A layered and highly porous poly-ionic liquid-based membrane as a gel polymer electrolyte for rechargeable lithium-ion batteries
Aparna S.
Potdar
ab,
Ujjwala P.
Chothe
b,
Murthy
Chavali
b,
Bharat B.
Kale
 *ab and
Milind V.
Kulkarni
*ab and
Milind V.
Kulkarni
 *a
*a
aCentre for Materials for Electronics Technology (C-MET), Panchawati, Off, Dr Homi Bhabha Road, Pune 411008, India. E-mail: milind@cmet.gov.in
bMIT World Peace University, Kothrud, Pune 411038, India. E-mail: bbkale1@gmail
First published on 13th November 2025
Abstract
Gel electrolytes have great significance in solid-state batteries; in this regard, lithium-ion conducting gel polymer electrolyte (GPE) membranes were developed using an optimized blend of the polymeric ionic liquid poly(diallyldimethylammonium)bis(trifluoromethanesulfonyl)imide (PDADMATFSI), poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP), and lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) salt. The resulting GPEs were systematically characterized for their morphological and electrochemical properties. The morphological study of the GPE membrane clearly shows a unique layered structure with distinct layers. The optimized membrane exhibited a high ionic conductivity of 1.48 × 10−3 S cm−1 at 25 °C, which increased to 3.17 × 10−3 S cm−1 at 65 °C, indicating efficient lithium-ion transport. It also demonstrated excellent mechanical flexibility, a broad electrochemical stability window of 4.8 V, and a lithium-ion transference number of 0.75. When employed in a Li|GPIL-3|LiFePO4 half-cell configuration, the membrane enabled a high initial discharge capacity of 127.09 mAh g−1 at 0.1C. which increased to 155.75 mAh g−1 by the fifth cycle. Moreover, it delivered 151 mAh g−1 at 1C with a capacity retention of 72.91% over 600 cycles, highlighting its robust rate capability and cycling stability. Ionic transport in the GPE membrane occurs primarily within the amorphous regions, where mobile Li+ cations coordinate with electronegative atoms along the polymer backbone, enabling stepwise migration between transient coordination sites and facilitating efficient conduction. Additionally, the membrane's layered structure is responsible for ion transport. Notably, the ionic conductivity achieved at room temperature surpasses previously reported values for PDADMA-based GPE systems. These findings underscore the potential of the optimized GPE composition for high-performance lithium-ion batteries.
1. Introduction
Over the past three decades, lithium-ion batteries (LIBs) have emerged as the foremost rechargeable batteries, owing to their high energy density, elevated operating voltage, low self-discharge rate, and excellent cycling stability.1,2 However, safety concerns persist due to the use of flammable, organic liquid electrolytes. Solid-state electrolytes (SSEs) have recently gained significant attention due to their non-flammable nature, broad electrochemical window, thermal stability, and compatibility with lithium metal anodes.3–5 Solid-state electrolytes (SSEs) fall into three major categories: inorganic ion conductors, solid polymer electrolytes (SPEs), and organic–inorganic hybrid composites. Among SSEs, SPEs have several advantages over inorganic ion conductors, including low interfacial resistance, excellent flexibility, and cost-effectiveness.6,7 Despite these benefits, their low ionic conductivity is a major drawback. Therefore, enhancing the ionic conductivity of SPEs is crucial for their practical use.8–10 Various strategies have been employed to enhance the room-temperature ionic conductivity of SPEs, including blending, copolymerization, adding inorganic fillers, and plasticizers. Depending on the materials used and the approach taken, these electrolytes are categorized as complex, blend, nanocomposite, or gel polymer electrolytes (GPEs).11–14 GPEs are highly regarded for comprising liquid-like transport properties with solid-like mechanical stability. These membrane-like systems offer excellent physical, chemical, thermal, electrical, and electrochemical properties, ensuring both performance and safety.15,16The polymer host in GPEs plays a crucial role in determining their mechanical properties and lithium-ion transport efficiency. Zhou et al. explore the potential of gel polymer electrolytes (GPEs) as a promising approach to improve the thermal stability and performance of rechargeable batteries over a wide temperature range.17 Voice et al. studied the gelation ability of various polymers with plasticizers. They discovered that poly(acrylonitrile) (PAN) and poly(vinylidene fluoride) (PVDF) hosts can create uniform gel structures with molecular-level dispersion of the salt and plasticizer.18 Polymers like poly(ethylene oxide) (PEO) and PMMA have also been used, achieving relatively high conductivities.19,20
Daems et al. demonstrated that introducing a second acrylate-based polymer into a cross-linked PAN network improved both mechanical flexibility and ionic conductivity.21 However, traditional polymers often exhibit strong interactions with Li+, which can restrict their mobility.18 On the other hand, PVDF, with its high polarity and large dielectric constant, and featuring the electron-withdrawing C–F group, is highly effective at dissolving lithium salts.22 Nonetheless, its high crystallinity at room temperature (40–70%) presents a significant challenge. PVDF-HFP stands out due to its exceptional electrochemical stability and degradation resistance, attributed to the strong electron-withdrawing nature of its fluorinated (–C–F) groups.23–25 Its high dielectric constant (ε = 8.4) enhances ion transport efficiency within the electrolyte matrix. Additionally, the inclusion of fluorinated propylene (–HFP) reduces the crystallinity of the polymer matrix, increasing ionic conductivity and promoting an amorphous nature. PVDF-HFP offers notable advantages for LIBs, including an enhanced dielectric constant, excellent chemical stability, and superior mechanical strength compared to the fabricated membranes.26 Moreover, incorporating gel polymer electrolytes leads to notable improvements in battery performance, as they ensure effective contact with the electrodes. However, the environmental friendliness of these electrolytes is limited due to the use of organic plasticizers. Two promising solutions to this issue are using ionic liquids and polyionic liquids (PILs) as safer plasticizers. Polymeric ionic liquids combine the beneficial properties of both polymers and ionic liquids. When a lithium salt is added, a PIL is formed. PILs with TFSI anions are particularly promising due to their wide charge distribution, high ionic conductivity, effective plasticizing properties, and compatibility with various electrodes.16 PDADMA TFSI has emerged as a leading polymeric ionic liquid matrix for gel polymer electrolytes incorporating pyrrolidonium and imidazolium-based ionic liquids.27–29 It demonstrates exceptional performance in applications such as lithium and lithium-sulfur batteries and supercapacitors.29–32 As a result, PILs are highly regarded as suitable polymer electrolytes and have been widely applied in battery technologies.32,33 Wang et al. reported that in the PDADMAFSI system with LiFSI, increasing the lithium salt ratio leads to anion Li+ aggregation, which may further enhance lithium-ion diffusion.34
The incorporation of PDADMA-TFSI into a PVDF-HFP and LiTFSI matrix has shown great promise for developing self-supporting gel polymer electrolytes (GPEs) tailored for lithium-ion batteries.35–37 This approach not only improves battery performance but also enhances overall system efficiency. The films formed from poly (DADMA-TFSI) and PVDF-HFP exhibit a semi-crystalline structure, where the addition of LiTFSI increases the amorphous regions, an essential factor for boosting ionic conductivity and electrochemical behaviour. Enhanced amorphosity facilitates reduced interfacial resistance and promotes improved electrochemical performance in the device. Sporadic Literature is available on this system; however, there is a vast ground to play with this system for better performance. Hence, we have put efforts into modifying the system for better lithium-ion transport with good reversibility.
