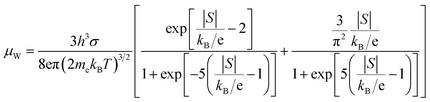Enhancing the thermoelectric performance of n-type polycrystalline SnSe with lead-free perovskite Cs2TiCl6
Wenjie
Li
a,
Zeqing
Hu
a,
Ziwang
Zhang
a,
Zehao
Lin
a,
Min
Ruan
a,
Jiahao
Jiang
a and
Jing
Shuai
 *ab
*ab
aSchool of Materials, Sun Yat-sen University, Shenzhen, 518107, China. E-mail: shuaij3@mail.sysu.edu.cn
bGuangdong Provincial Key Laboratory of Magnetoelectric Physics and Devices, Sun Yat-sen University, Guangzhou 510275, China
First published on 14th November 2025
Abstract
Enhancing the thermoelectric performance of n-type polycrystalline SnSe remains challenging due to the trade-off between carrier transport and lattice thermal conductivity. Here, we demonstrate that trace incorporation of the lead-free perovskite Cs2TiCl6 enables concurrent optimization of these factors in SnSe0.93. The [TiCl6]2− octahedral framework introduces effective electron donors, while Ti 3d states induce local band flattening, increasing the density-of-states effective mass without severely degrading weighted mobility. Consequently, the 0.5 wt% Cs2TiCl6 sample attains a high power factor of ∼532.1 µW m−1 K−2 at 823 K. Meanwhile, Ti-rich domains together with multi-scale structural defects (strain fields, dislocations, and twin boundaries) substantially intensify phonon scattering, driving the lattice thermal conductivity down to ∼0.32 W m−1 K−1 at 823 K—a 40.6% reduction relative to the pristine specimen. Benefiting from this synergistic carrier and phonon engineering, a peak ZT of ∼1.2 at 823 K is achieved for 0.5 wt% Cs2TiCl6, representing a 179.1% enhancement over undoped SnSe0.93 and outperforming most reported n-type polycrystalline SnSe-based materials. This work establishes trace lead-free perovskite doping as a dual-function strategy—simultaneously enabling band structure modulation and phonon mean free path suppression—offering a viable route toward environmentally benign, high-efficiency n-type SnSe thermoelectrics.
1. Introduction
The direct interconversion of heat and electricity offered by thermoelectric (TE) materials enables solid-state waste-heat harvesting, localized cooling, and self-powered sensing—functions that are increasingly relevant against the backdrop of rising global energy demand and carbon-neutrality goals.1–4 The performance of thermoelectric materials is mainly governed by the dimensionless figure of merit, ZT, defined as ZT = (σS2)T/κtot, where σ is the electrical conductivity, S is the Seebeck coefficient, κtot is the total thermal conductivity (comprising both the lattice and electronic components), and T is the absolute temperature.5–9 An ideal thermoelectric material should simultaneously possess a high Seebeck coefficient, high electrical conductivity, and low thermal conductivity to achieve efficient energy conversion.10–12 However, the strong interdependency among these parameters poses considerable challenges for improving ZT.13 To address these issues, multi-scale strategies have been developed to decouple and optimize these properties. Notable approaches include band engineering to tailor carrier effective mass,14–17 defect engineering to optimize carrier concentration,18–21 and nanostructuring to enhance phonon scattering.22–27 These methods have led to substantial enhancements in ZT across various thermoelectric material systems, such as Mg2(Bi, Sb)3,28–32 half-Heusler,33–35 Bi2Te3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 36–43 and Cu2Se.23,44–46
36–43 and Cu2Se.23,44–46
Tin selenide (SnSe) emerged as a landmark TE material after single crystals exhibited ultralow lattice thermal conductivity and very high ZT at elevated temperatures, attributed to strong anharmonicity and layered anisotropy.12,47–51 Nevertheless, translating these advantages to n-type polycrystalline SnSe has proven difficult: (i) intrinsically low electron carrier concentration limits the power factor, (ii) uncontrolled grain boundary phases and point defects degrade mobility, and (iii) conventional heavy-element or toxic dopants (e.g. Pb- and Bi-based) raise environmental and regulatory concerns.8,52–57 Reported strategies—band convergence density-of-states (DOS) engineering, resonant or aliovalent doping, defect and dislocation tailoring, nano/micro-structuring, and hierarchical phonon scattering architectures—have individually advanced aspects of performance but often incur trade-offs such as mobility loss, complex multi-step processing, poor dopant solubility, or diminished scalability.54,55,58–63 Consequently, a simple, environmentally benign route that simultaneously boosts electron concentration, preserves or enhances the Seebeck coefficient, and suppresses lattice thermal conductivity in n-type polycrystalline SnSe is still lacking.
Lead-free halide double perovskites have recently attracted attention as electronically versatile and chemically tunable additives. In particular, Cs2TiCl6 contains isolated [TiCl6]2− octahedra whose Ti4+ and Cl− constituents can, in principle, introduce donor defects when interfaced with chalcogenide matrices, while its volatile-free, earth-abundant composition is advantageous from a green manufacturing perspective.64–67 Beyond simple electron donation, localized Ti 3d orbital hybridization and nanoscale compositional fluctuations could reshape the electronic structure (e.g. increase DOS effective mass m* via local band flattening or multi-valley contribution) to mitigate the typical Seebeck penalty of raising carrier concentration.68,69 Simultaneously, limited interdiffusion during a short, energy-efficient micro-fusion (micro-melting) process can generate Ti-rich nanoregions, high densities of edge dislocations, strain field fluctuations, and twin boundaries.68 Such multiscale defects act as broadband phonon scatterers, lowering κlat without proportionally compromising carrier transport—particularly if the dominant scattering centers selectively target mid- to long-wavelength phonons while leaving electronic pathways percolative. Segregated Cs+ at grain boundaries may further modulate interfacial vibrational mismatch and assist in lattice thermal conductivity suppression.67
Here, we demonstrate that introducing only a minute amount (≤0.75 wt%) of lead-free Cs2TiCl6 into n-type polycrystalline SnSe0.93via an eco-friendly micro-fusion route achieves coordinated optimization of charge and phonon transport. Electron concentration is markedly elevated through TiSn and ClSe donor defects, while an increase in DOS effective mass helps sustain an enhanced Seebeck coefficient relative to the pristine low baseline. Concomitantly, the engineered microstructure—comprising Ti-enriched nanoscale domains, dense dislocations, strain fields, and twin boundaries—suppresses κlat at high temperature despite a modest low-temperature rise attributed to Ti-rich regions. As a result, the sample containing 0.5 wt% Cs2TiCl6 attains a maximum ZT of about 1.2 at 823 K, representing ∼179% improvement over undoped SnSe0.93, together with a substantially enhanced power factor.
