Simultaneous enhancement of thermoelectric performance and mechanical properties in lead-free cubic GeTe-based composite materials
Zihou
Xu†
a,
Zhentao
Guo†
a,
Pengyuan
Zhang
b,
Lankun
Wang
a,
Yu-Ke
Zhu
b,
Hao
Wu
b,
Fengkai
Guo
b,
Yuanbo
Yang
c,
Long
Jiang
*c,
Alexey
Kartsev
 d,
Wei
Cai
b,
Jiehe
Sui
d,
Wei
Cai
b,
Jiehe
Sui
 *b and
Zihang
Liu
*b and
Zihang
Liu
 *a
*a
aState Key Laboratory of Precision Welding & Joining of Materials and Structures, Harbin Institute of Technology, Harbin 150001, China. E-mail: zihangliu@hit.edu.cn
bNational Key Laboratory for Precision Hot Processing of Metals, Harbin Institute of Technology, Harbin 150001, China. E-mail: suijiehe@hit.edu.cn
cState Key Laboratory of Oil and Gas Equipment, CNPC Tubular Goods Research Institute, Xi'an, Shaanxi 710077, China. E-mail: jianglong003@cnpc.com.cn
dBauman Moscow State Technical University, Moscow, 105005, Russia
First published on 25th September 2025
Abstract
GeTe is a promising mid-temperature thermoelectric material that exhibits excellent thermoelectric performance. However, the rhombohedral-to-cubic phase transition occurring near 700 K causes a change in the coefficient of thermal expansion, leading to internal stresses in the device and affecting its working efficiency. In this work, we reported the alloying of Ge0.81Mn0.15Bi0.04Te with AgSbTe2 to further reduce the phase transition temperature and optimize thermoelectric performance. The co-doping of Ag and Mn increased the effective mass of the density of states, resulting in an enhancement of the Seebeck coefficient. Moreover, the lattice thermal conductivity decreased due to the combined effects of various phonon scattering mechanisms. After alloying with AgSbTe2, the average ZTave value increased to 1.18 from 300 K to 773 K in cubic (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 and, simultaneously, the Vickers microhardness was enhanced from 243 HV to 283 HV.
1. Introduction
Thermoelectric materials enable direct and reversible conversion between thermal and electrical energy owing to their unique physical properties, presenting a sustainable solution for energy and environmental challenges.1,2 Additionally, they have extensive applications in emerging technological fields such as wound healing,3 3D printing,4 and ambient energy harvesting.5 The performance of thermoelectric materials is conventionally evaluated using the dimensionless figure of merit ZT, defined as ZT = S2σT/κ, where S, σ, and κ denote the Seebeck coefficient, electrical conductivity, absolute temperature, and total thermal conductivity, respectively.6,7 The total thermal conductivity is expressed as κ = κe + κL, comprising the electronic thermal conductivity κe and the lattice thermal conductivity κL. Superior thermoelectric performance of a material directly enhances the energy-conversion efficiency of fabricated thermoelectric generators (TEGs) and therefore strategies for enhancing ZT have become the central focus of intensive research.8,9 The optimization of thermoelectric performance mainly involves two aspects: enhancing the electrical transport performance by increasing the power factor (PF, PF = S2σ) and reducing the thermal conductivity (κ) to maintain a temperature difference across the material.10 The interdependence of parameters like S, σ, and κe through the carrier concentration limits the large improvement of material performance. Current research focuses on manipulating chemical compositions and microstructural defects to decouple these parameters, with primary strategies encompassing carrier concentration optimization,11 band engineering,12,13 and phonon scattering engineering.14GeTe, a prominent IV–VI group compound, has emerged as a highly promising thermoelectric material, particularly for mid-temperature power generation applications.15 However, pristine GeTe transforms from a rhombohedral phase (R-GeTe) to a cubic phase (C-GeTe) at approximately 700 K,16–18 which leads to a change in the material's coefficient of thermal expansion. Internal stresses caused by the mismatch in thermal expansion coefficients can develop between the material and the electrodes, which leads to the formation of microcracks, ultimately resulting in the degradation or even failure of the thermoelectric device.19–21 Therefore, it is imperative to eliminate the structural phase transition to ensure the stable operation of GeTe-based thermoelectric devices. Doping with Mn lowered the phase transition temperature and optimized the electronic band structure.22 Entropy engineering suppressed phase transitions and enhanced atomic disorder, leading to enhanced ZT in Ge0.58Sb0.22Te0.8(AgSnSe2)0.2 (ref. 23) and Ge0.63Mn0.15Pb0.1Sb0.06Cd0.06Te.24 Nevertheless, the involvement of toxic Pb alloying in the aforementioned GeTe-based compounds limits their practical applications. Therefore, it is necessary to obtain lead-free cubic GeTe with high thermoelectric performance and environmental friendliness.