In the present study, poly(DADMA-TFSI) was synthesized through an anion exchange process and integrated with PVDF-HFP and LiTFSI to create a freestanding GPE membrane. This membrane demonstrated excellent compatibility with LiFePO4 cathodes, retaining 72.91% of its capacity after 600 cycles at 1C rate, all while maintaining high coulombic efficiency. These findings underscore the synergistic benefits of combining PDADMA-TFSI, PVDF-HFP, and LiTFSI for enhanced lithium-ion mobility, improved rate performance, and long-term cycling stability in Li-ion batteries.
2. Experimental section
2.1 Materials and methods
Poly(diallyl dimethylammonium)chloride solution (average Mw 400![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000–500
000–500![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000, 20 wt% in H2O) was acquired from Aldrich, poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP), poly(vinyl difluoride) (PVDF), lithium iron phosphate (LiFePO4), and N-methyl-2-pyrrolidone (NMP) were procured from Sigma-Aldrich chemicals and distilled before their use. Lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) salt was procured from TCI Chemicals and used as obtained. Carbon black was procured from Merck Life Sciences and MTI, respectively.
000, 20 wt% in H2O) was acquired from Aldrich, poly(vinylidene fluoride-co-hexafluoropropylene) (PVDF-HFP), poly(vinyl difluoride) (PVDF), lithium iron phosphate (LiFePO4), and N-methyl-2-pyrrolidone (NMP) were procured from Sigma-Aldrich chemicals and distilled before their use. Lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) salt was procured from TCI Chemicals and used as obtained. Carbon black was procured from Merck Life Sciences and MTI, respectively.
2.2 Synthesis of poly(diallyl dimethyl ammonium)bis(trifluoromethane sulfonyl)imide (PDDA-TFSI)
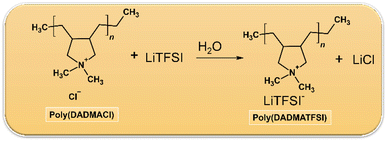
The synthesis of the PIL(PDADMA-TFSI) was achieved via an anion exchange reaction, as described in the literature.11,38Scheme 1 involved the use of commercially available poly(diallyldimethylammonium) chloride and a lithium salt. Initially, 8.52 g (29.68 mmol) of LITFSI was dissolved in 100 ml of distilled water. This solution was then added dropwise to another solution containing 4 g (24.74 mmol of monomer units) of poly(diallyldimethylammonium) chloride in 100 ml of distilled water. The reaction formed a white solid, and the mixture was stirred at room temperature for 30 minutes. Afterwards, the crude product was washed thrice with distilled water, and the collected precipitate was vacuum-dried at 60 °C for 24 h. The structure of the resulting material was confirmed by a 1H NMR study recorded in acetone-d6 solvent at 500 MHz, and is presented in Fig. SI1.2.3 Preparation of gel polymer electrolyte (GPE) membrane
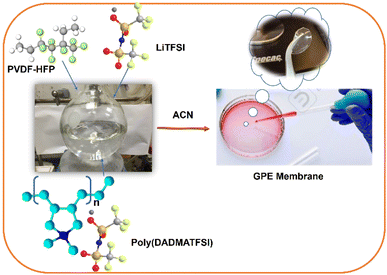
The GPE membrane was prepared by the simple casting approach method. In the synthesis of the GPE membrane, PVDF-HFP (0.2 g, 40 wt%) and lithium salt (0.2 g, 40 wt%) were dissolved in acetonitrile (ACN) (10 ml) at 60 °C. PDDA-TFSI (0.1 g, 20 wt%) was added to this mixture. The subsequent mixture was kept for ultrasonication to get a homogeneous solution, followed by transferring it into the Petri dish to obtain the flexible, free-standing film/membrane at room temperature. The GPE membrane was allowed to dry slowly through solvent evaporation at ambient temperature and finally, vacuum dried at 60 °C for 24 h. GPE membrane is uniform and flexible in appearance, as illustrated in Scheme 2.2.4 Characterization methods
Different characterization techniques were explored for the structural and morphological elucidation of the materials. The structure and purity of PIL were confirmed by 1H NMR spectroscopy. The morphological study of the as-synthesized membrane was carried out using a Field Emission Scanning Electron Microscope (FE-SEM TESCAN, CLARA). Fourier transform infrared (FT-IR) spectra were measured by using a PerkinElmer FTIR spectrometer in transmittance mode (within 400 and 4000 cm−1). The crystallographic features of the samples were analyzed using a Bruker D8 Advance X-ray diffractometer equipped with Cu Kα radiation. Thermogravimetric-differential thermal analysis (TG-DTA, Setsys, Setaram) was performed between 25 and 700 °C under a nitrogen atmosphere at a heating rate of 10 °C min−1, following sample degassing at 80 °C for 12 h. Raman spectra were obtained using a JASCO NRS 4500 Raman spectrometer equipped with a F012861984 Raman module. A green laser with an excitation wavelength of 531.93 nm served as the light source. The laser power was maintained at 20.1 mW, with a spectral resolution of 28.53 cm−1, and each spectrum represented an average of 1000 scans. All measurements were carried out at ambient temperature. Tensile testing of the GPIL-3 membrane was carried out at room temperature using dumbbell-shaped samples on a universal testing machine (model STS248, India).2.5 Electrode preparation and cell fabrication
The Li-ion half-cells (coin 2032 type) were fabricated using GPIL membrane to carry out electrochemical measurements and their performance. The electrodes were fabricated by formulating the slurry with a stoichiometric ratio of active material (LiFePO4), conducting carbon (C65, carbon black powder from Global Nanotech), and binder (polyvinylidene fluoride-PVDF) as 80![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10
10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 in N-methyl pyrrolidine solvent. The slurry was coated on a carbon-coated aluminium foil through the doctor blade technique, followed by overnight drying at 120 °C in a vacuum oven. The prepared GPE membrane and coated foil of electrode material were punched into 19 mm and 16 mm circular discs, respectively. The Li-ion coin cell was assembled in an ‘Ar’ filled glove box by placing the GPE membrane between the LFP cathode and Li metal foil (0.75 mm thickness) as an anode. Diethylene glycol dimethyl ether (Diglyme) was used as a solvent to have proper interfacial contact between the electrode–electrolyte interfaces during the fabrication of a Li-ion half-cell.
