Influence of material format and surface chemistry for the sustained delivery and efficacy of silk drug delivery systems in vivo
Christine Jurene O.
Bacal
 a,
Benjamin J.
Allardyce
a,
Benjamin J.
Allardyce
 *b and
Filippo
Valente
*b and
Filippo
Valente
 *a
*a
aEar Science Institute Australia, Ear Sciences Centre, School of Medicine, The University of Western Australia, Nedlands 6009, Australia. E-mail: Filippo.Valente@earscience.org.au
bInstitute for Frontier Materials, Deakin University, Geelong 3220, Australia
First published on 14th May 2025
Abstract
Silk fibroin materials are promising for use in controlled drug delivery in the field of tissue engineering and biomedical applications thanks to silk's generally established biocompatibility and tunable properties for implants and drug storage. Several factors must be considered in the materials design, including material format, drug properties and release kinetics, and the activity and stability of the drug after release. While numerous reviews described silk-based DDS that demonstrated controllable in vitro release, success in vivo has been limited, especially in some material formats. This review therefore aims to provide insight into the current material format and functionalization strategies to maximize in vivo performance by describing the in vivo activity of recently developed silk drug delivery systems. The review also aims to provide a fresh perspective on the suitable format and functionalization strategies for a target biomedical application. Based on the release behavior of drugs in various material formats, silk films, foams, and microneedles were better suited to serve as scaffolds for cell regeneration and improved recovery rate for biomedical applications involving wound healing and tissue engineering. Gels and particles could be incorporated within the films and foams but the purpose would be to serve as additional physical barriers towards drug diffusion in these types of application. For drugs or therapeutics that target internal organs (i.e. brain, liver, intestines, etc.), gels and particles were mainly used due to their size. In the event that the material format selection based on the target application does not contribute a lot to the prolonged release of drugs or therapeutic agents, hybrid functionalization strategies were adapted to make the surface chemistry of the material more responsive to the environmental stimuli for a more tunable silk DDS.
1. Introduction
Drug delivery systems (DDS) are devices or formulations that carry drugs or any therapeutic agents towards a certain target, which could be a pathogen, a cell, or a gene, for the improvement of the therapeutic efficiency, to improve specificity of the drug towards its target.1,2 DDS are designed based on both the pharmacokinetics (PK) and biodistribution (BD) of the drug and the environment where the therapeutic agent will be distributed.1,2 Depending on the desired application, DDS can consist of nanoparticles3 and liposomes,4 films,5 foams,6 hydrogels,7 injectables,8 inhalants,9 and even microneedle patches.10 Single dose DDS are useful to improve patient compliance in instances where delivery is required over prolonged periods.11 In addition, DDS have the potential to reduce the side effects from drug administration by providing local and targeted delivery at therapeutic doses, reducing the amount of systemic drugs that need to be administered over time.12 Despite their versatility and the tunability, development of DDSs that are highly resorbable and capable of mitigating undesired immunological responses such as hypersensitivity reactions13–15 to prevent invasive removal of non-resorbable DDS16,17 is yet to be achieved.Some biodegradable and resorbable materials approved by the United States Food and Drug Administration for clinical use include synthetic polymers such as poly(glycolic acid) (PGA), poly(lactic acid) (PLA), poly(lactic-co-glycolic acid) (PLGA), poly(caprolactone) (PCL), and poly(dioxanone) (PDS)18–20 and a natural polymer like collagen.21 However, these polymers still trigger inflammation,22–26 degradation of bone tissue (osteolysis) in bone implants,27,28 and reduced mechanical strength.11,29–31 Therefore, natural biopolymers such as chitosan, alginate, and silk are being increasingly explored.32
Among the natural biopolymers, silk has been extensively explored as a DDS because it was known to preserve the activity and stability of drugs and other therapeutic agents like temperature-sensitive vaccines33 and labile antibiotics.34 The inherent structure of silk resembles amphiphilic block copolymers which enables controllability of drug release via external stimuli (i.e. pH) owing to the changes in bond conformations and surface charges on the silk backbone.35 In addition, by altering the degree of physical crosslinking through a range of established methods like alcohol treatment36 or water vapor annealing,37 silk could be processed in various formats making it a versatile DDS.
Despite the advantages, silk's inherent amino acid sequence gives it a negative charge at neutral pH. As a result, compounds with a higher isoelectric point are typically released rapidly,38,39 which could be unfavorable when sustained release of these compounds is preferred. It is therefore necessary to explore a material configuration and functionalization method that will enable prolonged and controllable release of wider range of drugs and therapeutic agents in silk DDS.
In this review, the challenges of developing silk-based DDS that can deliver drugs with controlled release while ensuring the drug functionality were evaluated. Various functionalization strategies were presented to correlate how modifications in chemical functional groups affected the release profiles and other clinically-relevant silk properties. Finally, emerging smart (“stimuli-responsive”) and functional materials were evaluated to provide an insight on the novel materials that can be incorporated with various silk formats. The use of smart materials to improve surface chemistry were assessed on how they influenced the controllability of drug release.
2. Silk functionalization strategies for drug/therapeutic agent delivery
In designing a silk-based DDS, chemical/physical interactions between the drug and silk must be established based on the specific therapeutic agent to be delivered. In this section, techniques to incorporate active agents into silk were discussed as well as the available functionalization methods utilized to control binding and release.2.1. Conversion of silk fibers to silk solution
Silk fibers must be converted to silk solution prior to fabricating various silk drug delivery systems.40 The raw silk from the cocoons must be treated before removing the sericin. Sericin is the protein “glue” that binds silk filaments together into the cocoon.41 Silk sericin removal is called degumming and various reagents/solvents can be used such as sodium carbonate,42 calcium chloride/ethanol solution,43 sodium oleate,44 enzymes,45 and ionic liquid46 to boil the cocoons during pre-treatment. The selection of one or multiple solvents depends on the desired application as some could influence the resulting tensile properties.47 The degummed silk fibers are further treated with a chaotropic agent (e.g. lithium bromide,48 formic acid,49 lanthanide chlorides,50 or hexafluoroisopropanol51) to disrupt hydrogen bonding within the silk structure and dissolve silk resulting in processability of the solution into various forms (i.e. films, hydrogels, foams, etc.). Finally, the dissolved silk fibers are dialyzed against pure water to remove excess salts and centrifuged, resulting in an aqueous solution of pure silk fibroin that can be used to fabricate various silk material formats for drug delivery.2.2. Silk functionalization strategies in various silk drug delivery systems
Silk drug delivery systems, in any type of format, could be functionalized to control the release of the encapsulated therapeutic agent. This could be performed by tapping into the noncovalent interactions between silk and therapeutic agents or via chemical crosslinking.There are several different strategies to incorporate silk with therapeutic agent physically but this could be classified into two modes: by directly incorporating drugs within the silk solution and by coating physically. Strategies to directly incorporate drugs within the silk solution include embedding a drug/therapeutic agent within pure silk (bulk blending), using silk composite materials or by using interfacing or encapsulating agents. Bulk blending is by far, the simplest physical approach to sustain release in silk drug delivery systems (Fig. 1(a)). In this method, the drug is homogeneously blended with the silk solution prior to immobilization into a solid scaffold (i.e. casting to a film or lyophilizing to a foam) or direct administration to target site. Mixing drugs or therapeutic agents to silk blended with another polymer to form silk composite materials is another strategy to physically incorporate drugs or therapeutic agents.52 In this process, silk is mixed with another polymer to form a composite material with a different property than the original components (silk composite drug delivery systems) (Fig. 1(b)). Therapeutics are often internalized in this technique by adding them into a mixture of silk with another polymer. The drug is then physically adsorbed via electrostatic interaction or hydrogen bonding. Another strategy is to use interfacing or encapsulating agents such as an amphiphilic polymer, a surfactant, or a phospholipid before blending with silk solution (Fig. 1(c)). This technique is commonly applied in silk particles or hydrogels and has enabled slow release of drugs to facilitate sustained delivery over extended periods.52–54 In this same technique, the interaction between silk and the encapsulating agent prevents drug leaching due to the indirect interaction between the drug and the release bath.
 | ||
| Fig. 1 Functionalization methods via noncovalent interactions. (A) Bulk blending of silk and therapeutic agents. SNF (silk nanofibers), PTX (paclitaxel drug), DOX (doxorubicin drug) (reprinted with permission from Xiao, et al. ©2024 Wiley-VCH GmbH)55 (B) use of silk composite materials. PBS/EtOH (phosphate buffered saline/ethanol solvent system), SF (silk fibroin), PDLLA (poly(D,L-lactic acid)) (reprinted with permission from Deng, et al. ©2024 American Chemical Society)56 (C) use of encapsulating agents. (Reprinted with permission from Mao, et al. ©2017, Published by Elsevier B.V.).53 | ||
Meanwhile, coating (Fig. 2) is another functionalization technique which involved covering the silk biomaterial with a layer containing the encapsulated drug or another polymer containing the drug to slow down the release of the therapeutic agent.57 Strategies to coat the silk material physically are via dip coating to form a layer of surface coating or via the layer-by-layer (LbL) approach to form a multilayer assembly in silk drug delivery systems. Dip coating involves (Fig. 2(a)) silk substrate immersion into a solution that could form a surface barrier via electrostatic interactions which surrounds the silk drug delivery system and prevent the burst release of therapeutic agents. This solution could be pure silk itself, another polymer (forming silk composite coating), or a surfactant. Dip coating can also be applied repeatedly onto the same substrate to form a multilayered assembly in silk drug delivery systems. This technique is known as layer-by-layer (LbL) approach and manifested a more prolonged release behavior in most silk drug delivery systems developed compared to a single-layered coating (Fig. 2(b)).
 | ||
| Fig. 2 Physical coating strategies for silk drug delivery systems. (A) Dip coating to form a single surface layer. (B) Layer-by-layer coating approach. | ||
Based on early in vitro studies, it was hypothesized that merely applying bulk blending is insufficient to prolong the release of therapeutic agents for treatment of various diseases.58–60 However, recent in vivo studies have shown the efficacy of bulk blending in prolonging drug release in various animal models (Table 1, entries 1–8). Bulk blending was applied to develop silk drug delivery systems for combination chemotherapy,55 as vaccines for COVID-19,61 for tissue regeneration,62 for circadian rhythm regulation,63 for wound healing,64,65 for acute liver failure treatment,66 and for treatment of sensorineural hearing loss67 primarily since it is a facile approach and does not involve a lot of other materials or polymers aside from the therapeutic agent and silk solution minimizing the components that could counteract each other. For instance, when both doxorubicin and paclitaxel were incorporated in silk nanofiber hydrogels and administered intratumorally, tumor growth in female nude mice was inhibited for over 28 days (Table 1, entry 1).55 When receptor-binding domain (RBD) linked with fragment crystallizable (Fc) region of human immunoglobulin G1 (IgG1) (RBD-Fc) was directly incorporated with silk fibroin solution to form a silk fibroin hydrogel-based vaccine for COVID-19, it was found that the formulations had 1-1.3-fold increased immunogenicity in female ICR mice after 90 days since intramuscular injection compared with the silk hydrogel control without the therapeutic agent (Table 1, entry 2).61 However, burst release can still be observed upon transdermal administration of melatonin-containing silk fibroin microneedles for circadian rhythm regulation in Sprague–Dawley rats (Table 1, entry 4)63 as this has immediately induced sleep in the rats. Hence, other functionalization techniques can be explored to apply for this specific silk drug delivery system to prolong drug release.
