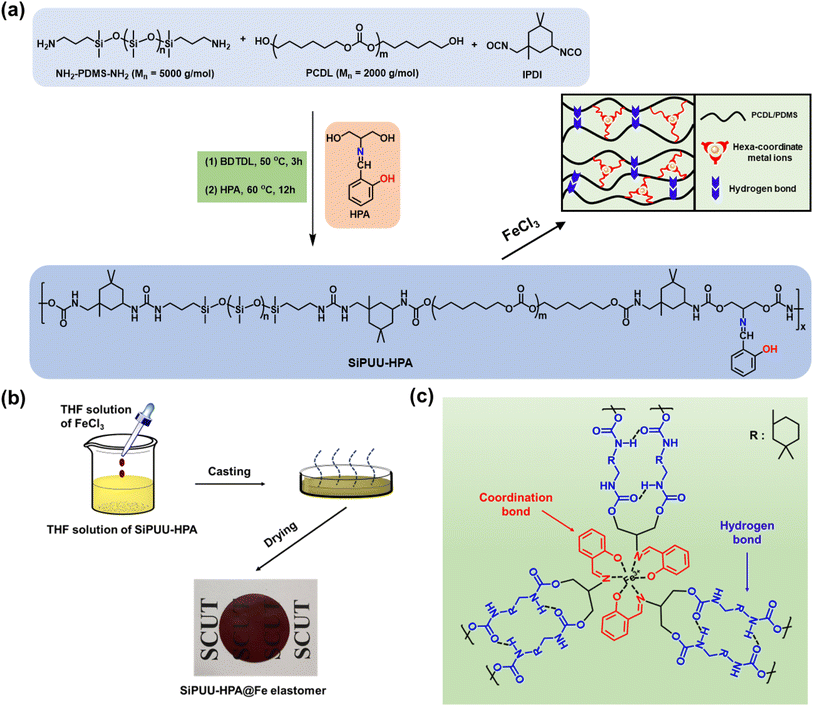
Multifunctional polyurethane elastomers with high mechanical robustness and exceptional crack tolerance performance based on bi-incompatible soft segments and dynamic coordination bonds
Xionghui
Wu
a,
Shuting
Chen
a,
Yaping
Sheng
a,
Anqiang
Zhang
 *a and
Yaling
Lin
*b
*a and
Yaling
Lin
*b
aSchool of Materials Science and Engineering, South China University of Technology, 381 Wushan Rd., Guangzhou 510641, Guangdong, China. E-mail: aqzhang@scut.edu.cn; linyaling@scau.edu.cn
bCollege of Materials and Energy, South China Agricultural University, 483 Wushan Rd., Guangzhou 510642, Guangdong, China
First published on 15th September 2025
Abstract
Elastomers with high strength and super toughness are widely used in aerospace, flexible electronics, biomedical, and other fields. However, the development of healable and recyclable elastomers with high strength, high toughness, and excellent crack resistance remains a significant challenge. In this work, we successfully synthesized a multifunctional silicone-based elastomer (SiPUU-HPA@Fe) with outstanding mechanical properties, self-healing capabilities and recyclability by combining biocompatible polycarbonate (PCDL) segments, flexible polydimethylsiloxane (PDMS) segments and metal coordination bonds. Owing to the presence of phase separation structures and dynamic coordination interaction, the optimized SiPUU-HPA2@Fe1/3 elastomer exhibits a high tensile strength of 45.5 MPa, an ultrahigh toughness of 412.5 MJ m−3, and an exceptional fracture energy of 179.3 kJ m−2. Additionally, the SiPUU-HPA2@Fe1/3 elastomers can be healed and recycled to regain their original mechanical strength and integrity under heating. Furthermore, the SiPUU-HPA2@Fe1/3 elastomer demonstrates excellent antibacterial properties and no cytotoxic effects. Finally, a soft SiPUU-HPA2@Fe1/3/Li based strain sensor was developed by combining SiPUU-HPA2@Fe1/3 with conductive ionic lithium bis(trifluoromethane)sulfonimide (LiTFSI) and demonstrated remarkable sensing capability to diverse human body motions. This research provides ideas for the design of polyurethane elastomers with high mechanical properties and multiple functions, and the elastomers are expected to be used in emerging fields such as biomedical and flexible electronics.
1 Introduction
In recent years, thermoplastic polyurethane (TPU) has been widely applied in soft robotics, biomedical materials, and flexible electronics due to its good biocompatibility, easy processing ability and tunable mechanical properties.1–10 Polysiloxane elastomers are an ideal substrate for the preparation of flexible electronic and biomedical devices, owing to their excellent biocompatibility, remarkable flexibility, high gas permeability, and exceptional thermal and oxidative stability.11–15 However, the non-polar and weak intermolecular interactions of PDMS lead to poor mechanical strength in silicone-based elastomers.16,17 At present, enhancing the strength, toughness and crack resistance of silicone-based elastomers remains a huge challenge that significantly limits their application range and service life. In addition, endowing elastomers with self-healing ability and recyclability can not only increase their service life but also reduce waste generation and environmental pollution.18–22 Therefore, the development of elastomers that exhibit high mechanical strength, toughness, exceptional crack resistance, recyclability, and self-healing properties is essential for building a sustainable society.Although developing a self-healing PDMS elastomer that combines high strength and toughness poses significant challenges, researchers have proposed various strategies to address the issue. For example, Wang et al. successfully synthesized a thermoplastic silicone-based elastomer by integrating the synergistic effects between bi-incompatible soft segments and multi-scale hydrogen bonds.19 The resulting silicone-based elastomer exhibited a tensile strength of 8.0 MPa, an elongation at break of 1910%, a high toughness of 67.8 MJ m−3, and an exceptional fracture energy of 31.8 kJ m−2. Sun et al. developed a healable and recyclable polyurethane elastomer with high strength (52.4 MPa), good toughness (363.8 MJ m−3) and exceptional fracture energy (192.9 kJ m−2) by incorporating dynamic hierarchical domains into a polymer matrix.23 Furthermore, the elastomers can be efficiently healed and recycled to regain their original mechanical properties and integrity, owing to the reversibility of the dynamic hierarchical domains and hydrogen and coordination bonds. Recently, Wang et al. synthesized a multifunctional polyurethane elastomer by employing PCDL and PDMS as soft segments and incorporating dynamic boron–oxygen and dynamic disulfide bonding into the polymer chain segments.24 This innovative design endowed the elastomer with outstanding mechanical properties, such as a tensile strength of 16.3 MPa, a remarkable elongation at break of 3300%, a toughness value of 278.8 MJ m−3, and a fracture energy of 234.9 kJ m−2. Therefore, the development of silicone-based elastomers with high mechanical strength and toughness, excellent tear resistance, self-healing and recyclability through rational molecular structure design remains a significant challenge.
In this work, a multifunctional polyurethane elastomer is developed through the introduction of two thermodynamically incompatible components, i.e., poly(dimethylsiloxane) (PDMS) and polycarbonate (PCDL), and metal coordination bonds. Owing to the synergistic effect of phase separation and hydrogen and coordination bonds, the optimized SiPUU-HPA2@Fe1/3 elastomer demonstrates exceptional mechanical properties including a tensile strength of 45.5 MPa, an ultrahigh toughness of 412.5 MJ m−3, and an exceptional fracture energy of 179.3 kJ m−2. Meanwhile, based on the reversibility and dynamic characteristics of hydrogen and coordination bonds, the SiPUU-HPA2@Fe1/3 elastomer can be healed and recycled to regain its original mechanical strength and integrity. Moreover, the PU elastomer exhibited good biocompatible and antibacterial properties, demonstrating its great potential for biomedical applications. Finally, its potential in flexible electronic sensors was demonstrated by combining the SiPUU-HPA2@Fe1/3 elastomer and lithium bis(trifluoromethane)sulfonimide (LiTFSI).
