Synergistic antibacterial drug elution from UHMWPE for load-bearing implants†
Nicoletta
Inverardi
 ab,
Maria F.
Serafim
a,
Anthony
Marzouca
a,
Keita
Fujino
ab,
Maria F.
Serafim
a,
Anthony
Marzouca
a,
Keita
Fujino
 a,
Matheus
Ferreira
a,
Mehmet D.
Asik
ab,
Amita
Sekar
a,
Matheus
Ferreira
a,
Mehmet D.
Asik
ab,
Amita
Sekar
 ab,
Orhun K.
Muratoglu
ab and
Ebru
Oral
ab,
Orhun K.
Muratoglu
ab and
Ebru
Oral
 *ab
*ab
aHarris Orthopaedic Laboratory, Massachusetts General Hospital, Boston, Massachusetts 02114, USA. E-mail: eoral@mgh.harvard.edu; Tel: +1 6177260657
bDepartment of Orthopaedic Surgery, Harvard Medical School, Boston, Massachusetts 02114, USA
First published on 13th January 2025
Abstract
Total joint replacement is a successful procedure for restoring the patient's musculoskeletal mobility and quality of life, but it carries the risk of severe peri-prosthetic joint infections (PJI) and is accompanied by post-operative pain. Cocktails of multiple drugs are often used for prevention/treatment of PJI and for addressing pain. Local drug delivery systems are promising for improving the outcome of the treatment and decreasing the side effects of systemic drugs. To this end, the ultra-high molecular weight polyethylene (UHMWPE) bearing surface of the joint implant is here proposed as a platform for simultaneous release of multiple therapeutics. The combined use of non-antibiotic drugs and antibiotics, and their incorporation into UHMWPE allows to obtain novel antibacterial implant materials. The combined elution of analgesics and antibiotics from UHMWPE is found to be synergistically effective in eradicating Staphylococcus aureus, as the non-antibiotic compound significantly enhances the antibacterial activity of the antibiotic. The drug properties and the employed method for their incorporation into UHMWPE are found to dictate the morphology, thus the mechanical properties of the resulting material. By adopting various fabrication methods, novel formulations showing an enhanced antibacterial activity and outstanding mechanical properties are here proposed to amplify the functionality of polymeric implant materials.
Introduction
Total joint arthroplasty is one of the most common surgical operations that can significantly improve the quality of life of arthritis patients by restoring the function of a joint with an artificial implant.1 While the benefits for the patient are manyfold and substantial, this surgical procedure is invasive and can have some significant complications.2,3Peri-prosthetic joint infection (PJI) is a severe complication of joint arthroplasty, commonly occurring within 3 months of the primary surgery. While its incidence is low after primary joint replacement (∼1–2%), it represents the major reason for the revision of joint replacements (∼25%),4 the annual cost of which exceeded $1.6B in 2020.5,6 The standard treatment comprises IV antibiotics, implant removal and debridement followed by insertion of a temporary bone cement joint spacer eluting antibiotics in the first stage surgery.7–9 The patient is largely immobilized for 6–8 weeks until the joint is clear of infection.10 In the second stage surgery, the spacer is removed, and a new joint replacement is performed.11 The treatment is effective in ∼75% of cases and subsequent surgeries have increased risk with often dire consequences such as arthrodesis or amputation.12 Recently, ultra-high molecular weight polyethylene (UHMWPE), which is the polymer used to manufacture one of the bearing surfaces of a joint implant device in total joint reconstructions, was also designed as a drug delivery platform for the local release of antibiotics to prolong antibiotic release and to improve load bearing by patients during treatment.13,14 In recent years, radiation and chemical cross-linking and antioxidant stabilization of this polymer have been developed to improve wear resistance,15 and oxidation resistance,16 respectively. Adding the functionality of drug elution to this highly successful implant material is desirable for treatment, but especially for prophylaxis due to its potential impact on the >3 million patients undergoing these surgeries annually.17
A challenge in the development of new sustained delivery devices for antibiotics is the concern of increasing antibacterial resistance by delivering sub-effective concentrations over long durations.18 Alternative approaches for improving antibacterial efficacy without increasing the risk of antibacterial resistance are based on the discovery and development of anti-microbial peptides,19 on anti-microbial polymers,20–22 and on the use of natural antibacterial compounds.23–25 Another interesting possibility is the repurposing of non-antibiotic drugs used in conjunction with antibiotics to enhance the antibacterial activity.26 One of the advantages of this approach is their potential faster and easier clinical translation compared to new drugs. The existing toxicological and safety data for these known compounds can be used in the initial stages of repurposing, but new safety and toxicological data will be nevertheless needed in relationship to the new indication.27
The antibacterial activity of topical anaesthetics was reported in dental practice,28 and the antimycobacterial properties of ion channel blockers were investigated clinically in pulmonary medicine.29 Post-operative pain associated with total joint replacement is usually addressed by oral, systemic, or intra-articular administration of multiple classes of analgesics, often including opioids.30 Local anaesthetics such as bupivacaine and ropivacaine are commonly used in intra-articular injections during the surgery.31,32 Recently, new formulation alternatives have been available (for example, Exparel®, an FDA approved liposomal carrier for releasing the non-opioid analgesic bupivacaine) for extending the release of the analgesic drug in the post-operative period.33
We hypothesized that the incorporation of both analgesic and antibiotic drugs into UHMWPE can be beneficial for providing a combined antibacterial effect by leveraging additive/synergistic drug combinations; and also sustaining analgesic delivery in the immediate post-operative period. A screening based on the fractional inhibitory concentration assay of various analgesics, NSAIDs and antibiotics used in orthopaedics revealed that combinations of the antibiotic gentamicin and the NSAID ketorolac, and the analgesics lidocaine and bupivacaine can show synergistic or additive effects towards Staphylococcus aureus, one of the main microorganisms involved in PJI.34–36 Recent mechanistic studies proposed the mechanism of synergy for gentamicin and ketorolac against Staphylococcus aureus and Staphylococcus epidermidis as consisting of a multitude of changes in the bacterial biofilm properties and morphology and of a stiffening effect exerted by ketorolac on the staphylococcal membrane.36 More in general, changes in the bacterial membrane permeability, stiffness and bacterial membrane rupture are reported in the literature as one of the probable mechanisms of antibacterial activity.26 To test our hypotheses, we incorporated the commonly used anaesthetic bupivacaine with vancomycin or gentamicin in UHMWPE and characterized the antibacterial activity, drug release and mechanical properties of the resulting material. We proposed different methods for incorporating the analgesic bupivacaine into the vancomycin/gentamicin-UHMWPE blend to improve mechanical properties. Tensile testing, impact toughness testing, and fatigue testing were performed to determine the suitability of these formulations as an implant material for load-bearing applications.
Materials and methods
Materials
Vancomycin hydrochloride (VC) (Hospira, Inc.), and gentamicin sulfate (GS) (Fujian Fukang Pharmaceutical Co.), bupivacaine hydrochloride (BP HCl) (bupivacaine hydrochloride 98.0+%, B392525G, TCI America™, Portland, OR) and bupivacaine free base (BP base) (16618, Cayman Chemical) were used, along with ultra-high molecular weight polyethylene (UHMWPE) (GUR® 1020, Celanese Corporation). Mueller Hinton broth (MHB; BD BBL™ B12322), BacTiter-Glo™ Microbial Cell Viability Assay (G8231, Promega), Tryptic soy agar (BD Difco™ 236950), Tryptic soy broth (BD Bacto™ 211825), and De-ionized water (751-628, Sigma-Aldrich) were used. LCMS grade water (W64), acetonitrile (AC615140025) and Heptafluorobutyric acid (53104) were purchased from Thermo Scientific.Bacterial culture and maintenance
Staphylococcus aureus ATCC 12600 (susceptible to GS, VC, and methicillin) was used as the control strain. All bacterial culture media and broth were prepared according to manufacturer's instructions in de-ionized water, autoclaved at 121 °C for 20 minutes, to ensure sterility. The glycerol stocks of S. aureus were thawed from −80 °C, cultured, and maintained on Tryptic Soy Agar (TSA) plates. The bacterial colonies were inoculated in 1 ml of Tryptic Soy Broth and incubated at 35 °C for 18–24 hours of optimal growth prior to all experiments.Materials preparation
UHMWPE-based compositions were prepared according to three categories: (i) virgin UHMWPE (i.e., without any drug loading); (ii) homogeneously consolidated drug loaded UHMWPE (antibiotic loaded UHMWPE, analgesic loaded UHMWPE, or dual drug loaded UHMWPE with antibiotic and analgesic); (iii) layered composite material with layers of virgin UHMWPE and of antibiotic and analgesic loaded UHMWPE; (iv) drug doped material after consolidation. The prepared compositions are listed in Table S1, ESI.† A schematic of the manufacturing is shown in Fig. 1.Blending drugs with UHMWPE
UHMWPE/drug blends were prepared by first sieving the respective drugs with a 75 μm mesh and then mixing the sieved drugs with virgin UHMWPE for 30 min by mechanical turbulation (Turbula®, WAB, Muttenz, Switzerland) according to the compositions reported in Table S1 (ESI†). Subsequently, the blends were dehydrated in a vacuum oven at 90 °C for 16–18 hours. The nominal weight percentage of the antibiotic was adjusted by considering its weight loss due to dehydration.Compression molding
Dehydrated blends were molded inside a custom-made mold (surface area: 50 × 85 mm2) by using a press (Carver Press, Carver, Wabash, IN) at a temperature of 170 °C under 20 MPa followed by cooling for at least 45 min down to room temperature under pressure. The molding time at 170 °C was 10 and 20 min for 5 mm and 10 mm thick blocks, respectively. Virgin UHMWPE blocks were also molded as control in the same custom-made mold at a temperature of 180 °C under 20 MPa.Diffusion doping of bupivacaine free base
1% VC loaded UHMWPE was also doped with BP base by diffusion doping, as described elsewhere.37 Briefly, the consolidated 1% VC loaded UHMWPE was machined according to the following testing (3 × 5 × 20 mm3 strips, 3.2 × 24 × 65 mm3 plates, 6.35 × 16 × 63.5 mm3) and soaked into molten BP base at 120 °C for 4 h, with mild agitation every 30 min and under inert gas purging (N2 gas).Manufacturing of layered composite materials
Composite blocks were obtained by a layering approach consisting of a section of virgin UHMWPE and a section of drug loaded UHWMPE, with varying ratio of the thickness of the drug loaded layer to the total thickness (i.e., 0, 0.25, 0.50, 1). The drug layer thickness ratio was calculated as the ratio between the thickness of the drug loaded layer to the total thickness of the block. Drug layer thickness ratio equal to 0 indicates virgin UHMWPE and 1 indicates full thickness of the drug loaded UHMWPE composition.Molding was performed by first filling the pre-heated mold at 130 °C with virgin UHMWPE powder, leveling its surface, pouring on top the dehydrated drug loaded blend and subsequently heating to 170 °C and applying 20 MPa pressure.
