equation for the response current has been expressed as follows:| Ipa (μA) = 0.0653x + 5.02585 × 10−6 (R2 = 0.998) | (3) |
3.7. Repeatability, reusability and stability
Reusability test of the AuNP@GCD/GCE sensor investigated the peak currents of dopamine on AuNP@GCD before and after cleaning (Fig. S14a, ESI†). The findings indicate that the current response was maintained at 98%. The DPV analysis response from five electrodes made with 0.1 M dopamine depicts the same current response, which confirm the reproducibility of the EC sensing technique. The peak currents of these separately made electrodes had a relative standard deviation of 1.24%, indicating the strong repeatability (Fig. S14b, ESI†). After 5 to 10 days, the current levels were reached at 90% and 82% of its initial current value, respectively (Fig. S14c, ESI†).3.8. Cytotoxicity assay on larval gut
The fruit flies (Drosophila melanogaster) were treated with AuNP@GCD, which directly interact with their intestinal epithelial cells as it passed through their intestines. The effects of AuNP@GCDs on cells was examined to assess the potential cytotoxic effects. DAPI was used to determine whether the AuNP@GCD caused any cell damage after the cell were exposed to harmful compounds. After staining larval intestines internally with DCFH-DA, a low level of ROS generation was observed in the control, 100 and 150 μg mL−1 groups (Fig. S15a–i, ESI†). In contrast, at the higher concentration of 200 μg mL−1, nuclear fragmentation is visible (Fig. S15m–p, ESI†). To verify the generation of ROS at higher concentration, the NBT assay was performed in the 3rd instar larval haemolymph (Fig. S16, ESI†). There was a quick increase in the ROS level when the amount of the probe was increased to 200 μg mL−1. These observations show that the AuNP@GCD nanoprobe is tolerable up to a concentration of 150 μg mL−1 in Drosophila melanogaster.3.9. Phenotype study
The wing vein pattern, eye color and bristle count on the thorax were examined to determine any phenotypic changes resulting from the nanocomposite treatment. The data presented in Fig. S17 (ESI†) show that there is no notable change between the control and treatment groups of D. melanogaster.3.10. Trypan blue study
The trypan blue image allows for the differentiation between live and dead cells. A positive trypan blue stain indicates that there is no damage to the gut after administering the AuNP@GCD nanocomposite in the food vial. Even at higher concentrations, the treatment of the AuNP@GCD nanocomposite did not result in any damage to the larval gut (Fig. S18, ESI†). All of the information suggests that our AuNP@GCD fluorescent probe is completely safe for use in bioimaging applications.3.11. Dopamine monitoring in the Drosophila brain
Dopamine is an essential neurotransmitter in the central nervous system of Drosophila fruit flies, where DA receptors are located in the mushroom bodies of the brain.47 In the nervous system of Drosophila, the mushroom body serves as a center for olfactory learning, whereas the lateral horn (LH) has traditionally been thought to play a role in innate olfactory behaviors (Michael Marquis et al. 2022).48 Research studies have revealed that dopamine and its receptors are present in the distinct area of the brain, suggesting that DA may change the odor response, olfactory processing, sensory processing and regulation of the locomotor response.49 The accurate monitoring of dopamine levels in the brain is vital for diagnosing neurological disorders and locomotory behaviour. Before preparing Drosophila for ex vivo dopamine imaging in the brain, all physiological movement assays such as climbing, crawling, light–dark assay and touch sensitivity were done (Fig. S19–S22, ESI†). The findings from all these assays confirm that there was no neuronal issue up to 150 μg mL−1. So, the ex vivo dopamine imaging study was conducted in Drosophila brain. The dissected brain treated with the AuNP@GCD nanoprobe showed the intense fluorescence in case of control group. The dopamine observed in the brain cell is highlighted by a white circle in Fig. 4. The weak fluorescence of AuNP@GCD was recovered when it interacted with dopamine. When the nanocomposite comes into contact with dopamine in the mushroom bodies of brain cell, that specific area of the brain shows a strong FL signal as compared to other regions (Fig. 4f–j). As GCD shows