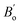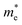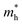Inorganic M3ACl3 (M = Ca, Sr, Ba, A = N, P, As) perovskite-derivatives for next-generation solar cells and optoelectronics: in-depth analysis of stability, optoelectronic features, and temperature-dependent carrier mobilities
Zia Ur
Rehman
 *a and
Zijing
Lin
*a and
Zijing
Lin
 *ab
*ab
aDepartment of Physics, University of Science and Technology of China, Hefei, 230026, China. E-mail: ziadesk_ustc@mail.ustc.edu.cn; zjlin@ustc.edu.cn
bHefei National Laboratory, University of Science and Technology of China, Hefei, 230088, China
First published on 18th September 2025
Abstract
The burgeoning realm of energy-harnessing technologies, coupled with significant advancements in optoelectronics, necessitates the exploration of compelling semiconductors that can effectively bridge existing technological divides. In this context, the present study meticulously examines M3ACl3 semiconductors to elucidate their multifaceted stability, encompassing structural, phase, dynamic, mechanical, and thermodynamic dimensions. Furthermore, this inquiry unveils insights into their optoelectronic properties, with a particular focus on temperature-induced carrier mobility behaviors. In this analysis, HSE06 calculations reveal the direct band gaps of the studied materials in the range of 1.7 to 2.9 eV. Specifically, Ba-containing materials possess the lowest band gaps, corresponding to the red region of the visible spectrum, while the others fall within the green and blue ranges. Moreover, these materials exhibit low effective masses, with Ba-containing variants showing the lowest effective masses and elevated relative effective mass ratios, indicating their pronounced charge carrier generation rates. In the optical domain, the absorption onset is observed in the visible range corresponding to the band gaps, displaying vis-UV light absorption characterized by high coefficients. Additionally, average mobilities range from 38.2 to 192.98 cm2 V−1 s−1 at 300 K for a carrier concentration of 1020 cm−3, showcasing remarkable electron and hole mobilities of 173 and 138.16 cm2 V−1 s−1, respectively, for Ba3PCl3. This study reveals the robust stabilities and pertinent optoelectronic features of the studied materials, indicating their potential applications in tandem solar cells and other optoelectronic devices such as RGB light-emitting diodes, semiconductor displays, and UV emitters and sensors.
1 Introduction
Green energy sources offer an eco-friendly, cost-effective, and sustainable power supply, which is a necessity of today.1 Solar energy with its global availability is one of the promising green energy sources, offering a mass-scale production of clean power. Currently, solar photovoltaics shares 5.4% of the total worldwide power generation and 8.3% of total renewable source generated power.2 In addition to harnessing this green source on a large scale, cutting-edge research of innovative materials is progressing. Compared to conventional solar cells with silicon of indirect band gap characteristics, metal lead-based halide perovskite materials with direct band gaps have exhibited exceptional power conversion efficiencies (PCE) surpassing 25%.3,4 However, despite their high efficiencies, low-cost production, lightweight designs, and stable structures, perovskite-driven solar cells are still on their way to widespread acceptance.5–7 The significant hurdles that prevent commercialization include lead-based toxicity and inferior stability at high temperatures, in moisture, and in electromagnetic environments. Lead is particularly problematic because of its bioaccumulation hazards.8,9The quest for lead-free perovskites and their derivatives is progressing rapidly, and recently, novel inorganic halide perovskite derivatives (A3BX3) have garnered the attention of researchers due to their exceptional structural attributes and direct band gap-tuned optoelectronic characteristics. Furthermore, the ability to modulate their band gaps under varying pressures and strains amplifies their versatility, facilitating the development of tunable optoelectronics.10–12 Haque et al. provides insights into the optoelectronic characteristics of Ba3SbX3 (X = F, Cl) compounds, showing band gaps of 0.943 and 0.908 eV for X = F and Cl, respectively, and revealing absorption maxima within the ultraviolet range. The authors remarked that these materials are promising for a range of optoelectronic applications, including UV photodetectors, deep-UV optical filters, and photovoltaic systems. Furthermore, the pressure-induced diminution of the band gaps in these materials underscores their potential for practicality in tunable optoelectronic devices.13 Apurba et al. investigated the thermoelectric and optoelectronic properties of Ca3PX3 (for X = Cl, Br, and I) within the framework of DFT-PBE, revealing direct band gaps of 1.4909, 1.9502, and 2.2058 eV, respectively. Their findings also indicated that the band gaps experience a contraction under compressive strain, while an expansion occurs under tensile strain, highlighting the excellent potential of these materials for enhancing light manipulation in solar energy applications and energy retention technologies.14
Additionally, the A3BX3 class of perovskite-derivatives shows high structural stability and thermal efficiencies.15 The shift in their electronic behavior under stress, such as the transition from semiconducting to metallic, further emphasizes their potential for innovative optoelectronic solutions.16 Furthermore, solar cells based on these materials exhibit a remarkable ability to sustain elevated open-circuit voltages.17 Also, the octahedral symmetry inherent in A3BX3 crystals may engender a shift in the p–s band edge state, which can markedly enhance the optical performance of the cells.18 Cumulatively, these investigations elucidate the significant potential of A3BX3 perovskite-derivatives and underscore their prospective applications across a diverse array of optoelectronic devices functioning within the visible and ultraviolet spectra.
Simulating solar cell devices necessitates intricately linked optoelectronic and transport properties, encompassing electron affinities, band gaps, dielectric constants, valence and conduction band density of states, charge carrier concentrations, and mobilities. The efficacy of optoelectronic devices depends on the carrier dynamics of semiconductors. In particular, the carrier mobilities are critical determinants of the interplay between carrier transport and recombination processes and thus play a vital role in determining optoelectronic performance.19–21 Numerous studies have revealed the finite limits of the intrinsic electron and hole mobilities of halide perovskites.22,23 In a theoretical investigation, Poncé et al. reported the electron and hole mobilities of CsPbI3 as 68 and 76 cm2 V−1 s−1, respectively, at 327 K, 41 cm2 V−1 s−1 for CsPbBr3, and a range of 30 to 80 cm2 V−1 s−1 for MAPbI3 halide perovskite. They examined that the mobilities of these materials are ultimately limited to 80 cm2 V−1 s−1 at ambient temperatures and are restrained due to the fluctuations in the Pb–I bond.22 Cucco et al. also computed the hole mobility of 41.6 cm2 V−1 s−1 at room temperature for bulk CsPbBr3.23 However, the exact carrier mobilities for A3BX3 compounds are not found in the prior investigations.
Due to a lack of exact theoretical or empirical data for mobilities of A3BX3, researchers have resorted to employing approximated or averaged mobilities in their analyses of optimizing A3BX3 based solar cells. For instance, Ria et al. have adopted mean electron and hole mobilities of 100 and 20 cm2 V−1 s−1, respectively, for perovskite-derived Ca3NF3, Ca3NCl3, Ca3NBr3, and Ca3NI3 semiconductors.24 Similarly, Islam et al. and Ghosh et al. have utilized an average mobility of 50 cm2 V−1 s−1 for Ca3NI3 and Ba3NCl3.25,26 In a parallel vein, Rahman et al. have employed the same statistics, 50 cm2 V−1 s−1, for the electron and hole mobilities of Ba3NCl3, Sr3NCl3, and Ca3NCl3 chlorides.27 However, the acquisition of experimentally validated or accurately predicted mobilities of these materials remains the most coveted attribute in the advancement of optoelectronics.
Viewing the substantial scope of inorganic A3BX3 halide perovskite-derivatives in solar cells, this study for the first time thoroughly investigated M3ACl3 (M = Ca, Sr, Ba, A = N, P, As) compounds that encompass the accurate prediction of their structural and dynamical stability characteristics and mechanical, thermodynamic, electronic, and optical properties employing the HSE06 functional, and most importantly, the prediction of carrier mobilities using the first-principles electron–phonon interaction physics, as implemented in Quantum Espresso coupled EPW (Electron–Phonon Wannier) code. Moreover, the impact of alkaline earth cations on the optoelectronic features of these materials is examined. Furthermore, this investigation is pioneering in its analysis of thermodynamic properties, the temperature-dependent dynamic stability derived from phonon dispersion spectra, and the temperature-dependent carrier mobilities of the studied pnictides. This study presents a foundational analysis of the optoelectronic properties of the materials studied, which are prerequisites for designing efficient solar cells. The explicit goal of this work is to establish a robust material-level understanding to inform subsequent device optimization studies.
2 Methodology
2.1 Framework for structural and electronic analysis
This investigation has conducted first-principles calculations using the Vienna Ab Initio Simulation Package (VASP).28–30 It has harnessed projected augmented wave potentials to adeptly account for ion–electron interactions. The PBE functional has been deployed to ascertain the structural parameters. However, for the assessment of optoelectronic properties, the Heyd–Scuseria–Ernzerhof (HSE06) hybrid functional,31 which encapsulates the Hartree–Fock exchange energy, has been adopted due to its remarkable accuracy in capturing exchange-correlation phenomena. The relaxation of crystal structures included relaxation of shape, volume, and ionic positions. Strict criteria for relaxation and energy calculations are adopted to ensure the convergence of static energies to within 1 × 10−7 eV, with maximum Hellmann forces on each atom restrained to less than 1 × 10−6 eV Å−1. Also, a gamma scheme sampled 6 × 6 × 6 k-point grid and a plane-wave basis with a cutoff energy of 350 eV, determined through convergence tests (see Fig. S1 and S2 of the SI for the details), have been utilized. Gaussian smearing with a smearing width of 0.02 is used for k-point sampling. However, the density of states is computed with a finer k-point grid of 8 × 8 × 8 for all studied cases. Structural optimization and static energy computations revealed the optimized lattice parameters, lattice energies, interatomic bond lengths, and electron density distributions.2.2 Framework for mechanical analysis
The mechanical attributes are meticulously scrutinized by using the energy-strain methodology, a preeminent technique for deducing theoretical elastic constants. This approach facilitates an exacting computation of the energy fluctuations induced by minute deformations relative to the equilibrium lattice configuration. For a given crystal system acted upon by a strain εi, the elastic energy (ΔE(V′, εi)) within the framework of harmonic approximation can be expressed using eqn (1).32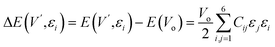 | (1) |
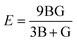 | (2) |
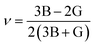 | (3) |
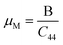 | (4) |
 | (5) |
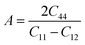 | (6) |
2.3 Framework for stability analysis
Structural stability assessments entail the meticulous computation of cohesive and formation energies to evaluate phase stability, while phonon spectra are scrutinized to ascertain dynamic stability. The cohesive and formation energies are determined by employing eqn (7) and (8).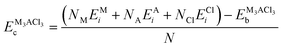 | (7) |
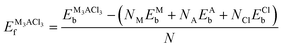 | (8) |
| N = NM + NA + NCl | (9) |
Furthermore, to assess the dynamic stability of the materials under investigation, the phonon dispersion relations across a temperature range of 200 to 500 K are computed. This meticulous evaluation facilitates a deeper understanding of the vibrational properties and stability characteristics of the materials at elevated temperatures. This analysis utilizes the temperature-dependent volumetric data derived from the QHDM. In this investigation, the super-cell method for determining phonon dispersion is adopted. It is a robust computational technique that facilitates the assessment of the dynamical matrix through finite difference approximations.36,37 This approach necessitates the construction of super-cells wherein atomic positions are made knowingly perturbed for a given crystal system. Then, energy calculations are performed systematically across all perturbed configurations that lead to the computation of force constants. Utilizing these force constants, phonon dispersion spectra are computed for the prescribed mesh sampling.
