 Open Access Article
Open Access ArticleNanoarchitectonics of carbon nitride/NiO/Zn3N2 heterointerfaces for bifunctional applications in electrocatalytic water splitting and coin cell supercapacitors
Tanuja
Singh
a,
Deepak
Deepak
a,
Abhishek
Panghal
a,
Shailendra Kumar
Saxena
 b,
Barnali
Mahato
c,
Abhishek S.
Shekhawat
b,
Barnali
Mahato
c,
Abhishek S.
Shekhawat
 b and
Susanta Sinha
Roy
b and
Susanta Sinha
Roy
 *a
*a
aDepartment of Physics, School of Natural Sciences, Shiv Nadar Institution of Eminence, Gautam Budh Nagar 201314, Uttar Pradesh, India. E-mail: susanta.roy@snu.edu.in
bDepartment of Physics and Nanotechnology, College of Engineering and Technology, SRM Institute of Science and Technology, Kattankulathur, Chennai, India
cInstrumentation and Applied Physics Department, Indian Institute of Science, Bangalore 560012, India
First published on 30th September 2025
Abstract
The advancement of effective and durable electrocatalysts for water splitting and high-performance supercapacitors is essential for sustainable energy conversion and storage. Integrating transition metal heteroatoms can be a pivotal technique to fabricate nanostructures for such bifunctional applications. In this regard, we report graphitic carbon nitride/NiO/Zn3N2 heterointerfaces through a single-step pyrolysis method for the oxygen evolution reaction (OER) and coin cell supercapacitor devices. The synergetic interaction between NiO and Zn3N2 advances charge transfer kinetics and augments the electronic structure, while g-C3N4 provides a conductive network and additional active sites. Optimized sample NZN400 showed exceptional OER performance with a low overpotential value of 350 mV at 50 mA cm−2, besides a low Tafel slope and high turnover frequency value. In addition, NZN400 electrodes showed a high specific capacitance value of 124 mF cm−2 at 2 mA cm−2 for the half-cell and 19.92 mF cm−2 at 0.2 mA cm−2 for the coin cell device. The fabricated device exhibited excellent cycling stability over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 GCD cycles with a capacitance retention of 95.7% and coulombic efficiency of 99.4% at 0.4 mA cm−2 and was able to power up several commercial LEDs, a digital hygrometer, and a digital stopwatch for prolonged durations. The results highlight an effective approach for integrating transition metal oxide/nitride-based compounds with carbon-based materials, aimed at developing economical and high-performance nanostructured materials for electrochemical energy applications.
000 GCD cycles with a capacitance retention of 95.7% and coulombic efficiency of 99.4% at 0.4 mA cm−2 and was able to power up several commercial LEDs, a digital hygrometer, and a digital stopwatch for prolonged durations. The results highlight an effective approach for integrating transition metal oxide/nitride-based compounds with carbon-based materials, aimed at developing economical and high-performance nanostructured materials for electrochemical energy applications.
1. Introduction
Electrochemical energy conversion and storage technologies play a pivotal role in the transition toward sustainable energy systems, addressing the global demand for clean and efficient power sources. Among these technologies, water electrolysis and energy storage systems, such as supercapacitors, have gained significant attention due to their potential to drive sustainable energy solutions.1,2 Water electrolysis enables green hydrogen production by splitting water into hydrogen and oxygen using renewable electricity, serving as a key pathway for clean fuel generation.3 Meanwhile, supercapacitors offer high-power energy storage with rapid charge–discharge capabilities, making them ideal for applications requiring burst energy delivery.4 Typical water splitting comprises two half-cell reactions: the hydrogen evolution reaction (HER) at the cathode and the oxygen evolution reaction (OER) at the anode. While both reactions are integral to overall efficiency, the OER remains a primary bottleneck due to its inherently sluggish kinetics, attributed to the involvement of multiple proton-coupled electron transfer steps.5 Traditionally, noble metal-based catalysts such as platinum (Pt), iridium (Ir), and ruthenium (Ru) have been employed for electrocatalysis; however, their exorbitant cost and limited terrestrial abundance severely restrict large-scale implementation.6,7 Consequently, the development of highly efficient, earth-abundant, and cost-effective electrocatalysts for the OER is imperative to enhance the overall efficiency of water splitting and render hydrogen production economically viable. Recent research efforts have focused on transition metal-based materials, heterointerfaces, and engineered nanostructures to augment catalytic activity, stability, and electronic conductivity. In this context, the exploration of novel material architectures and advanced synthesis strategies for OER catalysts is pivotal in advancing water-splitting technologies toward scalable and practical applications.8–10On the other hand, in the realm of energy storage, supercapacitors have emerged as vital electrochemical devices due to their rapid charge–discharge capability, high power density, and exceptional long-term stability.11 However, electrodes composed of a different material often exhibit high energy density, but they are lacking in terms of long-term stability, and fast charge–discharge rates, thereby restricting overall performance.12 To address this limitation, symmetric supercapacitors (SCs), which utilize the same electrode materials for the anode and cathode, have been extensively investigated. SCs offer a simplified fabrication and, due to the balanced charge storage mechanism, they exhibit higher stability and longer lifespan, making them a promising alternative for next-generation energy storage systems.13 Coin-cell symmetric supercapacitors, which are essential for assessing the electrochemical performance of electrode materials in a consistent and reproducible way, are an example of such systems.13The compact and robust design ensures accurate electrode spacing, optimal electrolyte distribution, and a tightly sealed compressed environment, thereby reducing external variables that may influence performance.14
The advancement of electrode materials is essential for improving energy storage and conversion performance, demanding high electrochemical activity, large surface area, superior areal-specific capacitance, and structural durability.15,16 Developing novel materials with tailored properties is crucial for optimizing efficiency and enabling practical energy storage applications. Nonetheless, the domain of material engineering in pursuit of a bifunctional nature in electrochemical energy conversion and storage continues to be underdeveloped.