10 in N-methyl pyrrolidine solvent. The slurry was coated on a carbon-coated aluminium foil through the doctor blade technique, followed by overnight drying at 120 °C in a vacuum oven. The prepared GPE membrane and coated foil of electrode material were punched into 19 mm and 16 mm circular discs, respectively. The Li-ion coin cell was assembled in an ‘Ar’ filled glove box by placing the GPE membrane between the LFP cathode and Li metal foil (0.75 mm thickness) as an anode. Diethylene glycol dimethyl ether (Diglyme) was used as a solvent to have proper interfacial contact between the electrode–electrolyte interfaces during the fabrication of a Li-ion half-cell.
2.6 Electrochemical characterization
The AC impedance method was used to assess the ion conductivity of the GPE membrane. The stainless-steel SS|GPE|SS symmetric stainless-steel device was fabricated and tested for electrochemical impedance spectra (EIS) in the frequency range of 1 MHz–1 Hz with a potential amplitude of 10 mV. The conductivity values (“σ”) were determined using the following equation| σ = l/RA | (1) |
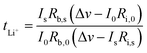 | (2) |
Cyclic voltammetry (CV) was performed using a Metrohm Autolab electrochemical workstation. For this, a coin cell with the configuration Li|GPIL-3|LFP was tested. The CV analysis was conducted within a potential range of 2.0–4.3 V with a scan rate of 0.1 mV s−1. To evaluate the electrochemical stability of the GPE membrane, linear sweep voltammetry (LSV) was carried out. The LSV profiles for Li|GPIL-3|SS cells were obtained at a scan rate of 0.1 mV s−1 over a voltage range of 0.0–6.0 V. Stability testing was also conducted using a cell configuration of Li|GPIL-3 GPE|Li, where the membrane was sandwiched between two lithium foil electrodes. The electrochemical performance of the GPIL-3 membrane was further tested with lithium metal foil over a potential range of −1.5 V to 6.5 V, also at a scan rate of 0.1 mV s−1. Finally, the galvanostatic charge–discharge cycling performance of a half-cell with the configuration Li|GPIL-3|LFP was examined using a battery analyser (MTI Corp., USA). These studies were conducted at various current densities within a potential 2.0–4.3 V window.
3. Results and discussions
The experimental section comprehensively describes the synthesis procedure for PIL, accompanied by a schematic illustration of the GPE membranes shown in Schemes 1 and 2, respectively. In this work, PILs were intentionally integrated into the system to improve key properties of the electrolyte, such as ionic conductivity, flexibility, and interfacial stability.The PIL-based GPE membranes underwent detailed characterization through various analytical methods and were utilized as electrolytes to enhance the ionic conductivity and safety of LIBs. Furthermore, GPE membranes were developed by adjusting the precursor compositions listed in Table 1. Various component ratios were prepared, and their ionic conductivities were analyzed using impedance spectroscopy (Fig. SI2).
| Name | PVDF-HFP (wt%) | LITFSI (wt%) | PIL (wt%) | Conductivity (S cm−1) |
|---|---|---|---|---|
| PFSI | 60 | 40 | — | 6.23 × 10−5 |
| GPIL-1 | 40 | 20 | 40 | 4.49 × 10−4 |
| GPIL-2 | 40 | 30 | 30 | 9.043 × 10−4 |
| GPIL-3 | 40 | 40 | 20 | 1.48 × 10−3 |
According to Table 1, GPE membranes were prepared through the blend of PILs, PVDF-HFP and LiTFSI salt. The GPIL membrane was developed by combining different proportions of PIL, LITFSI and PVDF-HFP polymer, as outlined in Table 1. The PFSI membrane without PIL exhibited relatively low ionic conductivity, i.e. 6.23 × 10−5 S cm−1 at room temperature. On the contrary, the GPIL-3 membrane displayed improved characteristics such as uniformity, transparency, and flexibility, with a notably higher ionic conductivity of 1.48 × 10−3 S cm−1 at 30 °C. Furthermore, changes in the concentrations of PIL and lithium salt negatively affected the film formation process, leading to poor or incomplete film development. Since the GPIL-3 exhibited the highest ionic conductivity relative to the other membranes, and was thus selected for subsequent electrochemical evaluations to determine its potential applicability in lithium-ion batteries.
The ionic conductivity of the GPIL-3 membrane was assessed at different temperatures, as shown in Fig. 1.
 | ||
| Fig. 1 (a) Nyquist plots of the GPE membrane at different temperatures, (b) Arrhenius plot of the ionic conductivity of the GPE membrane at different temperatures of the cell SS|GPIL-3|SS. | ||
Impedance measurements of the GPIL-3 membrane across a temperature range from 25 °C to 65 °C revealed a steady increase in ionic conductivity (Table SI1, SI). This increase in conductivity with temperature is attributed to the reduced polymer viscosity, which facilitates greater ionic mobility.
The plot of log![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) s versus 1000/T follows an Arrhenius-type trend, as expressed by eqn (3).
s versus 1000/T follows an Arrhenius-type trend, as expressed by eqn (3).
σ = σ0![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) exp(Ea/kT) exp(Ea/kT) | (3) |
The mechanism of ionic transport in the polymer membrane primarily occurs within the amorphous regions, where the polymer chains exhibit greater mobility, thereby facilitating ion movement. In this system, Li+ cations are transported through the porous membrane by coordinating with electronegative atoms, specifically the oxygen atoms in the sulfonyl groups of the TFSI anion from the polymeric ionic liquid (PIL), LITFSI, and the fluorine atoms in the PVDF-HFP polymer matrix. These coordination sites serve as temporary anchors that guide the Li+ ions through the membrane. The migration of Li+ occurs via an intrachain hopping mechanism, where the ions move stepwise from one coordination site to another along the polymer backbone, enabling efficient ionic conduction across the membrane mentioned in Fig. 2.
 | ||
| Fig. 2 Schematic presentation of the Li-ion conduction within the GPIL-3 membrane. Colour code: Ni atoms, blue; O atoms, red; H atoms, white; C atoms, cyan. F atoms, green; Li-ion, orange. | ||
The lithium-ion transference number was determined under anion-blocking conditions using the potentiostatic polarization method by fabricating a Li|GPIL-3|Li symmetric cell (Fig. 3).