2.4 Framework for thermodynamic analysis
Employing the Quasi-Harmonic Debye Model (QHDM),38 this study has examined the pressure and temperature effects of thermodynamic properties, including volume, bulk moduli, heat capacities, Debye temperatures, and thermal expansion coefficients. This model tackles anharmonic effects by employing a harmonic approximation for all crystalline structures, even those that exist out of equilibrium. Essentially, this provides quasi-anharmonicity by the volume dependence of phonon frequencies. The efficacy of this approach in forecasting thermodynamic properties and the phase stability of solids is well-established.39–41 This model incorporates the anharmonic effects through vibrational Helmholtz free energy function Fvib as given by eqn (10),38Fvib = nkBT(9ω/8 + 3![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) ln(1 − e−ω) − D(ω)) ln(1 − e−ω) − D(ω)) | (10) |
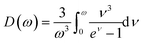 | (11) |
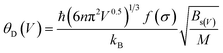 | (12) |
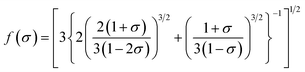 | (13) |
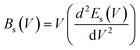 | (14) |
Poisson ratio σ is set to 0.25, a commonly used value for Cauchy solids.38
The calculation of the Debye temperature at the equilibrium volume for a given temperature and pressure leads to the computation of thermodynamic properties at that temperature and pressure, as determined from eqn (15) and (16).38,42
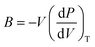 | (15) |
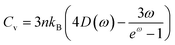 | (16) |
| Cp = Cv (1 + αγT) | (17) |
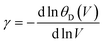 | (18) |
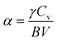 | (19) |
2.5 Framework for carrier mobility analysis
The carrier mobilities of the examined materials are computed utilizing the framework of density functional perturbation theory in conjunction with maximally localized Wannier functions, as executed in the EPW software.44,45 This methodology adeptly addresses electron–phonon interactions and facilitates the precise calculation of electron–phonon matrix elements across ultra-dense momentum grids. Subsequently, EPW employs the ab initio linearized Boltzmann Transport Equation (BTE), which elucidates the first-order response of the carrier distribution function (∂Eβfnk) to external perturbations, as delineated in eqn (20), to unveil the electronic transport properties.46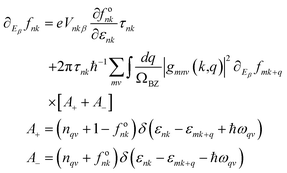 | (20) |
In this representation, fnk signifies the electron distribution function, wherein n refers to a Kohn–Sham band index and k denotes a wave vector. Vnkβ = ℏ−1∂εnk/∂kβ epitomizes the intra-band velocity matrix element associated with the eigenvalue εnk. The symbol δ denotes the Dirac delta function. The second term on the right-hand side represents an aggregate of Brillouin zone-integrated electron–phonon interactions, elucidating the scattering of electrons from state nk to state mk + q through the phonon of branch v and wave vector q. This linear response framework incorporates thermal influences derived from the Fermi–Dirac and Bose–Einstein equilibrium distribution functions for electrons (fonk) and phonons (nqv), respectively. In this formulation, τnk denotes the carrier relaxation time, which can be ascertained from eqn (21),46 delineating the scattering rates by Fermi's golden rule.
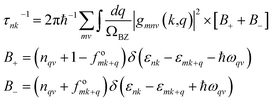 | (21) |
Subsequently, the conductivities (σαβ) and mobilities (μαβ) are calculated through eqn (22) and (23).46
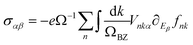 | (22) |
| μαβ = σαβ(eNc)−1 | (23) |
The prediction and effectiveness of this methodology are widely acknowledged within the materials science community.44,46–48 S. Poncé et al. developed software to benchmark experimental results for several semiconductors, including diamond, Si, GaAs, 3C-SiC, GaP, AlP, cubic BN, AlAs, AlSb, and SrO.44,47 Likewise, employing the same method, Lee et al. conducted a thorough benchmarking of the variation in electron and hole mobilities with temperature and carrier concentration in silicon semiconductors.46 Additionally, S. Poncé et al. reported comparable experimental results for the electron and hole mobilities in wurtzite gallium nitride.49 Recently, Yin et al. calculated the mobilities of CsSnBr3 inorganic halide perovskite, over a temperature range of 300 to 500 K, and found that their results align with experimental data.50
2.6 Framework for optical feature analysis
Utilizing the optical response theory as implemented within VASP, this study has elucidated the optical characteristics of the materials under scrutiny. The salient feature that delineates the optical response of a material to incident electromagnetic radiation is the complex dielectric function εω, which exhibits dependence on the incident photon frequency ω. The subsequent eqn (24) delineates the formulation of the dielectric function.32| εω = εrω + iεiω | (24) |
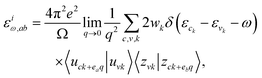 | (25) |
In this representation, Ω signifies the volume of the primitive cell, wk denotes the weights of the k-points ensuring the integral evaluates to 1, while ea and eb represent the unit vectors denoting the a and b bands, respectively. Furthermore, ck and vk epitomize the states of conduction and valence bands within the reciprocal k-space of the irreducible Brillouin zone, with zck and zvk indicating the periodic functions of the conduction and valence band states, respectively. At any given k-point, the index c traverses the unoccupied states of the conduction band, while the index v iterates over the occupied states of the valence band.32 In the presence of cubic crystalline symmetry, the off-diagonal components of the optical tensor dissipate, leaving only the diagonal components that are equivalent, thereby transforming the tensor into a scalar representation for cubic systems.52
Subsequently, by employing the Kramers–Kronig transformation as eqn (26) (ref. 51) describes, the real part of the dielectric function is computed.
 | (26) |
In this transformation, P represents the principal value. Subsequent calculations of optical characteristics utilize both real and imaginary parts. Eqn (27)–(31) describes the absorption coefficient (αω), extinction coefficient (Kω), refractive index (nω), reflectivity (Rω), and the energy-loss function (Lω), respectively.32,51,53
 | (27) |
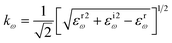 | (28) |
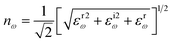 | (29) |
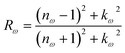 | (30) |
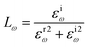 | (31) |
3 Results and discussion
3.1 Structural properties
The studied inorganic M3ACl3 (M = Ca, Sr, Ba, A = N, P, As) perovskite-derivatives crystallize in the Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m cubic crystal structures. Their optimized crystal structures are provided in Fig. S3 of the SI in detail. The unit cell of these structures comprises seven atoms. Each M2+ cation is coordinated with four co-planer Cl−1 ions, and two A−3 ions, where M–A bond lengths are equivalent to M–Cl, forming an octahedral geometry, as depicted in Fig. 1, while each A−3 ion is coordinated with 6 M2+ ions, forming an AM6 octahedral. The M2+, A−3, and Cl−1 ions are located at 3d (0, 0.5, 0), 1a (0, 0, 0), and 3c (0.5, 0.5, 0) Wyckoff positions, respectively. Γ, X, M, Γ, R, and X represent the high-symmetry k-points in the irreducible Brillouin zone of these structures, which are used to examine the phononic and electronic structures.
m cubic crystal structures. Their optimized crystal structures are provided in Fig. S3 of the SI in detail. The unit cell of these structures comprises seven atoms. Each M2+ cation is coordinated with four co-planer Cl−1 ions, and two A−3 ions, where M–A bond lengths are equivalent to M–Cl, forming an octahedral geometry, as depicted in Fig. 1, while each A−3 ion is coordinated with 6 M2+ ions, forming an AM6 octahedral. The M2+, A−3, and Cl−1 ions are located at 3d (0, 0.5, 0), 1a (0, 0, 0), and 3c (0.5, 0.5, 0) Wyckoff positions, respectively. Γ, X, M, Γ, R, and X represent the high-symmetry k-points in the irreducible Brillouin zone of these structures, which are used to examine the phononic and electronic structures.
 | ||
| Fig. 1 3D crystalline structure showing MA2Cl4 symmetry, and the k-path of IBZ of M3ACl3 (M = Ca, Ba, Sr A = N, P, As) perovskite-derivatives. | ||
Inter-atomic bond lengths at 0 and 300 K, listed in Table S1 of the SI, delineate equivalent M–A and M–Cl bond lengths. These bond lengths widen as A varies from N to P to As and M varies from Ca to Sr to Ba, which is related to the atomic radii of formula elements. These computed bond lengths agree with the results of prior investigations.12,57,58
Moreover, the plots of charge density distribution, as shown in Fig. 2, enlighten the nature of these bonds. These illustrate the contours of charge densities of studied compounds along the [200] crystallographic plane. The color bar on the right displays the scale of charge density intensity. The charge distribution illustrated in these plots reveals a pronounced overlap of electronic densities between the M and A atoms across the majority of these materials, with the notable exception of Ca3NCl3. This overlap diminishes the distinct characteristics of these related atoms, thereby indicating the formation of covalent bonds between the M and A species. In these structures, each A ion is encircled by six M ions, facilitating a shared charge among them. The plots further delineate varying extents of charge overlap, which correspond to the level of charges shared between the M and A atoms. Specifically, Ca3PCl3, Ca3AsCl3, and Sr3NCl3 exhibit a single level of charge overlap, whereas Sr3PCl3, Sr3AsCl3, Ba3NCl3, and Ba3AsCl3 demonstrate two levels of overlapping interactions. Notably, Ba3PCl3 is characterized by a triad of charge overlapping levels. Conversely, the absence of coincidental charge density contours between M and Cl suggests a predominantly ionic bonding character. It is also evident from the elongation edges of the charge density distribution of Cl towards the M atoms. However, the interactions between A and Cl atoms exhibit anti-bonding characteristics. Recent studies have highlighted the presence of various bonding types within these materials. For instance, Rahman et al. identified covalent bonds forming between strontium and nitrogen, alongside ionic bonds between strontium and chlorine, and anti-bonding characteristics in the compound Sr3NCl3.54 Similar findings have been noted in related compounds, such as Sr3PCl3,12 Sr3AsCl3,59 and Ba3NCl3,60 indicating a consistent pattern in the types of bonding among these materials.
 | ||
| Fig. 2 Charge density distributions of M3ACl3 (M = Ca, Sr, Ba, A = N, P, As) perovskite-derivatives along the [200] crystallographic plane. | ||
Fitting of the fourth-order Birch–Murnaghan equation of state, as expressed in eqn (32),61 to the energy–volume data of all studied materials, yielded the equilibrium energies and volumes (Eo, Vo), bulk moduli (B), and their derivatives (B′). The fitted energy–volume curves are provided in Fig. S4 of the SI. Additionally, the Debye temperatures are calculated using the equilibrium volumes and bulk moduli in eqn (12). Table 1 provides a summary of the lattice constants and all these structural properties, as calculated in the PBE computational domain.
| Materials | a (Å) | V st (Å)3 | V o (Å)3 | E st (eV) | E o (eV) | B o (GPa) | θ D (K) | E c (eV per atom) | E f (eV per atom) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Ca3NCl3 | 5.46 | 162.77 | 158.84 | −32.005 | −32.007 | 50.19 | 4.42 | 403.97 | 4.538 | −1.769 |
| (PBE)27 | 5.42 | |||||||||
| Ca3PCl3 | 5.76 | 190.97 | 186.78 | −30.832 | −30.834 | 40.17 | 4.20 | 358.87 | 4.369 | −2.024 |
| Ca3AsCl3 | 5.83 | 198.31 | 193.78 | −30.026 | −30.029 | 37.59 | 4.19 | 322.80 | 4.254 | −2.007 |
| Sr3NCl3 | 5.79 | 194.19 | 191.36 | −30.953 | −30.955 | 41.41 | 4.56 | 299.92 | 4.386 | −1.744 |
| (NC-PBE)54 | 5.74 | |||||||||
| Sr3PCl3 | 6.10 | 227.31 | 222.18 | −30.108 | −30.110 | 33.87 | 4.34 | 272.10 | 4.264 | −2.046 |
| (USP-PBE)12 | 6.05 | 221.35 | 30.42 | 2.56 | 282.07 | |||||
| Sr3AsCl3 | 6.16 | 233.24 | 229.93 | −29.391 | −29.393 | 31.77 | 4.33 | 251.61 | 4.162 | −2.041 |
| (NC-PBE)55 | 6.12 | 229.22 | 31.6 | −4.40 | ||||||
| Ba3NCl3 | 6.19 | 237.55 | 233.25 | −31.358 | −31.361 | 34.55 | 4.69 | 240.23 | 4.442 | −1.684 |
| Ba3PCl3 | 6.47 | 271.32 | 267.08 | −30.768 | −30.770 | 28.73 | 4.46 | 220.58 | 4.355 | −2.022 |
| (NC-PBE)56 | 6.44 | 27.745 | ||||||||
| Ba3AsCl3 | 6.55 | 280.61 | 275.51 | −30.149 | −30.151 | 27.00 | 4.45 | 206.84 | 4.268 | −2.032 |
The bulk modulus and its derivative, which measure a material's resistance to compressive forces and the variation of stress under pressure, are observed to range from 25 to 50 GPa, with the derivative from 4.1 to 4.7. This range of bulk moduli is typically preferred for applications involving relatively low-pressure forces, such as mass-scale optoelectronic devices, including solar cells, displays, optical sensors, dimming functions, optical amplifiers, and optocouplers. Materials possessing this range of bulk moduli may prove to be more economically viable and simpler to process than those exhibiting a high bulk modulus. Additionally, the positive values of B′ suggest that these materials become approximately four times harder when subjected to pressure. Moreover, the Debye temperatures are within the 205 to 405 K range which is a typical range for solids. These exhibit that rigidity in these materials decreases as volume expands due to the increased size of M and A atoms.