excitation-dependent emission, the red, green and blue emissions intensify at an excitation of 560, 488 and 405 nm, respectively. In the case of the Tau mutant, there was no increase in the emission signal (Fig. 4k–o). It has been well documented that the Tau burden in the brain areas is linked to dopaminergic pathways is associated with exacerbated dopaminergic dysfunction. To further establish the dopamine binding capability of our nanoprobe, the images of the mushroom bodies of the Drosophila of a Huntington variant has been captured after being treated with the AuNP@GCD probe. It is well reported that a dopamine imbalance in the striatum gives rise to pathological conditions like Huntington's diseases (HD). HD is a neurodegenerative disorder that is caused by an expansion of the CAG repeat region of the Huntingtin gene. It is characterized by multiple areas of brain degeneration, including the neurotransmitters dopamine.50 Moreover, the dopamine levels in patients with Huntington's disease are significantly lower than those in healthy individuals. In fact, the emission signal intensity from the mushroom bodies of Huntington variant Drosophila is notably reduced as observed in the confocal images (Fig. 4q–t). Among other potential interfering compounds, the FL response of the sensor towards glutamate, epinephrine and norepinephrine which has structural similarity with octopamine in Drosophila was verified. Although epinephrine and norepinephrine show a slight enhancement in the FL intensity, it is substantially less as compared to the intensity enhancement in the case of dopamine. Furthermore, in the above region of the mushroom (striatum) body, the particular neurons which secrete serotonin and octopamine are not present.51,52 Therefore, the enhanced fluorescence signal is due to the presence of dopamine. All these findings indicate that AuNP@GCD can be used to image dopamine in a living organism.4. Conclusion
A stable AuNP@GCD fluorescent nanoprobe has been designed, which shows specific nonenzymatic recognition towards dopamine by electrochemical and fluorescence techniques. The feeble fluorescence signal from the probe is substantially increased when it comes into contact with dopamine due to the disintegration of the AuNP@GCD FRET pair. In the electrochemical sensing, the current response of the AuNP@GCD-modified GCE in the presence of dopamine is 18 times higher than that of bare GCE, due to the improved conductivity, higher recognition sites and stability of the AuNP@GCD probe. The detection limit found in the electrochemical and fluorescence sensing were 0.0025 μM and 0.00026 μM, respectively. The developed fluorescent AuNP@GCD nanoprobe is safe and exhibits excellent performance for the identification of dopamine through ex vivo bioimaging, as demonstrated in the brain of Drosophila melanogaster.Author contributions
LB contributed to the investigation, data curation, analysis and writing the original draft. LM performed the validation experiments in Drosophila under the supervision of MM. SM is responsible for the conceptualization, fund acquisition, review and editing of the manuscript.Data availability
All data will be available upon request.Conflicts of interest
There are no conflicts to declare.Acknowledgements
This work is financially supported by SERB, DST, Govt. of India (CRG/2018/001717). LB and LM thank NIT Rourkela and CSIR, India, respectively, for providing doctoral research fellowships.References
- J. Jana, J. S. Chung and S. H. Hur, ACS Omega, 2019, 4, 17031–17038, DOI:10.1021/acsomega.9b02637.
- L. G. Mann and D. O. Claassen, Eur. J. Neurosci., 2024, 59, 1141–1152, DOI:10.1111/ejn.16209.
- I. Kawahata, D. I. Finkelstein and K. Fukunaga, Receptors, 2024, 3, 155–181, DOI:10.3390/receptors3020009.
- S. K. Sorooshyari, N. Ouassil, S. J. Yang and M. P. Landry, ACS Chem. Neurosci., 2023, 14, 2282–2293, DOI:10.1021/acschemneuro.3c00001.
- P. Belujon and A. A. Grace, Eur. J. Neurosci., 2017, 20, 1036–1046, DOI:10.1093/ijnp/pyx056.
- X. Pan, A. C. Kaminga, S. W. Wen, X. Wu, K. Acheampong and A. Liu, Front. Aging Neurosci., 2019, 11, 175, DOI:10.3389/fnagi.2019.00175.
- C. Cepeda, K. P. Murphy, M. Parent and M. S. Levine, Prog. Brain Res., 2014, 211, 235–254, DOI:10.1016/B978-0-444-63425-2.00010-6.
- J. Segura-Aguilar, I. Paris, P. Muñoz, E. Ferrari, L. Zecca and F. A. Zucca, J. Neurosci., 2014, 129, 898–915, DOI:10.1111/jnc.12686.
