Temperature and light dual-responsive hydrogels for anti-inflammation and wound repair monitoring†
Ji
Jiang‡
a,
Yuan
Tian‡
a,
Xiaoyang
Wu
a,
Mingze
Zeng
a,
Chengheng
Wu
 ab,
Dan
Wei
a,
Hongrong
Luo
a,
Jing
Sun
a,
Jie
Ding
*a and
Hongsong
Fan
ab,
Dan
Wei
a,
Hongrong
Luo
a,
Jing
Sun
a,
Jie
Ding
*a and
Hongsong
Fan
 a
a
aNational Engineering Research Center for Biomaterials, College of Biomedical Engineering, Sichuan University, Chengdu, 610065, P. R. China. E-mail: jie_ding@scu.edu.cn
bInstitute of Regulatory Science for Medical Devices, Sichuan University, Chengdu 610065, P. R. China
First published on 25th January 2025
Abstract
Wound healing is a complex and dynamic biological process that requires meticulous management to ensure optimal outcomes. Traditional wound dressings, such as gauze and bandages, although commonly used, often fall short in their frequent need for replacement, lack of real-time monitoring and absence of anti-inflammatory and antibacterial properties, which can lead to increased risk of infection and delayed healing. Here, we address these limitations by introducing an innovative hydrogel dressing, named PHDNN6, to combine wireless Bluetooth temperature monitoring and light-triggered nitric oxide (NO) release to enhance wound healing and management. The PHDNN6 hydrogel is based on a poly(N-isopropylacrylamide) (PNIPAM) matrix, integrated with methacrylated and dopamine-grafted hyaluronic acid (HA–MA–DA), which allows the dressing to be highly responsive to changes in wound temperature, enabling continuous and real-time monitoring of the wound microenvironment wirelessly. Besides, PHDNN6 is embedded with photothermal polydopamine nanoparticles (PDA NPs) that are loaded with a NO donor, N,N′-di-sec-butyl-N,N′-dinitroso-1,4-phenylenediamine (BNN6). When exposed to near-infrared (NIR) laser irradiation, these PDA@BNN6 nanoparticles release NO to provide potent antibacterial and anti-inflammatory effects. The integration of continuous wireless temperature monitoring with NO release within a single hydrogel dressing represents a significant advancement in clinical wound care. This dual-functional platform not only provides real-time diagnostic capabilities but also offers therapeutic interventions to manage wound infections and promote tissue regeneration. Our research highlights the potential of PHDNN6 to revolutionize wound management by offering a comprehensive solution that addresses both the diagnostic and therapeutic needs in wound healing.
1. Introduction
Wound healing is a multifaceted biological process that typically occurs in skin tissue following burns, trauma, or diabetic ulcers.1–3 Currently, conventional wound dressings like gauze and bandages are commonly used in clinical settings for wound care. However, these materials require frequent disinfection and replacement, as well as lack of real-time monitoring, and absence of anti-inflammatory and antibacterial properties, which can lead to increased risks of infection and delayed healing. In addition, extensive literature underscores the association between wound infection and local temperature elevation, with wound temperature being considered as a critical biomarker throughout the healing process.4,5 As a result, careful monitoring of wound temperature can improve understanding of the healing process and enable real-time interventions to protect against infection. Despite recent advances in the development of various wound temperature monitoring systems using touch and non-contact temperature sensors,6,7 infrared thermography,8 limitations such as low sensitivity, poor adhesion and biocompatibility issues remain. Thus, there is an urgent need for the development of an integrated diagnostic and therapeutic platform characterized by continuous, stable and sensitive temperature monitoring, coupled with superior antimicrobial properties activated by dynamic interventions, with profound implications for the field of wound healing and management.Recently, there has emerged interests in innovative temperature-responsive dressings, characterized by their high sensitivity, enhanced resolution, and unparalleled convenience, thereby facilitating intuitive and precise monitoring of temperature fluctuations at the wound interface. This trend underscores the pursuit of more efficient and user-friendly solutions for wound care management. In general, the fundamental requirements for wound dressing materials include biocompatibility, interface conformability, and the ability to treat infection, maintain moisture, and promote tissue regeneration. Besides, self-healing dressings formed by dynamic or non-covalent chemical bonds tend to have better wound closure, while also avoiding damage during use.9 Hydrogels, as polymeric materials with a three-dimensional network structure, exhibit excellent wound exudate absorption and high water retention,10,11 providing a suitable environment for wound healing. In particular, the use of intrinsic temperature-responsive hydrogel materials as temperature monitoring matrix not only enables more accurate temperature monitoring directly at the wound site, but also offers superior biocompatibility and tissue conformability compared to commercial electronic temperature sensors.12,13 As a prominent temperature-sensitive material, poly(N-isopropylacrylamide) (PNIPAM) has garnered widespread application owing to its remarkable capacity for volume reduction in chemically crosslinked hydrogels in response to rising temperatures. In addition, the phase transition temperature (VTT) of PNIPAM after crosslinking is around 32 °C, renders it an ideal candidate for temperature monitoring applications, as it closely aligns with the surface temperature of the human body.14 This unique property has made PNIPAM a versatile material in various biomedical applications. Notably, it has been explored extensively in drug delivery systems, where its temperature-sensitive behavior enables controlled release of therapeutic agents.15,16 Additionally, PNIPAM-based hydrogels have been utilized in biosensing technologies due to their responsive nature, which allows for real-time detection of biological signals.17–19 PNIPAM molecular chains can be easily adjusted by compositing with other molecules,20–22 which consequently enhances PNIPAM's comprehensive performance, encompassing mechanical robustness, adhesive capabilities, and biocompatibility, positioning it as a fitting choice for temperature-monitoring dressings in wound care applications. Hyaluronic acid (HA), a widely used biomaterial in the biomedical field, is a polysaccharide found in human connective tissues, eyeballs, and synovial fluid, among others, and exhibits excellent biocompatibility and biodegradability.23,24 Moreover, the extensive presence of hydroxyl and carboxyl groups on the HA molecular chains allows modifications such as aldehyde functionalization, methacrylation, and grafting of functional small molecules,25,26 providing favorable conditions for additional functionalization of PNIPAM hydrogels, such as adhesiveness and biocompatibility.
