The role of alkali doping in enhancing bulk and interface properties of Cu2CdSnS4 solar cells
Edwin
Julianto
 a,
Ha Kyung
Park
a,
Ha Kyung
Park
 b,
Ahmad
Ibrahim
b,
Ahmad
Ibrahim
 a,
Stener
Lie
a,
Stener
Lie
 a,
You Jung
Choi
c,
Geumha
Lim
c,
Mufti Ali
Ar-Royan
a,
Anupam
Sadhu
a,
You Jung
Choi
c,
Geumha
Lim
c,
Mufti Ali
Ar-Royan
a,
Anupam
Sadhu
 a,
Akhmad Herman
Yuwono
a,
Akhmad Herman
Yuwono
 d,
William
Jo
d,
William
Jo
 bc and
Lydia Helena
Wong
bc and
Lydia Helena
Wong
 *ae
*ae
aEnergy Research Institute @ NTU (ERI@N), Nanyang Technological University, 637553, Singapore. E-mail: lydiawong@ntu.edu.sg
bNew and Renewable Energy Research Center, Ewha Womans University, 03760, Republic of Korea
cDepartment of Physics, Ewha Womans University, 03760, Republic of Korea
dDepartment of Metallurgical and Materials Engineering, Faculty of Engineering, Universitas Indonesia, Kampus Baru UI, Depok 16424, Indonesia
eSchool of Materials Science and Engineering, Nanyang Technological University, 639798, Singapore
First published on 16th September 2025
Abstract
Alkali doping has been widely employed to enhance the performance of chalcopyrite and kesterite solar cells; however, its potential for Cu2CdSnS4 (CCTS) solar cells remains unexplored. This study systematically investigates the impact of all alkali dopants (Li, Na, K, Rb, Cs) on CCTS solar cells, providing new insights into their interaction with the CCTS structure and its subsequent impact on optoelectronic properties. Alkali doping increases carrier density by an order of magnitude without introducing detrimental recombination centres, as highlighted by the stable minority carrier lifetime. Kelvin probe force microscopy (KPFM) reveals a reduction in upward band bending at grain boundaries, minimizing majority carrier accumulation and enhancing carrier transport. Conductive-atomic force microscopy (c-AFM) further demonstrates an enhancement in intragrain conductivity, with nanoscale surface current increasing by an order of magnitude. Among the doped samples, Na-doped CCTS achieves the highest efficiency of 8.47%, attributed to its compact film morphology and improved charge transport, which collectively yield a higher fill factor and JSC. These findings establish alkali doping as a promising strategy for optimizing CCTS solar cells, with Na emerging as the most effective dopant to enhance device performance.
Introduction
Cu2CdSnS4 (CCTS) is one of the promising absorber materials for emerging thin film solar cells, inspired by the full substitution of Zn in kesterite Cu2ZnSnS4 (CZTS) with Cd. CCTS offers several advantages over CZTS. First, the Cd2+ ion in CCTS is tetrahedrally coordinated with an ionic radius of approximately 78 pm, which is about 25% larger than the ionic radius of Zn2+ (≈60 pm).1 This results in a stannite structure for CCTS,2 which reduces the probability of Cu–Zn antisite formation. Secondly, the formation energy of the deep electron trap defect cluster 2CuCd + SnCd in CCTS (≈0.44–0.72 eV) is higher than the corresponding defect in CZTS (2CuZn + SnZn, ≈0.28–0.67 eV). Furthermore, CCTS exhibits a lower formation energy for beneficial Cu-vacancy (VCu) defects (≈0.10–0.66 eV) compared to CZTS (≈0.17–0.74 eV),3 which suggests fewer detrimental defects in CCTS. Lastly, the bandgap of CCTS is reported to be 1.4 eV,4 closer to the ideal bandgap of 1.34 eV,5 which corresponds to the Shockley–Queisser efficiency limit for a single p–n junction, in contrast to the 1.5–1.6 eV bandgap of CZTS.6,7Recent studies have reported CCTS solar cell efficiencies ranging from 6.87% to 10.1%, demonstrating its potential for high performance.3,8,9 However, these values remain significantly below the theoretical limits of 28.3% based on the spectroscopic limited maximum efficiency (SLME) and 33.8% based on the Shockley–Queisser (SQ) limit,10 highlighting a discrepancy between experimental and theoretical performance that necessitates further optimization. Beyond the superior intrinsic defect properties of CCTS, both bulk optoelectronic and interface properties also play significant roles in achieving high-efficiency solar cells. Alkali doping has been extensively utilized to enhance both bulk and interface properties in chalcopyrite and kesterite solar cells, particularly in Cu(In,Ga)Se2 (CIGS) and Cu2ZnSn(S,Se)4 (CZTSSe). In CIGS, alkali incorporation has been pivotal in achieving record-high efficiencies. Alkali doping, typically introduced via post-deposition treatments (PDT) such as NaF, KF, RbF, and CsF, has been instrumental in optimizing the electronic and structural properties of CIGS absorbers. The impact of alkali was first noted by Hedstrom et al., who observed unintentional sodium (Na) diffusion from soda-lime glass improving open circuit voltage (VOC) and fill factor (FF).11 Subsequent PDT efforts have driven efficiency gains through various mechanism: NaF, RbF, and CsF PDT increase carrier density and intragrain conductivity by an order of magnitude.12 Heavier alkalis like K, Rb and Cs segregate at grain boundaries (GBs), forming (Alk)InSe2 compounds (e.g. RbInSe2) that passivate defects, suppress recombination and widen surface band gaps.13,14 This is also shown by recent advancements with RbF-PDT, reducing deep defects and enhancing p-type conductivity, resulting in a certified efficiency record of 23.64%.15 Similarly, CsF-PDT mitigates donor- and acceptor-like defects (e.g., VSe, InCu),16,17 while KF inhibits interface recombination.18 These tailored effects, varying with alkali size and treatment conditions, underscore their indispensable contribution to pushing CIGS toward its theoretical efficiency limits.
Similarly, in CZTSSe, alkali doping has been proven to improve bulk and interface properties, including its role in increasing hole concentrations (up to 1015 to 1020 cm−3), enhancing intragrain conductivity, and improving band bending.19–23 Extensive research, particularly on Na doping,24–28 highlights its critical role in promoting grain growth and decreasing recombination losses—often through unintentional diffusion from soda-lime glass substrates—contributing to efficiencies up to 12.51%.29 Li and K doping have also demonstrated effects in enhancing acceptor density and morphology,30 with Li, particularly when co-doped with Ag, enabling significant improvements in CZTSSe, achieving efficiencies of 14.91% efficiency and high fill factor of 74.30%.31 Heavier elements like Rb and Cs exhibit less pronounced effects but still contribute to increased acceptor concentrations.30
Despite the important roles of alkali doping in advancing the performance in CIGS and CZTSSe, its impact on the bulk and interface properties of CCTS remains largely unexplored. To date, only Ag doping has been reported to enhance bulk properties in CCTS, leading to improved device efficiency.8 Previous studies investigating a complete set of alkali dopants in CZTSSe have provided valuable insights into the varying effects of different alkali elements on bulk properties and device performance.30,32–35 Therefore, a systematic study of the effect of various alkali dopants on CCTS under consistent fabrication parameters is deemed essential to accelerate progress in the emerging kesterite-inspired materials.
