Advancing HT-PEM fuel cell technology: durability and performance under start–stop conditions
Nedjeljko
Seselj
a,
Lars N.
Cleemann
a,
Tomas
Torres
 bc and
Kobra
Azizi
bc and
Kobra
Azizi
 *a
*a
aBlue World Technologies, Egeskovvej 6C, Kvistgaard 3490, Denmark. E-mail: kazizi@agcbio.com; azizikobra@ymail.com
bDepartment of Organic Chemistry, Institute for Advanced Research in Chemical Sciences (IAdChem), Universidad Autónoma de Madrid (UAM), Madrid 28049, Spain
cIMDEA-Nanoscience, c/Faraday, 9, Ciudad Universitaria de Cantoblanco, Madrid 28049, Spain
First published on 14th October 2025
Abstract
High-temperature proton exchange membrane (HT-PEM) fuel cells represent a promising avenue for efficient and low-emission energy conversion, especially in automotive and distributed power applications. However, one of the critical challenges limiting their widespread deployment is performance degradation under dynamic operating conditions, particularly during frequent start–stop cycles. This review provides a comprehensive overview of recent advances in understanding and improving the durability and performance of HT-PEM fuel cells under these conditions. Key degradation mechanisms—such as catalyst layer degradation, membrane instability, and interfacial delamination—are examined alongside mitigation strategies, including novel material development, system design optimization, and operational control techniques. In addition, the review analyzes the coupling among these mechanisms, for instance, how membrane dehydration accelerates catalyst corrosion through acid redistribution, and how delamination locally distorts current density and mass transfer pathways, thus providing a more integrated degradation picture. The review highlights recent experimental and modeling studies that shed light on transient behavior and long-term reliability. Special emphasis is placed on recent disruptive technologies, such as hybrid inorganic–organic membranes, multifunctional catalysts, and digital-twin-based lifetime prediction models. By identifying current gaps and emerging solutions, this work aims to guide future research efforts and support the development of robust, high-performance HT-PEM systems suitable for real-world applications.
1. Introduction
The global challenges associated with energy consumption and climate change—largely driven by the use of fossil fuels—have prompted extensive research into clean, efficient, and sustainable energy technologies. Among the various alternatives, hydrogen and fuel cell (FC) technologies have been widely recognized as promising solutions and have matured to the point of practical implementation in both stationary and portable power systems.1Polymer electrolyte membrane fuel cells (PEMFCs) represent the dominant FC technology in automotive applications and are commercially available from multiple automotive manufacturers. These systems typically employ perfluorosulfonic acid (PFSA)-based membranes, such as Nafion, and are commonly referred to as low-temperature PEMFCs (LT-PEMFCs). PFSA membranes offer high proton conductivity, excellent chemical stability, mechanical robustness, and long-term durability; however, their operation is limited to highly hydrated conditions. As a result, LT-PEMFCs are generally restricted to operating temperatures around 80 °C under ambient pressure to ensure adequate membrane hydration.2
This temperature constraint introduces several operational challenges, including complex thermal and water management requirements, limited tolerance to fuel and oxidant impurities, and the need for a supporting hydrogen infrastructure. Water management becomes particularly critical at these temperatures, as the system operates near the boiling point of water, leading to a two-phase regime where water vaporizes at low humidity and condenses at high humidity. Thermal management is also demanding, since the required cooling capacity is determined by the ratio of heat generated by the stack to the temperature differential between the stack and the surrounding environment (Q/ΔT), necessitating effective cooling strategies within the 60–80 °C range.3
Furthermore, at these lower operating temperatures, catalyst poisoning due to contaminants such as CO, NOx, or sulfur species present in reformate fuel or ambient air poses a significant challenge. These limitations, along with inherently slower electrode reaction kinetics, are expected to improve at elevated temperatures.1
High-temperature PEMFCs (HT-PEMFCs), based on polybenzimidazole (PBI) membranes doped with phosphoric acid (PA), have gained increasing attention in this context. PBI is a heat-resistant, heterocyclic thermoplastic, while PA provides high intrinsic proton conductivity, thermal stability, and low volatility. HT-PEMFCs typically operate within the 150–200 °C range and demonstrate a markedly higher tolerance to carbon monoxide—up to approximately 3% in the fuel stream—compared to the 10–100 ppm limit for LT-PEMFCs at 80 °C. This high impurity tolerance is considered one of the most advantageous features of HT-PEMFCs, enabling operation with hydrogen-rich reformate gases containing CO, residual methanol, and other contaminants.4
Nevertheless, a key limitation of HT-PEMFCs is the sluggish oxygen reduction reaction (ORR) in the presence of PA, which strongly adsorbs onto the platinum (Pt) catalyst surface, a phenomenon well-documented in PA fuel cells (PAFCs). Consequently, HT-PEMFCs are generally characterized by lower power densities.
In addition to their use in stationary and remote power systems, HT-PEMFCs integrated with methanol reformers (Fig. 1A) have been explored for a range of other applications, including hydrogen purification.5 Among the various challenges facing the broader deployment of this technology, durability remains the most critical barrier to widespread commercialization.
 | ||
| Fig. 1 (A) Concept drawing of a commercial HT-PEMFC system,6 Reproduced with permission.7 Copyright 2019, Wiley-VCH. (B) Schematic of the HT-PEMFC electrode with the PA diffusing and leaching from the membrane [reproduced from ref. 8] (C) a representative start–stop cycle involving gas flow control, thermal and load cycling, and potential degradation mitigation by flushing. (D) Comparison of dynamic degradation with/without mitigation strategy, based on cycling profile represented in (C). | ||
During operation, all components of a fuel cell undergo performance degradation, which may be reversible or irreversible, ultimately resulting in a decline in power output or complete system failure. Chemically, these components are subjected to highly aggressive environments characterized by strong oxidizing and reducing conditions, as well as the presence of reactive species such as acids and radicals, particularly under realistic electrochemical potentials and elevated temperatures.
From a materials perspective, degradation phenomena include catalyst nanoparticle (NP) dissolution and sintering, corrosion of catalyst supports, polymer oxidation, loss of the acid dopant, and the degradation of various other structural and functional components. The durability of these materials is strongly affected by key operational parameters, such as temperature, pressure, fuel impurity levels, cell voltage and current, as well as thermal cycling, load cycling, and start–stop operations. The coupling of these degradation routes often amplifies damage, for example, polymer oxidation can enhance PA loss, which in turn accelerates Pt dissolution and interfacial delamination, highlighting the need for multi-scale degradation models.
At the system level, factors such as fuel supply fluctuations, delayed heat removal, and uneven gas distribution during transient operation can induce localized hot spots and current imbalances, further accelerating degradation.
The mechanisms of degradation, along with experimental methodologies for monitoring component aging and the influence of operational strategies, have been extensively discussed in the broader context of PEMFC technologies.9 In HT-PEMFC systems, the combination of PA and high operating temperatures exacerbates degradation processes. Previous studies have addressed the general durability challenges associated with this technology.4
Recent progress in the field has introduced multi-physics modeling frameworks coupling electrochemical kinetics, heat and mass transfer, and mechanical stress analysis to predict local hot-spot formation and stress concentration, offering quantitative insight into degradation under start–stop cycling.6,10–12 The present study offers a comprehensive examination of durability and mitigation strategies for HT-PEMFCs employing PA-doped polybenzimidazole (PBI) membranes under dynamic operating conditions, particularly start–stop cycling. Special emphasis is placed on recent advances in understanding the underlying mechanisms of material degradation in these conditions, as well as progress made in enhancing the long-term performance and operational lifespan of the technology.
Fuel cells (FCs) experience significantly greater stress when operated under dynamic conditions, including thermal, load, and start–stop cycling. These operating modes subject the cathode to elevated potentials, which can result in severe corrosion of carbon supports and agglomeration of catalyst NPs. Additionally, the repeated thermal expansion and contraction of the membrane and electrode layers induce mechanical stress at their interfaces, compromising the structural integrity of the membrane electrode assembly (MEA).
Dynamic operation also influences the distribution and behavior of PA within the system. Variations in water content can alter both the concentration and volume of PA in the membrane and electrodes, affecting proton conductivity and potentially leading to performance degradation. Furthermore, during start–stop cycles, liquid water may be present at lower temperatures, which is believed to accelerate acid leaching from the membrane, a known degradation mechanism.1
To support long-term deployment, durability targets have been defined by regulatory and funding bodies. The European Fuel Cell and Hydrogen 2 Joint Undertaking (FCH 2 JU), for example, has established a target operational lifetime exceeding 15 years (approximately 130![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 h) for stationary micro combined heat and power (m-CHP) systems. In the transportation sector, durability targets range from 6000 hours for passenger vehicles (with allowable power degradation under 10%) to 15
000 h) for stationary micro combined heat and power (m-CHP) systems. In the transportation sector, durability targets range from 6000 hours for passenger vehicles (with allowable power degradation under 10%) to 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hours for fuel cell buses.
000 hours for fuel cell buses.
Regardless of the application, stationary or automotive, ensuring stable and reliable performance under dynamic operating conditions is essential for the commercial viability of fuel cell technologies. To this end, the present review integrates key milestones from recent research, including system-integrated studies on transient fuel13 and heat management13 and cross-linked PA-retentive membranes,9 to ensure full coverage of recent high-impact contributions.
This review presents a comprehensive analysis of recent progress in enhancing the durability and performance of HT-PEMFCs under start–stop operating conditions. It addresses critical degradation phenomena, including catalyst layer deterioration, membrane instability, and interfacial delamination, while also discussing corresponding mitigation approaches such as the development of advanced materials, improvements in system architecture, and refined operational control strategies. Furthermore, the review synthesizes findings from recent experimental investigations and modeling efforts that elucidate the transient responses and long-term reliability of HT-PEMFC systems. By identifying existing knowledge gaps and highlighting emerging solutions, this work seeks to inform future research directions and facilitate the advancement of durable, high-performance HT-PEMFC technologies for real-world deployment.
2. HT-PEM fuel cell fundamentals
HT-PEMFCs utilize PBI membranes doped with PA to enable proton conduction without the need for liquid water. This configuration supports operation at elevated temperatures, typically ranging from 120 to 200 °C. Operating under these conditions offers several advantages, including improved utilization of waste heat, more effective water and thermal management, and significantly enhanced tolerance to impurities in the reactant gases.13In addition to the component-level advantages, recent studies have demonstrated that high-temperature operation also simplifies stack thermal management through enhanced temperature uniformity and reduced humidity control requirements.14 These system-level improvements translate to higher reliability and reduced balance-of-plant complexity,15 particularly in automotive applications experiencing rapid load changes.
Notably, this increased impurity tolerance allows the direct use of hydrogen-rich reformate gases, derived from methanol, natural gas, propane, or ammonia cracking, with minimal or, in some cases, no gas purification. This flexibility facilitates integration with a variety of fuel reformers and helps reduce system complexity and overall costs. Moreover, the ability to accommodate diverse fuel sources contributes to greater fuel flexibility,4 which is a key consideration for infrastructure development and widespread adoption.
Recent comparative studies have highlighted the influence of dopant chemistry and polymer architecture on conductivity and stability under dynamic conditions. For instance, phosphonated poly(ether ether ketone) (SPEEK) and PBI composites achieve proton conductivities of 0.08–0.12 S cm−1 at 160 °C, comparable to PA-doped PBI membranes.16,17 However, their acid retention and mechanical integrity remain inferior beyond 2000 hours of cycling, reinforcing the current dominance of PA-PBI systems.
As a result of their favorable properties, HT-PEMFCs with PA-doped PBI membranes have garnered growing interest. PBI is a class of thermally stable heterocyclic thermoplastics,18 while PA serves as a dopant that imparts high intrinsic proton conductivity, thermal stability, and low volatility.19 Among the various PBI derivatives, poly(2,5-benzimidazole) (AB-PBI), shown in its PA-doped form in represents the simplest polymer structure within this family. However, other forms such as poly(2,2′-(m-phenylene)-5,5′-bibenzimidazole) (m-PBI) and poly(2,2′-(p-phenylene)-5,5′-bibenzimidazole) (p-PBI) are more commonly employed due to their superior solubility in organic solvents and improved processability, Fig. 3A.1
Acid-doped PBI membranes are typically fabricated using one of two approaches. The first involves casting membranes from a solution of the polymer in an organic solvent, followed by post-doping in an aqueous PA solution. The second method entails direct casting from a polymer solution in polyphosphoric acid (PPA) via a sol–gel process, allowing in situ doping during membrane formation.
Further progress has been achieved using hybrid membranes that incorporate ionic liquids or nanofillers to stabilize PA uptake and reduce volatility under thermal cycling.18,20 These systems have demonstrated up to 30% lower acid loss over 1000 hours at 170 °C compared to conventional m-PBI.
The catalyst layers in HT-PEMFCs are similar to those used in low-temperature PEMFCs, consisting of carbon-supported Pt or Pt-alloy NPs dispersed with a polymeric binder. These layers are applied onto a porous substrate known as the gas diffusion layer (GDL), Fig. 1B. To enhance resistance to liquid penetration, a microporous layer is typically inserted between the catalyst layer and the GDL, improving water management and gas transport.21
When operating on methanol reformate, typically composed of approximately 69.2% H2, 22.3% CO2, 1.4% CO, and 6.9% H2O at ambient pressure, the state-of-the-art (SoA) performance of commercially available MEAs, such as those from Dapozol or Celtec, reaches around 0.67–0.70 V at a current density of 0.2 A cm−2. Peak power densities in the range of 0.45–0.50 W cm−2 are achieved at approximately 1.0 A cm−2, with relatively high platinum loadings of 0.7–1.0 mg cm−2 per electrode.22
Recent benchmark data from European research programs and industry demonstrate continued progress in durability and efficiency: Celtec-based MEAs have demonstrated multi-thousand-hour durability at 160 °C under reformed methanol operation, while new carbon-nanocage-supported PtCo catalysts achieve 20% higher mass activity.23 These results illustrate the advances in both MEA design and electrode architecture over the past three years.
Since the initial report of this technology in 1995,24 significant advancements have been made in HT-PEMFC systems, particularly in terms of their operational characteristics. These include minimal requirements for external humidification, high tolerance to carbon monoxide, and improved compatibility with fuel processing units due to simplified system integration. Moreover, the elevated operating temperature offers the potential for more effective utilization of waste heat, thereby enhancing overall system efficiency.
Beyond these macroscopic benefits, multi-physics simulation tools have recently been employed to quantify the interactions between electrochemical kinetics, heat generation, and mechanical stress under dynamic conditions. These studies reveal that localized hot spots and stress gradients can promote PA migration and membrane cracking, offering a predictive framework for lifetime optimization through thermal and load management strategies.25
3. Start–stop operation: challenges and implications
FCs experience significantly increased stress during dynamic operating modes, such as start–stop cycling that include thermal cycling and load cycling, Fig. 1C. These dynamic cycles expose the cathode to elevated potentials, which accelerates carbon support corrosion and leads to catalyst particle growth. Furthermore, repeated thermal expansion and contraction of membranes and electrodes generate mechanical stresses at the interfaces, compromising the integrity of the membrane electrode assembly (MEA). Variations in water content can alter the concentration and volume of PA within both membranes and electrodes. Additionally, during start–stop cycles, the presence of liquid water at lower temperatures is believed to exacerbate acid leakage.1Coupled degradation mechanisms are increasingly recognized as the dominant failure drivers under these conditions. For instance, recent in situ EIS and μ-CT studies demonstrated that membrane acid depletion can locally elevate cathode potential and accelerate carbon corrosion. Similarly, interfacial delamination induces non-uniform current density distributions that promote further acid migration and Pt dissolution. These findings underscore the synergistic nature of mechanical and chemical degradation pathways.26
Investigating degradation behavior under dynamic conditions is essential because fuel cells operate under varying loads in real-world scenarios, Fig. 1D. Yeon et al. evaluated multiple test protocols, including constant operation, start–stop cycling, and load cycling.27 Their findings demonstrated that catalyst degradation was most severe under load cycling, while membrane degradation was predominantly driven by start–stop cycling. Araya et al.28 proposed a novel method to mitigate PA loss by incorporating relaxation periods into the load cycling protocol. Start–stop cycling is widely recognized to induce durability challenges primarily through localized corrosion caused by high cathode potentials and electrolyte loss due to residual products. Leader et al. further reported that electrolyte redistribution, driven by temperature fluctuations during dynamic operation, results in volume expansion and contraction, contributing to degradation.29
To link these laboratory insights with practical applications, recent studies have evaluated system-integration aspects such as purge timing control and thermal delay compensation. Such control-oriented approaches reduce the frequency of reverse-current events by up to 70%, highlighting the growing importance of digital monitoring and adaptive start-up algorithms in stack-level durability.30,31
Given the flexibility of HT-PEMFCs with respect to fuel purity, recent research has increasingly examined performance with various reformed gas compositions. The mixed nature of reformed fuel gases, particularly from steam reforming, strongly influences fuel cell performance. For instance, synthetic reformate gas mixtures containing H2/CO2/CH4/CO (80/16/2/2 vol%) under start–stop cycling conditions have been shown to increase activation overpotentials and accelerate degradation.32 Zhou et al. investigated degradation using simulated reformate fuels composed of H2/H2O/methanol mixtures (79.11/17.95/2.94 vol%) and observed accelerated increases in ohmic and mass transport resistances.13 Numerous studies have also explored degradation mechanisms extensively.1,33
At the system level, fuel supply fluctuations and thermal lag within reformers are now recognized as major contributors to stack voltage instability under dynamic loads. Advanced control schemes using real-time feedback of temperature and gas composition have been proposed to mitigate these transients and extend stack life by stabilizing reactant distribution.34
Inert gases such as nitrogen constitute a notable fraction of reformed gases, approximately 27% in propane reformate, over 50% in natural gas reformate,35,36 and between 25% and 31.7% in ammonia-based fuel cell systems.37,38 Zhou et al.39 demonstrated that hydrogen dilution with nitrogen at concentrations between 20% and 50% significantly impairs fuel cell performance. Similar trends are reported in the literature, where decreasing hydrogen purity correlates with increased charge transfer and mass transport resistances.13 This dilution effect leads to immediate operational impacts and potentially contributes to accelerated long-term degradation of electrochemical performance and material stability. The versatility of HT-PEMFCs in utilizing diverse hydrogen sources, including those derived from ammonia, natural gas, and propane, underscores their strategic role in the transition toward cleaner and more efficient energy systems. Understanding the impact of dynamic stressors such as start–stop cycling, load cycling, and fuel dilution is vital for improving the durability and operational lifespan of these fuel cells.
In addition to conventional mitigation methods, emerging concepts such as self-healing acidic composite membranes, AI-assisted thermal control, and digital twin stack models are being explored to predict and counteract degradation.40–42 These disruptive approaches couple electrochemical, thermal, and mechanical domains to forecast localized stress and optimize operational parameters for extended durability.
Because operating fuel cells under normal conditions for thousands of hours is often impractical and costly, accelerated stress tests (ASTs) have been developed to expedite the evaluation of key materials and degradation pathways. The primary goal of ASTs is to assess the durability and performance of fuel cell components under controlled, intensified conditions to enable comparability of results. For HT-PEMFCs, AST protocols generally fall into four categories: (1) start–stop cycling; (2) open circuit voltage (OCV) hold; (3) load cycling at high or low current densities; and (4) thermal cycling.1,43
Start–stop protocols typically involve a single cycle of cell cold start, steady operation, and shutdown, with voltage degradation per cycle serving as a key durability metric. Load cycling tests are subdivided into low-load (0–0.2 A cm−2) and high-load (0.2–1.0 A cm−2) regimes, targeting catalyst/electrode and membrane-electrolyte evaluation, respectively.44 Low-load operation often includes exposure to OCV conditions, which occur naturally during fuel cell idling. Cells with effective sealing maintain elevated OCV during startup and cooldown, promoting carbon corrosion and Pt NP agglomeration. Thermal cycling subjects materials to repeated temperature variations, with high temperatures accelerating carbon corrosion, metal dissolution, and acid evaporation, while temperatures below 100 °C promote PA leaching due to liquid water washout.
To improve clarity and practical relevance, it is worth noting that the European Clean Hydrogen JU (2024) recommends standardized AST definitions: for instance, start–stop cycling between 0.4 V and 1.2 V for 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, load cycling between 0.1 A cm−2 and 0.8 A cm−2 for 1000 hours, and thermal cycling between 120 °C and 180 °C for 500 cycles. These targets harmonize HT-PEMFC durability assessment with DOE and EU benchmarks, promoting consistent comparability across laboratories.
000 cycles, load cycling between 0.1 A cm−2 and 0.8 A cm−2 for 1000 hours, and thermal cycling between 120 °C and 180 °C for 500 cycles. These targets harmonize HT-PEMFC durability assessment with DOE and EU benchmarks, promoting consistent comparability across laboratories.
In situ characterization methods are essential for monitoring these degradation phenomena in real time. Besides polarization (i–V) curves, electrochemical impedance spectroscopy (EIS) is particularly effective in tracking resistance changes and identifying the most affected cell components during dynamic operations.45 Additional post-mortem analyses such as scanning electron microscopy (SEM) for membrane and catalyst layer thinning, X-ray diffraction (XRD) and transmission electron microscopy (TEM) for Pt NP growth, micro-computed tomography (μ-CT) for structural analysis, X-ray photoelectron spectroscopy (XPS) for interfacial chemistry, and titration for PA doping level are widely employed to elucidate degradation mechanisms.45–47 To consolidate scattered experimental data, a comprehensive summary table comparing degradation rates and conditions has been added (Table 1). This table integrates key studies published until 2025, listing degradation rates in mV per cycle, along with test duration and reference.
| Test conditions | Lifetime | Degradation | Year | Ref. | |
|---|---|---|---|---|---|
| 150 °C, H2/air, daily start/stop (7/17 h) | 860 cycles, 3.5 years | 300 μV per cycle | 2004 | 48 | |
| 160 °C, daily on/off (12 h/12 h) | 240 cycles, 6000 h | 11 μV h−1, 200 μV per cycle | 2008 | 49 | |
| 160 °C | 2 min at OCV | 500 cycles | 34 μV per cycle | 2008 | 50 |
| 30 min at 0.2 A cm−2 | 28 μV per cycle | ||||
| 30 min at 0.6 A cm−2 | 41 μV per cycle | ||||
| 160 °C, load cycling 0.2–0.4 A cm−2 and start/stop, Celtec®P1000 | 24 cell stack | 658 h | 100 μV h−1 | 2010 | 51 |
| Single cell | 1105 h | 40 μV h−1 | |||
| 180 °C, simulated start–stop cycling, 0.2 A cm−2 | 157 cycles, 4000 h | 19 μV h−1, 480 μV per cycle | 2011 | 52 | |
| Thermal cycling 30 °C/30 min–180 °C/30 min | PBI membrane | 150 h | 14 μV h−1 | 2014 | 53 |
| PBI-PTFE composite membrane | 2 μV h−1 | ||||
| 165 °C, start/stop cycles, 0.25 A cm−2, 5-cell stack, Celtec P1100W (163 cm2) | 1562 cycles, 4160 h | 26 μV per cycle | 2015 | 54 | |
| Load cycling 0.0–0.5 A cm−2 for 16 s | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, 444 h 000 cycles, 444 h |
34 μV h−1 | 2015 | 55 | |
| Triangular sweep cycling 0.0–0.5 A cm−2 for 16 s, 2 s OCV | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, 500 h 000 cycles, 500 h |
44 μV h−1 | 2017 | 56 | |
| Constant load 0.22 A cm−2 | 30 μV h−1 | ||||
| 160 °C, low current cycling 16 min at 0.3 A cm−2, 4 min OCV | 70 h | 363 μV h | 2016 | 57 | |
| 216 min at 1.0 A cm−2 + 4 min at 0.6 A cm−2 + 10 min at OCV every 6 h | 330 μV h−1 | ||||
| 160 °C, 60 daily start/stop cycles, 0.3 A cm−2 | 12.6 μV h−1, 600 μV per cycle | 2016 | 58 | ||
| 160 °C, load cycling 0.2–0.8 A cm−2, Dapozol® MEAs | 16–195 μV h−1 | 2017 | 28 | ||
| 160 °C, start/stop cycling, λH2/air = 1.25/2.5, synthetic reformate (74.8% H2, 25% CO2, 0.2% CO), Celtec®-P1100W | 1562 cycles, 4042 h | 26 μV per cycle | 2018 | 59 | |
| Single cell start/stop, λH2/air = 1.5/2.0, 160 °C | 18 cycles, 1900 h | 60 μV h−1 | 2019 | 60 | |
| Potential cycling 0.5–0.9 V, 3 min at 0.5 A cm−2 (multiple serpentine) | 4821 cycles, 482 h | 40 μV per cycle | 2019 | 60 | |
| 140–160–180 °C thermal cycling at 0.2 A cm−2, 1 h interval | 100 h | 200 μV h−1 | 2019 | 61 | |
| High load cycling 0.2–0.6 A cm−2 | 103 μV h−1 | ||||
| Low load cycling 0.0–0.2 A cm−2 | 135 μV h−1 | ||||
| Load cycling 0.6 (4 min)–1.0 A cm−2 (16 min) | 1100 cycles, 500 h | 32 μV h−1 | 2019 | 62 | |
| Daily start/stop (12 h/12 h), 0.3 A cm−2 | 20 cycles, 500 h | 33 μV h−1 | |||
| Temperature & load cycling | 165 °C, 0.31 A cm−2 | 240 cycles, >4000 h | 480 μV per cycle | 2021 | 63 |
| 175 °C, 0.55 A cm−2 | 580 μV per cycle | ||||
| 160 °C, triangular sweep cycling | OCV – 0.5 A cm−2, 16 s | 100![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, 440 h 000 cycles, 440 h |
30 μV h−1 | 2021 | 64 |
| OCV – 0.5 A cm−2, 12 s + 2 s OCV | 125![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles, 550 h 000 cycles, 550 h |
30 μV h−1 | |||
| Start/stop cycling in 1.2 h, 0.22 A cm−2 | 300 cycles | 81 μV hv | |||
| 1000 cycles | 55 μV h−1 | ||||
| Start/stop cycling 12 h, 0.2 A cm−2 | 600 h | 29.7 μV h−1 | 2021 | 65 | |
| High load cycling 0.2–0.8 A cm−2, 20 min | 84 μV h−1 | ||||
| Low load cycling, 0.04–0.2 A cm−2, 20 min | 115 μV h−1 | ||||
| Thermal cycling, 140–160 °C, 0.15 A cm−2 | 700 cycles | < 30 μV h−1 | 2022 | 66 | |
| Load cycling, 0.6–1.0 A cm−2, 160 °C | 1000 h | 65 μV h−1 | 2024 | 67 | |
| Thermal & load cycling | 160 °C, 0.2 A cm−2, iconst. = 0.2 A cm−2 | 211 h | 14.2 μV h−1 | 2024 | 43 |
| 140–180 °C, i = 0.2 A cm−2 for 37 min | 197 h | 25.4–45.7 μV h−1 | |||
| 50–160 °C, OCV – 0.2 A cm−2 for 37 min | 260 h | 50 μV h−1 | |||
| 50–160 °C, OCV for 37 min | 238 h | 75.6 μV h−1 | |||
| Load cycling | 0.2 A cm−2 + 20 kHz triangular current ripples of 20% of amplitude | 2600 h | 4% less than ref. | 2025 | 47 |
Table 1 compiles literature on HT-PEMFC dynamic durability studies, organized by AST protocol categories, and the following discussion follows this structure. Start–stop cycling induces additional degradation relative to static operation. Reiser et al. identified a mechanism termed “reverse-current” degradation, Fig. 2E: during start or stop events, the hydrogen/air front sweeps through the anode compartment, generating a local reverse internal current and driving electrode potentials as high as 1.4–1.5 V.30,68 This elevated potential accelerates carbon corrosion in the catalyst layer, triggering platinum particle growth and a reduction in the active catalyst surface area. Consequences include loss of electrode hydrophobicity, increased mass transport resistance, catalyst layer thinning, and eventual cathode collapse. Hartnig et al.69 simulated 150 start–stop cycles by alternating hydrogen/air flow over the fuel electrode and quantified carbon corrosion via CO2 evolution at the air electrode. They correlated carbon corrosion with cathode performance degradation for catalysts supported on various materials, confirming mass transport loss as the primary effect of start–stop cycling.