| Entry no. | Silk drug delivery system | Drug/therapeutic agent carried | Application | Functionalization technique used | Material format | Delivery mode | Model organism | In vivo efficacy | Ref. |
|---|---|---|---|---|---|---|---|---|---|
| *Silk solutions were taken as fibroin solution unless explicitly stated to be sericin. | |||||||||
| 1 | Silk nanofibers | Doxorubicin and paclitaxel | Combination chemotherapy | Bulk blending | Hydrogel | In situ | Female nude mice | Inhibition of tumor growth over 28 days | 55 |
| 2 | Silk fibroin-based hydrogel | Receptor-binding domain (RBD) linked with fragment crystallizable (Fc) region of human immunoglobulin G1 (IgG1) (RBD-Fc vaccine) | Vaccine for COVID-19 | Bulk blending | Hydrogel | Intramuscular injection | Female ICR mice | SF hydrogel-based vaccine formulations had 1–1.3-fold increase immunogenicity after 90 days | 61 |
| 3 | Silk fibroin microspheres | Glucose | Tissue regeneration | Bulk blending | Particle (microspheres) | Subcutaneous implantation | Sprague–Dawley rats | Large amount of extracellular matrix distributed peripherally throughout the silk fibroin microspheres after 2-weeks | 62 |
| 4 | Silk fibroin microneedles | Melatonin | Circadian rhythm regulation | Bulk blending | Microneedles | Transdermal | Sprague–Dawley rats | Burst release upon administration as it induced sleep immediately, then melatonin concentration peaked after ∼1 h but sustaining release after 8 h | 63 |
| 5 | Omentum-extracellular matrix/silk fibroin hydroscaffold | Decellularized extracellular matrix | Treatment of nonhealing diabetic wounds | Bulk blending | Hydrogel/hydrogel scaffold hybrid | In situ | Wistar rats | Wounds treated with hydrogels and hydroscaffolds were completely closed after 28 days, untreated wounds showed delayed closure and took another 3–5 days | 64 |
| 6 | Silk fibroin nanofibers | Quercetin containing morpholine-pyridine motifs (QFM) | Wound healing | Bulk blending | Mat (membrane) | Transdermal | Wistar rats | 100% wound closure after 9 days | 65 |
| 7 | Silk fibroin electrospun scaffolds | Hepatocyte growth factor (HGF)/fibroblast growth factor-4 (FGF-4) | Acute liver failure treatment | Bulk blending | Scaffolds (membrane) | Implanted | BALB/C mice | Growth factor incorporated scaffolds showed greatest reduction in the biomarkers for hepatocyte damage after 14 days | 66 |
| 8 | Silk fibroin-based hydrogels | Dexamethasone and lipoic acid | Treatment of sensorineural hearing loss | Bulk blending | Hydrogel | Intratympanic injection | Guinea pigs | Dexamethasone undetected after 5 days in the perilymph after injecting dexamethasone loaded silk fibroin hydrogels; had no superior advantages than microcrystals | 67 |
| 9 | Silk/poly(D,L-lactic acid) microspheres | Doxorubicin hydrochloride | Anticancer | Using silk composite materials | Particle (microsphere) | In situ | BALB/C nude mice | Tumor volume is minimal after 15 days | 56 |
| 10 | Silk/poly(vinyl alcohol) microparticles | Metformin | Corneal neovascularization | Using silk composite materials | Particle (microparticle) | In situ | Rats | Reduced neovascularization and inflammation after 7 days | 68 |
| 11 | Silk fibroin/collagen nanofiber | Combination of silver (Ag) and gold (Au) nanoparticles | Wound healing | Using silk composite materials | Scaffolds (membrane) | Implanted | Sprague-Dawley rats | Healing percentage of ∼99% over the course of 21 days | 69 |
| 12 | Silk fibroin nanoparticle hydrogel | NK1R antagonist (CP-99,994) | Dry eye disease treatment | Using silk composite materials | Hydrogel | Topical administration | C57BL/6 mice | Significant recovery of Treg cell function and nanoparticle size retained after 14 days of treatment | 70 |
| 13 | Silk fibroin/chitosan-based scaffolds | Curcumin and 5-aminosalicylic acid | Anti-inflammatory and healing function for anal fistula | Using silk composite materials | Scaffold (membrane) | Subcutaneous implantation | Rats | Sustainable drug release above 400 h, better biocompatibility and anti-inflammatory effect than control groups | 71 |
| 14 | Silk nanofiber hydrogels | Asiaticoside and magnesium ions | Wound healing | Use of silk composite materials | Hydrogel | In situ | Sprague–Dawley rats | Above 60% of wounds exhibited better healing within 7 days compared to other hydrogel groups | 72 |
| 15 | Silk fibroin composite nanofiber membrane | Flavonoids | Wound healing | Use of silk composite materials | Membrane | Transdermal | Sprague–Dawley rats | After day 15, ∼93% and ∼97% wound shrinkage can be observed with modified groups compared to the ∼72% of the control group | 73 |
| 16 | Silk fibroin/alginate 3D-printed scaffold | Placental extracellular matrix | Wound healing | Use of silk composite materials | 3D printed scaffolds (hydrogel scaffolds) | Transdermal | C57 mice | Remarkable increase in the expression of pro-angiogenesis genes in the wound bed treated with the 3D-printed scaffold with extracellular matrix at both day 7 and day 21 post-surgery | 74 |
| 17 | Electrospun silk fibroin/poly(hydroxybutyrate-co-valerate) (PHBV) dressing | Berberine | Diabetic wound healing | Use of silk composite materials | Nanofibrous dressing (membrane) | Transdermal | Mice | Wound healed completely at a significantly higher rate (∼99.7%) than the control group (∼83–85%) after 18 days of treatment | 75 |
| 18 | Silk fibroin/poly(caprolactone) nanofibrous mat | Berberine | Diabetic alveolar bone regeneration | Use of silk composite materials | Nanofibrous mat (membrane) | Topical | Sprague–Dawley rats | Most regenerated bone formation in type 2 diabetes milletus rats compared to the control groups after 2–6 weeks | 76 |
| 19 | Cell-free chitosan/silk fibroin scaffolds | Bioactive glass | Regeneration of critical-size bone defects | Use of silk composite materials | Scaffold (foams) | In situ | Sprague–Dawley rats | Bone volume fraction was higher (∼46%) compared to the control (∼42%) at 12 weeks of treatment | 77 |
| 20 | Silk sericin hydrogel | Resveratrol | Wound healing | Use of silk composite materials | Hydrogel | Topical | Wistar albino rats | Maximum wound healing achieved at the end of day 14 compared to the toxic group | 78 |
| 21 | Silk fibroin mesh | Honey extract from bees | Wound healing | Dip coating in a mixture of poly(hydroxybutyrate-co-hydroxyvalerate) (PHBV) and poly(lactic acid) (PLA) or poly(caprolactone) (PCL) | Mesh (membrane) | Implanted | Sprague Dawley rats | ∼45% and ∼62% area deposition of extracellular matrix and collagen, respectively from ∼24% and ∼20% in the control PHBV mesh up to 28 days | 79 |
| 22 | Silk fibroin shell | siRNA | Antitumor | Layer-by-layer assembly | Particle | Subcutaneous injection | BALB/C nude mice | The treated group yielded the smallest tumor sizes (0.3 g) and highest tumor inhibition rate (56%) than the control group after 15 days | 80 |
| 23 | Dissolving microneedle system | Mupirocin and Ag nanoparticles | Treatment of delayed wound healing | Bulk blending/using silk composite materials | Microneedles | Transdermal | BALB/C mice | Wound contraction rate increases to ∼78%, significantly higher than control on day 11 | 81 |
| 24 | Silk fibroin and hydroxypropyl cellulose composite injectable hydrogel | Folate-modified AC16 cell-derived extracellular vesicles | Myocardial infarction repair | Use of silk composite materials/use of encapsulating agents | Hydrogel | In situ | Sprague–Dawley rats | Best cardiac repair result with left ventricular ejection fraction of ∼71% compared to the myocardial infarction control group with ∼43% after 28 days of treatment | 82 |
| 25 | Silk fibroin coating | Tumor targeting peptide | Cancer therapy | Layer-by-layer assembly/use of encapsulating agents | Particle (nanorods) | Intravenous injection | BALB/C nude mice | Decrease in tumor size after 14 days of irradiation in the treated group compared to the control | 83 |
| 26 | Porous chitosan–alginate hydrogel/electrospun PCL-silk sericin hydrogel scaffold | 10-hydroxydecanoic acid (queen bee acid) | Wound healing | Using silk composite materials/chemical crosslinking with glutaraldehyde for the porous chitosan–alginate layer | Hydrogel scaffold hybrid | Transdermal | Wistar rats | Fibrosis is less prominent with 10-HDA-containing wound dressing than controls after 14 days | 84 |
| 27 | Poly(vinyl alcohol)/silk fibroin nanofibrous membrane | Astaxanthin and silver nanoparticles | Wound dressing | Use of silk composite materials/crosslinking using glutaraldehyde | Membrane | Transdermal | Sprague–Dawley rats | Wound area reduced to ∼12% and ∼4% compared with the ∼22% of the negative control after 14 days | 85 |
| 28 | Self-healable and 3D-printable silk fibroin nanocomposite hydrogels | Oxidized salep (OS) and κ-carrageenan nanoparticles | Wound care | Using silk composite materials/Schiff base crosslinking between OS and κ-carrageenan nanoparticles | Hydrogel | Subcutaneous implantation | Wistar albino rats | All animals survived without any malignancy, infection, or abscess formation. No necrosis or muscle degeneration after 14 days | 86 |
| 29 | Conductive hydrogels based on tragacanth and silk fibroin | Vancomycin | Burn wound healing | Blending with another stimuli-responsive polymers/crosslinking using dopamine and to form a Schiff base/crosslinking using EDC | Hydrogels | In situ | Wistar rats | Wound treated with hydrogel dressings had nearly entirely healed with >60% of the wound area showing signs of recovery after 14 days | 87 |
| 30 | Silk fibroin/poly(vinyl alcohol)/ROS-scavenging dendrimer hydrogels | Gallic acid | Wound healing | Using silk composite materials/use encapsulating agents (dendrimers)/amide coupling reaction between gallic acid and the dendrimer using EDC/NHS | Hydrogel | In situ | Male BALB/C mice | Wound size of ∼30% in the dendrimer-modified silk compared to ∼56% in the control group after 10 days over 16-day treatment course | 88 |
| 31 | Silk fibroin/gelatin films | Gelatin | Regeneration of tympanic membrane perforation | Chemical crosslinking with genipin | Film | Surgical implantation | Sprague–Dawley rats | Tympanic membrane regeneration was 60% compared to 40% in control after 5 days | 89 |
| 32 | Silk protein hybrid hydrogel | Teicoplanin-decorated reduced graphene oxide | Diabetic wound healing | Chemical crosslinking with genipin | Hydrogel | Topical | Sprague–Dawley rats/BALB/C mice | Accelerated healing of noninfected wounds in diabetic rat and infected wounds in a diabetic mouse after 14 days of treatment | 90 |
| 33 | Silk fibroin modified decellularized porcine liver scaffolds | Gelatin | Tissue regeneration | Bulk blending/crosslinking using citric acid | Scaffolds (membrane) | Subcutaneous implantation | Wistar rats | Collagen remodeling within the 90-day time point | 91 |
| 34 | Silk fibroin/soy protein isolate hydrogels | Quercetin | Burn wound healing | Use of silk composite materials/chemical crosslinking between silk and soy protein isolate using epichlorohydrin | Hydrogel | In situ | Kunming mice | Wound closure rates were ∼79% for the hydrogel-treated group whereas it was only ∼48% for the control group after day 17 | 92 |
| 35 | Dual-network DNA-silk fibroin hydrogels | Bone marrow mesenchymal stem cells | Cartilage repair | Physical crosslinking via DNA base pairing/crosslinking via horseradish peroxidase (HRP)-mediated enzymatic reactions | Hydrogel | Implanted | Sprague–Dawley rats | Significantly accelerated cartilage regeneration after 14 days | 93 |
| 36 | Silk-based photocurable hydrogel | Platelet-rich plasma lysate | Meniscus treatment | Methacrylation via crosslinking (grafting) using lithium phenyl-2,4,6-trimethylbenzoylphosphinate (LAP) photoinitiator and poly(ethylene glycol diacrylate) (PEGDA) crosslinker | Hydrogel | In situ | New Zealand white rabbits | Treatment of full-thickness tears in rabbit menisci compared to the untreated counterparts after 8 weeks | 94 |
| 37 | Silk nanofibril/hyaluronic acid scaffold | Basic fibroblast growth factor | Spinal cord repair | Surface grafting of silk nanofibril/hyaluronic acid blends | Scaffold (membrane) | Implanted | Sprague–Dawley rats | Recovery of motor function 10 weeks after surgery compared to the control | 95 |
| 38 | Silk hydrogels | Adipose stem cells | Bone/wound healing | Photocrosslinking using riboflavin and hydrogen peroxide | Hydrogels (can be extended to microneedles, microcarriers, and bone screws) | Subcutaneous implantation | Sprague–Dawley rats | Bone screws did not cause serious inflammation and maintained bone screw shape after 14 days | 96 |
| 39 | Silk fibroin/hyaluronic acid hydrogel microneedles | Insulin | Diabetes management | Blending with another stimuli-responsive polymer (methacrylated hyaluronic acid)/photocrosslinking using riboflavin/layer-by-layer cumulative solution process | Hydrogel microneedles | Transdermal | C57BL/6 mice | Gentle regulation and prolonged maintenance of healthy blood glucose levels for close to 6 h compared to the blank group | 97 |
| 40 | Silk fibroin microgel scaffolds | Fluorescently tagged bovine serum albumin (BSA) | Tissue engineering and regenerative medicine | Photocrosslinking using tris(2,2′-bipyridyl)dichlororuthenium(II) hexahydrate (Ru) and sodium persulfate (SPS) | Hydrogels (microgel suspensions and scaffolds) | Subcutaneous implantation | C57/BL6 mice | Promoted cell and tissue ingrowth after day 10 | 98 |
| 41 | Dual-phase silk fibroin methacryloyl (SilMA) (hydrogel scaffold) (DPSH) | Neurotrophin-3 (NT-3) and angiotensin (1–7) (Ang-(1–7)) | Spinal cord repair | Use of encapsulating agents/UV crosslinking for silk fibroin methacryloyl | Hydrogel scaffolds (membrane) | Subcutaneous implantation | Mice | DPSH group promotes recovery of motor function following spinal cord injury after 8 weeks post implantation compared to the control groups | 99 |
| 42 | Silk fibroin hydrogels | Chondroitin sulfate methacrylate | Bone screw | Use of silk composite materials/UV crosslinking using methacrylic anhydride and poly(ethylene glycol diacrylate) (PEGDA) | Hydrogel | Implanted | White rabbit | Bone screws successfully implanted into the femur with appropriate match to the implantation site after day 7 | 100 |
| 43 | Silk fibroin/chitosan interpenetrating network hydrogel with microspheres | Methylprednisolone acetate (MPA) | Cartilage regeneration | Using silk composite materials/crosslinking using β-glycerophosphate and sodium dodecyl sulfate | Microsphere encapsulated in a hydrogel | In situ (intra-articular injection of precursor suspensions) | White rabbits | Improvement in locomotor function in treated groups compared to control groups after 12 weeks | 101 |
Silk composite materials have been increasingly used for delivering therapeutics for cancer treatment,56 corneal vascularization68 and dry eye disease treatment,70 wound healing,69,72–75,78 healing of anal fistula,71 diabetic alveolar bone regeneration76 and regeneration of critical-size bone defects (Table 1, entries 9–20).77 An in vivo study showed that by incorporating bioactive glass into a composite scaffold made from cell-free chitosan and silk fibroin, bone volume fraction of Sprague–Dawley rats was higher (∼46%) compared to the control scaffold (without bioactive glass) (∼42%) after 12 weeks of treatment (Table 1, entry 19). Thus, cell-free chitosan/silk fibroin composite has the potential to be an effective material to regenerate critical-size bone defects. In other studies though, the use of silk composite DDSs such as silk/poly(D,L-lactic acid) microspheres68 and silk nanofiber hydrogels72 were able to sustain the release of the effective concentration of therapeutic agents in vivo for a week in rats (Table 1, entries 10, 14–15). Hence, to utilize these materials for applications that require longer release of therapeutics, there must be careful consideration on the chemical properties of the incorporated components such as charge or hydrophilicity.
Dip coating of silk fibroin mesh incorporated with honey extract from bees in a mixture of poly(hydroxybutyrate-co-hydroxyvalerate) (PHBV) and poly(lactic acid) (PLA) or poly(caprolactone) (PCL) was utilized for wound healing and has shown more area deposition (∼45–62%) of extracellular matrix and collagen – indicators of healing process, as compared to the silk fibroin mesh dipped in PHBV alone (no honey extract) with ∼20–24% area deposition of extracellular matrix and collagen within 28 days of treatment administration in Sprague–Dawley rats (Table 1, entry 21).79 Since PHBV is a relatively large molecule, blending it with PLA and PCL increases the viscosity of the coating, decreasing the porosity of the mesh and therefore, adding a physical barrier to prevent fast diffusion of the honey extract towards the wound site. Layer-by-layer (LbL) coating strategy was adapted for inhibiting tumor growth in BALB/C mice with breast cancer (Table 1, entry 22).80 Here, small interfering RNA (siRNA) was loaded in a hollow mesoporous copper sulfide nanoparticle, to silence the PD-L1 gene expression in tumor cells thus, inhibiting tumor growth. The treated group yielded the smallest tumor sizes (0.3 g) and the highest tumor inhibition rate (56%) compared to the control group after 15 days of treatment. LbL assembly was applied by electrostatically adsorbing the copper sulfide nanoparticles onto a positively-charge polymer (poly(ethyleneimine)) (PEI) forming the first coating layer. Since siRNA were easily degraded, an outermost coating of silk was applied to the CuS-siRNA-PEI nanoparticle to stabilize the therapeutic agent. Both dip coating and LbL assembly, though facile, can cover pores especially in porous silk drug delivery systems. Therefore, it is essential to know the physical properties of the material used as a coating to know which physical coating strategy can be applied to achieve the target release profile.