In this work, we started with the Ge0.81Mn0.15Bi0.04Te compound from previous research and investigated the effects of alloying it with the ternary compound AgSbTe2 on the crystal structure and thermoelectric properties. By alloying AgSbTe2, the phase transition in Ge0.81Mn0.15Bi0.04Te is further suppressed, exhibiting a stable cubic phase structure above room temperature. Due to the significant increase in the effective mass of the density of states, the Seebeck coefficient of the samples increases markedly, offsetting the decrease in electrical conductivity and thereby maintaining the same level of PF. Meanwhile, the presence of point defects, dislocations, nanoscale second phases, and nanopores significantly scatters phonons across different frequency ranges, substantially reducing the lattice thermal conductivity. Finally, the thermoelectric performance of (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 is enhanced across the entire temperature range, achieving an average ZTave of 1.18 from 300 K to 773 K, approximately 18% higher than that of the initial Ge0.81Mn0.15Bi0.04Te, together with the enhanced Vickers microhardness.
2. Experiments
2.1. Materials synthesis
High-purity elements of Ge, Mn, Bi, Ag, Sb, and Te were weighed based on the nominal compositions (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x (x = 0, 0.10, 0.20, and 0.25) in a glove box filled with an argon atmosphere. These mixtures were placed in a stainless-steel jar and subjected to ball milling for five hours. Subsequently, the ball-milled powders were placed into a graphite die with a diameter of 12.7 mm and hot-pressed at 773 K for 30 minutes under a pressure of 60 MPa.2.2. Phase and structure characterization
The phase structure of all samples at room temperature was determined by powder X-ray diffraction (XRD) using an X'Pert PRO-PANalytical, the Netherlands (Cu Kα, 10–90°). The heat flow curves were measured by differential scanning calorimetry (DSC 204 F1, Germany). The morphology and microstructure were characterized using scanning electron microscopy (SEM, Zeiss Merlin Compact, Germany) and transmission electron microscopy (TEM, FEI Talos F200X, America). An energy dispersive X-ray spectrometer (EDS) was used to characterize the distribution of elements.2.3. Thermoelectric property characterization
The electrical conductivity and Seebeck coefficient were measured using a commercial electrical testing system (ZEM-3, Japan). The thermal conductivity was calculated using κ = DρCp, where D, ρ, and Cp are the thermal diffusivity coefficient, density, and specific heat capacity, respectively. The thermal diffusivity coefficient (D) was obtained using a laser flash diffusivity system (LFA-457, Germany). The density (ρ) was measured according to the Archimedes method. The specific heat capacity Cp was calculated using the Dulong–Petit law. The room-temperature Hall carrier concentration (nH) and mobility (μH) were calculated using nH = 1/eRH and μH = σRH, where e is the elementary charge, RH is the Hall coefficient measured by the van der Pauw technique, and σ is the electrical conductivity.2.4. Density functional theory calculations
The electronic band structure of GeTe-based materials was calculated using the generalized gradient approximation (GGA) of the Perdew–Burke–Ernzerhof (PBE) functional for the exchange-correlation potential. The calculations were performed using the Vienna Ab initio Simulation Package (VASP) with the projector augmented wave (PAW) method. We constructed a 2 × 2 × 2 cubic GeTe supercell and considered the effects of Mn, Ag, and Sb doping by substituting one Ge atom with one dopant atom, respectively. The plane wave cutoff energy was set to 450 eV, and the structures were fully relaxed until the atomic forces were less than 0.001 eV Å−1. The k-point mesh of 5 × 5 × 5 was used.3. Results and discussion