2 Experimental
2.1. Materials
Octamethylcyclotetrasiloxane (D4, > 99.5%), 1,3-bis(3-aminopropyl)-1,1,3,3-tetramethyldisiloxane (AMM, 97%), tetramethylammonium hydroxide pentahydrate (TMAH, 97%), 2-amino-1,3-propanediol (98%), salicylaldehyde (98%), isophorone diisocyanate (IPDI, 99%), lithium bis(trifluoromethane)sulfonimide (LiTFSI, 99%) and dibutyltin dilaurate (DBTDL) were obtained from Shanghai Macklin, China. Tetrahydrofuran (THF, 99.5%) and N,N-dimethylformamide (DMF, 99.5%) were obtained from Shanghai Macklin and dried using 3 Å molecular sieves before use. Polycarbonate diol (PCDL) with a molecular weight of 2000 g mol−1 was obtained from Shandong Liduo Chemical Co., Ltd (Shandong, China) and was evaporated under reduced pressure at 110 °C for 3 h before use. Escherichia coli (E. coli), Staphylococcus albus (S. albus) and mouse fibroblast cells (L929) were provided by South China Agricultural University.2.2. Synthesis of SiPUU-HPA@Fe elastomers
FTIR (ATR, cm−1): 3330 (–NH), 2962 (–CH3), 1640 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in urea), 1694 (C
O in urea), 1694 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in urethane), 1552 (N–H), 1731 (C
O in urethane), 1552 (N–H), 1731 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O in PCDL), 1625 (C
O in PCDL), 1625 (C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N in HPA), 1247 (C–O in PCDL), 1017 (–Si–O–Si–), 960 (C–O in PCDL), 797 (–Si–CH3).
N in HPA), 1247 (C–O in PCDL), 1017 (–Si–O–Si–), 960 (C–O in PCDL), 797 (–Si–CH3).
1H NMR (600 MHz, CDCl3, δ, ppm): 12.88 (s, Ph–OH in HPA), 8.39 (s, –CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N in HPA), 7.30–7.35 (m, Ph–H in HPA), 6.89–6.95 (m, Ph–H in HPA), 3.68–3.86 (m, –CH2–O in HPA), 3.35–3.49 (m, –NCH(CH2)2 in HPA), 4.05–4.15 (m, –O–CH2–(CH2)4–CH2–O– in PCDL), 1.58–1.75 (m, –O–CH2–CH2–(CH2)2–CH2–CH2–O– in PCDL), 1.24–1.45 (m, –O–(CH2)2–CH2–CH2–(CH2)2–O– in PCDL), −0.05–0.11 (m, –Si(CH3)2 in PDMS), 2.86–2.96 (m, –CH2–N in PDMS), 1.45–1.52 (m, –CH2–CH2–N in PDMS), 0.51 (s, –Si(CH3)2–CH2– in PDMS), 0.84 (s, –C(CH3)2), 1.06 (s, –C–CH3 in IPDI), 2.04 (m, –CH– in IPDI), 2.91 (m, –CH–CH2–N in IPDI).
N in HPA), 7.30–7.35 (m, Ph–H in HPA), 6.89–6.95 (m, Ph–H in HPA), 3.68–3.86 (m, –CH2–O in HPA), 3.35–3.49 (m, –NCH(CH2)2 in HPA), 4.05–4.15 (m, –O–CH2–(CH2)4–CH2–O– in PCDL), 1.58–1.75 (m, –O–CH2–CH2–(CH2)2–CH2–CH2–O– in PCDL), 1.24–1.45 (m, –O–(CH2)2–CH2–CH2–(CH2)2–O– in PCDL), −0.05–0.11 (m, –Si(CH3)2 in PDMS), 2.86–2.96 (m, –CH2–N in PDMS), 1.45–1.52 (m, –CH2–CH2–N in PDMS), 0.51 (s, –Si(CH3)2–CH2– in PDMS), 0.84 (s, –C(CH3)2), 1.06 (s, –C–CH3 in IPDI), 2.04 (m, –CH– in IPDI), 2.91 (m, –CH–CH2–N in IPDI).
2.3. Preparation of the SiPUU-HPA2@Fe1/3/Lix film
SiPUU-HPA2@Fe1/3/Lix was fabricated by mixing SiPUU-HPA2@Fe1/3 and LiTFSI with different ratios, where x represents the ratio of LiTFSI mass to SiPUU-HPA2@Fe1/3 mass. Taking the synthesis of SiPUU-HPA2@Fe1/3/Li80% as an example, a DMF solution of SiPUU-HPA (5.0 g) was mixed with a DMF solution of FeCl3 (the molar ratio of Fe3+ ions to HPA groups is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3). After stirring for 4 h at room temperature, LiTFSI (4.0 g, the mass ratio of LiTFSI to SiPUU-HPA2@Fe1/3 is 80%) was added into the above DMF solution, which was further stirred for another 4 h. Finally, the homogeneous solution was poured into a PTFE mold and then dried at 60 °C for 48 h to obtain the SiPUU-HPA2@Fe1/3/Li80% elastomer.
3). After stirring for 4 h at room temperature, LiTFSI (4.0 g, the mass ratio of LiTFSI to SiPUU-HPA2@Fe1/3 is 80%) was added into the above DMF solution, which was further stirred for another 4 h. Finally, the homogeneous solution was poured into a PTFE mold and then dried at 60 °C for 48 h to obtain the SiPUU-HPA2@Fe1/3/Li80% elastomer.
3 Results and discussion
3.1. Preparation and characterization of the SiPUU-HPA@Fe elastomer
The Schiff-base ligand HPA, which contains imine groups and phenol groups, demonstrates good biocompatibility. When a metal–ligand interaction involves multiple bonds, it forms a chelation bond and is stronger. Under the synergistic coordination of N and O atoms, coordination bonds easily form with various metal ions such as Fe3+ and Zn2+. In this work, focusing on potential biomedicine and wearable device applications, we chose biosafe PDMS and PCDL as soft segments, along with biologically relevant Fe3+ ions.The chemical structure of the HPA monomer was characterized by 1H NMR, as shown in Fig. S1. In the 1H NMR spectrum, the resonance peak at 8.49 ppm was assigned to the imine bond (CH![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N), confirming the successful reaction between the aldehyde of salicylaldehyde and the amino group of 2-amino-1,3-propanediol. In addition, the peak area ratio of the protons in HPA aligned with the theoretical calculations. These results indicate that the HPA monomer is successfully prepared.
N), confirming the successful reaction between the aldehyde of salicylaldehyde and the amino group of 2-amino-1,3-propanediol. In addition, the peak area ratio of the protons in HPA aligned with the theoretical calculations. These results indicate that the HPA monomer is successfully prepared.
The SiPUU-HPA@Fe elastomer is a multiblock polymer composed of PDMS and PCDL cross-linked with hydrogen bonds and coordination bonds between the HPA groups and Fe3+ ions. The synthesis routes and schematic diagram of the SiPUU-HPA@Fe elastomer are shown in Scheme 1. First, isocyanate-terminated PDMS and PCDL prepolymers were synthesized through the reaction of IPDI with a mixture of PCDL and PDMS. Then, the synthesized NCO-PDCL-NCO and NCO-PDMS-NCO were reacted with the HPA monomer to obtain SiPUU-HPA. Finally, the SiPUU-HPA@Fe elastomers were synthesized by adding FeCl3 into the THF solution of SiPUU-HPA under stirring. In terms of GPC analysis, the GPC trace of SiPUU-HPAx elastomers displayed a unimodal peak. Among them, the number-average molecular weight (Mn) of SiPUU-HPA2 was calculated to be 82.5 × 103 g mol−1 with a polydispersity index (PDI) of 1.64 (Fig. S2 and Table S2). As shown in the 1H NMR spectrum of SiPUU-HPA2, the characteristic peaks of the PCDL, PDMS, IPDI and HPA segments in the polymer chain were clearly observed (Fig. S3).