This study systematically investigates the impact of a complete set of alkali dopants on the bulk and interface properties of CCTS solar cells. Alkali doping is observed to enhance bulk properties by increasing carrier density, while simultaneously improving carrier transport at the interface by reducing band bending and increasing intragrain surface current. Notably, Na-doped CCTS exhibits superior film morphology, which further enhances fill factor, resulting in the highest device efficiency of 8.47% among the alkali doped samples. These findings provide key insights into the role of alkali doping in optimizing stannite-based solar cells.
Experimental
Precursor solution preparation
CCTS precursors are dissolved in dimethyl sulfoxide (≥99.9%, Sigma-Aldrich). The precursors are added individually until each is completely dissolved in sequence starting with thiourea (≥99.0%, Sigma-Aldrich), followed by SnCl2·2H2O (98%, Sigma-Aldrich), then followed by CuCl2·2H2O (≥99.95%, Sigma-Aldrich), and lastly CdCl2 (99.99%, Sigma-Aldrich) is added. The compositional ratios are kept at 0.87 for Cu/Cd + Sn and 1.25 for Cd/Sn. The appearance of the precursor solution is mainly clear transparent after each precursor is added (Fig. S1). Alkali chloride is then added into the precursor solution following the concentration needed with the atomic percentage calculated with respect to the total mol of CCTS. The following alkali chlorides are used: LiCl (≥99.98%, Sigma-Aldrich), NaCl (Sigma-Aldrich), KCl (≥99.0%, Sigma-Aldrich), RbCl (≥99.0%, Sigma-Aldrich), and CsCl (≥99.9%, Aladdin).Thin-film deposition
The precursor solution was spin-coated on Mo–glass substrates at 3000 rpm and annealed at 280 °C for 2 minutes on a hotplate. The spin-coating and annealing steps were repeated 12 times. The films were then sulfurized at 580 °C in a two-zone furnace for 40 minutes.Device fabrication
A thin layer of cadmium sulfide was deposited using chemical bath deposition at 80 °C for 8.5 minutes, followed by deposition of indium tin oxide using DC magnetron sputtering (75 W for 1 hour). Silver paste was used as the top contact point and individual solar cells were mechanically scribed to an area of 0.16 cm2. No antireflection coating was applied on any of the devices.Thin-film characterization
For Inductively Coupled Plasma Optical Emission Spectroscopy (ICP-OES) analysis, the absorber layer was mechanically peeled off from the Mo substrate and dissolved in 3 mL of HNO3 (≥69%, Honeywell). The solution was subsequently diluted with deionized water and analyzed using a PerkinElmer Avio 550 Max Oils. AC Hall measurement was done with a rotating parallel dipole line hall system under a magnetic field strength of 0.814 T.36 Time-resolved photoluminescence (TRPL) measurements were performed using a Horiba Scientific Fluorolog-3 spectrofluorometer equipped with a DeltaDiode-450L pulsed laser as excitation source, operating at a wavelength of 451 nm. The emission was recorded at 880 nm. Time-correlated single-photon counting (TCSPC) was employed to achieve high temporal resolution. The decay curves were analyzed using a biexponential decay model to extract the photoluminescence lifetime components. X-ray diffraction (XRD) measurement was conducted with Bruker D8 Advance using Cu-Kα radiation (40 kV, 40 mA) with grazing-incident setup with grazing angle of 3°. For the conductive-atomic force microscopy (c-AFM) and Kelvin probe force microscopy (KPFM) measurements, an MFP-3D Origin AFM model from Oxford Instruments was used. The scan rate was maintained below 0.3 Hz to prevent potential tip damage, with a pixel resolution of 256 × 256. c-AFM measurements were performed in contact mode using a 25 Pt300B tip from Park Systems. During the c-AFM measurement, an external DC bias of 1.0 V was applied to the sample, while the tip was grounded. For KPFM measurements, amplitude modulation (AM) operation in non-contact mode was employed, using an ASYELEC-01-R2 Ti/Ir-coated Si tip from Oxford Instruments. Scanning electron microscopy (SEM) images were taken using JEOL JSM-7600F with Oxford Instruments 50 mm2 X-Max EDS attached to it.Device characterizations
Current–voltage characteristics were measured using a Keithley 2612A source meter. An Xe-lamp-based VS-0852 solar simulator was used to simulate AM1.5G conditions using a certified standard Si cell as a reference. External Quantum Efficiency (EQE) were measured using a Bentham PVE300 system and a certified standard Si cell was used for calibration. Capacitance–voltage (C–V) measurements were performed using Autolab PGSTAT302N, and the devices were kept in dark 12 hours prior to the measurements.Results and discussion
To identify the trend on the various alkali dopant and the optimum amount of alkali dopant for further characterisation, a screening process based on the device performance was conducted. The device configuration is soda-lime glass (SLG)/Mo/CCTS/CdS/ITO/Ag. The efficiency screening results for alkali-doped CCTS devices are presented in Fig. 1a, with the corresponding JSC, VOC, and fill factor (FF) values detailed in Fig. S2. The screening includes Li-doped CCTS with doping concentrations of 2 at%, 4 at%, and 6 at%, and Na-, K-, Rb-, and Cs-doped CCTS with doping concentrations of 0.5 at%, 1 at%, and 1.5 at%. The optimal doping concentrations, indicated by dark red squares in Fig. 1a, were observed at 4 at% for Li-doped CCTS and 1 at% for Na-, K-, and Rb-doped CCTS, while Cs doping led to a reduction in efficiency across all tested concentrations. These results demonstrate that all alkali dopants, except Cs, improve device efficiency relative to the undoped control, primarily due to an increase in JSC. For subsequent characterisation and device applications, these optimal concentrations were employed. Hereafter, the terms CCTS, CCTS–Li, CCTS–Na, CCTS–K, CCTS–Rb, and CCTS–Cs refer to the undoped CCTS and CCTS doped with 4 at% Li, 1 at% Na, 1 at% K, 1 at% Rb, and 0.5 at% Cs, respectively.Fig. 1b summarizes the alkali content in each doped sample after sulfurization, as analysed using ICP-OES, with the alkali content represented as alkali/(Cu + Cd + Sn) ratio. ICP-OES confirms the presence of each alkali element in the respective samples, although some loss of alkali content occurs during the thin film deposition and sulfurization. CCTS–Li shows the most significant Li loss, despite having the highest precursor doping concentration of 4 at%, with only ∼0.1% Li/(Cu + Cd + Sn) (equivalent to 0.03 at% Li), suggesting minimal incorporation of Li into the CCTS. CCTS–Na, CCTS–K, and CCTS–Rb exhibit alkali/(Cu + Cd + Sn) ratios between 1.1% (0.39 at%) and 2.0% (0.7 at%), indicating that each alkali dopant has a distinct incorporation mechanism to the thin film. CCTS–Cs, with a lower precursor doping concentration of 0.5 at%, shows a lower Cs/(Cu + Cd + Sn) ratio of 0.87% (0.3 at% Cs).