However, it can also be broadly defined as the fraction of the total current carried by Li ions under these conditions. This makes it a crucial parameter for evaluating the transport properties of electrolyte materials in LIBs. The GPIL-3 membrane has a transference number of 0.75. An increased concentration of lithium salt slows down the decay of polarization current, which helps minimize concentration gradients, maintain conductivity, and enhance ion mobility.11,37
A high lithium-ion transference number is particularly beneficial for maximizing its contribution to charge transport and minimizing the influence of other ions, which ultimately stabilizes the solid electrolyte interphase (SEI). Together, these effects contribute to better stable cycling performance and improved overall battery health.39
The crystalline characteristics of the pristine poly(ionic liquid) (PIL) and the GPIL-3 composite membrane were analysed using X-ray diffraction (XRD), as shown in Fig. 4. The pristine poly (DADMATFSI) displays three distinct diffraction peaks at 2θ 12.38°, 17.46°, and 19.42°, corresponding to the crystalline regions of the polymeric ionic liquid. In contrast, the XRD pattern of LiTFSI exhibits the major diffraction peaks at 2θ 17.64° and 41.36° align well with JCPDS Card No. 01-082-2341 and correspond to the (011) and (411) lattice planes, respectively.36Additionally, the characteristic peaks at 2θ 20.38° and 26.70° are consistent with the (110) and (021) planes of semi-crystalline P(VDF-HFP), as reported in literature.11,41–43
In comparison, the GPIL-3 membrane exhibits broader and less intense peaks at corresponding 2θ values, indicating a significant reduction in crystallinity after the incorporation of LiTFSI and PVDF-HFP. The presence of these additives disrupts the regular packing of PIL chains, thereby enhancing the amorphous nature of the composite membrane.
To quantitatively evaluate these structural variations, Gaussian fitting of the diffraction peaks was carried out in Origin 2018 (see Fig. SI3), and the full width at half maximum (FWHM) values were used to calculate the crystallite sizes using the Scherrer equation
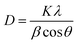 | (4) |
| Sample | 2(θ)° | FWHM (°) | Crystallite size (nm) |
|---|---|---|---|
| PIL-2 | 12.38 | 1.419 | 5.89 |
| 17.46 | 2.693 | 3.12 | |
| 41.07 | 5.188 | 1.71 | |
| GPIL-3 | 12.41 | 2.59 | 3.33 |
| 17.36 | 3.44 | 2.44 | |
| 41.93 | 5.69 | 1.56 |
GPIL-3 exhibits consistently smaller crystallite sizes compared with PIL (e.g., 5.89 to 3.33 nm, 3.12 to 2.44 nm, and 1.71 to 1.56 nm for corresponding peaks). The systematic broadening across all reflections confirms that the decrease in intensity is primarily due to reduced crystalline order rather than peak overlap.44
The degree of crystallinity (Xc) was further estimated using the integrated peak area method according to eqn (3).45
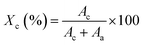 | (5) |
The enhanced amorphous phase within GPIL-3 is beneficial for its function as a gel polymer electrolyte, as it facilitates greater polymer segmental motion and thereby improves Li+ ion mobility. Consequently, the structural modifications induced by LiTFSI and PVDF-HFP are expected to increase ionic conductivity and contribute to more stable charge–discharge cycling in lithium-ion batteries (LIBs).
Fig. 5 presents the FTIR spectra of the Plane PFSI and GPIL-3 membranes, which were analyzed to investigate the interactions among the PVDF-HFP polymer, LiTFSI, and PIL.
The FTIR spectrum of the PFSI membrane exhibits distinct vibrational peaks at 1660, 1350, 1170, 1134, 1051, 884, and 742 cm−1. These peaks are attributed to various functional group vibrations: the peak at 1661 cm−1 corresponds to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C stretching of the polymer backbone, while the band at 1350 cm−1 is assigned to the S
C stretching of the polymer backbone, while the band at 1350 cm−1 is assigned to the S![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O stretching vibration from the TFSI anion. Peaks observed at 1170 cm−1 and 1130 cm−1 correspond to symmetrical stretching of the –SO2 group. A significant shift in the peak position is noticeable when PDADMA TFSI is combined with PVDF-HFP and LiTFSI. The band corresponding to the –SO2–N–SO2– stretching vibration, typically observed at 1130, 1050 cm−1 in the TFSI structure, shifts to 1134 cm−1 and 1057 cm−1. Asymmetric stretching peaks at 1355 cm−1 and 1329 cm−1, along with a symmetric stretching peak at 1144 cm−1, all corresponding to the SO2 functional group.48 Additionally, the peaks at 884 cm−1 and 738 cm−1 are attributed to CF2 bending vibrations in PVDF-HFP and C–S bond stretching in the CF3SO2 group, respectively.36,49
O stretching vibration from the TFSI anion. Peaks observed at 1170 cm−1 and 1130 cm−1 correspond to symmetrical stretching of the –SO2 group. A significant shift in the peak position is noticeable when PDADMA TFSI is combined with PVDF-HFP and LiTFSI. The band corresponding to the –SO2–N–SO2– stretching vibration, typically observed at 1130, 1050 cm−1 in the TFSI structure, shifts to 1134 cm−1 and 1057 cm−1. Asymmetric stretching peaks at 1355 cm−1 and 1329 cm−1, along with a symmetric stretching peak at 1144 cm−1, all corresponding to the SO2 functional group.48 Additionally, the peaks at 884 cm−1 and 738 cm−1 are attributed to CF2 bending vibrations in PVDF-HFP and C–S bond stretching in the CF3SO2 group, respectively.36,49
In contrast, the FTIR spectrum of the GPIL-3 membrane reveals characteristic features of PVDF-HFP, LiTFSI, and PIL, confirming its composite nature. However, when the Li salt is added, the peak intensity also increases. Notably, the GPIL-3 membrane displays a prominent broad band at 3502 cm−1, indicating the presence of –OH groups and suggesting its hygroscopic nature. Moreover, the peaks at 840 cm−1 and 745 cm−1 correspond to the –FSI anions, which can be attributed to the presence of PDADMA TFSI.16,36
Fig. 6 represents the Raman spectroscopy of GPIL-3 membrane and recovered GPIL-3 membrane after charge discharge, which further supports the proposed Li+–TFSI−/F coordination mechanism. The characteristic PVDF-HFP and LiTFSI band near ∼732.2 cm−1 progressively shifts to ∼747.8 cm−1 upon cycling, indicating stronger Li+–TFSI− interactions, consistent with previous studies.34,50–53 This coordination facilitates directional Li+ migration along the ordered domains of the layered structure, as clearly seen in the FE-SEM.