Machining
Molded blocks were machined into samples for elution studies (3 × 5 × 20 mm3) and antibacterial testing (3 × 5 × 10 mm3), tensile testing (3.2 × 50 × 85 mm3), Izod impact testing (6.35 × 12.7 × 63.5 mm3), and fatigue testing (compact specimen, C(T), ASTM Standard E647-15, 7 mm thick) by using a CNC machine (Shopbot Tools, Inc., Durham, NC).Physicochemical and morphological characterization
FTIR
FTIR was used to evaluate the diffusion profile of the doped drug in the thickness of the samples (for BP doped 1% VC). Samples for FTIR were obtained by microtoming a thin section (150 μm) from the cross-section of the doped samples (6.35 × 12.75 mm2, 3.2 × 5 mm2, 3.0 × 5 mm2). FTIR spectra were collected along the thickness every 0.20 mm by using an FTIR spectrometer with a microscope attachment (Agilent, Varian 670-IR with a Varian 620-IR FTIR microscope attachment; Agilent Technologies Inc., Wilmington, DE). For each material, three sections were analyzed, and each spectrum was an average of 32 scans. Virgin UHMWPE and VC loaded UHMWPE were also tested as controls. The peak selected for defining the FTIR BP index is shown in Fig. S1, ESI.† The index was defined as the ratio of the area under the drug peak (i.e., 960 cm−1, integration limit: 930–980 cm−1) and the polyethylene skeletal absorbance (i.e., 1895 cm−1, integration limit: 1850–1985 cm−1). The diffusion profile along the thickness was built by applying a spline interpolation to the three replicates of the scans for each material investigated (MATLAB, MathWorks).Morphological characterization
Cross-sections of the prismatic strips (3 × 5 mm2) were observed by optical microscopy (Olympus STM6-F00-3, Olympus Corporation, Tokyo, Japan) and scanning electron microscopy (SEM) (Gemini 560 Field Emission Scanning Electron Microscope, Zeiss, Germany). Prior to SEM, the surfaces were sputter coated with 5 nm of a platinum/palladium 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20 layer (EMS 150T S Metal Sputter Coater, Quorum Technologies, United Kingdom). The accelerating voltage was 3 kV and the Everhart–Thornley detector was used for secondary electrons.
20 layer (EMS 150T S Metal Sputter Coater, Quorum Technologies, United Kingdom). The accelerating voltage was 3 kV and the Everhart–Thornley detector was used for secondary electrons.
Elution experiments and analytical method for drug concentration quantification
Analytical methods
Concentration of drugs from the VC-BP compositions were measured by LCMS/MS (Ultivo Triple Quadrupole-2100b LC/MS system, Agilent Technologies, Santa Clara, CA). Samples were diluted with LCMS grade water, and 10 μl of samples were injected with the autosampler to the UPLC. The analytical column Poroshell 120 EC-C18 (dimensions: 2.7 μm × 2.1 mm × 100 mm, Agilent Technologies, CA) was used at 40 °C with a poroshell guard column at a flow rate of 0.4 ml min−1. The mobile phase was 10 mM Heptafluorobutyric acid in water (A) and acetonitrile (B). The gradient started at 85%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 15% (A
15% (A![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) B) and reached 5%
B) and reached 5%![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 95% (A
95% (A![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) B) at 4.5 minutes. After a 6-minute run, the device was re-equilibrated for 2.5 minutes. The source parameters were optimized for both compounds. The nitrogen gas temperatures and flows were 250 °C and 11 l min−1 and 290 with 8 l min−1 for sheath gas and the source, respectively. Capillary and nozzle voltages were set to 5000 V and 2000 V, respectively. We analyzed the samples using multiple reaction monitoring (MRM). 724.7 → 144.2 and 724.7 → 100.2 m/z were monitored for VC and 289.2→ 140.1 and 298.2 → 84 m/z for BP. VC and BP peaks were quantified by means of calibration with 7 calibration standards in Quant software (Agilent, technologies, CA). Concentration of drugs from the GS-BP compositions were measured spectrometrically by using a plate reader (Biotek, Agilent, Santa Clara, CA). The concentration of BP only in the drug eluent was determined by UV absorbance measurement at 250 nm, as GS does not show any UV/Vis absorbance. The concentration of GS was determined after reacting the drug eluent with o-pthaldialdehyde and measuring the resulting fluorescence, according to a protocol reported elsewhere.38 To ensure the independent measurement of GS only, the drug eluent was diluted to a concentration range in which the fluorescence contribution of BP reacted with o-pthaldialdehyde was negligible (<20 μg ml−1).
B) at 4.5 minutes. After a 6-minute run, the device was re-equilibrated for 2.5 minutes. The source parameters were optimized for both compounds. The nitrogen gas temperatures and flows were 250 °C and 11 l min−1 and 290 with 8 l min−1 for sheath gas and the source, respectively. Capillary and nozzle voltages were set to 5000 V and 2000 V, respectively. We analyzed the samples using multiple reaction monitoring (MRM). 724.7 → 144.2 and 724.7 → 100.2 m/z were monitored for VC and 289.2→ 140.1 and 298.2 → 84 m/z for BP. VC and BP peaks were quantified by means of calibration with 7 calibration standards in Quant software (Agilent, technologies, CA). Concentration of drugs from the GS-BP compositions were measured spectrometrically by using a plate reader (Biotek, Agilent, Santa Clara, CA). The concentration of BP only in the drug eluent was determined by UV absorbance measurement at 250 nm, as GS does not show any UV/Vis absorbance. The concentration of GS was determined after reacting the drug eluent with o-pthaldialdehyde and measuring the resulting fluorescence, according to a protocol reported elsewhere.38 To ensure the independent measurement of GS only, the drug eluent was diluted to a concentration range in which the fluorescence contribution of BP reacted with o-pthaldialdehyde was negligible (<20 μg ml−1).
Pharmacokinetic modeling
The Korsmeyer–Peppas semi-empirical equation39 reported in eqn (1) was used to approximate the cumulative drug mass release profiles: | (1) |
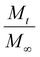 is the fraction of drug released at time t, K is the release rate constant, and n is the release rate exponent. Fickian diffusion corresponds to n ≤ 0.5, while non-Fickian diffusion (anomalous transport) is for 0.5 < n ≤ 1. Data points with
is the fraction of drug released at time t, K is the release rate constant, and n is the release rate exponent. Fickian diffusion corresponds to n ≤ 0.5, while non-Fickian diffusion (anomalous transport) is for 0.5 < n ≤ 1. Data points with 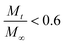 were fitted with a log-weighted linear regression for the identification of K and n.
were fitted with a log-weighted linear regression for the identification of K and n.
The prediction of the intraarticular concentration of the drugs resulting from elution from a 100 cm2 knee implant was obtained by applying a pharmacokinetic model with a single compartment (i.e., the knee joint) and an infinite sink. The drug decay was assumed to have a first-order kinetics and the half-life of the drugs was taken as 10 min for BP,40 and as 6 h for the antibiotics, which is the half-life for VC after intraarticular injection.41 On the curves of the predicted intraarticular concentration of the analgesics, we calculated the peak concentration (Cmax), the peak time (tmax) and the area under the curve (AUC) for different time periods and compared them with the results obtained for a bolus intraarticular injection of BP (50 mg) which is considered the recommended dose in the clinic.31
Antibacterial testing
MIC experiments were conducted per the Clinical and Laboratory Standards Institute (CLSI) protocol (M07-A10). Briefly, the drug stocks were prepared (VC, GS, BP HCl) in de-ionized water and were serially diluted in a 96-well clear plate using equal volume of MHB. Wells containing bacteria with no drug exposure and blank media served as positive and negative controls respectively. Diluted bacterial suspension (∼105 CFU ml−1) were added to all the wells except the blank media well and the plate was incubated for a period of 18–24 h at 35 °C. MIC of the drug was determined by visual assessment and absorbance measurement of growth inhibition at 600 nm by using a plate reader (Biotek, Agilent, Santa Clara, CA). The drug concentration that further showed no growth on a TSA plate was reported as MBC.
To determine FIC, the checkerboard assay was performed by subjecting S. aureus suspensions (∼105 CFU ml−1) to the cumulative MICs and further serial dilutions of antibiotic and analgesic drug combinations.36,42 The bacterial turbidity was determined after overnight incubation and the FIC indices were calculated using eqn (2).
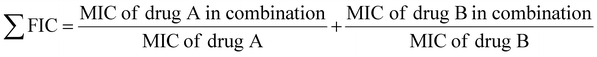 | (2) |
The numeric value of the ΣFIC determined the cumulative effect of the drug combinations: FIC < 0.5 is interpreted as synergistic effect, while FIC > 4 as antagonistic effect. For values in between, the interpretation is as follows: 0.5 < FIC < 1 is determined as partial synergy; 1 < FIC < 2 is determined as additive; 2 < FIC < 4 is determined as indifference.
Antibacterial activity of the drug loaded UHMWPE
The semi-dynamic antibacterial activity of the drug loaded UHMWPE was measured by using a purposely developed method based on an ATP-based real time microbial viability assay.43 A drug-eluting prismatic strip (3 × 5 × 10 mm3) was placed in a syringe and incubated with 1.35 ml of 105 CFU ml−1S. aureus in MHB at 37 °C, 100 rpm (n = 3). An aliquot of the bacterial suspension was collected at given timepoints (6 h, 1, 2, 3 and 7 days) and the real time microbial viability was measured as luminescence units using the BacTiterGlo™ reagent. The readings were converted to CFU ml−1 using a strain-specific calibration curve (limit of detection: ∼103 CFU ml−1). To confirm eradication, 10 μl of the bacterial broth was plated on tryptic soy agar and incubated for 24 hours. At each time point, the remaining bacterial solution was centrifuged at 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 × g for 10 min, the supernatant spent media was discarded, and the bacterial pellet was resuspended in fresh MHB. The renewed suspension was incubated with the drug-eluting strip until the next time point. Virgin UHMWPE incubated with bacteria served as positive control (n = 3).