The FTIR and EDS analysis verified the chemical structures and elemental compositions of SiPUU-HPA2@Fe elastomers. As shown in Fig. 1a, the coordination bond between HPA groups and Fe3+ ions in the SiPUU-HPA2 elastomer was confirmed through FTIR analysis. In the FTIR spectrum of SiPUU-HPA2, the peak at 1625 cm−1 is attributed to the stretching vibration of C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N in the HPA groups. This peak shifts to 1592 cm−1 in the FTIR spectrum of SiPUU-HPA2@Fe1/3, indicating the formation of coordination bonds between the Fe3+ ions and the HPA groups. The EDS mapping results showed that Si, O and Fe were uniformly distributed in the SiPUU-HPA2@Fe1/3 elastomer (Fig. 1b), indicating the homogeneous structure of the elastomer. This is beneficial for the formation of dynamic metal coordination between Fe3+ ions and HPA groups in the cross-linked network. Furthermore, the formation of coordination between Fe3+ ions and HPA groups was confirmed by XPS analysis. As shown in Fig. 1c, the Fe 2p XPS spectrum presented two signals corresponding to Fe 2p1/2 and Fe 2p3/2 at 725.6 and 710.8 eV, respectively.25,26 In addition, Fe signals were also found in the XPS fitting of N 1s and O 1s, which are located at 399.6 eV and 532.3 eV, respectively (Fig. 1d and e).27,28 These results demonstrate that the SiPUU-HPA2@Fe elastomer was successfully synthesized containing dynamic coordination bonds and hydrogen bonds. In addition, the thermal stability of the SiPUU-HPA2@Fe1/3 elastomer was characterized by TGA tests, as shown in Fig. S4. The Td, 5% values of SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers are 295 °C and 282 °C, respectively, indicating excellent thermal resistance properties. The residual carbon content of the SiPUU-HPA2@Fe1/3 elastomer is higher than that of the SiPUU-HPA2 elastomer, which may be attributed to the formation of Fe-containing compounds during the thermal degradation process.
N in the HPA groups. This peak shifts to 1592 cm−1 in the FTIR spectrum of SiPUU-HPA2@Fe1/3, indicating the formation of coordination bonds between the Fe3+ ions and the HPA groups. The EDS mapping results showed that Si, O and Fe were uniformly distributed in the SiPUU-HPA2@Fe1/3 elastomer (Fig. 1b), indicating the homogeneous structure of the elastomer. This is beneficial for the formation of dynamic metal coordination between Fe3+ ions and HPA groups in the cross-linked network. Furthermore, the formation of coordination between Fe3+ ions and HPA groups was confirmed by XPS analysis. As shown in Fig. 1c, the Fe 2p XPS spectrum presented two signals corresponding to Fe 2p1/2 and Fe 2p3/2 at 725.6 and 710.8 eV, respectively.25,26 In addition, Fe signals were also found in the XPS fitting of N 1s and O 1s, which are located at 399.6 eV and 532.3 eV, respectively (Fig. 1d and e).27,28 These results demonstrate that the SiPUU-HPA2@Fe elastomer was successfully synthesized containing dynamic coordination bonds and hydrogen bonds. In addition, the thermal stability of the SiPUU-HPA2@Fe1/3 elastomer was characterized by TGA tests, as shown in Fig. S4. The Td, 5% values of SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers are 295 °C and 282 °C, respectively, indicating excellent thermal resistance properties. The residual carbon content of the SiPUU-HPA2@Fe1/3 elastomer is higher than that of the SiPUU-HPA2 elastomer, which may be attributed to the formation of Fe-containing compounds during the thermal degradation process.
3.2. Mechanical performance of the SiPUU-HPA@Fe elastomer
The mechanical properties of SiPUU-HPAx elastomers were evaluated through a universal tensile test at a stretching speed of 100 mm min−1. As shown in Fig. S5, the tensile strength of the SiPUU-HPAx elastomers increased, whereas their elongation at break decreased as the HPA content increased. Among them, the SiPUU-HPA2 elastomer had a tensile strength of 9.2 MPa and an elongation at break of 1489%. This finding can be attributed to the increase in the number of hydrogen bonds and physical crosslinking points between polymer chains as the HPA content increases. Consequently, the tensile strength of the SiPUU-HPAx elastomers increased. However, an excess of physical crosslinking points limited the slippage of polymer chains, leading to a gradual decrease in the elongation at break of the elastomers.The excellent mechanical properties of the SiPUU-HPA2@Fex elastomers were evaluated via a uniaxial tensile test at a stretching speed of 100 mm min−1 at room temperature. The stress–strain curves of the SiPUU-HPA2@Fex elastomers are shown in Fig. 2a, while the mechanical properties are summarized in Table S3. All SiPUU-HPA2@Fex elastomers are typical elastomers because no yielding phenomena during elongation were observed. When Fe3+ ions were introduced, the mechanical strength of the SiPUU-HPA2@Fex elastomers gradually increased, while the elongation at break gradually decreased. This result demonstrates that the introduction of coordination interactions between Fe3+ ions and HPA groups increases the cross-linking density, significantly enhancing the mechanical properties of SiPUU-HPA2@Fex elastomers.
 | ||
| Fig. 2 (a) Typical stress–strain curve of the SiPUU-HPA2@Fex elastomers; (b) tensile strength and toughness histograms of the SiPUU-HPA2@Fex elastomers; (c) comparison of toughness of SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers; (d) comparison of the tensile strength and toughness of SiPUU-HPA2@Fe1/3 with data from other elastomers.4,22,29–37 (e) Schematic of the mechanism of enhanced mechanical properties of the SiPUU-HPA2@Fe elastomer. | ||
When the molar ratio of Fe3+ ions to HPA is 1![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3, the resulting SiPUU-HPA2@Fe1/3 elastomer exhibits the highest tensile strength and toughness, which are 45.5 MPa and 412.5 MJ m−3, respectively (Fig. 2b). In contrast, the tensile strength of SiPUU-HPA2 without Fe3+ ions was only 9.2 MPa and 58.1 MJ m−3, respectively (Fig. 2b). This is attributed to the addition of Fe3+ ions, which increases the cross-linking density and limits the mobility of polymer chains, thereby improving the mechanical strength of the SiPUU-HPA2@Fex elastomer. Furthermore, the toughness of the SiPUU-HPA2@Fe1/3 elastomer is 7 times greater than that of the SiPUU-HPA2 elastomer (58.1 MJ m−3) (Fig. 2c). To the best of our knowledge, the SiPUU-HPA2@Fe1/3 elastomer exhibits exceptional strength and toughness, surpassing those of the majority of polyurethane elastomers currently reported (Fig. 2d).4,22,29–37
3, the resulting SiPUU-HPA2@Fe1/3 elastomer exhibits the highest tensile strength and toughness, which are 45.5 MPa and 412.5 MJ m−3, respectively (Fig. 2b). In contrast, the tensile strength of SiPUU-HPA2 without Fe3+ ions was only 9.2 MPa and 58.1 MJ m−3, respectively (Fig. 2b). This is attributed to the addition of Fe3+ ions, which increases the cross-linking density and limits the mobility of polymer chains, thereby improving the mechanical strength of the SiPUU-HPA2@Fex elastomer. Furthermore, the toughness of the SiPUU-HPA2@Fe1/3 elastomer is 7 times greater than that of the SiPUU-HPA2 elastomer (58.1 MJ m−3) (Fig. 2c). To the best of our knowledge, the SiPUU-HPA2@Fe1/3 elastomer exhibits exceptional strength and toughness, surpassing those of the majority of polyurethane elastomers currently reported (Fig. 2d).4,22,29–37
To evaluate energy dissipation capability of SiPUU-HPA2@Fe elastomers, cyclic tensile tests were further performed. The cyclic tensile curves of the SiPUU-HPA2@Fe1/3 elastomer under different strains are depicted in Fig. 3a. The hysteresis loop gradually increased with increasing strain. As illustrated in Fig. 3b, the hysteresis areas at different strains were plotted as a function of the strain. The hysteresis area value represents the energy dissipation value. When the strain is below 300%, the hysteresis areas of the elastomer are small and the slope of the curve remains essentially constant. Within this range, elastic deformation mainly occurs, with an extremely small amount of hydrogen bonds and coordination bonds breaking. The hysteresis area rapidly increases when the strain exceeds 300%, indicating that energy can be effectively dissipated to enhance the toughness and crack tolerance of the elastomer. As illustrated in Fig. 2e, when the elastomer was stretched to small strains, the hydrogen bonds with lower bonding energy dissociated first. As the strain increased further, the coordination bond between Fe3+ and HPA groups, which has higher bonding energy, gradually dissociated.