2. Experimental details
2.1 Sample synthesis
SnSe0.93 + x wt% Cs2TiCl6 (x = 0, 0.25, 0.5, 0.75) bulk samples were synthesized using high-purity raw materials, including Sn pellets (99.99%, Aladdin), Se pellets (99.999%, Aladdin), and Cs2TiCl6. The Cs2TiCl6 precursor was prepared by sequentially adding 0.001 mol of TiCl4, 0.002 mol of CsCl, and 20 mL of concentrated hydrochloric acid into a 50 mL polytetrafluoroethylene (PTFE) liner placed inside a reaction autoclave. The mixture was magnetically stirred for 10 minutes to ensure thorough homogenization of the reactants. Subsequently, the autoclave was sealed and heated in an oven at 433 K for 24 hours. After the reaction was completed and the system cooled to room temperature, the resulting product was filtered, washed several times with ethanol, and dried. All materials were handled in an Ar-filled glove box to prevent oxidation and contamination. The prepared mixtures were sealed in double-walled quartz ampoules under a vacuum of 10−4 Pa. The ampoules were heated to 1223 K and maintained at this temperature for 24 hours, followed by cooling to room temperature. The resulting ingots were ground into fine powders and consolidated into dense bulk samples using spark plasma sintering (SPS) with a LABOX-110 system (Japan).2.2 Phase structure and microstructure characterization
The phase structure of the synthesized samples was analyzed using X-ray diffraction (XRD) on a Bruker D8 Advance system. Rietveld refinement of the XRD patterns was performed using HighScore Plus software (version 4.0) to extract detailed crystallographic information. Microstructural features were further examined using a 300 kV field-emission transmission electron microscope (FE-TEM, Tecnai G2 F30 S-Twin).2.3 Chemical valence of corresponding elements
The chemical valence states of the constituent elements were characterized using X-ray photoelectron spectroscopy (Kratos Supra+, Shimadzu, UK).2.4 Measurement of thermoelectric properties
Thermoelectric transport properties were measured as follows: the Seebeck coefficient (S) and electrical conductivity (σ) were simultaneously measured using a ZEM-3 system (Ulvac-Riko, Japan). Thermal diffusivity (D) was determined using a laser flash apparatus (LFA 467 HT, Netzsch, Germany) on disk-shaped specimens. Specific heat capacity (Cp) was estimated using the Dulong–Petit limit.70–72 The sample density (ρ) was measured via the Archimedes method. Thermal conductivity (κ) was calculated using the relationships κ = DCpρ. The electronic thermal conductivity (κe) was calculated using the Wiedemann–Franz law, κe = LσT, where the Lorenz number L was estimated using the simplified formula L = 1.5 + exp(−|S|/116),73 consistent with common practice for degenerate semiconductors. Lattice thermal conductivity (κlat) was calculated as κlat = κ − κe. Additionally, the Hall coefficient (RH) was measured at room temperature using a multifunctional semiconductor test system (NYMS-MS, Nengyi NYVac).3. Results and discussion
3.1 Phase composition and microstructural characterization
Fig. 1a shows the powder XRD patterns of the SnSe0.93 + x wt% Cs2TiCl6 samples (x = 0, 0.25, 0.5, 0.75). All dominant diffraction peaks are indexed to orthorhombic SnSe (PDF#48-1224, space group Pnma), accompanied by weak reflections attributable to metallic Sn. The pronounced deviation in relative intensity between the (111) and (400) reflections compared with the standard pattern indicates a strong preferred orientation (texture), consistent with the intrinsic layered anisotropy of SnSe and the directional grain alignment promoted during consolidation.74 For the (400) crystal plane, its texture coefficient F400 is greater than 0, indicating the presence of a pronounced [100] preferred orientation. Within the instrumental detection limit (∼1–2 wt%), no crystalline Ti, Cl, or Cs containing secondary phase is discerned. | ||
| Fig. 1 Crystal structure and crystallographic analysis of SnSe0.93 + x wt% Cs2TiCl6. (a) Crystal structure of SnSe at room temperature. (b) Rietveld refinement results of SnSe0.93 + 0.5 wt% Cs2TiCl6. | ||
Rietveld refinement of the SnSe0.93 + 0.5 wt% Cs2TiCl6 sample (Fig. 1b) confirms SnSe as the major phase and quantifies a metallic Sn secondary phase of approximately 4.2 wt%. The formation of the secondary metallic Sn phase is primarily attributed to the initial stoichiometric Se deficiency (SnSe0.93) and the partial Sn segregation that is difficult to completely suppress under the rapid micro-melting process. Although it has the aforementioned adverse effects on thermoelectric transport, its role should be viewed from a dialectical perspective. During the spark plasma sintering (SPS) process, this metallic Sn phase likely forms a transient liquid phase, which effectively promotes grain boundary sliding and particle rearrangement through a liquid-phase sintering mechanism. This plays a crucial and positive role in achieving a high relative density in the dense bulk material. Therefore, the presence of the metallic Sn phase can be regarded as a “trade-off” under the current processing conditions: while it introduces unfavorable parallel conductive pathways at the microscale, it contributes macroscopically to the high densification essential for achieving excellent mechanical properties and reliable performance measurements.75 Even at low volume fraction, metallic Sn is undesirable for thermoelectric performance because it can (i) partially short-circuit grains, diminishing the effective Seebeck coefficient through parallel conduction, while (ii) contributing minimally to phonon scattering owing to its limited dispersion scale. At room temperature, SnSe adopts a layered orthorhombic structure (Pnma) characterized by a stereochemically active 5s2 lone pair on Sn2+. This lone pair induces asymmetric coordination and a markedly distorted [SnSe7] polyhedral environment, resulting in pronounced bonding and elastic anisotropy.8,47,51 Such structural distortion underpins the well-documented dichotomy of nearly two-dimensional phonon transport versus more three-dimensionally percolating electronic transport in SnSe.47