Based on previous reports, PDADMA TFSI exhibits a glass transition temperature (Tg) of around 310 °C, while dynamic mechanical analysis (DMA) has determined 121 °C.54,55 Upon blending PDADMA with LiFSI in a molar ratio of PDADMA FSI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LiFSI = 1
LiFSI = 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 2, the Tg significantly decreases to below −50 °C,34 while pure PVDF-HFP shows a Tg near −35 °C.56 In our study, the GPIL-3 membrane (PIL
2, the Tg significantly decreases to below −50 °C,34 while pure PVDF-HFP shows a Tg near −35 °C.56 In our study, the GPIL-3 membrane (PIL![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) LiTFSI
LiTFSI![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) PVDF-HFP = 20
PVDF-HFP = 20![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40
40![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 40) demonstrates a further reduction in Tg due to the synergistic blending of PIL, PVDF-HFP, and LiTFSI. This reduction in Tg enhances polymer chain mobility within the amorphous regions, promoting Li+ migration. Raman analysis supports this observation, revealing strong coordination interactions between Li+ ions and the fluorine/oxygen sites of the polymer salt matrix. Under operating conditions where the temperature slightly exceeds Tg, Li+ conduction predominantly occurs via a hopping mechanism assisted by these coordinated sites.57
40) demonstrates a further reduction in Tg due to the synergistic blending of PIL, PVDF-HFP, and LiTFSI. This reduction in Tg enhances polymer chain mobility within the amorphous regions, promoting Li+ migration. Raman analysis supports this observation, revealing strong coordination interactions between Li+ ions and the fluorine/oxygen sites of the polymer salt matrix. Under operating conditions where the temperature slightly exceeds Tg, Li+ conduction predominantly occurs via a hopping mechanism assisted by these coordinated sites.57
Thermogravimetric analysis (Fig. 7) shows that both samples exhibit negligible mass loss below 100 °C, confirming low moisture content. The PIL remains stable up to ∼450 °C before undergoing a sharp, single-step decomposition, whereas GPIL-3 membrane shows a multi-stage degradation where a gradual loss occurs from ∼100–350 °C, a major decomposition region between ∼350–450 °C, and a substantial residual mass of ∼8–15% at 700 °C. The higher char yield of GPIL-3 is attributed to thermally stable fluorinated and inorganic species from PVDF-HFP and LiTFSI.
The differences in decomposition behaviour reflect the multi-component nature of GPIL-3. The early, gradual weight loss in GPIL-3 is attributed to the volatilisation of physically adsorbed species and the onset of decomposition of less thermally stable organic constituents introduced by PVDF-HFP and LiTFSI interactions with the PIL matrix. The main decomposition event for both samples corresponds to backbone degradation of the organic ionic liquid polymer; however, incorporation of PVDF-HFP and LiTFSI broadens and shifts the decomposition profile, producing a less abrupt mass-loss step and increasing the char yield. The higher residual mass of GPIL-3 relative to neat PIL is consistent with the presence of inorganic/ionic species (LiTFSI) and thermally stable fluorinated fragments from PVDF-HFP.58–60 More significantly, for battery applications, both materials are thermally stable well above the typical operating and abuse temperatures of lithium-ion cells (<100 °C), ensuring safe use.
Mechanical performance was evaluated by tensile testing (Fig. SI4). The GPIL-3 film exhibits elastic behaviour up to ∼6.5% strain with a maximum tensile stress of ∼3.2 MPa, followed by gradual stress relaxation while maintaining integrity up to ∼12% strain. A slight stress recovery at higher strain suggests polymer chain alignment or structural rearrangement. The combination of adequate strength and flexibility enables the membrane to maintain electrode contact and support stable charge–discharge cycling.61
Overall, the GPIL-3 GPE membrane combines reduced crystallinity, high thermal stability, and mechanical robustness, making it a promising candidate for both flexible and conventional lithium-ion battery applications. Fig. 8d.
 | ||
| Fig. 8 FE-SEM images of (a) PIL, (b) PFSI membrane (inset image cross-section), (c) GPIL-3 membrane, (d) GPIL-3 cross-section. | ||
Fig. 8 presents the FE-SEM images of the PIL (Fig. 8a), PFSI membrane (Fig. 8b), GPIL-3 membrane (Fig. 8c), and GPIL-3 cross-section (Fig. 8d), highlighting their morphological differences. The pure PIL membrane exhibits individual polymer aggregates and clusters, indicating phase separation within the material. In contrast, the PFSI membrane demonstrates the distribution of LiTFSI over the PVDF-HFP matrix, suggesting the successful incorporation of the lithium salt.58
The GPIL-3 membrane appears as a porous structure with a uniform distribution of LiTFSI, which is expected to collectively enhance ionic conductivity. Additionally, the cross-sectional FE-SEM image of the GPIL-3 membrane (Fig. 8d) reveals a layered structure. In contrast, the cross-section of the PFSI membrane (inset image of Fig. 8b) displays a relatively uniform and planar morphology. FE-SEM images clearly show ∼2–4 µm layers in the cross-section of the GPIL-3 membrane. These layered and porous structural characteristics influence the ion transport properties of the membranes of GPIL-3, potentially contributing to enhancing the electrochemical performance. The GPIL-3 membrane was tested for flammability using the vertical method (IS 15061:2002) and showed excellent flame–retardant properties (Fig. SI5). This suggests that GPIL-3 has strong self-extinguishing capabilities and demonstrates potential in safe and stable Li-ion batteries.
4. Characterization
4.1 Cyclic voltammetry (CV)
Cyclic voltammetry (CV) measurements were carried out on the GPIL-3 membrane by fabricating an LFP|GPIL-3|Li cell, in the potential range of 2.5 to 4.3 V (vs. Li/Li+) at room temperature. The CV data, recorded at a scan rate of 0.1 mV s−1 for four cycles, is presented in Fig. 9a. | ||
| Fig. 9 (a) Cyclic voltammetry profile of GPIL-3 membrane cycled at room temperature with 0.1 mV s−1 scan rate, (b) LSV curve of Li|GPIL-3|SS. | ||
The overlapping CV curves across all cycles indicate efficient intercalation and deintercalation of Li+ ions within the prepared GPIL-3 membrane. An anodic peak potential of 3.62 V and a cathodic peak potential of 3.24 V were observed, with both peaks consuming equal current. This symmetry highlights the excellent reversibility through the electrolyte during the electrochemical processes, which is consistent with the biphasic Li+ ion insertion/extraction mechanism from the LFP cathode. Additionally, no extraneous peaks were detected in the CV analysis, confirming the high purity of the developed electrolytes.62,63
The electrochemical stability window (ESW) of the GPE membrane was further evaluated using the linear sweep voltammetry (LSV) technique with a Li/GPIL-3/SS cell configuration (Fig. 9b). This analysis, performed over a potential range of 0 to 6 V (vs. Li/Li+) with a scan rate of 0.1 mV s−1 at room temperature, demonstrated an anodic stability limit of 4.8 V. This result indicates superior oxidative stability compared to conventional liquid electrolytes. The improved stability can be attributed to the incorporation of a PIL and the LiTFSI anion in the PVDF-HFP matrix, which is known for its excellent electrochemical stability. Throughout the LSV analysis, no fluctuations in current were observed, signifying the existence of high purity of the GPE membrane. These findings confirm that the GPE membrane is an electrochemically stable and suitable electrolyte for Li-ion battery applications.