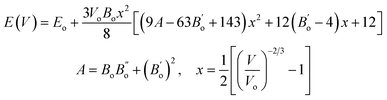 | (32) |
Moreover, the materials under investigation exhibit both phase stability and chemical stability as evidenced by their positive cohesive (Ec) and negative formation (Ef) energies listed in Table 1, as computed from eqn (7) and (8). Also, negative formation energies indicate the experimental formability and viability of the studied compounds. The comparison to prior investigations, which have employed Norm-Conserving (NC) and Ultra-Soft Pseudopotential (USP) with the PBE functional,12,54–56 shows that our results align with these.
3.2 Mechanical properties
This investigation conducted the computations for mechanical properties based on the strain–-energy theoretical framework. These computations provide the elastic stiffness tensor (Cij) comprising the elastic constants that are essential mechanical features for characterizing structural integrity and mechanical strength of materials. Essentially, these parameters signify the degree to which a substance deforms under an applied load and its capacity to revert to its original configuration upon the cessation of that load.62–64 The cubic Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m crystal structures of the studied compounds exhibit three independent elastic constants, namely C11, C12, and C44. These facilitate the examination of the material's anisotropic characteristics, mechanical stability, stiffness, and ductility or brittleness.
m crystal structures of the studied compounds exhibit three independent elastic constants, namely C11, C12, and C44. These facilitate the examination of the material's anisotropic characteristics, mechanical stability, stiffness, and ductility or brittleness.
By Born's criteria for mechanical stability, a material is deemed mechanically stable under zero stress if its second-order elastic stiffness tensor (Cij) is positive-definite, indicating that all its eigenvalues are strictly positive. More precisely, the requisite conditions include C11 − C12 > 0, C44 > 0, and C11 + 2C12 > 0.65 As listed in Table 2, the computed elastic constants are positive and manifest fulfilling the mentioned conditions and thus reveal the mechanical stability of the studied compounds. Moreover, the higher C11 than C12 and C44 also implies superior resistance towards compression along the x-axis.
| Materials | C 11 (GPa) | C 12 (GPa) | C 44 (GPa) | C 11 − C12 | C 11 + 2C12 | Pugh ratio | P c | ν | ζ | μ M | T m (K) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Ca3NCl3 | 113.08 | 18.80 | 28.22 | 94.28 | 150.69 | 1.45 | −9.4 | 0.22 | 0.35 | 1.78 | 1221.3 |
| Ca3PCl3 | 93.49 | 14.91 | 22.65 | 78.58 | 123.31 | 1.45 | −7.7 | 0.22 | 0.34 | 1.81 | 1105.5 |
| Ca3AsCl3 | 91.82 | 10.18 | 21.41 | 81.64 | 112.19 | 1.34 | −11.2 | 0.20 | 0.28 | 1.75 | 1109.57 |
| Ca3AsCl3 (ref. 58) | 97.7 | 11.87 | 20.94 | 0.108 | |||||||
| Sr3NCl3 | 94.27 | 14.74 | 20.47 | 79.53 | 123.75 | 1.54 | −5.7 | 0.23 | 0.34 | 2.02 | 1110.1 |
| Sr3PCl3 | 79.49 | 10.67 | 15.63 | 68.82 | 100.83 | 1.56 | −5.0 | 0.24 | 0.31 | 2.15 | 1022.8 |
| Sr3PCl3 (ref. 12) | 76.31 | 7.47 | 16.24 | 1.38 | −8.76 | 0.208 | 0.247 | 1.38 | 1004.02 | ||
| Sr3AsCl3 | 75.81 | 9.31 | 15.59 | 66.51 | 94.43 | 1.48 | −6.3 | 0.22 | 0.29 | 2.02 | 1001.1 |
| Sr3AsCl3 (ref. 55) | 75.07 | 10.03 | 15.21 | 1.529 | −5.18 | 0.231 | 0.284 | 2.08 | |||
| Ba3NCl3 | 78.09 | 12.57 | 13.59 | 65.52 | 103.22 | 1.76 | −1.0 | 0.26 | 0.34 | 2.53 | 1014.5 |
| Ba3NCl3 (ref. 60) | 77.90 | 12.97 | 14.01 | 1.76 | 0.27 | ||||||
| Ba3PCl3 | 70.37 | 10.34 | 10.97 | 60.03 | 91.04 | 1.82 | −0.6 | 0.27 | 0.32 | 2.77 | 968.9 |
| Ba3AsCl3 | 63.21 | 10.40 | 10.41 | 52.81 | 84.02 | 1.83 | 0.0 | 0.27 | 0.35 | 2.69 | 926.6 |
Theoretically, a Pugh ratio lower than 1.75,66 a negative Cauchy pressure (Pc),67 and a Poisson ratio of less than 0.26 all serve as criteria for distinguishing between brittle and ductile behavior in materials.68 The brittle behavior of the studied materials having Ca and Sr is established by their Pugh ratios of less than 1.75, negative Cauchy pressures, and Poisson ratios of less than 0.26. However, studied Ba-containing materials exhibit ductile behavior as manifested by their Pugh ratios higher than 1.75, Poisson ratios greater than or equal to 0.26, and approximately zero Cauchy pressures.
The graphical representation of the relationships between bulk, shear, and Young's moduli with respect to the investigated materials is depicted in Fig. 3. It illustrates a distinct correlation in which compounds containing Ca exhibit enhanced rigidity and stiffness, categorizing them as the most resilient among the studied materials. In contrast, those fortified with Sr display intermediate mechanical properties, while barium-containing compounds manifest the lowest degrees of rigidity and stiffness. Typically, pronounced rigidness and stiffness suggest a propensity for brittle behavior in materials, and ductile nature contrarily.69 Therefore, these findings also underscore the brittle character of Ca- and Sr-containing compounds and the ductile nature of Ba-containing compounds. Furthermore, it demonstrates that the linear compressibility increases along with a diminution in Vickers hardness for the sequence of examined compounds. These trends indicate that the studied Ba-containing materials exhibit greater compressibility and lower hardness, traits that are crucial for applications requiring enhanced malleability. These graphical illustrations further elucidate an inverse correlation between linear compressibility and bulk modulus, consistent with the understanding that materials exhibiting diminished compressibility generally manifest heightened bulk moduli and hardness.33 Additionally, the numerical data of these mechanical parameters, as described in Table S2 of the SI, are compared with the theoretical findings,12,55,58,60 showing close agreement with them.
 | ||
| Fig. 3 Graphical representation of bulk moduli (B), Young's moduli (E), shear moduli (G), linear compressibilities (β), and Vickers hardness (Hv) as a function of the studied compounds. | ||
Another essential mechanical metric is the dimensionless Kleinman parameter (ζ) that articulates the relative contribution of bond-bending and bond-stretching effects to the material's resistance against applied stress or strain, taking values from 0 to 1. A lower Kleinman parameter denotes a predominance of bond-bending contributing mechanical strength, often associated with brittle and rigid materials, while a higher value implies a primary contribution from bond-stretching, typically aligned with more ductile materials exhibiting reduced hardness.70,71 The materials studied display Kleinman parameter values in the range of 0.28 to 0.35, indicating a dominance of the bond-bending effect and elucidating their brittle character.
Moreover, the machinability index (μM), indicating how easily a material can be machined or shaped, is typically expressed as B/C44 for cubic crystals. A higher index suggests better machinability, meaning the material is more easily cut or molded.72,73 The machinability indices increase going from Ca3NCl3 to Ba3AsCl3 as shown in Table 2. It indicates the increasing cutting capacity or ease in shaping in this material sequence. So, this supports the brittleness of the studied Ca- and Sr-based materials and the ductile behavior of Ba-based ones.
Fine et al. determined a least squares fit formula from the elastic constant C11 and melting temperatures (Tm) for cubic elemental metals and intermetallic compounds,74 as shown in eqn (33).
| Tm = 553 + 5.91C11 | (33) |
Leveraging this formula, the melting temperatures of the studied cubic materials are determined. A decrease in Tm values going from Ca3NCl3 to Ba3AsCl3 can be observed in Table 2, which corresponds to a decreasing trend in their hardness. These values follow a pattern according to the hardness of these materials. Utilizing this empirical relation, Hosen has also determined the melting temperatures of cubic Sr3BCl3 (B = As, Sb),55 which aligns with our results.
Acoustic velocities have also been computed for the materials under investigation, highlighting the speed at which sound waves propagate through these materials. This phenomenon reflects the hardness and stiffness of the solids. Generally, higher acoustic velocities indicate a higher material's hardness and the inverse is true. Referring to the values in Table 3, it is evident that longitudinal wave velocities (LWV), also referred to as compressional wave speeds, surpass transverse wave velocities (TWV) or shear wave speeds. This disparity arises from the higher bulk moduli and lower shear moduli values that are used in computing these velocities. Furthermore, both types of wave velocities exhibit a decreasing trend from Ca3NCl3 to Ba3AsCl3, which corresponds with the observed hardness trends of these materials.
| Materials | A | UEA | LWV (km s−1) | TWV (km s−1) | AWV (km s−1) |
|---|---|---|---|---|---|
| Ca3NCl3 | 0.60 | 0.32 | 6.19 | 3.71 | 4.11 |
| Ca3PCl3 | 0.58 | 0.37 | 5.86 | 3.51 | 3.89 |
| Ca3AsCl3 | 0.53 | 0.52 | 5.36 | 3.28 | 3.62 |
| Sr3NCl3 | 0.52 | 0.55 | 4.81 | 2.84 | 3.14 |
| Sr3PCl3 | 0.45 | 0.79 | 4.57 | 2.69 | 2.98 |
| Sr3PCl3 (ref. 12) | 0.47 | 4.464 | 2.710 | 2.995 | |
| Sr3AsCl3 | 0.47 | 0.72 | 4.32 | 2.57 | 2.85 |
| Sr3AsCl3 (ref. 55) | 0.467 | ||||
| Ba3NCl3 | 0.42 | 0.99 | 3.99 | 2.27 | 2.52 |
| Ba3PCl3 | 0.37 | 1.32 | 3.89 | 2.19 | 2.44 |
| Ba3AsCl3 | 0.39 | 1.12 | 3.67 | 2.06 | 2.29 |
The Poisson ratio within the 0.25 to 0.5 range indicates that central force interactions predominantly govern the behavior of a crystal. In such a case, the interatomic forces are primarily directed toward the centers of the atoms, a characteristic commonly observed in cubic crystal structures. When the Poisson ratio falls outside this range, non-central force interactions become more significant as the characteristics of non-cubic or anisotropic crystals. Moreover, the Cauchy relations hold for central force crystals and deviate in the case of non-central crystals.75,76 This calculation shows that the Poisson ratios of the studied Ba-containing materials greater than 0.25 signify that these are central force crystals that also obey Cauchy relations. However, the Poisson ratios of Ca- and Sr-containing materials are closely below 0.25, suggesting that their anisotropic character originates from non-central interatomic forces. The elastic anisotropy of the studied materials reveals the directional dependence of their mechanical properties. In this regard, the Zener anisotropic factor (A) and the Universal Elastic Anisotropy (UEA) index offer a quantitative measure of the anisotropy of compounds. Specifically, the Zener anisotropic factor accounts for the anisotropy of cubic crystals.77,78 Factor A equals 1 indicating a state of zero anisotropy, and conversely, any deviation from 1 signifies the anisotropic behavior. In contrast, the UEA index is zero for isotropic materials and nonzero otherwise. Both parameters are detailed in Table 3, which illustrates the anisotropic behavior of the studied materials. Transitioning from Ca3NCl3 to Ba3AsCl3, the anisotropy indices gradually increase, highlighting more direction dependence of mechanical behavior. Fig. 4 presents 3D topological surface plots that depict the form of elastic anisotropy of Young's moduli, shear moduli, Poisson ratios, and linear compressibilities. Meanwhile, Fig. S5 of the SI shows the 2D contours of elastic anisotropy and highlights the minima, maxima, and the relevant anisotropy factor of these properties. These demonstrate isotropic behavior in linear compressibilities while showing directional dependence in Young's moduli, shear moduli, and Poisson ratios for all the materials studied. Additionally, although the anisotropic plots are identical for each material, the indices of anisotropy vary, and Ba3AsCl3 shows the highest indices of anisotropy.
 | ||
| Fig. 4 3D surface plots of elastic anisotropy of Young's moduli, linear compressibility, shear moduli, and Poisson ratios of the investigated materials. | ||
Our computed mechanical properties align with previous theoretical investigations.12,55,58,60 While no pertinent experimental work currently exists, our findings may prove instrumental in devising an experimental framework for these materials.