To evaluate the elastic behavior of the SiPUU-HPA2@Fe1/3 elastomer, the cyclic tensile tests were conducted at a strain of 500%, as shown in Fig. 3c. The first cyclic curve of SiPUU-HPA2@Fe1/3 exhibited a significant hysteresis loop (with 11.58 MJ m−3 dissipated energy), indicating that significant energy dissipation occurred attributed to the rupture of the dynamic crosslinking interactions within the polymer chains. As the number of cycles increased, the energy dissipation area gradually decreased, primarily because the dynamic hydrogen bonding and coordination bond interactions could not be quickly and fully restored to their original states. With the extension of waiting time, the cyclic tensile curves and hysteresis area gradually recovered and nearly reached the original state after a waiting time of 120 min. These results indicate that the SiPUU-HPA2@Fe1/3 elastomer exhibits excellent elastic restorability, significantly improving the material's fatigue resistance under deformation. To evaluate the fatigue resistance of the SiPUU-HPA2@Fe1/3 elastomer, a cyclic tensile test was further conducted. The results of the 500 continuous cyclic stretching tests at a strain of 100% are shown in Fig. 3d. As the number of cycles increases, the maximum force tends to be stable, indicating that hydrogen bonds and coordination bonds have achieved a dynamic balance between breakage and rapid association. These results demonstrate that the SiPUU-HPA2@Fe1/3 elastomer has excellent fatigue resistance.
The presence of phase separation structures and strong dynamic coordination interactions could also effectively improve the crack tolerance of the elastomer. Crack tolerance is regarded as a critical mechanical property parameter for elastomers. The crack tolerance of the SiPUU-HPA2@Fe elastomer was evaluated through notch stretching experiments. As shown in Fig. 4a, the notched SiPUU-HPA2@Fe1/3 elastomer can be stretched up to 11 times its original length, without rapid lateral propagation of the notch. This phenomenon could be primarily attributed to the dynamic dissociation and reassociation of hydrogen bonds and coordination bonds within the hard microdomains, resulting in a stick-slip motion of the polymer chains under external force. Consequently, the stress concentration at the notch tip is efficiently transmitted and dispersed into the entire polymer network, thereby effectively resisting the lateral propagation of the notch. Using the Greensmith method from the stress–strain curves, the fracture energy was calculated to be 179.3 kJ m−2, which is 18 times greater than that of natural rubber (approximately 10 kJ m−2). Additionally, the notched SiPUU-HPA2@Fe1/3 elastomer exhibited a fracture strength of 13.7 MPa. In contrast, the fracture energy of the SiPUU-HPA elastomer was only 31.1 kJ m−2, significantly lower than the values of the SiPUU-HPA2@Fe1/3 elastomer (Fig. 4b). Compared to most previously reported polyurethane elastomers, the SiPUU-HPA2@Fe1/3 elastomer demonstrated exceptional advantages in fracture energy (Fig. 4c).6,29,34,38–44 The above findings indicate that the elastomer exhibited excellent tear resistance, ensuring its safety in practical applications.
 | ||
| Fig. 4 (a) Notched tensile test curves of the SiPUU-HPA2@Fe1/3 elastomer. The inset images show the single-edge-notched elastomer at 1000% strain. (b) Comparison of fracture energy of SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers. (c) Comparison of the fracture energy of the SiPUU-HPA2@Fe1/3 elastomer with those of elastomers reported in the literature.6,29,34,38–44 | ||
3.3. Microstructure and thermal properties of the SiPUU-HPA2@Fe elastomer
To evaluate the relationship between the structure and properties of the SiPUU-HPA2@Fe elastomer, the microstructure of the elastomer was characterized using XRD, DSC, SAXS and AFM. The crystallization behavior of the SiPUU-HPA2@Fe1/3 elastomer was evaluated by XRD and DSC. As shown in Fig. 5b, the SiPUU-HPA2 and SiPUU-HPA2@Fe elastomers display two distinct diffraction peaks at about 12° and 20°, corresponding to the PCDL and random PDMS blocks, respectively, suggesting an amorphous structure. According to the DSC spectrum analysis, the glass transition temperatures (Tg, PCDL) of the SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers are −31.6 °C and −30.4 °C, respectively (Fig. 5a). Compared to SiPUU-HPA2, the Tg, PCDL of the SiPUU-HPA2@Fe1/3 elastomer decreased after coordination of Fe3+ ions, which could be attributed to the enhanced microphase separation. The introduction of Fe3+ ions affected the arrangement of polymer molecules, increasing the aggregation degree of the hard segment and consequently reducing the dispersion between hard and soft segments.45,46 | ||
| Fig. 5 (a) DSC curves, (b) XRD curves and (c) SAXS patterns of SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers. (d) AFM image of the SiPUU-HPA2@Fe1/3 elastomer. | ||
Furthermore, the microphase separation structure of the SiPUU-HPA2@Fe1/3 elastomer was investigated by SAXS. In the SAXS spectrum, the SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers exhibit a broad scattering peak (Fig. 5c), suggesting a microphase separation structure. The q value of the SiPUU-HPA2 elastomer is 0.153 nm−1, and the periodicity (D) of the phase-separated domains was calculated to be 26.8 nm. After coordination with Fe3+ ions, the q value of the SiPUU-HPA2@Fe1/3 elastomer increases, while the periodicity of the phase-separated domains decreases from 41 nm to 32 nm. These results demonstrate that the phase separation becomes more evident and the distance between hard segments becomes smaller after coordination.47 In brief, the incorporation of coordination bonds in hard segments draws the benzene rings closer to form more hydrogen bonds between adjacent carbamate bonds and modulates the microscopic phase structure of the elastomers. The phase separation of the elastomer was further analyzed using AFM. As shown in Fig. 5d and S6, both the SiPUU-HPA2 elastomer and the SiPUU-HPA2@Fe1/3 elastomer exhibited a two-phase structure, where the bright regions refer to hard domains and the dark regions refer to soft domains. After coordination with Fe3+ ions, the phase separation became more pronounced, because the interface between the dark and bright regions in the SiPUU-HPA2@Fe1/3 elastomer was more distinct compared to that in the SiPUU-HPA2 elastomer.