Besides each alkali dopants concentration, the amount of Na in the film was also measured to understand the effect of alkali dopants on the Na distribution which diffused from SLG. Fig. S3 presents the Na content in each sample measured with ICP-OES. Na is detected in all undoped and doped samples, which confirm that the Na is diffused from SLG into the absorber. However, the amount of Na varies depending on the type of dopant. CCTS–Li has the lowest Na content, while undoped CCTS and CCTS–Cs both have the highest Na content. This suggests that the diffusion of Na from SLG is affected and dependent on the type of alkali dopants that exist in the film. This kind of interplay among alkali dopants has also been reported in CZTSSe solar cell, where the introduction of one alkali element can either suppress or facilitate the incorporation of another, for example the saturation of Li content is promoted by the presence of Na in the CZTSSe absorber after annealing.37 Further studies using controlled Na doping strategies by using blocking layer or Na-free substrates could shed more insight into the role of Na and clarify how its interplay with other alkali elements affects CCTS properties.
The effect of alkali doping on the CCTS structure, as analyzed using XRD, is shown in Fig. 1c. The diffraction peaks of all samples are consistent with the reference CCTS structure (ICDD PDF 04-027-7840).38 No additional peaks associated with alkali-related secondary phases were observed in alkali-doped CCTS. However, CdS secondary phase is identified in all samples, a feature commonly reported for CCTS thin films.3,8,9Fig. 1d shows the magnified view of the (112) diffraction peak, revealing a shift toward higher 2θ angles in all alkali-doped samples except CCTS–Cs. This shift suggests the incorporation of alkali elements into the CCTS lattice, leading to lattice shrinkage. Despite the minimal Li element detected in CCTS–Li (Fig. 1b), it induces the most significant peak shift relative to the other alkalis, implying that the smaller radius size of Li and the similar ionic radii of Li+ to Cu+ (0.59 and 0.6 Å, respectively)1 help the incorporation into the CCTS lattice. The absence of peak shifts in the other alkali suggests that they are not incorporated into the CCTS lattice. Furthermore, the (112) peak of the Li-doped samples exhibits asymmetry with a shoulder peak around 27.7°, which corresponds to the (111) diffraction peak of digenite Cu1.8S secondary phase (ICDD PDF 00-056-1256).39 This suggests that Li is partially replacing Cu in the lattice which induces extra Cu to form a secondary phase.
The influence of alkali doping on the bulk electrical properties of CCTS was examined using AC Hall measurements employing the Van der Pauw method. Fig. 2a shows the majority carrier density of undoped and alkali-doped CCTS samples, all of which exhibit p-type conductivity. Corresponding hole mobility data are shown in Fig. S4a, while Table 1 summarizes the average hole densities and mobilities for all samples. Notably, alkali-doping increases carrier density by an order of magnitude compared to the undoped sample. This trend is consistent with previously reported findings for alkali-doped CZTSSe40,41 and CIGS.42,43 Among the alkali-doped samples, CCTS–Li exhibits the highest carrier density, reaching 8.33 × 1015 cm−3. The increase in carrier concentration could explain the increase in JSC for the alkali doped samples. While a slight reduction in hole mobility is observed upon alkali doping, the values remain comparable to those reported for CIGS and CZTS (μh ∼ 1 cm2 V−1 s−1).44–47 Also, both CCTS–Li and CCTS–Na achieve the highest conductivity values among all alkali dopants as shown in Fig. S4b (calculated from charge density and mobility, eqn (S1)). In contrast, the heavier alkalis exhibit inferior conductivity, which is also indicated by their lower JSC and FF values similar to the undoped CCTS. This highlights the complexity of doping effect on the carrier properties and its impact on device performance.48–50
 | ||
| Fig. 2 (a) Carrier density measured using AC Hall effect characterisation. (b) TRPL spectra for all samples. | ||
| p [cm−3] | μ h [cm2 V−1 s−1] | τ [ns] | |
|---|---|---|---|
| CCTS | 3.40 × 1014 | 2.60 | 0.16 |
| CCTS–Li | 8.33 × 1015 | 0.40 | 0.15 |
| CCTS–Na | 1.45 × 1015 | 1.06 | 0.15 |
| CCTS–K | 8.23 × 1014 | 0.93 | 0.16 |
| CCTS–Rb | 1.08 × 1015 | 1.05 | 0.15 |
| CCTS–Cs | 1.29 × 1015 | 0.81 | 0.16 |
To further explore the influence of alkali dopants on optoelectronic properties, the minority carrier lifetime of the samples was analysed using TRPL, with the normalized TRPL data presented in Fig. 2b. Biexponential fitting of the TRPL fast decay regime was performed to extract the weighted average decay time (τ), which represents the minority carrier lifetime. The values of τ1 (faster component) and τ2 (slower component) are listed in Table S1. Table 1 lists the τ values for each sample, all of which are approximately around 0.15 ns. The minimal variation in minority carrier lifetime across all alkali-doped samples suggests stable carrier collection, indicating that alkali incorporation has a negligible effect on the radiative recombination in CCTS. Moreover, minority carrier lifetime is directly related to the implicit VOC as described by eqn (1):51
 | (1) |
For a p-type semiconductor dominated by Shockley–Read–Hall (SRH) recombination, the reverse saturation current density J0 can be approximated as:51
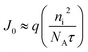 | (2) |
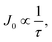 and consequently, VOC ∝ ln(τ). A similar minority carrier lifetime-as measured by TRPL-therefore leads to similar J0, and hence a similar VOC. This correlation is in-line with the finding that alkali doping has a minimal impact on VOC as shown from the screening study (Fig. S2).
and consequently, VOC ∝ ln(τ). A similar minority carrier lifetime-as measured by TRPL-therefore leads to similar J0, and hence a similar VOC. This correlation is in-line with the finding that alkali doping has a minimal impact on VOC as shown from the screening study (Fig. S2).