In addition, the modifiability and conformability of hydrogels provide a platform for varied technologies in the treatment of wound repair. As an emerging therapeutic modality, photothermal therapy (PTT), which converts light into localized heat, has shown remarkable antibacterial efficacy and attracted widespread attention in the treatment of wound infection.27,28 Many photothermal materials have been investigated for in vivo biomedical applications, encompassing inorganic constituents like gold-based nanomaterials, carbonaceous nanomaterials, and molybdenum disulfide (MoS2),29–31 alongside organic photothermal agents, which span polypyrrole (PPy), eumelanin, indocyanine green (ICG) and polydopamine (PDA).32–36 PDA-based nanoparticles (PDA NPs) stand out as compelling contenders owing to their exceptional biocompatibility, mussel-inspired adhesive properties, straightforward functionalization potential, and robust light absorption capabilities. Notably, PDA NPs boast a high surface-to-volume ratio, exceptional dispersibility, and unparalleled photothermal conversion efficiency, positioning them as favorable candidates for PTT applications.37 Meanwhile, the hydroxyl groups present in PDA NPs will engage in extensive hydrogen bonding interactions with hydrogel networks, promoting their uniform dispersion within the hydrogel network and providing additional adhesive properties to the hydrogel matrix.38 Nevertheless, it is noteworthy that employing PTT alone as an antibacterial strategy is insufficient to achieve the desired therapeutic effects; combining it with adjunctive therapies such as gas therapy, including hydrogen sulfide (H2S) carbon monoxide (CO) and nitric oxide (NO), is alternative for enhancing overall anti-bacterial efficacy.39,40 For example, hydrogel systems based on reactive oxygen species (ROS)-responsive release of H2S and CO have been reported in the literature,41,42 but ROS levels can fluctuate dramatically in wound environments, potentially leading to uncontrolled gas release and variable therapeutic outcomes. Among these, NO, the PTT-driven NO release system enables precise control over NO release through external temperature modulation using a light source, combined with temperature monitoring, allowing real-time feedback on wound conditions and dynamic adjustment of NO release. More recently, NO has garnered attention as a promising antibacterial agent, capable of combating a broad spectrum of bacteria, from Gram-positive to Gram-negative strains, by disrupting their membranes and DNA.39 Moreover, the regulation of macrophages is mostly induced by biological factors,43 which can easily cause systemic immune response or local long-term inflammation. NO is well-known for its dual role in inflammation, capable of promoting pro-inflammatory (M1) or anti-inflammatory (M2) phenotypes depending on its concentration and the microenvironment.44 This allows for a finely tuned regulation of macrophage activity, which is critical in wound healing and tissue regeneration. Besides, prior studies indicate that NO could accelerate wound repair processes by multifaceted mechanisms, including enhancing fibroblast proliferation, wound contraction, and collagen deposition.45,46 Consequently, integrating NIR laser-assisted PTT with the release of NO within a hydrogel system presents a promising approach for precise control of gas delivery at the wound site, thereby achieving antibacterial efficacy and augmenting wound repair.
Herein, in this work, as shown in Scheme 1, PNIPAM is used as a temperature-responsive hydrogel matrix and further composited with methacrylated and dopamine-grafted HA (HA–MA–DA). HA–MA–DA is expected to form stable covalent bonds with PNIPAM through methacrylate-induced vinyl crosslinking, thereby enhancing the mechanical and temperature-responsive properties of the composite hydrogel networks. In addition, the DA molecules in HA–MA–DA will provide PNIPAM with multiple hydrogen bonds, thereby imparting adhesiveness to ensure conformal contact with the wound geometry. Besides, the NO donor BNN6 is integrated with PDA NPs through π–π bonds to construct a light-responsive NO release system PDA@BNN6. Upon NIR irradiation (808 nm, 2 W cm−2), generated heat will trigger the decomposition of BNN6 molecules and lead to the release of NO. Furthermore, the PDA@BNN6 is incorporated into PNIPAM/HA–MA–DA (PH) to form a PHDNN6 composite hydrogel, and a wireless Bluetooth remote monitoring system is established to monitor wound temperature changes in real time, which can assess the degree of inflammation. The results show that the temperature-responsive PNIPAM matrix in PHDNN6 was able to monitor changes in wound temperature, while the light-responsive PDA@BNN6 NPs component could also respond to NIR heating and release NO for intracutaneous delivery, exhibiting antibacterial and anti-inflammatory effects, thereby promoting wound healing. Overall, the temperature and light dual-responsive hydrogel system with PTT and controllable NO release developed in our study promises to open new avenues for clinical wound healing treatment and dynamic wound management.
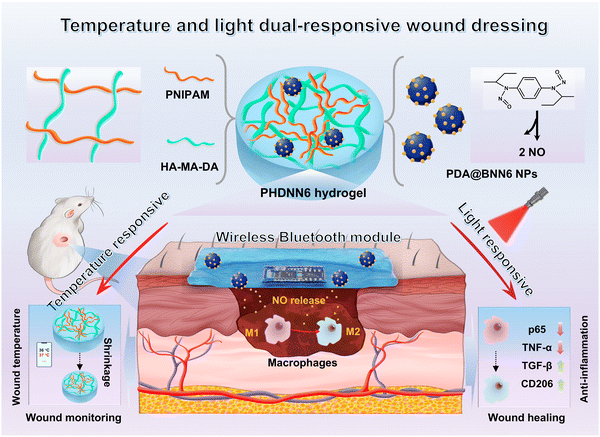 | ||
| Scheme 1 Schematic illustration of temperature and light dual-responsive hydrogel for immunomodulation and wound healing management. | ||
2. Materials and methods
2.1 Materials
Hyaluronic acid (HA, HA-TLM-20-40) were purchased from Bloomage Biotechnology Co., Ltd. Methacrylic anhydride (MA, 94%, AR), N-isopropyl acrylamide (NIPAM, 98%, AR), dopamine (DA, 98%, AR), N,N′-bis-sec-butylamino-p-phenylenediamine (BPA, 96%, AR), 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide (EDC, ≥95%, AR), N-hydroxysuccinimide (NHS, ≥98%, AR), NH4OH (28–30%, AR), N,N,N′,N′-tetramethylethylenediamine (TEMED, ≥96%, AR), ammonium persufate (APS), N,N′-methylenebis(acrylamide) (MBA, ≥95%, AR) were purchased from Aladdin Reagent Co., Ltd. Sodium nitrate (NaNO2, 99%, AR), hydrochloric acid (HCl, 36–38%, AR), were purchased from Kelon Reagent Co., Ltd. All reagents were used as supplied by the manufacturers, without additional purification, following the recommended dilutions.2.2 Synthesis of PDA@BNN6
2.3 Synthesis of PNIPAM/HA–MA–DA (PH) composite hydrogels
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 to the HA–MA monomer, were incorporated into the solution and thoroughly stirred for 30 minutes. Subsequently, 7.1 mg mL−1 of dopamine hydrochloride, also adhering to a 1.5
1 to the HA–MA monomer, were incorporated into the solution and thoroughly stirred for 30 minutes. Subsequently, 7.1 mg mL−1 of dopamine hydrochloride, also adhering to a 1.5![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 1 molar ratio with the HA–MA monomer, was introduced into the mixture. The reaction proceeded under pH conditions ranging from 4 to 5 for 10 hours. Post-reaction, the product, HA–MA–DA, underwent dialysis against distilled water for three consecutive days and was subsequently freeze-dried for final isolation.