000 × g for 10 min, the supernatant spent media was discarded, and the bacterial pellet was resuspended in fresh MHB. The renewed suspension was incubated with the drug-eluting strip until the next time point. Virgin UHMWPE incubated with bacteria served as positive control (n = 3).
Mechanical testing
Izod impact testing
Impact strength testing was performed according to Izod testing (ASTM F648-14, n = 3–6). Machined blocks (6.35 × 12.7 × 63.5 mm3) were first double notched with a manual custom-made notching machine and pictures of the notched surfaces were taken with a camera (PowerShot G10, Canon, Japan). For those samples obtained by the layering approach, the thickness of the drug layer and of the virgin UHMWPE layer was measured by using a software for image processing (ImageJ, National Institute of Health). The actual drug thickness ratio was calculated for each sample. The impact strength was measured by using the Instron pendulum CEAST 9050 (Impactor II, Instron, Norwood, MA). The fracture surface was observed by SEM (Gemini 560 Field Emission Scanning Electron Microscope, Zeiss, Germany) at an accelerating voltage of 3 kV and detecting secondary electrons, after sputter coating (EMS 150T S Metal Sputter Coater, Quorum Technologies, United Kingdom). Freeze-fracture surfaces for 0.5% GS + 9.5% BP base were also obtained by fracturing untested but pre-notched Izod samples after immersion in liquid nitrogen. Energy dispersive X-ray spectroscopy (EDX) during SEM was performed by using an Ultim Max EDS detector (Oxford Instruments, United Kingdom) at an accelerating voltage of 3 kV. The Kα1,2 signals for Carbon and the Kα1,2 signals for Nitrogen were recorded to identify the drug phase. Fracture surfaces were also observed by SEM after an extraction process aimed at fully extracting the drugs from 0.5% GS + 9.5% BP base without affecting the polymeric matrix. The extraction was performed by immersing the fracture surface in pure ethanol for several hours and refreshing with new solvent every two hours and allowing to complete dry overnight before sputter coating for SEM analysis.Fatigue testing
Resistance to fatigue crack propagation testing was performed according to ASTM standard E647 on type A1 compact tension specimens (7 mm thick; n = 3) by using a servohydraulic dynamometer (858 Mini Bionix, MTS Systems Corporation, Eden Prairie, MN) equipped with two high resolution cameras (Flea3 USB3 Model FL3-U3-120S3C-C, FLIR Systems, Inc., Wilsonville, OR) with zoom lenses (Computar MLH-10X manual varifocal lens, CBC America, NC). Compact tension specimens were pre-cracked by using a custom-made vise, according to the geometry prescribed in section 8.3 of ASTM E647 (pre-crack length of 1.875 mm). A custom software (Automated Fatigue Crack Propagation System (aFCP), Cambridge Polymer Group, Woburn, MA) was used for acquiring images throughout the test on both sides of the samples and for continuously measuring the crack length. Prior to testing, the imaging system was calibrated with a NIST-traceable dot grid. The sample was subjected to a cyclic load by applying a sinusoidal waveform at the frequency of 3 Hz and load ratio R = 0.1. The maximum force applied was 482.7 ± 2.5 N for virgin UHMWPE and 363.3 ± 5.8 N for 0.5% GS + 9.5% BP base. Thresholding and edge detection algorithms were used to determine the crack length from the location of the crack tip and the border of the sample. The crack lengths from each camera were averaged and used to create a crack length versus cycle count graph. Fatigue crack growth threshold was calculated according to section 9.4 of ASTM E647, with the lower bound, upper bound, and intercept set to 1 × 10−9, 3 × 10−8, and 1 × 10−9 respectively.Safety statement
Bupivacaine hydrochloride (CAS: 18010-40-7) and bupivacaine free base (CAS: 38396-39-3) are hazardous materials and need to be handled with care, by wearing the suitable PPEs, by working under a chemical fume hood and according to the precautionary statements of the safety datasheets.Statistical analysis
The results are presented with average and standard deviation, and the number of replicates used was n = 3, if not otherwise specified. A Student's t-test for two-tailed distribution of unequal variance was used to compare individual sets of data. The significance was assigned to p < 0.05.Results
Manufacturing of drug loaded UHMWPE and assessment of the distribution of bupivacaine in diffusion-doped UHMWPE
Consolidated blends of UHMWPE with the analgesic bupivacaine (BP) and the antibiotic vancomycin hydrochloride (VC) or gentamicin sulfate (GS) were prepared according to the formulations reported in Table S1, ESI† and to three manufacturing routes, as outlined in Fig. 1. While powder blended materials consolidated homogeneously were designed to have uniform distribution of drug, diffusion doping resulted in a non-uniform and thickness dependent drug distribution (Fig. 1). The profile of the diffused drug, i.e., bupivacaine base (BP base) in the BP doped 1% VC composition, was built after identifying an FTIR peak specific to BP (Fig. S1, ESI†) and showed the presence of drug-rich region at the surface of samples, regardless of the sample geometry, with a surface-to-bulk concentration ratio of 20–25![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 (Fig. 1(b) and (c)). The prismatic samples of BP doped 1% VC used for elution studies had drug intake equal to 27 ± 5.1 mg, corresponding to about 9.6% of drug loading. Samples with different geometries had a similar ratio of drug intake to surface area between 5.8–7.5 mg cm−2 (Table S2, ESI†).
1 (Fig. 1(b) and (c)). The prismatic samples of BP doped 1% VC used for elution studies had drug intake equal to 27 ± 5.1 mg, corresponding to about 9.6% of drug loading. Samples with different geometries had a similar ratio of drug intake to surface area between 5.8–7.5 mg cm−2 (Table S2, ESI†).
Determination of the real-time antibacterial activity for synergistic blends
The free antibiotic drugs vancomycin hydrochloride (VC) and gentamicin sulfate (GS) displayed similar minimum inhibitory concentration (MIC) values against S. aureus 12600, whereas there was a greater discrepancy in the value of the minimum bactericidal concentration (MBC), as GS required about eight times higher concentration to effectively eradicate this bacterial strain (Table 1). The local anaesthetic bupivacaine hydrochloride (BP HCl), showed about three orders of magnitude higher MIC and MBC than those of the antibiotics. The fractional inhibitory concentration (FIC) obtained by the checkboard assay was similar for the VC-BP and GS-BP combinations (Table 1). The FIC values reported were in the interval which is associated with an additive effect of the combined drugs (1 < FIC < 2).The antibacterial synergy of VC/GS with BP combinations was also tested for the eluted drugs from the implant material under a semi-dynamic approach. After an initial bacterial inoculum (i.e., 105 CFU ml−1) was incubated together with the eluting UHMWPE implant, the bacterial viability determined at various timepoints was significantly affected depending on the UHMWPE formulation used (Fig. 2(a) and (b)). Low antibiotic loading into UHMWPE, as in the case of 1% VC and 0.5% GS, was only capable of inhibiting the bacterial growth up to 1–2 days. After that, high levels of bacterial growth (i.e., 108 CFU ml−1) were observed, similar to the positive control used (i.e., virgin UHMWPE). Supplementing the antibiotic loading with 4–4.5 wt% of BP HCl (as in the case of 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl) allowed to achieve complete bacterial eradication within 2–3 days, as evaluated by the viability assay and confirmed by plating results (Fig. 2(a) and (b)). The use of BP base together with the antibiotic into UHMWPE also resulted in the perturbation of the bacterial viability. Doping 1% VC with BP base led to full bacterial eradication within 2 days (Fig. 2(a)), blending BP base with 0.5% GS led to bacterial inhibition up to 3 days for 0.5% GS + 4.5% BP base, and complete eradication within 7 days for 0.5% GS + 9.5% BP base (Fig. 2(b)).
The UHMWPE formulations with antibiotic loading only (i.e., 1% VC and 0.5% GS) displayed concentrations which quickly fell below their respective MIC and stayed at sub-MIC values for the remaining period of the study: for VC, after the burst release (∼3 μg ml−1) in the first time interval (0–6 h), the concentration was lower than 0.5 μg ml−1 thereafter; for GS, the concentration was less than 1 μg ml−1 at the 0–6 h burst period, and remained lower thereafter (Fig. 2(c)). The dual drug loaded compositions led to different antibiotic concentration in the assay media depending on the BP form. For compositions blended with BP HCl (i.e., 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl), the released antibiotic concentration was higher than MIC for the whole duration of the study, both with VC and GS. The antibiotic concentration was also greater than MBC for the whole duration of the study for the case of VC (from 5× to 20× MBC), while it was greater than MBC at the burst time period (0–6 h) and the last time period (3d–7d) for the case of GS (2.5× and 2× MBC, respectively). For compositions with BP base (i.e., BP doped 1% VC and 0.5% GS + 9.5% BP base), the released antibiotic concentrations quickly fell below MIC and remained lower than MIC for both VC and GS at the end of the study (∼0.1 μg ml−1) (Fig. 2(c)).
The concentration of the analgesic from the dual drug loaded UHMWPE also strongly depended on the incorporation method and the BP form; however, the analgesic concentration was lower than the MIC of BP against S. aureus 12600 for all compositions (Fig. 2(d) and Table 1). For compositions blended with BP HCl (i.e., 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl), the released analgesic concentration was very similar for the VC-based and GS-based formulations at all timepoints. After the burst release (∼100 μg ml−1 in the 0–6 h time period), the concentration decreased down to ∼10 μg ml−1 at the 2d–3d time period and ended at about ∼30 μg ml−1 during the last time period (3d–7d). For the composition with BP base by doping (i.e., BP doped 1% VC), the analgesic concentration remained stable around 20–30 μg ml−1 for all the time periods. For the composition blended with BP base (i.e., 0.5% GS + 9.5% BP base), the released analgesic concentration was also stable throughout the study; the values were about 1 order of magnitude lower than those of the BP doped 1% VC formulation (∼2–3 μg ml−1 for all the time periods) (Fig. 2(d)).