Following that, the influence of alkali doping on CCTS film morphology was investigated using SEM, with both top-view and cross-sectional images presented in Fig. 3. As shown in Fig. 3, CCTS–K, CCTS–Rb, and CCTS–Cs exhibit smaller grain sizes and a higher density of pinholes compared to CCTS, CCTS–Li, and CCTS–Na. The increased density of pinholes in these samples is expected to hinder carrier transport and potentially leading to higher series resistance (RS) and higher shunt conductance (GSH). These morphological variations indicate that the optimal absorber composition may be dopant-dependent, requiring tailored synthesis conditions to achieve a compact morphology.32 Notably, CCTS–Cs exhibits a highly porous morphology with a distinct bilayer structure in its cross-sectional SEM image, suggesting increased structural inhomogeneity. This inhomogeneity has a potential to further degrade carrier transport properties as evident by the lack of improvement with Cs doping. In contrast, CCTS–Na among the samples forms more compact morphology with a significantly lower pinhole density. This enhanced structural uniformity help promotes more efficient carrier transport, ultimately contributing to superior device performance.
 | ||
| Fig. 3 Top-view SEM images with corresponding cross-sectional SEM images (inset) of (a) CCTS, (b) CCTS–Li, (c) CCTS–Na, (d) CCTS–K, (e) CCTS–Rb, and (f) CCTS–Cs. | ||
In addition, the cationic elemental ratios of the thin films were evaluated using SEM-EDS, focusing on the Cu/(Cd + Sn) and Cd/Sn ratios (Table S2). The Cu/(Cd + Sn) ratio for all samples, including the undoped CCTS, is consistently close to 1.00—an increase from the Cu-poor precursor ratio of 0.87—indicating a shift towards Cu-rich conditions after processing. As for the Cd/Sn ratio, all samples retain the Cd-rich nature of the precursor solution. However, CCTS–Cs exhibits a notably lower Cd/Sn ratio, suggesting Cd loss during synthesis. This Cd deficiency may contribute to the poorer device performance observed for Cs-doped CCTS compared to the undoped sample. Overall, the elemental compositions of the doped films remain largely comparable to that of the undoped CCTS, indicating that alkali doping does not significantly alter the cation stoichiometry.
Continuing from the morphology study, the impact of alkali doping on the carrier transport at the absorber interface was also investigated using KPFM, c-AFM, and SEM. KPFM was employed to measure the contact potential difference (CPD) between intragrains (IGs) and grain boundaries (GBs) in both undoped and alkali-doped CCTS. The topographical image of undoped CCTS is presented in Fig. 4a, with the corresponding surface potential map shown in Fig. 4b. A linescan extracted along the yellow arrow in Fig. 4a and b is illustrated in Fig. 4c, providing a direct comparison between the topographical features and the corresponding CPD variations. The topographical images, surface potential maps, and linescans for the rest of alkali-doped CCTS are provided in Fig. S5. From the measurements, the histograms of CPDGB and CPDIG for all samples, presented in Fig. S6, show that CPDGB is consistently lower than CPDIG across all samples. This indicates that the GBs are more negatively charged which results in upward band bending at GBs, which attracts majority carriers (holes) while repelling minority carriers (electrons) at the GBs.52
The linescan also introduces the parameter ΔCPDGB, defined as the difference between the CPD at IGs (CPDIG) and at GBs (CPDGB) (ΔCPDGB = CPDIG − CPDGB), quantifying the band bending magnitude between IGs and GBs. Fig. 4d presents the ΔCPDGB values for each sample, showing a mean ΔCPDGB of approximately 25 mV for undoped CCTS, while alkali-doped CCTS samples (excluding CCTS–Rb) exhibit a reduced mean ΔCPDGB to around 18 mV. These values are notably lower than the reported mean ΔCPDGB values for CIGS and CZTSSe, which typically range from 40 mV to 150 mV.23,25,26,52–55 This reduction in ΔCPDGB indicates smaller upward band bending in CCTS–Li, CCTS–Na, CCTS–K, and CCTS–Cs compared to undoped CCTS. An illustration of the reduced upward band bending is provided in Fig. 4e and f. In contrast to previous reports on alkali doping in CIGS and CZTSSe, where alkali incorporation has been shown to increase band bending at GBs due to alkali introduction,25,26,53 the observed reduction in ΔCPDGB in alkali-doped CCTS suggests a distinct mechanism. This reduction likely arises from the interplay of alkali-related defects at GBs and IGs in CCTS. In CIGS or CZTSSe, the CPDGB is typically higher than CPDIG,56 and alkali doping, which commonly introduces acceptor-like defects (e.g. LiCu, LiCd+ or Lii+), tends to accumulate at GBs, enhancing the band bending.14,25,57 On the other hand, in CCTS, the similar incorporation leads to a flatter potential difference between the two regions (i.e., a reduced ΔCPDGB), and thus less pronounced upward band bending at the GBs. Consequently, the reduced upward band bending in alkali-doped CCTS minimizes the accumulation of majority carriers (holes) at GBs, promoting smoother carrier transport across the GBs to the absorber interface.
The c-AFM measurements were performed to investigate the nanoscale current characteristics in both undoped and alkali-doped CCTS. Fig. 5a depicts the topographic image of undoped CCTS, while Fig. 5b presents the corresponding surface current map acquired under an applied external bias of 1.0 V. A linescan extracted along the yellow arrow in Fig. 5a and b is shown in Fig. 5c, where the parameter Δcurrent represents the surface current measured within IGs. Topographical images, surface current maps, and linescans of alkali-doped CCTS samples are provided in Fig. S7. Fig. 5d summarizes the distribution of Δcurrent across all samples, showing that the mean Δcurrent for alkali-doped CCTS is higher by almost an order of magnitude compared to the undoped CCTS which suggest that alkali improve the IGs conductivity. This improvement in Δcurrent can be attributed to the increased CPDIG observed in alkali-doped CCTS (Fig. S6). The increased CPDIG in these samples can be caused by the occupation of negatively charged copper vacancies (VCu−) by the alkali leading to the formation of neutral alkali–Cu antisite defects. In CZTS and CIGS, the formation of these neutral defects are quite common, for example LiCu0 and NaCu0 which exhibit a low formation energy of 0.25 and 0.64 eV in CZTS, as reported in prior DFT studies.21,58,59 The presence of these neutral defects likely reduces the concentration of CdCu shallow donors too, passivating harmful defects, thereby enhancing surface current within IGs.23,60 The enhancement of Δcurrent in alkali-doped CCTS is consistent with the increase in hole density which indicates an overall improvement in intragrain conductivity, which facilitates more efficient carrier transport to the absorber interface.