Transition metal oxides, nitrides, and sulphides have attracted substantial attention for energy storage and conversion applications in recent years because of their structural tunability, high performance, and abundance.17–19 The incorporation of dual-metal systems improves catalytic performance by facilitating rapid charge transfer and leveraging synergistic effects, among other strategies, to enhance the properties of these materials. Nickel oxide (NiO) is a transition metal oxide that is extensively investigated due to its exceptional electrochemical properties.20 In a recent study, Deling Wang et al.21 published a CoN/Ni/NiO composite that exhibited impressive OER activity with an overpotential of 114 mV at 10 mA cm−2. In NiO, the Ni2+/Ni3+ redox couple is present, which facilitates rapid and reversible charge transfer, thereby improving the charge storage capability. Additionally, it demonstrates a high experimental and theoretical capacitance.22 The intrinsic stability of NiO guarantees long-term electrochemical performance, while its conductivity is substantially enhanced by nano-structuring.23 Furthermore, NiO functions as an effective catalyst for the oxygen evolution reaction (OER) in alkaline environments.24 NiO is frequently combined with metal nitrides, sulphides, or carbon-based materials to further improve its electrochemical kinetics and conductivity. Derived from earth-abundant precursors like urea and melamine, graphitic carbon nitride (g-C3N4) is one of the most promising carbon-based materials for energy applications.25 In a study conducted by Kousik Bhunia et al.26 the NiO NPs/g-C3N4 electrocatalyst exhibited an optimistic OER performance by achieving an overpotential of 360 mV at 10 mA cm−2 current density. Highly effective for both the hydrogen evolution reaction (HER) and the oxygen evolution reaction (OER), g-C3N4 is a metal-free electrocatalyst and support material with nitrogen-rich functional groups (–NH2 and –C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N) that provide plentiful active sites for redox processes. Nevertheless, the intrinsic conductivity of pure g-C3N4 is restricted; hence, metal doping (e.g., Ni, Co, and Zn) or coupling with conductive materials such as graphene and carbon nanotubes (CNTs) would help to improve electron transport properties.27,28 Often mixed with transition metal oxides, sulphides, or nitrides to improve charge storage performance, graphitic carbon nitride (g-C3N4) is a potential material for hybrid supercapacitors and water-splitting applications.29 Among several metal nitrides, Zn3N2 is among the least investigated despite its superior charge concentration, high electron conductivity, nitrogen-rich framework, outstanding stability in both alkaline and neutral environments, and cost-effectiveness.30 The manufacturing of a CoN/Zn3N2 composite for asymmetric supercapacitors (ASCs) leads to a specific capacitance of 75.4 F g−1, therefore highlighting its potential for advanced energy storage uses in a report by Ravikant Adalati et al.31
N) that provide plentiful active sites for redox processes. Nevertheless, the intrinsic conductivity of pure g-C3N4 is restricted; hence, metal doping (e.g., Ni, Co, and Zn) or coupling with conductive materials such as graphene and carbon nanotubes (CNTs) would help to improve electron transport properties.27,28 Often mixed with transition metal oxides, sulphides, or nitrides to improve charge storage performance, graphitic carbon nitride (g-C3N4) is a potential material for hybrid supercapacitors and water-splitting applications.29 Among several metal nitrides, Zn3N2 is among the least investigated despite its superior charge concentration, high electron conductivity, nitrogen-rich framework, outstanding stability in both alkaline and neutral environments, and cost-effectiveness.30 The manufacturing of a CoN/Zn3N2 composite for asymmetric supercapacitors (ASCs) leads to a specific capacitance of 75.4 F g−1, therefore highlighting its potential for advanced energy storage uses in a report by Ravikant Adalati et al.31
Huifang Yang32 reported a carbon nitride/NiO/CoN-based composite and showed its possible electrochemical uses. However, low stability and high cost of CoN limited the scalability and the practical applications. Inspired by this, herein, we report synthesis of a g-C3N4/NiO/Zn3N2 nanostructure using the one step pyrolysis method for the electrochemical oxygen evolution reaction and high-performance coin cell supercapacitors. In contrast to the above reported work, Zn3N2, which has a narrower bandgap than Co-based nitrides, facilitates increased charge transfer and electron mobility. While retaining outstanding electrochemical characteristics, this method provides a more cost-effective, durable, and high-performance alternative. Optimized nanostructure NZN400 showed a low overpotential value of 350 mV at 50 mA cm−2 and minimal Tafel slopes (85 mV per decade) for the oxygen evolution reaction in alkaline media. Additionally, NZN400 exhibited a high specific capacitance value of 124 mF cm−2 at 2 mA cm−2 for the half-cell and 19.92 mA cm−2 at 0.2 mA cm−2 for the coin cell symmetric supercapacitor device with a capacitance retention of 95.7% sustained after 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 GCD cycles, effectively powering a digital hygrometer, a stopwatch, and commercial LEDs for an extended duration.
000 GCD cycles, effectively powering a digital hygrometer, a stopwatch, and commercial LEDs for an extended duration.
2. Experimental section
2.1 Chemicals
All compounds used in this study were of analytical grade and used without additional purification. Zinc nitride hexahydrate, urea, and potassium hydroxide (KOH, 99%) were acquired from Sigma-Aldrich, whilst nickel foam (1.5 mm thickness) was sourced from Global Nanotech.2.2 Materials synthesis
2.3 Characterization details
XRD of fabricated electrodes was carried out in couple theta-two theta diffraction (Bruker D8 Discover) using Cu–K alpha radiation. Raman spectroscopy was conducted using a micro-Raman spectrometer (STR) with a 532 nm argon-ion laser source, 2.5 mW power output, and a 20× objective lens. Surface morphology was studied using a FESEM JEOL JSM PLUS. X-ray photoelectron spectroscopy (XPS) was conducted using a Kratos Analytical Axis Supra instrument equipped with a monochromatic Al Kα (1486.7 eV) X-ray excitation source.| ERHE = EAg/AgCl + 0.059 pH + 0.197 |
The long-term stability was assessed by a 22 h chronopotentiometry test at a current of 10 mA. All the electrochemical measurements were conducted at room temperature.
3. Results and discussion
3.1 X-ray diffraction (XRD) analysis
The XRD technique provides a hint towards crystallographic orientations and structural composition of synthesized nanostructures.33Fig. 1b shows a comparative XRD spectrum of NiO/Zn3N2/g-C3N4 heterointerfaces fabricated at various annealing temperatures. The diffraction peaks observed at 43.3° and 62.4° can be assigned to the (200) and (220) planes of cubic NiO present in NZN200, NZN400, and NZN600.34 These peaks become more noticeable as the annealing temperature increases from 200 °C to 400 °C, therefore suggesting increased crystallinity. Furthermore, peaks at 44.5°, 52.1° and 76.3° correspond to the (111), (200), and (220) planes of metallic Ni due to nickel foam.34 A slight hump at (∼26°) is attributed to g-C3N4.35 To further validate this, Fig. S1(b) presents the XRD spectrum of NZ400 (NiO/Zn3N2), illustrating the absence of this peak. The peak at 37.3° corresponds to the (400) plane owing to Zn3N2.36 However, at higher temperatures, the intensity of these peaks lowers due to lower nitrogen concentration (Fig. S1(b)). All JCPDS files with NZN heterostructures are shown in Fig. S26.3.2 Morphological characterization
3.3 Compositional analysis
The XPS spectra offer critical insights into the elemental composition, oxidation states, and chemical interactions of NiO/g-C3N4/Zn3N2 composites subjected to annealing at different temperatures. Fig. 3(a)–(e) represents the deconvoluted XPS spectra of Ni 2p, Zn 2p, C 1s, O 1s, and N 1s, respectively. The Ni 2p spectra (Fig. 3a) of all nanostructures exhibit two primary peaks at ∼855.97 eV and ∼873.72 eV, which correspond to Ni 2p3/2 and Ni 2p1/2, respectively, and indicate different oxidation states of NiO.39 Also, peaks at 854.12 eV and 872.18 eV correspond to the Ni2+ oxidation state, and peaks at 856.01 eV and 874.21 eV align with Ni3+ due to surface defects or phase formation of Ni2O3.40 Two additional satellite peaks are observed at 860.93 eV and 879.56 eV. Two prominent peaks in the Zn 2p spectra (Fig. 3b) at 1022.15 eV (Zn 2p3/2) and 1044.6 eV (Zn 2p1/2) point to Zn2+ in Zn3N2.41 The intensity of these peaks is higher in NZN400 than in NZN200, which implies that the optimal temperature is conducive to the enhanced incorporation of Zn3N2. NZN600 showed a slight peak broadening, which might be due to surface oxidation or nitrogen loss at higher temperatures. The C 1s spectra (Fig. 3c) illustrate peaks at 284.8 eV, 286.2 eV, and 288.5 eV attributed to C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C/C–N, C–O–C, and N–C
C/C–N, C–O–C, and N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N, respectively.42–44 In lower temperature ranges, i.e., for NZN200 and NZN400, the presence of N–C
N, respectively.42–44 In lower temperature ranges, i.e., for NZN200 and NZN400, the presence of N–C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) N indicates that graphitic carbon nitride is well preserved; however, decreased intensity in NZN600 signifies that it might have been deployed at high temperature. The O 1s spectra (Fig. 3d) of NZN200 show peaks at 531.42 eV and 532.76 eV, representing the oxidation state corresponding to Ni2O3 and surface adsorbed oxygen, respectively.40 Interestingly, NZN400 and NZN6000 show a lattice oxygen peak at 529.36 eV, confirming the significant phase formation of NiO.45 The N 1s spectra (Fig. 3e) exhibit two deconvoluted peaks at approximately 400.39 eV and 398.56 eV, which correspond to distinct nitrogen species. The pyridinic nitrogen in g-C3N4 is responsible for the peak at 398.56 eV.42–44 The presence of carbon nitride is confirmed by the peak at 400.39 eV,42–44 which is attributed to graphitic nitrogen. The intensity of the Zn–N peak decreases as the temperature increases, suggesting that nitrogen is lost at higher temperatures. The EDS analysis in Fig. S4 validates the compositional presence of NiO/g-C3N4/Zn3N2 as the temperature increases. Fig. S5 shows the elemental FESEM mapping analysis of NZN400, confirming the uniform distribution of Ni, Zn, C, O, and N. Fig. S6 shows the XPS survey spectrum of all nanostructures.