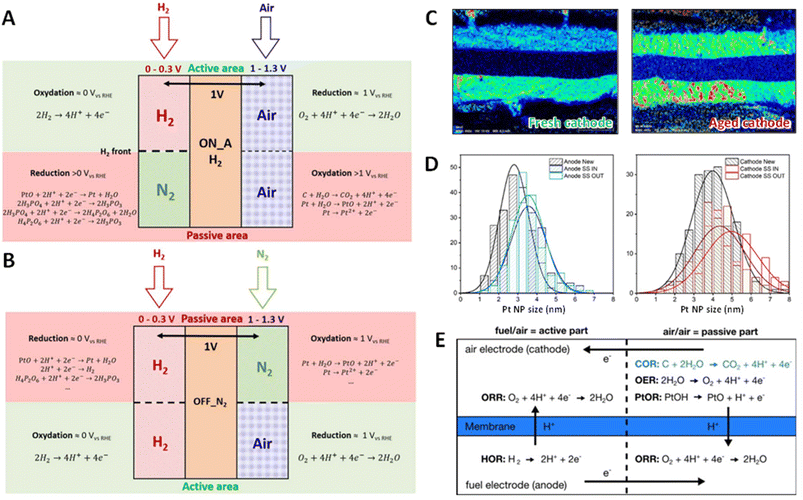 | ||
| Fig. 2 Illustrative schemes of degradation reactions, which could occur in the (A) startup and (B) the shutdown procedure (with N2 flushing). The reverse current in the passive area leads to PtOx formation and Pt dissolution at the cathode [reproduced from ref. 31]. (C and D) Pt cross-section mapping of fresh and aged air inlet of a MEA. Anode and cathode are respectively at the bottom and at the catalyst NPs from MEA in (C) [reproduced from ref. 31]. (E) Simplified scheme of start-up/shut-down protocol with reverse-current decay mechanism, highlighting the carbon oxidation reaction at the cathode during a gas feed exchange at the anode [reproduced from ref. 70]. | ||
These findings also indicate cross-coupled degradation: as carbon corrosion progresses, increased hydrophilicity of the electrode promotes PA flooding, reducing oxygen transport and locally increasing current density, which in turn accelerates further corrosion. Such feedback illustrates the interdependence of mechanical, chemical, and electrochemical degradation modes.
In real start–stop testing at 180 °C, cells operated at 0.2 A cm−2 for 16 hours followed by an 8-hour shutdown at room temperature without purging. After 4000 hours, corresponding to 157 daily start–stop cycles, voltage degradation averaged 19 μV per hour or approximately 480 μV per cycle. Compared to beginning-of-life performance, total losses included 31 mV attributed to increased ORR overpotential and 23 mV due to mass transport overpotential increases.69,71 In a similar test at 160 °C with overnight idling at 25 °C, Pinar et al. purged the cell with nitrogen during temperature excursions below 120 °C, observing a voltage loss rate of 12.6 μV per hour or 600 μV per cycle over 115 days.72
Overall, performance losses exceeding 200 mV per start–stop cycle are common in the absence of mitigation strategies, though some studies report lower degradation rates.73 Oyarce et al. evaluated different start–stop mitigation approaches, including hydrogen consumption (fuel shutoff under load), anode air purging, oxygen consumption (air shutoff under load), and cathode hydrogen purging.74 System-level strategies such as flow rate adjustments, cell potential control, and nitrogen purging have also been explored.13,30 Among these, oxygen consumption and cathode hydrogen purging demonstrated degradation rate reductions by an order of magnitude compared to no-purge operation, whereas hydrogen consumption and anode air purging provided limited protection.
Kannan et al. tested a five-cell stack equipped with Celtec P1100W MEAs (active area 163.5 cm2) using inert gas purging of the anode and oxygen consumption at the cathode. After 4160 hours, including over 1500 start–stop cycles of 15 minutes on/10 minutes off, the degradation rate was approximately 26 μV per cycle at 0.25 A cm−2. Considering steady-state degradation, performance loss attributable to start–stop cycling was estimated at 20 μV per cycle.75
Incorporating operational controls such as timed purge sequences and predictive shutdown protocols, as demonstrated here in system-integration studies, reduced degradation rates to 0.18–0.25 mV per cycle, confirming that stack-level management can be as influential as material design in improving overall durability.
Thermal cycling represents the most severe operational stress for evaluating the durability of PBI-based HT-PEM fuel cells. This cycling induces significant mechanical stresses on the membrane, electrodes, and their interfaces due to repeated expansion and contraction. The cell voltage fluctuates with changes in catalytic activity and proton conductivity, which contributes to catalyst degradation, as previously discussed in relation to load cycling.43
Operating at temperatures outside the typical range (usually around 150–160 °C) can exacerbate PA loss. High PA vapor pressure at elevated temperatures promotes increased acid evaporation.76 Conversely, at lower temperatures where liquid water is present, PA loss is also a concern. Lim et al. noted that the interaction between PA and water is stronger than that between PA and the polymer membrane, leading to PA segregation from the membrane when water is in the liquid state.66 In a triangular thermal cycling test between 80 and 160 °C with a temperature ramp of 10 °C min−1 and a constant current density of 0.15 A cm−2, the cell voltage dropped dramatically from 0.78 V to near zero after 70 cycles. This decline coincided with approximately 85% loss of PA from the membrane. In contrast, membranes exhibiting stronger acid–base interactions experienced only 40% acid loss even after 500 thermal cycles.
An earlier study subjected cells to cycling between 160 °C (2 h at 0.6 V) and 140 °C (2 h at 0.6 V), resulting in a power degradation rate of approximately 6% every 1000 hours, equating to roughly 200 mV loss per cycle.71 Park et al. performed thermal cycling between 180 °C (30 minutes at OCV) and 30 °C (30 minutes at OCV), with I–V curves recorded every 48 cycles. They observed an OCV decrease of 13 mV per cycle for standard PBI membranes after 50 cycles, while PTFE-reinforced membranes exhibited a much lower decline of 2 mV per cycle even after 150 cycles.71
Liu et al. conducted thermal cycling from 140 to 160 and then 180 °C at 0.2 A cm−2, maintaining each temperature step for 1 hour. Their results highlighted PA loss and membrane degradation as the dominant failure mechanisms, evidenced by a high PA loss rate of 94.2 ng H3PO4 per hour and increased hydrogen crossover.77
Comparing these various regimes, it is concluded that degradation during high-temperature thermal cycling (>170 °C) is dominated by PA volatilization, whereas membrane oxidation and interfacial delamination dominate at low-temperature cycling (<120 °C). This classification clarifies which mechanism prevails under specific operating conditions.
Load cycling, which involves periodic changes in load level, is a common strategy to accelerate fuel cell degradation, particularly targeting catalyst degradation. This cycling exposes the cell to severe carbon support corrosion and sintering of noble metal catalysts. Platinum catalyst dissolution, specifically the formation and dissolution of the metal surface oxide, is intensified during load cycling. The solubility of this metal oxide in the electrolyte follows a Nernstian dependence on the cathode potential.78
Yu et al. conducted one of the first studies on HT-PEMFCs and demonstrated that current cycling resulted in a degradation rate more than five times higher after just 500 hours compared to constant load operation over 2500 hours. Since then, both low and high load cycling have been recognized as effective stressors.79
Among all operating conditions, holding at OCV is the most damaging. Exposure to OCV can cause voltage degradation rates exceeding 400 mV h−1, accompanied by significant increases in both activation and mass transport resistances, and a marked reduction in the cathode's electrochemically active surface area (ECSA).80 After 245 hours at OCV, Pt NP sizes were found to increase more than fourfold compared to fresh cathodes.80 Similar findings were reported by Li et al.73 and further confirmed by Pinar et al.81
Recent multiphysics models coupling electrochemical kinetics, thermal gradients, and mechanical strain successfully reproduce these OCV degradation behaviors and predict local stress fields responsible for microcrack formation near the cathode–membrane interface. Such models provide quantitative insight into the coupling between electrical, thermal, and mechanical degradation.
Schonvogel et al. were among the first to perform AST under high load cycling. They cycled the load between 0.6 A cm−2 (4 minutes) and 1.0 A cm−2 (16 minutes), with 10-minute OCV idling periods every 6 hours. This test showed a high degradation rate of 330 μV h−1, comparable to low load cycling between 0 and 0.3 A cm−2, likely due to the OCV exposure during idling.82 Conversely, Büsselmann et al. reported a significantly lower degradation rate of 32 μV h−1 during high load cycling (0.6 to 1.0 A cm−2) without OCV exposure over 500 hours.83 Other studies also report similar rates of 30–40 μV h−1 for load cycling that avoids OCV excursions.79
Comparative studies using fast triangular sweep cycling (16 seconds per cycle) between 0.01 and 0.5 A cm−2, with and without short (2-second) dwells at OCV, revealed degradation rates of approximately 34 mV h−1 without OCV dwelling, and slightly higher rates of 44–45 mV h−1 with brief OCV exposure.84,85
An interesting investigation by Thomas et al. examined the effect of relaxation time (duration of low current operation) in load cycling between 0.2 and 0.8 A cm−2. By adjusting the high load duration to keep the average current at 0.55 A cm−2, they found that a 2-minute relaxation time yielded the best performance and a low degradation rate of 36 μV h−1, only 63% of that under constant load. This improvement was attributed to reduced PA loss during cycling.86
In addition to catalyst degradation, load cycling also mechanically stresses the membrane, as observed through μ-CT imaging and MEA thickness measurements.87 Acid loss is known to vary under high current densities, likely linked to increased water production, heat generation, and gas flow dynamics.88 However, load cycling tests, typically lasting a few hundred hours, show minimal membrane degradation, with little change in proton conductivity or hydrogen crossover resulting from acid loss or polymer degradation.
Complementing earlier work, several 2023–2025 studies from major research groups provide quantitative correlations between voltage decay, Pt particle growth, and ECSA loss across multiple cycling protocols. Their inclusion here ensures comprehensive coverage of milestone research in the past three years.14,31
The HT-PEMFC technology has been extensively validated under steady-state operating conditions. At a constant current density typically ranging from 0.2 to 0.4 A cm−2 and temperatures of 150–160 °C, voltage degradation rates on the order of a few microvolts per hour (μV h−1) have been consistently reported in recent years. These results highlight the technology's promising potential to meet the lifetime requirements for various applications. However, significant challenges remain when operating under dynamic conditions, particularly at elevated temperatures, higher current densities, or altered reactant stoichiometries. Durability under dynamic operation—such as during start–stop events, load cycling, and thermal cycling—remains the most critical concern.
Notably, idling at OCV or near-OCV (low load) conditions dramatically accelerates catalyst degradation, while high load operation predominantly exacerbates acid loss and membrane electrolyte deterioration. Start–stop cycling introduces additional stressors, including reverse current phenomena and elevated cathode potentials, which further accelerate catalyst degradation. Beyond these electrochemical effects, thermal cycling imposes mechanical stresses on membranes, electrodes, and their interfaces, amplifying PA loss through evaporation at elevated temperatures and washout at lower temperatures.1 Future investigations should aim to isolate and clarify the individual mechanisms and their impacts during OCV, start–stop, thermal, and load cycling conditions.
Catalyst degradation, manifesting as NP agglomeration, metal dissolution, and carbon support corrosion, plays a primary role in both initial and long-term performance decay throughout durability testing. This degradation leads to significant losses in the ECSA of the catalysts. Such phenomena have been extensively studied under both steady-state and dynamic load conditions. Although low degradation rates can be maintained during prolonged steady-state operation at moderate current densities, cells experience markedly accelerated degradation under variable operating modes. These include potentials at or near OCV, elevated temperatures, cell potential cycling, and during transient start-up and shutdown events. In the following section, durability results are categorized under dynamic operating conditions, followed by a discussion of an additional stress factor, fuel starvation.
Looking ahead, emerging directions such as digital-twin predictive modeling and machine-learning-guided degradation mapping are expected to play a crucial role in future HT-PEMFC design.89 These methods integrate large-scale experimental datasets with multiphysics models to forecast stack behavior and optimize lifetime under start–stop and thermal stress.
It is important to note that durability data and mitigation strategies at the stack or system level remain scarce. This review aims to stimulate further research efforts in HT-PEMFC durability, supporting the maturation and commercialization of this promising technology.
4. Degradation mechanisms under start–stop conditions
The degradation mechanisms affecting HT-PEMFC components, as outlined in the preceding section, include (1) pinhole formation in the membrane, (2) acid loss through evaporation, (3) membrane thinning due to polymer oxidation, (4) catalyst layer thinning resulting from carbon corrosion, (5) catalyst particle growth driven by physical and electrochemical Ostwald ripening, and (6) catalyst detachment, among others.90 While these degradation pathways, except for acid loss, are also observed in LT-PEMFCs, the elevated operating temperatures and the presence of PA in HT-PEMFCs exacerbate their effects. This intensification is particularly pronounced under dynamic operating conditions, such as thermal cycling, load cycling, and start–stop cycling, where elevated electrode potentials are encountered. In practice, these mechanisms rarely act in isolation; rather, polymer oxidation, PA redistribution, and carbon-support corrosion reinforce each other spatiotemporally, yielding localized hot spots of current density that accelerate further chemical and mechanical damage.The interplay of these mechanisms is highly complex and strongly dependent on operational parameters, complicating the isolation and identification of individual degradation processes. A thorough understanding of MEA lifetime limitations is critical for the development of effective mitigation strategies and for achieving the durability requirements of both vehicular and stationary fuel cell applications. Recent multi-cycle datasets indicate that under start–stop, carbon-corrosion-driven catalyst-layer thinning dominates voltage loss, whereas under sustained high load the rate-controlling process shifts toward PA evaporation and migration, with measurable increases in ohmic resistance. To connect material degradation to stack-level causes, we note that transient fuel-supply imbalances and thermal-management delays can briefly push local temperatures above 180–190 °C and produce λ variations that trigger starvation and reverse currents.
Electrolyte degradation, manifesting as acid loss and polymer oxidation, becomes significant primarily over extended periods, unless accelerated by harsh stressors such as elevated temperatures, high current densities, or extreme reactant stoichiometries. Conversely, catalyst degradation, including NP agglomeration, metal dissolution, and carbon support corrosion, primarily drives both initial and ongoing performance losses throughout durability testing. These effects lead to a reduction in the ECSA of the catalyst and have been extensively studied under both steady-state and dynamic load conditions.22 Coupled electro–chemo–mechanical models further show that interfacial delamination increases local contact resistance, focuses current pathways, and feeds back into accelerated peroxide formation and support corrosion.
Empirical evidence suggests that low degradation rates are achievable during prolonged steady-state operation at moderate current densities. However, more severe degradation is observed under variable operating regimes, including potentials at or near OCV, elevated temperatures, cell potential cycling, and transient start-up and shutdown events.1 For clarity across the review, we reference a consolidated comparison (Table 1/Fig. 6) that collates recent degradation rates (mV per cycle and μV h−1) under these regimes.
4.1 Membrane degradation
In HT-PEMFCs, membrane proton conductivity is primarily enabled through acid doping, with acid loss being a critical degradation mechanism affecting cell performance. Compared to LT-PEMFC technologies, PA-doped PBI cells typically exhibit higher ohmic resistance, which depends on both the intrinsic conductivity and the thickness of the membrane electrolyte. This section discusses the mechanisms of acid loss and their significance, focusing on acid doping, conductivity, migration, evaporation, and key operational parameters including temperature, current density, and gas stoichiometry. Numerically, representative through-plane conductivities at 160–180 °C are ≈0.09–0.12 S cm−1 for m-PBI/AB-PBI and up to ≈0.13–0.14 S cm−1 for recent cross-linked/composite PBI variants, underscoring the trade-off between conductivity and mechanical integrity.For PA-doped PBI membranes, proton conductivity is directly related to the acid doping level (ADL). Early studies using low molecular weight PBI reported an ADL of approximately 5–6 H3PO4 molecules per polymer repeating unit (PRU). With advancements to higher molecular weight polymers, the ADL has increased to current values around 11–12 H3PO4 molecules per PRU, resulting in proton conductivities near 0.1 S cm−1 at operating temperatures close to 160 °C.18,91 Although increasing the ADL enhances conductivity, it simultaneously compromises membrane mechanical integrity. Notably, while the conductivity of PA-doped PBI membranes approaches that of PFSA membranes, typical membrane thicknesses ranging from 40 to 100 μm yield relatively high ohmic resistances (∼100 mΩ cm2), exceeding those found in LT-PEMFCs.1 Under dynamic operation, small reductions in local ADL (via evaporation or washout) can raise area-specific resistance and shift local current to adjacent regions, magnifying heterogeneity and accelerating mechanical fatigue.
The proton exchange rate between PBI groups and PA is approximately nine orders of magnitude slower than the exchange between excess PA species. Consequently, acid doping far beyond the equimolar composition of 2 H3PO4 molecules per PRU is necessary to achieve ion conductivities suitable for practical applications. The ADL can be effectively controlled by adjusting the PA concentration in the doping solution, as exemplified for meta-PBI (m-PBI) in Fig. 3A. A typical ADL near 11 H3PO4 molecules per PRU is attained by equilibrating membranes in a 15 M (85 wt%) PA solution at room temperature.18
 | ||
| Fig. 3 (A) Ion conductivity at 160 °C for m-PBI as function of ADL under non-humidified conditions. The different markers refer to data sets from different sources, and the reader is referred to the original references for full experimental details.1 (B) Water vapor pressure in a H2O–P2O5 system as a function of composition and temperature, [Reproduced with permission,92 Copyright © 2016, Springer International Publishing Switzerland] (C) hydration isotherms of PA and PA-benzimidazole (bIm) mixtures [Reproduced with permission,1 Copyright 2023, Royal Society of Chemistry]. Data at 150 and 170 °C are from Vilčiauskas et al.93 while data at 182 °C are from Melchior et al.94 (D) average area-specific PA loss rate of PBI cells as a function of current density and the total (H2 + air) gas flow rate for operation at 160 °C.18 | ||
At fuel cell operating temperatures (150–160 °C), the acid vapor pressure has been estimated at approximately 0.7 mbar at 160 °C and increases to 3.7 mbar at 190 °C. Given continuous operation with significant airflow over several years, PA evaporation represents a non-negligible mode of acid loss. For example, at a current density of 0.2 A cm−2 and reactant stoichiometries of λH2/air = 1.2/2.0, these vapor pressures correspond to acid loss rates of approximately 3.6 mg m−2 s−1 at 160 °C and 19 mg m−2 s−1 at 190 °C.95–97 To reflect practical operation, we explicitly note that thermal-management delays and high-stoichiometry air sweeps during start/stop can transiently elevate evaporation flux, suggesting a control-oriented mitigation path (e.g., ramp-rate limiting and low-air cooldown).
A comprehensive review of acid loss data from PBI cells under various operational conditions is provided in Table 2.
| Membrane | Conditions | Time (h) | Acid loss (μg m−2 s−1) | Year | Ref. | |
|---|---|---|---|---|---|---|
| AB-PBI (ADL 3.5) | 160 °C, 0.2 A cm−2, 1000 h, λair = 2, λH2 = 2 | 180 | (A & C) 0.21 | 2008 | 98 | |
| p-PBI and 2OH-PBI (PPA process, ADL 26) | 80–190 °C, 0.2 A cm−2, λair = 2.0, λH2 = 1.2 | 160 °C | 1700–2500 | 0.02 | 2008 | 50 |
| 190 °C | 800 | 0.2–0.3 | ||||
| PBI (ADL 6.6) | Three-cell stack, 125 °C, 0.2 A cm−2, λair = 3, λH2 = 2 | 185 | 3.4 | 2011 | 99 | |
| 1.1 | ||||||
| Advent TPS® (not PBI-based) | 150 °C, 0.2 A cm−2 | 360 | 4.6–5.9 | 2013 | 100 | |
| Pyridine containing PBI copolymer (PPA process) | 180 °C, 0.2 A cm−2, λair = 2.0, λH2 = 1.2 | 2500 | 0.6 | 2014 | 101 | |
| 0.04 | ||||||
| m-PBI (ADL 9.3–9.6) | 160–180 °C, 0.2–0.8 A cm−2, λair = 4.0–25.5, λH2 = 2.0–22.6 | 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 000 |
2–59 | 2018 | 102 | |
| m-PBI membranes (ADL 9.5) | Varied GDLs, 180 °C, 0.2 A cm−2, λair = 2, λH2 = 1.5 | 5500 | 2.7–4.1 | 2018 | 103 | |
| m-PBI (layered, ADL 10–37) | 180 °C, 0.2 A cm−2, λair = 2, λH2 = 1.5 | 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 440 440 |
2.1–2.6 | 2020 | 104 | |
| PBI-based, PA-doped Advent Tech.® | 50–180 °C, 0.0–0.2 A cm−2, λair = 2, λH2 = 1.5 | 260 | 0.02–0.1 | 2024 | 43 | |
It is important to highlight that contact with liquid water induces washout of excess doping acid, as the interaction between water and PA facilitates segregation of PA from the membrane. Therefore, any water condensation must be strictly avoided during fuel cell operation, as well as during start-up or shutdown sequences, to prevent accelerated acid loss. System-level safeguards include purge scheduling, dew-point tracking, and predictive anti-condensation logic; these reduce the frequency of membrane rehydration–dehydration swings that drive PA redistribution.
