Thiol functionalized metal–organic framework for efficient silver adsorption and removal from aqueous solution
Simmy
Rathod
a,
Sofiia
Bercha
b,
M. Baris
Yagci
c,
Duygu
Yilmaz
 d,
Olena
Zavorotynska
d,
Olena
Zavorotynska
 b and
Sachin Maruti
Chavan
b and
Sachin Maruti
Chavan
 *a
*a
aDepartment of Chemistry, Bioscience and Environmental Engineering, University of Stavanger, P.O. Box 8600, Stavanger NO-4036, Norway. E-mail: Sachin.m.chavan@uis.no
bDepartment of Mathematics and Physics, University of Stavanger, P.O. Box 8600, Stavanger NO-4036, Norway
cKoç University Surface Science and Technology Center (KUYTAM), Istanbul 34450, Turkiye
dDepartment of Environmental Industrial Processes, Institute for Energy Technology, P.O. Box 40, Kjeller NO-2027, Norway
First published on 20th October 2025
Abstract
MOF-808, a zirconium-based metal–organic framework (Zr-MOF), allows desired functionalization using six monocarboxylate ligands. In this study, a thiol (–SH) group was incorporated into MOF-808 using thiolactic acid (ThA) as a modulator. MOF-808 with 25 molar equivalents of ThA (M-ThA-25) achieved a –SH loading of 5.62 per cluster, twice that of thioglycolic acid as a modulator. Due to strong Ag–S interactions predicted by the hard–soft-acid–base (HSAB) theory, M-ThA-25 demonstrated an excellent Ag+ adsorption capacity, with a maximum uptake of 365.47 mg g−1, which is well described by the Langmuir isotherm. The kinetics were impressive, achieving a 100% removal of 500 ppm of Ag+ in under 10 minutes. Post-adsorption powder X-ray diffraction and scanning electron microscopy images showed that chloride impurities can lead to the precipitation of AgCl as observed in MOF-808 with 50 equivalents of acetic acid (M-AA-50). In M-ThA-25, the adsorbed Ag is mainly in the form of Ag+. X-ray photoelectron spectroscopy analysis of M-ThA-25-Ag confirmed that the adsorption occurred via an ion-exchange mechanism, where incoming Ag ions replaced H+ in the –SH group. To evaluate selectivity, M-ThA-25 was exposed to a mixed-ion solution containing Al3+, Pb2+, and Ag+ at concentrations representative of those found in solar panel leachates. It showed that Ag+ uptake wasn't affected by the presence of competing Al3+. This highlights M-ThA-25's potential as an efficient silver adsorbent. However, regenerating silver is challenging due to the strong Ag–S bonds, so future work should focus on developing better desorption methods for practical recovery.
1. Introduction
Metal–organic frameworks (MOFs) represent a rapidly growing class of porous materials that have gained considerable attention as advanced adsorbents for selective metal separation. Their unique advantages include exceptionally high surface areas, tunable pore architectures, and the ability to introduce functional groups that enable specific binding interactions.1,2 Over the past decade, MOFs with tailored binding sites have been designed to capture toxic heavy metals such as lead, mercury, and cadmium from contaminated water, demonstrating great promise for environmental remediation.3,4 In parallel, extensive efforts have focused on developing MOFs for the separation and recovery of critical metals essential to clean energy technologies—including lithium, cobalt, rare earth elements, as well as precious metals—from energy-related waste, complex industrial effluents, and low-grade ores.5–8Among the broad family of MOFs, Zr-MOFs are particularly attractive for their high chemical and thermal stability.9,10 The remarkable robustness of this material is largely attributed to the strong interactions between carboxylate groups and zirconium ions, as well as the stability of Zr6 cluster [Zr6(μ3-O)4(μ3-OH)4]12+.9,11 This cluster serves as a versatile secondary building unit (SBU) that facilitates the construction of 3D MOFs with connectivity ranging from 6 to 12. The potential for missing linker vacancies and open coordination sites presents opportunities for targeted functionalization tailored to specific applications.20,21 MOF-808 has a spn topology and includes two trimesic acid linkers per cluster, which results in 12 unoccupied sites. These sites can be filled by up to six terminal monocarboxylate ligands or by a combination of monocarboxylic acids, hydroxides, chlorides, or water molecules.21
Monocarboxylic acids containing specific functional groups can be introduced into the pores of MOF-808 through either direct synthesis or solvent-assisted ligand exchange (SALE), also referred to as post-synthetic exchange (PSE).12–15Fig. 1 illustrates the possible modulators loaded on MOF-808 either through direct functionalization or PSE, along with their respective pKa values. Acetic acid (AA) and formic acid (FA) are commonly used modulators and can be grafted directly into MOF-808![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 12,15 while benzoic acid (BzA)12 and various amino acids14,15 could only be introduced into MOF-808 via the SALE method. This distinction cannot be explained solely by pKa values, as some amino acids with favorable pKa values cannot be incorporated through direct synthesis. A combination of factors, including pKa, solubility, coordination strength, and the concentration of modulator used, likely influences the final extent of modulator loading achieved via direct synthesis.
12,15 while benzoic acid (BzA)12 and various amino acids14,15 could only be introduced into MOF-808 via the SALE method. This distinction cannot be explained solely by pKa values, as some amino acids with favorable pKa values cannot be incorporated through direct synthesis. A combination of factors, including pKa, solubility, coordination strength, and the concentration of modulator used, likely influences the final extent of modulator loading achieved via direct synthesis.
 | ||
| Fig. 1 Modulators grafted on MOF-808 via direct functionalization or PSE with their –COOH pKa values and loading per cluster. | ||
In both approaches, a wide range of functional groups can be introduced into MOF-808, allowing the framework to be tailored for specific applications. For instance, Lyu et al. incorporated a series of amino acids into MOF via SALE for CO2 capture.15 Similarly, MOF-808 can be tailored with specific functionalities based on the hard–soft-acid–base (HSAB) theory for selective recovery of precious and hazardous metals. Furthermore, Valverde et al. functionalized MOF-808 with amino acids according to HSAB and tested for the recovery of hard, intermediate and soft metal ions from aqueous solutions, demonstrating promising results.14 A previous study has utilized thioglycolic acid as a source of –SH groups in the direct functionalization of MOF-808.16 However, the loading per cluster in this method was reported to be only 2.6, which is relatively low. To further improve SH loading, this study introduces thiolactic acid as a new modulator for the direct functionalization of MOF-808. According to the HSAB theory, Ag(I), a soft acid, will strongly interact with –SH, a soft base.17 The enhancement in loading of –SH is expected to significantly improve the Ag(I) uptake capacity compared to previously reported MOFs.