N indicates that graphitic carbon nitride is well preserved; however, decreased intensity in NZN600 signifies that it might have been deployed at high temperature. The O 1s spectra (Fig. 3d) of NZN200 show peaks at 531.42 eV and 532.76 eV, representing the oxidation state corresponding to Ni2O3 and surface adsorbed oxygen, respectively.40 Interestingly, NZN400 and NZN6000 show a lattice oxygen peak at 529.36 eV, confirming the significant phase formation of NiO.45 The N 1s spectra (Fig. 3e) exhibit two deconvoluted peaks at approximately 400.39 eV and 398.56 eV, which correspond to distinct nitrogen species. The pyridinic nitrogen in g-C3N4 is responsible for the peak at 398.56 eV.42–44 The presence of carbon nitride is confirmed by the peak at 400.39 eV,42–44 which is attributed to graphitic nitrogen. The intensity of the Zn–N peak decreases as the temperature increases, suggesting that nitrogen is lost at higher temperatures. The EDS analysis in Fig. S4 validates the compositional presence of NiO/g-C3N4/Zn3N2 as the temperature increases. Fig. S5 shows the elemental FESEM mapping analysis of NZN400, confirming the uniform distribution of Ni, Zn, C, O, and N. Fig. S6 shows the XPS survey spectrum of all nanostructures.
 | ||
| Fig. 3 Deconvoluted XPS spectra of all heterointerfaces: (a) Ni 2p, (b) Zn 2p, (c) C 1s, (d) O 1s and (e) N 1s; (f) Raman spectra of NZN200, NZN400, and NZN600. | ||
3.4 Raman spectroscopy and BET analysis
The Raman spectra of the NiO/Zn3N2 and graphitic carbon nitride (g-C3N4) hybrids show different characteristic vibrational modes. Fig. 3f shows a comparison of Raman analysis of NZN200, NZN400, and NZN600, depicting structural changes as a function of annealing temperature. A peak at about 385 cm−1 (in NZN200) indicates the presence of Zn3N2, either via defect-induced vibrations, intrinsic phonon modes, or probable oxygen incorporation producing a mixed phase46 A wide band from 500–550 cm−1 (i.e. in NZN400 and NZN600) indicates to F2g phonon mode and confirms the successful synthesis of nickel oxide.47 Interestingly, Zn3N2 exhibits a wide band in a similar range owing to the Zn–N vibrational mode.48 The Raman peaks within the range of 725 and 1045 cm−1 indicate the stretching vibrations of aromatic C–N heterocycle characteristic of graphitic carbon nitride (g-C3N4).49,50 Moreover, the strong peaks seen in the 1100–1300 cm−1 range indicate the skeletal vibrations of C–N bonds in g-C3N4; the G-band of sp2-hybridized carbon accounts for the observed peak in the 1500–1600 cm−1 range, further validating the presence of graphitic carbon nitride.49,50 Fig. S1(a) displays the Raman spectra of Zn3N2/NiO (NZ400) and g-C3N4/NiO (NU400) that have been annealed at an optimized temperature of 400 °C. With samples annealed at 600 °C (NZN600), the intensity and coherence of these peaks reflect a temperature-dependent pattern whereby most Raman characteristics indicate increased crystallinity of Zn3N2, NiO, and g-C3N4. Furthermore, increased oxygen consistency because of annealing influences the carbon–nitrogen bond, which is predominantly affected by the vibrations of nickel oxide and oxynitrides. Changing the annealing temperature affects the structural evolution of the composite, thereby improving phase purity and lowering amorphous contributions found in the lower-temperature nanostructures.3.5 BET analysis
Since the nanostructures were grown on self-supported nickel foam, we fragmented each NZN nanostructure into pieces and calculated their BET surface area (Fig. S13). NZN400 exhibited the highest surface area of 4.14 m2 g−1 as compared to NZN200 and NZN600, which show the values of 3.05 m2 g−1 and 1.3 m2 g−1, supporting the higher electrocatalytic properties of NZN400.3.6 Electrochemical energy conversion
To thoroughly assess the electrocatalytic properties, all the synthesized nanostructures have undergone electrochemical characterization procedures including cyclic voltammetry (CV), linear sweep voltammetry (LSV), electrochemical impedance spectroscopy (EIS), chronopotentiometry, and symmetric testing. A three-electrode system with 1 M KOH as the electrolyte was utilized for these studies. Ag/AgCl was used as the reference electrode, platinum as the counter electrode, and NZN200, NZN400, and NZN600 as working electrodes. Important characteristics such as overpotential, Tafel slope, ECSA, Rct, and Rs have been assessed to evaluate the OER performance. From a materials perspective, NiO is the main catalytic component for the OER since it supplies the main active sites for oxygen oxidation and adsorption.51 Because of the existence of Ni2+/Ni3+ redox couples, NiO offers a highly conductive network that improves charge transfer and fosters catalytic efficiency. Graphitic carbon nitride decreases overall resistance and facilitates charge transport via a conductive carbon matrix.52 The porous nature of C3N4 promotes the additional active sites required for catalytic activity. To enhance catalytic activity, C3N4 also introduces defect sites that modify the local charge density and the oxygen binding strength. Zn3N2 enhances mechanical integrity and accelerates reaction kinetics by altering the electronic structure of NiO and restricting excessive adsorption. The optimal proportion of NiO, Zn3N2, and g-C3N4 in NZN400 produces synergy, leading to enhanced long-term stability, increased active site exposure, ideal oxygen adsorption energy, and efficient charge transport. Given that the OER exhibits sluggish kinetics, it is imperative to develop an electrocatalyst with enhanced performance to augment overall efficiency.53 The charge transfer mechanism of the oxygen evolution reaction comprises four fundamental steps:| OH− + N* → OHN* + e− |
| OH− + OHN* → ON* + H2O + e− |
| ON* + OH− → OOHN* + e− |
| OOHN* + OH− → N* + O2 + H2O + e− |
In the NZN heterostructure, the Ni centre in NiO serves as the principal active site for the OER, the redox couples of Ni2+/Ni3+/Ni4+. Upon the electrochemical sweeps in alkaline electrolyte, Ni2+ is first oxidized to Ni3+, followed by Ni4+, producing the NiOOH species, which are considered as intermediates for OER-based electrocatalysts. The anodic peaks observed in the CV curves indicate the oxidation of Ni2+/Ni3+. In addition, post-OER XPS analysis shows the shift in Ni 2p B.E. values towards higher B.E., further confirming oxidation of Ni2+/Ni3+.
Zn3N2 exhibits Zn–N coordination sites, which contribute effectively to the OER activity in addition to NiO. The enriched nitrogen environment provides additional adsorption sites for OH− ions, which facilitates the formation of intermediate, i.e., OH*, O*, and OOH*. Zinc does not undergo major oxidation itself under the OER activity; however, its potential in modulating the electronic structure of the heterostructure and improving the OH− adsorption and charge redistribution is very crucial.
In addition, g-C3N4 has enriched nitrogen that increases the dispersion of both NiO and Zn3N2, further facilitating the charge transfer and stabilizing the heterostructure network. These three phases enable the close synergetic effects like increased conductivity, high ECSA, and increased exposed active sites.