 | ||
| Fig. 5 (a) Topographical image and (b) surface current map of undoped CCTS. (c) Linescan along the arrow in (a) and (b). (d) Boxplot of Δcurrent of alkali-doped CCTS. | ||
To further investigate the impact of alkali doping on CCTS solar cell performance, the key photovoltaic parameters for the optimally doped samples are summarized and compared in Fig. 6. Boxplots of JSC, VOC, FF, and power conversion efficiency (PCE) are presented, based on measurements from 10 devices per dopant, all fabricated without anti-reflection coating. As shown in Fig. 6a, alkali-doped samples exhibit an overall increase in JSC compared to the undoped CCTS, which can be attributed to the enhanced bulk carrier density (Fig. 2a) and improved charge transport to the absorber interface as indicated by the reduced surface potential difference (ΔCPD, Fig. 4d) and increase in nanoscale current in the grains (Δcurrent, Fig. 5d) from the KPFM and c-AFM measurements respectively. In contrast, the variations in VOC (Fig. 6b) remain minimal across all samples, consistent with the negligible changes in minority carrier lifetime observed for alkali-doped CCTS (Fig. 2b). The FF (Fig. 6c) follows a distinct trend: it increases for lighter alkali (Li and Na) but decreases for heavier ones (K, Rb, and Cs). This trend correlates with the film morphology (Fig. 3), where CCTS–Na exhibits a more compact and uniform structure, while CCTS–Cs shows a highly porous bilayer morphology. Notably, the trends in JSC and FF are aligned with the ΔCPD and Δcurrent measurements—both peaking around CCTS–Na—except for CCTS–Cs, which deviates, likely due to its poor morphology and compositional changes. Consequently, these factors are reflected in the PCE results (Fig. 6d), with CCTS–Na achieving the highest efficiency among all samples.
Table 2 details the photovoltaic parameters of the best-performing cell for each alkali dopant. The highest efficiency of 8.47% is obtained for CCTS–Na, attributed to its enhanced JSC and highest FF, followed in descending order by CCTS–Li, CCTS–Rb, CCTS–K, and CCTS–Cs. Diode parameters were extracted from light J–V curves (Fig. S8) using Lambert-W function fitting, revealing key metrics such as series resistance RS, shunt conductance GSH, ideality factor (n), and reverse saturation current density (J0). A distinct trend is observed for RS and GSH among CCTS–K, CCTS–Rb, and CCTS–Cs. The RS values for these samples are notably higher than those of undoped CCTS, CCTS–Li, and CCTS–Na. Similarly, the GSH values for the heavier alkali are higher than the lighter ones. These findings are consistent with the higher pinhole density in CCTS–K, CCTS–Rb, and CCTS–Cs, which hinders charge transport and decrease FF values.
| η [%] | J SC [mA cm−2] | V OC [mV] | FF [%] | R S [Ω cm2] | G SH [mS cm−2] | n | J 0 [mA cm−2] | J SC/JSC,SQ [%] | V OC/VOC,SQ [%] | |
|---|---|---|---|---|---|---|---|---|---|---|
| CCTS | 7.77 | 24.03 | 524.7 | 61.63 | 1.72 | 2.19 | 2.14 | 1.63 × 10−3 | 74.86 | 45.39 |
| Li | 8.35 | 25.81 | 522.1 | 61.98 | 1.20 | 0.98 | 2.50 | 7.45 × 10−3 | 80.40 | 45.16 |
| Na | 8.47 | 25.32 | 531.0 | 63.00 | 1.65 | 2.15 | 1.97 | 6.74 × 10−4 | 78.88 | 45.93 |
| K | 8.07 | 26.19 | 532.6 | 58.17 | 2.05 | 4.42 | 2.22 | 1.99 × 10−3 | 81.59 | 46.07 |
| Rb | 8.08 | 25.66 | 537.9 | 58.50 | 2.49 | 4.56 | 1.91 | 3.96 × 10−4 | 79.94 | 46.53 |
| Cs | 7.61 | 24.18 | 548.3 | 57.40 | 3.16 | 3.43 | 2.39 | 3.04 × 10−3 | 75.33 | 47.43 |
As for the ideality factor (n), only CCTS–Na and CCTS–Rb shows value less than 2 (n < 2), indicating that these two alkalis improve the CCTS recombination, which infer the dominant recombination mechanism is Shockley–Read–Hall (SRH) recombination in the depletion region, which is not ideal for a single diode junction, however it is more ideal than the undoped and other alkali with n > 2, which suggest more complex recombination are dominate in the junction, such as interface recombination or coupled defects recombination.61,62 Similarly, for J0, CCTS–Na and CCTS–Rb have the lowest value which indicates lower recombination and better diode behavior which lead to higher VOC than best undoped CCTS. This suggests that these two alkalis help to passivate defects more effectively in CCTS. These complementary behaviors point to co-doping strategies as a promising direction for further performance enhancement. For example, Li and Na improve JSC and FF through enhanced carrier transport and morphology, whereas Na and Rb reduce recombination losses and boost VOC. Combining such dopants could mitigate the trade-offs observed with individual species and potentially deliver synergistic improvements in efficiency. Co-doping thus emerges as an attractive design strategy for engineering both the bulk and interfacial properties of CCTS absorbers.
The external quantum efficiency (EQE) spectra for alkali-doped CCTS (Fig. 6e) exhibit minimal variation in the short-wavelength range (300–530 nm), suggesting uniform buffer and window layer across all samples. More noticeable differences appear in the 530–830 nm range, where alkali-doped CCTS samples exhibit enhanced EQE relative to the undoped CCTS, consistent with the JSC improvements observed in Fig. 6a.
The band gap of the films was extracted using the derivative d[−ln(1 − EQE)]/dE (Fig. S9a), as described by Mattheis et al.63,64 This approach allows the extraction of both the bandgap energy (Eg) and the standard deviation (σEg) associated with bandgap fluctuations. The extracted Eg for undoped CCTS was found to be approximately 1.44 eV, consistent with prior studies.3,8,9 Across all alkali-doped samples, minimal shifts in Eg were observed. The extracted σEg values, summarised in Table 3 and depicted in Fig. S9a, range from 39.21 to 45.05 meV across all samples. The EQE region beyond 900 nm was further analysed to examine the exponential decay associated with band tail states, quantified by the Urbach energy (EU). This parameter was determined by plotting ln[−ln(1 − EQE)] vs. E − Eg, as shown in Fig. S9b, and extracting the inverse slope of the linear region below the bandgap (Eg).65Table 3 details the EU values for each sample, while the inset of Fig. S9b provides a bar chart illustrating the distribution, which ranges from 41.22 meV to 51.24 meV. While the σEg values obtained for CCTS and alkali-doped CCTS are comparable to the 45.53 meV reported for the record 14.6% efficiency CZTSSe solar cell,66 the observed EU values are higher by 20–30 meV. This suggests higher band tailing and associated non-radiative recombination which limit VOC.