In conclusion, the remarkable mechanical properties of the SiPUU-HPA2@Fe elastomer could be attributed to several factors: (1) the microphase separation domain functioned as a rigid nanofiller; (2) the coordination interactions between Fe3+ ions and HPA groups increased the energy barrier for chain separation from the domain; (3) the dynamic dissociation and recombination behavior of coordination bonds and hydrogen bonds significantly improved energy dissipation.
3.4. Healability and recyclability of the SiPUU-HPA2@Fe elastomer
The viscoelastic properties of the SiPUU-HPA2@Fe1/3 elastomer at different temperatures were evaluated using rheological analysis. Fig. 6a and b illustrate the temperature-dependent oscillation rheology curve of SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers across a temperature range from 20 °C to 180 °C. As shown in Fig. 6a, when the temperature was raised to approximately 162 °C, the storage modulus (G′) intersected with the loss modulus (G′′), indicating that the viscous flow transition temperature of the SiPUU-HPA2 elastomer was 162 °C. This phenomenon was further investigated in terms of frequency dependency. By applying small-amplitude oscillatory shear at different temperatures ranging from 20 °C to 120 °C, we constructed a master curve (reference temperature = 100 °C) for the SiPUU-HPA2 elastomer, demonstrating the frequency dependency of G′ and G′′, as depicted in Fig. S7. A cross-point of G′ and G′′ at a critical shear frequency (ωc) was observed for the SiPUU-HPA2 elastomer, aligning with the results from temperature scanning. After the addition of Fe3+, the G′ of the SiPUU-HPA2@Fe1/3 elastomer decreases at a relatively slow rate and remains within a consistent order of magnitude as the temperature increases from 20 to 150 °C (Fig. 6b), demonstrating the excellent thermomechanical stability of the SiPUU-HPA2@Fe1/3 elastomer. When the temperature exceeds 150 °C, the G′ and G′′ decrease rapidly (Fig. 6b), indicating that a large number of the dynamic bonds in the elastomer are broken.48 However, the SiPUU-HPA2@Fe1/3 elastomer remained in a solid state up to 180 °C because its G′ is always higher than its G′′ in the tested temperature range. Rheological frequency sweep curves of SiPUU-HPA2@Fe1/3 were also measured at different temperatures. As shown in Fig. 6c, the rheological tests demonstrate that the G′ of the SiPUU-HPA2@Fe1/3 elastomer is higher than its G′′ in the entire frequency spectrum. The stress-relaxation experiments further demonstrated the dynamic viscoelasticity of the SiPUU-HPA2@Fe1/3 elastomer, as depicted in Fig. 6d. After coordination with Fe3+ ions, the relaxation time of the SiPUU-HPA2@Fe1/3 elastomer increases at room temperature. This result indicates that the formation of metal coordination bonds enhances the interactions between molecular chains and reduces the efficiency of chain movement.Polymer materials generate a considerable amount of waste after use or damage, resulting in significant resource waste and long-term environmental pollution. Therefore, the SiPUU-HPA2@Fe elastomers were designed to have a self-healing ability and recyclability. Taking SiPUU-HPA2@Fe1/3 as an example, the healing efficiency was studied at different temperatures and times. As shown in Fig. 7a, the healing efficiency of the SiPUU-HPA2@Fe1/3 elastomer gradually increases with the healing temperature or time. After being subjected to a temperature of 100 °C for 24 h, the tensile strength and elongation at break of the elastomer showed a self-healing efficiency of 88.3% and 95.7%, respectively (Fig. 7b). Moreover, Fig. 7a demonstrates that reducing the temperature significantly decreases the healing efficiency. The storage modulus (G′) of the SiPUU-HPA2@Fe1/3 elastomer at 25 °C and 100 °C is 0.54 MPa and 0.07 MPa, respectively (Fig. 6b). This finding indicates that the mobility of the polymer chains significantly increases when heated to 100 °C, which is attributed to the dynamic breaking of hydrogen bonds and coordination bonds and the high mobility of PDMS soft segments.23
Furthermore, the SiPUU-HPA2@Fe1/3 elastomer also demonstrated excellent recyclability due to the dynamic reversibility of metal coordination bonds and hydrogen bonds. As shown in Fig. 7c, the SiPUU-HPA2@Fe1/3 elastomer can be recovered by a hot-pressing process at 100 °C under a pressure of 10 MPa for 0.5 h. After three cycles of recycling, the stress–strain curves of the recycled SiPUU-HPA2@Fe1/3 elastomer nearly overlap with those of the original one (Fig. 7d), demonstrating its excellent recyclability and reprocessing performance. In summary, the material can maintain high mechanical performance even after three recycling processes, significantly reducing waste generation and consequently dissolving resource waste and environmental pollution.
3.5. Antibacterial properties and biocompatibility evaluation
Enhancing polyurethane materials with antimicrobial properties is essential in the practical application of medical devices to protect them from bacterial contamination and erosion. The antibacterial properties of the SiPUU-HPA2@Fe1/3 film were evaluated using representative Gram-negative Escherichia coli (E. coli) and Gram-positive Staphylococcus albus (S. albus). As shown in Fig. S8, the SiPUU-HPA2@Fe1/3 film exhibited excellent antibacterial performance, achieving antibacterial efficiencies of 97.82% and 99.99% against E. coli and S. albus, respectively. The Schiff-base ligand HPA contains imine groups and phenol groups, providing synergistic coordination of N and O atoms toward Fe3+ ions. The antibacterial mechanism of the SiPUU-HPA2@Fe1/3 elastomer can be summarized as follows: (1) after Fe3+ coordinates with the imine and phenolic hydroxyl groups of HPA, it disrupts the bacterial film structure and functions, hinders bacterial metabolism, and affects the metabolic processes, ultimately leading to bacterial apoptosis and exerting a bactericidal effect.49 (2) Schiff base compounds, known for their remarkable antibacterial activity, interact with the phospholipid bilayer in bacterial cell membranes, causing disruption of membrane integrity and ultimately leading to bacterial death.50 In summary, the SiPUU-HPA2@Fe1/3 film exhibits excellent antibacterial properties, attributed to the synergistic effects of Schiff base and metal–ligand bonds.The hemocompatibility of the SiPUU-HPA2@Fe1/3 film was evaluated by in vitro hemolysis testing. As illustrated in Fig. 8c and S9, compared to both negative and positive control groups, the SiPUU-HPA2@Fe1/3 film extracts with different immersion durations did not cause significant erythrocyte rupture, and their hemolysis rate was less than 2%, which is far lower than the standard requirement level of 5%. These findings demonstrate that SiPUU-HPA2@Fe1/3 exhibits excellent blood compatibility under in vitro conditions. Furthermore, the cytocompatibility of the SiPUU-HPA2@Fe1/3 film was evaluated using MTT assay and live/dead staining. The fluorescence images of L929 cells cultured in a dish containing SiPUU-HPA2@Fe1/3 leachate for 24 h and 72 h are displayed in Fig. 8a. It was observed that there was no significant difference in cell density between the control group and the experimental group, suggesting that SiPUU-HPA2@Fe1/3 does not have a significant effect on cell proliferation. The MTT assay results depicted in Fig. 8b demonstrate that the cell survival rate exceeded 85% during both 1-day and 3-day culture periods, demonstrating that the synthesized SiPUU-HPA2@Fe1/3 film exhibits low cytotoxicity. These results indicate that SiPUU-HPA2@Fe1/3 demonstrates excellent cytocompatibility and good biocompatibility and is expected to be used in the biomedical field.