Based on the room temperature X-ray diffraction patterns of (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x (x = 0, 0.10, 0.20, and 0.25) powders (Fig. 1a), the diffraction peaks correspond to cubic GeTe (PDF#00-052-0849). Moreover, weak peaks of elemental Ge were observed within the detection limit of the XRD instrument. Fig. 1b shows the XRD Rietveld refinements of the (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 sample, and the others are shown in Fig. S1. The Rietveld refinement results reveal a gradual increase in lattice parameters with higher AgSbTe2 alloying content x (Fig. 1c) in line with the left shifting of diffraction peaks. This is due to the differences in cationic radii, where the ionic radii of Ag+ (1.15 Å) and Sb3+ (0.76 Å) are larger than that of Ge2+ (0.73 Å). As illustrated in the heat flow curve (Fig. 1d), a sharp endothermic peak, which is characteristic of the rhombohedral-cubic phase transition, is observed in GeTe at approximately 670 K. In the cubic-phase Ge0.81Mn0.15Bi0.04Te, this endothermic peak disappears and is replaced by a minor thermal fluctuation observed at approximately 420 K. It exists in a metastable cubic structure at room temperature and undergoes local atomic motion during heating, transforming into a stable cubic structure, which leads to the emergence of thermal fluctuations. This is similar to the phenomenon observed by Zhu et al.25 in cubic AgSbSe2. Compared to the sharp peaks caused by the structural phase transition, such a slight thermal fluctuation only involves marginal atomic movements, rather than prompt atomic rearrangement. Alloying with AgSbTe2 enhances the symmetry of the crystal structure, transforming (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 into a non-distorted cubic structure, which leads to the disappearance of thermal fluctuations in the heat flow curve. This indicates that (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 exhibits a stable cubic structure above room temperature.The SEM image of the fresh fracture surface shows that the grain size increases from 0.5 μm in the Ge0.81Mn0.15Bi0.04Te sample (Fig. S2) to 1.0 μm in the (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 sample (Fig. 2a). Additionally, nanopores were observed in both x = 0 and x = 0.2 compositions. The medium-magnification TEM image of the (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 sample reveals nanometer-scale pores (Fig. S3). The diameter of the nanopores ranges from 10 to 100 nm. Considering that the powder prepared by high-energy ball milling has a smaller particle size, it results in a lower melting point. The melting point of Te (∼723 K) is lower than the hot-press sintering temperature (∼773 K), and the volatilization of Te during the sintering process may be the cause of the formation of nanopores.26 The SEM and EDS images of the polished surfaces of (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x samples reveal the presence of a distinct Ge second phase in all samples (Fig. S4). This phenomenon is common in GeTe thermoelectric materials.27 Due to the low formation energy of Ge vacancies in pristine C-GeTe, the composition of the matrix readily deviates from stoichiometry, resulting in the formation of Ge-rich regions within the material. In compositions with x = 0 and x = 0.1, elements other than Ge exhibit relatively uniform distributions. When x > 0.2, Ag enrichment is observed within the material. Fig. 2b shows the second phase observed in the medium-magnification TEM image. The EDS mapping in scanning transmission electron microscopy (STEM) mode in Fig. 2c–e reveals the distribution of Ag, Ge, and Te atoms. The selected area electron diffraction (SAED) pattern of the second phase is shown in Fig. 2f. The second phase composition was confirmed as Ag8GeTe6 through meticulous indexing of the diffraction patterns and elemental content analysis in the STEM-EDS mapping (Fig. S5). Fig. 2g shows the high-resolution TEM image (HRTEM) of the blue dashed