| E g [eV] | σ E g [meV] | E U [meV] | N CV [cm−3] | x [μm] | |
|---|---|---|---|---|---|
| CCTS | 1.44 | 41.20 | 47.27 | 1.35 × 1016 | 0.159 |
| CCTS–Li | 1.45 | 45.05 | 43.14 | 1.43 × 1016 | 0.154 |
| CCTS–Na | 1.44 | 42.46 | 49.04 | 4.18 × 1016 | 0.116 |
| CCTS–K | 1.44 | 39.21 | 51.24 | 7.36 × 1015 | 0.192 |
| CCTS–Rb | 1.44 | 40.11 | 41.22 | 1.57 × 1016 | 0.150 |
| CCTS–Cs | 1.44 | 42.75 | 43.88 | 1.85 × 1016 | 0.141 |
To evaluate the performance limit of our CCTS devices, we benchmarked them against the Shockley–Queisser (SQ) for a 1.44 eV bandgap absorber, corresponding to a theoretical JSC,SQ of 32.1 mA cm−2 and VOC,SQ of 1156 mV. The JSC/JSC,SQ ratio for the best-performing alkali-doped CCTS devices ranges from 75% to 82%, surpassing previously reported values for CCTS and Ag-doped CCTS,3,8,9 highlighting the effectiveness of alkali in improving JSC. However, the highest obtained VOC/VOC,SQ ratio is only 47.43%, which remains lower than that of the record CZTSSe solar cell. This VOC deficit is consistent based on the quite low minority carrier lifetime in TRPL and high Urbach tail. These findings indicate that non-radiative recombination, likely driven by band tailing and deep-level defects, remains a key bottleneck. Further efforts to improve absorber quality and interface passivation will be critical to unlocking higher VOC and pushing performance closer to the SQ limit.
In addition, capacitance–voltage (C–V) measurements were conducted to gain further insight into the electronic properties of alkali-doped CCTS devices (Fig. 6f). The apparent carrier density (NCV) and depletion width (x) for the samples are summarized in Table 3 to provide insight into their doping characteristics and junction properties. The NCV trends closely follow the carrier density variations presented in Fig. 2a, with alkali-doped CCTS exhibiting slightly higher NCV values than undoped CCTS. Among the doped samples, CCTS–Na exhibits the highest NCV, consistent with its high IGs conductivity which correlating well with its enhanced JSC and highest FF in this study.
Conclusions
This study systematically investigates the effects of a complete set of alkali doping on enhancing the photovoltaic performance of CCTS, with emphasis on the changes in bulk and interface properties. The results reveal that alkali doping increases carrier density by an order of magnitude while the minority carrier lifetimes remain the same. Both undoped and alkali-doped CCTS exhibit upward band bending at grain boundaries, which is reduced upon alkali doping, thereby minimizing majority hole carrier accumulation and enabling more efficient carrier transport across grain boundaries. Furthermore, alkali doping increases intragrain surface current by an order of magnitude, indicating improved intragrain conductivity. All of these contributed to the enhanced JSC in the alkali-doped samples.Among the alkali-doped samples, Na-doped CCTS exhibits a notably compact film morphology with fewer pinholes, further facilitating efficient carrier transport to the absorber interface. This results in lower RS and GSH, it also shows a low n and J0, leading to the highest FF among the samples. Combined with an improved JSC, CCTS–Na achieves the highest device efficiency of 8.47%, followed by CCTS–Li, CCTS–Rb, CCTS–K, and CCTS–Cs in descending order of performance. These findings highlight the potential of alkali doping as an effective approach to improving CCTS solar cell performance, with Na emerging as the most promising dopant.
Author contributions
Conceptualization: S. L., L. H. W., and W. J.; methodology: E. J., H. K. P., S. L., Y. J. C., G. L., M. A. A.-R., and A. S.; investigation: E. J., H. K. P., A. I., S. L., Y. J. C., G. L., M. A. A.-R., and A. S.; formal analysis: E. J., H. K. P., A. I., S. L., Y. J. C., G. L., M. A. A.-R., and A. S.; data curation: E. J., H. K. P., A. I., Y. J. C., G. L., and M. A. A.-R.; validation: E. J., H. K. P., A. I., S. L., A. S., A. H. Y., W. J., and L. H. W.; visualization: E. J., H. K. P., A. I., Y. J. C., G. L., and A. S.; writing – original draft: E. J., H. K. P., A. I., and S. L.; all authors contributed to review & editing; project administration: E. J., A. I., S. L., A. S., A. H. Y., W. J., and L. H. W.; resources: A. H. Y., W. J., and L. H. W.; supervision: S. L., A. H. Y., W. J., and L. H. W.; funding acquisition: A. I., S. L., A. H. Y., W. J., and L. H. W.Conflicts of interest
There are no conflicts to declare.Data availability
Data for this article are available in NTU research data repository DR-NTU. See DOI: https://doi.org/10.21979/N9/A5FZZW.Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta06518f.
Acknowledgements
This research was funded by the Indonesian Endowment Fund for Education (LPDP) on behalf of the Ministry of Education, Culture, Research, and Technology of Indonesia, and managed by Universitas Indonesia under the INSPIRASI Program (Grant No. PRJ-61/LPDP/2022 and 612/E1/KS.06.02/2022). Additional support was provided by the Singapore Ministry of Education Tier 1 grant (Award ID: RG155/23) and Tier 2 grant (MOE-T2EP50223-0007). It was also supported by national R&D programs through the National Research Foundation of Korea (Grant No. RS-2024-00355905 and NRF-2018R1A6A1A03025340). H. K. P. received fellowship support from the Hyundai Motor Chung Mong-Koo Foundation. We would like to express our gratitude to the Facility for Analysis, Characterization, Testing and Simulation (FACTS) at Nanyang Technological University, Singapore, for providing access to their SEM.References
- R. D. Shannon, Acta Crystallogr., Sect. A, 1976, 32, 751–767 CrossRef.
- I. V. Bodnar, I. A. Victorov, A. G. Karosa, E. K. Arushanov and S. Levcenko, Appl. Phys. Lett., 2020, 117, 182102 CrossRef.
- S. Hadke, S. Levcenko, G. S. Gautam, C. J. Hages, J. A. Márquez, V. Izquierdo-Roca, E. A. Carter, T. Unold and L. H. Wong, Adv. Energy Mater., 2019, 9, 1902509 CrossRef.
- H. Guan, J. Zhao, X. Wang and F. Yu, Chalcogenide Lett., 2013, 10, 367–372 Search PubMed.
- S. Rühle, Sol. Energy, 2016, 130, 139–147 CrossRef.
- S. Lie, M. Guc, V. Tunuguntla, V. Izquierdo-Roca, S. Siebentritt and L. H. Wong, J. Mater. Chem. A, 2022, 10, 9137–9149 RSC.
- A. Wang, J. Huang, C. Yan, G. He, X. Cui, X. Yuan, S. Zhou, M. He, T. Qiu, C. Zhao, M. A. Green, K. Sun and X. Hao, Adv. Funct. Mater., 2024, 34, 2407063 CrossRef.
- A. Ibrahim, S. Lie, J. M. R. Tan, R. Swope, A. G. Medaille, S. Hadke, E. Saucedo, R. Agrawal and L. H. Wong, J. Mater. Chem. A, 2024, 12, 2673–2679 RSC.