3.6. Application in strain sensors
Solid-state ionic conductive elastomers (SICEs) have effectively addressed the liquid leakage issues associated with gel-based ionic conductors due to their solid-state structure and exceptional environmental stability, making them key materials in flexible electronics. Ionic conductive polyurethane has advantages in terms of strength and toughness due to hydrogen bonds and microphase separation. However, achieving a balance between the mechanical properties and ionic conductivity of SICEs remains a significant challenge. In this work, a solid-state ionic conductive elastomer with good mechanical properties and high ionic conductivity was successfully prepared by introducing lithium salts into a SiPUU-HPA2@Fe1/3 elastomer utilizing metal–ligand coordination and hydrogen bonding interaction.The mechanical properties of SiPUU-HPA2@Fe1/3/Lix elastomers were evaluated through universal tensile experiments. As shown in Fig. S10, the tensile strength of SiPUU-HPA2@Fe1/3/Lix elastomers decreased sharply with increasing LiTFSI content, which is attributed to the destruction of the polyurethane microstructure caused by the introduction of LiTFSI. In particular, the tensile strength of the SiPUU-HPA2@Fe1/3/Li80% elastomer reached 4.3 MPa when the lithium salt was 80%. However, the Young's modulus of SiPUU-HPA2@Fe1/3/Li80% exhibited a significant reduction compared with that of SiPUU-HPA2@Fe1/3 (Fig. 9a), suggesting that the incorporation of ionic liquid imparted enhanced flexibility to the elastomer, thereby rendering it more suitable as a substrate for flexible sensing applications. This property is particularly beneficial for skin-mounted sensing, as the softened material demonstrates improved conformability to the inherent softness of human skin. Subsequently, we investigated the ionic conductivity of the SiPUU-HPA2@Fe1/3/Lix elastomer. As shown in Fig. S11, the amount of freely moving Li ions increased and the ionic conductivity of the films increased with increasing LiTFSI content. Among them, the SiPUU-HPA2@Fe1/3/Li80% film had an ionic conductivity of 0.24 mS cm−1. This could be attributed to the addition of LiTFSI, which enhanced the mobility of the chain segments to some extent, facilitating Li transport and thereby improving ionic conductivity of the elastomers. In summary, SiPUU-HPA2@Fe1/3/Li80% exhibited excellent mechanical properties and electrical conductivity, making it an ideal material for assembling flexible sensors.
The gauge factor (GF) value is a critical parameter for evaluating the sensitivity of a strain sensor. The slope of the fitting curve in Fig. 9b reveals that the GF of the SiPUU-HPA2@Fe1/3/Li80% strain sensor was 1.72 within the 0–300% strain range, demonstrating its broad detection range and stable sensitivity. Meanwhile, the response and relaxation times of the SiPUU-HPA2@Fe1/3/Li80% sensor were measured at 0.38 s and 0.31 s, respectively, demonstrating the rapid response behavior of the sensor (Fig. 9c). Owing to the outstanding sensing performance of SiPUU-HPA2@Fe1/3/Li across a wide strain range, the sensor can be used as a wearable soft sensor for monitoring human joint motion. The sensor was attached to a finger, and the ΔR/R0 signals gradually changed as the finger bent, indicating that it can accurately recognize the degree of finger bending (Fig. 9d). Moreover, the sensor was also used to monitor the signals of knee bending. Fig. 9e shows the changes in ΔR/R0 signals as the straightened knee gradually bent. The ΔR/R0 signal increased with the increasing bending angle of the knee. When the knee was straightened again, the ΔR/R0 signal returned to its original value. After being exposed to 25 °C for one month, the sensor maintains a consistent relative resistance change, demonstrating a long-term environmental stability (Fig. 9f). Additionally, we further investigated the sensing performance of SiPUU-HPA2@Fe1/3/Li80% at low temperatures. It was observed that the sensing performance exhibited no significant changes at −20 °C, as shown in Fig. 9f. In summary, SiPUU-HPA2@Fe1/3/Li enables real-time monitoring of ΔR/R0 signal variations, highlighting its potential for applications in flexible electronics.
4 Conclusions
In summary, we have successfully developed a thermoplastic silicone-based elastomer with outstanding mechanical performance, self-healing properties and recyclability by combining bi-incompatible soft segments and metal coordination bonds into the polymer chain segments. The presence of phase separation structures and strong dynamic coordination interactions significantly enhances its outstanding mechanical properties. The synthesized SiPUU-HPA2@Fe1/3 elastomer demonstrated a high tensile strength of 45.5 MPa, an ultrahigh toughness of 412.5 MJ m−3, and an exceptional fracture energy of 179.3 kJ m−2. Additionally, the SiPUU-HPA2@Fe1/3 elastomers can be healed and recycled to regain their original mechanical strength and integrity due to the reversibility of hydrogen and coordination bonds. Furthermore, the SiPUU-HPA2@Fe1/3 elastomer exhibits excellent antibacterial properties and biocompatibility, demonstrating its significant potential for use in biomedical applications. Finally, a strain sensor was developed by combining SiPUU-HPA2@Fe1/3 with conductive ionic lithium bis(trifluoromethane)sulfonimide (LiTFSI), demonstrating its potential applications in wearable electronics. This study provides ideas for the design of multifunctional polyurethane elastomers and broadens the application of high-performance polyurethane elastomers in biomedical and flexible electronic fields.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Author contributions
This manuscript was written through contributions of all authors. All authors have given approval to the final version of the manuscript.Data availability
The data supporting this article have been included as part of the SI. Supplementary information: experimental part, including the synthesis and characterization of HPA and NH2-PDMS-NH2, characterization of SiPUU-HPA and SiPUU-HPA2@Fe1/3 elastomers, antibacterial and hemolysis performance of SiPUU-HPA2 and SiPUU-HPA2@Fe1/3 elastomers, and mechanical properties and ionic conductivity of SiPUU-HPA2@Fe1/3/Li elastomers. See DOI: https://doi.org/10.1039/d5ta06699a.Acknowledgements
The authors acknowledge the financial support from the National Natural Science Foundation of China (52073098), the Natural Science Foundation of Guangdong Province (2023A1515011264) and the Open Fund for Key Lab of Guangdong High Property and Functional Macromolecular Materials, China (20240010).References
- M. W. M. Tan, G. Thangavel and P. S. Lee, Rugged soft robots using tough, stretchable, and self-healable adhesive elastomers, Adv. Funct. Mater., 2021, 31, 2103097, DOI:10.1002/adfm.202103097.
- K. Liu, M. Wang, C. Huang, Y. Yuan, Y. Ning, L. Zhang and P. Wan, Flexible bioinspired healable antibacterial electronics for intelligent human-machine interaction sensing, Adv. Sci., 2024, 11, 2305672, DOI:10.1002/advs.202305672.
- Y. Lin, N. Yu, S. Shan and A. Zhang, Synergistic enhancement of the robustness of multifunctional polyurethane via an ionic noncovalent cross-linking network and aromatic disulfides, Chem. Eng. J., 2024, 481, 148229, DOI:10.1016/j.cej.2023.148229.
- X. Guo, J. Liang, Z. Wang, J. Qin, Q. Zhang, S. Zhu, K. Zhang and H. Zhu, Tough, recyclable, and degradable elastomers for potential biomedical applications, Adv. Mater., 2023, 35, 2210092, DOI:10.1002/adma.202210092.
- Y. Xie, X. Shi, S. Gao, C. Lai, C. Lu, Y. Huang, D. Zhang, S. Nie, F. Xu and F. Chu, Biomimicking natural wood to fabricate isotropically super-strong, tough, and transparent hydrogels for strain sensor and triboelectric nanogenerator applications, J. Mater. Chem. A, 2024, 12, 5124–5132, 10.1039/D3TA08065J.