Silver, a precious metal widely utilized in industries such as electronics, photovoltaics, jewelry, and medicinal applications, is experiencing high demand and increasing costs.18,19 This situation has necessitated the development of efficient recovery methods for silver. Among secondary sources, end-of-life solar panels represent a particularly relevant reservoir of silver. While several conventional technologies exist for precious metal recovery,19 only a limited number of studies have examined the adsorption behavior of silver on MOFs. To date, a few functionalized MOFs have been reported for Ag(I) capture, including –SH, –NH2 and –S containing MOFs such as HS-MIL-53(Al),20 UiO-66-MAc,21 UiO-66-Rd(Rhodanine),17 NH2-MIL-125(Ti),22 and UiO-66-NH2-Rd.23 Among these, the highest adsorption capacity of Ag(I) was reported to be of NH2-MIL-125(Ti) with 192.5 mg g−1. Additionally, the MOF-oligomer composite MIL-127/PoPD (poly-o-phenylenediamine), which possesses amine functionality, achieved an adsorption capacity of 560 mg g−1, whereas the polymer PoPD alone provided a capacity of 360 mg g−1.24 The enhanced adsorption capacity of MOFs is often associated with the density of accessible functional groups within their framework. Building on this principle, the present work aims to improve Ag(I) uptake by introducing a high loading of thiol functionalities into MOF-808. Considering that highly acidic modulators are more readily grafted and often achieve higher loadings, thiolactic acid was selected for –SH source. This study focuses on optimizing synthesis conditions, characterizing the thiol incorporation, and evaluating the material's capacity for Ag+ adsorption.
2. Experimental
2.1. Materials
Trimesic acid (H3BTC, 95%), zirconyl chloride octahydrate (ZrOCl2·8H2O, 98%), thiolactic acid (CH3CH(SH)COOH, 100%), lead(II) nitrate (Pb(NO3)2, 99%), thiourea (CH4N2S, 98%) and aluminum nitrate nonahydrate (Al(NO3)3·8H2O, 98%) were acquired from Sigma-Aldrich. Acetic acid (CH3COOH, 100%), nitric acid (HNO3, 65%), and silver nitrate (AgNO3) were supplied by Supelco. Hydrochloric acid (HCl, 37%), sodium thiosulfate pentahydrate (Na2S2O3·8H2O, 100%), and sodium hydroxide (NaOH, 100%) were supplied by VWR chemicals. All chemicals were used as procured, without further purification or modification.2.2. High-throughput synthesis of MOF-808
The high-throughput synthesis was carried out using a stainless-steel block containing 24 reaction slots and a 4 ml screw-capped glass vial capable of withstanding high temperatures.MOF-808 procedure was adopted from the literature12 with some modifications. Initially, a specific amount of ZrOCl2·8H2O was dissolved in distilled water in a 4 ml screw-capped glass vial. An appropriate amount (10–50 molar equivalents with respect to Zr) of acetic acid (AA) or thiolactic acid (ThA) was subsequently added to the solution. The mixture was stirred for a few minutes to achieve homogeneity. A specific quantity of H3BTC was added, and the reaction mixture was heated for 72 h at 95 °C in a closed vial with stirring. The resulting solid product was separated by centrifugation and thoroughly washed four times with distilled water and twice with acetone. The final product was dried in an oven overnight at 60 °C. To investigate the effect of the amount of modulator, 10–50 molar equivalents of acetic acid and thiolactic acid were tested as indicated in Tables S1 and S2, respectively. M-ThA-X and M-AA-X are used as abbreviations for MOF-808-thiolactic acid and MOF-808-acetic acid, respectively. X denotes the molar equivalent of the modulator used.
2.3. Scale-up synthesis of MOF-808-ThA
The scale-up synthesis for 10, 25, and 40 molar equivalents was carried out in a closed round-bottom flask with stirring. The procedure was adopted from high-throughput synthesis.2.4. Characterization
PXRD was measured using Bruker D8 advance with Cu-Kα radiation with a wavelength of 1.5418 Å. The high-throughput samples were prepared using ethanol slurry and transferred to XRD plate with silicon wafers. The samples were measured in Bragg–Brentano geometry (40 kV, 25 mA) with a scan speed of 5.77° min−1 and 2θ range of 2–70°. The scaled-up synthesis samples were measured with a scan speed of 1.19° min−1 and 2θ range of 2–70°. TGA was performed using Mettler-Toledo TGA/DSC 3+ STARe system under synthetic air flow with 25 ml min−1 and a heating rate of 5 °C min−1 from room temperature to 900 °C. The morphology of MOF and the metal content were measured through SEM-EDS analysis using Jeol JSM-IT800 equipped with Oxford EDS detector. Surface area and porosity were studied using N2 sorption isotherms which were measured using Micromeritics TriStar II plus at 77 K. M-ThA-X was degassed at 120 °C for 3 h whereas M-AA-X was degassed at 160 °C for 3 h before the analysis. H1NMR spectra were recorded using Bruker Ascend™ 400Hz NMR spectrometer. Around 20 mg of MOF was digested in 1 ml of 1 M NaOH in D2O before NMR analysis. Ex situ Raman spectra were collected using a Renishaw inVia confocal Raman microscope equipped with a 532 nm laser, covering a sample area of approximately 60 μm, with a spectral resolution of 0.5 cm−1. Depending on the fluorescent background, exposure time and laser power were adjusted to optimize the signal-to-noise ratio, varying between 60–300 s and 25–2.5 mW, respectively. Data analysis was conducted using Wire 5.2 software for peak fitting, cosmic ray removal, background removal when necessary, and normalization. Zeta potential was measured using the Zetasizer Advanced Series Lab. 200 mg of M-ThA-25 was dispersed in 200 ml Ethanol. It was sonicated for a few minutes for better dispersion. 15 ml of dispersed solution was taken in a glass vial, and pH was changed using buffer solutions. ICP analysis was done using an Agilent 5800 ICP-OES instrument, which includes an SPS 4 autosampler. The samples were diluted utilizing a 2% HNO3 solution to ensure that the concentrations remain within the calibration range of 0 to 10 mg L−1. Transmission electron microscopy (TEM) was conducted using JEOL-2100 (LaB6 filament), which operates at 200 kV. The TEM is also equipped with Energy Dispersive Spectroscopy (EDS) for analysing composition.Synchrotron PXRD was performed at the BM31 beamline of the European Synchrotron Radiation Facility (ESRF). A small quantity of the powder sample was loaded into a 0.5 mm diameter capillary. The beam size used was 2 μm × 50 μm, and the wavelength (λ) was 0.025448 nm. In situ temperature stability was maintained with a heating ramp rate of 6 °C min−1 in an ambient atmosphere in the 22–227 °C range. Data integration was carried out using the SNBL toolbox software.25 XPS analyses were carried out by a Thermo Scientific K-Alpha XPS with Al K-alpha monochromatic radiation (1486.3 eV) using 400 μm X-ray spot size. The pass energy was set to 50 eV, corresponding to an energy resolution of roughly 0.5 eV. The take-off angle was set to 90°. All measured peaks were deconvoluted and fit by using avantage 5.9 software. The C 1s peak at 284.5 eV was designated for the charge correction and peak assessment.