1 molar ratio with the HA–MA monomer, was introduced into the mixture. The reaction proceeded under pH conditions ranging from 4 to 5 for 10 hours. Post-reaction, the product, HA–MA–DA, underwent dialysis against distilled water for three consecutive days and was subsequently freeze-dried for final isolation.
2.4 Synthesis of PHDNN6 composite hydrogels
The synthesis of PHDNN6 was accomplished in a one-step process, specifically, 10 mg of N-isopropyl acrylamide and 10 mg of HA–MA–DA and 0.2 mg of PDA@BNN6 were dissolved in 100 μL of deionized water at room temperature, followed by the addition of the initiator TEMED (15 μL), the cross-linker MBA (10 μL) and APS (10 μL). As for the synthesis of PNIPAM and PH hydrogels, the specific composition can be viewed in Table S1 (ESI†).2.5 Characterization
2.5.4.1 MTT. The biocompatibility of the control, PH, and PHDNN6 hydrogels was evaluated using an MTT assay. BMSCs were co-cultured with the hydrogels for 1, 3, and 5 days. Post-incubation, cells were exposed to a 10% MTT solution at 37 °C for 4 hours. Subsequently, the medium was replaced with DMSO, and the plates were gently shaken at a consistent temperature for 15 minutes. Absorbance was then measured at 490 nm using a Bio-Rad 550 microplate reader to assess cell viability.
2.5.4.2 FDA/PI staining. The viability of BMSCs cultured in α-MEM conditioned medium with various hydrogels was assessed. BMSCs at passage 3 were plated at a density of 2
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ×
× ![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 104 cells per well in a 24-well plate. The cells were incubated with conditioned medium containing control, PH, and PHDNN6 hydrogels, with standard α-MEM serving as the control medium. After 1 and 3 days of culture, cell viability was evaluated using FDA/PI double staining. Samples were rinsed with phosphate-buffered saline (PBS), incubated in the staining solution for 5 minutes, and then rinsed again with PBS. Cell morphology was also examined via FDA/PI staining and observed using an inverted fluorescence microscope (Leica DMi8A, Germany). Live cells exhibited green fluorescence, while dead cells showed red fluorescence.
104 cells per well in a 24-well plate. The cells were incubated with conditioned medium containing control, PH, and PHDNN6 hydrogels, with standard α-MEM serving as the control medium. After 1 and 3 days of culture, cell viability was evaluated using FDA/PI double staining. Samples were rinsed with phosphate-buffered saline (PBS), incubated in the staining solution for 5 minutes, and then rinsed again with PBS. Cell morphology was also examined via FDA/PI staining and observed using an inverted fluorescence microscope (Leica DMi8A, Germany). Live cells exhibited green fluorescence, while dead cells showed red fluorescence.
![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 103 for E. coli and 1
103 for E. coli and 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 105 for S. aureus) and plated on agar. After 24 hours of incubation at 37 °C, colony counts were recorded to calculate bacterial survival.
105 for S. aureus) and plated on agar. After 24 hours of incubation at 37 °C, colony counts were recorded to calculate bacterial survival.
Unclosed wound rate (%) = (At/A0) × 100%. Where At and A0 are the wound area on day t and day 0 after surgery, respectively.
3. Results and discussion
3.1 Synthesis and characterization of PDA@BNN6 NPs with NIR controlled release of NO
The light-responsive PDA@BNN6 NPs were synthesized as shown in Fig. 1a, where the NO donor BNN6 was synthesized mainly from N,N'-bis(1-methylpropyl)-1,4-phenylenediamine (BPA) as evidenced by 1H Nuclear Magnetic Resonance (1H NMR, δ = 7.52 (4H), 4.95–4.69 (2H), 2.00–1.84 (2H), Fig. S1, ESI†), the PDA NPs were prepared by a one-step method based on the formation of nanoparticles by self-assembly and π–π interactions of dopamine monomers. Subsequently, PDA NPs formed adsorption coupling with BNN6 on the surface through π–π stacking to obtain a self-assembled core–shell structured PDA@BNN6 NPs.47 As shown in Fig. 1b and c, the SEM and TEM images show that both the PDA and PDA@BNN6 NPs are homogeneous globular structure, with particle sizes of 130 nm and 145 nm, respectively, suggesting that the success loading of BNN6 on the PDA surface without changing the particle morphology. The Fourier transform infrared spectroscopy (FTIR) of the synthesized PDA, BNN6 and PDA@BNN6 NPs are shown in Fig. 1d. It was observed that a N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O absorption peak and a N–N
O absorption peak and a N–N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O absorption peaks appeared at 1560 cm−1 and 1376 cm−1 of PDA@BNN6, respectively, which matched the characteristic peaks of BNN6, indicating that BNN6 was successfully loaded onto the PDA NPs. In addition, the results of dynamic light scattering (DLS) analysis and zeta potential (Fig. 1e and f) show that the hydrated particle size of PDA is 134.8 ± 2 nm with a zeta potential of −22.4 ± 0.2 mV, which indicate that the PDA NPs are electronegative with uniform size, while that of PDA@BNN6 is 145.8 ± 1.9 nm with a Zeta potential of −20.3 mV. Subsequently, the appearance of –N
O absorption peaks appeared at 1560 cm−1 and 1376 cm−1 of PDA@BNN6, respectively, which matched the characteristic peaks of BNN6, indicating that BNN6 was successfully loaded onto the PDA NPs. In addition, the results of dynamic light scattering (DLS) analysis and zeta potential (Fig. 1e and f) show that the hydrated particle size of PDA is 134.8 ± 2 nm with a zeta potential of −22.4 ± 0.2 mV, which indicate that the PDA NPs are electronegative with uniform size, while that of PDA@BNN6 is 145.8 ± 1.9 nm with a Zeta potential of −20.3 mV. Subsequently, the appearance of –N![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O and –N–H peaks (402.1 eV and 400.2 eV) in the XPS spectra, as shown in Fig. 1g, h and Fig. S2 (ESI†), also further prove the successful coupling of BNN6 on PDA.
O and –N–H peaks (402.1 eV and 400.2 eV) in the XPS spectra, as shown in Fig. 1g, h and Fig. S2 (ESI†), also further prove the successful coupling of BNN6 on PDA.