Fuel starvation can arise under various conditions during fuel cell operation. For instance, fuel flow may fall below a critical threshold when the supply system cannot respond rapidly enough to sudden load changes. Additionally, uneven gas distribution within a stack can lead to fuel starvation in individual cells, thereby reducing the overall stack voltage. Other causes include start–stop cycling and malfunctions within the hydrogen supply system or methanol reformers. Zhou et al. conducted experiments on an HT-PEMFC operated at 150 °C and a constant current density of 0.4 A cm−2, cycling the hydrogen stoichiometry between 3.0 and 0.8 every two minutes.105 During the starvation period (λH2 = 0.8), the cell voltage dropped sharply and eventually reversed polarity. Upon restoring the hydrogen stoichiometry to 3.0, the cell performance continued to degrade, with the voltage declining from 0.5 V to 0.4 V after 19 cycles. A practical implication is that fuel-efficiency strategies (low λH2) must be balanced against starvation risk; supervisory controllers can minimize penalties by anticipating load steps with short battery assist rather than aggressive λ reduction.
This polarity reversal corresponds to the electrochemical generation of hydrogen at the cathode and oxygen at the anode, effectively causing water electrolysis. The detrimental effects of fuel starvation include corrosion of catalyst supports and other carbon-based components such as gas diffusion and backing layers, as well as agglomeration of the electrocatalyst particles.106 Collectively, these effects significantly shorten cell lifetime.107 A straightforward approach to mitigate fuel starvation involves supplying fuel in excess relative to the demanded power, maintaining hydrogen and air flow rates at stoichiometrically higher ratios than those required by the current load.108 Fuel starvation is widely recognized as a harmful phenomenon in PEMFC operation, and various mitigation strategies have been developed to prevent hydrogen and oxygen starvation in these systems.109
4.2 Catalyst layer degradation
The degradation of fuel cell performance is significantly influenced by the loss of ECSA of Pt catalysts. This loss primarily results from the growth of Pt particles, which reduces the available active surface area. Several mechanisms have been proposed to explain Pt particle dissolution and growth, as illustrated in Fig. 2A. One prominent mechanism is catalyst coarsening, which occurs through the migration and subsequent agglomeration or sintering of Pt particles on the carbon support. This process involves the movement of Pt adatoms between neighboring particles, favoring growth of larger particles at the expense of smaller ones, a phenomenon commonly referred to as Ostwald ripening.110Another significant contributor to Pt particle growth is electrochemical dissolution and redeposition, especially under changing gas fronts, Fig. 2B. Pt atoms dissolve into the electrolyte under certain electrochemical potentials and subsequently redeposit onto existing particles, leading to particle growth and morphological changes. This dissolution can occur through direct electrochemical oxidation of the Pt metal surface to Pt ions, followed by transport in the electrolyte and reduction back to metallic Pt.31 Additional mechanisms such as particle detachment from the carbon support and carbon corrosion-induced structural changes also contribute to the degradation of catalyst layer integrity and catalyst surface area.
The electrochemical dissolution of Pt can be a direct dissolution of the metal:111
| Pt → Pt2+ + 2e E (V) = 1.19 V + 0.029 V log[Pt2+] |
At elevated electrode potentials, a platinum oxide (PtOx) layer forms on the metal surface. The dynamics of this oxide layer, its formation and subsequent dissolution, are strongly influenced by the electrolyte pH and the applied electrode potential. In acidic environments typical of HT-PEMFCs, the formation of PtOx occurs at potentials above approximately 0.8 V versus the reversible hydrogen electrode (RHE). The oxide layer is typically composed of PtO and PtO2 species, which are electrochemically active and can undergo reduction during potential cycling.
The cyclic formation and reduction of this oxide layer lead to transient dissolution of Pt ions into the electrolyte. This dissolution process is potentiostatically dependent and follows a Nernstian behavior, where higher potentials promote oxide growth and metal dissolution, while reduction potentials favor Pt redeposition. Such redox cycling accelerates Pt loss and particle growth through a dissolution–redeposition mechanism, thereby contributing significantly to catalyst degradation during load and potential cycling in HT-PEMFCs.112
| Pt + H2O → PtO + 2H+ + 2e− E (V) = 0.98 V 0.59 V pH |
| PtO + H2O → PtO2 + 2H+ +2e− E (V) = 1.05 V 0.59 V pH |
The formed platinum oxides are inherently unstable in acidic electrolytes and can dissolve through both chemical and electrochemical pathways. The equilibria governing these dissolution processes can be represented as follows:113
Chemical dissolution:
| PtOx + 2H+ → Pt2+ + H2O |
Electrochemical dissolution:
| PtOx + ne− → Pt2+ + reduced species E (V) = 0.834 V 0.118 V pH 0.029 V log[Pt2+] |
These dissolution reactions contribute to the loss of electrochemically active platinum surface area by mobilizing platinum ions into the electrolyte. Subsequent redeposition of these ions can lead to particle growth and agglomeration, thereby diminishing catalyst performance over time. Coupling to the membrane is critical: dissolved Pt2+ can migrate into the ionomer, form a Pt band upon reduction by crossover hydrogen, and simultaneously catalyze peroxide formation that accelerates local polymer oxidation.
The solubility of Pt is influenced by several factors, including the electrode potential, electrolyte composition and pH, temperature, and the morphology of the Pt itself, such as NPs versus nanowires, Fig. 5A and B. Pt ions generated via dissolution are mobile within the electrolyte phase and can be redeposited on larger particles upon encountering electrons conducted through the carbon support (Fig. 2C).114 Additionally, mobile Pt ions can migrate into the ionomer phase, where they may encounter hydrogen permeating from the anode and subsequently be reduced inside the ion-conducting membrane. This results in the formation of a so-called “Pt band”, which has been associated with further degradation of both the membrane and the MEA, as extensively documented in LT-PEMFCs.115
Early studies on the stability of carbon-supported Pt NPs in concentrated PA electrolytes demonstrated minimal potential dependence of Pt surface area loss below 0.70 V versus RHE. However, at cathode potentials exceeding 0.70 V, a strong correlation between Pt surface area loss and electrode potential emerges, indicating an increased role of dissolution and redeposition mechanisms. More recent investigations by Prokop et al. have revisited this phenomenon, highlighting the acceleration of particle coarsening at elevated temperatures. Above 0.70 V, the degradation is dominated by a combination of agglomeration/sintering and Ostwald ripening processes.116–118 This phenomenon is more pronounced at cathode than at the anode (Fig. 2D) due to the electrode potentials evolved during the MEA operation. Operando tomography and multi-physics simulations further show that local stress concentrations at TPB-rich zones coincide with Pt coarsening hot spots during reverse-current transients.
In concentrated PA electrolytes at temperatures up to 200 °C, studies have reported the logarithmic dependence of Pt solubility (ranging from 10−7 to 10−4 M) on electrode potential between 0.8 and 1.0 V versus RHE, demonstrating Nernstian behavior. Specifically, in 96% PA at 176 and 196 °C, the Pt dissolution rate was found to increase by approximately nine orders of magnitude as the potential is swept from 0.6 to 1.0 V.119 For PBI cells, where the PA content in the membrane is limited, it is estimated that Pt dissolution under OCV conditions may account for 2–5% of the total Pt catalyst loading (0.5–1.0 mgPt cm−2) in the cathode. This level of dissolution is significant and likely contributes substantially to catalyst degradation, particularly under potential cycling conditions.
Carbon, which serves as the catalyst support, is vulnerable to corrosion under fuel cell operating conditions, Fig. 2E. The primary electrochemical reactions involved in carbon corrosion exhibit the following standard potentials versus the RHE:120
| C + 2H2O → CO2 + 4H+ + 4e− E0 = 0.207 V vs. RHE |
| C + H2O → CO + 2H+ + 2e E0 = 0.518 V vs. RHE |
These reactions highlight the oxidative degradation pathways of carbon supports, which can compromise catalyst layer integrity and overall fuel cell durability. Thermodynamically, carbon oxidation can occur at potentials above 0.207 V vs. RHE, leading to the release of CO2. Carbon corrosion results in the detachment of Pt particles, which subsequently promotes Pt agglomeration and growth, thus diminishing the ECSA of the catalyst. Additionally, carbon corrosion forms surface oxides that decrease the hydrophobicity of the catalyst layer, potentially causing PA flooding within the electrode. While corrosion rates of carbon at potentials below approximately 0.7 V vs. RHE remain relatively slow, they are significantly accelerated at elevated temperatures of HT-PEMFC and in the presence of Pt.121 Conversely, operational modes involving electrode exposure to high cell voltages near OCV substantially enhance carbon corrosion rates.68 Because these effects are operation-induced, system strategies such as oxygen-consumption shutdowns and cathode hydrogen purges have become essential to limit high-potential excursions in practice.
A critical scenario inducing rapid carbon corrosion is the start–stop cycling of fuel cells. During these events, transitions from hydrogen/air to air/air environments induce reverse currents, resulting in electrode potentials exceeding the OCV. This phenomenon, known as the reverse current effect, triggers accelerated carbon corrosion and catalyst degradation.122 Similar degradation patterns arise during fuel starvation, characterized by extraordinary performance losses including activation losses (due to Pt particle growth) and mass transport limitations caused by catalyst layer thinning or collapse. Extensive research on start–stop cycling effects and mitigation strategies are discussed in detail in Sections 6 and 7. We explicitly link these transient mechanisms to the dominant degradation hierarchy summarized in Table 1/Fig. 6 for quick comparison across protocols.
Elevated operating temperatures further exacerbate carbon corrosion by weakening Pt NP binding energies to the carbon support, thereby facilitating particle detachment. Carbon corrosion compromises catalyst layer integrity via three primary pathways: (1) loss of catalyst ECSA through particle detachment, (2) degradation of carbon electrical conductivity, and (3) increased mass transport resistance due to pore narrowing from oxide layer formation. As described previously, four irreversible mechanisms contribute to catalyst ECSA loss: carbon corrosion-induced NP agglomeration or detachment, Ostwald ripening-driven NP coarsening, Pt dissolution and subsequent recrystallization within the electrolyte matrix, and NP coalescence due to Pt crystallite mobility on carbon surfaces.22,123 Disruptive directions under investigation include graphitized carbons with engineered defect densities, PA-philic binders that stabilize TPB ionomer networks, and alloyed Pt (e.g., Pt–Co/Ni) that resists phosphate adsorption while maintaining ORR kinetics.
Bandlamudi et al. confirmed cathodic carbon corrosion by measuring CO2 emissions from the cathode, observing a monotonic increase in CO2 concentration with applied potential.124 Complementary studies by Borup et al. and Søndergaard et al. showed that Pt particle growth is accelerated at elevated potentials and that relative humidity acts as a stressor during ASTs, with higher humidity levels exacerbating Pt catalyst degradation.125,126
Carbon corrosion rates are influenced by several factors including temperature, relative humidity, cell potential (particularly OCV conditions), start–stop cycling, and fuel starvation. The physical structure of the carbon support also critically affects its susceptibility to oxidation. Amorphous carbons with high specific surface areas are more prone to corrosion compared to more crystalline, graphitized carbons, which demonstrate greater resilience at the expense of fewer anchoring sites for Pt NPs.127
Dynamic fuel cell operations such as start–stop cycling markedly accelerate cathodic carbon corrosion through the reverse current mechanism. Upon resuming fuel flow after shutdown, residual air mixed with anode gas induces cathodic transient potentials up to 1.6 V, precipitating rapid carbon corrosion and irreversible damage.122 Thermal cycling further induces mechanical stresses due to differential expansion and contraction at varying hydration levels, promoting carbon corrosion and Pt particle sintering. Multi-physics simulation (electrochemistry–transport–stress) captures these transients and predicts where delamination will initiate, consistent with post-mortem thickness gradients.128
Studies comparing various carbon supports under potential cycling conditions have identified that graphitized carbon blacks (GCB) exhibit superior stability and durability in HT-PEMFCs compared to conventional carbon blacks (CB) or multi-walled carbon nanotubes (MWCNTs).129 For example, Selvaganesh et al. demonstrated enhanced catalyst stability using GCB-supported Pt NPs relative to non-GCB supports.130 Recently, Pawlyta et al. explored the influence of carbon support structure by modifying CB through high-temperature heat treatment, linking structural properties to improved cell durability.131 Milestone reports from the last three years that track ECSA retention (>85–90% at 160 °C over ≥3000 h) on graphitized supports are highlighted in the comparative summary to ensure coverage of key contributions.
The catalyst layer (CL) is a complex system, as its operation requires efficient transport of mass, energy, and heat. To achieve high catalytic activity at the electrode, the CL must facilitate numerous “triple-phase boundary” (TPB) sites, locations where the gaseous reactant, the proton conductor (PA within the PBI matrix), and the electron conductor (carbon-supported metallic NPs) come into contact at the Pt surface. The formation of TPB sites strongly depends on the uniform distribution of both platinum and the binder within the CL. This distribution directly influences the CL's conductivity, affecting local current density and the layer's permeability.132 Together, the GDL, microporous layer (MPL), and catalyst layer (CL) form the gas diffusion electrode (GDE). The interplay of various carbon supports, PA, gaseous reactants and water at the Pt surface, under polarized conditions, results in propagation of complex degradation mechanisms. As previously discussed, this is exacerbated by dynamic operational protocols which amplify degradation processes of carbon oxidation, induce mass transport issues, PA loss and a loss of catalyst ECSA, thereby inhibiting ongoing electrochemical reactions and negatively impacting fuel cell performance. To mitigate various degradation mechanisms, such as metal dissolution and catalyst poisoning, HT-PEMFCs typically employ relatively high Pt loadings. This approach leads to the formation of comparatively thick catalyst layers (CLs), often on the order of 100 μm.22 While these thicker CLs improve durability, they also impede mass transport of reactants and reduce electronic conductivity within the CL, potentially accelerating degradation mechanisms. Despite the crucial role of the CL in HT-PEMFC performance, its electrical conductivity and permeability remain underexplored in the literature, unlike in LT-PEMFC systems where these properties have been more extensively characterized. Pt and its alloys remain the predominant catalysts for HT-PEMFCs due to their excellent activity and relative durability. The development of PGM-free cathode catalysts, such as Fe–N–C materials, could offer an alternative route to the Pt-related degradation in the CL.133 However, PGM-free materials present durability challenges that limit their practical application. Key degradation pathways for Fe–N–C catalysts include:
(1) Incorporation of radical-scavenging and Fe-stabilizing motifs (e.g., axial N-coordination, P-doping) and mesoporous architectures has recently reduced Fenton-type activity and mass-transport losses at ≥160 °C, though long-term validation remains outstanding.
(2) Metal leaching and structural breakdown:134 single-atom metal sites, notably Fe–N4 moieties, suffer from operation-induced Fe oxidation which weakens Fe–N bonds, resulting in their decomposition. Carbon corrosion further destabilizes the N–C bonds, especially at elevated potentials above 0.7 V, facilitating Fe atom clustering and loss of ECSA.
(3) Radical attack:112 peroxide-related radicals generated at the CL-membrane interface accelerate degradation of active sites and the surrounding carbon matrix, particularly at low operating voltages (<0.5 V).
(4.) Temperature effects:135 increased temperatures enhance the decomposition of H2O2, reducing the extent of Fe oxidation in HT-PEMFCs. Water flux in micropores can lead to Fe active center loss, although this phenomenon is less pronounced above 160 °C. Conversely, PA flooding limits gas diffusion due to poor oxygen solubility and diffusivity in PA, deactivating catalytic sites over time.
(5) Mass transport limitations:136 high mass transport resistance in Fe–N–C catalysts is linked to low gas diffusivity within micropores, which is a critical challenge for HT-PEMFC operation.
For Pt and Pt-based alloy catalysts, degradation mechanisms mirror those in low-temperature PEMFCs but are complicated by elevated operating temperatures and the presence of PA. Key considerations include:
Pt dissolution and redistribution:115 the acidic environment in HT-PEMFCs promotes dissolution of Pt ions, which can redeposit within the membrane, forming “Pt bands” visible via TEM. Elevated temperatures weaken Pt NP binding to carbon supports due to enhanced carbon corrosion.
Carbon support corrosion:124 this process leads to catalyst layer thinning, Pt particle detachment, and agglomeration, all contributing to ECSA loss. Elevated temperature accelerates carbon corrosion, but the reduced water activity and generally lower operating voltages in HT-PEMFCs may moderate this effect compared to LT-PEMFCs.
Catalyst layer thickness:18 high Pt loadings result in thick catalyst layers (∼100 μm), which while counteracting degradation, hinder mass transport and electronic conductivity, potentially promoting further catalyst degradation.
To mitigate these challenges, various strategies have been explored:
Carbon support improvements:137,138 graphitized carbon with fewer structural defects enhances support stability and catalyst durability, albeit with reduced surface area. Carbon nanotubes, especially when wrapped with PBI polymers, improve Pt NP immobilization, utilization, and durability, showing up to eightfold mass activity and fourfold durability improvements.
Alternative supports:139 materials such as tungsten carbides/oxides, silicon carbide (SiC), and SiC-titanium carbide (SiC-TiC) composites offer enhanced catalyst stability compared to traditional carbon supports.
Pt alloying:4,140,141 alloying Pt with transition metals (Co, Ni, Fe, Cu) forms Pt-rich NPs with modified surface energies and geometries, leading to improved ORR activity, enhanced durability, and reduced material costs via the so-called “anchor effect”.
Binder effects:142 the presence of benzimidazole-containing polymers such as PBI affects Pt dissolution rates in PA at elevated temperatures, suggesting that catalyst binders can influence durability. Recent alloy/binder pairings are shown to weaken phosphate adsorption while preserving OOH/OH scaling, and have demonstrated higher specific activity at ≥160 °C with reduced ECSA decay over 1000–3000 h.
Long-term durability studies reveal progressive Pt particle growth and carbon support loss during extended operation at moderate current densities and temperatures around 150 °C. For example, Oono et al.143 observed an increase in average Pt particle size from 4.6 nm to 7.9 nm after ∼17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 hours at 0.2 A cm−2, accompanied by a voltage decay of approximately 4 mV h−1 and catalyst layer thinning due to carbon oxidation. We highlight these as milestone datasets and cross-reference them in the summary comparison to ensure visibility alongside the latest results.
800 hours at 0.2 A cm−2, accompanied by a voltage decay of approximately 4 mV h−1 and catalyst layer thinning due to carbon oxidation. We highlight these as milestone datasets and cross-reference them in the summary comparison to ensure visibility alongside the latest results.
4.3 Electrode-membrane interface failures
Post-mortem characterization focused on the carbon support, especially at the interface with metal NPs, revealed key insights into the corrosion process.31 The initial stage of carbon corrosion involves the formation of carbon–oxygen surface functional groups, which precedes full carbon oxidation to CO2. Thermodynamically, carbon oxidation is feasible above 0.207 V vs. RHE, but kinetically it occurs only above ∼0.6 V vs. RHE in the presence of Pt NPs (and above 1.0 V vs. RHE without Pt catalysts). Carbon corrosion modifies the carbon support's morphology and porosity, potentially causing local collapses in the porous structure and changes in electrode thickness. Crucially, these chemical changes couple to mechanics: the loss of carbon backbone and growth of surface oxides increase local brittleness and contact resistance, which concentrates current in intact regions and further elevates local potentials, thus accelerating both oxide formation and delamination.SEM cross-sectional analyses of MEAs measured layer thicknesses before and after aging tests (averaged over at least four regions).30 While anode and membrane thicknesses remained mostly unchanged, consistent with the anode's lower susceptibility to carbon corrosion, the cathode CL exhibited a notable thickness reduction, particularly near the cell outlet. This thinning correlates with CO2 emissions observed during operation, indicating significant carbon corrosion. Consistent with multi-physics predictions, outlet-side thinning coincides with higher heat load and lower humidification, creating local hot spots and stress concentrations that promote interfacial failure.
Dimensional changes induce mechanical stresses between electrodes and the membrane, promoting catalyst layer delamination and increasing interfacial contact resistances. Notably, start–stop cycling accelerated the rise in ohmic resistance, suggesting that this dynamic operating mode exacerbates physical and chemical degradation within the MEA, reducing fuel cell efficiency and durability. At the stack/system level, brief thermal-management delays during start (fan/pump ramp) or non-uniform purges can aggravate these gradients; supervisory control that limits temperature ramp rates and enforces coordinated purge/airflow sequencing reduces the incidence of early-life interfacial resistance growth.
Introducing nitrogen (20 vol%) into the hydrogen fuel (80 vol%) led to increased kinetic and proton transfer resistances during testing.13,30 This indicates that nitrogen contamination impairs both the electrochemical reaction kinetics and proton conductivity, detrimentally impacting overall fuel cell performance. Practically, this argues for purge/recirculation strategies that avoid excessive N2 build-up and for controller set-points that trade a small fuel-efficiency penalty for reduced local starvation risk.
Load cycling with pure hydrogen, cycling current density between 0.2 and 0.4 A cm−2, was found to reduce acid loss and minimize degradation rates in fuel cells13,83 This suggests that controlled load cycling protocols can effectively mitigate catalyst and membrane degradation, thereby improving fuel cell longevity and operational stability. Under these conditions, activation-controlled losses dominate (moderate potentials), whereas under OCV-heavy or starvation-prone operation, support corrosion and Pt dissolution dominate; a summary comparison is referenced in Table 1/Fig. 6.
4.4 Bipolar plate and GDL issues
The GDL serves as a porous, electrically conductive interface positioned between the flow field (or bipolar plate, BPP) and the catalyst layer in MEAs. It fulfills multiple critical roles in MEA performance. Structurally, the GDL typically comprises two layers: a macroporous carbon fiber substrate and a MPL consisting of PTFE-bonded CB particles, which provides a smooth, supportive interface for the catalyst layer. Beyond materials selection, cell-to-stack integration (compression mapping, land/channel geometry, and cooling plate balance) significantly influences GDL stress states and wetting behavior during transients.The high porosity of the GDL facilitates efficient diffusion of reactant gases, hydrogen and oxygen (or air), from the flow field channels to the catalyst sites, where the hydrogen oxidation reaction (HOR) and ORR occur. In HT-PEMFCs, water produced during operation is removed through the GDL. The hydrophobicity of the GDL is thus essential not only for water management but also for PA retention and effective mass transport within the MEA.144
Despite its critical functions, the degradation of the GDL has received less attention compared to that of the membrane and catalyst layers. GDL degradation primarily manifests in two forms: (i) altered wetting properties due to changes in the carbon surface chemistry and loss of hydrophobicity,121 and (ii) structural degradation driven by mechanical stress and carbon corrosion.145 The hot-pressing process used in MEA fabrication can compromise the integrity of both the GDL and MPL, as the applied pressure often results in fiber breakage, structural displacement, and a consequent reduction in thickness and porosity. Recent disruptive approaches include corrosion-resistant coatings (e.g., PANI-based composites), tuned PTFE distributions, and micro-/laser-patterning to tailor local permeability and water pathways while minimizing PA hold-up.
During regular fuel cell operation, the GDL is exposed to mechanical erosion from gas and water flow, leading to the progressive loss of carbon material and PTFE. Furthermore, start–stop cycles and idling conditions exacerbate degradation through mechanisms such as carbon corrosion, freeze–thaw cycles, and radical-induced damage. These processes collectively reduce the hydrophobic character of the GDL surface, adversely affecting mass transport by increasing water accumulation and altering gas diffusion pathways. The degradation mechanisms are notably intensified under HT-PEMFC operating conditions.71 Coupled electrochemistry–transport–stress simulations reproduce these trends and help identify channel-land regions susceptible to early hydrophobicity loss; incorporating such models into design loops has begun to reduce variability in stack-to-stack durability.
According to Schmidt, in addition to structural failure and wettability loss, GDL degradation in HT-PEMFCs also contributes to PA redistribution and elevated mass transport overpotentials.146 Dynamic load cycling has been identified as a major accelerator of GDL degradation. In a load cycling study, Schmies et al. employed μ-CT to reveal PTFE binder deterioration and reduced tortuosity within the GDL.46 The decreased tortuosity, particularly in the cathodic GDL, likely results in shorter transport pathways that may compromise performance due to non-uniform current distribution. Li et al. further observed that carbon corrosion, affecting both the GDL and catalyst layer, significantly impairs gas-phase mass transport, thereby increasing polarization losses.73 We collate these findings (Table 1/Fig. 6) to clarify expected ranges of tortuosity change and associated voltage penalties under dynamic ASTs.