square area in Fig. 2b. The inverse fast Fourier transform (IFFT) analysis image (Fig. 2h) of the selected area in Fig. 2g reveals the presence of high-density dislocations within the sample. The inset shows the corresponding SAED pattern, in which two distinct sets of diffraction patterns can be observed, corresponding to cubic GeTe and Ag8GeTe6 phases. The geometric phase analysis (GPA) images reveal the distribution of stress and strain near the interface between the two phases (Fig. 2i). These multiscale defects, including nanopores, the Ag8GeTe6 second phase, and dislocation strain, can effectively scatter phonons, contributing to the reduction of the lattice thermal conductivity (κl).28–30
Fig. 3 shows the electrical transport properties of (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x. The electrical conductivity (σ) of all samples decreases with increasing temperature, as shown in Fig. 3a, exhibiting typical degenerate semiconductor characteristics. The increase in σ at high temperatures is due to intrinsic excitation. Compared to Ge0.81Mn0.15Bi0.04Te, the σ of (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x decreases gradually with the increase in AgSbTe2 content (x). Theoretically, monovalent Ag+ and trivalent Sb3+ incorporated into the GeTe lattice compensate for the charge of two Ge2+, producing an isoelectronic effect, which does not significantly alter the carrier concentration. The Hall measurement results indicate that the carrier concentration generally increases with the increase in AgSbTe2 content (Fig. 3b). The increase in the carrier concentration is related to the higher concentration of Ge vacancies in the cubic phase, which is attributed to its lower Ge vacancy formation energy compared to that of rhombohedral GeTe.27 AgSbTe2 alloying promotes the transformation of the crystal structure from a distorted cubic phase to a stable cubic phase, increasing the number of Ge vacancies in the crystal. Each Ge vacancy generates two holes, thereby significantly increasing the carrier concentration from 6.6 × 1020 cm−3 to 1.92 × 1021 cm−3. The decrease in the carrier concentration at x = 0.25 may be due to the increased content of the second phase (n-type Ag8GeTe6 (ref. 31)), which neutralizes some of the holes in the matrix with its electrons. Since alloying with AgSbTe2 leads to an increase in the carrier concentration, the strong scattering of carriers by point defects and nanopores results in a significant decrease in mobility, ultimately causing a reduction in σ.
The S increases with the increased AgSbTe2 alloying content x (Fig. 3c). The room temperature S increases from 130.94 μV K−1 for the x = 0 sample to 206.89 μV K−1 for the x = 0.25 sample. S is inversely proportional to nH; thus, with the increase in x, nH increases and S should decrease. To explain the increase in S, the band structures of Ge7MnTe8, Ge7AgTe8, and Ge7SbTe8 were calculated (Fig. 3d and S6). Compared to the original C-GeTe, Mn doping reduces the energy difference between the W point and the other valence band maximum (VBM) at the L and Σ points, and Ag doping reduces the difference among multiple valence band maxima. Alloying with Mn and Ag promotes band convergence, increases valley degeneracy (Nv), and enhances the density-of-states (DOS) effective mass m*. However, Sb doping does not change the energy difference between the valence band maximum and has no significant effect on valence band convergence (Fig. S6). Fig. 3e shows the room-temperature S as a function of nH for (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x. The m* and calculated Pisarenko plots are obtained based on a single parabolic band (SPB)32 model at room temperature. For the initial Ge0.81Mn0.15Bi0.04Te, m* is 5.37 me. In the samples alloyed with AgSbTe2, the room-temperature m* increases significantly from 5.37 me to 17.09 me. The Seebeck coefficient can be expressed as:33
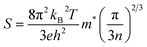 | (1) |