- J. P. Kesby, D. W. Eyles, J. J. McGrath and J. G. Scott, Transation Psychology, 2017, 8, 30, DOI:10.1038/s41398-017-0071-9.
- H. Chen, J. Li, Z. Huang, X. Fan, X. Wang, X. Chen and Y. Liu, Neuropharmacology, 2024, 260, 110133, DOI:10.1016/j.neuropharm.2024.110133.
- A. J. Steckl and P. Ray, ACS Sens., 2018, 3, 2025–2044, DOI:10.1021/acssensors.8b00726.
- A. Carlsson, T. Sharp, T. Zetterström and U. Ungerstedt, J. Chromatogr. A, 1986, 368, 299–308, DOI:10.1016/S0021-9673(00)91072-0.
- J. Bergquist and J. Silberring, Rapid Commun. Mass Spectrom., 1998, 12, 683–688, DOI:10.1002/(SICI)1097-0231(19980615)12:11<683::AID-RCM218>3.0.CO;2-N.
- F. N. Wu, J. Zhu, G. J. Weng, J. J. Li and J. W. J. Zhao, Sens. Actuators, B, 2024, 404, 135246, DOI:10.1016/j.snb.2023.135246.
- Y. Xu, J. Zhou, P. Sun, P. Li, D. Han, X. Wang and F. Chai, Sep. Purif. Technol., 2025, 353, 128396, DOI:10.1016/j.seppur.2024.128396.
- S. Batish and J. K. Rajput, ACS Appl. Nano Mater., 2024, 7, 15104–15115, DOI:10.1021/acsanm.4c01858.
- X. Zhang, X. Chen, S. Kai, H. Y. Wang, J. Yang, F. G. Wu and Z. Chen, Anal. Chem., 2015, 87, 3360–3365, DOI:10.1021/ac504520g.
- X. Huang, J. Song, B. C. Yung, X. Huang, Y. Xiong and X. Chen, Chem. Soc. Rev., 2018, 47, 2873–2920, 10.1039/C7CS00612H.
- Y. Xue, Q. Hassan, M. Noroozifar, R. M. A. Sullan and K. Kerman, Talanta, 2024, 266, 125030, DOI:10.1016/j.talanta.2023.125030.
- Y. Hao, T. Li, M. Tian, Q. Dai, F. Zhang and F. Chai, New J. Chem., 2024, 48, 1828–1836, 10.1039/D3NJ04627C.
- I. Soliman and C. Gunathilake, Next Mater., 2024, 3, 100160, DOI:10.1016/j.nxmate.2024.100160.
- W. Ling, X. Shang, C. Yu, C. Li, K. Xu, L. Feng and X. Huang, ACS Nano, 2024, 18, 10596–10608, DOI:10.1021/acsnano.4c00632.
- N. Zehra, A. H. Malik, R. Parui, S. Hussain and P. K. Iyer, Chem. – Asian J., 2024, 19, e202400544, DOI:10.1002/asia.202400544.
- Z. Meng, S. Sun, X. Pu, J. Wang, X. Liao, Z. Huang and G. Yin, Nanotechnology, 2024, 35, 235502, DOI:10.1088/1361-6528/ad2e49.
- R. Sangubotla, J. Paul and J. Kim, Appl. Surf. Sci., 2025, 687, 162275, DOI:10.1016/j.apsusc.2024.162275.
- S. R. Ahmed, A. G. Cardoso, A. H. V. Cobas, P. Das, A. Chen, S. Srinivasan and A. R. Rajabzadeh, ACS Appl. Nano Mater., 2023, 6, 8434–8443, DOI:10.1021/acsanm.3c00771.
- V. Sliesarenko, U. Bren and A. Lobnik, Sens. Actuators, Rep., 2024, 7, 100199, DOI:10.1016/j.snr.2024.100199.
- H. Chen, C. Liu and Y. Xia, Methods Appl. Fluoresc., 2017, 5, 014006, DOI:10.1088/2050-6120/aa5e2d.
- L. Ren, X. Hang, Z. Qin, P. Zhang, W. Wang, Y. Zhang and L. Jiang, Optik, 2020, 208, 163560, DOI:10.1016/j.ijleo.2019.163560.
- H. Li, J. Tian, L. Wang, Y. Zhang and X. Sun, J. Mater. Chem., 2011, 21, 824–828, 10.1039/C0JM02695F.