The PA exhibits inherently high thermal stability. However, at elevated temperatures, concentrated PA undergoes dehydration, forming oligomeric species such as dimers, trimers, and higher-order polymers through condensation reactions.4 This process liberates water and is reversible if the ambient water partial pressure is sufficiently high. Since water is generated during fuel cell operation and may also be introduced through the fuel stream, particularly when methanol reformate is used, the dynamic equilibrium between acid hydration and dehydration becomes critical. Variations in PA-water composition can significantly influence membrane proton conductivity, phase behavior, electrolyte acidity, and the kinetics of electrochemical reactions at the electrodes.4 From a controls perspective, dew-point tracking with guard margins and ramp-limited cooldowns reduces excursions into the dehydration regime; digital-twin implementations that forecast RH and heat flux have shown promise for supervisory scheduling.
Thermodynamic data regarding water vapor pressure over PA are frequently presented in temperature–composition phase diagrams for the binary H2O–P2O5 system, Fig. 3B.147 At operating temperatures of 150–170 °C, the equilibrium vapor pressure values are typically converted into relative humidity and plotted accordingly. For instance, Melchior et al. reported equilibrium water vapor pressures over pure PA and its mixtures with benzimidazole (bIm) at 182 °C, Fig. 3C.147 Their findings indicate that for neat PA, the equilibrium relative humidity is approximately 1.4% (∼150 mbar water vapor pressure). The addition of basic species such as bIm enhances PA condensation. At a H3PO4/bIm molar ratio of 9, a slightly higher vapor pressure is required to prevent dehydration. At a ratio of 3, the equilibrium vapor pressure increases by an order of magnitude.
At typical HT-PEMFC operating temperatures (∼160 °C), PA-doped PBI membranes at levels of 11–12 acid units per PRU is prone to condense into pyrophosphoric acid (H4P2O7) and higher oligomers (general formula Hn+2PnO3n+1) if the water vapor pressure falls below ∼120 mbar, corresponding to a relative humidity of ∼2%, which is close to actual cathode conditions under air stoichiometry of 2.0.1,93,94 When operating with reformate fuel, often containing residual water from the reformer, PA condensation is mitigated. However, in dry hydrogen environments at low stoichiometry or under OCV conditions, where cathodic water production is absent, condensation is more likely to occur. Although reversible, PA condensation proceeds with sluggish kinetics, and rehydration to orthophosphoric acid requires high water activity.148
Accordingly, we recommend ramp-down sequences that minimize time spent at dry OCV and prioritize brief, humidified holds before full shutdown to avoid deep condensation cycles.
In thermal cycling experiments between 80 and 160 °C (at 0.15 A cm−2), Lee et al. observed a threefold increase in area-specific ohmic resistance (from 0.14 to 0.45 Ω cm2) after 23 cycles, attributed to membrane swelling and interfacial degradation.149 Degradation in HT-PEMFCs operating on reformate is often exacerbated due to higher anode gas flow rates and increased water activity, leading to accelerated PA loss. Søndergaard et al. reported that the PA loss rate increased by 2.0–2.5 times when the temperature elevated from 160 to 180 °C, Fig. 3D.150 They also carried out a study of the MEA durability in a current range from 0.2 to 0.8 A cm−2, showing higher degradation at elevated current densities. A close correlation was found between the degradation and the PA loss. This is explained by an increased total gas flow of both hydrogen and air, carriers of the evaporated PA out of the cell, since a high current density is demanded from the MEA. An alternative reason is the electrochemical migration of PA through the MEA towards the anode, driven by parasitic movement of the PA-based ions due to the small fraction of the vehicular mechanism.150,151 Additionally, dynamic load cycling imposes high cathodic potentials that promote carbon support corrosion and catalyst coarsening. Repeated thermal expansion and contraction stress the MEA interfaces, weakening structural integrity. Variations in water content also alter PA concentration and volume in both the membrane and electrodes, impacting electrochemical stability. These observations underpin our suggested minimum AST elements for HT-PEMFCs (thermal cycle window, RH bounds, and gas-stoichiometry limits) to improve comparability across studies.
During start–stop events, the presence of liquid water at low temperatures is believed to enhance PA leaching. Thermal cycling exacerbates degradation, with high temperatures accelerating carbon corrosion, metal dissolution, and acid volatilization, while temperatures below 100 °C increase PA leaching due to washout by liquid water.1 Moreover, electrolyte redistribution arises from volume changes in PA caused by temperature-dependent expansion and water absorption. Low relative humidity and high temperatures yield highly concentrated PA; conversely, low temperatures and high humidity dilute PA due to its hygroscopicity. Schmidt et al. noted that polymer-based PBI membranes may buffer against electrolyte redistribution more effectively than inorganic matrices like SiC, as used in PAFCs.152 From a system standpoint, shutdown scripts that (i) purge to inert, (ii) control stack cooldown rate, and (iii) cap anode/cathode dew points mitigate both washout and over-concentration events.
In early start–stop tests, daily operation for 7 h at 150 °C using H2/air, followed by passive cooling over 17 h, resulted in an average voltage decay rate of 0.3 mV per cycle over a period of three years (∼900 cycles).153 Continuous operation at 0.2 A cm−2 with and without start–stop cycling showed that degradation increased from 5 to 11 mV h−1, with the cycling condition contributing a rate of 0.2 mV per cycle. Although ohmic resistance and ORR-related losses remained similar, cathodic mass transport losses increased significantly due to support corrosion.152 These values are included in the cross-study summary to benchmark contemporary results and to highlight how mitigation strategies shift the dominant loss from mass-transport to ohmic/activation components.
Besides catalyst degradation, mechanical fatigue induced by load cycling has also been confirmed via μ-CT and MEA thickness monitoring.46,154 Acid loss under high current densities correlates with elevated water production, thermal stress, and increased gas flow rates. Lee et al. proposed that PA interacts more strongly with water than with the polymer matrix, leading to acid segregation from the membrane.149 Zhou et al. subjected a cell to stoichiometric hydrogen cycling between 3.0 and 0.8 every 2 minutes at 150 °C and 0.4 A cm−2.155 During fuel starvation (λH2 = 0.8), the cell voltage dropped sharply and reversed polarity, indicating water electrolysis and reverse gas generation at the electrodes. This behavior leads to corrosion of carbon components (support, GDL, backing) and catalyst agglomeration, ultimately accelerating cell degradation and shortening service life. We therefore recommend system-level safeguards (starvation alarms from ΔP/ΔV signatures, brief battery assist on load steps, and enforced λ minima) to reduce reverse-current events.
At typical HT-PEMFC operating conditions (150–160 °C), the vapor phase above PA solutions consists predominantly of water vapor. However, the presence of volatile PA in the gas phase is non-negligible, Fig. 3B. The partial pressure of PA in the vapor phase has been estimated to be approximately 0.7 μbar at 160 °C and increases to about 3.7 μbar at 190 °C.4,18 Over long-term operation with continuous airflow, these values translate into measurable acid loss. For example, at a current density of 0.2 A cm−2 and reactant stoichiometries of λH2/air = 1.2/2.0, the corresponding acid loss rates are approximately 3.6 mg m−2 s−1 at 160 °C and 19 mg m−2 s−1 at 190 °C. To align studies, we propose reporting PA loss as μg cm−2 h−1 with standardized stoichiometry and temperature annotations, and to perform symmetrical sampling of both anode/cathode exhausts to reduce entrapment bias.
Experimental quantification of acid loss is typically performed by collecting trace amounts of acid carried in the anode and cathode exhaust gases. While extensive data on acid loss from PBI-based fuel cells are available in the literature, these results vary widely depending on cell design, operational parameters, and measurement methodology, as compiled in Table 2. Accurate assessment of acid loss at the scale of a few μg m−2 s−1 or μg cm−2 h−1 remains challenging. Minor acid entrapment in system components, including the GDLs, BPPs, flow fields, and tubing, can introduce significant error, as evidenced by inconsistencies among published values. We therefore add a minimum measurement guideline (sample-line heating, condensate traps, blank tubing tests, and instrument calibration with PA-water standards) to encourage reproducibility across labs.
Under steady-state conditions at a typical current density of 0.2 A cm−2, with low stoichiometry reactant flows and operating temperatures in the range of 150–160 °C, acid loss values generally fall within a narrow range of several μg m−2 s−1, Fig. 3D.88 In their degradation model fitting at 150, 170, and 190 °C, Oono et al. referenced acid loss data from PAFCs, which are notably higher than those observed for PBI-based systems.156 These benchmark values are integrated into the degradation-rate comparison (Table 1/Fig. 6) for quick cross-reference.
A clear dependence of acid loss on operating temperature is evident, correlating with the exponential increase in PA vapor pressure.79 This trend confirms that evaporation is the dominant mechanism driving acid loss, which is further influenced by gas flow rate and stoichiometry, acting as carriers for PA vapor. Therefore, both elevated temperatures and high gas flow rates significantly exacerbate acid loss. System controls (air λ caps during idle, staged cooldown, and minimized dry-air sweeps) are thus central to limiting evaporative flux without major efficiency penalties.
In addition to evaporative loss, another degradation pathway exists in systems using direct-feed organic fuels such as methanol or dimethyl ether. In such configurations, PA can react chemically to form organic phosphate derivatives (e.g., alkyl phosphates) within the membrane.157,158 These compounds are both more volatile and less conductive than orthophosphoric acid, contributing to accelerated performance decay in direct methanol and direct dimethyl ether HT-PEMFCs.159 However, due to the relatively low methanol crossover from reformate in HT-PEMFCs, this effect appears to have minimal impact on long-term durability.160 It is also important to highlight that liquid-phase water in contact with the membrane can result in the physical leaching of excess doped PA. Consequently, any condensation within the cell during operation, start-up, or shutdown must be carefully avoided to preserve electrolyte integrity. We flag this chemistry as an area for further disruptive concepts (e.g., acid-retentive binders, PA-tethering moieties, or reactive scavengers) to be tested under representative start/stop profiles.
To address the issue of PA loss, recent efforts have focused on improving acid retention using polymer membranes functionalized with quaternary ammonium (QA) groups, Fig. 4E.149,161 These polymers form ion pairs with PA (QA-H2PO4), effectively reducing acid mobility within the membrane. Exposure of such membranes to inert atmospheres with varying relative humidities has demonstrated improved PA retention compared to conventional PA-doped PBI membranes. We highlight these QA-functionalized systems, together with cross-linked/composite PBIs, as recent high-impact directions (past three years) that directly target start/stop-induced acid redistribution while preserving high-temperature conductivity.
 | ||
Fig. 4 (A) Mass loss of low (23–33 kDa) and high molecular weight (48–105 kDa) m-PBI, and modified (cross-linked, blended or copolymerized) PBI, during Fenton testing in 3 wt% H2O2 containing 4 ppm (mol) Fe(II) at 68–70 °C, [Reproduced with permission.18 Copyright © 2020, Springer Nature] (B) membrane thinning during the durability test for m-PBI and cross-linked ABPBI. Cross-sectional MEA images with m-PBI membranes (C) before and (D) after 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 h operation at 0.2 A cm−2 at 150 °C, [Reproduced with permission.162 Copyright © 2012 Elsevier B.V.] (E) chemical structures of PA-PBI, PA-QAPOH, PA-QASOH, PA-doped SnP2O7, and PWN70 ionomers. (F) Anhydrous proton conductivity of ionomers as a function of temperature. (G) Proton conductivity as a function of RH, 80 °C. [Reproduced with permission from ref. 163 Copyright Springer Nature 2020]. 800 h operation at 0.2 A cm−2 at 150 °C, [Reproduced with permission.162 Copyright © 2012 Elsevier B.V.] (E) chemical structures of PA-PBI, PA-QAPOH, PA-QASOH, PA-doped SnP2O7, and PWN70 ionomers. (F) Anhydrous proton conductivity of ionomers as a function of temperature. (G) Proton conductivity as a function of RH, 80 °C. [Reproduced with permission from ref. 163 Copyright Springer Nature 2020]. | ||
5. Recent advances in materials and component design
The membrane is a critical component in HT-PEMFCs, directly influencing proton conductivity, mechanical integrity, and chemical stability under harsh operating conditions. Traditional PA-doped PBI membranes have been widely used due to their excellent thermal and chemical stability.4 However, these membranes face significant challenges, particularly under dynamic operating conditions such as start/stop cycling, which is common in automotive and backup power applications. These cycles introduce rapid temperature fluctuations, humidity changes, and oxidative stress, all of which can accelerate membrane degradation, Fig. 4C and D. To address these limitations, two major strategies have emerged: crosslinking of PBI and the development of composite membranes. In what follows, we connect materials choices to their system-level implications (purge logic, ramp rates, stoichiometry limits) and indicate where multi-physics models (electrochemistry–transport–mechanics) explain observed hot spots and stress concentrations during start/stop.Crosslinking PBI involves introducing covalent bonds between polymer chains to form a three-dimensional network structure. This approach significantly enhances the mechanical strength, dimensional stability, chemical stability (Fig. 4A), and acid retention of the membrane. For example, tetrafunctional epoxy compounds such as N,N,N′,N′-tetraglycidyl-4,4′-diaminodiphenylmethane (TGDDM) have been used to crosslink PBI, forming a robust matrix that resists thermal and mechanical stress. A recent study introduced a novel proton-conducting additive, zirconium ethylenediamine tetramethylene phosphonate (ZrEDTMPS), which contains –PO3H2 groups capable of forming hydrogen bonds and facilitating proton transfer under low relative humidity (RH). When incorporated into a crosslinked PBI matrix, the resulting membrane, mPBI-TGDDM (10%) with ZrEDTMPS (30%), demonstrated outstanding performance, achieving proton conductivities of 0.122 S cm−1 at 100% RH, 0.050 S cm−1 at 50% RH, and 0.029 S cm−1 at 0% RH at 180 °C. Additionally, the membrane exhibited excellent oxidative stability, with only 2.1% mass loss in Fenton's reagent, and significantly reduced methanol permeability (6.38 × 10−8 cm2 s−1 at 70 °C), nearly an order of magnitude lower than Nafion 117.164 For context, representative sulfonated alternatives such as SPEEK or SPEEK-based composites typically show ∼10−3–10−2 S cm−1 at 160–180 °C under low RH, with markedly higher swelling; thus, cross-linked PBI platforms close the conductivity gap while maintaining dimensional stability at HT.
A novel HT-PEM system was developed using a flexible branched polymer (broPBI), a water-soluble proton conductor (PBSA), and a multifunctional crosslinker rich in imidazole rings (BrABPBI). The branched architecture of broPBI introduces a large free volume and a high density of terminal groups, which enhances proton transport and mechanical strength. BrABPBI facilitates covalent crosslinking between broPBI and PBSA, forming a robust, membrane-wide network that improves mechanical integrity and oxidative resistance while minimizing PBSA leaching. Additionally, the crosslinking process converts secondary amines in broPBI and PBSA into tertiary amines, increasing their basicity and boosting the membrane's acid doping capacity. Critically, multi-physics simulations indicate that these cross-linked/branched architectures reduce start/stop-induced interfacial shear by limiting membrane swelling hysteresis, which helps prevent early delamination near inlet lands where heat and current concentrate.
However, excessive branching or crosslinking can negatively impact membrane performance, so a balanced integration of both strategies was employed. The optimized composite membrane, broPBI-BrABPBI (20%)–PBSA (50%), demonstrated excellent proton conductivity at 180 °C: 0.140 S cm−1 at 100% RH, 0.0701 S cm−1 at 50% RH, and 0.0310 S cm−1 at 0% RH. These results highlight the membrane's strong mechanical properties, high conductivity, and oxidative stability, making it a promising candidate for HT-PEMFC applications.165 These properties are particularly advantageous under start/stop conditions. During startup, the membrane experiences thermal shock as the temperature rapidly increases from ambient to operating levels. The crosslinked network structure of PBI membranes provides enhanced dimensional and thermal stability, reducing the risk of cracking or deformation. Moreover, the crosslinking minimizes PA leaching, which is critical because acid redistribution during thermal cycling can lead to uneven conductivity and localized degradation. During shutdown and cooldown, condensation and rehydration can occur, potentially leading to acid dilution or migration. The improved acid retention of crosslinked membranes helps maintain consistent proton conductivity and prevents performance loss during these transitions.1,166 To support practitioners, we map these materials behaviors to controller set-points: limiting cooldown rates, enforcing minimum dew-point margins, and avoiding prolonged dry-OCV holds, operating choices that our review shows to reduce the dominance of ohmic losses relative to activation losses after cycling.
Composite membranes offer another powerful strategy to enhance the performance of PBI-based systems, Fig. 4E–G. These membranes incorporate inorganic or organic fillers into the polymer matrix to improve mechanical strength, acid retention, and proton conductivity. Inorganic fillers such as silica (SiO2), zirconium phosphate (ZrP), and graphene oxide (GO) are commonly used due to their ability to interact with PA and create additional proton conduction pathways, Fig. 5J and K. For instance, functionalized GO can form stable hydrogen-bond networks with PBI and PA, enhancing both conductivity and structural integrity. Another promising approach involves the integration of phosphonic acid-functionalized siloxane into sulfonated and imidazole-functionalized poly(2,6-dimethyl-1,4-phenylene oxide) (PPO) matrices. These materials form Si–O–Si crosslinked networks that significantly improve thermal and oxidative stability. The resulting membranes have demonstrated high proton conductivities of 0.1131 S cm−1 at 80 °C and 0.1049 S cm−1 at 160 °C under low RH conditions, making them suitable for a wide operational temperature range.167 Compared side-by-side, these composite PPO systems rival PA-PBI conductivity at 160 °C while offering lower PA mobility; however, their start/stop stability still hinges on filler–polymer interfacial adhesion under thermal gradients, an area where standardized ASTs are needed for fair benchmarking. Under start/stop cycling, composite membranes provide several advantages. The inorganic fillers act as acid reservoirs, buffering the membrane against acid loss during dry-hot conditions and rehydration during cooldown. They also enhance mechanical robustness, allowing the membrane to withstand repeated expansion and contraction without delamination or pinhole formation. Furthermore, the oxidative stability of these membranes is improved, reducing the risk of radical-induced degradation during transient phases when both hydrogen and oxygen may be present in the same chamber.43 The integration of crosslinking and composite strategies is increasingly being explored to create multifunctional membranes that combine the benefits of both approaches. Such hybrid membranes are expected to offer superior performance under the demanding conditions of HT-PEMFCs, including high temperatures, low humidity, and long operational lifetimes. Future research is focusing on the development of self-healing membranes, green synthesis methods, and scalable fabrication techniques to facilitate commercial deployment. We flag these as disruptive concepts relative to traditional PA-PBI, particularly (i) PA-tethering chemistries, (ii) self-healing crosslinks that re-form after thermal strain, and (iii) greener P-sources, each of which directly targets acid redistribution and interfacial fatigue observed during start/stop.
 | ||
Fig. 5 (A) The HAADF-STEM images of the Pt3Co hierarchical NWs with (B) the corresponding EDS.168 The specific and mass activities of the (C) hierarchical Pt3Co NWs/C catalyst before and after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, 15 000, 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 and 20 000 and 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 potential cycles, compared to (D) the commercial Pt/C catalyst. The durability tests were carried out at room temperature in 0.1 M HClO4 and a scan rate of 100 mV s−1.168 (E) The HR-TEM image Pt/CNC with (F) polarization curves showing catalyst degradation before and after accelerated durability tests.169 (G) Schematic representation of the CNT@SiO2-Pt in the HT-PEMFC CL, with (J) MEA performance showing the initial polarization and power density curves of Pt/C, CNT-Pt, CNT@SiO2-Pt, and (K) 100-h durability test at a constant current density of 0.2 A cm−2, 160 °C in H2/O2 [Reproduced with permission from ref. 170 Copyright © 2021, Springer-Verlag GmbH Germany] (H) the TEM image of gC3N4 nanosheets, with (I) MEA durability tests at a constant load of 0.2 A cm−2 for 100 h, 150 °C [Reproduced with permission from ref. 138 Copyright © 2020, American Chemical Society]. 000 potential cycles, compared to (D) the commercial Pt/C catalyst. The durability tests were carried out at room temperature in 0.1 M HClO4 and a scan rate of 100 mV s−1.168 (E) The HR-TEM image Pt/CNC with (F) polarization curves showing catalyst degradation before and after accelerated durability tests.169 (G) Schematic representation of the CNT@SiO2-Pt in the HT-PEMFC CL, with (J) MEA performance showing the initial polarization and power density curves of Pt/C, CNT-Pt, CNT@SiO2-Pt, and (K) 100-h durability test at a constant current density of 0.2 A cm−2, 160 °C in H2/O2 [Reproduced with permission from ref. 170 Copyright © 2021, Springer-Verlag GmbH Germany] (H) the TEM image of gC3N4 nanosheets, with (I) MEA durability tests at a constant load of 0.2 A cm−2 for 100 h, 150 °C [Reproduced with permission from ref. 138 Copyright © 2020, American Chemical Society]. | ||
Efforts to optimize the crystal phase, particle size, morphology, and surface structure of Pt NPs have brought significant improvements in intrinsic activity and stability, helping to reduce material costs. However, such optimization is approaching its practical limits. Although novel nanostructures and complex synthesis methods have demonstrated excellent catalytic performance, their scalability and cost-effectiveness for commercial deployment remain significant challenges. Consequently, research has shifted toward nanostructured Pt-alloy electrocatalysts, which have shown promising performance for the ORR and offer more practical prospects for industrial application.141 Moreover, under the conditions most damaging in start/stop (high potentials, transient air fronts), alloy choice and support graphitization primarily govern whether Pt dissolution or carbon corrosion dominates, guidance we make explicit in the comparative notes linked to Table 1/Fig. 6.
 | ||
| Fig. 6 (A) Compiled literature on HT-PEMFC MEA durability performances and corresponding degradation rates under different operating conditions (in A cm−2).171 (B) The average HT-PEMFC membrane temperatures at different voltages.172 (C) The stress distributions on the middle plane of the membrane at the average membrane temperature 40, 80, and 120 °C (left to right).172 (D) Performance of a residential microgrid with HT-PEMFC-based micro-CHP system, showing electrical performance under the supervised case study. (E) The structure of micro-CHP system.14 The electrical and thermal demand of the system in a (F) winter, and a (G) summer day.14 | ||
Currently, Pt alloys, along with core–shell and Pt-skin architectures, represent the leading catalyst designs for ORR.173 Bimetallic and trimetallic Pt-based systems provide an effective route for enhancing both ORR activity and catalyst durability while decreasing overall Pt usage. The choice of alloying elements is often based on their inherent catalytic properties and their ability to synergistically interact with Pt through bifunctional mechanisms. Alloying Pt with transition metals such as Co, Ni, Fe, and Cu can enhance catalytic performance and durability via the so-called “anchor effect”, Fig. 5C and D.140 These transition metals are typically selected based on volcano plots that correlate ORR activity with the binding energies of *OH and *OOH intermediates. Important for HT-PEMFCs in PA media, alloying that downshifts the Pt d-band (e.g., PtCo/PtNi) tends to weaken phosphate adsorption and improves ORR at high temperature; alloys that geometrically disrupt three-fold phosphate binding (e.g., PtAu skins) similarly mitigate poisoning, both effects relevant to start/stop when PA redistribution alters local acidity.
The underlying mechanism involves electronic structure modification. The subsurface alloy layer can tune the d-band center of surface Pt atoms, shifting it upward or downward depending on the secondary metal, which in turn modifies the adsorption strength of ORR intermediates.174 This fine-tuning of adsorption energetics can enhance catalytic activity in PEMFCs.
In HT-PEMFCs, two key concerns with Pt-alloy catalysts are (1) particle stability, and (2) altered adsorption behavior at the active sites. Under HT-PEMFC operating conditions, Pt alloys with non-noble metals tend to undergo dealloying to varying extents.18 Pt alloys with refractory metals such as Cr and Zr, originally considered chemically stable, have shown resistance to corrosion but are still electrochemically unstable, as evidenced by metal leaching in PAFCs.175 Thus, selection should weigh ORR gains against start/stop dealloying liability; we recommend reporting both ECSA retention and alloy composition drift after standardized start/stop ASTs to enable apples-to-apples comparisons.
Furthermore, alloying Pt with a secondary metal affects molecular adsorption properties due to scaling relationships.176 These relationships govern the adsorption energies of O2 and phosphate species on Pt-based surfaces. Achieving weaker binding for both O2 and phosphate ions is desirable, as it leads to enhanced ORR kinetics and reduced susceptibility to phosphate poisoning. This provides a pathway for developing Pt-alloy catalysts with improved performance and lower Pt loadings.