Thus, the OER mechanism in our composite involves the following key steps at Ni sites:
| Ni2+–OH− → Ni3+–OH* + e− |
| Ni3+–OH* → Ni4+–O* + H+ + e− |
| Ni4+–O* + OH− → Ni4+–OOH* + e− |
| Ni4+–OOH* → Ni2+ + O2 + OH− + e− |
Zn3N2 modulates the electronic density at the sites of Ni through the interactions of Ni–N–Zn. In addition, it also provides Lewis basic N sites which can directly absorb the OH− ions which further assists in the formation of OH* and OOH*. It also prevents the excess oxidation of NiO by stabilizing Ni3+/Ni4+. Overall, Zn3N2 introduces Zn–N moieties which can serve as the secondary adsorption sites for the OH−. The coupling between Zn–N and Ni–O redistributes the charge density which further lowers the activation barrier for water splitting.
g-C3N4 participates directly through the pyridinic and graphitic N sites which interact with OH− and serve as electron donating groups which overall accelerate the Ni2+/Ni3+ redox conversion. The robust coupling at the heterointerface of NiO/g-C3N4 generates the defect rich active centres which facilitate the water splitting process.
This redox cycle is supported and accelerated by Zn–N and g-C3N4 sites, which assist in intermediate adsorption and charge mobility (Fig. 8a).
Since NZN400 has a higher degree of crystallization at an optimal temperature, which results in a higher proportion of nitrogen and oxygen vacancies, it is anticipated to have the best OER activity among all. The LSV polarisation curves of NZN200, NZN400, and NZN600 at a scan rate of 20 mV s−1 are shown in Fig. 4a. NZN400 showed the least overpotential value of 350 mV at 50 mA cm−2 as compared to NZN200 and NZN600, which showed 470 mV and 600 mV, respectively. NZN200 shows saturation behaviour beyond 1.6 V, which might be due to:
1. At higher overpotentials, due to deprived porosity and phase development, mass transport limitations arise, which hinder the performance.
2. In NZN200, accumulation of OER intermediates like OOH* or the evolution of gas bubbles cannot be efficiently desorbed or removed from the surface (catalyst passivation), leading to low current or saturated current response.
3. An inadequate phase leads to instability of the catalyst at high anodic potentials which further decreases the electrocatalytic activity.
Fig. S7a shows the LSV comparison profiles of pristine materials and heterointerfaces. A comparison bar graph depicting overpotential values at 50 mA cm−2 is shown in Fig. 4b. The Tafel slope provides information on the kinetics of the process and reaction pathway. The reaction kinetics are better when the Tafel slope value is lower.54Fig. 4c shows the Tafel slope values of NZN200, NZN400, and NZN600. NZN400 shows the least Tafel slope value of 85 mV dec−1 as compared to NZN200 and NZN600, which showed slope values of 175 mV dec−1 and 376 mV dec−1, respectively. This parameter demonstrates better electrocatalytic activity of NZN400 over other catalysts. Generally, a Tafel slope value close to 120 mV dec−1 indicates the first electron transfer step with the formation of *OH from OH− as the rate-limiting step, and a Tafel slope value of 60 mV dec−1 indicates the second electron transfer step with the formation of *O from *OH as the rate-determining step. A Tafel slope above 200 mV dec−1 reflects sluggish kinetics caused by mass transport resistance or weak intermediate adsorption.
For NZN400, the observed Tafel slope (85 mV dec−1) indicates stable binding energy of OER intermediates and increased charge transfer kinetics. This behaviour can be supported by the close synergetic effects of NiO, Zn3N2, and g-C3N4 at 400 °C, which ensures the increased crystallinity, modulation of the electronic structure, and enriched redox couple sites of Ni2+/Ni3+. Overall, these factors combined accelerate the rate kinetics by rapid formation of intermediates and transformation of OER intermediates like OH*, O*, and OOH*. In contrast, NZN200 exhibits a higher Tafel slope value of 175 mV dec−1, lacks proper phase formation, and exhibits lower conductivity due to inadequate crystallization at 200 °C. This phenomenon results in sluggish kinetics, allowing the first electron transfer step to become the rate-limiting step. NZN600 is annealed at a very high temperature and shows a Tafel slope value of 376 mV dec−1 and decreased electrocatalytic activity due to structural deformation or collapse of the g-C3N4 network and nitrogen loss from Zn3N2, which leads to poor intermediate stabilization and decreased active site density.
In summary, the Tafel slope variations for NZN heterostructures underline the dependence of the OER mechanism on morphology, crystallinity, and modulated electronic conductivity and available active sites.
The CV plots of NZN200, NZN400, and NZN600 at a scan rate of 100 mV s−1 are shown in Fig. 4d. NZN400 has a very high enclosed area that directly indicates towards a large charge storing capacity and can be determined from the CV plots. In addition, NZN400 exhibits a higher current density than NZN200 and NZN600. Electric double layer capacitance is an additional parameter used in the analysis of catalyst activity and calculation of electrochemical active surface area (ECSA). Fig. 4e shows the Cdl values of all catalysts, depicting the highest value for NZN400 as compared to NZN200 and NZN600. Based on this value, ECSA for the nanostructures was calculated (SI eqn (6)) using CV curves at scan rates of 20, 40, 60, 80, and 100 mV s−1 and found in the order of NZN400 > NZN200 > NZN600, which is 10.00 cm2 for NZN400, 2.11 cm2 for NZN200, and 1.36 cm2 for NZN600. ECSA normalized LSV profiles of NZN200, NZN400, and NZN600 can be found in Fig. S15. Fig. S7(b–d) shows the CV curves of all nanostructures in non-faradaic regions.
Fig. 4f depicts the Electrochemical Impedance Spectroscopy (EIS) spectra for NZN200, NZN400, and NZN600. EIS is a highly effective technique for gaining useful insights into an electrocatalyst's charge transfer resistance and ion/electron transport characteristics.55 The equivalent fitted circuit is shown in SI Fig. S8. The Rs value, which is the resistance given by the electrolyte and electrical contacts, is the lowest for NZN400, at 1.23 Ω, followed by NZN200 at 2.84 Ω and NZN600 at 3.179 Ω. In addition, the Rct value is utilized to determine charge transfer at the electrode–electrolyte interface. For NZN400, the Rct is 1.45 Ω, in contrast to NZN200 and NZN600, which showed Rct values of 2.12 Ω and 6.36 Ω, respectively, indicating improved charge transfer kinetics and thus superior OER kinetics. Thus, EIS reveals that NZN400 exhibits quick charge transfer and low resistance, making it an effective OER catalyst. Furthermore, symmetric device testing is performed to create a baseline assessment of the electrochemical behaviour under ideal conditions, employing NZN400 electrodes at both the anode and cathode (Fig. 4g). The LSV curve demonstrated an overpotential value of 750 mV in 1 M KOH at a current density of 20 mA cm−2 and a scan rate of 2 mV s−1. Also, this testing showed that the catalyst requires a cell voltage of 1.85 V at 10 mA cm−2. To assess the long-term durability of the optimized NZN400 electrode, a chronopotentiometry test was performed for 22 hours at a constant current density of 10 mA cm−2, as shown in Fig. 4h. The voltage retention for this sample was found to be 99.80%, showing good stability over long test periods. Notably, the turnover frequency (TOF) is calculated using an additional SI eqn (4). NZN400 showed the highest TOF value of 0.018 s−1 as compared to NZN200 and NZN600, which showed TOF values of 0.012 s−1 and 0.005 s−1, respectively. Fig. 4i shows the TOF vs. overpotential variation. Furthermore, to investigate the catalytic effectiveness of the catalyst material per unit mass, mass activity was calculated using SI eqn (5). NZN400 showed the highest mass activity of 16.27 A g−1 among others, demonstrating superior OER performance. Consistently, the optimized sample NZN400 had significantly higher mass activity values, which were consistent with earlier electrochemical tests. Fig. S7(e) shows the iR drop for all samples estimated using (Galvanostatic Charge Discharge) GCD, which reflects the voltage drop across the electrolyte. Notably, NZN400 showed the lowest slope, indicating negligible voltage loss during the OER process. Table 1 lists all the parameter values. Fig. S16(a)–(i) shows the electrocatalytic performance parameter testing of NZN nanostructures in the Hg/HgO reference electrode configuration. These findings demonstrate NZN400's stability and efficiency, indicating its potential as a viable catalyst for electrochemical applications.