Elemental mapping (TEM-EDS, Fig. 2) reveals local enrichment of Ti despite its concentration being below the XRD detection threshold. The solid-state micro-fusion route used here is intrinsically non-equilibrium; limited thermal dwell and diffusion distance mean that Cs2TiCl6 may not completely decompose and homogenize to the atomic level within the SnSe lattice. Instead, partial decomposition and restricted interdiffusion lead to nanoscale Ti-rich regions and associated segregation of decomposition products at grain boundaries or lattice defects.69 The surrounding elastic strain fields and compositional perturbations can locally modulate the electronic band structure, including valence (or near-valence) band flattening, which helps sustain a relatively high Seebeck coefficient even as carrier concentration increases. A trade-off is possible: transient or localized increases in lattice order (or reduced point-defect randomness) near these clusters may slightly elevate thermal conductivity in certain temperature windows, although the net multiscale defect scattering (discussed below) ultimately favors lattice thermal conductivity suppression.68
Transmission electron microscopy (TEM) provides deeper insight into these hierarchical features. A low-magnification TEM image of the SnSe0.93 + 0.5% wt Cs2TiCl6 specimen (Fig. 3a) displays a dense polycrystalline morphology. The corresponding HRTEM image (Fig. 3b) from Area 1 shows well resolved lattice fringes; the measured interplanar spacing of ∼2.86 Å in Area 2 matches the (400) planes of orthorhombic SnSe, consistent with XRD indexing. The SAED pattern (Fig. 3c) further corroborates the principal crystallographic orientations of SnSe without additional ordered secondary diffraction rings or spots. To elucidate defect structures, inverse fast Fourier transform (IFFT) reconstruction of Area 3 (Fig. 3d1) and geometric phase analysis (GPA, Fig. 3d2 and d3) highlight a high density of edge dislocations accompanied by periodic tensile and compressive strain lobes. These dislocations and their long-range strain fields generate broad-spectrum phonon scattering centers, analogous to effects reported in (Bi,Sb)2Te,72,76 Cu2Se,46 PbTe,77 Mg3Bi2,78 and single-crystal SnSe.50 Concurrently, twin boundaries identified in Area 4 of Fig. 3b act as coherent planar defects that can (i) scatter mid- to long-wavelength phonons while (ii) preserving charge carrier mobility due to minimal electronic potential discontinuity, and (iii) enhance mechanical robustness via impeded dislocation motion.38
Overall, the structural picture comprises: (1) a textured primary SnSe matrix with a minor metallic Sn fraction; (2) nanoscale Ti-rich segregations below XRD detectability; (3) dense dislocation arrays and associated anisotropic strain fields; and (4) coherent twin boundaries. This hierarchical defect architecture is poised to deliver multiscale phonon scattering (point defects and local strain for high-frequency modes; dislocations and twins for intermediate and long wavelengths) while mitigating excessive degradation of electronic transport. The combined effects rationalize the maintained Seebeck coefficient under elevated carrier density and lay the structural foundation for the transport behavior addressed in subsequent sections.
3.2 Analysis of chemical states
X-ray photoelectron spectroscopy (XPS) was employed to elucidate the chemical states of the constituent elements (Fig. 4). The survey spectrum of the SnSe0.93 + 0.5% wt Cs2TiCl6 sample (Fig. 4a) confirms the presence of Sn, Se, Cs, Ti, and Cl. All XPS spectra were calibrated by setting the C 1s peak to 284.8 eV. In the Sn 3d region (Fig. 4b), two spin–orbit doublets are resolved: peaks at 486.1 eV (3d5/2) and 494.4 eV (Sn 3d3/2) correspond to Sn2+, while the components at 487.4 eV and 495.7 eV are assigned to metallic Sn, consistent with the minor Sn secondary phase identified by XRD. The Se 3d spectrum (Fig. 4c) exhibits Se 3d5/2 and Se 3d3/2 features at 54.1 eV and 54.9 eV, characteristic of Se2−. The Cs 3d spectrum (Fig. 4d) shows Cs 3d5/2 and Cs 3d3/2 at 725.1 eV and 738.9 eV, consistent with Cs+. In the Ti 2p region (Fig. 4e), peaks at 459.2 eV (Ti 2p3/2) and 464.8 eV (Ti 2p1/2) are indicative of Ti4+. The Cl 2p spectrum (Fig. 4f) displays Cl 2p3/2 and Cl 2p1/2 at 194.9 eV and 198.9 eV, assignable to Cl−. Collectively, these data confirm the coexistence of Sn2+, Se2−, Cs+, Ti4+, Cl−, and a metallic Sn fraction.Given the effective ionic radii of Ti4+ (∼0.61 Å)79 and Sn2+ (∼1.18 Å),54 substitution of Ti4+ on Sn sites is plausible, forming donor defects denoted as 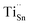 . Each
. Each 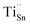 contributes extra electrons, thereby increasing the electron carrier concentration n.68 Similarly, the presence of Cl− implies partial substitution on Se2− sites, generating single-donor defects
contributes extra electrons, thereby increasing the electron carrier concentration n.68 Similarly, the presence of Cl− implies partial substitution on Se2− sites, generating single-donor defects 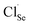 .80 The combined donor action of
.80 The combined donor action of 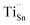 and
and 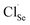 constitutes the primary origin of the enhanced n-type conductivity observed in subsequent transport measurements. A portion of the charge compensation may also involve a reduced concentration of native acceptor defects (e.g. Sn vacancies), further reinforcing electron density. The large ionic radius of Cs+ (∼1.67 Å)81 relative to both Sn2+ and Se2− renders substitutional incorporation into the SnSe lattice energetically unfavorable. Cs is therefore expected to segregate preferentially at grain boundaries or interfacial defect regions. While Cs+ does not directly donate additional free electrons in this context, its interfacial presence can (i) modify local electrostatic potentials, (ii) introduce elastic and mass contrast, and (iii) contribute to phonon scattering, thereby assisting in the reduction of lattice thermal conductivity κlat without severely compromising carrier mobility.