4.2 Cycling study
To evaluate the cycling performance of the GPIL-3 membrane, a half-coin cell was fabricated using an LiFePO4|GPIL-3|Li configuration. The electrochemical behaviour of the cell was tested within a voltage range of 2.0 to 4.3 V (vs. Li/Li+) under varying current rates at ambient temperature. Fig. 10a shows the cycling performance obtained at various C-rates, while Fig. 10b illustrates the galvanostatic charge–discharge curves at different current rates. The curves displayed a prominent plateau near 3.4 V versus Li/Li+, a typical feature of LiFePO4 cathode materials.This plateau signifies the characteristic two-phase reaction mechanism of LiFePO4, where lithium-ion insertion and extraction occur with minimal voltage variation, demonstrating its stable electrochemical behaviour.
Initially, the cell delivered a first discharge capacity of approximately 127.09 mAh g−1 at a current rate of 0.1C. From the second cycle onward, the capacity gradually increased, reaching 155.74 mAh g−1 by the fifth cycle. This enhancement in capacity during the initial cycling can be attributed to electrochemical stabilization processes within the cell. The open-circuit voltage (OCV) profile indicates the occurrence of initial side reactions at the electrode–electrolyte interface, likely involving the partial trapping of lithium ions within the polymer matrix or the formation of a solid electrolyte interphase (SEI)-like layer.
As cycling progresses, these previously immobilized lithium ions are gradually released, thereby contributing to the observed increase in discharge capacity.64 In terms of rate performance, the LFP|GPIL-3|Li half-cell demonstrated discharge capacities of 155.7, 152.70, 154.49, 151.07, and 119.97 mAh g−1 when tested at 0.1C, 0.2C, 0.5C, 1C, and 2C, respectively. When the cell was retained at a 0.1C current rate, it maintained a stable capacity of 151.44 mAh g−1, indicating good reversibility (Fig. 10a). Incorporating PIL and LITFSI in PVDF-HFP polymer has proven highly effective in improving the rate capability of GPE, which is a vital factor for practical applications. As shown in Fig. 10b, the rate performance was evaluated across various current rates ranging from 0.1 to 2C. The LFP|GPIL-3|Li cell demonstrated superior discharge performance compared to literature data available to date (Table SI2). Notably, when the current rate was reverted to 0.1C, the cell's capacity nearly retained its initial value, demonstrating its remarkable cyclic stability and excellent reversibility.
The cycling performance of the GPIL membrane was assessed at a 1C rate, the cell exhibited an initial discharge capacity of 151.03 mAh g−1 with a high coulombic efficiency of 99.66%. Throughout 200 cycles, the capacity gradually declined to 135.16 mAh g−1, corresponding to a capacity retention of 89.4% (Fig. 10c). In Fig. 10c, the charging peak observed at cycles 40–60 is likely associated with structural reorganization within the PIL-based membrane or transient changes in the electrode–electrolyte interface, such as partial SEI reformation, which can temporarily enhance ion transport. This is a known and often benign behaviour as the system stabilizes.65
With extended cycling, the discharge capacity decreased to about 110.13 mAh g−1 after 600 cycles, corresponding to a capacity retention of 72.91%. Although some decline is expected at higher cycles under 1C operation, the coulombic efficiency remained steady at ∼98%. The drop is expected at a 1C rate. More importantly, the coulombic efficiency stays close to ∼98%, suggesting the reactions are stable and reversible, with minimal side reactions. This ultimately shows a good long-term stability of the electrolyte material.
Subsequently, the cell's performance was evaluated at a 2C rate, delivering an initial discharge capacity of 119.57 mAh g−1. Remarkably, the capacity remained stable throughout the 300 cycles (113.87 mAh g−1), with 95.85% coulombic efficiency and 95.23% capacity retention (Fig. 10d).
The fluctuations in charge/discharge capacity at higher cycles (2C) are likely caused by changes at the interface between the electrode and electrolyte, especially at higher current rates. These small variations are typical at high rates and don't necessarily mean the material contains impurities or forms unwanted byproducts. The coulombic efficiency remains high throughout, showing the battery continues to operate reliably.66 Overall, both figures show that the electrolyte material performs well over long-term cycling and handles high charge/discharge rates effectively. The changes observed are common in practical systems and do not indicate any major issues with the GPIL membrane.
To the best of our knowledge, no prior studies have reported on PADADMA and PVDF-HFP-based gel polymer electrolytes, which demonstrate high ionic conductivity and excellent cycling performance at room temperature (Table SI2).11,34,51,63 In a nutshell, the proposed GPE has great potential in high-voltage safe lithium batteries because of its low flammability characteristics, high ionic conductivity, adequate flexibility, and excellent electrochemical stability.
5. Conclusion
The present work demonstrates the successful development of a GPE membrane with outstanding electrochemical performance. The optimized GPE composition achieved notable ionic conductivities of 1.48 × 10−3 S cm−1 at 25 °C and 3.17 × 10−3 S cm−1 at 65 °C, confirming its capability to support effective lithium-ion transport. Structural modification through the incorporation of LiTFSI into the semi-crystalline poly (DADMATFSI) matrix enhanced its amorphous characteristics, which play a vital role in improving ion mobility, reducing interfacial resistance, and boosting overall electrochemical behaviour. Morphological analysis confirmed a layered and porous structure as well as an extended electrochemical voltage window up to 4.8 V with a handsome transference number of 0.75, highlighting its potential for practical application in high-voltage LIBs. The half-cell further demonstrated consistent discharge capacities across varying current densities, validating the strong rate capability of the material. Overall, these results underscore the potential of this GPE system as a viable and effective component for next-generation high-voltage LIBs, offering a promising path toward safe, efficient, and sustainable energy storage technologies.Author contributions
A. P.: investigation; methodology; writing – original draft, U. C. & M. C.: formal analysis; validation. B. K & M. K.: conceptualization; supervision; writing – review & editing.Conflicts of interest
There are no conflicts to declare.Abbreviations
| LIBs | Lithium-ion batteries |
| GPE | Gel polymer electrolyte |
| LiTFSI | Lithium bis(trifluoromethanesulfonyl)imide |
| PDADMATFSI | Poly(diallyldimethylammonium)bis(trifluoromethanesulfonyl)imide |
| PIL | Poly ionic liquid |
Data availability
The data supporting the findings of this study are included in the manuscript.The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: 1H NMR spectrum of poly(diallyl dimethylammonium)bis(trifluoromethanesulfonic)imide (PDADMATFSI) in acetone D6 (Fig. SI1), ionic conductivity of prepared membrane (Fig. SI2), impedance study of GPIL-3 membrane at a different temperature (Table SI1), XRD peak fitting image of (a) PIL, (b) GPIL-3 (Fig. SI3), flame retardant test GPIL-3 membrane (a and b) (Fig. SI5), stress–strain study of GPIL-3 membrane (Fig. SI4), the comparative literature data on the reported system (Table SI2). See DOI: https://doi.org/10.1039/d5ta06822c.