To investigate whether the 808 nm NIR laser can control the NO production by PDA@BNN6, the amount of NO produced by PDA@BNN6 at a concentration of 400 μg mL−1 under the irradiation of the 808 nm NIR laser was quantified using the Griess reagent. As showcased in Fig. 1i, only a small amount of NO was produced without light illumination, but once NIR illumination was applied, the PDA@BNN6 NPs rapidly released NO, and the maximum amount of NO released could be more than 300 μM. Furthermore, Fig. 1j shows the NO release from the PDA@ BNN6 under the “on–off” switching condition of the 808 nm NIR laser. When the NIR laser was turned on, NO was produced continuously and the accumulated amount increased; when the laser was turned off, the production of NO stopped immediately; and further turned on the laser, NO continued to be produced and the accumulated amount increased, showing that the NO production can be schemed controlled through the “on–off” switching of the laser. The above results indicate that we successfully synthesized PDA@BNN6 NPs with light-responsive controlled release of NO.
3.2 Temperature and light dual-responsive PHDNN6 composite hydrogels with controlled NO release and temperature sensing
The temperature-responsive hydrogel matrix was composed of PNIPAM and methacrylated (–MA)/dopamine (–DA) double grafted hyaluronic acid (HA–MA–DA), the resulting composite hydrogel was abbreviated as PH. As illustrated in Fig. 2a, the methacrylate-modified HA was chosen to form the network structure by double crosslinking with PNIPAM hydrogels. Meanwhile, the modification of DA enhanced the adhesion properties of the composite hydrogel and ensured a conformal contact with tissues. In addition, PDA@BNN6 also forms hydrogen bonding interactions with PNIPAM and HA–MA–DA. Consequently, as shown in Fig. 2b, the swelling ratio of PH hydrogel increased compared to the pure PNIPAM hydrogel, which can be attributed to the higher water retention ability of HA itself. After further compounding PDA@BNN6, the swelling ratio changed little due to the low content of PDA@BNN6. Moreover, the results in Fig. 2c and Fig. S3 (ESI†) demonstrate the compressive strength properties of several groups of hydrogels, in which pure PNIPAM shows the compressive strength of 0.15 MPa, while the compressive strength of PH hydrogel was increased to 0.16 MPa due to the formation of covalent interaction and hydrogen bonding interactions between HA–MA–DA and PNIPAM. The compressive strength of PHDNN6 was increased to 0.18 MPa after the compositing of PDA@BNN6 due to the additional hydrogen bonding from PDA. The dynamic rheological properties of the hydrogels were further investigated to examine the viscoelasticity of the hydrogels. As shown in Fig. 2d, both PH and PHDNN6 showed energy storage modulus (G′) and loss modulus (G′′) in the scanning frequency range from 0.1 to 100 rad s−1. The rheological properties of all hydrogel samples exhibited G′ > G′′, indicating that the samples had gel properties and formed a stable elastic crosslinked network. Furthermore, as Fig. 2e shown, all the hydrogels showed a classical porous structure, and the composite of PDA@BNN6 NPs into PHDNN6 hydrogel did not result in the destruction of the porous structure.We further quantified the NO content released from the PHDNN6 hydrogel in Fig. 2f. The PHDNN6 + NIR group showed a significant increase in NO release compared with the group without irradiation, which indicates that NIR can accelerate the decomposition of BNN6 to produce NO. Meanwhile, the release of NO in the hydrogel gradually slowed down with the increase of light time, and only a small amount of NO was produced. The rapid and then slow release of NO shown by PHDNN6 hydrogel under NIR excitation provides a favorable basis for further application in wound healing. Similarly, the PHDNN6 hydrogel also exhibited a controllable NO release from under the “on–off” switching of the NIR (Fig. 2g).
The electrical performance and temperature sensing performance of PHDDN6 hydrogel is the key to realize the temperature monitoring. Due to the inherent presence of ions in the hydrogel, it has the ability to transport electrons without the addition of conductive components.48 As shown in Fig. 2h, the conductivity of the several groups of hydrogels are nearly 0.6 S m−1, which meet the requirements for using as temperature sensors.49 Furthermore, the results of the V–I curves of the hydrogel showed that the current of PHDNN6 increased with the increase of voltage (Fig. 2i), suggesting that the resistive response behavior of PHDNN6 provides the basis for the sensing performance of the hydrogel. In addition, as shown in Fig. 2j, in vitro temperature response testing of the PHDNN6 hydrogel indicated that the resistance change (R/R0) was up to 35% under temperature change, and this good temperature response performance of the PHDNN6 hydrogel shows advantages in wound monitoring. As depicted in Fig. 2k, we evaluated the resistance stability of the hydrogel under cyclic temperature variations between 25 °C and 42 °C (n = 10). The outcomes indicate minimal resistance fluctuations and commendable long-term stability, showcasing its stability against temperature fluctuations. As a result, we have obtained a temperature and light dual-responsive hydrogel dressing with both physical and chemical properties that can be applied to the wound repair interface.
3.3 Biocompatibility and antibacterial properties of PHDNN6 composite hydrogel
To assess the biocompatibility of the hydrogels, the growth of cultured BMSCs cells in each group of materials was observed by fluorescein diacetate/propidium iodide (FDA/PI) staining. As shown in Fig. 3a, none of the hydrogel groups showed significant cytotoxicity with increasing incubation time. To further evaluate the cytotoxicity of the hydrogels, we utilized 3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide (MTT) assay to assess cell proliferation. As depicted in Fig. 3b, after 1, 4, and 7 days of incubation, all experimental groups exhibited significant cell proliferation, with no notable differences observed among them. This finding underscores the excellent biocompatibility of the hydrogels.Further, the antibacterial properties of the hydrogels were investigated. The results of agar plate culture of bacterial suspensions are shown in Fig. 3c. Almost no colonies were formed on E. coli agar plates treated with PHDNN6 group and PHDNN6 + NIR group with a power of 2 W cm−2, while colonies were still formed in the control and PH groups alone. Similarly, experiments with growing S. aureus agar plates showed a similar trend, PHDNN6 + NIR group hydrogel resulted in virtually less colony formation on S. aureus plates compared with control, PH groups. Meanwhile, as can be seen from the statistics of relative bacterial viability of E. coli and S. aureus (Fig. 3d and e), both PHDNN6 and PHDNN6 + NIR groups exhibited antimicrobial activity, but the antimicrobial activity of PHDNN6 + NIR group was still higher than that of PHDNN6 group, this is attributed to the fact that NIR light can promote the production of NO on the one hand, and on the other hand, the photothermal effect of PDA can generate high temperature at the same time, which damage the bacterial structure.