- P. Fan, J. Lin, J. Hu, Z. Yu, Y. Zhao, S. Chen, Z. Zheng, J. Luo, G. Liang and Z. Su, Adv. Funct. Mater., 2022, 32, 2207470 CrossRef.
- N. Sarmadian, R. Saniz, B. Partoens and D. Lamoen, J. Appl. Phys., 2016, 120, 085707 CrossRef.
- J. Hedstrom, H. Ohlsen, M. Bodegard, A. Kylner, L. Stolt, D. Hariskos, M. Ruckh and H.-W. Schock, in Conference Record of the Twenty Third IEEE Photovoltaic Specialists Conference - 1993 (Cat. No.93CH3283-9), IEEE, 1993, pp. 364–371 Search PubMed.
- H. K. Park, K. Yim, J. Lee, Y. Cho, I. Jeong, D. Shin, J. Gwak, A. Walsh, K. Kim and W. Jo, npj Flexible Electron., 2024, 8, 58 CrossRef.
- S. Siebentritt, E. Avancini, M. Bär, J. Bombsch, E. Bourgeois, S. Buecheler, R. Carron, C. Castro, S. Duguay, R. Félix, E. Handick, D. Hariskos, V. Havu, P. Jackson, H. P. Komsa, T. Kunze, M. Malitckaya, R. Menozzi, M. Nesladek, N. Nicoara, M. Puska, M. Raghuwanshi, P. Pareige, S. Sadewasser, G. Sozzi, A. N. Tiwari, S. Ueda, A. Vilalta-Clemente, T. P. Weiss, F. Werner, R. G. Wilks, W. Witte and M. H. Wolter, Adv. Energy Mater., 2020, 10, 1903752 CrossRef.
- N. Nicoara, R. Manaligod, P. Jackson, D. Hariskos, W. Witte, G. Sozzi, R. Menozzi and S. Sadewasser, Nat. Commun., 2019, 10, 3980 CrossRef PubMed.
- J. Keller, K. Kiselman, O. Donzel-Gargand, N. M. Martin, M. Babucci, O. Lundberg, E. Wallin, L. Stolt and M. Edoff, Nat. Energy, 2024, 9, 467–478 CrossRef.
- H. Lee, Y. Jang, S. W. Nam, C. Jung, P. P. Choi, J. Gwak, J. H. Yun, K. Kim and B. Shin, ACS Appl. Mater. Interfaces, 2019, 11, 35653–35660 CrossRef CAS PubMed.
- I. Khatri, P. Santos, P. Anacleto, D. Sharma, M. Belmoubarik, N. Nicoara, M. Sugiyama and S. Sadewasser, Phys. Status Solidi RRL, 2022, 16, 2100462 CrossRef CAS.
- A. Chirilg, P. Reinhard, F. Pianezzi, P. Bloesch, A. R. Uhl, C. Fella, L. Kranz, D. Keller, C. Gretener, H. Hagendorfer, D. Jaeger, R. Erni, S. Nishiwaki, S. Buecheler and A. N. Tiwari, Nat. Mater., 2013, 12, 1107–1111 CrossRef.
- G. Lim, H. K. Park, W. H. Kim, S. H. Kim, K. J. Yang, J. K. Kang, D. H. Kim and W. Jo, J. Mater. Chem. A, 2023, 11, 25555–25562 RSC.
- A. Cabas-Vidani, S. G. Haass, C. Andres, R. Caballero, R. Figi, C. Schreiner, J. A. Márquez, C. Hages, T. Unold, D. Bleiner, A. N. Tiwari and Y. E. Romanyuk, Adv. Energy Mater., 2018, 8, 1801191 CrossRef.
- C. M. Sutter-Fella, J. A. Stückelberger, H. Hagendorfer, F. La Mattina, L. Kranz, S. Nishiwaki, A. R. Uhl, Y. E. Romanyuk and A. N. Tiwari, Chem. Mater., 2014, 26, 1420–1425 CrossRef CAS.
- S. G. Haass, C. Andres, R. Figi, C. Schreiner, M. Bürki, A. N. Tiwari and Y. E. Romanyuk, AIP Adv., 2018, 8, 015133 CrossRef.
- H. Xin, S. M. Vorpahl, A. D. Collord, I. L. Braly, A. R. Uhl, B. W. Krueger, D. S. Ginger and H. W. Hillhouse, Phys. Chem. Chem. Phys., 2015, 17, 23859–23866 RSC.
- K. Yang, S. Kim, S. Kim, D. Son, J. Lee, Y. Kim, S. Sung, D. Kim, T. Enkhbat, J. Kim, J. Kim, W. Jo and J. Kang, Adv. Funct. Mater., 2021, 31, 2102238 CrossRef CAS.
- W. L. Jeong, K. P. Kim, J. Kim, H. K. Park, J. H. Min, J. S. Lee, S. H. Mun, S. T. Kim, J. H. Jang, W. Jo and D. S. Lee, Adv. Sci., 2020, 7, 1903085 CrossRef CAS PubMed.
- J. Kim, G. Y. Kim, T. T. T. Nguyen, S. Yoon, Y. K. Kim, S. Y. Lee, M. Kim, D. H. Cho, Y. D. Chung, J. H. Lee, M. J. Seong and W. Jo, Phys. Chem. Chem. Phys., 2020, 22, 7597–7605 RSC.
- L. Dong, S. Tao, M. Zhao, D. Zhuang, Q. Gong, H. Zhu, Y. Wang, Y. Li, H. Wang, M. Jia and J. Li, Sol. RRL, 2023, 7, 2300061 CrossRef CAS.
- H. Xu, R. Meng, X. Xu, Y. Liu, Y. Sun and Y. Zhang, Small, 2024, 20, 2408122 CrossRef CAS.
- W. Yang, Y. Ji, W. Chen, Y. Pan, Z. Chen, S. Wu, S. P. Russo, Y. Xu, T. A. Smith, A. Chesman, P. Mulvaney and F. Liu, Small, 2024, 20, 2307807 CrossRef CAS PubMed.
- A. Mule, B. Vermang, M. Sylvester, G. Brammertz, S. Ranjbar, T. Schnabel, N. Gampa, M. Meuris and J. Poortmans, Thin Solid Films, 2017, 633, 156–161 CrossRef CAS.
- X. Zhong, J. Wang, L. Han, J. J. Chi, T. Liu, D. Kou, W. Zhou, Z. Zhou, S. Yuan, Y. Meng, Y. Qi, Q. Meng and S. Wu, Adv. Funct. Mater., 2025, 35, 2418548 CrossRef CAS.
- S. G. Haass, C. Andres, R. Figi, C. Schreiner, M. Bürki, Y. E. Romanyuk and A. N. Tiwari, Adv. Energy Mater., 2018, 8, 1701760 CrossRef.