In some cases, two or more physical functionalization strategies can be applied to silk drug delivery systems to prolong the release and enable maximum in vivo efficacy (Table 1, entries 22–24).81,82,97 Silk fibroin and hydroxypropyl cellulose (HPC) composite hydrogel was used to deliver folate-modified AC16 cells encapsulated in extracellular vesicles in situ in Sprague–Dawley rats for myocardial infarction repair (Table 1, entry 23).82 Best cardiac repair result was recorded for the rats treated with silk/HPC composite hydrogel loaded with folate-modified extracellular vesicles with left ventricular ejection fraction of ∼71% compared to the untreated group with ∼45% after 28 days of treatment. Extracellular vesicles (EVs) are cell membrane particles around 30 nm to 10 μm in diameter that contain lipids, proteins, and nucleic acids and are recently discovered to regulate myocardial fibrosis after infarction.102–104 In this study, EVs were used to encapsulate folate-modified AC16 cells – stem cells that could differentiate into mature cardiac cells before incorporating in the silk/HPMC composite material and delivering to the target site in the form of a hydrogel. EVs are good encapsulating agents as they are naturally formed by human cells hence they are stable inside the body, naturally biocompatible, and possess low immunogenicity.104,105 However, some of the limitations of using EVs include low drug loading, lack of standardized procedures for isolation and purification, and insufficient methods to upscale production.106 Hence, therapeutic agents that can be loaded in EVs should have therapeutic dose that only require nano- to micromolar amounts and other encapsulating agents can be considered if a higher drug dosage is required.
| Entry no. | Type of reaction | Chemical crosslinker | Type of bond/products formed | Ref. |
|---|---|---|---|---|
| a Similar mechanism as with enzyme-catalyzed crosslinking. | ||||
| 1 | Glutaraldehyde crosslinking |

|
|
113 |
| 2 | Carbodiimide coupling |

|
|
113 |
| 3 | Genipin crosslinking |

|

|
114 and 115 |
| 4 | Epoxide ring opening |
|

|
116 |
| 5 | Enzyme-catalyzed crosslinking |

|

|
117 and 118 |
| 6 | Photocrosslinking |

|

|
49 |
| 7 | Photocrosslinking |

|

|
49 and 119 |
| 8 | Thiol–ene “click” reaction |

|

|
120 |
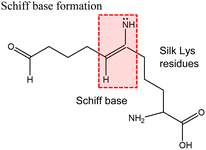
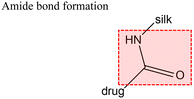
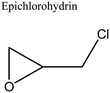
For instance, dopamine is known to form a covalent bond via Michael addition or Schiff base formation of the catechol, imine, and amine groups of dopamine with free oxygen radicals and therefore, reactive oxygen species (ROS) is decreased when used as crosslinker in silk drug delivery systems which helps in diabetic wound healing.121,122 Epichlorohydrin (ECH) can react with the hydroxyl and amino groups of various polymers including silk via epoxide ring opening or nucleophilic attack on hydroxyl groups, resulting in a silk drug delivery system with better mechanical stability which could be particularly useful as implants or scaffolds for tissue engineering (Table 2, entry 4).123,124 Horseradish peroxidase catalyzes crosslinking reactions under mild conditions during chemical functionalization of silk drug delivery systems. Silk, having aromatic phenol group, amine, and phenolic acids, can act as a reducing substrate whereas hydrogen peroxide can be added to the solution to act as the oxidizing substrate to drive the formation of covalent bonds via a cationic radical polymerization using the heme group of horseradish peroxidase (Table 2, entry 5).117 Depending on the amount of the crosslinker and hydrogen peroxide, the mechanical strength and biodegradability of the silk drug delivery system can be tuned for the desired application. Since the polymerization reaction was performed under mild conditions to preserve enzymatic activity, the activity of the therapeutic agent can be preserved as well. Riboflavin, when exposed to UV, is excited to form reactive singlet oxygen (1O2) which attacks the tyrosine residue of silk to form a tyrosine radical intermediate. Hydrogen peroxide acts as an electron acceptor forming dityrosine linkages in silk drug delivery systems (Table 2, entry 6).125,126 Sodium persulfate is another crosslinker that can be used to form dityrosine bonds in silk. It acts as an electron acceptor upon activation of a photoinitiator ruthenium(II) hexahydrate (Ru(II)(bpy)2+) (Ru) which could propagate by attacking the silk tyrosine residue to form a tyrosine radical intermediate (Table 2, entry 7).119 Since silk is mainly composed of tyrosine residues (∼5.1 mol% or ∼277 residues per protein),35 there will be more bonding sites for riboflavin and ruthenium, increasing the polymerization duration which might not be suitable for in situ applications.96 However, the dityrosine linkages formed make the silk or silk composite material more robust and mechanically stable.127 Poly(ethylene glycol diacrylate) (PEGDA) has also been used as crosslinkers in silk drug delivery systems because of its ability to be photocrosslinked and to form three-dimensional (3D) polymer structures that can swell and absorb water. These 3D structures are formed by thiol–ene click reaction of PEGDA with silk. In this reaction, the acrylate groups in PEGDA will form radicals upon UV exposure and will react with the thiol groups from the cysteine residues of silk (Table 2, entry 8).120 Unlike when riboflavin is used, thiol–ene click reactions with PEGDA are rapid since cysteine residues in silk are not as abundant as tyrosine residues and therefore, much applicable to in situ applications. The specificity of the click reactions between PEGDA and silk can also enable the functionalization of smart polymers (“stimuli-responsive”) materials which allows for a much controllable release of encapsulated therapeutics. Glutaraldehyde is regarded as the most commonly used chemical crosslinker in silk drug delivery systems because it is readily accessible and does not involve several precursor steps to bind the target molecule of interest.107–109 The crosslinking mechanism involves formation of Schiff bases between the two carbonyl ends of glutaraldehyde and positively-charged amino groups on the lysine residues of the silk protein (Table 2, entry 1). Meanwhile, 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) is another crosslinker that couples N-hydroxysuccinamide (NHS) with silk to form amide bonds (Table 2, entry 2).128 In this crosslinking reaction, EDC activates the carboxyl group of silk to form an unstable O-acylisourea intermediate. The primary amines in silk attacks the O-acylisourea intermediate via a nucleophilic substitution reaction to form amide bonds. NHS is often added to form a more stable ester with the O-acylisourea intermediate which can also react with the primary amines of silk. As a result, a higher crosslinking efficiency is achieved and with less toxicity than glutaraldehyde.129 Genipin, a crosslinker derived from gardenia fruits, has a covalent interaction with silk via the olefinic carbon at C3 position in genipin which is attacked by the amino group of silk under acidic and neutral conditions. Subsequently, a second condensation reaction occurs wherein the dihydropyran ring in genipin opens and the secondary amino group attacks the newly formed aldehyde group (Table 2, entry 3).112,130,131 Genipin is the least toxic crosslinker among glutaraldehyde and EDC and has been increasingly used for silk drug delivery systems applied in vivo.
Recent strategies used to form covalent bonds in silk drug delivery systems for prolonged drug release include grafting another layer of polymer containing the therapeutic agent on the silk surface to form a covalently-bonded coating (Fig. 3) or direct attachment of the therapeutic agent towards the silk backbone using chemical crosslinkers. Grafting involves rearrangement of chemical bonds (“grafting to” and “grafting through”) (Fig. 3(a) and (c)) or formation of radicals (radical polymerization, “grafting from”) (Fig. 3(b)) on the polymer containing the therapeutic agent which could be triggered by various stimuli such as temperature, pH, UV light, plasma, or enzyme catalyst132 before incorporating with silk. On the contrary, direct attachment of the therapeutic agent towards the silk backbone involves activation of the drug to enable binding sites for the chemical crosslinkers. The crosslinkers serve as a bridge that connects the drug towards the silk substrate via covalent bonds such as amide bonds using EDC or Schiff bases using glutaraldehyde to reversibly or irreversibly bind silk to the drug adding a degree of controllability to the release of therapeutics incorporated within the silk drug delivery system while enabling slower release of drugs.
In vivo studies have presented the use of chemical crosslinking to fabricate more stable silk drug delivery systems in tandem with other physical functionalization techniques to further prolong drug release. Glutaraldehyde crosslinking has been employed to fabricate silk scaffolds as wound dressings (Table 1, entries 26, 27).84,85 Glutaraldehyde crosslinked the porous chitosan–alginate layer to form the bottom hydrogel layer of the scaffold which incorporates the active therapeutic agent, 10-hydroxydecanoic acid (10-HDA), also known as the “queen bee acid”. It is found exclusively in royal jelly and has anti-inflammatory,133 antimicrobial,134 and immune system-modulating properties.135 Electrospinning was utilized to form the poly(caprolactone)/silk sericin surface layer for the glutaraldehyde-crosslinked chitosan–alginate hydrogel scaffold. It was observed that fibrosis was less prominent with the 10-HDA-containing wound dressing than the control (without 10-HDA) after 14 days from transdermal application in Wistar rats (Table 1, entry 26).84 When astaxanthin, an antioxidant, and silver nanoparticles, an antimicrobial were incorporated in electrospun poly(vinyl alcohol)/silk fibroin nanocomposite membrane, wound area induced in Sprague–Dawley rats was reduced to ∼4–12% as compared to the membranes without the therapeutic agents with wound area reduced to ∼22% after 14 days. In these membranes, glutaraldehyde was used to crosslink poly(vinyl alcohol), silk fibroin, and the therapeutic agent. Meanwhile, crosslinking using EDC/NHS in silk drug delivery systems has also allowed for the wound treatment in Wistar rats and BALB/C mice for approximately two weeks. For the treatment of burn wounds in Wistar rats (Table 1, entry 29),87 vancomycin – a potent antibiotic that inhibits growth of methicillin-resistant Staphylococcus aureus (MRSA), was loaded in conductive hydrogels based on tragacanth – an anionic polysaccharide that accelerates wound regeneration,136 and silk fibroin. EDC was used to conjugate vancomycin with silk fibroin and to functionalize the silk fibroin/tragacanth composite with a conductive carboxyl aniline pentamer. Meanwhile, EDC was used to conjugate gallic acid to a reactive oxygen species scavenging dendrimer prior to blending with poly(vinyl alcohol) and silk fibroin composite to form a hydrogel that can promote wound healing in BALB/C mice (Table 1, entry 30).88 Genipin was used to crosslink silk fibroin with gelatin to form silk fibroin/gelatin films (Table 1, entry 31)89 and teicoplanin-decorated reduced graphene oxide to form silk protein hybrid hydrogels (Table 1, entry 32)90 to regenerate the tympanic membrane and accelerate diabetic wound healing in Sprague–Dawley rats and BALB/C mice, respectively. There was a 60% tympanic membrane regeneration compared to the control after 5 days89 and an accelerated healing of infected wounds in diabetic BALB/C mice was observed after 14 days of treatment.90 Quercetin loaded in epichlorohydrin-crosslinked silk/soy protein isolate hydrogel facilitated wound closure rates of ∼79% compared to ∼48% in the control group after 17 days of treatment (Table 1, entry 34).92 When bone marrow mesenchymal stem cells were loaded in dual network DNA-silk fibroin hydrogels, it was observed that there was significantly accelerated cartilage regeneration after 14 days in Sprague–Dawley rats (Table 1, entry 35).93 Since enzyme crosslinking using horseradish peroxidase did not involve harsh chemicals that could influence the chemical properties of both the silk/silk composite substrate and the therapeutic agent (stem cells), significant improvement was observed in vivo. PEGDA has been used in a couple of studies as a crosslinker in a photo-initiated functionalization of hydrogels (Table 1, entries 36 and 42).94,100 Platelet-rich plasma lysate was loaded in a silk-based photocurable hydrogel for the treatment of knee cartilage (meniscus) tears in New Zealand rabbits.94 Methacrylate functional groups have been added to the silk and gelatin composite substrate using lithium phenyl-2,4,6-trimethylbenzoylphosphinate as initiator and PEGDA crosslinker which aided in better distribution and defect coverage of the hydrogel in rabbit menisci (Table 1, entry 36).94 Chondroitin sulfate, a building block of human cartilage,137 was also methacrylated using methacrylic anhydride and PEGDA to form silk fibroin/chondroitin sulfate methacrylate hydrogel bone screws for the treatment of osteoarthritis (Table 1, entry 42)100 By UV crosslinking of chondroitin sulfate with silk using PEGDA, it was observed that bone screws were successfully implanted into the femur of white rabbits and were integrated well into the implantation site after a week. This could be attributed to the formation of macromolecular network framework which incorporated beta-sheet crystalline building blocks and preserved the mechanical integrity of the bone screws. Riboflavin has been an effective crosslinker as well as it enabled bone/wound healing and diabetes management by carrying adipose stem cells and insulin, respectively (Table 1, entries 38, 39).96,97 Silk hydrogel bone screws formed from photocrosslinking silk substrate using riboflavin in the presence of hydrogen peroxide did not induce serious inflammation and maintained bone screw shape after 14 days after subcutaneous implantation of the adipose stem cell-carrying hydrogel in Sprague–Dawley rats (Table 1, entry 38).96 When silk fibroin was blended with methacrylated hyaluronic acid (HAMA) – a stimuli-responsive polymer, and were photocrosslinked with using riboflavin, insulin was regulated and healthy blood glucose levels were maintained close to 6 h in C57BL/6 mice compared to the control group without insulin due to the regulation of the macromolecular network density, which swells upon absorbing tissue fluid after insertion into the subcutaneous tissue (Table 1, entry 39).97 Layer-by-layer assembly was the adapted technique to the blend to ensure that there was controlled regulation of insulin after transdermal application of the hydrogel microneedles.
Chemical crosslinkers stoichiometrically reacted with a certain compound138 and excess unreacted drugs could influence release rates through initial burst release. A striking observation when chemical crosslinkers were used to bind drugs to the silk material was some drugs were only partially released despite the prolonged period of drug release monitoring which could be quite counterproductive as this could hinder reaching the drug therapeutic window when applied in vivo. Despite the recorded sustained release of drugs in various silk formats using these crosslinkers, careful evaluation of the release profiles upon consideration of various factors such as the bonds and the by-products formed, the reversibility of the release, and the drug activity, biocompatibility, and biodegradability of the material after crosslinking must be performed to ensure that there are no cytotoxic by-products formed, the release was prolonged, and the drug activity was preserved even after covalent interaction with the chemical crosslinker.