Morphology of the drug domains in UHMWPE
The size and dispersion of the drug domains in the UHMWPE matrix strongly depended on the drug and drug loading for single antibiotic loaded UHMWPE and for the dual drug loaded UHMWPE with BP HCl (Fig. 3). Drug domains for 5% GS looked circular and dispersed in the matrix (Fig. 3(d)) in contrast to those of 5% VC, which looked thinner and more elongated with partial interconnection of different domains throughout the matrix (Fig. 3(a)). For the dual drug loaded UHMWPE with BP HCl (i.e., 1% VC + 4% BP HCl, 0.5% GS + 4.5% BP HCl), the drug domains appeared to be distributed around the grain boundaries of UHMWPE leading to an interconnected and percolated domain structure (Fig. 3(b) and (e)).For the dual drug loaded UHMWPE with BP base, the observed drug domains were ascribed only to the antibiotics as they had similar morphology to their respective counterpart in single antibiotic loaded UHMWPE – circular for the case of 0.5% GS + 9.5% BP base (Fig. 3(f)) and more elongated for the case of BP doped 1% VC (Fig. 3(c)).
By SEM imaging, spherical GS particles with a size distribution of 5–50 μm were observed for 5% GS (Fig. 4(d)) and, in a lower amount, also for 0.5% GS + 9.5% BP base (Fig. 4(f)). Prismatic plate-like particles (principal dimension <75 μm) were observed for VC in 5% VC (Fig. 4(a)) and, in a lower amount, also for BP doped 1% VC (Fig. 4(c)). The morphological features for 1% VC + 4% BP HCl (Fig. 4(b)) and 0.5% GS + 4.5% BP HCl (Fig. 4(e)) were largely due to BP HCl: the rectangular cross-sections (aspect ratio 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, width around 10 μm) of the drug fibers were visible at the polymer grain boundaries. The domain size of the BP base was substantially different compared to that of BP HCl
2, width around 10 μm) of the drug fibers were visible at the polymer grain boundaries. The domain size of the BP base was substantially different compared to that of BP HCl![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) BP base appeared to be in the shape of needle-like fibers with cross-sections in the nanometer scale and length in the micrometer scale. These needle-like fibers were evident at the interface between the polymeric matrix and the antibiotic domains (Fig. 4(c) and (f)).
BP base appeared to be in the shape of needle-like fibers with cross-sections in the nanometer scale and length in the micrometer scale. These needle-like fibers were evident at the interface between the polymeric matrix and the antibiotic domains (Fig. 4(c) and (f)).
Characterization of drug elution and pharmacokinetic modelling
The cumulative drug mass for the antibiotic released from strips (3 × 5 × 20 mm3) containing BP HCl (i.e., 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl) was similar for VC and GS-blended UHMWPE, with a similar initial burst of about 70 μg (6 h) and a cumulative mass of 0.2–0.3 mg at the end of the study (28d) (Fig. 5(a)). The antibiotic released from compositions with BP base (i.e., BP doped 1% VC and 0.5% GS + 9.5% BP base) was significantly lower than that from the compositions with BP HCl; the burst amount was about 9–10 μg (6 h) and the cumulative amount was 16–18 μg (Fig. 5(a)). The release behavior of BP HCl was similar regardless of the antibiotic used (for 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl): the burst amount was about 0.6 mg, and the cumulative amount was about 1.5–1.7 mg (Fig. 5(b)). The release profile for BP base was substantially different with a significantly lower burst (∼0.1–0.2 mg). The cumulative BP base amount was greater from the doped material (1.5 mg) compared to that from the blended one (∼0.8 mg) (Fig. 5(b)).Compositions blended with BP HCl released a similar percentage of the initial BP HCl (12–14%) (Fig. 5(d)), whereas the antibiotic release was significantly higher for GS (∼18% from 0.5% GS + 4.5% BP HCl) compared to that of VC (∼7% from 1% VC + 4% BP HCl) (Fig. 5(c)). The cumulative fractional release in the compositions supplemented with BP base was significantly lower both for the antibiotic (∼1.2% from 0.5% GS + 9.5% BP base and ∼0.6% from BP doped 1% VC) (Fig. 5(c)) and for the analgesic (∼6% from BP doped 1% VC and ∼3% from 0.5% GS + 9.5% BP base) (Fig. 5(d)) compared to the ones with BP HCl.
Drug release was Fickian for all cases, as the release exponent, n, was lower than 0.5 (Table S3, ESI†). The release exponent for the antibiotic was higher in the case of the antibiotic eluting from compositions with BP HCl compared to that eluting from compositions with BP base. The release exponent for the analgesic was higher for BP base compared to BP HCl (Table S3, ESI†).
Intraarticular concentrations of the drugs eluted from a full implant component (surface area of 100 cm2) were predicted by using a pharmacokinetic model with a single compartment (the knee compartment with a volume of 2 ml) and exponential decrease of the concentration based on the half-life of the drug. The predicted intraarticular antibiotic concentrations were above their respective MICs for three weeks for compositions with BP HCl; at day 21 both the GS concentration (from 0.5% GS + 4.5% BP HCl) and the VC concentration (from 1% VC + 4% BP HCl) were 20× of their respective MICs (Fig. 5(e)). The predicted intraarticular antibiotic concentration released from compositions with BP base decayed much faster and remained higher than MIC for about two weeks for 0.5% GS + 9.5% BP base and for about 18 days for BP doped 1% VC. The analgesic concentrations had a lower Cmax, ranging from 0.4 to 2.7 mg ml−1 depending on the composition compared to the bolus 50 mg intraarticular injection of BP used as a comparison, which was quickly cleared out. The area under the curve (AUC) evaluated between 0 and 14 days was similar for the compositions containing BP HCl compared to the bolus injection (Fig. 5(f) and Table S4, ESI†).
Mechanical properties
Mechanical properties measured under tensile conditions were significantly dependent on the total drug loading, the incorporation method, and the drug form (Fig. S2, ESI†). When considering single drug loaded UHMWPE with antibiotics or the analgesic BP HCl, there was a linear decrease (R2 = 0.800) of UTS as a function of drug loading with the maximum decrease (at 10 wt% drug loading) around 40–60% compared to UHMWPE without drugs. A value of UTS close to 30 MPa can be obtained by limiting the drug loading to 7 wt% or lower (Fig. S2, ESI†).44 The ductility measured as EAB was less impacted, as the percentage decrease was about 16–29% with a weaker dependence on the drug loading (R2 = 0.418) and EAB values were higher than 300% except for the case of 10% BP HCl (Fig. S2, ESI†). For single drug loaded UHMWPE with 10% BP base, UTS and EAB were not statistically different than those of virgin UHMWPE (p > 0.05).For dual drug loaded UHMWPE with the antibiotic and BP HCl at a total loading of 5 wt% (i.e., 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl), the UTS and EAB were not statistically different than those of the single drug loaded 5% BP HCl (p > 0.05). For a total drug loading of 7 wt% (i.e., 1% VC + 6% BP HCl and 0.5% GS + 6.5% BP HCl), there was a significant decrease in UTS and EAB compared to the properties for the single drug loaded 7% BP HCl (p < 0.05).
Supplementing 1% VC with BP base by diffusion doping instead of by blending with 4% BP HCl resulted in an increased UTS (p < 0.05). The UTS and EAB of the BP doped 1% VC were similar to those of 1% VC (p > 0.05), in contrast to 1% VC + 4% BP whose UTS and EAB were decreased compared to 1% VC (p < 0.05).
Supplementing 0.5% GS with 4.5% BP base by blending resulted in increased UTS and EAB compared to 0.5% GS + 4.5% BP HCl (p < 0.05). The UTS and EAB of the 0.5% GS + 4.5% BP base were similar to those of the 0.5% GS (p > 0.05), in contrast to 0.5% GS + 4.5% BP HCl whose UTS and EAB were decreased comparatively (p < 0.05). Further, the UTS for 0.5% GS + 4.5% BP base was similar to that of 0.5% GS + 9.5% BP base (p > 0.05); both the UTS and EAB of the 0.5% GS + 9.5% BP base were similar to those of the 10% BP base (p > 0.05).
Toughness was significantly affected by the selection of the drugs, their incorporation method and the design of the composites, as measured by Izod impact strength (Fig. 6). Compositions obtained by blending with BP HCl (i.e., 1% VC + 4% BP HCl, 0.5% GS + 4.5% BP HCl) were prepared both as homogenously consolidated blocks and as layered composites with a drug loaded layer of varying thickness and a virgin UHMWPE layer (Fig. 1). Their Izod impact strength had a strong linear correlation with the ratio of the drug layer to the total thickness (R2 ∼ 0.94–0.97). A ratio smaller than 0.5 was found to provide an Izod impact strength above 100 kJ m−2, while a full thickness drug loaded UHMWPE (i.e., drug layer thickness ratio equal to 1) resulted in Izod impact strength close to 60 kJ m−2 (Table 2), regardless of the antibiotic used (p > 0.05 when comparing 1% VC + 4% BP HCl to 0.5% GS + 4.5% BP HCl).
| UTS (MPa) | EAB (%) | Izod impact strength (kJ m−2) | Stress intensity factor at crack inception, ΔKincep (MPa m1/2) | |
|---|---|---|---|---|
| a N.t.: not tested. | ||||
| Virgin UHMWPE | 50.8 ± 2.5 | 448 ± 34 | 147.2 ± 3.3 | 1.72 ± 0.18 |
| 1% VC + 4% BP HCl loaded UHWPE | 32.5 ± 0.5 | 336 ± 17 | 62.9 ± 3.7 | N.t.a |
| BP doped 1% VC loaded UHMWPE | 37.9 ± 1.4 | 362 ± 20 | 141.3 ± 7.7 | N.t.a |
| 0.5% GS + 4.5% BP HCl loaded UHMWPE | 32.9 ± 0.5 | 333 ± 10 | 60.0 ± 8.4 | N.t.a |
| 0.5% GS + 9.5% BP base loaded UHMWPE | 37.7 ± 2.5 | 403 ± 22 | 140.9 ± 4.0 | 1.39 ± 0.04 |
Compositions obtained by doping BP base or blending BP base (i.e.EBP doped 1% VC, 0.5% GS + 9.5% BP base) in a homogeneously consolidated block led to high values of Izod impact strength, which were almost unchanged compared to that of virgin UHMWPE (p > 0.05 for the case of BP doped 1% VC as opposed to virgin UHMWPE) (Fig. 6 and Table 2).
Fatigue resistance was also tested by measuring the resistance of fatigue crack growth propagation for 0.5% GS + 9.5% BP base compared to virgin UHMWPE: the stress intensity factor at crack inception, ΔKincep, resulted to be about 19% decreased for this drug loaded UHMPWE compared to virgin UHMWPE (Table 2).