constitutes the primary origin of the enhanced n-type conductivity observed in subsequent transport measurements. A portion of the charge compensation may also involve a reduced concentration of native acceptor defects (e.g. Sn vacancies), further reinforcing electron density. The large ionic radius of Cs+ (∼1.67 Å)81 relative to both Sn2+ and Se2− renders substitutional incorporation into the SnSe lattice energetically unfavorable. Cs is therefore expected to segregate preferentially at grain boundaries or interfacial defect regions. While Cs+ does not directly donate additional free electrons in this context, its interfacial presence can (i) modify local electrostatic potentials, (ii) introduce elastic and mass contrast, and (iii) contribute to phonon scattering, thereby assisting in the reduction of lattice thermal conductivity κlat without severely compromising carrier mobility.
3.3 Electronic transport properties
Fig. 5 summarizes the electrical transport behavior of the Cs2TiCl6-doped SnSe0.93 ceramics. Incorporation of trace amounts of Cs2TiCl6 markedly enhances the electrical conductivity relative to the pristine sample over the entire temperature range (Fig. 5a). The electrical conductivity of all samples initially decreases but subsequently increases within the temperature range of ∼475–675 K, which is indicative of a phase transition in SnSe occurring within this interval.51 At 323 K, the sample containing 0.5 wt% Cs2TiCl6 attains σ ∼17.71 S cm−1, corresponding to a 153.7% increase over pristine SnSe0.93. At 823 K, this composition also maintains the highest conductivity (∼35.9 S cm−1). Hall measurements (Fig. 5b) reveal a monotonic increase in carrier concentration with rising Cs2TiCl6 content, reaching ∼9.7 × 1018 cm−3 for 0.75 wt%, accompanied by a concomitant decline in mobility µ. Hence, the net enhancement of σ is primarily driven by the elevated n. The [TiCl6]2− octahedral framework in Cs2TiCl6, constructed via corner-sharing Cl− ions,64 introduces effective electron donors into the SnSe lattice, thereby increasing carrier density.As shown in Fig. 5c, the absolute Seebeck coefficient |S| is moderately improved relative to the pristine sample despite the higher carrier concentrations. The 0.75 wt% Cs2TiCl6 sample delivers an S of ∼144.9 µV K−1 at 323 K, a 44.9% enhancement over the undoped counterpart. DFT calculations69 indicate that incorporation of Ti 3d states induces local band flattening, increasing the density-of-states effective mass m*, which offsets the usual decrease of S at elevated n. Pisarenko analysis at 300 K based on the single parabolic band (SPB) model82 further confirms that m* is significantly larger in the doped samples. Accordingly, the improvement in S is attributed to the increased m*. The weighted mobility µW of SnSe0.93 + x wt% Cs2TiCl6 (x = 0, 0.25, 0.5, 0.75) (Fig. 5e) is likewise enhanced after Cs2TiCl6 incorporation, indicating superior overall electronic transport. The formula for the weighted mobility is as follows:83
3.4 Thermal conductivity and ZT
Fig. 6 shows the evolution of thermal transport in SnSe0.93 + x wt% Cs2TiCl6. Upon light Cs2TiCl6 incorporation, the total thermal conductivity (κtot) first exceeds that of pristine SnSe0.93 and then decreases with further dopant addition, reaching ∼0.36 W m−1 K−1 at 823 K for x = 0.75 (Fig. 6a). The electronic contribution (κe) (Fig. 6b) rises monotonically with dopant content, consistent with the enhanced electrical conductivity, indicating that the net reduction of κtot at higher content is governed by suppression of the lattice term κlat (κtot = κe + κlat). As temperature increases, κlat decreases for all samples. Although the room-temperature κlat values of the doped specimens are slightly higher than that of pristine SnSe0.93, they decline progressively with increasing Cs2TiCl6 loading. The 0.75 wt% sample shows a room-temperature κlat of ∼0.93 W m−1 K−1, comparable to the pristine value (∼0.92 W m−1 K−1). At 823 K, κlat is reduced from ∼0.45 W m−1 K−1 (pristine) to ∼0.32 W m−1 K−1 (doped), corresponding to a 40.6% decrease. EDS mapping (Fig. 2) reveals Ti-rich regions; at low temperature, the intrinsically higher thermal conductivity of Ti-containing domains can transiently elevate κlat.84,85 With increasing temperature, however, multi-scale structural perturbations—including dislocations, local strain fields, and twin boundaries (Fig. 3)—generate high-density phonon scattering centers,33,38,77,86 thereby overwhelming the initial increase and markedly depressing κlat.Benefiting from the concurrent optimization of carrier transport and lattice thermal resistance, the dimensionless figure of merit ZT is substantially improved across the full temperature span (Fig. 6c). A peak ZT of ∼1.2 at 823 K is achieved for the sample with 0.5 wt% Cs2TiCl6, representing a 179.1% enhancement relative to the pristine specimen. This value surpasses previously reported n-type polycrystalline SnSe-based materials (Fig. 6d),55,75,87,88 underscoring that trace lead-free perovskite (Cs2TiCl6) doping is an effective route to elevate the thermoelectric performance of n-type polycrystalline SnSe.