3. Silk formats used in drug delivery systems
Several different material formats could be molded from the silk solution by controlling its physico-chemical properties and conformation. Some of the most common forms utilized in current silk drug delivery systems are gels, films/membranes, particles/spheres, and microneedle patches (Fig. 4). | ||
| Fig. 4 Various silk formats adapted as drug delivery systems. Silk can be prepared as (a) hydrogels (reprinted with permission from Wang, et al. ©2014, The Author(s)109), (b) aerogels (reprinted with permission from Maleki, et al. ©2019, American Chemical Society139), (c) films (reprinted with permission from Wang, et al. ©2022, The Author(s)140), (d) electrospun membranes (reprinted with permission from Xie, et al. ©2019, The Author(s)141), (e) particles or spheres (reprinted with permission from Wang, et al. ©2009, Elsevier Ltd. All rights reserved.142), and (f) microneedle patches (reprinted with permission from Gao, et al. ©2019, American Chemical Society.143). | ||
3.1. Gels
Gels are semisolid three-dimensional polymer networks, with a solid phase dispersed in a liquid (hydrogels) or gas phase (aerogels/foams).144 Although the liquid/gas phase comprise the majority of the gel composition, these materials exhibited more solid-like properties.144 | ||
Fig. 5 Formation of injectable silk hydrogels. (A) Temperature-induced formation of Bombyx mori and Antheraea assamensis-blended silk fibroin hydrogels (reprinted with permission from Jaiswal, et al., ©2022, Elsevier B.V. All rights reserved.145). (B) Polymer additive-induced formation of silk hydrogels using ethylene glycol/tri(ethylene) glycol (formulation of silk fibroin![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ethylene glycol ethylene glycol![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) tri(ethylene glycol) is 60 tri(ethylene glycol) is 60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) : :![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20) (reprinted with permission from Maity, et al., ©2020, American Chemical Society148). (C) Sonication-induced formation of silk fibroin hydrogels with miR-675-loaded stem cell-derived exosomes (reprinted with permission from Han, et al., ©2019, The Author(s). Published by Elsevier B.V.151). 20) (reprinted with permission from Maity, et al., ©2020, American Chemical Society148). (C) Sonication-induced formation of silk fibroin hydrogels with miR-675-loaded stem cell-derived exosomes (reprinted with permission from Han, et al., ©2019, The Author(s). Published by Elsevier B.V.151). | ||
Doxorubicin (chemotherapeutic drug) and desferrioxamine (drug for neovascularization) release were sustained for up to two months after administering the temperature-induced silk hydrogel intratumorally or in situ in female BALB/c nude mice146 and Sprague–Dawley adult male rats.147 The results could be attributed to the more ordered structure of the β-sheets which trapped the drugs upon heating. Although promising, the use of temperature in silk hydrogel formation was not applicable to incorporate drugs or therapeutic agents that were unstable or lose activity at very high temperatures. Therefore, other methods of silk hydrogel formation could be adapted such as the use of biocompatible polymer additives and sonication to deliver temperature-sensitive drugs or therapeutic agents. When polymer additive-induced silk hydrogel delivered insulin in Wistar rats,148 and vancomycin subcutaneously and in situ149 as treatment for diabetes and chronic osteomyelitis (bone infection), respectively, release of therapeutics was recorded to be after five days to a week. Typically, diabetes treatment for type 2 patients lasts for up to two days and the silk-EG/TEG hydrogel drug delivery system being able to sustain the release of insulin for up to five days promotes glucose homeostasis in diabetic rats.148 A sustained release of ≥3 months for bevacizumab, a therapeutic agent (monoclonal antibody) that has an affinity against vascular endothelial growth factor (VEGF), was recorded in Dutch-belted rabbits when sonication-induced silk hydrogels were used to encapsulate this therapeutic agent for the potential treatment of age-related macular degeneration (AMD).150 Low ultrasonic energy was applied to ensure that there was minimal increase in temperature during sonication that induced rapid gelation, allowing for more interaction between bevacizumab and silk. The therapeutic threshold for typical hydrogel formulations for bevacizumab was one injection per month which meant that sonication was able to prolong the release of the anti-VEGF, bevacizumab. Significantly enhanced blood perfusion was also observed in C57BL6 mice on day 28 when sonication-induced silk hydrogel was used to encapsulate miR-675, a modulator of aging process via a stem cell-derived exosome for the treatment of vascular diseases although it should be taken note of that a temperature of 37 °C was applied too (Fig. 5(c)).151
A non-injectable, sonication-induced heparinized-silk fibroin hydrogel was used to encapsulate fibroblast growth factor (FGF-1) for wound healing applications.157 A burst release (43.7%) was recorded in the first 12 h after application on the wound site on Sprague–Dawley rats and a slow release (70%) was observed over the course of 5 days. A study also showed that a 3D-bioprinted non-injectable silk fibroin/gelatin hydrogel delivered trans-dermally was able to reduce the wound area of wounded Kunming mice compared to the control group after two weeks which could be attributed to the reduced viability of bacteria S. aureus and E. coli only after 20 min in vitro.159
Despite the preference on silk hydrogels over other formats, silk hydrogels remain to have limitations on its mechanical integrity and therefore, optimization is still necessary for the formulation of silk hydrogels that enables better mechanical strength in vivo while ensuring degradation rates are rapid enough to minimize adverse physiological reactions.
In vitro release studies showed that some silk foams were capable of sustaining release for longer periods depending on the fabrication method which influences foam morphology, the functionalization technique applied on the material which influences material-therapeutic agent interaction, and the chemical properties of the therapeutic agent which influences diffusion of the drug from the material. For instance, silk fibroin xerogels sustained the delivery of estradiol (Fig. 6(a)), a hormone used for contraception and hormone replacement therapy (HRT) by enabling slow release (∼90%) after 129 days (∼four months).162 Silk xerogel morphology for the delivery of estradiol (Fig. 6(a)) showed a compact surface with pores sparsely distributed on the xerogel surface which could have prevented initial burst release. Meanwhile, the therapeutic level for estradiol had a wide range with hardly any published literature for its specific therapeutic index.168,169 Estradiol was observed to form crystals when the lyophilized foams were treated with ethanol for β-sheet formation, with estradiol lipophilicity also contributing to its slow diffusion in the aqueous release bath. Aerogels fabricated using scCO2 drying were also utilized to deliver ibuprofen (Fig. 6(b)) and a release of ∼90% after 6 h was observed.163 A highly porous surface morphology with more evenly distributed pores could have contributed to this result (Fig. 6(b)). Ibuprofen also had a wide therapeutic range (800–1200 mg day−1 at low, over-the-counter doses; 1800–2400 mg day−1 at prescription doses),170,171 and the drug release behavior must be controlled depending on whether it was used acutely, as an antipyretic or temporary pain reliever or chronically, for the long-term treatment of arthritis, ankylosing spondylitis, and other long-term conditions.171 For ibuprofen-loaded aerogels, the majority of the drug was released within a short time period (6 h) since physical adsorption between the silk and the drug was the prevailing interaction for the whole system which was ideal since ibuprofen should not exist in increased levels in the body for a prolonged period to prevent hepatotoxicity.172 Lyogels were also previously used to deliver murine anti-TGFβ monoclonal antibody and it was observed that using this silk format, sustained release of the antibodies (∼15–70%) can be achieved until 38 days.165 Lyogels, most particularly those with higher silk content, were more resistant to swelling due to its solid structure. In addition, the silk-antibody interaction was strengthened upon water removal resulting in prolonged antibody release. It was also found that using this silk format, murine anti-TGFβ monoclonal antibody activity lie within the expected range which meant that the released antibody was stable even after 38 days. Ciprofloxacin and a protease enzyme were also loaded into a cryogel (Fig. 6(c)) and showed a controllable drug release (∼20%) until about 50 days when the drug in powder form was loaded.166 Cross-sectional micrographs (Fig. 6(c)) showed a stacked sheet-like structure which could have served as a barrier for the antibodies that were bound to the innermost region of the lyogel enabling the release to be sustained for around a month. The antibiotic also has retained its activity as manifested in the clearing zones observed from the antibacterial assay. 3D-printed aerogels were also used to deliver sorafenib (SFN) (Fig. 6(d)) during photothermal bone cancer therapy,167 and it was observed that ∼70–90% was released for up to around seven hours. 3D printing allowed for the fabrication of a dual network structure leading to the attainment of a hierarchically organized porosity for a more controlled release (Fig. 6(d)). Still, careful consideration must be given to the method of drug incorporation (i.e. physical blending or chemical crosslinking),173 the composite materials to be added for better dissolution or dispersion of the drug (i.e. silica, MXenes),167,174–176 and the solubility of the drug towards the chosen solvent (i.e. use of binary solvents)177 for the silk bio-ink to ensure that more therapeutics were internalized within the 3D-printed structure.
 | ||
| Fig. 6 Morphological characteristics of silk (a) xerogels loaded with estradiol (reprinted with permission from Krizman, et al., ©2021, Published by Elsevier B.V.),162 (b) aerogels made from drying using supercritical carbon dioxide loaded with ibuprofen (reprinted with permission from Marin, et al., ©2014, Elsevier B.V. Published by Elsevier B.V. All rights reserved.),163 (c) cryogels loaded with ciprofloxacin hydrochloride (reprinted with permission from Chambre, et al., ©2020, American Chemical Society.),166 (d) lyophilized 3D printed aerogels loaded with bismuth sulfide nanorods (reprinted with permission from Al-Jawuschi, et al., ©2023, American Chemical Society).167 | ||
A current study on the in vivo bone regeneration in Sprague–Dawley rats showed that bone volume fraction was higher (∼46%) in cell-free chitosan/silk fibroin composite scaffolds loaded with bioactive glass compared to the control (without bioactive glass) (∼42%) at 12 weeks of treatment. The sustained release could be attributed to the very complex pore architecture brought by the fabrication method (radial freezing technique) which prevented the ready diffusion of bioglass from the composite scaffold (Table 1, entry 19).77
The silk foam format is mainly for the purposes of increased drug loading compared with other silk formats (i.e. aerogel porosity) and mechanical stability (i.e. flexibility). The three-dimensional (3D), porous structure of silk aerogels results in a high surface area (∼200–400 m2 g−1)164 which can be utilized to load higher drug concentrations for localized delivery of therapeutics.167 Opting for silk aerogel format would be beneficial if there was an involvement of organic solvent or other stimuli such as extreme temperature178 and pH179 during the fabrication process which could alter the mechanical stability of the silk aerogel upon delivery of a drug or therapeutic agent to the target site.
3.2. Films/membranes
Silk films/membranes (Fig. 4(c) and (d)) are two-dimensional drug delivery systems. Like foams, silk films are highly applicable as scaffolds for bone and tissue regeneration or as implants or dressings for wound healing because of its sheet-like geometry and higher mechanical integrity than hydrogels (Table 1, entries 6, 7, 11, 13, 15, 17, 18, 21, 26, 27, 31, 33, 37, and 41). This silk format was primarily fabricated via the conventional dry casting wherein the silk solution is drop-casted in a poly(styrene) Petri dish,108,180,181 a glass plate,52 or relatively inert mold182 and is allowed to dry in ambient conditions but in some applications (i.e. corneal tissue engineering183 and scaffolds for neuronal growth184), centrifugal casting technique (centrifugal force is applied on an aluminum tube with silk fibroin solution), and electrospinning185 are also utilized. Electrospinning141 is another common method used to fabricate silk membranes, which can be also loaded with drugs. Homogeneity/solubility of the therapeutic agent on the spinning solution was mainly important for slower release of the drug.141,184,185 Film/membrane structure is more controllable in electrospinning than solution casting since formation of solid matrix occurs inside an electrospinning machine wherein the main driving force of film/membrane formation is the tunable electric current whereas in solution casting, it happens mainly via solvent evaporation. However, a more mechanically stable film/membrane can be formed via solution casting than electrospinning since silk proteins can self-assemble during casting therefore reinforcing film/membrane mechanical properties compared to irregularly oriented fibers than can be obtained on the electrospun mats.186 In both cases, drug chemistry, particularly its solubility and charge, has a significant effect in its incorporation inside the film/membrane which can ultimately influence its release. In terms of release behavior, the key to prevent burst release and sustaining the amount of drugs released over time for this material format was to increase the number of β-sheets to improve silk film crystallinity and increase the interpenetrating networks (physical crosslinks) which could trap the small drug molecules and restrict film swelling thus hindering fast diffusion of drugs towards the release bath.187 The most common solvents used to form silk films/membranes with high β-sheet content were formic acid,188 and 1,1,1,3,3,3-hexafluoroisopropanol (HFIP).189 Conformational change that promoted tighter interactions between silk molecules causing increased number of β-sheets in silk films was brought by the hydrogen bonds that could be formed between the hydroxyl groups of formic acid,188 HFIP,190 and silk upon the evaporation of the solvents.Annealing is another strategy used to increase the β-sheets in the films by providing sufficient thermal energy for large-scale molecular motion of protein chains; therefore, allowing for faster crystallization due to the kinetic motion of the molecules.191 The annealing process could be water-based37,181 or alcohol-based (commonly methanol/ethanol).181,192 Glycerol or poly(vinyl alcohol) could be added as a plasticizer to improve film flexibility for annealed films.193,194
A number of current studies has incorporated flavonoids in silk nanofibrous mats for wound healing in rats (Table 1, entries 6 and 15).65,73 Flavonoids are a class of plant-derived compounds characterized by polyphenol groups in their chemical structure which are found to have wound–healing properties attributed to their anti-inflammatory, angiogenesis (formation of new blood vessels), re-epithelialization (restoration of skin barrier), and antioxidant effects.195 Full wound closure can be observed from 9–15 days after transdermal application of the silk nanofibrous mats. In other studies, silver nanoparticles has been incorporated with silk composite materials for better wound healing in rats (Table 1, entries 11 and 27).69,85 Silver nanoparticles has been known to aid in wound healing by controlling the release of cytokine (proteins that act as immune system messengers) which are responsible for inflammation.196 A healing percentage of ∼88–99% was recorded after 14–21 days of treatment when silver was combined with gold nanoparticles69 or astaxanthin, a carotenoid found to have high antioxidant properties and promotes collagen production85 prior to incorporation with silk fibroin/collagen blend or silk fibroin/poly(vinyl alcohol) composite, respectively. Berberine, also a plant-derived compound that belong to alkaloid class with similar functions as flavonoids, has been loaded in electrospun silk fibroin blended with biodegradable polymer such as poly(hydroxybutyrate-co-valerate) (PHBV) or poly(caprolactone) (PCL) for wound healing in diabetic mice (Table 1, entries 17 and 18). It was observed when berberine-loaded electrospun silk fibroin/poly(hydroxybutyrate-co-valerate) (PHBV) wound dressing was applied on diabetic mice, wound healed completely at a significantly higher rate (∼99.7%) than the control group (∼83–85%) after 18 days treatment (Table 1, entry 17).75 A more sustained release was also recorded for berberine-loaded silk fibroin/poly(caprolactone) (PCL) nanofibrous mat where bone regeneration can be observed after 2–6 weeks (Table 1, entry 18).76 The differences in release rates could be due to the faster degradation rate exhibited by PHBV than PCL which enabled more berberine to be released at a higher dose at a shorter period of time. Another therapeutic agent such as honey extract from bees loaded in silk fibroin mesh fabricated by dip coating silk membrane in a mixture of poly(hydroxybutyrate) (PHBV) or poly(caprolactone) (PCL) also resulted in faster wound healing 28 days after implantation (Table 1, entry 21).79
Gelatin was blended with silk fibroin to form composite films that were used for tissue regeneration in rats (Table 1, entries 31 and 33).89,91 It was observed that tympanic membrane has regenerated and 60% of the perforation has been covered compared to 40% in control five days after surgical implantation (Table 1, entry 31).
Growth factors have been also incorporated within silk and silk composite scaffolds for acute liver failure treatment and spinal cord repair in rats and mice (Table 1, entries 7, 37, and 41).66,95,99 A combination of growth factors such as hepatocyte growth factor (HGF)/fibroblast growth factor-4 (FGF-4) loaded in silk electrospun scaffolds aided in reducing the biomarker concentration for hepatocyte damage in BALB/C mice after 14 days of transplantation on injured livers (Table 1, entry 7).66 Basic fibroblast growth factor was also loaded in silk nanofibril/hyaluronic acid scaffold for spinal cord repair in Sprague–Dawley rats (Table 1, entry 37). In the case of spinal cord injury repair, motor function was recovered 8–10 weeks after subcutaneous implantation of silk nanofibril/hyaluronic acid scaffold loaded with basic fibroblast growth factor (Table 1, entry 37)95 and dual-phase silk fibroin methacryloyl (SilMA) hydrogel scaffold (DPSH) loaded with neurotrophin-3 (NT-3) and angiotensin (1–7) (Ang-(1–7)) (Table 1, entry 41),99 respectively.