3.4 Anti-inflammatory and macrophage M2-promoting effects of PHDNN6 composite hydrogels in vitro
In the natural course of skin wound healing, the cessation of cytokine supply after an inflammatory response triggers the programmed death of inflammatory cells, known as apoptosis.50 Conversely, in the case of chronic wounds, the unregulated activation of key inflammatory players, notably neutrophils and macrophages, prompts the overproduction of cytokines. This excessive release fuels an intensified inflammatory reaction, fostering a persistent and detrimental cycle of chronic inflammation.51 Nuclear factor κB (NF-κB) p65 serves as a pivotal nuclear transcription factor within cells, orchestrating the inflammatory response by fine-tuning the expression of inflammatory genes. It constitutes a prominent pro-inflammatory signaling cascade that governs numerous pro-inflammatory mediators, including but not limited to IL-1β, IL-6, and IL-18.52,53 In an inflammatory milieu, NF-κB p65 undergoes phosphorylation, facilitating its translocation to the nucleus. This translocation subsequently fosters the transcription of inflammatory biomarkers, thereby contributing to inflammatory damage within the wound, representing a crucial mechanism in the regulation of the inflammatory process. To study the macrophage activation in vivo, RAW264.7 cells were initially stimulated with lipopolysaccharide (LPS) for 2 hours, followed by treatment with various hydrogels-PH, PHDNN6, and PHDNN6 + NIR.We explored the anti-inflammatory of different hydrogel group treatments after LPS inflammation induction by immunofluorescence staining. The fluorescence staining and corresponding statistical analysis in Fig. 4a and b show that the expression of p65 protein increased in the LPS group, PH group, and PHDNN6 group, while the PHDNN6 + NIR group exhibited a marked decrease in NF-κB p65 levels, attributed to the released NO's ability to inhibit NF-κB transcription by blocking p65's phosphorylation and nuclear translocation. Furthermore, TNF-α, a key inflammatory marker downstream of the NF-κB pathway, serves as an indicator of inflammation intensity, and its expression levels reflect this status to a certain extent.54 Moreover, the expression of TGF-β responds to the degree of activity of immune cells, and it can be considered as a response to the degree of cellular anti-inflammation.55 Consequently, immunofluorescence staining of TNF-α- and TGF-β shows that the expression was almost unchanged in the LPS treatment group and those with the treatment of PH and the PHDNN6 groups. However, TNF-α expression decreased while TGF-β expression increased after treatment with PHDNN6 + NIR, suggesting the release of NO in the PHDNN6 + NIR group inhibited the inflammatory response, promoted the reduction of inflammatory cells, and allowed the wound to enter the proliferation and remodeling stage. Additionally, macrophages occupy a central role among immune cells at the wound, with their transformation from the M1 phenotype (pro-inflammation) to the M2 phenotype (anti-inflammation) being pivotal for mitigating inflammation and facilitating wound healing.56,57 To assess the modulatory effect on macrophage polarization, immunofluorescence results of CD86 and C206 showed that PHDNN6 + NIR hydrogels completely counteracted the pro-inflammatory LPS-induced decrease in M2 macrophages (CD206+). In addition, the expression of intracellular iNOS did not change significantly throughout the inflammatory process, which indicates that the production of endogenous NO was not altered, the cells themselves did not produce NO after hydrogel treatment in all groups, and the NO that produced anti-inflammatory effects only came from the NIR-controlled NO release of PHDNN6 hydrogel. The above results indicate that the ability of our prepared PHDNN6 hydrogels to release NO under NIR irradiation has significant anti-inflammatory effects and promotes M2 polarization of macrophages, providing favorable conditions for use in wound repair.
3.5 NIR-induced NO release from PHDNN6 hydrogel promotes wound repair in vivo
To evaluate whether PHDNN6 hydrogel has the effect of promoting the healing of infected wounds, animal experiments were conducted in a rat whole skin wound model (Fig. 5a). Four experimental groups were assessed, including the control group, the PH hydrogel group, the PHDNN6 hydrogel group, and the PHDNN6 + NIR group, in which the PHDNN6 + NIR group was irradiated for 10 minutes at the 2nd day after surgery. The photographs of the wounds (Fig. 5b, c and Fig. S4, ESI†) and the remaining area of the wounds (%) as the number of days of repair are shown in Fig. 5d. The wounds of all groups decreased gradually with the increase of the number of days of the experiment. On the 3rd day after surgery, the wounds in the PHDNN6 + NIR group were smaller than that of the control, PH, and PHDNN6 groups (P < 0.05), demonstrating the ability of PDA@BNN6 to promote wound healing under NIR irradiation. On the 10th postoperative day, the wound in the PHDNN6 + NIR group was almost completely closed, and there was an obvious difference between the wound areas in the control, PH, and PHDNN6 groups (P < 0.05). During the wound repair period, the wound healing speed of the PHDNN6 group was faster than the control group, demonstrating that the PHDNN6 hydrogel itself has good ability to promote wound healing. And when the PHDNN6 hydrogel was irradiated by NIR to release a large amount of NO, the ability of wound healing was more significant. The above results indicate that the combined effect of PHDNN6 + NIR can most significantly promote infected wound healing.Wound healing is divided into a total of three stages at the cellular level, which are inflammation, proliferation and maturation, so we can assess the wound healing by observing the degree of inflammation and the number of collagen fibers. After 14 days implantation, H&E staining was utilized to assess the wound growth and inflammation levels after wound healing. As depicted in Fig. 5e, the control group exhibited a significant presence of inflammatory cells (highlighted by red arrows), whereas compared to the control group, the PH hydrogel group, though showing some improvement, still maintained a notable count of inflammatory cells. Moreover, the PHDNN6 group displayed a decreased immune cell count, suggesting NO's immunosuppressive effect. Notably, the PHDNN6 + NIR group demonstrated a pronounced decrease in inflammatory cells, highlighting PDA@BNN6's ability to swiftly release lots of NO under NIR irradiation, thereby inhibiting inflammation and enhancing wound healing. Masson staining, which highlights collagen fibers in blue, was employed to assess collagen deposition. Fig. 5f and g reveal that both the control and pH hydrogel groups exhibited low collagen content, indicative of limited wound repair. In contrast, the PHDNN6 + NIR hydrogel group stood out with the highest collagen deposition, thickest fibers, and a collagen fiber structure resembling that of healthy tissue, arranged in an orderly fashion. The PHDNN6 hydrogel group, however, did not significantly outperform the PH group in this regard. Besides, PHDNN6 + NIR group also showed a lowest epidermal thickness compared with other groups (Fig. 5h). These results indicate that under the effect of NO, the wound healing in the PHDNN6 + NIR hydrogel group entered the maturation stage, and the collagen was reorganized to repair the scar tissue.