4. Conclusions
Trace incorporation (≤0.75 wt%) of lead-free Cs2TiCl6 into n-type polycrystalline SnSe0.93via an energy-efficient micro-fusion (micro-melting) route enables concurrent optimization of electronic and phononic transport. The isolated [TiCl6]2− octahedra act as an effective donor reservoir, generating TiSn and ClSe donor defects that markedly elevate electron concentration and enhance electrical conductivity. Simultaneously, hybridization involving localized Ti 3d states induces local band (DOS) flattening, increasing the density-of-states effective mass while avoiding severe mobility degradation, thereby sustaining an improved Seebeck coefficient at higher carrier concentrations. Benefiting from this coupled carrier engineering, the 0.5 wt% sample delivers a maximum power factor of ∼532.1 µW m−1 K−2 at 823 K. In parallel, a hierarchically engineered microstructure—Ti-rich nanodomains, dense dislocations, strain fields, and twin boundaries—intensifies multi-scale phonon scattering, reducing the lattice thermal conductivity to ∼0.32 W m−1 K−1 (a ∼40.6% decrease relative to the pristine material). These synergistic effects culminate in a peak ZT of ∼1.2 at 823 K, corresponding to a ∼179.1% enhancement over undoped SnSe0.93. This study establishes trace, lead-free halide perovskite doping as a dual-function, environmentally benign, and scalable strategy for simultaneously modulating the band structure and suppressing phonon mean free paths in SnSe. The approach provides a generalizable paradigm for advancing other thermoelectric materials constrained by the conventional trade-off between carrier transport and lattice thermal conductivity.Author contributions
Wenjie Li: conceptualization, investigation, validation, methodology, formal analysis, data curation, and writing – original draft. Zeqing Hu: validation, methodology, and data curation. Ziwang Zhang: methodology and data curation. Zehao Lin: validation and methodology. Min Ruan: methodology. Jiahao Jiang: methodology and formal analysis. Jing Shuai: resources, writing – review and editing, visualization, supervision, project administration, and funding acquisition.Conflicts of interest
All authors declare no conflict of interest in this work.Data availability
All data generated or analyzed during this study are included in this published article. The datasets used and analyzed during the current study are available from the corresponding author on reasonable request. Detailed experimental methods (materials, synthesis of SnSe bulks); structural characterization (XRD, FE-TEM, XPS); characterization of SnSe-based samples at different doping concentrations; key parameters for all the bulk samples.Acknowledgements
This work was supported by the Guangdong Innovative and Entrepreneurial Research Team Program (grant no. 2021ZT09L227) and the National Natural Science Foundation of China (grant no. 52471247).References
- G. J. Snyder and E. S. Toberer, Nat. Mater., 2008, 7, 105–114 CrossRef
.
- B. Qin, M. G. Kanatzidis and L.-D. Zhao, Science, 2024, 386, eadp2444 CrossRef
.
- X. Shi and L. Chen, Nat. Mater., 2016, 15, 691–692 CrossRef PubMed
.
- X.-L. Shi, N.-H. Li, M. Li and Z.-G. Chen, Chem. Rev., 2025, 125, 7525–7724 CrossRef PubMed
.
- A. V. Powell, P. Vaqueiro, S. Tippireddy and J. Prado-Gonjal, Nat. Rev. Chem., 2025, 1–20 Search PubMed
.
- N. Nandihalli, D. H. Gregory and T. Mori, Adv. Sci., 2022, 9, e2106052 CrossRef PubMed
.
- D. Mao, Y. Zhou, Y. Yu, Y. Wang, M. Han, Q. Meng, Y. Lu, J. Feng, M. Kong, H. Yang, Q. Gan, X. Xu, L. Xie, G. W. Ho and J. He, Joule, 2024, 8, 3313–3323 CrossRef
.
- S. Byun, B. Ge, H. Song, S.-P. Cho, M. S. Hong, J. Im and I. Chung, Joule, 2024, 8, 1520–1538 CrossRef
.
- M. Rakshit, D. Jana and D. Banerjee, J. Mater. Chem. A, 2022, 10, 6872–6926 RSC
.
- D. Liu, D. Wang, T. Hong, Z. Wang, Y. Wang, Y. Qin, L. Su, T. Yang, X. Gao, Z. Ge, B. Qin and L.-D. Zhao, Science, 2023, 380, 841–846 CrossRef PubMed
.
- S. Wang, Y. Qiu, C. Chang and L.-D. Zhao, Sci. China Mater., 2024, 68, 780–784 CrossRef
.
- Y. Wang, S. Bai, H. Shi, Q. Cao, B. Qin and L.-D. Zhao, J. Mater. Chem. A, 2024, 12, 144–152 RSC
.
- S. Roychowdhury, T. Ghosh, R. Arora, M. Samanta, L. Xie, N. K. Singh, A. Soni, J. He, U. V. Waghmare and K. Biswas, Science, 2021, 371, 722–727 CrossRef PubMed
.
- J. Cai, R. Wang, S. Zhuang, F. Gao, M. Zhang, Z. Zhang, X. Tan, G. Liu and J. Jiang, Adv. Funct. Mater., 2024, 34, 2311217 CrossRef
.
- X. Yang, Y. Liu, R. Min, X. Jiang, Y. Liu, Y. Zhang and H. Chen, Acta Mater., 2025, 284, 120626 CrossRef
.
- Q. Zhang, Q. Song, X. Wang, J. Sun, Q. Zhu, K. Dahal, X. Lin, F. Cao, J. Zhou, S. Chen, G. Chen, J. Mao and Z. Ren, Energy Environ. Sci., 2018, 11, 933–940 RSC
.