Ultimately, the severity of the wound or tissue damage will determine how the silk film/membrane can be fabricated to ensure the highest therapeutic agent loading and delivery towards the target site for faster recovery. Sustained release of therapeutic agents is necessary for diabetic wound healing and spinal cord repair hence the use of materials that could form composites with silk must not degrade at a relatively fast rate. Otherwise, if the silk film/membrane is to be applied for normal wound healing, faster degradation rate of the material is preferable.
3.3. Particles
Silk particles (Fig. 4(e)) are one-dimensional formats used to encapsulate the drug/therapeutic agent of interest. They can exist from nano- to micro- range and display fast release rates when directly applied to the target site due to high surface area.197–199 Silk particles can also be embedded or incorporated within scaffolds/implants to sustain the release of the therapeutic agent for a longer duration.200–202Silk particles can be synthesized via salting out,203 nanoprecipitation,198,204,205 emulsification,200,206 coaxial electrospraying,201 spray drying/spray freeze drying,202 and semi-batch silk desolvation. Salting out is a process wherein silk particles are formed by adding high concentrations of salt in the silk solution.207 The salt ion can be a kosmotropic agent which competitively interacts with the water molecules that forms hydrogen bonds with the amide moieties in silk, therefore facilitating dehydration and favoring precipitation of the protein resulting in the formation of silk particles.207 By selecting the proper type of salt to use during the “salting out” process, the dynamic transition of the aggregate and the controllability of particle formation can be achieved.208 Nanoprecipitation is another method of silk particle formation wherein the aqueous silk solution is dissolved in a partially miscible organic solvent from which the heavy-chain hydrophilic blocks have low solubility.209 Organic solvents can also serve as kosmotropic agents as they can induce protein aggregation by lowering dielectric constant and increasing protein–protein interactions.210 Emulsification is another technique of silk particle formation wherein an aqueous phase is dispersed in an oil phase to form water-in-oil (W/O), oil-in-water (O/W), or water-in-oil-in-water (W/O/W) emulsions upon application of an external force (i.e. ultrasonication). For the fabrication of silk particles using this technique, silk aqueous solutions are often dispersed in a continuous oil phase, or another immiscible liquid, to form the particles or spheres.211 Silk, being an amphiphilic multi-block copolymer by itself, serves as an emulsion stabilizer as it could interact and self-assemble between two phases, decreasing interfacial tension.212 Coaxial electrospraying involves ejecting the silk polymer solution via electrostatic forces forming droplets which eventually turn into nanoparticles upon solvent evaporation.201 This technique is promising as it does not require the emulsification step or further purification which could alter silk properties and reduce the activity of the encapsulated therapeutic agent.201 Meanwhile, for spray drying, a spray dryer is used to aerosolize the silk solution and spray it into a heated drying chamber to evaporate the solvent and leave a solid particle. In spray freeze drying, silk solution is atomized using a nozzle and sprayed into liquid nitrogen prior to lyophilization.
Silk particles has been mostly used to encapsulate drugs to treat tumors that cause cancer (Table 1, entries 9, 22, and 25).56,80,83 Doxorubicin hydrochloride and small interfering RNAs (siRNAs) were loaded in silk/poly(D,L-lactic acid) microspheres and silk fibroin shell, respectively whereas a tumor-targeting peptide was coated with silk to form nanorods as cancer treatment in BALB/C nude mice. When these were administered intratumorally, tumor volume is minimal after about two weeks.
Metformin-loaded silk/poly(vinyl alcohol) microparticles were used to treat corneal neovascularization (abnormal growth of blood vessels in a typically avascular cornea) in rats and showed reduced neovascularization and inflammation after seven days (Table 1, entry 10).68 Methylprednisolone acetate (MPA), a type of corticosteroid, was also loaded in silk fibroin/chitosan interpenetrating network hydrogel for cartilage regeneration in white rabbits (Table 1, entry 43).101 It was found that locomotor function in treated groups has improved after 12 weeks. Silk particles can typically sustain the release of drugs for at least two weeks and the introduction of a hydrogel network during fabrication can sustain the release of MPA from silk fibroin particles for up to 12 weeks.
Functionalized silk particles allowed for easier achievement of therapeutic dose since more drugs could be encapsulated or loaded and due to their size, they could also penetrate cell membranes or even the blood–brain barrier which was highly favorable for targeted or localized drug delivery.197,213 However, their size could also pose some disadvantages when it came to introducing the drug.214 Since cells internalized these materials easily at this form,213 careful optimization of the maximum noncytotoxic concentration of the drug loaded must be performed prior to and upon encapsulating with silk particles to ensure that the drugs would not agglomerate resulting in increased size.
3.4. Microneedles
Microneedles (Fig. 4(f)) are an array of micron-sized needles typically ranging from 150–1500 μm in length, 50–250 μm in width, and 1–25 μm in thickness attached on top of a supporting component that allow these needles to penetrate through the skin and deliver therapeutic agents transdermally without incurring pain to the patient.215 This format can be solid, coated, hollow, or dissolvable depending on the required mode of delivery. Solid silk microneedles (Fig. 7(a)) are often administered following a “poke-and-patch approach”216 wherein the microneedles directly puncture the skin surface.217 One limitation of solid microneedles was it was difficult to maintain therapeutic dose for this microneedle type due to the inherent ability of skin to heal over time.218 Silk-coated microneedles (Fig. 7(b)) is another type of microneedle applied using the “coat-and-poke approach”216 wherein therapeutic agents are coated on the tip of the microneedle before administration. This technique might not be the best for some drugs that were attached only via electrostatic interaction or physical adsorption since the premature release could occur unless initial burst release was sufficient to deliver the therapeutic level of the drug.219,220 For instance, immobilization of a peptide within a coated silk microneedle for insulin delivery only required 15 min for a therapeutic level to be delivered. Dissolvable microneedles (Fig. 7(c)) have tips made up of soluble polymers with encapsulated therapeutic agents.216,221 Although less expensive and enabled higher dose of drugs via encapsulation than coated microneedles, the “poke-and-dissolve approach” has its limitations in terms of other “non-drug” components which can also be degraded during the delivery process.222 Finally, silk swelling or hydrogel microneedles also existed (Fig. 7(d)) wherein a swellable material was used to control drug release.216,223 This microneedle type was particularly interesting to use in smart release systems triggered by various stimuli due to the maneuverability of its degree of swelling.223,224 However, one downside to this approach was that highly tunable silk hydrogel microneedles had trade-offs in mechanical integrity. To counter this, a new class of porous microneedle had emerged using a mechanically stable photocurable resin as substrate resulting in the preservation of the hardness of the microneedle during delivery.222 | ||
| Fig. 7 Silk microneedle types. (a) Solid microneedles for levonorgestrel delivery (reprinted with permission from Yavuz, et al., ©2020, American Chemical Society217), (b) coated microneedles for Rhodamine B (model drug) delivery (reprinted with permission from Lin, et al., ©2020, The Author(s)25), (c) dissolvable microneedles loaded with antigen for vaccine delivery (reprinted with permission from Boopathy, et al., ©2019, The Author(s)221), (d) swellable microneedles for FITC-Dextran (model drug) release (reprinted with permission from Yin, et al., ©2018, Elsevier B.V. All rights reserved.223). | ||
Silk fibroin microneedles loaded with melatonin for circadian rhythm regulation in Sprague–Dawley rats have exhibited burst release upon administration as it induced sleep immediately but was able to sustain the release after 8 h after melatonin concentration peaked after about an hour (Table 1, entry 4).63 A dissolving silk microneedle system loaded with a topical antibiotic mupirocin and silver nanoparticles was used to treat delayed wound healing in BALB/C mice and it was observed that wound contraction rate increases to ∼78% 11 days post-treatment (Table 1, entry 23).81 When insulin was loaded in silk fibroin/hyaluronic acid hydrogel microneedles for diabetes management in C57BL/6 mice, it was reported that there was a gentle regulation and prolonged maintenance of healthy glucose blood levels for close to six hours compared to the blank group (Table 1, entry 39).97 Silk microneedles used in current in vivo studies did not require high doses of therapeutic agents for effective activity which made it suitable for these applications. In addition, a rapid effect is required for some instances (i.e. sleep induction or diabetes management) hence, burst release is not an issue. However due to the high surface area brought by the morphology of silk microneedles (pointed geometry), it is recommended to maximize the utility of this format in terms of drug/therapeutic agent loading for a more effective and rapid drug action.
4. Conclusions and perspectives
Based on recent studies, most silk functionalization methods are oriented more towards blending or coating another biocompatible/biodegradable material with silk rather than conjugating silk with the drug or therapeutic agent for sustained release in vivo. This technique is highly attributed to the effect of the interpenetrating networks between silk and the other material in “caging” the drug of interest and thereby slowing down its release. Although this strategy is effective in prolonging the release in some studies, maximum efficiency could not be achieved at a shorter period of time which could ultimately result in decreasing the overall treatment duration resulting in better patient comfort. Some factors that hinder the maximum efficiency of these silk drug delivery systems include limitations in drug loading due to the morphological properties of material format selected for a certain type of application resulting in the inability to reach the therapeutic threshold, the impact of the reagents used in functionalization of the drug delivery system in the mechanical stability of the material, and the biodegradability of the drug delivery system considering the standardized treatment protocol for a certain disease. There has also been an inadequate evaluation of the biocompatibility of other components aside from silk that were utilized to coat the therapeutic agent or the substrate. For instance, in the case of using chemical crosslinkers, increased concentration may probably be required to bind the therapeutic agent and the silk or silk composite substrate together to form a homogeneous surface coating or to sustain the release in vivo. Even though there are already individual studies suggesting the limited biocompatibility of some crosslinkers (e.g. EDC225 and genipin226), in vivo assessment of the concentration that could have leached from the substrate is lacking.To overcome these limitations in the practicality of current silk drug delivery systems towards in vivo testing, therapeutic agents or silk drug delivery systems were modified using hybrid techniques such as combining a coating strategy with other surface modification techniques (i.e. coating the silk substrate after embedding the therapeutic agent via physical means) to allow for higher drug loading concentrations consequently ensuring that the target release profile and the drug therapeutic level could be achieved. The concentration of toxic components that could leach from the sample could be minimized as well.
Interestingly, the preference on the use of silk hydrogel drug delivery systems in current in vivo studies than films or foams could be attributed to silk hydrogel biocompatibility. Aqueous silk solutions, which is the precursor of most silk hydrogels, could straightforwardly be treated with heat or sonication to drive the silk hydrogel formation and do not involve potentially harsh solvents or other polymers which could have a cytotoxic by-product. When it comes to aerogels (foams), 3D printing is gaining traction as a precise technique to achieve the desired material format and to also mitigate the use of excess chemical reagents (i.e. organic solvents used for the polymer dope solution) for enhanced biocompatibility since the amount of reagents can be automatically input in a 3D printing software.176 Despite the increasing popularity of 3D printing in aerogel fabrication, success in vivo is yet to be demonstrated since the form of the drug (i.e. pure powder or as conjugates) that could be encapsulated with foams are very limited.166 It might be necessary to synthesize several forms of drug to ensure maximum solubility or dispersibility with the silk solution hence leading to better release behaviors.
It must also be emphasized that consideration of drug properties and target application (i.e. skin patch, implant, inhalant, etc.) is highly significant. Despite silk's well-established reputation as versatile biomaterial, designing the most appropriate silk drug delivery system for a particular application is quite challenging since the silk format (i.e. gels, films, particles, etc.) and silk physico-chemical properties (i.e. molecular weight, isoelectric point, temperature stability, etc.) must be tailored to the drug's pharmacokinetics and intended application via various functionalization strategies to attain the desired release profile. Not to mention, it must be ensured that the activity of the drug/therapeutic agent after release was preserved.
Drug properties such as the size of the drug, lipophilicity, solubility, surface charge, half-life, isoelectric point, and acid-dissociation constant were some of the factors that must be considered along with the selection of the silk material format. For instance, the charge at a certain pH must be determined to know whether it would electrostatically bind to silk with a pI of 4.5 at that certain pH.227 During internalization of the drug with the silk, it would also be beneficial to know whether the drug would aggregate at the silk fabrication pH as this could possibly affect the drug release profile later on. By carefully studying the chemistry of the components and their compatibility with silk, it is possible to combine two or more therapeutic agents in one format. Selecting a simple approach to functionalize the silk drug delivery system (i.e. bulk blending) does not necessarily lead to burst release of drugs or therapeutic agents from the silk DDS but by comparing bulk-blended silk DDS over silk composite materials showed that shorter release times can be recorded from silk composite DDS, mainly due to insufficient understanding of the chemical properties of the two polymers being combined prior to encapsulating the therapeutic agent. Layer-by-layer technique can be very much applicable in nonporous silk drug delivery systems, wherein the release will be activated upon the degradation of the material. Dip coating can be used if there is a need to preserve the porosity of the substrate for the faster diffusion of drugs to the target site. Crosslinking between silk and another polymer is common to facilitate formation of drug delivery systems with better mechanical strength but studies showing crosslinking between silk and the therapeutic agent is less prominent because as this can alter the drug structure and hence, its activity/effectivity. Therefore, these factors must be considered in designing the preclinical models for the continuously emerging silk drug delivery systems.
Data availability
No primary research results, software or code have been included and no new data were generated or analyzed as part of this review.Conflicts of interest
There are no conflicts to declare.Acknowledgements
Dr Filippo Valente and Dr Christine Bacal would like to acknowledge Ear Science Institute Australia and the Channel 7 Telethon Kids Grant of Dr Filippo Valente for the support in the completion of this article.References
- A. M. Vargason, A. C. Anselmo and S. Mitragotri, Nat. Biomed. Eng., 2021, 5, 951–967 CrossRef PubMed.
- T. M. Allen and P. R. Cullis, Science, 2004, 303, 1818–1822 CrossRef CAS PubMed.
- J. K. Patra, G. Das, L. F. Fraceto, E. V. R. Campos, M. d P. Rodriguez-Torres, L. S. Acosta-Torres, L. A. Diaz-Torres, R. Grillo, M. K. Swamy, S. Sharma, S. Habtemariam and H.-S. Shin, J. Nanobiotechnol, 2018, 16, 71 CrossRef PubMed.
- D. Guimarães, A. Cavaco-Paulo and E. Nogueira, Int. J. Pharm., 2021, 601, 120571 CrossRef PubMed.
- S. Karki, H. Kim, S.-J. Na, D. Shin, K. Jo and J. Lee, Asian J. Pharm. Sci., 2016, 11, 559–574 Search PubMed.
- D. Hoc and D. Haznar-Garbacz, Eur. J. Pharm. Biopharm., 2021, 167, 73–82 CrossRef CAS PubMed.
- J. Li and D. J. Mooney, Nat. Rev. Mater., 2016, 1, 16071 CrossRef CAS PubMed.
- J. Banks, IEEE Pulse, 2023, 14, 15–17 Search PubMed.
- N. R. Labiris and M. B. Dolovich, Br. J. Clin. Pharmacol., 2003, 56, 600–612 CrossRef CAS PubMed.
- B. H. J. Gowda, M. G. Ahmed, A. Sahebkar, Y. Riadi, R. Shukla and P. Kesharwani, Biomacromolecules, 2022, 23, 1519–1544 CrossRef CAS PubMed.