- Y.-T. Hsieh, Q. Han, C. Jiang, T.-B. Song, H. Chen, L. Meng, H. Zhou and Y. Yang, Adv. Energy Mater., 2016, 6, 1502386 CrossRef.
- G. Altamura, M. Wang and K. L. Choy, Sci. Rep., 2016, 6, 22109 CrossRef CAS.
- S. López-Marino, Y. Sánchez, M. Espíndola-Rodríguez, X. Alcobé, H. Xie, M. Neuschitzer, I. Becerril, S. Giraldo, M. Dimitrievska, M. Placidi, L. Fourdrinier, V. Izquierdo-Roca, A. Pérez-Rodríguez and E. Saucedo, J. Mater. Chem. A, 2016, 4, 1895–1907 RSC.
- O. Gunawan, Y. Virgus and K. F. Tai, Appl. Phys. Lett., 2015, 106, 062407 CrossRef.
- S. Moser, A. N. Tiwari and R. Carron, Sol. Energy Mater. Sol. Cells, 2023, 250, 112094 CrossRef CAS.
- K. A. Rosmus, J. A. Brant, S. D. Wisneski, D. J. Clark, Y. S. Kim, J. I. Jang, C. D. Brunetta, J. H. Zhang, M. N. Srnec and J. A. Aitken, Inorg. Chem., 2014, 53, 7809–7811 CrossRef CAS PubMed.
- G. Will, E. Hinze and A. R. M. Abdelrahman, Eur. J. Mineral., 2002, 14, 591–598 CrossRef CAS.
- T. Prabhakar and N. Jampana, Sol. Energy Mater. Sol. Cells, 2011, 95, 1001–1004 CrossRef CAS.
- Y. E. Romanyuk, S. G. Haass, S. Giraldo, M. Placidi, D. Tiwari, D. J. Fermin, X. Hao, H. Xin, T. Schnabel, M. Kauk-Kuusik, P. Pistor, S. Lie and L. H. Wong, J. Phys.: Energy, 2019, 1, 044004 CAS.
- T. Nakada, D. Iga, H. O. H. Ohbo and A. K. A. Kunioka, Jpn. J. Appl. Phys., 1997, 36, 732 CrossRef CAS.
- Y. Sun, S. Lin, W. Li, S. Cheng, Y. Zhang, Y. Liu and W. Liu, Engineering, 2017, 3, 452–459 CrossRef CAS.
- T. Umehara, S. Iinuma, A. Sadono, Y. Kurokawa and A. Yamada, Jpn. J. Appl. Phys., 2015, 54, 018001 CrossRef CAS.
- T. Hayakawa, T. Nishimura, H. Sugiura, N. Suyama, K. Nakada and A. Yamada, IEEE J. Photovoltaics, 2018, 8, 1841–1846 Search PubMed.
- O. Gunawan, K. F. Tai and Y. Virgus, in 2016 IEEE 43rd Photovoltaic Specialists Conference (PVSC), IEEE, 2016, pp. 2002–2005 Search PubMed.
- S. Lie, W. Li, S. W. Leow, D. M. Bishop, O. Gunawan and L. Helena Wong, Sol. RRL, 2020, 4, 1900521 CrossRef CAS.
- K. Timmo, M. Pilvet, K. Muska, M. Altosaar, V. Mikli, R. Kaupmees, R. Josepson, J. Krustok, M. Grossberg-Kuusk and M. Kauk-Kuusik, Mater. Adv., 2023, 4, 4509–4519 RSC.
- E. Cho, S. J. Sung, K. J. Yang, J. Lee, V. Q. Hoang, B. Kadiri-English, D. K. Hwang, J. K. Kang and D. H. Kim, J. Mater. Chem. A, 2025, 13, 8507–8517 RSC.
- S. Yang, B. Yu and H. Yu, Small Methods, 2025, 9, 2401307 CrossRef CAS.
- S. M. Sze and K. K. Ng, Physics of Semiconductor Devices, Wiley, 2006 Search PubMed.
- O. Cojocaru-Mirédin, M. Raghuwanshi, R. Wuerz and S. Sadewasser, Adv. Funct. Mater., 2021, 31, 2103119 CrossRef.
- J. Hwang, H. K. Park, D. Shin, I. Jung, I. Hwang, Y. J. Eo, A. Cho, J. H. Park, S. Song, Y. Cho, J. Gwak, H. S. Jang, W. Jo and K. Kim, J. Mater. Chem. A, 2023, 11, 19546–19555 RSC.
- L. Cao, Z. Zhou, W. Zhou, D. Kou, Y. Meng, S. Yuan, Y. Qi, L. Han, Q. Tian, S. Wu and S. Liu, Small, 2024, 20, 2304866 CrossRef CAS.
- E. M. Lanzoni, T. Gallet, C. Spindler, O. Ramírez, C. K. Boumenou, S. Siebentritt and A. Redinger, Nano Energy, 2021, 88, 106270 CrossRef CAS.
- L. Yang, Y. Wang, X. Wang, S. Shafique, F. Zheng, L. Huang, X. Liu, J. Zhang, Y. Zhu, C. Xiao and Z. Hu, Small, 2024, 20, 2304362 CrossRef CAS.
- J. B. Li, V. Chawla and B. M. Clemens, Adv. Mater., 2012, 24, 720–723 CrossRef CAS PubMed.
- Y. Yang, L. Huang and D. Pan, ACS Appl. Mater. Interfaces, 2017, 9, 23878–23883 CrossRef CAS PubMed.
- T. Maeda, A. Kawabata and T. Wada, Phys. Status Solidi C, 2015, 12, 631–637 CrossRef CAS.
- A. Nagaoka, H. Miyake, T. Taniyama, K. Kakimoto, Y. Nose, M. A. Scarpulla and K. Yoshino, Appl. Phys. Lett., 2014, 104, 152101 CrossRef.
- M. Saad and A. Kassis, Sol. Energy Mater. Sol. Cells, 2003, 79, 507–517 CrossRef CAS.
- A. Schenk and U. Krumbein, J. Appl. Phys., 1995, 78, 3185–3192 CrossRef CAS.
- J. Mattheis, U. Rau and J. H. Werner, J. Appl. Phys., 2007, 101, 113519 CrossRef.
- U. Rau, B. Blank, T. C. M. Müller and T. Kirchartz, Phys. Rev. Appl., 2017, 7, 044016 CrossRef.
- M. Troviano and K. Taretto, Sol. Energy Mater. Sol. Cells, 2011, 95, 821–828 CrossRef CAS.
- J. Shi, J. Wang, F. Meng, J. Zhou, X. Xu, K. Yin, L. Lou, M. Jiao, B. Zhang, H. Wu, Y. Luo, D. Li and Q. Meng, Nat. Energy, 2024, 9, 1095–1104 CAS.
| This journal is © The Royal Society of Chemistry 2025 |