2.5. Adsorption studies
M-ThA-25 was activated at 120 °C for 3 h, and M-AA-50 was activated at 160 °C for 3 h in oven before adsorption experiments. Various concentrations of Ag+ ions were prepared by dissolving appropriate quantities of silver nitrate (AgNO3) in distilled water. Batch adsorption experiments were conducted by adding 50 mg of activated MOF into 10 ml of the metal ion solution and it was stirred at room temperature (RT) for 24 h. The mixture was centrifuged at 4000 rpm for 5 min. The supernatant was collected and analyzed using ICP-OES to determine the residual metal ion concentration. The effect of pH on adsorption efficiency was investigated under two different conditions: (1) by adjusting the pH of the initial 500 ppm Ag+ solution before the addition of the MOF, and (2) by altering the pH after the MOF was introduced into the silver solution. The pH adjustments were performed using 0.1 M HNO3 and 0.1 M NaOH, respectively.The adsorption capacity (qeq, mg g−1) and removal efficiency (R%) were calculated using eqn (1) and (2), respectively.
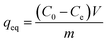 | (1) |
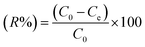 | (2) |
Adsorption isotherm: 50 mg of activated MOF was added to 10 ml of aqueous solution with varied concentrations of Ag+. The mixture was stirred at RT for 1 h. The sample was collected through centrifugation and filtered using a 0.22 μm PVDF membrane filter prior to ICP analysis. The data was compared with the Langmuir and the Freundlich models. The Langmuir adsorption isotherm presumes a homogeneous adsorption surface, wherein adsorbed species exhibit no interactions among them. It is expressed as:
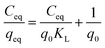 | (3) |
Furthermore, the Freundlich isotherm, addressing multilayer adsorption, is expressed as follows:
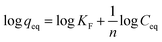 | (4) |
The equation for the Sips model, which combines both the Langmuir and Freundlich models, is expressed as:
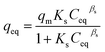 | (5) |
Kinetic study: 50 mg of activated MOF was dispersed in 10 ml of 500 ppm Ag+ solution. Before adding MOF, the solution's pH was set to 2, 3, and 4 to investigate the correlation of pH with kinetics. The mixture was stirred at room temperature for the desired time. The sample was collected through centrifugation and filtered with a 0.22 μm PVDF membrane filter prior to ICP analysis. The pseudo-first-order (eqn (6)) and pseudo-second-order kinetic models (eqn (7)) describe the adsorption kinetics of the Ag+ ion.
In(qe − qt) = In![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) qe − k1t qe − k1t | (6) |
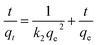 | (7) |
The intra-particle diffusion model (Weber–Morris model) (eqn (8)) helps in determining the rate-limiting step in the adsorption kinetics.
| qt = kit0.5 + C | (8) |
2.6. Selectivity study
To assess the selectivity of M-ThA-25 towards Ag, a mixed metal solution was prepared, comprising Pb2+, Ag+, and Al3+ using respective nitrate salts at concentrations of 21.4 ppm, 200 ppm, and 1440 ppm, respectively, as found in the actual solar panel leachate.26 50 mg of activated MOF was added to 10 ml of this solution without altering the pH. The resultant mixture was stirred for 2 h at 25 °C. Subsequently, the solution was filtered using a 0.22 μm PVDF membrane filter before ICP analysis.2.7. Regeneration test
The adsorption cycles were performed by adding 100 mg of activated MOF to 20 ml of 500 ppm of Ag+ solution, which was stirred for 30 min at 25 °C. The desorption experiment procedure was adopted from the literature with some modifications.23,27,28 The experiments were conducted using 10 ml of 0.4 M sodium thiosulfate solution, stirred at 25 °C for 2 h. For comparison, 10 ml of 0.1 M HNO3 containing 0.1% thiourea was also used for the desorption study up to two cycles. After every adsorption–desorption cycle, the MOF was washed multiple times with distilled water and dried overnight at 60 °C before the next cycle.3. Results
3.1. High-throughput synthesis and characterization
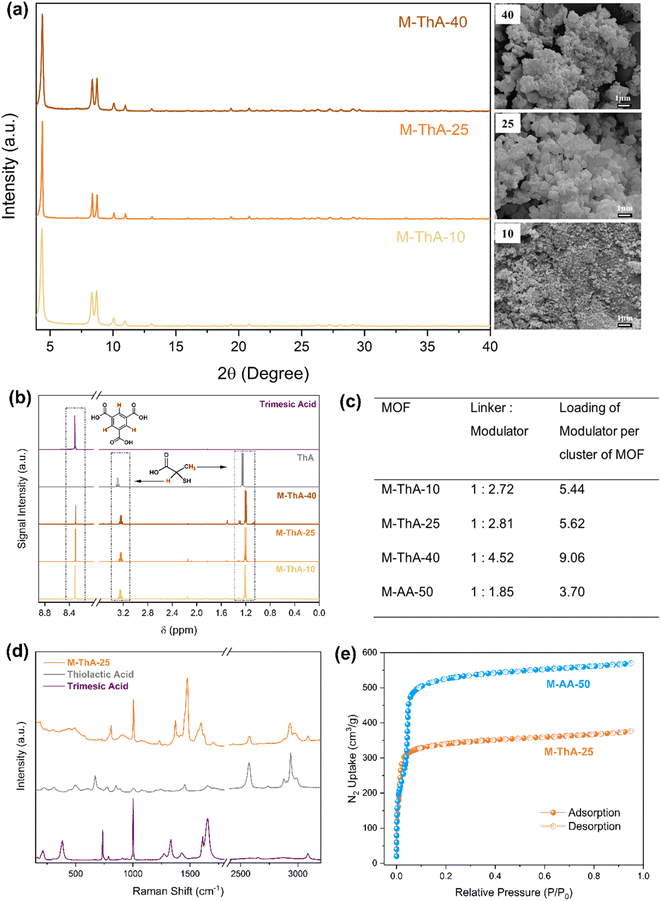
The incorporation of thiol-functionalized modulator onto MOF-808 was investigated via a one-pot synthesis approach outlined in Fig. 2a. The effects of various molar equivalents of thiolactic acid (ThA), ranging from 10 to 50, were investigated using the high-throughput setup depicted in Fig. 2b. For comparison, a parallel synthesis was conducted using acetic acid (AA) as a modulator. It was observed that the modulator plays a critical role in the synthesis of MOF-808. Without the modulator, gel formation occurred, which could be due to a rapid nucleation process, resulting in the agglomeration of nanocrystals as reported by Hou et al.29 Additionally, increasing the modulator quantity beyond 30 equivalents resulted in a noticeably lower yield. The X-ray diffraction (XRD) peaks of MOF-808-ThA (M-ThA) and MOF-808-AA (M-AA) are shown in Fig. 2c and S1, respectively. The XRD patterns of both M-ThA and M-AA exhibit all the characteristic peaks with no shift relative to the simulated XRD of MOF-808. The consistency of the XRD results with previously reported data in the literature12 confirms the successful formation of pure MOF-808, regardless of the type or amount of modulator used. The average lattice parameters of M-AA and M-ThA unit cells were approximately 35.33 ± 0.02 and 35.19 ± 0.06 Å, respectively, which closely matched the reported values.30 However, slight peak broadening was observed at lower 2θ in M-ThA-10 and M-ThA-20, linked to the smaller crystallite size visible in the SEM images in Fig. S2.The SEM images demonstrate that as the quantity of modulator increases, the particle size increases. However, at 40 and 50 equivalents of ThA, a heterogeneous distribution of particle sizes was observed. This suggests that higher concentrations of ThA may require longer reaction times to achieve a uniform crystal size distribution. At lower equivalents (10 to 20) of ThA, the particles display a uniform morphology, albeit with smaller sizes below 200 nm. It was clearly observed that at higher modulator concentrations, the synthesis solution remained clear for a longer duration. In contrast, at lower modulator concentrations, the solution became turbid more rapidly, leading to the earlier formation of a white precipitate. This behavior can be attributed to the role of modulators in slowing down nucleation and crystal growth.31
3.2. Characterization of scale-up synthesis
To investigate how different amounts of modulator affect the yield and loading of ThA in MOF, three syntheses were scaled up using 10, 25, and 40 molar equivalents of ThA. The quantities of reagents and the percentage yield for the scaled-up synthesis are provided in Table S3. The lower yields of M-ThA-10 and M-ThA-40 may result from an insufficient or excessive amount of a modulator, which influenced the kinetics of crystallization. In contrast, M-ThA-25 achieved the highest yield of 74.15%, suggesting that the modulator amount was suitable for MOF synthesis. Furthermore, up to 15 g of M-ThA-25 was produced with minimal effects on yield.The XRD pattern in Fig. 3a is identical to the one obtained in the high-throughput synthesis. Slight peak broadening was observed, like the high-throughput synthesis at lower 2θ in M-ThA-10, which is linked to the smaller particle size visible in the SEM images in Fig. 3a. In the upscaled synthesis, the particle size of M-ThA-10 is approximately 100 nm or less, but uniform in size, which agrees with the XRD pattern. M-ThA-25 has a uniform particle size (∼400 nm) with a clearly visible octahedral shape. However, M-ThA-40 is observed to have both large and small-sized particles, which corresponds to incomplete crystallization. This phenomenon may be attributed to the substantial quantity of the modulator present.