Fracture morphology
A strong and periodic lineage with ridges perpendicular to the direction of crack propagation during impact could be observed for virgin UHMWPE with buckled features visible at the high magnification (Fig. 7(a)). The compositions with BP HCl displayed mixed fracture modes with similar ridge features ascribed to UHMWPE recognizable only in limited areas surrounded by drug particles after their brittle fracture – for example, with some truncated plate-like particles of VC (Fig. 7(b) – second row) and some truncated rod-shape particles for BP HCl (Fig. 7(b) – third row, Fig. 7(c) – second and third rows). Compositions supplemented with BP base displayed a (impact) fracture surface, which was very similar to that of virgin UHMWPE at low magnification. For both BP doped 1% VC (Fig. 7(d)) and 0.5% GS + 9.5% BP base (Fig. 7(e)), it was possible to observe a similar periodic lineage perpendicular to the direction of crack propagation, with the additional feature of a unique surface texturing. Higher magnification for both cases showed that this feature consists of a dispersion of needle-like fibers with cross-section on the nanometer scale (Fig. 7(d) – third row and Fig. 7(e)). For BP doped 1% VC, this surface texture was visible close to the lateral surfaces (Fig. 7(d) second row displays the central part of the cross-section, while the third row displays the part closer to the lateral surface), while for 0.5% GS + 9.5% BP base this feature was found throughout the whole cross-section (Fig. 7(e)).In a comparison of the impact and freeze-fracture surfaces of 0.5% GS + 9.5% BP base before and after drug extraction (Fig. S3, ESI†), the freeze-fracture surfaces showed some topological features typical of a brittle fracture (Fig. S3a first row, ESI†); while nanometer-sized fibers were also present, their amount was less compared to the impact fracture (Fig. S3b first row, ESI†). EDX of the impact fracture surface before drug extraction showed the wide-spread presence of carbon, which was ascribed mainly to the polymer, and of nitrogen, which was ascribed mainly to BP (Fig. S3c, ESI†). After drug extraction, the impact surface did not have any fibers (Fig. S3b second row, ESI†) and revealed underneath the lineage morphology perpendicular to the direction of crack propagation typical of virgin UHMWPE.
The morphology of the fatigue surfaces was observed close to the crack initiation region for virgin UHMWPE (Fig. 8(a)) and for 0.5% GS + 9.5% BP base (Fig. 8(b)). In both cases, the typical morphology for fatigue surfaces of UHMWPE characterized by a diamond-like pattern (45° striations),45,46 with the major axis of the diamond aligned with the direction of crack propagation, was observed at low magnification. Higher magnification allowed to highlight the presence of lines perpendicular to the direction of crack propagation and the region at the edge of the diamond pattern, which displayed buckled edges and finer lines parallel to the direction of the propagating crack. The drug loaded 0.5% GS + 9.5% BP base had all these features, with the addition of the presence of some holes (<100 μm, dimension and shape close to those of the GS particles) visible at low magnification (Fig. 8(b) first row) and of distributed nanometer-sized fibers (Fig. 8(b) second row).
Discussion
Persistent and hard-to-eradicate bacterial infections are a serious healthcare problem that may worsen in the next years due to the growing concern related to antimicrobial resistance and the emergence of multidrug-resistant microorganisms.47 The rate at which bacteria are acquiring resistance to the current available antibiotics is not met by the rate of discovery of new treatment modes including new classes of antibiotics. Traditional de novo drug discovery and development are known to be very lengthy, expensive, and risky.48 An interesting alternative strategy for extending the life of current antibiotics involves the combination of non-antibiotic drugs with antibiotic drugs to improve antibacterial efficacy.49 This approach can also be enhanced by the repurposing of currently used drugs approved for other indications.48,50 Major benefits of this strategy are the possibility of considerably shortening the timeline to the clinic and minimizing the risk for the patients due to existing information on the safety, toxicological and pharmacokinetic profiles of the repurposed drugs.In PJI, where the main causative pathogen is Staphylococcus aureus, increasing antibiotic resistance is a concern as resistance against β-lactams (MRSA – Methicillin-resistant Staphylococcus aureus) is already widespread, especially in revision surgeries.51,52 In this work, we are proposing to use the nonconventional antibiotic local anaesthetic bupivacaine to improve the antibacterial efficacy of gentamicin and vancomycin, which are commonly employed in orthopaedics to control PJI. Our approach is to fashion an antibacterial drug delivery device out of the UHMWPE bearing surface used in total joint replacement to elute an effective combination of these drugs.
Our hypothesis that bupivacaine could synergistically enhance the antibacterial activity of gentamicin and vancomycin was strongly supported. The FIC checkboard assay suggested additive efficacy of the selected drug combinations in their free forms (Table 1). The feasibility study for demonstrating that the drugs could act synergistically also when eluting from the polymeric matrix was performed with very small samples and also low antibiotic concentrations to ensure limited antibiotic activity with limited antibiotic elution from the UHMWPE. We used a real-time testing of antibacterial activity we developed for the drug loaded material incubated with the same bacterial population over 7 days,43 and showed that the polymeric material incorporated with additional bupivacaine showed strong synergistic antibacterial activity with the antibiotics (Fig. 2(a) and (b)). For the combinations with BP HCl (i.e., 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl), the synergistic antibacterial activity was influenced also by the synergistic elution of the two molecules: the presence of BP HCl helped obtain a higher elution of the antibiotics, which resulted in concentrations higher than their respective MBCs for 1 day and their respective MICs for 7 days (Fig. 2(c)). In contrast, for combinations with BP base (i.e., BP doped 1% VC and 0.5% GS + 9.5% BP base), the synergistic antibacterial activity resulted only from the combined action of the two drugs, as both the eluted antibiotic and analgesic concentrations were lower than their respective individual MICs (Fig. 2(c) and (d)), while their antibacterial activity resulted in the eradication of S. aureus within 7 days. These initial results showed that using BP HCl or BP base improved the antibacterial activity of GS and VC against S. aureus when eluted from UHMWPE.
Bupivacaine was chosen as the non-antibiotic compound because it is often administered intraarticularly for the management of post-operative pain after total joint replacement surgery. This molecule offers also a secondary benefit related to its availability both in a salt form (i.e., BP HCl) and in a free base form; these two drug forms have very different physicochemical properties, for example, in terms of solubility (BP HCl is hydrophilic versus BP base is hydrophobic) and melting point (the melting point, Tm, is greater than 200 °C for BP HCl versus ∼108 °C for BP base). We leveraged these differences to manufacture UHMWPE loaded with a small amount of antibiotic and supplemented with BP by using different manufacturing approaches (Fig. 1). In the first approach, powder blending and molding of the mixed blend was used, where the molding temperature is below the melting temperature of BP HCl, preventing its phase transition during the compression molding cycle (for 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl) and above the melting temperature of BP base enabling its melting and recrystallization during consolidation (for 0.5% GS + 9.5% BP base). For BP base, it was also possible to exploit its lower melting point to promote doping of the drug into UHMWPE by diffusion (for BP doped 1% VC).37
Compositions with BP HCl, which were obtained by powder blending and molding without any phase transition of the drug, had an evident phase-separated morphology, in which drug domains are clearly visible on the micrometer scale at the grain boundaries of the UHMWPE matrix (Fig. 3(b), (e) and 4(b), (e)). This morphology was largely observed previously for antibiotics,13,44 analgesics,53,54 and NSAID in UHMWPE,55 and also for conductive composites of UHMWPE with carbon nanotubes or graphite.56,57 The 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl displayed an interconnected morphology of the drug domains at the grain boundaries (Fig. 3(b) and (e)). This morphology was very similar to that of 5% BP HCl loaded UHMWPE as observed by optical microscopy in a previous work.54 The similarity of these morphologies is in agreement with the composition of these two dual drug loaded UHMWPEs, where the main component in weight percentage is BP HCl. On the other hand, the use of BP base led to a morphology in which the phase-separation on the micrometer scale was minimal and could be ascribed to the small amount of antibiotic domains present (Fig. 3(c), (f) and 4(c), (f)). At the higher magnification attained by SEM, the prismatic-like particles characteristic of VC and the sphere particles characteristic of GS were less in number and more isolated compared to the 5% VC and 5% GS, and they were surrounded by nanometer fibers of BP base. The interconnection of the drug domains in blends with BP HCl presumably was the main factor leading to higher cumulative release and higher total fractional release for both the antibiotic and the analgesic drugs (Fig. 5) through improved percolation. The highly interconnected drug domains create an access to water and solvents to penetrate inside the material in these otherwise fully consolidated UHMWPE blends and initiate drug dissolution. There was thus a favorable path for both BP HCl and the antibiotic for increased elution out of the polymeric matrix. The antibiotic release was low for the less phase-separated blends based on BP base due to the relative isolation of the drug domains in this more homogenous matrix (Fig. 3(c) and (f)). For a more quantitative correlation between the morphology of the materials and the elution kinetics of the drugs, the optical microscopy here used may need to be coupled to volumetric techniques, like micro-CT,56 which could be useful to assess the interconnection of the domains in the material volume and to obtain the volume fractions of the drug domains.
The antibacterial and drug elution studies were performed on small samples, with a total surface area which is lower than or equal to 4%, respectively, of that of an idealized full knee implant with a surface area of 100 cm2. Spatially limiting the drug-eluting material on specified surfaces of the implant component may be possible to obtain antibacterial efficacy, but the manufacturing of a full-scale implant may be more practical. Thus, pharmacokinetic modeling was used to predict the intraarticular concentration in the knee joint resulting from drug elution from the full-sized tibial knee insert using a one-compartment model with a clearance based on the half-life of the drugs. This latter parameter is often unknown, but for the case of intraarticular administration of VC it was found to be between 3 to 6 h.41,58 The higher release of the antibiotics from the percolated structure obtained using BP HCl enabled predicted intraarticular concentrations of the antibiotics above their respective MIC for the full duration of the study whereas the elution at this level was sustained for about 2 weeks from the more homogenous blends with BP base (Fig. 5(e)). In this study, the loading of the antibiotic was intentionally limited to a small amount for the aim of exploring combination of drugs showing synergistic antibacterial activity. If a longer duration of the sustained antibiotic release is desired, the antibiotic loading could be increased.