- Y. Pei, H. Wang and G. J. Snyder, Adv. Mater., 2012, 24, 6125–6135 CrossRef CAS PubMed
.
- H. Xie, S. Hao, T. P. Bailey, S. Cai, Y. Zhang, T. J. Slade, G. J. Snyder, V. P. Dravid, C. Uher, C. Wolverton and M. G. Kanatzidis, J. Am. Chem. Soc., 2021, 143, 5978–5989 CrossRef CAS
.
- Y. Xiao, L. Xu, T. Hong, H. Shi, S. Wang, X. Gao, X. Ding, J. Sun and L.-D. Zhao, Energy Environ. Sci., 2022, 15, 346–355 RSC
.
- Y. Chen, M. Li, X. Wang, W. Chen, S. Liu, M. Zhang, W. Lyu, N. Li, H. Gao, W. Liu, X.-L. Shi and Z.-G. Chen, Nat. Commun., 2025, 16, 6796 CrossRef CAS PubMed
.
- S. Sepahban Shahgoli, M. Ozen, D. Zavanelli, G. Aliyeva, A. B. Burcak, U. Burkhardt, E. Isotta, G. J. Snyder and U. Aydemir, J. Mater. Chem. A, 2025, 13, 31740–31754 RSC
.
- H. Yang, B. Jia, L. Xie, D. Mao, J. Xia, J. Yang, M. Yuan, Q. Gan, X. Liu, M. Hu, J. Shuai and J. He, Joule, 2024, 8, 2667–2680 CrossRef CAS
.
- P. Ying, Q. Jian, Y. Gong, T. Song, Y. Yang, Y. Geng, J. Huang, R. Sun, C. Chen, T. Shen, Y. Li, W. Dou, C. Liang, Y. Liu, D. Xiang, T. Feng, X. Fei, Y. Zhang, K. Song, Y. Zhang, H. Wu and G. Tang, Nat. Commun., 2025, 16, 3305 CrossRef CAS
.
- X. Xu, D. Mao, M. O. Liedke, M. Butterling, J. Cui, J. Wang, Y. Luo, Z. Ge, E. Hirschmann, A. Wagner, J. He, K. Nielsch and R. He, Adv. Energy Mater., 2025, 15, 2500838 CrossRef CAS
.
- J. Zhong, X. Yang, T. Lyu, G. Liang, S. Zhang, C. Zhang, W. Ao, F. Liu, P. Nan, B. Ge and L. Hu, Sci. Bull., 2024, 69, 1037–1049 CrossRef CAS
.
- Z. Li, X. Yang, T.-E. Shi, Y.-W. Wang, W.-Q. Bao, S.-J. Huang, Y.-X. Zhang, J. Feng, Z.-H. Ge and L.-D. Zhao, J. Mater. Chem. A, 2025, 13, 30519–30530 RSC
.
- Y. Gu, W. Ai, J. Chen, A. Zhao, X. Hu, P. Zong, L. Pan, C. Lu, C. Wan and Y. Wang, J. Mater. Chem. A, 2023, 11, 1447–1454 RSC
.
- J. W. Li, H. Gao, Z. Han, J. Yu, H. L. Zhuang, L. Chen, H. Li, Y. Jiang, Z. Wang, Q. Zheng and J. F. Li, Adv. Mater., 2025, 2503665 CrossRef CAS PubMed
.
- Y. Sun, Y. Zhu, H. Wu, N. Qu, L. Xie, J. Zhu, Z. Liu, Q. Zhang, W. Cai, F. Guo and J. Sui, Energy Environ. Sci., 2024, 17, 738–747 RSC
.
- Z. Zhang, C. Ming, Q. Song, L. Wang, H. Chen, J. Liao, C. Wang, L. Chen and S. Bai, Acta Mater., 2025, 287, 120806 CrossRef CAS
.
- Q. Cao, J. Lyu, M. Yang, M. Yuan, J. Jiang, Z. Lin, Z. Zhao, J. Shuai and Y. Hou, Sci. China Mater., 2025 DOI:10.1007/s40843-025-3407-0
.
- B. Tian, Y. Liao, F. Xu, X. Qiu, F. Zhang and R. Ang, J. Mater. Chem. A, 2023, 11, 23319–23329 RSC
.
- S. Duan, X. Bao, J. Huang, R. Shi, L. Fei, W. Xue, H. Yao, X. Li, J. Wang, X. Liu, J. Mao, F. Cao, Y. Wang and Q. Zhang, Joule, 2025, 9, 101854 CrossRef CAS
.
- S. Ye, S. Zhi, X. Ma, X. Bao, P. Zhao, J. Cheng, S. Duan, C. Lin, Z. Wu, S. Chen, J. Qiu, L. Yin, X. Zhang, Y. Zhou, F. Jiang, Z. Chen, F. Cao, Y. Fu, Q. Zhang and J. Mao, Nat. Commun., 2025, 16, 1812 CrossRef CAS
.
- C. Fu, T. Zhu, Y. Liu, H. Xie and X. Zhao, Energy Environ. Sci., 2015, 8, 216–220 RSC
.
- Y. Li, S. Bai, Y. Wen, Z. Zhao, L. Wang, S. Liu, J. Zheng, S. Wang, S. Liu, D. Gao, D. Liu, Y. Zhu, Q. Cao, X. Gao, H. Xie and L. D. Zhao, Sci. Bull., 2024, 69, 1728–1737 CrossRef CAS
.
- J. W. Ha, V. Rathinam, E. H. Go and S. J. Hong, Adv. Eng. Mater., 2025, 27, 2400859 CrossRef CAS
.
- Q. Zhang, M. Yuan, K. Pang, Y. Zhang, R. Wang, X. Tan, G. Wu, H. Hu, J. Wu, P. Sun, G. Q. Liu and J. Jiang, Adv. Mater., 2023, 35, 2300338 CrossRef CAS
.