3.3 Dynamic stability analysis
Phonon dispersion spectra are shown in Fig. 5, emphasizing the dynamic stability of materials under investigation. This figure illustrates the dispersion spectra at high-symmetry points in the first Brillouin zone over a temperature range from 200 to 500 K, with a step size of 50 K. These spectra evidence that the N-containing materials lack dynamic stability, as indicated by the presence of negative frequency modes across the entire temperature range. Contrarily, the Sr- and Ba-based materials exhibit positive phonon frequencies, highlighting their dynamic stability. These spectra demonstrate that phonon frequencies decrease as the temperature increases, which transpires due to the rising Grüneisen parameters and thermal expansion coefficients with temperature rise of the investigated materials. Despite frequency decreases with temperature, the dynamic stability of the Sr- and Ba-based materials is not disregarded by any temperature change. | ||
| Fig. 5 Graphical representations of temperature-dependent phonon dispersions of the examined compounds. | ||
As can be observed, optical phonon frequencies decrease with an increase in temperature, while acoustic phonon frequencies remain largely unaffected. These spectra indicate that the highest optical frequencies observed are 249.13, 217.04, 207.97, 164.12, 158.41, and 123.47 cm−1 for Ca3PCl3, Ca3AsCl3, Sr3PCl3, Sr3AsCl3, Ba3PCl3, and Ba3AsCl3, respectively. These frequencies correspond to temperatures of 358.44, 312.27, 299.22, 236.13, 227.92, and 177.65 K. Furthermore, multiplying these temperatures by 3/2 yields values of 537.66, 468.405, 448.83, 354.195, 341.88, and 266.475 K, at which point the heat capacities of these materials at constant volume approximately approach the Dulong–Petit limit of 3R. These values indicate that the examined materials reach their maximum heat capacity at relatively low temperatures. Additionally, a steeper slope in the acoustic modes suggests increased acoustic velocities within these crystals. All the dispersion spectra are alike along the k-path but with differences in frequencies. Looking into the Ba3AsCl3 spectra as a case, among the acoustic modes, two low-frequency transverse acoustic (TA) modes correspond to transverse waves and shear speeds in solids, while the longitudinal acoustic (LA) mode with a steeper slope signifies a longitudinal wave, indicative of a higher longitudinal speed. Likewise, the lower-frequency transverse optical (TO) modes represent transverse waves, and the higher-frequency longitudinal optical (LO) modes are associated with the most energetic longitudinal waves. Other spectra have a resemblance with differences in frequencies.
The lack of dynamic stability in studied N-based materials indicates the off-centering of the B-site cation (N-site), which is a common concern in perovskite structures, leading to distortions and the prospect of low-symmetry tilted structures. Essentially, the relatively small radius of N atoms in these structures causes substantial voids within the lattice, undermining structural compactness and leading to the emergence of imaginary frequency phonon modes. This inherent dynamic instability of M3NCl3 materials signifies a propensity for structural degradation, rendering them susceptible to phase transitions or structural decomposition. Furthermore, their instability can preclude electron–phonon coupling and compromise carrier mobility calculations, thereby making it futile to predict transport properties. Due to these implications, the M3NCl3 materials are dropped for further analysis in this work. However, the low-symmetry tetragonal and rhombohedral configurations, as potential distortions of their tilted structures, necessitate a parallel comprehensive investigation.
3.4 Thermodynamic properties
Utilizing QHDM, this study has computed the thermodynamic properties under varying pressure and temperature conditions where pressure ranges from 0 to 10 GPa, and temperature ranges from 0 to 500 K. Fig. 6 illustrates the cell volumes, isothermal bulk moduli, and Debye temperatures for the given pressure range at 300 K and for the given temperature range at a static pressure of 0 GPa. These characteristics demonstrate that the investigated structures exhibit typical contraction under the influence of applied pressure while they undergo an expansion with increasing temperature. Under compressive forces, atomic constituents are compelled into closer proximity, effectively diminishing the overall crystallographic volume and augmenting its hardness. This improvement of hardness is evident by the observable elevation in both the bulk modulus and Debye temperatures. Conversely, temperature elevation induces dilation of the lattice parameters, culminating in an overall enlargement of the crystalline volume. This thermal expansion is attributable to the enhanced vibrational motion of atoms within the lattice structure, which subsequently leads to a reduction in the material's hardness. Declining trends in both bulk modulus and Debye temperature with temperature rise also substantiate this reduction in hardness. The relationship delineated by eqn (12) elucidates the intricate dependence of the Debye temperature, θD, on the bulk modulus. It translates that an elevation in the bulk modulus under applied pressure leads to a heightened θD. This increase signifies enhanced material stiffness and elevated melting points. Conversely, an increase in temperature induces a decrement in the bulk modulus, thereby resulting in a concomitant reduction of θD. | ||
| Fig. 6 Thermodynamic trends of unit cell volumes, bulk moduli, and Debye temperatures of M3ACl3 (M = Ca, Sr, Ba, A = P, As) compounds. | ||
Fig. 7 illustrates the molar heat capacities (Cp and Cv) per formula unit. It shows that the heat capacities are elevating when moving from Ca3NCl3 to Ba3AsCl3. This trend is attributed to the lowering of the frequencies of the acoustic phonons in this series of materials, as shown in Fig. 5. Additionally, the elevated heat capacities correspond to the increased number of electrons near the Fermi level that enhances the electronic contribution to heat capacities. However, the electronic contribution is always smaller than the phonon contribution. Consequently, the studied Ba-based materials are comparatively more stable structures that can absorb, withstand, and store more thermal energy.
 | ||
| Fig. 7 Thermodynamic behavior of molar heat capacities (Cp, Cv) of M3ACl3 (M = Ca, Sr, Ba, A = P, As) compounds. | ||
Under elevated pressure conditions, both molar heat capacities exhibit a decrement, thereby diminishing their thermal capacity. It indicates that the material's temperature will rise quickly with minimal thermal energy input under high-pressure regimes. This phenomenon occurs due to the augmented stiffness of the studied materials, which is reflected in their enhanced bulk moduli and elevated Debye temperatures, as illustrated in Fig. 6. Furthermore, the lattice vibrational frequencies mount under pressure. These high-frequency oscillations, primarily associated with optical phonons, contribute less to the overall heat capacity in comparison to the low-frequency acoustic phonons.
Upon increasing the temperature, the molar heat capacities increase rapidly up to 100 K, primarily due to the significant contribution of low-frequency acoustic phonons. Beyond this point, both heat capacities gradually rise to saturation values where the contribution of acoustic phonon modes to heat capacity becomes less, while optical phonons contribute. The Debye temperatures, which indicate the highest degree of vibrational motion in solids, as summarized in Table 1 are 403.97, 358.87, 322.80, 299.92, 272.10, 251.61, 240.23, 220.58, and 206.84 K traversing from Ca3NCl3 to Ba3AsCl3, respectively. Theoretically, the heat capacities of these materials will reach the Dulong–Petit limit at temperatures exceeding their Debye temperatures. The Dulong–Petit limit is equal to 3NR, which for these materials amount to 174.59 J mol K−1. Considering the Debye temperatures, the barium-based materials will reach their Dulong–Petit limit at lower temperatures than the others. Specifically, Ba3AsCl3, with the lowest Debye temperature, will saturate at low temperature, while Ca3NCl3, having the highest Debye temperature, will reach saturation at a higher temperature, as Fig. 7 depicts.
Fig. 8 delineates the influence of pressure and temperature on thermal expansion coefficients (α) and entropies (S) of the studied materials, revealing no discontinuities during thermal fluctuations, indicative of their thermal stability. An inverse correlation between α and compressive forces is observed, as characterized by eqn (19). Increased phonon frequencies from compression enhance material hardness, thereby reducing α, which benefits in mitigating crack propagation at thermal interfaces. In contrast, elevated temperatures induce volumetric expansion, yielding softening behavior accompanied by reduced bulk moduli and Debye temperatures, which enhances α in line with heat capacity trends. The increase in α above 100 K is attributed to anharmonic lattice vibrations prevailing at elevated temperatures, consistent with the Grüneisen relation α ∝ γCv/3B, where γ amplifies with bond softening. However, the high α in the range of 8 to 9 × 10−5 K−1 at 300 K of the studied materials exhibits mismatch of nearly Δα ≈ 7.5 × 10−5 K−1 with standard substrates that are generally used in silicon or thin-film solar cells (e.g., soda lime glass with α ≈ 9.1–9.5 × 10−6 K−1,79 engendering interfacial stresses, which could lead to delamination unless mitigated by negative thermal expansion interlayers or buffer layers with intermediate coefficients.80
 | ||
| Fig. 8 Thermodynamic behavior of thermal expansion coefficients (α), and entropies (S) of M3ACl3 (M = Ca, Sr, Ba, A = P, As) compounds. | ||
Moreover, the entropies of the investigated systems decrease under heightened pressure and increase with temperature. Elevated pressure constrains molecular motion, resulting in a more ordered configuration and reduced entropy due to a decrease in accessible microstates. Conversely, increased temperature augments crystal volume and kinetic energies, boosting the number of microstates and fostering disorder, thereby elevating entropy. The pronounced entropy at higher temperatures indicates phase stability against decomposition, impeding halide migration in solar cells during operational heating.
Additionally, the thermodynamic variables of the studied materials at ambient temperature are delineated in Table S3. These reveal lower values of B and θD, and higher values of Cp, Cv, and α for Ba3PCl3 and Ba3AsCl3 than others, which underscores their comparatively lower hardness. This investigation found no theoretical or experimental record of the thermodynamic properties of the studied inorganic halide perovskite derivatives. Our research bridges this gap and presents for the first time a comprehensive understanding of the thermodynamic variables of the investigated materials. These thermodynamic characteristics manifest a consistent and predictable behavior devoid of any anomalous trends or discontinuities, thereby highlighting the thermal stability of the studied material. Such stability is necessary in applications where materials are subjected to fluctuating thermal environments, ensuring reliability and performance under varying conditions.
3.5 Electronic properties
Following structural optimization and QHDM calculation of lattice volumes at 300 K, the electronic properties, including band structures, the partial density of states (PDOS), carrier effective masses, and carrier mobilities, are determined. Since the accuracy and efficacy of HSE06 calculations have been agreed upon by the scientific community,81–84 this study utilized the HSE06 exchange-correlation functional to ensure the accuracy of results. | ||
| Fig. 9 Graphical representations of the band structures (HSE06) along the high symmetry k-path in IBZ of the examined M3ACl3 (M = Ca, Sr, Ba, A = P, As) compounds at 300 K. | ||
Moreover, the band gaps are observed to diminish as the atomic size of the alkaline earth metal (M) progresses from Ca to Sr to Ba in these structures. This attenuation is attributed to the augmented orbital overlap resulting from the increased atomic dimensions. Such pronounced overlap consequently narrows the disparity in energy between the valence and conduction bands.95
Table 4 exhibits a comparative analysis of the band gaps, demonstrating that our results are in close alignment with previously calculated findings using the HSE06 methodology. Given that these materials are novel and have only recently begun to be scrutinized through theoretical investigations, no precise experimental comparisons are currently available for validation. Typically, band gaps less than or about 1.7 eV are deemed optimal for the top cells in tandem photovoltaic systems.96 In contrast, band gaps ranging from 2.5 to 2.9 eV present an excellent choice for optoelectronic applications, including light-emitting diodes (LEDs), as well as laser diodes and ultraviolet sensors. Specifically, for the development of solid-state lighting, semiconductor displays, and multi-color red-green-blue LEDs, semiconductors with band gaps in the 1.7 to 3.1 eV range are desirable.97 Consequently, the studied Ba-containing variants emerge as particularly advantageous for solar cell applications, whereas Ca- and Sr-containing compounds offer promising prospects for a variety of other optoelectronic devices.
 | ||
| Fig. 10 Graphical representation of partial density of states (HSE06) of the examined M3ACl3 (M = Ca, Sr, Ba, A = P, As) compounds at 300 K. | ||
Like the orbital interactions in the studied Ca-containing materials, the Sr- and Ba-containing compounds exhibit p–d bonding interactions and p–p anti-bonding interactions. In the bottom line, for M3ACl3 compounds, the hybridized A-p and M-d states interact at the conduction band edge, constituting a covalent bond between them while the interactions among A-p and M-p states, as well as A-p and Cl-p states, exhibit anti-bonding characteristics. Other states contribute negligibly to valence and conduction band edges. Moreover, the Ba-5d states are more delocalized due to the screening effect of electrons as compared to the Ca-3d and Sr-4d states, therefore the Ba-5d states hybridize more strongly with P-3p or As-4p orbitals. This not only results in diminished band gaps of studied Ba-based studied but the stronger p–d bonding interactions also stabilize these compounds.