| S. no. | Sample name | Overpotential (η50) (mV) | Tafel slope (mV dec−1) | ECSA (cm2) | Mass activity (A g−1) | TOF (s−1) |
|---|---|---|---|---|---|---|
| 1 | NZN200 | 470 | 175 | 2.11 | 11.71 | 0.012 |
| 2 | NZN400 | 350 | 85 | 10.00 | 16.27 | 0.018 |
| 3 | NZN600 | 600 | 376 | 1.36 | 5.09 | 0.005 |
3.7 Electrochemical half-cell supercapacitor performance
The electrochemical half-cell energy storage characteristics of the synthesized NiO/Zn3N2/C3N4 (NZN) nanostructure were evaluated using a three-electrode setup. The self-supported NZN (200, 400, and 600) heterointerfaces served as the working electrode, Ag/AgCl as the reference electrode, and a platinum (Pt) electrode as the counter electrode. All measurements were performed in 1 M Na2SO4 electrolyte. Electrochemical performance was evaluated using techniques like CV, GCD, and EIS. Fig. 5a shows the CV profile comparison at 10 mV s−1 of NZN200, NZN400, and NZN600. NZN400 exhibited the highest enclosed area under the CV curve, depicting the maximum specific capacitance. Also, CV curves demonstrated significant redox peaks for NZN400, implying a pseudocapacitive nature due to faradaic redox reactions (Ni2+ ↔ Ni3+ + e−). In contrast, NZN200 showed less intense redox peaks, possibly due to inadequate phase formation or insufficient crystallization, resulting in reduced electrochemical performance. On the other hand, NZN600 exhibits suppressed peaks, which might be due to excessive grain growth or a wrinkled agglomerated structure, leading to reduced active sites at high temperature.NZN400 shows the highest enclosed area in the CV profile with prominent redox peaks suggesting a strong pseudocapacitive behaviour, which results from the Ni2+/Ni3+ redox transformations. The close synergetic effects of NiO, Zn3N2, and g-C3N4 result in efficient ion/electron transport, which results in effective faradaic reactions, leading to the highest specific capacitance. NZN200, on the other hand, has decreased crystallinity and inadequate phase formation in comparison, and shows weak redox peaks and reduced enclosed area in CV, depicting a combination of electric double layer capacitance and pseudo capacitance. The decreased active sites and lower conductivity result in decreased specific capacitance. NZN600, at last, which is annealed at very high temperatures, exhibits a repressed CV curve and diminished redox activity. Deformation of g-C3N4 and nitrogen loss from Zn3N2 leads to a decreased number of active sites and conductivity. Furthermore, the distorted morphology will lead to decreased ion transport and decreased specific capacitance. The observations indicate that the different capacitive behaviour depends on proper phase formation, interface quality, and defect density. SI Fig. S9a and b display additional CV curves of NZN200 and NZN600 at scan rates ranging from 10 mV s−1 to 100 mV s−1. Fig. 5c shows the GCD profiles of composite structures. NZN400 exhibits the longest charge–discharge duration, which confirms its highest capacitance. The observation of a minor voltage plateau, which aligns well with the nature of CV curves and well in line with CV analysis, further supports the pseudocapacitive behaviour associated with faradaic reactions.56 The nearly symmetric charge–discharge curves, which also suggest a strong coulombic efficiency, are indicative of excellent electrochemical reversibility. Fig. S9c shows the specific capacitance comparison of NZN200, NZN400, and NZN600 with scan rates. The GCD profiles for NZN200 and NZN600 are illustrated in SI Fig. S9d & e and were acquired at current densities ranging from 2 to 7 mA cm−2. Fig. 5e illustrates the rate capacity of these nanostructure electrodes by illustrating the change in specific capacitance as a function of varied current densities. NZN400 showed a specific capacitance value of 124 mF cm−2 at 2 mA cm−2, which was found to be higher than that of NZN200 and NZN600, which exhibited a capacitance value of 39.6 mF cm−2 and 15.4 mF cm−2 at similar current densities. Fig. 5b shows the CV cycles of the NZN400 nanostructure, which demonstrate capacitive behaviour as the scan rate increases. The area under the CV curve increases with scan rate due to effective interaction of the electrolyte ions with the electrode materials.57Fig. 5d shows the GCD profiles of NZN00 at different current densities. To conduct a more thorough estimation of the electrochemical performance, EIS measurements were carried out (Fig. 5f). The Nyquist plot demonstrates that NZN400 exhibits the minimum charge transfer resistance (Rct) of 19.7 Ω, suggesting enhanced kinetics for electron and ion transport. In addition, NZN400 demonstrated outstanding electrolyte accessibility and solution kinetics, with a solution resistance (Rs) of 3.42 Ω. The electrochemical parameters for each heterointerface are presented in Table 2. The findings suggest that NZN400 is a promising electrode material for high-performance supercapacitors, attributed to its exceptional electrochemical stability, remarkable rate capability, and improved specific capacitance.
| S. no. | Sample | Specific capacitance (mF cm−2) | R ct (Ω) | R s (Ω) |
|---|---|---|---|---|
| 1 | NZN200 | 39.6 | 675 | 5.10 |
| 2 | NZN400 | 124 | 19.7 | 3.42 |
| 3 | NZN600 | 15.4 | 681 | 5.50 |
3.8 Coin cell supercapacitor performance
The electrochemical performance of the NiO/Zn3N2/graphitic carbon nitride coin cell supercapacitor was examined employing various electrochemical approaches. Fig. 6a illustrates the cyclic voltammetry (CV) cycles of NZN400 at various scan rates. The CV curves exhibit distinct redox peaks, signifying pseudocapacitive behaviour resulting from faradaic redox transitions of Ni2+/Ni3+. Fig. 6b shows the GCD profiles of NZN400 at different current densities. The nonlinear charge–discharge nature of the curves may account for the pseudocapacitive behaviour of the nanostructure. Fig. 6c shows the specific capacitance vs. scan rate of NZN400. It showed the highest specific capacitance value of 19.92 mF cm−2 at 0.2 mA cm−2. Fig. 6d illustrates the Nyquist plot of the NZN400 device, displaying a slight semicircle in the high-frequency domain, with a low charge transfer resistance (Rct) of 175 Ω, which, as compared to the half-cell, is high due to the use of a separator that causes kinetic constraints. In the low-frequency regime, a sharp curve signifies the optimum capacitive performance. The inset of Fig. 6d shows the equivalent fitted circuit comprised of solution resistance with a value of 1.75 Ω, and charge transfer resistance (Rct) with two constant phase elements (CPE1 and CPE2). Fig. S10 also shows the equivalent circuit modelling, besides EIS performance parameters for both half-cell and coin cell supercapacitor devices.
Fig. 6e shows the Ragone plot, which represents the relationship between energy density and power density. The NZN400 device showed an energy density of 2.7 μW h cm−2 at 0.2 mA cm−2 and a power density of 0.2 mW cm−2 at 0.2 mA cm−2. This trend of decrease in energy density with increasing power density highlights the trade-off between rapid charge–discharge capability and total charge storage. Fig. 6f demonstrates the cycling stability and coulombic efficiency of the NZN400 device. Excellent stability over 10![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 cycles with a retention rate of 95.7% reveals the durability and robustness of the fabricated device. Additionally, the coulombic efficiency of 99.4% suggests high reversibility of charge storage mechanisms with bare or no material degradation.
000 cycles with a retention rate of 95.7% reveals the durability and robustness of the fabricated device. Additionally, the coulombic efficiency of 99.4% suggests high reversibility of charge storage mechanisms with bare or no material degradation.