The presence of ThA and its incorporation into the MOF via upscaled synthesis were confirmed through NMR (Fig. 3b). For M-ThA-25, the ThA loading was calculated to be 5.62 per cluster, which is the closest to the maximum modulator loading for MOF-808. In M-ThA-10, the modulator loading was determined to be 5.44 per cluster, whereas in M-ThA-40, the loading exceeded the theoretically expected maximum for incorporation at a cluster. Considering the good yield, uniform particle size, and high loading of thiol functionality obtained for M-ThA-25, this material was further investigated for Ag+ uptake. The loading of the modulator is also nearly twice the reported amount of thioglycolic acid as a modulator.16 The Raman spectra are presented in Fig. 3d. The characteristic peak at 2580 cm−1 corresponds to the –SH stretching mode, indicating that the thiol group remains uncoordinated, making it available for metal adsorption.32,33 The bands observed at 1594 cm−1 and 1474 cm−1 are associated with the asymmetric and symmetric stretching modes of the carboxyl (–COOH) group present in trimesic acid, while the band at 1565 cm−1 is indicative of the asymmetric stretching of the carboxyl (–COOH) group from thiolactic acid.32,34,35 The additional characteristic bands and their respective assignments are presented in Table S4.
The N2 adsorption–desorption curve presented in Fig. 3e indicates a reduction in Brunauer–Emmett–Teller (BET) surface area when ThA is used as a modulator. Specifically, BET surface area dropped from 2065 m2 g−1 for M-AA-50 to 1357 m2 g−1 for M-ThA-25. Furthermore, M-AA-50 exhibits a two-step isotherm that transitions to a type 1 isotherm with a single step upon the introduction of ThA as a modulator as observed in the literature.12 The BET surface area of M-ThA-25 was higher than that reported for MOF-808 using thioglycolic acid (1260.9 m2 g−1)16 as a modulator. Since acetic acid and formic acid have smaller molecular sizes, they offer a greater surface area for MOF-808. Table S5 summarizes the BET surface area for commonly used modulators.
3.3. Thermal stability of M-ThA-25
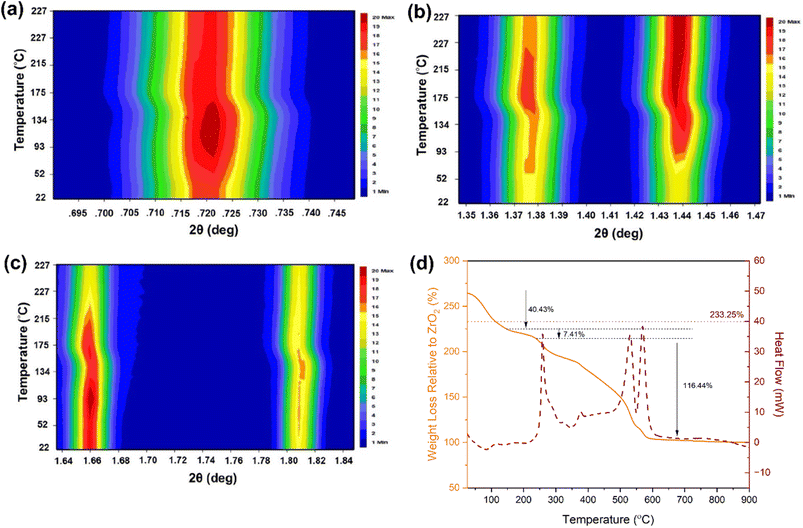
The stability of MOFs with respect to temperature and pH is a crucial property for adsorption experiments. The thermal stability of M-ThA-25 was examined using variable-temperature synchrotron PXRD, as depicted in a 2D contour plot in Fig. 4a–c, and TGA, as shown in Fig. 4d. The contour plot shows the intensity changes observed in the peaks at lower 2θ values corresponding to the (111) (Fig. 4a), (311) and (222) (Fig. 4b), and (400) and (311) (Fig. 4c) planes, as the temperature gradually increased. Both a change in the peak intensity and the shift towards lower angles were observed during the initial heating stage (up to 100 °C). This behavior is typically associated with the desorption of physisorbed solvent molecules (water and acetone) from the pores. This removal of these molecules reduces electron density, altering the scattering power and affecting peak intensities. These findings are further supported by TGS results, which show a corresponding weight and associated endothermic peaks observed in this temperature range.36,37Further increases in temperature (between 130 °C and 227 °C) lead to a significant downshift of all peaks, accompanied by a subsequent drop in intensity, and the emergence of new reflections at higher angles (Fig. S3). These changes indicate that M-ThA-25 undergoes a phase transition in this temperature range. Correspondingly, the TGA shows the first exothermic event in this temperature range, which is unique to M-ThA-25 and absent for M-AA-50 (Fig. S5). The strong exothermic signal, combined with negligible weight loss in the TGA, suggests some transformation of the modulator, potentially involving S–S bond formation that could alter the size of the unit cell. While this hypothesis requires further in-depth investigation, the results clearly show that the thiol functionality is retained up to 200 °C and the MOF structure remains intact up to 227 °C, as indicated by PXRD (Fig. S4).