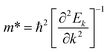 | (34) |
| Materials | D | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Γ–M | Γ–R | Γ–X | Avg. | Γ–M | Γ–R | Γ–X | Avg. | Γ–M | Γ–R | Γ–X | Avg. | |
| Ca3PCl3 | 0.736 | 0.537 | 2.071 | 1.114 | 0.913 | 0.654 | 0.933 | 0.833 | 0.806 | 0.821 | 2.219 | 1.337 |
| Ca3AsCl3 | 0.745 | 0.540 | 2.056 | 1.113 | 0.866 | 0.614 | 0.886 | 0.788 | 0.860 | 0.879 | 2.321 | 1.412 |
| Sr3PCl3 | 0.690 | 0.516 | 1.863 | 1.023 | 1.014 | 0.564 | 1.038 | 0.872 | 0.680 | 0.915 | 1.794 | 1.173 |
| Sr3AsCl3 | 0.699 | 0.515 | 1.910 | 1.041 | 0.936 | 0.523 | 0.958 | 0.805 | 0.747 | 0.985 | 1.994 | 1.293 |
| Ba3PCl3 | 0.680 | 0.502 | 1.978 | 1.053 | 0.893 | 0.432 | 0.921 | 0.748 | 0.761 | 1.162 | 2.148 | 1.407 |
| Ba3AsCl3 | 0.692 | 0.503 | 2.051 | 1.082 | 0.828 | 0.404 | 0.854 | 0.695 | 0.836 | 1.245 | 2.401 | 1.557 |
It is noted that both types of carriers – electrons and holes – show different effective masses depending on the direction. Notably, all studied materials exhibit lower electron effective masses than holes along the Γ–M and Γ–R directions that are associated with relatively high edge conduction band dispersion and a flat valence band at Γ, respectively. However, the exception is witnessed for the studied Ba-based materials, which have lower hole masses than electrons along the Γ–R direction. Contrarily, along the Γ–X direction, electrons have a larger effective mass than holes in all studied materials, which is associated with a relatively more flat conduction band than the valence band in this direction. Specifically, the effective masses of both carriers along the Γ–M and Γ–R directions are lower compared to the Γ–X direction. It indicates the higher carrier mobilities along the diagonal directions but lower mobilities along b2-direction as depicted by the high symmetry path in Fig. 1. This behavior is contingent upon the spatial distribution of the electrostatic potential within the material. The elevated effective masses of carriers along the Γ–X directions suggest a more pronounced potential along the b2 direction compared to alternative pathways. Consequently, carriers exhibit preferential mobility along the remaining two directions rather than along the b2 axis.99 Liu et al. reported similar band structures and effective masses of Ba3MX3 (M = As, Sb, X = Cl, Br, I). They observed higher electron effective mass than holes along the Γ–X direction, which were due to the flat conduction band dispersion.100 Our computed effective masses and their trends along different directions for the Ba3AsCl3 exactly match Liu et al. findings for the same material.
Moreover, both electron and hole effective masses along the Γ–M direction are moderate and reasonable in all materials. But remarkably, the effective masses along the Γ–R direction are small and highly desirable for thin-film solar cells and other optoelectronic applications. On the other hand, the average electron effective masses are greater than those of holes, indicating high hole mobilities and efficient hole transport in these semiconductors. This suggests that they can function effectively as hole transport layers in optoelectronic devices, particularly in solar applications. Hosen has also reported observing higher electron effective masses and lower hole effective masses for Sr3AsCl3 and Sr3SbCl3.55 However, both types of carrier masses are below 1.5, indicating higher mobility, lower recombination rates, and improved optoelectronic performance of the studied materials.101–103
The relative ratio of the effective masses of photo-induced electrons and holes 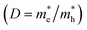 is noteworthy for evaluating the rates of recombination of charge carriers. This ratio is also crucial for illustrating the generation process of charge carriers and analyzing the photocatalytic performance. The deviation of D from 1 attributes the difference in electron and hole mobilities, indicating higher rates of recombination of charge carriers and poor optoelectronic efficiencies.99,104Table 5 shows that the D values along all directions deviate from 1, which infers the lower recombination rates of electron–hole pairs in the investigated compounds. Specifically, along the Γ–X direction, the ratio deviates more than in the other two directions. It indicates lower recombination rates in this direction, resulting in a higher generation of photo-induced carriers and a longer lifespan for these carriers.105 Zhang et al. reported the D values for bismuth oxyhalides (BiOX, X = Cl, Br, I) to comprehend their photocatalytic properties. They reported the D values in the range from about 0.7 to 21 along various directions of k vectors. They found that the facets with D values 1 are ill-suited for photocatalytic activity.104
is noteworthy for evaluating the rates of recombination of charge carriers. This ratio is also crucial for illustrating the generation process of charge carriers and analyzing the photocatalytic performance. The deviation of D from 1 attributes the difference in electron and hole mobilities, indicating higher rates of recombination of charge carriers and poor optoelectronic efficiencies.99,104Table 5 shows that the D values along all directions deviate from 1, which infers the lower recombination rates of electron–hole pairs in the investigated compounds. Specifically, along the Γ–X direction, the ratio deviates more than in the other two directions. It indicates lower recombination rates in this direction, resulting in a higher generation of photo-induced carriers and a longer lifespan for these carriers.105 Zhang et al. reported the D values for bismuth oxyhalides (BiOX, X = Cl, Br, I) to comprehend their photocatalytic properties. They reported the D values in the range from about 0.7 to 21 along various directions of k vectors. They found that the facets with D values 1 are ill-suited for photocatalytic activity.104
In summary, the electronic characteristics of the investigated compounds reveal band gaps situated within the visible spectrum, underscoring their potential utility in vis-UV-driven optoelectronic applications. Furthermore, the effective masses and D ratios signify superior carrier mobilities and diminished recombination rates of electron–hole pairs, culminating in enhanced optoelectronic efficacy. Notably, the Ba3PCl3 and Ba3AsCl3 compounds exhibit greater promise for solar cell applications, attributable to their smaller band gaps, lower effective masses, and elevated D values.
Fig. 11 illustrates the fundamental scattering mechanisms that influence the relationship between mobilities and carrier concentrations, using arbitrary values for illustration. In regime I, which corresponds to low-to-moderate carrier concentrations up to 1019 cm−3, unscreened electron–phonon scattering dominates the transport phenomena. The long-range electron–phonon interactions remain unaffected by the rise in carrier concentration, and the scattering rate shows invariance, resulting in constant mobility. Beyond this threshold, in regime II, the screening effect becomes dominant, overwhelming other scattering mechanisms, until very high carrier concentrations. The influence of carrier screening substantially reduces polar optical phonon scattering, also known as Fröhlich interactions. At high concentrations, within regime III, mobility decreases because of band non-parabolicity,106 which causes the Fermi level to drop deeper into the conduction band. This shift increases the effective masses of the carriers, leading to reduced mobility. Moreover, optical phonon scattering becomes more significant, and inelastic emission of LO phonons becomes prominent. Moreover, acoustic phonon scattering, a short-range interaction, is independent of carrier concentration, as depicted. Therefore, the mobility does not depend on low-energy acoustic phonon scattering.
 | ||
| Fig. 11 Schematic illustration of the scattering mechanism and mobility driven by carrier concentration. | ||
Our results align well with previous studies. Jacoboni et al. documented similar peak mobility of GaAs at a concentration of 3 × 1018 cm−3 due to the screening effect, followed by a drop from non-parabolicity.107 Wu et al. have reported theoretical results of carrier concentration-dependent electron–hole mobilities of CsBX3, Cs2BX6, and for Cs3B2X9 compounds. The authors witnessed that the mobilities for each compound remain nearly invariant within the carrier concentration range of 1016 to 1019 cm−3.108 In a couple of experimental investigations, Tanase et al. observed analogous phenomena concerning the hole transport layers of field-effect transistors and light-emitting diodes.109,110 Their findings reveal that mobilities remain static up to a threshold of 1022 cm−3, after which they exhibit an ascendant trend, adhering to a power law described by the expression (μ ∼ NTexp,on/T−1), as articulated in reference eqn (3).109 Here, Texp,on signifies a quantification of the breadth of the exponential density of states. Likewise, Li et al. analytically corroborated the presence of analogous effects concerning mobility versus carrier concentrations, specifically the escalation of mobility following surpassing a threshold concentration.
Furthermore, all three components of hole mobilities are higher than electron mobilities for Ca3PCl3, Sr3PCl3, and Ba3AsCl3 compounds, which is due to their lower hole effective masses along the Γ–X direction of the Brillouin zone, as Table 5 expresses. Zhang et al. also reported higher hole mobilities than electron mobilities of BSb semiconductors at 300 K and elucidated that higher mobilities are due to small effective masses, high sound speeds, and high optical phonon frequencies.111 The zz-component of electron mobility is notably higher in four structures: Ca3AsCl3, Sr3PCl3, Sr3AsCl3, and Ba3PCl3, which is due to their lower electron effective masses along Γ–X. Fig. 12 also illustrates that Ba3AsCl3 demonstrates isotropic behavior across all tensor components, exhibiting uniform characteristics for both electron and hole mobilities. Moreover, as delineated in Table 6, Ca3PCl3 exhibits the congruent maximum mobilities for holes and electrons at a concentration of 1020 cm3, and Ba3PCl3 holds the highest electron mobility within the low to intermediate concentration range.
| Materials | N h (cm−3) | μ maxh (cm2 V−1 s−1) | N e (cm−3) | μ maxe (cm2 V−1 s−1) |
|---|---|---|---|---|
| Ca3PCl3 | 1020 | 233.56 | 1020 | 233.56 |
| Ca3AsCl3 | 1021 | 31.57 | 1022 | 78.63 |
| Sr3PCl3 | 1020 | 94.86 | 1020 | 104.36 |
| Sr3AsCl3 | 1020 | 52.59 | 1020 | 128.15 |
| Ba3PCl3 | 1020 | 158.10 | 1013 to 1017 | 515.27 |
| Ba3AsCl3 | 1022 | 108.37 | 1022 | 67.76 |
Fig. 13 elucidates the temperature dependence of carrier mobilities across various concentrations. Conventionally, carrier mobilities exhibit a decrement with an elevation in temperature,47 a trend that our findings corroborate. This phenomenon arises due to the weakening of electron–phonon coupling at elevated temperatures, which diminishes the scattering rates and consequently leads to reduced mobilities. However, concurrently, the density of phonons augments according to the Bose–Einstein statistics, whereby the occupancy of phonons in a specified energy state proliferates with increasing temperature.112 The proliferation of phonons amplifies phonon–phonon interactions, commonly referred to as Umklapp scattering, which becomes increasingly prominent at elevated temperatures and predominantly contributes to the attenuation of carrier mobility.113
 | ||
| Fig. 13 Carrier mobilities of the studied materials as a function of temperature for various carrier concentrations (N). | ||
These findings elucidate that Ba3PCl3 possesses the most elevated carrier mobilities among all evaluated materials, showcasing a peak zz-component of electron mobility at an extraordinary 1170 cm2 V−1 s−1 at a concentration of 1019 cm−3 at 200 K. Conversely, Ca3AsCl3 demonstrates the lowest mobilities at the same concentration level. These results underscore a pronounced transition from superior hole mobilities to enhanced electron mobilities correlated with variations in concentration levels. At 1019 cm−3, Ca3PCl3, Ca3AsCl3, Sr3PCl3, and Ba3AsCl3 manifest superior hole mobilities, a trend that undergoes inversion at 1022 cm−3 for Ca3PCl3, at 1020 cm−3 for both Ca3AsCl3 and Sr3PCl3, and at 1021 cm−3 for Ba3AsCl3. Notably, Ba3PCl3 consistently displays higher electron mobilities across all examined concentration levels. In probing MoS2, Poncé et al. identified superior hole mobilities compared to electron mobilities for temperatures beneath approximately 250 K.114 In parallel, Xia et al. carried out an experimental inquiry into the electrical mobility of defect-free single crystals of MAPbI3, reporting μe = 33 cm2 V−1 s−1 and μh = 50 cm2 V−1 s−1 at 300 K. They also performed mobility calculations employing the Boltzmann Transport Equation, which also indicated higher hole mobilities than electron mobilities for temperatures spanning 0 to 400 K.115
Moreover, our findings demonstrate that hole mobilities exhibit a rapid decline in comparison to electron mobilities as temperature increases. This phenomenon is attributable to the pronounced increase in the effective mass of holes with rising temperatures. A comparable observation is documented by Zhou et al., who conducted a study on the mobilities of GaP, InP, GaAs, InAs, GaSb, and InSb within a temperature range of 100 to 500 K. They discovered that electron mobilities decay by one order of magnitude across this temperature range, while hole mobilities decline at a fast rate.48 Herz cataloged the mobilities of various films, single crystals, thin films, and poly-crystalline specimens of halide perovskites, ranging from a mere 0.2 cm2 V−1 s−1 for FAPbI3 films to an impressive 2320 cm2 V−1 s−1 for MASnI3 poly-crystalline materials, while the majority of measurements fall below 50 cm2 V−1 s−1.116 This compilation of data reveals significant discrepancies in mobilities, presenting a wide spectrum of mobility values for a single material. For instance, reference Table 1 (ref. 116) illustrates that for single crystalline MAPbI3, reported mobility values fluctuate between 2.5 and 164 cm2 V−1 s−1 for hole mobility and from 115 to cm2 V−1 s−1 for the electron–hole sum mobility, indicative of a staggering discrepancy of nearly two orders of magnitude. Given that identical single crystals sharing the same stoichiometry and crystallographic structure should yield consistent charge-carrier mobilities, and this extensive variation can be primarily attributed to experimental uncertainties.