The quantitative diffusion properties of the NZN400 device are examined using Dunn's method. This method indicates the difference between surface capacitive and diffusion-controlled contributions at different scan rates at the applied potential. At a constant potential, a changing current can be represented by using the equation58
| I(V) = K1(v) + K2(v)1/2 | (1) |
| I = avb | (2) |
The determination of the diffusion-controlled contribution and the capacitive contribution depends on the value of b. When the CV shape is rectangular and the value of b equals 1, the electrode material demonstrates EDLC behaviour.59 When the CV shape resembles a rectangle with several points of inflection and the value of b is close to 0.5, the material demonstrates pseudocapacitive behaviour due to fast redox reactions.59 The NZN400 device showed a slope b = 1.08 (Fig. 6g). Fig. 6i shows the capacitive and diffusion-controlled contribution of the NZN400 device with scan rate. At lower scan rates (1–3 mV s−1), the contribution from diffusion control is more significant (i.e. 32.49% at 2 mV s−1). The increase in scan rate leads to a systematic increase in the capacitive contribution, from 59.52% at 1 mV s−1 to 78.26% at 6 mV s−1. At higher scan rates (7–10 mV s−1), the capacitive behaviour dominates, reaching 82.29% at 10 mV s−1. Fig. S11(a) shows the specific capacitance variation of the NZN400 device with scan rate, and Fig. S11(b) shows the CV curves of the same recorded at low scan rates.
Furthermore, a fabricated coin cell supercapacitor was tested for real-time applications (Fig. 6j–l), including charging of different commercial LEDs, a digital hygrometer for 5 minutes, and a digital stopwatch over 50 minutes (SI Videos 1 & 2). The robust performance of NZN400 in real-time operations underscores its promise for futuristic energy storage technologies. Table S3 shows the comparison of the electrochemical energy density of the present work with similar materials.
Apart from the above-mentioned claims and experiments, the exceptional energy conversion and storage capabilities of NZN400 are mostly due to the following material characteristics.
• Using a balanced mechanism of EDLC and faradaic reactions, the integration of NiO, Zn3N2, and g-C3N4 enhances charge storage.
• Employing nickel foam as a substrate improves interfacial contact with active materials, hence reducing interfacial resistance and enhancing charge transfer kinetics.60
• The coexistence of mixed oxidation states (Ni2+/Ni3+ and Zn2+) alongside surface oxygen species augments electronic conductivity and charge redistribution, leading to a notable enhancement in energy storage and OER kinetics.
4. Electrochemical kinetics, ex situ and in situ analysis
A higher rate constant value substantially influences the catalytic efficiency and overall performance by facilitating rapid electron or ion movement. Fig. 7a–c shows the anodic and cathodic peak current vs. the square root of the scan rate of NZN heterostructures.The rate constant k0 and transfer coefficient α can be calculated using the Nicolson dimensional number and Laviron equation61,62 respectively using the following equations:
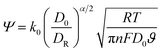 | (3) |
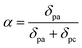 | (4) |
In eqn (4), δpa and δpc are the slopes calculated from currents recorded from anodic and cathodic peaks vs. the logarithm of scan rate plots, respectively.
If we consider D0 ≅ DR, then eqn (3) can be rewritten as:
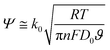 | (5) |
Additionally, Ψ can be calculated as:
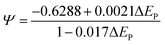 | (6) |
Utilizing eqn (5) and (6), the value of k0 can be calculated as the slope of Ψ versus v−1/2.
Fig. 7d–f shows the anodic and cathodic peak current slopes against the log of scan rates of NZN200, NZN400, and NZN600, respectively. The corresponding Ψ and rate constant variations are shown in Fig. 7g and h, respectively. NZN400 shows a rate constant value of 0.001 s−1, which is higher as compared to that of NZN200 and NZN600, which show a value of 0.009 s−1 and 0.0008 s−1, suggesting faster kinetics in NZN400. Fig. S18 presents the CV of NZN200, NZN400, and NZN600 in the redox active electrolyte. Fig. 7i shows the post-mortem Raman spectra of NZN400, showing no change and robustness of the electrocatalyst synthesized. Fig. S17 shows the fitted Cdl slope of NZN nanostructures in the Ag/AgCl reference electrode. Fig. S18 shows CV of NZN200, NZN400, and NZN600 in redox active electrolyte.
4.1 Post-mortem and ex situ characterization studies
Fig. S14 shows the postmortem FESEM and EDS of the NZN400 nanostructure after the electrochemical stability test, revealing the robust nature that maintains the structural and elemental integrity with little agglomeration due to K ions. Fig. 8b shows the deconvoluted Ni 2p XPS spectrum for NZN400 before and after the stability test. A small shift in Ni 2p B.E. values towards higher B.E. after stability can be attributed to oxidation of Ni2+/Ni3+. Fig. 8c shows the deconvoluted Zn 2p spectra with minimal change in the B.E., depicting the robustness of zinc metal. Fig. 8d shows a comparison of C 1s spectra, which shows an additional peak at 289.5 eV for NZN400 after electrochemical measurements which can be assigned to the C![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) O bond (oxidation) due to prolonged cycles. In the O 1s spectra (Fig. 8e), the reduced intensity of the Ni2+ signal can be attributed to the partial oxidation of Ni2+ species in NiO to Ni3+/Ni4+, resulting in the formation of highly active NiOOH-like intermediates. Fig. 8f shows the minimal shift in the C–N–H bond, which corresponds to the stretching due to long-term cycling tests. Fig. 8g shows the XRD spectrum before and after electrochemical stability testing, showing no change or any additional peaks, further confirming the robustness of the nanostructures. Fig. S19–S21 shows the effect of nitrogen environment on the adsorption of OH− ions via wetting, double layer capacitance calculation and LSV profiles, showing that the nitrogen environment can effectively alter the overall electrochemical performance. Fig. S24 shows EIS, I–V and sheet resistance of NZN400 and without g-C3N4. Fig. S26 presents the JCPDS file no assigned to NZN heterostructures.
O bond (oxidation) due to prolonged cycles. In the O 1s spectra (Fig. 8e), the reduced intensity of the Ni2+ signal can be attributed to the partial oxidation of Ni2+ species in NiO to Ni3+/Ni4+, resulting in the formation of highly active NiOOH-like intermediates. Fig. 8f shows the minimal shift in the C–N–H bond, which corresponds to the stretching due to long-term cycling tests. Fig. 8g shows the XRD spectrum before and after electrochemical stability testing, showing no change or any additional peaks, further confirming the robustness of the nanostructures. Fig. S19–S21 shows the effect of nitrogen environment on the adsorption of OH− ions via wetting, double layer capacitance calculation and LSV profiles, showing that the nitrogen environment can effectively alter the overall electrochemical performance. Fig. S24 shows EIS, I–V and sheet resistance of NZN400 and without g-C3N4. Fig. S26 presents the JCPDS file no assigned to NZN heterostructures.
 | ||
| Fig. 8 (a) Mechanistic visualization of OER activity, postmortem XPS spectra of NZN400: (b) Ni 2p, (c) Zn 2p, (d) C 1s, (e) O 1s, and (f) N 1s, and (g) postmortem XRD of NZN400. | ||
4.2 Reaction order analysis
We have measured the LSV profiles of NZN400 at different electrolyte concentrations (0.1 M, 0.2 M, 0.3 M, 0.4 M, 0.6 M, 1 M, 2 M and 4 M) and plotted the log (current density) vs. log (OH−) to get the corresponding slope (Fig. S22). The value of the slope generally indicates the reaction order. The obtained slope 1.02 provides the reaction order corresponding to hydroxide ions. It means j ∝ [OH−]1.02 or first order dependence on OH− in the tested range. This value indicates the involvement of one OH− as a reactant in the elementary rate determining state. In alkaline media, this suggests OH adsorption (* + OH− → *OH + e−) or the deprotonation/dehydroxylation of an adsorbed hydroxyl (*OH + OH− → *O + H2O + e−).4.3 Kinetic isotope effect
To further analyse the information about the intermediate's behaviours, we calculated the kinetic isotope effect parameter KIE via testing of NZN400 in 1 M KOH and 1 M D2O solutions (Fig. S23). KIE = KH/KD with a value of 3.4 in a different overpotential range indicates proton involvement implying OH bond stretching and contributing directly to the rate determining step. The semiclassical approximation of this value leads to a zero-point energy difference ΔZPE = RT![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln
ln![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 3.4 ≅ 3.0 kJ mol−1. This KIE values outclass the RDS agreements of purely electron transfer limited step or entirely chemical O–O coupling which exhibits typically a KIE value of 1.