- J. Peng, D. Liu, S. Bai, Y. Wen, H. Liang, L. Su, X. Qian, D. Wang, X. Gao, Z. Ding, Q. Cao, Y. Pei, B. Qin and L. D. Zhao, Adv. Energy Mater., 2025, 15, 2404653 CrossRef CAS
.
- Y.-C. Shi, J. Yang, Y. Wang, Z.-G. Li, T.-Y. Zhong, Z.-H. Ge, J. Feng and J. He, Energy Environ. Sci., 2024, 17, 2326–2335 RSC
.
- J. Liu, F. Liu, Y. Li, B. Ying, Y. Wu, Z. Tang, Y. Cao, T. Zhu and C. Fu, Sci. China Mater., 2024, 68, 920–927 CrossRef
.
- E. Lee, J. Ko, J.-Y. Kim, W.-S. Seo, S.-M. Choi, K. H. Lee, W. Shim and W. Lee, J. Mater. Chem. C, 2016, 4, 1313–1319 RSC
.
- S. Ma, C. Li, P. Wei, W. Zhu, X. Nie, X. Sang, Q. Zhang and W. Zhao, J. Mater. Chem. A, 2020, 8, 4816–4826 RSC
.
- H. Hu, Y. Ju, J. Yu, Z. Wang, J. Pei, H.-C. Thong, J.-W. Li, B. Cai, F. Liu, Z. Han, B. Su, H.-L. Zhuang, Y. Jiang, H. Li, Q. Li, H. Zhao, B.-P. Zhang, J. Zhu and J.-F. Li, Nat. Mater., 2024, 23, 527–534 CrossRef CAS
.
- J. Yu, X. Liu, H. Hu, Y. Jiang, H.-L. Zhuang, H. Li, B. Su, J.-W. Li, Z. Han, Z. Wang, L. Chen, K. Hayashi, Y. Miyazaki, B. L. Mehdi and J.-F. Li, Joule, 2024, 8, 2652–2666 CrossRef CAS
.
- C. Xiao, L. Wu, K. Fang, Z. Xia, N. Jiang, B. Duan, X. Feng, L. Zhou, P. Zhai, G. Li and Q. Zhang, Acta Mater., 2025, 286, 120761 CrossRef CAS
.
- C. Chang, M. Wu, D. He, Y. Pei, C.-F. Wu, X. Wu, H. Yu, F. Zhu, K. Wang, Y. Chen, L. Huang, J.-F. Li, J. He and L.-D. Zhao, Science, 2018, 360, 778–783 CrossRef CAS
.
- Y. Xiao and L.-D. Zhao, Science, 2020, 367, 1196–1197 CrossRef CAS PubMed
.
- B. Qin, D. Wang, X. Liu, Y. Qin, J.-F. Dong, J. Luo, J.-W. Li, W. Liu, G. Tan, X. Tang, J.-F. Li, J. He and L.-D. Zhao, Science, 2021, 373, 556–561 CrossRef CAS
.
- H. Shi, Y. Wen, S. Bai, C. Chang, L. Su, T. Gao, S. Liu, D. Liu, B. Qin, Y. Qin, H. Liang, X. Qian, Z. Hou, X. Gao, T. Zhou, Q. Tan and L. D. Zhao, Nat. Commun., 2025, 16, 1788 CrossRef CAS PubMed
.
- L.-D. Zhao, S.-H. Lo, Y. Zhang, H. Sun, G. Tan, C. Uher, C. Wolverton, V. P. Dravid and M. G. Kanatzidis, Nature, 2014, 508, 373–377 CrossRef CAS PubMed
.
- B. Qin and L.-D. Zhao, Science, 2022, 378, 832–833 CrossRef CAS
.
- L. Su, D. Wang, S. Wang, B. Qin, Y. Wang, Y. Qin, Y. Jin, C. Chang and L.-D. Zhao, Science, 2022, 375, 1385–1389 CrossRef CAS PubMed
.
- Y. Gong, W. Dou, B. Lu, X. Zhang, H. Zhu, P. Ying, Q. Zhang, Y. Liu, Y. Li, X. Huang, M. F. Iqbal, S. Zhang, D. Li, Y. Zhang, H. Wu and G. Tang, Nat. Commun., 2024, 15, 4231 CrossRef CAS PubMed
.
- W.-H. Gu, Y.-X. Zhang, J. Guo, J.-F. Cai, Y.-K. Zhu, F. Zheng, L. Jin, J. Xu, J. Feng and Z.-H. Ge, J. Alloys Compd., 2021, 864, 158401 CrossRef CAS
.
- X. Zhang, Y. Wang, G. Zhang, C. Wang and Y. Yan, J. Alloys Compd., 2019, 772, 1061–1066 CrossRef CAS
.
- V. Q. Nguyen, T. H. Nguyen, V. T. Duong, J. E. Lee, S.-D. Park, J. Y. Song, H.-M. Park, A. T. Duong and S. Cho, Nanoscale Res. Lett., 2018, 13, 200 CrossRef
.
- R. Zhang, Z. Zhou, Q. Yao, N. Qi and Z. Chen, Phys. Chem. Chem. Phys., 2021, 23, 3794–3801 RSC
.
- C. Chang, D. Wang, D. He, W. He, F. Zhu, G. Wang, J. He and L. D. Zhao, Adv. Energy Mater., 2019, 9, 1901334 CrossRef
.
- T. Lanigan-Atkins, S. Yang, J. L. Niedziela, D. Bansal, A. F. May, A. A. Puretzky, J. Y. Y. Lin, D. M. Pajerowski, T. Hong, S. Chi, G. Ehlers and O. Delaire, Nat. Commun., 2020, 11, 4430 CrossRef CAS
.
- S. Chandra, U. Bhat, P. Dutta, A. Bhardwaj, R. Datta and K. Biswas, Adv. Mater., 2022, 34, e2203725 CrossRef PubMed
.