PtCo, PtNi, PtFe, and PtAu systems have demonstrated improved ORR activity alongside decreased adsorption of poisoning species such as phosphate and CO, making them suitable for HT-PEMFC applications.177–179 For example, PtCo and PtNi exhibit a downshift in the d-band center relative to pure Pt, resulting in weaker acid adsorption. In contrast, PtAu alloys display an upshifted d-band center, which, coupled with geometric effects (e.g., disruption of threefold phosphate binding sites due to Pt atom isolation by Au), leads to lower phosphate coverage and enhanced ORR activity.180 In practice, PtCo/PtNi show strong performance at high load cycling (less OCV exposure), whereas PtAu-like skins can be preferable where OCV/air fronts are frequent, reflecting different dominance of dissolution vs. poisoning mechanisms.
Ruthenium (Ru), known for its oxophilic nature, is frequently incorporated into binary or polymetallic catalysts to enhance CO tolerance.181 However, high cost of Ru, and its scarcity, render PtRu alloys impractical for large-scale applications. To address this, Co has been explored as a more abundant alternative to Ru, particularly in PtCo alloys designed to mitigate CO poisoning.182
Yano et al. synthesized monodisperse Pt and PtM (M = V, Cr, Fe, Co, Ni) alloy NPs supported on carbon via a one-pot method, achieving particle sizes between 2.0–2.5 nm, independent of metal loading (10–55 wt%).183 The specific ORR activity of these PtM catalysts was 1.3–1.8 times higher than that of pure Pt (0.82 mA cm−2 at 0.8 V vs. RHE), with the activity trend as follows: Pt < PtNi < PtFe < PtCo < PtV < PtCr. We include these as representative high-impact contributions and note that post-2022 reports extend this trend to PA media at 160–180 °C with improved phosphate tolerance.
Beyond transition metals, Pt is also alloyed with noble metals when performance or long-term durability is prioritized. For example, PtPd and PtRh alloys have been extensively studied in various nanostructured forms.184–186
While bimetallic catalysts remain the focus of significant industrial and academic interest, recent work has turned toward polymetallic (ternary and quaternary) systems to further address the challenges associated with HT-PEMFCs.187–189 The vast compositional space of multimetallic systems remains largely unexplored, but ternary Pt-based alloys have already shown enhanced catalytic activity, stability, and resistance to poisoning compared to commercial Pt/C catalysts. These ternaries are a promising disruptive direction; however, we emphasize manufacturability and composition control under start/stop (avoiding preferential leaching) as gating criteria for practical adoption.
A representative example is the carbon-supported PtRuCo ternary catalyst, which demonstrated resilience to CO poisoning during air injection into a CO-contaminated hydrogen stream.190 Similarly, Almeida et al. synthesized PtRhNi catalysts via a wet-chemistry route and investigated their ethanol electrooxidation behavior.191 XRD revealed lattice distortions due to the differing atomic radii of Rh and Ni, and catalytic selectivity was found to depend strongly on surface composition, highlighting the importance of composition control during synthesis.
Vedyagin et al. studied a PtPdRh ternary alloy and reported that metal ratios significantly influence both catalytic activity and thermal stability.192 Across most alloy systems studied, improvements in ORR performance, stability, or resistance to poisoning have been consistently observed, substantiating alloying as a robust strategy for advancing HT-PEMFC catalyst design.
Catalyst supports play a critical role in determining the performance, stability, and cost-effectiveness of fuel cell (FC) electrocatalysts. They influence the dispersion, utilization, and stability of catalyst NPs, directly impacting particle size distribution, degree of alloying, and overall electrocatalytic activity. In membrane electrode assemblies (MEAs), the support material also governs mass transport by facilitating effective contact between reactants and active catalytic sites.193,194 Consequently, the choice of support material strongly affects catalyst activity, durability, and fuel cell efficiency.195
In high-temperature proton exchange membrane fuel cells (HT-PEMFCs), carbon (C) remains the most widely employed support material for Pt-based electrocatalysts due to its high surface area and excellent electrical conductivity. However, carbon is prone to electrochemical oxidation under the harsh operating conditions of HT-PEMFCs, elevated temperatures and the presence of concentrated PA, leading to NP detachment and severe performance degradation.196 For start/stop, the balance of mechanisms is clear: graphitized carbons suppress support corrosion (delaying mass-transport loss), while less-ordered carbons enable higher dispersion but corrode sooner, shifting degradation toward ECSA loss and CL thinning.
Given these challenges, extensive efforts have been dedicated to developing more robust and chemically stable support materials. Strong adhesion between the precious metal catalyst and the support is essential for improving long-term durability. In addition to physical anchoring, the support can electronically influence the catalyst by modifying the density of states in the metal's d-band, thereby altering catalytic properties.
CB with its graphitic and mesoporous structure, remains a common choice. Typical examples include Vulcan XC-72, Black Pearls 2000, Ketjen EC 300J, and EC 600JD, all of which offer high surface areas (>100 m2 g−1) and good electronic conductivity. CB is generally classified into acetylene black (AB) and furnace black (FB), based on the synthesis route.197
To enhance the structural order and chemical stability of carbon supports, GCB is produced by recrystallizing CB at 2500–3000 °C in an inert atmosphere, yielding crystalline domains with surface areas in the range of 100–300 m2 g−1.198 Increased graphitization is associated with improved corrosion resistance and durability under HT-PEMFC conditions.199
Hollow graphitic spheres (HGS) and carbon nanocages (CNCs) are 3D carbon nanostructures that have demonstrated enhanced stability as catalyst supports in PEM fuel cells, Fig. 5E and F.169 The electrochemical durability of CNCs is notably improved by a high nitrogen content and a high degree of graphitization. In particular, the Pt/CNC (1000) catalyst exhibited negligible changes in composition and morphology following AST, maintaining performance over ∼3000 hours of operation, Fig. 5F. This exceptional stability is attributed to the synergistic effects of CNCs' graphitized framework, robust interaction with Pt NPs, and strong resistance to oxidative degradation. We identify these as recent high-impact advances that specifically address start/stop carbon corrosion while keeping electronic pathways intact.
Carbon nanotubes (CNTs) have attracted substantial interest due to their excellent electrical conductivity, mechanical strength, and chemical stability, Fig. 5G.200,201 However, pristine CNT surfaces are chemically inert and often incompatible with Pt NPs, leading to agglomeration and poor dispersion.202 Surface functionalization techniques, such as acid treatment, high-temperature KOH activation, and annealing, are employed to introduce anchoring sites and improve metal–support interactions.203
Graphitized CNTs (GCNTs) offer even higher electrochemical durability compared to standard CNTs Fig. 5E and F. A comparative study using electrochemical stress testing (triangle-wave AST) showed that Pt/GCNT systems exhibited the highest Pt utilization, electrochemical stability, and overall HT-PEMFC performance among tested carbon supports, despite a higher electrical resistance than Pt/CNT and Pt/CB analogues.204
Incorporating nitrogen-functionalized carbon supports, bearing groups such as amides, imides, or lactams, has shown promise in both conventional and high-temperature PEMFCs. These N-groups can interact with sulfonate or phosphate groups in the ionomer or electrolyte, promoting uniform ionomer distribution and improving oxygen mass transport.205 In HT-PEMFCs, the basic N-sites also facilitate PA retention via acid–base interactions, forming a stable PA network across the catalyst layer. While this can improve performance, careful control of acid content is necessary to avoid excessive absorption, which may alter local conductivity and water management. At the system level, these supports can reduce the need for aggressive humidification transients during startup, easing control complexity and limiting fuel-efficiency penalties from purges.
Graphene-based materials have emerged as another class of promising catalyst supports, owing to their exceptional properties: high surface area, thermal and electrical conductivity, mechanical strength, and chemical inertness, Fig. 5H and I.206 However, the intrinsic two-dimensional stacking behavior of pristine graphene often limits gas permeability and blocks access to catalytic sites at high current densities.207
Heteroatom doping of carbon materials (e.g., N, S, B, P) is a widely used strategy to tune their surface chemistry and electronic structure, enabling tailored interactions with metal catalysts and improved performance in energy conversion devices, Fig. 5J and K.208 We highlight N-doped, graphitized frameworks as a convergent design that balances dispersion, PA retention, and corrosion resistance, particularly relevant to mitigating start/stop reverse-current damage.
The durability and performance of HT-PEMFCs under dynamic start–stop conditions are critically affected by the behavior of the GDLs and BPPs. These components are exposed to thermal cycling, oxidative stress, and PA migration, leading to mechanical degradation, loss of conductivity, and compromised interfacial integrity. Recent research has focused on developing corrosion-resistant materials, optimizing transport properties, and enabling scalable manufacturing strategies for long-term operation above 120 °C in PA-rich environments. We also discuss real-world integration constraints, cost of coatings, contact-resistance targets, and control complexity, so that component innovations can be evaluated alongside system-level trade-offs.
Conventional GDLs typically employ PTFE-coated carbon substrates for hydrophobicity and acid resistance. However, alternative coatings, such as polyaniline (PANI)-based composites, are now being explored for their enhanced thermal stability, acid retention, and electrical conductivity under HT-PEMFC conditions.209 PANI-modified carbon paper has demonstrated superior performance and stability, particularly under high-temperature and high-acid-loading environments. We classify these as disruptive GDL concepts that couple materials and wetting control, aligning with field-relevant start/stop transients.
Advanced GDL designs also incorporate laser-perforation and microporous structuring to improve mass transport and water management.210 Laser-patterned GDLs have been shown to promote localized accumulation and accelerated removal of product water on the cathode side, thereby enhancing oxygen diffusivity and mitigating flooding. Tailoring porosity, channel geometry, and pore alignment has resulted in up to 7–8% improvement in peak power output under dynamic loading conditions.211
Investigations using synchrotron radiography and environmental-SEM (ESEM) have enabled visualization of water distribution changes following microstructural modifications of the GDL and MPL. While laser perforation improves dry-condition performance due to the creation of hydrophilic regions, it also leads to PTFE loss and serious flooding at high relative humidity. In contrast, mechanically machined perforations offer a more balanced water distribution and superior performance under humidified conditions. To assist practitioners, we recommend reporting a minimal set of metrics – contact angle, tortuosity, PTFE content before/after AST, and interfacial resistance, to standardize comparisons across GDL concepts.
Simulations using the ANSYS PEM Fuel Cell Module support these findings, showing that increasing the GDL contact angle to ∼150° improves water evacuation and overall performance. However, gains plateau beyond this point. Experimental validation confirms that PTFE-coated GDLs with high hydrophobicity outperform uncoated variants, particularly under wet operating conditions. Additionally, increasing GDL porosity from 0.4 to 0.6 enhances gas transport and liquid water removal, further improving cell performance.212 These results reinforce the utility of multi-physics/digital-twin workflows to forecast start/stop hot spots and guide control set-points (e.g., airflow limits, ramp rates) that minimize flooding or PA washout.
Incorporating materials such as TiC, iridium (Ir), or titanium dioxide (TiO2) at the GDL/MPL interface has been shown to reduce interfacial contact resistance and facilitate thermal regulation. In one study, stable operation at 1.14 A cm−2 for 50 hours was achieved using a TiO2-coated GDL under low-humidity conditions.
BPPs must retain high electrical conductivity while withstanding PA corrosion and electrochemical transients during start–stop cycles. Current strategies focus on hybrid materials, combining stainless steel or polymer substrates with carbon-based or ceramic coatings, such as graphite, titanium nitride (TiN), or TiC. These coatings improve both mechanical strength and corrosion resistance, while maintaining low interfacial contact resistance. From a stack perspective, uniform clamping pressure and coolant distribution are as important as coating choice; we therefore recommend documenting pressure maps and coolant ΔT in durability studies to clarify system-level contributions to BPP and GDL aging.
Improved BPP materials and coatings not only extend component lifetime but also contribute to more stable voltage output, reduced voltage degradation rates, and enhanced MEA durability under thermal and electrochemical cycling. Continued innovation in surface engineering, microstructural tuning, and materials integration is essential to meet the demanding requirements of next-generation HT-PEMFC systems. Finally, we connect materials advances to the broader need for AST standardization: reporting common start/stop protocols and durability KPIs (e.g., mV per cycle, % ECSA loss, μg cm−2 h−1 PA loss) will make these innovations comparable and actionable for practitioners.
6. System and operational strategies for durability
A comprehensive understanding of MEA lifetime limitations is essential for developing effective mitigation strategies and achieving the durability targets required for both vehicular and stationary HT-PEMFC applications. System-level and operational strategies have emerged as crucial tools to extend cell lifetime and maintain high performance over prolonged operation. These strategies include thermal management protocols, optimized reactant stoichiometry, humidity control, voltage regulation, and intelligent start–stop procedures. Some results of comprehensive studies on degradation mitigation are presented in Table 1 and Fig. 6A. Additionally, advances in materials engineering, such as hydrophobic and corrosion-resistant coatings, tailored porosity in GDLs, and acid-retentive membranes, must be complemented by robust operational frameworks to fully realize their benefits. To aid practitioners and improve comparability, we recommend reporting a minimal durability KPI set (e.g., mV per cycle, % ECSA loss per 100 h, μg cm−2 h−1 PA loss, and hours to 10% power fade) alongside each strategy, and mapping each operating window to the dominant degradation mode (low-load/OCV → carbon corrosion & Pt dissolution; high-load → PA evaporation/migration).This section provides an overview of the system and operational strategies developed to enhance the durability of HT-PEMFCs. Emphasis is placed on the interplay between component-level innovations and system-level controls, with a focus on mitigating key degradation pathways through optimized fuel cell operation. Where appropriate, we also point to multi-physics modeling (electrochemistry–transport–thermal–mechanical) as a means to identify local hot spots and stress concentrations that guide controller set-points (purge timing, ramp rates, stoichiometry limits).
Unlike previous studies primarily focused on pure hydrogen-based micro combined heat and power (μ-CHP) systems, Xie et al. designed and modeled a methanol-fed μ-CHP system based on HT-PEMFC, Fig. 6D–G.14 To enable coordinated operation between the μ-CHP system and other energy and storage components within a residential microgrid, a novel two-layer control and management system is developed. This approach differs from existing literature, which often separates control from energy management systems (EMS) and focuses exclusively on either real-time operation or long-term planning. The proposed system integrates both aspects within a unified framework. We highlight this as a representative system-level milestone because it links stack-transient constraints (e.g., start/stop exposure) to household-level scheduling—precisely the integration gap that often drives real-world degradation.
The control architecture consists of an (1) upper layer, an optimization-based EMS responsible for day-ahead hourly generation scheduling, with the objective of minimizing the economic cost associated with purchasing electricity and methanol; and a (2) lower layer, a decentralized real-time control scheme that manages power sharing, maintains stable DC bus voltage, and regulates the hot water tank (HWT) temperature. This layer is designed to operate under both supervised and unsupervised modes. In the unsupervised mode, it autonomously manages energy flows, while in the supervised mode, it follows generation schedules provided by the upper layer. From a durability perspective, such hierarchical control can explicitly constrain OCV dwell time, enforce purge-before-idle rules, and cap stoichiometry overshoot during load steps, thereby limiting reverse-current transients during start/stop.
Key contributions include (1) the detailed design and dynamic modeling of the methanol-reformed HT-PEMFC-based μ-CHP system, incorporating both electrical and thermal dynamics suitable for system-level analysis (Fig. 6F and G), (2) the development of the two-layer control and management system that addresses both daily energy reservation and real-time operational demands, and (3) the implementation of control algorithms for both supervised and unsupervised scenarios (Fig. 6D), providing flexible operation adaptable to diverse user requirements, while maintaining a simple and scalable control structure. We note that embedding degradation-aware costs (e.g., penalizing OCV time or temperature excursions) into the upper-layer objective function is a practical route to convert lab insights into operational savings.
The thermal control subsystem is engineered to maximize utilization of recoverable fuel cell waste heat, while maintaining the HWT temperature within a prescribed optimal range. This is achieved through two coordinated controllers: one managing the coolant radiator and another managing the heat pump. The radiator activates only when the HWT temperature reaches its maximum allowable limit (Ttank,max), thereby preventing overheating by dissipating excess heat and limiting further charging. Conversely, the heat pump operates when the HWT temperature falls to its lower limit (Ttank,min), adjusting its power input to ensure the temperature remains within the safe operating window. Both controllers operate at high temporal resolution within the lower control layer, enabling rapid response to fluctuating thermal demands. Thermal management also governs PA volatility and membrane stress; we therefore recommend logging stack ΔT, coolant flow, and temperature ramp rates in durability reports to link transient heat loads to acid loss and interfacial delamination.
The integration of the HT-PEMFC μ-CHP system with ancillary components including battery storage, grid interface, hot water tank, and heat pump creates a comprehensive multi-energy residential microgrid. Coordinated operation of this system is achieved through the proposed two-layer control scheme: the upper layer employs optimization based on forecasted energy prices and consumption patterns to plan energy generation, while the lower layer uses rule-based control to allocate power instantaneously across energy sectors. In practice, this coordination buffers fast load changes with the battery, reducing start/stop frequency and OCV dwell – direct levers on catalyst and carbon-support degradation.
Compared to prior research, this study thoroughly characterizes the μ-CHP system's behavior, demonstrating substantial efficiency improvements through waste heat recovery, which mitigates the traditionally low efficiency of fuel cells at high power outputs and enables flexible operation. Extensive simulation case studies validate the proposed control methodology, showing notable benefits in fuel consumption reduction, real-time power tracking, and energy reservation. The supervised control scenario consistently outperforms others, delivering the best overall operational efficiency. Seasonal analysis indicates additional economic advantages during winter months due to increased thermal demand. We encourage authors of similar studies to co-report degradation KPIs and control variables (purge gas consumption, efficiency penalty) to clarify cost–durability trade-offs.
Future research will focus on enhancing next-day energy consumption prediction models and conducting a comprehensive 4E (energy, economic, environmental, and engineering) assessment. Experimental validation will be pursued using a Hardware-in-the-Loop (HIL) residential testbed currently under construction, to further substantiate the practical feasibility and performance of the developed control strategies and system configurations. HIL testbeds are also ideal for validating digital-twin durability models (coupled electrochemistry–CFD–FEM), enabling on-line lifetime prediction and predictive maintenance triggers.
To mitigate the inherently slow dynamic response of the HT-PEMFC system and accommodate the variability of renewable energy sources, battery storage is incorporated to provide auxiliary power support within the microgrid. Power converters play a critical role in this system by enabling efficient electrical energy conversion between different forms. Specifically, a buck/boost DC/DC converter is deployed for the fuel cell system, while the grid interface utilizes both AC/DC and buck converters. Key semiconductor devices such as Insulated Gate Bipolar Transistors (IGBTs) and Metal-Oxide-Semiconductor Field-Effect Transistors (MOSFETs) serve as the primary switching components, with their switching frequencies critically influencing the precision and efficiency of energy conversion. Converter control should also enforce voltage-rate limits at the stack terminals to cap transient potential spikes during start/stop and load steps.
The overall control and management framework is designed to coordinate the operation of the μ-CHP system with the broader microgrid. Based on differing objectives and sampling times, the system architecture is stratified into two layers: an upper management layer and a lower controlling layer. The upper layer employs an optimization-based EMS, which assumes availability of next-day forecasts for energy prices and household consumption. It generates day-ahead hourly generation schedules aimed at minimizing the combined cost of electricity and methanol procurement. The EMS output includes on/off status signals and power setpoints for both the fuel cell and the grid, which are subsequently communicated to the lower layer for real-time execution. Including “health-aware” constraints (maximum OCV minutes per day, maximum thermal cycles per day) in the EMS is a pragmatic pathway to stack life extension with minimal cost penalty.
Conversely, the lower controlling layer operates based on instantaneous system information to allocate load dynamically across multiple energy sources. This layer supports two operational scenarios: supervised and unsupervised control. The supervised control mode integrates the upper-layer EMS's pre-calculated plans with real-time measurements to manage power distribution effectively. The unsupervised mode enables autonomous operation of the system relying solely on instantaneous information, providing robustness in cases where the EMS input is unavailable due to absent price signals or consumption data. We suggest logging controller actions (purges, ramps, idles) as part of durability datasets to connect control decisions with observed degradation.
Numerous studies have investigated the degradation effects of start–stop cycling and load variations on HT-PEMFCs, revealing critical insights into performance loss mechanisms and mitigation strategies, Fig. 7C. To consolidate scattered results, we provide (Table 1/Fig. 6A) a cross-study roll-up of typical ranges: start–stop without mitigation ≈0.2–0.4 V per cycle vs. ≤1 mV per cycle with optimized purges; low-load cycling with OCV dwells ≈100–400 μV h−1; high-load cycling without OCV ≈ 30–40 μV h−1; thermal cycling rates scale with temperature swing and ramp rate.
 | ||
| Fig. 7 (A) The scheme of (a) the reverse-current mechanism, with the corresponding dual-cell mechanism during a start–stop protocol. (B) The potential profile along a fuel cell gas channel [(A and B) Reprinted with permission from ref. 213 Copyright©2020, Elsevier Ltd]. (C) Summary of fuel cell degradation mitigation strategies under start–stop protocol. [Reprinted with permission from ref. 30 Copyright©2022, Elsevier Ltd]. (D) The HT-PEMFC stack polarization curves during load cycling with reformate gas operation, with gas-purging strategy.34 (E) The voltage evolution comparison of HT-PEMFC MEAs during start–stop cycling, under degradation mitigation strategy of varying pure H2 and N2-diluted feeds.13 | ||
In a daily start–stop cycling test conducted by Pinar et al., the cell operated at 0.2 A cm−2 and 160 °C, idling overnight at 25 °C.72 During temperature excursions below 120 °C, the cell was purged with N2/N2, resulting in an overall voltage loss rate of 12.6 μV h−1, or approximately 600 μV per cycle over 115 days. Without mitigation, typical performance losses per cycle often exceed 200 mV, although some reports suggest lower degradation rates. Oyarce et al. explored various start–stop strategies, including hydrogen consumption (fuel shutoff under load), anode air purge, oxygen consumption (air shutoff under load), and cathode hydrogen purge, and found that oxygen consumption and cathode hydrogen purge reduced degradation rates by an order of magnitude compared to no-purge conditions.74 Similarly, Kannan et al. reported a degradation rate as low as 26 μV per cycle over more than 1500 cycles in a 5-cell stack operating at 0.25 A cm−2, estimating approximately 20 mV loss per cycle including steady-state degradation. These results underscore that dominant mechanisms shift with protocol: start/stop → reverse-current carbon corrosion; low-load with OCV → Pt dissolution/oxide cycling; high-load → PA loss and ohmic rise.54
Load cycling studies commonly involve low current densities (0–0.2 to 0.5 A cm−2) with frequent dwelling at OCV. Higher degradation rates (100–400 μV h−1) occur under OCV or near-OCV conditions, with activation polarization identified as the dominant degradation mechanism. Potential cycling between 0.6–0.7 V and 1.0 V corroborates these findings, emphasizing catalyst degradation in high potential regimes. It needs to be further considered that local areas in a MEA can also experience varying potential profiles across the cell due to the changing gas fronts, Fig. 7B. Gas management, therefore, has to go hand-in-hand with the gas distribution system. Thomas et al. further highlighted the importance of relaxation time during low load cycling, showing a 2-minute relaxation time reduced degradation to 36 μV h−1, 63% of the degradation under constant load.214 Li et al. studied effects of varying voltages (0.9 V, 0.7 V and 0.5 V) and flow arrangement (co-flow and counter flow) on temperature, current density, proton conductivity and stress distributions of HT-PEMFC, Fig. 6B and C.172 They found that the maximum stress increased when a lower voltage was adopted, and the counter-flow arrangement provided a more uniform stress distribution than that of co-flow arrangement. We recommend AST reports include gas-front mapping (e.g., segmented cell or reference electrodes) to capture these local potential excursions that standard bulk metrics miss.
Thermal cycling introduces additional degradation pathways. Liu et al. demonstrated that cycling between 140 °C and 180 °C at 0.2 A cm−2 caused significant loss of PA and membrane degradation, evidenced by high PA loss rates (94.2 ng H3PO4 per h) and increased hydrogen crossover.77 Hjuler et al. reported a degradation rate of 0.48 mV per cycle during thermal cycles between 50 °C and 175 °C, over 240 cycles (∼4000 h).63 Qi et al. proposed a mitigation strategy involving a resistor insertion and nitrogen purging during OCV periods that maintained cell voltage and limited cathode Pt particle growth during thermal cycling.80 Standardizing thermal-AST parameters (ΔT, ramp rate, dwell humidity) would substantially reduce inter-study scatter.