- M. Prakasam, J. Locs, K. Salma-Ancane, D. Loca, A. Largeteau and L. Berzina-Cimdina, J. Funct. Biomater., 2017, 8, 44 CrossRef PubMed.
- Z. Zhang, X. Li, W. Zhang and D. S. Kohane, Adv. Funct. Mater., 2021, 31, 2008701 CrossRef CAS PubMed.
- D. S. Alberts and D. J. Garcia, Drugs, 1997, 54, 30–35 CrossRef CAS PubMed.
- J. Szebeni, L. Baranyi, S. Savay, J. Milosevits, R. Bunger, P. Laverman, J. M. Metselaar, G. Storm, A. Chanan-Khan, L. Liebes, F. M. Muggia, R. Cohen, Y. Barenholz and C. R. Alving, J. Liposome Res., 2002, 12, 165–172 CrossRef CAS PubMed.
- W. Jiskoot, R. M. F. van Schie, M. G. Carstens and H. Schellekens, Pharm. Res., 2009, 26, 1303–1314 CrossRef CAS PubMed.
- A. S. Almutairi, F1000Res, 2018, 7, 685 Search PubMed.
- P. TÖrmälä, T. Pohjonen and P. Rokkanen, Proc. Inst. Mech. Eng., Part H, 1998, 212, 101–111 CrossRef PubMed.
- Y. K. Sung and S. W. Kim, Biomater. Res., 2020, 24, 12 CrossRef CAS PubMed.
- P. C. Pires, F. Mascarenhas-Melo, K. Pedrosa, D. Lopes, J. Lopes, A. Macário-Soares, D. Peixoto, P. S. Giram, F. Veiga and A. C. Paiva-Santos, Eur. Polym. J., 2023, 187, 111868 CrossRef CAS.
- D. Pappalardo, T. Mathisen and A. Finne-Wistrand, Biomacromolecules, 2019, 20, 1465–1477 CrossRef CAS PubMed.
- FDA Approves XARACOLL® (bupivacaine HCl) Implant, a Non-opioid, Drug-device Treatment Option for Acute Postsurgical Pain Relief for up to 24 Hours Following Open Inguinal Hernia Repair in Adults, https://www.innocoll.com/news-and-media/2020/08/fda-approves-xaracoll-bupivacaine-hcl-implant-a-non-opioid-drug-device-treatment-option-for-acute-postsurgical-pain-relief-for-up-to-24-hours-following-open-inguinal-hernia-repair-in-adults/, (accessed July 19, 2024).
- K. Ceonzo, A. Gaynor, L. Shaffer, K. Kojima, C. A. Vacanti and G. L. Stahl, Tissue Eng., 2006, 12, 301–308 CrossRef CAS PubMed.
- K. S. Stankevich, A. Gudima, V. D. Filimonov, H. Klüter, E. M. Mamontova, S. I. Tverdokhlebov and J. Kzhyshkowska, Mater. Sci. Eng., C, 2015, 51, 117–126 CrossRef CAS PubMed.
- S. J. Yoon, S. H. Kim, H. J. Ha, Y. K. Ko, J. W. So, M. S. Kim, Y. I. Yang, G. Khang, J. M. a Rhee and H. B. Lee, Tissue Eng., Part A, 2008, 14, 539–547 CrossRef CAS PubMed.
- S. L. Lin and M. O. Christen, J. Cosmet., Dermatol., 2020, 19, 1907–1914 CrossRef PubMed.
- J. A. Martins, A. A. Lach, H. L. Morris, A. J. Carr and P. A. Mouthuy, J. Biomater. Appl., 2020, 34, 902–916 CrossRef PubMed.
- G. J. Galano, K. N. Jiang, R. J. Strauch, M. P. Rosenwasser and P. Tang, Hand, 2010, 5, 307–312 CrossRef PubMed.
- M. A. Schrumpf, A. T. Lee and A. J. Weiland, J. Hand Surg., 2011, 36, 1769–1773 CrossRef PubMed.
- S. O. Sarrigiannidis, J. M. Rey, O. Dobre, C. González-García, M. J. Dalby and M. Salmeron-Sanchez, Mater. Today Bio, 2021, 10, 100098 CrossRef CAS PubMed.
- M. Sanguedolce, G. Rotella, F. Curcio, R. Pelaccia, V. Siciliani, R. Cassano, L. Orazi and L. Filice, Int. J. Adv. Des. Manuf. Technol., 2023, 125, 4621–4629 CrossRef.
- R. Ahmad Raus, W. M. F. Wan Nawawi and R. R. Nasaruddin, Asian J. Pharm. Sci., 2021, 16, 280–306 Search PubMed.
- F. Corduas, D. A. Lamprou and E. Mancuso, Bio-Des. Manuf., 2021, 4, 278–310 CrossRef.
- S. Hasanzadeh, M. Farokhi, M. Habibi, M. A. Shokrgozar, R. Ahangari Cohan, F. Rezaei, M. R. Asadi Karam and S. Bouzari, ACS Biomater. Sci. Eng., 2020, 6, 4573–4582 CrossRef CAS PubMed.
- A. B. Li, J. A. Kluge, N. A. Guziewicz, F. G. Omenetto and D. L. Kaplan, J. Controlled Release, 2015, 219, 416–430 CrossRef CAS PubMed.
- C. Z. Zhou, F. Confalonieri, M. Jacquet, R. Perasso, Z. G. Li and J. Janin, Proteins: Struct., Funct., Bioinf., 2001, 44, 119–122 CrossRef CAS PubMed.
- S. Kaewpirom and S. Boonsang, RSC Adv., 2020, 10, 15913–15923 RSC.
- X. Hu, K. Shmelev, L. Sun, E.-S. Gil, S.-H. Park, P. Cebe and D. L. Kaplan, Biomacromolecules, 2011, 12, 1686–1696 CrossRef CAS PubMed.
- A. Lammel, M. Schwab, M. Hofer, G. Winter and T. Scheibel, Biomaterials, 2011, 32, 2233–2240 CrossRef CAS PubMed.
- D. J. Hines and D. L. Kaplan, Macromol. Chem. Phys., 2013, 214, 280–294 CrossRef CAS.
- D. N. Rockwood, R. C. Preda, T. Yücel, X. Wang, M. L. Lovett and D. L. Kaplan, Nat. Protoc., 2011, 6, 1612–1631 CrossRef CAS PubMed.
- C. Vepari and D. L. Kaplan, Prog. Polym. Sci., 2007, 32, 991–1007 CrossRef CAS PubMed.
- K. A. S. Usman, Y. Yao, C. J. O. Bacal, J. Zhang, K. L. Jarvis, P. A. Lynch, P. Mota-Santiago, S. Qin, M. Naebe, L. C. Henderson, D. Y. Hegh, B. J. Allardyce and J. M. Razal, Adv. Mater. Interfaces, 2023, 10, 2201634 CrossRef CAS.
- P. Dorishetty, R. Balu, S. S. Athukoralalage, T. L. Greaves, J. Mata, L. de Campo, N. Saha, A. C. W. Zannettino, N. K. Dutta and N. R. Choudhury, ACS Sustainable Chem. Eng., 2020, 8, 2375–2389 CrossRef CAS.
- J. H. Lee, B. K. Park and I. C. Um, Int. J. Mol. Sci., 2022, 23, 11344 CrossRef CAS PubMed.
- X. Liu, Q. Huang, P. Pan, M. Fang, Y. Zhang, S. Yang, M. Li and Y. Liu, Polymers, 2023, 15, 3383 CrossRef CAS PubMed.
- S. K. Vyas and S. R. Shukla, J. Text. Inst., 2020, 111, 1364–1370 CrossRef CAS.
- K. Nultsch, L. K. Bast, M. Näf, S. E. Yakhlifi, N. Bruns and O. Germershaus, Macromol. Mater. Eng., 2018, 303, 1800408 CrossRef.
- M. Bayattork, R. Rajkhowa, B. J. Allardyce, X. Wang and J. Li, J. Colloid Interface Sci., 2023, 631, 46–55 CrossRef CAS PubMed.
- B. J. Allardyce, R. Rajkhowa, R. J. Dilley, S. L. Redmond, M. D. Atlas and X. Wang, Mater. Sci. Eng., C, 2017, 80, 165–173 CrossRef CAS PubMed.
- G. Rizzo, V. Petrelli, T. Sibillano, L. De Caro, M. M. Giangregorio, M. Lo Presti, F. G. Omenetto, C. Giannini, P. Mastrorilli and G. M. Farinola, ACS Omega, 2023, 8, 24165–24175 CrossRef CAS PubMed.
- A. K. Brooks and V. K. Yadavalli, Adv. Eng. Mater., 2025, 27, 2402401 CrossRef CAS.
- X.-Y. Liu, C.-C. Zhang, W.-L. Xu and C.-X. Ouyang, Mater. Lett., 2009, 63, 263–265 CrossRef CAS.
- K.-L. Mao, Z.-L. Fan, J.-D. Yuan, P.-P. Chen, J.-J. Yang, J. Xu, D.-L. ZhuGe, B.-H. Jin, Q.-Y. Zhu, B.-X. Shen, Y. Sohawon, Y.-Z. Zhao and H.-L. Xu, Colloids Surf., B, 2017, 160, 704–714 CrossRef CAS PubMed.
- E. Wenk, A. J. Meinel, S. Wildy, H. P. Merkle and L. Meinel, Biomaterials, 2009, 30, 2571–2581 CrossRef CAS PubMed.
- L. Xiao, H. Yang, Q. Lu and D. L. Kaplan, Macromol. Biosci., 2024, 24, 2300523 CrossRef CAS PubMed.
- Q. Deng, P. Lin, H. Gu, X. Zhuang and F. Wang, Biomacromolecules, 2024, 25, 1527–1540 CrossRef CAS PubMed.
- C. J. O. Bacal, J. W. Maina, H. H. Nandurkar, M. Khaleel, R. Guijt, Y. Chang, K. M. Dwyer and L. F. Dumée, Mater. Adv., 2021, 2, 7210–7236 RSC.
- E. Wenk, A. J. Wandrey, H. P. Merkle and L. Meinel, J. Controlled Release, 2008, 132, 26–34 CrossRef CAS PubMed.
- N. Singh, S. P. Gautam, A. Harjaskaran, L. Singh, A. Verma and S. Rani, Ars Pharm., 2016, 57, 137–142 CrossRef CAS.
- Z. Hu, K. An, J. Wang, X. Xu, Z. Chen, J. Hu and L. Yang, Fibers Polym., 2020, 21, 252–261 CrossRef CAS.
- N. Soomherun, R. Kriangsaksri, W. Tanticharakunsiri, N. Foongsawat, W. Phoolcharoen, S. Tawinwung, S. Keeratihattayakorn and J. Ratanavaraporn, J. Drug Delivery Sci. Technol., 2024, 95, 105574 CrossRef CAS.
- J. Ding, Z. Cheng, Y. Ma, T. Zhang, L. Du, X. Jiang, M. Zhu, W. Li and B. Xu, Adv. Healthcare Mater., 2025, 14, 2402932 CrossRef CAS PubMed.
- X. Li, Y. Chen, H. Shao, Z. Yan, Y. Wang and S. Lu, Int. J. Biol. Macromol., 2025, 301, 140500 CrossRef CAS PubMed.
- B. Bhar, P. Ranta, P. K. Samudrala and B. B. Mandal, ACS Biomater. Sci. Eng., 2024, 10, 1090–1105 CrossRef CAS PubMed.
- G. Sabarees, V. Velmurugan, S. Gouthaman, V. R. Solomon and S. Kandhasamy, Pharmaceutics, 2024, 16, 462 CrossRef CAS PubMed.
- X. Liu, X. Shi, D. Zhang, S. Zhao, J. Hu, Q. Ouyang, J. Yin, X. Yao, Y. Zhang and L. Yan, Composites, Part B, 2025, 296, 112260 CrossRef CAS.
- H. Xiong, P. Zhang, D. Wang, Z. Zhou, J. Sun and M. Diao, Colloids Surf., B, 2024, 237, 113855 CrossRef CAS PubMed.
- H. K. Polat, E. Aytekin, N. F. Karakuyu, Y. A. Çaylı, S. Çalamak, N. Demirci, S. Ünal, N. Kurt, R. Çırak, E. Erkan, B. Mokhtare, A. S. Mendil and S. Bozdağ Pehlivan, J. Drug Delivery Sci. Technol., 2024, 96, 105625 CrossRef CAS.
- M. Arumugam, B. Murugesan, D. K. Chinnalagu and S. Mahalingam, J. Drug Delivery Sci. Technol., 2024, 95, 105620 CrossRef CAS.
- B. Gong, Y. Liu, H. Li, X. Ju, D. Li, Y. Zou, X. Guo, K. Dong, J. Xiao, W. Wu, R. Chai, R. Zhang and M. Yu, Adv. Sci., 2025, 12, e2404835 CrossRef PubMed.
- J. Chen, L. Zhang, Z. Han, X. Meng, X. Sun, Y. Zhong, M. Zhi, D. Huang and G. Li, Int. J. Biol. Macromol., 2024, 281, 135927 CrossRef CAS PubMed.
- G. Yang, L. Liu, L. Xiao, S. Ke, H. Yang and Q. Lu, J. Mater. Chem. B, 2024, 12, 11670–11684 RSC.
- X. Chen, J. Liu, Y. Lu, H. Liu, L. Cheng, Z. Li and F. Dai, Int. J. Mol. Sci., 2024, 25, 9263 CrossRef CAS PubMed.
- Z. Khosrowpour, Z. Bashiri, D. Jafari, S. Alizadeh, Z. Keshtkaran, B. Rezaei, F. Bargrizaneh, M. Abdollahpour-Alitappeh and M. Gholipourmalekabadi, Polym. Adv. Technol., 2024, 35, e6637 CrossRef CAS.
- H. Lv, S. Wang, M. He, X. Zhu, P. He, R. Sun, Z. Jia, Z. Wang, W. Zhao, Z. Zhong and Y. Han, Mater. Des., 2025, 251, 113574 CrossRef CAS.
- Y. Ming, H. Xinyi, Z. Zhenxing, M. Xuehuan, Z. Ye, T. Hao, Y. Guoyin, H. Yun and L. Zheng, Int. J. Nanomed., 2024, 19, 10263–10282 CrossRef PubMed.
- X. Zhang, Y. Xia, J. Xu, J. Kang, X. Li, Y. Li, W. Yan, F. Tian, B. Zhao, B. Li, C. Wang and L. Wang, Carbohydr. Polym., 2024, 332, 121945 CrossRef CAS PubMed.
- A. Pandey, P. Yadav, S. Mishra, S. Srivastava and P. Parashar, J. Drug Delivery Sci. Technol., 2024, 101, 106145 CrossRef CAS.
- N. Bokhari, A. Ali, A. Yasmeen, R. Khan, S. Haider and F. Sharif, Appl. Mater. Today, 2024, 38, 102261 CrossRef.
- Z. Li, L. Cheng, X. Xu, R. Jia, S. Zhu, Q. Zhang, G. Cheng, B. Wu, Z. Liu, X. Tong, B. Xiao and F. Dai, Mater. Today Bio, 2024, 29, 101298 CrossRef CAS PubMed.