- P. Suresh Kumar, S. Megarajan, G. Rajendra Kumar Reddy and V. Anbazhagan, IET Nanobiotechnol., 2018, 12, 909–914, DOI:10.1049/iet-nbt.2018.0048.
- Z. Aryan, H. Khajehsharifi and S. Shahrokhian, Microchem. J., 2024, 198, 110087, DOI:10.1016/j.microc.2024.110087.
- A. S. Anand, U. Gahlot, D. N. Prasad, A. Chauhan and E. Kohli, Nanotoxicology, 2019, 13, 977–989, DOI:10.1080/17435390.2019.1602680.
- J. Peng, C. Hu, X. Zhou, D. Wu, X. Sun, A. Tang and J. Tian, Chem. Phys., 2025, 594, 112658, DOI:10.1016/j.chemphys.2025.112658.
- B. Barik, L. Behera and S. Mohapatra, Ind. Eng. Chem. Res., 2023, 62, 10849–10860, DOI:10.1021/acs.iecr.3c01012.
- N. Sharma, G. S. Das and K. Yun, Appl. Microbiol. Biotechnol., 2020, 104, 7187–7200, DOI:10.1007/s00253-020-10726-5.
- L. Behera, D. Pati, B. B. Sahu and S. Mohapatra, Colloids Surf., A, 2022, 653, 130002, DOI:10.1016/j.colsurfa.2022.130002.
- S. Mohapatra and R. K. Das, Anal. Chem. Acta, 2019, 1058, 146–154, DOI:10.1016/j.aca.2019.01.021.
- A. Thamilselvan, P. Manivel, V. Rajagopal, N. Nesakumar and V. Suryanarayanan, Colloids Surf., B, 2019, 180, 1–8, DOI:10.1016/j.colsurfb.2019.04.034.
- B. C. Pattnayak and S. Mohapatra, Spectrochim. Acta, Part A, 2023, 286, 122009, DOI:10.1016/j.saa.2022.122009.
- Z. Liang, M. Kang, G. F. Payne, X. Wang and R. Sun, ACS Appl. Mater. Interfaces, 2016, 8, 17478–17488, DOI:10.1021/acsami.6b04826.
- W. Cheng, S. Dong and E. Wang, Langmuir, 2002, 18, 9947–9952, DOI:10.1021/la026022b.
- H. Fang, H. Yu, Q. Lu, X. Fang, Q. Zhang, J. Zhang and Q. Ma, Anal. Chem., 2020, 92, 12825–12832, DOI:10.1021/acs.analchem.0c01147.
- N. Kalcec, A. Ljulj, L. Božičević, V. Vrček, D. Marson, S. Pricl and I. Vinković Vrček, Inorg. Chem., 2022, 61, 10781–10791, DOI:10.1021/acs.inorgchem.2c00996.
- S. Saisree, J. S. Arya Nair and K. Y. Sandhya, J. Mater. Chem. B, 2022, 10, 3974–3988, 10.1039/D1TB02368C.
- H. S. Jang, D. Kim, C. Lee, B. Yan, X. Qin and Y. Piao, Inorg. Chem. Commun., 2019, 105, 174–181, DOI:10.1016/j.inoche.2019.05.009.
- B. Silva, S. Hidalgo and J. M. Campusano, PLoS One, 2020, 15, e0229671, DOI:10.1371/journal.pone.0229671.
- M. Marquis and R. I. Wilson, Curr. Biol., 2022, 32, 5406–5414, DOI:10.1016/j.cub.2022.11.008.
- N. Fuenzalida-Uribe and J. M. Campusano, Neuroscience, 2018, 371, 433–444, DOI:10.1016/j.neuroscience.2017.12.032.
- J. Y. Chen, E. A. Wang, C. Cepeda and M. S. Levine, Front. Neurosci., 2013, 7, 114, DOI:10.3389/fnins.2013.00114.
- O. V. Alekseyenko and E. A. Kravitz, Fly, 2014, 8, 200–205, DOI:10.1080/19336934.2015.1045171.
- S. Busch, M. Selcho, K. Ito and H. Tanimoto, J. Comp. Neurol., 2009, 513, 643–667, DOI:10.1002/cne.21966.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d5tb00265f |
| This journal is © The Royal Society of Chemistry 2025 |