Dynamic operation studies typically use dry H2 and air with stoichiometries around 2.0/4.0; however, higher stoichiometries (e.g., 22.6/25.2) significantly accelerate PA loss due to evaporation, reducing cell durability by more than 50% at 0.2–0.6 A cm−2 and temperatures of 160–180 °C. At elevated current densities, PA concentration gradients develop across the MEA, exacerbating PA loss and accelerating degradation. Degradation mitigation strategy of reformate gas flushing the anode compartment before the cell start-up and after the cell shutdown showed promising MEA durability, Fig. 7D. This is explained by the deliberate CO-poisoning of the Pt surface and, therefore, protection against PA adsorption. We flag this as a practical system-level tactic; the fuel-efficiency penalty and control complexity should be reported so operators can weigh costs vs. durability gains.
Load cycling stress can be categorized by current density ranges: low load (0–0.2/0.5 A cm−2) and high load (0.2–1.0 A cm−2). Low load cycling incurs high degradation primarily due to catalyst agglomeration and carbon support corrosion, driven by high electrode potentials near OCV. Consequently, degradation rates can reach 100–300 mV h−1. Under high load cycling, PA loss dominates, leading to membrane deterioration such as reduced proton conductivity and increased hydrogen crossover, which can lead to e.g. reverse-current degradation mechanisms (Fig. 7A), although such effects may be less evident in short-term tests. Prolonged OCV exposure is particularly detrimental, causing voltage degradation exceeding 400 mV h−1 due to ECSA loss and catalyst layer damage. This critical comparison clarifies which mechanism dominates when, answering the call for prescriptive guidance rather than summary only.
Start–stop and thermal cycling accelerate degradation by mechanically stressing the membrane, electrodes, and interfaces. During start–stop events, potential excursions up to 1.4–1.5 V occur from hydrogen/air fronts on the anode, driving carbon corrosion and catalyst agglomeration. Without mitigation, these cycles can cause performance losses exceeding 200 mV per cycle. Thermal cycling exacerbates catalyst degradation, while operation below 100 °C introduces PA leaching from liquid water formation, further challenging durability. We recommend minimum start/stop AST elements: defined purge recipes, controlled cooldown rate, documented OCV dwell, and reporting of CO2 evolution and ECSA loss per 100 cycles.
7. Experimental studies and modeling approaches
Baudy et al. investigated the catalyst degradation mechanisms in MEAs aged within a HT-PEMFC stack operated at 160 °C for approximately 900 hours, incorporating deliberate start-up and shutdown cycling protocols.31 Through detailed physicochemical and electrochemical characterization, well-established degradation pathways were confirmed. At the cathode, PtNi NPs experienced growth, dissolution, migration, coalescence, and detachment, all following corrosion of the carbon support. This degradation was primarily driven by the constant load operation between start-up and shutdown phases, as well as by the distinct start/stop procedures examined. Specifically, loss of ECSA at the cathode, due to Pt and Ni dissolution via Ostwald ripening, occurred during both constant load operation and ON_C_Air/OFF_N2 cycling phases. This led to a marked decrease in ORR activity and consequently lower cell performance, particularly pronounced at the cell inlet. The dissolution of Ni from the PtNi alloy was more severe at the inlet, attributed to the region's higher oxidant and acidity levels, thereby diminishing the catalytic effectiveness of the alloy. Moreover, carbon corrosion was predominantly observed at the cathode outlet, especially under the ON_A_H2 startup condition and, to a lesser extent, via H2O2 formation during OFF_Air cycling, resulting in NP agglomeration between carbon grains. On the anode side, an increase in ECSA at the outlet was linked to the redeposition of Pt2+ species originating from cathode degradation, diffusing during OCV conditions or migrating during operation. Additionally, anode carbon functionalization was detected and attributed to chemical interactions with PA and its reduced species under low potential, as evidenced by Raman and XPS analyses. Although PA leaching was not directly characterized in this study, it was suggested to contribute significantly to the observed degradation phenomena. Operationally, the study identified that the ON_A_H2 startup procedure was the most damaging to cathode carbon support, while the ON_C_Air/OFF_N2 procedure promoted Pt2+ formation and subsequent interaction with PA. The inclusion of nitrogen purging (N2 flush) during these steps was found critical to mitigating, though not entirely preventing degradation, Fig. 7E. This work, together with Pinar, Oyarce, and Kannan, anchors our proposed minimum AST set and provides recent high-impact validation under realistic cycling.Complementary to these findings, Zhou et al. examined the degradation effects of fuel composition and dynamic operation on PBI-based HT-PEMFCs by comparing pure hydrogen with nitrogen-diluted hydrogen feeds under start–stop and load cycling conditions.13 Their results demonstrated that load cycling under pure hydrogen yielded relatively stable performance, with a degradation rate of approximately 88.2 μV h−1 at 0.4 A cm−2. In contrast, start–stop cycling significantly accelerated degradation, showing rates of 306 μV h−1 under pure hydrogen and an alarming 3.129 mV h−1 when 20 vol% nitrogen was present in the fuel. Notably, start–stop dynamics induced a pronounced increase in the cell's ohmic resistance, suggesting that internal resistive elements within the MEA grew rapidly due to combined physical and chemical alterations. The introduction of nitrogen adversely affected both kinetic and proton transfer resistances, thereby reducing the efficiency of electrochemical reactions and proton conductivity. These observations highlight nitrogen contamination as a critical factor in accelerated degradation. Interestingly, load cycling with pure hydrogen, involving current density modulation between 0.2 and 0.4 A cm−2, effectively reduced acid loss, indicating that controlled dynamic loading can mitigate degradation and enhance fuel cell durability. System designers should therefore consider N2 contamination budgets and install diagnostics (e.g., anode lambda tracking) to preempt N2-induced ohmic rise during start/stop.
Further insights were provided by Todorovski et al., who studied MEA degradation following 50 start–stop cycles using dry hydrogen and air over approximately 90 to 100 hours of operation.89 Their analysis revealed several degradation mechanisms consistent with earlier reports by Mlakar et al. The results showed that PA dehydration during start–stop cycling contributed to performance loss, while damage to the porous structure surrounding Pt particles led to narrowing or blocking of gas pathways, thereby impeding reactant transport. Moreover, product water management was adversely affected, and transient load changes induced mechanical stress on the membrane. The study also confirmed catalyst degradation through agglomeration, diffusion, and coarsening of Pt particles. An increase in ohmic resistance was attributed to the conversion of PA into species that poison the catalyst. The degradation rates measured initially were high, 3.6 mV per cycle for one MEA and 2.4 mV per cycle for another, but decreased markedly over time, suggesting the attainment of a semi-equilibrium state after approximately 40 cycles. This stabilization was proposed to result from redistribution of free acid within the MEA, which initially caused severe performance losses but later mitigated further degradation. The higher initial degradation rates, compared to those reported for fresh MEAs, were likely due to the older MEAs' reduced structural and chemical stability. These observations reinforce our earlier coupling narrative: electrolyte redistribution and interfacial mechanics feed back into transport losses, emphasizing the need for coupled models and targeted ASTs.
Taken together, these studies emphasize that dynamic operational stresses such as start–stop cycling and load variation significantly influence HT-PEMFC durability by accelerating catalyst dissolution, carbon corrosion, and PA-related degradation. The presence of nitrogen in the fuel stream exacerbates these effects by increasing resistive losses and impairing electrochemical reactions. Mitigation strategies including nitrogen purging, careful control of startup procedures, and optimized load cycling have been demonstrated as effective approaches to slow degradation and extend fuel cell lifetimes. We encourage authors to report mitigation costs (extra purge gas, efficiency impact) alongside durability benefits to guide system-level decisions.
In situ material characterization techniques play a crucial role in understanding the dynamic degradation phenomena occurring within HT-PEMFCs, as they enable real-time monitoring of the evolving physical and electrochemical states, Fig. 8B–E. EIS stands out as a powerful diagnostic tool that complements traditional current–voltage (i–V) curve measurements. EIS provides detailed insights into the sources and locations of resistance buildup during operation, allowing identification of the cell components most affected by specific operational stresses. For example, Søndergaard et al. utilized EIS to monitor HT-PEMFCs employing a thermally cross-linked PBI membrane compared to a reference linear PBI membrane without crosslinking.215 Operating at 160 °C and 0.2 A cm−2 for up to 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hours, the study minimized disturbance to steady-state operation by relying solely on in-line EIS diagnostics, demonstrating the effectiveness of this approach for long-term durability assessment. To accelerate learning across labs, we suggest an EIS reporting template (frequency window, perturbation amplitude, EC model elements) and periodic “health snapshots” during ASTs.
000 hours, the study minimized disturbance to steady-state operation by relying solely on in-line EIS diagnostics, demonstrating the effectiveness of this approach for long-term durability assessment. To accelerate learning across labs, we suggest an EIS reporting template (frequency window, perturbation amplitude, EC model elements) and periodic “health snapshots” during ASTs.
 | ||
| Fig. 8 (A) A flow chart of the approach Zhou et al. used for modeling the HT-PEMFC performance degradation.216In situ characterization techniques: (B) line scan of STM over Pt(111) facet in fuel cell.217 (C) In situ XRD test under various potentials for Pt/C catalyst, and (D) the corresponding function of NP size versus the potentials. (E) The corresponding Pt L-edge spectrum of PtCo under the varying potentials. (B–E) Reproduced from ref. 218. Copyright © 2016, American Chemical Society. | ||
In contrast to in situ methods, many characterization techniques focus on post-mortem analysis to elucidate the structural and chemical changes accumulated during fuel cell operation. SEM is commonly used to assess membrane thinning and morphological changes (Fig. 4C), while XRD and TEM provide detailed information on Pt NP growth and agglomeration. The μ-CT technique allows three-dimensional visualization of microstructural evolution, including porosity and tortuosity changes within GDLs. Complementary spectroscopic techniques such as Raman spectroscopy and XPS reveal chemical interactions between components, and titration methods enable quantitative determination of PA doping levels within the membrane. Combining in situ EIS with scheduled post-mortem (TEM/μ-CT/XPS) on matched cells provides the cross-validation needed to attribute observed voltage loss to specific mechanisms.
Oono et al. conducted a long-term comparative study of cells based on linear PBI and crosslinked AB-PBI membranes, both operated at 150 °C and 0.2 A cm−2 for periods extending to 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 800 hours.90 The linear PBI membrane experienced a significant reduction in thickness, with the membrane thickness at end-of-life being approximately half of the initial value after 13
800 hours.90 The linear PBI membrane experienced a significant reduction in thickness, with the membrane thickness at end-of-life being approximately half of the initial value after 13![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hours. In contrast, the crosslinked AB-PBI membrane exhibited only a modest thickness decrease after 17
000 hours. In contrast, the crosslinked AB-PBI membrane exhibited only a modest thickness decrease after 17![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 500 hours, indicating that chemical crosslinking effectively suppresses polymer degradation, Fig. 4B. Despite these promising results, direct in situ evidence correlating polymer degradation with long-term fuel cell operation remains limited. Most ex situ degradation studies rely on harsh chemical stressors, such as Fenton reagents, which do not fully replicate the actual operational environment, thereby questioning their relevance. To bridge this gap, we propose thermal-cycle EIS signatures (ohmic vs. charge-transfer arcs) as proxies for gradual polymer thinning vs. catalyst loss, later confirmed by post-mortem.
500 hours, indicating that chemical crosslinking effectively suppresses polymer degradation, Fig. 4B. Despite these promising results, direct in situ evidence correlating polymer degradation with long-term fuel cell operation remains limited. Most ex situ degradation studies rely on harsh chemical stressors, such as Fenton reagents, which do not fully replicate the actual operational environment, thereby questioning their relevance. To bridge this gap, we propose thermal-cycle EIS signatures (ohmic vs. charge-transfer arcs) as proxies for gradual polymer thinning vs. catalyst loss, later confirmed by post-mortem.
Mechanical degradation of the membrane induced by load cycling has also been documented. For instance, Schmies et al. measured thicknesses, revealing physical changes attributable to mechanical stress from dynamic loading, underscoring the importance of mechanical factors alongside chemical and electrochemical degradation processes.46 Together, these in situ and post-mortem characterization techniques provide comprehensive insight into the multifaceted degradation mechanisms in HT-PEMFCs, facilitating the development of strategies to improve durability and performance under realistic operating conditions. This explicitly addresses the coupling of chemistry, mechanics, and mass transport.
7.1 Modeling efforts to simulate start–stop behavior and predict lifespan
Zhou et al. conducted a comprehensive literature review to identify degradation trends and develop mathematical expressions describing key degradation mechanisms in HT-PEMFCs, Fig. 8A.216 Building upon this survey, they formulated a degradation model that integrates with an HT-PEMFC performance model by incorporating voltage loss contributions from major operating parameters and underlying mechanisms. The model successfully reflected established patterns in the literature regarding the degradation behaviors of HT-PEMFCs. We view this as groundwork for a digital twin that fuses measured inputs (flow, temperature, impedance) with a physics-informed degradation state estimator to predict remaining useful life in real time.Specifically, the degradation model accounted for two principal contributors to voltage loss: activation losses and ohmic losses. These were represented through time-dependent mathematical expressions derived from reported experimental data. The loss of ECSA, modeled as catalyst particle coalescence, was identified as the dominant factor influencing total voltage degradation. Meanwhile, membrane conductivity loss, attributed to acid evaporation, exhibited a significant relative increase over time; however, its absolute impact on overall voltage loss remained comparatively minor. This aligns with our mechanism-dominance map (activation at low-load/OCV vs. ohmic at sustained high-load), reinforcing controller priorities under different duty cycles.
Temperature was shown to exert a dual influence on cell performance and durability. While fuel cell performance initially improved linearly with increasing temperature, the rate of voltage decay escalated exponentially. Through this analysis, Zhou et al. identified an optimal operating temperature window between 160 °C and 170 °C.43 Within this range, the fuel cell benefits from enhanced performance relative to lower temperatures (<140 °C) without incurring the accelerated degradation and reduced lifetime observed at higher temperatures (>180 °C). We recommend codifying such windows as EMS constraints and validating with HIL platforms to quantify lifetime extensions.
In conclusion, the study highlighted that FC degradation is multifactorial, influenced by various interrelated parameters. Therefore, degradation models and lifetime prognostics that comprehensively capture these effects are critical for accurately assessing the feasibility and reliability of HT-PEMFC technology across different application scenarios. Adopting common AST protocols and shared KPI reporting will be essential to train and benchmark these models across laboratories.
8. Outlook and future research directions
Despite significant advancements in HT-PEMFC technology, several critical knowledge gaps remain that hinder widespread deployment, particularly under start–stop operational conditions. One major limitation is the lack of long-term field data that captures real-world degradation patterns, making it difficult to validate laboratory findings and model lifetime performance accurately. Additionally, the coupled degradation mechanisms, such as interactions between catalyst sintering, carbon support corrosion, and membrane thinning, are still not fully understood, especially under dynamic load cycles. To address these challenges, future research should prioritize the development of novel materials with enhanced resilience, such as doped carbon supports or alternative catalyst architectures. Moreover, integrating real-time diagnostic tools capable of monitoring key performance indicators (e.g., voltage decay, impedance shifts) can enable predictive maintenance strategies, reducing downtime and extending system life. These efforts will be instrumental in bridging the gap between laboratory innovation and field reliability. We specifically call for multi-lab round-robin start/stop ASTs with unified reporting, and for deployment of stack-level digital twins that couple electrochemistry–transport–mechanics to operational data for lifetime prediction.Building on these overarching research priorities, a critical area of focus lies in the development and optimization of advanced materials that can withstand the rigors of start–stop cycling. In particular, innovations in catalyst design and carbon support structures offer promising pathways to enhance both durability and performance. The following section explores recent progress and future prospects in material engineering, highlighting how tailored compositions and morphologies can mitigate degradation and extend operational lifetimes. We also highlight disruptive directions – PGM-free cathodes with improved HT stability, self-healing PA-tethered membranes, and corrosion-proof graphitized frameworks, as priority areas to address cost and durability simultaneously.
8.1 Materials – catalysts
The consensus in the literature supports that Pt alloys generally exhibit enhanced catalytic performance compared to pure Pt, primarily because the incorporation of additional metals can modify the electronic properties and surface structure of Pt. While bi-metallic catalysts have attracted significant attention and practical application from both academic and industrial sectors, recent research efforts have expanded to explore polymetallic electrocatalysts to further improve catalytic activity and address persistent challenges in HT-PEMFC technology. The combinatorial complexity of ternary and quaternary alloy catalysts, however, means that much of this compositional space remains largely unexplored. To ensure recent milestones (≤3 years) are visible, we relate alloy selections to PA-rich media at 160–180 °C and emphasize composition retention under start/stop as a key screening metric.Different metal combinations influence not only catalytic activity but also particle stability and reaction pathways. For example, distinct CO oxidation mechanisms have been identified on PtRu and PtMo catalysts at different potentials. When combined into a ternary alloy catalyst, these mechanisms can produce a co-catalytic effect, Fig. 9A. Electrochemical studies on ternary PtRuMo alloy catalysts demonstrated that they exhibited superior activity for CO and methanol oxidation compared to their binary counterparts. This enhancement was attributed to a shift in the reaction pathway favoring direct fuel oxidation, thereby minimizing CO intermediate formation, due to synergistic interactions among Pt, Ru, and Mo, particularly through strengthened Mo–Pt and Mo–Ru bonds. Further experimental studies have gone to quaternary alloyed catalysts, although it is debatable on whether these approaches are commercially scalable, Fig. 9D and E. Experimental and modeling studies on some of these “exotic” catalysts are still selected on their activity based on a volcano plot, e.g.Fig. 9C. We caution that scale-up and alloy integrity under start/stop (dealloying) remain the principal hurdles for ternaries/quaternaries despite activity gains.
 | ||
| Fig. 9 (A) Stability simulation snapshots of the ternary core@shell nanocatalyst Pd25Au25@Pt50 [Reprinted with permission from ref. 219 Copyright©2016, Elsevier Ltd]. (B) Performance of a low PGM-loaded ternary catalyst compared to C–Pt for ORR.220 (C) A “volcano-type” dependence of the ORR activity on the Pt d-band center, relative to the Fermi level [Reprinted with permission from ref. 221 Copyright © 2021, American Chemical Society]. (D) The HR-TEM images of a novel quaternary hexapod nano-skeletons with HIFs, offering high ORR activity [Reprinted with permission from ref. 222 Copyright © 2019 Elsevier B.V]. (E) The performance screening of the quaternary Pt34Ru30Ir13Ni23 catalyst [Reprinted with permission from ref. 223 Copyright © 2013 Elsevier Inc.]. | ||
Similarly, Park et al. investigated PtNiCu ternary catalysts synthesized by electrodeposition, revealing that alloy composition significantly affected the morphology and crystalline structure.224 Electrochemical testing under conditions containing phosphate anion poisoning showed that the PtNiCu catalyst achieved a 5.5-fold increase in specific ORR activity compared to pure Pt. Furthermore, the PtNiCu alloy demonstrated superior durability during potential cycling in PA environments, suggesting enhanced resistance to phosphate adsorption alongside improved catalytic performance.
Nan et al. explored core–shell catalysts consisting of PdM cores (M = Ni, Co, Fe) coated with an atomic-thin Pt shell.225 Among these, PdFe@Pt exhibited the highest ORR activity, delivering current densities of 0.88 mA cm−2 and 1.14 A mgPt−1 at 0.9 V vs. RHE, representing a 5.4-fold increase over commercial Pt catalysts. In addition, Vedyagin et al. examined ternary alloys composed solely of noble metals like Pt, Pd, and Rh, revealing that the relative metal ratios critically influenced both catalytic activity and thermal stability.226 Across most studies of alloy catalysts, alloying has consistently resulted in performance improvements relative to pure Pt. These studies constitute high-impact contributions; we recommend durability reporting include alloy composition drift and ECSA retention after start/stop ASTs to align with proposed standard KPIs.
Researchers continue to focus on designing high-efficiency electrocatalysts by exploring novel catalyst support materials to enhance both activity and durability in HT-PEMFCs. Among these, alternative supports such as tungsten carbides and oxides, SiC, and SiC-TiC composites have shown improved catalyst stability compared to conventional carbon supports. Incorporation of metal oxide supports has also been investigated. Ignaszak et al. studied TiC and core–shell TiC@TiO2 as catalyst supports, revealing that while TiC-supported Pt and Pt3Pd catalysts suffered rapid ECSA loss, 78% and 94% after just 500 cycles respectively, core–shell TiC@TiO2 significantly enhanced electrochemical stability.227 Similarly, Lobato et al. observed that SiC-based supports, when tested in MEAs at 160 °C, led to Pt NP agglomeration that improved fuel cell stability, albeit at the expense of reduced ECSA and overall system performance.228 Despite these benefits, metal oxides generally suffer from poor electrical conductivity and limited chemical stability under high-temperature and acidic HT-PEMFC conditions, restricting their practical utility as catalyst supports. We therefore prioritize highly graphitized, N-doped carbon frameworks as near-term candidates balancing corrosion resistance, PA retention, and conductivity.
Graphitic carbon nitride (g-C3N4) nanosheets and acid-treated CB have been explored as alternative supports, demonstrating marked improvements in catalyst durability. For example, TEM analyses showed the formation of small (∼1.8 nm) Pt NPs on g-C3N4 nanosheets, and AST revealed that Pt-g-C3N4-C retained 87.6% of its initial ECSA after 5000 cycles, significantly outperforming commercial Pt–C, which retained only 54%. Additionally, composite supports such as nitrogen-doped, electrochemically reduced exfoliated GO intercalated with CB (Pt-NrEGO/CB) have been developed to serve as three-dimensional physical barriers that inhibit Pt leaching and enhance gas transport. The N-doping further improves Pt NP anchoring and suppresses carbon corrosion, leading to improved performance and stability not only in LT-PEMFCs but also in HT-PEMFCs, even at reduced Pt loadings. These results are consistent with our coupling analysis: improved anchoring reduces Pt dissolution at high potentials, while graphitization slows reverse-current carbon corrosion during start/stop.
Graphene-based materials have attracted considerable attention due to their exceptional electrical conductivity, large surface area, thermal conductivity, mechanical strength, and chemical stability. However, their practical application remains limited by complex synthesis routes, high costs, and intrinsic drawbacks such as two-dimensional stacking, which can block Pt active sites and hinder gas diffusion, particularly at high current densities. Doping strategies, especially nitrogen functionalization of carbon supports, have proven effective in enhancing the stability and performance of HT-PEMFC cathodes. Nitrogen groups promote the formation of a PA network throughout the catalyst layer via acid–base interactions, improving acid distribution and mass transport. While functionalized nitrogen-doped catalysts typically exhibit increased ORR resistance due to enhanced reaction kinetics and oxygen diffusion near the catalyst sites, their higher acid uptake necessitates careful acid management within the membrane electrode assembly to optimize long-term operation. Pimperi et al. demonstrated that even modest nitrogen-containing functional groups improve MEA performance by facilitating more uniform PA dispersion, although the optimization of this approach for extended durability remains a challenge.229 This points to a materials–systems co-design space: tune N-site density for PA retention and set controller humidity/stoichiometry to avoid over-filling of PA in the CL during transients.
Based on the current understanding and extensive literature review, promising research directions for advancing HT-PEMFC catalysts include: (1) the development of highly robust catalyst supports such as more graphitized carbon materials capable of withstanding elevated temperatures; (2) improving catalyst–support interactions to stabilize Pt-based NPs and enhance catalytic activity; and (3) investigating novel catalyst formulations, including durable platinum-group-metal (PGM)-free catalysts, to achieve exceptional robustness and cost-effectiveness. We additionally suggest co-reporting techno-economic metrics (USD per kW, Pt-group g kW−1) with durability KPIs to connect lab innovations to deployment.
Over the years, various in-line characterization techniques have been developed to monitor fuel cell degradation in real time. Hartnig et al.69 conducted a study simulating 150 start–stop cycles by alternately passing hydrogen and air flows through the fuel electrode, during which they measured carbon corrosion products (CO2) at the air electrode. This study established a correlation between carbon corrosion and cathode performance degradation across different catalyst support materials, highlighting mass transport loss as a major consequence of start–stop cycling. Despite the value of such detailed analyses, there remains a demand for facile and easily implementable in-line diagnostic tools that offer high informational value with minimal operational disturbance. Among these, EIS stands out as a powerful and accessible technique capable of capturing the effects of operational events on cell performance, such as identifying where resistance accumulates and which cell components are most affected. We therefore recommend a practical “diagnostics core set” for HT-ASTs: i–V, EIS, outlet CO2, and periodic H2 crossover, with reporting templates to ease cross-study synthesis.