- X. Wu, Q. Tang, Y. Hu, Y. Lin and A. Zhang, Healable and recyclable supramolecular poly(urethane-urea) elastomers with high mechanical robustness, low hysteresis, fatigue resistance and antibacterial based on dynamic double crosslinked networks, Polymer, 2025, 334, 128745, DOI:10.1016/j.polymer.2025.128745.
- M. Khatib, O. Zohar, W. Saliba, S. Srebnik and H. Haick, Highly efficient and water-insensitive self-healing elastomer for wet and underwater electronics, Adv. Funct. Mater., 2020, 30, 1910196, DOI:10.1002/adfm.201910196.
- C. Jiang, L. Zhang, Q. Yang, S. Huang, H. Shi, Q. Long, B. Qian, Z. Liu, Q. Guan, M. Liu, R. Yang, Q. Zhao, Z. You and X. Ye, Self-healing polyurethane-elastomer with mechanical tunability for multiple biomedical applications in vivo, Nat. Commun., 2021, 12, 4395, DOI:10.1038/s41467-021-24680-x.
- X. Nie, Y. Xie, X. Ding, L. Dai, F. Gao, W. Song, X. Li, P. Liu, Z. Tan, H. Shi, C. Lai, D. Zhang and Y. Lai, Highly elastic, fatigue-resistant, antibacterial, conductive, and nanocellulose-enhanced hydrogels with selenium nanoparticles loading as strain sensors, Carbohydr. Polym., 2024, 334, 122068, DOI:10.1016/j.carbpol.2024.122068.
- N. Yu, Y. Meng, R. Li, D. Mai, S. Shan, X. Wu, Y. Lin and A. Zhang, Simultaneously enhancing the mechanical robustness and conductivity of ionogels by in situ formation of coordination complexes as physical crosslinks, J. Mater. Chem. A, 2024, 12, 12134–12145, 10.1039/D4TA01001A.
- A. Turco, E. Primiceri, M. Frigione, G. Maruccio and C. Malitesta, An innovative, fast and facile soft-template approach for the fabrication of porous PDMS for oil–water separation, J. Mater. Chem. A, 2017, 5, 23785–23793, 10.1039/C7TA06840A.
- D. Qi, K. Zhang, G. Tian, B. Jiang and Y. Huang, Stretchable electronics based on PDMS substrates, Adv. Mater., 2021, 33, 2003155, DOI:10.1002/adma.202003155.
- X. Wu, Y. Hu, Y. Xia, Y. Lin and A. Zhang, Siloxane-based segmented poly(urethane-urea) elastomers with enhanced mechanical properties, hydrophobicity and anti-calcification based on hierarchical phase separation for potential applications of polymeric heart valve, Eur. Polym. J., 2024, 218, 113349, DOI:10.1016/j.eurpolymj.2024.113349.
- Z. Lin, H. Deng, Y. Hou, X. Liu, R. Xu, H. Xiang, Z. Peng, M. Rong and M. Zhang, Dual-crosslinking side chains with an asymmetric chain structure: a facile pathway to a robust, self-healable, and re-dissolvable polysiloxane elastomer for recyclable flexible devices, J. Mater. Chem. A, 2022, 10, 11019–11029, 10.1039/D2TA01535H.
- T. Liu, C. Li, H. Yao, F. Sun, L. Wang, B. Yao, J. Xu and J. Fu, Extremely strengthening fatigue resistance, elastic restorability and thermodynamic stability of a soft transparent self-healing network based on a dynamic molecular confinement-induced bioinspired nanostructure, Mater. Horiz., 2023, 10, 2968–2979, 10.1039/D3MH00358B.
- H. Chen, J. J. Koh, M. Liu, P. Li, X. Fan, S. Liu, J. C. C. Yeo, Y. Tan, B. C. K. Tee and C. He, Super tough and self-healable poly(dimethylsiloxane) elastomer via hydrogen bonding association and its applications as triboelectric nanogenerators, ACS Appl. Mater. Interfaces, 2020, 12, 31975–31983, DOI:10.1021/acsami.0c08213.
- F. Wang, C. Zhang, Y. Han, G. Yi, Z. Mao, J. Li, Z. Li and W. Wang, A hybrid reversible crosslinked polysiloxane elastomer with high toughness and recyclability, J. Polym. Sci., 2024, 62, 2147–2156, DOI:10.1002/pol.20240019.
- X. Wang, Y. Li, Y. Qian, H. Qi, J. Li and J. Sun, Mechanically robust atomic oxygen-resistant coatings capable of autonomously healing damage in low earth orbit space environment, Adv. Mater., 2018, 30, 1803854, DOI:10.1002/adma.201803854.
- W. Zheng, C. Zhang, Y. Han, W. Wang and Z. Li, Highly durable silicone-based elastomers achieved through the synergy of bi-incompatible soft segments and multi-scale hydrogen bonds, Small, 2024, 20, 2402124, DOI:10.1002/smll.202402124.
- X. Wu, X. Xiong, Y. Hu, Q. Tang, Y. Lin and A. Zhang, Healable and recyclable poly(urethane-urea) elastomers with high mechanical strength, extreme toughness, and excellent crack tolerance via a supramolecular self-assembly strategy for strain sensor application, Polymer, 2025, 318, 127994, DOI:10.1016/j.polymer.2024.127994.
- Y. Yanagisawa, Y. Nan, K. Okuro and T. Aida, Mechanically robust, readily repairable polymers via tailored noncovalent cross-linking, Science, 2018, 359, 72–76, DOI:10.1126/science.aam7588.
- X. Xiang, L. Zhang, D. Sheng, X. Yang, X. Qi, S. Wei and H. Dai, Healable and recyclable polyurea–urethane elastomer with high mechanical robustness, superhigh elastic restorability, and exceptional crack tolerance, Adv. Funct. Mater., 2024, 34, 2312571, DOI:10.1002/adfm.202312571.
- X. Wang, S. Zhan, Z. Lu, J. Li, X. Yang, Y. Qiao, Y. Men and J. Sun, Healable, recyclable, and mechanically tough polyurethane elastomers with exceptional damage tolerance, Adv. Mater., 2020, 32, 2005759, DOI:10.1002/adma.202005759.
- H. Tian, W. Lu, C. Wang, R. Wang, P. Zhou, F. Fei, M. Xu and J. Wang, Development of highly robust polyurethane elastomers possessing self-healing capabilities for flexible sensors, Mater. Horiz., 2025, 12, 3949–3964, 10.1039/D5MH00022J.
- Y. Chen, P. G. Miller, X. Ding, C. E. Stowell, K. M. Kelly and Y. Wang, Chelation crosslinking of biodegradable elastomers, Adv. Mater., 2020, 32, 2003761, DOI:10.1002/adma.202003761.
- L. Zhang, Q. Guan, A. Shen, R. E. Neisiany, Z. You and M. Zhu, A topological polymer network with Cu(II)-coordinated reversible imidazole-urea locked unit constructs an ultra-strong self-healing elastomer, Sci. China: Chem., 2022, 65, 363–372, DOI:10.1007/s11426-022-1437-5.
- W. Xu, H. Liu, J. He, B. Bao, J. Jiang, H. Ren, B. Zeng, C. Yuan and L. Dai, Fabrication of multifunction polyurethane with robust, self-healing and anti-corrosion performance based multiple dynamic bonds strategy, Prog. Org. Coat., 2024, 192, 108513, DOI:10.1016/j.porgcoat.2024.108513.
- Y. Sheng, M. Wang, K. Zhang, Z. Wu, Y. Chen and X. Lu, An “inner soft external hard”, scratch-resistant, self-healing waterborne poly(urethane-urea) coating based on gradient metal coordination structure, Chem. Eng. J., 2021, 426, 131883, DOI:10.1016/j.cej.2021.131883.