3.4 ≅ 3.0 kJ mol−1. This KIE values outclass the RDS agreements of purely electron transfer limited step or entirely chemical O–O coupling which exhibits typically a KIE value of 1.
4.4 In situ EIS analysis
In situ EIS was recorded after every 5 GCD cycles at 4 mA cm−2 to understand the Na+ transport behaviour under operating conditions (Fig. S25). The Nyquist spectra at 0, 5, 10, 15, 20, 25 and 30 cycles show almost no major change. A slight increase in resistance is observed after the initial 5 cycles compared to the fresh cell which may be due to initial wetting/activation of the electrode–electrolyte interface. After that, the spectra remain stable with very small variations.63,64 The high-frequency intercept (Rs) remained constant which suggests stable Na+ conduction in the electrolyte and at the electrode/electrolyte interface. The semicircle diameter (Rct) also did not change much, indicating fast and reversible adsorption desorption of Na+ ions at the redox active sites. The low frequency region line remains nearly vertical which shows that Na+ diffusion in porous electrode channels and capacitive charge storage was maintained even after several cycles. These results clearly prove that the NZN400 electrode provides stable Na+ ion transport pathways and supports the excellent cycling stability of the device.5. Conclusion
In summary, we have integrated transition metal heteroatoms to fabricate a hybrid nanostructure, which is further utilised for bifunctional applications in energy conversion and storage. Synergetic interactions between such heterointerfaces facilitate the charge transfer characteristics and increase the active sites. The optimized catalyst NZN400 demonstrated excellent OER activity with a low overpotential of 350 mV in addition to remarkable coin cell device performance, which exhibited a specific capacitance value of 19.92 mF cm−2 and excellent stability with real-time applications. The presence of mixed oxidation states (Ni2+/Ni3+, Zn2+) in conjunction with surface oxygen species enhances electronic conductivity and charge redistribution, resulting in a significant improvement in energy storage and OER kinetics. These hybrid nanostructures offer significant insights for the advancement of efficient, durable, and affordable catalysts designed for commercial storage and electrolysis technology.Author contributions
Tanuja Singh: conceptualization, methodology, formal analysis, data curation, writing – original draft, writing – review & editing, validation. Deepak Deepak: conceptualization, methodology, investigation, validation. Abhishek Panghal: methodology, investigation, software, visualization. Shailendra Kumar Saxena: investigation, data curation, formal analysis. Barnali Mahato: investigation, visualization, formal analysis. Abhishek S Shekhawat: investigation, data curation. Susanta Sinha Roy: conceptualization, investigation, formal analysis, data curation, supervision, validation, visualization, writing – review & editing.Conflicts of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.Data availability
The results of this research are corroborated by the data which can be obtained from the corresponding author upon request. The data which support these findings can be found in the paper.Supplementary information: FESEM images, EDS mapping, HRTEM images, XPS spectra, CV curves at different scan rates, LSV curves, GCD, equivalent circuit modelling, electrochemical performance parameters in Hg/HgO, postmortem studies, reaction order, etc. Fig. S1–S26. See DOI: https://doi.org/10.1039/d5ta05959c.
Acknowledgements
Tanuja Singh acknowledges the financial support from the Shiv Nadar Institution of Eminence (SNIoE), India, through a scholarship. Authors acknowledge the Nanotechnology Research Centre (NRC) SRMIST, KTR, for providing the facilities for XPS measurements. We also acknowledge support from the FIST Project (Sanction Number SR/FST/PS-I/2017/6(C)) for the Raman measurements.References
- Z. Sun, L. Sun, S. W. Koh, J. Ge, J. Fei, M. Yao, W. Hong, S. Liu, Y. Yamauchi and H. Li, Carbon Energy, 2022, 4, 1262–1273 CAS.
- G. M. Tomboc, J. Kim, Y. Wang, Y. Son, J. Li, J. Y. Kim and K. Lee, J. Mater. Chem. A, 2021, 9, 4528–4557 RSC.
- H. Tüysüz, Acc. Chem. Res., 2024, 57, 558–567 Search PubMed.
- A. G. Olabi, Q. Abbas, A. Al Makky and M. A. Abdelkareem, Energy, 2022, 248, 123617 CrossRef CAS.
- T. Singh, D. Deepak, A. Panghal and S. S. Roy, Int. J. Hydrogen Energy, 2024, 84, 939–950 CrossRef CAS.
- S. Mohanapriya, P. Dhanasekaran and S. V. Selvaganesh, Noble Metal-Free Electrocatalysts: Fundamentals and Recent Advances in Electrocatalysts for Energy Applications, Am. Chem. Soc., 2022, 1431, 4–73 Search PubMed.
- A. Udayakumar, P. Dhandapani, S. Ramasamy, C. Yan and S. Angaiah, Ionics, 2024, 30, 61–84 CrossRef CAS.
- D. Deepak, T. Singh, A. Mahapatra, A. Panghal, C. Nagesh and S. S. Roy, Adv. Sustain. Syst., 2025, 9, 2400656 CrossRef CAS.
- M. Sohail, S. Rauf, M. Irfan, A. Hayat, M. M. Alghamdi, A. A. El-Zahhar, D. Ghernaout, Y. Al-Hadeethi and W. Lv, Nanoscale Adv., 2024, 6, 1286–1330 RSC.
- G. G. Njema and J. K. Kibet, Prog. Eng. Sci., 2024, 100018 CrossRef.
- F. Wang, X. Wu, X. Yuan, Z. Liu, Y. Zhang, L. Fu, Y. Zhu, Q. Zhou, Y. Wu and W. Huang, Chem. Soc. Rev., 2017, 46, 6816–6854 RSC.
- M. Hepel, Electrochem. Sci. Adv., 2023, 3, e2100222 CrossRef CAS.
- S. V Prabhakar Vattikuti, K. C. Devarayapalli, M. Chandra Sekhar, N. Kumar Reddy Nallabala, N. Nguyen Dang and J. Shim, Mater. Lett., 2021, 285, 129203 CrossRef.
- B. Tang, Y. Wei, R. Jia, F. Zhang and Y. Tang, Small, 2024, 20, 2308126 CrossRef CAS.
- A. Panghal, T. Singh, D. Deepak, R. Kumar and S. S. Roy, J. Energy Storage, 2025, 115, 115981 CrossRef.
- A. Panghal, D. Sahoo, D. Deepak, S. Deshmukh, B. Kaviraj and S. S. Roy, ACS Appl. Nano Mater., 2024, 7, 5358–5371 CrossRef CAS.
- J. Rehman, K. Eid, R. Ali, X. Fan, G. Murtaza, M. Faizan, A. Laref, W. Zheng and R. S. Varma, ACS Appl. Energy Mater., 2022, 5, 6481–6498 CrossRef CAS.
- B. Gao, X. Li, K. Ding, C. Huang, Q. Li, P. K. Chu and K. Huo, J. Mater. Chem. A, 2019, 7, 14–37 RSC.
- D. Deepak, J. Raghav, A. Panghal, T. Singh, S. Roy and S. S. Roy, J. Alloys Compd., 2024, 978, 173391 CrossRef CAS.
- R. Paulose, R. Mohan and V. Parihar, Nano-Struct. Nano-Objects, 2017, 11, 102–111 CrossRef CAS.
- D. Wang, C. Zhang, J. Hu, T. Zhuang and Z. Lv, J. Colloid Interface Sci., 2024, 655, 217–225 CrossRef CAS PubMed.
- A. Nashim, S. Pany and K. Parida, RSC Adv., 2024, 14, 233–244 RSC.
- Y.-Y. Xie, H.-J. Chen, Q.-Y. Bai, X.-L. Wang, G. Wu, S.-C. Chen and Y.-Z. Wang, Electrochim. Acta, 2021, 390, 138772 CrossRef CAS.