While the primary role of the addition of BP in these blends is to enhance the antibacterial activity of the incorporated antibiotic, we investigated the possible analgesic effect of BP elution from these blends. The exposure of the knee compartment to BP eluted from the idealized knee implant made of the antibiotic/analgesic blends, evaluated as the area under the curve (AUC) of the predicted concentration profiles (Fig. 5(f)), were substantially similar to that of a bolus injection for 14 days, suggesting that a similar analgesic effect is expected. The maximum intraarticular concentration was lower in the case of the drug delivery device, which may be an advantage for decreasing acute local and systemic toxicity (Table S4, ESI†).
For the clinical use of these antibacterial UHMWPEs as a full-size joint component, state-of-the-art mechanical properties are required as they are correlated with the successful outcome and longevity of the implant material. Tensile strength, ductility, toughness, and fatigue resistance are usually the parameters evaluated for determining the mechanical and structural behavior for these components which undergo loading under complex modes, including cyclic compression, tension and shear in vivo. We first performed tensile testing as a screening analysis for multiple formulations. Clinically available materials generally have an ultimate tensile strength (UTS) above 30 MPa, an elongation at break (EAB) greater than 250%;59 thus, we based our screening using these values as thresholds. A significant difference in the tensile performance was found depending on the form of BP used: compositions with the salt form had significantly lower properties than compositions with the free base form (Fig. S2, ESI†). For compositions with BP HCl, limiting the total drug loading to 5 wt% was needed to achieve the threshold values for UTS and EAB, while for blending or diffusion doping of the BP base there was no decrease based on the drug loading, for the studied loading range. This difference in the mechanical properties is strictly correlated to the morphological differences of the composite material: standard blending techniques led to phase-separation clearly observed by optical microscopy and scanning electron microscopy. This case is characterized by poor interfacial adhesion between the polymeric matrix and the drug domains, which are brittle and may be considered as stress concentrators. By modifying the drug incorporation methods and by leveraging the drug properties, we were able to significantly reduce the phase-separation previously observed on the micrometer scale. This led to outstanding mechanical properties with high strength and high ductility for the compositions with BP base, as measured by tensile testing. High ductility is also correlated to high toughness;60 thus, we performed Izod impact strength testing to screen for toughness.
Limiting the thickness of the drug layer of compositions with BP HCl to less than 40% was a viable strategy to achieve values of Izod impact strength equal or higher than 100 kJ m−2, which is well above the minimum used for clinical grades materials;61 however, for compositions with BP base, the layering approach was not required, as the full thickness samples had impact strength which was unchanged to that of virgin UHMWPE (Table 2 and Fig. 6). While the compositions with BP HCl (i.e., 1% VC + 4% BP HCl and 0.5% GS + 4.5% BP HCl) could be used as part of a layered design, the compositions with BP base (i.e., BP doped 1% VC and 0.5% GS + 9.5% BP base) can easily be used for a full-size joint implant. Other methods used to increase the toughness of UHMWPE such as high temperature melting62 are not a viable strategy for the drugs used in this work as the high temperature required for this post-treatment (i.e., 280–320 °C) would result in significant degradation of the incorporated drugs.44
These enhanced mechanical properties obtained for blends with BP base were correlated with the morphology of the materials by performing fracture analysis by SEM: while the phase-separated blends showed signs of mixed-mode fracture under impact with ductile features of UHMWPE mixed with brittle features coming from the drug and drug-to-polymer interface (Fig. 7(b) and (c)), for non-phase separated materials, a similar fracture morphology to that of virgin UHMWPE was observed with the addition of nanometer drug fibers homogenously dispersed in the polymeric matrix (Fig. 7(d) and (e)). These nanometer-sized fibers were confirmed to be BP base domains using SEM/EDX (Fig. S3c, ESI†), and the cause of this morphology was ruled out to be deformation by exhaustive drug extraction (Fig. S3b, ESI†). We demonstrated that it was also possible to control the distribution of the drug nanometer needle-like fibers by manufacturing. Powder blending of BP base (i.e., 0.5% GS + 9.5% BP base) allowed to obtain a material with homogenous dispersion of the drug throughout the material thickness, as evident from the fracture surfaces (Fig. 7(e)), while diffusion doping of BP base (i.e., BP doped 1% VC) resulted in a material with thickness-dependent drug distribution as confirmed by FTIR analysis (Fig. 1(b) and (c)) and by the observation of nanometer-sized needle-like BP fibers close to the doping surfaces of the sample (Fig. 7(d)). The difference in the morphology of the powder blends of BP HCl and BP base was presumably responsible also for the excellent fatigue toughness properties of 0.5% GS + 9.5% BP base loaded UHMWPE. Composites of UHMWPE with poor interfacial strength between the polymeric matrix and the reinforcing agent, such as in the case of carbon fibers, have displayed low fatigue crack propagation resistance, due to discontinuities in the matrix acting as stress concentrators triggering crack initiation.45 In our case, the decrease in the stress intensity factor at crack inception for 0.5% GS + 9.5% BP base was only less than 20% compared to virgin UHMWPE (Table 2) and its value was well above the values displayed by crosslinked UHMWPEs (e.g., ∼0.6–0.8 MPa m1/2,60). The decrease is likely caused by the domains of GS, which act as interrupters of the polymeric matrix (Fig. 8).
This study represents a significant advancement in the field of antibacterial implant materials as the proposed approach resulted in materials that meet the state-of-the-art properties required for UHMWPE, combined with a novel formulation for stronger antibacterial efficacy for treatment of infections. In addition, this formulation may be used as an additional pain management tool after surgery. Further work will involve radiation sterilization and crosslinking for achieving optimal wear properties, and antioxidant stabilization. The structure of the drug and the drug–polymer interactions could also be further investigated by means of solid-state NMR spectroscopy, as it is known that different forms of BP (e.g., salt versus free base forms) exist as polymorphs, hence they have very different crystal arrangement, which can influence relevant properties, such as solubility, bioavailability and efficacy, and safety profile.63,64 Of interest, will be a mechanistic interpretation of the antimicrobial synergy for combination of drugs, that could help in discovering new synergistic drug combinations and could assist in a successful repositioning of currently available drugs for improving the treatment of bacterial infections. For a future clinical translation, animal testing will be required to assess the safety and efficacy of these drug delivery devices under in vivo conditions.
Conclusions
This study reports a novel approach for the obtainment of antibacterial implant materials based on UHMWPE, which is commonly employed as the bearing component in total joint replacement. Combinations of non-antibiotic and antibiotic drugs were incorporated into UHMWPE following various manufacturing methods. The drugs elution from the polymeric matrix and their antibacterial activity were found to not only be synergistic and effective in eradicating S. aureus, but also to provide concentration of analgesics which are expected to be effective in pain relieving after the surgery. The drugs’ physicochemical properties were leveraged during manufacturing to obtain tailored morphologies which resulted in high mechanical properties. High strength, ductility, toughness, and fatigue resistance, which are necessary for the long-term success of implant materials, were achieved for the optimal compositions. These findings combined are promising for the development of novel antibacterial implant materials, paving the way to the use of drug combinations including the repositioning of non-antibiotic drugs for an effective antibacterial effect.Author contributions
Nicoletta Inverardi: conceptualization, data curation, formal analysis, investigation, methodology, supervision, visualization, writing – original draft, writing – review & editing. Maria F. Serafim: data curation, formal analysis, investigation, methodology, writing – review & editing. Anthony Marzouca: formal analysis, investigation, writing – review & editing. Keita Fujino: investigation, writing – review & editing. Matheus Ferreira: data curation, investigation, methodology, writing – review & editing. Mehmet D. Asik: data curation, investigation, methodology, writing – review & editing. Amita Sekar: conceptualization, methodology, supervision, writing – review & editing. Orhun K. Muratoglu: conceptualization, resources, supervision, writing – review & editing. Ebru Oral: conceptualization, data curation, methodology, funding acquisition, project administration, resources, supervision, writing – original draft, writing – review & editing.Data availability
Data are available upon request from the corresponding author. The data that support the findings of this study are partially included in the ESI† and will be made openly available after publication on the Zenodo open data repository at the DOI: https://doi.org/10.5281/zenodo.14606659.Conflicts of interest
MDA: Stock or Stock Options: Corlamlabs. OKM: Royalties – Corin, Mako, Iconacy, Renovis, Arthrex, ConforMIS, Meril Healthcare, Exactech, Cambridge Polymer Group; Stake/Equity-Cambridge Polymer Group, Orthopedic Technology Group, Alchimist. EO: Royalties-Corin, Iconacy, Renovis, Arthrex, ConforMIS, Meril Healthcare, Exactech; Paid consultant – WL Gore & Assoc; Editorial Board – JBMR; Officer/Committee-SFB, ISTA. None of these are in direct conflict with the study.Acknowledgements
This work was supported by the National Institutes of Health grant R01AR077023 (EO). This work was performed in part at the Harvard University Center for Nanoscale Systems (CNS); a member of the National Nanotechnology Coordinated Infrastructure Network (NNCI), which is supported by the National Science Foundation under NSF award no. ECCS-2025158. The authors would like to thank The Coastal Milling Works Group (Medford, MA) for help with CNC machining. The authors would like to thank Parker Jones and Jean Yuh for helping with the bacterial culture.References
- A. J. R. Registry, American Joint Replacement Registry, Rosemont, IL, 2022 Search PubMed.
- F. Canovas and L. Dagneaux, Quality of life after total knee arthroplasty, Orthop. Traumatol.: Surg. Res., 2018, 104, S41–S46 CAS.
- S. M. Heo, I. Harris, J. Naylor and A. M. Lewin, Complications to 6
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) months following total hip or knee arthroplasty: observations from an Australian clinical outcomes registry, BMC Musculoskeletal Disord., 2020, 21, 602 CrossRef PubMed.
months following total hip or knee arthroplasty: observations from an Australian clinical outcomes registry, BMC Musculoskeletal Disord., 2020, 21, 602 CrossRef PubMed. - J. P. M. Masters, N. A. Smith, P. Foguet, M. Reed, H. Parsons and A. P. Sprowson, A systematic review of the evidence for single stage and two stage revision of infected knee replacement, BMC Musculoskeletal Disord., 2013, 14, 222 CrossRef.
- S. Kurtz, E. Lau, H. Watson, J. Schmier and J. Parvizi, Economic burden of periprosthetic joint infection in the United States, J. Arthroplasty, 2012, 27, 61–65 CrossRef.
- A. J. Tande and R. Patel, Prosthetic joint infection, Clin. Microbiol. Rev., 2014, 27, 302–345 CrossRef PubMed.