- H. Shi, L. Su, S. Bai, B. Qin, Y. Wang, S. Liu, C. Chang and L.-D. Zhao, Energy Environ. Sci., 2023, 16, 3128–3136 RSC
.
- L. Su, H. Shi, S. Wang, D. Wang, B. Qin, Y. Wang, C. Chang and L. D. Zhao, Adv. Energy Mater., 2023, 13, 2300312 CrossRef CAS
.
- Q. Mahmood, M. Hassan, N. Yousaf, A. A. AlObaid, T. I. Al-Muhimeed, M. Morsi, H. Albalawi and O. A. Alamri, Mater. Sci. Semicond. Process., 2022, 137, 106180 CrossRef CAS
.
- R. Ksouri, R. Maizi, A. G. Boudjahem, N. Cheghib, I. Djaghout and M. Derdare, Russ. J. Inorg. Chem., 2024, 69, 1370–1375 CrossRef CAS
.
- Y. Nouri, B. Hartiti, A. Batan, S. Fadili, A. Ziti, H. Labrim and P. Thévenin, Solid State Commun., 2023, 363, 115087 CrossRef CAS
.
- K. Bouferrache, M. A. Ghebouli, B. Ghebouli, M. A. Habila, T. Chihi, M. Fatmi, A. Djemli and M. Sillanpaa, Results Phys., 2024, 56, 107138 CrossRef
.
- F. Li, W. Wang, X. Qiu, Z. Zheng, P. Fan, J. Luo and B. Li, Inorg. Chem. Front., 2017, 4, 1721–1729 RSC
.
- J. Lu, L. Guo, G. Xiang, Y. Nie and X. Zhang, J. Electron. Mater., 2019, 49, 290–296 CrossRef
.
- T.-R. Wei, M. Guan, J. Yu, T. Zhu, L. Chen and X. Shi, Joule, 2018, 2, 2183–2188 CrossRef
.
- R. Li, W. Ou, J. Zhu, Q. Deng, X. Tan, Q. Zhao, T. Lu, S. Gao, H. Ma, H. Wu, Q. Sun and R. Ang, Chem. Eng. J., 2024, 497, 154624 CrossRef CAS
.
- R. Li, G. Wu, Q. Zhang, Q. Pan, M. Wang, K. Pang, L. Miao, X. Tan, P. Sun, H. Hu, J. Wu, G. Q. Liu and J. Jiang, Adv. Funct. Mater., 2025, 2502535 CrossRef CAS
.
- H.-S. Kim, Z. M. Gibbs, Y. Tang, H. Wang and G. J. Snyder, APL Mater., 2015, 3, 041506 CrossRef
.
- P. P. Shang, J. Dong, J. Pei, F. H. Sun, Y. Pan, H. Tang, B. P. Zhang, L. D. Zhao and J. F. Li, Research, 2019, 9253132 CAS
.
- X. Yang, X.-Y. Ma, T.-E. Shi, W.-Q. Bao, J. Wang, Z.-Y. Wang, Y.-X. Zhang, J. Feng and Z.-H. Ge, Ceram. Int., 2024, 50, 20515–20524 CrossRef CAS
.
- W. Chen, X.-L. Shi, M. Li, T. Liu, Y. Mao, Q. Liu, M. Dargusch, J. Zou, G. Q. M. Lu and Z.-G. Chen, Science, 2024, 386, 1265–1271 CrossRef CAS PubMed
.
- B. Jia, D. Wu, L. Xie, W. Wang, T. Yu, S. Li, Y. Wang, Y. Xu, B. Jiang, Z. Chen, Y. Weng and J. He, Science, 2024, 384, 81–86 CrossRef CAS PubMed
.
- P. Zhao, W. Xue, Y. Zhang, S. Zhi, X. Ma, J. Qiu, T. Zhang, S. Ye, H. Mu, J. Cheng, X. Wang, S. Hou, L. Zhao, G. Xie, F. Cao, X. Liu, J. Mao, Y. Fu, Y. Wang and Q. Zhang, Nature, 2024, 631, 777–782 CrossRef CAS PubMed
.
- D. Pang, M. Chen, T. Zhang and G. Huang, J. Electroceram., 2025, 1–12 Search PubMed
.
- X. Yang, W.-H. Gu, W.-J. Li, Y.-X. Zhang, J. Feng and Z.-H. Ge, J. Phys. Chem. Solids, 2023, 172, 111077 CrossRef CAS
.
- D. Liu, W. Du, X. Liu, R. Tian and H. Li, J. Phys. Chem. C, 2019, 123, 2157–2164 CrossRef CAS
.
- J. Zhu, X. Zhang, M. Guo, J. Li, J. Hu, S. Cai, W. Cai, Y. Zhang and J. Sui, npj Comput. Mater., 2021, 7, 116 CrossRef CAS
.
- G. J. Snyder, A. H. Snyder, M. Wood, R. Gurunathan, B. H. Snyder and C. Niu, Adv. Mater., 2020, 32, e2001537 CrossRef
.
- E. B. Dolgusheva, Phys. Solid State, 2023, 64, 489–492 CrossRef
.
- X. Guo, Y. Liu, X. Tian, Z. Tao, X. Yan and Z. Liu, RSC Adv., 2023, 13, 6075–6086 RSC
.
- J. Guo, Y.-X. Zhang, Z.-Y. Wang, F. Zheng, Z.-H. Ge, J. Fu and J. Feng, Nano Energy, 2020, 78, 105227 CrossRef CAS
.
- W.-j. Li, W.-H. Gu, J. Guo, Y.-X. Zhang, J. Feng, Q. Shan and Z.-H. Ge, J. Solid State Chem., 2022, 310, 123037 CrossRef CAS
.
- H. Liu, S. Zhang, Y. Zhang, S. Zong, W. Li, C. Zhu, F. Luo, J. Wang and Z. Sun, ACS Appl. Energy Mater., 2022, 5, 15093–15101 CrossRef CAS
.
| This journal is © The Royal Society of Chemistry 2025 |