- Y. Li, J.-Y. Gong, P. Wang, H. Fu, F. Yousef, R. Xie, W. Wang, Z. Liu, D.-W. Pan, X.-J. Ju and L.-Y. Chu, J. Colloid Interface Sci., 2024, 661, 123–138 CrossRef CAS PubMed.
- Y. Hua, Z. He, Y. Ni, L. Sun, R. Wang, Y. Li, X. Li and G. Jiang, Biomed. Phys. Eng. Express, 2024, 10, 045001 CrossRef PubMed.
- J. Wang, N. Jin, Z. Xie, Q. Cheng, B. Jiang, Y. Shuai, Z. Xu, Q. Wan, Y. Chen, C. Mao and M. Yang, Nanoscale, 2025, 17, 4624–4635 RSC.
- M. Nazemoroaia, F. Bagheri, S. Z. Mirahmadi-Zare, F. Eslami-kaliji and A. Derakhshan, Int. J. Pharm., 2025, 668, 124976 CrossRef CAS PubMed.
- Z. Mai, P. Wang, C. Gong, J. Wen, W. Zheng, Y. Xia, F. Ko, H. Zhao and W. Zhou, J. Appl. Polym. Sci., 2024, 141, e55518 CrossRef CAS.
- S. Sheybanikashani, N. Zandi, D. Hosseini, R. Lotfi and A. Simchi, J. Mater. Chem. B, 2024, 12, 784–799 RSC.
- M. Babaluei, Y. Mojarab, F. Mottaghitalab, M. R. Saeb and M. Farokhi, Int. J. Biol. Macromol., 2024, 261, 129932 CrossRef CAS PubMed.
- M. R. Islam, M. S. Manir, M. Razzak, M. A. Mamun, M. F. Mortuza, M. J. Islam, S. Yang, H. Pan, A. K. M. M. Alam and Q. T. H. Shubhra, Int. J. Biol. Macromol., 2024, 280, 135567 CrossRef CAS PubMed.
- M. Aleemardani, Z. Akbarnejad, M. Jalessi, H. Chahsetareh, Z. Hajmohammadi, S. Hassanzadeh, R. Najafi, R. Alizadeh, M. Farhadi and Z. Bagher, J. Drug Delivery Sci. Technol., 2024, 95, 105642 CrossRef CAS.
- B. M. Bakadia, R. Zheng, A. A. Qaed Ahmed, Z. Shi, B. L. Babidi, T. Sun, Y. Li and G. Yang, Adv. Healthcare Mater., 2024, 13, 2304572 CrossRef CAS PubMed.
- P. V. Jesna, S. L. Devika and P. R. Umashankar, Int. J. Biol. Macromol., 2025, 306, 141804 CrossRef CAS PubMed.
- J. Li, Y. Li, C. Guo and X. Wu, Chem. Eng. J., 2024, 481, 148458 CrossRef CAS.
- Z. Zhou, P. Song, Y. Wu, M. Wang, C. Shen, Z. Ma, X. Ren, X. Wang, X. Chen, Y. Hu, Z. Li, Q. Zhang, M. Li, Z. Geng and J. Su, Mater. Horiz., 2024, 11, 1465–1483 RSC.
- A. Bandyopadhyay, B. Ghibhela, S. Shome, D. Pal, S. K. Nandi and B. B. Mandal, Appl. Mater. Today, 2024, 37, 102111 CrossRef.
- A. Huang, Y. Huang, W. Yang, L. Wang, R. You, J. Wang, S. Yan and Q. Zhang, Int. J. Biol. Macromol., 2024, 263, 130287 CrossRef CAS PubMed.
- R. Huang, J. Hua, M. Ru, M. Yu, L. Wang, Y. Huang, S. Yan, Q. Zhang and W. Xu, ACS Nano, 2024, 18, 15312–15325 CrossRef CAS PubMed.
- J. Hua, R. Huang, M. Yu, R. You, L. Wang, S. Yan, Y. Huang and Q. Zhang, Int. J. Biol. Macromol., 2025, 298, 140357 CrossRef CAS PubMed.
- F. Karimi, N. Farbehi, F. Ziaee, K. Lau, M. Monfared, M. Kordanovski, H. Joukhdar, T. G. Molly, R. Nordon, K. A. Kilian, M. H. Stenzel, K. S. Lim and J. Rnjak-Kovacina, Adv. Funct. Mater., 2024, 34, 2313354 CrossRef CAS.
- R. Zhang, M. Zhang, L. Chen, L. Jiang, C. Zou, N. Li, H. Zhou and S. Feng, Front. Bioeng. Biotechnol., 2024, 12, 1501488 CrossRef PubMed.
- M. Yu, R. Huang, J. Hua, M. Ru, R. You, Y. Huang, S. Yan and Q. Zhang, Int. J. Biol. Macromol., 2024, 267, 131519 CrossRef CAS PubMed.
- H. Shaygani, A. Shamloo, K. Akbarnataj and S. Maleki, Int. J. Biol. Macromol., 2024, 270, 132126 CrossRef CAS PubMed.
- P. Cheng, L. Cheng, H. Han, J. Li, C. Ma, H. Huang, J. Zhou, J. Feng, Y. Huang, Y. Lv, H. Huang, Y. Wang, L. Hou, Y. Chen and G. Li, Adv. Healthcare Mater., 2022, 11, 2200971 CrossRef CAS PubMed.
- E. I. Buzas, Nat. Cell Biol., 2022, 24, 1322–1325 CrossRef CAS PubMed.
- M. D. Hade, C. N. Suire, J. Mossell and Z. Suo, Med. Res. Rev., 2022, 42, 2102–2125 CrossRef CAS PubMed.
- L. M. Doyle and M. Z. Wang, Cells, 2019, 8, 727 CrossRef CAS PubMed.
- W. Meng, C. He, Y. Hao, L. Wang, L. Li and G. Zhu, Drug Delivery, 2020, 27, 585–598 CrossRef CAS PubMed.
- P. Sungkhaphan, J. Ratanavaraporn and S. Damrongsukkul, Eng. J., 2017, 21, 139–153 CrossRef CAS.
- R. Rujiravanit, S. Kruaykitanon, A. M. Jamieson and S. Tokura, Macromol. Biosci., 2003, 3, 604–611 CrossRef CAS.
- Z. Wang, Y. Zhang, J. Zhang, L. Huang, J. Liu, Y. Li, G. Zhang, S. C. Kundu and L. Wang, Sci. Rep., 2014, 4, 7064 CrossRef PubMed.
- O. Bayraktar, Ö. Malay, Y. Özgarip and A. Batıgün, Eur. J. Pharm. Biopharm., 2005, 60, 373–381 CrossRef CAS PubMed.
- A. R. Murphy, P. St John and D. L. Kaplan, Biomaterials, 2008, 29, 2829–2838 CrossRef CAS PubMed.
- Z. Wang, H. Liu, W. Luo, T. Cai, Z. Li, Y. Liu, W. Gao, Q. Wan, X. Wang, J. Wang, Y. Wang and X. Yang, J. Tissue Eng., 2020, 11, 2041731420974861 CrossRef PubMed.
- J. Chen, H. Venkatesan and J. Hu, Adv. Eng. Mater., 2018, 20, 1700961 CrossRef.
- C. Ninh, A. Iftikhar, M. Cramer and C. J. Bettinger, J. Mater. Chem. B, 2015, 3, 4607–4615 RSC.
- V. Hasirci, P. Yilgor, T. Endogan, G. Eke and N. Hasirci, 1.121-polymer fundamentals: polymer synthesis, Elsevier, Oxford, 2011 Search PubMed.
- I. A. Udoetok, R. M. Dimmick, L. D. Wilson and J. V. Headley, Carbohydr. Polym., 2016, 136, 329–340 CrossRef CAS PubMed.
- J. W. Bae, J. H. Choi, Y. Lee and K. D. Park, J. Tissue Eng. Regener. Med., 2015, 9, 1225–1232 CrossRef CAS PubMed.
- E. Gantumur, S. Sakai, M. Nakahata and M. Taya, Soft Matter, 2019, 15, 2163–2169 RSC.
- X. Mu, J. K. Sahoo, P. Cebe and D. L. Kaplan, Polymers, 2020, 12, 2936 CrossRef CAS PubMed.
- J. Liang, X. Zhang, Z. Chen, S. Li and C. Yan, Polymers, 2019, 11, 2102 CrossRef CAS PubMed.
- H. Feinberg and T. W. Hanks, Polym. Int., 2022, 71, 578–582 CrossRef CAS.
- X. Bao, J. Zhao, J. Sun, M. Hu and X. Yang, ACS Nano, 2018, 12, 8882–8892 CrossRef CAS PubMed.
- Z. Tu, M. Shi, X. Zhang, P. Liu, Y. Wu and X. Hu, J. CO2 Util., 2021, 46, 101473 CrossRef CAS.
- Y. Zhao, M. He, L. Zhao, S. Wang, Y. Li, L. Gan, M. Li, L. Xu, P. R. Chang, D. P. Anderson and Y. Chen, ACS Appl. Mater. Interfaces, 2016, 8, 2781–2795 CrossRef CAS PubMed.
- S. Sel, N. Nass, S. Pötzsch, S. Trau, A. Simm, T. Kalinski, G. I. Duncker, F. E. Kruse, G. U. Auffarth and H. J. Brömme, Redox Rep., 2014, 19, 72–79 CrossRef CAS PubMed.
- N. Johari, L. Moroni and A. Samadikuchaksaraei, Eur. Polym. J., 2020, 134, 109842 CrossRef CAS.
- H. A. Tran, T. T. Hoang, A. Maraldo, T. N. Do, D. L. Kaplan, K. S. Lim and J. Rnjak-Kovacina, Mater. Today, 2023, 65, 244–259 CrossRef CAS.
- M. J. E. Fischer, in Surface Plasmon Resonance: Methods and Protocols, ed. N. J. Mol and M. J. E. Fischer, Humana Press, Totowa, NJ, 2010, pp. 55–73 DOI:10.1007/978-1-60761-670-2_3.
- A. Duangpakdee, C. Laomeephol, D. Jindatip, P. Thongnuek, J. Ratanavaraporn and S. Damrongsakkul, Molecules, 2021, 26, 3191 CrossRef CAS PubMed.
- J. S. Yoo, Y. J. Kim, S. H. Kim and S. H. Choi, Korean J. Thorac. Cardiovasc. Surg., 2011, 44, 197–207 CrossRef PubMed.
- A. Oryan, A. Kamali, A. Moshiri, H. Baharvand and H. Daemi, Int. J. Biol. Macromol., 2018, 107, 678–688 CrossRef CAS PubMed.
- P. Purohit, A. Bhatt, R. K. Mittal, M. H. Abdellattif and T. A. Farghaly, Front. Bioeng. Biotechnol., 2022, 10, 1044927 CrossRef PubMed.
- C. Kibungu, P. P. Kondiah, P. Kumar and Y. E. Choonara, Front. Mater., 2021, 8, 681960 CrossRef.
- K. Gao, B. Su, J. Dai, P. Li, R. Wang and X. Yang, Molecules, 2022, 27, 1485 CrossRef CAS PubMed.
- S. Botezan, G.-M. Baci, L. Bagameri, C. Pasca and D. S. Dezmirean, Molecules, 2023, 28, 1510 CrossRef CAS PubMed.
- B. Singh and J. Singh, Carbohydr. Polym. Technol. Appl., 2021, 2, 100058 CAS.
- M. Iovu, G. Dumais and P. du Souich, Osteoarthritis Cartilage, 2008, 16, S14–S18 CrossRef PubMed.
- B. Jayachandran, T. N. Parvin, M. M. Alam, K. Chanda and B. MM, Molecules, 2022, 27, 8124 CrossRef CAS PubMed.
- H. Maleki, M.-A. Shahbazi, S. Montes, S. H. Hosseini, M. R. Eskandari, S. Zaunschirm, T. Verwanger, S. Mathur, B. Milow, B. Krammer and N. Hüsing, ACS Appl. Mater. Interfaces, 2019, 11, 17256–17269 CrossRef CAS PubMed.
- Y. Wang, G. Boero, X. Zhang and J. Brugger, ACS Appl. Mater. Interfaces, 2022, 14, 40418–40426 CrossRef CAS PubMed.
- X. Xie, J. Yu, Z. Zhao, Z. Zheng, M. Xie, X. Wang, Z. Han and G. Li, Adsorpt. Sci. Technol., 2019, 37, 412–424 CrossRef CAS.
- X. Wang, T. Yucel, Q. Lu, X. Hu and D. L. Kaplan, Biomaterials, 2010, 31, 1025–1035 CrossRef CAS PubMed.
- Y. Gao, M. Hou, R. Yang, L. Zhang, Z. Xu, Y. Kang and P. Xue, Biomacromolecules, 2019, 20, 1334–1345 CrossRef CAS PubMed.
- J. das Neves and M. F. Bahia, Int. J. Pharm., 2006, 318, 1–14 CrossRef CAS PubMed.
- C. Jaiswal, T. Gupta, P. K. Jadi, J. C. Moses and B. B. Mandal, Biomater. Adv., 2023, 145, 213224 CrossRef CAS PubMed.
- H. Wu, S. Liu, L. Xiao, X. Dong, Q. Lu and D. L. Kaplan, ACS Appl. Mater. Interfaces, 2016, 8, 17118–17126 Search PubMed.
- Z. Ding, M. Zhou, Z. Zhou, W. Zhang, X. Jiang, X. Lu, B. Zuo, Q. Lu and D. L. Kaplan, ACS Biomater. Sci. Eng., 2019, 5, 4077–4088 CrossRef CAS PubMed.
- B. Maity, S. Samanta, S. Sarkar, S. Alam and T. Govindaraju, ACS Appl. Bio Mater., 2020, 3, 3544–3552 CrossRef CAS PubMed.
- Y. Sun, P. Zhang, F. Zhang, M. Pu, W. Zhong, Y. Zhang, Y. Shen and B. Zuo, J. Drug Delivery Sci. Technol., 2022, 75, 103596 CrossRef CAS.
- M. L. Lovett, X. Wang, T. Yucel, L. York, M. Keirstead, L. Haggerty and D. L. Kaplan, Eur. J. Pharm. Biopharm., 2015, 95, 271–278 CrossRef CAS PubMed.
- C. Han, J. Zhou, B. Liu, C. Liang, X. Pan, Y. Zhang, Y. Zhang, Y. Wang, L. Shao, B. Zhu, J. Wang, Q. Yin, X.-Y. Yu and Y. Li, Mater. Sci. Eng., C, 2019, 99, 322–332 CrossRef CAS PubMed.
- R. Fernández-Serra, A. Lekouaghet, L. Peracho, M. Yonesi, A. Alcázar, M. Chioua, J. Marco-Contelles, J. Pérez-Rigueiro, F. J. Rojo, F. Panetsos, G. V. Guinea and D. González-Nieto, Biomacromolecules, 2024, 25, 5233–5250 CrossRef PubMed.
- W. Hwang, B. H. Kim, R. Dandu, J. Cappello, H. Ghandehari and J. Seog, Langmuir, 2009, 25, 12682–12686 CrossRef CAS PubMed.