Acknowledgements
The authors are thankful for the financial support from Dr Vishwanath Karad, MIT World Peace University. BK and MK acknowledge the facility of the Centre of Excellence for rechargeable batteries at C-MET Pune. AP is also thankful to Nanomaterials & Nanocomposite/Battery Laboratory, C-MET, Pune, India.References
- Z. Yang, J. Wang, C. Cui, Y. Jin, G. Zhang, H. Zhou and W. Qian, J. Power Sources, 2022, 524, 230977 CrossRef CAS.
- C. Wang, C. Yang and Z. Zheng, Adv. Sci., 2022, 9, 2105213 CrossRef CAS.
- A. Manthiram, X. Yu and S. Wang, Nat. Rev. Mater., 2017, 2, 16103 CrossRef CAS.
- Q. Zhao, S. Stalin, C.-Z. Zhao and L. A. Archer, Nat. Rev. Mater., 2020, 5, 229–252 CrossRef CAS.
- L. Fan, S. Wei, S. Li, Q. Li and Y. Lu, Adv. Energy Mater., 2018, 8, 1702657 CrossRef.
- Y. An, X. Han, Y. Liu, A. Azhar, J. Na, A. K. Nanjundan, S. Wang, J. Yu and Y. Yamauchi, Small, 2022, 18, 2103617 CrossRef CAS.
- L. Long, S. Wang, M. Xiao and Y. Meng, J. Mater. Chem. A, 2016, 4, 10038–10069 RSC.
- F. Gambino, M. Gastaldi, A. Jouhara, S. Malburet, S. Galliano, N. Cavallini, G. Colucci, M. Zanetti, A. Fina, G. A. Elia and C. Gerbaldi, J. Power Sources Adv., 2024, 30, 100160 CrossRef CAS.
- W. Zhou, Z. Wang, Y. Pu, Y. Li, S. Xin, X. Li, J. Chen and J. B. Goodenough, Adv. Mater., 2019, 31, 1805574 CrossRef PubMed.
- Z. Ruan, Y. Du, H. Pan, R. Zhang, F. Zhang, H. Tang and H. Zhang, Polymers, 2022, 14(10), 1950 CrossRef CAS PubMed.
- X. Hu, R. Muchakayala, S. Song, J. Wang, J. Chen and M. Tan, Int. J. Hydrogen Energy, 2018, 43, 3741–3749 CrossRef.
- K. Aruchamy, S. Ramasundaram, S. Divya, M. Chandran, K. Yun and T. H. Oh, Gels, 2023, 9, 585 CrossRef PubMed.
- D. T. Hallinan and N. P. Balsara, Annu. Rev. Mater. Res., 2013, 43, 503–525 CrossRef.
- N. N. M. Radzir, S. A. Hanifah, A. Ahmad, N. H. Hassan and F. Bella, J. Solid State Electrochem., 2015, 19, 3079–3085 CrossRef.
- I. Osada, H. de Vries, B. Scrosati and S. Passerini, Angew. Chem., Int. Ed., 2016, 55, 500–513 CrossRef PubMed.
- X. Wang, H. Zhu, G. M. A. Girard, R. Yunis, D. R. MacFarlane, D. Mecerreyes, A. J. Bhattacharyya, P. C. Howlett and M. Forsyth, J. Mater. Chem. A, 2017, 5, 23844–23852 RSC.
- X. Zhou, Y. Zhou, L. Yu, L. Qi, K.-S. Oh, P. Hu, S.-Y. Lee and C. Chen, Chem. Soc. Rev., 2024, 53, 5291–5337 RSC.
- A. M. Voice, G. R. Davies and I. M. Ward, Polym. Gels Networks, 1997, 5, 123–144 CrossRef.
- T. Kataoka, Y. Ishioka, M. Mizuhata, H. Minami and T. Maruyama, ACS Appl. Mater. Interfaces, 2015, 7, 23346–23352 CrossRef.
- J. Feng, L. Wang, Y. Chen, P. Wang, H. Zhang and X. He, Nano Convergence, 2021, 8, 1–12 CrossRef PubMed.
- K. Daems, P. Yadav, K. Dermenci, J. Van Mierlo and M. Berecibar, Renewable Sustainable Energy Rev., 2024, 191, 114136 CrossRef.
- T. Liu, Z. Chang, Y. Yin, K. Chen, Y. Zhang and X. Zhang, Solid State Ionics, 2018, 318, 88–94 CrossRef.
- Y. Li, W. Zhang, Q. Dou, K. W. Wong and K. M. Ng, J. Mater. Chem. A, 2019, 7, 3391–3398 RSC.
- W. Zhang, J. Nie, F. Li, Z. L. Wang and C. Sun, Nano Energy, 2018, 45, 413–419 CrossRef.
- J. Li, L. Zhu, J. Zhang, M. Jing, S. Yao, X. Shen, S. Li and F. Tu, Int. J. Energy Res., 2021, 45, 7663–7674 CrossRef.
- Y. Wu, Y. Li, Y. Wang, Q. Liu, Q. Chen and M. Chen, J. Energy Chem., 2022, 64, 62–84 CrossRef CAS.
- G. A. Tiruye, D. Muñoz-Torrero, J. Palma, M. Anderson and R. Marcilla, J. Power Sources, 2016, 326, 560–568 CrossRef CAS.
- D. Zhou, R. Liu, J. Zhang, X. Qi, Y.-B. He, B. Li, Q.-H. Yang, Y.-S. Hu and F. Kang, Nano Energy, 2017, 33, 45–54 CrossRef CAS.
- G. A. Tiruye, D. Munoz-Torrero, J. Palma, M. Anderson and R. Marcilla, J. Power Sources, 2015, 279, 472–480 CrossRef.
- G. Appetecchi, G.-T. Kim, M. Montanino, M. Carewska, R. Marcilla, D. Mecerreyes and I. De Meatza, J. Power Sources, 2010, 195, 3668–3675 CrossRef CAS.
- M. Safa, A. Chamaani, N. Chawla and B. El-Zahab, Electrochim. Acta, 2016, 213, 587–593 CrossRef CAS.