At temperatures above 300 °C, the decomposition of the MOF initiates, resulting in the loss of the linker and residual modulator, ultimately, yielding ZrO2 as the final product.37–39 Notably, M-ThA-25 exhibits two distinct exothermic peaks in the decomposition region, whereas M-AA-50 shows only one (Fig. S5), highlighting their overall different thermal behaviors. The horizontal dotted line in TGA at 233.25% represents ideal weight percent plateau of M-ThA-25 (Zr6O4(OH)4(BTC)2(ThA)6). Experiment results show only minor deviations from this plateau, indicating the presence of very few structural defects in M-ThA-25. Detailed calculations of weight loss are presented in Tables S6 and S7 for M-ThA-25 and M-AA-50, respectively.
3.4. Chemical stability and adsorption studies
The chemical stability of MOF under different pH environments is a crucial parameter for metal adsorption studies. The PXRD patterns of the dried MOF samples after the pH stability tests are shown in Fig. 5a. The XRD patterns obtained at pH 2, 3, 4, and 5 display identical features, confirming the excellent stability of the framework under these conditions. Although most of the characteristic peaks remained present as the pH increased to 7 and 8, they became broader and less intense. At pH 10, a complete loss of crystallinity was observed, indicating full collapse of the framework. Based on these results, and to avoid the risk of AgOH precipitation40 under alkaline conditions, all adsorption experiments were conducted at pH values below 6. Another significant parameter is the surface charge, which is determined by the Zeta potential, and was examined for M-ThA-25 within a pH range of 2.0 to 5.0, as demonstrated in Fig. 5b. It was observed that the surface charge remains positive at pH values below 3.0 and transitions to negative at pH values exceeding 3.0. The point of zero charge (pHPZC) for M-ThA-25 is determined to be 3.01.The impact of pH on Ag+ adsorption at MOF was explored using two methods within a pH range of 2–6: (1) setting the pH of the initial solution before adding the MOF, and (2) altering the pH post-MOF addition to the metal solution. The equilibrium pH was recorded after 24 h of stirring and is shown in Fig. 5c and d. The pH of M-ThA-25 in the AgNO3 solution stabilized around 2.5. Importantly, the initial pH of the AgNO3 solution did not affect the final pH after the MOF was added, which consistently fell to 2.4–2.5 (Fig. 5c). The pH of the AgNO3 solution before changing ranged between 5.2 and 5.4. Therefore, when the initial pH was set between 2 and 5, extra H+ ions were introduced. Since MOF-808 contains acidic protons in its cluster, the pH remained stable at 2.4 to 2.5, regardless of the initial pH of the solution. However, when the pH was adjusted after adding MOF to the solution, the equilibrium pH dropped by 1 unit due to the introduction of more OH− ions, as the initial pH of the mixture was approximately 2.5. These findings suggest that the method used to establish the initial pH can significantly impact adsorption studies that depend on pH.
Despite differences in pH control strategy, final Ag+ uptake remained unaffected. This indicates that the thiol functionality grafted onto MOF-808 remains unaffected by pH and exhibits a high affinity for Ag, in line with the hard–soft-acid–base (HSAB) theory.41 The color of the M-ThA-25 changed from white to beige and brown upon adsorption, as shown in Fig. S6. The color change in thiol-functionalized MOF can serve as an indicator for Ag adsorption. According to the Zeta potential data, the surface charge of M-ThA-25 remained negative above pH 3. However, no difference in adsorption capacity was observed at both positive and negative Zeta potentials due to the affinity of Ag for –SH. It also indicates that surface charge doesn't play any role in the adsorption of Ag+ ions. A similar pH- and surface charge-independent adsorption of Ag has also been reported previously by Xue et al.24 To assess the influence of functional groups on adsorption behavior, similar experiments were conducted using M-AA-50, where the pH was adjusted post-MOF addition to minimize changes in the desired pH. This MOF also exhibited 100% removal efficiency in the absence of any functional group, as shown in Fig. S7. Post-adsorption characterization was conducted to determine whether Ag+ was adsorbed or precipitated as AgCl. M-AA-50 contains Cl impurities based on the SEM-EDS analysis presented in Fig. S8, which indicates that Ag was precipitated as AgCl instead of being adsorbed.
3.5. Adsorption kinetics
To further assess the impact of pH on adsorption capacity, kinetic studies were conducted at initial pH levels of 2, 3, and 4 for AgNO3 solutions. The adsorption process demonstrated rapid kinetics, achieving a complete removal efficiency in less than 10 minutes for all pH values, indicating that pH does not affect kinetics. The kinetic data were analyzed using both pseudo-first-order and pseudo-second-order models, as shown in Table 1. The adsorption capacity as a function of time exhibited an exceptional fit to the pseudo-second-order kinetic model, with correlation coefficients (R2) ranging from 0.999 to 1 across all pH levels (Fig. 6a, b, S10 and S11), thereby indicating that the adsorption of Ag+ onto SH predominantly occurs through chemisorption.22 The adsorption data were further analyzed using the intra-particle diffusion model to identify the rate-limiting step. According to the Weber–Morris model, if the plot of qtvs. t0.5 is linear and passes through the origin, intra-particle diffusion is the sole rate-controlling mechanism. However, in this study, the plot (Fig. S12a–c) was neither linear nor did it pass through the origin, indicating that boundary layer (film) diffusion plays a dominant role.22,42 Thus, adsorption proceeded rapidly and was primarily governed by external film diffusion, resulting in a quick equilibrium. Moreover, the adsorption kinetics observed in this study were remarkably fast compared to those of previously reported MOFs,17,22,23 highlighting the material's efficiency in achieving rapid uptake due to the presence of abundant active sites. The positive surface charge observed from Zeta potential measurements may act as an initial diffusion barrier for Ag+ approaching the MOF surface. However, once Ag+ ions reach the surface, the large pore size and readily accessible thiol groups facilitate rapid intraparticle diffusion, enabling efficient adsorption.| Sorbent (pH) | Pseudo first order model | Psuedo second order model | |||||
|---|---|---|---|---|---|---|---|
| q e (mg g−1) | k 1 (min−1) | R 2 | q e (mg g−1) | k 2 (g mg−1 min−1) | R 2 | ||
| M-ThA-25 (pH 2) | 1.88 | −3.2 × 10−6 | 0.0141 | 86.96 | 0.0088 | 1 | |
| M-ThA-25 (pH 3) | 1.56 | −8.3 × 10−7 | 0.0304 | 89.29 | 0.0291 | 1 | |
| M-ThA-25 (pH 4) | 1.65 | −4.6 × 10−7 | 0.0108 | 89.29 | 0.0104 | 0.9997 | |
3.6. Adsorption isotherm
The adsorption isotherm (Fig. 6c) was obtained using initial Ag+ concentrations ranging from 300 ppm to 6000 ppm. The data were analyzed using the Langmuir, Freundlich, and Sips models. The Langmuir data provided the best fit (Fig. 6d, R2 = 0.9988), indicating monolayer adsorption of Ag+ by M-ThA-25. The Freundlich model (Fig. S13) was applied to assess the possibility of adsorption occurring at heterogeneous sites such as the Zr-cluster of the MOF; however, the fit was poor, indicating negligible contribution from such sites. The Sips model, which combines features of both Langmuir and Freundlich equations, offered a reasonable fit, with the heterogeneity parameter n < 1 suggesting some degree of surface heterogeneity. Nevertheless, the dominance of the Langmuir fit underscores monolayer adsorption as the prevailing process. The corresponding model parameters are summarized in Table 2. Based on the Langmuir analysis, the maximum adsorption capacity of M-ThA-25 for Ag+ was determined to be 365.47 mg g−1, which is close to the theoretically calculated adsorption capacity of MOF (351.5 mg g−1). The adsorption capacity surpasses the values reported for thiol or –S– functionalized MOFs, including HS-MIL-53(Al),20 UiO-66-MAc,21 UiO-66-Rd,17 and UiO-66-NH2-Rd.23 A detailed comparison is provided in Table S8. These findings demonstrate that M-ThA-25 offers high silver uptake capacity, highlighting its potential as an efficient adsorbent.| Langmuir | Freundlich | Sips | |||||||