Table 7 lists the averaged mobilities and their corresponding Fermi levels at 300 K for a concentration level of 1020 cm−3. In an ab initio method study, Poncé et al. documented the electron and hole mobilities of CsPbI3 iodides to be 68 and 76 cm2 V−1 s−1, respectively, at 327 K, and an average 41 cm2 V−1 s−1 for CsPbBr3 at room temperature. In the case of MAPbI3, the electron and hole mobility averages at room temperature fluctuate within a range of 30 to 80 cm2 V−1 s−1 at ambient temperature, attributable to the fluctuations in the Pb–I bond structure.22 Similarly, Cucco et al. computed the hole mobility of 41.6 cm2 V−1 s−1 for bulk CsPbBr3 at room temperature.23 This comparison illustrates reasonable agreement between our results and findings of ABX3 type halide perovskites. Instead, Ba3PCl3 exhibits more distinguished electron and hole mobilities of 173.0 and 138.16 cm2 V−1 s−1, respectively. In this view, our findings indicate the commendable performance of the evaluated semiconductors and provide a substantial reference point for future investigations.
| Materials | ε eF (eV) |
![[small mu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0cd.gif) e (cm2 V−1 s−1)
e (cm2 V−1 s−1) |
ε hF (eV) |
![[small mu, Greek, macron]](https://www.rsc.org/images/entities/i_char_e0cd.gif) h (cm2 V−1 s−1)
h (cm2 V−1 s−1) |
|---|---|---|---|---|
| Ca3PCl3 | 8.039 | 82.23 | 5.247 | 192.98 |
| Ca3AsCl3 | 8.425 | 38.20 | 5.394 | 25.06 |
| Sr3PCl3 | 7.692 | 80.96 | 5.212 | 82.11 |
| Sr3AsCl3 | 7.954 | 80.78 | 5.327 | 43.43 |
| Ba3PCl3 | 7.249 | 173.00 | 5.447 | 138.16 |
| Ba3AsCl3 | 7.399 | 60.32 | 5.455 | 90.33 |
| CsPbI3 (ref. 22) | 68 | 76 | ||
| CsPbBr3 (ref. 22) | 41 | 41 | ||
| CsPbBr3 (ref. 23) | 41.6 |
3.6 Optical properties
This investigation entails the dielectric functions (εrω, εiω), absorption (αω) and extinction coefficients (κω), along with the reflective (Rω) and refractive (nω) indices, and energy-loss functions of the materials under examination. This analysis employs hybrid functional (HSE06) energy calculations leveraging the QHDM-computed lattice parameters at 300 K. It is observed that in these calculations, the off-diagonal tensor components of the dielectric function diminish, and the diagonal elements are all equal due to the cubic structural symmetry of these materials. Therefore, all optical properties manifest isotropic functions of incident photon frequency (ω). The dielectric function plays a pivotal role in elucidating the optical characteristics of materials. It delineates the material's response to incident photons or applied electric fields. Typically, the real part of the dielectric function elucidates the polarizability that occurs due to incident photons or in the presence of an electric field.117,118 Moreover, the peaks in the real part correlate with the material's resonance frequencies, indicating electronic polarization that occurs in the ultraviolet region or ionic polarization that is generally evident in the infrared domain. These peaks signify that materials exhibit pronounced responsiveness to incident photons whose frequencies coincide with the resonance frequencies. Conversely, the imaginary component encapsulates the optical transitions between the valence and conduction bands in reaction to incident photons. The peaks in the imaginary component signify a substantial optical transition density.Fig. 14 delineates that the εrω functions are non-zero at null frequency or under static conditions, and this value is characterized as the dielectric constant (εo) of the material. The computed values for Ca3PCl3, Ca3AsCl3, Sr3PCl3, Sr3AsCl3, Ba3PCl3, and Ba3AsCl3 are 2.68, 2.38, 2.61, 2.68, 2.35, and 2.41, respectively. This parameter is critically significant in the design and optimization of solar cells and optoelectronic devices. Generally, elevated dielectric constant values indicate diminished electron–hole pair recombination rates. Also, a higher εo effectively mitigates the coulombic interaction between electrons and holes.119–121 Furthermore, the dielectric constant has an inverse relation with band gaps so a higher dielectric constant is indicative of a lower band gap.122 Consequently, superior dielectric constants are intrinsically linked to the enhanced optoelectronic performance of materials.123 The values exhibited by the studied materials are commendable and promising for optoelectronic applications. The peak values of the εrω function are 4.20, 3.89, 3.96, 4.12, 3.21, and 3.35 at frequencies of 3.19, 3.18, 3.01, 2.80, 2.10, and 2.10 eV for Ca3PCl3, Ca3AsCl3, Sr3PCl3, Sr3AsCl3, Ba3PCl3, and Ba3AsCl3, respectively, and they are within the visible spectrum demonstrating a robust interaction with visible light that leads to augmented polarization effects. Conversely, the shallow region displaying a negative εrω function manifests a pronounced light reflection, particularly in the Ca- and Sr-containing variants within the energy range of approximately 9 to 12 eV. In contrast, the Ba-based compounds exhibit no negative εrω function, resulting in stronger interactions with incident light and minimal reflection. These trends are further elucidated by the Rω function, which reveals minimal reflection in Ba-based materials, while prominent reflection peaks correspond to the negative region of the εrω function.
The spectra of εiω, αω, and κω elucidate the intrinsic mechanisms underlying optical transitions and the initiation of absorption in the visible spectrum. These demonstrate the onset of optical transitions and absorption in the visible range of light. The onset frequencies are concordant with the band gap energies investigated, aligning with the red, green, and blue segments of the visible spectrum for the studied Ca-, Sr-, and Ba-based materials, respectively. The transition peaks exhibited in the εiω functions demonstrate pronounced maxima within the visible spectrum and extend into the proximal violet region. Conversely, the more elevated peaks of absorption lie in the ultraviolet domain. This contrast is attributable to the optical transitions encompassing both rates of photon absorption and emission processes; hence, a prominent peak does not necessarily imply elevated absorption. In the instances of Ca3PCl3, Ca3AsCl3, Sr3PCl3, and Sr3AsCl3, the absorption peaks are situated near the violet edge and progress into the ultraviolet range. However, the studied Ba-containing compounds feature an initial albeit diminutive peak of εiω within the visible domain, specifically around the green wavelength at approximately 2.5 eV, while the subsequent peaks reside in the ultraviolet region near 5 eV and 8.5 eV. Thus, Ba-based variants demonstrate relatively adequate absorption capabilities within the visible spectrum. Although each of the investigated materials exhibits absorption within the visible range, their peak responses predominantly linger in the ultraviolet realm. This characteristic renders them promising candidates for vis-UV driven optoelectronic applications.
The refractive index (n0) is a critical design parameter in the realm of optoelectronic devices. The utilization of semiconductors in optoelectronic devices is profoundly influenced by the intrinsic characteristics and magnitude of both the refractive index and band gap of materials, which are instrumental in evaluating device performance.124 Various theoretical models, including the Penn model,125 delineate an inverse correlation between the refractive index and band gap energy, suggesting that a diminished band gap is indicative of a heightened refractive index.126 The refractive index serves as a metric for the transparency or opacity of materials to incident light. The frequency dependence of the refractive index, nω, exhibits trends that closely mirror those of the real component of the dielectric functions, as they are interconnected through eqn (29). At zero frequency, the refractive indices recorded are 1.64, 1.54, 1.62, 1.64, 1.53, and 1.55 for Ca3PCl3, Ca3AsCl3, Sr3PCl3, Sr3AsCl3, Ba3PCl3, and Ba3AsCl3, respectively. It underscores the notion that these perovskite-derivatives exhibit opacity under static conditions. Moreover, their opaqueness intensifies with increasing incident photon energies within the visible spectrum, while their refractive indices decrease in the ultraviolet region, particularly for higher frequencies from 10 to 12 eV, signifying substantial reflective properties in that domain.
The energy loss function, derived from the imaginary component of the inverse of the complex dielectric function (−Im[1/ε(ω)]), elucidates the dissipation of electron energy during electronic excitation phenomena within semiconductors. This construct is paramount for a meticulous quantitative assessment of inelastic scattering during electron transport.127 The most elevated peak of this function corresponds to the plasma frequency.128,129 The plasma frequencies observed for the materials under examination are 11.68, 10.5, 11.28, 11.21, 9.16, and 8.87 eV for Ca3PCl3, Ca3AsCl3, Sr3PCl3, Sr3AsCl3, Ba3PCl3, and Ba3AsCl3, respectively, as illustrated in Fig. 15. These frequencies indicate that incident photons at these energies will primarily be expended in generating inelastic scattering, thereby impeding the generation of the electron–hole pairs. Consequently, one can observe a conspicuous reduction of the absorption coefficients at these corresponding frequencies. In a theoretical study, Gosh et al. recorded peaks at 9.54, 9.44, and 9 eV for Sr3AsF3, Sr3AsCl3, and Sr3AsBr3, respectively.59 Our findings show alignment with these observations. Beyond these plasma frequencies, the materials exhibit transparency, which can be checked in our results by the asymptotically approaching values of refractive index to 1.
 | ||
| Fig. 15 Energy loss functions of the examined M3ACl3 (M = Ca, Sr, Ba, A = P, As) perovskite-derivatives. | ||
The band gap range for the top cell of tandem solar cells is documented as 1.5–2.2 eV.130,131 The calculated band gaps of Ba3PCl3 and Ba3AsCl3 fall within this spectrum. With their ideal band gaps and notable optical properties, these compounds demonstrate considerable promise for application in solar cell technologies, particularly in the top cell of tandem heterojunction cells.132 Their impressive absorption coefficients across the visible spectrum further indicate their favorable optical performance.
Other examined materials demonstrate band gaps spanning 2.5 to 2.9 eV, signifying their potential application in photovoltaic cells for selective UV absorption. Furthermore, their elevated UV absorption coefficients facilitate effective photon capture within the crucial short-wavelength spectrum, thereby diminishing thermalization losses.133,134 Conversely, the relatively low refractive indices, ranging from 1.53 to 1.64, reduce reflection losses at interfaces, thereby enhancing light coupling between sub-cells. The notable peaks in UV absorption suggest their effectiveness for UV emitters, particularly in UV-based LED applications such as sterilization and purification systems,135 where excitonic absorption can improve the efficiency of radiative recombination. The dielectric constants in the 2.35 to 2.68 range indicate compatibility with common charge-transport layers (e.g., TiO2, ZnO), reducing interfacial defects in heterostructures. Moreover, absorption coefficients within the visible spectrum facilitate the utilization of down-conversion phosphors in converted LED technologies.136,137 The combination of low εo values in the 2.3–2.68 range and moderate refractive indices between 1.53 and 1.64 makes these materials suitable for anti-reflection coatings with reduced parasitic absorption, which is critical for photodetectors.
4 Conclusions
The scrutinized M3ACl3 (M = Ca, Sr, Ba, A = N, P, As) perovskite-derivatives manifest covalent interactions between M and A atoms alongside the ionic bonding of M with Cl atoms and display anti-bonding characteristics between A and Cl atoms, as delineated through their charge density distributions and density of states analyses, except Ca3NCl3 which exhibits all ionic bonds. Within these compounds, a hybridization of A-p and M-d electron states engenders bonding interactions that constitute the valence and conduction band edges. The electronic band gaps range from 1.7 to 2.9 eV, as determined through HSE06 calculations, facilitating direct optical transitions between valence and conduction bands at the high-symmetry Γ point. The delocalized Ba-5d electrons significantly enhance p–d interactions, culminating in reduced band gaps for Ba3PCl3 and Ba3AsCl3 as compared to Ca- and Sr-containing materials.The mechanical properties underscore the inherent brittleness of the Ca- and Sr-containing materials as evidenced by their Pugh ratios of less than 1.75, negative Cauchy pressures, and Poisson ratios of less than 0.26, contrasting with the ductile behavior exhibited by Ba-based compounds. Furthermore, these structures exhibit both structural and phase stability, as evidenced by their negative formation energies and positive cohesive energies. Mechanical stability is affirmed through adherence to Born's stability criteria, while dynamic stability is underscored by the phonon dispersion spectra observed between 200 and 500 K. Contrarily, the N-based structures examined lack dynamic stability, attributable to the negative frequencies of their phonon modes across all temperatures.