The total thermal conductivity (κ) of (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x samples first decreases and then increases with rising temperature (Fig. 4a). The κ decreases significantly with increasing AgSbTe2 content (x), reducing from 1.49 W m−1 K−1 for x = 0 to 0.84 W m−1 K−1 for x = 0.25 at 300 K. This reduction is attributed to the simultaneous decrease in both the electronic thermal conductivity (κe) and the lattice thermal conductivity (κL). The κe is calculated according to the Wiedemann–Franz law defined as κe = LσT, where the Lorenz number (L) is estimated based on an SPB model with acoustic phonon scattering. Owing to the decrease in σ, the κe also exhibits a pronounced decline, as shown in Fig. S8. The lattice thermal conductivity (κL) decreases significantly with increasing AgSbTe2 alloying content x (Fig. 4b). The room temperature κL decreases from 1.02 W m−1 K−1 for initial Ge0.81Mn0.15Bi0.04Te to 0.68 W m−1 K−1 for (Ge0.81Mn0.15Bi0.04Te)0.75(AgSbTe2)0.25. Additionally, within the entire temperature range of 300–773 K, the κL of the AgSbTe2 alloyed samples remains below 0.8 W m−1 K−1. The κL reaches a minimum value of 0.48 W m−1 K−1 in the sample with x = 0.25 at 773 K, which approaches the theoretical limit calculated according to the Cahill model.34 To further elucidate the influence of distinct phonon-scattering mechanisms on the κL, the Debye–Callaway model29,35–37 was employed to simulate the κL of (Ge0.81Mn0.15Bi0.04Te)0.80(AgSbTe2)0.20. As shown in the TEM observations, Umklapp process scattering (U), grain-boundary scattering (GB), point-defect scattering (PD), dislocation scattering (DS), second phase scattering (SP), and nanopore scattering (NP) mechanisms are incorporated into the Debye–Callaway model (shown in the SI). The scattered symbols indicate the experimental κL of (Ge0.81Mn0.15Bi0.04Te)0.80(AgSbTe2)0.20, while the solid curve shows the estimated values obtained by taking into account defect-related phonon scattering mechanisms (Fig. 4c). It can be seen that the calculated κL, which includes contributions from U, GB, PD, DS, SP, and NP scattering, demonstrates a good agreement with the experimental κL value. Inferring from the frequency dependence of spectral lattice thermal conductivity (κS) (Fig. 4d), point defects predominantly scatter high-frequency phonons. Dislocations, second phases, and nanopores enhance scattering of mid-frequency phonons. Low-frequency phonons are predominantly scattered by grain boundaries. Therefore, the alloying of AgSbTe2 introduces several phonon scattering mechanisms that significantly reduce κL.
Fig. 5a shows the temperature dependence of the ZT value for the (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x samples. Due to the increased Seebeck coefficient and the significant decrease in thermal conductivity, the AgSbTe2-alloyed samples exhibit a markedly higher ZT than the initial Ge0.81Mn0.15Bi0.04Te within the entire temperature range. The (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 sample exhibits a ZT value of ∼0.5 at room temperature, with a peak ZT of ∼1.58 achieved at 748 K. The average ZTave of the (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 sample is 1.18 for the 300–773 K range, which is 18% higher than the average ZTave (∼1.0) of the initial Ge0.81Mn0.15Bi0.04Te. Furthermore, this average ZTave value is comparable to or higher than that of most reported lead-free cubic GeTe-based materials.23,38–40 In addition to superior thermoelectric performance, thermoelectric materials are also required to have good mechanical stability to meet the service requirements of practical thermoelectric devices. The (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 sample exhibits a Vickers microhardness of 283 HV, much higher than that of the Ge0.81Mn0.15Bi0.04Te sample (243 HV) and surpassing that of other reported GeTe-based materials41–44 (Fig. 5b).