|---|---|---|---|---|---|---|---|---|---|
| b (L mg−1) | q max (mg g−1) | R 2 | K F | n | R 2 | q max (mg g−1) | K | n | R 2 |
| 0.285 | 365.47 | 0.9988 | 225.50 | 13.33 | 0.5685 | 367.44 | 13.676 | 0.678 | 0.9801 |
3.7. Post adsorption analysis
The samples were analyzed using PXRD, and SEM to determine whether the Ag ions are adsorbed or precipitated. The PXRD pattern of M-ThA-25 (Fig. 7a) shows no additional peaks or significant changes in intensity compared to the pristine MOF and the Ag+ adsorbed MOF. Conversely, the PXRD pattern of M-AA-50 reveals notable changes, including the emergence of new peaks at higher 2θ values. To compare these peaks with the AgCl precipitate, the AgNO3 solution was treated with HCl, and the PXRD pattern of the resulting precipitate was recorded. This confirms the presence of AgCl in M-AA-50, attributed to the presence of chlorine in the framework. However, no such precipitation was observed in M-ThA-25 post-adsorption. These findings suggest that Ag+ was successfully adsorbed by M-ThA-25 due to its affinity for thiol groups. This conclusion is further supported by the SEM images shown in Fig. 7b–e, which demonstrate the morphological differences between pristine MOF and MOF with Ag.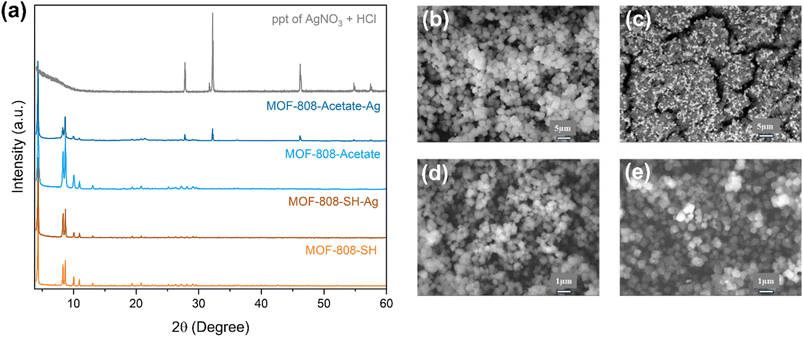 | ||
| Fig. 7 (a) PXRD pattern of M-ThA-25 and M-ThA-50 post Ag+ adsorption experiment at pH 3 (pH change after adding MOF). SEM images of (b) M-AA-50 (c) M-AA-50 with Ag (d) M-ThA-25 (e) M-ThA-25 with Ag. | ||
The SEM images of pristine M-AA-50 (Fig. 7b) reveal an octahedral morphology, which is characteristic of the parent MOF structure. Upon treatment with Ag+ ions (Fig. 7c), a distinct surface layer attributed to AgCl precipitate is observed. This observation is corroborated by EDS mapping data (Fig. S14), which demonstrates a spatial overlap between Ag and Cl signals, confirming the formation and deposition of AgCl on the MOF surface. In contrast, M-ThA-25 does not exhibit such surface precipitation following Ag+ adsorption (Fig. 7d and e), and its morphological features remain intact. EDS mapping of Ag-loaded M-ThA-25 (Fig. S15) reveals colocalization of Ag and S, suggesting a strong interaction between Ag+ and thiol groups. Although trace amounts of Cl are detected, no corresponding AgCl phase is evident in the XRD patterns, further supporting the conclusion that Ag+ is primarily adsorbed via coordination with thiol functionalities rather than precipitating as AgCl.
3.8. Adsorption mechanism
The adsorption mechanism and the oxidation state of the adsorbed Ag were studied using XPS analysis, as presented in Fig. 8a–d. The wide-scan XPS spectra of M-ThA-25-Ag (Fig. 8b) exhibit the characteristic peak of Ag 3d; however, it is nearly undetectable in M-AA-50 (Fig. 8a), suggesting that Ag is adsorbed onto M-ThA-25. The S 2p binding energy of M-ThA-25 (Fig. 8c) shifted from 163.3 eV to 162.9 eV upon Ag+ adsorption, signifying a strong interaction between S and Ag atoms.39 The two peaks of S 2p spectra at 164.38 eV and 163.18 eV correspond to S 2p1/2 and S 2p3/2, respectively,39 in M-ThA-25 (Fig. 8c). In addition to these peaks, two new peaks emerge after Ag adsorption at 162.78 eV and 161.58 eV, corresponding to S 2p1/2 and S 2p3/2, respectively, in M-ThA-25-Ag. The Ag 3d XPS was recorded for M-ThA-25 to determine the oxidation state of adsorbed Ag (Fig. 8d). The two principal split peaks at 367.98 eV and 374.08 eV correspond to Ag 3d5/2 and Ag 3d3/2, respectively.24,43 These peaks are ascribed to Ag–S–R, with Ag in the +1 oxidation state.24 Additionally, two other split peaks of low intensity are observed at 369.48 eV and 375.48 eV, which represent metallic Ag0. In the adsorption experiment with respect to pH, the pH of the solution drops when M-ThA-25 was added, indicating the ion exchange mechanism for Ag adsorption. This aligns well with the XPS data, which represents Ag+ coordinated with thiols. The presence of metallic Ag0 can be attributed to the formation of AgCl precipitates, arising from trace Cl− impurities in M-ThA-25 as confirmed by EDS (Fig. S9). Under light exposure, AgCl may undergo photoreduction to yield Ag0. TEM images of M-ThA-25 after Ag+ adsorption (Fig. S16a) clearly reveal the presence of a small quantity of Ag nanoparticles distributed on the MOF surface. The high-resolution TEM (Fig. S16b) further confirms the presence of Ag0 through a lattice spacing of 2.32 Å, corresponding to the (111) plane of metallic silver.22 In contrast, TEM images of M-AA-50 (Fig. S16c) show significant agglomeration of Ag, indicating precipitation.3.9. Selectivity and regeneration
The selectivity of M-ThA-25 toward Ag+ was assessed by examining its adsorption behavior in a mixed-ion system containing Pb2+, Ag+, and Al3+ at weight ratios representative of those found in end-of-life solar panels.26 Since the surface charge did not significantly affect the adsorption capacity, the experiment was conducted without any pH adjustment. As illustrated in Fig. 9a, both Ag+ and Pb2+ ions were adsorbed entirely by the MOF, with no noticeable selectivity observed. In contrast, no uptake of Al3+ was detected, and its concentration remained unchanged after the adsorption experiment. Due to the strong affinity of Ag and Pb with thiol groups,41,44 both metal ions were completely removed. The concentration of Pb2+ in the simulated solution is significantly less compared to Ag+. The dynamic experiment could further separate Ag and Pb.Regeneration of adsorbed ions is crucial for developing sustainable metal recovery technology. Therefore, in this study, silver recovery from adsorbed MOF was attempted using two different desorbents: sodium thiosulfate solution (Fig. 9b) and HNO3 with thiourea (Fig. S17). The percentage of removal decreased with each cycle during the adsorption process due to the saturation of the MOF's uptake capacity. Conversely, desorption is noted to be exceedingly low, albeit showing a gradual increase with each subsequent cycle. Nevertheless, both cycles of adsorption exhibited comparable removal rates when utilizing HNO3–thiourea as the desorbent. The results of desorption were also consistent across both cycles. In this study, both desorbents were unsuccessful in completely removing adsorbed Ag. The concentration of desorbents used is lower than typically reported or used; therefore, a higher concentration of desorbent is required for efficient desorption. In previous studies,17,23,28,45 the regeneration of silver has primarily been evaluated based on adsorption efficiency or removal percentage of silver from the initial solution rather than desorption performance. The reported multi-cycle adsorption capabilities are often attributed to the fact that regeneration experiments are conducted using silver concentrations lower than the maximum adsorption capacity of the adsorbent. This approach enables continued adsorption over multiple cycles but does not necessarily reflect the actual regeneration efficiency. Furthermore, the absence of desorption data in these studies limits the ability to accurately evaluate and compare the regeneration performance with the findings presented in this work. Therefore, further optimization studies are needed to investigate desorption with higher concentrations of desorbents.