Additionally, the average carrier mobilities of the analyzed materials span from 38.2 to 192.98 cm2 V−1 s−1 at 300 K and a carrier concentration of 1020 cm−3. All the materials investigated demonstrate reasonably good mobilities; however, Ba3PCl3 stands out with exceptional electron and hole mobilities of 173 and 138.16 cm2 V−1 s−1, respectively. These impressive mobilities highlight its superior optoelectronic capabilities, making it a noteworthy candidate in the field.
The optical spectra reveal that the initiation of optical transitions and absorption occurs within the visible spectrum, corresponding to their respective band gap energies, thus facilitating the absorption of red, green, and blue light for Ca-, Sr-, and Ba-containing compounds, respectively. Specifically, the Ba-containing variant demonstrates promising absorption coefficients of 1.616 and 1.675 × 105 cm−1 at 2.6 eV. However, these spectra exhibit peaks in the ultraviolet range. Consequently, the optical characteristics accentuate the applicability of these materials in devices operating across the visible to UV spectrum. Specifically, Ba-based materials show exceptional promise for photovoltaics, particularly in tandem solar cells, while the others are suitable for a spectrum of optoelectronic applications, including RGB light-emitting diodes, thin-film transistors, diode lasers, semiconductor displays, and UV emitters and sensors.
Moreover, the elucidated optoelectronic properties of the examined materials furnish essential design parameters, encompassing band gaps, carrier mobilities, effective masses of charge carriers, dielectric constants, absorption spectra, and refractive indices, among others, for the computation of PCE of optimized solar cells constructed from these materials.
Conflicts of interest
There is no conflict of interest.Data availability
The authors confirm that the data presented in the main article, as well as in the SI, which support the findings of this study, are available upon reasonable request.Supplementary information is available. See DOI: https://doi.org/10.1039/d5ta05368d.
Acknowledgements
The work was sponsored by the National Natural Science Foundation of China (No. 12374017) and the Innovation Program for Quantum Science and Technology (2021ZD0303303). The computing time of the Supercomputing Center of the University of Science and Technology of China is acknowledged.References
- A. Androniceanu and O. M. Sabie, Energies, 2022, 15, 8573 Search PubMed.
- https://www.iea.org/energy-system/renewables/solar-pv .
- G. Kim, H. Min, K. S. Lee, D. Y. Lee, S. M. Yoon and S. I. Seok, Science, 2020, 370, 108–112 Search PubMed.
- E. Rezaee, W. Zhang and S. R. P. Silva, Small, 2021, 17, 2008145 CrossRef CAS PubMed.
- H. Zhang, X. Ji, H. Yao, Q. Fan, B. Yu and J. Li, Sol. Energy, 2022, 233, 421–434 CrossRef CAS.
- Y.-F. Gu, H.-J. Du, N.-N. Li, L. Yang and C.-Y. Zhou, Chin. Phys. B, 2019, 28, 048802 Search PubMed.
- D. Ju, X. Zheng, J. Yin, Z. Qiu, B. Turedi, X. Liu, Y. Dang, B. Cao, O. F. Mohammed and O. M. Bakr, et al. , ACS Energy Lett., 2018, 4, 228–234 Search PubMed.
- M. Ren, X. Qian, Y. Chen, T. Wang and Y. Zhao, J. Hazard. Mater., 2022, 426, 127848 CrossRef CAS PubMed.
- G. Schileo and G. Grancini, J. Mater. Chem. C, 2021, 9, 67–76 RSC.
- I. G. G. Apurba, M. R. Islam, M. A. Hossain, J. Y. Al-Humaidi and M. M. Rana, Energy Sci. Eng., 2025 DOI:10.1002/ese3.70205.
- M. F. Rahman, M. A. Rahman, M. R. Islam, A. Ghosh, B. Shanto, M. Abul, M. Chowdhury, M. Al Ijajul Islam, M. H. Rahman and M. K. Hossain, et al. , AIP Adv., 2023, 13(8), 085329 CrossRef CAS.
- S. Joifullah, M. A. Hossain, M. Al Yeamin, M. M. Haque, R. K. Pingak, N. F. A. Mohammad, M. S. Abu-Jafar, A. A. Mousa and A. Hosen, Opt. Quant. Electron., 2024, 56, 1463 CrossRef CAS.
- M. M. Haque, A. I. Shimul, A. Ghosh, N. S. Awwad and A. R. Chaudhry, Opt. Express, 2025, 33, 30486–30504 CrossRef PubMed.
- I. G. G. Apurba, M. R. Islam, M. S. Rahman, M. F. Rahman and J. Park, Heliyon, 2024, 10(7), e29144 CrossRef CAS PubMed.
- M. B. H. Parosh, M. Saiduzzaman, J. Islam, N. J. Nisha and I. A. Ovi, RSC Adv., 2025, 15, 10085–10105 RSC.
- M. A. U. Islam, O. Das, D. B. Khadka, M. R. Islam, M. F. Rahman, S. Kato and T. Soga, ACS Omega, 2024, 9, 8005 Search PubMed.
- L. Zhang, T. Hu, J. Li, L. Zhang, H. Li, Z. Lu and G. Wang, Front. Mater., 2020, 6, 330 Search PubMed.
- M. S. Reza, M. F. Rahman, A. Kuddus, M. K. Mohammed, A. K. Al-Mousoi, M. R. Islam, A. Ghosh, S. Bhattarai, R. Pandey and J. Madan, et al. , RSC Adv., 2023, 13, 31330–31345 RSC.
- A. Baumann, J. Lorrmann, D. Rauh, C. Deibel and V. Dyakonov, Adv. Mater., 2012, 24, 4381–4386 CrossRef CAS PubMed.
- C. M. Proctor, J. A. Love and T.-Q. Nguyen, Adv. Mater., 2014, 26, 5957–5961 CrossRef CAS.
- N. K. Tailor, R. Ranjan, S. Ranjan, T. Sharma, A. Singh, A. Garg, K. S. Nalwa, R. K. Gupta and S. Satapathi, et al. , J. Mater. Chem. A, 2021, 9, 21551–21575 RSC.
- S. Poncé, M. Schlipf and F. Giustino, ACS Energy Lett., 2019, 4, 456–463 Search PubMed.
- B. Cucco, J. Leveillee, V.-A. Ha, J. Even, M. Kepenekian, F. Giustino and G. Volonakis, PRX Energy, 2024, 3, 023012 CrossRef.
- M. S. I. Ria, A. Ghosh, M. A. Rahman, J. Y. Al-Humaidi, N. S. Awwad, H. A. Ibrahium and M. M. Rahman, Mater. Sci. Eng., B, 2024, 310, 117674 CrossRef.
- M. S. Islam, M. F. Rahman, M. R. Islam, A. Ghosh, M. A. Monnaf, M. S. Reza, M. K. Hossain, A. Zaman, S. Ezzine and L. B. Farhat, J. Phys. Chem. Solids, 2024, 185, 111791 CrossRef CAS.
- A. Ghosh, A. A. H. Newaz, A. Al Baki, N. S. Awwad, H. A. Ibrahium, M. S. Hossain, M. M. R. Sonic, M. S. Islam and M. K. Rahman, RSC Adv., 2024, 14, 24066–24081 RSC.
- M. A. Rahman, M. F. Rahman, L. Marasamy, M. Harun-Or-Rashid, A. Ghosh, A. R. Chaudhry and A. Irfan, Energy Fuels, 2024, 38, 8199–8217 CrossRef CAS.
- G. Kresse and J. Hafner, Phys. Rev. B: Condens. Matter Mater. Phys., 1993, 47, 558–561 CrossRef CAS PubMed.
- G. Kresse and J. Furthmüller, Phys. Rev. B: Condens. Matter Mater. Phys., 1996, 54, 11169–11186 CrossRef CAS.
- G. Kresse and D. Joubert, Phys. Rev. B: Condens. Matter Mater. Phys., 1999, 59, 1758–1775 CrossRef CAS.
- J. Heyd and G. E. Scuseria, J. Chem. Phys., 2004, 121, 1187–1192 CrossRef CAS PubMed.
- V. Wang, N. Xu, J.-C. Liu, G. Tang and W.-T. Geng, Comput. Phys. Commun., 2021, 267, 108033 CrossRef CAS.
- R. Gaillac, P. Pullumbi and F.-X. Coudert, J. Phys.: Condens. Matter, 2016, 28, 275201 CrossRef PubMed.
- X.-Q. Chen, H. Niu, D. Li and Y. Li, Intermetallics, 2011, 19, 1275–1281 CrossRef CAS.
- Y. Tian, B. Xu and Z. Zhao, Int. J. Refract. Metals Hard Mater., 2012, 33, 93–106 CrossRef CAS.
- A. Togo, L. Chaput, T. Tadano and I. Tanaka, J. Phys. Condens. Matter, 2023, 35, 353001 CrossRef CAS PubMed.
- A. Togo, J. Phys. Soc. Jpn., 2023, 92, 012001 CrossRef.
- A. Otero-de-la Roza, D. Abbasi-Pérez and V. Luaña, Comput. Phys. Commun., 2011, 182, 2232–2248 CrossRef CAS.
- P. Carrier, R. Wentzcovitch and J. Tsuchiya, Phys. Rev. B: Condens. Matter Mater. Phys., 2007, 76, 064116 CrossRef.
- B. B. Karki and R. M. Wentzcovitch, Phys. Rev. B: Condens. Matter Mater. Phys., 2003, 68, 224304 CrossRef.
- H. Wang, J. Zhuang, Z. Zhang, Q. Zhang and R. Wentzcovitch, AGU Fall Meeting Abstracts, 2021, pp. MR45A0066 Search PubMed.
- M. Blanco, E. Francisco and V. Luana, Comput. Phys. Commun., 2004, 158, 57–72 CrossRef CAS.
- N. Merhav, in Vibrations in a Solid – Phonons and Heat Capacity, Springer International Publishing, Cham, 2018, pp. 95–102 Search PubMed.
- S. Poncé, E. R. Margine and F. Giustino, Phys. Rev. B, 2018, 97, 121201 CrossRef.
- F. Giustino, M. L. Cohen and S. G. Louie, Phys. Rev. B Condens. Matter, 2007, 76, 165108 CrossRef.
- H. Lee, S. Poncé, K. Bushick, S. Hajinazar, J. Lafuente-Bartolome, J. Leveillee, C. Lian, J.-M. Lihm, F. Macheda and H. Mori, et al. , npj Comput. Mater., 2023, 9, 156 CrossRef CAS.
- S. Poncé, F. Macheda, E. R. Margine, N. Marzari, N. Bonini and F. Giustino, Phys. Rev. Res., 2021, 3, 043022 CrossRef.
- J. Zhou, S. Poncé and J.-C. Charlier, Phys. Rev. B, 2024, 110, 125304 CrossRef CAS.
- S. Poncé, D. Jena and F. Giustino, Phys. Rev. B, 2019, 100, 085204 CrossRef.
- J. Yin, O. Hellman and S. Poncé, arXiv, 2025, preprint, arXiv:2505.20092, DOI:10.48550/arXiv.2505.20092.
- Z. U. Rehman and Z. Lin, J. Mater. Chem. A, 2024, 12, 8846–8861 RSC.
- Z. Mao, W. Li and J. Tan, npj Comput. Mater., 2024, 10, 265 CrossRef.
- S. A. Dar, R. Sharma, V. Srivastava and U. K. Sakalle, RSC Adv., 2019, 9, 9522–9532 RSC.
- M. F. Rahman, M. H. Rahman, M. R. Islam, M. K. Hossain, A. Ghosh, M. S. Islam, M. M. Islam, M. Harun-Or-Rashid, H. Albalawi and Q. Mahmood, J. Mater. Sci., 2023, 58, 13100–13117 CrossRef CAS.
- A. Hosen, Heliyon, 2024, 10(16), e35855 CrossRef CAS PubMed.
- M. A. I. Islam, M. F. Rahman, M. M. Islam, M. K. Mohammed and A. Irfan, Phys. Chem. Chem. Phys., 2025, 27(4), 1861–1883 RSC.
- M. R. Islam, A. Zahid, M. A. Rahman, M. F. Rahman, M. Islam, M. K. Hossain, M. Ali, M. A. Iqbal, F. I. Bakhsh and S. Ahmad, J. Phys. Chem. Solids, 2024, 184, 111722 CrossRef CAS.
- K. Mishra, S. Chahar and R. Sharma, Solid State Commun., 2024, 390, 115623 Search PubMed.
- A. Ghosh, M. F. I. Buian, N. L. Dey, M. S. I. Ria, A. A. Baki, A. A. Miazee, N. S. Awwad, R. I. C. Robin and H. A. Ibrahium, Phys. Chem. Chem. Phys., 2024, 26, 25890–25909 RSC.