- M. A. Roof, V. K. Aggarwal and R. Schwarzkopf, The Economics of Revision Arthroplasty for Periprosthetic Joint Infection, Arthroplast. Today, 2023, 23, 101213 CrossRef.
- A. Visperas, D. Santana, A. K. Klika, C. A. Higuera-Rueda and N. S. Piuzzi, Current treatments for biofilm-associated periprosthetic joint infection and new potential strategies, J. Orthop. Res., 2022, 40, 1477–1491 CrossRef CAS PubMed.
- Y. S. Lee and A. F. Chen, Two-stage reimplantation in infected total knee arthroplasty, Knee Surg. Relat. Res., 2018, 30, 107–114 CrossRef PubMed.
- R. Sousa, A. Carvalho, D. Soares and M. A. Abreu, Interval between two-stage exchanges: what is optimal and how do you know?, Arthroplasty, 2023, 5, 33 CrossRef.
- G. Cierny III and D. DiPasquale, Periprosthetic total joint infections: staging, treatment, and outcomes, Clin. Orthop. Relat. Res., 2002, 403, 23–28 CrossRef.
- T. Trouillez, P. A. Faure, P. Martinot, H. Migaud, E. Senneville, G. Pasquier, J. Dartus and S. Putman, Above–the–knee amputation versus knee arthrodesis for revision of infected total knee arthroplasty: Recurrent infection rates and functional outcomes of 43 patients at a mean follow-up of 6.7 years, Orthop. Traumatol.: Surg. Res., 2021, 107, 102914 Search PubMed.
- V. J. Suhardi, D. A. Bichara, S. J. J. Kwok, A. A. Freiberg, H. Rubash, H. Malchau, S. H. Yun, O. K. Muratoglu and E. Oral, A Fully Functional Drug-Eluting Joint Implant, Nat. Biomed. Eng., 2017, 1, 0080 CrossRef CAS PubMed.
- D. Gil, A. Atici, R. Connolly, S. Hugard, S. Shuaev, K. Wannomae, E. Oral and O. Muratoglu, Addressing prosthetic joint infections via gentamicin-eluting UHMWPE spacer, Bone Joint J., 2020, 102, 151–157 CrossRef.
- P. Bracco, A. Bellare, A. Bistolfi and S. Affatato, Ultra-High Molecular Weight Polyethylene: Influence of the Chemical, Physical and Mechanical Properties on the Wear Behavior. A Review, Materials, 2017, 10, 791 CrossRef.
- P. Bracco and E. Oral, Vitamin E-stabilized UHMWPE for Total Joint Implants: A Review, Clin. Orthop. Relat. Res., 2011, 469, 2286–2293 CrossRef.
- S. P. Ryan, J. B. Stambough, J. I. Huddleston and B. R. Levine, Highlights of the 2023 American Joint Replacement Registry Annual Report, Arthroplast. Today, 2024, 26, 101325 CrossRef.
- C. R. MacNair, S. T. Rutherford and M.-W. Tan, Alternative therapeutic strategies to treat antibiotic-resistant pathogens, Nat. Rev. Microbiol., 2024, 22, 262–275 CrossRef CAS.
- G. Li, Z. Lai and A. Shan, Advances of Antimicrobial Peptide-Based Biomaterials for the Treatment of Bacterial Infections, Adv. Sci., 2023, 10, 2206602 CrossRef CAS.
- H. Özçelik, N. E. Vrana, A. Gudima, V. Riabov, A. Gratchev, Y. Haikel, M.-H. Metz-Boutigue, A. Carradò, J. Faerber, T. Roland, H. Klüter, J. Kzhyshkowska, P. Schaaf and P. Lavalle, Harnessing the Multifunctionality in Nature: A Bioactive Agent Release System with Self-Antimicrobial and Immunomodulatory Properties, Adv. Healthcare Mater., 2015, 4, 2026–2036 CrossRef.
- A. Mutschler, L. Tallet, M. Rabineau, C. Dollinger, M.-H. Metz-Boutigue, F. Schneider, B. Senger, N. E. Vrana, P. Schaaf and P. Lavalle, Unexpected Bactericidal Activity of Poly(arginine)/Hyaluronan Nanolayered Coatings, Chem. Mater., 2016, 28, 8700–8709 CrossRef CAS.
- L. Tallet, V. Gribova, L. Ploux, N. E. Vrana and P. Lavalle, New Smart Antimicrobial Hydrogels, Nanomaterials, and Coatings: Earlier Action, More Specific, Better Dosing?, Adv. Healthcare Mater., 2021, 10, 2001199 CrossRef CAS.
- F.-Y. Wang, Y. Ren, R.-T. Lan, W.-Q. Fu, Z.-J. Chen, S. Huang, R. M. Gul, J. Wang, J.-Z. Xu and Z.-M. Li, Controlled bacteriostasis of tea polyphenol loaded ultrahigh molecular weight polyethylene with high crosslink density and oxidation resistance for total joint replacement, Mater. Sci. Eng., C, 2021, 124, 112040 CrossRef CAS PubMed.
- Y. Ren, J.-C. Lv, S.-P. Zhao, H. Fu, Z.-B. Sun, L. Li, S. Huang, J.-Z. Xu and Z.-M. Li, Construction of a cross-bedded tea polyphenol delivery structure in ultrahigh molecular weight polyethylene against bacteria for joint replacement, Compos. Sci. Technol., 2024, 254, 110689 CrossRef CAS.
- G. Graziani, D. Ghezzi, F. Nudelman, E. Sassoni, F. Laidlaw, M. Cappelletti, M. Boi, G. Borciani, S. Milita, M. Bianchi, N. Baldini and G. Falini, A natural biogenic fluorapatite as a new biomaterial for orthopedics and dentistry: antibacterial activity of lingula seashell and its use for nanostructured biomimetic coatings, J. Mater. Chem. B, 2024, 12, 2083–2098 RSC.
- V. S. Foletto, T. F. da Rosa, M. B. Serafin, A. Bottega and R. Hörner, Repositioning of non-antibiotic drugs as an alternative to microbial resistance: a systematic review, Int. J. Antimicrob. Agents, 2021, 58, 106380 CrossRef CAS.
- J.-P. Jourdan, R. Bureau, C. Rochais and P. Dallemagne, J. Pharm. Pharmacol., 2020, 72, 1145–1151 CrossRef CAS.
- K. Pelz, M. Wiedmann-Al-Ahmad, C. Bogdan and J.-E. Otten, Analysis of the antimicrobial activity of local anaesthetics used for dental analgesia, J. Med. Microbiol., 2008, 57, 88–94 CrossRef CAS.
- D. Machado, D. Pires, J. Perdigão, I. Couto, I. Portugal, M. Martins, L. Amaral, E. Anes and M. Viveiros, Ion Channel Blockers as Antimicrobial Agents, Efflux Inhibitors, and Enhancers of Macrophage Killing Activity against Drug Resistant Mycobacterium tuberculosis, PLoS One, 2016, 11, e0149326 CrossRef PubMed.
- Z. Dawson, S. S. Stanton, S. Roy, R. Farjo, H. A. Aslesen, B. R. Hallstrom and M. C. Bicket, Opioid Consumption After Discharge From Total Knee and Hip Arthroplasty: A Systematic Review and Meta-Analysis, J. Arthroplasty, 2024, 39, 2130–2136 CrossRef PubMed.
- Q.-B. Sun, S.-D. Liu, Q.-J. Meng, H.-Z. Qu and Z. Zhang, Single administration of intra-articular bupivacaine in arthroscopic knee surgery: a systematic review and meta-analysis, BMC Musculoskeletal Disord., 2015, 16, 21 CrossRef PubMed.
- E. Marret, M. Gentili, M. P. Bonnet and F. Bonnet, Intra-articular ropivacaine 0.75% and bupivacaine 0.50% for analgesia after arthroscopic knee surgery: a randomized prospective study, Arthroscopy, 2005, 21, 313–316 CrossRef PubMed.
- M. Yayac, W. T. Li, A. C. Ong, P. M. Courtney and A. Saxena, The Efficacy of Liposomal Bupivacaine Over Traditional Local Anesthetics in Periarticular Infiltration and Regional Anesthesia During Total Knee Arthroplasty: A Systematic Review and Meta-Analysis, J. Arthroplasty, 2019, 34, 2166–2183 CrossRef PubMed.
- D. Gil, K. Daffinee, R. Friedman, B. Bhushan, O. K. Muratoglu, K. LaPlante and E. Oral, Synergistic antibacterial effects of analgesics and antibiotics against Staphylococcus aureus, Diagn. Microbiol. Infect. Dis., 2020, 96, 114967 CrossRef CAS.
- D. Gil, S. Grindy, O. Muratoglu, H. Bedair and E. Oral, Antimicrobial effect of anesthetic-eluting ultra-high molecular weight polyethylene for post-arthroplasty antibacterial prophylaxis, J. Orthop. Res., 2019, 37, 981–990 CrossRef CAS PubMed.
- A. Sekar, D. Gil, P. Tierney, M. McCanne, V. Daesety, D. Trendafilova, O. K. Muratoglu and E. Oral, Synergistic use of anti-inflammatory ketorolac and gentamicin to target staphylococcal biofilms, J. Transl. Med., 2024, 22, 102 CrossRef CAS PubMed.
- N. Inverardi, S. Lekkala, M. F. Serafim, A. Sekar, K. K. Wannomae, B. Micheli, H. Bedair, O. K. Muratoglu and E. Oral, Diffusion doping of analgesics into UHMWPE for prophylactic pain management, J. Mater. Chem. B, 2024, 12, 10332–10345 RSC.
- J. Gubernator, Z. Drulis-Kawa and A. Kozubek, A simply and sensitive fluorometric method for determination of gentamicin in liposomal suspensions, Int. J. Pharm., 2006, 327, 104–109 CrossRef CAS PubMed.
- P. L. Ritger and N. A. Peppas, A simple equation for description of solute release I. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs, J. Controlled Release, 1987, 5, 23–36 CrossRef CAS.
- S. L. Barry, S. A. Martinez, N. M. Davies, C. M. Remsberg, C. L. Sayre and A. Bachelez, Synovial fluid bupivacaine concentrations following single intra-articular injection in normal and osteoarthritic canine stifles, J. Vet. Pharmacol. Ther., 2015, 38, 97–100 CrossRef CAS.