4. Conclusion
In this study, thiol-functionalized MOF-808 was successfully synthesized via a one-pot approach using thiolactic acid as a modulator. Structural characterization by NMR and Raman spectroscopy confirmed the successful incorporation of thiol groups into the MOF framework. The M-ThA-25 material demonstrated thermal stability up to 220 °C, as evidenced by in situ PXRD and TGA analyses. Increased thiol loading was directly correlated with enhanced silver adsorption capacity, with M-ThA-25 exhibiting a maximum uptake of 365.47 mg g−1—one of the highest values reported for MOF-based adsorbents. Moreover, Ag(I) adsorption was found to be pH-independent, indicating an ion-exchange mechanism as supported by XPS analysis. The adsorption kinetics were notably rapid, achieving complete (100%) removal of Ag(I) in less than 10 minutes. The material also showed high selectivity for Ag(I) and Pb(II) over Al(III) in a simulated solution representative of the metal composition found in end-of-life solar panels. However, further investigation is required to optimize the regeneration and recovery of adsorbed silver. The regeneration process could potentially be improved by employing desorbents with a high concentration of thiol groups or reported potential desorbents such as ascorbic acid, thereby enhancing the material's applicability for efficient Ag recovery from waste streams. Overall, this work establishes MOF-808-ThA as a highly efficient and scalable platform for silver adsorption, combining fast kinetics, high adsorption capacity, and robust structural stability under application-relevant conditions.Author contributions
Simmy Rathod: conceptualization, investigation and writing – original draft. Sofiia Bercha: investigation, formal analysis, writing – review and editing. M. Baris Yagci: resources and investigation. Duygu Yilmaz: resources and writing – review and editing. Olena Zavorotynska: resources and validation. Sachin Maruti Chavan: conceptualization, funding acquisition, project administration, and writing – review & editing. All authors contributed to the overall discussion of the results and assisted in preparing the manuscript.Conflicts of interest
There are no conflicts to declare.Data availability
The original/raw data for this article, including X-ray diffraction, Raman and IR Spectra, N2 sorption at 77K, NMR, XPS and SEM images are available at National Science Archive (NVA) at https://sikt.no/tjenester/nasjonalt-vitenarkiv-nva according to the policy at the University of Stavanger.The data supporting this article have been included as part of the supplementary information (SI). Supplementary information: Fig. S1–S17 and Tables S1–S8. See DOI: https://doi.org/10.1039/d5ta06248a.
Acknowledgements
The primary research work presented was supported by the MOFsorbMET project (code 314746), funded by the Norwegian Research Council. Additional analysis was made possible through access from the RETRIEVE project (EU HE, Grant Agreement No. 101122332), which provides insights into silver recovery from photovoltaic waste. The authors would like to acknowledge Wakshum Mekonnen Tucho, University of Stavanger, for his contributions to the SEM-EDS and TEM analyses of all the samples. Synchrotron PXRD was performed at the BM31 beamline of the European Synchrotron Radiation Facility (ESRF) under proposal number A31-1-229. The authors gratefully acknowledge Dr Dragos Stoian for his valuable assistance during the XRD measurements.References
- H. Furukawa, K. E. Cordova, M. O'Keeffe and O. M. Yaghi, Science, 2013, 341, 1230444 CrossRef.
- O. M. Yaghi, G. Li and H. Li, Nature, 1995, 378, 703–706 CrossRef CAS.
- J. Wen, Y. Fang and G. Zeng, Chemosphere, 2018, 201, 627–643 CrossRef CAS PubMed.
- F. Ayesh Alharthi, A. A. Abutaleb, M. M. Aljohani and M. Alshareef, Comments Inorg. Chem., 2025, 45, 245–282 CrossRef CAS.
- Z. Han, J. Rushlow, Y. Yang, J. Huo, W. Shi and H.-C. Zhou, Inorg. Chem. Front., 2024, 11, 8589–8601 RSC.
- J. Piątek, T. M. Budnyak, S. Monti, G. Barcaro, R. Gueret, E. S. Grape, A. Jaworski, A. K. Inge, B. V. M. Rodrigues and A. Slabon, ACS Sustain. Chem. Eng., 2021, 9, 9770–9778 CrossRef.
- P. Bhadane, D. Menon, P. Goyal, M. Reza Alizadeh Kiapi, B. Kanta Satpathy, A. Lanza, I. Mikulska, R. Scatena, S. Michalik, P. Mahato, M. Asgari, X. Chen, S. Chakraborty, A. Mishra, I. Lynch, D. Fairen-Jimenez and S. K. Misra, Sep. Purif. Technol., 2025, 360, 130946 Search PubMed.
- Z. Zuhra, M. Haris Khan, S. Ali, M. Ahmad and Z. Zhaowei, Chem. Eng. J., 2024, 500, 157105 Search PubMed.
- M. Taddei, Coord. Chem. Rev., 2017, 343, 1–24 Search PubMed.
- C. L. Hobday, R. J. Marshall, C. F. Murphie, J. Sotelo, T. Richards, D. R. Allan, T. Düren, F.-X. Coudert, R. S. Forgan, C. A. Morrison, S. A. Moggach and T. D. Bennett, Angew. Chem., Int. Ed., 2016, 55, 2401–2405 CrossRef CAS.
- D. Feng, Z.-Y. Gu, J.-R. Li, H.-L. Jiang, Z. Wei and H.-C. Zhou, Angew. Chem., Int. Ed., 2012, 51, 10307–10310 CrossRef CAS PubMed.