 | ||
| Fig. 5 Thermoelectric figure of merit and microhardness. (a) Temperature dependence of ZT for (Ge0.81Mn0.15Bi0.04Te)1−x(AgSbTe2)x, with the inset showing the ZTave values from 300 K to 773 K from this work and other ref. 23 and 38–40. (b) Room-temperature Vickers microhardness values from this work and other ref. 41–44. | ||
4. Conclusions
In this work, we successfully optimized the thermoelectric performance of lead-free cubic phase Ge0.81Mn0.15Bi0.04Te through alloying it with AgSbTe2. Alloying with AgSbTe2 further reduces the phase transition temperature, yielding a stable cubic GeTe-based thermoelectric material above room temperature. Doping with Mn and Ag reduces the energy difference between the multiple valence bands in C-GeTe, increasing the effective mass of the density of states from 5.37 me to 17.09 me, which significantly enhances the Seebeck coefficient. Moreover, various defects that are present act as phonon scattering centers, strongly scattering phonons across different frequency ranges and thereby reducing the lattice thermal conductivity. As a result, after alloying with AgSbTe2, the ZT of Ge0.81Mn0.15Bi0.04Te increases significantly and the average ZTave from 300 K to 773 K increases to 1.18, which is a relatively high level for other lead-free cubic GeTe-based materials. The Vickers microhardness of the (Ge0.81Mn0.15Bi0.04Te)0.8(AgSbTe2)0.2 sample reaches 283 HV, surpassing that of other reported GeTe-based materials. This work lays the foundation for the development of high-performance lead-free cubic GeTe-based thermoelectric devices.Author contributions
Zihou Xu and Zhentao Guo: writing – original draft, visualization, investigation, methodology and data curation. Pengyuan Zhang and Lankun Wang: software and data curation. Yu-Ke Zhu and Hao Wu: investigation and data curation. Fengkai Guo, Yuanbo Yang and Alexey Kartsev: writing – review & editing. Wei Cai: resources, supervision. Long Jiang, Jiehe Sui and Zihang Liu: writing – review & editing, conceptualization and project administration.Conflicts of interest
The authors declare no conflict of interest.Data availability
All relevant data are available in this article and the SI. Supplementary information: Fig. S1–8; Table S1 formula S1–18. See DOI: https://doi.org/10.1039/d5ta06837a.Acknowledgements
This work was funded by the National Natural Science Foundation of China (No. 22409043 and 52271206). The authors are thankful for the support from the State Key Laboratory of Precision Welding & Joining of Materials and Structures (MSWJ-24-Z-15). The authors acknowledge the Beijing Super Cloud Computing Center (BSCC) for providing HPC resources that have contributed to the research results reported in this paper. URL: http://www.blsc.cn/.References
- I. Petsagkourakis, K. Tybrandt, X. Crispin, I. Ohkubo, N. Satoh and T. Mori, Sci. Technol. Adv. Mater., 2018, 19, 836–862 CrossRef CAS PubMed.
- L. E. Bell, Science, 2008, 321, 1457–1461 CrossRef CAS PubMed.
- S. Gao, Y. Rao, X. Wang, Q. He, M. Yang, Y. Wang, Y. Zhou, J. Guo and F. Yan, Adv. Funct. Mater., 2024, 35, 2415085 CrossRef.
- S. Choo, J. Lee, B. Şişik, S.-J. Jung, K. Kim, S. E. Yang, S. Jo, C. Nam, S. Ahn, H. S. Lee, H. G. Chae, S. K. Kim, S. LeBlanc and J. S. Son, Nat. Energy, 2024, 9, 1105–1116 CAS.
- J. Cao, J. Dong, J. Wu and A. Suwardi, ACS Nano, 2025, 19, 26249–26258 CrossRef CAS PubMed.
- J. He and T. M. Tritt, Science, 2017, 357, eaak9997 CrossRef PubMed.
- G. Tan, L.-D. Zhao and M. G. Kanatzidis, Chem. Rev., 2016, 116, 12123–12149 CrossRef CAS PubMed.
- Y. Jin, Y. Qiu, C. Pan and L. D. Zhao, Adv. Mater., 2025, 37, 2500802 CrossRef CAS PubMed.
- Q. Yan and M. G. Kanatzidis, Nat. Mater., 2021, 21, 503–513 CrossRef PubMed.
- N. Li, G. Wang, Z. Zhou, G. Wang, G. Han, X. Lu and X. Zhou, Adv. Funct. Mater., 2024, 34, 2405158 CrossRef CAS.
- X. Zhang, J. Li, X. Wang, Z. Chen, J. Mao, Y. Chen and Y. Pei, J. Am. Chem. Soc., 2018, 140, 15883–15888 CrossRef CAS PubMed.