- M. Baloch, A. Vizintin, R. K. Chellappan, J. Moskon, D. Shanmukaraj, R. Dedryvère, T. Rojo and R. Dominko, J. Electrochem. Soc., 2016, 163, A2390 CrossRef CAS.
- A. Eftekhari and T. Saito, Eur. Polym. J., 2017, 90, 245–272 CrossRef CAS.
- X. Wang, F. Chen, G. M. Girard, H. Zhu, D. R. MacFarlane, D. Mecerreyes, M. Armand, P. C. Howlett and M. Forsyth, Joule, 2019, 3, 2687–2702 CrossRef CAS.
- K. Abraham, Z. Jiang and B. Carroll, Chem. Mater., 1997, 9, 1978–1988 CrossRef CAS.
- X. Hu, R. Muchakayala, S. Song, J. Wang, J. Chen and M. Tan, Int. J. Hydrogen Energy, 2018, 43, 3741–3749 CrossRef CAS.
- T. G. Paiva, M. Klem, S. L. Silvestre, J. Coelho, N. Alves, E. Fortunato, E. J. Cabrita and M. C. Corvo, ChemSusChem, 2025, 18, e202401710 CrossRef CAS.
- T. G. Paiva, M. Klem, S. L. Silvestre, J. Coelho, N. Alves, E. Fortunato, E. J. Cabrita and M. C. Corvo, ChemSusChem, 2024, e202401710 Search PubMed.
- S. M. George, S. Sampath and A. J. Bhattacharyya, Batteries Supercaps, 2022, 5, e202200252 CrossRef CAS.
- B. M. Wiers, M. L. Foo, N. P. Balsara and J. R. Long, J. Am. Chem. Soc., 2011, 133, 14522–14525 CrossRef CAS PubMed.
- D. Saikia and A. Kumar, Electrochim. Acta, 2004, 49, 2581–2589 CrossRef CAS.
- J. Wilson, G. Ravi and M. A. Kulandainathan, Polímeros, 2006, 16, 88–93 CrossRef CAS.
- S. Abbrent, J. Plestil, D. Hlavata, J. Lindgren, J. Tegenfeldt and Å. Wendsjö, Polymer, 2001, 42, 1407–1416 CrossRef CAS.
- D. Balzar, J. Res. Natl. Inst. Stand. Technol., 1993, 98, 321 CrossRef CAS.
- B. Rao, N. Maramu, E. Rao, N. Rao and K. R. Prasad, Res. Rev.: J. Phys., 2018, 2, 1–4 Search PubMed.
- J. D. Kamminga and L. J. Seijbel, J. Res. Natl. Inst. Stand. Technol., 2004, 109, 65–74 CrossRef.
- C. F. Holder and R. E. Schaak, ACS Nano, 2019, 13(7), 7359–7365 CrossRef CAS PubMed.
- P. L. Stigliano, A. Gallastegui, C. Villacis-Segovia, M. Amores, A. Kumar, L. A. O'Dell, J. Fang, D. Mecerreyes, C. Pozo-Gonzalo and M. Forsyth, Batteries, 2024, 10, 125 CrossRef CAS.
- M. Celik, A. Kızılaslan, M. Can, T. Cetinkaya and H. Akbulut, Electrochim. Acta, 2021, 371, 137824 CrossRef CAS.
- S. Das and A. Ghosh, J. Appl. Phys., 2016, 119, 095101 CrossRef.
- S. Kondou, M. Abdullah, I. Popov, M. L. Martins, L. A. O'Dell, H. Ueda, F. Makhlooghiazad, A. Nakanishi, T. Sudoh and K. Ueno, J. Am. Chem. Soc., 2024, 146, 33169–33178 CrossRef CAS.
- Z. Zhao, F. Li, J. Zhao, G. Ding, J. Wang, X. Du, Q. Zhou, G. Hou and G. Cui, Adv. Funct. Mater., 2020, 30, 2000347 CrossRef CAS.
- J. Pitawala, A. Martinelli, P. Johansson, P. Jacobsson and A. Matic, J. Non-Cryst. Solids, 2015, 407, 318–323 CrossRef CAS.
- S. Malunavar, A. Gallastegui, X. Wang, F. Makhlooghiazad, D. Mecerreyes, M. Armand, M. Galceran, P. C. Howlett and M. Forsyth, ACS Appl. Polym. Mater., 2022, 4, 8977–8986 CrossRef CAS.
- M. Brinkkötter, E. I. Lozinskaya, D. O. Ponkratov, P. S. Vlasov, M. P. Rosenwinkel, I. A. Malyshkina, Y. Vygodskii, A. S. Shaplov and M. Schönhoff, Electrochim. Acta, 2017, 237, 237–247 CrossRef.
- L. F. Malmonge, J. A. Malmonge and W. K. Sakamoto, Mater. Res., 2003, 6, 469–473 CrossRef CAS.
- Z. Li, J. Fu, X. Zhou, S. Gui, L. Wei, H. Yang, H. Li and X. Guo, Adv. Sci., 2023, 10, 2201718 CrossRef CAS.
- V. K. Singh and R. K. Singh, J. Mater. Chem. C, 2015, 3, 7305–7318 RSC.
- L. Yu, L. Yu, Q. Liu, T. Meng, S. Wang and X. Hu, Adv. Funct. Mater., 2022, 32, 2110653 CrossRef CAS.
- S. Caimi, H. Wu and M. Morbidelli, ACS Appl. Energy Mater., 2018, 1, 5224–5232 CAS.
- T. K. L. Nguyen and T.-N. Pham-Truong, Polymers, 2024, 16, 2506 CrossRef CAS.
- N. Karaman, M. Aliefendić, S. Pljuco, D. Kozlica, N. Nalić, F. Korać and S. Gutić, Bull. Chem. Technol. Bosnia Herzegovina, 2015, 45, 19–22 CAS.
- X. Song, H. Zhang, D. Jiang, L. Yang, J. Zhang, M. Yao, X. Ji, G. Wang and S. Zhang, Electrochim. Acta, 2021, 368, 137581 CrossRef CAS.
- S. Menkin, C. A. O'Keefe, A. B. Gunnarsdóttir, S. Dey, F. M. Pesci, Z. Shen, A. Aguadero and C. P. Grey, J. Phys. Chem. C., 2021, 125, 16719–16732 CrossRef CAS.
- Z. Katcharava, A. Marinow and W. H. Binder, Chem. Commun., 2025, 61, 3250–3270 RSC.
- Q. Wang, Z. A. Grady, C. R. Bowen and J. I. Roscow, J. Materiomics, 2025, 11, 100912 CrossRef.
| This journal is © The Royal Society of Chemistry 2025 |