During testing, EIS measurements are typically performed with minimal disruption to steady-state operation, providing real-time insight into degradation mechanisms. The impedance spectra are commonly interpreted using equivalent circuit (EC) models, which enable deconvolution of various electrochemical processes contributing to cell degradation. The EC model applied in this context comprises several components: a parallel resistor (R1) and inductor (L1) representing high-frequency inductive effects; the ohmic resistance (R_ohm), which reflects changes in proton conductivity; a parallel constant phase element (CPE) and resistor (R_hf) associated primarily with charge transfer losses at the anode and cathode interfaces; and another CPE-resistor pair (R_lf) capturing low-frequency mass transport losses related to diffusion and migration of reactants and products within the cell. The characteristic semicircular shapes in the Nyquist plots generated from EIS data correspond to these processes, with the diameter of each semicircle providing quantitative information on charge transfer resistance and the peak positions reflecting reaction kinetics or diffusion limitations. Given its non-invasive, non-destructive nature and ease of implementation, EIS serves as a straightforward and effective in-line diagnostic tool to assess the health and lifetime of individual cells or stacks, thereby enabling continuous monitoring and predictive maintenance strategies for HT-PEMFC systems. We connect these elements to controller actions: rising R_lf → increase airflow or reduce current; rising R_ohm → check PA retention/humidity; rising R_hf → monitor catalyst degradation/OCV exposure.
The European Fuel Cell and Hydrogen 2 Joint Undertaking (FCH 2 JU) has set ambitious lifetime targets for fuel cell systems, recommending over 15 years (approximately 4![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 130
130![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hours) of operation for stationary micro combined heat and power (μ-CHP)14 applications.1 In contrast, lifetime goals for transportation vary, with passenger cars expected to achieve around 6000 hours of operation while maintaining less than 10% nominal power loss, and buses aiming for approximately 15
000 hours) of operation for stationary micro combined heat and power (μ-CHP)14 applications.1 In contrast, lifetime goals for transportation vary, with passenger cars expected to achieve around 6000 hours of operation while maintaining less than 10% nominal power loss, and buses aiming for approximately 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 hours. Across both stationary and automotive sectors, maintaining stable and reliable cell performance under dynamic operating conditions is essential.1 However, the practicalities of operating fuel cells continuously under normal conditions for thousands of hours render long-term durability testing both costly and time-consuming. To address this, AST protocols have been developed to rapidly evaluate critical materials and degradation mechanisms. These ASTs aim to simulate and accelerate real-world aging phenomena in a compressed timeframe, facilitating comparative analysis of durability and performance data. Aligning HT-ASTs with EU/DOE targets (e.g., % power loss thresholds, catalyst mass activity retention) would strengthen relevance and accelerate down-selection of materials and controls.
000 hours. Across both stationary and automotive sectors, maintaining stable and reliable cell performance under dynamic operating conditions is essential.1 However, the practicalities of operating fuel cells continuously under normal conditions for thousands of hours render long-term durability testing both costly and time-consuming. To address this, AST protocols have been developed to rapidly evaluate critical materials and degradation mechanisms. These ASTs aim to simulate and accelerate real-world aging phenomena in a compressed timeframe, facilitating comparative analysis of durability and performance data. Aligning HT-ASTs with EU/DOE targets (e.g., % power loss thresholds, catalyst mass activity retention) would strengthen relevance and accelerate down-selection of materials and controls.
For HT-PEMFCs, AST protocols typically fall into four categories: (1) start–stop cycling, (2) OCV exposure, (3) load cycling at either high or low current densities, and (4) thermal cycling. Start–stop tests simulate operational cycles involving cold start, steady-state operation, and shutdown, with the degradation in cell voltage per cycle serving as a key durability metric. Load cycling protocols are differentiated by current density, with low-load cycles (0–0.2 A cm−2) primarily focusing on catalyst and electrode stability, and high-load cycles (0.2–1.0 A cm−2) targeting membrane and electrolyte durability. Notably, low-load operation often involves prolonged exposure to OCV conditions, which inherently occur during fuel cell idling. Under these conditions, well-sealed cells tend to retain reformate gases, sustaining high OCV values that can accelerate degradation mechanisms such as carbon corrosion and Pt NP agglomeration. Thermal cycling further stresses fuel cell components through temperature fluctuations, where elevated temperatures promote carbon corrosion, metal dissolution, and acid evaporation, while temperatures below 100 °C can exacerbate PA leaching due to liquid water washout. We propose a concise “minimum HT-AST” set in Table 1 with explicit set-points (voltages, ΔT, ramp rates, stoichiometries) and required diagnostics to drive standardization.
In situ material characterization techniques are critical in these studies due to their ability to monitor degradation phenomena in real time, Fig. 8B–E. However, durability testing of HT-PEMFCs under realistic operating conditions remains both lengthy and costly, reinforcing the role of ASTs as essential tools for evaluating degradation pathways and ensuring reproducibility. These protocols are designed to selectively accelerate the degradation of targeted cell components while monitoring relevant physical parameters. For example, the Fuel Cell Commercialization Conference of Japan (FCCJ) has proposed separate AST protocols for assessing the relative contributions of carbon corrosion and catalyst degradation under start–stop and load-cycle conditions in fuel cell vehicles. Evaluations of catalyst durability frequently involve measuring changes in ECSA and particle size during potential cycling, with the comparison of theoretical ECSA loss, estimated from particle size changes, and experimentally observed ECSA loss providing insight into dominant degradation mechanisms. Publishing both theoretical and measured ECSA loss during HT-ASTs would directly answer the field's call for mechanism attribution.
Despite ongoing research, considerable variation exists among AST protocols, as summarized in Table 1, complicating direct comparisons of cell performance and durability across different studies. This lack of standardization underscores the critical need for unified AST protocols to enable consistent and comprehensive optimization of HT-PEMFC technology. Table 1 further outlines suggested testing protocols aimed at systematically studying various aspects of cell activity and lifetime to foster greater alignment within the research community. We believe these steps – standard KPIs, minimum HT-AST set, and diagnostics templates, will materially improve clarity, depth, and practical impact of future HT-durability studies.
9. Conclusion
This review has highlighted significant advancements in the field of high-temperature proton exchange membrane (HT-PEM) fuel cells, particularly focusing on their durability and performance under start–stop conditions. Key findings from the review include:Degradation mechanisms: the primary degradation mechanisms affecting HT-PEM fuel cells during start–stop cycles have been identified as catalyst layer degradation, membrane instability, and interfacial delamination. These mechanisms contribute to the overall performance decline and reduced lifespan of the fuel cells.
Mitigation strategies: various strategies have been explored to mitigate these degradation effects. These include the development of novel materials with enhanced stability, optimization of system design to reduce stress on components, and implementation of advanced operational control techniques to manage dynamic conditions effectively.
Experimental and modeling studies: recent experimental and modeling studies have provided deeper insights into the transient behavior and long-term reliability of HT-PEM fuel cells. These studies are crucial for understanding the complex interactions within the fuel cell and for developing more robust systems.
The importance of developing durable HT-PEM systems for practical applications cannot be overstated. As the demand for efficient and low-emission energy solutions grows, particularly in automotive and distributed power applications, the ability to maintain high performance under dynamic operating conditions becomes critical. By addressing the identified degradation mechanisms and implementing effective mitigation strategies, the development of HT-PEM fuel cells can be significantly advanced.
Future research should continue to focus on bridging the current gaps in knowledge and technology. This includes further material innovation, refined system designs, and improved operational protocols. By doing so, we can support the deployment of HT-PEM fuel cells in real-world applications, contributing to a sustainable and energy-efficient future.
Conflicts of interest
There are no conflicts to declare.Data availability
No new data were created or analyzed in this study. Data sharing is not applicable to this article. All data supporting this article are derived from previously published sources, which are cited throughout the manuscript. All figures have been properly cited, and copyright permissions have been obtained for any reproduced material.References
- N. Seselj, D. Aili, S. Celenk, L. N. Cleemann, H. A. Hjuler, J. O. Jensen, K. Azizi and Q. Li, Chem. Soc. Rev., 2023, 52, 4046–4070 RSC.
- Y. Wang, D. F. Ruiz Diaz, K. S. Chen, Z. Wang and X. C. Adroher, Mater. Today, 2020, 32, 178–203 CrossRef CAS.
- S. Rashidi, N. Karimi, B. Sunden, K. C. Kim, A. G. Olabi and O. Mahian, Prog. Energy Combust. Sci., 2022, 88, 100966 CrossRef.
- Q. Li, D. Aili, H. A. Hjuler and J. O. Jensen, High Temperature Polymer Electrolyte Membrane Fuel Cells: Approaches, Status, and Perspectives, Springer, 2016 Search PubMed.
- J. H. Bitter and A. Asadi Tashvigh, Ind. Eng. Chem. Res., 2022, 61, 6125–6134 CrossRef CAS.
- N. Li, X. Cui, J. Zhu, M. Zhou, V. Liso, G. Cinti, S. L. Sahlin and S. S. Araya, Renewable Sustainable Energy Rev., 2023, 182, 113395 CrossRef CAS.
- M. S. Herdem, M. Y. Sinaki, S. Farhad and F. Hamdullahpur, Int. J. Energy Res., 2019, 43, 5076–5105 CrossRef CAS.
- Q. Meyer, C. Yang, Y. Cheng and C. Zhao, Electrochem. Energy Rev., 2023, 6, 16 CrossRef CAS.
- M. Shahjahan Kabir Chowdury, Y. Park, S. Bum Park and Y. Park, J. Electroanal. Chem., 2024, 975, 118712 CrossRef CAS.
- A. Samir, M. S. Maowwad, M. A. Farahat and M. Talaat, Sci. Rep., 2025, 15, 5318 CrossRef CAS PubMed.
- K. Foniok, L. Drozdova, L. Prokop, F. Krupa, P. Kedron and V. Blazek, Energies, 2025, 18, 2117 CrossRef CAS.
- A. M. Dafalla, L. Wei, F. Hu, W. Cao, J. Guo and F. Jiang, Results Eng., 2025, 27, 107004 CrossRef CAS.
- M. Zhou, N. Li, D. Wang, S. S. Araya and V. Liso, Int. J. Hydrogen Energy, 2024, 65, 639–647 CrossRef CAS.
- P. Xie, F. Zhou, S. Tan, V. Liso and S. L. Sahlin, Appl. Energy, 2025, 381, 125118 CrossRef CAS.
- P. Xie, S. S. Araya, J. M. Guerrero and J. C. Vasquez, in IECON Proceedings (Industrial Electronics Conference), IEEE Computer Society, 2023 Search PubMed.
- W. Cui, Y. Lv, P. Sun, Z. Li, H. Pei and X. Yin, ACS Appl. Energy Mater., 2020, 3, 12115–12126 CrossRef CAS.
- P. Li, H. Guo, P. Sun, L. Zhang, X. Zhi and Z. Li, J. Membr. Sci., 2023, 666, 121159 CrossRef CAS.
- D. Aili, D. Henkensmeier, S. Martin, B. Singh, Y. Hu, J. O. Jensen, L. N. Cleemann and Q. Li, Electrochem. Energy Rev., 2020, 3, 793–845 CrossRef CAS.
- L. Vilčiauskas, M. E. Tuckerman, G. Bester, S. J. Paddison and K.-D. Kreuer, Nat. Chem., 2012, 4, 461–466 CrossRef.
- J. Ying, T. Liu, Y. Wang, M. Guo, Q. Shen, Y. Lin, J. Yu and Z. Yu, Energy Fuels, 2024, 38, 6613–6643 CrossRef CAS.
- H. Tang, K. Geng, D. Aili, Q. Ju, J. Pan, G. Chao, X. Yin, X. Guo, Q. Li and N. Li, Nat. Commun., 2022, 13, 7577 CrossRef CAS.
- N. Seselj, S. M. Alfaro, E. Bompolaki, L. N. Cleemann, T. Torres and K. Azizi, Adv. Mater., 2023, 35(40), 2302207 CrossRef CAS.
- J. Lee, M. Kim, B. Lee, J. Jang, S. Lee, D. J. You, J. Song and N. Jung, Nanomaterials, 2025, 15, 657 CrossRef CAS.
- J. S. Wainright, J.-T. Wang, D. Weng, R. F. Savinell and M. Litt, J. Electrochem. Soc., 1995, 142, L121 CrossRef CAS.
- S. Thomas, Operational strategies for longer durability of HT-PEM fuel cells operating on reformed methanol, Aalborg Universitetsforlag, 2017 Search PubMed.
- T. Zierdt, J. Müller-Hülstede, H. Schmies, D. Schonvogel, P. Wagner and K. A. Friedrich, ChemElectroChem, 2024, 11, e202300583 CrossRef CAS.
- Y. Jeon, H. Na, H. Hwang, J. Park, H. Hwang and Y. Shul, Int. J. Hydrogen Energy, 2015, 40, 3057–3067 CrossRef.
- S. Thomas, C. Jeppesen, T. Steenberg, S. S. Araya, J. R. Vang and S. K. Kær, Int. J. Hydrogen Energy, 2017, 42, 27230–27240 CrossRef.
- J. O. Leader, Y. Yue, M. R. Walluk and T. A. Trabold, Int. J. Hydrogen Energy, 2022, 47, 18820–18830 CrossRef.
- Z. Liu, H. Chen and T. Zhang, Appl. Energy, 2022, 327, 120058 CrossRef.
- A. Baudy, M. Durand, F. Roussel-Dherbey, G. Berthomé, N. Sergent, C. Turpin, J. Regnier, A. Jaafar, L. Dubau, M. Scohy and M. Chatenet, J. Electrochem. Soc., 2024, 171, 104513 CrossRef.
- J. Kim, M. Kim, B.-G. Lee and Y.-J. Sohn, Int. J. Hydrogen Energy, 2015, 40, 7769–7776 CrossRef.
- K. Hooshyari, B. Amini Horri, H. Abdoli, M. Fallah Vostakola, P. Kakavand and P. Salarizadeh, Energies, 2021, 14, 5440 CrossRef.
- N. Li, S. L. Sahlin, M. Zhou, V. Liso and S. S. Araya, Next Energy, 2024, 3, 100118 CrossRef.
- Y. Yue, Performance and Durability of High-Temperature Proton Exchange Membrane Fuel Cells Operated on Propane Reformate, Rochester Institute of Technology, 2019 Search PubMed.
- G. C. Bandlamudi, M. Saborni, P. Beckhaus, F. Mahlendorf and A. Heinzel, J. Fuel Cell Sci. Technol., 2010, 7, 014501 CrossRef.
- G. Cinti, V. Liso, S. L. Sahlin and S. S. Araya, Energies, 2020, 13, 4689 CrossRef CAS.
- H. M. A. Hunter, J. W. Makepeace, T. J. Wood, O. S. Mylius, M. G. Kibble, J. B. Nutter, M. O. Jones and W. I. F. David, J. Power Sources, 2016, 329, 138–147 CrossRef CAS.
- X. Zhou, Z. Ma, Y. Liu and A. Li, Int. J. Automot. Technol., 2023, 24, 1383–1393 CrossRef.
- Z. Yu, T. Ding, X. Huang, Y. Wang and G. Chen, in SAE 2024 Vehicle Powertrain Diversification Technology Forum, SAE International, 2025 Search PubMed.
- W. Shen, H. Su, J. Gao, L. Fan, G. Zhang and S. Zhou, Energies, 2025, 18, 2443 CrossRef CAS.
- X. Zhang, X. Ma, Z. Zhang, H. Du, Z. Wu, Z. Li and S. Shuai, Renew. Energy, 2025, 244, 122716 CrossRef CAS.
- M. Zhou, A. Ali, J. Zhu, S. S. Araya and V. Liso, J. Power Sources, 2024, 623, 235411 CrossRef CAS.
- S. Celenk, High Temperature Polymer Electrolyte Membrane Fuel Cells – Performance and Durability Studies, Technical University of Denmark, 2023 CAS.
- J.-R. Kim, J. S. Yi and T.-W. Song, J. Power Sources, 2012, 220, 54–64 CrossRef CAS.
- H. Schmies, D. Schonvogel, J. Büsselmann, P. Wagner and A. Dyck, ECS Meeting Abstracts, 2020, MA2020-02, p. 2161 Search PubMed.
- M. Stodel, M. Pons, T. Jarry, A. Jaafar, F. Lacressonniere, C. Turpin, R. Chattot, P. Lenormand and C. Tenailleau, Int. J. Hydrogen Energy, 2025, 138, 175–182 CrossRef CAS.
- Q. Li, R. He, J. O. Jensen and N. J. Bjerrum, Fuel Cells, 2004, 4, 147–159 CrossRef CAS.
- T. J. Schmidt and J. Baurmeister, J. Power Sources, 2008, 176, 428–434 CrossRef CAS.
- S. Yu, L. Xiao and B. C. Benicewicz, Fuel Cells, 2008, 8, 165–174 CrossRef CAS.
- P. Mocotéguy, B. Ludwig, J. Scholta, Y. Nedellec, D. J. Jones and J. Rozière, Fuel Cells, 2010, 10, 299–311 CrossRef.
- C. Hartnig and T. J. Schmidt, J. Power Sources, 2011, 196, 5564–5572 CrossRef CAS.
- J. Park, L. Wang, S. G. Advani and A. K. Prasad, Electrochim. Acta, 2014, 120, 30–38 CrossRef CAS.
- A. Kannan, A. Kabza and J. Scholta, J. Power Sources, 2015, 277, 312–316 CrossRef CAS.
- R. Kerr, H. R. García, M. Rastedt, P. Wagner, S. M. Alfaro, M. T. Romero, C. Terkelsen, T. Steenberg and H. A. Hjuler, Int. J. Hydrogen Energy, 2015, 40, 16860–16866 CrossRef CAS.
- R. Taccani, T. Chinese and M. Boaro, Int. J. Hydrogen Energy, 2017, 42, 1875–1883 CrossRef CAS.
- D. Schonvogel, M. Rastedt, P. Wagner, M. Wark and A. Dyck, Fuel Cells, 2016, 16, 480–489 CrossRef CAS.
- M. Rastedt, F. J. Pinar, N. Pilinski, A. Dyck and P. Wagner, ECS Trans., 2016, 75, 455–469 CrossRef CAS.
- A. Kannan, J. Kaczerowski, A. Kabza and J. Scholta, Fuel Cells, 2018, 18, 287–298 CrossRef CAS.
- X. Sun, Y. Li, F. Qi, F. Jing, H. Sun and G. Sun, ChemistrySelect, 2019, 4, 12313–12319 CrossRef CAS.
- S. Liu, M. Rasinski, Y. Rahim, S. Zhang, K. Wippermann, U. Reimer and W. Lehnert, J. Power Sources, 2019, 439, 227090 CrossRef CAS.
- J. Büsselmann, M. Rastedt, V. Tullius, K. Yezerska, A. Dyck and P. Wagner, Int. J. Hydrogen Energy, 2019, 4, 19384–19394 CrossRef.
- H. A. Hjuler, K. Azizi, N. Seselj, S. Martinez Alfaro, H. R. Garcia, D. Gromadskyi, L. Hromadska, S. Primdahl, J. O. Jensen, Q. Li, S. Celenk and L. Cleemann, ECS Trans., 2021, 104, 403–413 CrossRef CAS.
- T. Chinese, F. Ustolin, B. Marmiroli, H. Amenitsch and R. Taccani, Energies, 2021, 14, 6737 CrossRef CAS.
- J. Li, L. Yang, Z. Wang, H. Sun and G. Sun, Int. J. Hydrogen Energy, 2021, 46, 24353–24365 CrossRef CAS.
- K. H. Lim, A. S. Lee, V. Atanasov, J. Kerres, E. J. Park, S. Adhikari, S. Maurya, L. D. Manriquez, J. Jung, C. Fujimoto, I. Matanovic, J. Jankovic, Z. Hu, H. Jia and Y. S. Kim, Nat. Energy, 2022, 7, 248–259 CrossRef CAS.
- D. Schonvogel, J. Belack, J. Vidakovic, H. Schmies, L. M. Uhlig, H. Langnickel, P. K. Man Tung, Q. Meyer, C. Zhao and P. Wagner, J. Power Sources, 2024, 591, 233835 CrossRef CAS.
- C. A. Reiser, L. Bregoli, T. W. Patterson, J. S. Yi, J. D. Yang, M. L. Perry and T. D. Jarvi, Electrochem. Solid-State Lett., 2005, 8, A273 CrossRef CAS.
- C. Hartnig and T. J. Schmidt, J. Power Sources, 2011, 196, 5564–5572 CrossRef CAS.
- T. Engl, L. Gubler and T. J. Schmidt, Energy Technol., 2016, 4, 65–74 CrossRef CAS.
- J. Park, L. Wang, S. G. Advani and A. K. Prasad, Electrochim. Acta, 2014, 120, 30–38 CrossRef CAS.
- F. Javier Pinar, M. Rastedt, N. Pilinski and P. Wagner, Int. J. Hydrogen Energy, 2016, 41, 19463–19474 CrossRef CAS.
- J. Li, L. Yang, Z. Wang, H. Sun and G. Sun, Int. J. Hydrogen Energy, 2021, 46, 24353–24365 CrossRef CAS.
- A. Oyarce, E. Zakrisson, M. Ivity, C. Lagergren, A. B. Ofstad, A. Bodén and G. Lindbergh, J. Power Sources, 2014, 254, 232–240 CrossRef CAS.
- A. Kannan, A. Kabza and J. Scholta, J. Power Sources, 2015, 277, 312–316 CrossRef CAS.
- N. Bevilacqua, M. G. George, S. Galbiati, A. Bazylak and R. Zeis, Electrochim. Acta, 2017, 257, 89–98 CrossRef CAS.
- S. Liu, M. Rasinski, Y. Rahim, S. Zhang, K. Wippermann, U. Reimer and W. Lehnert, J. Power Sources, 2019, 439, 227090 CrossRef CAS.
- P. Mardle, I. Cerri, T. Suzuki and A. El-kharouf, Catalysts, 2021, 11, 965 CrossRef CAS.
- S. Yu, L. Xiao and B. C. Benicewicz, Fuel Cells, 2008, 8, 165–174 CrossRef CAS.
- Z. Qi and S. Buelte, J. Power Sources, 2006, 161, 1126–1132 CrossRef CAS.
- F. J. Pinar, N. Pilinski and P. Wagner, AIChE J., 2016, 62, 217–227 CrossRef CAS.
- D. Schonvogel, M. Rastedt, P. Wagner, M. Wark and A. Dyck, Fuel Cells, 2016, 16, 480–489 CrossRef CAS.
- J. Büsselmann, M. Rastedt, V. Tullius, K. Yezerska, A. Dyck and P. Wagner, Int. J. Hydrogen Energy, 2019, 44, 19384–19394 CrossRef.
- R. Taccani, T. Chinese and M. Boaro, Int. J. Hydrogen Energy, 2017, 42, 1875–1883 CrossRef CAS.
- T. Chinese, F. Ustolin, B. Marmiroli, H. Amenitsch and R. Taccani, Energies, 2021, 14, 6737 CrossRef CAS.
- S. Thomas, C. Jeppesen, T. Steenberg, S. S. Araya, J. R. Vang and S. K. Kær, Int. J. Hydrogen Energy, 2017, 42, 27230–27240 CrossRef CAS.
- D. Schonvogel, M. Rastedt, P. Wagner, M. Wark and A. Dyck, Fuel Cells, 2016, 16, 480–489 CrossRef CAS.
- T. Søndergaard, L. N. Cleemann, H. Becker, T. Steenberg, H. A. Hjuler, L. Seerup, Q. Li and J. O. Jensen, J. Electrochem. Soc., 2018, 165, F3053 CrossRef.
- F. Todorovski, M. Sekavcnik, M. Mori and A. Lotric, in 9th International Youth Conference on Energy, IYCE 2024, Institute of Electrical and Electronics Engineers Inc., 2024 Search PubMed.
- Y. Oono, A. Sounai and M. Hori, J. Power Sources, 2012, 210, 366–373 CrossRef CAS.
- D. Aili, J. Yang, K. Jankova, D. Henkensmeier and Q. Li, J. Mater. Chem. A, 2020, 8, 12854–12886 RSC.
- Q. Li, A. David, H. A. Hjuler and J. O. Jensen, High Temperature Polymer Electrolyte Membrane Fuel Cells: Approaches, Status, and Perspectives, Springer International Publishing, Switzerland, 2016 Search PubMed.
- L. Vilčiauskas, M. E. Tuckerman, J. P. Melchior, G. Bester and K.-D. Kreuer, Solid State Ionics, 2013, 252, 34–39 CrossRef.
- J.-P. Melchior, G. Majer and K.-D. Kreuer, Phys. Chem. Chem. Phys., 2017, 19, 601–612 RSC.
- Z. Guo, M. Perez-Page, J. Chen, Z. Ji and S. M. Holmes, J. Energy Chem., 2021, 63, 393–429 CrossRef CAS.
- F. N. Büchi, M. Inaba and T. J. Schmidt, Polymer Electrolyte Fuel Cell Durability, Springer, 2009 Search PubMed.