3.6 Temperature monitoring assists wound repair management and the anti-inflammatory mechanism of NO controlled release
The wound healing process is highly correlated with cellular activities regulated by cytokines, and we choose three representative indicators, TNF-α, TGF-β and p65, to assess the degree of inflammation in the wounds of different experimental groups, and two representative indicators, CD86 and CD206, to assess the degree of macrophage polarization in the wounds of different experimental groups. Consequently, at day 3 of wound healing, the immunofluorescence images and corresponding protein expression statistics of all groups showed insignificant differences (Fig. S5, ESI†). As the repair progressed to day 7, as depicted in Fig. 6a and b, immunofluorescence imaging and quantitative assessment revealed a notable decline in the expression of p65 and TNF-α within the PHDNN6 + NIR group, compared to the remaining three groups, suggesting a gradual alleviation of the inflammatory response at the wound site. Parallelly, as the repair time extended, TGF-β expression augmented solely in the PHDNN6 group, while no significant alterations were observed in the other three groups. Furthermore, utilizing CD86 as a marker for M1-type macrophages and CD206 for M2-type macrophages, our results indicated minimal variation in CD86 and CD206 expression across the control, PH, and PHDNN6 groups. However, in the PHDNN6 + NIR group, a significant reduction in CD86 expression and a concurrent increase in CD206 expression were observed, pointing to the successful transition of macrophages from the M1 to the M2 phenotype under NIR induction. Besides, wound temperature as an important indicator of wound inflammation, in our previous research work, it has been confirmed that the wound inflammation is highest on the 3rd day, and the temperature monitoring results recorded from Bluetooth-integrated hydrogel system also confirmed that the wound temperature is highest on the 3rd day.13 Therefore, throughout the wound healing process, we also conducted real-time temperature monitoring of the wounds, the photographs of assemble PHDNN6 hydrogel matrix with real-time and remote monitoring module on a rat was shown in Fig. S6 and S7 (ESI†). When the temperature of the rat's wound changes, it will affect the volume of the temperature-responsive hydrogel, and the change in electrical resistance due to the hydrogel's volume shift can be detected by the Bluetooth connected to it, and ultimately transmitte to the smartphone by wireless communication. As shown in Fig. 6c, for the PHDNN6 + NIR group, the wound temperature started to decrease at about 4000 minutes (about 3 days), indicating that the inflammation of the wound in the PHDNN6 + NIR group reached its peak at 4000 minutes, and the inflammation was in a state of subsiding in the following time. And due to the anti-inflammatory treatment by releasing NO at the early stage of inflammation, we can observe that the maximum value of wound temperature in the PHDNN6 + NIR group is significantly lower than that in the other two groups, which also indicates that the PHDNN6 + NIR hydrogel can be anti-inflammatory by releasing a large amount of NO under NIR irradiation, thus promoting the healing of the wound.The aforementioned findings underscore the pivotal role of the NF-κB signaling pathway in immune response and inflammation.58 Activation of this pathway occurs through phosphorylation of p65 and subsequent formation of the NF-κB-p65 dimer. Consequently, a marked elevation in NF-κB p65 expression is evident in cells exposed to LPS or wound-induced inflammation. Additionally, the introduction of exogenous NO into the system triggers a cascade activation of the NO/cGMP/PKG/K+ ATP signaling pathway, resulting in a substantial increase in cGMP (guanosine cyclic phosphate) levels.59 Notably, the activation of the cGMP/PKG signaling pathway effectively inhibits p65 phosphorylation, thereby disrupting the NF-κB inflammatory response signaling pathway. Therefore, when the PHDNN6 hydrogel generates a large amount of NO under NIR irradiation, a significant decrease in the expression of p65 can be clearly observed and its phosphorylation is inhibited. This led to a decrease in the expression of the downstream inflammation-related factor TNF-α, as well as an elevation of TGF-β expression. It also stimulated the conversion of M1-type macrophages to M2, thereby inhibiting the inflammatory response (Fig. 6d).
4. Conclusions
In this study, we successfully synthesized light-responsive PDA@BNN6 nanoparticles capable of NIR controlled NO release. Furthermore, we developed a novel temperature and light dual-responsive hydrogel dressing, PHDNN6, by incorporating PDA@BNN6 NPs into a PNIPAM-based hydrogel matrix with the inclusion of HA–MA–DA. The PHDNN6 hydrogel not only monitored wound temperature changes effectively but also released NO in a controlled manner upon NIR irradiation, enabling real-time wound management. In vitro and in vivo studies revealed that the PHDNN6 hydrogel exhibited excellent biocompatibility, antibacterial properties, and anti-inflammatory effects through NO-mediated inhibition of the NF-κB signaling pathway. Specifically, NO released from PHDNN6 upon NIR irradiation reduced inflammation, promoted macrophage polarization towards the M2 phenotype, and accelerated wound healing in rat models. Additionally, real-time wound temperature monitoring facilitated early detection of inflammation and guided therapeutic interventions. Overall, our findings suggest that the temperature and light dual-responsive PHDNN6 hydrogel represents a promising platform for advanced wound care and dynamic wound management.Author contributions
Ji Jiang: conceptualization, methodology, investigation, writing – original draft, writing – review & editing. Yuan Tian: methodology, formal analysis, writing – original draft, data curation. Xiaoyang Wu: methodology, investigation, validation. Mingze Zeng: data curation, formal analysis. Chengheng Wu: methodology, investigation, validation. Dan Wei: data curation, formal analysis, investigation, funding acquisition. Hongrong Luo: conceptualization, methodology. Jing Sun: visualization, formal analysis, funding acquisition. Jie Ding: methodology, investigation, validation, data curation, formal analysis, writing – original draft, writing – review & editing, funding acquisition. Hongsong Fan: methodology, investigation, visualization, formal analysis, writing – review & editing, funding acquisition.Data availability
We confirm that all the relevant research data is contained with the manuscript and electronic ESI.† No databases have been used and no references to such databases are contained in the manuscript or ESI.†Conflicts of interest
The authors declare no conflict of interest.Acknowledgements
This work is supported by the National Natural Science Foundation of China (no. 52273141 and 52373150), Natural Science Foundation of Sichuan Province (no. 2023NSFSC0338 and 2024NSFSC1014).References
- O. A. Peña and P. Martin, Nat. Rev. Mol. Cell Biol., 2024, 25, 599–616 CrossRef PubMed.
- L. Zhou, Z. Zeng, S. Liu, T. Min, W. Zhang, X. Bian, H. Du, P. Zhang and Y. Wen, Adv. Funct. Mater., 2022, 32, 2207466 CrossRef CAS.
- V. Falanga, R. R. Isseroff, A. M. Soulika, M. Romanelli, D. Margolis, S. Kapp, M. Granick and K. Harding, Nat. Rev. Dis. Primers, 2022, 8, 50 CrossRef PubMed.