- M. Colleoni, H. J. Litman, M. Castiglione-Gertsch, W. Sauerbrei, R. D. Gelber, M. Bonetti, A. S. Coates, M. Schumacher, G. Bastert, C. M. Rudenstam, C. Schmoor, J. Lindtner, J. Collins, B. Thürlimann, S. B. Holmberg, D. Crivellari, C. Beyerle, R. L. Neumann and A. Goldhirsch, Br. J. Cancer, 2002, 86, 1705–1714 CrossRef CAS PubMed.
- F. C. DeCroos, B. Todorich, R. Alshareef, M. Khuthaila, S. Fekrat, A. C. Ho, C. D. Regillo and M. J. Spirn, J. Ophthalmic Vision Res., 2014, 9, 461–468 CrossRef PubMed.
- M. Floren, C. Migliaresi and A. Motta, J. Funct. Biomater., 2016, 7, 26 CrossRef PubMed.
- S. He, D. Shi, Z. Han, Z. Dong, Y. Xie, F. Zhang, W. Zeng and Q. Yi, Biomed. Eng. Online, 2019, 18, 97 CrossRef PubMed.
- X. Wang, J. A. Kluge, G. G. Leisk and D. L. Kaplan, Biomaterials, 2008, 29, 1054–1064 CrossRef CAS PubMed.
- Z. Li, A. Zheng, Z. Mao, F. Li, T. Su, L. Cao, W. Wang, Y. Liu and C. Wang, Int. J. Bioprint., 2023, 9, 773 CrossRef PubMed.
- M. Viola, C. Chartier, M. Mihajlovic, S. Buwalda, C. Pradille, T. Budtova and T. Vermonden, Eur. Polym. J., 2024, 205, 112722 CrossRef CAS.
- T. Abdullah, E. Su and A. Memić, Biomimetics, 2022, 8, 5 CrossRef PubMed.
- K. Križman, S. Novak, J. Kristl, G. Majdič and N. Drnovšek, J. Drug Delivery Sci. Technol., 2021, 65, 102701 CrossRef.
- M. A. Marin, R. R. Mallepally and M. A. McHugh, J. Supercrit. Fluids, 2014, 91, 84–89 CrossRef CAS.
- R. R. Mallepally, M. A. Marin, V. Surampudi, B. Subia, R. R. Rao, S. C. Kundu and M. A. McHugh, Biomed. Mater., 2015, 10, 035002 CrossRef PubMed.
- N. Guziewicz, A. Best, B. Perez-Ramirez and D. L. Kaplan, Biomaterials, 2011, 32, 2642–2650 CrossRef CAS PubMed.
- L. Chambre, R. N. Parker, B. J. Allardyce, F. Valente, R. Rajkhowa, R. J. Dilley, X. Wang and D. L. Kaplan, ACS Appl. Bio Mater., 2020, 3, 2466–2472 CrossRef CAS PubMed.
- N. Al-Jawuschi, S. Chen, N. Abie, T. Fischer, S. Fare and H. H. Maleki, Langmuir, 2023, 39, 4326–4337 CrossRef CAS PubMed.
- J. S. Bergum, J. K. Prescott, R. W. Tejwani, T. P. Garcia, J. Clark and W. Brown, Pharm. Eng., 2014, 34, 1–10 Search PubMed.
- P. Y. Muller and M. N. Milton, Nat. Rev. Drug Discovery, 2012, 11, 751–761 CrossRef CAS PubMed.
- L. L. Mazaleuskaya, K. N. Theken, L. Gong, C. F. Thorn, G. A. FitzGerald, R. B. Altman and T. E. Klein, Pharmacogenet. Genomics, 2015, 25, 96–106 CrossRef CAS PubMed.
- K. D. Rainsford, Inflammopharmacology, 2009, 17, 275–342 CrossRef CAS PubMed.
- M. E. Zoubek, M. I. Lucena, R. J. Andrade and C. Stephens, Aliment. Pharmacol. Ther., 2020, 51, 603–611 CrossRef CAS PubMed.
- N. Karamat-Ullah, Y. Demidov, M. Schramm, D. Grumme, J. Auer, C. Bohr, B. Brachvogel and H. Maleki, ACS Biomater. Sci. Eng., 2021, 7, 4545–4556 CrossRef CAS PubMed.
- H. Maleki, S. Montes, N. Hayati-Roodbari, F. Putz and N. Huesing, ACS Appl. Mater. Interfaces, 2018, 10, 22718–22730 CrossRef CAS PubMed.
- P. Ng, A. R. Pinho, M. C. Gomes, Y. Demidov, E. Krakor, D. Grume, M. Herb, K. Lê, J. Mano, S. Mathur and H. Maleki, Macromol. Biosci., 2022, 22, 2100442 CrossRef CAS PubMed.
- H. K. Pektas, Y. Demidov, A. Ahvan, N. Abie, V. S. Georgieva, S. Chen, S. Farè, B. Brachvogel, S. Mathur and H. Maleki, ACS Mater. Au, 2023, 3, 711–726 CrossRef CAS PubMed.
- H. Maleki and N. Huesing, J. Sol-Gel Sci. Technol., 2021, 98, 430–438 CrossRef CAS PubMed.
- S. Du, W. Zhou, X. Jin, Y. Zhang, X. Chen, J. Zhang, J. Li and X. Wang, Fibers Polym., 2021, 22, 373–381 CrossRef CAS.
- X. Tang, X. Qiao and K. Sun, Colloids Surf., A, 2015, 486, 86–95 CrossRef CAS.
- Y. Qi, H. Wang, K. Wei, Y. Yang, R. Y. Zheng, I. S. Kim and K. Q. Zhang, Int. J. Mol. Sci., 2017, 18, 237 CrossRef PubMed.
- Q. Lu, X. Wang, X. Hu, P. Cebe, F. Omenetto and D. L. Kaplan, Macromol. Biosci., 2010, 10, 359–368 CrossRef CAS PubMed.
- J. M. Coburn, E. Na and D. L. Kaplan, J. Controlled Release, 2015, 220, 229–238 CrossRef CAS PubMed.
- M. C. Lee, D. K. Kim, O. J. Lee, J. H. Kim, H. W. Ju, J. M. Lee, B. M. Moon, H. J. Park, D. W. Kim, S. H. Kim and C. H. Park, J. Biomed. Mater. Res., Part B, 2016, 104, 508–514 CrossRef CAS PubMed.
- S. Madduri, M. Papaloïzos and B. Gander, Biomaterials, 2010, 31, 2323–2334 CrossRef CAS PubMed.
- J. Xue, T. Wu, Y. Dai and Y. Xia, Chem. Rev., 2019, 119, 5298–5415 CrossRef CAS PubMed.
- K. Ghosal, A. Chandra, P. G, S. S, S. Roy, C. Agatemor, S. Thomas and I. Provaznik, Sci. Rep., 2018, 8, 5058 CrossRef PubMed.
- K. A. Karve, E. S. Gil, S. P. McCarthy and D. L. Kaplan, J. Membr. Sci., 2011, 383, 44–49 CrossRef CAS PubMed.
- I. C. Um, H. Y. Kweon, K. G. Lee and Y. H. Park, Int. J. Biol. Macromol., 2003, 33, 203–213 CrossRef CAS PubMed.
- P. Wongpanit, H. Ueda, Y. Tabata and R. Rujiravanit, J. Biomater. Sci., Polym. Ed., 2010, 21, 1403–1419 CrossRef CAS PubMed.
- H. Li, S. Si, K. Yang, Z. Mao, Y. Sun, X. Cao, H. Yu, J. Zhang, C. Ding, H. Liang and L. Wu, Prog. Org. Coat., 2023, 184, 107881 CrossRef CAS.
- X. Hu, D. Kaplan and P. Cebe, Macromolecules, 2008, 41, 3939–3948 CrossRef CAS.
- M. Puerta, M. C. Arango, N. Jaramillo-Quiceno, C. Álvarez-López and A. Restrepo-Osorio, SN Appl. Sci., 2019, 1, 1443 CrossRef CAS.
- M. Choi, D. Choi and J. Hong, Biomacromolecules, 2018, 19, 3096–3103 CrossRef CAS PubMed.
- C. Geão, A. R. Costa-Pinto, C. Cunha-Reis, V. P. Ribeiro, S. Vieira, J. M. Oliveira, R. L. Reis and A. L. Oliveira, J. Mater. Sci.: Mater. Med., 2019, 30, 27 CrossRef PubMed.
- N. Zulkefli, C. N. M. Che Zahari, N. H. Sayuti, A. A. Kamarudin, N. Saad, H. S. Hamezah, H. Bunawan, S. N. Baharum, A. Mediani, Q. U. Ahmed, A. F. H. Ismail and M. N. Sarian, Int. J. Mol. Sci., 2023, 24, 4607 CrossRef CAS PubMed.
- R. Abdel-Gawad, R. Osman, G. A. S. Awad and N. Mortada, Carbohydr. Polym., 2024, 324, 121526 CrossRef CAS PubMed.
- I. Khan, K. Saeed and I. Khan, Arabian J. Chem., 2019, 12, 908–931 CrossRef CAS.
- H. Rahmani, A. Fattahi, K. Sadrjavadi, S. Khaledian and Y. Shokoohinia, Adv. Pharm. Bull., 2019, 9, 601–608 CrossRef CAS PubMed.
- A. S. Gobin, R. Rhea, R. A. Newman and A. B. Mathur, Int. J. Nanomed., 2006, 1, 81–87 CrossRef CAS PubMed.
- B. B. Mandal and S. C. Kundu, Nanotechnology, 2009, 20, 355101 CrossRef PubMed.
- Y. Cao, F. Liu, Y. Chen, T. Yu, D. Lou, Y. Guo, P. Li, Z. Wang and H. Ran, Sci. Rep., 2017, 7, 11913 CrossRef PubMed.
- S. Y. Kim, D. Naskar, S. C. Kundu, D. P. Bishop, P. A. Doble, A. V. Boddy, H.-K. Chan, I. B. Wall and W. Chrzanowski, Sci. Rep., 2015, 5, 11878 CrossRef CAS PubMed.
- L. Xiao, G. Lu, Q. Lu and D. L. Kaplan, ACS Biomater. Sci. Eng., 2016, 2, 2050–2057 CrossRef CAS PubMed.
- F. Rezaei, S. Damoogh, R. L. Reis, S. C. Kundu, F. Mottaghitalab and M. Farokhi, Biofabrication, 2021, 13, 015005 CrossRef CAS PubMed.
- P. C. Bessa, E. R. Balmayor, H. S. Azevedo, S. Nürnberger, M. Casal, M. van Griensven, R. L. Reis and H. Redl, J. Tissue Eng. Regener. Med., 2010, 4, 349–355 CrossRef CAS PubMed.
- T. Imsombut, M. Srisa-ard, P. Srihanam and Y. Baimark, e-Polymers, 2011, 11, 088 Search PubMed.
- J. G. Hardy, L. M. Römer and T. R. Scheibel, Polymer, 2008, 49, 4309–4327 CrossRef CAS.
- B. Kang, H. Tang, Z. Zhao and S. Song, ACS Omega, 2020, 5, 6229–6239 CrossRef CAS PubMed.
- S. A. L. Matthew, J. D. Totten, S. Phuagkhaopong, G. Egan, K. Witte, Y. Perrie and F. P. Seib, ACS Biomater. Sci. Eng., 2020, 6, 6748–6759 CrossRef CAS PubMed.
- R. M. Kramer, V. R. Shende, N. Motl, C. N. Pace and J. M. Scholtz, Biophys. J., 2012, 102, 1907–1915 CrossRef CAS PubMed.
- P. Wilde, A. Mackie, F. Husband, P. Gunning and V. Morris, Adv. Colloid Interface Sci., 2004, 108–109, 63–71 CrossRef CAS PubMed.
- J. Wen, J. Yao, X. Chen and Z. Shao, ACS Omega, 2018, 3, 3396–3405 CrossRef CAS PubMed.
- V. Pandey, T. Haider, A. R. Chandak, A. Chakraborty, S. Banerjee and V. Soni, Int. J. Biol. Macromol., 2020, 164, 2018–2027 CrossRef CAS PubMed.
- P. Naserzadeh, S. A. Mortazavi, K. Ashtari, E. Seydi and J. Pourahmad, Iran J. Pharm. Res., 2018, 17, 134–145 Search PubMed.
- T. Waghule, G. Singhvi, S. K. Dubey, M. M. Pandey, G. Gupta, M. Singh and K. Dua, Biomed. Pharmacother., 2019, 109, 1249–1258 Search PubMed.
- Z. Qi, Z. Yan, G. Tan, T. Jia, Y. Geng, H. Shao, S. C. Kundu and S. Lu, Pharmaceutics, 2023, 15, 355 Search PubMed.
- B. Yavuz, L. Chambre, K. Harrington, J. Kluge, L. Valenti and D. L. Kaplan, ACS Appl. Bio Mater., 2020, 3, 5375–5382 CrossRef CAS PubMed.
- Y. Mizuno, K. Takasawa, T. Hanada, K. Nakamura, K. Yamada, H. Tsubaki, M. Hara, Y. Tashiro, M. Matsuo, T. Ito and T. Hikima, Biomed. Microdevices, 2021, 23, 38 CrossRef CAS PubMed.
- L. Liang, Y. Chen, B. L. Zhang, X. P. Zhang, J. L. Liu, C. B. Shen, Y. Cui and X. D. Guo, J. Drug Delivery Sci. Technol., 2020, 55, 101464 CrossRef CAS.
- Y. Kapoor, M. Milewski, L. Dick, J. Zhang, J. R. Bothe, M. Gehrt, K. Manser, B. Nissley, I. Petrescu, P. Johnson, S. Burton, J. Moseman, V. Hua, T. Grunewald, M. Tomai and R. Smith, Biomed. Microdevices, 2019, 22, 7 Search PubMed.
- A. V. Boopathy, A. Mandal, D. W. Kulp, S. Menis, N. R. Bennett, H. C. Watkins, W. Wang, J. T. Martin, N. T. Thai, Y. He, W. R. Schief, P. T. Hammond and D. J. Irvine, Proc. Natl. Acad. Sci. U. S. A., 2019, 116, 16473–16478 Search PubMed.
- A. Sadeqi, G. Kiaee, W. Zeng, H. Rezaei Nejad and S. Sonkusale, Sci. Rep., 2022, 12, 1853 CrossRef CAS PubMed.
- Z. Yin, D. Kuang, S. Wang, Z. Zheng, V. K. Yadavalli and S. Lu, Int. J. Biol. Macromol., 2018, 106, 48–56 Search PubMed.
- Z. Zhu, J. Wang, X. Pei, J. Chen, X. Wei, Y. Liu, P. Xia, Q. Wan, Z. Gu and Y. He, Sci. Adv., 2023, 9, eadh2213 Search PubMed.
- S. Li, P. Wu, Z. Ji, Y. Zhang, P. Zhang, Y. He and Y. Shen, J. Biomater. Sci., Polym. Ed., 2023, 34, 482–496 CrossRef CAS PubMed.
- Y. Yu, S. Xu, S. Li and H. Pan, Biomater. Sci., 2021, 9, 1583–1597 RSC.
- W. S. Denham and W. Brash, J. Text. Inst., Trans., 1927, 18, T520–T525 CrossRef.
| This journal is © The Royal Society of Chemistry 2025 |