- C. Wannek, B. Kohnen, H.-F. Oetjen, H. Lippert and J. Mergel, Fuel Cells, 2008, 8, 87–95 CrossRef CAS.
- C. Wannek, B. Kohnen, H. F. Oetjen, H. Lippert and J. Mergel, Fuel Cells, 2008, 8, 87–95 CrossRef CAS.
- F. J. Pinar, P. Cañizares, M. A. Rodrigo, D. Úbeda and J. Lobato, J. Power Sources, 2011, 196, 4306–4313 CrossRef CAS.
- H. S. Oh, Y. Cho, W. H. Lee and H. Kim, J. Mater. Chem. A, 2013, 1, 2578–2581 RSC.
- M. A. Molleo, X. Chen, H. J. Ploehn, K. J. Fishel and B. C. Benicewicz, Fuel Cells, 2014, 14, 16–25 CrossRef.
- T. Søndergaard, L. N. Cleemann, H. Becker, T. Steenberg, H. A. Hjuler, L. Seerup, Q. Li and J. O. Jensen, J. Electrochem. Soc., 2018, 165, F3053–F3062 CrossRef.
- A. Kannan, Q. Li, L. N. Cleemann and J. O. Jensen, Fuel Cells, 2018, 18, 103–112 CrossRef.
- A. Kannan, D. Aili, L. N. Cleemann, Q. Li and J. O. Jensen, Int. J. Hydrogen Energy, 2020, 45, 1008–1017 CrossRef.
- F. Zhou, S. J. Andreasen, S. K. Kær and D. Yu, Int. J. Hydrogen Energy, 2015, 40, 2833–2839 CrossRef.
- N. Yousfi-Steiner, P. Moçotéguy, D. Candusso and D. Hissel, J. Power Sources, 2009, 194, 130–145 CrossRef.
- M. A. Danzer, S. J. Wittmann and E. P. Hofer, J. Power Sources, 2009, 190, 86–91 CrossRef.
- A. Dalvi and M. Guay, Control Eng. Pract., 2009, 17, 924–938 CrossRef.
- W. R. W. Daud, R. E. Rosli, E. H. Majlan, S. A. A. Hamid, R. Mohamed and T. Husaini, Renew. Energy, 2017, 113, 620–638 CrossRef.
- F. Ruiz-Zepeda, M. Gatalo, A. Pavlišič, G. Dražić, P. Jovanovič, M. Bele, M. Gaberšček and N. Hodnik, Nano Lett., 2019, 19, 4919–4927 CrossRef PubMed.
- S. Cherevko, N. Kulyk and K. J. J. Mayrhofer, Nano Energy, 2016, 29, 275–298 CrossRef.
- A. A. Topalov, S. Cherevko, A. R. Zeradjanin, J. C. Meier, I. Katsounaros and K. J. J. Mayrhofer, Chem. Sci., 2014, 5, 631–638 RSC.
- P. P. Lopes, D. Tripkovic, P. F. B. D. Martins, D. Strmcnik, E. A. Ticianelli, V. R. Stamenkovic and N. M. Markovic, J. Electroanal. Chem., 2018, 819, 123–129 CrossRef.
- Z. Kong, J. Wu, Z. Liu, D. Yan, Z.-P. Wu and C.-J. Zhong, Exploration, 2025, 5, 20230052 CrossRef CAS PubMed.
- S. Liu, M. Rasinski, Y. Lin, K. Wippermann, A. Everwand and W. Lehnert, Electrochim. Acta, 2018, 289, 354–362 CrossRef CAS.
- M. Prokop, T. Bystron, P. Belsky, O. Tucek, R. Kodym, M. Paidar and K. Bouzek, Electrochim. Acta, 2020, 363, 137165 CrossRef CAS.
- M. Prokop, R. Kodym, T. Bystron, M. Drakselova, M. Paidar and K. Bouzek, Electrochim. Acta, 2020, 333, 135509 CrossRef CAS.
- M. Prokop, R. Kodym, T. Bystron, M. Paidar and K. Bouzek, Electrochim. Acta, 2019, 313, 352–366 CrossRef CAS.
- J. Aragane, H. Urushibata and T. Murahashi, J. Appl. Electrochem., 1996, 26, 147–152 CrossRef CAS.
- F. Maillard, W. O. Silva, L. Castanheira, L. Dubau and F. H. B. Lima, ChemPhysChem, 2019, 20, 3106–3111 CrossRef CAS PubMed.
- D. A. Stevens and J. R. Dahn, Carbon, 2005, 43, 179–188 CrossRef CAS.
- C. A. Reiser, L. Bregoli, T. W. Patterson, J. S. Yi, J. D. Yang, M. L. Perry and T. D. Jarvi, Electrochem. Solid-State Lett., 2005, 8, A273 CrossRef CAS.
- J. C. Meier, C. Galeano, I. Katsounaros, A. A. Topalov, A. Kostka, F. Schüth and K. J. J. Mayrhofer, ACS Catal., 2012, 2, 832–843 CrossRef.
- V. Bandlamudi, P. Bujlo, C. Sita and S. Pasupathi, Mater. Today: Proc., 2018, 5, 10602–10610 Search PubMed.
- R. L. Borup, J. R. Davey, F. H. Garzon, D. L. Wood and M. A. Inbody, J. Power Sources, 2006, 163, 76–81 CrossRef.
- T. Søndergaard, L. N. Cleemann, L. Zhong, H. Becker, T. Steenberg, H. A. Hjuler, L. Seerup, Q. Li and J. O. Jensen, Electrocatalysis, 2018, 9, 302–313 CrossRef.
- M. Pawlyta, S. Smykała, B. Liszka, A. Blacha-Grzechnik, M. Krzywiecki, K. Jurkiewicz and A. Jakóbik-Kolon, Appl. Surf. Sci., 2023, 611, 155637 CrossRef.
- F. Bai, H.-B. Quan, R.-J. Yin, Z. Zhang, S.-Q. Jin, P. He, Y.-T. Mu, X.-M. Gong and W.-Q. Tao, Appl. Energy, 2022, 324, 119763 CrossRef.
- M. Endo, Y. A. Kim, T. Hayashi, T. Yanagisawa, H. Muramatsu, M. Ezaka, H. Terrones, M. Terrones and M. S. Dresselhaus, Carbon, 2003, 41, 1941–1947 CrossRef.
- S. V. Selvaganesh, P. Sridhar, S. Pitchumani and A. K. Shukla, J. Electrochem. Soc., 2012, 160, F49 CrossRef.
- M. Pawlyta, S. Smykała, B. Liszka, A. Blacha-Grzechnik, M. Krzywiecki, K. Jurkiewicz and A. Jakóbik-Kolon, Appl. Surf. Sci., 2023, 611, 155637 CrossRef.
- M. Prokop, M. Vesely, P. Capek, M. Paidar and K. Bouzek, Electrochim. Acta, 2022, 413, 140133 CrossRef.
- A. Zitolo, V. Goellner, V. Armel, M.-T. Sougrati, T. Mineva, L. Stievano, E. Fonda and F. Jaouen, Nat. Mater., 2015, 14, 937–942 CrossRef.
- G. Bae, S. Han, H.-S. Oh and C. H. Choi, Angew. Chem., Int. Ed., 2023, 62, e202219227 CrossRef PubMed.
- D. Menga, Y.-S. Li, A. M. Damjanović, O. Proux, F. E. Wagner, T.-P. Fellinger, H. A. Gasteiger and M. Piana, ChemElectroChem, 2024, 11, e202400228 CrossRef CAS.
- J. Müller-Hülstede, T. Zierdt, H. Schmies, D. Schonvogel, Q. Meyer, C. Zhao, P. Wagner and M. Wark, J. Power Sources, 2022, 537, 231529 CrossRef.
- S. V. Selvaganesh, P. Sridhar, S. Pitchumani and A. K. Shukla, J. Electrochem. Soc., 2012, 160, F49 CrossRef.
- W. Zhang, Z. Cao, J. Zhang, K. Peng, Q. Ma, Q. Xu and H. Su, ACS Sustain. Chem. Eng., 2020, 8, 9195–9205 CrossRef CAS.
- M. Perchthaler, T. Ossiander, V. Juhart, J. Mitzel, C. Heinzl, C. Scheu and V. Hacker, J. Power Sources, 2013, 243, 472–480 CrossRef CAS.
- Y. Shao, G. Yin, Z. Wang and Y. Gao, J. Power Sources, 2007, 167, 235–242 CrossRef CAS.
- N. Shroti and M. K. Daletou, Int. J. Hydrogen Energy, 2022, 47, 16235–16248 CrossRef CAS.
- H. Tang, K. Geng, D. Aili, Q. Ju, J. Pan, G. Chao, X. Yin, X. Guo, Q. Li and N. Li, Nat. Commun., 2022, 13, 7577 CrossRef CAS PubMed.
- Y. Oono, A. Sounai and M. Hori, J. Power Sources, 2013, 241, 87–93 CrossRef CAS.
- M. Prokop, R. Kodym, T. Bystron, M. Drakselova, M. Paidar and K. Bouzek, Electrochim. Acta, 2020, 333, 135509 CrossRef CAS.
- Y. Pan, H. Wang and N. P. Brandon, J. Power Sources, 2021, 513, 230560 CrossRef CAS.
- T. J. Schmidt, ECS Trans., 2006, 1, 19 CrossRef CAS.
- J.-P. Melchior, K.-D. Kreuer and J. Maier, Phys. Chem. Chem. Phys., 2017, 19, 587–600 RSC.
- C. B. Spainhour, in Encyclopedia of Toxicology, ed. P. Wexler, Academic Press, Oxford, 3rd edn, 2014, pp. 916–919 Search PubMed.
- A. S. Lee, Y.-K. Choe, I. Matanovic and Y. S. Kim, J. Mater. Chem. A, 2019, 7, 9867–9876 RSC.
- T. Søndergaard, L. N. Cleemann, H. Becker, T. Steenberg, H. A. Hjuler, L. Seerup, Q. Li and J. O. Jensen, J. Electrochem. Soc., 2018, 165, F3053 CrossRef.
- D. Aili, H. Becker, U. Reimer, J. W. Andreasen, L. N. Cleemann, J. O. Jensen, C. Pan, X. Wang, W. Lehnert and Q. Li, J. Electrochem. Soc., 2020, 167, 134507 CrossRef CAS.
- T. J. Schmidt and J. Baurmeister, J. Power Sources, 2008, 176, 428–434 CrossRef CAS.
- Q. Li, R. He, J. O. Jensen and N. J. Bjerrum, Fuel Cells, 2004, 4, 147–159 CrossRef CAS.
- M. Prokop, M. Vesely, P. Capek, M. Paidar and K. Bouzek, Electrochim. Acta, 2022, 413, 140133 CrossRef.
- F. Zhou, S. J. Andreasen, S. K. Kær and D. Yu, Int. J. Hydrogen Energy, 2015, 40, 2833–2839 CrossRef.
- Y. Oono, T. Fukuda, A. Sounai and M. Hori, J. Power Sources, 2010, 195, 1007–1014 CrossRef.
- D. Aili, A. Vassiliev, J. O. Jensen, T. J. Schmidt and Q. Li, J. Power Sources, 2015, 279, 517–521 CrossRef.
- A. Vassiliev, A. K. Reumert, J. O. Jensen and D. Aili, J. Power Sources, 2019, 432, 30–37 CrossRef.
- J. Neutzler, G. Qian, K. Huang and B. Benicewicz, J. Power Sources, 2012, 216, 471–474 CrossRef.
- S. Thomas, J. R. Vang, S. S. Araya and S. K. Kær, Appl. Energy, 2017, 192, 422–436 CrossRef.
- K.-S. Lee, J. S. Spendelow, Y.-K. Choe, C. Fujimoto and Y. S. Kim, Nat. Energy, 2016, 1, 16120 CrossRef.
- Y. Oono, A. Sounai and M. Hori, J. Power Sources, 2012, 210, 366–373 CrossRef.
- V. Atanasov, A. S. Lee, E. J. Park, S. Maurya, E. D. Baca, C. Fujimoto, M. Hibbs, I. Matanovic, J. Kerres and Y. S. Kim, Nat. Mater., 2021, 20, 370–377 CrossRef PubMed.
- P. Sun, Z. Xia, Z. Li, Z. Fan, F. He, Q. Liu and X. Yin, Mater. Today Chem., 2022, 23, 100653 CrossRef.
- H. Ding, P. Sun, Y. Wang, Z. Xia, P. Li, C. Cui, M. Li and Z. Li, J. Membr. Sci., 2023, 685, 121911 CrossRef.
- Z. Xu, N. Chen, S. Huang, S. Wang, D. Han, M. Xiao and Y. Meng, Molecules, 2024, 29, 4480 CrossRef PubMed.
- Z. Wu, C. Shen, S. Gao, X. Zhu, M. Zhang and A. Ding, Macromol. Res., 2021, 29, 199–210 CrossRef.
- L. Bu, S. Guo, X. Zhang, X. Shen, D. Su, G. Lu, X. Zhu, J. Yao, J. Guo and X. Huang, Nat. Commun., 2016, 7, 11850 CrossRef CAS.
- X. X. Wang, Z. H. Tan, M. Zeng and J. N. Wang, Sci. Rep., 2014, 4, 4437 CrossRef PubMed.
- G. Huang, Y. Li, S. Du, Y. Wu, R. Chen, J. Zhang, Y. Cheng, S. Lu, L. Tao and S. Wang, Sci. China Chem., 2021, 64, 2203–2211 CrossRef CAS.
- N. Li, X. Cui, J. Zhu, M. Zhou, V. Liso, G. Cinti, S. L. Sahlin and S. S. Araya, Renewable Sustainable Energy Rev., 2023, 182, 113395 CrossRef CAS.
- S. Li, C. Peng, Q. Shen, Y. Cheng, C. Wang and G. Yang, Membranes, 2021, 11, 817 CrossRef CAS PubMed.
- L. Huang, M. Wei, R. Qi, C.-L. Dong, D. Dang, C.-C. Yang, C. Xia, C. Chen, S. Zaman, F.-M. Li, B. You and B. Y. Xia, Nat. Commun., 2022, 13, 6703 CrossRef CAS PubMed.
- R. Zeis, Beilstein J. Nanotechnol., 2015, 6, 68–83 CrossRef.
- P. Stonehart, Ber. Bunsenges. Phys. Chem. oder Ber. Phys. Chem., 1990, 94, 913–921 CrossRef CAS.
- I. E. L. Stephens, A. S. Bondarenko, U. Grønbjerg, J. Rossmeisl and I. Chorkendorff, Energy Environ. Sci., 2012, 5, 6744–6762 RSC.
- M. Mamlouk and K. Scott, J. Power Sources, 2011, 196, 1084–1089 CrossRef CAS.
- J.-E. Lim, U. J. Lee, S. H. Ahn, E. Cho, H.-J. Kim, J. H. Jang, H. Son and S.-K. Kim, Appl. Catal., B, 2015, 165, 495–502 CrossRef CAS.
- K.-S. Lee, S. J. Yoo, D. Ahn, S.-K. Kim, S. J. Hwang, Y.-E. Sung, H.-J. Kim, E. Cho, D. Henkensmeier, T.-H. Lim and J. H. Jang, Electrochim. Acta, 2011, 56, 8802–8810 CrossRef CAS.
- Q. Zhang, R. Yue, F. Jiang, H. Wang, C. Zhai, P. Yang and Y. Du, Gold Bull., 2013, 46, 175–184 CrossRef.
- Z. Jusys, T. J. Schmidt, L. Dubau, K. Lasch, L. Jörissen, J. Garche and R. J. Behm, J. Power Sources, 2002, 105, 297–304 CrossRef.
- H. Liu, C. Li, D. Chen, P. Cui, F. Ye and J. Yang, Sci. Rep., 2017, 7, 11421 CrossRef PubMed.
- H. Yano, M. Kataoka, H. Yamashita, H. Uchida and M. Watanabe, Langmuir, 2007, 23, 6438–6445 CrossRef PubMed.
- P. He, T. Cheng, R. Bashyam and S. Knights, ECS Meeting Abstracts, 2010, MA2010-01, p. 566 Search PubMed.
- X. Deng, S. Yin, X. Wu, M. Sun, Z. Li, Z. Xie, Y. Liang and Q. Huang, Int. J. Hydrogen Energy, 2019, 44, 2752–2759 CrossRef.
- M. G. H. Al-Tememy and Y. Devrim, Int. J. Energy Res., 2021, 45, 3343–3357 CrossRef.
- P. Strasser, Q. Fan, M. Devenney, W. H. Weinberg, P. Liu and J. K. Nørskov, J. Phys. Chem. B, 2003, 107, 11013–11021 CrossRef.
- W. Tu, W. Luo, C. Chen, K. Chen, E. Zhu, Z. Zhao, Z. Wang, T. Hu, H. Zai, X. Ke, M. Sui, P. Chen, Q. Zhang, Q. Chen, Y. Li and Y. Huang, Adv. Funct. Mater., 2020, 30, 1908230 CrossRef.
- N. Seselj, S. M. Alfaro, E. Bompolaki, L. N. Cleemann, T. Torres and K. Azizi, Adv. Mater., 2023, 35(40), 2302207 CrossRef.
- O. V Tripachev, A. D. Modestov, O. V Korchagin, V. A. Bogdanovskaya, V. A. Vasilenko and M. V Radina, Russ. J. Appl. Chem., 2020, 93, 1743–1749 CrossRef.
- C. V. S. Almeida, N. A. Galiote, K. I. B. Eguiluz, G. R. Salazar-Banda, V. Del Colle and G. Tremiliosi-Filho, Electrochim. Acta, 2020, 351, 136223 CrossRef.
- A. A. Vedyagin, Y. V Shubin, R. M. Kenzhin, P. E. Plyusnin and V. O. Stoyanovskii, Processes, 2020, 8, 928 CrossRef CAS.
- M. A. Abdelkareem, T. Wilberforce, K. Elsaid, E. T. Sayed, E. A. M. Abdelghani and A. G. Olabi, Int. J. Hydrogen Energy, 2021, 46, 23529–23547 CrossRef.
- S. A. Grigoriev, V. N. Fateev, A. S. Pushkarev, I. V. Pushkareva, N. A. Ivanova, V. N. Kalinichenko, M. Yu. Presnyakov and X. Wei, Materials, 2018, 11, 1405 CrossRef.
- Y. Devrim, E. D. Arıca and A. Albostan, Int. J. Hydrogen Energy, 2018, 43, 11820–11829 CrossRef.
- S. S. Araya, F. Zhou, V. Liso, S. L. Sahlin, J. R. Vang, S. Thomas, X. Gao, C. Jeppesen and S. K. Kær, Int. J. Hydrogen Energy, 2016, 41, 21310–21344 CrossRef.
- E. Antolini, R. R. Passos and E. A. Ticianelli, J. Power Sources, 2002, 109, 477–482 CrossRef.
- J. Wang, W. Zhu, X. He and S. Yang, Catal. Commun., 2008, 9, 2163–2167 CrossRef.
- M. Endo, Y. A. Kim, T. Hayashi, T. Yanagisawa, H. Muramatsu, M. Ezaka, H. Terrones, M. Terrones and M. S. Dresselhaus, Carbon, 2003, 41, 1941–1947 CrossRef.
- Z. Liu, L. M. Gan, L. Hong, W. Chen and J. Y. Lee, J. Power Sources, 2005, 139, 73–78 CrossRef.
- D. R. Kauffman and A. Star, Analyst, 2010, 135, 2790 RSC.
- C. Kim, Y. J. Kim, Y. A. Kim, T. Yanagisawa, K. C. Park, M. Endo and M. S. Dresselhaus, J. Appl. Phys., 2004, 96, 5903–5905 CrossRef.
- H. Chu, Y. Shen, L. Lin, X. Qin, G. Feng, Z. Lin, J. Wang, H. Liu and Y. Li, Adv. Funct. Mater., 2010, 20, 3747–3752 CrossRef.
- Y. Devrim and E. D. Arıca, Int. J. Hydrogen Energy, 2020, 45, 3609–3617 CrossRef.
- A. Orfanidi, P. Madkikar, H. A. El-Sayed, G. S. Harzer, T. Kratky and H. A. Gasteiger, J. Electrochem. Soc., 2017, 164, F418–F426 CrossRef.
- D. A. C. Brownson and C. E. Banks, Analyst, 2010, 135, 2768 RSC.
- A. Marinkas, F. Arena, J. Mitzel, G. M. Prinz, A. Heinzel, V. Peinecke and H. Natter, Carbon, 2013, 58, 139–150 CrossRef CAS.
- H. Wang, T. Maiyalagan and X. Wang, ACS Catal., 2012, 2, 781–794 CrossRef CAS.
- Y. Huang, F. Bao, M. Ji, Y. Hu, L. Huang, H. Liu, J. Yu, G. Cong, C. Zhu and J. Xu, Chem. Commun., 2021, 57, 13792–13795 RSC.
- F. C. Lee, M. S. Ismail, D. B. Ingham, K. J. Hughes, L. Ma, S. M. Lyth and M. Pourkashanian, Renewable Sustainable Energy Rev., 2022, 166, 112640 CrossRef CAS.
- R. Alink, J. Haußmann, H. Markötter, M. Schwager, I. Manke and D. Gerteisen, J. Power Sources, 2013, 233, 358–368 CrossRef CAS.
- M. Arif, S. C. P. Cheung and J. Andrews, Energy Fuels, 2020, 34, 13010–13022 CrossRef CAS.
- P. Ren, P. Pei, Y. Li, Z. Wu, D. Chen and S. Huang, Prog. Energy Combust. Sci., 2020, 80, 100859 CrossRef.
- S. Thomas, C. Jeppesen, T. Steenberg, S. S. Araya, J. R. Vang and S. K. Kær, Int. J. Hydrogen Energy, 2017, 42, 27230–27240 CrossRef CAS.
- T. Søndergaard, L. N. Cleemann, H. Becker, D. Aili, T. Steenberg, H. A. Hjuler, L. Seerup, Q. Li and J. O. Jensen, J. Power Sources, 2017, 342, 570–578 CrossRef.
- M. Zhou, S. Frensch, V. Liso, N. Li, S. L. Sahlin, G. Cinti and S. Simon Araya, Energies, 2022, 15, 5651 CrossRef CAS.
- R. Rizo, E. Herrero and J. M. Feliu, Phys. Chem. Chem. Phys., 2013, 15, 15416–15425 RSC.
- K. Sasaki, N. Marinkovic, H. S. Isaacs and R. R. Adzic, ACS Catal., 2016, 6, 69–76 CrossRef CAS.
- P. Brault, C. Coutanceau, P. C. Jennings, T. Vegge, J. Berndt, A. Caillard, S. Baranton and S. Lankiang, Int. J. Hydrogen Energy, 2016, 41, 22589–22597 CrossRef.
- Z. Wang, X. Yao, Y. Kang, D. Xia and L. Gan, Catalysts, 2019, 9, 569 CrossRef.
- F. Ando, T. Gunji, T. Tanabe, I. Fukano, H. D. Abruña, J. Wu, T. Ohsaka and F. Matsumoto, ACS Catal., 2021, 11, 9317–9332 CrossRef CAS.
- L. Huang, M. Wei, N. Hu, P. Tsiakaras and P. Kang Shen, Appl. Catal., B, 2019, 258, 117974 CrossRef.
- J. H. Kim, S. Y. Kwon, D. Bhattacharjya, G. S. Chai and J.-S. Yu, J. Catal., 2013, 306, 133–145 CrossRef.
- H. Park, D.-K. Kim, H. Kim, S. Oh, W. S. Jung and S.-K. Kim, Appl. Surf. Sci., 2020, 510, 145444 CrossRef.
- H. Nan, Y. Q. Su, C. Tang, R. Cao, D. Li, J. Yu, Q. Liu, Y. Deng and X. Tian, Sci. Bull., 2020, 65, 1396 CrossRef PubMed.
- A. A. Vedyagin, Y. V Shubin, R. M. Kenzhin, P. E. Plyusnin and V. O. Stoyanovskii, Processes, 2020, 8, 928 CrossRef.
- A. Ignaszak, C. Song, W. Zhu, J. Zhang, A. Bauer, R. Baker, V. Neburchilov, S. Ye and S. Campbell, Electrochim. Acta, 2012, 69, 397–405 CrossRef.
- J. Lobato, H. Zamora, J. Plaza and M. A. Rodrigo, ChemCatChem, 2016, 8, 848–854 CrossRef.
- N. Pimperl, N. Bevilacqua, M. A. Schmid, P. A. Loichet Torres, H. A. El-Sayed, R. Zeis and K. P. Zeyer, J. Power Sources, 2021, 507, 229971 CrossRef.
| This journal is © The Royal Society of Chemistry 2025 |