- E. Aunan, C. W. Affolter, U. Olsbye and K. P. Lillerud, Chem. Mater., 2021, 33, 1471–1476 CrossRef CAS.
- R. Thür, N. Van Velthoven, V. Lemmens, M. Bastin, S. Smolders, D. De Vos and I. F. J. Vankelecom, ACS Appl. Mater. Interfaces, 2019, 11, 44792–44801 Search PubMed.
- A. Valverde, G. I. Tovar, N. A. Rio-López, D. Torres, M. Rosales, S. Wuttke, A. Fidalgo-Marijuan, J. M. Porro, M. Jiménez-Ruiz, V. García Sakai, A. García, J. M. Laza, J. L. Vilas-Vilela, L. Lezama, M. I. Arriortua, G. J. Copello and R. Fernández de Luis, Chem. Mater., 2022, 34, 9666–9684 CrossRef CAS.
- H. Lyu, O. I.-F. Chen, N. Hanikel, M. I. Hossain, R. W. Flaig, X. Pei, A. Amin, M. D. Doherty, R. K. Impastato, T. G. Glover, D. R. Moore and O. M. Yaghi, J. Am. Chem. Soc., 2022, 144, 2387–2396 CrossRef CAS.
- X. Zhao, L. Pei, Y.-N. Zhang, H. Huang, X. Zheng, B. Liu and M. Tong, Green Chem. Eng., 2022, 3, 405–412 CrossRef CAS.
- L. Ding, M. Xiong, X. Yin, L. Fang, L. Liu, W. Ren, F. Deng and X. Luo, Sep. Purif. Technol., 2023, 314, 123541 CrossRef CAS.
- K. Komoto, J.-S. Lee, J. Zhang, D. Ravikumar, P. Sinha, A. Wade and G. A. Heath, End-of-life Management of Photovoltaic Panels: Trends in PV Module Recycling Technologies, National Renewable Energy Laboratory (NREL), Golden, CO (United States), 2018 Search PubMed.
- A. Divya, T. Adish, P. Kaustubh and P. S. Zade, Sol. Energy Mater. Sol. Cells, 2023, 253, 112151 CrossRef CAS.
- X. Cheng, M. Liu, A. Zhang, S. Hu, C. Song, G. Zhang and X. Guo, Nanoscale, 2015, 7, 9738–9745 RSC.
- B. Moll, T. Müller, C. Schlüsener, A. Schmitz, P. Brandt, S. Öztürk and C. Janiak, Mater. Adv., 2021, 2, 804–812 RSC.
- X. Ren, C.-C. Wang, Y. Li, C.-Y. Wang, P. Wang and S. Gao, Chem. Eng. J., 2022, 442, 136306 CrossRef CAS.
- L. Ding, P. Shao, Y. Luo, X. Yin, S. Yu, L. Fang, L. Yang, J. Yang and X. Luo, Chem. Eng. J., 2020, 382, 123009 CrossRef CAS.
- T. Xue, L. Peng, O. A. Syzgantseva, M. A. Syzgantseva, P. Guo, H. Lai, R. Li, J. Chen, S. Li, X. Yan, S. Yang, J. Li, B. Han and W. L. Queen, Angew. Chem., Int. Ed., 2023, 62, e202309737 CrossRef CAS.
- K. Klementiev, K. Norén, S. Carlson, K. S. Clauss and I. Persson, J. Phys.: Conf. Ser., 2016, 712, 012023 CrossRef.
- J. M. Mwase, S. Wahl, S. Meyer and D. Yilmaz, Processes, 2024, 12, 2454 CrossRef CAS.
- Q. Wang, M. Li, M. Xi, M. Zhao, X. Wang, X. Chen and L. Ding, Toxics., 2024, 12, 351 CrossRef CAS PubMed.
- C. Hu, S. Kang, B. Xiong, S. Zhou and K. Tang, Environ. Sci. Pollut. Res., 2019, 26, 14214–14225 CrossRef CAS.
- J. Hou, A. F. Sapnik and T. D. Bennett, Chem. Sci., 2020, 11, 310–323 RSC.
- K. D. Nguyen, N. T. Vo, K. T. Le, K. V. Ho, N. T. Phan, P. H. Ho and H. V. Le, New J. Chem., 2023, 47, 6433–6447 RSC.
- D. Jiang, C. Huang, J. Zhu, P. Wang, Z. Liu and D. Fang, Coord. Chem. Rev., 2021, 444, 214064 CrossRef CAS.
- A. Kudelski, Surf. Sci., 2002, 502–503, 219–223 CrossRef CAS.
- F. C. Marques, G. P. Oliveira, R. A. R. Teixeira, R. M. S. Justo, T. B. V. Neves and G. F. S. Andrade, Vib. Spectrosc., 2018, 98, 139–144 CrossRef CAS.
- K. Kumar and S. Murugesan, J. Saudi Chem. Soc., 2018, 22, 16–26 CrossRef CAS.
- R. Paz, H. Viltres, N. K. Gupta, A. Romero-Galarza and C. Leyva, Environ. Sci.: Adv., 2022, 1, 182–191 CAS.
- Y. Arellano, C. Pazo, V. Roa, Y. Hidalgo-Rosa, X. Zarate, J. Llanos, N. Escalona and E. Schott, Dalton Trans., 2024, 53, 16397–16406 RSC.
- P.-H. Chang, C.-Y. Chen, R. Mukhopadhyay, W. Chen, Y.-M. Tzou and B. Sarkar, J. Colloid Interface Sci., 2022, 623, 627–636 CrossRef CAS PubMed.
- Y. Tao, B. Yang, F. Wang, Y. Yan, X. Hong, H. Xu, M. Xia and F. Wang, Sep. Purif. Technol., 2022, 300, 121825 CrossRef CAS.
- C. Ji, Y. Ren, H. Yu, M. Hua, L. Lv and W. Zhang, Chem. Eng. J., 2022, 430, 132960 CrossRef CAS.
- M.-Y. Sun, L. Zhang, Y. Li, C.-C. Wang, P. Wang, X. Ren and X.-H. Yi, Chin. Chem. Lett., 2025, 36, 110035 CrossRef CAS.
- R. G. Pearson, J. Am. Chem. Soc., 1963, 85, 3533–3539 CrossRef CAS.
- K. H. Chu, M. A. Hashim, M. H. Zawawi and J.-C. Bollinger, J. Environ. Chem. Eng., 2025, 13, 117266 CrossRef CAS.
- A. Regiel-Futyra, M. Kus-Liśkiewicz, V. Sebastian, S. Irusta, M. Arruebo, A. Kyzioł and G. Stochel, RSC Adv., 2017, 7, 52398–52413 RSC.
- F. Ke, J. Jiang, Y. Li, J. Liang, X. Wan and S. Ko, Appl. Surf. Sci., 2017, 413, 266–274 CrossRef CAS.
- S. Mao, T. Shen, Q. Zhao, T. Han, F. Ding, X. Jin and M. Gao, Chin. J. Chem. Eng., 2023, 57, 319–328 CrossRef CAS.
| This journal is © The Royal Society of Chemistry 2025 |