- Q. Pang, F. Yang, Z. Jiang, K. Wu, R. Hou and Y. Zhu, Mater. Des., 2023, 229, 111917 CrossRef CAS.
- G. Yao, X. Mo, C. Yin, W. Lou, Q. Wang, S. Huang, L. Mao, S. Chen, K. Zhao, T. Pan, L. Huang and Y. Lin, Sci. Adv., 2022, 8, eabl8379 CrossRef CAS PubMed.
- P. Mostafalu, A. Tamayol, R. Rahimi, M. Ochoa, A. Khalilpour, G. Kiaee, I. K. Yazdi, S. Bagherifard, M. R. Dokmeci, B. Ziaie, S. R. Sonkusale and A. Khademhosseini, Small, 2018, 14, 1703509 CrossRef PubMed.
- J. Shi, S. Kim, P. Li, F. Dong, C. Yang, B. Nam, C. Han, E. Eig, L. L. Shi, S. Niu, J. Yue and B. Tian, Science, 2024, 384, 1023–1030 CrossRef CAS PubMed.
- J. Saminathan, M. Sasikala, V. B. Narayanamurthy, K. Rajesh and R. Arvind, Infrared Phys. Technol., 2020, 105, 103219 CrossRef.
- D. Rybak, C. Rinoldi, P. Nakielski, J. Du, M. A. Haghighat Bayan, S. S. Zargarian, M. Pruchniewski, X. Li, B. Strojny-Cieślak, B. Ding and F. Pierini, J. Mater. Chem. B, 2024, 12, 1905–1925 RSC.
- D. Yang, Chem. Mater., 2022, 34, 1987–1989 CrossRef CAS.
- X. Ma, L. Lin, H. Luo, Q. Zheng, H. Wang, X. Li, Z. Wang, Y. Feng and Y. Chen, Adv. Sci., 2024, 2403362 CrossRef CAS PubMed.
- J. Sun, W. Jia, H. Qi, J. Huo, X. Liao, Y. Xu, J. Wang, Z. Sun, Y. Liu, J. Liu, M. Zhen, C. Wang and C. Bai, Adv. Mater., 2024, 36, 2312440 CrossRef CAS PubMed.
- J. Jiang, J. Ding, X. Wu, M. Zeng, Y. Tian, K. Wu, D. Wei, J. Sun, Z. Guo and H. Fan, J. Mater. Chem. B, 2023, 11, 4934–4945 RSC.
- Y. Hiruta, Polym. J., 2022, 54, 1419–1430 CrossRef CAS.
- P. Nakielski, S. Pawłowska, C. Rinoldi, Y. Ziai, L. De Sio, O. Urbanek, K. Zembrzycki, M. Pruchniewski, M. Lanzi, E. Salatelli, A. Calogero, T. A. Kowalewski, A. L. Yarin and F. Pierini, ACS Appl. Mater. Interfaces, 2020, 12, 54328–54342 CrossRef CAS PubMed.
- M. A. Haghighat Bayan, Y. J. Dias, C. Rinoldi, P. Nakielski, D. Rybak, Y. B. Truong, A. L. Yarin and F. Pierini, J. Polym. Sci., 2023, 61, 521–533 CrossRef CAS.
- Y. Ziai, F. Petronella, C. Rinoldi, P. Nakielski, A. Zakrzewska, T. A. Kowalewski, W. Augustyniak, X. Li, A. Calogero, I. Sabała, B. Ding, L. De Sio and F. Pierini, NPG Asia Mater., 2022, 14, 18 CrossRef CAS.
- Y. Ziai, C. Rinoldi, F. Petronella, A. Zakrzewska, L. De Sio and F. Pierini, Nanoscale, 2024, 16, 13492–13502 RSC.
- Y. Ziai, C. Rinoldi, P. Nakielski, L. De Sio and F. Pierini, Curr. Opin. Biomed. Eng., 2022, 24, 100413 CrossRef CAS.
- Y. Chen, Y. Gao, L. P. da Silva, R. P. Pirraco, M. Ma, L. Yang, R. L. Reis and J. Chen, Polym. Chem., 2018, 9, 4063–4072 RSC.
- B. L. Ekerdt, C. M. Fuentes, Y. Lei, M. M. Adil, A. Ramasubramanian, R. A. Segalman and D. V. Schaffer, Adv. Healthcare Mater., 2018, 7, 1800225 CrossRef PubMed.
- I. K. Kwon and T. Matsuda, Biomaterials, 2006, 27, 986–995 CrossRef CAS PubMed.
- A. Dalla Pietà, D. Carpanese, A. Grigoletto, A. Tosi, S. Dalla Santa, G. K. Pedersen, D. Christensen, L. Meléndez-Alafort, V. Barbieri, P. De Benedictis, G. Pasut, I. M. Montagner and A. Rosato, Cell. Mol. Immunol., 2021, 18, 1197–1210 CrossRef PubMed.
- H. Kim, S. Choi, Y. Hong, J. Chung, J. Choi, W.-K. Choi, I. W. Park, S. H. Park, H. Park, W.-J. Chung, K. Heo and M. Lee, Appl. Mater. Today, 2021, 22, 100920 CrossRef.
- S. A. Bencherif, A. Srinivasan, F. Horkay, J. O. Hollinger, K. Matyjaszewski and N. R. Washburn, Biomaterials, 2008, 29, 1739–1749 CrossRef CAS PubMed.
- D. Zhou, S. Li, M. Pei, H. Yang, S. Gu, Y. Tao, D. Ye, Y. Zhou, W. Xu and P. Xiao, ACS Appl. Mater. Interfaces, 2020, 12, 18225–18234 CrossRef CAS PubMed.
- H. Liu, F. Xing, Y. Zhou, P. Yu, J. Xu, R. Luo, Z. Xiang, P. Maria Rommens, M. Liu and U. Ritz, Mater. Des., 2023, 233, 112231 CrossRef CAS.
- L. He, D. Di, X. Chu, X. Liu, Z. Wang, J. Lu, S. Wang and Q. Zhao, J. Controlled Release, 2023, 363, 180–200 CrossRef CAS PubMed.
- B. Muzzi, M. Albino, A. Gabbani, A. Omelyanchik, E. Kozenkova, M. Petrecca, C. Innocenti, E. Balica, A. Lavacchi, F. Scavone, C. Anceschi, G. Petrucci, A. Ibarra, A. Laurenzana, F. Pineider, V. Rodionova and C. Sangregorio, ACS Appl. Mater. Interfaces, 2022, 14, 29087–29098 CrossRef CAS PubMed.
- S. Zhao, L. Yan, M. Cao, L. Huang, K. Yang, S. Wu, M. Lan, G. Niu and W. Zhang, ACS Appl. Mater. Interfaces, 2021, 13, 53610–53617 Search PubMed.