- M. Bue, M. B. Thomassen, O. H. Larsen, A. R. Jørgensen, M. Stilling, K. Søballe and P. Hanberg, Local Vancomycin Concentrations after Intra-articular Injection into the Knee Joint: An Experimental Porcine Study, J. Knee Surg., 2021, 34, 936–940 CrossRef PubMed.
- C. D. Doern, When Does 2 Plus 2 Equal 5? A Review of Antimicrobial Synergy Testing, J. Clin. Microbiol., 2014, 52, 4124–4128 CrossRef.
- A. Sekar, S. Lekkala and E. Oral, A Novel Method to Determine the Longitudinal Antibacterial Activity of Drug-Eluting Materials, J. Visualized Exp., 2023, 193, e64641 Search PubMed.
- S. Lekkala, N. Inverardi, J. Yuh, K. K. Wannomae, P. Tierney, A. Sekar, O. K. Muratoglu and E. Oral, Antibiotic-Loaded Ultrahigh Molecular Weight Polyethylenes, Macromol. Biosci., 2024, 24, 2300389 CrossRef CAS.
- G. M. Connelly, C. M. Rimnac, T. M. Wright, R. W. Hertzberg and J. A. Manson, Fatigue crack propagation behavior of ultrahigh molecular weight polyethylene, J. Orthop. Res., 1984, 2, 119–125 Search PubMed.
- F. Ansari, B. Gludovatz, A. Kozak, R. O. Ritchie and L. A. Pruitt, Notch fatigue of ultrahigh molecular weight polyethylene (UHMWPE) used in total joint replacements, J. Mech. Behav. Biomed. Mater., 2016, 60, 267–279 CrossRef CAS PubMed.
- C. J. L. Murray, K. S. Ikuta, F. Sharara, L. Swetschinski, G. Robles Aguilar, A. Gray, C. Han, C. Bisignano, P. Rao, E. Wool, S. C. Johnson, A. J. Browne, M. G. Chipeta, F. Fell, S. Hackett, G. Haines-Woodhouse, B. H. Kashef Hamadani, E. A. P. Kumaran, B. McManigal, S. Achalapong, R. Agarwal, S. Akech, S. Albertson, J. Amuasi, J. Andrews, A. Aravkin, E. Ashley, F.-X. Babin, F. Bailey, S. Baker, B. Basnyat, A. Bekker, R. Bender, J. A. Berkley, A. Bethou, J. Bielicki, S. Boonkasidecha, J. Bukosia, C. Carvalheiro, C. Castañeda-Orjuela, V. Chansamouth, S. Chaurasia, S. Chiurchiù, F. Chowdhury, R. Clotaire Donatien, A. J. Cook, B. Cooper, T. R. Cressey, E. Criollo-Mora, M. Cunningham, S. Darboe, N. P. J. Day, M. De Luca, K. Dokova, A. Dramowski, S. J. Dunachie, T. Duong Bich, T. Eckmanns, D. Eibach, A. Emami, N. Feasey, N. Fisher-Pearson, K. Forrest, C. Garcia, D. Garrett, P. Gastmeier, A. Z. Giref, R. C. Greer, V. Gupta, S. Haller, A. Haselbeck, S. I. Hay, M. Holm, S. Hopkins, Y. Hsia, K. C. Iregbu, J. Jacobs, D. Jarovsky, F. Javanmardi, A. W. J. Jenney, M. Khorana, S. Khusuwan, N. Kissoon, E. Kobeissi, T. Kostyanev, F. Krapp, R. Krumkamp, A. Kumar, H. H. Kyu, C. Lim, K. Lim, D. Limmathurotsakul, M. J. Loftus, M. Lunn, J. Ma, A. Manoharan, F. Marks, J. May, M. Mayxay, N. Mturi, T. Munera-Huertas, P. Musicha, L. A. Musila, M. M. Mussi-Pinhata, R. N. Naidu, T. Nakamura, R. Nanavati, S. Nangia, P. Newton, C. Ngoun, A. Novotney, D. Nwakanma, C. W. Obiero, T. J. Ochoa, A. Olivas-Martinez, P. Olliaro, E. Ooko, E. Ortiz-Brizuela, P. Ounchanum, G. D. Pak, J. L. Paredes, A. Y. Peleg, C. Perrone, T. Phe, K. Phommasone, N. Plakkal, A. Ponce-de-Leon, M. Raad, T. Ramdin, S. Rattanavong, A. Riddell, T. Roberts, J. V. Robotham, A. Roca, V. D. Rosenthal, K. E. Rudd, N. Russell, H. S. Sader, W. Saengchan, J. Schnall, J. A. G. Scott, S. Seekaew, M. Sharland, M. Shivamallappa, J. Sifuentes-Osornio, A. J. Simpson, N. Steenkeste, A. J. Stewardson, T. Stoeva, N. Tasak, A. Thaiprakong, G. Thwaites, C. Tigoi, C. Turner, P. Turner, H. R. van Doorn, S. Velaphi, A. Vongpradith, M. Vongsouvath, H. Vu, T. Walsh, J. L. Walson, S. Waner, T. Wangrangsimakul, P. Wannapinij, T. Wozniak, T. E. M. W. Young Sharma, K. C. Yu, P. Zheng, B. Sartorius, A. D. Lopez, A. Stergachis, C. Moore, C. Dolecek and M. Naghavi, Global burden of bacterial antimicrobial resistance in 2019: a systematic analysis, Lancet, 2022, 399, 629–655 CrossRef CAS.
- T. T. Ashburn and K. B. Thor, Drug repositioning: identifying and developing new uses for existing drugs, Nat. Rev. Drug Discovery, 2004, 3, 673–683 CrossRef CAS PubMed.
- M. Tyers and G. D. Wright, Drug combinations: a strategy to extend the life of antibiotics in the 21st century, Nat. Rev. Microbiol., 2019, 17, 141–155 CrossRef CAS PubMed.
- C. G. Begley, M. Ashton, J. Baell, M. Bettess, M. P. Brown, B. Carter, W. N. Charman, C. Davis, S. Fisher, I. Frazer, A. Gautam, M. P. Jennings, P. Kearney, E. Keeffe, D. Kelly, A. F. Lopez, M. McGuckin, M. W. Parker, C. Rayner, B. Roberts, J. S. Rush and M. Sullivan, Drug repurposing: Misconceptions, challenges, and opportunities for academic researchers, Sci. Transl. Med., 2021, 13, eabd5524 CrossRef CAS.
- D. Campoccia, L. Montanaro and C. R. Arciola, The significance of infection related to orthopedic devices and issues of antibiotic resistance, Biomaterials, 2006, 27, 2331–2339 CrossRef CAS PubMed.
- B. Li and T. J. Webster, Bacteria antibiotic resistance: New challenges and opportunities for implant-associated orthopedic infections, J. Orthop. Res., 2018, 36, 22–32 CrossRef.
- S. C. Grindy, D. Gil, J. V. Suhardi, O. K. Muratoglu, H. Bedair and E. Oral, Delivery of bupivacaine from UHMWPE and its implications for managing pain after joint arthroplasty, Acta Biomater., 2019, 93, 63–73 CrossRef CAS.
- S. Lekkala, N. Inverardi, S. C. Grindy, S. Hugard, O. K. Muratoglu and E. Oral, Irradiation Behavior of Analgesic and Nonsteroidal Anti-Inflammatory Drug-Loaded UHMWPE for Joint Replacement, Biomacromolecules, 2024, 25, 2312–2322 CrossRef CAS.
- D. Gil, S. Hugard, S. Grindy, N. Borodinov, O. S. Ovchinnikova, O. K. Muratoglu, H. Bedair and E. Oral, Structural and antibacterial properties of NSAID-loaded ultra-high molecular weight polyethylene, Materialia, 2020, 12, 100662 CrossRef CAS.
- H. J. Favreau, K. I. Miroshnichenko, P. C. Solberg, I. I. Tsukrov and D. W. Van Citters, Shear enhancement of mechanical and microstructural properties of synthetic graphite and ultra-high molecular weight polyethylene carbon composites, J. Appl. Polym. Sci., 2022, 139, 52175 Search PubMed.
- P.-G. Ren, Y.-Y. Di, Q. Zhang, L. Li, H. Pang and Z.-M. Li, Composites of Ultrahigh-Molecular-Weight Polyethylene with Graphene Sheets and/or MWCNTs with Segregated Network Structure: Preparation and Properties, Macromol. Mater. Eng., 2012, 297, 437–443 CrossRef CAS.
- M. E. Roy, M. P. Peppers, L. A. Whiteside and R. M. LaZear, Vancomycin concentration in synovial fluid: direct injection into the knee vs. intravenous infusion, J. Arthroplasty, 2014, 29, 564–568 CrossRef.
- J. P. Collier, B. H. Currier, F. E. Kennedy, J. H. Currier, G. S. Timmins, S. K. Jackson and R. L. Brewer, Comparison of Cross-Linked Polyethylene Materials for Orthopaedic Applications, Clin. Orthop. Relat. Res., 2003, 414, 289–304 CrossRef.
- B. Doshi, J. S. Ward, E. Oral and O. K. Muratoglu, Fatigue toughness of irradiated vitamin E/UHMWPE blends, J. Orthop. Res., 2016, 34, 1514–1520 CrossRef CAS PubMed.
- E. Oral, B. N. Doshi, K. Fung, C. O'Brien, K. K. Wannomae and O. K. Muratoglu, Chemically Cross-Linked UHMWPE With Superior Toughness, J. Orthop. Res., 2019, 37, 2182–2188 CrossRef PubMed.
- J. Fu, B. W. Ghali, A. J. Lozynsky, E. Oral and O. K. Muratoglu, Ultra high molecular weight polyethylene with improved plasticity and toughness by high temperature melting, Polymer, 2010, 51, 2721–2731 CrossRef CAS.
- J. W. Lubach, B. E. Padden, S. L. Winslow, J. S. Salsbury, D. B. Masters, E. M. Topp and E. J. Munson, Solid-state NMR studies of pharmaceutical solids in polymer matrices, Anal. Bioanal. Chem., 2004, 378, 1504–1510 CrossRef CAS.
- H. Hamaed, J. M. Pawlowski, B. F. T. Cooper, R. Fu, S. H. Eichhorn and R. W. Schurko, Application of Solid-State 35Cl NMR to the Structural Characterization of Hydrochloride Pharmaceuticals and their Polymorphs, J. Am. Chem. Soc., 2008, 130, 11056–11065 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4tb02672a |
| This journal is © The Royal Society of Chemistry 2025 |