- C. Zhou, Y. Yu, Y. K. Lee, O. Cojocaru-Mirédin, B. Yoo, S.-P. Cho, J. Im, M. Wuttig, T. Hyeon and I. Chung, J. Am. Chem. Soc., 2018, 140, 15535–15545 CrossRef CAS PubMed.
- X. Dong, J. Zhu, Y.-k. Zhu, M. Liu, L. Xie, N. Qu, Y. Jin, X. Jiang, F. Guo, Z. Liu, W. Cai and J. Sui, Phys. Rev. B, 2024, 109, 155203 CrossRef CAS.
- A. R. Muchtar, B. Srinivasan, S. L. Tonquesse, S. Singh, N. Soelami, B. Yuliarto, D. Berthebaud and T. Mori, Adv. Energy Mater., 2021, 11, 2101122 CrossRef CAS.
- J. E. Boschker, R. Wang and R. Calarco, CrystEngComm, 2017, 19, 5324–5335 RSC.
- M. Hong, J. Zou and Z. G. Chen, Adv. Mater., 2019, 31, 1807071 CrossRef PubMed.
- X. Zhang, Z. Bu, S. Lin, Z. Chen, W. Li and Y. Pei, Joule, 2020, 4, 986–1003 CrossRef CAS.
- T. Xing, Q. Song, P. Qiu, Q. Zhang, X. Xia, J. Liao, R. Liu, H. Huang, J. Yang, S. Bai, D. Ren, X. Shi and L. Chen, Natl. Sci. Rev., 2019, 6, 944–954 CrossRef CAS PubMed.
- J. Cheng, L. Yin, X. Wang, S. Duan, P. Zhao, X. Ma, X. Li, X. Bao, S. Zhi, J. Mao, F. Cao and Q. Zhang, Small, 2024, 20, 2312145 CrossRef CAS PubMed.
- L. Xie, C. Ming, Q. Song, C. Wang, J. Liao, L. Wang, C. Zhu, F. Xu, Y.-Y. Sun, S. Bai and L. Chen, Sci. Adv., 2023, 9, eadg7919 CrossRef CAS PubMed.
- L. Xie, L. Yin, Y. Yu, G. Peng, S. Song, P. Ying, S. Cai, Y. Sun, W. Shi, H. Wu, N. Qu, F. Guo, W. Cai, H. Wu, Q. Zhang, K. Nielsch, Z. Ren, Z. Liu and J. Sui, Science, 2023, 382, 921–928 CrossRef CAS PubMed.
- Z. Liu, J. Sun, J. Mao, H. Zhu, W. Ren, J. Zhou, Z. Wang, D. J. Singh, J. Sui, C.-W. Chu and Z. Ren, Proc. Natl. Acad. Sci. U. S. A., 2018, 115, 5332–5337 CrossRef CAS PubMed.
- G. Liang, T. Lyu, L. Hu, W. Qu, S. Zhi, J. Li, Y. Zhang, J. He, J. Li, F. Liu, C. Zhang, W. Ao, H. Xie and H. Wu, ACS Appl. Mater. Interfaces, 2021, 13, 47081–47089 CrossRef CAS PubMed.
- S. Zhi, J. Li, L. Hu, J. Li, N. Li, H. Wu, F. Liu, C. Zhang, W. Ao, H. Xie, X. Zhao, S. J. Pennycook and T. Zhu, Adv. Sci., 2021, 8, 2100220 CrossRef CAS PubMed.
- H. Zhu, T. Zhao, B. Zhang, Z. An, S. Mao, G. Wang, X. Han, X. Lu, J. Zhang and X. Zhou, Adv. Energy Mater., 2021, 11, 2003304 CrossRef CAS.
- Y. K. Zhu, Y. Jin, J. Zhu, X. Dong, M. Liu, Y. Sun, M. Guo, F. Li, F. Guo, Q. Zhang, Z. Liu, W. Cai and J. Sui, Advanced Science, 2023, 10, 2206395 CrossRef CAS PubMed.