- A. A. Hassan, M. S. I. Ria, A. Ghosh, H. Alrafai, A. Al Baki, S. K. A. Abdelrahim, J. Y. Al-Humaidi, R. I. C. Robin, M. M. Rahman and M. Maniruzzaman, Mater. Sci. Eng., B, 2024, 308, 117559 CrossRef CAS.
- https://aoterodelaroza.github.io/gibbs2/manual/eos/ .
- R. Akter, M. Khan, M. N. M. Nobin, M. S. Ali, M. M. Hossain, M. Z. Rahaman and M. L. Ali, Mater. Today Commun., 2023, 36, 106630 CrossRef CAS.
- M. K. Alam, M. N. M. Nobin and M. L. Ali, RSC Adv., 2024, 14, 1267–1283 RSC.
- D.-M. Han, X.-J. Liu, S.-H. Lv, H.-P. Li and J. Meng, Phys. B, 2010, 405, 3117–3119 CrossRef CAS.
- F. Mouhat and F.-X. Coudert, Phys. Rev. B: Condens. Matter Mater. Phys., 2014, 90, 224104 CrossRef.
- S. Muslov and S. Arutyunov, Inorg. Mater. Appl. Res., 2023, 14, 1028–1033 CrossRef.
- R. Thompson and W. Clegg, Curr. Opin. Solid State Mater. Sci., 2018, 22, 100–108 CrossRef CAS.
- C. J. Price and S. P. Hepplestone, J. Mater. Chem. C, 2023, 11, 14278–14291 RSC.
- S. S. Murugan, Int. J. Mod. Stud. Mech. Eng, 2020, 6, 28–38 Search PubMed.
- M. Naher and S. Naqib, Sci. Rep., 2021, 11, 5592 Search PubMed.
- S. Daoud, Acta Phys. Pol., A, 2018, 133, 23–27 CrossRef CAS.
- M. Khan and M. L. Ali, Adv. Eng. Mater., 2024, 26, 2400614 CrossRef CAS.
- M. N. M. Nobin, Z. Hasan, M. Z. Rahaman and M. L. Ali, J. Mater. Res. Technol., 2024, 30, 5334–5345 Search PubMed.
- M. Fine, L. Brown and H. Marcus, Scripta Metall., 1984, 18, 951–956 CrossRef CAS.
- M. M. Rahaman, M. H. Rubel, M. A. Rashid, M. A. Alam, K. M. Hossain, M. I. Hossain, A. A. Khatun, M. M. Hossain, A. A. Islam and S. Kojima, et al. , J. Mater. Res. Technol., 2019, 8, 3783–3794 Search PubMed.
- T. Hailing, G. Saunders, Y. Yogurtcu, H. Bach and S. Methfessel, J. Phys. C: Solid State Phys., 1984, 17, 4559 CrossRef CAS.
- C. M. Zener and S. Siegel, J. Phys. Chem., 1949, 53, 1468 Search PubMed.
- H. Ledbetter and A. Migliori, J. Appl. Phys., 2006, 100(6), 063516 Search PubMed.
- G. Ruseska, E. Fidancevska and J. Bossert, Sci. Sinter., 2006, 38, 245–253 Search PubMed.
- W. Zhang, L. He, Y. Zhou, D. Tang, B. Ding, C. Zhou, P. J. Dyson, M. K. Nazeeruddin and X. Li, Chem. Eng. J., 2023, 457, 141216 CrossRef CAS.
- J. E. Moussa, P. A. Schultz and J. R. Chelikowsky, J. Chem. Phys., 2012, 136(20), 204117 CrossRef PubMed.
- M. Islam, Nucl. Mater. Energy, 2024, 38, 101631 Search PubMed.
- Y. Duan, L. Qin, L. Shi and G. Tang, Comput. Mater. Sci., 2015, 101, 56–61 CrossRef CAS.
- A. J. Garza and G. E. Scuseria, J. Phys. Chem. Lett., 2016, 7, 4165–4170 Search PubMed.
- P. Sreedevi, R. Vidya and P. Ravindran, Sol. Energy, 2019, 190, 350–360 CrossRef CAS.
- Z. U. Rehman and Z. Lin, J. Mater. Chem. C, 2025, 13, 8151–8168 RSC.
- J. S. Manser, J. A. Christians and P. V. Kamat, Chem. Rev., 2016, 116, 12956–13008 Search PubMed.
- A. S. Mohamed, Optik, 2023, 295, 171505 CrossRef CAS.
- M. Bhullar, A. Akinpelu and Y. Yao, Comput. Mater. Today., 2024, 2, 100009 CrossRef.
- P. King, T. D. Veal, P. Jefferson, C. F. McConville, H. Lu and W. Schaff, Phys. Rev. B:Condens. Matter Mater. Phys., 2007, 75, 115312 CrossRef.
- L. Petit, T. C. Schulthess, A. Svane, Z. Szotek, W. M. Temmerman and A. Janotti, Phys. Rev. B:Condens. Matter Mater. Phys., 2006, 73, 045107 CrossRef.
- M. Nolan and S. D. Elliott, Phys. Chem. Chem. Phys., 2006, 8, 5350–5358 RSC.
- M. F. Rahman, M. M. Islam, M. R. Islam, A. Ghosh, M. A. Rahman, M. H. Rahman, M. A. I. Islam, M. Islam, H. Albalawi and Q. Mahmood, J. Solid State Chem., 2023, 328, 124341 CrossRef CAS.
- H.-J. Feng and Q. Zhang, Appl. Phys. Lett., 2021, 118(11), 111902 CrossRef CAS.
- M. B. Pramanik, M. A. Al Rakib, M. A. Siddik and S. Bhuiyan, Eur. J., Eng. Sci., Tech., 2024, 9, 10–15 Search PubMed.
- A. M. Ganose, D. O. Scanlon, A. Walsh and R. L. Hoye, Nat. Commun., 2022, 13, 4715 CrossRef CAS PubMed.
- C.-Z. Ning, L. Dou and P. Yang, Nat. Rev. Mater., 2017, 2, 1–14 Search PubMed.
- M. Laurien, H. Saini and O. Rubel, Phys. Chem. Chem. Phys., 2021, 23, 7418–7425 RSC.
- K. Ding, B. Chen, Y. Li, Y. Zhang and Z. Chen, J. Mater. Chem. A, 2014, 2, 8294–8303 RSC.
- D. Liu, H. Ruan and R. Sa, Mater. Sci. Semicond. Process., 2025, 186, 109029 CrossRef CAS.
- N. Vukmirović, Phys. Rev. B, 2021, 104, 085203 CrossRef.
- S. Al Bacha, S. Saitzek, P. Roussel, M. Huvé, E. E. McCabe and H. Kabbour, Chem. Mater., 2023, 35, 9528–9541 CrossRef CAS.
- J. Yang, Z. Zhao, H. Geng, C. Cheng, J. Chen, Y. Sun, L. Shi, Y. Yi, Z. Shuai and Y. Guo, et al. , Adv. Mater., 2017, 29, 1702115 Search PubMed.
- H. Zhang, L. Liu and Z. Zhou, RSC Adv., 2012, 2, 9224–9229 Search PubMed.
- H. Zhang, L. Liu and Z. Zhou, Phys. Chem. Chem. Phys., 2012, 14, 1286–1292 Search PubMed.
- O. Madelung, Introduction to Solid-State Theory, Springer, 1978, pp. 175–227 Search PubMed.
- C. Jacoboni and L. Reggiani, Rev. Mod. Phys., 1983, 55, 645 Search PubMed.
- Y. Wu, L. Liu, J. Ning, D. Qiu, S. Wang, J. Xi and J. Yang, J. Mater. Sci. Technol., 2025, 204, 245–254 CrossRef CAS.
- C. Tanase, P. W. Blom, E. J. Meijer and D. M. de Leeuw, Organic Field Effect Transistors II, 2003, pp. 80–86 Search PubMed.
- C. Tanase, P. Blom, D. De Leeuw and E. Meijer, Phys. Status Solidi, 2004, 201, 1236–1245 CrossRef CAS.
- C. Zhang, R. Wang, H. Mishra and Y. Liu, Phys. Rev. Lett., 2023, 130, 087001 CrossRef CAS PubMed.
- R. Schley, A. Berkovitz, S. Rinott, I. Shammass, A. Blumkin and J. Steinhauer, Phys. Rev. Lett., 2013, 111, 055301 CrossRef CAS PubMed.
- J. Chen and L. Meng, ACS Omega, 2022, 7, 20186–20194 CrossRef CAS PubMed.
- S. Poncé, M. Royo, M. Gibertini, N. Marzari and M. Stengel, Phys. Rev. Lett., 2023, 130, 166301 CrossRef PubMed.
- C. Q. Xia, J. Peng, S. Poncé, J. B. Patel, A. D. Wright, T. W. Crothers, M. Uller Rothmann, J. Borchert, R. L. Milot and H. Kraus, et al. , J. Phys. Chem. Lett., 2021, 12, 3607–3617 CrossRef CAS PubMed.
- L. M. Herz, ACS Energy Lett., 2017, 2, 1539–1548 CrossRef CAS.
- T. Dakin, IEEE Electr. Insul. Mag., 2006, 22, 11–28 Search PubMed.
- T. D. Huan, S. Boggs, G. Teyssedre, C. Laurent, M. Cakmak, S. Kumar and R. Ramprasad, Prog. Mater. Sci., 2016, 83, 236–269 Search PubMed.
- M. P. Hughes, Understanding the Dielectric Constant in Organic Photovoltaic Materials, University of California, Santa Barbara, 2019 Search PubMed.
- C. Wang, Z. Zhang, S. Pejic, R. Li, M. Fukuto, L. Zhu and G. Sauvé, Macromolecules, 2018, 51, 9368–9381 Search PubMed.
- J. M. Gutiérrez-Villarreal, H. A. Pineda-León, M. F. Suzuki Valenzuela, I. Sosa-Tinoco and S. J. Castillo, Photonics, 2023, 994 CrossRef.
- P. Sharma and S. Auluck, Phys. Rev. B: Condens. Matter Mater. Phys., 1981, 23, 874 CrossRef CAS.
- X. Liu, B. Xie, C. Duan, Z. Wang, B. Fan, K. Zhang, B. Lin, F. J. Colberts, W. Ma and R. A. Janssen, et al. , J. Mater. Chem. A, 2018, 6, 395–403 RSC.
- N. Ravindra, P. Ganapathy and J. Choi, Infrared Phys. Technol., 2007, 50, 21–29 CrossRef CAS.
- D. R. Penn, Phys. Rev., 1962, 128, 2093 CrossRef CAS.
- S. Tripathy, Opt. Mater., 2015, 46, 240–246 Search PubMed.
- H. Shinotsuka, H. Yoshikawa and S. Tanuma, First-principles calculations of optical energy loss functions for 30 compound and 5 elemental semiconductors, e-J. Surf. Sci. Nanotech., 2021, 19, 70–87 CrossRef CAS.
- J. Zhu and Y. Pan, Mater. Sci. Eng., B, 2024, 308, 117554 CrossRef CAS.
- R. Saniz, L.-H. Ye, T. Shishidou and A. Freeman, Phys. Rev. B:Condens. Matter Mater. Phys., 2006, 74, 014209 CrossRef.
- L. Y. Ming, A. Los and G. Xiong, J. Phys Energy, 2023, 5, 042002 CrossRef.
- J. Tong, Q. Jiang, F. Zhang, S. B. Kang, D. H. Kim and K. Zhu, ACS Energy Lett., 2020, 6, 232–248 CrossRef PubMed.
- R. King, D. Bhusari, A. Boca, D. Larrabee, X.-Q. Liu, W. Hong, C. Fetzer, D. Law and N. Karam, Prog. Photovoltaics Res. Appl., 2011, 19, 797–812 CrossRef CAS.
- C. Chen, S. Zheng and H. Song, Chem. Soc. Rev., 2021, 50, 7250–7329 RSC.
- B. McKenna and R. C. Evans, Adv. Mater., 2017, 29, 1606491 CrossRef PubMed.
- Y. Kebbi, A. I. Muhammad, A. S. Sant'Ana, L. do Prado-Silva, D. Liu and T. Ding, Compr. Rev. Food Sci. Food Saf., 2020, 19, 3501–3527 CrossRef PubMed.
- J. Zhao, B. Han, J. Wang, L. Chen, L. Xu, X. Wang and J. Song, Adv. Opt. Mater., 2024, 12, 2400277 CrossRef CAS.
- A. Mayavan, ACS Omega, 2024, 9, 16880–16892 CAS.
| This journal is © The Royal Society of Chemistry 2025 |