- W. Hu, T. Xiao, D. Li, Y. Fan, L. Xing, X. Wang, Y. Li, X. Shi and M. Shen, Adv. Sci., 2021, 8, 2100165 CrossRef CAS PubMed.
- X. Liu, N. Xu, X. Pu, J. Wang, X. Liao, Z. Huang and G. Yin, J. Mater. Chem. B, 2022, 10, 4605–4614 RSC.
- B. Zhou, C. Yin, Q. Feng, Y. Wu, X. Pan, C. Liu, J. Tian, S. Geng, K. Wang, J. Xing, Y. Cao, P. Shou, Z. Yu and A. Wu, Nanoscale, 2021, 13, 19085–19097 RSC.
- X. Qi, Y. Huang, S. You, Y. Xiang, E. Cai, R. Mao, W. Pan, X. Tong, W. Dong, F. Ye and J. Shen, Adv. Sci., 2022, 9, 2106015 CrossRef CAS PubMed.
- M. A. Haghighat Bayan, C. Rinoldi, A. Kosik-Kozioł, M. Bartolewska, D. Rybak, S. S. Zargarian, S. A. Shah, Z. J. Krysiak, S. Zhang, M. Lanzi, P. Nakielski, B. Ding and F. Pierini, Adv. Mater. Technol., 2024, 2400450 CrossRef.
- M. Bartolewska, A. Kosik-Kozioł, Z. Korwek, Z. Krysiak, D. Montroni, M. Mazur, G. Falini and F. Pierini, Adv. Healthcare Mater., 2025, 2402431 CrossRef CAS PubMed.
- B. Poinard, S. Z. Y. Neo, E. L. L. Yeo, H. P. S. Heng, K. G. Neoh and J. C. Y. Kah, ACS Appl. Mater. Interfaces, 2018, 10, 21125–21136 CrossRef CAS PubMed.
- W. Cheng, X. Zeng, H. Chen, Z. Li, W. Zeng, L. Mei and Y. Zhao, ACS Nano, 2019, 13, 8537–8565 CrossRef CAS PubMed.
- L. M. Estes, P. Singha, S. Singh, T. S. Sakthivel, M. Garren, R. Devine, E. J. Brisbois, S. Seal and H. Handa, J. Colloid Interface Sci., 2021, 586, 163–177 CrossRef CAS PubMed.
- L. K. Wareham, R. K. Poole and M. Tinajero-Trejo, J. Biol. Chem., 2015, 290, 18999–19007 CrossRef CAS PubMed.
- Z. Yuan, W. Zhang, C. Wang, C. Zhang, C. Hu, L. Liu, L. Xiang, S. Yao, R. Shi, D. Fan, B. Ren, G. Luo and J. Deng, Regener. Biomater., 2025, 12, rbae134 Search PubMed.
- X. Jin, Z. Ou, X. Huang, L. Shi, R. Shi, J. Wang, J. Yang, K. Fang, J. Yang, D. Lv, C. Wang, Z. Yuan, W. li, W. Liu, G. Luo, J. Deng and W. Wang, Nano Today, 2023, 51, 101898 CrossRef CAS.
- X. Ge, J. Hu, Y. Peng, Z. Zeng, D. He, X. Li, Y. Chen, G. Luo, J. Deng, Z. Xu and S. He, Biomaterials, 2023, 301, 122254 Search PubMed.
- X. Lv, Y. Xu, X. Ruan, D. Yang, J. Shao, Y. Hu, W. Wang, Y. Cai, Y. Tu and X. Dong, Acta Biomater., 2022, 146, 107–118 Search PubMed.
- C. He, Y. Wang, C. Chen, L. Yuan, W. Dong, X. Yang, Y. Wu, J. Liu, Q. Kong, J. He and B. Yan, Mater. Des., 2024, 238, 112719 CrossRef CAS.
- J. He, Z. Li, J. Chen, J. Wang, L. Qiao, B. Guo and J. Hu, Chem. Eng. J., 2024, 492, 152249 Search PubMed.
- G. Liu, L. Wang, Y. He, L. Wang, Z. Deng, J. Liu, D. Peng, T. Ding, L. Lu, Y. Ding, J. Zhang, P. Liu and K. Cai, Adv. Healthcare Mater., 2021, 10, 2101476 CrossRef CAS PubMed.
- H. Zhang, T. Gao, L. Jiang, X. Meng, J. Wang, N. Ma, H. Wei and X. Zhang, Eur. Polym. J., 2022, 173, 111266 Search PubMed.
- Y. Shi, C. Ma, L. Peng and G. Yu, Adv. Funct. Mater., 2015, 25, 1219–1225 CrossRef CAS.
- Z. Zhou, T. Deng, M. Tao, L. Lin, L. Sun, X. Song, D. Gao, J. Li, Z. Wang, X. Wang, J. Li, Z. Jiang, L. Luo, L. Yang and M. Wu, Biomaterials, 2023, 299, 122141 CrossRef CAS PubMed.
- O. Soehnlein, S. Steffens, A. Hidalgo and C. Weber, Nat. Rev. Immunol., 2017, 17, 248–261 CrossRef CAS PubMed.
- H. Yu, L. Lin, Z. Zhang, H. Zhang and H. Hu, Signal Transduction Targeted Ther., 2020, 5, 209 CrossRef CAS PubMed.
- T. Liu, L. Zhang, D. Joo and S.-C. Sun, Signal Transduction Targeted Ther., 2017, 2, 17023 CrossRef PubMed.
- G. van Loo and M. J. M. Bertrand, Nat. Rev. Immunol., 2023, 23, 289–303 CrossRef CAS PubMed.
- Z. Deng, T. Fan, C. Xiao, H. Tian, Y. Zheng, C. Li and J. He, Signal Transduction Targeted Ther., 2024, 9, 61 CrossRef PubMed.
- Y. Yuan, D. Fan, S. Shen and X. Ma, Chem. Eng. J., 2022, 433, 133859 CrossRef CAS.
- J. Sapudom, S. Karaman, W. K. E. Mohamed, A. Garcia-Sabaté, B. C. Quartey and J. C. M. Teo, npj Regener. Med., 2021, 6, 83 CrossRef CAS PubMed.
- K. Taniguchi and M. Karin, Nat. Rev. Immunol., 2018, 18, 309–324 Search PubMed.
- Y. Zhao, P. M. Vanhoutte and S. W. S. Leung, J. Pharmacol. Sci., 2015, 129, 83–94 Search PubMed.
Footnotes |
| † Electronic supplementary information (ESI) available. See DOI: https://doi.org/10.1039/d4tb02555e |
| ‡ These authors contributed equally to this work. |
| This journal is © The Royal Society of Chemistry 2025 |