- Z. Liu, N. Sato, Q. Guo, W. Gao and T. Mori, NPG Asia Mater., 2020, 12, 66 CrossRef CAS.
- P. Zhang, M. Liu, Y. Zhu, H. Liang, Z. Ge, X. Dong, K. Yu, M. Lan, M. Guo, F. Guo, Z. Liu, W. Cai and J. Sui, Trans. Mater. Res., 2025, 1, 100062 CrossRef.
- J. Du, Y. Sun, F. Guo, H. Tong, Z. Yu, Z. Liu, J. Zhu and J. Sui, Mater. Today Phys., 2025, 53, 101705 CrossRef CAS.
- G. Wu, Z. Guo, Q. Zhang, X. Wang, L. Chen, X. Tan, P. Sun, G.-Q. Liu, B. Yu and J. Jiang, J. Mater. Chem. A, 2021, 9, 13065–13070 RSC.
- M. Fujikane, K. Kurosaki, H. Muta and S. Yamanaka, J. Alloys Compd., 2005, 396, 280–282 CrossRef CAS.
- J. Zhu, X. Zhang, M. Guo, J. Li, J. Hu, S. Cai, W. Cai, Y. Zhang and J. Sui, npj Comput. Mater., 2021, 7, 116 CrossRef CAS.
- N. Jia, J. Cao, X. Y. Tan, J. Dong, H. Liu, C. K. I. Tan, J. Xu, Q. Yan, X. J. Loh and A. Suwardi, Mater. Today Phys., 2021, 21, 100519 CrossRef CAS.
- D. G. Cahill, S. K. Watson and R. O. Pohl, Phys. Rev. B: Condens. Matter Mater. Phys., 1992, 46, 6131–6140 CrossRef CAS PubMed.
- J. Callaway and H. C. von Baeyer, Phys. Rev., 1960, 120, 1149–1154 CrossRef CAS.
- K. Chen, Q. Sun, H. Li, X. Tan, J. Cai, Z. Guo, Z. Zhang, G. Liu, J. Wu and J. Jiang, Small, 2025, 21, 2502510 CrossRef CAS PubMed.
- Y. Jiang, J. Dong, H. L. Zhuang, J. Yu, B. Su, H. Li, J. Pei, F. H. Sun, M. Zhou, H. Hu, J. W. Li, Z. Han, B. P. Zhang, T. Mori and J. F. Li, Nat. Commun., 2022, 13, 6087 CrossRef CAS PubMed.
- H.-S. Kim, P. Dharmaiah, B. Madavali, R. Ott, K.-H. Lee and S.-J. Hong, Acta Mater., 2017, 128, 43–53 CrossRef CAS.
- M. Samanta, S. Roychowdhury, J. Ghatak, S. Perumal and K. Biswas, Chemistry, 2017, 23, 7438–7443 CrossRef CAS PubMed.
- F. Guo, M. Liu, J. Zhu, Z. Liu, Y. Zhu, M. Guo, X. Dong, Q. Zhang, Y. Zhang, W. Cai and J. Sui, Mater. Today Phys., 2022, 27, 100780 CrossRef CAS.
- J. Zhu, F. Zhang, Y. Tai, X. Tan, Q. Deng, P. Nan, R. Cheng, C. Xia, Y. Chen, B. Ge and R. Ang, InfoMat, 2024, 6, e12514 CrossRef CAS.
- S. Perumal, S. Roychowdhury and K. Biswas, Inorg. Chem. Front., 2016, 3, 125–132 RSC.
- J. Davidow and Y. Gelbstein, J. Electron. Mater., 2012, 42, 1542–1549 CrossRef.
- Q. Sun, K. Chen, X. Tan, H. Li, G. Wu, Z. Guo, J. Cai, G. Q. Liu, J. Wu and J. Jiang, Small, 2025, 21, 2500333 CrossRef CAS PubMed.
Footnote |
| † Zihou Xu and Zhentao Guo made equal contributions. |
| This journal is © The Royal Society of Chemistry 2025 |