- M. Zhang, Y. Wang, M. Yang, Y. Deng, W. Zhou, H. Wang, X. Jian and Y. Chen, Fabrication of mechanical strong supramolecular waterborne polyurethane elastomers with the inspiration of hierarchical dynamic structures of scallop byssal threads, Adv. Funct. Mater., 2025, 35, 2413083, DOI:10.1002/adfm.202413083.
- Y. Yao, B. Liu, Z. Xu, J. Yang and W. Liu, An unparalleled H-bonding and ion-bonding crosslinked waterborne polyurethane with super toughness and unprecedented fracture energy, Mater. Horiz., 2021, 8, 2742–2749, 10.1039/D1MH01217G.
- Y. Xu, S. Zhou, Z. Wu, X. Yang, N. Li, Z. Qin and T. Jiao, Room-temperature self-healing and recyclable polyurethane elastomers with high strength and superior robustness based on dynamic double-crosslinked structure, Chem. Eng. J., 2023, 466, 143179, DOI:10.1016/j.cej.2023.143179.
- X. Chen, Q. Zhong, C. Cui, L. Ma, S. Liu, Q. Zhang, Y. Wu, L. An, Y. Cheng, S. Ye, X. Chen, Z. Dong, Q. Chen and Y. Zhang, Extremely tough, puncture-resistant, transparent, and photoluminescent polyurethane elastomers for crack self-diagnose and healing tracking, ACS Appl. Mater. Interfaces, 2020, 12, 30847–30855, DOI:10.1021/acsami.0c07727.
- X. Guo, J. Wang, L. Chen, Z. Wang, Y. Zhang and L. Fang, Liquefied chitin-derived super tough, sustainable, and anti-bacterial polyurethane elastomers, Chem. Eng. J., 2023, 465, 143074, DOI:10.1016/j.cej.2023.143074.
- Y. Li, W. Li, A. Sun, M. Jing, X. Liu, L. Wei, K. Wu and Q. Fu, A self-reinforcing and self-healing elastomer with high strength, unprecedented toughness and room-temperature reparability, Mater. Horiz., 2021, 8, 267–275, 10.1039/D0MH01447H.
- Z. Ping, F. Xie, X. Gong, F. Zhang, J. Zheng, Y. Liu and J. Leng, Tailoring photoweldable shape memory polyurethane with intrinsic photothermal/fluorescence via engineering metal-phenolic systems, Adv. Funct. Mater., 2024, 34, 2402592, DOI:10.1002/adfm.202402592.
- Q. Zhong, X. Chen, Y. Yang, C. Cui, L. Ma, Z. Li, Q. Zhang, X. Chen, Y. Cheng and Y. Zhang, Hydrogen bond reinforced, transparent polycaprolactone-based degradable polyurethane, Mater. Chem. Front., 2021, 5, 5371–5381, 10.1039/D1QM00476J.
- Z. Wu, J. Dong, H. Guo, R. Shang, X. Qin, Y. Xia, X. Li, X. Zhao, C. Ji and Q. Zhang, Robust, self-healing, and multi-use poly(urethane-urea-imide) elastomer as a durable adhesive for thermal interface materials, Small, 2024, 20, 2401815, DOI:10.1002/smll.202401815.
- Y. Qian, F. Dong, S. Wang, Y. Jiang, X. Xu and H. Liu, Ultrarobust, stretchable, and highly elastic supramolecular elastomer with hydrogen-bond interactions via sp2 hybridized boron-urethane bonds, Angew. Chem., 2025, 137, e202421099, DOI:10.1002/anie.202421099.
- D. Wang, J. Xu, J. Chen, P. Hu, Y. Wang, W. Jiang and J. Fu, Transparent, mechanically strong, extremely tough, self-recoverable, healable supramolecular elastomers facilely fabricated via dynamic hard domains design for multifunctional applications, Adv. Funct. Mater., 2020, 30, 1907109, DOI:10.1002/adfm.201907109.
- X. Liu, X. Liu, W. Li, Y. Ru, Y. Li, A. Sun and L. Wei, Engineered self-healable elastomer with giant strength and toughness via phase regulation and mechano-responsive self-reinforcing, Chem. Eng. J., 2021, 410, 128300, DOI:10.1016/j.cej.2020.128300.
- X. Wang, J. Xu, X. Zhang, Z. Yang, Y. Zhang, T. Wang and Q. Wang, Molecularly engineered unparalleled strength and supertoughness of poly(urea-urethane) with shape memory and clusterization-triggered emission, Adv. Mater., 2022, 34, 2205763, DOI:10.1002/adma.202205763.
- H. Wu, H. Wang, M. Luo, Z. Yuan, Y. Chen, B. Jin, W. Wu, B. Ye, H. Zhang and J. Wu, Mechanically robust, self-reporting and healable polyurethane elastomers by incorporating symmetric/asymmetric chain extenders, Mater. Horiz., 2024, 11, 1548–1559, 10.1039/D3MH01987J.
- J. Shi, T. Zheng, Z. Wang, P. Wang, H. Yang, J. Guo, D. Wang, B. Guo and J. Xu, Filler effects inspired high performance polyurethane elastomer design: segment arrangement control, Mater. Horiz., 2024, 11, 4747–4758, 10.1039/D4MH00648H.
- Y. Han, K. Xi, C. Zhang, W. Wang and Z. Li, Tough silicone-based elastomers enhanced by synergistic Zn(ii)-carboxylate interactions and weak hydrogen bonds between incompatible soft segments, J. Mater. Chem. A, 2025, 13, 13305–13315, 10.1039/D5TA01214G.
- J. Dou, P. Li, Y. Zhao, L. Zhou, X. Li, J. Wang and N. Huang, Copper-mediated polyurethane materials with enzyme-like catalysis for biocompatibility improvement in blood environments, Biosurface and Biotribology, 2021, 7, 30–41, DOI:10.1049/bsb2.12009.
- A. Shen, H. Xuan, Y. Jia, S. Gu, R. E. Neisiany, W. Shu, W. Sun and Z. You, Dynamic healing-assembly for biocompatible, biodegradable, stretchable and self-healing triboelectric nanogenerators, Chem. Eng. J., 2024, 491, 151896, DOI:10.1016/j.cej.2024.151896.
- Y. Hou, H. Liu, Y. Peng, J. Zhang, G. Huang, Q. Wu and J. Wu, A fast self-healing and mechanical-enhanced polyurethane via Cu-pyridine coordination, Polymer, 2022, 257, 125266, DOI:10.1016/j.polymer.2022.125266.
- X. Wang, Y. L. Wang, X. Yang, Z. Lu, Y. Men and J. Sun, Skin-inspired healable conductive elastomers with exceptional strain-adaptive stiffening and damage tolerance, Macromolecules, 2021, 54, 10767–10775, DOI:10.1021/acs.macromol.1c01976.
- Y. Li, Z. Sun, S. Chai, Z. Zhang, Y. Li, C. Zhu and D. Fan, Tissue-like soft dual-network hydrogel patch with instant/tunable adhesion and microenvironment modulation for infected wound healing, ACS Nano, 2025, 19, 27714–27730, DOI:10.1021/acsnano.5c07573.
- S. Liu, R. Chen, Q. Liu, J. Liu, J. Zhu, G. Sun, H. Zhang and J. Wang, Schiff base modified polyurethane coating with enhanced mechanical property for marine antifouling application, Prog. Org. Coat., 2025, 203, 109138, DOI:10.1016/j.porgcoat.2025.109138.
| This journal is © The Royal Society of Chemistry 2025 |