- J. Nama and S. Sharma, ACS Appl. Energy Mater., 2024, 7, 10407–10416 CrossRef CAS.
- H. M. Tofail, R. Ghazi, F. A. Ghaeb, R. N. Dara, I. Kebaili, I. Boukhris, H. Ding and Z. U. Rehman, Sustainable Energy Fuels, 2025, 9, 2900–2927 RSC.
- K. Bhunia, S. Khilari, M. Chandra, D. Pradhan and S.-J. Kim, J. Alloys Compd., 2023, 935, 167842 CrossRef CAS.
- Y. Li, V. Verma, H. Su, X. Zhang, S. Zhou, T. Lawson, J. Li, R. Amal, Y. Hou and L. Dai, Adv. Energy Mater., 2024, 14, 2401341 CrossRef CAS.
- N. AbouSeada, M. G. Elmahgary, S. O. Abdellatif and K. Kirah, J. Alloys Compd., 2024, 1002, 175325 CrossRef CAS.
- A. A. Ensafi, M. M. Abarghoui and B. Rezaei, Ceram. Int., 2019, 45, 8518–8524 CrossRef CAS.
- D. Laniel, A. A. Aslandukova, A. N. Aslandukov, T. Fedotenko, S. Chariton, K. Glazyrin, V. B. Prakapenka, L. S. Dubrovinsky and N. Dubrovinskaia, Inorg. Chem., 2021, 60, 14594–14601 CrossRef CAS PubMed.
- R. Adalati, A. Kumar, M. Sharma, P. Tiwari and R. Chandra, Appl. Phys. Lett., 2020, 117, 123904 CrossRef CAS.
- H. Yang, H. Guo, K. Pang, P. Fan, X. Li, W. Ren and R. Song, Nanoscale, 2020, 12, 7024–7034 RSC.
- R. Naik, H. P. Nagaswarupa, B. H. M. Darukesha and D. M. Tejashwini, Advances in Space Radiation Detection: Novel Nanomaterials and Techniques, Springer Nature Switzerland, Cham, 2024, pp. 23–36 Search PubMed.
- N. Srinivasa, J. P. Hughes, P. S. Adarakatti, C. Manjunatha, S. J. Rowley-Neale, S. Ashoka and C. E. Banks, RSC Adv., 2021, 11, 14654–14664 RSC.
- M. R. Islam, A. K. Chakraborty, M. A. Gafur, M. A. Rahman and M. H. Rahman, Res. Chem. Intermed., 2019, 45, 1753–1773 CrossRef CAS.
- A. Trapalis, J. Heffernan, I. Farrer, J. Sharman and A. Kean, J. Appl. Phys., 2016, 120, 205102 CrossRef.
- P. Kathiravan, K. Thillaivelavan, G. Viruthagiri and N. Shanmugam, J. Indian Chem. Soc., 2024, 101, 101171 CrossRef CAS.
- E. Scattolin, M. Benedet, G. A. Rizzi, A. Gasparotto, O. I. Lebedev, D. Barreca and C. Maccato, ChemSusChem, 2024, 17, e202400948 CrossRef CAS PubMed.
- W. Huang, S. Ding, Y. Chen, W. Hao, X. Lai, J. Peng, J. Tu, Y. Cao and X. Li, Sci. Rep., 2017, 7, 5220 CrossRef PubMed.
- Y. S. Chen, J. F. Kang, B. Chen, B. Gao, L. F. Liu, X. Y. Liu, Y. Y. Wang, L. Wu, H. Y. Yu, J. Y. Wang, Q. Chen and E. G. Wang, J. Phys. D Appl. Phys., 2012, 45, 65303 CrossRef.
- D. S. Mann, S.-N. Kwon, S. Thakur, P. Patil, K.-U. Jeong and S.-I. Na, Small, 2024, 20, 2311362 CrossRef CAS PubMed.
- H. Wang, X. Li, Q. Ruan and J. Tang, Nanoscale, 2020, 12, 12329–12335 Search PubMed.
- M. Ge, X. Li, M. Zhang and Z. Liu, J. Inorg. Organomet. Polym. Mater., 2022, 32, 678–686 CrossRef CAS.
- A. Dogra, A. Kumar, M. Kapoor and N. Gupta, ChemistrySelect, 2021, 6, 7118–7122 CrossRef CAS.
- S. Yadav, A. Singh and A. K. Choubey, J. Alloys Compd., 2024, 979, 173422 CrossRef CAS.
- P.-C. Wei, S.-C. Tong, C.-M. Tseng, C.-C. Chang, C.-H. Hsu and J.-L. Shen, J. Appl. Phys., 2014, 116, 143507 CrossRef.
- A. Mahapatra, R. S. Ajimsha, D. Deepak and P. Misra, Sci. Rep., 2024, 14, 11871 CrossRef CAS PubMed.
- A. J. Addie, M. A. Mohammed and R. A. Ismail, Mater. Sci. Semicond. Process., 2022, 145, 106664 CrossRef CAS.
- H. Wang, X. Zhang, J. Xie, J. Zhang, P. Ma, B. Pan and Y. Xie, Nanoscale, 2015, 7, 5152–5156 RSC.
- Q. Xiang, J. Yu and M. Jaroniec, J. Phys. Chem. C, 2011, 115, 7355–7363 CrossRef CAS.
- N. Zhang, Y. Hu, L. An, Q. Li, J. Yin, J. Li, R. Yang, M. Lu, S. Zhang and P. Xi, Angew. Chem., 2022, 134, e202207217 CrossRef.
- A. Haruna, K. B. Dönmez, S. Hooshmand, E. Avcı, M. Qamar, S. A. Zaidi, F. Shahzad, T. S. Miller, B. K. Chakrabarti, C. A. Howard and M. K. Bayazıt, Carbon, 2024, 226, 119177 CrossRef CAS.
- X. Zhang, L. Zhang, Y. Zhu, Z. Li, Y. Wang, T. Wågberg and G. Hu, ChemSusChem, 2021, 14, 467–478 CrossRef CAS PubMed.
- D. Antipin and M. Risch, Electrochem. Sci. Adv., 2023, 3, e2100213 CAS.
- M. Y. Perdana, B. A. Johan, M. Abdallah, M. E. Hossain, M. A. Aziz, T. N. Baroud and Q. A. Drmosh, Chem. Rec., 2024, 24, e202400007 CrossRef CAS PubMed.
- Y. Li, T. Liu, Y. Liu, F. Meng and Z. Cao, Chem. Eng. J., 2024, 479, 147906 CAS.
- T. Kedara Shivasharma, A. C. Mendhe, R. Sahu and B. R. Sankapal, J. Colloid Interface Sci., 2024, 676, 739–754 CAS.
- D. B. Bailmare, P. Tripathi, A. D. Deshmukh and B. K. Gupta, Sci. Rep., 2022, 12, 3084 CAS.
- S. Sharma and P. Chand, Results Chem., 2023, 5, 100885 CAS.
- L. Lyu, J. Kang, K. Seong, C. W. Kim, J. Lim and Y. Piao, J. Alloys Compd., 2021, 872, 159610 CAS.
- M. C. C. G. Carneiro, F. T. C. Moreira, R. A. F. Dutra, R. Fernandes and M. G. F. Sales, Microchem. J., 2018, 138, 35–44 CAS.
- I. Lavagnini, R. Antiochia and F. Magno, Electroanalysis, 2004, 16, 505–506 CAS.
- P. Xu, M. Xu, J. Zhang, J. Zou, Y. Shi, D. Luo, D. Wang, H. Dou and Z. Chen, Angew. Chem., Int. Ed., 2024, 63, e202407909 CAS.
- Y. Shao, W. Lu, T. Zhang, B. Yin, B.-B. Xie, J. Ning and Y. Hu, Carbon Energy, 2025, 7, e701 CAS.
| This journal is © The Royal Society of Chemistry 2025 |






